Natural product-derived compounds in clinical trials and drug approvals
Mark S.
Butler
 *a,
Robert J.
Capon
*a,
Robert J.
Capon
 b,
Mark A. T.
Blaskovich
b,
Mark A. T.
Blaskovich
 ac and
Ian R.
Henderson
ac
ac and
Ian R.
Henderson
ac
aCentre for Superbug Solutions, Australia. E-mail: m.butler2@imb.uq.edu.au
bCentre for Chemistry and Drug Discovery, Australia
cARC Training Centre for Environmental and Agricultural Solutions to Antimicrobial Resistance, Institute for Molecular Bioscience, The University of Queensland, St. Lucia, Queensland 4072, Australia
First published on 9th September 2025
Abstract
Covering: January 2014–June 2025. Previous review: Natural Product Reports, 2014, 31, 1612
Natural products (NPs) have long been foundational in medicine, from ancient herbal remedies to the discovery of transformative drugs like morphine and quinine. The mid-20th century marked a ‘golden age’ for antibiotic discovery from natural sources, which then expanded into other therapeutic areas. However, by the late 20th century, other technological advances had shifted NPs from being a central component of the discovery process to one of several options. This review explores the current role of NPs in pharmaceuticals by analysing NP-derived (NP-D) drugs approved since 2014 and clinical candidates in development as of the end of 2024. 58 NP-related drugs launched between January 2014 and June 2025 were identified, which included 45 NP and NP-D new chemical entities (NCEs) and 13 NP-antibody drug conjugates (NP-ADCs). Next, all 579 drugs—388 (67%) of which were NCEs and 191 (33%) were new biological entities (NBEs)—approved globally from 2014 to 2024 were analysed. In total, 56 (9.7%) of these 579 drugs were classified as NPs or NP-Ds using this review's NP definition: 44 NCEs (7.6% overall; 11.3% of NCEs) and 12 NP-ADCs (2.1% overall; 6.3% of NBEs). The number of new NP-D NCEs and NP-ADCs has fluctuated between 0 and 8 annually since 2014, with an average of five approvals per year. Next, 125 NP and NP-D compounds were identified that were undergoing clinical trials or in the registration phase at the end of December 2024. Thirty-three new pharmacophores not previously found in approved drugs are now in development; however, only one has been discovered in the past 15 years. This review highlights the enduring promise of NPs, despite their diminished role in drug discovery, and advocates for renewed emphasis on bioassay-guided isolation and mode of action studies to identify new drug leads.
1. Introduction
Natural products (NPs) have a rich history in medicine, with crude preparations serving as traditional remedies for centuries. These medicines remain vital, especially in developing countries, where they continue to play a significant role in healthcare. The 19th century marked a pivotal moment in drug discovery, with the advent of purification technologies leading to the isolation of compounds like morphine, quinine, pilocarpine, digitoxin, salicin, and ephedrine from plants. The early to mid-20th century saw the rise of synthetic drugs, creating a dynamic interplay between natural products, synthetic chemistry, and vaccines in modern medicine. This is exemplified by the explosion in antibiotic research following the discovery of synthetic sulfonamides in the 1930s, which inspired the development of penicillin, first used clinically in 1942. This sparked a search for new microbially derived antibiotics, leading to the discovery of tyrothricin, gramicidin S, gramicidin D, bacitracin, streptomycin, and chlortetracycline in the 1940s. This period, often referred to as the ‘golden age of antibiotics’, saw about 30 years of intensive NP-focused research into antibiotics and other therapeutic areas. However, in the late 20th century, advancements in technology transformed drug discovery and development. NPs, once the primary source of drug leads, became one tool among many in the discovery arsenal. In the first quarter of the 21st century, biologic drugs (Table 1) saw a remarkable rise, now rivalling small molecules in prominence. For example, biologic drugs represented 10 (21.3%) of the 47 drugs approved globally in 2014, which increased to 32 (42.7%) of the 75 drugs in 2024 (Fig. 1).1,2 Notably, NPs have found new roles as warheads in antibody–drug conjugates (ADCs). Against this backdrop, a crucial question arises: where does NP lead discovery and drug development fit in this ever-evolving pharmaceutical landscape?| Categories | Abbrev. | Definition |
|---|---|---|
| Regulatory | NCE | New chemical entities, typically small molecules but can include peptides, carbohydrates, and polymers |
| NBE | New biological entities, biological products such as proteins, antibodies (including ADCs), microorganisms, viruses and vaccines | |
| Compound | NP | Natural products, isolated from natural sources |
| NP-D | Natural product-derived, prepared by chemical modification of a NP or by total synthesis of a compound inspired from a NP | |
| NP-ADC | Antibody drug conjugate with a NP-derived warhead | |
| Manufacturing | I | Isolated from a natural source and used as isolated |
| SS | Semi-synthesis, involving modification of an advanced natural product precursor | |
| TS | Total synthesis |
 | ||
| Fig. 1 Global NCEs and NBEs approved from 2014 to 2024 divided into NP-D NCEs, NP-ADCs and other NCEs and NBEs. | ||
In this review, the term NP refers to pure, well-defined small molecules—such as terpenes, polyketides, peptides, lipids, and compounds of mixed biogenesis—isolated from natural sources, including marine and terrestrial plants, microbes and marine animals. Molecules typically associated with primary metabolism or primarily serving nutritional roles, such as steroids, bile acids, amino acids, sialic acids, prostaglandins, retinoids, hormones, nucleosides, and human proteins and protein fragments, are excluded. In the figures, NP structures are shown in blue, while NP-derived compounds are depicted in black. Repurposing efforts involving approved NP-derived drugs are discussed in Section 2.2 but not included in overall analyses. The compounds reviewed here are categorized into three compound (NP, NP-D and NP-ADC) and three manufacturing categories (Table 1), along with the definitions of new chemical entities (NCEs) and new biological entities (NBEs). The separation of compound and manufacturing categories marks a departure from previous reviews in this series,3–6 a change made to simplify the analysis, while allowing for a more detailed exploration of manufacturing processes. Compounds were excluded from this analysis if they met any of the following criteria: (1) the clinical trial registry indicated that the trial was completed on or before 31 December 2021, without evidence of an active development plan, (2) clinical development had been terminated, or (3) the compounds had been removed from the developing organization's pipeline without being licensed. The disruptive impact of COVID-19 on clinical trials and company funding was also taken into consideration. Despite the comprehensive analysis of publicly available data, some NP and NP-D compounds may have been overlooked, particularly those in the early stages of clinical development. Additionally, the clinical trial landscape and company involvement can evolve rapidly, so readers are advised to consult company websites, clinical trial registries, and recent literature for the most up-to-date information.
This review provides an overview of NP, NP-D and NP-ADCs drugs launched globally between January 2014 and June 2025 (Section 2, Tables 2 and 3). It then describes NP, NP-D, and NP-ADCs undergoing clinical evaluation or registration as of 31 December 2024, categorized by disease area: infectious diseases (Section 3), neurological diseases (Section 4), cardiovascular and metabolic diseases (Section 5), immunological, inflammatory, and related diseases (Section 6), and oncology (Section 7). ADCs containing NP-derived warheads in phase II trials and above are discussed in Section 7.3. Additionally, the review details the routes of administration and the original lead NP structures for these compounds (Section 8). Section 9 summarises NP-D compounds in late-stage development, along with clinical candidates featuring novel drug pharmacophores.
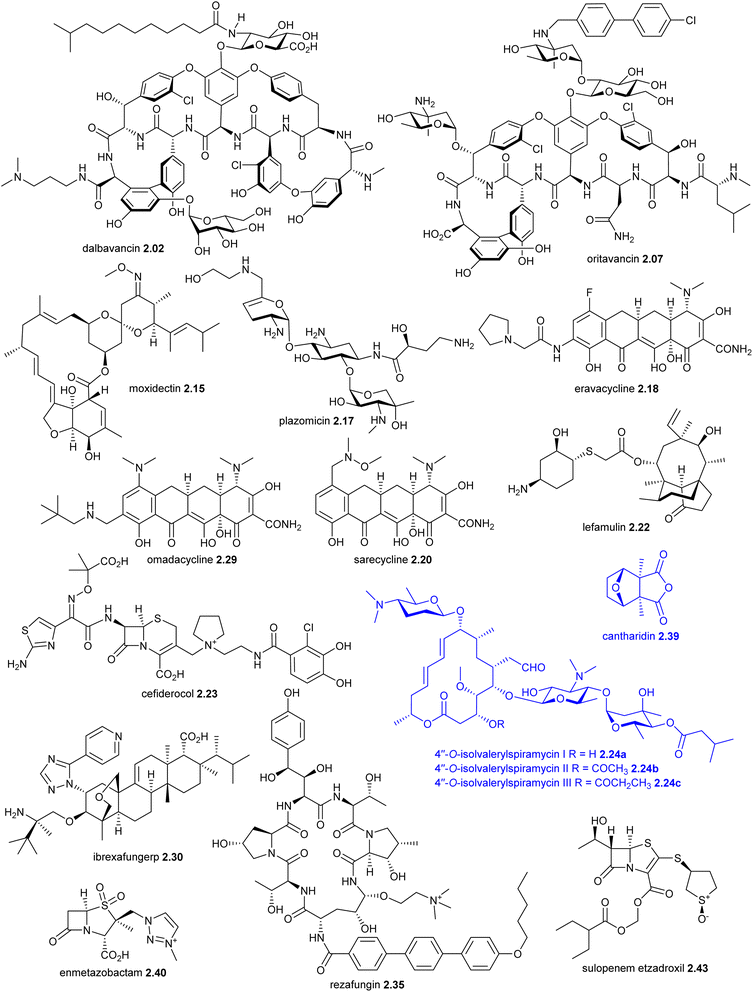
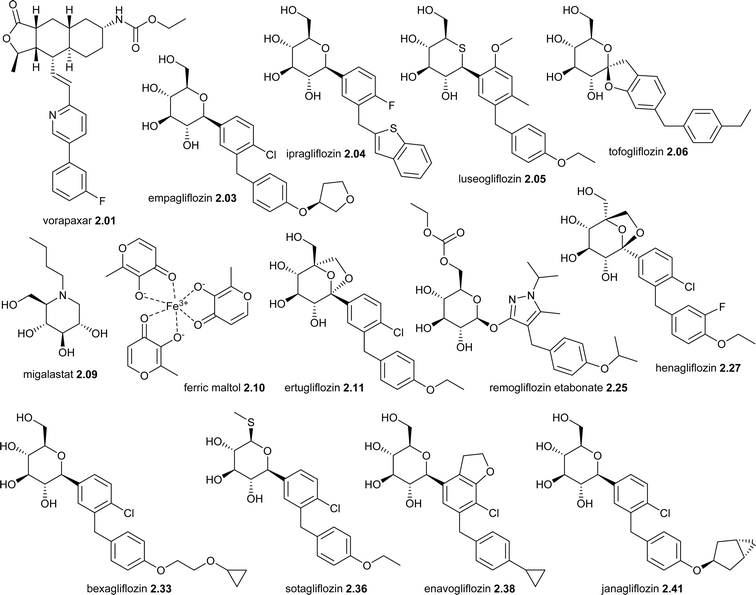
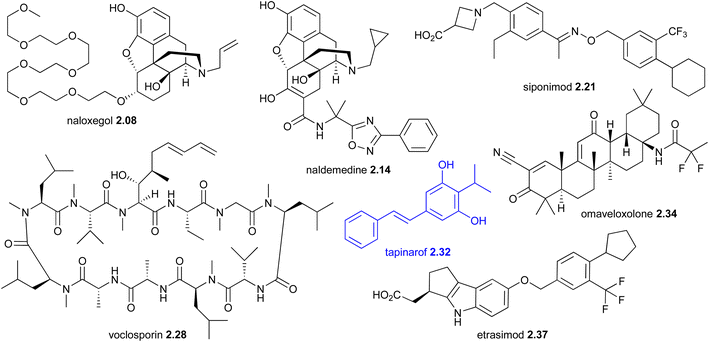
| Year | Drug name (brand names, company) | Lead compound (source) | Mode of action | 1st | Classif | Manuf | Disease area | Launch | Admina |
|---|---|---|---|---|---|---|---|---|---|
| a IM, intramuscular; IV, intravenous; IV/O, both intravenous and oral; O, oral; O-NS, oral, non-systemic; T, topical. b 1-Deoxynojirimycin (moranoline) has been isolated from both plants and bacteria.28 c Moxidectin 2.15 was first approved in 1990 as an animal anthelmintic before its repurposing. d Plitidepsin 2.16 and trabectedin 7.45 were originally isolated from ascidians but are produced by bacteria.29 e Carrimycin is a mixture of three macrolide antibiotics, isovalerylspiramycin I 2.24a, II 2.24b and III 2.24c. Abbreviations: ABSSI, acute bacterial skin and skin structure infections; AhR, aryl hydrocarbon receptor; AML, acute myeloid leukaemia; CABP, community-acquired bacterial pneumonia; eEF1A2, eukaryotic elongation factor 1A2; FLT3, Fms-like tyrosine kinase 3; G-ve, Gram negative; G+ve, Gram positive; IAI, intra-abdominal infections; Nrf2, nuclear factor erythroid 2-related factor 2; PAR-1, protease-activated receptor-1; PBP, penicillin binding protein; SGLT1, sodium-glucose co-transporter 1; SGLT2, sodium-glucose co-transporter 2; S1P, sphingosine-1-phosphate; TRPM8, transient receptor potential melastatin; UTI, urinary tract infection. | |||||||||
| 2014 | Vorapaxar 2.01 (Zontivity®, Merck & Co) | Himbacine (plant) | PAR-1 | Y | NP-D | TS | Cardiovascular | USA | O |
| 2014 | Dalbavancin 2.02 (Dalvance®, Durata Therapeutics) | A40926 (actinomycete) | D-Ala-D-Ala binding | NP-D | SS | Antibacterial | USA | IV | |
| 2014 | Empagliflozin 2.03 (Jardiance®, Boehringer Ingelheim and Eli Lilly & Co) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | USA | O | |
| 2014 | Ipragliflozin 2.04 (Suglat®, Astellas & Kotobuki) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | Japan | O | |
| 2014 | Luseogliflozin 2.05 (Lusefi®, Taisho Pharmaceutical) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | Japan | O | |
| 2014 | Tofogliflozin 2.06 (Deberza®/Apleway®, Chugai Pharmaceutical) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | Japan | O | |
| 2014 | Oritavancin 2.07 (Orbactiv®, The Medicines Company) | Chloroeremomycin (actino.) | D-Ala-D-Ala binding | NP-D | SS | Antibacterial | USA | IV | |
| 2014 | Naloxegol 2.08 (Movantik®, AstraZeneca) | Morphine (plant) | μ-Opioid receptor | NP-D | SS | Opioid-induced constipation | USA | O-NS | |
| 2016 | Migalastat 2.09 (Galafold®, Amicus Therapeutics) | 1-Deoxynojirimycin (plantb) | α-Galactosidase A stabilisation | NP-D | TS | Faby disease | EU | O | |
| 2016 | Ferric maltol 2.10 (Feraccru®/Accrufer®, Shield Therapeutics) | Maltol (plant) | Iron replacement | NP-D | TS | Iron deficiency | EU | O | |
| 2017 | Ertugliflozin 2.11 (Steglatro®, Merck & Co & Pfizer | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | USA | O | |
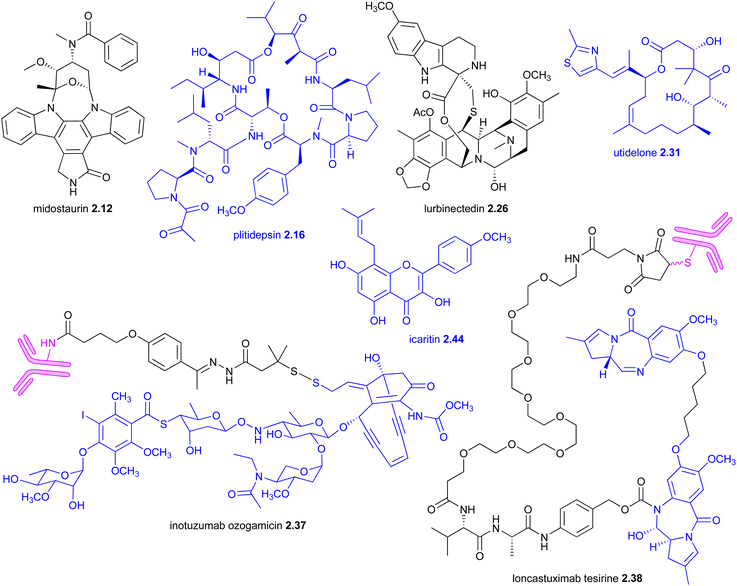
| 2017 | Midostaurin 2.12 (Rydapt®, Novartis) | Staurosporine (actinomycete) | FLT3 & other kinases | Y | NP-D | TS | Oncology | USA | O |
| 2017 | Dinalbuphine sebacate 2.13 (Naldebain®, Lumosa Therapeutics) | Morphine (plant) | κ- and partial μ-opioid receptors | NP-D | SS | Postsurgical pain | Taiwan | IM | |
| 2017 | Naldemedine 2.14 (Symproic®, Shionogi & Co) | Morphine (plant) | μ-, δ-, and κ-opioid receptors | NP-D | SS | Opioid-induced constipation | USA | O-NS | |
| 2018 | Moxidectin 2.15 (Moxidectin®, Medicines Development for Global Health) | Nemadectin (actinomycete) | Glutamate-gated chloride channels | Y | NP-D | SS | Antiparasiticc | USA | O |
| 2018 | Plitidepsin 2.16 (Aplidin®, PharmaMar) | Plitidepsin 2.16 (ascidiand) | eEF1A2 and other mechanisms | Y | NP | I | Oncology | Australia | IV |
| 2018 | Plazomicin 2.17 (Zemdri®, Achaogen) | Sisomicin (actinomycete) | Protein synthesis | NP-D | SS | Antibacterial | USA | IV | |
| 2018 | Eravacycline 2.18 (Xerava®, Tetraphase Pharmaceuticals) | Tetracycline (actinomycete) | Protein synthesis | NP-D | TS | Antibacterial | USA | IV | |
| 2018 | Omadacycline 2.19 (Nuzyra®, Paratek Pharmaceuticals) | Tetracycline (actinomycete) | Protein synthesis | NP-D | SS | Antibacterial | USA | IV/O | |
| 2018 | Sarecycline 2.20 (Seysara®, Almirall) | Tetracycline (actinomycete) | Protein synthesis | NP-D | SS | Antibacterial | USA | O | |
| 2019 | Siponimod 2.21 (Mayzent®, Novartis) | Myriocin (fungus) | S1P receptor | NP-D | TS | Multiple sclerosis | USA | O | |
| 2019 | Lefamulin 2.22 (Xenleta®, Nabriva Therapeutics) | Pleuromutilin (fungus) | Bacterial protein synthesis | NP-D | SS | Antibacterial | USA | IV/O | |
| 2019 | Cefiderocol 2.23 (Fetroja®, Shionogi & Co) | Cephalosporin (fungus) | PBP | NP-D | SS | Antibacterial | USA | IV | |
| 2019 | Carrimycin 2.24a–2.24c (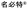 , Shenyang Tonglian Pharmaceutical) , Shenyang Tonglian Pharmaceutical) |
Spiramycin I (actinomycete) | Bacterial protein synthesis | NPe | I | Antibacterial | China | O | |
| 2019 | Remogliflozin etabonate 2.25 (Remo®, Glenmark Pharmaceuticals) | Phlorizin (plant) | SGLT1 and SGLT2 | NP-D | TS | Type 2 diabetes | India | O | |
| 2020 | Lurbinectedin 2.26 (Zepzelca®, PharmaMar) | Trabectedin 7.45 (ascidianc) | DNA binding | NP-D | TS | Oncology | USA | IV | |
| 2021 | Henagliflozin 2.27 (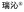 , Guangzhou Baiyunshan Pharmaceutical) , Guangzhou Baiyunshan Pharmaceutical) |
Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | China | O | |
| 2021 | Voclosporin 2.28 (Lupkynis®, Aurinia Pharmaceuticals) | Cyclosporin (fungus) | Calcineurin inhibition | NP-D | SS | Lupus nephritis | USA | O | |
| 2021 | Samidorphan 2.29 + olanzapine (Lybalvi®, Alkermes) | Morphine (plant) | μ-, δ-, and κ-opioid receptors | NP-D | SS | Schizophrenia and bipolar | USA | O | |
| 2021 | Ibrexafungerp 2.30 (Brexafemme®, SCYNEXIS) | Enfumafungin (fungus) | 1,3-β-D-Glucan synthesis | Y | NP-D | SS | Antifungal | USA | IV/O |
| 2021 | Utidelone 2.31 (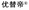 , Shanghai Fosun Pharmaceutical) , Shanghai Fosun Pharmaceutical) |
Epothilone (myxobacteria) | Microtubule stabilisation | NP | I | Oncology | China | IV | |
| 2022 | Icaritin 2.44 (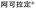 , Beijing Shenogen Pharmaceutical) , Beijing Shenogen Pharmaceutical) |
Icaritin 2.44 (plant) | Signal pathway mediation | NP | I | Oncology | China | O | |
| 2022 | Tapinarof 2.32 (Vtama®, Dermavant Sciences) | Tapinarof 2.32 (bacteria) | AhR activation | Y | NP | TS | Plaque psoriasis | USA | T |
| 2022 | Bexagliflozin 2.33 (Brenzavvy®, TheracosBio) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | USA | O | |
| 2023 | Omaveloxolone 2.34 (Skyclarys®, Reata Pharmaceuticals) | Oleanonic acid (plant) | Nrf2 pathway activation | Y | NP-D | SS | Friedreich's ataxia | USA | O |
| 2023 | Rezafungin 2.35 (Rezzayo®, Cidara Therapeutics) | Echinocandin (fungus) | 1,3-β-D-Glucan synthesis | NP-D | SS | Antifungal | USA | IV | |
| 2023 | Sotagliflozin 2.36 (Inpefa®, Lexicon pharmaceuticals) | Phlorizin (plant) | SGLT2 | NP-D | TS | Heart failure | EU | O | |
| 2023 | Etrasimod 2.37 (Velsipity®, Arena pharmaceuticals) | Myriocin (fungus) | S1P receptor | NP-D | TS | Ulcerative colitis | USA | O | |
| 2023 | Enavogliflozin 2.38 (Envlo®, Daewoong Pharmaceutical) | Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | S. Korea | O | |
| 2023 | Cantharidin 2.39 (Ycanth®, Verrica Pharmaceuticals) | Cantharidin 2.39 (beetle) | Skin blistering | Y | NP | TS | Antiviral | USA | T |
| 2024 | Enmetazobactam 2.40 + cefepime (Exblifep®, Allecra Therapeutics) | Clavulanic acid (actinomycete) | β-Lactamase | NP-D | TS | Antibacterial | EU | O | |
| 2024 | Janagliflozin 2.41 ( , Sihuan Pharmaceutical) , Sihuan Pharmaceutical) |
Phlorizin (plant) | SGLT2 | NP-D | TS | Type 2 diabetes | China | O | |
| 2024 | Xanomeline 2.42 + trospium (Cobenfy®, Karuna Therapeutics) | Arecoline (plant) | Muscarinic receptor | Y | NP-D | TS | Schizophrenia | USA | O |
| 2024 | Sulopenem etzadroxil 2.43 + probenecid (Orlynvah®, Iterum Therapeutics) | Thienamycin (actinomycete) | PBP inhibition | NP-D | TS | Antibacterial | USA | O | |
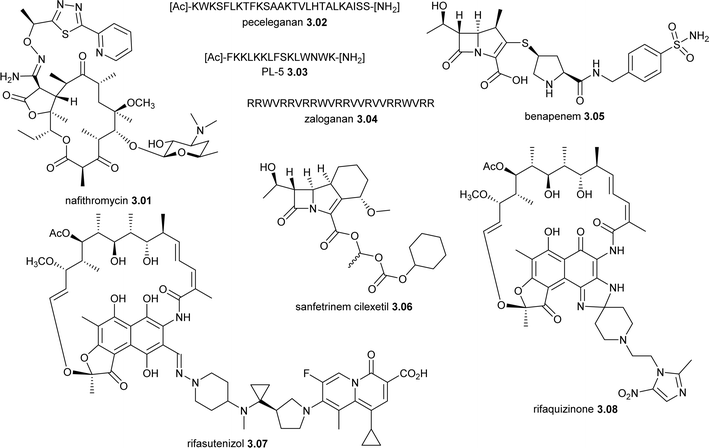
| 2025 | Nafithromycin 3.01 (Miqnaf®, Wockhardt) | Erythromycin (actinomycete) | Protein synthesis | NP-D | SS | Antibacterial | India | O | |
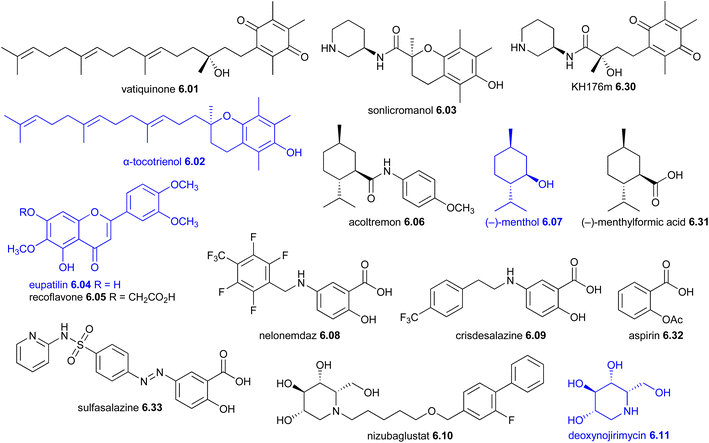
| 2025 | Acoltremon 6.06 (Tryptyr®, Alcon) | Menthol (plant) | TRPM8 protein stimulant | NP-D | SS | Dye eye disease | USA | T | |
| Year | Drug name (brand names, company) | Lead compound, warhead (source)/antigen | Warhead MoA | 1st | Cancer | Launch | Admin |
|---|---|---|---|---|---|---|---|
| a Dolastatin 10 was originally isolated from a sea hare but is biosynthesised by cyanobacteria.30,31 b The first ADC biosimilar, trastuzumab deruxtecan (Ujvira®), was approved in India in 2021. c Belantamab mafodotin was withdrawn from the US market in November 2022 upon request by FDA as a confirmatory phase III trial did not meet the requirements of their Accelerated Approval regulations. d Maytansine was originally isolated from a plant32 but it is also produced by bacteria.33 Abbreviations: ALL, acute lymphoblastic leukaemia; DLBCL, diffuse large B-cell lymphoma. | |||||||
| 2017 | Inotuzumab ozogamicin 2.37 (Besponsa®, Pfizer) | Calicheamicin γI1 (actinomycete)/CD22 | DNA cleavage | B cell precursor ALL | USA | IV | |
| 2019 | Polatuzumab vedotin (Polivy®, Genentech) | Dolastatin 10, MMAE (sea harea)/CD79b | Microtubule inhibition | DLBCL | USA | IV | |
| 2019 | Enfortumab vedotin (Padcev®, Seattle Genetics) | Dolastatin 10, MMAE (sea harea)/NECTIN4 | Microtubule inhibition | Urothelial | USA | IV | |
| 2019 | Trastuzumab deruxtecan (Enhertu®, Daiichi Sankyo) | Camptothecin, DXd (plant)/HER2 | DNA topoisomerase I | Y | HER2-positive breast | USAb | IV |
| 2020 | Sacituzumab govitecan (Trodelvy®, Immunomedics) | Camptothecin, SN38 (plant)/TROP2 | DNA topoisomerase I | Triple-negative breast | USA | IV | |
| 2020 | Belantamab mafodotin (Blenrep®, GlaxoSmithKline) | Dolastatin 10, MMAE (sea harea)/BCMA | Microtubule inhibition | Multiple myeloma | EUc | IV | |
| 2021 | Loncastuximab tesirine 2.38 (Zynlonta®, ADC Therapeutics) | Anthramycin, PBD (actinomycete)/CD19 | DNA binding | Y | DLBCL | USA | IV |
| 2021 | Tisotumab vedotin (Tivdak®, Seagen) | Dolastatin 10, MMAE (sea harea)/TF | Microtubule inhibition | Cervical | USA | IV | |
| 2021 | Disitamab vedotin (Aidexi®,  , RemeGen) , RemeGen) |
Dolastatin 10, MMAE (sea harea)/HER2 | Microtubule inhibition | Gastric | China | IV | |
| 2022 | Mirvetuximab soravtansine (Elahere®, ImmunoGen) | Maytansine, DM4 (plantd)/FRα | Microtubule inhibition | Ovarian | USA | IV | |
| 2024 | Sacituzumab tirumotecan ( , Sichuan Kelun-Biotech) , Sichuan Kelun-Biotech) |
Camptothecin, KL610023 (plant)/TROP2 | DNA topoisomerase I | Triple-negative breast | China | IV | |
| 2024 | Datopotamab deruxtecan (Datroway®, Daiichi Sankyo & AstraZeneca) | Camptothecin, DXd (plant)/TROP2 | DNA topoisomerase I | HR+/HER2- breast | Japan | IV | |
| 2025 | Telisotuzumab vedotin 7.55 (Emrelis, AbbieVie) | MMAE (dolastatin)/MET | Microtubule inhibition | NSCLC | USA | IV | |
1.1 Development pathway summary
Understanding the drug development process requires familiarity with key terms related to drug approval.7 Before clinical trials can begin, an investigational new drug application (IND) must be submitted to the appropriate regulatory agency, such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), Chinese National Medical Products Administration (NMPA), Japanese Pharmaceuticals and Medical Devices Agency (PMDA), Indian Central Drugs Standard Control Organization (CDSCO), Australian Therapeutic Goods Administration (TGA), or their equivalents. In Europe, this application is called an Investigational Medicinal Product Dossier (IMPD).Phase I clinical trials focus on evaluating drug candidate safety, metabolism, and excretion in healthy volunteers. These trials are sometimes subdivided into phase Ia (single ascending dose) and phase Ib (multiple ascending dose) studies. Phase II trials assess the drug candidate's efficacy and side effects in a larger patient group affected by the target disease or condition, establishing a preliminary risk/benefit profile and identifying appropriate dosing levels. Phase III trials involve even larger patient populations and compare the drug candidate's effectiveness against standard or equivalent treatments, while also monitoring for adverse reactions. Hybrid trials, such as phase Ib/II or phase II/III, may also be conducted to streamline the development process. Upon successful completion of clinical trials, the applicant submits a New Drug Application (NDA) or Biologics License Application (BLA) to obtain marketing approval. In Europe, the equivalent submission is the Marketing Authorisation Application (MAA). Drugs approved via an NDA are referred to as NCEs (Table 1), typically small molecules but sometimes including peptides, carbohydrates, or polymers. Approvals from a BLA pertain to NBEs (Table 1), which are biological products such as proteins, antibodies (including ADCs), microorganisms, viruses, and vaccines. Even after approval, drugs undergo continuous monitoring in phase IV trials to identify long-term or rare adverse effects, ensuring ongoing safety for the general population.
1.2 Data sources
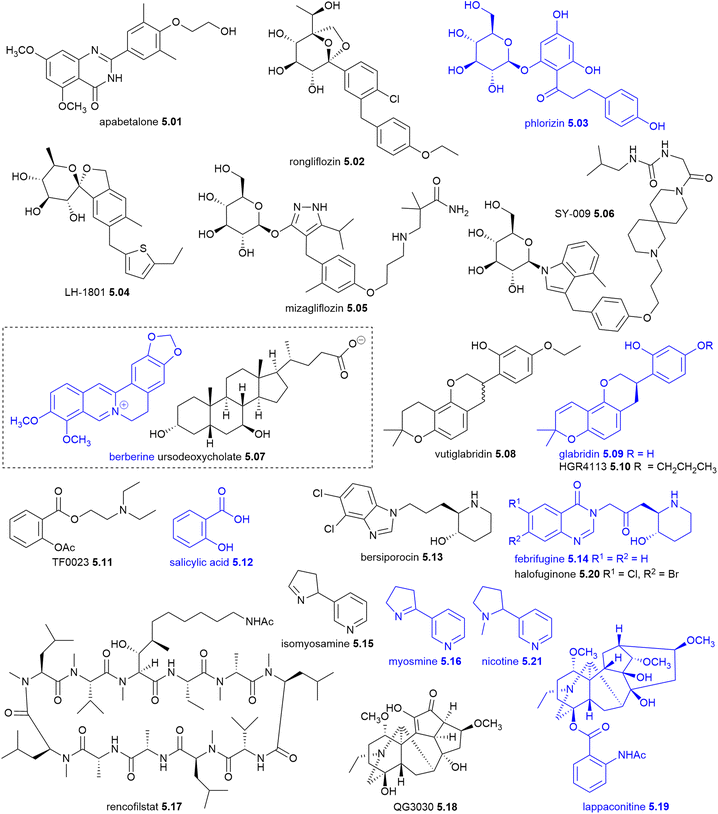
The data for this review was gathered from a range of sources, including scientific literature (reviews, research papers, books, and patents), publicly available conference presentations, clinical trial registries such as the US NIH's https://clinicaltrials.gov/, WHO International Clinical Trials Registry, ISRCTN Registry, Europe, Japan, China, India, South Korea, and Australia and New Zealand, and biotechnology news aggregators such as ADIS Insights, Synapse, ChemLinked, STAT News, Fierce Biotech and BioSpace. Information was also sourced from Research organisations and company websites (pipeline disclosures, press releases, investor presentations and regulatory filings), as well as reports written by companies, governments and non-government organisations (NGOs).2. NP, NP-D and NP-ADC drugs approved globally from January 2014 to June 2025
2.1 NP-derived drugs
From January 2014 to June 2025, a total of 58 NP related drugs were approved globally: 45 were NP and NP-D NCEs (Table 2) and 13 were NP-ADCs (Table 3). Among the 45 NCEs, six were NPs and 39 were NP-Ds with three being manufactured from biomass, 18 were semi-synthetic and 24 by total synthesis. Nine of the 45 NCEs were from new human drug classes (Table 2), while two of the thirteen NP-ADCs had first-in-class warheads (Table 3). A brief overview of these 55 NP-derived drugs launched between 2014 and 2024, along with their structures, is provided by therapeutic area in Sections 2.1.1–2.1.5 with the three NP-D drugs approved to June 2025, nafithromycin 3.01 acoltremon 6.06 and telisotuzumab vedotin 7.55 in Sections 3.1, 6 and 7.3 respectively.All of the NCE and NBE drugs approved globally between 2014 and 2024,1,2,8–27 were categorised into NP and NP-D NCEs, NP-ADCs and other NCEs and NBEs (Fig. 1). Between 2014 and 2024, 579 new drugs were approved globally, which comprised 388 (67%) NCEs and 191 (33%) NBEs. Of the 579 new drugs, 44 NCEs (7.6% total, 11.3% NCEs) were NP and NP-D, while 12 NBEs (2.1% total, 6.3% NBEs) were NP-ADCs. In total, 56 (9.7%) of the 579 drugs were NPs, NP-D and NP-ADCs using this review's NP definition. This percentage would be higher if steroids, bile acids, amino acids, sialic acids, prostaglandins, retinoids, hormones, nucleosides were included. Several key trends emerge from Fig. 1:
(1) The number of NCEs has slightly increased over the past decade compared to most of the previous 14 years, with reduced approval numbers in 2022 and 2023 likely due to the COVID-19 pandemic.
(2) There has been a significant rise in NBE approvals over the last 10 years.
(3) From 2014 to 2024, the number of NP-derived NCEs and NP-ADCs has fluctuated between 0 and 8 annually, with an average of five approvals per year.
(4) Since 2014, 13 NP-ADCs have been approved (Table 3), and this number is expected to rise, given the many candidates in late-stage clinical trials and registration (Section 7.3).
Fifteen new anti-infective drugs were launched, encompassing 11 antibacterials (including one β-lactamase inhibitor), two antifungals, one antiviral, and one antiparasitic. The antibacterial drugs belonged to several previously approved classes: three tetracyclines (eravacycline 2.18,34 sarecycline 2.20 (ref. 35) and omadacycline 2.29 (ref. 36)), two glycopeptides (dalbavancin 2.02 (ref. 37) and oritavancin 2.07 (ref. 38)), two β-lactams (cefiderocol 2.23 (ref. 39) and sulopenem etzadroxil 2.43 (ref. 40)), one β-lactam β-lactamase inhibitor (enmetazobactam 2.40 (ref. 41)), one aminoglycoside (plazomicin 2.17),42 one pleuromutilin (lefamulin 2.22)43 and one macrolide mixture (carrimycin 2.24a–2.24c).44,45 The two approved antifungal drugs, ibrexafungerp 2.30 (ref. 46 and 47) and rezafungin 2.35,48 belong to the new fungerp49 and the established echinocandin classes,50 respectively. Cantharidin 2.39, a topical antiviral, was approved for treating molluscum contagiosum (a DNA poxvirus infection).51 Historically, it has also been used in traditional Chinese medicine52 and as an anti-wart treatment in Europe and the US in the 1950s.53 The antiparasitic drug moxidectin 2.15 is a repurposed veterinary anthelmintic.54
From 2014 to 2024, 12 ADCs were launched (Table 2), building on the foundation laid by three previously approved ADCs: gemtuzumab ozogamicin (first launch: 2000, USA, warhead: calicheamicin γ1I/mAb target: CD33), brentuximab vedotin (2011, USA, dolastatin MMAE/CD30), and trastuzumab emtansine 7.56 (2013, USA, maytansine DM1/HER2). Seven of the 12 ADCs incorporated these NP-D warheads, calicheamicin × 1, dolastatin × 5, maytansine × 1, while the remaining five used new NP-derived warheads (camptothecin × 4 and anthramycin × 1). The structures of inotuzumab ozogamicin 2.37 and loncastuximab tesirine 2.38 are provided in this section, with representative examples of the other NP-derived warheads detailed in Section 7.3.
2.2 Selected previously approved NP and NP-D drugs in late-stage development
Tebipenem pivoxil (GSK3778712, SPR-994),82 first launched in Japan in 2009, is now being investigated by Spero Therapeutics and GSK in a phase III trial (NCT06059846) for the treatment of complicated urinary tract infections (cUTIs), with the goal of securing approval in markets outside of Asia. The trial was stopped early in May 2025 due to an interim analysis confirming non-inferiority to IV imipenem-cilastatin.83 A combination of the monobactam aztreonam (first launch 1984) and the β-lactamase inhibitor avibactam (launched 2015), which is called Emblaveo®, was approved by the EMA in March 2024 to treat complicated intra-abdominal infections (IAI), urinary tract infections (UTIs), hospital-acquired pneumonia (HAP) and infections due to aerobic Gram-negative (G-ve) organisms in people with limited treatment options.84,85 An intravenous formulation of rifabutin (BV100) developed by BioVersys AG has completed a phase II trial (NCT05685615) as a potential treatment of severe infections caused by carbapenem-resistant Acinetobacter baumannii.86 Cannabidiol and dronabinol are being evaluated in multiple phase II and III trials, while cytisine (cytisinicline) is being evaluated in several phase II and III trials for smoking withdrawal around the world. Cytisine has been used in the USSR and Eastern Europe since the 1960s87 and is also approved in a few other countries.88,893. Compounds undergoing evaluation in infectious diseases
NPs have played a pivotal role in the development of new anti-infective drugs. However, despite pressing clinical needs and the growing threat of resistance, large pharmaceutical companies have reduced or abandoned their anti-infective drug development efforts. Progress in antibacterial drug discovery has stalled, especially for agents active against G-ve bacteria,90–92 with poor financial incentives contributing to the decline.93–95 As a result, the field now relies heavily on small- to mid-sized biotech companies (however several of these companies such as Achaogen and Tetraphase shut down after securing approval of a new antibiotic due to lack of revenue), public–private partnerships, and non-profit initiatives to drive innovation and sustain the pipeline. One emerging alternative is the development of indirect-acting and biologic antibacterials, often referred to as non-traditional agents90,96–99 Examples of non-traditional approaches include directly or indirectly acting bacteriophages, proteins, microorganisms and monoclonal antibodies (mAbs), as well as other indirectly acting agents that function through virulence suppression, resistance mitigation, restoration of the gut microbiome, or enhancement of host immunity to promote infection clearance. At the same time, antifungal drug innovation remains limited, with only a small number of new classes reaching clinical trials or approval in recent decades.49,100 In contrast, antiviral drug development has been more dynamic, driven by an urgent response to emerging viral threats such as SARS-CoV-2.101 Despite recent progress, most antiviral development has been virus-specific, underscoring the ongoing need for effective broad-spectrum agents. Antiparasitic drug pipelines also remain relatively limited, with only a few novel therapies emerging beyond established treatments.102,103 The NP and NP-D antibacterials in clinical trials are described in Section 3.1 (Table 4), while antiparasitic, antiviral and antifungal drug candidates are discussed in Section 3.2 (Table 5).| Compound (synonym) | Lead compound (class,a source) | Classif | Manuf | Mode of action | Development status (administration), disease | Developer |
|---|---|---|---|---|---|---|
| a Antibiotic class if different from the lead compound. b Also in clinical trials as an antifungal (Section 3.3). c Structure not publicly available. Abbreviations: ABSSSI, acute bacterial skin and skin structure infections; CABP, community-acquired bacterial pneumonia; cUTI, complicated urinary tract infections; PJI, periprosthetic joint infection; TB, tuberculosis. | ||||||
| Nafithromycin 3.01 (WCK-4873) | Erythromycin (actinomycete) | NP-D | SS | Protein synthesis | Approved 2 January 2025 in India (oral), CABP | Wockhardt |
| Peceleganan 3.02 (PL-5, V681) | Cecropins (AMP, insects, moths) and melittin (bees) | NP-D | TS | Membrane disruption | NDA in China (topical), wound infections; phase II, diabetic foot infections | Jiangsu ProteLight |
| PL-18 3.03 | H. pylori ribosomal protein L1 (AMP, bacteria) | NP-D | TS | Membrane disruption | Phase I (suppository), bacterial vaginosis; antifungalb | Jiangsu ProteLight |
| Zaloganan 3.04 (PLG0206) | LL37 (AMP, human) | NP-D | TS | Membrane disruption | Phase I (topical), PJI | Peptilogics |
| HRS-8427c | Cefiderocol 2.23 (cephalosporin, fungus) | NP-D | SS | Penicillin binding protein, iron transport mediated cell entry | Phase III (IV), cUTI | Jiangsu Hengrui Medicine |
| Benapenem 3.05 | Thienamycin (carbapenem, actinomycete) | NP-D | TS | Penicillin binding protein | Phase II/III (IV), cUTI | Xuanzhu Biopharm |
| Sanfetrinem cilexetil 3.06 (GV118819) | Thienamycin (trinem β-lactam, actinomycetes) | NP-D | TS | Penicillin binding protein | Phase II (oral), TB | GSK/TASK Applied Science |
| Rifasutenizol 3.07 (TNP-2198) | Rifamycin-nitroimidazole hybrid (actinomycete) | NP-D | SS | RNA polymerase, reactive nitrogen species that can damage bacterial DNA and cells | Phase III (oral, non-systemic), H. pylori | TenNor Therapeutics |
| Rifaquizinone 3.08 (TNP-2092) | Rifamycin-quinolizinone hybrid (actinomycete) | NP-D | SS | RNA polymerase, DNA gyrase (GyrA) and Topo IV (ParC) | Phase II (oral, non-systemic and IV), PJI and G+ve ABSSSI | TenNor Therapeutics |
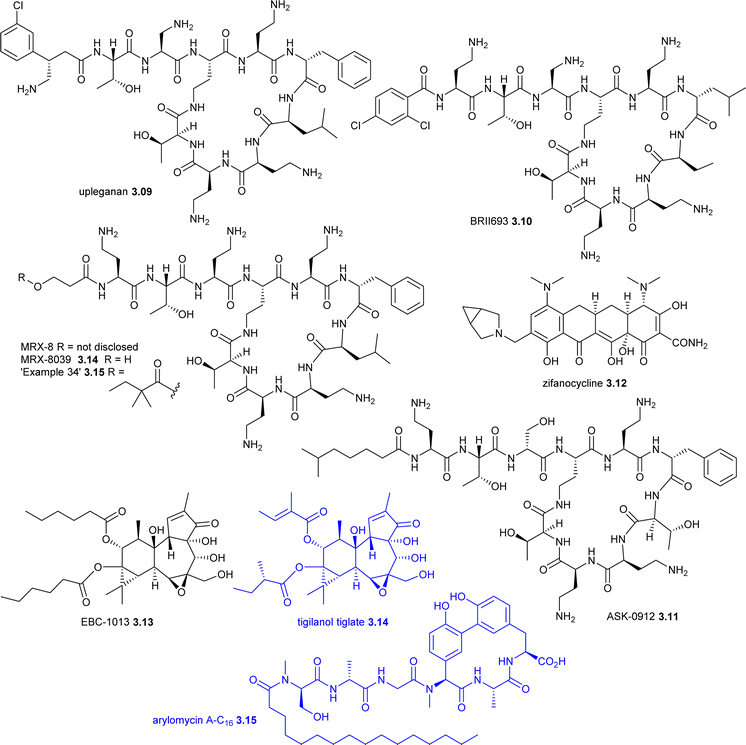
| Upleganan 3.09 (SPR206) | Polymyxin (bacteria) | NP-D | TS | Membrane disruption | Phase I (IV), G-ve infections | MicuRx |
| MRX-8c | Polymyxin (bacteria) | NP-D | TS | Membrane disruption | Phase I (IV), G-ve infections | Spero Therapeutics |
| BRII-693 3.10 (QPX-9003) | Polymyxin (bacteria) | NP-D | TS | Membrane disruption | Phase I (IV), G-ve infections | Brii Biosciences |
| ASK0912 3.11 (polymyxin S2, IMB-0912) | Polymyxin (bacteria) | NP | TS | Membrane disruption | Phase I(IV), G-ve infections | Jiangsu Aosaikang |
| Zifanocycline 3.12 (KBP-7072) | Tetracycline (actinomycete) | NP-D | SS | Protein synthesis | Phase I (IV/oral), G+ve and G-ve infections | KBP Biosciences |
| RG6436c (GGDC-0829) | Arylomycin (actinomycete) | NP-D | TS | LepB (SPase I) | Phase I (IV), cUTI | Genentech (Roche) |
| EBC-1013 3.13 | Tigilanol tiglate 3.14 (plant) | NP-D | SS | Bacterial and biofilm modulation, followed by immune cell activation | Phase I (topical), wound healing | QBiotics |
| Compound (synonyms) | Lead compound (source) | Classif | Manuf | Mode of action | Development status (administration), disease(s) | Developer |
|---|---|---|---|---|---|---|
| a Q-Griffithsin is a recombinantly produced lectin composed of 121 amino acids (MWt 12.7 kDa). b Transcrocetin 3.19 also in clinical trials in oncology (Section 7.1). c Structure not publicly available. d PL-18 3.03 is also in antibacterial clinical trials (Section 2.1). | ||||||
| Antiparasitic | ||||||
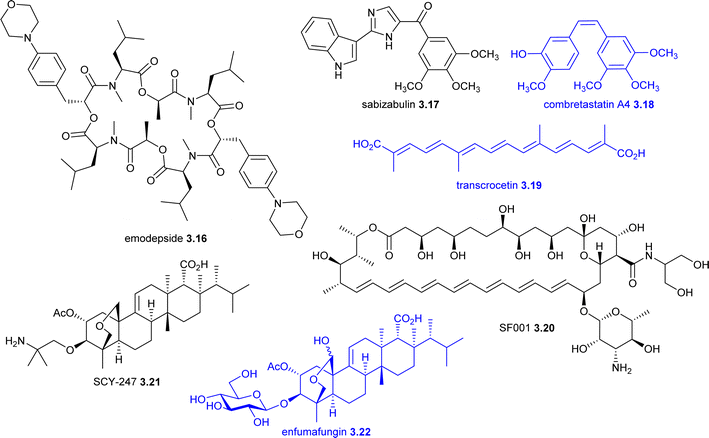
| Emodepside 3.16 | PF1022A (fungus) | NP-D | SS | SLO-1 potassium channel, latrophilin receptor | Phase II (oral), threadworm; onchocerciasis; hookworm and whipworm | Drugs for neglected diseases initiative |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Antiviral | ||||||
| Q-griffithsina (M78Q griffithsin) | Griffithsin (red algae) | NPa | Ia | Mannose N-linked glycan binding | Phase I (nasal spray), COVID-19 prophylaxis | University of Louisville |
| Sabizabulin 3.17 (VERU-111, ABI-231) | Combretastatin A4 3.18 (plant) | NP-D | TS | Tubulin binding | Phase III (oral), ARDS | Veru Inc |
| Transcrocetin 3.19 (LEAF-4L6715) | Transcrocetin (plant) | NP | TS | Oxygen diffusion enhancement | Phase I/II (IV), ARDS; oncologyb | LEAF4Life |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Antifungal | ||||||
| HRS9432c | Echinocandin (fungus) | NP-D | SS | 1,3-β Glucan synthase | Phase II (IV), candidemia and/or invasive candidiasis | Fujian Shengdi Pharmaceutical Co (Jiangsu Hengrui Medicine) |
| PL-18 3.03 | H. pylori ribosomal protein L1 (bacteria) | NP-D | TS | Membrane disruption | Phase Ib/II (suppository), uncomplicated vulvovaginal candidiasis; antibacteriald | Jiangsu ProteLight |
| SF001 3.20 (AM-2-19) | Amphotericin B (actinomycete) | NP-D | SS | Ergosterol binding | Phase I (IV), aspergillosis | Elion Therapeutics |
| SCY-247 3.21 | Enfumafungin 3.22 (fungus) | NP-D | SS | 1,3-β Glucan synthase | Phase I (oral), antifungal | SCYNEXIS |
3.1 Antibacterials
Nafithromycin 3.01 (WCK 4873) is a ketolide with broad spectrum antibacterial activity104–106 being developed by Wockhardt Limited that successfully completed a phase III trial (CTRI/2019/11/021964) in India for community-acquired bacterial pneumonia (CABP).107 On 2 January 2025 (just missing the review's cut-off date), Wockhardt announced the approval of nafithromycin 3.01 for the treatment of CABP in India.108Jiangsu ProteLight Pharmaceutical and Biotechnology are developing two antimicrobial peptides (AMPs), peceleganan 3.02 (PL-5, V681) and PL-18 3.03. Peceleganan 3.02 is a 26-mer α-helical AMP hybrid peptide of cecropin A and melittin B.109,110 It has completed one phase III trial (ChiCTR2100047202)111 and is being investigated in another (ChiCTR2300071255) for the treatment of skin wound infections. An NDA for wound infections has been submitted to the NMPA in China. Peceleganan 3.02 is also being evaluated against diabetic foot infections (phase II, NCT06189638).112 PL-18 3.03 is a 15-mer α-helical AMP derived109,110 from the N-terminus of the Helicobacter pylori ribosomal protein L1 (RpL1)110,113 that has completed a phase-I trial (NCT05340790) for bacterial vaginosis using suppository administration and is currently being evaluated in a phase Ib/II trial (CTR20232467) for uncomplicated vulvovaginal candidiasis.
Zaloganan 3.04 (PLG0206, WLBU2) is a 24 membered AMP being evaluated by Peptilogics that was inspired by LL37, a human AMP, that has broad spectrum activity against Gram-positive (G+ve) and G-ve bacteria, including biofilms.114–117 Zaloganan 3.04 was evaluated in a phase I trial (NCT05137314) for periprosthetic joint infection (PJI) used intraoperatively by local irrigation, along with an IV administered phase I trial (ACTRN12618001920280).118 CARB-X recently announced that they are supporting the development of a slow release formulation of 3.04 to treat fracture-related infections.119
HRS-8427 (structure not disclosed) is a cefiderocol 2.23 derivative120–122 being evaluated a phase III trial for cUTI (NCT06569056) by Jiangsu Hengrui Medicine Co. Cefiderocol 2.23 (Section 2.1) is a cephalosporin that incorporates an iron siderophore to enhance cell uptake first approved in 2019 for the treatment of cUTIs.123,124
Benapenem 3.05 is an IV administered carbapenem structurally related to ertapenem that has completed a phase-II/III trial for cUTI in May 2020 (NCT04505683) in China.125,126 Shanghai Pharmaceuticals Holdings licensed 3.05 in 2021 and it is listed as in development in their 2023 Annual Report.127
Sanfetrinem cilexetil 3.06 (GV-104326) is a prodrug of the synthetic tricyclic carbapenem (trinem class),128,129 which successfully completed a phase II trial in the 1990s for respiratory infections but development was halted for commercial considerations.130 An early bactericidal activity (EBA) phase II trial (NCT05388448) of 3.06 started in May 2022.131 There has been other interest in re-purposing β-lactams for the treatment of TB.132,133
Rifasutenizol 3.07 (TNP-2198) is a rifamycin-metronidazole hybrid134 being developed by TenNor, which is orally delivered but not systematically absorbed. Rifasutenizol 3.07 recently completed a phase III trial (NCT05857163) in combination with rabeprazole and amoxicillin for patients with treatment of participants with H. pylori infections.135
Rifaquizinone 3.08 (TNP-2092, CBR 2092) is a rifamycin-quinolizine (lead ABT-719, fluoroquinolone-like) hybrid136–138 being developed by TenNor Therapeutics for the treatment of G+ve infections. Rifaquizinone 3.08 has completed phase II trials using IV dosing for G+ve acute bacterial skin and skin structure infections (ABSSSI) (NCT03964493) and PJI (NCT04294862) and phase III trials are planned for both indications.139 Rifaquizinone 3.08 has also been dosed orally for the treatment of gut related issues like cirrhotic hepatic encephalopathy (phase I/II, NCT06135675) and diarrhoea-predominant irritable bowel syndrome (phase I). The mode of action (MoA) of 3.08 is a combination of its hybrid components: RNA polymerase (rifamycin) and DNA gyrase and topoisomerase IV (quinolizinone).137
Upleganan 3.09 (EVER206, SPR206) is a polymyxin analogue being developed by Spero Therapeutics (USA), Everest Medicines (Greater China, South Korea and certain Southeast Asian countries) and Pfizer Europe (rest of the world) for the treatment of MDR G-ve pathogens. Upleganan 3.09 has completed four phase I trials (NCT03792308, NCT04868292, NCT04865393 and ChiCTR2200056692)140–142 and phase II trials are planned.
MRX-8 (structure not disclosed) is a polymyxin analogue being developed by MicuRx with a fatty acid tail linked via a polar ester group to form a ‘soft’ prodrug, which hydrolyses to MRX-8039 3.14.143–145 An example from patent US9971394 is compound 34 3.15.146 MRX-8 has completed two phase-I trials: NCT04649541 and CRT20221866.147
BRII-693 3.10 (QPX-9003, F365) is a synthetic polymyxin derivative discovered by researchers at Monash University that is being developed as a treatment for P. aeruginosa and A. baumannii infections.148 A phase I trial (NCT04808414) was competed in July 2022 by Qpex Pharma (now part of Shionogi), who subsequently licensed the worldwide development to Brii Biosciences.149
ASK0912 3.11 (polymyxin S2, IMB-0912) is a naturally occurring polymyxin, which was shown to have lower renal cytotoxicity and acute toxicity than polymyxins B and E.150 ASK0912 3.11 showed promising in vivo activity in an A. baumannii mouse model151 and has been evaluated by Jiangsu Aosaikang Pharmaceutical in phase I trials: CTR20243311 (ongoing) and CTR20222379 (completed).122,152
Zifanocycline 3.12 (KBP-7072) is an aminomethylcycline tetracycline derivative being developed by KBP BioSciences and has now finished four phase I trials (latest was NCT05507463). As with other tetracyclines, 3.12 is a protein synthesise inhibitor with broad spectrum antibacterial activity.153–155
EBC-1013 3.13, a semi-synthetic derivative156 of tigilanol tiglate 3.14 (Section 7.1),157,158 is being evaluated by QBiotics in a phase-I trial (ACTRN12624000544572) as a potential treatment for wound healing in venous leg ulcers.156 This is the only non-traditional NP-D antibacterial currently in clinical trials. EBC-1013 3.13 is also under development for managing acute and chronic wounds in canines and horses. Mechanistically, 3.13 interacts with bacterial cell walls, causing permeabilization of both G+ve and G-ve cell walls, disrupting established biofilms by targeting the extracellular polymeric substance matrix, and activating immune cells to produce reactive oxygen species.156 In a diabetic chronic wound mouse model, EBC-1013 3.13 treatment led to increased expression of host-defence peptides, modulated cytokine levels, stimulated immune cell activation and enhanced wound closure.156
RG6436 (GGDC-0829) (structure not disclosed) is an arylomycin derivative that is being evaluated in a phase I trial (ISRCTN18049481) by Genentech (Roche).159–161 Arylomycins such as arylomycin A-C163.15 are Streptomyces-derived inhibitors of SPase,162,163 an enzyme critical for bacterial viability and virulence that hydrolyses the N-terminal signal peptides of secreted proteins.164–166 RG-6436 is an inhibitor of LepB, which is an E. coli Type I signal peptidase (SPase), and is being developed as an IV therapeutic for cUTI.167 Another arylomycin LepB inhibitor, RG-6319 (structure not disclosed), completed two phase I trials (ISRCTN15259645 and ISRCTN16073754) but development was discontinued in favour of RG6436.168
3.2 Antiparasitics, antivirals and antifungals
Emodepside 3.16 (BAY 44-4400) is a bis-morpholine derivative of PF1022A being developed as a human anthelmintic by the Drugs for Neglected Diseases initiative (DNDi). Emodepside 3.16 has been used in combination with praziquantel for the treatment of parasitic nematodes in cats and dogs since 2005.169 Emodepside 3.16 is being evaluated in a phase II trial (NCT06373835) against threadworm (Strongyloides stercoralis) and a phase II trial (NCT05180461) against onchocerciasis,170–172 while it has completed phase II trials for the treatment of human whipworm (Trichuris trichiura), hookworm (NCT05017194) and hookworm alone (NCT05538767).173 PF1022A was first reported in 1992 from Rosellinia sp.174 with in vivo activity against the roundworm Ascaridia galli but no in vitro against bacteria, yeasts and other fungi.175–177 The mode of action of PF1022A primarily involves the activation of the SLO-1 potassium channels178,179 and interaction with latrophilin receptors,180,181 which leads to worm paralysis and GABAergic transmission modulation.182Griffithsin is a 121 amino acid lectin isolated from the New Zealand red algae Griffithsia sp. with picomolar antiviral activity against HIV, herpes simplex type 2 virus, and hepatitis C virus. Griffithsin, which can be produced recombinantly,183,184 inhibits viral entry by initially binding to terminal mannose residues of mannose-rich oligosaccharides on the virion surface, followed by glycan crosslinking.185,186 An intranasal spray of Q-griffithsin (M78Q mutation for improved oxidative stability) has been evaluated by the University of Louisville in two phase I trials (NCT05437029 and NCT05122260) for COVID-19 prophylaxis.187 Griffithsin has an excellent safety profile and has been tested earlier in combination with carrageenan for potential use against sexually transmitted infections in a phase-I trial (NCT02875119).188
Sabizabulin 3.17 (VERU-111, ABI-231)189 is a combretastatin A4 3.18 inspired analogue being developed by Veru Inc that completed a phase III trial (NCT04842747) in mid-2022 for in COVID-19 patients at high risk for acute respiratory distress syndrome (ARDS). An interim analysis showed that 3.17 treatment resulted in a 24.9% absolute reduction in deaths compared to a placebo,190 but further development will rely upon co-funding and clinical need. Sabizabulin 3.17 was also evaluated as a treatment for metastatic castration-resistant prostate cancer191 but a phase III trial (NCT04844749) was halted in 2023 for strategic reasons. Sabizabulin 3.17 disrupts the microtubule polymerisation (colchicine binding site), which leads to leads to cell cycle arrest and apoptosis of rapidly dividing cells.189 This mechanism can interfere with the distribution of coronavirus S proteins, which impedes intracellular viral particle transport, assembly and replication.192,193
Transcrocetin 3.19, which is also being evaluated in anti-cancer trials (Section 7.1), and its glycosylated analogues are yellow apocarotenoids derived from plants such as Gardenia jasminoides (Cape Jasmine) and Crocus sativus (saffron).194 These compounds are biosynthesised through enzymatic cleavage of zeaxanthin, followed by aldehyde reduction.195–197 A liposomal formulation of 3.19 (LEAF-4L6715), developed by Leaf4Life, has completed a phase I/II trial (NCT04378920) for COVID-19-related ARDS,198 and a phase III trial (NCT06640777) has been registered but has not yet commenced. Transcrocetin 3.19 has been shown to enhance systemic oxygen diffusion by reducing plasma viscosity and potentially altering the structure and organization of water molecules in plasma.194,199 This modification promotes the formation of additional hydrogen bonds among water molecules, facilitating more efficient oxygen transport along diffusion gradients in both water and plasma. The disodium salt of 3.19 underwent a phase II trial (NCT04573322) for COVID-19,200 sponsored by Diffusion Pharmaceuticals (now part of CervoMed). However, further development of this compound appears to have been discontinued.
SF001 3.20 (AM-2-19), an N-1,3-dihydroxypropan-2-yl amide derivative of amphotericin B, is being evaluated in a phase I trial by Elion Therapeutics using an IV formulation as a potential treatment for invasive aspergillosis.201,202 Oral formulations are also under evaluation. It has been proposed that cholesterol extraction contributes to the human renal toxicity of amphotericin B, and developing a polyene with selective ergosterol extraction properties could significantly reduce these side effects.203,204 In line with this selectivity objective, 3.20 has shown enhanced ergosterol extraction properties, along with renal-sparing characteristics in mice and primary human renal cells.205
HRS9432 (structure not disclosed) is an anidulafungin derivative121 being evaluated by Fujian Suncadia Pharmaceuticals Co in a phase-II (NCT06194201) for candidemia and/or invasive candidiasis. Other echinocandins have been approved including rezafungin 2.35 (Section 2) in 2023.48
SCY-247 3.21,206–208 a second-generation fungerp,49 is a derivative of the fungus-derived triterpene enfumafungin 3.22.209 Scynexis announced the start of phase 1 trials (ACTRN12624001393549 and ACTRN12624001392550) for invasive fungal infections in December 2024.210 Ibrexafungerp 2.30, the first antifungal fungerp, received its initial approval in 2021 (Section 2).47,49
4. Compounds undergoing evaluation in neurological diseases
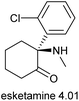
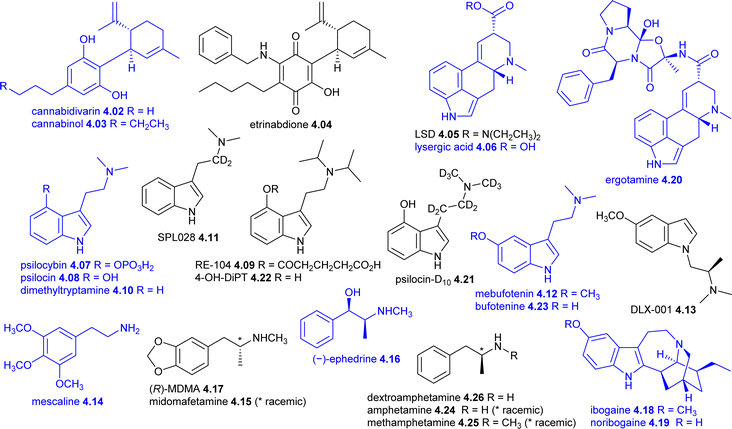
Since the last review,6 there has been a notable rise in the number of psychedelic and hallucinogenic compounds undergoing clinical evaluation. Following research conducted after World War II, psychedelics showed significant potential for treating mental health disorders such as anxiety, depression, and substance use disorder.211 However, this momentum declined in the 1960s due to factors including stricter clinical trial regulations and widespread legal prohibitions. The 2019 approval of esketamine 4.01 as a nasal spray antidepressant by the US FDA, though not NP-D, played a pivotal role in revitalizing interest in the field. This review categorizes compounds under investigation for neurological disorders into two groups: hallucinogens/psychedelics (Section 4.1, Table 6) and other related compounds (Section 4.2, Table 7).| Compound (synonyms) | Lead compound (source) | Classif | Manuf | Mode of action | Development status (administration), disease(s) | Developer |
|---|---|---|---|---|---|---|
| a Abbreviations: ADHD, attention-deficit hyperactivity disorder; CB1, cannabinoid receptor CB1; CB2, cannabinoid receptor CB2; DAT dopamine transporter; 5-HT; 5-hydroxytryptamine (serotonin) receptor; mAChRs, muscarinic acetylcholine receptors; NMDA, N-methyl-D-aspartate receptor; PPARγ, peroxisome proliferator-activated receptor gamma; PTSD, post-traumatic stress disorder; SERT, serotonin reuptake transporter; TRPV1, transient receptor potential cation channel subfamily V member 1. | ||||||
| Cannabidivarin 4.02 (CBDV, GWP42006) | Cannabinol 4.03 (plant) | NP | TS | CB1 and TRPV1 | Phase II (oral), autistic spectrum disorder, Prader–Willi syndrome | Montefiore Medical Center |
| Etrinabdione 4.04 (EHP-101; VCE-004.8) | Cannabinol 4.03 (plant) | NP-D | TS | CB2 and PPARγ receptor agonist | Phase II (oral), peripheral arterial disorders | VivaCell Biotechnology España |
| LSD 4.05 | Lysergic acid 4.06 (fungus) | NP-D | SS | 5-HT2A | Phase III (oral), generalised anxiety disorder; phase II, ADHD | MindMed and MindBio Therapeutics |
| Psilocybin 4.07 | Psilocybin 4.07 (fungus) | NP | TS | 5-HT2A | Phase I to phase III (oral), treatment-resistant depression | Various companies |
| Psilocin 4.08 (ELE-101, ELE-Psilo) | Psilocin 4.08 (fungus) | NP | TS | 5-HT2A | Phase I/IIa (IV), treatment-resistant depression; also, oral dosing | Beckley Psytech, Alera Pharma and Tryptamine Therapeutics |
| CYB003 (deuterated psilocin, structure not disclosed) | Psilocin 4.08 (fungus) | NP | TS | 5-HT2A | Phase I/IIa (oral), major depressive disorder | Cybin |
| RE-104 4.09 | Psilocin 4.08 (fungus) | NP-D | TS | 5-HT2A | Phase II (sc), moderate-to-severe postpartum depression | Reunion Neuroscience |
| GM-2505 (structure not disclosed) | Psilocybin 4.07/DMT 4.10 (plants) | NP-D | TS | 5-HT2A and 5-HT2C | Phase II (IV), major depressive disorder | Gilgamesh Pharmaceuticals |
| Dimethyltryptamine 4.10 (DMT, VLS-01, SPL026) | DMT 4.10 (plant) | NP | TS | 5-HT2A | Phase I (oral), treatment-resistant depression | Atai Life Sciences and others |
| CYB004 (deuterated DMT, structure not disclosed) | DMT 4.10 (plant) | NP | TS | 5-HT2A | Phase II (oral), generalised anxiety disorder | Cybin |
| SPL028 4.11 (D2-DMT fumarate) | DMT 4.10 (plant) | NP | TS | 5-HT2A | Phase I/II trial (IM and IV), major depressive disorder | Cybin (Small Pharma) |
| Mebufotenin 4.12 (5-MeO-DMT) | Mebufotenin 4.12 (plant) | NP | TS | 5-HT2A | Phase III (oral), treatment-resistant depression | Atai Life Sciences |
| DLX-1 4.13 | DMT 4.10 (plant) | NP-D | TS | 5-HT2A | Phase I (oral), major depressive disorder | Delix Therapeutics |
| Mescaline 4.14 | Mescaline 4.14 (plant) | NP | TS | 5-HT2A | Phase I (oral), psychometric tool | University Hospital, Basel |
| Midomafetamine 4.15 (MDMA) | Ephedrine 4.16 (plant) | NP-D | TS | 5-HT2A | Phase III (oral), PTSD | Lykos Therapeutics and others |
| EMP-01 and MM402 4.17 (R-MDMA) | Ephedrine 4.16 (plant) | NP-D | TS | 5-HT2A | Phase I (oral), major depressive disorder | Atai Life Sciences and MindMed |
| Ibogaine 4.18 (DMX-1002, IBX-210) | Ibogaine 4.18 (plant) | NP | TS/SS | NMDA, κ, μ and δ opioid, σ-1 and σ-2, M1 and M2 mAChRs, SERT and DAT | Phase II (oral), alcoholism and phase I (oral), opioid related disorders | University of Sao Paulo and Atai Life Sciences |
| Noribogaine 4.19 | Ibogaine 4.18 (plant) | NP | TS/SS | NMDA, κ, μ and δ opioid, σ-1 and σ-2, M1 and M2 mAChRs, SERT and DAT | Phase I (oral), alcoholism | Atai Life Sciences |
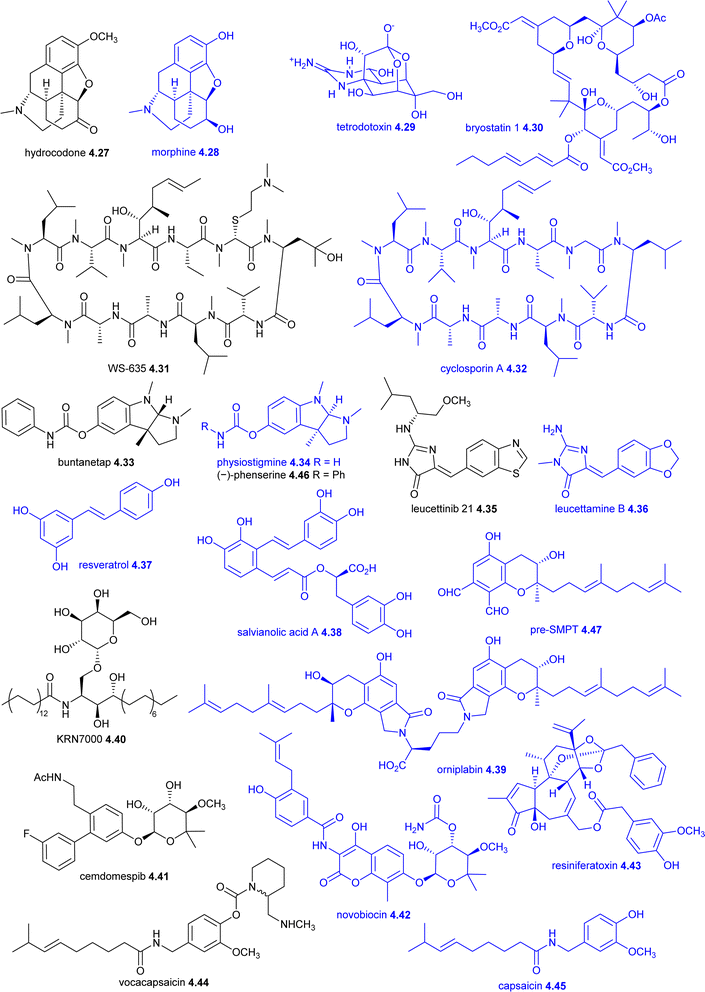
| Compound (synonyms) | Lead compound (source) | Classif | Manuf | Mode of action | Development status (administration), disease(s) | Developer |
|---|---|---|---|---|---|---|
| a These assignments are tentative. Abbreviations: DYRK, dual-specificity tyrosine-regulated kinase; TRPV1, transient receptor potential cation channel subfamily V member 1. | ||||||
| O2P hydrocodone 4.27 prodrugs ETR028 and ETR029 (structures not disclosed) | Morphine 4.28 (plant) | NP-D | SS | μ opioid agonist (predominant), also δ opioid agonist | Phase I (oral), acute pain | Elysium Therapeutics |
| Tetrodotoxin 4.29 | Tetrodotoxin 4.29 (pufferfish and other organisms) | NP | I | Nav1.7 and Nav1.6 sodium channel blocker | Phase II (sc), chemotherapy induced neuropathic and cancer related pain | Dogwood Therapeutics |
| Bryostatin 1 4.30 | Bryostatin 1 4.30 (bryozoan/bacteria) | NP | I/TS | Protein kinase C | Phase II (IV), Alzheimer's disease | Synaptogenix |
| WS635 4.31 (SCY635) | Cyclosporin 4.32 (fungus) | NP-D | TS | Cyclophilin inhibitor | Phase I (oral), postoperative delirium | Waterstone Pharmaceuticals |
| Buntanetap 4.33 (ANVS401, (+)-phenserine) | (+)-physostigmine 4.34 (plant) | NP-D | TS | Suppresses translation of amyloid precursor protein, tau, α-synuclein and other neurotoxic aggregating proteins | Phase III (oral), Alzheimer's disease, Parkinson's disease | Annovis Bio |
| LCTB-21 4.35 (leucettinib 21) | Leucettamine B 4.36 (sponge) | NP-D | TS | DYRK (kinase) | Phase I (oral), Alzheimer's disease; down syndrome | Perha pharmaceuticals |
| Trans-resveratrol 4.37 (JNS108, JNS101, JNS102, JNS107) | Trans-resveratrol 4.37 (plant) | NP | TS | Various | Phase II (oral), mild cognitive impairment/early Alzheimer's disease, Friedreich's ataxia | Jupiter Neurosciences |
| Salvianolic acid A 4.38 | Salvianolic acid A 4.38 (plant) | NP | Ia | Anti-inflammatory | Phase II (IV), acute ischemic cerebrovascular disease | Shanghai Pharmaceuticals Holding/Chinese Academy of Medical Sciences |
| Orniplabin 4.39 (JX10, TMS-007, BIIB-131, SMTP-007) | Orniplabin 4.39 (fungus) | NP | I | Plasminogen-fibrin binding enhancement | Phase II (IV), stroke | Corxel Pharmaceuticals/TMS |
| TMS-008 (structure not disclosed) | Orniplabin 4.39 (fungus) | NPa | Ia | Plasminogen-fibrin binding enhancement | Phase I (IV), acute kidney injury | TMS |
| KRN 7000 4.40 (RGI-2001, αGalCer, AGL 582) | Agelasphin (sponge) | NP-D | TS | Natural killer T cells activation | Phase II (IV), prevention of graft-versus-host disease | REGiMMUNE |
| NLY01 (C40-tPEG50K-Ex4-Cys, pegylated exenatide-4) | Exendin-4 (lizard) | NP-D | TS | Glucagon-like peptide-1 receptor (GLP-1R) agonist | Phase II (IV), Parkinson's disease; phase I (IV), Alzheimer's | Neuraly |
| Cemdomespib 4.41 (RTA 901, KU-596) | Novobiocin 4.42 (actinomycete) | NP-D | TS | HSP90 heat-shock inhibitor | Phase II (oral), diabetic neuropathies | Biogen |
| Resiniferatoxin 4.43 (Lopain; MTX-071; RTX; RTX-GRT7039; MCP 101). | Resiniferatoxin 4.43 (plant) | NP | I | TRPV1 ion channel | Phase II (intra-articular injection) musculoskeletal pain | Grünenthal/Shionogi |
| Vocacapsaicin 4.44 (CA-008) | Capsaicin 4.45 (plant) | NP-D | TS | TRPV1 ion channel | Phase II (topical/IV), postoperative pain | Concentric Analgesics |
4.1 Hallucinogens/psychedelics
Cannabidivarin 4.02 (CBDV, GWP42006) is an alkyl homologue of cannabidiol 4.03 found in most Cannabis strains212,213 with similar biological properties to cannabidiol 4.03.214,215 It is currently being investigated in phase II clinical trials by Montefiore Medical Center (NY, USA) for children with autism spectrum disorder (NCT03202303) and individuals with Prader–Willi syndrome (NCT03848481). Previously, Jazz Pharmaceuticals evaluated cannabidivarin 4.02 in a phase II trial (NCT02365610) for participants with inadequately controlled focal seizures; although it was well tolerated, no efficacy was observed, potentially due to a high placebo response.216Etrinabdione 4.04 (EHP-101; VCE-004.8) is a synthetic cannabidiol 4.03-related cannabinoquinone being evaluated by VivaCell Biotechnology España in a phase II trial (CTIS2023-509452-34-00) for patients with peripheral arterial disease. Previously, its clinical development for diffuse scleroderma (NCT04166552) was discontinued in September 2023 following the closure of Emerald Health Pharmaceuticals. Etrinabdione 4.04 has been characterized as a dual agonist of the CB2 and PPARγ receptors,217 and its discovery has been documented.218 Recent studies suggest it may also be a potential candidate for delayed treatment of ischemic stroke.219
MindMed is developing LSD 4.05 (lysergic acid diethylamide, lysergide) as its D-tartrate salt (code MM-120) and have completed phase II trials for ADHD (NCT05200936) and generalised anxiety disorder (NCT05407064).220 Two phase III trials (NCT06809595 and NCT06741228) for generalised anxiety disorder started recently. Another company, MindBio Therapeutics is investigating microdoses of LSD 4.05 (coded MB22001) for Major Depressive Disorder (open label phase IIa), and phase IIb trials for Major Depressive Disorder and for helping to decrease end of life distress related depression and anxiety for patients with advanced stage cancer.221 Like other psychedelics, LSD 4.05 acts on multiple receptors in the brain, with its primary effects mediated through the serotonin 5-hydroxytryptamine 2A receptor (5-HT2AR).222 It has played a significant role in the resurgence of interest in psychedelic substances.223,224 The history of LSD's discovery is well documented. Ergot alkaloids are derived from Claviceps purpurea, a fungus that readily grows on grain grasses that can cause severe poisoning when consumed.225–227 Sandoz isolated the first pure ergot alkaloid, ergotamine 4.20, in 1917.228 Research on ergot alkaloids continued with Albert Hofmann, who first synthesised LSD from lysergic acid 4.06 in 1938 but only discovered its psychedelic properties upon resynthesising it in 1943.229–231 Sandoz started to supply LSD 4.05 for research purposes in 1947 under the brand name Delysid but it was never commercially marketed.229,232 While LSD 4.05 initially showed promise for psychiatric applications, its use was widely prohibited by the late 1960s.211
Psilocybin 4.07 is the phosphate derivative of psilocin 4.08 (8-hydroxytrypamine), a psychedelic compound found in Psilocybe spp. (‘magic mushrooms’) and other fungi with psychoactive properties.233 Used by various cultures for millennia,234 psilocybin 4.07 was first isolated from P. mexicana in 1958 by Hofmann and colleagues.235–238 Psilocybin 4.07 is a prodrug that rapidly hydrolyses to psilocin 4.08 after ingestion and interacts with several different serotonin neuroreceptors, including the 5-HT1A, 5-HT1D, 5-HT2A, 5HT2B, and 5-HT2C subtypes.234 As with other psychedelics, psilocybin 4.07 and psilocin 4.08 were classified as controlled substances in the 1960s, limiting research and their development.223,224 However, interest has resurged, with over 200 clinical trials registered on https://clinicaltrials.gov/, most initiated in the past five years. Australia became the first country to permit registered psychiatrists to prescribe psilocybin 4.07 (for treatment-resistant depression) though this does not constitute formal drug approval. Several companies and research organisations are conducting advanced clinical studies:
• Compass pathways is evaluating psilocybin 4.07 (code COMP360) in two phase III trials for treatment-resistant depression (NCT05624268, NCT05624268) and phase II trials for post-traumatic stress disorder (PTSD) (NCT05312151) and anorexia nervosa (NCT05481736).239
• Usona Institute, a non-profit, recently launched a phase III trial (NCT06308653) for major depressive disorder.
• Sunstone Medical is conducting a phase II trial (NCT05947383) for major depressive disorder.
• Tryptamine Therapeutics has three ongoing phase II trials (code TRP-8802) for fibromyalgia (NCT05128162), binge eating disorder (NCT05035927), and irritable bowel syndrome (NCT06206265).
• Clairvoyant Therapeutics is leading a phase II trial (NCT05646303) on psilocybin-assisted psychotherapy for alcohol use disorder.240
• Apex Laboratories (code APEX-90) has begun a phase II trial (NCT06141876) investigating psilocybin-assisted psychotherapy for severe depression in patients with PTSD.
• Incannex Healthcare and Monash University (code PSX-001) completed a phase II trial (ACTRN12621001358831) in Australia for psilocybin-assisted psychotherapy in generalized anxiety disorder and plan US and UK phase II studies.241
• Nova Mentis Life Science initiated a phase IIa trial (NCT05832255) for fragile X syndrome, though it is currently on hold.
The benzoate salt of psilocin 4.08 is being evaluated in a phase I/IIa trial (NCT05434156) by Beckley Psytech (code ELE-101) for the treatment-resistant depression using an IV formulation. The mucic acid salt (Alera Pharma and Lobe Sciences, code L-130) completed a phase I trial (NCT06035900) in 2023 using oral dosing and there are plans for a further study against chronic cluster headaches. Tryptamine Therapeutics also has an IV formulation (code TRP-8803) in phase I (ACTRN12624000547549). In addition, Cybin is advancing CYB003, a deuterated psilocin that recently completed a phase I/II trial (NCT05385783) for MDD.242 While its structure has not been disclosed, Cybin's patents include psilocin-D10 4.21 and related analogues.243
RE-104 4.09 (FT-104, isoprocin glutarate),244 which is a fast acting prodrug of the psilocin analogue 4-hydroxy-N,N-diisopropyltryptamine 4.22 (4-OH-DiPT),245,246 being developed by Reunion Neuroscience. RE-104 4.09 is being evaluated in a phase II clinical trial (NCT06342310) for participants with moderate-to-severe postpartum depression. The intellectual property surrounding RE-104 4.09 was the subject of contention, with two companies asserting ownership.247
GM-2505 (structure not disclosed) is a 5-HT2A/5-HT2C receptor agonist with 5-HT releasing activity being developed by Gilgamesh Pharmaceuticals.248 GM-2505 is a psilocybin/tryptamine analogue with a shorter psychedelic experience249,250 being evaluated a phase-II clinical trial (NCT06236880) for major depressive disorder.
Dimethyltryptamine 4.10 (DMT) and mebufotenin 4.12 (5-OMe-DMT, O-methyl-bufotenin) are components of plant-derived products used in Central and South America, often as part of religious and shamanic rituals,251 being investigated in a variety of clinical trials. Mebufotenin 4.12 and bufotenine 4.23 (5-OH-DMT) are also produced in the parotid gland secretions of toads.252,253 Dimethyltryptamine 4.10 is being evaluated for depression in a phase II trial (NCT06094907, vaporizer device administration) by the Universidade Federal do Rio Grande do Norte and a phase II trial (code VLS-01, NCT06524830, buccal administration) by atai Life Sciences.
CYB004 (structure not disclosed) is a deuterated dimethyltryptamine 4.10 derivative being evaluated by Cybin in a phase II trial (NCT06051721, IM) with participants with generalized anxiety disorder. Cybin acquired Small Pharma in October 2023 who were developing SPL026 (DMT 4.10 fumarate) and its deuterated analogue SPL028 4.11 (D2-DMT fumarate).254 SPL026 has completed a phase I/II trial (NCT04673383, IM and IV) in patients with major depressive disorder.255,256 SPL028 4.11 (IM and IV) was well tolerated in a phase I trial and the average psychedelic experience was around 90 min, compared to around 45 min for the non-deuterated SPL026.242
Mebufotenin 4.12 is being evaluated in the following clinical trials: phase II (NCT05660642, intranasal, code BPL-003) for treatment-resistant depression by Beckley Psytech (atai Life Sciences); phase II (NCT05839509, inhalation, code GH001) for bipolar II disorder, phase II (NCT05804708, inhalation) for postpartum depression, and phase I/II trial (NCT04698603, inhalation) for treatment-resistant depression by GH Research; and phase I (NCT05753956, IV, code GH002) by GH Research.
DLX-1 4.13 (AAZ-A 154) is an isotryptamine derivative257,258 being developed by Delix Therapeutics that has successfully completed a phase I trial showing brain activity with no hallucinogenic effects.259
Mescaline 4.14 (trimethoxyphenylethylamine) is found in several cacti, most notably the North American peyote (Lophophora williamsii) and the South American wachuma (Trichocereus pachanoi, T. peruvianus, and T. bridgesii), which has been used as a hallucinogen for thousands of years.260,261 The hallucinogenic activities of mescaline 4.14 and other phenylethylamines are mediated through the serotonin 2A receptor (5-HT2AR).261–263 University Hospital in Basel have evaluated mescaline 4.14 in phase I trials as a potential psychometric tool alongside LSD 4.05 and psilocybin 4.07 (NCT04227756), as well as exploring the role of the 5-HT2AR in psychedelic effects (NCT04849013). Other organisations are planning to evaluate mescaline in clinical trials.
Lykos Therapeutics has been developing midomafetamine 4.15 (MDMA, ‘ecstasy’) and has completed several phase III clinical trials for PTSD (NCT01793610, NCT04077437 and NCT04714359), culminating in an NDA submission.264 However, in August 2024, the US FDA declined the application and requested additional clinical trials.265,266 Meanwhile, the University of Southern California (NCT06189027) and San Diego Veterans Healthcare System (NCT05979844) are conducting phase III trials for PTSD, while there are many other phase I and II trials in progress. Prior to this regulatory setback, Australia became the first country to permit registered psychiatrists to prescribe midomafetamine 4.15 (for PTSD) in July 2023, although this does not constitute formal drug approval.267,268 The R enantiomer of midomafetamine, (R)-MDMA 4.17,268 is currently being investigated in phase I trials as a potential treatment for major depressive disorder by MindMed (ISRCTN13877948, code MM402) and atai Life Sciences (code EMP-01). Although midomafetamine 4.15 was first synthesised in 1912 when looking for adrenaline- and ephedrine-like compounds, it wasn't until the 1970s that it was more widely explored for its psychotomimetic/empathogenic activity.269,270 Additionally, several organisations are exploring MDMA-like empathogens in preclinical development. How amphetamines are categorised as NP-D is detailed as follows. Ephedrine 4.16 is a phenylethylamine derivative first reported from the traditional Chinese medicine Ephedra sinica (ma huang) in 1887, which also contains pseudoephedrine and norephedrine.271,272 Eli Lilly introduced ephedrine 4.16 onto the market in the US in 1926 for nasal congestion relief. In 1929, Gordon Alles synthesised amphetamine 4.24 and patented its salt form.273 Researchers at Smith, Kline & French launched the free base form of amphetamine 4.24 in 1933, which was administered using inhalation. In 1934, Alles finalised a licensing deal with Smith, Kline & French and Merck for 5% royalties on sales of these salts.273 Prior to Alles' work, Akira Ogata synthesised a crystalline form of deoxyephedrine, also known as methamphetamine 4.25, in 1919 (ref. 274) and was introduced into the German market in 1938 as Pervitin. Amphetamine 4.24 and dextroamphetamine 4.26 were used by Allied forces during the Second World War as stimulants, while the Germans used Pervitin.275,276 Quite a number amphetamine derivatives were marketed as weight loss drugs in the next 25 years including dextroamphetamine 4.26, which was approved in 1944 and is still used for narcolepsy.277,278
Ibogaine 4.18 is an indole alkaloid isolated from the West Central African bush, (Tabernanthe iboga syn. T. manii), and is used by the Bwiti and Mbiri as a remedy for fatigue, hunger, and thirst at lower doses, and for initiation rites and religious ceremonies at higher doses.279–282 Ibogaine 4.18 is considered to be an ‘oneirogen’, as it produces and/or enhances dreamlike states of consciousness. Ibogaine 4.18 was first isolated from root bark of T. iboga in 1901, its structure was determined in 1958 and was confirmed in 1960 by X-ray crystallography.280,283,284 A T. iboga root bark-derived alkaloid extract called Lambarène, which was named after the capital city of Moyen-Ogooué in modern day Gabon, was used in France from 1930 as a “neuromuscular stimulant” recommended for indications that included fatigue, depression, and infectious disease recovery.280,281 However, sales of Lambarène were discontinued in 1970 when 4.18 was classed by the International Olympic Committee as a doping agent.280 Ibogaine 4.18 was also categorised by the US FDA as a Schedule I drug (‘drugs with no currently accepted medical use and a high potential for abuse’) in the same year.280 Despite evidence from the 1960s that showed 4.18 had promise for the treatment of substance abuse, these restrictions stymied clinical research for many years.211,223,224,282 Ibogaine 4.18 and its major metabolite noribogaine 4.19 have pharmacologically complex MoAs with moderate-to-weak affinity for a number of neurotransmitter receptors such as N-methyl-D-aspartate (NMDA), κ, μ and δ opioid, σ-1 and σ-2, M1 and M2 muscarinic, nicotinic acetylcholine, serotonin transporter (SERT) and dopamine transporter (DAT).282 Presently, ibogaine 4.18 is being clinically evaluated by several different organisations, but higher doses have been associated with neurological and cardiac (QT interval, hERG channel inhibition) side effects.224,285,286 University of Sao Paulo researchers are evaluating ibogaine 4.18 in a phase II trial (NCT03380728) as a treatment for alcoholism, while atai Life Sciences plan to start a phase I/IIa trial for opioid related disorders using IV dosing (code DMX-1002).287 Atai acquired DemeRx IB in October 2023 who had started a similar trial (NCT05029401) in 2021 with oral administration (code DMX-IB 201).286 A open-label clinical study (NCT04313712) of ibogaine 4.18 (oral) and magnesium sulfate (IV), which was used to decrease the QT interval, of military veterans with traumatic brain injuries showed promising results.288
Noribogaine 4.19 (DMX-1002, 12-hydroxy-ibogamine) has been evaluated by DemeRx NB (atai Life Sciences) in a phase I clinical trial (NCT06480981) with the aim of using it to help combat alcoholism.
4.2 Other compounds undergoing evaluation in neurological diseases
Recently, Elysium Therapeutics completed a phase I trial (NCT05572190) evaluating two hydrocodone 4.27 prodrugs, ETR028 and ETR029 (structures not disclosed), as well as their combination for acute pain. Hydrocodone 4.27, a semi-synthetic derivative of morphine 4.28, is commonly used for pain management and as a cough suppressant in various drug formulations.289 According to an Elysium press release,290 the prodrugs were well-tolerated in the trial and successfully delivered therapeutically relevant hydrocodone doses while maintaining drug levels designed to reduce the risk of abuse and fatal overdose.Tetrodotoxin 4.29 is a neurotoxin first isolated from the pufferfish and later from other organisms with high affinity for voltage-gated sodium (Nav) channels.291–293 A 1922 paper reported that a 1% tetrodotoxin 4.29 solution was used in Japan to empirically treat various diseases including pain relief.294 Tetrodotoxin 4.29 (Halneuron®) is being evaluated by Dogwood Therapeutics (formally WEX Pharmaceuticals) in a phase II trial for chemotherapy induced neuropathic pain (NCT05359133) using sc dosing. Previous phase II and III trials have been conducted, and a meta-analysis found that 4.29 exhibited strong analgesic activity without signs of euphoria, addiction, or withdrawal.295 However, while the risk of serious adverse effects was not elevated, there was an increased incidence of non-severe side effects.295
Bryostatin 1 4.30 is a macrocyclic lactone anticancer agent first reported from the bryozoan Bulgula neritina by Pettit and co-workers in 1982,296,297 which was later found to be produced by a bacterial symbiont.298,299 Bryostatin 1 4.30 is a potent protein kinase C (PKC) inhibitor, which has been investigated in over 35 phase I and II anticancer trials, but was not considered to be effective enough to continue development.300,301 PKC is involved in other cellular processes and bryostatin 1 4.30 has also been evaluated in other therapeutic areas, primarily Alzheimer's disease.302,303 Synaptogenix (previously Neurotrope Bioscience) has evaluated bryostatin 4.30 in several phase II trials for patients with moderately severe to severe Alzheimer's disease (NCT00606164, NCT02431468, NCT03560245 and NCT04538066) between 2008 and 2022. Data from the most recent trial, reported in late 2023 showed no significant cognitive decline for the bryostatin 1 4.30 moderately severe cohort throughout the 10-month trial period, while patients receiving the placebo displayed cognitive decline.304,305 Synaptogenix and The Cleveland Clinic are also evaluating bryostatin 1 4.30 in a phase I trial (NCT06190912) for multiple sclerosis (Section 6). Bryostatin-1 4.30 used in clinical trials is produced by isolation or total synthesis.306,307
The cyclosporin 4.32 analogue WS635 4.31 (SCY635) is a non-immunosuppressive cyclophilin inhibitor308–311 that completed a hepatitis C phase II (NCT01265511) in 2011. In 2014, Scynexis licensed the worldwide rights for 4.31 to Waterstone Pharmaceuticals,312 who recently registered a phase I trial (CTR20243892) in China to evaluate 4.31 as a treatment for post-operative delirium.313
Buntanetap 4.33 (ANVS401, (+)-phenserine, posiphen) is an enantiomeric analogue314–316 of the plant alkaloid (+)-physostigmine 4.34 (ref. 317 and 318) being developed by Annovis Bio. Buntanetap 4.33 has completed a phase II/III trial (NCT05686044) for patients with Alzheimer's disease, a phase III trial (NCT05357989) with patients with early Parkinson's disease and has just started a new phase III trial (NCT06709014) in participants with early Alzheimer's disease.319,320 Buntanetap 4.33 suppresses the translation of the mRNAs of amyloid-β precursor protein (APP), tau, α-synuclein and other neurotoxic aggregating proteins by enhancing the binding of the atypical iron response element in the mRNAs' 5′UTR regions of these neurotoxic proteins to iron regulatory protein 1 (IRP1).321–323 The enantiomer (−)-phenserine 4.46 is a potent acetylcholinesterase (AChE) inhibitor whose development for Alzheimer's disease was stopped after failing to improve cognition or clinical measures in two phase III trials. Buntanetap 4.33 has weaker AChE activity and can be administered at higher doses.316,324
Leucettinib 21 4.35 (LCTB-21) is a tyrosine phosphorylation-regulated kinase (DYRK) inhibitor that was derived from leucettamine B 4.36,325–327 which was originally isolated from the marine sponge Leucetta microraphis.328 DYRK1A is the primary target but there are also some ‘off-target’ effects that may contribute to its biological activity.329 Recently, Perha Pharmaceuticals started a phase I trial (NCT06206824) evaluating 4.35 in healthy volunteers and patients with Down syndrome or Alzheimer's disease.
Trans-resveratrol 4.37 is a plant-derived stilbene, also found in low concentrations in red wine, that has been evaluated in clinical trials in various therapeutic areas: atherosclerosis, cancer, diabetes, hypertension, obesity, and neurodegenerative diseases.330,331 Jupiter Neurosciences are evaluating an oral formulation332 of resveratrol 4.37 in phase II trials for mild cognitive impairment/early Alzheimer's Disease (code JNS108) and Friedreich's Ataxia (code JNS101), as well as phase I trials in mucopolysaccharidosis type 1 (code JNS102) and amyotrophic lateral sclerosis (code JNS107).333
Salvianolic acid A 4.38 was first reported from the traditional Chinese medicine Salvia miltiorrhiza in 1984 (ref. 334) and has anti-inflammatory activity.335,336 Salvianolic acid A 4.38 is being developed by Shanghai Pharmaceuticals Holding and completed a phase II trial (CTR20210544) in June 2023 for diabetic peripheral neuropathy.
There have been over 60 Stachybotrys microspora triprenyl phenol (SMTP) analogues reported from the fungus Stachybotrys microspore,337 which include orniplabin 4.39 (JX10, TMS-007, SMTP-7, BIIB 131),337–340 an ornithine linked dimer of pre-SMTP 4.47.341 The absolute configuration of orniplabin 4.39 was determined by microcrystal electron diffraction (MicroED)342 and confirmed by total synthesis.343 Orniplabin 4.39 has been shown to relax plasminogen conformation and enhance plasminogen-fibrin binding, which leads to plasminogen activation and clot clearance, as well as anti-inflammatory activity through inhibition of soluble epoxide hydrolase.337 TMS have completed a phase IIa trial (JapicCTI-183842) in 2021, though only the phase I data has been published.344 Initially licensed to Biogen in 2021, orniplabin 4.39 has since been re-licensed to Corxel Pharmaceuticals (formerly Ji Xing Pharmaceuticals), which now holds worldwide rights, excluding Japan, where TMS maintains ownership.345,346 Additionally, TMS recently initiated a phase I trial (JPRN-jRCT2031240084) of another SMTP, TMS-008 (structure not disclosed), as a potential treatment for acute kidney injury.347,348
KRN7000 4.40 (αGalCer, AGL 582) is a synthetic α-galactosylceramide349,350 based on a series of agelasphin-type lipids isolated from a marine sponge Agelas mauritianus.351,352 KRN7000 4.40 can activate invariant natural killer T cells, which are responsible for its anticancer and immunology actives.353,354 KRN7000 4.40 was previously evaluated in clinical trials as a potential anticancer and anti-hepatitis B and C agent,355 but development was halted around 2013. REGiMMUNE has developed a liposomal formulation of 4.40, coded as RGI-2001. This formulation has completed a phase II trial (NCT04014790) for the prevention of acute graft-versus-host disease following allogeneic haematopoietic stem cell transplantation.356
NLY01 (C40-tPEG50K-Ex4-Cys)357 is a pegylated exenatide-4 derivative being developed by Neuraly, which was shown to be protective in Parkinson's358 and Alzheimer's disease359 mouse models. Like the parent peptide exenatide-4 (Section 5), NLY01 is a glucagon-like peptide-1 (GLP-1) agonist and has a human half-life of 12.5 days when administered subcutaneously.360 Results recently published from a phase II trial (NCT04154072) for NLY01 in patients with early Parkinson's disease did not reach statistical significance.361–363 NLY01 has also completed a phase I trial in Alzheimer's disease and a phase II trial (NCT04159766) in type 2 diabetes.
Cemdomespib 4.41 (RTA 901, KU-596) is a novobiocin 4.42 inspired HSP90 heat-shock inhibitor (‘novologue’)364 that Biogen (Reata) has been evaluating in a phase II trial (NCT05895552) for diabetic peripheral neuropathic pain. Cemdomespib 4.41 and other novologues have been found to bind to the C-terminus of Hsp90 and promote the expression of Hsp70,365–367 as well as improved neuromuscular function in animal models.368
Resiniferatoxin 4.43 (RTX-GRT7039, MTX-071, MCP 101) is a daphnane-type diterpene with both ortho ester and ester groups that was first isolated from Euphorbia resinifera and E. unispina in 1975 (ref. 369) and later from E. poisonii.370 The structure of resiniferatoxin 4.43 was revised in 1982.371 Resiniferatoxin 4.43 was discovered as a mouse ear irritant that was 1000 times more active than 12-O-tetradecanoylphorbol-13-acetate (TPA),371 which was later shown to be a potent agonist of transient receptor potential cation channel subfamily V member 1 (TRPV1) like capsaicin 4.45.372 Grünenthal and Shionogi are evaluating resiniferatoxin 4.43 in three phase III trials (NCT05248386, NCT05377489 and NCT05449132) for pain associated with knee arthritis using intra-articular injection. Sorrento Therapeutics were also developing resiniferatoxin 4.43 in a phase II trial (NCT04885972) but the development status is uncertain due to the company filing for bankruptcy in February 2023.
Vocacapsaicin 4.44 (CA-008) is a prodrug being developed by Concentric Analgesics, which hydrolyses to capsaicin 4.45 and an inactive cyclic urea after administration.373 Capsaicin 4.45 is the hot tasting compound present in chillies and is an agonist of TRPV1, a non-opioid pain target.374 Patients with osteoarthritis, post-herpetic neuralgia and diabetic polyneuropathy can be treated with high-dose capsaicin 4.45 patches and injections. Vocacapsaicin 4.44 has been locally administered to surgical wounds in phase II clinical trials, with successful outcomes reported for total knee arthroplasty (NCT03731364) and bunionectomy (NCT03599089).375,376
5. Compounds undergoing evaluation in cardiovascular and metabolic diseases
NPs have played a pivotal role in shaping cardiovascular and metabolic disease management. For example, the cholesterol-lowering statins were inspired by the fungal metabolite mevastatin,377,378 the first GLP-1 receptor agonist is exendin-4, a 39-amino acid peptide found in Gila monster lizard (Heloderma suspectum) saliva,357,379 and sodium-glucose co-transporter (SGLT) inhibitors were developed from a plant-derived glycosylated chalcone lead compound, phlorizin 5.03 (Section 2 and this section).380,381 Additionally, plant-based polyphenols, flavonoids, and ω-3 fatty acids provide anti-inflammatory, antioxidant, and lipid-lowering benefits, contributing to the prevention and treatment of atherosclerosis, hypertension, and diabetes. A list of NP-D compounds being developed to treat cardiovascular and metabolic diseases are summarised in Table 8 and described below.| Compound (synonyms) | Lead compound (source) | Classif | Manuf | Mode of action | Development status (administration), disease(s) | Developer |
|---|---|---|---|---|---|---|
| a Not reported whether berberine is isolated or synthesised for clinical trials. Abbreviations: MASH, metabolic dysfunction-associated steatohepatitis (previously named non-alcohol related steatohepatitis, NASH); T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus. | ||||||
| Apabetalone 5.01 (RVX-000222; RVX-208) | Flavonoid (plant) | NP-D | TS | BET bromodomain antagonist | Phase II/III (oral), long COVID; phase II, hypertension | Resverlogix Corporation |
| Rongliflozin 5.02 (DJT1116PG) | Phlorizin 5.03 (plant) | NP-D | TS | SGLT2 inhibitor | NDA (oral), T2DM | HEC Pharma/Sunshine Lake Pharma |
| LH-1801 5.04 | Phlorizin 5.03 (plant) | NP-D | TS | SGLT2 inhibitor | Phase III (oral), T2DM | Jiangsu Lianhuan Pharmaceutical |
| Mizagliflozin 5.05 (KWA 0711, GSK 1614235) | Phlorizin 5.03 (plant) | NP-D | TS | SGLT1 inhibitor | Phase II (oral), post-bariatric hypoglycaemia | Vogenx |
| SY-009 5.06 | Phlorizin 5.03 (plant) | NP-D | TS | SGLT1 inhibitor | Phase II (oral), T2DM | Suzhou Yabao Pharmaceutical |
| YG1699 (structure not disclosed) | Phlorizin 5.03 (plant) | NP-D | TS | SGLT1 and SGLT2 inhibitor | Phase II (oral), T1DM | Youngene Therapeutics |
| THDBH 101 (structure not disclosed) | Phlorizin 5.03 (plant) | NP-D | TS | SGLT1 and SGLT2 inhibitor | Phase I (oral), T2DM | Dongbao Zixing Biopharmaceutical |
| Berberine ursodeoxycholate 5.07 (HTD1801) | Berberine 5.04 (plant) | NP | Ia | Multiple mechanisms | Phase III (oral), T2DM | HighTide Biopharma |
| Vutiglabridin 5.08 (HSG4112) | Glabridin 5.09 (plant) | NP-S | TS | Paraoxonase 2 agonist, anti-inflammatory | Phase II (oral), obesity | Glaceum |
| HGR4113 5.10 | Glabridin 5.09 (plant) | NP-S | TS | Anti-inflammatory | Phase I (oral), MASH | Glaceum |
| TF0023 5.11 (aspirin trelamine) | Salicylic acid 5.12 (plant) | NP-D | TS | Thrombin inhibitor | Phase II (topical spray), ischaemic stroke and COVID | Techfields Pharma |
| Bersiporocin 5.13 (CSP-088; DWN-12088; ODP 2301) | Febrifugine 5.14 (plant) | NP-D | TS | Amino acyl tRNA synthetase | Phase II (oral), idiopathic pulmonary fibrosis | Daewoong Pharmaceutical |
| Isomyosamine 5.15 (MYMD-1) | Myosmine 5.16 (plant) | NP-D | TS | TNF-α inhibitor | Phase II (oral), chronic inflammation | TNF Pharmaceuticals |
| Rencofilstat 5.17 (CRV431) | Cyclosporin 4.32 (fungus) | NP-D | TS | Cyclophilin inhibitor | Phase II (oral), MASH | Hepion pPharmaceuticals |
| QG3030 5.18 | Lappaconitine 5.19 (plant) | NP-D | SS | Not disclosed | Phase I (oral), osteoporosis and fractures | Qgenetics |
Apabetalone 5.01 (RVX-208, RVX000222) is an antagonist of bromodomain and extra-terminal domain (BET) proteins,382,383 a class of epigenetic regulators that influence gene expression related to inflammation, lipid metabolism, and vascular disease.384 Developed by Resverlogix Corp, apabetalone 5.01 specifically targets the BRD2, BRD3, and BRD4 proteins, which recognise acetylated lysines on histones and control the transcription of genes involved in inflammation and atherosclerosis.383 Resverlogix Corp plans to evaluate 5.01 in a phase II/III trial (NCT06590324) in type 2 diabetes mellitus (T2DM) patients taking dapagliflozin with long-COVID and phase II trial (NCT04915300) for pulmonary arterial hypertension. Previously, a phase III trial (NCT02586155) did not demonstrate a significant reduction in major adverse cardiovascular events among high-risk T2DM patients.384,385 Apabetalone 5.01 was identified through phenotypical screening aimed at enhancing ApoA-I mRNA expression.382 While the original lead compound remains undisclosed, its structure was designed based on plant-derived polyphenols.386
Phlorizin 5.03 is a plant-derived dihydrochalcone glycoside, which was shown to be a non-selective SGLT inhibitor.387–389 There are two main SGLT subtypes: SGLT1, which is mainly expressed in the small intestine which decreases glucose uptake and alters postprandial blood glucose excursion, and SGLT2, which is responsible for the reabsorption of glucose filtered through the renal glomerulus. There have been 13 phlorizin 5.03-inspired SGLT2 selective anti-diabetes drugs and one dual SGLT1/SGLT2 inhibiter (sotagliflozin 2.36, Section 2.1.3), collectively known as ‘gliflozins’, approved since 2012. Rongliflozin 5.02 (DJT1116PG) is a gliflozin being developed as the L-pyroglutamic acid salt390 by Yichang East Sunshine Changjiang Pharmaceutical (part of HEC Holdings) as a T2DM treatment. An NDA for 5.02 was filed with the Chinese NMPA on 11 January 2024 (CXHS2400002 and CXHS2400003).
LH-1801 5.04 is a SGLT2 selective gliflozin391,392 being evaluated by Jiangsu Lianhuan Pharmaceutical in two phase III trials alone (CTR20240399) and in combination with metformin (CTR20240385).
Mizagliflozin 5.05 (KWA-0711, GSK 1614235, DSP-3235, KGA-3235) is a selective SGLT1 inhibitor,393,394 which was originally evaluated as treatment for chronic constipation.395 Vogenx licensed the right to develop 5.05 outside of Japan, Korea, and Taiwan from Kissei Pharmaceuticals in 2022 (ref. 396) and recently announced positive results from a phase II trial (NCT05541939) for postprandial glucose and insulin in post-bariatric hypoglycaemia subjects.397,398 Mizagliflozin 5.05 is also being evaluated in a phase II trial (NCT05721729) for postprandial glucose excursions.
SY-009 5.06 is a SGLT1 inhibitor399 being developed by Suzhou Yabao Pharmaceutical Research and Development Co, which completed a phase II trial (NCT05426018) for T2DM in September 2023.
YG1699 (structure not disclosed) is a dual SGLT1/SGLT 2 inhibitor400,401 being developed by Youngene Therapeutics. A phase IIa trial (NCT04956263) in people with T1DM on insulin pump therapy (sc administration) showed that oral YG1699 reduced post-prandial glucose compared to dapagliflozin.402 THDBH 101 (structure not disclosed) is another dual SGLT1 and SGLT2 inhibitor being developed by Dongbao Zixing (Hangzhou) Biomedical Co that completed a phase I trial (CTR20220031) in 2023.
Berberine ursodeoxycholate 5.07 (HTD-1801)403–405 is the ursodeoxycholic acid salt of berberine being developed by HighTide Biopharma. Berberine ursodeoxycholate is being evaluated in three phase III trials (NCT06350890, NCT06353347 and NCT06415773) for T2DM. Berberine is a plant-derived quaternary ammonium isoquinoline alkaloid with a variety of activities that include anti-inflammatory, antimicrobial, cholesterol lowering and antitumour,406,407 while ursodeoxycholic acid is used to treat chronic cholestatic liver diseases such as primary biliary cholangitis.408
Vutiglabridin 5.08 (HSG4112) is a racemic glabridin 5.09 analogue with improved bioavailability being developed by Glaceum.409 Glabridin 5.09 is a prenylated isoflavan found in the roots of liquorice (Glycyrrhiza glabra), which has antioxidant, anti-inflammatory, and anti-atherogenic activities.410 Glaceum completed a phase II trial (NCT05197556) of vutiglabridin 5.08 for obesity in 2023,411 and have registered a phase II Parkinson's disease trial (NCT06329141) that has yet to start. Recently, vutiglabridin 5.08 has been shown to be an agonist of paraoxonase 2 (PON2), which is known for its protective role against oxidative stress.412
Glaceum also recent started a phase I trial (NCT05642377) of another glabridin 5.09 analogue, HGR4113 5.10,413 as a potential T2DM treatment. Vutiglabridin 5.08 and HGR4113 5.10 both have anti-inflammatory activity.414
TF0023 5.11 (aspirin trelamine) is a salicylic acid 5.12 prodrug being developed by Techfields Pharma.415 TF0023 5.11 was in a phase II trial (NCT02785120) for the treatment of ischemic stroke but this has been suspended due to enrolment challenges and business operations. TF0023 5.11 was also evaluated in a phase II trial (NCT05212818) for the relief of the signs and symptoms of ARDS and pneumonia caused by COVID-19, but this trial was recently halted due to a lack of participants.
Bersiporocin 5.13 (DWN-12088, CSP-088, ODP 2301) is a febrifugine 5.14 analogue being developed by Daewoong Pharmaceutical in a phase II trial (NCT05389215) for the treatment of idiopathic pulmonary fibrosis. Bersiporocin 5.13 is a prolyl-tRNA synthetase 1 (PARS1) inhibitor, which binds to catalytic site of each protomer of PARS1 dimer in an asymmetric mode with different affinity.416,417 Bersiporocin 5.13 was designed based on halofuginone 5.20, a halogenated derivative of febrifugine 5.14, which is an antimalarial quinazolinone alkaloid originally isolated from the traditional Chinese medicine, Dichroa febrifuga.418
Isomyosamine 5.15 (isomiosamine, MYMD-1) is a myosmine 5.16 and nicotine 5.21 analogue being developed by TNF Pharmaceuticals (formally MyMD Pharmaceuticals).419,420 In 2023, the company completed a phase II clinical trial (NCT05283486) investigating its use to treat chronic inflammation associated with sarcopenia and frailty. Isomyosamine 5.15 has been shown to inhibit the production of TNF-α, a signalling protein in immune cells and inflammatory cytokine that plays a key role in initiating and sustaining the inflammatory response.421 TNF Pharmaceuticals also plans to evaluate 5.15 for the treatment of Hashimoto's thyroiditis and rheumatoid arthritis.422
Rencofilstat 5.17 (CRV431) is a semi-synthetic cyclosporin 4.32 (Section 4) derivative423 being evaluated in phase II trials NCT04480710![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 424 and NCT05402371 by Hepion Pharmaceuticals for non-alcoholic steatohepatitis (NASH), which is now called metabolic dysfunction-associated steatohepatitis (MASH).425–427 Like cyclosporin A 4.32, rencofilstat 5.17 is a cyclophilin inhibitor.
424 and NCT05402371 by Hepion Pharmaceuticals for non-alcoholic steatohepatitis (NASH), which is now called metabolic dysfunction-associated steatohepatitis (MASH).425–427 Like cyclosporin A 4.32, rencofilstat 5.17 is a cyclophilin inhibitor.
QG3030 5.18 is a semi-synthetic lappaconitine 5.19 derivative428,429 developed by Qgenetics that recently started a phase I trial as a potential treatment for osteoporosis and fractures.430 Lappaconitine 5.18 is a diterpene alkaloid isolated from Aconitum sinomontanum with antiarrhythmic, anti-inflammatory and anti-hypersensitivity in chronic pain properties.429,431
6. Compounds undergoing evaluation in immunological, inflammatory and related diseases
NPs have been instrumental in developing innovative treatments for immunological and inflammatory diseases. For example, the fungal-derived cyclosporin 4.32 and mycophenolate mofetil, along with the Streptomyces-derived sirolimus and tacrolimus, have been extensively used as immunosuppressants to help prevent organ rejection in transplant recipients.432,433 The plant alkaloid colchicine has long been used to manage gout, an inflammatory condition caused by uric acid crystal deposition in joints. In recent years (Section 2), the stilbene tapinarof 2.32 was approved for plaque psoriasis,71 a chronic autoimmune skin disease, while migalastat 2.09 was approved to treat Fabry disease, a rare X-linked lysosomal storage disorder caused by GLA gene mutations that results in multi-organ damage. Additionally, the cyclosporin 4.32 derivative voclosporin 2.28 has been approved for lupus nephritis,70 a serious kidney complication of systemic lupus erythematosus. NP-D compounds being investigated in a range of clinical trials focused on immunological, inflammatory and related diseases, which are listed in Table 9.| Compound (synonym) | Lead compound (source) | Classif | Manuf | Mode of action | Development status (administration), disease(s) | Developer(s) |
|---|---|---|---|---|---|---|
| a Not disclosed. Abbreviations: AR, androgen receptor; Hsf1, heat shock factor protein 1; mPGES-1, microsomal prostaglandin E synthase-1; NMDAR2B/NR2B, N-methyl-D-aspartate receptor subtype 2B antagonist; Nrf1 and NFR2, nuclear respiratory factor 1 and 2, PGE2, prostaglandin E2; TRPM8, transient receptor potential melastatin 8. | ||||||
| Vatiquinone 6.01 (EPI-743, PTC-743) | α-Tocopherol 6.02 (plant) | NP-D | TS | 15-Lipoxygenase inhibitor | NDA (oral), Friedreich's ataxia | PTC Therapeutics |
| Sonlicromanol 6.03 (KH 176) | α-Tocopherol 6.02 (plant) | NP-D | TS | Antioxidant, reduction of PGE2 levels | Phase II (oral), mitochondrial diseases | Khondrion |
| Recoflavone 6.04 (AJU-S56, GLH8NDE, DA-6034) | Eupatilin 6.05 (plant) | NP-D | TS | Mucus secretion increase | Phase III (topical), dry eye disease | AJU Pharm Co (GL Pharm Tech Corporation) |
| Acoltremon 6.06 (AR-15512; AVX 012; WS-12) | (−)-Menthol 6.07 (plant) | NP-D | SS | TRPM8 ion channel agonist | Approved by US FDA on 28 May 2025 (topical), dry eye disease | Alcon (Aerie Pharmaceuticals) |
| Nelonemdaz 6.08 (Neu2000) | Salicylic acid 5.12 (plant) | NP-D | TS | NMDAR2B antagonist, antioxidant | Phase III (oral), stroke | AmKor Pharma (GNT Pharma) |
| Crisdesalazine 6.09 (AAD-2004) | Salicylic acid 5.12 (plant) | NP-D | TS | mPGES-1 inhibition, antioxidant | Phase I (oral), Alzheimer's disease | AmKor Pharma (GNT Pharma) |
| Nizubaglustat 6.10 (AZ-3102) | Deoxynojirimycin 6.11 (plant and bacteria) | NP-D | TS | Ceramide glucosyltranferase and non-lysosomal neutral glucosylceramidase inhibitor | Phase II (oral), Niemann-Pick disease type C and GM2 gangliosidoses | Azafaros |
| Mocravimod 6.12 (KRP-203) | Myriocin 6.13 (fungus) | NP-D | TS | Sphingosine-1-phosphate (S1P) | Phase III (oral), AML patients undergoing allogeneic hematopoietic cell transplantation | Priothera |
| Vonifimod 6.14 (SLB736, NXC736) | Myriocin 6.13 (fungus) | NP-D | TS | Sphingosine-1-phosphate (S1P) | Phase II (oral), alopecia areata | Nextgen Bioscience |
| Icanbelimod 6.15 (CBP-307) | Myriocin 6.13 (fungus) | NP-D | TS | Sphingosine-1-phosphate (S1P) | Phase II (oral), ulcerative colitis | Suzhou Connect Biopharmaceutical |
| Vibozilimod 6.16 (SCD-044) | Myriocin 6.13 (fungus) | NP-D | TS | Sphingosine-1-phosphate (S1P) | Phase II (oral), atopic dermatitis; plaque psoriasis | Sun Pharmaceutical Industries |
| Amiselimod 6.17 (MT-1303; SLX-G1018) | Myriocin 6.13 (fungus) | NP-D | TS | Sphingosine-1-phosphate (S1P) | Phase II (oral), ulcerative colitis | Bausch Health Companies |
| Zetomipzomib 6.18 (KZR-616) | Epoxomicin 6.19 (actinomycete) | NP-D | TS | Proteasome inhibitor | Phase II (sc), autoimmune hepatitis | Kezar Life Sciences/Everest Medicines |
| Rosolutamide 6.20 (AJ201, ASC-JM17, ALZ003) | Curcumin 6.21 (plant) | NP-D | TS | PolyQ AR degradation and Nrf1, Nrf2 and Hsf1 activation | Phase I/II (oral), spinal and bulbar muscular atrophy | Avenue Therapeutics/AnnJi Pharmaceutical |
| Glasmacinal 6.22 (EP395) | Erythromycin 6.23 (actinomycete) | NP-D | SS | Enhancement of endothelial barrier function and inhibition of inflammatory cell recruitment | Phase II (oral), chronic obstructive pulmonary disease (COPD) | EpiEndo Pharmaceuticals |
| Progerinin 6.24 (SLC-D011) | Decursin 6.25 (plant) | NP-D | SS | Progerin-lamin A binding inhibitor | Phase II (oral), Hutchinson-Gilford progeria syndrome (HGPS) | PRG Science & Technology |
| Pegmispotide 6.26 (ADS-051, BT051) | Cyclosporin 4.32 (fungus) | NP-D | SS | Inhibits colon neutrophil trafficking | Phase II (oral, non-systemic), ulcerative colitis | Adiso Therapeutics |
| Exaluren 6.27 (ELX-02; NB-124) | Paromomycin 6.28 (actinomycete) | NP-D | TS | Ribosomal readthrough of premature stop codons | Phase II (sc), cystinosis; hereditary nephritis | Eloxx Pharmaceuticals |
| ENV-294 (structure not disclosed) | NP-D | Phase I (oral), atopic dermatitis | Enveda Biosciences | |||
| Bryostatin 1 4.30 | Bryostatin 1 4.30 (bryozoan/bacteria) | NP | I/TS | Protein kinase C | Phase I (IV), multiple sclerosis | Synaptogenix (Cleveland Clinic) |
| ZKN-0013 (structure not disclosed) | ‘Macrolide’a | NP-D | Ribosomal readthrough of premature stop codons | Phase I (oral), epidermolysis bullosa and familial adenomatous polyposis (FAP) | Eloxx Pharmaceuticals/Almirall | |
| Epigallocatechin 3-gallate 6.29 (EGCG) | EGCG 6.29 (plant) | NP | I | Anti-inflammatory, antioxidant | Phase I (oral), pulmonary fibrosis | University of California, San Francisco |
Vatiquinone 6.01 (PTC-743, EPI-743, ATQ3, α-tocotrienol quinone) is being developed by PTC Therapeutics and has completed phase IIb/III trials for Friedreich's ataxia (NCT04577352) and mitochondrial disorders in participants with refractory epilepsy (NCT04378075).434 PTC Therapeutics filed an NDA with the FDA for Friedreich's ataxia in December 2024.435 There are also two ongoing phase III studies for Friedreich's ataxia (NCT05515536) and inherited mitochondrial disease (NCT05218655). Vatiquinone 6.01 is a metabolic product of α-tocotrienol 6.02,436 which has potent antioxidant activity.436,437 Vatiquinone 6.01 has been reported to inhibit 15-lipoxygenase, which is a key component of ferroptosis that has been shown to be activated in several diseases including epilepsy.438,439
Sonlicromanol 6.03 (KH 176) is an α-tocopherol 6.02 derivative being developed by Khondrion BV that is being evaluated in a phase II trial (NCT04846036) for mitochondrial diseases in the paediatric population; interim results from this study have been published.440 A phase III trial (NCT06451757) for primary mitochondrial diseases has a planned start date of January 2025. Sonlicromanol 6.03, which is metabolised to KH176m 6.30in vivo, has antioxidant properties441,442 that inhibit lipopolysaccharide (LPS)-induced or interleukin-1β (IL-1β)-induced prostaglandin E2 production in skin fibroblasts and the mouse macrophage-like RAW264.7 cell line.443 KH176m 6.30 also inhibited microsomal prostaglandin E synthase-1 (mPGES-1) transcription.443,444
Recoflavone 6.04 (AJU-S56, GLH8NDE, DA-6034) is a eupatilin 6.05 derivative445 being evaluated by AJU Pharm Co (GL Pharm Tech Corporation) in a phase III trial (NCT06291194) for dry eye disease.446,447 Recoflavone 6.04 had been previously evaluated by Dong-A Pharmaceutical in other anti-inflammatory trials448 and has been proposed to increase mucus secretion via prostaglandin production.449–451
Acoltremon 6.06 (AR-15512; AVX 012; WS-12) is a (−)-menthol 6.07 derivative being developed by Alcon that completed a phase III trial (NCT05493111) in early 2024 for treatment of dry eye disease.452 On 28 May 2025, the US FDA approved an ophthalmic solution of acoltremon 6.06 for the treatment of the signs and symptoms of dry eye disease.453 Three further phase III trials (NCT06544694, NCT06544707 and NCT06660290) are ongoing. Acoltremon 6.06 was first described as a semi-synthetic product via (−)-menthylformic acid 6.31 with menthol-like cooling properties in a 1971 patent454 and a 1978 publication.455 It was later demonstrated to be an agonist of the calcium-permeable ion channel transient receptor potential melastatin 8 (TRPM8), which serves as the principal detector of cold in humans.456,457 TRPM8 is also involved with the regulation of tear production and blink rate.458
Nelonemdaz 6.08 (Neu2000) and crisdesalazine 6.09 (AAD-2004) are salicylic acid 5.12 (Section 5) derivatives that both were designed by GNT Pharma from aspirin 6.32 and sulfasalazine 6.33 for their anti-inflammatory and antioxidant activities. Nelonemdaz 6.08 is a selective NMDA receptor subtype 2B antagonist and antioxidant,459–461 which has completed a phase III trial (NCT05041010) in acute ischemic stroke patients before endovascular thrombectomy.462 An additional multinational stroke phase III trial is planned.
Crisdesalazine 6.09 has completed a phase I trial for Alzheimer's disease and could be evaluated in other neurodegenerative brain diseases.463 Crisdesalazine 6.09 has strong antioxidant activity and also inhibits microsomal prostaglandin E synthase-1 (mPGES-1) activity464 and was approved in 2021 in South Korea to treat dogs with cognitive dysfunction syndrome (brand name Gedacure®).465
Nizubaglustat 6.10 (AZ-3102) is a deoxynojirimycin 6.11 derivative466 that interferes with glucosylceramide metabolism being developed by Azafaros, which is being evaluated in a phase II trial (NCT05758922) for GM2 gangliosidoses or Niemann-Pick disease type C.467–469
Myriocin 6.13 (ISP-1), isolated from the fungus Isaria sinclairii,470 exhibits antifungal and immunosuppressive properties.471 Extensive SAR led to fingolimod 6.34, a prodrug approved in the USA in 2010 for multiple sclerosis, which requires phosphorylation to modulate the sphingosine-1-phosphate (S1P) receptor.472,473 Fingolimod 6.34 inspired the development of siponimod 2.21 (ref. 474) and etrasimod 2.37 (ref. 475) (Section 2) and there are five related compounds currently in development. Priothera is evaluating mocravimod 6.12 (KRP-203), a S1P agonist476 and phosphoinositide kinase (PIK) modulator,477 in a phase III trial (NCT05429632) as an adjunctive and maintenance treatment in adult acute myeloid leukaemia (AML) patients undergoing allogeneic hematopoietic cell transplantation.478 Vonifimod 6.14 (NXC736, SLB736), which is being developed by Nextgen Bioscience, has selective S1P1 and S1P4 isoform activity that attenuates NLRP3.479,480 Vonifimod 6.14 recently started a phase II trial (NCT06104839) in severe alopecia areata (hair loss patches). Icanbelimod 6.15 (CBP-307) is an S1P receptor agonist being developed by Suzhou Connect Biopharmaceuticals that has completed a phase II (NCT04700449) for ulcerative colitis.481,482 Connect Biopharmaceuticals is actively seeking to out-license icanbelimod 6.15 for future trials in ulcerative colitis and Crohn's disease.483 Vibozilimod 6.16 (SCD-044), a selective S1P agonist being developed by Sun Pharmaceutical Industries, is being evaluated in phase II trials for plaque psoriasis (NCT04566666) and atopic dermatitis (NCT04684485).484 Amiselimod 6.17 (MT-1303, SLX-G1018)485 is being evaluated by Bausch Health Companies in a phase II trial (NCT04857112) for ulcerative colitis.486
Zetomipzomib 6.18 (KZR-616) is a proteasome inhibitor487–489 under development by Kezar Life Sciences and Everest Medicines that is being evaluated in a phase II trial for autoimmune hepatitis (NCT05569759). The lead compound was the actinomycetes-derived epoxomicin 6.19,490–492 which also served as the basis for the development of carfilzomib 6.35, an anticancer drug approved by the FDA for the treatment of multiple myeloma in 2012.493
Rosolutamide 6.20 (AJ201, ASC-JM17, ALZ003) is a curcumin 6.21 analogue being developed by Avenue Therapeutics and AnnJi Pharmaceutical that has completed a phase I/II trial (NCT05517603) for spinal and bulbar muscular atrophy (SBMA, Kennedy's Disease).494 Curcumin 6.21 (Section 7.1) is a phenolic diarylheptanoid, which has both diketone and keto-enol tautomers.495 Found in the rhizomes of turmeric (Curcuma longa) and other Curcuma species, it exhibits anti-inflammatory, antioxidant, anticancer, and antimicrobial properties.496 Curcumin 6.21 has also been evaluated in clinical trials, mostly as a dietary supplements. Rosolutamide 6.20 has an FDA Orphan Drug Designation for the polyQ diseases SBMA, Huntington's disease and spinocerebellar ataxia. Rosolutamide 6.20 is a next generation analogue of dimethylcurcumin 6.36 (ASC-J9),497,498 which is an androgen receptor (AR) antagonist developed for prostate cancer and later evaluated for acne vulgaris. Dimethylcurcumin 6.36 was shown to disrupt the interaction between ARs and coregulators, which also increases cell survival by decreasing AR-polyQ nuclear aggregation and increasing AR-polyQ degradation in cultured cells.499 Similarly, rosolutamide 6.20 acts on Nrf1, Nrf2 and Hsf1 to enhance the expression of proteasome subunits, antioxidant enzymes and molecular chaperones, while also disrupting the AR.500–503
Glasmacinal 6.22 (EP395) is a non-antibacterial azithromycin 6.37 ketolide derivative (erythromycin 6.23 derivative)504 with anti-inflammatory properties that is being developed by EpiEndo Pharmaceuticals. It has completed a phase II trial (NCT05572333) in patients with chronic obstructive pulmonary disease (COPD).505 Glasmacinal 6.22 is the first member of non-antibacterial macrolide agents with anti-inflammatory activity with the newly designated WHO international nonpropriety name (INN) suffix -macinal.
Progerinin 6.24 (SLC-D011, KC2) is a decursin 6.25 derivative506 being evaluated by PRG Science & Technology in a phase II trial (NCT06775041) for the treatment of Hutchinson-Gilford progeria syndrome (HGPS, a genetic disorder that causes children to age rapidly). A phase II trial (NCT05847179) is also planned for Werner syndrome (autosomal recessive disorder characterised by premature aging). The prenyl-coumarins decursin 6.25 and decursin angelate 6.38 are the major bioactive components of the Korean traditional medicine Cham-Dang-Gui (Angelica gigas).507 Progerinin 6.24 is an inhibitor of progerin, a mutant form of the lamin A protein implicated in HGPS508,509 and Werner syndrome.510
Pegmispotide 6.26 (ADS-051, BT051) is a pegylated cyclosporin 4.32 derivative511 being developed by Adiso Therapeutics that successfully completed a phase Ib trial (NCT05084261) for ulcerative colitis.512 Pegmispotide 6.26 blocks neutrophil trafficking into and activation within the colon mediated by multidrug resistance protein 2 (MRP2)513 and formyl peptide receptor 1 (FPR1) mechanisms.511,514
Exaluren 6.27 (ELX-02; NB-124)515 is being developed by Eloxx Pharmaceuticals and has completed several phase II trials using subcutaneous (SC) administration: Alport Syndrome (NCT05448755) and CF patients with a G542X allele (NCT04135495 and NCT04126473).516,517 Eloxx plan to continue development of 6.27 for Alport Syndrome patients with nonsense mutations.518 In 1985, it was found that paromomycin 6.28 and geneticin 6.39 (G418) could induce the mammalian ribosome to suppress disease-causing nonsense mutations and partially restore the expression of functional proteins.519 Exaluren 6.27 is a synthetic aminoglycoside designed to have selectivity for cytoplasmic versus mitochondrial ribosomes and weak antibacterial activity, which was identified by comparing screening of translational read-through of premature STOP codons with diminished binding to the bacterial ribosome compared to paromomycin 6.28 and geneticin 6.39.515,520 Other aminoglycosides and other classes of compounds have demonstrated similar properties.521
Eloxx Pharmaceuticals and Almirall are developing the macrolide derivative ZKN-0013 (structure not disclosed),522 which completed a phase I trial (ACTRN12624000632594) in 2024. ZKN-0013 is being investigated for the treatment of nonsense mutation recessive dystrophic epidermolysis bullosa (RDEB), junctional epidermolysis bullosa (JEB), and familial adenomatous polyposis (FAP).523
Synaptogenix is evaluating bryostatin 1 4.30 in a phase I trial (NCT06190912) in multiple sclerosis patients not receiving any disease modifying therapy due to its potential neuroprotective and anti-inflammatory effects.524,525 Bryostatin 1 4.30 is also being investigated as a Alzheimer's disease treatment (Section 4.2).
ENV-294, which is being developed by Enveda Biosciences, recently started phase I trials for atopic dermatitis.526 Although there is little publicly disclosed information on ENV-294,527 Enveda have patented the plant sesquiterpene nootkatone 6.40 as a treatment for psoriasis or atopic dermatitis.528 (+)-Nootkatone 6.40 was first reported in 1962,529 its structure revised in 1965,530 and is a valued food flavouring due to its grapefruit-like odour and slightly bitter taste.531,532
Epigallocatechin 3-gallate 6.29 (EGCG) is an abundant polyphenol present in green tea (Camellia sinensis) and other plants with anti-inflammatory and antioxidant activities. Epigallocatechin 3-gallate 6.29 has been reported to have various biological activities including against neurological disorders,533 cancer,534 and viruses;533 although care needs to be taken as 6.29 can act promiscuously in biological assays and has suboptimal systemic distribution.535,536 There are over 110 studies registered on https://www.clinicaltrials.gov/ that have evaluated epigallocatechin 3-gallate 6.29 (including supplements) and two phase I trials (NCT05195918 and NCT06265532) have recently started for the of treatment idiopathic pulmonary fibrosis.
7. Compounds undergoing evaluation in oncology
NPs have been the source of many cancer drugs, particularly before the emergence of kinase inhibitors, mAbs, and CAR-T therapies.537 There are still at least 35 NP-D compounds in oncology clinical trials (Tables 10 and 11, Sections 7.1 and 7.2), as well at least 46 NP-ADCs in phase II and beyond (Tables 11 and 12, Section 7.3).| Name (synonym) | Lead compound/s | Classif. | Manuf. | Mode of action | Development status (administration), cancer | Developer |
|---|---|---|---|---|---|---|
| Gimatecan 7.01 (ST-1481)541,542,566,567 | Camptothecin 7.02 | NP-D | TS | Topoisomerase I | Phase II (oral), ovarian, pancreatic, SCLC | Lee's Pharmaceuticals |
| Locnartecan 7.03 (HDC SN-38; PEN-866; STA-12-8666; STA-8666) | Camptothecin 7.02 | NP-D | TS | Topoisomerase I | Phase I/II (IV), solid tumours | SciClone Pharmaceuticals/NCI |
| Orenasitecan 7.04 (VIP236) | Camptothecin 7.02 | NP-D | TS | Topoisomerase I | Phase I (IV), solid tumours | Vincerx Pharma |
| Epigallocatechin-3-gallate 6.29 (EGCG) | Epigallocatechin-3-gallate 6.29 | NP | I | Anti-inflammatory | Phase II (oral), hepatocellular carcinoma chemoprevention | University of Texas Southwestern Medical Center and others |
| Genistein 7.05 (BIO-300) | Genistein 7.05 | NP | TS | Signalling pathway modulation and tyrosine kinase inhibition | Phase I/II (oral) | Humanetics and others |
| ME-344 7.06 + bevacizumab | Genistein 7.05 + mAb | NP-D | TS | OXPHOS inhibition, VDAC 1 and 2 inhibition, purine biosynthesis | Phase I (IV), colorectal | MEI Pharma |
| Foslinanib 7.07 (CVM-1118, TRX-818) | Flavone 7.08 | NP-D | TS | TRAP1 inhibition | Phase II (oral), hepatocellular carcinoma, neuroendocrine tumours | TaiRx |
| Voruciclib 7.09 (P1446A) | Rohitukine 7.10 | NP-D | TS | CDK9 inhibition | Phase I (oral), DLBCL, AML | MEI Pharma |
| LEAF-4L6715 (liposomal transcrocetin 3.19) | Transcrocetin 3.19 | NP | TS | Various mechanism | Phase I/II (IV), glioblastoma; phase III planned | LEAF4Life |
| Patidegib 7.11 (saridegib, BBP-009; FIN-5; IPI-926) | Cyclopamine 7.12 | NP-D | SS | SMO and Hedgehog | Phase III (topical), basal cell nevus syndrome | Sol–Gel Technologies |
| Tigilanol tiglate 3.14 (EBC-46) | Tigilanol tiglate 3.14 | NP | I | PKC activation | Phase II (intratumorally), sarcoma, head and neck | QBiotics |
| Tuvatexib 7.13 (VDA-1102) | Jasmonic acid 7.14 | NP-D | TS | Dual voltage-dependent anion channel (VDAC) and hexokinase II (HK II) modulation | Phase II (topical), actinic keratosis | Vidac Pharma |
| Paclitaxel obaluronate 7.15 (Oncofid-P-B) | Paclitaxel 7.16-hyaluronan | NP-D | SS | Tubulin stabilisation | Phase III (intravesical), bladder | Fidia Farmaceutici |
| Sudocetaxel zendusortide 7.17 (TH1902) | Paclitaxel 7.16 peptide conjugate | NP-D | SS | Tubulin stabilisation | Phase I (IV), solid tumours | Theratechnologies |
| Hypericin 7.18 (SGX301) | Hypericin 7.18 | NP | TS | ROS production followed by inflammatory response | Phase II (topical), mycosis fungoides-cutaneous T-cell lymphoma | Soligenix |
| Ivospemin 7.19 (SBP-101) | Spermine 7.20 | NP-D | TS | Polyamine metabolism modulator (ornithine decarboxylase and spermidine/spermine N1-acetyltransferase activity) | Phase II/III (sc), pancreatic | Panbela Therapeutics |
| Curcumin 6.21 | Curcumin 6.21 | NP | TS | Anti-inflammatory and anti-oxidative properties | Phase I/II (oral), glioma; phase II (oral), gastric | SignPath Pharma; NCI and others |
| (±)-gossypol 7.21 | (±)-gossypol 7.21 | NP | I | LRPPRC degrader (and other mechanisms) | Phase II (oral), breast | Zhejiang Cancer Hospital |
| Resveratrol 4.37 | Resveratrol 4.37 | NP | TS | Antioxidant (and other mechanisms) | Phase II (oral), lymphangioleiomyomatosis | University of Cincinnati and others |
| Name (synonym) and references | Lead compound/s (source) | Classif. | Manuf. | Mode of action | Development status (administration), cancer | Developer |
|---|---|---|---|---|---|---|
| a Halichondrins are probably produced by dinoflagellates living or filtered through the sponges. b Trabectedin 7.45 was originally isolated from an ascidian but are produced by bacteria.29 c Ecubectedin 7.44 and PM54 7.46 are manufactured by total synthesis using the bacterially-derived cyanosafracin B as a key intermediate, like trabectedin 7.45 and lurbinectedin 2.36.76,77 | ||||||
| Aldoxorubicin 7.22 | Doxorubicin 7.23 (actinomycete) | NP-D | SS | Topoisomerase II | Phase II (IV), pancreatic; phase III completed, soft tissue sarcoma; other cancers | LadRx Corporation (ImmunityBio) |
| Berubicin 7.24 (RTA 744, WP744) | Doxorubicin 7.23 (actinomycete) | NP-D | SS | Topoisomerase II | Phase II (IV), glioblastoma | CNS Pharmaceuticals/WPD Pharmaceuticals |
| Naxtarubicin 7.25 (L-annamycin, AR-522) | Doxorubicin 7.23 (actinomycete) | NP-D | TS | Topoisomerase II | Phase I/II (IV), soft tissue sarcomas | Moleculin Biotech |
| Plinabulin 7.26 | Phenylahistin 7.27 (fungus) | NP-D | TS | Tubulin polymerisation inhibition | Phase III (IV), NSCLC and cancer-induced neutropenia | BeyondSpring |
| Evexomostat 7.28 (SDX-7320) | Fumagillin 7.29 (fungus) | NP-D | SS | Methionine aminopeptidase 2 | Phase II (IV), breast | SynDevRx |
| Orellanine 7.30 | Orellanine 7.30 (fungus) | NP | TS | Antioxidant, not fully elucidated | Phase I/II (IV), renal cell carcinoma | Oncorena AB |
| Irofulven 7.31 | Illudin S 7.32 (fungus) | NP-D | SS | DNA alkylation and adduct formation | Phase II (IV), prostate | Lantern Pharma |
| LP-184 7.33 | Illudin S 7.32 (fungus) | NP-D | SS | DNA alkylation and adduct formation | Phase I (IV), solid tumours | Lantern Pharma |
| LP-284 7.34 | Illudin S 7.32 (fungus) | NP-D | TS | DNA alkylation and adduct formation | Phase I (IV), lymphomas and solid tumours | Lantern Pharma |
| Antroquinonol 7.35 | Antroquinonol 7.35 (fungus) | NP | TS | Downregulates signalling pathways including PI3K/AKT/β-catenin, Ras and Rho, and Nrf-2 activation | Phase I/II (IV), pancreatic; phase II, AML and NSCLC; also, hepatitis B, COVID-19 and atopic dermatitis | Golden Biotech |
| NUC-7738 7.36 | Cordycepin 7.37 (fungus) | NP-D | TS | Downregulates signalling pathways including NF-κB and Wnt/β-catenin | Phase I/II (IV), solid tumours and lymphoma | NuCana |
| E6201 7.38 | 5Z-7-oxo-zeaenol 7.39 (fungus) | NP-D | TS | MEK1, MEK kinase-1 and FLT3 inhibitor | Phase I (IV), melanoma | Spirita Oncology/JS InnoPharm (Eisai) |
| E7130 7.40 | Halichondrin B 7.41 (spongea) | NP-D | TS | Microtubule-destabilisation (near colchicine binding site) | Phase I (IV), solid tumours | Eisai |
| PM534 7.42 | PM742 7.43 (sponge) | NP-D | TS | Microtubule-destabilisation (colchicine binding site) | Phase I (IV), solid tumours | PharmaMar |
| Ecubectedin 7.44 | Trabectedin 7.45 (ascidian)b | NP-D | TSc | DNA minor groove binding, adduct formation, and interference with DNA repair and transcription | Phase I/II (IV), soft tissue sarcoma and solid tumours | PharmaMar |
| PM54 7.46 | Trabectedin 7.45 (ascidian)b | NP-D | TSc | DNA minor groove binding, adduct formation, and interference with DNA repair and transcription | Phase I (IV), solid tumours | PharmaMar |
| Name (synonyms) | Warhead (class)/antigena | Development status (cancer)b | Developer/s |
|---|---|---|---|
| a Antigen synonyms TROP2 = TACSTD2; HER2 = ERBB2; HER3 = ERBB3, bispecific antigens are separated by the × symbol. b Only the phase III trial target cancers are listed. c Trastuzumab mafodotin has also been called caxmotabart entudotin. d Structure of HS-9265 has not been disclosed but is likely to be SHR9265 (rezetecan). Abbreviations: DLBCL, diffuse large B cell lymphoma; HNSCC, head and neck squamous cell carcinoma; NSCLC, non-small cell lung cancer; PBD, pyrrolobenzodiazepine; SCLC, small cell lung cancer; TN, triple negative. | |||
| Telisotuzumab vedotin 7.55 (ABBV-399) | MMAE (dolastatin)/MET | Approved by the US FDA on 14 May 2025 (NSCLC) | AbbieVie (BMS) |
| Patritumab deruxtecan 7.57 (HER3-DXd, MK-1022) | Deruxtecan (DXd) (camptothecin)/ERBB3 | Registration, phase III (NSCLC) | Daiichi Sankyo/Merck & Co |
| Becotatug vedotin (MRG003) | MMAE (dolastatin)/EGFR | Phase III (HNSCC) | Shanghai Miracogen |
| Bulumtatug fuvedotin (9MW2821) | MMAE (dolastatin)/NECTIN4 | Phase III (urothelial, cervical) | Mabwell (Shanghai) Bioscience |
| Garetatug rezetecan (SHR-A1904) | Rezetecan (SHR9265) (camptothecin)/CLDN18.2 | Phase III (gastric, adenocarcinoma) | Shanghai Hengrui Pharmaceutical |
| Ifinatamab deruxtecan (I-DXd, MK-2400, DS-7300) | Deruxtecan (DXd) (camptothecin)/CD276 | Phase III (NSCLC, RSCLC, breast) | Daiichi Sankyo/Merck & Co |
| Izalontamab brengitecan (BL-B01D1) | Brengitecan (Ed-04) (camptothecin)/HER3 × EGFR | Phase III (NSCLC, RSCLC, nasopharyngeal, breast, squamous cell) | Sichuan Biokin Pharmaceutical/BMS/SystImmune |
| Rinatabart sesutecan (GEN1184, PRO1184) | Sesutecan (camptothecin)/FRα | Phase III (fallopian tube, ovarian; peritoneal) | Genmab |
| Ruzaltatug rezetecan (SHR-A2009) | Rezetecan (SHR9265) (camptothecin)/HER3 | Phase III (NSCLC) | Suzhou Suncadia Biopharmaceuticals |
| Sigvotatug vedotin (PF-08046047, SGN-B6A) | MMAE (dolastatin)/IB6 | Phase III (NSCLC) | Pfizer |
| Sonesitatug vedotin (AZD-0901, CMG901) | MMAE (dolastatin)/CLDN18.2 | Phase III (gastric) | AstraZeneca/KeyMed Biosciences |
| Tecotabart vedotin (LM-302, TPX-4589, BMS-986476) | MMAE (dolastatin)/CLDN18.2 | Phase III (adenocarcinoma) | LaNova Medicines Zhejiang |
| Telisotuzumab adizutecan (ABBV-400) | Adizutecan (camptothecin)/cMET | Phase-III (colorectal) | AbbieVie |
| Tizetatug rezetecan (SHR-A1921) | Rezetecan (SHR9265) (camptothecin)/TROP2 | Phase III (epithelial) | Suzhou Suncadia Biopharmaceuticals |
| Trastuzumab envedotin (DP-303c) | MMAE (dolastatin)/HER2 | Phase III (breast) | CSPC ZhongQi Pharmaceutical Technology |
| Trastuzumab mafodotinc (FS-1502, IKS-014, LCB-14) | MMAE (dolastatin)/HER2 | Phase III (breast) | Shanghai Fosun Pharmaceutical/Iksuda Therapeutics |
| Trastuzumab rezetecan (SHR-A1811) | Rezetecan (SHR9265) (camptothecin)/HER2 | Phase III (colorectal, gastric) | Jiangsu Hengrui Pharmaceutical |
| Trastuzumab pamirtecan (BNT-323, DB-1303) | Pamirtecan (P1003) (camptothecin)/HER2 | Phase III (breast) | BioNTech/Duality Biologics |
| Trastuzumab vedotin (MRG002) | MMAE (dolastatin)/HER2 | Phase III (urothelial, breast) | Shanghai Miracogen |
| Zilovertamab vedotin (MK-2140, VLS-101) | MMAE (dolastatin)/ROR1 | Phase III (DLBCL) | Merck |
| ESG-401 (STI-3258, OQY-3258) | Govitecan (SN-38) (camptothecin)/TROP2 | Phase III (HER2- breast) | Shanghai Escugen Biotechnology |
| FDA018-ADC | Govitecan (SN-38) (camptothecin)/TROP2 | Phase III (TN breast) | Shanghai Fudan-Zhangjiang Bio-Pharmaceutical |
| HS-20093 (GSK5764227) | HS-9265d (camptothecin)/B7H3 | Phase III (SCLC) | Shanghai Hansoh Biomedical/GSK |
| IBI-343 | Exatecan (DX8951f) (camptothecin)/CLDN18.2 | Phase III (adenocarcinoma) | Innovent Biologics |
| BL-M07D1 | Brengitecan (Ed-04) (camptothecin)/HER2 | Phase III (breast) | SystImmune/Sichuan Baili Pharmaceutical |
| YL-201 | YL0010014 (camptothecin)/B7H3 | Phase III (nasopharyngeal, SCLC) | MediLink Therapeutics (Suzhou) Co |
| Anvatabart opadotin (ARX-788) | AS29 (dolastatin)/HER2 | Phase II/III (HER2+ breast) | Ambrx/Zhejiang Medicine |
| Vobramitamab duocarmazine 7.59 (MGC 018) | Duocarmycin (duocarmycin)/B7H3 | Phase II/III (prostate) | MacroGenics |
| Luveltamab tazevibulin 7.58 (STRO-002) | SC209 (hemiasterlin)/FOLR1 | Phase II/III (ovarian, fallopian tube) | Sutro Biopharma |
7.1 Plant-derived
As mentioned in Section 6, epigallocatechin 3-gallate 6.29 (EGCG) is a plant polyphenol with anti-inflammatory and antioxidant activities (Section 6), which also has promise in cancer chemoprevention.533–535,538 It is being evaluated in a phase II trial (NCT06015022) by the University of Texas Southwestern Medical Center for hepatocellular carcinoma chemoprevention,539,540 and by the Shandong Cancer Hospital and Institute in a phase II (NCT06398405, oral) for the treatment of dysphagia (difficulty in swallowing) in oesophageal squamous cancer patients and a phase I/II (NCT06524609, topical) for the treatment of taxane-induced peripheral neurotoxicity. There are also other ongoing phase I trials and clinical studies, as well as combination and dietary supplement trials.Gimatecan 7.01 (ST-1481) is a O-tert-butyl oxime derivative541,542 of camptothecin 7.02 being developed by China Oncology Focus, a subsidiary of Lee's Pharmaceutical Holdings.543 Camptothecin 7.02 is a plant alkaloid derived topoisomerase I inhibitor first reported from Camptotheca acuminata in 1966.544,545 Gimatecan 7.01 has been evaluated in China in three phase II trials: ovarian (NCT0484684), pancreatic (NCT04571489) and SCLC (NCT04501029).
Locnartecan 7.03 (HDC SN-38, PEN-866, STA-8666) is being developed by SciClone Pharmaceuticals and the NCI in a phase I/II (NCT04890093) in combination with vincristine and temozolomide for the treatment of solid tumours. Locnartecan 7.03 has previously completed a phase I/II trial (NCT03221400) in 2023 for solid tumours.546 Locnartecan 7.03 contains a hydrolysable camptothecin 7.02 derivative SN-38 unit linked to an Heat Shock Protein 90 (HSP90) inhibitor, which interacts with topoisomerase I to induce double strand and single strand DNA breaks.547,548
Orenasitecan 7.04 (VIP236) is a small molecule drug conjugate (SMDC) being developed by Vincerx Pharma that has competed a phase I trial (NCT05712889) in solid tumours in late 2024. SMDCs have targeting ligand, a spacer, a cleavable bridge and a therapeutic payload, which are designed to aid transport to the tumour and release the payload in the tumour microenvironment.549 Orenasitecan 7.04 has an αVβ3 integrin-targeting moiety and a neutrophil elastase-cleavable linker with a camptothecin derivative VIP126 as the cytotoxic payload.550
Genistein 7.05 is a plant isoflavone known for its antioxidant, anti-inflammatory, and potential anticancer properties via cell signalling pathway modulation and tyrosine kinase inhibition. Genistein 7.05 has been being evaluated in several clinical trials,551 mostly as part of dietary supplements.552 Humanetics Corporation is developing a nanosuspension of genistein 7.05 (code BIO-300)553 that has completed a phase I/II trial (NCT02567799) for reducing radiation injuries in combination with standard dose radiation therapy and chemotherapy in patients with NSCLC.554 BIO-300 is being investigated in a phase II trial (NCT04482595) for its effectiveness in protecting lung tissues against the long-term effects of COVID-19. Finally, a phase I trial (NCT04650555) for acute radiation syndrome has been completed and there is a plan to seek approval using the FDA's Animal Rule.555,556
ME-344 7.06 in combination with the anti-VEGF-A monoclonal bevacizumab557 is being evaluated in a phase I trial (NCT05824559) in patients with previously treated metastatic colorectal cancer by MEI Pharma. ME-344 7.06 is a derivative of idronoxil 7.46, which is a discontinued oncology clinical candidate based on isoflavones like genistein 7.05.558,559 ME-344 7.06 has been shown to inhibit mitochondrial oxidative phosphorylation (OXPHOS),560,561 as well as disrupt purine biosynthesis561 and voltage-dependent anion channels 1 and 2 (VDAC 1 and 2).562
Foslinanib 7.07 (CVM-1118, TRX-818), a flavone 7.08 derivative prodrug563,564 being developed by TaiRx, is currently under evaluation in two phase II trials: (1) in combination with the anti-PD-1 monoclonal nivolumab for hepatocellular carcinomas (NCT05257590) and (2) advanced neuroendocrine tumours (NCT03600233). Recent studies report that foslinanib 7.07 binds to TNF receptor associated protein 1 (TRAP1), leading to the inhibition of succinate accumulation and destabilisation of HIF-1α.565 This mechanism induces cellular apoptosis, cell cycle arrest at G2/M, and suppression of vasculogenic mimicry formation.565
Voruciclib 7.09 (ME-522, P1446A) is currently being developed by MEI Pharma and is a flavonoid-based CDK inhibitor related to alvocidib 7.47 (flavopiridol).6 The progenitor compound for alvocidib 7.47, which was subsequently found to be a pan cyclin-dependant kinase (CDK) inhibitor,568,569 was rohitukine 7.10, first reported in 1979 from Aphanamixis polystachya and later from Dysoxylum binectariferum.570–572 Despite being evaluated in over 65 clinical trials, alvocidib 7.47 did not receive approval, primarily due to adverse events.573 Voruciclib 7.09 was shown to potently block CDK9, which is the transcriptional regulator of MCL-1 that along with Bcl-2 are frequently over-expressed in DLBCL.574,575 Voruciclib 7.09 is currently being evaluated as a monotherapy or in combination with venetoclax (Bcl-2 inhibitor) in a phase I trial (NCT03547115) for B-Cell malignancies or AML.
Transcrocetin 3.19 is an apocarotenoid usually isolated from Gardenia jasminoides and Crocus sativus (saffron).194 Transcrocetin 3.19 has a variety of anti-cancer mechanisms include cell proliferation inhibition, apoptosis induction, angiogenesis suppression, and potentiation of chemotherapy, which occur via various pathways that include PI3K/Akt, MAPK and NF-κB.194,576 LEAF-4L6715, a liposomal formulation of transcrocetin 3.19,198,577 is scheduled to be evaluated by the Institut de cancérologie Strasbourg Europe in a phase III trial (NCT06477939) in combination with radiotherapy and temozolomide followed by adjuvant temozolomide in new diagnosed glioblastoma, and a phase II trial (NCT06476704) in combination with radiotherapy for soft-tissue sarcoma. LEAF-4L6715 is also being clinically evaluated for COVID-19 ARDS (Section 2.3).198,200 Diffusion Pharmaceuticals (now part of CervoMed) completed a phase III glioblastoma trial (NCT03393000) of transcrocetinate sodium in late 2020, but development has been halted.578
Patidegib 7.11 (SGT-610, saridegib, IPI-926) is a semi-synthetic derivative579,580 of cyclopamine 7.12 (11-deoxojervine) with improved potency and physicochemical properties.581,582 The steroidal alkaloid cyclopamine 7.12, isolated from Veratrum californicum, causes teratogenic effects such as cyclopia and facial malformations in lambs when ingested by pregnant ewes.583 Cyclopamine 7.12 was later shown to be a specific inhibitor of Hedgehog (Hh) signalling, functioning through direct antagonism of the transmembrane receptor Smoothened (SMO).583 Patidegib 7.11 is being evaluated by Sol–Gel Technologies in phase III trial (NCT06050122) as a topical prevention of new basal cell carcinoma lesions in patients with Gorlin syndrome.584
Tigilanol tiglate 3.14 (EBC 46) is a diterpene ester isolated from Fontainea picrosperma157,158,585 being developed by QBiotics that is evaluated in phase-II trials for soft tissue sarcoma (NCT05755113) and head and neck cancer (NCT05608876). Tigilanol tiglate 3.14 is marketed as Stelfonta® in the EU, UK, USA and Australia to treat canine mast cell tumours,586,587 and is being evaluated to treat canine melanoma and soft tissue sarcoma. The MoA of 3.14 starts with PKC activation with induces local inflammation and tumour vasculature.585,588 The immune response also induces wound healing, which was also found with its analogue EBC-1013 3.13 (Section 2.2).589
Tuvatexib 7.13 (VDA-1102)590 is a synthetic derivative of jasmonic acid 7.14, a plant-derived endogenous growth regulator with known anticancer properties.591,592 Developed by Vidac Pharma, tuvatexib 7.13 has completed two phase II trials (NCT03538951 and NCT02844777) for actinic keratosis with a phase IIb trial recently announced for advanced actinic keratosis.593 Its anticancer mechanism involves dual modulation of the voltage-dependent anion channel (VDAC) and hexokinase II (HK II).590,594
Paclitaxel obaluronate 7.15 (ONCOFID®-P-B) is a paclitaxel 7.16-hyaluronan (∼200 kDa, paclitaxel ∼20% w/w) bioconjugate595–597 being developed by Fidia Farmaceutici that is being evaluated in a phase III trial (NCT05024773, intravesical administration) for bladder cancer. Paclitaxel obaluronate 7.15 was found to enter human bladder cancer cells via a CD44, which is a cell surface adhesion protein involved in cellular interactions, where it accumulates into lysosomes.598 The ester bonds are then hydrolysed to release hyaluronan and paclitaxel 7.16, which interferes β-tubulin leading to cell death.
Sudocetaxel zendusortide 7.17 (TH1902) is a bis-docetaxel 7.48-TH19P01 peptide drug conjugate with succinic ester linkages599,600 being developed by Theratechnologies that is being evaluated in a phase I trial (NCT04706962) against solid tumours. TH19P01 specifically interacts with sortilin (SORT1; also known as neurotensin receptor-3), which is a transmembrane protein involved with protein sorting and the trafficking of targeted proteins and peptides to distinct intracellular destinations, which has been associated with cancer progression.599–601 Sudocetaxel zendusortide 7.17 binds to SORT-1 on the cell surface, then internalised and docetaxel 7.48 is released inside the cancer cell.600
Hypericin 7.18 (SGX301, HyBryte™, SGX302) is a red naphthodianthrone pigment first isolated from St. John's Wort (Hypericum perforatum),602–604 but also present other Hypericum spp. and some fungi.605 Soligenix is developing synthetic hypericin 7.18 as a topical photodynamic therapy in oncology (code SGX-301) and psoriasis (code SGX-302). A phase III trial (NCT02448381)606 combining SGX-301 with fluorescent light for treatment of cutaneous T-cell lymphoma (CTCL, mycosis fungoides) has been completed and a phase III trial (NCT06470451) has begun. A phase II trial with SGX-302 (NCT05442190) for mild-to-moderate psoriasis is ongoing. Hypericin 7.18 can preferentially accumulate in cancer cells and produces light activated reactive oxygen species, which can lead to cellular component damage, while its psoriasis activity against is predominantly via anti-inflammatory and immunomodulatory mechanisms rather than direct cell killing.605,607,608
Ivospemin 7.19 (SBP-101, diethyl dihydroxyhomospermine)609 is a spermine 7.20/homospermine 7.49 polyamine derivative being developed by Panbela Therapeutics. Polyamines are produced by plants,610 animals, bacteria and fungi and play a pivotal role cellular functions.611,612 A combination of ivospemin 7.19, nab-paclitaxel (abraxane) and gemcitabine is being evaluated in a phase II/III (NCT05254171) against pancreatic cancer. Polyamines are essential for growth, proliferation, and survival in mammalian cells611 and their uptake is disrupted by ivospemin 7.19. Specifically, 7.19 has been shown in vitro to decrease ornithine decarboxylase and increase spermidine/spermine N1-acetyltransferase activity.613
Curcumin 6.21 is a diarylheptanoid found in Curcuma longa (turmeric) rhizomes with anti-inflammatory (Section 6), antioxidant, anticancer and antimicrobial activities.496 Liposomal curcumin 6.21 is being evaluated in combination with radiotherapy and temozolomide by SignPath Pharma in a phase I/II (NCT05768919) glioma trial. The NCI are evaluating 6.21 in a phase II trial (NCT02782949) for gastric cancer (chronic atrophic gastritis and/or gastric intestinal metaplasia). Other trials involving curcumin supplements and chemoprevention are ongoing or have been completed.
(±)-Gossypol 7.21, a component in cottonseed oil, is being evaluated as an acetic acid salt by the Zhejiang Cancer Hospital in a phase II trial (NCT06133088) for HR-positive and HER2-negative breast cancer after failure of CDK4/6 treatment. Gossypol has been reported to exhibit a range of activities and mechanisms of action.614–616 Notably, researchers at Zhejiang Cancer Hospital have shown that it promotes degradation of Leucine Rich Pentatricopeptide Repeat Containing (LRPPRC), thereby enhancing sensitivity to CDK4/6 inhibitors both in vitro and in vivo.617 During the 1980s, (±)-gossypol 7.21 was investigated as a male contraceptive in China, but it was discontinued due to its narrow toxicity window,618 while (R)-(−)-gossypol 7.50 (code AT-101) has been evaluated multiple phase I and II oncology trials.614,619
Resveratrol 4.37 is a plant-derived stilbene with antioxidant, anti-inflammatory and anti-cancer properties, which has had a chequered history of clinical development.331,620,621 Researchers at the University of Cincinnati evaluated resveratrol 4.37 in combination with sirolimus in a phase II trial (NCT03253913) for lymphangioleiomyomatosis.622 Resveratrol 4.37 is also being clinically evaluated for mild cognitive impairment/early Alzheimer's disease, and Friedreich's ataxia (Section 7.2).
7.2 Microorganism- and marine invertebrate-derived
Aldoxorubicin 7.22 (DOXO-EMCH, INNO-206) is an acid sensitive doxorubicin 7.23 derivative623 that completed a phase III trial (NCT02049905) in 2017 for the treatment of soft tissue sarcoma by ImmunityBio.624 Doxorubicin 7.22 is an actinomyetes-derived topoisomerase II inhibitor that has been used clinically since the early 1970s.625,626 ImmunityBio did further development work but returned the development rights to LadRx Corporation in June 2024,627 who later announced that they are planning to submit an NDA for soft tissue sarcoma to the FDA.628Berubicin 7.24 (RTA 744, WP744) is a 4′-O-benzylated doxorubicin 7.23 derivative being developed by CNS Pharmaceuticals, which has reduced P-glycoprotein (P-gp) and multidrug resistant protein-1 (MRP1)-mediated efflux, and the ability to cross the brain blood barrier.629–631 Berubicin 7.24 is currently being evaluated in a phase II trial (NCT04762069) for the treatment of recurrent glioblastoma multiforme.
Naxtarubicin 7.25 (L-annamycin) is a synthetic doxorubicin 7.23 derivative632 being developed by Moleculin Biotech, which has a high affinity for lipid membranes633 and reduced cardiotoxicity.634 A liposomal formulation635 of naxtarubicin 7.25 is being evaluated in a phase II trial (NCT05518526) in patients with previously treated soft-tissue sarcomas, while a phase II/III trial (NCT06788756) for AML in combination with cytarabine started in March 2025.
Plinabulin 7.26 (BPI 2358, NPI 2358), being developed by BeyondSpring Pharmaceuticals, is a synthetic derivative636 of the diketopiperazine (−)-phenylahistin 7.27, originally isolated from Aspergillus ustus.637 Both compounds act as tubulin polymerisation inhibitors, binding near the colchicine binding site.638–640 Recently, it was reported that a combination of plinabulin 7.26 and docetaxel 7.16 successfully completed a phase III trial (NCT02504489) for advanced or metastatic non-small-cell lung cancer (NSCLC).641 Plinabulin 7.26 has also been evaluated in other phase III trials: docetaxel 7.48 and plinabulin 7.26versus docetaxel 7.48 and pegfilgrastim in NSCLC (NCT03102606)642 and for the prevention of docetaxel 7.48, doxorubicin 7.23 and cyclophosphamide induced neutropenia in combination with pegfilgrastim (NCT03294577).
Evexomostat 7.28 (SDX-7320) is a fumagillin 7.29 related polymer conjugate prodrug643 being developed by SynDevRx. Fumagillin 7.29 is a fungal metabolite isolated from Aspergillus fumigatus.644–646 It was first introduced clinically in the USA in 1953 as an ameobicide277 and later approved in France in 2005 for the treating diarrhoea associated with intestinal microsporidiosis.647,648 Fumagillin 7.29 irreversibly binds selectively to methionine aminopeptidase 2, and several of its analogues have been evaluated in oncology-related clinical trials.649,650 Although fumagillin 7.29 is available to treat Nosema infections in honeybees,651 it has not been commercially manufactured for human use for several years,649 which has caused issues.652 Evexomostat 7.28 is being evaluated in two breast cancer clinical trials: a phase II trial (NCT05570253) in combination with eribulin 7.51 and a phase I/II trial (NCT05455619) in combination with fulvestrant (ER, estrogen receptor inhibitor) and one of alpelisib (PI3K inhibitor) or capivasertib (AKT inhibitor). Evexomostat 7.28 has a fumagillol 7.52 derivative SDX-7359 7.53 as its active component that is attached to a Gly-Phe-Leu-Gly linker, which is lysosomal protease (e.g. cathepsin B) substrate, and a mixed polymer derived from methacrylic acid and N-(2-hydroxypropyl)-methacrylamide.643
The mushroom Cortinarius orellanus was identified as the causative agent of a mass poisoning in Poland in 1957, affecting 135 people, and its toxic component, orellanine 7.30, was isolated in 1962 with its structure reported in 1979.653,654 Orellanine 7.30 also occurs as mono- and di-glucosylated derivatives.655 Accidental consumption of these mushrooms—often mistaken for edible species—leads to severe kidney and liver damage. Orellanine 7.30 exerts its toxicity over several days, ultimately causing irreversible damage to proximal tubule cells.654,656 Orellanine 7.30 was found to specifically target renal clear cell carcinoma.657 Based on this property, Oncorena AB is sponsoring a phase I/II clinical trial (NCT05287945) evaluating a synthetic form of 7.30 (code ONC175) as a potential treatment for renal cancer.658
Irofulven 7.31 (LP100, MG114) is a semi-synthetic derivative of illudin S 7.32 (lampterol).659,660 Illudin S 7.32 was first reported from the fungus Omphalotus (Clitocybe) illudens in 1950 (ref. 661) and later from Lampteromyces japonicus.662,663 The cytotoxicity of illudins is due to DNA alkylation and DNA adduct formation.662,664,665 Irofulven 7.31 was studied extensively in the early 2000s but its development was halted due to a lack of efficacy in phase III trials.662,666 Allarity Therapeutics completed a phase II trial (NCT03643107) of 7.31 in combination with prednisolone for the treatment of docetaxel-pretreated metastatic castration-resistant prostate cancer in late 2022. In 2021, the global rights were transferred to Lantern Pharma, who are also developing two other illudin derivatives, LP184 7.33 and LP-284 7.34.
LP184 7.33 (STAR-001) is a semi-synthetic illudin S 7.32 derivative and irofulven 7.31 analogue667–670 being evaluated by Lantern Pharma in a phase I trial (NCT05933265) for advanced solid tumours (later focus will be on pancreatic and high-grade gliomas). A subsidiary of Lantern, Starlight Therapeutics, will develop 7.33 in CNS cancers.
LP-284 7.34 is a synthetically produced enantiomer671–673 of LP184 7.33 being evaluated in a phase I trial (NCT06132503) against relapsed or refractory lymphomas and solid tumours (mantle cell and high-grade B-cell lymphoma).
Antroquinonol 7.35, which is being developed by Golden Biotech, is a cytotoxic agent isolated in 2007 from a fermentation of the mushroom Antrodia camphorata (A. cinnamomea),674,675 which has been used traditionally in Taiwan for the treatment of intoxication, liver injury, cancer, and inflammation.676,677 Antroquinonol has a complex MoA including targeting PI3K/AKT/β-catenin signalling,678,679 Nrf-2 activation,680 and Ras and Rho signaling.681 Antroquinonol 7.35 is being evaluated in a phase I/II trial (NCT03310632) in metastatic pancreatic cancer682 and has completed in phase II trials in AML (NCT03823352) and NSCLC (NCT02047344). Antroquinonol 7.35 also recently completed phase II studies for hepatitis B (NCT03625102), COVID-19 (NCT04523181) and atopic dermatitis (NCT03622463).
NUC-7738 7.36 is an aryloxy phosphoramidate prodrug683 of cordycepin 7.37 (3′-deoxy-adenosine)684,685 being developed by NuCana. It is being evaluated in a phase I/II trial (NCT03829254) in patients with advanced solid tumours and lymphoma. Cordycepin 7.37 is one of the active components in Cordyceps militaris, which is used as a traditional Chinese medicine to treat inflammatory diseases and cancer.686,687 NUC-7738 7.36 is rapidly converted in vivo to cordycepin 7.37, which then downregulates signalling pathways involved in cell survival and proliferation, particularly the NF-κB and Wnt/β-catenin pathways.688,689
E6201 7.38 (ER 806201) is a synthetic analogue690 of the fungal metabolite (5Z)-7-oxo-zeaenol 7.39 (LL-Z1640-2) discovered by Eisai,691 being developed by Spirita Oncology and JS InnoPharm.692 E6201 7.38 inhibits ATP binding of several kinases involved in cancer development including MEK1, MEK kinase-1 and FLT3.692–694 After showing promising activity against solid tumours in a phase I trial (NCT00794781),695 the Mayo Clinic started a phase I melanoma trial (NCT05388877) of a combination of E6201 7.38 and dabrafenib in 2022. E6201 7.38 was also evaluated in a phase I/II trial (NCT02418000) for advanced hematologic malignancies with FLT3 and/or Ras Mutations, which was halted due to insufficient efficacy.
E7130 7.40 is a totally synthetic C52-halichondrin-B amine analogue, developed by Kishi's team at Harvard University in collaboration with Eisai researchers.696 It is currently being evaluated in a phase I trial (NCT03444701) for solid tumours.697 Remarkably, E7130 7.40 can be synthesized at a scale that supports its clinical progression, despite its intricate structure.698,699 The halichondrins (e.g. halichondrin B 7.41) are extremely potent cytotoxins first identified in the marine sponge Halichondria okadai in 1985 (ref. 700 and 701) and later in other sponges,702–705 which bind to the GTP binding side of microtubules, although halichondrin aquaculture was trialled on a New Zealand sponge Lissodendoryx sp.,706,707 the discovery of active, truncated synthetic derivatives by Kishi's group reduced interest in sponge-based supply.708,709 Optimisation led to the development of eribulin 7.51,710,711 which was approved by the FDA for the treatment of late-stage breast cancer in 2010 and liposarcoma in 2016. An eribulin containing ADC, farletuzumab ecteribulin 7.54, is also in clinical development (Section 7.3).
PM534 7.42 is a synthetic analogue of PM742 7.43 being developed by PharmaMar,712 which was isolated from the marine sponge Discodermia du Bocage. PM534 7.42 is a microtubule-destabilisation at the colchicine binding site712,713 that is being evaluated in a phase I trial (NCT05835609) for solid tumours.
Trabectedin 7.45 was first reported from the ascidian Ecteinascidia turbinate,714 but later shown to be produced by a symbiotic marine bacterium Candidatus Endoecteinascidia frumentensis.715 Trabectedin 7.45 was first approved by the EMA in 2007 for the treatment of advanced soft tissue sarcoma, while the analogue lurbinectedin 2.26 was first approved by the US FDA in 2020 for the treatment of metastatic small-cell lung cancer (SCLC). PharmaMar are evaluating two additional analogues, eubectedin 7.44 (PM14) and PM54 7.46,716,717 in clinical trials: ecubectedin 7.44 in a phase I/II trial (NCT05146440) in soft tissue sarcomas and other solid tumours alone and in combination with radiotherapy, and PM54 7.46 in a phase I trial (NCT05841563) for solid tumours. Note that the stereochemistry of secondary alcohol (starred) in the PM54 7.46 was drawn analogously to ecubectedin 7.44.
7.3 NP-antibody anticancer conjugates (ADCs)
ADCs are targeted therapeutics consisting of a mAb conjugated to multiple copies of a cytotoxic payload, enabling precise drug delivery to specific cells. ADCs have been extensively reviewed,718–721 including analyses of the pipeline.722–726 Although ADCs can be designed for a range of diseases, the majority are currently being developed for oncology indications. In these cases, the mAb binds selectively to an antigen on cancer cells, directing the cytotoxic payload to tumours while minimising harm to healthy tissue. ADCs comprise three key components: the mAb, the linker, and the warhead. The mAb is optimised for high specificity and affinity to the target antigen, minimal off-target binding, efficient tumour penetration, and low immunogenicity. Linkers, which connect the payload to the mAb, must remain stable in circulation and are classified as cleavable or non-cleavable. Cleavable linkers release the payload in response to specific intracellular conditions, such as enzymatic activity, pH changes, or redox potential. Non-cleavable linkers require ADC internalisation, followed by lysosomal degradation, to release the payload. Warheads are designed to be highly cytotoxic upon release, with good solubility, stability, and minimal off-target effects. Optimising each component is crucial to enhancing anti-cancer efficacy while minimising toxicity to normal cells.As detailed in Section 2.1.4 and Table 2, there have been fifteen ADCs approved globally with NP-derived warheads: dolastatin (×6), camptothecin (×4), calicheamicin (×2), maytansine (×2) and anthramycin (×1) (Fig. 2). ADCs with NP warheads in later stages of clinical development are listed along with their antigen targets in Table 12 (registration, phase III and phase II/III) and Table 13 (phase II), with two ADCs in the registration phase, 24 in phase III, three in phase II/III and 16 in phase II. ADCs in the earlier stages of development were excluded from this review because (i) publicly available information on their warheads is often lacking, and (ii) the number of ADCs in early development is substantial. For instance, a 2023 review estimated that as of January 1, 2023, there were 93 ADCs in phase I and 38 in phase I/II trials, the majority of which contained NP-D warheads.723
 | ||
| Fig. 2 Approved ADC drugs and ADC clinical candidate in registration and phase III trials, and phase II/III and phase II trials by NP warhead at the end of 2024. | ||
| Name (synonyms) | Warhead (class)/antigena | Development status (cancer) | Developer/s |
|---|---|---|---|
| a Antigen synonyms HER2 = ERBB2; HER3 = ERBB3. b Structure of HS-9265 has not been disclosed but is likely to be SHR9265 (rezetecan). Abbreviations: DLBCL, diffuse large B cell lymphoma; HNSCC, head and neck squamous cell carcinoma; NSCLC, non-small cell lung cancer. | |||
| Farletuzumab ecteribulin 7.54 (FZEC, MORAb-202) | Ecteribulin (eribulin, halichondrin)/FOLR1 | Phase II (NSCLC, ovarian, peritoneal, fallopian tube) | Eisai |
| Ixotatug vedotin (TORL-1-23) | MMAE (dolastatin)/Claudin6 | Phase II (ovarian) | TORL Biotherapeutics |
| Mecbotamab vedotin (CAB-AXL, BA-3011) | MMAE (dolastatin)/AXL | Phase II (NSCLC) | BioAlta |
| Misitatug blivedotin (RC88) | MMAE (dolastatin)/MSLN | Phase II (fallopian tube, ovarian, peritoneal) | RemeGen |
| Naratuximab emtansine (Debio1562M, IMGN-529) | DM1 (maytansine)/CD37 | Phase II (DLBCL, non-Hodgkin lymphoma) | Debiopharm |
| Ozuriftamab vedotin (BA-3021) | MMAE (dolastatin)/ROR2 | Phase II (HNSCC) | BioAtla |
| Raludotatug deruxtecan (R-DXd, DS-6000) | Deruxtecan (DXd, camptothecin)/CDH6 | Phase II (NSCLC, lung, solid tumour) | Daiichi Sankyo/Merck & Co |
| Trastuzumab botidotin (A166) | Botidotin (dolastatin)/HER2 | Phase II (breast) | Kelun Biotech (Klus Pharma) |
| BAT8006 | Exatecan (camptothecin)/FOLR1 | Phase II (solid tumours) | Bio-Thera Solutions |
| BB-1701 | Eribulin (halichondrin)/HER2 | Phase II (breast) | BlissBio/Eisai |
| DX126-262 7.60 | Tub114 (tubulysin)/HER2 | Phase II (breast) | Hangzhou DAC Biotechnology |
| FDA022 (trastuzumab)-BB05 | Deruxtecan (DXd, camptothecin)/HER2 | Phase II (solid tumours) | Shanghai Fudan-Zhangjiang Bio-pharmaceutical |
| HS-20089 | HS-9265b (camptothecin)/B7H4 | Phase II (ovarian, endometrial, fallopian tube) | Shanghai Hansoh Biomedical/GSK |
| RC108-ADC | MMAE (dolastatin)/cMET | Phase II (digestive) | RemeGen |
| TQB-2102 | DDDXd (camptothecin)/HER2 × ECD2/ECD4 | Phase II (breast, gynaecological, gastroesophageal) | Chia Tai Tianqing Pharmaceutical |
| YL-202 | YL0010014 (camptothecin)/HER3 | Phase II (breast, NSCLC, HNSCC) | MediLink Therapeutics (Suzhou) Co |
At the end of December 2024 there were that 22 camptothecin-derived warheads (e.g. patritumab deruxtecan 7.57) and 17 dolastatin derived warheads (e.g. telisotuzumab vedotin 7.55) in phase II and beyond, accounting for 39 of the 45 ADCs. Of the remaining six, two were derived from halichondrin B 7.41/eribulin 7.51 (e.g. farletuzumab ecteribulin 7.54), while one each originated from hemiasterlin (e.g. luveltamab tazevibulin 7.58), duocarmycin (e.g. vobramitamab duocarmazine 7.59), maytansine (e.g. trastuzumab emtansine 7.47) and tubulysin (DX126-262 7.60) (Fig. 2). These data show that the majority of NP-D warheads belong to the dolastatin and camptothecin classes.
Representative ADCs from each warhead class are discussed below. Telisotuzumab vedotin 7.55 has the dolastatin 10 analogue MMAE attached to a linker containing an enzyme-cleavable valine-citrulline (Val-Cit) unit727 and was approved by the US FDA on 14 May 2025. The linker and payload are conjugated to the cysteine residues on the mAb. Trastuzumab emtansine 7.56 has the maytansinoid DM1 attached to a bifunctional non-cleavable linker, succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC), which conjugates to the mAb through lysine residues.728 Patritumab deruxtecan 7.57 has a exatecan-type camptothecin warhead attached to a cleavable tetrapeptide (Ala-Phe-Ala-Ala) linker that is connected to the mAb using maleimide chemistry.729 In farletuzumab ecteribulin 7.54, the terminal amine of eribulin 7.51 (Section 7.2) is attached to a cleavable Cit-Val linker that is in turn attached by maleimide chemistry.730 Luveltamab tazevibulin 7.58 is synthesised using an azide–alkyne cycloaddition-based (SPAAC) copper-free reaction between the dibenzocyclooctyne group (DBCO), which is part of the cleavable 3-aminophenyl hemiasterlin linker-warhead SC239, with para-azidomethyl-L-phenylalanine (pAMF) amino acid residues engineered into the mAb luveltamab.731,732 Vobramitamab duocarmazine 7.59 has a duocarmycin derivative, seco-DUocarmycin hydroxyBenzamide Azaindole (DUBA), attached to a cleavable Val-Cit containing linker, which is attached to the mAbvia its cysteine residues. DX126-262 7.60 incorporates a tubulysin B derivative,733 Tub114, as the warhead attached to a non-cleavable linker that branches into pegylated side chain and a maleimide group, which is used for mAb attachment.734,735
There is another ADC, pamlectabart tismanitin (HDP-101),736,737 that targets the B-cell maturation antigen (BCMA) with a cleavable Ala–Val linker and an α-amanitin warhead being evaluated in a phase I/II trial (NCT04879043) in relapsed refractory multiple myeloma. α-Amanitin is an extremely potent toxin, isolated from several mushrooms, including the death cap (Amanita phalloides), that causes severe liver damage by inhibiting RNA polymerase II and III.738 There have also been other ADCs with NP-D warheads that are no longer in development: anthracyclines, vinca alkaloids and docetaxel.
8. Analysis of natural product-derived compounds in clinical trials
At the end of 2024, 129 NP-derived small molecules were undergoing clinical trials (125 unique, with transcrocetin 3.19, bryostatin 1 4.30, resveratrol 4.37 and curcumin 6.21 being evaluated in multiple therapeutic areas). The majority were being investigated for oncology (35, 27.1%, Section 7.1) and neurology (33, 25.6%, Section 3) (Fig. 3). The remaining were distributed across anti-infectives (24, 18.6%), immunological and anti-inflammatory diseases (22, 17.1%) and cardiovascular and metabolic diseases (15, 11.6%). Since the 2014 review,6 there has been a slight increase in the overall number of NP-D compounds from 100, which consisted of 38 in oncology, six in neurology, 26 in anti-infectives, 22 in immunological and anti-inflammatory diseases and 19 in cardiovascular and metabolic diseases. The significant increase in neurology has been driven by a resurgence of interest in psychedelics and related compounds. | ||
| Fig. 3 Classification of the 129 NP-D compounds in clinical development at the end of 2024 by therapeutic area. | ||
Of the 129 NP-D small molecules in clinical development, 39 were in phase I, 65 in phases I/II and II, 21 in phase II/III and III and four with an NDA filed in China or the USA or approved in January 2025 (Fig. 4). The late-stage development candidates in phase III and NDA are listed in Table 14.
 | ||
| Fig. 4 NP-derived compounds in clinical development at the end of 2024 by clinical trial development phase and therapeutic area. | ||
| Compound (synonym) | Classif./Manuf. | Development status, disease (administration) |
|---|---|---|
| a Abbreviations: AML, acute myelogenous leukaemia; ARDS, acute respiratory distress syndrome; CABP, community-acquired bacterial pneumonia; cUTI, complicated urinary tract infections; HCT, haematopoietic cell transplantation; NDA, New Drug Application; NSCLC, non-small cell lung cancer; PTSD, post-traumatic stress disorder. | ||
| Nafithromycin 3.01 | NP-D/SS | Approved 2 January 2025 (India), CABP (oral) |
| Peceleganan 3.02 | NP-D/TS | NDA in China, wound infections (topical) |
| Rongliflozin 5.02 | NP-D/TS | NDA in China, type 2 diabetes mellitus (oral) |
| Vatiquinone 6.01 | NP-D/TS | NDA in the USA, Friedreich's ataxia (oral) |
| HRS-8427 | NP-D/SS | Phase III, cUTI (IV) |
| Rifasutenizol 3.07 | NP-D/SS | Phase III, H. pylori infections (oral, non-systemic) |
| Sabizabulin 3.17 | NP-D/TS | Phase III, ARDS (oral) |
| LSD 4.05 | NP-D/SS | Phase III, generalised anxiety disorder (oral) |
| Psilocybin 4.07 | NP-D/SS | Phase III, treatment-resistant depression (oral) |
| Mebufotenin 4.12 | NP/TS | Phase III, treatment-resistant depression (oral) |
| Midomafetamine 4.15 | NP-D/TS | Phase III, PTSD (oral) |
| Buntanetap 4.33 | NP-D/TS | Phase III, Alzheimer's disease, Parkinson's disease (oral) |
| LH-1801 5.04 | NP-D/TS | Phase III, type 2 diabetes mellitus (oral) |
| Berberine ursodeoxycholate 5.07 | NP-D/TS/I | Phase III, type 2 diabetes mellitus (oral) |
| Recoflavone 6.04 | NP-D/TS | Phase III, dry eye disease (topical) |
| Acoltremon 6.06 | NP-D/SS | Approved 28 May 2025 (USA), dry eye disease (topical) |
| Nelonemdaz 6.08 | NP-D/TS | Phase III, stroke (oral) |
| Mocravimod 6.12 | NP-D/TS | Phase III, AML patients undergoing allogeneic HCT (oral) |
| Patidegib 7.11 | NP-D/SS | Phase III, basal cell nevus syndrome (topical) |
| Paclitaxel obaluronate 7.15 | NP-D/SS | Phase III, bladder cancer (intravesical) |
| Aldoxorubicin 7.22 | NP-D/SS | Phase III, soft tissue sarcoma (IV) |
| Plinabulin 7.26 | NP-D/TS | Phase III, NSCLC and cancer-induced neutropenia (IV) |
9. New natural product drug pharmacophores
There are 38 compounds in development that have 33 new pharmacophores that are not present in currently approved drugs (Table 15). Over half (17) are in oncology, nine in neurology, seven in anti-infectives, three in cardiovascular and metabolic diseases and two in immunological and anti-inflammatory diseases. Bryostatin 1 4.30 is being developed for Alzheimer's disease and multiple sclerosis, while epigallocatechin 3-gallate 6.29 is under investigation for pulmonary fibrosis and cancer chemoprevention. PharmaMar's PM534 7.42, a synthetic analogue712,713 of the sponge-derived PM742 7.43 patented in 2019,739 is notable as the only newly discovered pharmacophore in the past 15 years currently being evaluated in clinical trials.| Compound | Lead compound (source) | Disease area |
|---|---|---|
| a Abbreviations: AML, acute myelogenous leukaemia; DLBCL, diffuse large B-cell lymphoma; NSCLC, non-small cell lung cancer. | ||
| Peceleganan 3.02 | Cecropin (insects, moths) and melittin (bees) | Wound and diabetic foot infections |
| PL-18 3.03 | H. pylori ribosomal protein L1 (bacteria) | Bacterial vaginosis and fungal infections |
| Zaloganan 3.04 | LL37 (human) | Periprosthetic joint infection |
| RG6436 | Arylomycin (actinomycete) | Complicated urinary tract infections |
| Emodepside 3.16 | PF1022A (fungus) | Threadworm and onchocerciasis |
| Q-griffithsin | Griffithsin (red algae) | COVID-19 prophylaxis |
| Sabizabulin 3.17 | Combretastatin A4 3.18 (plant) | Acute respiratory distress syndrome |
| Ibogaine 4.18 | Ibogaine 4.18 (plant) | Alcoholism and opioid related disorders |
| Noribogaine 4.19 | Ibogaine 4.18 (plant) | Alcoholism |
| Tetrodotoxin 4.29 | Tetrodotoxin 4.29 (pufferfish) | Neuropathic and cancer related pain |
| Bryostatin 1 4.30 | Bryostatin 1 4.30 (bryozoan/bacteria) | Alzheimer's disease and multiple sclerosis |
| LCTB-21 4.35 | Leucettamine B 4.36 (sponge) | Alzheimer's disease and down syndrome |
| Orniplabin 4.39 | Orniplabin 4.39 (fungus) | Stroke |
| TMS-008 | Orniplabin 4.39 (fungus) | Acute kidney injury |
| KRN 7000 4.40 | Agelasphin (sponge) | Prevention of graft-versus-host disease |
| Resiniferatoxin 4.43 | Resiniferatoxin 4.43 (plant) | Musculoskeletal pain |
| Vutiglabridin 5.08 | Glabridin 5.09 (plant) | Obesity |
| HGR4113 5.10 | Glabridin 5.09 (plant) | Metabolic dysfunction-associated steatohepatitis |
| QG3030 5.18 | Lappaconitine 5.19 (plant) | Osteoporosis and fractures |
| Rosolutamide 6.20 | Curcumin 6.21 (plant) | Spinal and bulbar muscular atrophy |
| Curcumin 6.21 | Curcumin 6.21 (plant) | Spinal and bulbar muscular atrophy |
| Epigallocatechin 3-gallate 6.29 | Epigallocatechin 3-gallate 6.29 (plant) | Pulmonary fibrosis and cancer chemoprevention |
| Voruciclib 7.09 | Rohitukine 7.10 (plant) | DLBCL and AML |
| Patidegib 7.11 | Cyclopamine 7.12 (plant) | Basal cell nevus syndrome |
| Hypericin 7.18 | Hypericin 7.18 (plant) | Mycosis fungoides-cutaneous T-cell lymphoma |
| (±)-Gossypol 7.21 | (±)-Gossypol 7.21 (plant) | Breast cancer |
| Ivospemin 7.19 | Spermine 7.20 (plant and others) | Pancreatic cancer |
| Plinabulin 7.26 | Phenylahistin 7.27 (fungus) | NSCLC and cancer-induced neutropenia |
| Orellanine 7.30 | Orellanine 7.30 (fungus) | Renal cell carcinoma |
| Irofulven 7.31 | Illudin S 7.32 (fungus) | Prostate cancer |
| LP-184 7.33 | Illudin S 7.32 (fungus) | Solid tumours |
| LP-284 7.34 | Illudin S 7.32 (fungus) | Lymphomas and solid tumours |
| Antroquinonol 7.35 | Antroquinonol 7.35 (fungus) | Pancreatic cancer, AML, NSCLC, hepatitis B, COVID-19 and atopic dermatitis |
| NUC-7738 7.36 | Cordycepin 7.37 (fungus) | Solid tumours and lymphoma |
| E6201 7.38 | 5Z-7-oxo-zeaenol 7.39 (fungus) | Melanoma |
| PM534 7.42 | PM742 7.43 (sponge) | Solid tumours |
| Luveltamab tazevibulin 7.58 | Hemiasterlin warhead (sponge) | Ovarian and fallopian tube cancer |
| Vobramitamab duocarmazine 7.59 | Duocarmycin warhead (actinomycete) | Prostate cancer |
| DX126-262 7.60 | Tubulysin warhead (myxobacteria) | Breast cancer |
10. Conclusions
Natural products have been a cornerstone of medicine for centuries, from ancient remedies to landmark drugs like morphine and quinine, with their impact peaking during the mid-20th century ‘golden age’ of antibiotic discovery and beyond. However, by the late 20th century, advances in synthetic chemistry, molecular biology, pharmacology, combined with the rise of biologics, transformed the drug discovery landscape, relegating NPs from their central role to one of many discovery approaches. So, where do NPs fit in this ever-evolving pharmaceutical landscape today?Analysis of all drugs launched globally between January 2014 and December 2024 identified 56 NP-related drugs, which included 44 NP and NP-derived NCEs (Table 2) and 12 NP-ADCs (Table 3). It is notable that first approvals are now increasingly being obtained by small to mid-sized pharmaceutical companies, with a growing number based in China and, to a lesser extent, India. During this 11-year period, there has been an average of five new NP-D and NP-ADC first drug approvals per year. By the end of June 2025, there have been two and one more NCEs and NP-ADC approvals respectively (Tables 2 and 3). Nine of the 46 launches since 2014 belong to new human drug classes (Tables 2 and 3). Additionally, an analysis of 388 NCEs and 191 NBEs launched globally between 2014 and 2024 revealed that 44 NCEs (11.3%) were NP or NP-derived, while 12 NBEs (6.3%) were NP-ADCs, based on this review's NP definition (Fig. 1 and Section 2). This indicates that 56 or 9.7% of the 579 total drugs approved between 2014 and 2024 were NP-D.
This analysis identified 125 unique NP and NP-D compounds were being evaluated in clinical trials or in the registration phase at the end of 2024, with transcrocetin 3.19, bryostatin 1 4.30, resveratrol 4.37 and curcumin 6.21 being evaluated in two different therapeutic areas. These can be categorised into the following therapeutic areas: oncology (35, 27.1%), neurology (33, 25.6%) anti-infectives (24, 18.6%), immunology and inflammation (22, 17.1%), and cardiovascular and metabolic diseases (15, 11.6%). Renewed interest in psychedelics has contributed to a resurgence in neurology clinical trials. As of 2025, there were 23 small molecules in phase III trials or registration and two approved (Fig. 4 and Table 14). In parallel, 25 NP-ADCs were in phase III, with one in registration and one approved in 2025 (Table 12). These figures suggest that several additional NP-D and NP-ADC drugs are likely to be launched in the coming years. Although 33 new pharmacophores not found in currently approved drugs are in clinical development (Table 15), PharmaMar's PM534 7.42, a synthetic analogue712,713 of the sponge-derived PM742 7.43 patented in 2019,739 stands out as the only newly discovered pharmacophore in the past 15 years in active development.
In conclusion, although biologically focused NP lead discovery has declined within the pharmaceutical industry, a modest yet steady number of NP-derived drugs continues to gain approval each year. Development and regulatory progress in this area now largely depend on small to medium-sized enterprises, which are often narrowly focused on specific therapeutic areas. As a result, broad screening against novel targets is no longer routine. Although 33 new pharmacophores are in development, only one has been discovered in the past 15 years, indicating a worrying slowdown in innovation. NPs remain a vital reservoir of structurally complex and mechanistically diverse molecules, but without the discovery of new bioactive NPs or novel modes of action for known compounds, this reservoir risks depletion. The loss of this source of innovation would significantly hinder our capacity to address emerging and drug-resistant diseases.
11. Conflicts of interest
There are no conflicts of interest to declare.12. Data availability
The data used to produce Fig. 1–4 can be obtained from the SI. See DOI: https://doi.org/10.1039/d5np00031a.13. Acknowledgements
The Australian Research Council Industrial Transformation Training Centre for Environmental and Agricultural Solutions to Antimicrobial Resistance (IC220100050) is funded by its Partners and the Australian Government.14. Notes and references
- E. Araujo, B. Bartels, I. M. Bell, G. Castanedo, M. Zeng, T. G. Murali Dhar, N. Holmberg-Douglas, E. R. Welin, D. C. Koester, B. León, J. R. Manning, J. Robert Merritt, K. M. Peese, S. Shockley, A. Tran and J. J. Bronson, in 2024 Medicinal Chemistry Reviews, MEDI, Inc. Published by American Chemical Society, 2024, vol. 59, ch. 19, p. 417 Search PubMed.
- S. A. Lim, N. J. Agard, R. L. Kelly, B. N. Bell, S.-J. Chen, A. M. Gram, I. Moench, K. Pance and T. Petojevic, in 2024 Medicinal Chemistry Reviews, MEDI, Inc. Published by American Chemical Society, 2024, vol. 59, ch. 20, p. 553 Search PubMed.
- M. S. Butler, Nat. Prod. Rep., 2005, 22, 162 RSC.
- M. S. Butler, Nat. Prod. Rep., 2008, 25, 475 RSC.
- M. S. Butler, in Natural Product Chemistry for Drug Discovery, ed. A. D. Buss and M. S. Butler, The Royal Society of Chemistry, 2009, ch. 11, p. 321, 10.1039/9781847559890-00321.
- M. S. Butler, A. A. B. Robertson and M. A. Cooper, Nat. Prod. Rep., 2014, 31, 1612 RSC.
- R. Ng, Drugs: From Discovery to Approval, 3rd Edition, John Wiley & Sons, Hoboken, NJ, USA, 2015 Search PubMed.
- J. Bronson, K. Peese, A. Black, M. Dhar, A. Pashine, B. A. Ellsworth and J. R. Merritt, in 2015 Medicinal Chemistry Reviews, ed. C. D. Manoj, MEDI, Inc. Published by American Chemical Society, 2015, vol. 50, ch. 19, p. 461 Search PubMed.
- J. Bronson, K. Peese, M. Dhar, A. Pashine, F. J. Duclos, B. A. Ellsworth, R. Garcia and J. R. Merritt, in 2016 Medicinal Chemistry Reviews, ed. C. D. Manoj, MEDI, Inc. Published by American Chemical Society, 2016, vol. 51, ch. 25, p. 439 Search PubMed.
- C. A. Bolger, T. G. M. Dhar, A. Pashine, P. S. Dragovich, W. Mallet, K. M. Peese and J. R. Merritt, in 2017 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2017, vol. 52, p. 531 Search PubMed.
- C. A. Bolger, J. E. Carpenter, T. G. M. Dhar, A. Pashine, P. S. Dragovich, J. H. Cook, E. P. Gillis, K. M. Peese and J. R. Merritt, in 2016 Medicinal Chemistry Reviews, ed. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2018, vol. 53, p. 587 Search PubMed.
- E. Araujo, M.-G. Braun, P. S. Dragovich, A. Converso, P. G. Nantermet, A. J. Roecker, T. G. M. Dhar, P. Haile, A. Hurtley, J. R. Merritt and K. M. Peese, in 2019 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2019, vol. 54, p. 469 Search PubMed.
- C. A. Bolger, S. A. Kahn, D. Lipovšek, W. Mallet and J. Wieler, in 2019 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2019, vol. 54, ch. 23, p. 597 Search PubMed.
- N. J. Agard, P. S. Dragovich, A. M. Beal, C. A. Bolger, S. A. Kahn, B. Hubbard and D. Lipovšek, in 2020 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2020, p. 749 Search PubMed.
- E. Araujo, C. A. Bolger, M.-G. Braun, G. Castanedo, M. Zeng, A. K. Charnley, A. Converso, I. T. Raheem, A. J. Roecker, A. A. Shah, T. G. M. Dhar, D. C. Grünenfelder, J. R. Merritt and K. M. Peese, in 2020 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2020, vol. 55, p. 625 Search PubMed.
- N. J. Agard, P. S. Dragovich, R. L. Kelly, S. A. Lim, A. M. Beal, I. Moench, A. M. Gram, P.-P. Kung and D. Lipovšek, in 2020 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2021, ch. 22, p. 841 Search PubMed.
- M.-G. Braun, G. Castanedo, S. A. Green, M. Zeng, J. Carpenter, A. Converso, I. T. Raheem, A. J. Roecker, A. A. Shah, L. Debien, T. G. M. Dhar, L. M. Holder, J. R. Merritt, K. M. Peese, D. C. Grünenfelder and J. C. Lo, in 2020 Medicinal Chemistry Reviews, ed. J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2021, ch. 21, p. 683 Search PubMed.
- N. J. Agard, P. S. Dragovich, R. L. Kelly, S. A. Lim, A. M. Beal, I. Moench, S.-J. Chen, A. M. Gram and P.-P. Kung, in 2022 Medicinal Chemistry Reviews, ed. J. Rudolph and J. J. Bronson, MEDI, Inc. Published by American Chemical Society., 2022, vol. 57, ch. 25, p. 733 Search PubMed.
- E. Araujo, I. M. Bell, A. Burckle, D. C. Koester, J. R. Manning, R. D. Caldwell, G. Castanedo, S. A. Green, M. Zeng, L. Debien, T. G. M. Dhar, N. Holmberg-Douglas, E. R. Welin, J. R. Merritt, K. M. Peese, D. C. Grünenfelder and J. C. Lo, in 2022 Medicinal Chemistry Reviews, ed. J. Rudolph and J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2022, vol. 57, ch. 24, p. 587 Search PubMed.
- E. Araujo, I. M. Bell, A. Burckle, D. C. Koester, J. R. Manning, G. Castanedo, M. Zeng, T. G. Murali Dhar, N. Holmberg-Douglas, E. R. Welin, J. R. Merritt, K. M. Peese and J. J. Bronson, in 2023 Medicinal Chemistry Reviews, ed. J. Rudolph and J. J. Bronson, MEDI, Inc. Published by American Chemical Society, 2023, vol. 58, ch. 24, pp. 595 Search PubMed.
- S. A. Lim, N. J. Agard, R. L. Kelly, B. N. Bell, S.-J. Chen, A. M. Gram, J. R. Merritt, I. Moench, K. Pance and T. Petojevic, in 2023 Medicinal Chemistry Reviews, ed. J. Rudolph and J. J. Bronson, MEDI, Inc. Published by American Chemical Society., 2023, vol. 58, ch. 25, pp. 713 Search PubMed.
- A. Mullard, Nat. Rev. Drug Discovery, 2024, 23, 88 CrossRef CAS.
- A. Papapetropoulos, S. Topouzis, S. P. H. Alexander, M. Cortese-Krott, D. A. Kendall, K. A. Martemyanov, C. Mauro, N. Nagercoil, R. A. Panettieri Jr, H. H. Patel, R. Schulz, B. Stefanska, G. J. Stephens, M. M. Teixeira, N. Vergnolle, X. Wang and P. Ferdinandy, Br. J. Pharmacol., 2024, 181, 1553 CrossRef CAS PubMed.
- A. Mullard, Nat. Rev. Drug Discovery, 2025, 24, 75 CrossRef CAS PubMed.
- X. Zhi, Q. Li and L. Shao, Nat. Rev. Drug Discovery, 2025, 24, 160 CrossRef CAS.
- S. Topouzis, A. Papapetropoulos, S. P. H. Alexander, M. Cortese-Krott, D. A. Kendall, K. Martemyanov, C. Mauro, N. Nagercoil, R. A. Panettieri Jr, H. H. Patel, R. Schulz, B. Stefanska, G. J. Stephens, M. M. Teixeira, N. Vergnolle, X. Wang and P. Ferdinandy, Br. J. Pharmacol., 2025, 182, 1416 CrossRef CAS.
- KEGG, New drug approvals in the USA, Europe and Japan, https://www.genome.jp/kegg/drug/br08328.html, accessed 20 March 2025.
- K. Gao, C. Zheng, T. Wang, H. Zhao, J. Wang, Z. Wang, X. Zhai, Z. Jia, J. Chen, Y. Zhou and W. Wang, Molecules, 2016, 21, 1600 CrossRef.
- C. M. Rath, B. Janto, J. Earl, A. Ahmed, F. Z. Hu, L. Hiller, M. Dahlgren, R. Kreft, F. Yu, J. J. Wolff, H. K. Kweon, M. A. Christiansen, K. Håkansson, R. M. Williams, G. D. Ehrlich and D. H. Sherman, ACS Chem. Biol., 2011, 6, 1244 CrossRef CAS.
- H. Luesch, R. E. Moore, V. J. Paul, S. L. Mooberry and T. H. Corbett, J. Nat. Prod., 2001, 64, 907 CrossRef CAS.
- D. Kallifidas, D. Dhakal, M. Chen, Q.-Y. Chen, S. Kokkaliari, N. A. Colon Rosa, R. Ratnayake, S. D. Bruner, V. J. Paul, Y. Ding and H. Luesch, Org. Lett., 2024, 26, 1321 CrossRef CAS.
- S. M. Kupchan, Y. Komoda, W. A. Court, G. J. Thomas, R. M. Smith, A. Karim, C. J. Gilmore, R. C. Haltiwanger and R. F. Bryan, J. Am. Chem. Soc., 1972, 94, 1354 CrossRef CAS PubMed.
- S. Kusari, M. Lamshöft, P. Kusari, S. Gottfried, S. Zühlke, K. Louven, U. Hentschel, O. Kayser and M. Spiteller, J. Nat. Prod., 2014, 77, 2577 CrossRef CAS.
- L. J. Scott, Drugs, 2019, 79, 315 CrossRef CAS PubMed.
- E. D. Deeks, Drugs, 2019, 79, 325 CrossRef CAS.
- A. Markham and S. J. Keam, Drugs, 2018, 78, 1931 CrossRef.
- L. J. Scott, Drugs, 2015, 75, 1281 CrossRef CAS PubMed.
- K. D. Brade, J. M. Rybak and M. J. Rybak, Infect. Dis. Ther., 2016, 5, 1 CrossRef.
- Y. Y. Syed, Drugs, 2021, 81, 1559 CrossRef CAS PubMed.
- G. G. Zhanel, M. Pozdirca, A. R. Golden, C. K. Lawrence, S. Zelenitsky, L. Berry, F. Schweizer, D. Bay, H. Adam, M. A. Zhanel, P. Lagacé-Wiens, A. Walkty, N. Irfan, K. Naber, J. P. Lynch and J. A. Karlowsky, Drugs, 2022, 82, 533 CrossRef CAS PubMed.
- S. J. Keam, Drugs, 2024, 84, 737 CrossRef CAS PubMed.
- K. M. Shaeer, M. T. Zmarlicka, E. B. Chahine, N. Piccicacco and J. C. Cho, Pharmacotherapy, 2019, 39, 77 CrossRef CAS.
- G. G. Zhanel, C. Deng, S. Zelenitsky, C. K. Lawrence, H. J. Adam, A. Golden, L. Berry, F. Schweizer, M. A. Zhanel, N. Irfan, D. Bay, P. Lagacé-Wiens, A. Walkty, L. Mandell, J. P. Lynch and J. A. Karlowsky, Drugs, 2021, 81, 233 CrossRef CAS.
- W. He, C. Yang, X. Zhao and Y. Wang, Bioorg. Med. Chem. Lett., 2017, 27, 4576 CrossRef CAS PubMed.
- W. Zhu, Y. Tian, L. Xiang, L. Cao and L. He, Infect. Drug Resist., 2023, 16, 2365 CrossRef.
- J. M. Apgar, R. R. Wilkening, D. L. Parker, D. Meng, K. J. Wildonger, D. Sperbeck, M. L. Greenlee, J. M. Balkovec, A. M. Flattery, G. K. Abruzzo, A. M. Galgoci, R. A. Giacobbe, C. J. Gill, M.-J. Hsu, P. Liberator, A. S. Misura, M. Motyl, J. Nielsen Kahn, M. Powles, F. Racine, J. Dragovic, W. Fan, R. Kirwan, S. Lee, H. Liu, A. Mamai, K. Nelson and M. Peel, Bioorg. Med. Chem. Lett., 2021, 32, 127661 CrossRef CAS.
- K. N. Barnes, A. M. Yancey and A. B. Forinash, Ann. Pharmacother., 2023, 57, 99 CrossRef CAS PubMed.
- Y. Y. Syed, Drugs, 2023, 83, 833 CrossRef CAS.
- F. Vicente, F. Reyes and O. Genilloud, Nat. Prod. Rep., 2024, 41, 1835 RSC.
- G. Garcia-Effron, J. Fungi, 2020, 6, 262 CrossRef CAS PubMed.
- S. J. Keam, Pediatr. Drugs, 2024, 26, 95 CrossRef PubMed.
- G.-S. Wang, J. Ethnopharmacol., 1989, 26, 147 CrossRef CAS PubMed.
- L. Moed, T. A. Shwayder and M. W. Chang, Arch. Dermatol., 2001, 137, 1357 CAS.
- P. Milton, J. I. D. Hamley, M. Walker and M.-G. Basáñez, Expert Rev. Anti Infect. Ther., 2020, 18, 1067 CrossRef CAS PubMed.
- C.-J. Li, M.-Y. Ku, C.-Y. Lu, Y.-E. Tien, W. H. Chern and J.-d. Huang, Int. J. Pharm., 2017, 531, 306 CrossRef CAS.
- J. Paik, Drugs, 2021, 81, 1431 CrossRef CAS PubMed.
- P. Sauerberg, P. H. Olesen, S. Nielsen, S. Treppendahl, M. J. Sheardown, T. Honore, C. H. Mitch, J. S. Ward and A. J. Pike, J. Med. Chem., 1992, 35, 2274 CrossRef CAS PubMed.
- A. D. Volgin, A. Bashirzade, T. G. Amstislavskaya, O. A. Yakovlev, K. A. Demin, Y.-J. Ho, D. Wang, V. A. Shevyrin, D. Yan, Z. Tang, J. Wang, M. Wang, E. T. Alpyshov, N. Serikuly, E. A. Wappler-Guzzetta, A. M. Lakstygal and A. V. Kalueff, ACS Chem. Neurosci., 2019, 10, 2176 CrossRef CAS PubMed.
- H. Madersbacher and E. Rovner, Expert Opin. Pharmacother., 2006, 7, 1373 CrossRef CAS PubMed.
- A. Breier, S. K. Brannan, S. M. Paul and A. C. Miller, Psychopharmacology, 2023, 240, 1191 CrossRef CAS.
- L. Nikola, T. M. Mirjana, G. Zoran, R. Manfredi, R. Djordje, S. S. Jasjit and R. I. Esma, Curr. Med. Chem., 2024, 31, 4781 CrossRef.
- E. H. McCafferty and L. J. Scott, Drugs, 2019, 79, 543 CrossRef CAS PubMed.
- S. Chackalamannil, Y. Wang, W. J. Greenlee, Z. Hu, Y. Xia, H.-S. Ahn, G. Boykow, Y. Hsieh, J. Palamanda, J. Agans-Fantuzzi, S. Kurowski, M. Graziano and M. Chintala, J. Med. Chem., 2008, 51, 3061 CrossRef CAS PubMed.
- J. E. Frampton, Drugs, 2015, 75, 797 CrossRef CAS.
- A. Khoury, K. A. Pagan and M. Z. Farland, Ann. Pharmacother., 2021, 55, 222 CrossRef CAS.
- K. P. Garnock-Jones, Drugs, 2015, 75, 419 CrossRef CAS.
- H. A. Blair, Drugs, 2019, 79, 1241 CrossRef CAS PubMed.
- L. J. Scott, CNS Drugs, 2020, 34, 1191 CrossRef CAS PubMed.
- M. Shirley, Drugs, 2024, 84, 247 CrossRef CAS PubMed.
- T. van Gelder, E. Lerma, K. Engelke and R. B. Huizinga, Expert Rev. Clin. Pharmacol., 2022, 15, 515 CrossRef CAS PubMed.
- J. I. Silverberg, M. Boguniewicz, F. J. Quintana, R. A. Clark, L. Gross, I. Hirano, A. M. Tallman, P. M. Brown, D. Fredericks, D. S. Rubenstein and K. A. McHale, J. Allergy Clin. Immunol., 2024, 154, 1 CrossRef CAS.
- A. Lee, Drugs, 2023, 83, 725 CrossRef CAS PubMed.
- E. S. Kim, Drugs, 2017, 77, 1251 CrossRef CAS PubMed.
- Pharma Mar, APLIDIN®, https://pharmamar.com/en/products/aplidin/, accessed 7 April 2025.
- S. Alonso-Álvarez, E. Pardal, D. Sánchez-Nieto, M. Navarro, M. D. Caballero, M. V. Mateos and A. Martín, Drug Des., Dev. Ther., 2017, 11, 253 CrossRef.
- J. F. M. Leal, M. Martínez-Díez, V. García-Hernández, V. Moneo, A. Domingo, J. A. Bueren-Calabuig, A. Negri, F. Gago, M. J. Guillén-Navarro, P. Avilés, C. Cuevas, L. F. García-Fernández and C. M. Galmarini, Br. J. Pharmacol., 2010, 161, 1099 CrossRef CAS PubMed.
- C. Cuevas and A. Francesch, Nat. Prod. Rep., 2009, 26, 322 RSC.
- Y. Shi, G. Chen, Y. Zhao, J. Zhao and L. Lin, Cancer Pathog. Ther., 2024, 2, 103 CrossRef.
- N. T. Huong and N. T. Son, Phytochemistry, 2023, 213, 113772 CrossRef CAS.
- R. Guo, Z. Yan, R. Wang, T. Guo, H. Li, M. Kong and W. Guo, Am. J. Chin. Med., 2025, 53, 179 CrossRef CAS PubMed.
- S. Xie, F. Zhan, J. Zhu, S. Xu and J. Xu, Expert Opin. Drug Discovery, 2025, 20, 827 CrossRef CAS.
- B. R. Mangum, J. M. Pogue and K. E. Barber, Curr. Infect. Dis. Rep., 2024, 26, 139 CrossRef.
- GSK, PIVOT-PO phase III study for tebipenem HBr stopped early for efficacy following review by Independent Data Monitoring Committee, Press Release, 28 May 2025, https://www.gsk.com/en-gb/media/press-releases/pivot-po-phase-iii-study-for-tebipenem-hbr-stopped-early-for-efficacy-following-review-by-independent-data-monitoring-committee/, accessed 10 July 2025.
- Y. Carmeli, J. M. Cisneros, M. Paul, G. L. Daikos, M. Wang, J. Torre-Cisneros, G. Singer, I. Titov, I. Gumenchuk, Y. Zhao, R.-M. Jiménez-Rodríguez, L. Liang, G. Chen, O. Pyptiuk, F. Aksoy, H. Rogers, M. Wible, F. F. Arhin, A. Luckey, J. L. Leaney, R. Pypstra, J. W. Chow, M. Bonten, J. Rodríguez-Baño, C. Lammens, H. Goossens and S. Malhotra-Kumar, Lancet Infect. Dis., 2025, 25, P218 CrossRef.
- A. Zasheva, E. Batcheva, K. D. Ivanova and A. Yanakieva, Antibiotics, 2024, 13, 1077 CrossRef CAS PubMed.
- V. Trebosc, C. Kemmer, S. Lociuro, M. Gitzinger and G. E. Dale, Drug Discovery Today, 2021, 26, 2099 CrossRef CAS.
- J. Rouden, M.-C. Lasne, J. Blanchet and J. Baudoux, Chem. Rev., 2013, 114, 712 CrossRef PubMed.
- D. Lipanovic, Pharm. J., 2023, 311, 7980 Search PubMed.
- Quit Centre, Cytisine for cessation, https://www.quitcentre.org.au/news/cytisine-for-cessation, accessed 7 April 2025.
- M. S. Butler, I. R. Henderson, R. J. Capon and M. A. T. Blaskovich, J. Antibiot., 2023, 76, 431 CrossRef CAS PubMed.
- W. V. M. S. Butler, E. C. A. Goodall, R. J. Capon, I. R. Henderson and M. A. T. Blaskovich, ACS Infect. Dis., 2024, 10, 3440 CrossRef PubMed.
- E. M. Duffy, E. T. Buurman, S. L. Chiang, N. R. Cohen, M. Uria-Nickelsen and R. A. Alm, ACS Infect. Dis., 2021, 7, 2043 CrossRef CAS.
- K. Outterson, Health Aff., 2021, 40, 1758–1765 CrossRef PubMed.
- A. Engel, ACS Infect. Dis., 2020, 6, 1311 CrossRef CAS PubMed.
- C. Leonard, N. Crabb, D. Glover, S. Cooper, J. Bouvy, M. Wobbe and M. Perkins, Appl. Health Econ. Health Pol., 2023, 21, 365 CrossRef.
- J. H. Rex, H. Fernandez Lynch, I. G. Cohen, J. J. Darrow and K. Outterson, Nat. Commun., 2019, 10, 3416 CrossRef PubMed.
- U. Theuretzbacher and L. J. V. Piddock, Cell Host Microbe, 2019, 26, 61 CrossRef CAS.
- D. M. Shlaes, ACS Infect. Dis., 2021, 7, 2027 CrossRef CAS.
- M. S. Butler, V. Gigante, H. Sati, S. Paulin, L. Al-Sulaiman, J. H. Rex, P. Fernandes, C. A. Arias, M. Paul, G. E. Thwaites, L. Czaplewski, R. A. Alm, C. Lienhardt, M. Spigelman, L. L. Silver, N. Ohmagari, R. Kozlov, S. Harbarth and P. Beyer, Antimicrob. Agents Chemother., 2022, 66, e01991 CAS.
- C. F. Neoh, W. Jeong, D. C. M. Kong and M. A. Slavin, Expert Rev. Anti Infect. Ther., 2023, 21, 577 CrossRef CAS PubMed.
- A. von Delft, M. D. Hall, A. D. Kwong, L. A. Purcell, K. S. Saikatendu, U. Schmitz, J. A. Tallarico and A. A. Lee, Nat. Rev. Drug Discovery, 2023, 22, 585 CrossRef CAS PubMed.
- M. Bello-Akinosho, K. O. Afolabi, H. Chandra, D. K. Mayashinta, Y. D. Setia, A. Prakash Mishra and C. Pohl-Albertyn, in Parasitic Infections, 2023, ch. 11, p. 227, DOI:10.1002/9781119878063.ch11.
- X. Shang, L. Dai, X. Cao, Y. Ma, I. Gulnaz, X. Miao, X. Li and X. Yang, Nat. Prod. Rep., 2025 10.1039/D5NP00007F , In press.
- M. A. Hackel, J. A. Karlowsky, D. Dressel and D. F. Sahm, J. Clin. Microbiol., 2017, 55, 3021 CrossRef CAS PubMed.
- S. Kohlhoff and M. R. Hammerschlag, Antimicrob. Agents Chemother., 2021, 65, e00585 CrossRef CAS.
- M. Zhou, L. Wu, W. Kang, Y. Li, G. Zhang, J. Zhang, S. Duan, J. Li, T. Wang, Y. Xu and Y. Gu, JAC-Antimicrob. Resist., 2022, 4, dlac103 CrossRef PubMed.
- Wockhardt Limited, Wockhardt announces successful completion of pivotal Phase 3 pneumonia study of its macrolide antibiotic Nafithromycin WCK 4873 (Press Release 11 December 2023), https://www.wockhardt.com/wp-content/uploads/2023/12/wockhardt-announces-successful-completion-of-pivotal-phase-3-pneumonia-study-of-its-macrolide-antibiotic-nafithromycin-wck-4873.pdf, accessed 7 April 2025.
- Wockhardt Limited, Indian Drug Regulator approves Wockhardt's new generation oral antibiotic Miqnaf® (Nafithromycin) for the treatment of community-acquired bacterial pneumonia (CABP), Press Release 2 January 2025, https://www.wockhardt.com/wp-content/uploads/2025/01/press-release.pdf, accessed 7 April 2025.
- Y. Chen, C. T. Mant, S. W. Farmer, R. E. W. Hancock, M. L. Vasil and R. S. Hodges, J. Biol. Chem., 2005, 280, 12316 CrossRef CAS.
- Q. Feng, Y. Huang, M. Chen, G. Li and Y. Chen, Eur. J. Clin. Microbiol. Infect. Dis., 2015, 34, 197 CrossRef CAS.
- Y. Wei, Y. Li, X. Li, Y. Zhao, J. Xu, H. Wang, X. Rong, J. Xiong, X. Chen, G. Luo, G. Lv, C. Lin, C. Han, H. Yu, Y. Zhang, S. Tang, Y. Fan, J. Tu, C. Xia, H. Zu, W. Liu, C. Liu, J. Liu, B. Zhang, Q. Nong, T. Li, L. Wang, G. Song, Y. Su, Z. Chen, W. Lai, Y. Fu, J. a. Yu, P. Zhang, W. Yang, G. Yao, H. Zhang, K. Fan, H. Dong, Y. Chen, J. Wu and PL-5 Investigators, JAMA Netw. Open, 2024, 7, e2415310 CrossRef PubMed.
- L. Qian, A. Wang, W. Hu, G. Wang, S. Xinjuan and Y. Liu, Diabetes, 2024, 73, 173 CrossRef.
- L. Zhao, Y. Huang, S. Gao, Y. Cui, D. He, L. Wang and Y. Chen, Sci. China: Chem., 2013, 56, 1307 CrossRef CAS.
- B. Deslouches, K. Islam, J. K. Craigo, S. M. Paranjape, R. C. Montelaro and T. A. Mietzner, Antimicrob. Agents Chemother., 2005, 49, 3208 CrossRef CAS PubMed.
- B. Deslouches, J. D. Steckbeck, J. K. Craigo, Y. Doi, J. L. Burns and R. C. Montelaro, Antimicrob. Agents Chemother., 2015, 59, 1329 CrossRef CAS PubMed.
- D. B. Huang, K. M. Brothers, J. B. Mandell, M. Taguchi, P. G. Alexander, D. M. Parker, D. Shinabarger, C. Pillar, I. Morrissey, S. Hawser, P. Ghahramani, D. Dobbins, N. Pachuda, R. Montelaro, J. D. Steckbeck and K. L. Urish, PLoS One, 2022, 17, e0274815 CrossRef CAS PubMed.
- D. Huang, N. Pachuda, J. M. Sauer, D. Dobbins and J. Steckbeck, Antibiotics, 2022, 11, 41 CrossRef CAS.
- D. Huang, D. Dobbins, P. Ghahramani, I. Friedland and J. Steckbeck, Antimicrob. Agents Chemother., 2022, 66, e01441 CAS.
- CARB-X, CARB-X funds Peptilogics to develop a novel, broad-spectrum therapeutic to treat fracture-related infections, Press Rlease 9 January 2025, https://carb-x.org/carb-x-news/carb-x-funds-peptilogics-to-develop-a-novel-broad-spectrum-therapeutic-to-treat-fracture-related-infections/, accessed 7 April 2025.
- J. Huang, L. Zhu, Y. Zou and C. Zhang, US Pat., US20240132518A1, 2022 Search PubMed.
- X. Yan, Y. Huang, J. Xie, Q. Wu, S. Yang, X. Yang, H. Chen, J. Huang and G. Yang, Antimicrob. Agents Chemother., 2024, 68, e00524 Search PubMed.
- X. Yang, C. Li, X. Wang, Z. Zheng, P. Sun, C. Xu, L. Chen, J. Jiang, S. Normark, B. Henriques-Normark and X. You, Engineering, 2024, 38, 52 CrossRef CAS.
- T. Sato and K. Yamawaki, Clin. Infect. Dis., 2019, 69, S538 CrossRef CAS PubMed.
- L. Wang, J. Zhu, L. Chen and H. Du, Drug Resistance Updates, 2024, 72, 101034 CrossRef CAS.
- C.-Y. Zhao, Y. Lv, Y. Zhu, M.-J. Wei, M.-Y. Liu, X.-W. Ji, Z.-S. Kang, Y.-H. Xia, J.-H. Tian, Y. Ma and Y. Liu, Antimicrob. Agents Chemother., 2019, 63, e02188 CAS.
- H. Yang, M. Zhang, Y. Chen, H. Ren, H. Zhang, C. Yu, J. Lu, L. You, J. Yu, H. Liang, C. Xiao, Z. He, J. Wu, J. Xue and J. Zhang, Eur. J. Clin. Pharmacol., 2022, 78, 1079 CrossRef CAS PubMed.
- Shanghai Pharmaceuticals Holding Co, Shanghai Pharmaceuticals Holding Co., Ltd 2023 Annual Report, https://www.hkexnews.hk/listedco/listconews/sehk/2024/0426/2024042600622.pdf, accessed 7 April 2025.
- G. V. Doern, G. Pierce and A. B. Brueggemann, Diagn. Microbiol. Infect. Dis., 1996, 26, 39 CrossRef CAS PubMed.
- S. Biondi, E. Piga, T. Rossi and G. Vigelli, Bioorg. Med. Chem. Lett., 1997, 7, 2061 CrossRef CAS.
- Working Group on TB Drugs, Sanfertrinem https://www.newtbdrugs.org/pipeline/compound/sanfetrinem, accessed 7 April 2025.
- S. Ramón-García, R. González del Río, M. P. Arenaz-Callao, H. I. Boshoff, J. Rullás, S. Anca, M. C. Izquierdo, E. Porras de Francisco, E. P. Herrán, A. Santos-Villarejo, A. Mendoza-Losana, S. Ferrer-Bazaga, C. J. Thompson, D. B. Aguirre and R. H. Bates, Drug Resistance Updates, 2024, 90, 101213 Search PubMed.
- A. H. Diacon, L. van der Merwe, M. Barnard, F. von Groote-Bidlingmaier, C. Lange, A. L. García-Basteiro, E. Sevene, L. Ballell and D. Barros-Aguirre, N. Engl. J. Med., 2016, 375, 393 CrossRef PubMed.
- B. Gold, J. Zhang, L. L. Quezada, J. Roberts, Y. Ling, M. Wood, W. Shinwari, L. Goullieux, C. Roubert, L. Fraisse, E. Bacqué, S. Lagrange, B. Filoche-Rommé, M. Vieth, P. A. Hipskind, L. N. Jungheim, J. Aubé, S. M. Scarry, S. L. McDonald, K. Li, A. Perkowski, Q. Nguyen, V. Dartois, M. Zimmerman, D. B. Olsen, K. Young, S. Bonnett, D. Joerss, T. Parish, H. I. Boshoff, K. Arora, C. E. Barry III, L. Guijarro, S. Anca, J. Rullas, B. Rodríguez-Salguero, M. S. Martínez-Martínez, E. Porras-De Francisco, M. Cacho, D. Barros-Aguirre, P. Smith, S. J. Berthel, C. Nathan and R. H. Bates, ACS Infect. Dis., 2022, 8, 557 CrossRef CAS.
- Z. Ma, S. He, Y. Yuan, Z. Zhuang, Y. Liu, H. Wang, J. Chen, X. Xu, C. Ding, V. Molodtsov, W. Lin, G. T. Robertson, W. J. Weiss, M. Pulse, P. Nguyen, L. Duncan, T. Doyle, R. H. Ebright and A. S. Lynch, J. Med. Chem., 2022, 65, 4481 CrossRef CAS PubMed.
- X. Li, Y. Liu, M. Wang, L. Gao, J. Liu, H. Zhang, M. Wu, H. Chen, J. Lou, J. Wang, J. Chen, G. Geng, Z. Ma and Y. Ding, Lancet Infect. Dis., 2024, 24, 650 CrossRef CAS PubMed.
- Z. Ma and A. S. Lynch, J. Med. Chem., 2016, 59, 6645 CrossRef CAS PubMed.
- G. T. Robertson, E. J. Bonventre, T. B. Doyle, Q. Du, L. Duncan, T. W. Morris, E. D. Roche, D. Yan and A. S. Lynch, Antimicrob. Agents Chemother., 2008, 52, 2313 CrossRef CAS.
- R. Thakare, A. Dasgupta and S. Chopra, Drugs Future, 2021, 46, 129 CrossRef.
- T. Dai, C. Ma, F. Zhang, H. Wang, Z. Ma, H. Wang, Y. Wen and L. Chen, J. Infect. Dis., 2024, 229, 1658 CrossRef CAS.
- J. Bruss, T. Lister, K. Gupta Vipul, E. Stone, L. Morelli, Y. Lei and D. Melnick, Antimicrob. Agents Chemother., 2021, 65, e0073921 CrossRef.
- S. Li, X. Zhu, G. Cao, J. Shen, X. Zhu, J. Yu, X. Wu, J. Wu, H. Yang, N. Li, Y. Hu, J. Wang, H. Huang and J. Zhang, Antimicrob. Agents Chemother., 2024, 68, e0156323 CrossRef PubMed.
- B. Bruss Jon, J. Bader and A. Hamed Kamal, Antimicrob. Agents Chemother., 2023, 67, e00505 Search PubMed.
- A. J. Lepak, W. Wang and D. R. Andes, Antimicrob. Agents Chemother., 2020, 64, e01517 CAS.
- S. Wu, D. Yin, P. Zhi, Y. Guo, Y. Yang, D. Zhu and F. Hu, Front. Cell. Infect. Microbiol., 2022, 12, 829592 CrossRef CAS.
- X. Qu, C. Guo, S. Liu, X. Li, L. Xi, X. Liu and J. Zhang, Antibiotics, 2024, 13, 354 CrossRef CAS PubMed.
- M. F. Gordeev, J. Liu, X. Wang and Z. Yuan, US Pat., US9771394B1, 2019 Search PubMed.
- MicuRx Pharmaceuticals, MicuRx Pharmaceuticals completed the first subject dosing of MRX-8 (a new drug against drug-resistant bacteria) in China(Press Release 9 November 2022), https://www.micurx.com/1210.html, accessed 7 April 2025.
- K. D. Roberts, Y. Zhu, M. A. K. Azad, M.-L. Han, J. Wang, L. Wang, H. H. Yu, A. S. Horne, J.-A. Pinson, D. Rudd, N. H. Voelcker, N. A. Patil, J. Zhao, X. Jiang, J. Lu, K. Chen, O. Lomovskaya, S. J. Hecker, P. E. Thompson, R. L. Nation, M. N. Dudley, D. C. Griffith, T. Velkov and J. Li, Nat. Commun., 2022, 13, 1625 CrossRef CAS.
- Brii Biosciences, Brii Biosciences announces entry into definitive agreements from sale of its equity interest in Qpex Biopharma and acquiring exclusive global rights for BRII-693, https://www.briibio.com/en/media/press-release/202306260730/, accessed 7 April 2025.
- A. L. Cui, X.-X. Hu, Y. Gao, J. Jin, H. Yi, X.-K. Wang, T.-Y. Nie, Y. Chen, Q.-Y. He, H.-F. Guo, J.-D. Jiang, X.-F. You and Z.-R. Li, J. Med. Chem., 2018, 61, 1845 CrossRef CAS.
- C. Xie, P. Wang, H. Wu, X. Hu, T. Nie, X. Li, P. Pang, G. Li, Y. Lu, X. Yang, X. Wang, C. Li and X. You, Biomed. Pharmacother., 2023, 164, 114965 CrossRef CAS.
- Jiangsu Aosaikang Pharmaceutical Co, ASK0912, an innovative first-in-class drug under development by Aosaikang, has completed dosing of patients in the first dose group (Press Release 8 December 2022), https://www.ask-pharm.com/detail/925.html, accessed 16 January 2025.
- L. Li, X. Tan, T. Zhou, S. Chi, Y. Zhu, Q. Liu, Y. Chen and J. Zhang, J. Infect. Chemother., 2024, 30, 34 CrossRef CAS.
- M. A. Pfaller, L. Li, Q. Liu, J. Zhang, M. D. Huband, J. M. Lindley and R. E. Mendes, JAC-Antimicrob. Resist., 2021, 3, dlab177 CrossRef PubMed.
- M. D. Huband, J. D. Thompson, N. D. Gurung, Q. Liu, L. Li, J. Zhang, J. M. Streit and M. Castanheira, Antimicrob. Agents Chemother., 2022, 66, e01397 CrossRef CAS.
- L. C. Powell, J. K. Cullen, G. M. Boyle, T. De Ridder, P.-Y. Yap, W. Xue, C. J. Pierce, M. F. Pritchard, G. E. Menzies, M. Abdulkarim, J. Y. M. Adams, J. Stokniene, L. W. Francis, M. Gumbleton, J. Johns, K. E. Hill, A. V. Jones, P. G. Parsons, P. Reddell and D. W. Thomas, Sci. Transl. Med., 2022, 14, eabn3758 CrossRef CAS PubMed.
- G. Chianese, H. I. M. Amin, C. Maioli, P. Reddell, P. Parsons, J. Cullen, J. Johns, H. Handoko, G. Boyle, G. Appendino, O. Taglialatela-Scafati and S. Gaeta, J. Nat. Prod., 2022, 85, 1959 CrossRef CAS.
- R. W. Lamont, G. C. Conroy, P. Reddell and S. M. Ogbourne, BMC Plant Biol., 2016, 16, 57 CrossRef CAS.
- Roche, Product Development Portfolio Last update: 23rd of October 2024, https://www.roche.com/solutions/pipeline, accessed 7 April 2025.
- P. A. Smith, M. F. T. Koehler, H. S. Girgis, D. Yan, Y. Chen, Y. Chen, J. J. Crawford, M. R. Durk, R. I. Higuchi, J. Kang, J. Murray, P. Paraselli, S. Park, W. Phung, J. G. Quinn, T. C. Roberts, L. Rougé, J. B. Schwarz, E. Skippington, J. Wai, M. Xu, Z. Yu, H. Zhang, M.-W. Tan and C. E. Heise, Nature, 2018, 561, 189 CrossRef CAS.
- M. F. T. Koehler, Y.-C. Chen, Y. Chen, Y. Chen, J. J. Crawford, M. R. Durk, K. Garland, E. J. Hanan, R. I. Higuchi, H. Hu, C. Q. Ly, P. G. Paraselli, T. C. Roberts, J. B. Schwarz, P. A. Smith, Z. Yu and C. E. Heise, ACS Med. Chem. Lett., 2023, 14, 1524 CrossRef CAS PubMed.
- A. Höltzel, D. G. Schmid, G. J. Nicholson, S. Stevanovic, J. Schimana, K. Gebhardt, H.-P. Fiedler and G. Jung, J. Antibiot., 2002, 55, 571 CrossRef.
- J. Schimana, K. Gebhardt, A. Höltzel, D. G. Schmid, R. Süssmuth, J. Müller, R. Pukall and H.-P. Fiedler, J. Antibiot., 2002, 55, 565 CrossRef CAS.
- Y. X. Tan, D. S. Peters, S. I. Walsh, M. Holcomb, D. Santos-Martins, S. Forli and F. E. Romesberg, J. Nat. Prod., 2020, 83, 2112 CrossRef CAS.
- N. Szałaj, A. Benediktsdottir, D. Rusin, A. Karlén, S. L. Mowbray and A. Więckowska, Eur. J. Med. Chem., 2022, 238, 114490 CrossRef PubMed.
- M. Paetzel, J. J. Goodall, M. Kania, R. E. Dalbey and M. G. P. Page, J. Biol. Chem., 2004, 279, 30781 CrossRef CAS.
- Medical Countermeasures and CBRN MCM Dashboard, GDC-0829, https://medicalcountermeasures.gov/barda/cbrn/, accessed 7 April 2025.
- N. P. Taylor, Roche's pipeline rethink hits 20% of new molecules as cancer candidates join discard pile (24 April 2024), https://www.fiercebiotech.com/biotech/roches-pipeline-rethink-hits-20-new-molecules-cancer-candidates-join-discard-pile, accessed 7 April 2025.
- J. Krücken, A. Harder, P. Jeschke, L. Holden-Dye, V. O'Connor, C. Welz and G. von Samson-Himmelstjerna, Trends Parasitol., 2012, 28, 385 CrossRef PubMed.
- J. Krücken, L. Holden-Dye, J. Keiser, R. K. Prichard, S. Townson, B. L. Makepeace, M. P. Hübner, S. R. Hahnel, I. Scandale, A. Harder and D. Kulke, PLoS Pathog., 2021, 17, e1009682 CrossRef.
- F. Risch, A. Kazakov, S. Specht, K. Pfarr, P. U. Fischer, A. Hoerauf and M. P. Hübner, Trends Parasitol., 2024, 40, 829 CrossRef CAS PubMed.
- T. Karpstein, V. Pasche, C. Häberli, I. Scandale, A. Neodo and J. Keiser, Parasites Vectors, 2019, 12, 226 CrossRef.
- L. Taylor, A. A. Ahmada, M. S. Ali, S. M. Ali, J. Hattendorf, I. S. Mohammed and J. Keiser, Lancet, 2024, 404, 683 CrossRef CAS.
- S. Miyadoh, H. Kawasaki, K. Aoyagi, T. Yaguchi, T. Okada and J. Sugiyama, Jpn. J. Mycol., 2000, 41, 183 CAS.
- T. Sasaki, M. Takagi, T. Yaguchi, S. Miyadoh, T. Okada and M. Koyama, J. Antibiot., 1992, 45, 692 CrossRef CAS PubMed.
- M. Ohyama, Y. Okada, M. Takahashi, O. Sakanaka, M. Matsumoto and K. Atsumi, Biosci. Biotechnol. Biochem., 2011, 75, 1354 CrossRef CAS.
- A. A. Salim, M. S. Butler, M. A. T. Blaskovich, I. R. Henderson and R. J. Capon, Nat. Prod. Rep., 2023, 40, 1754 RSC.
- G. Geßner, S. Meder, T. Rink, G. Boheim, A. Harder, P. Jeschke, J. Scherkenbeck and M. Londershausen, Pestic. Sci., 1996, 48, 399 CrossRef.
- T. Raisch, A. Brockmann, U. Ebbinghaus-Kintscher, J. Freigang, O. Gutbrod, J. Kubicek, B. Maertens, O. Hofnagel and S. Raunser, Nat. Commun., 2021, 12, 7164 CrossRef CAS PubMed.
- B. Saeger, H.-P. Schmitt-Wrede, M. Dehnhardt, W. P. M. Benten, J. Krücken, A. Harder, G. von Samson-Himmelstjerna, H. Wiegand and F. Wunderlich, FASEB J., 2001, 15, 1332 CrossRef CAS PubMed.
- R. Dornetshuber, M. R. Kamyar, P. Rawnduzi, I. Baburin, K. Kouri, E. Pilz, T. Hornbogen, R. Zocher, W. Berger and R. Lemmens-Gruber, Biochem. Pharmacol., 2009, 77, 1437 CrossRef CAS.
- S. M. Miltsch, J. Krücken, J. Demeler, I. J. I. Janssen, N. Krüger, A. Harder and G. von Samson-Himmelstjerna, Int. J. Parasitol., 2012, 42, 761 CrossRef CAS.
- T. Mori, B. R. O'Keefe, R. C. Sowder, S. Bringans, R. Gardella, S. Berg, P. Cochran, J. A. Turpin, R. W. Buckheit, J. B. McMahon and M. R. Boyd, J. Biol. Chem., 2005, 280, 9345 CrossRef CAS.
- J. L. Fuqua, K. Hamorsky, G. Khalsa, N. Matoba and K. E. Palmer, Plant Biotechnol. J., 2015, 13, 1160 CrossRef PubMed.
- S. Lusvarghi and C. A. Bewley, Viruses, 2016, 8, 296 CrossRef.
- C. Lee, Mar. Drugs, 2019, 17, 567 CrossRef CAS.
- E. Cash, K. Deitz, K. L. Potts, H. W. Nabeta, M. Zahin, S. N. Rai, G. W. Dryden and K. E. Palmer, BMJ Open, 2023, 13, e073735 CrossRef PubMed.
- N. Teleshova, M. J. Keller, J. A. Fernández Romero, B. A. Friedland, G. W. Creasy, M. G. Plagianos, L. Ray, P. Barnable, L. Kizima, A. Rodriguez, N. Cornejal, C. Melo, G. Cruz Rodriguez, S. Mukhopadhyay, G. Calenda, S. U. Sinkar, T. Bonnaire, A. Wesenberg, S. Zhang, K. Kleinbeck, K. Palmer, M. Alami, B. R. O'Keefe, P. Gillevet, H. Hur, Y. Liang, G. Santone, R. N. Fichorova, T. Kalir and T. M. Zydowsky, PLoS One, 2022, 17, e0261775 CrossRef CAS PubMed.
- J. Chen, S. Ahn, J. Wang, Y. Lu, J. T. Dalton, D. D. Miller and W. Li, J. Med. Chem., 2012, 55, 7285 CrossRef CAS.
- K. G. Barnette, M. S. Gordon, D. Rodriguez, T. G. Bird, A. Skolnick, M. Schnaus, P. K. Skarda, S. Lobo, E. Sprinz, G. Arabadzhiev, P. Kalaydzhiev and M. Steiner, NEJM Evidence, 2022, 1, EVIDoa2200145 CrossRef.
- M. C. Markowski, R. Tutrone, C. Pieczonka, K. G. Barnette, R. H. Getzenberg, D. Rodriguez, M. S. Steiner, D. R. Saltzstein, M. A. Eisenberger and E. S. Antonarakis, Clin. Cancer Res., 2022, 28, 2789 CrossRef CAS.
- A.-T. Rüdiger, P. Mayrhofer, Y. Ma-Lauer, G. Pohlentz, J. Müthing, A. von Brunn and C. Schwegmann-Weßels, Virology, 2016, 497, 185 CrossRef.
- A. A. Mironov, M. A. Savin and G. V. Beznoussenko, Int. J. Mol. Sci., 2023, 24, 4523 CrossRef CAS PubMed.
- Z.-L. Guo, M.-X. Li, X.-L. Li, P. Wang, W.-G. Wang, W.-Z. Du, Z.-Q. Yang, S.-F. Chen, D. Wu and X.-Y. Tian, Front. Pharmacol., 2022, 12, 745683 CrossRef PubMed.
- R. Castillo, J.-A. Fernández and L. Gómez-Gómez, Plant Physiol., 2005, 139, 674 CrossRef CAS PubMed.
- A. Ji, J. Jia, Z. Xu, Y. Li, W. Bi, F. Ren, C. He, J. Liu, K. Hu and J. Song, Front. Plant Sci., 2017, 8, 00518 Search PubMed.
- J. H. Lee, S.-R. Lee, S. Y. Lee and P. C. Lee, Microb. Cell Fact., 2024, 23, 10 CrossRef CAS PubMed.
- P.-M. Mertes, O. Collange, P. Coliat, M. Banerjee, M.-C. Diringer, A. Roche, X. Delabranche, V. Chaban, M. Voegelin, A. Bernard, V. Sartori, N. Laurent, M. Velten, N. Dhindsa, J. Defuria, G. Kim, Z. H. Xu, M. Theodorou, Z. R. Huang, K. Khalifa, B. Geng, C. Niyikiza, V. Moyo, P. Gizzi, P. Villa, A. Detappe and X. Pivot, J. Controlled Release, 2021, 336, 252 CrossRef CAS.
- J. L. Gainer, Expert Opin. Invest. Drugs, 2008, 17, 917 CrossRef CAS PubMed.
- A. Streinu-Cercel, O. Săndulescu, V. D. Miron, A.-A. Oană, M. M. Motoi, C. D. Galloway and A. Streinu-Cercel, medRxiv, 2021, preprint, 2021.10.08.21264719, DOI:10.1101/2021.10.08.21264719.
- A. J. Lepak, B. VanScoy, C. Rubino, P. G. Ambrose and D. R. Andes, Antimicrob. Agents Chemother., 2024, 68, e01631 Search PubMed.
- Elion Therapeutics, Developing next-generation polyene therapy, https://eliontx.com/our-pipeline/, accessed 7 April 2025.
- B. C. Wilcock, M. M. Endo, B. E. Uno and M. D. Burke, J. Am. Chem. Soc., 2013, 135, 8488 CrossRef CAS.
- X. Guo, J. Zhang, X. Li, E. Xiao, J. D. Lange, C. M. Rienstra, M. D. Burke and D. A. Mitchell, ACS Cent. Sci., 2021, 7, 781 CrossRef CAS.
- A. Maji, C. P. Soutar, J. Zhang, A. Lewandowska, B. E. Uno, S. Yan, Y. Shelke, G. Murhade, E. Nimerovsky, C. G. Borcik, A. S. Arango, J. D. Lange, J. P. Marin-Toledo, Y. Lyu, K. L. Bailey, P. J. Roady, J. T. Holler, A. Khandelwal, A. M. SantaMaria, H. Sanchez, P. R. Juvvadi, G. Johns, M. J. Hageman, J. Krise, T. Gebremariam, E. G. Youssef, K. Bartizal, K. A. Marr, W. J. Steinbach, A. S. Ibrahim, T. F. Patterson, N. P. Wiederhold, D. R. Andes, T. V. Pogorelov, C. D. Schwieters, T. M. Fan, C. M. Rienstra and M. D. Burke, Nature, 2023, 623, 1079 CrossRef CAS PubMed.
- S. Chu, L. Long, S. McCormick Thomas, K. Borroto-Esoda, S. Barat and A. Ghannoum Mahmoud, Antimicrob. Agents Chemother., 2021, 65, e01989 CAS.
- S. Chu, L. Long, R. Sherif, S. McCormick Thomas, K. Borroto-Esoda, S. Barat and A. Ghannoum Mahmoud, Antimicrob. Agents Chemother., 2021, 65, e01988 CAS.
- J. M. Apgar, R. R. Wilkening, M. L. Greenlee, J. M. Balkovec, A. M. Flattery, G. K. Abruzzo, A. M. Galgoci, R. A. Giacobbe, C. J. Gill, M. J. Hsu, P. Liberator, A. S. Misura, M. Motyl, J. Nielsen Kahn, M. Powles, F. Racine, J. Dragovic, B. Habulihaz, W. Fan, R. Kirwan, S. Lee, H. Liu, A. Mamai, K. Nelson and M. Peel, Bioorg. Med. Chem. Lett., 2015, 25, 5813 CrossRef CAS.
- F. Peláez, A. Cabello, G. Platas, M. T. Díez, A. G. del Val, A. Basilio, I. Martán, F. Vicente, G. F. Bills, R. A. Giacobbe, R. E. Schwartz, J. C. Onishi, M. S. Meinz, G. K. Abruzzo, A. M. Flattery, L. Kong and M. B. Kurtz, Syst. Appl. Microbiol., 2000, 23, 333 CrossRef.
- SCYNEXIS, SCYNEXIS initiates dosing in phase 1 trial of SCY-247, a second-generation fungerp candidate for invasive fungal infections (Press Release 18 December 2024), https://ir.scynexis.com/news-events/press-releases/detail/342/scynexis-initiates-dosing-in-phase-1-trial-of-scy-247-a, accessed 7 April 2025.
- W. Hall, Psychol. Med., 2022, 52, 26 CrossRef.
- L. Vollner, D. Bieniek and F. Korte, Tetrahedron Lett., 1969, 10, 145 CrossRef.
- N. D. Sarma, A. Waye, M. A. ElSohly, P. N. Brown, S. Elzinga, H. E. Johnson, R. J. Marles, J. E. Melanson, E. Russo, L. Deyton, C. Hudalla, G. A. Vrdoljak, J. H. Wurzer, I. A. Khan, N.-C. Kim and G. I. Giancaspro, J. Nat. Prod., 2020, 83, 1334 CrossRef CAS.
- C. Russo, M. Lavorgna, R. Nugnes, E. Orlo and M. Isidori, Sci. Rep., 2021, 11, 22494 CrossRef CAS.
- P. B. Sampson, J. Nat. Prod., 2021, 84, 142 CrossRef CAS.
- M. J. Brodie, P. Czapinski, L. Pazdera, J. W. Sander, M. Toledo, M. Napoles, F. Sahebkar, A. Schreiber, T. Nezadal, J. Slonkova, A. Altman, K. Füle, Á. Horváth, A. Kelemen, S. Komoly, M. Banach, I. Kurkowska-Jastrzebska, P. Lisewski, M. Falip, R. Rocamora and M. Bagary, Cannabis Cannabinoid Res., 2021, 6, 528 CrossRef CAS.
- A. García-Martín, M. Garrido-Rodríguez, C. Navarrete, D. Caprioglio, B. Palomares, J. DeMesa, A. Rollland, G. Appendino and E. Muñoz, Biochem. Pharmacol., 2019, 163, 321 CrossRef.
- D. Caprioglio, D. Mattoteia, O. Taglialatela-Scafati, E. Muñoz and G. Appendino, Biomolecules, 2021, 11, 991 CrossRef CAS PubMed.
- B. P. Lavayen, C. Yang, J. Larochelle, L. Liu, R. J. Tishko, A. C. P. de Oliveira, E. Muñoz and E. Candelario-Jalil, Neurochem. Int., 2023, 165, 105508 CrossRef CAS.
- MindMed, Clinical Focus, https://mindmed.co/clinical-digital-trials/, accessed 7 April 2025.
- MindBio Therapeutics, Clinical Trials Pipeline, https://www.mindbiotherapeutics.com/clinical-trials, accessed 7 April 2025.
- S. T. Slocum, J. F. DiBerto and B. L. Roth, J. Neurochem., 2022, 162, 24 CrossRef CAS PubMed.
- A. Gomez-Escolar, D. Folch-Sanchez, J. Stefaniuk, Z. Swithenbank, A. Nisa, F. Braddick, N. Idrees Chaudhary, P. B. van der Meer and A. Batalla, CNS Drugs, 2024, 38, 771 CrossRef.
- J. M. Mitchell and B. T. Anderson, Neuropsychopharmacology, 2024, 49, 96 CrossRef PubMed.
- M. R. Lee, J. Roy. Coll. Phys. Edinb., 2009, 39, 179 CrossRef CAS.
- M. R. Lee, J. Roy. Coll. Phys. Edinb., 2009, 39, 365 CrossRef CAS.
- J.-J. Chen, M.-Y. Han, T. Gong, J.-L. Yang and P. Zhu, RSC Adv., 2017, 7, 27384 RSC.
- L. Ruzicka, Biogr. Mem. Fellows R. Soc., 1972, 18, 566 CrossRef.
- A. Hofmann, LSD, My Problem Child, McGraw-Hill, 1980 Search PubMed.
- A. Stoll and A. Hofmann, Helv. Chim. Acta, 1943, 26, 922 CrossRef CAS.
- M. R. Lee, J. Roy. Coll. Phys. Edinb., 2010, 40, 77 CrossRef CAS PubMed.
- N. Ohler, Tripped. Nazi Germany, the CIA, and the Dawn of the Psychedelic Age, Atlantic Books, London, UK, 2024 Search PubMed.
- M. Meyer and J. Slot, Fungal Genet. Biol., 2023, 167, 103812 CrossRef CAS PubMed.
- P. Sharma, Q. A. Nguyen, S. J. Matthews, E. Carpenter, D. B. Mathews, C. A. Patten and C. J. Hammond, J. Psychopharmacol., 2023, 37, 849 CrossRef PubMed.
- A. Hofmann, R. Heim, A. Brack and H. Kobel, Experientia, 1958, 14, 107 CrossRef CAS.
- H. Weidmann, M. Taeschler and H. Konzett, Experientia, 1958, 14, 378 CrossRef CAS PubMed.
- A. Hofmann, A. Frey, H. Ott, T. Petrzilka and F. Troxler, Experientia, 1958, 14, 397 CrossRef CAS PubMed.
- A. Hofmann, R. Heim, A. Brack, H. Kobel, A. Frey, H. Ott, T. Petrzilka and F. Troxler, Helv. Chim. Acta, 1959, 42, 1557 CrossRef CAS.
- J. Williamson, A. J. Spicer, E. Louramo and J. Jalkanen, Drug Discovery Today, 2024, 29, 103919 CrossRef PubMed.
- M. P. Bogenschutz, S. Ross, S. Bhatt, T. Baron, A. A. Forcehimes, E. Laska, S. E. Mennenga, K. O'Donnell, L. T. Owens, S. Podrebarac, J. Rotrosen, J. S. Tonigan and L. Worth, JAMA Psychiatry, 2022, 79, 953 CrossRef PubMed.
- Incannex Healthcare, FDA review of PsiGAD2 IND complete; clinical trial for psilocybin assisted psychotherapy in patients with generalised anxiety disorder to proceed (Press Release 5 August 2024), https://ir.incannex.com/news-releases/news-release-details/fda-review-psigad2-ind-complete-clinical-trial-psilocybin, accessed 7 April 2025.
- Cybin, CYB003: Deuterated psilocin program with FDA breakthrough therapy designation, https://cybin.com/cyb003/, accessed 7 April 2025.
- A. Nivorozhkin, M. Palfreyman, P. Pathare, K. L. Avery, M. Shukoor, J. H. Huang, M. E. Morgan and J. M. Krakowsky, World Pat., WO2022195011A1, 2022 Search PubMed.
- N. Bryson, R. Alexander, A. Asnis-Alibozek and M. D. Ehlers, ACS Chem. Neurosci., 2024, 15, 2386 CrossRef CAS PubMed.
- A. Shulgin and A. Shulgin, TiHKAL: The continuation, Transform Press, Berkeley, CA, 1997 Search PubMed.
- T. J. Kelly, E. M. Bonniwell, L. Mu, X. Liu, Y. Hu, V. Friedman, H. Yu, W. Su, J. D. McCorvy and Q.-s. Liu, Neuropsychopharmacology, 2024, 49, 854 CrossRef CAS PubMed.
- Psychedelic Alpha, Reunion sues Mindset, claims it copied RE104 (15 March 2023), https://psychedelicalpha.com/news/reunion-sues-mindset-claims-it-copied-re104, accessed 7 April 2025.
- Z. Hughes, A. Klein, D. Dvorak, E. Austin, L. Kiss, G. Marek, J. Sporn and A. Kruegel, Biol. Psychiatry, 2023, 93, S102 CrossRef.
- M. Peplow, Nat. Biotechnol., 2024, 42, 827 CrossRef CAS PubMed.
- A. C. Kruegel, World Pat., WO2022235927A1, 2022 Search PubMed.
- J. Ott, J. Psychoact. Drugs, 1999, 31, 171 CrossRef CAS PubMed.
- T. Lyttle, D. Goldstein and J. Gartz, J. Psychoact. Drugs, 1996, 28, 267 CrossRef CAS.
- M. V. Uthaug, R. Lancelotta, K. van Oorsouw, K. P. C. Kuypers, N. Mason, J. Rak, A. Šuláková, R. Jurok, M. Maryška, M. Kuchař, T. Páleníček, J. Riba and J. G. Ramaekers, Psychopharmacology, 2019, 236, 2653v2666 CrossRef PubMed.
- M. Layzell, P. Rands, M. Good, Z. Joel, R. Cousins, T. Benway, E. James and C. Routledge, ACS Med. Chem. Lett., 2023, 14, 1216 CrossRef CAS.
- E. James, D. Erritzoe, T. Benway, Z. Joel, C. Timmermann, M. Good, C. Agnorelli, B. M. Weiss, T. Barba, G. Campbell, M. Baker Jones, C. Hughes, H. Topping, M. Boyce and C. Routledge, Front. Psychiatry, 2024, 14, 1305796 CrossRef.
- M. Good, Z. Joel, T. Benway, C. Routledge, C. Timmermann, D. Erritzoe, R. Weaver, G. Allen, C. Hughes, H. Topping, A. Bowman and E. James, Eur. J. Drug Metab. Pharmacokinet., 2023, 48, 311 CrossRef CAS.
- F. Wagner, N. A. Powell, M. Chytil and D. E. Olson, US Pat., US20230219969A1, 2023 Search PubMed.
- C. Dong, C. Ly, L. E. Dunlap, M. V. Vargas, J. Sun, I.-W. Hwang, A. Azinfar, W. C. Oh, W. C. Wetsel, D. E. Olson and L. Tian, Cell, 2021, 184, 2779 CrossRef CAS.
- Delix Therapeutcis, Delix announces DLX-001 demonstrates evidence of CNS penetration and brain activity without psychotomimetic, dissociative, or hallucinogenic effects in ongoing phase 1 trial, Press Release 13 May 2024, https://www.delixtherapeutics.com/news/delix-announces-dlx-001-demonstrates-evidence-of-c/, accessed 7 April 2025.
- H. Copes, A. Hochstetler, J. Ragland and P. S. Hendricks, J. Drug Issues, 2024 DOI:10.1177/00220426241274737 , in press.
- B. K. Cassels and P. Sáez-Briones, ACS Chem. Neurosci., 2018, 9, 2448 CrossRef CAS.
- W. Duan, D. Cao, S. Wang and J. Cheng, Chem. Rev., 2024, 124, 124 CrossRef CAS.
- D. E. Nichols, in Behavioral Neurobiology of Psychedelic Drugs, ed. A. L. Halberstadt, F. X. Vollenweider and D. E. Nichols, Springer Berlin Heidelberg, Berlin, Heidelberg, 2018, p. 1, DOI:10.1007/7854_2017_475.
- J. M. Mitchell, M. Ot'alora G, B. van der Kolk, S. Shannon, M. Bogenschutz, Y. Gelfand, C. Paleos, C. R. Nicholas, S. Quevedo, B. Balliett, S. Hamilton, M. Mithoefer, S. Kleiman, K. Parker-Guilbert, K. Tzarfaty, C. Harrison, A. de Boer, R. Doblin, B. Yazar-Klosinski and Mapp Study Collaborator Group, Nat. Med., 2023, 29, 2473 CrossRef CAS.
- Lykos Therapeutics, Lykos Therapeutics Announces Complete Response Letter for Midomafetamine Capsules for PTSD (Press Release 9 August 2024), https://news.lykospbc.com/2024-08-09-Lykos-Therapeutics-Announces-Complete-Response-Letter-for-Midomafetamine-Capsules-for-PTSD, accessed 7 April 2025.
- B. Y. Fong, FDA rejects MDMA-assisted therapy for PTSD treatment – a drug researcher explains the challenges psychedelics face (The Conversation, 12 August 2024), https://theconversation.com/fda-rejects-mdma-assisted-therapy-for-ptsd-treatment-a-drug-researcher-explains-the-challenges-psychedelics-face-236383, accessed 7 April 2025.
- Australian Therapeutic Goods Administration, MDMA and psilocybine hub, https://www.tga.gov.au/products/unapproved-therapeutic-goods/mdma-and-psilocybine-hub, accessed 11 February 2025.
- K. D. Lewis, G. A. Pullella, H. C. Loh, B. W. Skelton, G. R. Flematti and M. J. Piggott, Aust. J. Chem., 2023, 76, 299 CrossRef CAS.
- R. W. Freudenmann, F. Öxler and S. Bernschneider-Reif, Addiction, 2006, 101, 1241 CrossRef.
- A. Shulgin and A. Shulgin, PiHKAL: A chemical love story, Transform Press, Berkeley, CA, 1991 Search PubMed.
- N. Nagai, Pharm. Ztg., 1887, 32, 700 Search PubMed.
- G. Appendino, A. Minassi and O. Taglialatela-Scafati, Nat. Prod. Rep., 2014, 31, 880 RSC.
- N. Rasmussen and J. Hist, Med. Allied Sci., 2006, 61, 288 CrossRef.
- A. Ogata, Yakugaku Zasshi, 1919, 451, 751 CAS.
- M. Morelli and E. Tognotti, Exp. Neurol., 2021, 342, 113754 CrossRef CAS PubMed.
- N. Rasmussen, On speed: The many lives of amphetamine, NYU Press, 2008 Search PubMed.
- P. de Haen, Pharmacy Times, 1976, 40 Search PubMed.
- B. Pires, L. M. Rosendo, A. T. Brinca, A. Y. Simão, M. Barroso, T. Rosado and E. Gallardo, Life, 2023, 13, 2180 CrossRef CAS.
- R. N. Iyer, D. Favela, G. Zhang and D. E. Olson, Nat. Prod. Rep., 2021, 38, 307 RSC.
- K. R. Alper, in The Alkaloids: Chemistry and Biology, Academic Press, 2001, vol. 56, ch. 1, p. 1 Search PubMed.
- C. Lavaud and G. Massiot, in Progress in the Chemistry of Organic Natural Products 105, ed. A. D. Kinghorn, H. Falk, S. Gibbons and J. i. Kobayashi, Springer International Publishing, Cham, Switzerland, 2017, p. 89, DOI:10.1007/978-3-319-49712-9_2.
- M. J. Wasko, P. A. Witt-Enderby and C. K. Surratt, ACS Chem. Neurosci., 2018, 9, 2475 CrossRef CAS.
- M. F. Bartlett, D. F. Dickel and W. I. Taylor, J. Am. Chem. Soc., 1958, 80, 126 CrossRef CAS.
- G. Arai, J. Coppola and G. A. Jeffrey, Acta Crystallogr., 1960, 13, 553 CrossRef CAS.
- T. Knuijver, A. Schellekens, M. Belgers, R. Donders, T. van Oosteren, K. Kramers and R. Verkes, Addiction, 2022, 117, 118 CrossRef PubMed.
- D. C. Mash, C. A. Kovera, J. Pablo, R. F. Tyndale, F. D. Ervin, I. C. Williams, E. G. Singleton and M. Mayor, Ann. N. Y. Acad. Sci., 2000, 914, 394 CrossRef CAS PubMed.
- ATAI Life Sciences, ATAI Life Sciences N.V. 2023 Annual Reporthttps://ir.atai.life/static-files/46fe4570-d7ba-490c-b062-70e83afc45f7, accessed 7 April 2025.
- K. N. Cherian, J. N. Keynan, L. Anker, A. Faerman, R. E. Brown, A. Shamma, O. Keynan, J. P. Coetzee, J.-M. Batail, A. Phillips, N. J. Bassano, G. L. Sahlem, J. Inzunza, T. Millar, J. Dickinson, C. E. Rolle, J. Keller, M. Adamson, I. H. Kratter and N. R. Williams, Nat. Med., 2024, 30, 373 CrossRef CAS PubMed.
- L. Cardia, G. Calapai, D. Quattrone, C. Mondello, V. Arcoraci, F. Calapai, C. Mannucci and E. Mondello, Front. Pharmacol., 2018, 9, 1122 CrossRef CAS.
- Elysium Therapeutics, Elysium Therapeutics announces compelling human proof-of-concept data for its SMART™ opioid, O2P™ hydrocodone prodrug for acute pain that could disrupt the industry by establishing new standards for opioid safety, Press Release 5 March 2024, https://www.elysiumrx.com/press, accessed 7 April 2025.
- C. Y. Kao, Pharmacol. Rev., 1966, 18, 997 CrossRef CAS.
- G. M. Bucciarelli, M. Lechner, A. Fontes, L. B. Kats, H. L. Eisthen and H. B. Shaffer, Toxins, 2021, 13, 517 CrossRef CAS.
- Y. Zhang, S. Zou, S. Yin and T. Wang, Toxin Rev., 2023, 42, 727 CrossRef CAS.
- K. Iwakawa and S. Kimura, Arch. Exp. Path. Pharm., 1922, 93, 305 CrossRef CAS.
- M. Á. Huerta, J. de la Nava, A. Artacho-Cordón and F. R. Nieto, Mar. Drugs, 2023, 21, 316 CrossRef CAS.
- G. R. Pettit, C. L. Herald, D. L. Doubek, D. L. Herald, E. Arnold and J. Clardy, J. Am. Chem. Soc., 1982, 104, 6846 CrossRef CAS.
- G. R. Pettit, D. L. Herald, F. Gao, D. Sengupta and C. L. Herald, J. Org. Chem., 1991, 56, 1337 CrossRef CAS.
- A. E. Trindade-Silva, G. E. Lim-Fong, K. H. Sharp and M. G. Haygood, Curr. Opin. Biotechnol., 2010, 21, 834 CrossRef CAS PubMed.
- J. Miller Ian, N. Vanee, S. Fong Stephen, E. Lim-Fong Grace and C. Kwan Jason, Appl. Environ. Microbiol., 2016, 82, 6573 CrossRef CAS.
- D. K. Armstrong, J. A. Blessing, K. Y. Look, R. Schilder and E. R. Nunez, Invest. New Drugs, 2003, 21, 373 CrossRef CAS PubMed.
- P. Kollár, J. Rajchard, Z. Balounová and J. Pazourek, Pharm. Biol., 2014, 52, 237 CrossRef PubMed.
- R. Wu, H. Chen, N. Chang, Y. Xu, J. Jiao and H. Zhang, Chem.–Eur. J., 2020, 26, 1166 CrossRef CAS PubMed.
- Z. Tian, X.-T. Lu, X. Jiang and J. Tian, Front. Pharmacol., 2023, 14, 1187411 CrossRef CAS.
- D. L. Alkon, M.-K. Sun, A. J. Tuchman and R. E. Thompson, J. Alzheimer's Dis., 2023, 96, 759 CAS.
- R. E. Thompson, A. J. Tuchman and D. L. Alkon, J. Alzheimer's Dis., 2022, 86, 1221 CAS.
- P. R. Newswrire, Neurotrope announces strategic partnership with BryoLogyx for supply of synthetic bryostatin-1 and continuation of the National Cancer Institute (NCI) trial for childhood leukemia (Press Release 10 June 2020), https://www.prnewswire.com/news-releases/neurotrope-announces-strategic-partnership-with-bryologyx-for-supply-of-synthetic-bryostatin-1-and-continuation-of-the-national-cancer-institute-nci-trial-for-childhood-leukemia-301073668.html, accessed 7 April 2025.
- P. A. Wender, C. T. Hardman, S. Ho, M. S. Jeffreys, J. K. Maclaren, R. V. Quiroz, S. M. Ryckbosch, A. J. Shimizu, J. L. Sloane and M. C. Stevens, Science, 2017, 358, 218 CrossRef CAS.
- U. Chatterji, M. Bobardt, S. Selvarajah, F. Yang, H. Tang, N. Sakamoto, G. Vuagniaux, T. Parkinson and P. Gallay, J. Biol. Chem., 2009, 284, 16998 CrossRef CAS.
- S. Hopkins, B. Scorneaux, Z. Huang, M. G. Murray, S. Wring, C. Smitley, R. Harris, F. Erdmann, G. Fischer and Y. Ribeill, Antimicrob. Agents Chemother., 2010, 54, 660 CrossRef CAS.
- S. Hopkins, M. Bobardt, U. Chatterji, J. A. Garcia-Rivera, P. Lim and P. A. Gallay, Antimicrob. Agents Chemother., 2012, 56, 3888 CrossRef CAS.
- S. Hopkins, B. DiMassimo, P. Rusnak, D. Heuman, J. Lalezari, A. Sluder, B. Scorneaux, S. Mosier, P. Kowalczyk, Y. Ribeill, J. Baugh and P. Gallay, J. Hepatol., 2012, 57, 47 CrossRef CAS PubMed.
- SCYNEXIS, SCYNEXIS, Inc. enters into worldwide agreement with Waterstone Pharmaceutical for the development and commercialization of SCY-635 for viral diseases (Press Release 3 November 2014), https://ir.scynexis.com/news-events/press-releases/detail/34/scynexis-inc-enters-into-worldwide-agreement-with, accessed 7 April 2025.
- J. Lu, F. Liang, P. Bai, C. Liu, M. Xu, Z. Sun, W. Tian, Y. Dong, Y. Zhang, Q. Quan, A. Khatri, Y. Shen, E. Marcantonio, G. Crosby, D. J. Culley, C. Wang, G. Yang and Z. Xie, Alzheimer's Dement., 2023, 19, 4110 CrossRef CAS PubMed.
- A. Brossi and Q.-s. Yu, Heterocycles, 1988, 27, 745 CrossRef.
- Q.-s. Yu, X.-F. Pei, H. W. Holloway, N. H. Greig and A. Brossi, J. Med. Chem., 1997, 40, 2895 CrossRef CAS.
- J. Klein, Expert Opin. Invest. Drugs, 2007, 16, 1087 CrossRef CAS PubMed.
- A. Proudfoot, Toxicol. Rev., 2006, 25, 99 CrossRef CAS PubMed.
- A. M. Arens and T. Kearney, J. Med. Toxicol., 2019, 15, 184 CrossRef CAS PubMed.
- C. Fang, P. Hernandez, K. Liow, E. Damiano, H. Zetterberg, K. Blennow, D. Feng, M. Chen and M. Maccecchini, J. Prev. Alzheimers Dis., 2023, 10, 25 CrossRef CAS.
- M. L. Maccecchini, M. Y. Chang, C. Pan, V. John, H. Zetterberg and N. H. Greig, J. Neurol. Neurosurg. Psychiat., 2012, 83, 894 CrossRef.
- H.-H. Cho, C. M. Cahill, C. R. Vanderburg, C. R. Scherzer, B. Wang, X. Huang and J. T. Rogers, J. Biol. Chem., 2010, 285, 31217 CrossRef CAS PubMed.
- J. T. Rogers, S. Mikkilineni, I. Cantuti-Castelvetri, D. H. Smith, X. Huang, S. Bandyopadhyay, C. M. Cahill, M. L. Maccecchini, D. K. Lahiri and N. H. Greig, J. Neural Transm., 2011, 118, 493 CrossRef CAS.
- X.-Q. Chen, C. A. Barrero, R. Vasquez-Del Carpio, E. P. Reddy, C. Fecchio, S. Merali, A. Deglincerti, C. Fang, J. Rogers and M. L. Maccecchini, Pharmaceutics, 2021, 13, 2109 CrossRef CAS.
- S. Batool, T. Furqan, M. S. Hasan Mahmood, D. Tweedie, M. A. Kamal and N. H. Greig, ACS Pharmacol. Transl. Sci., 2022, 5, 70 CrossRef CAS PubMed.
- E. Deau, M. F. Lindberg, F. Miege, D. Roche, N. George, P. George, A. Krämer, S. Knapp and L. Meijer, J. Med. Chem., 2023, 66, 10694 CrossRef CAS.
- N. Loaëc, E. Attanasio, B. Villiers, E. Durieu, T. Tahtouh, M. Cam, R. A. Davis, A. Alencar, M. Roué, M.-L. Bourguet-Kondracki, P. Proksch, E. Limanton, S. Guiheneuf, F. Carreaux, J.-P. Bazureau, M. Klautau and L. Meijer, Mar. Drugs, 2017, 15, 316 CrossRef PubMed.
- L. Meijer, E. Chrétien, D. Ravel, P. I. Moreira, J. Avila, D. Galimberti, M. A. Pappolla, G. Plascencia-Villa, A. A. Sorensen, X. Zhu and G. Perry, J. Alzheimer's Dis., 2024, 101, S95 CAS.
- G. W. Chan, S. Mong, M. E. Hemling, A. J. Freyer, P. H. Offen, C. W. DeBrosse, H. M. Sarau and J. W. Westley, J. Nat. Prod., 1993, 56, 116 CrossRef CAS.
- M. F. Lindberg, E. Deau, F. Miege, M. Greverie, D. Roche, N. George, P. George, L. Merlet, J. Gavard, S. J. T. Brugman, E. Aret, P. Tinnemans, R. de Gelder, J. Sadownik, E. Verhofstad, D. Sleegers, S. Santangelo, J. Dairou, Á. Fernandez-Blanco, M. Dierssen, A. Krämer, S. Knapp and L. Meijer, J. Med. Chem., 2023, 66, 15648 CrossRef CAS PubMed.
- M. Farhan and A. Rizvi, Nutrients, 2023, 15, 4486 CrossRef CAS PubMed.
- K. Brown, D. Theofanous, R. G. Britton, G. Aburido, C. Pepper, S. Sri Undru and L. Howells, Int. J. Mol. Sci., 2024, 25, 747 CrossRef PubMed.
- C. Kemper, D. Behnam, S. Brothers, C. Wahlestedt, C.-H. Volmar, D. Bennett and M. Hayward, AAPS Open, 2022, 8, 11 CrossRef PubMed.
- Jupiter Neurosciences, Pipeline, https://jupiterneurosciences.com/pipeline/, accessed 10 September 2024.
- L. Lian-niang, T. Rui and C. Wei-ming, Planta Med., 1984, 50, 227 CrossRef CAS.
- L. Cai, Y. Chen, H. Xue, Y. Yang, Y. Wang, J. Xu, C. Zhu, L. He and Y. Xiao, J. Ethnopharmacol., 2024, 319, 117354 CrossRef CAS PubMed.
- H. Yang, M. M. Ibrahim, S. Zhang, Y. Sun, J. Chang, H. Qi and S. Yang, Front. Immunol., 2024, 15, 1433590 CrossRef CAS PubMed.
- K. Hasumi and E. Suzuki, Int. J. Mol. Sci., 2021, 22, 954 CrossRef CAS PubMed.
- C. Shinohara, K. Hasumi, W. Hatsumi and A. Endo, J. Antibiot., 1996, 49, 961 CrossRef CAS PubMed.
- W. Hu, S. Ohyama and K. Hasumi, J. Antibiot., 2000, 53, 241 CrossRef CAS PubMed.
- W. Hu, R. Narasaki, S. Ohyama and K. Hasumi, J. Antibiot., 2001, 54, 962 CrossRef CAS PubMed.
- R. Iwama, Y. Sasano, T. Hiramatsu, S. Otake, E. Suzuki and K. Hasumi, J. Fungi, 2022, 8, 975 CrossRef CAS.
- B. Wang and Y. Lin, Chem. Commun., 2022, 58, 13071 RSC.
- D.-I. A. Kwok, R. Huang, D. Shang, Y. Xue and D. G. Walker, Org. Lett., 2024, 26, 697 CrossRef CAS.
- T. Moritoyo, N. Nishimura, K. Hasegawa, S. Ishii, K. Kirihara, M. Takata, A. K. Svensson, Y. Umeda-Kameyama, S. Kawarasaki, R. Ihara, C. Sakanaka, Y. Wakabayashi, K. Niizuma, T. Tominaga, T. Yamazaki and K. Hasumi, Br. J. Clin. Pharmacol., 2023, 89, 1809 CrossRef CAS.
- Ji Xing, Pharmaceuticals, JIXING acquires BIIB131 from Biogen to treat acute ischemic stroke (Press Release 11 January 2024), https://www.corxelbio.com/en/press-releases/jixing-acquires-biib131-from-biogen-to-treat-acute-ischemic-stroke/, accessed 7 April 2025.
- Ji Xing, Pharmaceuticals, TMS and JIXING announce a series of strategic collaborations (press Release 11 January 2024), https://www.corxelbio.com/en/press-releases/tms-and-jixing-announce-a-series-of-strategic-collaborations/, accessed 7 April 2025.
- TMS, Notice of dosing to the first subject in TMS-008 phase I clinical trial (Press Release 19 June 2024), https://www.tms-japan.co.jp/en/ir/news/auto_20240619531866/pdfFile.pdf, accessed 7 April 2025.
- TMS, Technology, https://www.tms-japan.co.jp/en/business/r-and-d/technology.html, accessed 7 April 2025.
- K. Motoki, E. Kobayashi, T. Uchida, H. Fukushima and Y. Koezuka, Bioorg. Med. Chem. Lett., 1995, 5, 705 CrossRef CAS.
- M. Morita, K. Motoki, K. Akimoto, T. Natori, T. Sakai, E. Sawa, K. Yamaji, Y. Koezuka, E. Kobayashi and H. Fukushima, J. Med. Chem., 1995, 38, 2176 CrossRef CAS.
- T. Natori, Y. Koezuka and T. Higa, Tetrahedron Lett., 1993, 34, 5591 CrossRef CAS.
- T. Natori, M. Morita, K. Akimoto and Y. Koezuka, Tetrahedron, 1994, 50, 2771 CrossRef CAS.
- X. Laurent, B. Bertin, N. Renault, A. Farce, S. Speca, O. Milhomme, R. Millet, P. Desreumaux, E. Hénon and P. Chavatte, J. Med. Chem., 2014, 57, 5489 CrossRef CAS PubMed.
- C. Romanò and M. H. Clausen, Eur. J. Org Chem., 2022, 2022, e202200246 CrossRef.
- F. L. Schneiders, R. J. Scheper, B. M. E. von Blomberg, A. M. Woltman, H. L. A. Janssen, A. J. M. van den Eertwegh, H. M. W. Verheul, T. D. de Gruijl and H. J. van der Vliet, Clin. Immunol., 2011, 140, 130 CrossRef CAS PubMed.
- D. Y.-B. Chen, S. Farhan, L. J. Lekakis, G. J. Schiller, J. A. Yared, M. Y. Mapara, A. Assal, H. Choe, Z. DeFilipp, D. D. Lee, H. Lane, L. J. Burns, M.-J. Zhang, M. Bye, T. A. Gooley and A. Saad, Transplant. Cell. Ther., 2024, 30, S29 CrossRef.
- T. H. Kim, H. H. Jiang, S. M. Lim, Y. S. Youn, K. Y. Choi, S. Lee, X. Chen, Y. Byun and K. C. Lee, Bioconjugate Chem., 2012, 23, 2214 CrossRef CAS.
- S. P. Yun, T.-I. Kam, N. Panicker, S. Kim, Y. Oh, J.-S. Park, S.-H. Kwon, Y. J. Park, S. S. Karuppagounder, H. Park, S. Kim, N. Oh, N. A. Kim, S. Lee, S. Brahmachari, X. Mao, J. H. Lee, M. Kumar, D. An, S.-U. Kang, Y. Lee, K. C. Lee, D. H. Na, D. Kim, S. H. Lee, V. V. Roschke, S. A. Liddelow, Z. Mari, B. A. Barres, V. L. Dawson, S. Lee, T. M. Dawson and H. S. Ko, Nat. Med., 2018, 24, 931 CrossRef CAS PubMed.
- J.-S. Park, T.-I. Kam, S. Lee, H. Park, Y. Oh, S.-H. Kwon, J.-J. Song, D. Kim, H. Kim, A. Jhaldiyal, D. H. Na, K. C. Lee, E. J. Park, M. G. Pomper, O. Pletnikova, J. C. Troncoso, H. S. Ko, V. L. Dawson, T. M. Dawson and S. Lee, Acta Neuropathol. Commun., 2021, 9, 78 CrossRef CAS PubMed.
- M. Gharagozloo, M. D. Smith, E. S. Sotirchos, J. Jin, K. Meyers, M. Taylor, T. Garton, R. Bannon, H.-N. Lord, T. M. Dawson, V. L. Dawson, S. Lee and P. A. Calabresi, Neurotherapeutics, 2021, 18, 1834 CrossRef CAS PubMed.
- A. McGarry, S. Rosanbalm, M. Leinonen, C. W. Olanow, D. To, A. Bell, D. Lee, J. Chang, J. Dubow, R. Dhall, D. Burdick, S. Parashos, J. Feuerstein, J. Quinn, R. Pahwa, M. Afshari, A. Ramirez-Zamora, K. Chou, A. Tarakad, C. Luca, K. Klos, Y. Bordelon, M.-H. St Hiliare, D. Shprecher, S. Lee, T. M. Dawson, V. Roschke and K. Kieburtz, Lancet Neurol., 2024, 23, 37 CrossRef CAS.
- T. Müller and J. D. Möhr, Lancet Neurol., 2024, 23, 558 CrossRef.
- A. McGarry, Lancet Neurol., 2024, 23, 559 CrossRef CAS PubMed.
- M. Anyika, M. McMullen, L. K. Forsberg, R. T. Dobrowsky and B. S. J. Blagg, ACS Med. Chem. Lett., 2016, 7, 67 CrossRef CAS.
- J. Ma, P. Pan, M. Anyika, B. S. J. Blagg and R. T. Dobrowsky, ACS Chem. Neurosci., 2015, 6, 1637 CrossRef CAS PubMed.
- Y. A. Rodriguez, S. Kaur, E. Nolte, Z. Zheng, B. S. J. Blagg and R. T. Dobrowsky, ACS Chem. Neurosci., 2021, 12, 3049 CrossRef CAS PubMed.
- S. Ghosh, Y. Liu, G. Garg, M. Anyika, N. T. McPherson, J. Ma, R. T. Dobrowsky and B. S. J. Blagg, ACS Med. Chem. Lett., 2016, 7, 813 CrossRef CAS.
- R. M. Lang, R. Chawla, S. Patel, C. K. Abrams and R. T. Dobrowsky, ACS Pharmacol. Transl. Sci., 2025, 8, 124 CrossRef CAS PubMed.
- M. Hergenhahn, W. Adolf and E. Hecker, Tetrahedron Lett., 1975, 16, 1595 Search PubMed.
- F. J. Evans and R. J. Schmidt, Phytochemistry, 1976, 15, 333 CrossRef CAS.
- W. Adolf, B. Sorg, M. Hergenhahn and E. Hecker, J. Nat. Prod., 1982, 45, 347 CrossRef CAS.
- D. C. Brown, Pharmaceuticals, 2016, 9, 47 CrossRef PubMed.
- T. Knotts, K. Mease, L. Sangameswaran, M. Felx, S. Kramer and J. Donovan, J. Orthop. Res., 2022, 40, 2281 CrossRef CAS.
- A.-P. Koivisto, M. G. Belvisi, R. Gaudet and A. Szallasi, Nat. Rev. Drug Discovery, 2022, 21, 41 CrossRef CAS PubMed.
- S. L. Shafer, S. L. Teichman, I. J. Gottlieb, N. Singla, H. S. Minkowitz, D. Leiman, B. Vaughn and J. F. Donovan, Anesthesiology, 2024, 141, 250 CrossRef CAS PubMed.
- Concentric Analgesics, Concentric Analgesics announces data highlighting two weeks of durable pain relief and earlier cessation of opioids in two surgical procedures (Press Release 6 August 2024), https://www.concentricanalgesics.com/announcing-data-highlighting-two-weeks-of-durable-pain-relief-and-earlier-cessation-of-opioids, accessed 7 April 2025.
- A. Endo, Proc. Jpn. Acad., Ser. B, 2010, 86, 484 CrossRef CAS.
- J. A. Tobert, Nat. Rev. Drug Discovery, 2003, 2, 517 CrossRef CAS PubMed.
- M. K. K. Yap and N. Misuan, Basic Clin. Pharmacol. Toxicol., 2019, 124, 513 CrossRef CAS PubMed.
- J. R. White Jr, Clin. Diabetes, 2010, 28, 5 CrossRef.
- Ö. Helvaci and B. Helvaci, Exp. Clin. Transplant., 2023, 21, 105 CrossRef PubMed.
- K. G. McLure, E. M. Gesner, L. Tsujikawa, O. A. Kharenko, S. Attwell, E. Campeau, S. Wasiak, A. Stein, A. White, E. Fontano, R. K. Suto, N. C. W. Wong, G. S. Wagner, H. C. Hansen and P. R. Young, PLoS One, 2014, 8, e83190 CrossRef.
- S. Picaud, C. Wells, I. Felletar, D. Brotherton, S. Martin, P. Savitsky, B. Diez-Dacal, M. Philpott, C. Bountra, H. Lingard, O. Fedorov, S. Müller, P. E. Brennan, S. Knapp and P. Filippakopoulos, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 19754 CrossRef CAS.
- H. Dhulkifle, M. I. Diab, M. Algonaiah, H. M. Korashy and Z. H. Maayah, ACS Pharmacol. Transl. Sci., 2024, 7, 546 CrossRef CAS PubMed.
- K. K. Ray, S. J. Nicholls, K. A. Buhr, H. N. Ginsberg, J. O. Johansson, K. Kalantar-Zadeh, E. Kulikowski, P. P. Toth, N. Wong, M. Sweeney, G. G. Schwartz and B. Investigators and Committees, JAMA, 2020, 323, 1565 CrossRef CAS PubMed.
- H. C. Hansen, US Pat., US8053440B1, 2011 Search PubMed.
- J. H. Miller, J. M. Mullin, E. McAvoy and A. Kleinzeller, Biochim. Biophys. Acta, Biomembr., 1992, 1110, 209 CrossRef CAS.
- V. Jörgens, Acta Diabetol., 2019, 56, 29 CrossRef PubMed.
- J. R. Ehrenkranz, N. G. Lewis, C. R. Kahn and J. Roth, Diabetes Metab. Res. Rev., 2005, 21, 31 CrossRef CAS PubMed.
- D.-Q. Chen, Z. Gu, Y.-S. Wu, W.-H. Yuan and Z. Li, CrystEngComm, 2021, 23, 8033 RSC.
- Y. Wang, Y. Lou, J. Wang, D. Li, H. Chen, T. Zheng, C. Xia, X. Song, T. Dong, J. Li, J. Li and H. Liu, Eur. J. Med. Chem., 2019, 180, 398 CrossRef CAS PubMed.
- G. Chen, T. Liu and X. Ji, China Pat., CN113429379A, 2021 Search PubMed.
- T. Inoue, M. Takemura, N. Fushimi, Y. Fujimori, T. Onozato, T. Kurooka, T. Asari, H. Takeda, M. Kobayashi, H. Nishibe and M. Isaji, Eur. J. Pharmacol., 2017, 806, 25 CrossRef CAS PubMed.
- Y. Niu, W. Cui, R. Liu, S. Wang, H. Ke, X. Lei and L. Chen, Nat. Commun., 2022, 13, 6440 CrossRef CAS PubMed.
- S. Fukudo, Y. Endo, M. Hongo, A. Nakajima, T. Abe, H. Kobayashi, T. Nakata, T. Nakajima, K. Sameshima, K. Kaku, E. Shoji, K. Tarumi, Y. Nagaoka, T. Ooshima, K. Ozawa, T. Majima, S. Kamata, T. Tada, H. Ishii, Y. Segawa, S. Miyazaki, T. Yamamoto, Y. Yagi, H. Sawada, S. Shirota, S. Otsuka, N. Yamada, R. Suzuki, H. Kurakata, K. Nakai, Y. Syuji, T. Usui, M. Yamamura, T. Oishi and H. Tanaka, Lancet Gastroenterol. Hepatol., 2018, 3, 603 CrossRef.
- Vogenx, Vogenx announces Series A financing and exclusive license agreement (Press Release 1 February 2022), https://vogenx.com/vogenx-inc-announces-initiation-of-phase-2-clinical-study-of-mizagliflozin-for-post-bariatric-hypoglycemia-copy/, accessed 7 April 2025.
- H. Margaret Lawler, T. L. McLaughlin, S. Shakeri, E. Frederick Stortz, A. Gupta, V. Singh, N. Turk, S. Walker, B. Cheatham and W. Wilkison, J. Endocr. Soc., 2023, 7, bvad114.829 CrossRef.
- Vogenx, Vogenx announces positive results from second phase 2 study of mizagliflozin in post-bariatric hypoglycemia (Press Release 26 June 2024), https://vogenx.com/vogenx-announces-positive-results-in-second-phase-2-study-of-mizagliflozin-in-post-bariatric-hypoglycemia/, accessed 7 April 2025.
- L. Huang, B. Cao, Y. Geng, X. Zhou, Y. Yang, T. Ma, H. Lin, Z. Huang, L. Zhuo and J. Li, Eur. J. Pharm. Sci., 2024, 192, 106644 CrossRef CAS PubMed.
- C. Li, T. Li, F. Lee, R. H. Xu and H. Wang, Diabetes, 2020, 69, 138 CrossRef.
- Y. Wang, C. Liu and H. Wang, China Pat., CN113004349A, 2021 Search PubMed.
- P. Lapuerta, S. Urbina, J. He, A. Wittle, C. Li, T. Li, H. Wang and M. Hompesch, Clin. Pharm. Therap., 2024, 115, 1383 CrossRef CAS PubMed.
- A. M. Di Bisceglie, G. F. Watts, P. Lavin, M. Yu, R. Bai and L. Liu, Lipids Health Dis, 2020, 19, 239 CrossRef CAS PubMed.
- K. V. Kowdley, L. Forman, B. Eksteen, N. Gunn, V. Sundaram, C. Landis, S. A. Harrison, C. Levy, A. Liberman, A. M. Di Bisceglie and G. M. Hirschfield, Am. J. Gastroenterol., 2022, 117, 1805 CrossRef CAS PubMed.
- S. A. Harrison, N. Gunn, G. W. Neff, A. Kohli, L. Liu, A. Flyer, L. Goldkind and A. M. Di Bisceglie, Nat. Commun., 2021, 12, 5503 CrossRef CAS.
- Z. Qin, R. Tang, J. Liang and X. Jia, Int. Immunopharmacol., 2024, 137, 112422 CrossRef CAS.
- X.-F. Luo, H. Zhou, P. Deng, S.-Y. Zhang, Y.-R. Wang, Y.-Y. Ding, G.-H. Wang, Z.-J. Zhang, Z.-R. Wu and Y.-Q. Liu, Bioorg. Med. Chem., 2024, 112, 117880 CrossRef CAS.
- A. Tanaka, X. Ma, A. Takahashi and J. M. Vierling, Lancet, 2024, 404, 1053 CrossRef CAS PubMed.
- L. S. Choi, I. G. Jo, K. S. Kang, J. H. Im, J. Kim, J. Kim, J. W. Chung and S.-K. Yoo, Int. J. Obes., 2021, 45, 130 CrossRef CAS PubMed.
- J. Zhang, X. Wu, B. Zhong, Q. Liao, X. Wang, Y. Xie and X. He, Drug Des., Dev. Ther., 2023, 17, 15 CrossRef CAS.
- K. Min, B. Oh, H. Y. Koo, Y.-H. Kim, J.-W. Lee, S. Lee, Y. Kim and H. Kwon, Front. Pharmacol., 2023, 14, 1177539 CrossRef CAS PubMed.
- G.-C. Shin, H. M. Lee, N. Kim, J. Hur, S.-K. Yoo, Y. S. Park, H. S. Park, D. Ryu, M.-H. Park, J. H. Park, S.-U. Seo, L. S. Choi, M. R. Madsen, M. Feigh, K. P. Kim and K.-H. Kim, Br. J. Pharmacol., 2024, 181, 3717 CrossRef CAS PubMed.
- E. J. Ha, J. I. Seo, S. U. Rehman, H. S. Park, S.-K. Yoo and H. H. Yoo, Pharmaceutics, 2023, 15, 1684 CrossRef CAS PubMed.
- J. Shin, L. S. Choi, H. J. Jeon, H. M. Lee, S. H. Kim, K.-W. Kim, W. Ko, H. Oh and H. S. Park, Molecules, 2023, 28, 2135 CrossRef CAS.
- C. Yu and L. Xu, World Pat., WO2023134732A1, 2023 Search PubMed.
- I. Yoon, S. Kim, M. Cho, K. A. You, J. Son, C. Lee, J. H. Suh, D. J. Bae, J. M. Kim, S. Oh, S. Park, S. Kim, S. H. Cho, S. Park, K. Bang, M. Seo, J. H. Kim, B. Lee, J. S. Park, K. Y. Hwang and S. Kim, EMBO Mol. Med., 2023, 15, e16940 CrossRef CAS.
- M. Y. Park, S. Bae, J. A. Heo, M. Park, Y. Kim, J. Han, I.-J. Jang, K.-S. Yu and J. Oh, Clin. Transl. Sci., 2023, 16, 1163 CrossRef CAS PubMed.
- N. P. McLaughlin, P. Evans and M. Pines, Bioorg. Med. Chem., 2014, 22, 1993 CrossRef CAS PubMed.
- J. Brager, C. Chapman, L. Dunn and A. Kaplin, Drug Res., 2022, 73, 95 Search PubMed.
- J. D. Glenn, I. M. Pantoja, P. Caturegli and K. A. Whartenby, J. Neuroimmunol., 2020, 339, 577115 CrossRef CAS PubMed.
- G. Di Dalmazi, P. Chalan and P. Caturegli, J. Immunol., 2019, 202, 1350 CrossRef CAS PubMed.
- TNF Pharmaceuticals, TNF Pharmaceuticals prepares to advance lead clinical program targeting age-related decline (Press Release 21 August 2024), https://tnfpharma.com/news/tnf-pharmaceuticals-prepares-to-advance-lead-clinical, accessed 7 April 2025.
- F.-X. Li, Q.-Z. Zhang, S.-J. Li, G. Lin, X.-Y. Huo, Y. Lan and Z. Yang, Org. Lett., 2021, 23, 3421 CrossRef CAS PubMed.
- S. A. Harrison, P. R. Mayo, T. M. Hobbs, C. Canizares, E. P. Foster, C. Zhao, D. R. Ure, D. J. Trepanier, J. A. Greytok and R. T. Foster, Hepatol. Commun., 2022, 6, 3379 CrossRef CAS PubMed.
- W. T. Stauffer, A. Z. Goodman, M. Bobardt, D. R. Ure, R. T. Foster and P. Gallay, PLoS One, 2024, 19, e0298211 CrossRef CAS PubMed.
- E. Remenchik, P. R. Mayo, T. M. Hobbs, J. A. Greytok, E. P. Foster, C. Zhao, D. Ure, D. J. Trepanier and R. T. Foster, Clin. Pharmacol. Drug Dev., 2023, 12, 287 CrossRef CAS.
- D. R. Ure, D. J. Trepanier, P. R. Mayo and R. T. Foster, Expert Opin. Invest. Drugs, 2020, 29, 163 CrossRef CAS PubMed.
- C. S. Moon, H. M. Kang, Y. Nam, J. Lim, J. Kim, T.-H. Lee, J. Lee, M. S. Chang and J. Y. Lee, Org. Lett., 2024, 26, 6535 CrossRef CAS PubMed.
- H. M. Kang, C. S. Moon, Y. Nam, J. Lim, J. Kim, T.-H. Lee, J. Lee, M. S. Chang and J. Y. Lee, Org. Process Res. Dev., 2024, 28, 4328 CrossRef CAS.
- Qgenetics, Qgenetics company webpage, http://www.qgenetics.co.kr/, accessed 7 April 2025.
- W. Zhang, S. Mi, X. He, J. Cui, K. Zhi and J. Zhang, Int. J. Mol. Sci., 2024, 25, 8255 CrossRef CAS PubMed.
- D. Cantarovich, D. Kervella, G. Karam, J. Dantal, G. Blancho, M. Giral, C. Garandeau, A. Houzet, S. Ville, J. Branchereau, F. Delbos, C. Guillot-Gueguen, C. Volteau, M. Leroy, K. Renaudin, J.-P. Soulillou and M. Hourmant, Am. J. Transplant., 2020, 20, 1679 CrossRef CAS PubMed.
- B. Tönshoff, in Pediatric Pharmacotherapy, ed. W. Kiess, M. Schwab and J. van den Anker, Springer International Publishing, Cham, 2020, p. 441, DOI:10.1007/164_2019_331.
- T. Zesiewicz, J. L. Salemi, S. Perlman, K. L. Sullivan, J. D. Shaw, Y. Huang, C. Isaacs, C. Gooch, D. R. Lynch and M. B. Klein, Neurodegener. Dis. Manag., 2018, 8, 233 CrossRef PubMed.
- PTC Therapeutics, PTC Therapeutics announces vatiquinone NDA submission to FDA for the treatment of children and adults living with Friedreich ataxia (Press Release 19 December 2024), https://ir.ptcbio.com/news-releases/news-release-details/ptc-therapeutics-announces-vatiquinone-nda-submission-fda, accessed 7 April 2025.
- W. D. Shrader, A. Amagata, A. Barnes, G. M. Enns, A. Hinman, O. Jankowski, V. Kheifets, R. Komatsuzaki, E. Lee, P. Mollard, K. Murase, A. A. Sadun, M. Thoolen, K. Wesson and G. Miller, Bioorg. Med. Chem. Lett., 2011, 21, 3693 CrossRef CAS PubMed.
- Á. Cores, N. Carmona-Zafra, J. Clerigué, M. Villacampa and J. C. Menéndez, Antioxidants, 2023, 12, 1464 CrossRef PubMed.
- A. H. Kahn-Kirby, A. Amagata, C. I. Maeder, J. J. Mei, S. Sideris, Y. Kosaka, A. Hinman, S. A. Malone, J. J. Bruegger, L. Wang, V. Kim, W. D. Shrader, K. G. Hoff, J. C. Latham, E. A. Ashley, M. T. Wheeler, E. Bertini, R. Carrozzo, D. Martinelli, C. Dionisi-Vici, K. A. Chapman, G. M. Enns, W. Gahl, L. Wolfe, R. P. Saneto, S. C. Johnson, J. K. Trimmer, M. B. Klein and C. R. Holst, PLoS One, 2019, 14, e0214250 CrossRef CAS PubMed.
- F. Feng, R. Luo, D. Mu and Q. Cai, Mol. Neurobiol., 2024, 61, 7354 CrossRef CAS PubMed.
- J. Smeitink, R. van Maanen, L. de Boer, G. Ruiterkamp and H. Renkema, BMC Neurol., 2022, 22, 158 CrossRef CAS PubMed.
- J. Beyrath, M. Pellegrini, H. Renkema, L. Houben, S. Pecheritsyna, P. van Zandvoort, P. van den Broek, A. Bekel, P. Eftekhari and J. A. M. Smeitink, Sci. Rep., 2018, 8, 6577 CrossRef PubMed.
- Y. Xiao, K. Yim, H. Zhang, D. Bakker, R. Nederlof, J. A. M. Smeitink, H. Renkema, M. W. Hollmann, N. C. Weber and C. J. Zuurbier, Cardiovasc. Drugs Ther., 2021, 35, 745 CrossRef CAS PubMed.
- X. Jiang, H. Renkema, B. Pennings, S. Pecheritsyna, J. C. Schoeman, T. Hankemeier, J. Smeitink and J. Beyrath, Sci. Rep., 2021, 11, 880 CrossRef CAS PubMed.
- X. Jiang, H. Renkema, J. Smeitink and J. Beyrath, PLoS One, 2021, 16, e0254315 CrossRef CAS PubMed.
- Y. Kim, M. Son, J. Ko, H. Cho, M. Yoo, W. Kim, I. Song and C. Kim, Arch. Pharmacal Res., 1999, 22, 354 CrossRef CAS PubMed.
- E. Kim, J. S. Park and T. Y. Chung, Invest. Ophthalmol. Vis. Sci., 2023, 64, 3980 Search PubMed.
- J. Jung, K. Y. Huh, X. Jin, A. Ha, K. H. Park, J. S. Park, E. Kim, J. Lee, I.-J. Jang and H. Lee, Clin. Transl. Sci., 2022, 15, 343 CrossRef CAS PubMed.
- J. Lee, K.-H. Shin, J.-R. Kim, K. S. Lim, I.-J. Jang and J.-Y. Chung, Clin. Drug Invest., 2014, 34, 37 CrossRef CAS PubMed.
- Y. W. Kim, W. H. Lee, S. M. Choi, Y. Y. Seo, B. O. Ahn, S. H. Kim and S. G. Kim, J. Gastroenterol. Hepatol., 2012, 27, 397 CrossRef CAS PubMed.
- S. M. Choi, J. H. Shin, K. K. Kang, B. O. Ahn and M. Yoo, Dig. Dis. Sci., 2007, 52, 3075 CrossRef CAS PubMed.
- H. Lee, E. K. Kim, J. Y. Kim, Y.-M. Yang, D. M. Shin, K. K. Kang and T.-i. Kim, Invest. Ophthalmol. Vis. Sci., 2014, 55, 6565 CrossRef CAS PubMed.
- Alcon, Alcon announces positive topline results from phase 3 COMET trials of AR-15512, a novel topical drug candidate for dry eye (Press Release 9 January 2024), https://www.alcon.com/media-release/alcon-announces-positive-topline-results-phase-3-comet-trials-ar-15512-novel-topical/, accessed 6 February 2025.
- A. Mullard, Nat. Rev. Drug Discovery, 2025, 24, 491 CrossRef CAS PubMed.
- H. R. Watson, D. G. Rowsell and J. H. D. Browning, UK Pat., GB1351761A, 1971 Search PubMed.
- H. R. Watson, R. Hems, D. G. Rowsell and D. J. Spring, J. Soc. Cosmet. Chem., 1978, 29, 185 CAS.
- M. A. Sherkheli, A. K. Vogt-Eisele, D. Bura, L. R. Beltrán Márques, G. Gisselmann, H. Hatt and J. Pharm, Pharm. Sci., 2010, 13, 242 CAS.
- Y. Yin, S. C. Le, A. L. Hsu, M. J. Borgnia, H. Yang and S.-Y. Lee, Science, 2019, 363, eaav9334 CrossRef CAS PubMed.
- A. Parra, R. Madrid, D. Echevarria, S. del Olmo, C. Morenilla-Palao, M. C. Acosta, J. Gallar, A. Dhaka, F. Viana and C. Belmonte, Nat. Med., 2010, 16, 1396 CrossRef CAS PubMed.
- S. I. Cho, U. J. Park, J.-M. Chung and B. J. Gwag, Drug News Perspect., 2010, 23, 548 Search PubMed.
- N. P. Visavadiya, M. L. McEwen, J. D. Pandya, P. G. Sullivan, B. J. Gwag and J. E. Springer, Toxicol. In Vitro, 2013, 27, 788 CrossRef CAS PubMed.
- V. Dammavalam, S. Lin, S. Nessa, N. Daksla, K. Stefanowski, A. Costa and S. Bergese, Int. J. Mol. Sci., 2024, 25, 891 CrossRef CAS PubMed.
- J. S. Lee, H. G. Kang, S. H. Ahn, T.-J. Song, D.-I. Shin, H.-J. Bae, C. H. Kim, S. H. Heo, J.-K. Cha, Y. B. Lee, E. G. Kim, M. S. Park, H.-K. Park, J. Kim, S. Yu, H. Mo, S. I. Sohn, J. H. Kwon, J. G. Kim, Y. S. Kim, J. C. Choi, Y.-H. Hwang, K. H. Jung, S.-K. Kim, W. K. Seo, J. H. Seo, J. Yoo, J. Y. Chang, M. Park, J. S. Lee, C. San An, B. J. Gwag, D. W. Choi and S. U. Kwon, JAMA Netw. Open, 2025, 8, e2456535 CrossRef PubMed.
- GNT Pharma, Crisdesalazine, http://www.gntpharma.com/EN/en2_2_b.php, accessed 5 February 2025.
- J. H. Shin, Y. A. Lee, J. K. Lee, Y. B. Lee, W. Cho, D. S. Im, J. H. Lee, B. S. Yun, J. E. Springer and B. J. Gwag, J. Neurochem., 2012, 122, 952 CrossRef CAS PubMed.
- Business Wire, New drug showing symptomatic relief and disease-modifying potential in canine cognitive dysfunction syndrome (Press Release 10 February 2021), https://www.businesswire.com/news/home/20210210005036/en, accessed 7 April 2025.
- A. T. Ghisaidoobe, R. J. B. H. N. van den Berg, S. S. Butt, A. Strijland, W. E. Donker-Koopman, S. Scheij, A. M. C. H. van den Nieuwendijk, G.-J. Koomen, A. van Loevezijn, M. Leemhuis, T. Wennekes, M. van der Stelt, G. A. van der Marel, C. A. A. van Boeckel, J. M. F. G. Aerts and H. S. Overkleeft, J. Med. Chem., 2014, 57, 9096 CrossRef CAS PubMed.
- Azafaros, Azafaros announces positive topline Phase 2 study data with nizubaglustat in GM2 gangliosidosis and Niemann-Pick disease type C (Press Release 16 July 2024), https://www.azafaros.com/news/azafaros-announces-positive-topline-phase-2-study-data-with-nizubaglustat-in-gm2-gangliosidosis-and-niemann-pick-disease-type-c/l187c14, accessed 7 April 2025.
- C. Paquet Luzy, E. Doppler, T. M. Polasek and R. Giorgino, Mol. Genet. Metab., 2024, 141, 108113 CrossRef CAS PubMed.
- T. Bremova-Ertl and S. Schneider, Expert Opin. Pharmacother., 2023, 24, 1229 CrossRef CAS PubMed.
- J. Bagli, D. Kluepfel and M. St-Jacques, J. Org. Chem., 1973, 38, 1253 CrossRef PubMed.
- T. Fujita, K. Inoue, S. Yamamoto, T. Ikumoto, S. Sasaki, R. Toyama, K. Chiba, Y. Hoshino and T. Okumoto, J. Antibiot., 1994, 47, 208 CrossRef CAS PubMed.
- C. R. Strader, C. J. Pearce and N. H. Oberlies, J. Nat. Prod., 2011, 74, 900 CrossRef CAS PubMed.
- V. Brinkmann, A. Billich, T. Baumruker, P. Heining, R. Schmouder, G. Francis, S. Aradhye and P. Burtin, Nat. Rev. Drug Discovery, 2010, 9, 883 CrossRef CAS PubMed.
- S. Pan, N. S. Gray, W. Gao, Y. Mi, Y. Fan, X. Wang, T. Tuntland, J. Che, S. Lefebvre, Y. Chen, A. Chu, K. Hinterding, A. Gardin, P. End, P. Heining, C. Bruns, N. G. Cooke and B. Nuesslein-Hildesheim, ACS Med. Chem. Lett., 2013, 4, 333 CrossRef CAS PubMed.
- D. J. Buzard, S. H. Kim, L. Lopez, A. Kawasaki, X. Zhu, J. Moody, L. Thoresen, I. Calderon, B. Ullman, S. Han, J. Lehmann, T. Gharbaoui, D. Sengupta, L. Calvano, A. G. Montalban, Y.-A. Ma, C. Sage, Y. Gao, G. Semple, J. Edwards, J. Barden, M. Morgan, W. Chen, K. Usmani, C. Chen, A. Sadeque, R. J. Christopher, J. Thatte, L. Fu, M. Solomon, D. Mills, K. Whelan, H. Al-Shamma, J. Gatlin, M. Le, I. Gaidarov, T. Anthony, D. J. Unett, A. Blackburn, J. Rueter, S. Stirn, D. P. Behan and R. M. Jones, ACS Med. Chem. Lett., 2014, 5, 1313 CrossRef CAS PubMed.
- H. Shimizu, M. Takahashi, T. Kaneko, T. Murakami, Y. Hakamata, S. Kudou, T. Kishi, K. Fukuchi, S. Iwanami, K. Kuriyama, T. Yasue, S. Enosawa, K. Matsumoto, I. Takeyoshi, Y. Morishita and E. Kobayashi, Circulation, 2005, 111, 222 CrossRef CAS PubMed.
- Y. Ikeda, M. I. Davis, K. Sumita, Y. Zheng, S. Kofuji, M. Sasaki, Y. Hirota, R. Pragani, M. Shen, M. B. Boxer, K. Takeuchi, T. Senda, A. Simeonov and A. T. Sasaki, Biochem. Biophys. Res. Commun., 2023, 679, 116 CrossRef CAS PubMed.
- S. Dertschnig, P. Gergely, J. Finke, U. Schanz, E. Holler, U. Holtick, G. Socié, M. Medinger, J. Passweg, T. Teshima, C. Stylianou, S. Oehen, D. Heim and C. Bucher, Transplant. Cell. Ther., 2023, 29, 41.e1 CrossRef CAS PubMed.
- C. H. Hong, M. S. Ko, J. H. Kim, H. Cho, C.-H. Lee, J. E. Yoon, J.-Y. Yun, I.-J. Baek, J. E. Jang, S. E. Lee, Y. K. Cho, J. Y. Baek, S. J. Oh, B. Y. Lee, J. S. Lim, J. Lee, S. M. Hartig, L. Conde de la Rosa, C. Garcia-Ruiz, K.-U. Lee, J. C. Fernández-Checa, J. W. Choi, S. Kim and E. H. Koh, Cell. Mol. Gastroenterol. Hepatol., 2022, 13, 925 CrossRef PubMed.
- S. Y. Kim, S. Park, R. Cui, H. Lee, H. Choi, M. E.-A. Farh, H. I. Jo, J. H. Lee, H. J. Song, Y.-J. Lee, Y.-S. Lee, B. Y. Lee and J. Cho, Int. J. Mol. Sci., 2023, 24, 16265 CrossRef CAS PubMed.
- J. Lickliter, X. Yang, J. Guo, W. Pan and Z. Wei, Front. Immunol., 2024, 15, 1380975 CrossRef CAS PubMed.
- L. Yu, L. He, B. Gan, R. Ti, Q. Xiao, X. Yang, H. Hu, L. Zhu, S. Wang and R. Ren, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2117716119 CrossRef CAS PubMed.
- Connect Biopharma, Annual Filings (Form 20-F) (Files 16 April 2024), https://investors.connectbiopharm.com/financials-filings/sec-filings, accessed 7 April 2025.
- Sun Pharma, Investor Presentation – July 2024, https://sunpharma.com/wp-content/uploads/2024/07/SPIL-IR-Presentation-July2024-INR.pdf, accessed 30 September 2024.
- K. Sugahara, Y. Maeda, K. Shimano, A. Mogami, H. Kataoka, K. Ogawa, K. Hikida, H. Kumagai, M. Asayama, T. Yamamoto, T. Harada, P. Ni, S. Inoue and A. Kawaguchi, Br. J. Pharmacol., 2017, 174, 15 CrossRef CAS PubMed.
- K. Shimano, Y. Maeda, H. Kataoka, M. Murase, S. Mochizuki, H. Utsumi, K. Oshita and K. Sugahara, PLoS One, 2019, 14, e0226154 CrossRef CAS PubMed.
- H. W. B. Johnson, E. Lowe, J. L. Anderl, A. Fan, T. Muchamuel, S. Bowers, D. C. Moebius, C. Kirk and D. L. McMinn, J. Med. Chem., 2018, 61, 11127 CrossRef CAS PubMed.
- T. Muchamuel, R. A. Fan, J. L. Anderl, D. J. Bomba, H. W. B. Johnson, E. Lowe, B. B. Tuch, D. L. McMinn, B. Millare and C. J. Kirk, Front. Immunol., 2023, 14, 1043680 CrossRef CAS PubMed.
- Y. Fang, H. Johnson, J. L. Anderl, T. Muchamuel, D. McMinn, C. Morisseau, B. D. Hammock, C. Kirk and J. Wang, Drug Metab. Dispos., 2021, 49, 810 CrossRef CAS PubMed.
- K. Sugawara, M. Hatori, Y. Nishiyama, K. Tomita, H. Kamei, M. Konishi and T. Oki, J. Antibiot., 1990, 43, 8 CrossRef CAS PubMed.
- M. Hanada, K. Sugawara, K. Kaneta, S. Toda, Y. Nishiyama, K. Tomita, H. Yamamoto, M. Konishi and T. Oki, J. Antibiot., 1992, 45, 1746 CrossRef CAS PubMed.
- L. Meng, R. Mohan, B. H. B. Kwok, M. Elofsson, N. Sin and C. M. Crews, Proc. Natl. Acad. Sci. U. S. A., 1999, 96, 10403 CrossRef CAS PubMed.
- K. B. Kim and C. M. Crews, Nat. Prod. Rep., 2013, 30, 600 RSC.
- AnnJi Pharmaceutical, Avenue Therapeutics announces last patient last visit in phase 1b/2a clinical trial of AJ201 for the treatment of spinal and bulbar muscular atrophy (Kennedy's disease) (Press Release 16 May 2024), https://www.ajpharm.com/2024/05/22/avenue-therapeutics-announces-last-patient-last-visit-in-phase-1b-2a-clinical-trial-of-aj201-for-the-treatment-of-spinal-and-bulbar-muscular-atrophy-kennedys-disease/, accessed 7 April 2025.
- D. Prasad, A. Praveen, S. Mahapatra, S. Mogurampelly and S. R. Chaudhari, Food Chem., 2021, 360, 130000 CrossRef CAS PubMed.
- K. Kaur, A. K. Al-Khazaleh, D. J. Bhuyan, F. Li and C. G. Li, Antioxidants, 2024, 13, 1092 CrossRef CAS PubMed.
- H. Ohtsu, Z. Xiao, J. Ishida, M. Nagai, H.-K. Wang, H. Itokawa, C.-Y. Su, C. Shih, T. Chiang, E. Chang, LeeLee, M.-Y. Tsai, C. Chang and K.-H. Lee, J. Med. Chem., 2002, 45, 5037 CrossRef CAS PubMed.
- L. R. C. Barclay, M. R. Vinqvist, K. Mukai, H. Goto, Y. Hashimoto, A. Tokunaga and H. Uno, Org. Lett., 2000, 2, 2841 CrossRef CAS PubMed.
- Z. Yang, Y.-J. Chang, I. C. Yu, S. Yeh, C.-C. Wu, H. Miyamoto, D. E. Merry, G. Sobue, L.-M. Chen, S.-S. Chang and C. Chang, Nat. Med., 2007, 13, 348 CrossRef CAS PubMed.
- L. C. Bott, N. M. Badders, K.-l. Chen, G. G. Harmison, E. Bautista, C. C. Y. Shih, M. Katsuno, G. Sobue, J. P. Taylor, N. P. Dantuma, K. H. Fischbeck and C. Rinaldi, Hum. Mol. Genet., 2016, 25, 1979 CrossRef CAS PubMed.
- C. Grunseich and K. H. Fischbeck, Curr. Opin. Neurol., 2020, 33, 629 CrossRef CAS PubMed.
- G.-Y. Lai, H. Chan, T.-C. Chen, W.-J. Lee and Y.-S. Ho, J. Cancer Res. Pract., 2022, 9, 1 CrossRef.
- Y.-L. Wu, J.-C. Chang, Y.-C. Chao, H. Chan, M. Hsieh and C.-S. Liu, Antioxidants, 2022, 11, 1389 CrossRef CAS PubMed.
- J. Kricker and V. Norris, World Pat., WO2024194494A1, 2024 Search PubMed.
- BioSpace, EpiEndo reports Phase 2A results for lead asset, EP395, in COPD (10 April 2024), https://www.biospace.com/epiendo-reports-phase-2a-results-for-lead-asset-ep395-in-copd, accessed 7 April 2025.
- W. Lee, O. Yuseok, C. Lee, S. Y. Jeong, J.-H. Lee, M.-C. Baek, G.-Y. Song and J.-S. Bae, Biochem. Pharmacol., 2019, 163, 260 CrossRef CAS PubMed.
- Z. He, Y. Wang, Y. Chen, F. Geng, Z. Jiang and X. Li, Biochem. Syst. Ecol., 2023, 111, 104717 CrossRef CAS.
- S.-m. Kang, M.-H. Yoon, J. Ahn, J.-E. Kim, S. Y. Kim, S. Y. Kang, J. Joo, S. Park, J.-H. Cho, T.-G. Woo, A.-Y. Oh, K. J. Chung, S. Y. An, T. S. Hwang, S. Y. Lee, J.-S. Kim, N.-C. Ha, G.-Y. Song and B.-J. Park, Commun. Biol., 2021, 4, 5 CrossRef CAS PubMed.
- S.-m. Kang, S. Seo, E. J. Song, O. Kweon, A.-h. Jo, S. Park, T.-G. Woo, B.-H. Kim, G. T. Oh and B.-J. Park, Cells, 2023, 12, 1232 CrossRef CAS PubMed.
- S.-m. Kang, M.-H. Yoon, S.-J. Lee, J. Ahn, S. A. Yi, K. H. Nam, S. Park, T.-G. Woo, J.-H. Cho, J. Lee, N.-C. Ha and B.-J. Park, Sci. Rep., 2021, 11, 9122 CrossRef CAS PubMed.
- C. K. Murphy, B. Dixit, F. B. Oleson, R. E. Dolle, R. Farquhar and B. A. McCormick, FEBS Open Bio, 2023, 13, 1434 CrossRef CAS PubMed.
- J. R. Allegretti, A. S. Cheifetz, P. S. Dulai, A. C. Stevens, J. Chapas-Reed, L. Chesnel, B. Dixit, R. Farquhar, P. Ghahramani, B. W. Miller, C. K. Murphy, M. Quintas, R. Tanase, T. Telia, B. Woźniak-Stolarska and R. Gupta, Am. J. Gastroenterol., 2025, 120, 1624 CrossRef PubMed.
- R. L. Szabady, C. Louissaint, A. Lubben, B. Xie, S. Reeksting, C. Tuohy, Z. Demma, S. E. Foley, C. S. Faherty, A. Llanos-Chea, A. J. Olive, R. J. Mrsny and B. A. McCormick, J. Clin. Invest., 2018, 128, 4044 CrossRef PubMed.
- D. A. Dorward, C. D. Lucas, G. B. Chapman, C. Haslett, K. Dhaliwal and A. G. Rossi, Am. J. Pathol., 2015, 185, 1172 CrossRef CAS PubMed.
- J. Kandasamy, D. Atia-Glikin, E. Shulman, K. Shapira, M. Shavit, V. Belakhov and T. Baasov, J. Med. Chem., 2012, 55, 10630 CrossRef CAS PubMed.
- E. Kerem, Expert Opin. Invest. Drugs, 2020, 29, 1347 CrossRef CAS PubMed.
- D. K. Crawford, J. Mullenders, J. Pott, S. F. Boj, S. Landskroner-Eiger and M. M. Goddeeris, J. Cystic Fibrosis, 2021, 20, 436 CrossRef CAS PubMed.
- Global News Wire, Eloxx Pharmaceuticals provides pipeline and financing updates (Press Release 11 July 2024), https://www.globenewswire.com/news-release/2024/07/11/2911999/0/en/Eloxx-Pharmaceuticals-Provides-Pipeline-and-Financing-Updates, accessed 6 February 2025.
- J. F. Burke and A. E. Mogg, Nucleic Acids Res., 1985, 13, 6265 CrossRef CAS PubMed.
- W. J. Friesen, B. Johnson, J. Sierra, J. Zhuo, P. Vazirani, X. Xue, Y. Tomizawa, R. Baiazitov, C. Morrill, H. Ren, S. Babu, Y.-C. Moon, A. Branstrom, A. Mollin, J. Hedrick, J. Sheedy, G. Elfring, M. Weetall, J. M. Colacino, E. M. Welch and S. W. Peltz, PLoS One, 2018, 13, e0206158 CrossRef PubMed.
- S. Li, J. Li, W. Shi, Z. Nie, S. Zhang, F. Ma, J. Hu, J. Chen, P. Li and X. Xie, Biomolecules, 2023, 13, 988 CrossRef CAS PubMed.
- M. R. Graf, S. Apte, E. Terzo, S. Padhye, S. Shi, M. K. Cox, R. B. Clark, V. Modur and V. Badarinarayana, J. Mol. Med., 2023, 101, 375 CrossRef CAS PubMed.
- Almirall, Almirall and Eloxx Pharmaceuticals Enter into Exclusive Agreement to license ZKN-013 for rare dermatological diseases (Press Release 13 March 2024), https://www.almirall.com/newsroom/news/almirall-and-eloxx-pharmaceuticals-enter-into-exclusive-agreement-to-license-zkn-013-for-rare-dermatological-diseases, accessed 7 April 2025.
- M. D. Kornberg, M. D. Smith, H. A. Shirazi, P. A. Calabresi, S. H. Snyder and P. M. Kim, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 2186 CrossRef CAS PubMed.
- F. Safaeinejad, S. Bahrami, H. Redl and H. Niknejad, Front. Pharmacol., 2018, 9, 625 CrossRef PubMed.
- Business Wire, Enveda announces first candidate discovered from nature using AI advances to clinical trials (13 November 2024), https://www.businesswire.com/news/home/20241113334257/en/Enveda-Announces-First-Candidate-Discovered-From-Nature-Using-AI-Advances-to-Clinical-Trials, accessed 7 April 2025.
- Enveda Biosciences, Differentiated medicines, developed at record pace, https://enveda.com/pipeline/, accessed 7 April 2025.
- V. Colluru, D. Wee, M. Gaetz, H. Gordon, M. Yu and M. Deeg, World Pat., WO2024123825A1, 2023 Search PubMed.
- H. Erdtman and Y. Hirose, Acta Chem. Scand., 1962, 16, 1311 CrossRef CAS.
- W. D. MacLeod, Tetrahedron Lett., 1965, 6, 4779 CrossRef.
- M. A. Fraatz, R. G. Berger and H. Zorn, Appl. Microbiol. Biotechnol., 2009, 83, 35 CrossRef CAS PubMed.
- X. Deng, Z. Ye, J. Duan, F. Chen, Y. Zhi, M. Huang, M. Huang, W. Cheng, Y. Dou, Z. Kuang, Y. Huang, G. Bian, Z. Deng, T. Liu and L. Lu, New Phytol., 2024, 241, 779 CrossRef CAS PubMed.
- A. Rani, V. Saini, P. Patra, T. Prashar, R. K. Pandey, A. Mishra and H. C. Jha, ACS Chem. Neurosci., 2023, 14, 2968 CrossRef CAS PubMed.
- M. Alam, S. Ali, G. M. Ashraf, A. L. Bilgrami, D. K. Yadav and M. I. Hassan, Food Chem., 2022, 379, 132135 CrossRef CAS PubMed.
- D. Mereles and W. Hunstein, Int. J. Mol. Sci., 2011, 12, 5592 CrossRef CAS PubMed.
- J. B. Baell, J. Nat. Prod., 2016, 79, 616 CrossRef CAS PubMed.
- D. J. Newman and G. M. Cragg, J. Nat. Prod., 2020, 83, 770 CrossRef CAS PubMed.
- M. Kciuk, M. Alam, N. Ali, S. Rashid, P. Głowacka, R. Sundaraj, I. Celik, E. B. Yahya, A. Dubey, E. Zerroug and R. Kontek, Molecules, 2023, 28, 5246 CrossRef CAS PubMed.
- D. Li, D. Cao, Y. Cui, Y. Sun, J. Jiang and X. Cao, Front. Pharmacol., 2023, 14, 1201085 CrossRef CAS PubMed.
- P. C. Agioutantis, V. Kotsikoris, F. N. Kolisis and H. Loutrari, Comput. Struct. Biotechnol. J., 2020, 18, 686 CrossRef CAS PubMed.
- S. Dallavalle, A. Ferrari, B. Biasotti, L. Merlini, S. Penco, G. Gallo, M. Marzi, M. O. Tinti, R. Martinelli, C. Pisano, P. Carminati, N. Carenini, G. Beretta, P. Perego, M. De Cesare, G. Pratesi and F. Zunino, J. Med. Chem., 2001, 44, 3264 CrossRef CAS PubMed.
- M. De Cesare, G. Pratesi, P. Perego, N. Carenini, S. Tinelli, L. Merlini, S. Penco, C. Pisano, F. Bucci, L. Vesci, S. Pace, F. Capocasa, P. Carminati and F. Zunino, Cancer Res., 2001, 61, 7189 CAS.
- Lee's Pharmaceutical Holdings, 2024 Interim Report, https://www.leespharm.com/wp-content/uploads/2024/09/2024091901105.pdf, accessed 7 April 2025.
- M. E. Wall, M. C. Wani, C. E. Cook, K. H. Palmer, A. T. McPhail and G. A. Sim, J. Am. Chem. Soc., 1966, 88, 3888 CrossRef CAS.
- N. H. Oberlies and D. J. Kroll, J. Nat. Prod., 2004, 67, 129 CrossRef CAS PubMed.
- G. S. Falchook, J. C. Bendell, S. V. Ulahannan, S. Sen, R. Vilimas, K. Kriksciukaite, L. Mei, G. Jerkovic, N. Sarapa, M. Bilodeau, J. Bloss and A. Thomas, J. Clin. Oncol., 2020, 38, 3515 Search PubMed.
- D. A. Proia, D. L. Smith, J. Zhang, J.-P. Jimenez, J. Sang, L. S. Ogawa, M. Sequeira, J. Acquaviva, S. He, C. Zhang, V. Khazak, I. Astsaturov, T. Inoue, N. Tatsuta, S. Osman, R. C. Bates, D. Chimmanamada and W. Ying, Mol. Cancer Ther., 2015, 14, 2422 CrossRef CAS PubMed.
- A. Y. Deneka, L. Haber, M. C. Kopp, A. V. Gaponova, A. S. Nikonova and E. A. Golemis, PLoS One, 2017, 12, e0176747 CrossRef PubMed.
- C. Zhuang, X. Guan, H. Ma, H. Cong, W. Zhang and Z. Miao, Eur. J. Med. Chem., 2019, 163, 883 CrossRef CAS PubMed.
- H.-G. Lerchen, B. Stelte-Ludwig, M. Heroult, D. Zubov, K. M. Gericke, H. Wong, M. M. Frigault, A. J. Johnson, R. Izumi and A. Hamdy, Cancers, 2023, 15, 4381 CrossRef CAS PubMed.
- H. Joshi, D. S. Gupta, N. K. Abjani, G. Kaur, C. D. Mohan, J. Kaur, D. Aggarwal, I. Rani, S. Ramniwas, H. S. Abdulabbas, M. Gupta and H. S. Tuli, Naunyn-Schmiedeberg's Arch. Pharmacol., 2023, 396, 2893 CrossRef CAS PubMed.
- S. Rasheed, K. Rehman, M. Shahid, S. Suhail and M. S. H. Akash, J. Food Biochem., 2022, 46, e14228 CrossRef CAS PubMed.
- V. K. Singh and T. M. Seed, Expert Opin. Invest. Drugs, 2020, 29, 429 CrossRef CAS PubMed.
- C. B. Simone, A. A. Serebrenik, E. M. Gore, P. Mohindra, S. L. Brown, D. Wang, I. J. Chetty, Z. Vujaskovic, S. Menon, J. Thompson, G. Fine, M. D. Kaytor and B. Movsas, Int. J. Radiat. Oncol. Biol. Phys., 2024, 118, 404 CrossRef PubMed.
- V. K. Singh, A. A. Serebrenik, S. Y. Wise, S. A. Petrus, O. O. Fatanmi and M. D. Kaytor, Mil. Med., 2024, 189, 390 CrossRef PubMed.
- A. A. Serebrenik, O. O. Fatanmi, S. Y. Wise, S. A. Petrus, M. D. Kaytor and V. K. Singh, Int. J. Mol. Sci., 2024, 25, 8818 CrossRef CAS PubMed.
- M. Quintela-Fandino, S. Morales, A. Cortés-Salgado, L. Manso, J. V. Apala, M. Muñoz, A. Gasol Cudos, J. Salla Fortuny, M. Gion, A. Lopez-Alonso, J. Cortés, J. Guerra, D. Malón, E. Caleiras, F. Mulero and S. Mouron, Clin. Cancer Res., 2020, 26, 35 CrossRef CAS PubMed.
- P. Kate, F. Walter Douglas, L. Olivier, D. Frederic and W. John, Curr. Cancer Drug Targets, 2020, 20, 341 CrossRef PubMed.
- Noxopharm, Noxopharm Annual Report Year Ended 30 June 2024, https://www.noxopharm.com/pdf/cd0e86f1-a924-4af7-aa5d-1a4e14846ffc/Annual-Report-to-shareholders.pdf, accessed 7 April 2025.
- L. Zhang, J. Zhang, Z. Ye, Y. Manevich, L. E. Ball, J. R. Bethard, Y.-L. Jiang, A.-M. Broome, A. C. Dalton, G. Y. Wang, D. M. Townsend and K. D. Tew, Cancer Res., 2019, 79, 4072 CrossRef CAS PubMed.
- K. H. Hurrish, Y. Su, S. Patel, C. L. Ramage, J. Zhao, B. R. Temby, J. L. Carter, H. Edwards, S. A. Buck, S. E. Wiley, M. Hüttemann, L. Polin, J. Kushner, S. H. Dzinic, K. White, X. Bao, J. Li, J. Yang, J. Boerner, Z. Hou, G. Al-Atrash, S. N. Konoplev, J. Busquets, S. Tiziani, L. H. Matherly, J. W. Taub, M. Konopleva, Y. Ge and N. Baran, Biochem. Pharmacol., 2024, 220, 115981 CrossRef CAS PubMed.
- L. Zhang, D. M. Townsend, M. Morris, E. N. Maldonado, Y.-L. Jiang, A.-M. Broome, J. R. Bethard, L. E. Ball and K. D. Tew, J. Pharmacol. Exp. Ther., 2020, 374, 308 CrossRef CAS PubMed.
- S. C. Kuo, H. Z. Lee, J. P. Juang, Y. T. Lin, T. S. Wu, J. J. Chang, D. Lednicer, K. D. Paull and C. M. Lin, J. Med. Chem., 1993, 36, 1146 CrossRef CAS PubMed.
- L.-C. Chou, M.-T. Tsai, M.-H. Hsu, S.-H. Wang, T.-D. Way, C.-H. Huang, H.-Y. Lin, K. Qian, Y. Dong, K.-H. Lee, L.-J. Huang and S.-C. Kuo, J. Med. Chem., 2010, 53, 8047 CrossRef CAS PubMed.
- L. Shen, Y.-L. Chen, C.-C. Huang, Y.-C. Shyu, R. E. B. Seftor, E. A. Seftor, M. J. C. Hendrix, D.-S. Chien and Y.-W. Chu, Pathol. Oncol. Res., 2023, 29, 1611038 CrossRef CAS PubMed.
- M. Zucchetti, D. Meco, A. Francesco, T. Servidei, V. Patriarca, G. Cusano, M. D'Incalci, D. Forestieri, C. Pisano and R. Riccardi, Cancer Chemother. Pharmacol., 2010, 66, 635 CrossRef CAS PubMed.
- J. Hu, P. Y. Wen, L. E. Abrey, C. E. Fadul, J. Drappatz, N. Salem, J. G. Supko and F. Hochberg, J. Neurooncol., 2013, 111, 347 CrossRef CAS PubMed.
- B. Carlson, T. Lahusen, S. Singh, A. Loaiza-Perez, P. J. Worland, R. Pestell, C. Albanese, E. A. Sausville and A. M. Senderowicz, Cancer Res., 1999, 59, 4634 CAS.
- P. Bose, G. L. Simmons and S. Grant, Expert Opin. Invest. Drugs, 2013, 22, 723 CrossRef CAS PubMed.
- A. D. Harmon, U. Weiss and J. V. Silverton, Tetrahedron Lett., 1979, 20, 721 CrossRef.
- R. G. Naik, S. L. Kattige, S. V. Bhat, B. Alreja, N. J. de Souza and R. H. Rupp, Tetrahedron, 1988, 44, 2081 CrossRef CAS.
- G. Kaur, M. Stetler-Stevenson, S. Sebers, P. Worland, H. Sedlacek, C. Myers, J. Czech, R. Naik and E. Sausville, J. Natl. Cancer Inst., 1992, 84, 1736 CrossRef CAS PubMed.
- M. J. Mughal, K. Bhadresha and H. F. Kwok, Semin. Cancer Biol., 2023, 88, 106 CrossRef CAS PubMed.
- J. Dey, T. L. Deckwerth, W. S. Kerwin, J. R. Casalini, A. J. Merrell, M. O. Grenley, C. Burns, S. H. Ditzler, C. P. Dixon, E. Beirne, K. C. Gillespie, E. F. Kleinman and R. A. Klinghoffer, Sci. Rep., 2017, 7, 18007 CrossRef PubMed.
- D. A. Luedtke, Y. Su, J. Ma, X. Li, S. A. Buck, H. Edwards, L. Polin, J. Kushner, S. H. Dzinic, K. White, H. Lin, J. W. Taub and Y. Ge, Signal Transduction Targeted Ther., 2020, 5, 17 CrossRef CAS PubMed.
- W. Koch, A. Wawruszak, W. Kukula-Koch, M. Zdziebło, P. Helon, Z. M. Almarhoon, B. Al-Omari, D. Calina and J. Sharifi-Rad, Naunyn-Schmiedeberg's Arch. Pharmacol., 2024, 397, 1455 CrossRef CAS PubMed.
- M.-C. Diringer, P. Coliat, C. Mathieu, N. Laurent, C. Mura, M. Banerjee, C. Zhu, A. Grabowska, A. Ritchie, P. Clarke, A. Bernard, C. Vit, H. Burckel, G. Noel, P. Harvey, X. Pivot and A. Detappe, Small, 2023, 19, 2205961 CrossRef CAS PubMed.
- B. Pająk, Biomedicines, 2022, 10, 1001 CrossRef PubMed.
- K. P. Olive, M. A. Jacobetz, C. J. Davidson, A. Gopinathan, D. McIntyre, D. Honess, B. Madhu, M. A. Goldgraben, M. E. Caldwell, D. Allard, K. K. Frese, G. DeNicola, C. Feig, C. Combs, S. P. Winter, H. Ireland-Zecchini, S. Reichelt, W. J. Howat, A. Chang, M. Dhara, L. Wang, F. Rückert, R. Grützmann, C. Pilarsky, K. Izeradjene, S. R. Hingorani, P. Huang, S. E. Davies, W. Plunkett, M. Egorin, R. H. Hruban, N. Whitebread, K. McGovern, J. Adams, C. Iacobuzio-Donahue, J. Griffiths and D. A. Tuveson, Science, 2009, 324, 1457 CrossRef CAS PubMed.
- M. R. Tremblay, A. Lescarbeau, M. J. Grogan, E. Tan, G. Lin, B. C. Austad, L.-C. Yu, M. L. Behnke, S. J. Nair, M. Hagel, K. White, J. Conley, J. D. Manna, T. M. Alvarez-Diez, J. Hoyt, C. N. Woodward, J. R. Sydor, M. Pink, J. MacDougall, M. J. Campbell, J. Cushing, J. Ferguson, M. S. Curtis, K. McGovern, M. A. Read, V. J. Palombella, J. Adams and A. C. Castro, J. Med. Chem., 2009, 52, 4400 CrossRef CAS PubMed.
- T. Masamune, Y. Mori, M. Takasugi and A. Murai, Tetrahedron Lett., 1964, 5, 913 CrossRef.
- R. F. Keeler and W. Binns, Teratology, 1968, 1, 5 CrossRef CAS PubMed.
- J. K. Chen, Nat. Prod. Rep., 2016, 33, 595 RSC.
- H. Zhu and D. J. Lewis, Expert Opin. Pharmacother., 2022, 23, 739 CrossRef CAS PubMed.
- G. M. Boyle, M. M. A. D'Souza, C. J. Pierce, R. A. Adams, A. S. Cantor, J. P. Johns, L. Maslovskaya, V. A. Gordon, P. W. Reddell and P. G. Parsons, PLoS One, 2014, 9, e108887 CrossRef PubMed.
- J. Miller, J. Campbell, A. Blum, P. Reddell, V. Gordon, P. Schmidt and S. Lowden, Front. Vet. Sci., 2019, 6, 106 CrossRef PubMed.
- M. L. Musser, P. D. Jones, T. L. Goodson, E. Roof and C. M. Johannes, J. Vet. Intern. Med., 2024, 38, 3162 CrossRef PubMed.
- J. K. Cullen, P.-Y. Yap, B. Ferguson, Z. C. Bruce, M. Koyama, H. Handoko, K. Hendrawan, J. L. Simmons, K. M. Brooks, J. Johns, E. S. Wilson, M. M. A. de Souza, N. Broit, P. Stewart, D. Shelley, T. McMahon, S. M. Ogbourne, T. H. Nguyen, Y. C. Lim, A. Pagani, G. Appendino, V. A. Gordon, P. W. Reddell, G. M. Boyle and P. G. Parsons, J. ImmunoTher. Cancer, 2024, 12, e006602 CrossRef PubMed.
- R. L. Moses, E. L. Woods, J. Dally, J. P. Johns, V. Knäuper, G. M. Boyle, V. Gordon, P. Reddell, R. Steadman and R. Moseley, Biochem. Pharmacol., 2024, 230, 116607 CrossRef CAS PubMed.
- B. Liu, Y. Lu, A. Taledaohan, S. Qiao, Q. Li and Y. Wang, Molecules, 2024, 29, 75 CrossRef CAS PubMed.
- M. Ghorbel, F. Brini, A. Sharma and M. Landi, Plant Cell Rep., 2021, 40, 1471 CrossRef CAS PubMed.
- A. Ghasemi Pirbalouti, S. E. Sajjadi and K. Parang, Arch. Pharm., 2014, 347, 229 CrossRef CAS PubMed.
- Vidac Pharma, Vidac Pharma receives reinvestment from shareholders to drive clinical trials of its cancer drug candidates (Press Release 7 November 2024), https://vidacpharma.com/en/press-releases/255-vidac-pharma-receives-reinvestment-from-shareholders-to-drive-clinical-trials-of-its-cancer-drug-candidates, accessed 7 April 2025.
- P. Cramer and E. Stockfleth, Expert Opin. Emerg. Drugs, 2020, 25, 49 CrossRef CAS PubMed.
- F. Leonelli, A. La Bella, A. Francescangeli, R. Joudioux, A.-L. Capodilupo, M. Quagliariello, L. M. Migneco, R. M. Bettolo, V. Crescenzi, G. De Luca and D. Renier, Helv. Chim. Acta, 2005, 88, 154 CrossRef CAS.
- A. Rosato, A. Banzato, G. De Luca, D. Renier, F. Bettella, C. Pagano, G. Esposito, P. Zanovello and P. Bassi, Urol. Oncol.: Semin. Orig. Invest., 2006, 24, 207 CrossRef CAS PubMed.
- P. F. Bassi, A. Volpe, D. D'Agostino, G. Palermo, D. Renier, S. Franchini, A. Rosato and M. Racioppi, J. Urol., 2011, 185, 445 CrossRef CAS PubMed.
- I. M. Montagner, A. Banzato, G. Zuccolotto, D. Renier, M. Campisi, P. Bassi, P. Zanovello and A. Rosato, Urol. Oncol.: Semin. Orig. Invest., 2013, 31, 1261 CrossRef CAS PubMed.
- M. Demeule, C. Charfi, J.-C. Currie, A. Larocque, A. Zgheib, S. Kozelko, R. Béliveau, C. Marsolais and B. Annabi, Cancer Sci., 2021, 112, 4317 CrossRef CAS PubMed.
- M. Demeule, J.-C. Currie, C. Charfi, A. Zgheib, I. Cousineau, V. Lullier, R. Béliveau, C. Marsolais and B. Annabi, Front. Immunol., 2024, 15, 1355945 CrossRef CAS PubMed.
- Y.-J. Li, C.-B. Fang, S.-S. Wang, X.-Q. Chen, Y. Li, Q. Liu, Y.-K. Qi and S.-S. Du, Bioorg. Med. Chem., 2024, 111, 117869 CrossRef CAS PubMed.
- H. Brockmann, M. N. Haschad, K. Maier and F. Pohl, Naturwissenschaften, 1939, 27, 550 CrossRef CAS.
- H. Brockmann, E.-H. F. v. Falkenhausen and A. Dorlars, Naturwissenschaften, 1950, 37, 540 CrossRef CAS.
- H. Brockmann and F. Kluge, Naturwissenschaften, 1951, 38, 141 CrossRef CAS.
- Z. Jendželovská, R. Jendželovský, B. Kuchárová and P. Fedoročko, Front. Plant Sci., 2016, 7, 560 Search PubMed.
- E. J. Kim, A. R. Mangold, J. A. DeSimone, H. K. Wong, L. Seminario-Vidal, J. Guitart, J. Appel, L. Geskin, E. Lain, N. J. Korman, N. Zeitouni, N. Nikbakht, K. Dawes, O. Akilov, J. Carter, M. Shinohara, T. M. Kuzel, W. Piette, N. Bhatia, A. Musiek, D. Pariser, Y. H. Kim, D. Elston, E. Boh, M. Duvic, A. Huen, T. Pacheco, J. P. Zwerner, S. T. Lee, M. Girardi, C. Querfeld, K. Bohjanen, E. Olsen, G. S. Wood, A. Rumage, O. Donini, A. Haulenbeek, C. J. Schaber, R. Straube, C. Pullion, A. H. Rook and B. Poligone, JAMA Dermatol., 2022, 158, 1031 CrossRef PubMed.
- X. Dong, Y. Zeng, Z. Zhang, J. Fu, L. You, Y. He, Y. Hao, Z. Gu, Z. Yu, C. Qu, X. Yin, J. Ni and L. J. Cruz, J. Pharm. Pharmacol., 2021, 73, 425 CrossRef PubMed.
- Z. Peng, J. Lu, K. Liu, L. Xie, Y. Wang, C. Cai, D. Yang, J. Xi, C. Yan, X. Li and M. Shi, Phytother. Res., 2023, 37, 5639 CrossRef CAS PubMed.
- C. E. Holbert, J. R. Foley, R. A. Casero and T. M. Stewart, Biomedicines, 2024, 12, 1157 CrossRef CAS PubMed.
- J. Qiao, W. Cai, K. Wang, E. Haubruge, J. Dong, H. R. El-Seedi, X. Xu and H. Zhang, J. Agric. Food Chem., 2024, 72, 5089 CrossRef CAS PubMed.
- C. E. Holbert, R. A. Casero and T. M. Stewart, Discov. Onc., 2024, 15, 173 CrossRef PubMed.
- Y.-J. Shi, J. Zhang, Y.-W. Wang, K. Ding, Y. Yan, C.-Y. Xia, X.-X. Li, J. He, W.-K. Zhang and J.-K. Xu, Eur. J. Med. Chem., 2022, 240, 114600 CrossRef CAS PubMed.
- C. E. Holbert, J. R. Foley, T. Murray Stewart and R. A. Casero, Int. J. Mol. Sci., 2022, 23, 6798 CrossRef CAS PubMed.
- J. B. Fiveash, X. Ye, D. M. Peerboom, T. Mikkelsen, S. Chowdhary, M. Rosenfeld, G. J. Lesser, J. Fisher, S. Desideri, S. Grossman, L. Leopold and L. B. Nabors, PLoS One, 2024, 19, e0291128 CrossRef CAS PubMed.
- R.-L. Du, H.-Y. Chow, Y. W. Chen, P.-H. Chan, R. A. Daniel and K.-Y. Wong, Front. Microbiol., 2023, 13, 1080308 CrossRef PubMed.
- Q. Yu and Y. Sun, Drug Des., Dev. Ther., 2021, 15, 1 CrossRef PubMed.
- W. Zhou, W. Wang, Y. Liang, R. Jiang, F. Qiu, X. Shao, Y. Liu, L. Fang, M. Ni, C. Yu, Y. Zhao, W. Huang, J. Li, M. J. Donovan, L. Wang, J. Ni, D. Wang, T. Fu, J. Feng, X. Wang, W. Tan and X. Fang, Nat. Commun., 2023, 14, 4212 CrossRef CAS PubMed.
- S. Qian and Z. Wang, Annu. Rev. Pharmacol. Toxicol., 1984, 24, 329 CrossRef CAS PubMed.
- O. Renner, M. Mayer, C. Leischner, M. Burkard, A. Berger, U. M. Lauer, S. Venturelli and S. C. Bischoff, Pharmaceuticals, 2022, 15, 144 CrossRef CAS PubMed.
- A. Y. Berman, R. A. Motechin, M. Y. Wiesenfeld and M. K. Holz, npj Precision Onc., 2017, 1, 35 CrossRef PubMed.
- C. Schmidt, Nat. Biotechnol., 2010, 28, 185 CrossRef CAS PubMed.
- N. Gupta, B. Zhang, Y. Zhou, F. X. McCormack, R. Ingledue, N. Robbins, E. J. Kopras, S. McMahan, A. Singla, J. Swigris, A. G. Cole and M. K. Holz, Chest, 2023, 163, 1144 CrossRef CAS PubMed.
- D. Willner, P. A. Trail, S. J. Hofstead, H. D. King, S. J. Lasch, G. R. Braslawsky, R. S. Greenfield, T. Kaneko and R. A. Firestone, Bioconjugate Chem., 1993, 4, 521 CrossRef CAS PubMed.
- S. P. Chawla, K. N. Ganjoo, S. Schuetze, Z. Papai, B. A. V. Tine, E. Choy, D. A. Liebner, M. Agulnik, S. Chawla, S. Wieland and D. J. Levitt, J. Clin. Oncol., 2017, 35, 11000 Search PubMed.
- S. Y. van der Zanden, X. Qiao and J. Neefjes, FEBS J., 2021, 288, 6095 CrossRef CAS PubMed.
- M. Sittig, Pharmaceutical Manufacturing Encyclopedia, Second Edition, Volume 1, A-K, Noyes PublicationsWestwood, NJ, USA, 1988 Search PubMed.
- LadRx Corporation, LadRx and ImmunityBio mutually agree to terminate aldoxorubicin license (Press Release 3 June 2024), https://www.ladrxcorp.com/wp-content/uploads/2024/06/Aldoxorubicin-License-Announcement-XOMA.pdf, accessed 7 April 2025.
- LadRx Corporation, LadRx planning NDA submission under 505(b)(2) for aldoxorubicin and other updates (Press Release 11 December 2024), https://www.ladrxcorp.com/wp-content/uploads/2024/12/LADX-to-Initiate-505b2.pdf, accessed 7 April 2025.
- T. A. Brooks, K. L. O'Loughlin, H. Minderman, B. N. Bundy, L. A. Ford, M. R. Vredenburg, R. J. Bernacki, W. Priebe and M. R. Baer, Invest. New Drugs, 2007, 25, 115 CrossRef CAS PubMed.
- T. H. Inge, N. L. Harris, J. Wu, R. G. Azizkhan and W. Priebe, J. Surg. Res., 2004, 121, 187 CrossRef CAS PubMed.
- C. Alifieris and D. T. Trafalis, Pharmacol. Ther., 2015, 152, 63 CrossRef CAS PubMed.
- D. Horton, W. Priebe and O. Varela, Carbohydr. Res., 1984, 130, C1 CrossRef CAS.
- Y. Zou, W. Priebe, Y.-H. Ling and R. Perez-Soler, Cancer Chemother. Pharmacol., 1993, 32, 190 CrossRef CAS PubMed.
- W. C. M. Dempke, R. Zielinski, C. Winkler, S. Silberman, S. Reuther and W. Priebe, Eur. J. Cancer, 2023, 185, 94 CrossRef CAS PubMed.
- L. Gil, A. Wierzbowska, T. Wróbel, E. Lech-Maranda, S. Milczarek, B. Machalinski, C. C. Abbate, E. Wasyl, R. Shepard and J. P. Waymack, Br. J. Cancer, 2023, 6, 613 Search PubMed.
- B. Nicholson, G. K. Lloyd, B. R. Miller, M. A. Palladino, Y. Kiso, Y. Hayashi and S. T. C. Neuteboom, Anti-Cancer Drugs, 2006, 17, 25 CrossRef CAS PubMed.
- K. Kanoh, S. Kohno, T. Asari, T. Harada, J. Katada, M. Muramatsu, H. Kawashima, H. Sekiya and I. Uno, Bioorg. Med. Chem. Lett., 1997, 7, 2847 CrossRef CAS.
- K. Kanoh, S. Kohno, J. Katada, J. Takahashi and I. Uno, J. Antibiot., 1999, 52, 134 CrossRef CAS PubMed.
- Y. Yamazaki, K. Kohno, H. Yasui, Y. Kiso, M. Akamatsu, B. Nicholson, G. Deyanat-Yazdi, S. Neuteboom, B. Potts, G. K. Lloyd and Y. Hayashi, ChemBioChem, 2008, 9, 3074 CrossRef CAS PubMed.
- G. La Sala, N. Olieric, A. Sharma, F. Viti, F. de Asis Balaguer Perez, L. Huang, J. R. Tonra, G. K. Lloyd, S. Decherchi, J. F. Díaz, M. O. Steinmetz and A. Cavalli, Chem, 2019, 5, 2969 CAS.
- B. Han, T. Feinstein, Y. Shi, G. Chen, Y. Yao, C. Hu, J. Shi, J. Feng, H. Wu, Y. Cheng, Q.-s. Guo, Z. Jie, F. Ye, Y. Zhang, Z. Liu, W. Mao, L. Zhang, J. Lu, J. Zhao, L. Bazhenova, J. Ruiz, G. H. Kloecker, K. R. Sujith, I. A. Oliff, M. Wong, B. Liu, Y. Wu, L. Huang, Y. Sun, B. Han, T. Feinstein, Y. Shi, G. Cheng, Y. Yao, C. Hu, J. Shi, J. Feng, H. Wu, Y. Cheng, Q.-s. Guo, Z. Jie, F. Ye, Y. Zhang, Z. Liu, W. Mao, L. Zhang, J. Lu, J. Zhao, L. Bazhenova, J. Ruiz, G. Kloecker, R. S. Kalmadi, I. Oliff, M. Wong, B. Liu, Y. Wu, L. Huang, Y. Sun, F. Luo, J.-y. Zhou, H. Pan, H. Wang, C. Liu, L. Cao, C. H. Huang, E. Thara, X. Li, D. Jiang, R. B. Mowat, J. S. Hrom, A. Nagrial, V. Jain, N. A. Karim, L. V. Shunyakov, A. Kiberu, R. Jennens, K. Lloyd, Z. Wang, L. Du and H. Li, Lancet Respir. Med., 2024, 12, 775 CrossRef CAS PubMed.
- D. W. Blayney, M. Chang, L. Huang and R. Mohanlal, Blood, 2021, 138, 2056 CrossRef.
- P. Cornelius, B. A. Mayes, J. S. Petersen, D. J. Turnquist, P. J. Dufour, A. J. Dannenberg, J. M. Shanahan and B. J. Carver, Mol. Cancer Ther., 2024, 23, 595 CrossRef CAS PubMed.
- F. R. Hanson and T. E. Eble, J. Bacteriol., 1949, 58, 527 CrossRef CAS PubMed.
- M. C. McCowen, M. E. Callender and J. F. Lawlis, Science, 1951, 113, 202 CrossRef CAS PubMed.
- D. S. Tarbell, R. M. Carman, D. D. Chapman, S. E. Cremer, A. D. Cross, K. R. Huffman, M. Kunstmann, N. J. McCorkindale, J. G. McNally, A. Rosowsky, F. H. L. Varino and R. L. West, J. Am. Chem. Soc., 1961, 83, 3096 CrossRef CAS.
- EMA, Fumagillin. EU/3/01/081 - orphan designation for treatment of diarrhoea associated with intestinal microsporidial infection, https://www.ema.europa.eu/en/medicines/human/orphan-designations/eu-3-01-081, accessed 7 April 2025.
- A. Maillard, A. Scemla, B. Laffy, N. Mahloul and J.-M. Molina, J. Antimicrob. Chemother., 2021, 76, 487 CrossRef CAS PubMed.
- B. C. Das, P. Chokkalingam, M. A. Shareef, S. Shukla, S. Das, M. Saito and L. M. Weiss, J. Eukaryot. Microbiol., 2024, 71, e13036 CrossRef CAS PubMed.
- X. Guruceaga, U. Perez-Cuesta, A. Abad-Diaz de Cerio, O. Gonzalez, R. M. Alonso, F. L. Hernando, A. Ramirez-Garcia and A. Rementeria, Toxins, 2020, 12, 7 CrossRef CAS PubMed.
- M. Peirson and S. F. Pernal, Insects, 2024, 15, 29 CrossRef PubMed.
- A.-G. Moulun, French hospitals manufacture discontinued drug to save liveshttps://www.medscape.com/viewarticle/977962, accessed 7 April 2025.
- W. Z. Antkowiak and W. P. Gessner, Tetrahedron Lett., 1979, 20, 1931 CrossRef.
- M. J. Lyons, C. Ehrhardt and J. J. Walsh, J. Nat. Prod., 2023, 86, 1620 CrossRef CAS PubMed.
- A. Herrmann, H. Hedman, J. Rosén, D. Jansson, B. Haraldsson and K.-E. Hellenäs, J. Nat. Prod., 2012, 75, 1690 CrossRef CAS PubMed.
- H. Hedman, J. Holmdahl, J. Mölne, K. Ebefors, B. Haraldsson and J. Nyström, BMC Nephrol., 2017, 18, 121 CrossRef CAS PubMed.
- L. Buvall, H. Hedman, A. Khramova, D. Najar, L. Bergwall, K. Ebefors, C. Sihlbom, S. Lundstam, A. Herrmann, H. Wallentin, E. Roos, U. A. Nilsson, M. Johansson, J. Törnell, B. Haraldsson and J. Nyström, Oncotarget, 2017, 8, 91085 CrossRef PubMed.
- S. Lundstam, B. Haraldsson, J. Nystrom and J. Yachnin, J. Clin. Oncol., 2024, 42, TPS486 CrossRef.
- T. C. McMorris, M. J. Kelner, W. Wang, J. Yu, L. A. Estes and R. Taetle, J. Nat. Prod., 1996, 59, 896 CrossRef CAS PubMed.
- T. C. McMorris, Bioorg. Med. Chem., 1999, 7, 881 CrossRef CAS PubMed.
- M. Anchel, A. Hervey and W. J. Robbins, Proc. Natl. Acad. Sci. U. S. A., 1950, 36, 300 CrossRef CAS PubMed.
- M. Tanasova and S. J. Sturla, Chem. Rev., 2012, 112, 3578 CrossRef CAS PubMed.
- T. C. McMorris and M. Anchel, J. Am. Chem. Soc., 1965, 87, 1594 CrossRef CAS PubMed.
- P. Le, M. B. Nodwell, J. Eirich and S. A. Sieber, Chem.–Eur. J., 2019, 25, 12644 CrossRef CAS PubMed.
- L. Casimir, S. Zimmer, F. Racine-Brassard, P.-É. Jacques and A. Maréchal, DNA Repair, 2023, 122, 103433 CrossRef CAS PubMed.
- R. Williams, Expert Opin. Invest. Drugs, 2013, 22, 1627 CrossRef CAS PubMed.
- M. D. Staake, A. Kashinatham, T. C. McMorris, L. A. Estes and M. J. Kelner, Bioorg. Med. Chem. Lett., 2016, 26, 1836 CrossRef CAS PubMed.
- A. Kulkarni, J. Zhou, N. Biyani, U. Kathad, P. P. Banerjee, S. Srivastava, Z. Prucsi, K. Solarczyk, K. Bhatia, R. B. Ewesuedo and P. Sharma, Cancer Res. Commun., 2024, 4, 1199 CrossRef CAS PubMed.
- D. Restifo, J. R. McDermott, D. Cvetkovic, T. Dos Santos, C. Ogier, A. Surumbayeva, E. A. Handorf, C. Schimke, C. Ma, K. Q. Cai, A. J. Olszanski, U. Kathad, K. Bhatia, P. Sharma, A. Kulkarni and I. Astsaturov, Mol. Cancer Ther., 2023, 22, 1182 CrossRef CAS PubMed.
- B. Lal, A. Kulkarni, J. McDermott, R. Rais, J. Alt, Y. Wu, H. Lopez-Bertoni, S. Sall, U. Kathad, J. Zhou, B. S. Slusher, K. Bhatia and J. Laterra, Clin. Cancer Res., 2023, 29, 4209 CrossRef CAS PubMed.
- G. J. Tobin, S. T. Blumberg and A. D. Malakhov, US Pat., US11739043B2, 2021 Search PubMed.
- J. Zhou, D. Sturtevant, C. Love, A. Kulkarni, N. Biyani, U. Kathad, E. Thacker, S. Dave and K. Bhatia, Oncotarget, 2023, 14, 597 CrossRef PubMed.
- J. Zhou, A. Kulkarni, C. Love, L. Happ, D. Sturtevant, N. Biyani, R. L. Peru y Colón De Portugal, K. Bhatia and S. S. Dave, Blood, 2022, 140, 4940 CrossRef.
- T.-H. Lee, C.-K. Lee, W.-L. Tsou, S.-Y. Liu, M.-T. Kuo and W.-C. Wen, Planta Med., 2007, 73, 1412 CrossRef CAS PubMed.
- M. T. Villaume, E. Sella, G. Saul, R. M. Borzilleri, J. Fargnoli, K. A. Johnston, H. Zhang, M. P. Fereshteh, T. G. M. Dhar and P. S. Baran, ACS Cent. Sci., 2016, 2, 27 CrossRef CAS PubMed.
- B.-B. Zhang, P.-F. Hu, J. Huang, Y.-D. Hu, L. Chen and G.-R. Xu, J. Agric. Food Chem., 2017, 65, 10395 CrossRef CAS PubMed.
- Y. Kuang, B. Li, Z. Wang, X. Qiao and M. Ye, Nat. Prod. Rep., 2021, 38, 83 RSC.
- H.-C. Lin, M.-H. Lin, J.-H. Liao, T.-H. Wu, T.-H. Lee, F.-L. Mi, C.-H. Wu, K.-C. Chen, C.-H. Cheng and C.-W. Lin, J. Agric. Food Chem., 2017, 65, 51 CrossRef CAS PubMed.
- V. B. Kumar, T.-C. Yuan, J.-W. Liou, C.-J. Yang, P.-J. Sung and C.-F. Weng, Mutat. Res., 2011, 707, 42 CrossRef CAS PubMed.
- K. J. S. Kumar, F.-H. Chu, H.-W. Hsieh, J.-W. Liao, W.-H. Li, J. C.-C. Lin, J.-F. Shaw and S.-Y. Wang, J. Ethnopharmacol., 2011, 136, 168 CrossRef CAS PubMed.
- C.-L. Ho, J.-L. Wang, C.-C. Lee, H.-Y. Cheng, W.-C. Wen, H. H.-Y. Cheng and M. C.-M. Chen, Biomed. Pharmacother., 2014, 68, 1007 CrossRef CAS PubMed.
- GoldenBiotech, ASCO GI 2024: Golden Biotech's antroquinonol shows significantly prolonged survival in untreated metastatic pancreatic cancer patients (Yahoo Finance) (Press Release 19 January 2024), https://www.goldenbiotech.com/en/news.php?act=view%26id=34, accessed 7 April 2025.
- M. Serpi, V. Ferrari, C. McGuigan, E. Ghazaly and C. Pepper, J. Med. Chem., 2022, 65, 15789 CrossRef CAS PubMed.
- K. G. Cunningham, W. Manson, F. S. Spring and S. A. Hutchinson, Nature, 1950, 166, 949 CrossRef CAS PubMed.
- E. A. Kaczka, N. R. Trenner, B. Arison, R. W. Walker and K. Folkers, Biochem. Biophys. Res. Commun., 1964, 14, 456 CrossRef CAS PubMed.
- R. He and W. Zhou, Mol. Med. Rep., 2024, 30, 161 CrossRef CAS PubMed.
- J. Zeng, Y. Zhou, M. Lyu, X. Huang, M. Xie, M. Huang, B.-X. Chen and T. Wei, Biotechnol. Adv., 2024, 74, 108396 CrossRef CAS PubMed.
- H. Schwenzer, E. De Zan, M. Elshani, R. van Stiphout, M. Kudsy, J. Morris, V. Ferrari, I. H. Um, J. Chettle, F. Kazmi, L. Campo, A. Easton, S. Nijman, M. Serpi, S. Symeonides, R. Plummer, D. J. Harrison, G. Bond and S. P. Blagden, Clin. Cancer Res., 2021, 27, 6500 CrossRef CAS PubMed.
- A. M. Shahid, I. H. Um, M. Elshani, Y. Zhang and D. J. Harrison, PLoS One, 2022, 17, e0278209 CrossRef CAS PubMed.
- Y. Shen, R. Boivin, N. Yoneda, H. Du, S. Schiller, T. Matsushima, M. Goto, H. Shirota, F. Gusovsky, C. Lemelin, Y. Jiang, Z. Zhang, R. Pelletier, M. Ikemori-Kawada, Y. Kawakami, A. Inoue, M. Schnaderbeck and Y. Wang, Bioorg. Med. Chem. Lett., 2010, 20, 3155 CrossRef CAS PubMed.
- G. A. Ellestad, F. M. Lovell, N. A. Perkinson, R. T. Hargreaves and W. J. McGahren, J. Org. Chem., 1978, 43, 2339 CrossRef CAS.
- M. Goto, J. Chow, K. Muramoto, K.-i. Chiba, S. Yamamoto, M. Fujita, H. Obaishi, K. Tai, Y. Mizui, I. Tanaka, D. Young, H. Yang, Y. J. Wang, H. Shirota and F. Gusovsky, J. Pharmacol. Exp. Ther., 2009, 331, 485 CrossRef CAS PubMed.
- W. Zhang, G. Borthakur, C. Gao, Y. Chen, H. Mu, V. R. Ruvolo, K. Nomoto, N. Zhao, M. Konopleva and M. Andreeff, Cancer Res., 2016, 76, 1528 CrossRef CAS PubMed.
- S. Kuttikrishnan, K. S. Prabhu, A. H. Al Sharie, Y. O. Al Zu'bi, F. Q. Alali, N. H. Oberlies, A. Ahmad, T. El-Elimat and S. Uddin, Drug Discovery Today, 2022, 27, 547 CrossRef CAS PubMed.
- R. Tibes, M. J. Borad, C. E. Dutcus, L. Reyderman, K. Feit, A. Eisen, D. A. Verbel and D. D. Von Hoff, Br. J. Cancer, 2018, 118, 1580 CrossRef CAS PubMed.
- S. Kawano, K. Ito, K. Yahata, K. Kira, T. Abe, T. Akagi, M. Asano, K. Iso, Y. Sato, F. Matsuura, I. Ohashi, Y. Matsumoto, M. Isomura, T. Sasaki, T. Fukuyama, Y. Miyashita, Y. Kaburagi, A. Yokoi, O. Asano, T. Owa and Y. Kishi, Sci. Rep., 2019, 9, 8656 CrossRef PubMed.
- T. Doi, N. Matsubara, Y. Naito, Y. Kuboki, K. Harano, M. Ono, T. Urasaki, A. Ohmoto, T. Kawanai, T. Hisai, H. Ikezawa, S. Shiba, K. Ito, T. Semba, O. Asano and S. Takahashi, Cancer, 2023, 129, 2348 CrossRef CAS PubMed.
- Y. Kaburagi, K. Kira, K. Yahata, K. Iso, Y. Sato, F. Matsuura, I. Ohashi, Y. Matsumoto, M. Isomura, T. Sasaki, T. Fukuyama, Y. Miyashita, H. Azuma, D. Iida, T. Ishida, W. Itano, M. Matsuda, M. Matsukura, N. Murai, S. Nagao, M. Seki, A. Yamamoto, Y. Yamamoto, N. Yoneda, Y. Watanabe, A. Kamada, A. Kayano, K. Tagami, O. Asano, T. Owa and Y. Kishi, Org. Lett., 2024, 26, 2837 CrossRef CAS PubMed.
- T. Sasaki, K. Yahata, M. Isomura, I. Ohashi, T. Fukuyama, Y. Miyashita, Y. Watanabe, N. Murai, M. Matsuda, A. Kamada, Y. Kaburagi, K. Kira, K. Iso, Y. Sato, F. Matsuura, Y. Matsumoto, H. Azuma, D. Iida, T. Ishida, W. Itano, S. Nagao, M. Seki, A. Yamamoto, Y. Yamamoto, N. Yoneda, M. Matsukura, O. Asano, A. Kayano, K. Tagami, T. Owa and Y. Kishi, Org. Process Res. Dev., 2024, 28, 2077 CrossRef CAS.
- D. Uemura, K. Takahashi, T. Yamamoto, C. Katayama, J. Tanaka, Y. Okumura and Y. Hirata, J. Am. Chem. Soc., 1985, 107, 4796 CrossRef CAS.
- Y. Hirata and D. Uemura, Pure Appl. Chem., 1986, 58, 701 CrossRef CAS.
- G. R. Pettit, R. Tan, F. Gao, M. D. Williams, D. L. Doubek, M. R. Boyd, J. M. Schmidt, J. C. Chapuis and E. Hamel, J. Org. Chem., 1993, 58, 2538 CrossRef CAS.
- G. R. Pettit, Y. Ichihara, G. Wurzel, M. D. Williams, J. M. Schmidt and J.-C. Chapuis, J. Chem. Soc., Chem. Commun., 1995, 383, 10.1039/C39950000383.
- M. Litaudon, J. B. Hart, J. W. Blunt, R. J. Lake and M. H. G. Munro, Tetrahedron Lett., 1994, 35, 9435 CrossRef CAS.
- S. J. H. Hickford, J. W. Blunt and M. H. G. Munro, Bioorg. Med. Chem., 2009, 17, 2199 CrossRef CAS PubMed.
- M. H. G. Munro, J. W. Blunt, E. J. Dumdei, S. J. H. Hickford, R. E. Lill, S. Li, C. N. Battershill and A. R. Duckworth, J. Biotechnol., 1999, 70, 15 CrossRef CAS PubMed.
- J. B. Hart, R. E. Lill, S. J. H. Hickford, J. W. Blunt and M. H. G. Munro, in Drugs from the sea, ed. N. Fusetani, Karger, Basel, 2000, p. 134 Search PubMed.
- W. Zheng, B. M. Seletsky, M. H. Palme, P. J. Lydon, L. A. Singer, C. E. Chase, C. A. Lemelin, Y. Shen, H. Davis, L. Tremblay, M. J. Towle, K. A. Salvato, B. F. Wels, K. K. Aalfs, Y. Kishi, B. A. Littlefield and M. J. Yu, Bioorg. Med. Chem. Lett., 2004, 14, 5551 CrossRef CAS PubMed.
- T. D. Aicher, K. R. Buszek, F. G. Fang, C. J. Forsyth, S. H. Jung, Y. Kishi, M. C. Matelich, P. M. Scola, D. M. Spero and S. K. Yoon, J. Am. Chem. Soc., 1992, 114, 3162 CrossRef CAS.
- M. J. Yu, W. Zheng, B. M. Seletsky, B. A. Littlefield and Y. Kishi, in Annu. Rep. Med. Chem., ed. J. E. Macor, Academic Press, Amsterdam, 2011, vol. 46, ch. 14, p. 227 Search PubMed.
- M. J. Yu, W. Zheng and B. M. Seletsky, Nat. Prod. Rep., 2013, 30, 1158 RSC.
- P. G. Cruz, R. Fernández, R. Rodríguez-Acebes, M. Martínez-Díez, G. Santamaría-Núñez, M. Pérez and C. Cuevas, Mar. Drugs, 2024, 22, 339 CrossRef CAS PubMed.
- D. Lucena-Agell, M. J. Guillén, R. Matesanz, B. Álvarez-Bernad, R. Hortigüela, P. Avilés, M. Martínez-Díez, G. Santamaría-Núñez, J. Contreras, I. Plaza-Menacho, J. F. Giménez-Abián, M. A. Oliva, C. Cuevas and J. F. Díaz, J. Med. Chem., 2024, 67, 2619 CrossRef CAS PubMed.
- K. L. Rinehart, T. G. Holt, N. L. Fregeau, J. G. Stroh, P. A. Keifer, F. Sun, L. H. Li and D. G. Martin, J. Org. Chem., 1990, 55, 4512 CrossRef CAS.
- M. M. Schofield, S. Jain, D. Porat, G. J. Dick and D. H. Sherman, Environ. Microbiol., 2015, 17, 3964 CrossRef CAS PubMed.
- M. Cigrang, J. Obid, M. Nogaret, L. Seno, T. Ye, G. Davidson, P. Catez, P. Berico, C. Capelli, C. Marechal, A. Zachayus, C. Elly, T.-K. Li, E. Compe, P. Avilés, I. Davidson, J.-M. Egly, C. Cuevas and F. Coin, bioRxiv, 2024, preprint, 2024.03.26.586754, DOI:10.1101/2024.03.26.586754.
- I. C. Salaroglio, P. Aviles, J. Kopecka, A. Merlini, F. Napoli, L. Righi, S. Novello, H. Sullivan, C. Cuevas, G. V. Scagliotti and C. Riganti, J. Exp. Clin. Cancer Res., 2024, 43, 327 CrossRef CAS PubMed.
- D. J. Newman, J. Nat. Prod., 2021, 84, 917 CrossRef CAS.
- W. Dong, W. Wang and C. Cao, ChemMedChem, 2024, 19, e202400109 CrossRef CAS PubMed.
- L. Conilh, L. Sadilkova, W. Viricel and C. Dumontet, J. Hematol. Oncol., 2023, 16, 3 CrossRef CAS PubMed.
- K. C. Nicolaou and S. Rigol, Angew. Chem., Int. Ed., 2019, 58, 11206 CrossRef CAS PubMed.
- X. Jiang, W. N. Nik Nabil, Y. Ze, R. Dai, Z. Xi and H. Xu, Phytother. Res., 2025, 39, 789 CrossRef CAS PubMed.
- H. Maecker, V. Jonnalagadda, S. Bhakta, V. Jammalamadaka and J. R. Junutula, mAbs, 2023, 15, 2229101 CrossRef.
- L. Shen, X. Sun, Z. Chen, Y. Guo, Z. Shen, Y. Song, W. Xin, H. Ding, X. Ma, W. Xu, W. Zhou, J. Che, L. Tan, L. Chen, S. Chen, X. Dong, L. Fang and F. Zhu, Nucleic Acids Res., 2023, 52, D1097 CrossRef PubMed.
- P. Flynn, S. Suryaprakash, D. Grossman, V. Panier and J. Wu, Nat. Rev. Drug Discovery, 2024, 23, 577 CrossRef CAS PubMed.
- NJ Bio, Recent Advances in ADCs (September 2024), https://njbio.com/antibody-drug-conjugates/, accessed 14 March 2025.
- D. R. Camidge, D. Morgensztern, R. S. Heist, M. Barve, E. Vokes, J. W. Goldman, D. S. Hong, T. M. Bauer, J. H. Strickler, E. Angevin, M. Motwani, A. Parikh, Z. Sun, B. A. Bach, J. Wu, P. B. Komarnitsky and K. Kelly, Clin. Cancer Res., 2021, 27, 5781 CrossRef CAS PubMed.
- J. M. Lambert and R. V. J. Chari, J. Med. Chem., 2014, 57, 6949 CrossRef CAS PubMed.
- K. Koyama, H. Ishikawa, M. Abe, Y. Shiose, S. Ueno, Y. Qiu, K. Nakamaru and M. Murakami, PLoS One, 2022, 17, e0267027 CrossRef CAS.
- X. Cheng, J. Li, K. Tanaka, U. Majumder, A. Z. Milinichik, A. C. Verdi, C. J. Maddage, K. A. Rybinski, S. Fernando, D. Fernando, M. Kuc, K. Furuuchi, F. Fang, T. Uenaka, L. Grasso and E. F. Albone, Mol. Cancer Ther., 2018, 17, 2665 CrossRef CAS PubMed.
- E. S. Zimmerman, T. H. Heibeck, A. Gill, X. Li, C. J. Murray, M. R. Madlansacay, C. Tran, N. T. Uter, G. Yin, P. J. Rivers, A. Y. Yam, W. D. Wang, A. R. Steiner, S. U. Bajad, K. Penta, W. Yang, T. J. Hallam, C. D. Thanos and A. K. Sato, Bioconjugate Chem., 2014, 25, 351 CrossRef CAS PubMed.
- X. Li, S. Zhou, C. L. Abrahams, S. Krimm, J. Smith, K. Bajjuri, H. T. Stephenson, R. Henningsen, J. Hanson, T. H. Heibeck, D. Calarese, C. Tran, G. Yin, R. L. Stafford, A. Y. Yam, T. Kline, V. I. De Almeida, A. K. Sato, M. Lupher Jr, K. Bedard and T. J. Hallam, Mol. Cancer Ther., 2023, 22, 155 CrossRef CAS PubMed.
- B. C. Murray, M. T. Peterson and R. A. Fecik, Nat. Prod. Rep., 2015, 32, 654 RSC.
- X. Cai, M. Cao, Q. Yang, X. Yu, X.-H. Fen and R. Y. Zhao, Am. J. Cancer Res., 2024, 14, 5752 CrossRef CAS.
- H. Guo, H. Xie, Y. Huang, J. Jia, X. Kong, Q. Yang, S. Gai, W. Li, L. Bai, L. Zhang, X. Chen, Z. Ye, H. Ye, L. Zhao, Y. Xu, Y. Du, X. Zhang, M. Chen, X. Zhou and R. Y. Zhao, Front. Pharmacol., 2025, 16, 1532104 CrossRef CAS PubMed.
- V. Figueroa-Vazquez, J. Ko, C. Breunig, A. Baumann, N. Giesen, A. Pálfi, C. Müller, C. Lutz, T. Hechler, M. Kulke, C. Müller-Tidow, A. Krämer, H. Goldschmidt, A. Pahl and M. S. Raab, Mol. Cancer Ther., 2021, 20, 367 CrossRef CAS PubMed.
- D. Ning, J. Xue, X. Lou, R. Shao, Y. Liu and G. Chen, Arch. Toxicol., 2024, 98, 1705 CrossRef CAS PubMed.
- J. Vetter, Molecules, 2023, 28, 5932 CrossRef CAS PubMed.
- M. J. Martín López, R. Rodríguez Acebes, P. G. Cruz López, A. M. Francesch Solloso and M. d. C. Cuevas Marchante, World Pat., WO2020127194, 2020 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |