 Open Access Article
Open Access ArticleThe glass transition width and its dependence on fragility, nonexponentiality and nonlinearity
Jiří
Málek

Department of Physical Chemistry, Faculty of Chemical Technology, University of Pardubice, Studentská 573, Pardubice 532 10, Czech Republic. E-mail: jiri.malek@upce.cz
First published on 17th March 2026
Abstract
This paper presents a comprehensive and critical analysis of the glass transition width 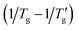 or the reduced width
or the reduced width 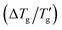 observed in simulated differential scanning calorimetry (DSC) heating and cooling scans. The study employs the Tool-Narayanaswamy-Moynihan model (TNM) for 24 diverse materials, encompassing inorganic glasses, organic polymers and molecular glassy systems. The analysis reveals an important novel finding. The width (or the reduced width) of the glass transition cooling scan is shown to be inversely proportional to the activation energy (h*/R), or fragility index (m), as well as the sum the non-exponentiality β and nonlinearity x parameters, following the relationship: [(h*/R) × (β + x)]−1 or [m × (β + x)]−1. With precise determinations of Tg and
observed in simulated differential scanning calorimetry (DSC) heating and cooling scans. The study employs the Tool-Narayanaswamy-Moynihan model (TNM) for 24 diverse materials, encompassing inorganic glasses, organic polymers and molecular glassy systems. The analysis reveals an important novel finding. The width (or the reduced width) of the glass transition cooling scan is shown to be inversely proportional to the activation energy (h*/R), or fragility index (m), as well as the sum the non-exponentiality β and nonlinearity x parameters, following the relationship: [(h*/R) × (β + x)]−1 or [m × (β + x)]−1. With precise determinations of Tg and 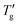 , the estimated sum of (β + x) achieves an accuracy comparable to Hutchinson's established peak shift method.
, the estimated sum of (β + x) achieves an accuracy comparable to Hutchinson's established peak shift method.
1. Introduction
The glass transition, defined as the continuous transformation of a supercooled liquid into a glassy state upon cooling to sufficiently low temperatures, is an essential feature of all glassy materials, including organic polymers, oxides, chalcogenides, metallic alloys, and molecular systems. This phenomenon is primarily kinetic; nonetheless, it exhibits distinct thermodynamic characteristics – such as a step change in heat capacity or thermal expansion coefficient – resulting from the loss of configurational degrees of freedom as molecular motion is arrested.Over 30 years ago, Moynihan's work1 established a correlation between the width of the glass transition region (as measured by Differential Scanning Calorimetry, DSC) and the activation energy (h*/R) for the structural relaxation of high-Tg inorganic glasses. Moynihan defined a dimensionless parameter, C, as a constant specific to certain inorganic glasses.1,2 This parameter is derived from the reciprocal temperatures at the onset (1/Tg) and end 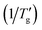 of the glass transition on a DSC thermogram:
of the glass transition on a DSC thermogram:
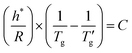 | (1) |
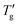 for a given glass remain invariant, within experimental error, as long as the cooling/heating rate ratio stays within the range of 0.2 to 5. This correlation was later extended to molecular glasses by combining DSC and dielectric relaxation data and utilizing the F1/2 fragilities.3
for a given glass remain invariant, within experimental error, as long as the cooling/heating rate ratio stays within the range of 0.2 to 5. This correlation was later extended to molecular glasses by combining DSC and dielectric relaxation data and utilizing the F1/2 fragilities.3
However, this extension of the Moynihan correlation has been challenged. Johari et al.4 used simulations to show that a simple relationship between the DSC endotherm width and the non-Arrhenius temperature dependence of a liquid's kinetic property requires explicitly including the distribution of relaxation times. The method's general applicability was further questioned by Hancock et al.,5 who tested it on various pharmaceutical glass-forming materials. They concluded that the conditions required for eqn (1) to be universally applicable with a constant C are not met for a wide range of glassy materials.
Pikal et al.6 later provided a detailed analysis of experimental data for (poly)vinyl pyrrolidone, sucrose, and trehalose. Their approach, which combined various experimental procedures combined with simulations of theoretical curves, was equivalent to the comprehensive Tool-Narayanaswamy-Moynihan (TNM) formalism. Within this work, Pikal et al. found that a modification of Moynihan's original equation provides a much better correlation for both their experimental and simulated data.6 This modified equation incorporates the stretched exponential parameter β (a measure of non-exponentiality) to more accurately describe the relaxation behaviour of these glassy materials. They reformulated eqn (1) to account for this non-exponentiality:
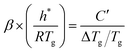 | (2) |
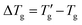 and C′ is a constant.
and C′ is a constant.
Building on this, Chen et al.7 reported that the reduced glass transition width, ΔTg/Tg, is influenced by the fragility index, m = (h*/RTg)/ln(10), and the stretched exponential parameter, β. In contrast, they found no significant relationship between the nonlinearity parameter (x of the TNM model) and ΔTg/Tg. More recently, Bogdanova and Kocherbitov8 applied this formalism to a sucrose–water system and arrived at very similar conclusions, corroborating the findings of Chen et al. This convergence suggests that the relationship between fragility, non-exponentiality, and the width of the glass transition may hold for a broader class of glassy materials.
In his seminal papers,9,10 Donth utilized the glass transition width determined from DSC thermograms even earlier than the aforementioned studies.1–8 By assuming mean temperature fluctuation within cooperatively rearranging regions (CRRs), Donth proposed a method to estimate average size of these thermodynamic subsystems based on the shape of thermal relaxation spectrum. This approach has since been applied to the analysis of conventional, modulated, and fast DSC data for polymers, a-Se,11 and metallic glasses.12 Recently, Schawe et al.,13 compared the characteristic length of dynamic heterogeneities across a wide range of glass-formers. Their work discussed the correlation between chemical structure, the size of dynamic heterogeneities, and the macroscopic kinetics of the glass transition, revealing nearly universal behaviour among studied systems.
Previous studies1–8 were restricted to analyzing heating DSC curves, typically measured or simulated immediately after a prior cooling from equilibrium supercooled liquid well above the glass transition. This paper provides a comprehensive and critical analysis of the glass transition width within a DSC thermal cycle, utilizing simulated cooling and subsequent heating curves based on TNM parameters for 24 diverse materials, including inorganic glasses to organic polymers and some molecular systems. The results indicate that the width of the cooling DSC curve exhibits an inverse proportionality to the material's fragility and to the combined sum of the non-exponentiality (β) and nonlinearity (x) parameters.
2. The TNM model
The Tool-Narayanaswamy-Moynihan (TNM) formalism, an important model for structural relaxation in glassy materials, integrates three fundamental concepts. The first concept, introduced by Tool,14,15 posits that relaxation time depends on both temperature T and the material's actual structure, represented by the fictive temperature Tf. The fictive temperature essentially defines the temperature at which an equilibrium supercooled liquid would possess the same structure as the non-equilibrium glass. It is defined by an empirical equation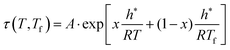 | (3a) |
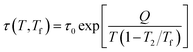 | (3b) |
The second essential concept was introduced by Narayanaswamy.18 According to his model, the structural relaxation should be linear in terms of the material (or reduced) time defined by
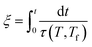 | (4) |
The third fundamental concept integrates Tool's fictive temperature with the material-time concept. Mazurin et al.22 and Moynihan et al.,23,24 employed the stretched exponential function, ϕ, to account for the nonexponentiality of the structural relaxation, as shown in eqn (5).
| ϕ = exp(−ξβ) | (5) |
3. Simulation of DSC curves in the glass transition range
The structural relaxation is usually simulated in terms of evolution of the fictive temperature for a well-defined time-temperature protocol. This protocol captures the entire thermal history of a glassy material from the very first moment of its departure from metastable equilibrium supercooled liquid well above the glass transition. The structural relaxation can be described by combining the material-time concept (eqn (4)), and the Boltzmann superposition principle for any given time-temperature protocol.24For practical implementation, the protocol is often discretized into several consecutive steps, which may involve a heating/cooling ramp or isothermal annealing. For the TNM model, the evolution of the fictive temperature during continuous heating or cooling at a constant rate qk = dT/dt (negative for cooling) then can be expressed22 by eqn (6a) and (6b):
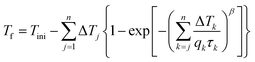 | (6a) |
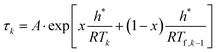 | (6b) |
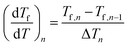 | (7) |
Fig. 1b shows a thermal cycle, illustrating the simulated cooling (#1) and subsequent heating (#2) curves from eqn (6) and (7) using an identical scanning rate q = ±10 K min−1. The cooling (dTf/dT) curve exhibits a simple sigmoidal shape, contrasting sharply with the more complicated heating curve, which features kinetic phenomena such as a minimum (undershoot) and maximum (overshoot). Fig. 1a clearly indicates that the limiting fictive temperature Tlim is identical for both scans – a characteristic result for this type of thermal cycle. Tlim is situated close to the inflectional point of the cooling curve and the extrapolated onset of the heating curve (Tg).
 | ||
| Fig. 1 The fictive temperature Tf and its derivative dTf/dT, calculated using eqn (6) and (7) for the TNM parameters: h*/R = 51.2 kK, ln(A/s) = 90.8, x = 0.96, β = 0.84. The temperature program used was Tini = 310 °C, qc = −10 K min−1, qh = +10 K min−1. Open triangles mark inflection points. Full circles and full squares indicate the extrapolated inflectional tangent intersection with dTf/dT = 0 and dTf/dT = 1 lines, respectively. Inflectional tangents for heating data are represented by dashed lines, while those for cooling data use solid line. Arrow indicates limiting fictive temperature. | ||
The derivative dTf/dT in fact corresponds to the heat capacity CNp, which is equivalent to a DSC curve scaled between 0 and 1:
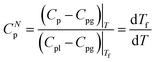 | (8) |
To demarcate the lower end of the glass transition region for DSC heating curve Moynihan1 used “extrapolated temperature of onset of rapid rise of the Cpvs. T curve, Tg”, as shown in Fig. 1b. To demarcate upper end of the glass transition for heating DSC data “the extrapolated temperature of completion of the overshoot, 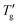 was used.” However, this manual approach is inadequate for the precise numerical analysis of simulated DSC data. To overcome this limitation, we propose a refined numerical procedure. Step 1: the inflection points of the simulated DSC heating data are first determined numerically; step 2: tangents are then calculated at these inflection points using linear regression, based on 10 to 20 adjacent data points to ensure a correlation coefficient greater than 0.9999; step 3: the intersections of these tangents define the characteristic temperatures: Tg is the intersection with dTf/dT = 0, and
was used.” However, this manual approach is inadequate for the precise numerical analysis of simulated DSC data. To overcome this limitation, we propose a refined numerical procedure. Step 1: the inflection points of the simulated DSC heating data are first determined numerically; step 2: tangents are then calculated at these inflection points using linear regression, based on 10 to 20 adjacent data points to ensure a correlation coefficient greater than 0.9999; step 3: the intersections of these tangents define the characteristic temperatures: Tg is the intersection with dTf/dT = 0, and 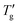 is the intersection with dTf/dT = 1. The dashed lines in Fig. 1b illustrate this; the same procedure was similarly employed for the analysis of simulated DSC cooling data, which is represented by the solid line in Fig. 1b.
is the intersection with dTf/dT = 1. The dashed lines in Fig. 1b illustrate this; the same procedure was similarly employed for the analysis of simulated DSC cooling data, which is represented by the solid line in Fig. 1b.
4. Results and discussion
Table 1 presents a selection of previously reported Tool-Narayanaswamy-Moynihan (TNM) parameter sets for 24 diverse materials, encompassing a wide range of properties. The apparent activation energies of these materials span from 12 to 225 kK. Similarly, the nonlinearity parameter x varies from 0.10 to 0.96, and the non-exponentiality parameter β ranges from 0.23 to 0.86. This broad range ensures a comprehensive test of validity of the eqn (1) and (2) across different material types.| Material | h*/R [kK] | −ln(A/s) | x | β | Ref. |
|---|---|---|---|---|---|
| PVC | 225.0 | 622.0 | 0.10 | 0.23 | 17 |
| PMMA | 138.0 | 357.8 | 0.19 | 0.35 | 17 |
| PS bulk | 83.4 | 212.5 | 0.18 | 0.43 | 25 |
| ZBLA | 165.0 | 282.6 | 0.19 | 0.50 | 26 |
| Li2O·SiO2 | 111.8 | 150.8 | 0.27 | 0.48 | 27 |
| PC | 150.0 | 353.6 | 0.22 | 0.54 | 28 |
| Glycerol | 16.0 | 81.0 | 0.29 | 0.51 | 29 |
| EG gel | 12.0 | 75.5 | 0.46 | 0.39 | 26 |
| PVAc | 71.3 | 223.6 | 0.35 | 0.57 | 30 |
| PS | 126.6 | 334.7 | 0.29 | 0.69 | 31 |
| Se | 42.8 | 133.0 | 0.42 | 0.58 | 32 |
| TPD bulk | 109.5 | 321.0 | 0.64 | 0.37 | 33 |
| B2O3 | 45.0 | 75.6 | 0.40 | 0.65 | 24 |
| LiCl gel | 12.0 | 70.5 | 0.67 | 0.39 | 26 |
| ASAHI plate glass | 73.0 | 83.8 | 0.45 | 0.62 | 34 |
| As2S3 | 32.4 | 62.1 | 0.31 | 0.82 | 35 |
| Se70Te30 | 34.5 | 100.0 | 0.43 | 0.73 | 36 |
| As2Se3 | 41.0 | 85.5 | 0.49 | 0.67 | 37 |
| (GeSe2)30(Sb2Se3)70 | 62.1 | 120.0 | 0.52 | 0.67 | 38 |
| (GeS2)30(Sb2S3)70 | 60.2 | 113.0 | 0.57 | 0.72 | 39 |
| NBS711 | 45.0 | 57.4 | 0.65 | 0.65 | 40 |
| P-SK57 | 82.2 | 101.5 | 0.75 | 0.77 | 41 |
| (GeS2)50(Sb2S3)50 | 51.2 | 90.8 | 0.75 | 0.86 | 42 |
| (GeTe4)60(GaTe3)40 | 40.9 | 91.9 | 0.96 | 0.84 | 43 |
A complete DSC thermal cycle, – specifically, cooling scan #1 (−10 K min−1) from the equilibrium supercooled liquid at Tini to a temperature well below Tg, followed by subsequent heating scan #2 (+10 K min−1) back to Tini – was calculated using eqn (6) and (7) for all TNM parameter sets listed in Table 1 (see SI). These simulated curves are analyzed separately in the following sections.
4.1. Analysis of simulated DSC heating scans
Moynihan's proposed eqn (1) suggests that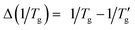 , derived from simulated DSC heating scans (+10 K min−1), multiplied by the reduced activation energy of structural relaxation h*/R, should equal a constant parameter, C. However, our analysis of 24 diverse materials from Table 1 demonstrates that C depends on the non-exponentiality parameter β (Fig. 2a), a finding previously observed by several authors.6–8 For the specific range of β the values of C approximate Moynihan's proposed constant (C ≅ 4.8).1
, derived from simulated DSC heating scans (+10 K min−1), multiplied by the reduced activation energy of structural relaxation h*/R, should equal a constant parameter, C. However, our analysis of 24 diverse materials from Table 1 demonstrates that C depends on the non-exponentiality parameter β (Fig. 2a), a finding previously observed by several authors.6–8 For the specific range of β the values of C approximate Moynihan's proposed constant (C ≅ 4.8).1
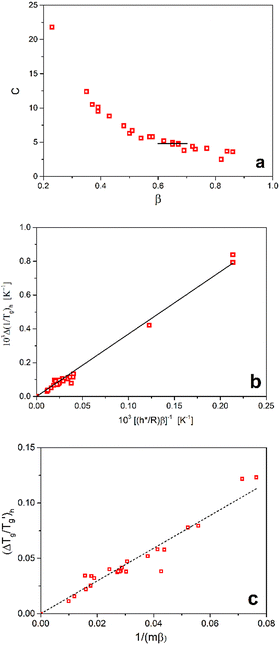 | ||
| Fig. 2 Analysis of heating DSC scans (qh = +10 K min−1) following cooling from equilibrium supercooled liquid (qc = −10 K min−1), simulated using the TNM parameters from Table 1 (points). (a) Dependence of parameter C (eqn (1)) on β. (b) Glass transition width (eqn (9)) as a function of [(h*/R) × β]−1. (c) Reduced glass transition width (eqn (10)) as a function of [m × β]−1. Solid and dashed lines represent linear regressions, with slopes D = 3.69 ± 0.06 and D′ = 1.48 ± 0.04, respectively. | ||
Eqn (1) can thus be reformulated into a different form
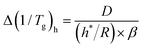 | (9) |
Alternatively, the reduced glass transition width can be expressed 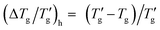 . Eqn (1) can then be rewritten in the following form
. Eqn (1) can then be rewritten in the following form
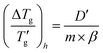 | (10) |
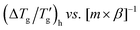 should yield a straight line passing through the origin (Fig. 2c). Linear regression validates this relationship, with the proportionality constant D′ found to be 1.48 ± 0.04 (R2 = 0.980).
should yield a straight line passing through the origin (Fig. 2c). Linear regression validates this relationship, with the proportionality constant D′ found to be 1.48 ± 0.04 (R2 = 0.980).
4.2. Analysis of simulated DSC cooling scans
A similar relationship to eqn (1) can be assumed for cooling DSC scans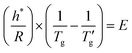 | (11) |
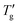 followed the procedure detailed in Section 3. The inflection point was first calculated numerically from the simulated DSC cooling data. A tangent was then fitted to this point via linear regression of twenty nearest data points, ensuring a correlation coefficient better than 0.9999. As illustrated by a solid line in Fig. 1b, Tg and
followed the procedure detailed in Section 3. The inflection point was first calculated numerically from the simulated DSC cooling data. A tangent was then fitted to this point via linear regression of twenty nearest data points, ensuring a correlation coefficient better than 0.9999. As illustrated by a solid line in Fig. 1b, Tg and 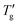 are defined by the intersection of this tangent line with dTf/dT = 0 and dTf/dT = 1, respectively.
are defined by the intersection of this tangent line with dTf/dT = 0 and dTf/dT = 1, respectively.
In contrast to the heating scan analysis, Fig. 3a illustrates that the parameter E for cooling process is dependent on the sum of both nonexponentiality and nonlinearity parameters, specifically (β + x). Consequently, eqn (11) can be rewritten as
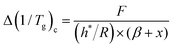 | (12) |
 | ||
| Fig. 3 Analysis of cooling DSC scans (qc = −10 K min−1) from equilibrium supercooled liquid, simulated using the TNM parameters from Table 1 (points). (a) Dependence of parameter E (eqn (11)) on (β + x). (b) Glass transition width (eqn (12)) as a function of [(h*/R) × (β + x)]−1. (c) Reduced glass transition width (eqn (13)) as a function of [m × (β + x)]−1. Solid and dashed lines represent linear regressions, with slopes F = 9.7 ± 0.2 and F′ = 4.10 ± 0.07, respectively. | ||
Similarly, mirroring the observation for the heating scan, the reduced glass transition width for cooling can be expressed as
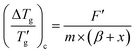 | (13) |
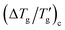 versus [m×(β + x)]−1 should also produce a straight line passing through the origin (Fig. 3c). The proportionality constant F′ found by this linear regression is 4.10 ± 0.07 (R2 = 0.992).
versus [m×(β + x)]−1 should also produce a straight line passing through the origin (Fig. 3c). The proportionality constant F′ found by this linear regression is 4.10 ± 0.07 (R2 = 0.992).
If the glass transition width Δ(1/Tg)c is obtained from the cooling scan, eqn (12) can potentially be used to estimate the value of the sum (β + x), provided the activation energy is known. However, such an estimated value is often distorted (usually overestimated), carrying an error margin of about 0.2. Very similar results are obtained when applying eqn (13). Applying the same procedure to heating scans provides even less reliable results; the error margins for the values of β obtained from eqn (9) and (10) exceed 0.2. The primary challenge across all these estimations lies in the uncertainties associated with the accurate determination of Δ(1/Tg)h and 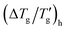 . It appears that even the modified procedure detailed earlier in this paper does not provide satisfactory results for accurately determining the nonexponentiality and nonlinearity parameters.
. It appears that even the modified procedure detailed earlier in this paper does not provide satisfactory results for accurately determining the nonexponentiality and nonlinearity parameters.
As already pointed out by Johari et al.,4 it is advantageous to use a more self-consistent method for determining Δ(1/Tg) and 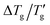 . In this approach, Tg is defined as the temperature at which the normalized heat capacity (CNp ≡ dTf/dT) equals a specific value z. Subsequently,
. In this approach, Tg is defined as the temperature at which the normalized heat capacity (CNp ≡ dTf/dT) equals a specific value z. Subsequently, 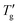 is determined as the temperature where the normalized heat capacity reaches a value 1 − z.Fig. 4 illustrates this determination for DSC cooling scans at different scanning rates calculated for PVC and As2S3.
is determined as the temperature where the normalized heat capacity reaches a value 1 − z.Fig. 4 illustrates this determination for DSC cooling scans at different scanning rates calculated for PVC and As2S3.
 | ||
Fig. 4 Determination of the Tg and 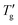 (for z = 0.15) from simulated cooling DSC scans for PVC (a) and As2S3 (b). Curves are shown for various scanning rates using TNM parameters listed in Table 1. (for z = 0.15) from simulated cooling DSC scans for PVC (a) and As2S3 (b). Curves are shown for various scanning rates using TNM parameters listed in Table 1. | ||
Fig. 5a represents a plot of Δ(1/Tg)cversus [(h*/R) × (β + x)]−1, determined using DSC cooling scan at −10 K min−1 and a value z = 0.15 for all TNM parameter sets listed in Table 1. Linear regression of this data yields the proportionality constant F = 9.33 ± 0.05 (R2 = 0.999). Similarly, Fig. 5b shows a plot of 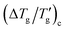 versus [m×(β + x)]−1 for the same TNM parameter sets and z value. The resulting proportionality constant F′ is 3.98 ± 0.03 (R2 = 0.998). These dimensionless constants, F and F′ ≅ F/ln(10) are invariant across different cooling rates and TNM parameter sets. They solely depend on quantity z that has been used for their determination. Table 2 provides their values for selected values of z.
versus [m×(β + x)]−1 for the same TNM parameter sets and z value. The resulting proportionality constant F′ is 3.98 ± 0.03 (R2 = 0.998). These dimensionless constants, F and F′ ≅ F/ln(10) are invariant across different cooling rates and TNM parameter sets. They solely depend on quantity z that has been used for their determination. Table 2 provides their values for selected values of z.
 | ||
Fig. 5 Analysis of simulated cooling DSC scans (qc = −10 K min−1) using TNM parameters from Table 1 (points). (a) Glass transition width defined by eqn (12). (b) Reduced glass transition width defined by eqn (13). Lines represent linear fits with F = 9.33 ± 0.05 (solid line) and F′ = 3.98 ± 0.03 (dashed line). Tg and 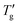 values were determined using the procedure defined in Fig. 4 with z = 0.15. values were determined using the procedure defined in Fig. 4 with z = 0.15. | ||
| z | F | F′ |
|---|---|---|
| 0.01 | 25.2 ± 0.4 | 10.8 ± 0.3 |
| 0.05 | 16.1 ± 0.1 | 6.9 ± 0.1 |
| 0.10 | 11.89 ± 0.06 | 5.08 ± 0.05 |
| 0.15 | 9.33 ± 0.05 | 3.98 ± 0.03 |
| 0.20 | 7.39 ± 0.04 | 3.15 ± 0.02 |
| 0.25 | 5.84 ± 0.04 | 2.49 ± 0.02 |
| 0.30 | 4.45 ± 0.03 | 1.90 ± 0.01 |
| 0.35 | 3.24 ± 0.03 | 1.38 ± 0.01 |
| 0.40 | 2.13 ± 0.02 | 0.904 ± 0.008 |
4.3. Estimation of nonexponentiality and nonlinearity
Utilizing the method detailed in Fig. 4 (for z ≥ 0.15) for cooling DSC scans allows for estimation of (β + x) values from eqn (12) and (13) with significantly higher precision than achievable with Moynihan's method, assuming the activation energy (h*/R) or fragility m is known (Fig. 3 and 5). The error margin for the resulting (β + x) values is typically less than 0.1. This accuracy aligns with the typical error in the nonlinearity parameter x estimated via the peak shift method proposed by Hutchinson,45,46 and validated in recent extensive testing.47 Paradoxically, the method suggested by Johari et al.4 does not perform adequately for the heating scans for which it was originally proposed; the linear fit is considerably poorer and the error margins significantly exceed those of the corrected Moynihan's method.Moynihan et al.23,48 established that the glass transition temperature Tg is related to cooling rate qc by the following equation:
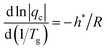 | (14) |
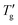 data, confirming that both linear dependences are parallel. Consequently, their differences remain constant regardless of the scanning rate: Δ(1000/Tg)c = 0.1270 ± 0.0002 K−1 for PVC and 0.2676 ± 0.0001 K−1 for As2S3. By inserting these values Δ(1/Tg)c, (h*/R) and F (for z = 0.15) into eqn (12) we estimate (β + x) values of 0.33 for PVC and 1.08 for As2S3. Similarly, we can find the reduced glass transition widths:
data, confirming that both linear dependences are parallel. Consequently, their differences remain constant regardless of the scanning rate: Δ(1000/Tg)c = 0.1270 ± 0.0002 K−1 for PVC and 0.2676 ± 0.0001 K−1 for As2S3. By inserting these values Δ(1/Tg)c, (h*/R) and F (for z = 0.15) into eqn (12) we estimate (β + x) values of 0.33 for PVC and 1.08 for As2S3. Similarly, we can find the reduced glass transition widths:  for PVC and 0.1153 ± 0.0059 for As2S3. By inserting these values, fragility m and the proportionality constant F′ (for z = 0.15) into eqn (13) we estimate (β + x) values of 0.32 for PVC and 1.09 for As2S3. All calculated values exhibit good agreement with the established parameters for these materials listed in Table 1.
for PVC and 0.1153 ± 0.0059 for As2S3. By inserting these values, fragility m and the proportionality constant F′ (for z = 0.15) into eqn (13) we estimate (β + x) values of 0.32 for PVC and 1.09 for As2S3. All calculated values exhibit good agreement with the established parameters for these materials listed in Table 1.
 | ||
| Fig. 6 Determination of the activation energy (h*/R) and the glass transition width Δ(1000/Tg)c using the cooling curves for PVC and As2S3 presented in Fig. 5. | ||
Combining eqn (12) and (14) yields the following relationship:
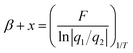 | (15) |
 | ||
Fig. 7 (a) Determination of Tg and 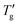 (at z = 0.3) from simulated cooling DSC scans of (GeTe4)60(GaTe3)40 glass at various scanning rates using TNM parameters from Table 1. (b) Evaluation of ln|q1| and ln|q2| at 1000/T = 2.4 K−1 based on the cooling curves presented in Fig. 7a. (at z = 0.3) from simulated cooling DSC scans of (GeTe4)60(GaTe3)40 glass at various scanning rates using TNM parameters from Table 1. (b) Evaluation of ln|q1| and ln|q2| at 1000/T = 2.4 K−1 based on the cooling curves presented in Fig. 7a. | ||
Fig. 7a displays the calculated curves for various cooling rates of the (GeTe4)60(GaTe3)40 chalcogenide glass,43 including the determination of the Tg and 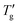 values for z = 0.3. Fig. 7b presents the linear dependencies ln|qc| versus 1000/T, from which ln|q1| and ln|q2| are determined. Linear regression of these plots yields ln|q1/q2| = 2.40 at 1000/T = 2.4 K−1. Using eqn (15) and the factor F corresponding to z = 0.3 (Table 2), an estimate of (β + x) = 1.85 ± 0.03 is obtained, which agrees well with the parameters for this material listed in Table 1. Similarly, ln|q1/q2| values of 8.92 for PVC and 28.76 for As2S3 are estimated from the data shown in Fig. 4. Applying eqn (15) with F corresponding to z = 0.15, yields (β + x) estimate of 0.32 ± 0.03 for PVC and 1.05 ± 0.03 for As2S3; these results closely match the values in Table 1. While the nonlinearity and nonexponentiality parameters cannot be extracted separately from the glass transition width of cooling DSC scans alone, they can easily be resolved from the temperature down-jump and up-jump experiments of the same magnitude.50,51
values for z = 0.3. Fig. 7b presents the linear dependencies ln|qc| versus 1000/T, from which ln|q1| and ln|q2| are determined. Linear regression of these plots yields ln|q1/q2| = 2.40 at 1000/T = 2.4 K−1. Using eqn (15) and the factor F corresponding to z = 0.3 (Table 2), an estimate of (β + x) = 1.85 ± 0.03 is obtained, which agrees well with the parameters for this material listed in Table 1. Similarly, ln|q1/q2| values of 8.92 for PVC and 28.76 for As2S3 are estimated from the data shown in Fig. 4. Applying eqn (15) with F corresponding to z = 0.15, yields (β + x) estimate of 0.32 ± 0.03 for PVC and 1.05 ± 0.03 for As2S3; these results closely match the values in Table 1. While the nonlinearity and nonexponentiality parameters cannot be extracted separately from the glass transition width of cooling DSC scans alone, they can easily be resolved from the temperature down-jump and up-jump experiments of the same magnitude.50,51
Measuring DSC cooling curves across a wide range of scanning rates can be challenging. The primary difficulty lies in achieving high cooling rates, particularly at lower temperatures. Conversely, at low cooling rates, a significant level of noise can complicate experimental data, especially for systems exhibiting only a small change in ΔCp in the glass transition region. Given these limitations, dilatometric experiments, especially modern implementations like spectroscopic ellipsometry,52 appear to be more suitable. If the glass transition cannot be reliably measured across different cooling rates, then eqn (15) is inapplicable. However, the parameter E, defined by eqn (11), remains applicable for (β + x) estimation in a single-cooling-rate experiment if the activation energy is known. Fig. 8a illustrates the dependence of E as a function of 1/(β + x) for all materials listed in Table 1 (points corresponding to data sets shown in Fig. 4 and 7 are highlighted).
 | ||
Fig. 8 (a) Dependence of the parameter E (calculated by eqn (11)) on 1/(β + x). Green points represent data for all materials listed in Table 1. Solid lines represent linear regression fits. Tg and 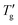 values were determined from cooling scans using the procedure shown in Fig. 4 for z = 0.15 (R2 = 0.996) and z = 0.30 (R2 = 0.992). Open triangles represent data reported by Chen et al.7 (b) Dependence of the parameter C (calculated by the eqn (1) using data shown in Table 1) on 1/β (red points). Tg and values were determined from cooling scans using the procedure shown in Fig. 4 for z = 0.15 (R2 = 0.996) and z = 0.30 (R2 = 0.992). Open triangles represent data reported by Chen et al.7 (b) Dependence of the parameter C (calculated by the eqn (1) using data shown in Table 1) on 1/β (red points). Tg and 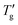 values were determined from heating scans according to the procedure described in Section 3. The solid line represents a linear regression fit (R2 = 0.975). Open squares represent data and corresponding linear fit (R2 = 0.857) extracted from Chen et al.7 Highlighted points represent data sets shown in Fig. 4 and 7. values were determined from heating scans according to the procedure described in Section 3. The solid line represents a linear regression fit (R2 = 0.975). Open squares represent data and corresponding linear fit (R2 = 0.857) extracted from Chen et al.7 Highlighted points represent data sets shown in Fig. 4 and 7. | ||
The solid lines in Fig. 8a represent the result of linear regression: E = −0.42 + 9.61/(β + x) for z = 0.15 and E = 0.47 + 3.88/(β + x) for z = 0.30. For a rough estimate of the nonexponentiality, the parameter C, defined by eqn (1), can be used analogously. Fig. 8b illustrates its dependence on 1/β for all materials listed in Table 1. The determination of Tg and  was performed according to the numerical procedure detailed in Section 3. The solid line in Fig. 8b represents the linear regression result: C = −3.84 + 5.57/β.
was performed according to the numerical procedure detailed in Section 3. The solid line in Fig. 8b represents the linear regression result: C = −3.84 + 5.57/β.
In their study, Chen et al.7 utilized the original Moynihan1 graphical method with DSC experiments at a 20 K min−1 heating rate to determine the Tg and 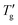 values for approximately 70 molecular, metallic, oxidic, and other glass-forming systems, and reported their TNM parameters. The open data points shown in Fig. 8b represent the parameter C calculated for this dataset. The linear regression C = −4.84 + 6.59/β of Chen's data set is weaker (R2 = 0.857) compared to stronger correlation observed for materials in Table 1 (R2 = 0.975). More importantly, the slope of the C vs. 1/β dependence is steeper compared to our dataset (dashed line, Fig. 8b). This suggests that, the specific proportionality constant C might depend slightly on the methodology used to determine Tg and
values for approximately 70 molecular, metallic, oxidic, and other glass-forming systems, and reported their TNM parameters. The open data points shown in Fig. 8b represent the parameter C calculated for this dataset. The linear regression C = −4.84 + 6.59/β of Chen's data set is weaker (R2 = 0.857) compared to stronger correlation observed for materials in Table 1 (R2 = 0.975). More importantly, the slope of the C vs. 1/β dependence is steeper compared to our dataset (dashed line, Fig. 8b). This suggests that, the specific proportionality constant C might depend slightly on the methodology used to determine Tg and 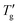 values.
values.
Chen et al.7 do not provide measured cooling DSC curves; however, these curves can easily be obtained by simulation using the TNM parameters they report. In Fig. 8a, the open points represent the parameter E obtained by eqn (11), using the Tg and 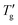 values determined via the Johari method (as described in Section 4.2, with z = 0.15 and 0.30, for simulated cooling curves at qc = −10 K min−1). The results show excellent agreement with both the data and the linear fit for all materials listed in Table 1. This demonstrates that the Johari method provides highly robust and reliably reproducible Tg and
values determined via the Johari method (as described in Section 4.2, with z = 0.15 and 0.30, for simulated cooling curves at qc = −10 K min−1). The results show excellent agreement with both the data and the linear fit for all materials listed in Table 1. This demonstrates that the Johari method provides highly robust and reliably reproducible Tg and 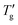 values, facilitating meaningful comparison across different materials. Consequently, the proportionality constant E remains invariant for a given value of z. In contrast, the analysis of heating scans may introduce systematic inaccuracies in the glass transition width that significantly exceed typical expectations.
values, facilitating meaningful comparison across different materials. Consequently, the proportionality constant E remains invariant for a given value of z. In contrast, the analysis of heating scans may introduce systematic inaccuracies in the glass transition width that significantly exceed typical expectations.
5. Conclusions
The glass transition width Δ(1/Tg) or the reduced width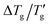 derived from DSC heating and cooling scans was comprehensively analyzed across 24 diverse materials, including inorganic glasses, organic polymers, and select molecular systems. An analysis employing the Tool-Narayanaswamy-Moynihan (TNM) model revealed significant differences between the cooling and heating processes.
derived from DSC heating and cooling scans was comprehensively analyzed across 24 diverse materials, including inorganic glasses, organic polymers, and select molecular systems. An analysis employing the Tool-Narayanaswamy-Moynihan (TNM) model revealed significant differences between the cooling and heating processes.
The width of the glass transition during cooling scans from metastable equilibrium supercooled liquid is independent on scanning rate. Instead, it is inversely proportional to the activation energy or the fragility index m, and the sum of the nonexponentiality β and nonlinearity x parameters: [(h*/R) × (β + x)]−1 or [m × (β + x)]−1. Notably, applying the method proposed by Johari4 for cooling DSC scans allows for the estimation of the (β + x) value with an accuracy comparable to that achieved via the established Hutchinson's peak shift method.45,46
In contrast the glass transition width of heating scan is influenced by both the current heating rate and the previous cooling rate. This width is inversely proportional to the activation energy or fragility, and solely the nonexponentiality parameter: [(h*/R) × β]−1 or [m × β]−1. However, the β value estimated from the glass transition width of heating scans is considerably less accurate than the (β + x) derived from cooling scans.
Conflicts of interest
The author declare that he has no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
A significant part of the data generated during this study is included in this article and the rest of the datasets is included in supplementary information (SI) (Fig. S1–S24). Supplementary information provide simulated DSC cooling and heating scans including the fictive temperature dependences for all materials analysed and covariance and regression matrices for linear fits. See DOI: https://doi.org/10.1039/d5ma01473e.Acknowledgements
This work was supported by the Selected Research Teams Program of the Faculty of Chemical Technology, University of Pardubice.References
- C. T. Moynihan, Correlation between the Width of the Glass Transition Region and the Temperature Dependence of the Viscosity of High-Tg Glasses, J. Am. Ceram. Soc., 1993, 76, 1081–1087 CrossRef CAS.
- C. T. Moynihan, K. S. Lee, M. Tatsumisago and T. Minami, Estimation of activation energies for structural relaxation and viscous flow from DTA and DSC experiments, Thermochim. Acta, 1996, 280/281, 153–162 CrossRef CAS.
- K. Ito, C. T. Moynihan and C. A. Angell, Thermodynamic determination of fragility in liquids and a fragile-to-strong liquid transition in water, Nature, 1999, 398, 492–495 CrossRef CAS.
- G. P. Johari, M. Beiner, C. MacDonald and J. Wang, The glass-softening temperature range and non-Arrhenius dynamics: the case of vitrified water, J. Non-Cryst. Solids, 2000, 278, 58–68 CrossRef CAS.
- B. C. Hancock, C. R. Dalton, M. J. Pikal and S. L. Shamblin, A Pragmatic Test of a Simple Calorimetric Method for Determining the Fragility of Some Amorphous Pharmaceutical Materials, Pharmaceutical Res., 1998, 15, 762–767 CrossRef CAS PubMed.
- M. J. Pikal, L. Chang and X. Tang, Evaluation of Glassy-State Dynamics from the Width of the Glass Transition: Results from Theoretical Simulation of Differential Scanning Calorimetry and Comparison with Experiment, J. Pharm. Sci., 2004, 93, 981–994 CrossRef CAS PubMed.
- Z. Chen, L. Zhao, W. Tu, Z. Li, Y. Gao and L. Wang, Dependence of calorimetric glass transition profiles on relaxation dynamics in non-polymeric glass-formers, J. Non-Cryst. Solids, 2016, 433, 20–27 CrossRef CAS.
- E. Bogdanova and V. Kocherbitov, Assessment of activation energy of enthalpy relaxation in sucrose-water system: effects of DSC cycle type and sample thermal history, J. Therm. Anal. Calorim., 2022, 147, 9695–9709 CrossRef CAS.
- K. Schneider, K. Schonhals and E. Donth, Uber die Grosse der kooperativen Umlagerungsbereiche am thermischen Glasubergang amorpher Polymere, Acta Polym., 1981, 32, 471–475 CrossRef CAS.
- E. Donth, The size of cooperatively rearranging regions at the glass transition, J. Non-Cryst. Solids, 1982, 53, 325–330 CrossRef CAS.
- K. Hallavant, M. Mejres, J. E. K. Schawe, A. Esposito and A. Saiter-Fourcin, J. Phys. Chem. Lett., 2024, 15, 4508–4514 CrossRef CAS PubMed.
- J. E. K. Schawe, M. Kyung Kwak, M. Stoica, E. Soo Park and J. F. Loffler, J. Phys. Chem. Lett., 2025, 16, 948–954 Search PubMed.
- J. E. K. Schawe, K. Hallavant, A. Esposito, J. F. Loffler and A. Saiter-Fourcin, J. Phys. Chem. Lett., 2025, 16, 12255–12265 CrossRef CAS PubMed.
- A. Q. Tool, Relation between Inelastic Deformability and Thermal Expansion of Glass in its Annealing Range, J. Am. Ceram. Soc., 1946, 29, 240–253 CrossRef CAS.
- A. Q. Tool, Effect of Heat-Treatment on the Density and Constitution of High-Silica Glasses of the Borosilicate Type, J. Am. Ceram. Soc., 1948, 31, 177–186 CrossRef CAS.
- G. Adam and J. H. Gibbs, On the Temperature Dependence of Cooperative Relaxation Properties in Glass-Forming Liquids, J. Chem. Phys., 1965, 43, 139–146 Search PubMed.
- I. M. Hodge, Effects of Annealing and Prior History on Enthalpy Relaxation in Glassy Polymers. 6. Adam-Gibbs Formulation of Nonlinearity, Macromolecules, 1987, 20, 2897–2908 Search PubMed.
- O. S. Narayanaswamy, A Model of Structural Relaxation in Glass, J. Am. Ceram. Soc., 1971, 54, 491–498 Search PubMed.
- J. C. Dyre, Narayanaswamy's 1971 aging theory and material time, J. Chem. Phys., 2015, 143, 114507 CrossRef PubMed.
- I. L. Hopkins, Stress Relaxation or Creep of Linear Viscoelastic Substances under Varying Temperature, J. Polym. Sci., 1958, 28, 631–633 Search PubMed.
- I. M. Douglass and J. C. Dyre, Distance-as-time in physical aging, Phys. Rev. E, 2022, 106, 054615 Search PubMed.
- O. V. Mazurin, S. M. Rekhson and Y. K. Startsev, The Role of Viscosity in the Calculation of the Properties of Glass in the Glass-Transition Region, Fiz. Khim. Stekla, 1975, 1, 412–416 Search PubMed . (English transl.).
- C. T. Moynihan, A. J. Easteal, M. A. DeBolt and J. Tucker, Dependence of the Fictive Temperature of Glass on Cooling Rate, J. Am. Ceram. Soc., 1976, 59, 12–16 CrossRef CAS.
- M. A. DeBolt, A. J. Easteal, P. B. Macedo and C. T. Moynihan, Analysis of Structural Relaxation in Glass Using Rate Heating Data, J. Am. Ceram. Soc., 1976, 59, 16–21 CrossRef CAS.
- Y. Guo, C. Zhang, C. Lai, R. D. Priestley, M. D'Acunzi and G. Fytas, Structural Relaxation of Polymer Nanospheres under Soft and Hard Confinement: Isobaric versus Isochoric Conditions, ACS Nano, 2011, 5, 5365–5373 CrossRef CAS PubMed.
- I. M. Hodge, Enthalpy relaxation and recovery in amorphous materials, J. Non-Cryst. Solids, 1994, 169, 211–266 CrossRef CAS.
- Y. Han, A. D'Amore and N. Nicolais, Analysis of Structural Relaxation in a Li2O·2SiO2 Glass using Rate Heating Approach, J. Mater. Sci., 1999, 34, 1899–1904 CrossRef.
- I. M. Hodge, Effects of Annealing and Prior History on Enthalpy Relaxation in Glassy Polymers. 4. Comparison of Five Polymers, Macromolecules, 1983, 16, 898–902 CrossRef CAS.
- S. L. Simon and G. B. McKenna, Interpretation of the dynamic heat capacity observed in glass-forming liquids, J. Chem. Phys., 1997, 107, 8678–8685 CrossRef CAS.
- H. Sasabe and C. T. Moynihan, Structural Relaxation in Poly(vinyl Acetate), J. Polym. Sci., 1978, 16, 1447–1457 CAS.
- Y. P. Koh and S. L. Simon, Enthalpy Recovery of Polystyrene: Does a Long-Term Aging Plateau Exist?, Macromolecules, 2013, 46, 5815–5821 CrossRef CAS.
- J. Málek, R. Svoboda, P. Pustková and P. Čičmanec, Volume and enthalpy relaxation of a-Se in the glass transition region, J. Non-Cryst. Solids, 2009, 355, 264–272 CrossRef.
- J. Málek and R. Svoboda, Remarkable difference in structural relaxation dynamics of conventionally prepared bulk glass and vapor-deposited thin films, J. Chem. Phys., 2024, 161, 074507 CrossRef PubMed.
- G. W. Scherer, Volume relaxation far from equilibrium, J. Am. Ceram. Soc., 1986, 69, 374–381 CrossRef.
- J. Málek, Dilatometric study of structural relaxation in arsenic sulfide glass, Thermochim. Acta, 1998, 311, 183–198 CrossRef.
- R. Svoboda, P. Honcová and J. Málek, Enthalpic structural relaxation in Se-Te glassy system, J. Non-Cryst. Solids, 2011, 357, 2163–2169 CrossRef CAS.
- A. J. Easteal, J. A. Wilder, R. K. Mohr and C. T. Moynihan, Heat Capacity and Structural Relaxation of Enthalpy in As2Se3 Glass, J. Am. Ceram. Soc., 1977, 60, 134–138 CrossRef CAS.
- R. Svoboda, J. Málek and M. Liška, Correlation between the structure and structural relaxation data for (GeSe2)y(Sb2Se3)1−y glasses, J. Non-Cryst. Solids, 2019, 505, 162–169 CrossRef CAS.
- R. Svoboda, J. Málek and M. Liška, Correlation between the structure and relaxation dynamics of (GeS2)y(Sb2S3)1−y glassy matrices, J. Non-Cryst. Solids, 2018, 479, 113–119 CrossRef CAS.
- S. N. Crichton and C. T. Moynihan, Structural relaxation of lead silicate glass, J. Non-Cryst. Solids, 1988, 102, 222–227 CrossRef CAS.
- S. Gaylord, B. Ananthasayanam, B. Tincher, L. Petit, C. Cox, U. Fotteringham, P. Joseph and K. Richardson, Thermal and Structural Property Characterization of Commercially Moldable Glasses, J. Am. Ceram. Soc., 2010, 93, 2207–2214 CrossRef CAS.
- R. Svoboda, M. Fraenkel, B. Frumarová, T. Wágner and J. Málek, Thermokinetic behaviour of Ag-doped (GeS2)50(Sb2S3)50 glasses, J. Non-Cryst. Solids, 2016, 449, 12–19 CrossRef CAS.
- R. Svoboda, M. Setnička, Z. Zmrhalová, D. Brandová and J. Málek, Structural interpretation of the enthalpy relaxation kinetics of (GeTe4)y(GaTe3)1−y far-infrared glasses, J. Non-Cryst. Solids, 2016, 447, 110–116 CrossRef CAS.
- A. M. Abd-Elnaiem and G. Abbady, A thermal analysis study of melt-quenched Zn5Se95 chalcogenide glass, J. Alloys Compd., 2020, 818, 152 CrossRef 880.
- J. M. Hutchinson and A. J. Kovacs, Effects of thermal history on structural recovery of glasses during isobaric heating, Polym. Eng. Sci., 1984, 24, 1087–1103 CrossRef CAS.
- J. M. Hutchinson and M. Ruddy, Thermal cycling of glasses. II. Experimental evaluation of the structure (or nonlinearity) parameter x, J. Polym. Sci., Part B, 1988, 26, 2341–2366 CrossRef CAS.
- R. Svoboda, The peak-shift method for the description of structural relaxation in glassy materials: a critical review, J. Am. Ceram. Soc., 2023, 106, 5233–5247 CrossRef CAS.
- C. T. Moynihan, A. J. Easteal, J. Wilder and J. Tucker, Dependence of the Glass Transition Temperature on Heating and Cooling Rate, J. Phys. Chem., 1974, 78, 2673–2677 CrossRef CAS.
- R. Svoboda, How to determine activation energy of glass transition, J. Therm. Anal. Calorim., 2014, 118, 1721–1732 CrossRef CAS.
- J. Málek, Volume and Enthalpy Relaxation Rate in Glassy Materials, Macromolecules, 1998, 31, 8312–8322 CrossRef.
- J. Málek, How to Distinguish Nonexponentiality and Nonlinearity in Isothermal Structural Relaxation of Glass-Forming Materials, J. Phys. Chem. B, 2024, 128, 8074–8083 CrossRef PubMed.
- Y. Zhang, E. C. Glor, M. Li, T. Liu, K. Wahid, W. Zhang, R. A. Riggleman and Z. Fakhraai, Long-range correlated dynamics in ultra-thin molecular glass films, J. Chem. Phys., 2016, 145, 114502 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |
