Open Access Article
Open Access ArticleA comparative study on heterogeneous CeO2 and homogeneous 1,8-diazabicyclo[5.4.0]-7-undecene catalysts for conversion of CO2 and monoethanolamine/ethylenediamine into cyclic compounds
Hiroki
Sato
a,
Mizuho
Yabushita
 *a,
Yingai
Li
a,
Yoshinao
Nakagawa
*a,
Yingai
Li
a,
Yoshinao
Nakagawa
 a and
Keiichi
Tomishige
a and
Keiichi
Tomishige
 *ab
*ab
aDepartment of Applied Chemistry, Graduate School of Engineering, Tohoku University, 6-6-07 Aoba, Aramaki, Aoba-ku, Sendai, Miyagi 980-8579, Japan. E-mail: m.yabushita@tohoku.ac.jp; tomishige@tohoku.ac.jp
bAdvanced Institute for Materials Research (WPI-AIMR), Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai, Miyagi 980-8577, Japan
First published on 6th March 2026
Abstract
Heterogeneous CeO2 and homogeneous 1,8-diazabicyclo[5.4.0]-7-undecene (DBU) catalysts have been employed for non-reductive conversion of CO2; however, their activity and catalysis have not yet been clarified clearly in identical reactions or conditions. Here, we conducted a kinetic study on the reactions between CO2 and monoethanolamine (MEA) (or ethylenediamine (EDA)) producing 2-oxazolidinone (or 2-imidazolidinone) with the two catalysts under high CO2-pressure conditions. The conversion of MEA with CeO2 proceeded faster than that with DBU under all the conditions tested in this study owing to the better affinity between CeO2 and the possible intermediate of CO2-captured MEA. In contrast, the conversion of EDA over CeO2 became slower at higher CO2 pressure, while the same reaction with DBU was accelerated and became faster than that with CeO2 upon the increase of CO2 pressure. These contrasting behaviors of CeO2- and DBU-catalyzed reactions with change in CO2 pressure were rationalized by the different effects of gaseous CO2 on the possible intermediate species. In the case of CeO2, excess CO2 capped the reactive amino group of an EDA-derived intermediate chemisorbed on the CeO2 surfaces, decreasing the nucleophilicity of this functional group for the cyclization reaction and lessening the reaction rate. Meanwhile, the DBU-catalyzed reactions under high pressure CO2 enabled the formation of the zwitterionic pair of CO2-captured DBU and CO2-captured EDA species, and such a strong electrostatic interaction between these species led to better reaction progress compared to low CO2-pressure conditions.
Green foundation1. The kinetic and comparable insights into both homogeneous DBU and heterogeneous CeO2 catalysts in non-reductive transformation of CO2 to directly synthesize cyclic organic carbamates and urea derivatives are precious guidelines for maximizing the efficiency of reaction systems.2. The catalytic performance of DBU and CeO2 has been shown via a detailed kinetic study to be drastically impacted by CO2 pressure, positing the great importance of selecting appropriate catalysts in accordance with the operating CO2 pressure. 3. Life cycle assessment and techno-economic analysis will bring remaining issues to improve the feasibility of each catalytic system under its corresponding optimum conditions into sharp relief. Such information should be helpful for further improvement of feasibility of each catalytic system. |
1. Introduction
Non-reductive conversion of carbon dioxide (CO2) has been recognized as a promising means of directly producing useful chemicals such as organic carbonates, organic carbamates, and urea derivatives from CO2 with alcohols and/or amines.1–9 The non-toxic and non-flammable nature of CO2 makes this means advantageous over conventional approaches for synthesizing such functional compounds that use hazardous carbonyl sources such as phosgene and carbon monoxide10–12 from a safety perspective. Yet, there are two well-known obstacles in most of the non-reductive CO2 conversion processes—severe thermodynamic limitation and low reactivity of CO2. For the former thermodynamic constraints, direct reactions of CO2 with aliphatic/aromatic alcohols into organic carbonates are highly endergonic and thus suffer from quite low equilibrium yields of target products (at most a few percent),1,4,12–14 and such a severe equilibrium limitation can be overcome by removing co-produced water via dehydrating agents1,4,12–15 and a gas stripping technique.16,17 Meanwhile, equilibrium limitation is generally not severe in the production of organic urea derivatives from CO2 and aliphatic amines owing to the high nucleophilicity of such amines, and high-yielding production of target compounds is possible without any dehydration technique.7 For the latter obstacle of low reactivity of CO2, this gaseous compound needs to be activated with catalysts for rapid syntheses of the aforementioned useful chemicals, in contrast to the conventional phosgene method that proceeds well without catalysts.Both heterogeneous and homogeneous catalysts have been reported to accelerate the non-reductive CO2 conversion with other reactants (e.g., alcohols and/or amines) thus far. Among various heterogeneous catalysts reported, CeO2-based materials have been shown to exhibit excellent catalytic activity because of their acid–base bifunctionality,9,18–34 and exemplified reactions are listed in Table S1. Meanwhile, an organic and strongly basic compound, 1,8-diazabicyclo[5.4.0]-7-undecene (hereafter, abbreviated as DBU), was employed as one of the typical homogeneous catalysts for the non-reductive CO2 conversion to produce cyclic carbamates from alkanolamines (Table S1),35,36 urea from ammonia,37 and polyureas from aliphatic diamines.38 Due to the strongly basic nature originating from the amidine moiety, DBU chemically captures and activates CO2, which undergoes intermolecular reactions with ammonia and aliphatic amines. A DBU-immobilized catalyst was also developed for the two-step synthesis of organic carbamates from CO2, aliphatic amines, and alkyl halides, the latter of which were added to reaction mixtures after certain times.39 DBU has also served as a catalyst for activating CO2 to trigger cycloaddition with epoxides into cyclic carbonates,40–42 polymerization with diamine into polyurea,43 and carboxylative coupling with propargyl chlorides and primary aliphatic amines into organic carbamates.42 In addition, the adduct of DBU and CO2 was used for synthesizing organic carbamates44 and urea derivatives,45 and DBU-derived salts worked as catalysts in cycloaddition of CO2 with epoxides.46 These previous reports posit DBU to be the key homogeneous catalyst/mediator for converting CO2 into value-added chemicals.
As alluded to above, both CeO2 and DBU have been employed as a heterogeneous and homogeneous catalyst, respectively, not only in the production of organic carbamates directly from CO2, amines, and alcohols but also in other reactions including CO2 as a reactant. From a practical perspective, CeO2 is advantageous over DBU because of the ease of separation and reuse as well as the applicability to continuous-flow reactions. Meanwhile, from academic viewpoints such as kinetic and mechanistic studies, it remains unclear whether CeO2 has further advantages beyond the general merits as a heterogeneous catalyst. The reason for this remaining issue is a lack of comparable reaction results acquired in the presence of CeO2 and DBU for the same substrates under identical conditions, as seen in Table S1. Therefore, in this study, such a comparative study was targeted with CeO2 and DBU to reveal the characteristics of each catalyst. Considering that the presence of three reactants (i.e., CO2, amine, and alcohol) with either catalyst in the production of organic carbamates makes the understanding of each reaction difficult, a single molecule possessing two terminal functional groups—2-aminoethanol (also called monoethanolamine (MEA))—was employed as a substrate to simplify the reaction system in this study. The target compound of this reaction is a cyclic organic carbamate, 2-oxazolidinone (eqn (1)). In addition, the structural analog of MEA, ethylenediamine (EDA), was also employed as a substrate to examine the catalysis of CeO2 and DBU in the production of a cyclic urea derivative, 2-imidazolidinone (eqn (2)). For both reactions of MEA19,47 and EDA48,49 as well as their related substrates of CO2-captured amines (i.e., alkylcarbamic acids),50–56 CeO2 was reported to exhibit higher catalytic activity compared to other metal oxides and afford good yields of 2-oxazolidinone and 2-imidazolidinone without dehydrating agents. Meanwhile, the absence of any catalyst failed to produce 2-oxazolidinone and 2-imidazolidinone from MEA + CO2 (investigated at 423 K, 2 MPa of CO2)19 and EDA + CO2 (at 433 K, 0.5 MPa of CO2),48 respectively. In addition, high CO2 pressure was shown to decrease the rates of substrate conversion because of the capping of the terminal functional group (i.e., hydroxy group for MEA and amino group for EDA) with excess CO2, while the maximum yield of target compounds was enhanced at higher CO2 pressures due to the shift of equilibrium.19,48,57
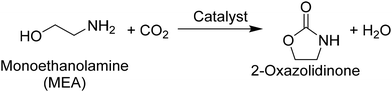 | (1) |
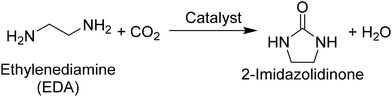 | (2) |
We note that the non-reductive conversion of CO2 with another structural analog of MEA, ethylene glycol, was not subject to this comparative study since this reaction suffers from severe reaction equilibrium (e.g., the equilibrium yield of the target product (ethylene carbonate) was only 1.2% at 423 K),58 and thus, the precise comparison between CeO2 and DBU under the kinetic-controlled region is quite difficult. We also note that no dehydrating agent was employed in this study to fairly compare the activity and catalysis of CeO2 and DBU due to the following two reasons. The first reason for no employment of dehydrating agents is given by a previously reported fact that some dehydrating agents such as 2-cyanopyridine not only trigger equilibrium shift but also promote reactions via the generation of highly basic sites at the interface between CeO2 and a chemisorbed 2-cyanopyridine molecule.59,60 Such additional effects of dehydrating agents make the precise elucidation and comparison of catalytic activity between CeO2 and DBU complicated or even impossible. Another reason originated from the not so severe equilibrium in the two target reactions operated in this study; indeed, over 80% yields of target products can be obtained even without dehydrating agents (exemplified data are shown later).
2. Experimental
2.1. Reagents
CeO2 (HS grade) was purchased from Daiichi Kigenso Kagaku Kogyo and used after its calcination in air in an electric furnace at 873 K for 3 h, which were previously found as the optimum pretreatment conditions for the non-reductive conversion of CO2 and amines (+alcohols) to produce urea derivatives (or organic carbamates).18,19,50,52,54 Acetonitrile, ethylenediamine (EDA), and tert-butyl alcohol were obtained from FUJIFILM Wako Pure Chemical. 1,8-Diazabicyclo[5.4.0]-7-undecene (DBU), 2-aminoethanol (MEA), and D2O were obtained from Tokyo Chemical Industry.2.2. Catalytic tests for the kinetic study
All the catalytic tests in this study were operated in a stainless-steel autoclave (HIRO Company, inner volume: 190 mL). Typically, 10 mmol of substrate, 1–5 mmol of either CeO2 or DBU as a catalyst, 500 mmol of acetonitrile as a solvent, and a stirring bar were charged into the autoclave. After sealing, the autoclave was purged and pressured with Ar to 1 MPa at room temperature. The reactor was heated to the target temperature with magnetic stirring at 250 rpm. When the inner temperature of the reactor, which was monitored using a K-type thermocouple, reached the designated one, the autoclave was pressured with CO2 to 2 MPa. This procedure for introducing CO2 into the reactor at the target temperature enabled us to suppress the undesired reaction progress during the temperature-ramping process and acquire kinetic data precisely.61 The time when the pressurization with CO2 was completed was defined as 0 h of reaction time. After the reaction operation for a specific time, the autoclave was immersed in a water bath to be quickly cooled to room temperature. The reaction mixture was collected with distilled water and filtered using a polytetrafluoroethylene (PTFE) syringe filter (0.2 μm mesh). For all the reactions except for those with an MEA substrate, the liquid phase was analyzed using a gas chromatograph with a flame-ionization detector (GC-FID; Shimadzu, GC-2014) equipped with an InertCap for Amines capillary column (GL Sciences, ∅0.32 mm × 30 m). In the case of MEA, the liquid phase was mixed with D2O and tert-butyl alcohol, the latter of which was used as an internal standard and analyzed by 1H nuclear magnetic resonance spectroscopy (1H NMR; Bruker, AV400, superconducting magnet 9.4 T, 1H 400 MHz). The 1H NMR spectra of typical reaction mixtures as well as authentic samples are shown in Fig. S1–S8. The spent CeO2 catalyst was employed for thermogravimetry and differential thermal analyses (TG-DTA; Thermo Plus EVOII, Rigaku); for the measurement, ca. 10 mg of the catalyst was heat-treated in an air flow (500 mL min−1) with a ramp rate of 10 K min−1 from room temperature to 1173 K. The fresh and spent catalysts were analyzed by powder X-ray diffraction measurement (XRD; MiniFlex600, Rigaku, Cu Kα radiation at 40 kV and 40 mA) and scanning transmission electron microscopy (STEM; HD-2700, Hitachi).3. Results and discussion
3.1. Comparison of CeO2 and DBU in the conversion of MEA and CO2
The catalytic activity of CeO2 and DBU was initially compared in the conversion of MEA and CO2 (eqn (1)) at 403 K under various conditions. It should be noted that all the data in this study were acquired in the kinetic region to fairly compare the activity of the two catalysts by taking the difference of their activity into account; hence, such a difference made us employ each appropriate catalyst amount in order to adjust the reaction progress within the kinetically controlled level and compare their catalytic activity on the basis of reaction rate per catalyst amount. In this reaction, the CeO2 catalyst was previously shown to exhibit good reusability in its four-time reuse after washing with methanol, drying at 383 K, and calcination in air at 873 K,19 with the preservation of its structural properties as confirmed by XRD (Fig. S9) and STEM (Fig. S10); within this context, neither deactivation nor structural change of the CeO2 catalyst needs to be considered in the kinetic study discussed below. We also note that the high-yielding production of the target product (i.e., 2-oxazolidinone) was possible by using both CeO2 and DBU catalysts (≥80%; Fig. S11A and Table S2). In these reactions for the high-yielding production of 2-oxazolidinone, the reactor was pressurized to 2 MPa with CO2 at room temperature (r.t.) and then heated to 403 K. In stark contrast, all the kinetic data were acquired in this study with the more precise experimental procedure as we employed previously;61 the reactor was initially heated to the desired temperatures and then pressurized with CO2 to eliminate the contributions of reaction progress during temperature ramp.As listed in Table 1 (the second data point from the right in Fig. 1A; detailed information is also available in Fig. S12 and Table S4), CeO2 exhibited 0.80 mmol mmolcat−1 h−1 formation rate of 2-oxazolidinone (entry 1), which is the target product in this reaction, in 380 mM MEA solution at 2 MPa of CO2 pressure. This rate was 17-fold higher than that given by DBU (entry 2). Considering that the formation of 2-oxazolidinone proceeds on the surface of CeO2 where 0.97 mmol g−1 Ce atoms are calculated to be present (detailed calculation procedure is described in the SI), its formation rate based on the number of Ce atoms at the surface (i.e., turnover frequency (TOF)) was estimated to be 5.9 mmol mmolCe−1 h−1, which was 125-fold higher than that in the case of DBU. Likewise, even at the other reaction temperatures ranging from 393 K to 423 K (Fig. 1A and Fig. S12 and Table S4), higher formation rates of 2-oxazolidinone were given by CeO2 rather than by DBU. These data clearly showed the higher catalytic activity of CeO2 for this reaction compared to DBU within the whole temperature range in this study. This observed rank of catalytic activity between the two catalysts was also reflected by the lower apparent activation energy (Ea) for CeO2 (64 kJ mol−1) than that for DBU (91 kJ mol−1).
| Entry | Substrate | Product | Catalyst | Formation rate (403 K, CO2 – 2 MPa) [mmol mmol−1 h−1] | Reaction orderb |
E
a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c [kJ mol−1] c [kJ mol−1] |
|
|---|---|---|---|---|---|---|---|
| P(CO2) | Substrate conc. | ||||||
| a The Arrhenius plots and double logarithmic plots for elucidation of the reaction order are depicted in Fig. 1–3, which are drawn on the basis of the reaction data summarized in Fig. S12–S14 and S16–S19 as well as Tables S4–S6 and S7–S10. b At 403 K. c Apparent activation energy at 2 MPa of CO2 pressure. d The values in parentheses were determined at 0.2 MPa of CO2 pressure. | |||||||
| 1 | MEA | 2-Oxazolidinone | CeO2 | 0.80 | 0.0 | 0.0 | 64 |
| 2 | DBU | 0.047 | 0.3 | 1.1 | 91 | ||
| 3 | EDA | 2-Imidazolidinone | CeO2 | 0.11 (0.54)d | –0.5 | –0.2 | 117 (76)d |
| 4 | DBU | 0.11 | 0.5 | 0.3 | 95 | ||
The reaction orders with respect to the CO2 pressure and MEA concentration in the presence of either CeO2 or DBU catalyst were then examined at 403 K by varying these parameters, as shown in Fig. 1B–E and Fig. S13 and S14 (detailed data are listed in Tables S5 and S6). Under all the conditions investigated here, the activity of the CeO2 catalyst was superior to that of DBU. In the case of CeO2, zero-order dependence was observed for both CO2 pressure and MEA concentration (entry 1 in Table 1 and Fig. 1B and D). Considering that amines readily capture CO2 chemically as alkylcarbamic acids,62–64 CO2-captured MEA (i.e., (2-hydroxyethyl)carbamic acid; hereafter, denoted as MEA-CA) was assumed to be an intermediate in this reaction; indeed, related studies often reported alkylcarbamic acids as intermediates in the formation of urea derivatives from amines and CO2.48,65–68 Besides, the catalytic transformation of MEA-CA in MEA solvent over CeO2 was shown recently to produce a linear urea derivative via 2-oxazolidinone as an intermediate.56 The observed zero-order dependence with respect to both CO2 pressure and MEA concentration thus indicated that the surfaces of the CeO2 catalyst were saturated by MEA-CA. In contrast, in the DBU-catalyzed conversion of MEA, the reaction orders with respect to the CO2 pressure and MEA concentration were determined to be 0.3 and 1.1, respectively (entry 2 in Table 1 and Fig. 1C and E). These positive reaction orders indicated the weak interaction between DBU and MEA-CA. Due to the presence of excess CO2 against MEA, the concentration of the plausible intermediate MEA-CA was dependent on the amount of MEA, not on the CO2 pressure. This fact could be connected to the observation of the larger positive reaction order with respect to MEA concentration compared to CO2 pressure. Yet, the slightly positive reaction order with respect to CO2 pressure still pointed to the importance of CO2 in the gas phase for the reaction progress. Altogether, CeO2 was superior to DBU as a catalyst in terms of the activity for the production of 2-oxazolidinone from MEA and CO2 under all the reaction conditions ranging from low to high CO2 pressure.
3.2. Comparison of CeO2 and DBU in the conversion of EDA and CO2
The kinetic study on the conversion of EDA and CO2 (eqn (2)) was performed with CeO2 and DBU. We note that the yield of the target product (i.e., 2-imidazolidinone) reached as high as 94% in the presence of the DBU catalyst; meanwhile, the reaction with CeO2 became quite slow with a long reaction time, and the yield of 2-imidazoildinone was 45% even at 96 h (Fig. S11B and Table S3). This behavior of CeO2 was due to the catalyst deactivation by the surface deposition of organic species, as confirmed by the observation of weight decrease with exothermic peaks in TG-DTA (Fig. S15). Such catalyst deactivation was also found previously in the conversion of CO2-absorbed ethylenediamine (i.e., (2-aminoethyl)carbamic acid, abbreviated as EDA-CA) in EDA solvent, where insoluble polyurea-like compounds were accumulated on the catalyst surfaces and led to the deactivation.52 The structural change of the CeO2 catalyst was not the reason for such deactivation shown by no obvious difference in its XRD pattern (Fig. S9) and STEM image (Fig. S10). Furthermore, appropriate treatment consisting of washing with methanol, drying at 383 K, and calcination in air at 873 K was demonstrated in our former study to regenerate the catalytic activity of CeO2.48 To avoid the underestimation of the catalytic activity of CeO2 and enable a fair comparison between DBU and CeO2 catalysts, the very initial stage of reaction time course was carefully examined in this study.Akin to the aforementioned reaction of MEA and CO2, the activity of these catalysts was initially elucidated and compared in 380 mM EDA solution at 403 K at 2 MPa of CO2 pressure (entries 3 and 4 in Table 1, the first data points from the right in Fig. 2A; detailed information is also available in Fig. S16 and Table S7). In this reaction, the formation rates of the target product 2-imidazolidinone given by CeO2 and DBU were both 0.11 mmol mmolcat−1 h−1, indicating similar mole-basis activity of these catalysts under the given conditions. On the basis of the surface Ce atoms (vide supra), the formation rate of 2-imidazolidinone over CeO2 was calculated to be 0.81 mmol mmolCe−1 h−1. At the higher temperatures of 413 K and 423 K, the formation of 2-imidazolidinone over CeO2 was faster than that over DBU (Fig. 2A). The negatively steeper slope for CeO2 in this Arrhenius plot provided a higher Ea of 117 kJ mol−1 (entry 3 in Table 1) relative to the 95 kJ mol−1 for DBU (entry 4). One of the possible factors for the high Ea value for CeO2 is its stronger affinity for EDA molecules that possess two basic amino groups due to its amphoteric nature, compared to basic DBU. This trend was opposite to that in the reaction between MEA and CO2 to produce 2-oxazolidinone with CeO2 and DBU (see section 3.1) and can be rationalized by the dependence of activity of CeO2 on CO2 pressure and the reaction mechanism. As seen in Fig. 2B, the negative reaction order with respect to CO2 pressure (i.e., −0.5, entry 3 in Table 1; more detailed data are available in Fig. S17 and Table S8) was observed in the reaction between EDA and CO2 over CeO2, indicating the inhibitory effect of CO2 in the gas phase. The double logarithmic plot in Fig. 2 clearly shows that the formation rate of 2-imidazolidinone over CeO2 became lower than that with DBU at high CO2 pressure. As proposed previously, the free amino group in the possible intermediate, CO2-absorbed EDA (i.e., EDA-CA), chemisorbed on the surfaces of CeO2 is possibly capped with gaseous CO2via an acid–base reaction in the presence of excess CO2, resulting in the decrease of nucleophilicity of the amino group that is a prerequisite for the cyclization to form 2-imidazolidinone (detailed description and scheme are provided later).48,50,57 Due to the requirement of the removal of such capping CO2, the reactions with CeO2 had a relatively high Ea. Indeed, the Ea at a low CO2 pressure (i.e., 0.2 MPa, which is equivalent to EDA (10 mmol)) was determined from the Arrhenius plot in Fig. 3 to be 76 kJ mol−1 (entry 3 in Table 1; more detailed data are available in Fig. S18 and Table S9), which was by 41 kJ mol−1 lower than that at 2 MPa of CO2 pressure. The formation rate of 2-imidazolidinone at 0.2 MPa of CO2 pressure was 0.54 mmol mmolcat−1 h−1, which was closer to that of 2-oxazolidinone over the same catalyst (entry 1). These Ea values showed the significant inhibitory effect of excess CO2 on the CeO2-catalyzed reaction of EDA. The reaction order with respect to EDA concentration determined from the raw data in Fig. S19 and Table S10 was −0.2, which was close to zero-order dependence and suggested the saturation of CeO2 surfaces with the above-mentioned intermediate, EDA-CA.
 | ||
| Fig. 3 Arrhenius plot for the conversion of EDA and CO2 over the CeO2 catalyst at a CO2 pressure of 0.2 MPa (i.e., equivalent to EDA). Detailed data are shown in Fig. S18 and Table S9. | ||
In stark contrast to the CeO2-catalyzed conversion of EDA and CO2, the reaction order with respect to CO2 pressure in the presence of the DBU catalyst was positively large (i.e., 0.5, entry 4 in Table 1 and Fig. 2B; more detailed data are available in Fig. S17 and Table S8). The presence of excess CO2 was, therefore, preferable for the reaction progress and should offer the ease of formation of key intermediate species (detailed discussion is given later). Due to such dependence, as described above, the use of DBU as a catalyst was favorable over that of CeO2 in the conversion of EDA operated under high pressure CO2. The reaction order with respect to EDA concentration was slightly positive (entry 4 in Table 1; more detailed reaction data are available in Fig. S19 and Table S10), indicating the relatively strong interaction between DBU and the plausible intermediate EDA-CA. Overall, the appropriate catalyst needs to be chosen for the production of 2-imidazolidinone from EDA and CO2 with consideration of CO2 pressure; that is, the desire to operate reactions at low CO2 pressure makes CeO2 a more suitable catalyst, while DBU is a preferable catalyst for reactions operated at high CO2 pressure.
3.3. The plausible reaction mechanism of the conversion of MEA/EDA and CO2 in the presence of CeO2/DBU
On the basis of the aforementioned kinetic results for the conversion of MEA/EDA with CO2 in the presence of either CeO2 or DBU as a catalyst (Table 1), the reaction mechanisms for each reaction are proposed in this section. In the CeO2-catalyzed reactions, the dependence on CO2 pressure was significantly different between the reaction of MEA (zero-order) and that of EDA (negative order). The former zero-order dependence with respect to CO2 pressure (and also the MEA concentration) can be explained by the saturation of CeO2 surfaces with the intermediate species that are readily produced from CO2 and MEA (i.e., MEA-CA). On the CeO2 surfaces, such chemisorbed and activated MEA-CA underwent the intermolecular nucleophilic attack by the terminal hydroxy group toward the carbonyl carbon to form the cyclic compound 2-oxazolidinone (Fig. 4A). Due to the neutral nature of the hydroxy group, acidic CO2 in the gas phase does not influence the reactivity of this functional group, and therefore, the negative impact of CO2 was not observed in this reaction. | ||
| Fig. 4 Plausible differences between the cyclization of each substrate in the presence of excess CO2 over CeO2 catalyst: (A) MEA and (B) EDA. | ||
Even in the case of EDA conversion over CeO2, the intermediate species EDA-CA, which is easily formed via chemical absorption of CO2 into EDA,50,51,69 should readily interact with the CeO2 surfaces as reported previously.51,52 Yet, the highly basic terminal amino group in this intermediate was problematic in the presence of excess CO2. This functional group needs to attack the carbonyl carbon as illustrated in the left side of Fig. 4B for the cyclization to form 2-imidazolidinone; however, this amino group should readily react with gaseous CO2via an acid–base reaction and lose its nucleophilicity (the right side of Fig. 4B), and the same rationale was provided in previous reports.48,50,57 In other words, the activation of amino groups via the removal of capping CO2 is necessary for the production of 2-imidazolidinone. This discussion can also be rationalized by the higher Ea value at 2 MPa of CO2 pressure (i.e., excess CO2 conditions) than that at 0.2 MPa (i.e., equivalent CO2 conditions).
In the reactions catalyzed by DBU, the positive reaction orders with respect to CO2 pressure were observed to a greater or lesser extent, indicating the favorability of high CO2-pressure conditions. According to previous studies, the chemical absorption of CO2 by a mixture of DBU and amine readily generated an ion pair of a protonated DBU cation and a carbamate anion.70–73 In the same manner as these reports, such an ion pair was also formed initially in our reactions using DBU as a catalyst (see the first step in Fig. 5). Under the high CO2-pressure conditions, another CO2 molecule was assumed to be captured at the DBU side along with the transfer of the proton from protonated DBU to the amino group in another MEA molecule in the MEA conversion (or the counter EDA-CA anion in the EDA conversion), as shown in the second step in Fig. 5. Such capture of additional CO2 molecule(s) in the presence of enough CO2 was also reported for various CO2-capturing systems using sole amine or multiple amines, resulting in the formation of multivalent ionic species.74–78 This step generated the zwitterionic DBU species possessing delocalized positive and negative charges at the amidine and captured CO2 moieties, respectively. In the conversion of EDA, both DBU- and EDA-derived species were in the zwitterionic state, and two pairs of positive and negative charges led to a strong electrostatic interaction between these species (Fig. 6B). Meanwhile, in the conversion of MEA, due to the presence of single negative charge in the MEA-derived species, the electrostatic interaction between such species and DBU-derived zwitterionic species could be weak. The degree of interaction strength between DBU- and substrate-derived species was indeed reflected by the large difference between the reaction orders with respect to the concentration of each substrate (i.e., 1.1 for MEA versus 0.3 for EDA; Table 1). The strong basic nature of DBU possibly enabled it to capture additional CO2 and be beneficial for the operation of reactions (namely, EDA conversion) at high CO2 pressure. In contrast, in the case of acid–base bifunctional CeO2 whose basicity is weaker than amines, the highly basic amino moiety of chemisorbed EDA-CA was capped with CO2, resulting in the negative impact of CO2 pressure on the formation rate of 2-imidazolidinone.
 | ||
| Fig. 5 Plausible ion pairs of DBU and each substrate in the presence of excess CO2: (A) MEA and (B) EDA. | ||
 | ||
| Fig. 6 Possible interaction between zwitterionic CO2-captured DBU and the (zwitter)ionic CO2-captured substrate: (A) MEA and (B) EDA. | ||
4. Conclusions
This work compared the activity and catalysis of heterogeneous CeO2 and homogeneous 1,8-diazabicyclo[5.4.0]-7-undecene (DBU) catalysts, both of which have been employed for various non-reductive CO2 conversions thus far, in the two model reactions using either monoethanolamine (MEA) or ethylenediamine (EDA) as a substrate under high CO2-pressure conditions. In the former reaction, the activity of the CeO2 catalyst was superior to that of DBU regardless of reaction temperature, CO2 pressure, or MEA concentration. Meanwhile, the activity of DBU became higher than that of CeO2 at high CO2 pressure in the conversion of EDA because of the inhibitory effect of excess CO2 on the CeO2-catalyzed EDA conversion. In the conversion of MEA over CeO2, the zero-order dependence for both CO2 pressure and MEA concentration was observed, suggesting that the possible intermediate of CO2-captured MEA (i.e., (2-hydroxyethyl)carbamic acid) interacted well with the CeO2 surfaces. On the other hand, the negative reaction order with respect to CO2 pressure was found for the CeO2-catalyzed conversion of EDA, along with a small dependence on EDA concentration. The inhibitory effect shown by gaseous CO2 can be explained by a decline in the nucleophilicity of the amino group in the key intermediate of CO2-captured EDA (i.e., (2-aminoethyl)carbamic acid) via its capping with CO2. The DBU-catalyzed reactions of both MEA and EDA were positively dependent on CO2 pressure and substrate concentration, while the degree of dependence was related to the interaction strength of the plausible intermediate species. In both reactions, the excess CO2 led to the formation of zwitterionic CO2-captured DBU possessing positive and negative charges at amidine and captured CO2 moieties, respectively. This DBU-derived species could interact better as a zwitterionic pair with the zwitterionic species derived from EDA and CO2, compared to the single anionic species derived from MEA and CO2, making the EDA conversion more favorable at higher CO2 pressure. The kinetic insights into these catalytic reactions with the two different catalysts, CeO2 and DBU, are precious guidelines for selecting an appropriate catalyst as well as suitable conditions for each target reaction.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc06699a.Acknowledgements
This work was financially supported by the following grants: a Grant-in-Aid for Specially Promoted Research and International Leading Research from the Japan Society for the Promotion of Science (JSPS KAKENHI; grant no. 23H05404 and 23K20034); the ENEOS TonenGeneral Research/Development Encouragement & Scholarship Foundation; and the International Polyurethane Technology Foundation.References
- K. Tomishige, M. Tamura and Y. Nakagawa, Chem. Rec., 2019, 19, 1354–1379 CrossRef CAS PubMed
.
- X. Zhao, S. Yang, S. Ebrahimiasl, S. Arshadi and A. Hosseinian, J. CO2 Util., 2019, 33, 37–45 CrossRef CAS
.
- S. Dabral and T. Schaub, Adv. Synth. Catal., 2019, 361, 223–246 CrossRef CAS
.
- K. Tomishige, Y. Gu, Y. Nakagawa and M. Tamura, Front. Energy Res., 2020, 8, 117 CrossRef
.
- H. Li, H. Cheng and F. Zhao, Asian J. Org. Chem., 2022, 11, e202200338 CrossRef CAS
.
- Q.-W. Song, R. Ma, P. Liu, K. Zhang and L.-N. He, Green Chem., 2023, 25, 6538–6560 RSC
.
- M. Yabushita, R. Fujii, Y. Nakagawa and K. Tomishige, ChemCatChem, 2024, 16, e202301342 CrossRef CAS
.
- K. Takeuchi, J. Jpn. Pet. Inst., 2024, 67, 45–51 CrossRef CAS
.
-
K. Tomishige, M. Yabushita and Y. Nakagawa, in Specialist Periodical Reports – Catalysis, ed. D. Shekhawat, Royal Society of Chemistry, Cambridge, 2025, vol. 36, pp. 45–90 Search PubMed
.
- H. Babad and A. G. Zeiler, Chem. Rev., 1973, 73, 75–91 CrossRef CAS
.
- A.-A. G. Shaikh and S. Sivaram, Chem. Rev., 1996, 96, 951–976 CrossRef CAS PubMed
.
- B. A. V. Santos, V. M. T. M. Silva, J. M. Loureiro and A. E. Rodrigues, ChemBioEng Rev., 2014, 1, 214–229 CrossRef CAS
.
- M. Honda, M. Tamura, Y. Nakagawa and K. Tomishige, Catal. Sci. Technol., 2014, 4, 2830–2845 RSC
.
- M. Tamura, Y. Nakagawa and K. Tomishige, Asian J. Org. Chem., 2022, 11, e202200445 CrossRef CAS
.
- H. Ohno, M. Ikhlayel, M. Tamura, K. Nakao, K. Suzuki, K. Morita, Y. Kato, K. Tomishige and Y. Fukushima, Green Chem., 2021, 23, 457–469 RSC
.
- Y. Gu, M. Tamura, Y. Nakagawa, K. Nakao, K. Suzuki and K. Tomishige, Green Chem., 2021, 23, 5786–5796 RSC
.
- P. Chen, H. Lin, Y. Kita, K. Nakao, T. Arai, A. Nakayama and M. Tamura, Energy Fuels, 2025, 39, 20506–20516 CrossRef CAS
.
- M. Honda, S. Sonehara, H. Yasuda, Y. Nakagawa and K. Tomishige, Green Chem., 2011, 13, 3406–3413 RSC
.
- M. Tamura, M. Honda, K. Noro, Y. Nakagawa and K. Tomishige, J. Catal., 2013, 305, 191–203 CrossRef CAS
.
- A. Bansode and A. Urakawa, ACS Catal., 2014, 4, 3877–3880 CrossRef CAS
.
- R. Zhang, L. Guo, C. Chen, J. Chen, A. Chen, X. Zhao, X. Liu, Y. Xiu and Z. Hou, Catal. Sci. Technol., 2015, 5, 2959–2972 RSC
.
- D. Stoian, A. Bansode, F. Medina and A. Urakawa, Catal. Today, 2017, 283, 2–10 CrossRef CAS
.
- M. Tamura, A. Miura, M. Honda, Y. Gu, Y. Nakagawa and K. Tomishige, ChemCatChem, 2018, 10, 4821–4825 CrossRef
.
- D. Stoian, F. Medina and A. Urakawa, ACS Catal., 2018, 8, 3181–3193 CrossRef CAS
.
- Y. Gu, A. Miura, M. Tamura, Y. Nakagawa and K. Tomishige, ACS Sustainable Chem. Eng., 2019, 7, 16795–16802 CrossRef CAS
.
- K. A. Alferov, Z. Fu, S. Ye, D. Han, S. Wang, M. Xiao and Y. Meng, ACS Sustainable Chem. Eng., 2019, 7, 10708–10715 CrossRef CAS
.
- G. S. More and R. Srivastava, Ind. Eng. Chem. Res., 2021, 60, 12492–12504 CrossRef CAS
.
- Y. Gu, M. Tamura, Y. Nakagawa, E. Ando and K. Tomishige, Catal. Today, 2023, 410, 19–35 CrossRef CAS
.
- F.-Y. Tu, H.-J. Lai, T.-A. Chiu, J.-C. Jiang and W.-Y. Yu, Surf. Interfaces, 2023, 43, 103597 CrossRef CAS
.
- J.-Y. Chuang, K.-T. Liu, M. M. Lin, W.-Y. Yu, R.-J. Jeng and M. Leung, J. CO2 Util., 2023, 76, 102592 CrossRef CAS
.
- T. Wei, L. Wang, J. Zhang, Y. Cao, X. Hui, J. Shi, S. Xu, L. Zhao, P. He and H. Li, Ind. Eng. Chem. Res., 2024, 63, 4338–4352 CrossRef CAS
.
- C. Li, C. Che, F. Li, W. Xue, J. Li, X. Zhao and Y. Wang, Ind. Eng. Chem. Res., 2024, 63, 17757–17766 CrossRef CAS
.
- Z. Yang, M. M. Lin, X. Lu, J. T. Zheng, W.-Y. Yu, Y. Zhu, H. Chang and Y. Xia, Chem. Eng. J., 2024, 486, 150339 CrossRef CAS
.
- Z. Yang, J. T. Zheng, X. Lu, M. M. Lin, D. Cai, Y. Wang, W.-Y. Yu, Y. Zhu and Y. Xia, Mater. Adv., 2024, 5, 6605–6617 RSC
.
- J. K. Mannisto, J. Heikkinen, J. Puumi, A. Sahari, P. R. Veliz and T. Repo, Org. Lett., 2025, 27, 5971–5976 CrossRef CAS PubMed
.
- J. Puumi, A. Sahari, A. Žáková, J. K. Mannisto, N. M. Maier and T. Repo, ACS Omega, 2025, 10, 33843–33849 CrossRef CAS PubMed
.
- Y. Manaka, Y. Nagatsuka and K. Motokura, Sci. Rep., 2020, 10, 2834 CrossRef CAS PubMed
.
- S. Zhang and L. Yan, ACS Appl. Polym. Mater., 2024, 6, 13350–13358 CrossRef CAS
.
- J. A. M. Vargas and A. C. B. Burtoloso, ChemSusChem, 2023, 16, e202300936 CrossRef PubMed
.
- N. Fanjul-Mosteirín, C. Jehanno, F. Ruipérez, H. Sardon and A. P. Dove, ACS Sustainable Chem. Eng., 2019, 7, 10633–10640 CrossRef
.
- A. W. Kleij, Curr. Opin. Green Sustainable Chem., 2020, 24, 72–81 CrossRef
.
- W. Schilling and S. Das, ChemSusChem, 2020, 13, 6246–6258 CrossRef CAS PubMed
.
- P.-X. Wu, H.-Y. Cheng, R.-H. Shi, S. Jiang, Q.-F. Wu, C. Zhang, M. Arai and F.-Y. Zhao, Adv. Synth. Catal., 2019, 361, 317–325 CrossRef CAS
.
- E. R. Pérez, M. O. da Silva, V. C. Costa, U. P. Rodrigues-Filho and D. W. Franco, Tetrahedron Lett., 2002, 43, 4091–4093 CrossRef
.
- Z.-Z. Yang, L.-N. He, Y.-N. Zhao, B. Li and B. Yu, Energy Environ. Sci., 2011, 4, 3971–3975 RSC
.
- L. Guo, K. J. Lamb and M. North, Green Chem., 2021, 23, 77–118 RSC
.
- R. Juárez, P. Concepción, A. Corma and H. García, Chem. Commun., 2010, 46, 4181–4183 RSC
.
- M. Tamura, K. Noro, M. Honda, Y. Nakagawa and K. Tomishige, Green Chem., 2013, 15, 1567–1577 RSC
.
- A. Primo, E. Aguado and H. Garcia, ChemCatChem, 2013, 5, 1020–1023 CrossRef CAS
.
- J. Peng, M. Tamura, M. Yabushita, R. Fujii, Y. Nakagawa and K. Tomishige, ACS Omega, 2021, 6, 27527–27535 CrossRef CAS PubMed
.
- J. Peng, M. Yabushita, Y. Li, R. Fujii, M. Tamura, Y. Nakagawa and K. Tomishige, Appl. Catal., A, 2022, 643, 118747 CrossRef CAS
.
- R. Fujii, M. Yabushita, D. Asada, M. Tamura, Y. Nakagawa, A. Takahashi, A. Nakayama and K. Tomishige, ACS Catal., 2023, 13, 1562–1573 CrossRef CAS
.
- R. Fujii, M. Yabushita, Y. Li, Y. Nakagawa and K. Tomishige, ACS Catal., 2023, 13, 11041–11056 CrossRef CAS
.
- S. Mihara, M. Yabushita, Y. Nakagawa and K. Tomishige, ChemSusChem, 2024, 17, e202301436 CrossRef CAS PubMed
.
- D. Su, H. Chen, Q. Yang, H. Liu, M. Lv, Y. Zhao, J. Zhou, Q. Zhang, Z. Lu and L. Chen, J. CO2 Util., 2024, 83, 102793 CrossRef CAS
.
- S. Mihara, N. Mizutani, H. Terada, M. Yabushita, T. Endo, Y. Nakagawa, A. Nakayama and K. Tomishige, Green Chem., 2025, 27, 14852–14872 RSC
.
- M. Tamura, M. Honda, Y. Nakagawa and K. Tomishige, J. Chem. Technol. Biotechnol., 2014, 89, 19–33 CrossRef CAS
.
- K. Tomishige, H. Yasuda, Y. Yoshida, M. Nurunnabi, B. Li and K. Kunimori, Green Chem., 2004, 6, 206–214 RSC
.
- M. Tamura, R. Kishi, Y. Nakagawa and K. Tomishige, Nat. Commun., 2015, 6, 8580 CrossRef CAS PubMed
.
- M. Tamura, R. Kishi, A. Nakayama, Y. Nakagawa, J. Hasegawa and K. Tomishige, J. Am. Chem. Soc., 2017, 139, 11857–11867 CrossRef CAS PubMed
.
- T. Chang, M. Yabushita, Y. Nakagawa, N. Fukaya, J.-C. Choi, T. Mishima, S. Matsumoto, S. Hamura and K. Tomishige, Catal. Sci. Technol., 2023, 13, 5084–5093 RSC
.
- D. B. Dell'Amico, F. Calderazzo, L. Labella, F. Marchetti and G. Pampaloni, Chem. Rev., 2003, 103, 3857–3898 CrossRef PubMed
.
- C. Gouedard, D. Picq, F. Launay and P. L. Carrette, Int. J. Greenhouse Gas Control, 2012, 10, 244–270 CrossRef CAS
.
- A. C. Forse and P. J. Milner, Chem. Sci., 2021, 12, 508–516 RSC
.
- A. Ion, V. Parvulescu, P. Jacobs and D. De Vos, Green Chem., 2007, 9, 158–161 RSC
.
- T. Jiang, X. Ma, Y. Zhou, S. Liang, J. Zhang and B. Han, Green Chem., 2008, 10, 465–469 RSC
.
- C. Wu, H. Cheng, R. Liu, Q. Wang, Y. Hao, Y. Yu and F. Zhao, Green Chem., 2010, 12, 1811–1816 RSC
.
- M. Marchegiani, M. Nodari, F. Tansini, C. Massera, R. Mancuso, B. Gabriele, M. Costa and N. Della Ca’, J. CO2 Util., 2017, 21, 553–561 CrossRef CAS
.
- H. Koizumi, K. Takeuchi, K. Matsumoto, N. Fukaya, K. Sato, M. Uchida, S. Matsumoto, S. Hamura and J.-C. Choi, Commun. Chem., 2021, 4, 66 CrossRef CAS PubMed
.
- X. Zhu, M. Song and Y. Xu, ACS Sustainable Chem. Eng., 2017, 5, 8192–8198 CrossRef CAS
.
- X.-C. Chen, K.-C. Zhao, Y.-Q. Yao, Y. Lu and Y. Liu, Catal. Sci. Technol., 2021, 11, 7072–7082 RSC
.
- H. Koizumi, K. Takeuchi, K. Matsumoto, N. Fukaya, K. Sato, M. Uchida, S. Matsumoto, S. Hamura and J.-C. Choi, ACS Sustainable Chem. Eng., 2022, 10, 5507–5516 CrossRef CAS
.
- H. Koizumi, K. Takeuchi, K. Matsumoto, N. Fukaya, K. Sato, M. Uchida, S. Matsumoto, S. Hamura, J. Hirota, M. Nakashige and J.-C. Choi, J. Org. Chem., 2023, 88, 5015–5024 CrossRef CAS PubMed
.
- V. Ermatchkov, Á.P.-S. Kamps and G. Maurer, J. Chem. Thermodyn., 2003, 35, 1277–1289 CrossRef CAS
.
- W. Conway, D. Fernandes, Y. Beyad, R. Burns, G. Lawrance, G. Puxty and M. Maeder, J. Phys. Chem. A, 2013, 117, 806–813 CrossRef CAS PubMed
.
- P. V. Kortunov, L. S. Baugh, M. Siskin and D. C. Calabro, Energy Fuels, 2015, 29, 5967–5989 CrossRef CAS
.
- J. K. Mannisto, L. Pavlovic, T. Tiainen, M. Nieger, A. Sahari, K. H. Hopmann and T. Repo, Catal. Sci. Technol., 2021, 11, 6877–6886 RSC
.
- X. E. Hu, Q. Yu, F. Barzagli, C. Li, M. Fan, K. A. M. Gasem, X. Zhang, E. Shiko, M. Tian, X. Luo, Z. Zeng, Y. Liu and R. Zhang, ACS Sustainable Chem. Eng., 2020, 8, 6173–6193 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |