 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Next-generation multiphase systems for homogeneous catalyst recycling: overcoming the reactivity vs. separation dilemma
Tim B.
Riemer†
 ,
Anna
Kampwerth†
,
Anna
Kampwerth†
 ,
Tim
Sinnhoffer
,
Moritz
Prange
,
Marc
Häusler
,
Dieter
Vogt
,
Tim
Sinnhoffer
,
Moritz
Prange
,
Marc
Häusler
,
Dieter
Vogt
 and
Thomas
Seidensticker
and
Thomas
Seidensticker
 *
*
Department for Biochemical and Chemical Engineering, Laboratory for Industrial Chemistry, TU Dortmund University, Emil-Figge-Str. 66, 44265 Dortmund, Germany. E-mail: thomas.seidensticker@tu-dortmund.de
First published on 3rd March 2026
Abstract
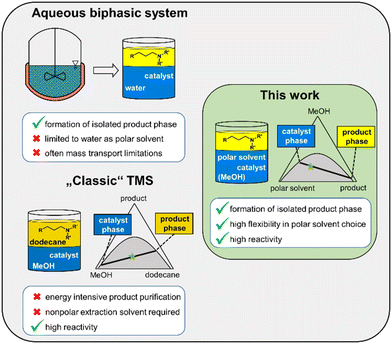
A new generation of thermomorphic multiphase systems combines the crucial advantages of classic homogeneous catalyst recycling methods: the reaction occurs under monophasic conditions in green solvents without mass transport limitations. Meanwhile, as in aqueous biphasic systems, the products form a separate and ideally solvent-free phase after reaction, enabling efficient separation from both the homogeneous catalyst and the solvents. This principle is demonstrated in three next-generation multiphase systems using the green solvents water, methanol, ethylene glycol, and γ-valerolactone, applying the rhodium-catalyzed hydroaminomethylation of 1-decene as a model reaction. Using a structured approach, phase-behavior experiments supported by LLE predictions via PC-SAFT enabled efficient identification of suitable solvent compositions. Validating their reaction performance in single- and recycling-batch experiments, all systems were then upscaled to continuous miniplant operation, where 268 hours of cumulative runtime revealed stability and high separation efficiency. An ethylene glycol/methanol solvent system stood out by combining high yields (79%) and selectivities (83%), very low catalyst loss into the product phase at just 0.8 mg rhodium per kg of product, and a directly attainable product – an amine purity of 72%, reducing subsequent purification efforts. These findings highlight the strong potential of thermomorphic systems to advance sustainable catalytic processes and streamline product purification in industrial applications.
Green foundation1. For the successful design of green chemical processes, several criteria – high reactivity, use of green solvents, catalyst recycling, and product separation – should be considered. We developed a novel type of switchable solvent system that combines all four criteria into one single concept. While in this study, these systems were designed for the sustainable production of aliphatic amines, this work also serves as a case study for the development of any other homogeneously catalyzed reaction.2. We demonstrated these next-generation multiphase systems in the rhodium-catalyzed hydroaminomethylation of 1-decene. Three of these newly developed solvent systems were upscaled to miniplant operation, providing reliable information on excellent performance in terms of reaction, catalyst recycling, and product purification. Compared to the product phase of the state-of-the art multiphase systems with the nonpolar extracting agent n-dodecane, the solvent content was reduced by 83% in this work, without additional purification steps. This significantly reduces product purification efforts since product amine purities of at least 72.3% can be obtained after just one decantation step. 3. This study focuses on proving the general principle of these new systems. Future work should apply and optimize these systems for actual applications in the chemical industry. |
Introduction
Homogeneous catalysis is a powerful tool for developing sustainable processes, as highly selective conversions to the desired products under typically very mild reaction conditions are achieved. Selecting the reaction medium (i.e., the solvent system) is a critical part of developing homogeneously catalyzed processes.1 Besides the chemical reaction, a number of aspects must be carefully balanced. Therefore, we suggest the following four criteria for the successful design of a chemical process:• reactivity;
• green solvents;
• catalyst stability and separation; and
• product separation.
First, the reactivity must be given, which often largely depends on the solvent.1,2 Furthermore, green solvents should be preferred, and hazardous and environmentally harmful solvents should be discarded as soon as possible (criterion 2). While these two criteria are usually considered in academic research, criteria 3 and 4 are often neglected or at least underrepresented.
Due to the expensive nature of homogeneous catalysts, their separation from the reaction mixture and their recycling and reuse are often essential for an economical process (criterion 3). Furthermore, the separation and purification of the products should already be considered at an early stage of process development (criterion 4), since downstream operations are often energy- and, hence, cost-intensive.
While products with low boiling points can still be separated from the catalyst and solvent by distillation, other concepts must be considered for higher-boiling-point products, as most homogeneous transition metal catalysts do not exhibit extensive temperature stability.3
Frequently used concepts for long-chain products are aqueous biphasic systems such as microemulsion systems (MESs)4,5 or aqueous systems with cyclodextrins6,7 as mass transport intensification agents. In these systems, substrates and products reside in a nonpolar organic phase, while the catalyst is immobilized in the aqueous phase.
Although such immobilization in different, immiscible phases allows simple separation after the reaction (criteria 3 and 4), mass transport limitations cause losses in reactivity (criterion 1).
Furthermore, a more demanding ligand design is usually required, and compatibility of the reaction system with water must be ensured, which leads to restrictions in the available systems.8
One efficient solution to this problem is the utilization of switchable liquid/liquid systems. Here, the reaction is performed under monophasic reaction conditions, leading to high reaction rates without mass-transport limitations. After the reaction, the phase behavior is switched to biphasic conditions, enabling easy phase separation via decantation. If the temperature is used as a phase switch, the systems are referred to as Thermomorphic Multiphase Systems (TMSs).9
TMSs utilize the temperature-dependent miscibility gap of a mixture involving three liquid components. During the reaction, the mixture is outside the miscibility gap in the monophasic area. Decreasing the temperature after the reaction widens the miscibility gap, separating the solution into a polar catalyst phase and a nonpolar product phase (Fig. 1).
The efficiency of this principle has already been demonstrated in various reaction and solvent systems.10–23 One particularly efficient TMS consists of the two solvents methanol and n-dodecane. Due to its high robustness, it has already been used in a continuous multipurpose plant to produce a wide range of alkyl amines.23
In this classic TMS, a nonpolar solvent, n-dodecane, is used alongside the polar solvent to adjust the miscibility gap. After the reaction, the nonpolar solvent acts as an extracting agent, separating the products from the polar catalyst phase.
However, this also results in the product phase being highly diluted, consisting of up to 70% n-dodecane, which leads to energy-intensive downstream processing via distillation. Processes with solvent-free product phases and significantly lower purification costs have the potential to significantly increase the sustainability of processes. For the first time, we present a new generation of thermomorphic multiphase solvent systems that meet all the criteria mentioned.
Here, as in previous TMSs, a temperature-dependent miscibility gap is adjusted using green solvents (criterion 2) to enable monophasic conditions with high reactivities during the reaction (criterion 1) and simple separation of the catalyst phase via subsequent cooling of the reaction mixture (criterion 3).
The crucial difference, however, is the elimination of the nonpolar extraction agent, since the thermomorphic miscibility gap is only adjusted with two polar solvents (a polar solvent and a medium polar solvent). The absence of a nonpolar extracting agent leads to an ideally solvent-free product phase (criterion 4), which allows considerable savings in product purification costs (Fig. 2, right side).
 | ||
| Fig. 2 Comparison of classic TMSs consisting of one polar solvent and one nonpolar extracting solvent (e.g., methanol and n-dodecane)23 and the newly developed TMSs of this work, omitting the nonpolar extracting agent. | ||
Consequently, as in the aqueous biphasic systems, both the catalyst and the products can be effectively separated after the reaction. In contrast to these, however, our system can achieve higher reactivities while simultaneously allowing for the use of solvents other than water. Even in comparison with the ex situ extraction procedure, which is often performed in industry, our system has the advantage that no separation of a nonpolar extracting agent is required after the simple decantation step.
The first step in developing these systems is to select suitable solvent candidates and to check the phase behavior of the selected solvents with the product, especially under separation conditions. Then, the chosen solvent systems are evaluated with regard to reaction activity and selectivity in smaller batch reactors.
The rhodium-catalyzed hydroaminomethylation (HAM) of 1-decene with diethylamine (DEA) to the aliphatic amine N,N-diethylundecylamine (Fig. 3) is chosen as the model reaction for this work. On the one hand, amines are a valuable substance class in the chemical industry and used in the production of various products, including surfactants, polymers, and pharmaceuticals.24,25 On the other hand, this reaction system has already been used in previous investigations of conventional methanol/n-dodecane TMS. Hence, having a suitable benchmark enables a reliable evaluation of the developed solvent systems at the end of this study.
 | ||
| Fig. 3 Reaction network for the Rh-catalyzed hydroaminomethylation (HAM) of 1-decene and diethylamine. | ||
After investigating the reaction performance, recycling runs are performed in small batch reactors to obtain initial estimates of the catalyst retention in its polar catalyst phase, as well as the general accumulation of components.
If a system remains promising after this step, it is upscaled into continuous miniplant operation. In this way, reliable information on the accumulation of components, catalyst leaching, and, in general, long-term stability is obtained. Moreover, the removal of coproducts and side products can be further investigated. The coproduct water was removed in this study via an organic solvent nanofiltration membrane since this method proved very efficient in previous works.21,22
Results and discussion
Since the second step in our procedure of developing a switchable solvent system is evaluating its reaction performance, the reaction network is discussed below (Fig. 3). Hydroaminomethylation is an orthogonal tandem reaction for the atom economical production of valuable aliphatic amines, which consists of hydroformylation and reductive amination. First, an aldehyde is formed from an alkene and syngas. This aldehyde reacts further via reductive amination, which includes condensation with a primary or secondary amine to an enamine/imine, following its hydrogenation to the corresponding amine. In HAM, the hydrogenation of 1-decene and the hydrogenation of aldehydes to the respective alcohols are typical side reactions. However, the alcohol formation is suppressed by the presence of carbon monoxide in the gas phase.4,21,26 Furthermore, isomerization of 1-decene can take place. Starting from the isomers, branched aldehydes, enamines, and tertiary amines can be formed following the reaction pathway, while aldol condensates may form in a consecutive reaction. Here, the aldehyde condensates with enamine under the cleavage of diethylamine. Since the formation of aldol condensates can be reduced via rapid hydrogenation of the enamine, a highly active catalyst system should be preferred. Therefore, a catalyst system consisting of rhodium and the ligand sulfoxantphos is used in this study, which has already demonstrated high activities and selectivities in previous works. Furthermore, the ligand's sulfonate groups enable the active catalyst's immobilization in the polar catalyst phase.The selectivity towards the product amines can be further increased by keeping the concentrations of the intermediates, namely the aldehyde and enamine, consistently low through the use of a Continuous Stirred Tank Reactor (CSTR).
The flowsheet of the continuous miniplant process, which has also already been used in previous TMS studies, is shown in Fig. 4. This miniplant setup mainly consists of the CSTR (B1), a decanter (B2), and a membrane unit (M1). A more detailed description of the setup can be found in the SI.
 | ||
| Fig. 4 Flowsheet of the continuous miniplant for the hydroaminomethylation of 1-decene and diethylamine. | ||
The first step is to select the potential solvents for the temperature-switchable systems. The main criteria are polarity, sustainability, and, if known, their suitability for performing the hydroaminomethylation in terms of reactivity. For the latter, the reaction activity in enamine hydrogenation was also considered.2 We selected water, methanol, ethylene glycol and γ-valerolactone. When it comes to sustainability, water is the solvent of choice.27 Furthermore, water has already proved to be a suitable solvent for hydroaminomethylation.6,7,28,29 In addition, from a process engineering point of view, a solvent system with water offers several advantages: first of all, water is formed during the reaction and, therefore, is already present in the system. Hence, using it as a reaction solvent reduces the number of different substances in the process, which must be considered during subsequent downstream processing. Furthermore, the coproduct water is separated via an OSN membrane to enable a constant water concentration. Since the effectiveness of the water separation increases with the water feed concentration, a sufficient membrane separation can already be achieved with a considerably smaller membrane area if the process involves a consistently high water concentration. Moreover, Perturbed-Chain Statistical Associating Fluid Theory (PC-SAFT) predictions of enamine thermodynamic activities by Huxoll et al. showed that small amounts of water positively influence the activity in reductive amination and hydroaminomethylation.30
Alcohols, such as methanol, and polyhydric alcohols, such as ethylene glycol, are also considered green solvents and are categorized as “recommended” in the CHEM21 solvent selection guide.27
Additionally, various alcohols have already been proved to be suitable solvents for hydroaminomethylation and reductive amination.29,31–34 Methanol, in particular, has already been successfully used with n-dodecane as a TMS for both reactions.23 Methanol ought to be an excellent solvent for hydroaminomethylation, as in the reductive amination it was shown that methanol coordinates to the aldehyde and accelerates the condensation step to the enamine, which exhibits high activity in methanol, thereby favoring hydrogenation to the tertiary amine at the same time.1
In addition to methanol, high thermodynamic activities of the enamine were also predicted in DMF (N,N-dimethylformamide) and NMP (N-methyl-2-pyrrolidone).2 However, these cannot be considered green and sustainable solvents and are even reprotoxic. Therefore, as γ-valerolactone is considered a green substitute for DMF and NMP, it is used as a sustainable solvent for the solvent systems developed in this work.35,36
Next, the solubility and phase behavior of the selected solvents with the product N,N-diethylundecylamine were determined experimentally and computationally via PC-SAFT predictions. Liquid–liquid equilibrium (LLE) experiments with the product amine as a second component most accurately represent the phase behavior in the actual process since the product is the component with the highest concentration in the CSTR, besides the solvents. Furthermore, it must be guaranteed that effective separation of the product and the polar catalyst phase occurs after the reaction at a high product concentration.
Considering the LLE of γ-valerolactone and the product amine (see Fig. 5), it is apparent that the desired phase behavior of monophasic conditions at a reaction temperature of 125 °C and biphasic conditions at 10 °C can be achieved with only the binary system. Hence, γ-valerolactone can be used without a second solvent.
 | ||
| Fig. 5 Liquid–liquid equilibrium (LLE) of γ-valerolactone and the product amine with the selected operation point of the reactor and the decanter. T. Averbeck from our working group calculated the data from Perturbed-Chain Statistical Associating Fluid Theory (PC-SAFT) predictions using the novel 3D-DMF-H method published in 2024.37 | ||
For the other solvents, the desired phase behavior must be adjusted by combination with a second solvent. The combination of water/methanol (for the LLE, see Fig. 6) and ethylene glycol/methanol (the LLE is provided in the SI) proved promising for a switchable solvent system.
 | ||
| Fig. 6 Ternary diagram of water, methanol and the product amine. The green star marks the selected operation point for the reaction. The PC-SAFT predictions were calculated by F. Huxoll from the Laboratory for Thermodynamics at TU Dortmund University with the already published PC-SAFT binary interaction parameters.30 | ||
As shown in Fig. 6, the investigated solvent system, consisting of methanol, water, and the amine product, exhibits a sufficiently temperature-dependent miscibility gap according to PC-SAFT predictions. The predicted data for 20 °C almost perfectly match the measured values at 10 °C. The selected operating point is in the biphasic region at 20 °C and in the monophasic region at higher temperatures.
Thus, three next-generation TMSs for the HAM of 1-decene with DEA were identified. In the first system, thermomorphic behavior is achieved with only a single polar solvent, γ-valerolactone. The second system achieves the desired phase behavior by combining water and methanol as an additional medium-polarity solvent. The third system uses the polar solvent ethylene glycol in combination with methanol as a phase mediator to adjust the thermomorphic miscibility gap (LLE in the SI).
Next, it is necessary to examine whether sufficient reaction activities can be achieved in the hydroaminomethylation. The results are shown in Fig. 7. The previously published solvent system, which consists of methanol and dodecane, is also included here for comparison.
Satisfactory product yields were achieved with all three solvent systems. The L-amine yield of the switchable ethylene glycol/methanol solvent system slightly exceeded that of the previous solvent system by 2%. This is particularly interesting as this solvent system is not monophasic under the selected reaction conditions. However, despite the two-phase conditions, excellent reaction yields were achieved.
In addition, the leaching of rhodium was halved, and 7 times less ligand was lost in the case of the ethylene glycol/methanol system. The γ-valerolactone solvent system also outperformed the previous solvent system in terms of leaching. However, significantly more aldol condensates were formed with the water/methanol and the γ-valerolactone solvent systems since, in the presence of water, the equilibrium of the amination reaction is shifted to the aldehydes, which can then be irreversibly converted via aldol condensation.38
As intended, all systems show significantly improved product phase purities compared to the previous system, as presented in Fig. 8. In particular, with γ-valerolactone, an almost solvent-free product phase consisting of 95 wt% of the amine product was achieved. In comparison, the product phase in the previously published system of methanol and dodecane consists of only 29.5 wt% amine product. The remaining 70 wt% are the two solvents, mostly n-dodecane, which have to be removed in energy-intensive downstream processes.
 | ||
| Fig. 8 Product phase composition after the hydroaminomethylation of 1-decene with diethylamine in the next-generation solvent systems. | ||
At least five batch recycling experiments were performed with all three systems. In the γ-valerolactone system (Fig. 9a), high product amine yields of about 75% were achieved in the initial reaction run. However, immediately after the initial run, a severe drop in selectivity could be observed, and exceedingly high yields of the aldol condensates of up to 60% were observed in the fifth recycling run. Further investigations showed that this behavior is caused by the accumulation of the coproduct water in the recycle phase (see the SI).
The water/methanol system (Fig. 9b) showed the highest stability over the five recycling runs. Here, the catalyst activity, expressed by the turnover frequency (TOF) at 20% conversion, was maintained throughout all recycling runs with yields of the tertiary product amine of up to 74%. The catalyst retention was also stable, ranging from 0.4% to 0.75% loss of Rh, during the batch recycling experiments. Again, ethylene glycol showed the highest reaction yields in the recycling runs with up to 84% of product amine and product selectivities of up to 85% during recycling (Fig. 9c), even under biphasic conditions. However, with this system, a significant drop in the reaction activity was observed over the recycling runs, which may be caused by oxygen contamination in the system in between the batch recycling runs. The rhodium leaching, which increased from 0.22% to over 4% from run three onwards, may also be caused by oxygen contamination.
All three solvent systems showed promising retention of the rhodium catalyst in the batch recycling runs. However, since precise and reliable values for the catalyst leaching and the accumulation of components can only be obtained on a larger continuous scale, the final evaluation of the solvent systems can only be done after upscaling them to the continuous TMS miniplant.
As expected from the recycling experiments of the γ-valerolactone system, the yields and selectivities of the tertiary amine rapidly decrease in continuous miniplant operation (see Fig. 10) due to the negative effect of water, which accumulates to 3.7 wt% in the catalyst phase. Furthermore, in the catalyst phase, the accumulation of up to 14 wt% of an unknown component was detected, which was later identified as N,N-diethyl-4-hydroxypentanamide and is formed in a ring-opening reaction of diethylamine with the solvent γ-valerolactone. Chalid et al. described a similar reaction of γ-valerolactone with other secondary amines as early as 2012.39
 | ||
| Fig. 10 Continuous miniplant operation of the γ-valerolactone solvent system in the hydroaminomethylation of 1-decene and diethylamine. Conditions: Treactor = 125–140 °C, p = 36–46 bar, pH2/pCO = 2, τreactor = 3 h, N = 800 rpm, nLig/nCat = 3.5, ccat, reactor = 0.69 mmol L−1, n1-decene/ncat = 1250, nDEA/n1-decene = 1, w1-decene = 12.5 wt%, m1-decene = 13.4 g h−1, mGVL/m1-decene = 6.46. Membrane: Nanopro S-3012. Yields were determined via the product phase composition in the decanter using GC-FID (more details are given in ref. 21). Dashed gray area: start-up procedure. | ||
The inability to observe this undesired side-reaction in the previous small-scale recycling experiments underlines the importance of performing continuous miniplant experiments with longer operation times. In these recycling experiments, no significant amount of side product had yet been formed. However, due to its highly polar nature, a drastic accumulation in the catalyst phase was observed in the miniplant operation. Given that this system does not exhibit stable reaction performance and γ-valerolactone turns out not to be inert, it is unsuitable for the investigated hydroaminomethylation.
The resulting reaction yields of the continuous miniplant experiment of the water/methanol solvent system are shown in Fig. 11. Aldol condensate formation significantly decreased when the operation mode was changed from batch to CSTR, since lower concentrations of both the aldehyde and the enamine are present during the reaction in the CSTR. As a result, the aldol condensate selectivity could be reduced from up to 21% to 2.8% by transferring the reaction to a continuous operation mode. Furthermore, the diethylamine feed was increased to potentially push the condensation equilibrium towards the enamine. However, an increase in the DEA/1-decene ratio from 1.25 to 1.75 showed no further change in selectivity. Over the whole miniplant operation, product amine yields of up to 73.2% with a selectivity of 76.4% were achieved.
 | ||
| Fig. 11 Reaction yields for the continuous miniplant operation of the switchable water/methanol solvent system for the hydroaminomethylation of 1-decene with diethylamine. Conditions: Treactor = 125–135 °C, p = 36 bar, pH2/pCO = 2, τreactor = 2–4 h, N = 800 rpm, nLig/nCat = 3.5, ccat, reactor = 0.73 mmol L−1, n1-decene/ncat = 960, nDEA/n1-decene = 1.25–1.75, m1-decene = 8.1–16.3 g h−1, w1-decene = 12.5 wt%, mmethanol/mwater = 5.15, mmethanol+water/m1-decene = 6.46. Membrane: Nanopro S-3012. Yields were determined via the product phase composition in the decanter using GC-FID (more details are given in ref. 21). Dashed gray area: start-up procedure. | ||
Besides the high reaction performance, the foreseen increase in water separation through the OSN membrane was also observed. In this way, it was possible to consistently remove more water from the process than was being produced during the reaction (detailed results are presented in the SI).
The third solvent system investigated in the continuous miniplant is the ethylene glycol/methanol TMS. The continuous experiment was conducted in both the liquid biphasic (MeOH/EG = 0.96) and the liquid monophasic (MeOH/EG = 1.8) reaction regimes (the phase diagram is provided in the SI) to evaluate the stability of this solvent system. The reaction yields of this miniplant experiment are shown in Fig. 13.
The ethylene glycol/methanol solvent system shows outstanding performance in both the reaction yields and the stability of the system for over 98 hours of miniplant operation. Product amine yields of up to 79% with selectivities of 83.4% were achieved.
Furthermore, changing the solvent ratio to switch from the biphasic (0–45 h) to the monophasic phase regime (60–75 h) in the reactor showed no remarkable effect on the reaction performance and underlines the robustness of this system. In addition to achieving excellent reaction performance in the methanol/ethylene glycol TMS, water was successfully separated from the process via an OSN membrane, limiting water accumulation in the recycle stream to only 6 wt%. More information about the influence of the solvent ratio on reaction performance, as well as about the membrane separation, can be found in the SI.
Besides the reactivity of the developed systems, their product purity is an important criterion for their evaluation. The solvent systems’ product phase should contain only a tiny fraction of solvents and preferably a large proportion of the tertiary product amine in order to reduce downstream processing costs. The product phase purities over the miniplant operation of the water/methanol and ethylene glycol/methanol systems are shown in Fig. 12. With up to 61.1 wt% product amine and only 19.5 wt% solvent, the water/methanol system achieved a significantly increased purity of the product phase. When comparing these two solvent systems, the ethylene glycol/methanol system achieved even higher purities with up to 72.3 wt% product amine and a minimum solvent content of only 12.3 wt%. Here, reducing the MeOH/EG ratio was beneficial for reducing the solvent content, since an increased ethylene glycol content results in less methanol dissolving in the product phase.
 | ||
| Fig. 12 Product phase purities of the miniplant operation of the solvent systems of water/methanol (A) and ethylene glycol/methanol (B). Conditions: Tdecanter = 10 °C, Tbuffer tank = 35 °C. | ||
 | ||
| Fig. 13 Reaction yields for the continuous miniplant operation of the switchable ethylene glycol/methanol solvent system for the hydroaminomethylation of 1-decene with diethylamine. Conditions: Treactor = 125–135 °C, p = 36 bar, pH2/pCO = 2, τreactor = 2–4 h, N = 800 rpm, nLig/nCat = 3.5, ccat, reactor = 0.76 mmol L−1, n1-decene/ncat = 960, nDEA/n1-decene = 1, m1-decene = 8.4–16.9 g h−1, w1-decene = 11.5 wt%, mmethanol/mEG = 0.74–1.87, mmethanol+EG/m1-decene = 7.15. Membrane: Nanopro S-3012. Yields were determined via the product phase composition in the decanter using GC-FID (more details are given in ref. 21). Dashed gray area: start-up procedure. | ||
In addition to the high reactivity and product purity of these systems, a high retention of the expensive rhodium catalyst is also crucial for the economic feasibility of these solvent systems (see Table 1).
| Species | Loss per houra [% h−1] | Loss per product [mg kg−1] | |||
|---|---|---|---|---|---|
| Water/MeOH | EG/MeOH | Water/MeOH | EG/MeOH | ||
| a Percentage losses refer to the initial mass of Rh and SX in the miniplant. | |||||
| Rh | TMS | 0.053 | 0.010 | 3.5 | 0.7 |
| OSN | 0.018 | 0.012 | 1.3 | 0.8 | |
| ∑ | 0.071 | 0.022 | 4.8 | 1.5 | |
| SX | TMS | 0.016 | 0.003 | 29.1 | 5.4 |
| OSN | 0.018 | 0.011 | 34.6 | 19.9 | |
| ∑ | 0.035 | 0.014 | 63.7 | 25.3 | |
The first measure is the hourly percentage loss of their initial mass. Furthermore, the loss of rhodium and the ligand per mass of the produced product amine is stated. Both solvent systems are characterized by their low catalyst loss, and in total, 4.4 wt% of the initial rhodium was lost via the product phase for the water/MeOH system and only 0.9 wt% with EG/MeOH. Measuring only 0.7 mg of rhodium loss per kg of the produced product amine, the EG/MeOH system particularly stands out for its low leaching.
Besides the lowest catalyst leaching per kilogram of product amine (Table 2), the ethylene glycol TMS also obtained the highest space–time yield (STY). Most notably, however, the ethylene glycol TMS stands out for its drastically increased product phase purity. Compared to the product phase of the conventional TMS with the nonpolar extracting agent n-dodecane, the solvent content was reduced by 83% in this work, without additional purification steps. This significantly reduces product purification efforts since product amine purities of at least 72.3% can already be obtained after just one decantation step.
| a In the product phase. b Heat duty required for heating and evaporating the solvents per kg of product amine. The heat duty was estimated by calculating the heating and evaporation energy for each solvent separately and then summing their contributions according to their mass fractions (more details are given in the SI). c Loss via the product phase of the TMS. |
|---|

|
Due to the increased product purity, the heat required to heat and evaporate the solvents in the product phase per kilogram of product amine (Qsolv/mprod) is reduced by 91.4% compared to the conventional MeOH/dodecane TMS.
Hence, developing this new class of solvent systems allows substantial improvements in all critical areas, such as catalyst retention, reaction performance, the space–time yield, and, in particular, product purification.
Materials and methods
Batch autoclave experiments
The batch experiments were performed in a 300 mL stirred tank reactor. A gas reservoir (V = 1 L) was used to provide a continuous hydrogen![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) carbon monoxide (2
carbon monoxide (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio) pressure of 36 bar in the reactor. The gas reservoir was pressurized to 75 bar with a mixture of hydrogen and carbon monoxide and a pressure controller was used to reduce the pressure to the reactor. The pressure drop in the gas storage tank caused by the reaction was measured with a digital pressure sensor. To fill 1-decene into the reaction mixture, the reactor was equipped with a dropping funnel with pressure equilibration.
1 ratio) pressure of 36 bar in the reactor. The gas reservoir was pressurized to 75 bar with a mixture of hydrogen and carbon monoxide and a pressure controller was used to reduce the pressure to the reactor. The pressure drop in the gas storage tank caused by the reaction was measured with a digital pressure sensor. To fill 1-decene into the reaction mixture, the reactor was equipped with a dropping funnel with pressure equilibration.
Batch recycling experiments
For the recycling experiments, the reactor was cooled in an ice bath after the reaction and carefully depressurized to less than 5 bar. A separation funnel with a glass flask for the catalyst phase was inertized with argon. The reaction solution from the reactor was transferred to the separating funnel using a slight overpressure and argon counter-flow and then separated at 10 °C for 10 minutes. Afterwards, the catalyst phase was released into the inertized glass flask and, from there, transferred to the reactor with a slight argon overpressure. After adding the appropriate amount of the amine substrate and solvent into the reactor and 1-decene into the dropping funnel, the next recycling run was started. A sample was taken from the product phase for GC-TCD and FID analysis as well as for ICP-OES to determine the concentration of rhodium and the ligand.Continuous miniplant experiments
The continuous miniplant experiments were carried out in a 1000 mL continuous stirred tank reactor (CSTR) with a blade stirrer and a baffle. The substrate 1-decene was purified from peroxides using an Al2O3 column. Furthermore, 1-decene, diethylamine, and the solvents (water/methanol or ethylene glycol/methanol) were degassed with argon in an ultrasonic bath for 30 minutes. The precursor Rh(acac)cod and the ligand sulfoxantphos were dissolved in the degassed polar solvent mixture in an ultrasonic bath for 1 hour. The entire plant was flushed with nitrogen several times, and the membrane module was connected in nitrogen counter-flow to the miniplant. Afterwards, the system was flushed with nitrogen again, and the degassed polar catalyst solution was drawn into the reactor under vacuum. The catalyst solution was then preformed in the reactor at 125 °C and 36 bar synthesis gas (H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO = 2
CO = 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for one hour.
1) for one hour.
Meanwhile, the two degassed substrates, 1-decene and diethylamine, were transferred into the dropping funnel under vacuum. Once this was finished, part of the catalyst solution was transferred to the other miniplant sections, the decanter and the buffer tank of the membrane loop, and the decanter was set to its operating temperature of 10 °C. After disconnecting the reactor from the rest of the system, the substrates in the dropping funnel were transferred into the reactor. At the end of the two-hour batch reaction, the reactor and membrane module were connected to the remaining miniplant by opening the respective valves. The recycling, membrane, and substrate pumps were started, and the continuous dosing of carbon monoxide and hydrogen to their desired ratio and pressure was set via two MFCs to start continuous operation. GC samples of the product phase in the decanter were taken automatically every hour and analyzed via online GC. In addition, samples from the product, recycling stream, and membrane permeate stream were analyzed offline every few hours to determine their composition via GC-FID/TCD and ICP-OES. When the ICP samples had rhodium or phosphorus concentrations below the detection limit of 1 wppm, they were concentrated by evaporation and re-measured to obtain their precise concentration. The constant gas composition during the operation was maintained via two Mass Flow Controllers (MFCs).
Additionally, the gas composition in the reactor was verified via GC-TCD over the miniplant experiment. The mass of the recycling phase was determined by measuring the mass of the buffer tank of the membrane loop. The mass of the catalyst phase and its solvent ratio can be adjusted by pumping the polar solvents via a pump into the membrane buffer tank.
Conclusions
In this work, a novel type of thermomorphic multiphase system (TMS) was developed to overcome the reactivity versus separation dilemma in homogeneous catalysis. These systems differ from other “conventional” TMSs because the adjustment of the temperature-dependent miscibility gap is only realized via polar solvents. Hence, the use of nonpolar extracting agents is omitted. As a result, these extraction agent-free TMSs are able to combine the key advantages of other established catalyst recycling methods. Similar to conventional TMSs or the classic combination of reaction and subsequent extraction, the reaction can occur under monophasic conditions. In other words, high reaction activities can be achieved without liquid–liquid mass transport limitations. Simultaneously, as with the aqueous biphasic systems, the products form a separate and (ideally) solvent-free product phase after the reaction and can, thus, be easily separated from the homogeneous catalyst and the solvents via a simple decantation step.The working principle of these TMSs was demonstrated in a systematic investigation involving phase behavior experiments, batch autoclave reactions, and continuous miniplant runs, as well as PC-SAFT calculations for the determination of liquid–liquid equilibria. During this process, three different thermomorphic solvent systems – γ-valerolactone, water and methanol, and ethylene glycol and methanol – were successfully identified. Since high reactivity and catalyst retention were achieved in the batch autoclave experiments with all three TMSs, all systems were subsequently investigated in continuous miniplant operation. The conducted study of the γ-valerolactone system underlines the importance of miniplant experiments in the development of novel chemical processes. Not only could the adverse effects of water accumulation on the reaction performance in this system be verified, but also the accumulation of a side product from the reaction of the γ-valerolactone solvent with the secondary amine not previously observed could be detected. While the γ-valerolactone system was thus excluded for hydroaminomethylation, the other two solvent systems showed excellent performance in continuous miniplant operation. The water/methanol solvent system was characterized by its effective separation of the coproduct water via an OSN membrane, as well as high product amine yields of 73.2% with a low catalyst loss of only 3.5 mg of rhodium per kg of the product amine produced. Yet the ethylene glycol/methanol TMS performed particularly well. In its continuous miniplant operation, product amine yields of up to 79% with selectivities of 83.4% were achieved. Excellent values were also obtained in terms of catalyst retention, and only 0.7 mg kg−1 of rhodium per product amine was lost via the product phase. Most notably, high product amine purities of 72.3% were achieved by adjusting the solvent ratio, reducing the heat duty for solvent heating and evaporation per kilogram of product amine by 91.4% compared to the conventional nonpolar extracting agent TMS.
The outstanding performance in catalyst retention, reaction performance, and product purification underlines the high potential of this new TMS class to combine the four criteria – reactivity, use of green solvents, catalyst separation, and product separation – into one single concept.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are available in the supplementary information (SI). Supplementary information: chemicals, analytics, miniplant setup, experimental procedure of phase investigation experiments, additional results of the miniplant experiments (including phase compositions, LLEs, and membrane performance), further experiments on the influence of the solvent ratio on the reactivity, and additional information on determining the heat duty for solvent separation. See DOI: https://doi.org/10.1039/d5gc06802a.The authors have cited additional references within the SI.40
Acknowledgements
Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – TRR 63 “Integrated Chemical Processes in Liquid Multiphase Systems” (subproject D3) 56091768.We would like to thank Fabian Huxoll from the Laboratory for Thermodynamics (TU Dortmund University) and Tobias Averbeck from our working group for the PC-SAFT calculations.
References
- F. Jameel and M. Stein, The many roles of solvent in homogeneous catalysis – The reductive amination showcase, J. Catal., 2022, 405, 24 Search PubMed.
- F. Huxoll, F. Jameel, J. Bianga, T. Seidensticker, M. Stein, G. Sadowski and D. Vogt, Solvent selection in homogeneous catalysis—Optimization of kinetics and reaction performance, ACS Catal., 2021, 11, 590 CrossRef CAS.
- D. J. Cole-Hamilton and R. P. Tooze, in Catalyst Separation, Recovery and Recycling: Chemistry and Process Design, ed. D. J. Cole-Hamilton and R. P. Tooze, Springer Netherlands, Dordrecht, 2006, pp. 1–8 Search PubMed.
- A. Weber, L. Porthun and R. Schomäcker, Rh-Catalyzed reductive amination of undecanal in an aqueous microemulsion system using a non-ionic surfactant, Catalysts, 2021, 11, 1223 CrossRef CAS.
- K. Duch, V. Kozachynskyi, L. Porthun, M. Illner and J.-U. Repke, Continuous homogeneously catalyzed production of fatty amines in microemulsions—Proof of concept in a mini-plant, Ind. Eng. Chem. Res., 2024, 63, 8916 CrossRef CAS.
- T. Roth, R. Evertz, N. Kopplin, S. Tilloy, E. Monflier, D. Vogt and T. Seidensticker, Continuous production of amines directly from alkenes via cyclodextrin-mediated hydroaminomethylation using only water as the solvent, Green Chem., 2023, 25, 3680 RSC.
- K. U. Künnemann, D. Weber, C. Becquet, S. Tilloy, E. Monflier, T. Seidensticker and D. Vogt, Aqueous biphasic hydroaminomethylation enabled by methylated cyclodextrins: Sensitivity analysis for transfer into a continuous Process, ACS Sustainable Chem. Eng., 2021, 9, 273 CrossRef.
- J. Diekamp and T. Seidensticker, Synthesis strategies towards tagged homogeneous catalysts to improve their separation, Angew. Chem., Int. Ed., 2023, e202304223 CAS.
- J. Bianga, K. U. Künnemann, T. Gaide, A. J. Vorholt, T. Seidensticker, J. M. Dreimann and D. Vogt, Thermomorphic multiphase systems – Switchable solvent mixtures for the recovery of homogeneous catalysts in batch and flow processes, Chem. – Eur. J., 2019, 25, 11586 CrossRef CAS PubMed.
- J. Bianga, K. U. Künnemann, L. Goclik, L. Schurm, D. Vogt and T. Seidensticker, Tandem catalytic amine synthesis from alkenes in continuous flow enabled by integrated catalyst recycling, ACS Catal., 2020, 10, 6463 CrossRef CAS.
- K. U. Künnemann, J. Bianga, R. Scheel, T. Seidensticker, J. M. Dreimann and D. Vogt, Process development for the rhodium-catalyzed reductive amination in a thermomorphic multiphase system, Org. Process Res. Dev., 2020, 24, 41 CrossRef.
- A. Kampwerth, T. B. Riemer, J. Pöttker-Menke, N. Oppenberg, A. M. Windisch, D. Vogt and T. Seidensticker, Hydroaminomethylation of methyl 10-undecenoate with integrated catalyst recycling via a thermomorphic multiphase system for the continuous production of renewable amines, RSC Sustain., 2024, 2, 1797 RSC.
- A. R. Gottu Mukkula, T. B. Riemer, A. Kühl, D. Vogt and S. Engell, Iterative real-time optimization of a reductive amination process in a thermomorphic multiphase system, Chem. Eng. Sci., 2024, 287, 119662 CrossRef CAS.
- S. Huang, H. Bilel, F. Zagrouba, N. Hamdi, C. Bruneau and C. Fischmeister, Olefin metathesis transformations in thermomorphic multicomponent solvent systems, Catal. Commun., 2015, 63, 31 CrossRef CAS.
- M. Jokiel, N. M. Kaiser, P. Kováts, M. Mansour, K. Zähringer, K. D. P. Nigam and K. Sundmacher, Helically coiled segmented flow tubular reactor for the hydroformylation of long-chain olefins in a thermomorphic multiphase system, Chem. Eng. J., 2019, 377, 120060 CrossRef CAS.
- L.-E. Meyer, M. B. Andersen and S. Kara, A deep eutectic solvent thermomorphic multiphasic system for biocatalytic applications, Angew. Chem., Int. Ed., 2022, 61, e202203823 CrossRef CAS PubMed.
- K. H. Rätze, M. Jokiel, N. M. Kaiser and K. Sundmacher, Cyclic operation of a semi-batch reactor for the hydroformylation of long-chain olefins and integration in a continuous production process, Chem. Eng. J., 2019, 377, 120453 CrossRef.
- M. S. Shaharun, H. Mukhtar and B. K. Dutta, Selectivity of rhodium-catalyzed hydroformylation of 1-octene in a thermomorphic solvent system, J. Appl. Sci., 2011, 11, 1157 CrossRef CAS.
- Y. Yang, J. Jiang, Y. Wang, C. Liu and Z. Jin, A new thermoregulated PEG biphasic system and its application for hydroformylation of 1-dodecene, J. Mol. Catal. A: Chem., 2007, 261, 288 CrossRef CAS.
- Y. Zeng, Y. Wang, Y. Xu, Y. Song, J. Zhao, J. Jiang and Z. Jin, Rh nanoparticles catalyzed hydroformylation of olefins in a thermoregulated ionic liquid/organic biphasic system, Chin. J. Catal., 2012, 33, 402 CrossRef CAS.
- T. B. Riemer, P. Lapac, D. Vogt and T. Seidensticker, Stable and continuous production of amines via reductive amination in a green switchable solvent system with efficient water removal, ACS Sustainable Chem. Eng., 2023, 11, 12959 CrossRef CAS.
- S. Schlüter, K. U. Künnemann, M. Freis, T. Roth, D. Vogt, J. M. Dreimann and M. Skiborowski, Continuous co-product separation by organic solvent nanofiltration for the hydroaminomethylation in a thermomorphic multiphase system, Chem. Eng. J., 2021, 409, 128219 CrossRef.
- T. B. Riemer, A. M. Windisch, D. Vogt and T. Seidensticker, Robust and flexible continuous operation of homogeneous catalysis in thermomorphic multiphase systems – On-stream switching of substrates and reactions in amine production, Chem. Eng. J., 2024, 497, 154643 CrossRef CAS.
- O. I. Afanasyev, E. Kuchuk, D. L. Usanov and D. Chusov, Reductive amination in the synthesis of pharmaceuticals, Chem. Rev., 2019, 119, 11857 CrossRef CAS PubMed.
- R. Tripathi, S. Verma, J. Pandey and V. Tiwari, Recent development on catalytic reductive amination and applications, Curr. Org. Chem., 2008, 12, 1093 CrossRef CAS.
- M. Strohmann, A. J. Vorholt and W. Leitner, Branched tertiary amines from aldehydes and α-olefins by combined multiphase tandem reactions, Chem. – Eur. J., 2022, 28, e202202081 CrossRef CAS PubMed.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, CHEM21 selection guide of classical- and less classical-solvents, Green Chem., 2016, 18, 288 RSC.
- E. Monciatti, F. Migliorini, G. Romagnoli, M. Vogt, R. Langer, M. L. Parisi and E. Petricci, Micellar catalysis for a sustainable hydroaminomethylation process in water, ACS Sustainable Chem. Eng., 2023, 11, 13387 CrossRef CAS.
- T. A. Faßbach, F. O. Sommer and A. J. Vorholt, Hydroaminomethylation in aqueous solvent systems – an efficient pathway to highly functionalized amines, Adv. Synth. Catal., 2018, 360, 1473 CrossRef.
- F. Huxoll, A. Kampwerth, T. Seidensticker, D. Vogt and G. Sadowski, Predicting solvent effects on homogeneity and kinetics of the hydroaminomethylation: A thermodynamic approach using PC-SAFT, Ind. Eng. Chem. Res., 2022, 61, 2323 CrossRef CAS.
- M. Ahmed, A. M. Seayad, R. Jackstell and M. Beller, Amines made easily: A highly selective hydroaminomethylation of olefins, J. Am. Chem. Soc., 2003, 125, 10311 CrossRef CAS PubMed.
- J. Bianga, N. Kopplin, J. Hülsmann, D. Vogt and T. Seidensticker, Rhodium-catalysed reductive amination for the synthesis of tertiary amines, Adv. Synth. Catal., 2020, 362, 4415–4424 CrossRef CAS.
- A. de Oliveira Dias, M. G. Gutiérrez, J. A. Villarreal, R. L. Carmo, K. C. Oliveira, A. G. Santos, E. N. dos Santos and E. v. Gusevskaya, Sustainable route to biomass-based amines: Rhodium catalyzed hydroaminomethylation in green solvents, Appl. Catal., A, 2019, 574, 97 CrossRef CAS.
- A. Behr, D. Levikov and E. Nürenberg, Rhodium-catalyzed hydroaminomethylation of cyclopentadiene, RSC Adv., 2015, 5, 60667 RSC.
- F. Kerkel, M. Markiewicz, S. Stolte, E. Müller and W. Kunz, The green platform molecule gamma-valerolactone – Ecotoxicity, biodegradability, solvent properties, and potential applications, Green Chem., 2021, 23, 2962 RSC.
- F. Valentini, G. Brufani, B. Di Erasmo and L. Vaccaro, γ-Valerolactone (GVL) as a green and efficient dipolar aprotic reaction medium, Curr. Opin. Green Sustainable Chem., 2022, 36, 100634 CrossRef CAS.
- T. Averbeck, G. Sadowski, C. Held and T. Seidensticker, Neural network-based tensor completion: Advancing predictions of activity coefficients and beyond, Ind. Eng. Chem. Res., 2024, 63, 12648 CrossRef CAS.
- S. Kirschtowski, F. Jameel, M. Stein, A. Seidel-Morgenstern and C. Hamel, Kinetics of the reductive amination of 1-undecanal in thermomorphic multicomponent system, Chem. Eng. Sci., 2021, 230, 116187 CrossRef CAS.
- M. Chalid, H. J. Heeres and A. A. Broekhuis, Ring-opening of γ-valerolactone with amino compounds, J. Appl. Polym. Sci., 2012, 123, 3556 CrossRef CAS.
- T. Gaide, J. M. Dreimann, A. Behr and A. J. Vorholt, Overcoming phase-transfer limitations in the conversion of lipophilic oleo compounds in aqueous media – A thermomorphic approach, Angew. Chem., Int. Ed., 2016, 55, 2924 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |



