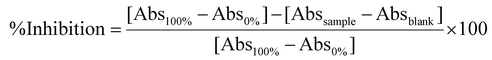Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Functional potential of crackers formulated with an innovative fermented cooked chickpea flour
Manuela Flavia
Chiacchio
a,
Silvia
Tagliamonte
 a,
Giuseppe
Blaiotta
a,
Angela
Pazzanese
a,
Giuseppe
Blaiotta
a,
Angela
Pazzanese
 a,
Cristina
Juan
b,
Ana
Juan-García
a,
Cristina
Juan
b,
Ana
Juan-García
 b and
Paola
Vitaglione
b and
Paola
Vitaglione
 *a
*a
aDepartment of Agricultural Sciences, University of Naples Federico II, Parco Gussone Ed. 84, 80055 Portici, Italy. E-mail: paola.vitaglione@unina.it; Tel: +39 081 2539357
bLaboratory of Food Chemistry and Toxicology, Faculty of Pharmacy and Food Science, University of Valencia, Avda. Vicent Andrés Estellés, s/n, 46100 Burjassot, Spain
First published on 11th March 2026
Abstract
Incorporating legume ingredients into staple foods offers a sustainable strategy to promote legume intake while improving the nutritional quality. Fermentation can further enhance the nutritional and functional properties of chickpea flour. This study evaluated the use of cooked chickpea flour (CF) and Lentilactobacillus diolivorans 13-4A-fermented cooked chickpea flour (FF) as functional ingredients in crackers, replacing refined wheat flour at 10% or 25% levels. Four enriched formulations (C10CF, C25CF, C10FF, C25FF) were compared to a conventional wheat cracker (CW). All products underwent in vitro digestion to obtain bioaccessible fractions (BFs) and digestion residues (RFs). BFs were analyzed for polyphenol bioaccessibility and bioavailability (using a Caco-2 cell model), total antioxidant capacity (TAC), and inhibitory activity against α-amylase, α-glucosidase, and dipeptidyl peptidase-IV (DPP-IV), while RFs were assessed for residual TAC. Fermentation tripled CF polyphenol content, with FF-enriched crackers, particularly C25FF, showing the highest polyphenol levels. Pyrogallol was detected only in FF and related crackers. FF exhibited the highest pre-digestion TAC, while post-digestion, C25FF and C10CF had the highest bioaccessible and residue TAC, respectively. CF or FF enrichment (10%) most effectively inhibited α-amylase (∼50%), whereas FF and C25FF showed the strongest α-glucosidase inhibition (∼8%). CF demonstrated the strongest DPP-IV inhibition. All BFs were non-cytotoxic; FF-derived BF enhanced the transcellular permeability and polyphenol transport in Caco-2 cells. Pyrogallol and sinapic/protocatechuic acids glucosides were the most bioavailable (40–100%). Overall, Lent. diolivorans 13-4A-fermented chickpea flour demonstrates strong potential to improve the nutritional and functional properties of crackers, contributing to the development of healthier cereal-based snacks.
1. Introduction
In recent decades, there has been a growing interest in dietary strategies that support healthier lifestyles, including the development of functional foods rich in fiber and plant-based proteins.1,2 Legumes have gained considerable attention due to their association with improved metabolic health, positive modulation of the gut microbiota, and their potential as sustainable alternatives to anima-based proteins.3 Despite their nutritional and environmental advantages, legume consumption remains well below the recommended 50–100 g per day intake established by the Food and Agriculture Organization (FAO).4 One promising approach to promote legume intake is the incorporation of legume-derived ingredients, such as chickpea flour, into widely consumed products like snacks and baked goods. Chickpeas (Cicer arietinum L.) are particularly valued for their high-quality protein content, slowly digestible carbohydrates, dietary fiber, and naturally gluten-free composition.5–7 Their inclusion in cereal-based products has been shown to enhance nutritional profiles by improving amino acid balance, increasing antioxidant activity, and contributing bioactive phytochemicals of products such as bread,8–10 snacks,2 muffins,11 and pasta.12 However, the incorporation of chickpea flour in food products poses several technological and sensory challenges, including the presence of anti-nutritional factors (e.g., phytic acid, fermentable oligosaccharides), off-flavors (e.g., earthy, beany), and undesirable textural changes.3,13 Fermentation has emerged as an effective, non-invasive and biotechnological process to overcome these limitations by modifying the food matrix. It can improve the organoleptic qualities of legumes while also enhancing the bioavailability of functional compounds such as bioactive peptides (BAPs), phenolic compounds, and antioxidant molecules.14,15 Notably, the functionality of these bioactives is influenced not only by their concentration but also by their release from the food matrix and bioaccessibility during digestion.16 Polyphenols, for example, are often bound within complex structures of dietary fiber and proteins, which can limit their release and subsequent absorption.17 Moreover, the interactions between bioactive compounds and the food matrix during processing may alter their biological properties, underscoring the importance of evaluating both the ingredient and the final product in functional food development.18 Crackers represent a convenient, widely accepted, versatile, food product, making them an ideal model-system for functional ingredient incorporation. Previous studies have shown that legume flour enrichment can enhance the nutritional value of crackers without negatively affecting sensory attributes at moderate inclusion levels (<30%).19 Moreover, fermentation of legume-enriched products has demonstrated improvements in protein digestibility, antioxidant capacity, and the release of phenolics and essential amino acids in vitro.14,15 We recently screened 14 lactic acid bacteria strains for their growth performance in chickpea-based purees and their ability to modulate key metabolites, including polyphenols, bioactive peptides, and phytic acid.20 Each strain generated distinct chemical and functional profiles, enabling the identification of candidates with multifunctional potential. Metabolomic analysis of the resulting lyophilized powders showed that fermentation with Lentilactobacillus diolivorans 13-4A produced a flour characterized with a well-balanced combination of phenolic and proteolytic features. Principal component analysis positioned this fermented flour within an intermediate cluster, highlighting its multifunctional properties. Moreover, Lent. diolivorans 13-4A exhibited robust growth and adaptability in the chickpea matrix, supporting its suitability as a starter culture. Beyond its balanced metabolomic profile, this strain is also known to produce exopolysaccharides, which may contribute to improved texture and stability in food formulations.21 Based on these findings, we hypothesized that chickpea powder fermented with Lent. diolivorans 13-4A could function as a multifunctional ingredient in baked products, enhancing nutritional and functional properties without compromising overall product quality. The aim of this study was therefore to evaluate the functional potential of Lent. diolivorans 13-4A-fermented chickpea powder as a novel ingredient in cracker. To this end, crackers were formulated by partially replacing wheat flour at 10% and 25% (w/w) with either fermented or non-fermented chickpea powder and compared with a conventional wheat-based control. The ingredients and resulting crackers were evaluated for antioxidant capacity, polyphenol bioaccessibility and intestinal bioavailability using a Caco-2 cell model, as well as for their in vitro inhibitory activity against digestive enzymes involved in glycemic control. Overall, this study provides a step forward in translating fermented chickpea-based ingredients, optimized through targeted microbial fermentation, into practical food formulations with validated functional and nutritional properties.2. Materials and methods
2.1 Chemicals and reagents
All the solvents used were high-pressure liquid chromatography (HPLC) and mass spectrometry (MS) grade or analytical grade and were purchased from Sigma-Aldrich (Milan, Italy) as were water, acetonitrile, methanol, formic acid (99–100%), acetic acid glacial. The standard 4-hydroxyphenyl acetic acid (≥95%), p-hydroxybenzoic acid (≥99%), pyrogallol (≥98%) and 2,2-difenil-1-picrylidrazyl (DPPH), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), Trizma, starch azure, Gly-Pro p-nitroanilide hydrochloride, para-nitrophenyl-α-D-glucopyranoside and sodium phosphate were purchased from Sigma Aldrich (Milan, Italy). Potassium chloride, sodium chloride, magnesium chloride, ammonium bicarbonate, and potassium dihydrogen phosphate were purchased from Merck (Darmstadt, Germany). The enzymes α-amylase from porcine pancreas (13 U mg−1 solid), α-glucosidase from Saccharomyces cerevisiae (≥10 U mg−1 solid), pepsin from porcine gastric mucosa (599 U mg−1 solid), pancreatin from porcine pancreas (100 U mg−1 solid), human dipeptidyl peptidase 4 (≥10 U mg−1 solid), and the bile salts were purchased from Sigma Aldrich (Milan, Italy). Cellulose powder from spruce was obtained from Fluka (Taufkirchen, Germany). For Caco-2 cell culture, Dulbecco's Modified Eagle's Medium (DMEM), fetal bovine serum (FBS), non-essential amino acids (NEAA), N-2-hydroxyethylpiperazine-N-2-ethane sulfonic acid (HEPES) buffer, antibiotic solution (penicillin–streptomycin), antimycotic solution (fungizone), phosphate buffered solution (PBS) and trypsin-EDTA were purchased from Gibco Invitrogen (Carlsbad, USA). While for the assays 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenylthiazolium bromide (MTT), dimethyl-sulfoxide (DMSO) were purchased from Sigma Aldrich (St Louis, MO, USA).2.2 Chickpea flour preparation
Chickpeas (Cicer arietinum L.) of the Cicerale cultivar, a traditional variety from the Campania region (Southern Italy) and recognized as a Slow Food Presidium, were used in this study. Chickpea seeds (harvest 2023) were obtained from a local producer (Azienda Agricola Iuorio Luigia, Villamina, Campania region, Italy). The seeds, cultivated throughout the Campania region, were processed to prepare cooked and freeze-dried fermented and unfermented chickpea powders. Since both ingredients were incorporated as wheat flour substitutes in the formulation of innovative crackers (section 2.3), they are hereafter referred to as cooked chickpea flour (CF) and fermented cooked chickpea flour (FF), respectively. The preparation has been previously described.20 Briefly, 500 g of chickpeas were cleaned with 500 mL tap water for 2 min at room temperature and then cooked at 121 °C for 20 min (Vapour Line 80B, VWR International PBI srl) in deionized water (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water-to-seed ratio). Cooked chickpeas were blended to obtain a 20% w/v puree and cooled to 30 °C. The chickpea-puree concentration (20%, w/w) was selected based on a previous screening study showing that this level was sufficient to support microbial growth, adaptation, and stable fermentation performance.20 For CF, the puree was directly lyophilized. For FF, the cooled puree was inoculated with Lent. diolivorans 13-4A (previously isolated from Sardinian fermented milk) at a concentration of ∼107 CFU mL−1 and incubated at 30 °C for 48 h prior to lyophilization. Both CF and FF were sieved through a 2 mm mesh and stored until use.
1 water-to-seed ratio). Cooked chickpeas were blended to obtain a 20% w/v puree and cooled to 30 °C. The chickpea-puree concentration (20%, w/w) was selected based on a previous screening study showing that this level was sufficient to support microbial growth, adaptation, and stable fermentation performance.20 For CF, the puree was directly lyophilized. For FF, the cooled puree was inoculated with Lent. diolivorans 13-4A (previously isolated from Sardinian fermented milk) at a concentration of ∼107 CFU mL−1 and incubated at 30 °C for 48 h prior to lyophilization. Both CF and FF were sieved through a 2 mm mesh and stored until use.
Prior to fermentation, the strain was precultured in MRS broth (Oxoid, Thermo Fisher Scientific) at 30 °C for 48 h. The pH and microbial loads were determined at the beginning of fermentation (after inoculum) and at the end of the fermentation period by pH measurement and standard plate counting on MRS agar (Oxoid), respectively. In addition, High-Performance Liquid Chromatography (HPLC) analyses were performed as previously described,20 to assess the availability of fermentable sugars (glucose and fructose) for LAB growth and to quantify the main metabolites produced during fermentation.
2.3 Cracker design and development
The detailed formulations of all crackers are provided in Table 1. Wheat flour (type 00′, SELEX S.p.A, Italy), sodium bicarbonate (CRASTAN S.p.A), and fine salt (Italkali, S.p.A) were used to produce the crackers. All these ingredients were purchased by a local supermarket (Sole 365 S.p.a). CF and FF produced as described above (section 2.2), were used to formulate four novel cracker prototypes, which were evaluated for their functional properties in comparison with a conventional wheat-based cracker (CW). The CW was prepared by adapting the recipe reported by Gangola et al.22 All the ingredients were mixed for 10 min, and the resulting dough was rolled to a thickness of ∼0.5 mm. Baking was carried out in a forced-air convection oven (fan speed level 2 on a 1–5 scale) to ensure uniform heat distribution. Baking conditions were optimized through preliminary trials by testing different temperature (170, 180, and 190 °C) and time (10, 15, and 20 min) combinations to obtain crackers with colour and crispness comparable to those of commercially available products (data not shown). Based on these trials, the final baking conditions were set at 180 °C for 15 min. After cooling, crackers were freeze-dried (Thermo scientific Heto, Denmark), ground using a GRINDOMIX 2000 (Retsch Italia, Verdere Scientific S.r.l, Bergamo, Italy) and sieved through a 2 mm mesh prior to analysis.| Ingredient | CW | C10CF | C25CF | C10FF | C25FF |
|---|---|---|---|---|---|
| CW: 100% wheat-based cracker; C10CF: 10% cooked chickpea flour-enriched cracker; C25CF: 25% cooked chickpea flour-enriched cracker; C10FF: 10% fermented cooked chickpea flour-enriched cracker; C25FF: 25% fermented cooked chickpea flour-enriched cracker. | |||||
| Wheat flour (WF) | 102.2 | 92.0 | 76.7 | 92.0 | 76.7 |
| Cooked chickpea flour (CF) | — | 10.2 | 25.6 | — | — |
| Fermented cooked chickpea flour (FF) | — | — | — | 10.2 | 25.6 |
| Baking soda | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 |
| Salt | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Water | 46.0 | 46.0 | 46.0 | 46.0 | 46.0 |
| Total | 150.0 | 150.0 | 150.0 | 150.0 | 150.0 |
Previous studies on bakery products containing chickpea flour have shown that enrichment levels below 30–50% can significantly increase the protein and fiber content while maintaining acceptable sensory quality and desirable rheological and textural properties of the final product.19,23,24 The nutritional composition of the crackers is reported in Table S1. It was calculated from the dough formulations (Table 1) using nutrient data for the individual ingredients obtained from the CREA food composition database (dry-matter basis). Total nutrient contents were adjusted to account for water loss during baking (33% for CW; 35% for C10CF and C10FF; 36% for C25CF and C25FF). Nutritional values were expressed per 100 g of finished product, based on the experimentally determined moisture content (lyophilization in triplicate), as reported in Table S1.
In the novel formulations, replacement of wheat flour with FF or CF at 10% and 25% (w/w) allowed the products to meet the “source of protein” and “source of fiber” nutrition claims as defined by Regulation (EC) no. 1924/2006, as amended by Regulation (EU) no. 1047/2012.25 At substitution levels of 10% and 25%, proteins contributed approximately 14% and 16% of the total energy value, respectively, while fiber content reached 3.5 g and 5.2 g per 100 g of product. Moreover, these substitution levels ensured satisfactory dough-handling properties, enabling evaluation of the effects of different replacement levels on the functional characteristics of the final products.
2.4 In vitro digestion
To investigate the bioaccessibility of polyphenols, in vitro gastrointestinal digestion was performed on CF, FF, and the corresponding crackers using the standardized INFOGEST method.26 Simulant salivary (SSF), gastric (SGF), and intestinal fluids (SIF) were prepared accordingly. For the oral phase, 2.5 g of each freeze-dried sample were mixed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with water to form a paste-like bolus, to which SSF (pH 7.0), amylase (75 U mL−1), and CaCl2 (1.5 Mm in SSF) were added. The mixtures were incubated for 2 min at 37 °C and 160 rpm before proceeding to the simulation of gastric phase. After 2 min, SGF (pH 3.0) and CaCl2 (0.15 mM in SGF), were added and the pH was adjusted to 3 with HCl 1 M prior adding pepsin (2000 U mL−1). The samples were then incubated for 2 h at 37 °C at 130 rpm. For the intestinal phase, SIF (pH 7.0), pancreatin (800 U mL−1), bile salts (10 mM), and CaCl2 were added. The pH was adjusted to 7.0 with NaOH 1 M, and the samples were incubated for 2 h at 37 °C and 100 rpm. At the end of intestinal phase, samples were centrifuged at 4200g for 15 min at 4 °C to separate the supernatant (bioaccessible fraction) from the pellet (residual fraction). The pellet was freeze-dried and gently ground to obtain a homogeneous powder, which was subsequently used to determine the residual antioxidant activity retained by the undigested fraction, as described in section 2.5.3. Aliquots (1 mL) of each digested sample were freeze-dried and used for the analyses reported below. For the in vitro bioavailability study, 5 mL of each supernatant were collected, freeze-dried, and subsequently resuspended in 2.5 mL of DMSO prior to the experiments described in section 2.8.
1 with water to form a paste-like bolus, to which SSF (pH 7.0), amylase (75 U mL−1), and CaCl2 (1.5 Mm in SSF) were added. The mixtures were incubated for 2 min at 37 °C and 160 rpm before proceeding to the simulation of gastric phase. After 2 min, SGF (pH 3.0) and CaCl2 (0.15 mM in SGF), were added and the pH was adjusted to 3 with HCl 1 M prior adding pepsin (2000 U mL−1). The samples were then incubated for 2 h at 37 °C at 130 rpm. For the intestinal phase, SIF (pH 7.0), pancreatin (800 U mL−1), bile salts (10 mM), and CaCl2 were added. The pH was adjusted to 7.0 with NaOH 1 M, and the samples were incubated for 2 h at 37 °C and 100 rpm. At the end of intestinal phase, samples were centrifuged at 4200g for 15 min at 4 °C to separate the supernatant (bioaccessible fraction) from the pellet (residual fraction). The pellet was freeze-dried and gently ground to obtain a homogeneous powder, which was subsequently used to determine the residual antioxidant activity retained by the undigested fraction, as described in section 2.5.3. Aliquots (1 mL) of each digested sample were freeze-dried and used for the analyses reported below. For the in vitro bioavailability study, 5 mL of each supernatant were collected, freeze-dried, and subsequently resuspended in 2.5 mL of DMSO prior to the experiments described in section 2.8.
2.5 Total antioxidant capacity (TAC)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 v/v), vortexed for 30 s and centrifuged at 4 °C for 10 min at 21
30 v/v), vortexed for 30 s and centrifuged at 4 °C for 10 min at 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100g. The resulting supernatant was separated from the pellet and filtered using 0.45 µm PTFE filters prior analysis. For the extraction of bioaccessible fractions, the aliquots (1 mL) of supernatant collected at the end of digestion were freeze-dried, resuspended in 1 mL of a methanol/water solution (70
100g. The resulting supernatant was separated from the pellet and filtered using 0.45 µm PTFE filters prior analysis. For the extraction of bioaccessible fractions, the aliquots (1 mL) of supernatant collected at the end of digestion were freeze-dried, resuspended in 1 mL of a methanol/water solution (70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 v/v), and extracted as described in this section for the undigested products.
30 v/v), and extracted as described in this section for the undigested products.
2.6 Determination of polyphenols by HPLC-UV/VIS analysis
Phenolic compounds were characterized in the extracted samples and bioaccessible fractions as reported by Chiacchio et al.20 Chromatographic analysis was performed using an HPLC SHIMADZU equipped with a detector UV/VIS SPD 20° set at wavelength of 280 nm and a Prodigy ODS3 100 Å column (250 mm × 4.6 mm, particle size 5 μm) (Phenomenex, CA, USA). The eluents were HPLC-grade water with 0.2% (v/v) formic acid (solvent A) and a mixture of acetonitrile/methanol (60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 v/v) (solvent B). The gradient was set as follows: 20% B (2 min), 30% B (8 min), 40% B (18 min), 50% B (26 min), 90% B (34 min), 90% B (37 min), 20% B (39 min), and 20% B (43 min). The injection volume was 20 μL and the flow rate of 1 mL min−1. The analysis was performed in triplicate for each sample, and the results were expressed as µg g−1 of dry weight using a calibration curve built with the corresponding standard compound. This analysis allowed for the identification of p-hydroxybenzoic acid, 4-hydroxyphenyl acetic acid, and pyrogallol. The limits of detection (LOD), quantification (LOQ), and the retention times of the identified polyphenols are reported in Table S2. Protocatechuic-4-O-glucoside, sinapoyl D-glucoside, and kaempferol 3-O-glucoside were identified through a high-resolution mass spectrometry (LC-HMRS) analysis as we previously reported.20 Molecular formula, m/z, and retention time of polyphenols identified by LC-HRMS are reported in Table S3.
40 v/v) (solvent B). The gradient was set as follows: 20% B (2 min), 30% B (8 min), 40% B (18 min), 50% B (26 min), 90% B (34 min), 90% B (37 min), 20% B (39 min), and 20% B (43 min). The injection volume was 20 μL and the flow rate of 1 mL min−1. The analysis was performed in triplicate for each sample, and the results were expressed as µg g−1 of dry weight using a calibration curve built with the corresponding standard compound. This analysis allowed for the identification of p-hydroxybenzoic acid, 4-hydroxyphenyl acetic acid, and pyrogallol. The limits of detection (LOD), quantification (LOQ), and the retention times of the identified polyphenols are reported in Table S2. Protocatechuic-4-O-glucoside, sinapoyl D-glucoside, and kaempferol 3-O-glucoside were identified through a high-resolution mass spectrometry (LC-HMRS) analysis as we previously reported.20 Molecular formula, m/z, and retention time of polyphenols identified by LC-HRMS are reported in Table S3.
2.7 Potential anti-diabetic activity: inhibition of digestive enzymes
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100g for 10 min. To evaluate the maximum activity of the enzyme (100%), the same reaction was reproduced by replacing the sample with the buffer Tris-HCl 0.1 M. Similarly, to evaluate the minimum activity of the enzyme (0%), the sample and the enzyme were replaced with Tris-HCl 0.1 M. For each sample, a blank was prepared, in which the buffer Tris-HCl 0.1 M was added instead of the enzyme. The absorbance of the resulting supernatants was measured at 595 nm with a UV-VIS spectrophotometer (PG Instruments, UK). Each determination was conducted in triplicate, and the results were expressed as %inhibition using the following formula:
100g for 10 min. To evaluate the maximum activity of the enzyme (100%), the same reaction was reproduced by replacing the sample with the buffer Tris-HCl 0.1 M. Similarly, to evaluate the minimum activity of the enzyme (0%), the sample and the enzyme were replaced with Tris-HCl 0.1 M. For each sample, a blank was prepared, in which the buffer Tris-HCl 0.1 M was added instead of the enzyme. The absorbance of the resulting supernatants was measured at 595 nm with a UV-VIS spectrophotometer (PG Instruments, UK). Each determination was conducted in triplicate, and the results were expressed as %inhibition using the following formula:where Abs100% is the absorbance of the sample containing the TRIS-HCl buffer instead of the digests; Abs0% is the absorbance of the samples containing TRIS-HCl buffer instead of the digests and the enzyme; Absblank is the absorbance of the samples containing TRIS-HCl buffer instead the enzyme.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100g for 10 min at 4 °C. After this period, the absorbance was measured at 410 nm with a UV-VIS spectrophotometer (PG Instruments, UK). To evaluate the maximum activity of the enzyme (100%), the same reaction was reproduced by replacing the sample with the phosphate buffer solution 0.1 M. Similarly, to evaluate the minimum activity of the enzyme (0%), the sample and the enzyme were replaced with phosphate buffer solution 0.1 M. For each sample, a blank was prepared, in which the phosphate buffer solution 0.1 M was added instead of the enzyme. Each determination was conducted in triplicate, and the results were expressed as %inhibition using the following formula:
100g for 10 min at 4 °C. After this period, the absorbance was measured at 410 nm with a UV-VIS spectrophotometer (PG Instruments, UK). To evaluate the maximum activity of the enzyme (100%), the same reaction was reproduced by replacing the sample with the phosphate buffer solution 0.1 M. Similarly, to evaluate the minimum activity of the enzyme (0%), the sample and the enzyme were replaced with phosphate buffer solution 0.1 M. For each sample, a blank was prepared, in which the phosphate buffer solution 0.1 M was added instead of the enzyme. Each determination was conducted in triplicate, and the results were expressed as %inhibition using the following formula:where Abs100% is the absorbance of the sample containing the phosphate buffer solution 0.1 M instead of the digests; Abs0% is the absorbance of the samples containing phosphate buffer solution 0.1 M instead of the digests and the enzyme; Absblank is the absorbance of the samples containing phosphate buffer solution 0.1 M instead the enzyme.
where Abs1 is the absorbance of the sample containing TRIS-HCl buffer instead of the digests; Abs2 is the absorbance of the digested sample; Abs3 is absorbance of the sample containing Tris-HCl buffer instead of the enzyme solution and the potential inhibitor.
2.8 Bioavailability of polyphenols in vitro
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 to 1
0 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 256 (1
256 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 dilution). The plates were incubated for 24 h and 48 h at 37 °C. After 24 h and 48 h, the media containing the extracts was removed and 200 μL of fresh medium along with 50 μL of MTT solution were added. This assay allows to measure the viability of cells that are metabolically active by determining the reduction of the yellow soluble tetrazolium salt to an insoluble purple formazan crystal via a mitochondrial reaction which reflects the viability of cells.30 Therefore, after 4 h of incubations at 37 °C, formazan crystals were dissolved in 200 μL of DMSO and 25 μL of Sorensen's solution (glycine 0.4 μmol L−1 + 0.1 μmol L−1 NaCl, pH 10.5) were added. The amount of converted dye was determined by measuring absorbance at 570 nm wavelength with the ELISA plate reader Multiskan EX (Thermo Scientific, MA, USA). Cell viability was expressed in percent relative to control cells which were treated with 1% DMSO.
2 dilution). The plates were incubated for 24 h and 48 h at 37 °C. After 24 h and 48 h, the media containing the extracts was removed and 200 μL of fresh medium along with 50 μL of MTT solution were added. This assay allows to measure the viability of cells that are metabolically active by determining the reduction of the yellow soluble tetrazolium salt to an insoluble purple formazan crystal via a mitochondrial reaction which reflects the viability of cells.30 Therefore, after 4 h of incubations at 37 °C, formazan crystals were dissolved in 200 μL of DMSO and 25 μL of Sorensen's solution (glycine 0.4 μmol L−1 + 0.1 μmol L−1 NaCl, pH 10.5) were added. The amount of converted dye was determined by measuring absorbance at 570 nm wavelength with the ELISA plate reader Multiskan EX (Thermo Scientific, MA, USA). Cell viability was expressed in percent relative to control cells which were treated with 1% DMSO.
| TEER (Ω cm2) = (R1 − R0) × A |
where concentration in BL is the phenolic concentration in the basolateral compartment after the time of incubation at 1 h, 2 h, 3 h, and 4 h; concentration in AP is the phenolic concentration in the apical compartment.
2.9 Statistical analysis
All the analysis were performed in three replicates and the results were expressed as mean ± SD. The results of cell viability (MTT test) were expressed as mean ± SD of eight independent replicates. Statistical analysis was performed using statistical software SPSS (version 20.0, SPSS, Inc., Chicago, IL, USA). The differences between samples were assessed by T-test and one-way ANOVA and Tukey's post hoc tests (p < 0.05). The visualization of polyphenols abundance with heatmap was performed using R version 4.0.3 (https://www.r-project.org) ComplexHeatmap package.323. Results and discussion
3.1 Antioxidant capacity of the flours and crackers
Lent. diolivorans 13-4A showed a very good growth performances during chickpea puree fermentation, reaching 9.00 ± 0.13 log CFU mL−1 and lowing the pH from 6.23 to 4.33 (Table S4). The chickpea puree before fermentation (t0 h) contained about 6 g L−1 of monosaccharides (glucose and fructose) (Table S4). Fermentation depleted about half of the initial monosaccharides and produced lactic acid (6.22 ± 0.17 g L−1), acetic acid (0.76 ± 0.11 g L−1) and ethanol (1.32 ± 0.09 g L−1) as e consequence of obligate hetero-fermentative metabolism of Lent. diolivorans.33 The high residual sugar, in relation to the high lactic acid produced, suggests the fermentation of some oligosaccharides. In fact, Lent. diolivorans strains able to ferment raffinose were described in literature.33,34 In addition, the Lent. diolivorans 13-4A was originally isolated from Sardinian fermented milk as raffinose fermenting (data not shown). The TAC of fermented chickpea flour (FF) was compared with that of unfermented chickpea flour (CF) and results are reported in the Fig. 1A (green bars). Fermentation resulted in a substantial increase in TAC, with FF exhibiting approximately 11-fold higher than CF. This finding aligns with previous studies reporting enhanced antioxidant capacity, up to 3-fold, in chickpea-based products fermented with LAB strains including Lactiplantibacillus plantarum CRL2211, Weissella paramesenteroides CRL2182, Lactobacillus plantarum 299v.24,35–37 This enhancement is likely due to microbial enzymatic activity during fermentation, which promotes structural modification of the matrix and the release of antioxidant compounds such as free phenols, bioactive peptides, and amino acids.38,39 Following the in vitro digestion, soluble antioxidant activity decreased in both CF and FF bioaccessible fractions (Fig. 1A, blue bars), by approximately 7-fold and 2.8-fold, respectively, compared to the products. However, FF retained markedly higher TAC in the pellet (non-solubilized residual) fraction, (Fig. 1B) which corresponds to the material that could reach the colon and interact with the gut microbiota in vivo.Among the five cracker formulations, C10CF (10% unfermented chickpea flour) showed the highest TAC, followed by the conventional product (CW) (Fig. 1, green bars). In contrast, Sáez et al.24 observed an improved antioxidant activity in crackers produced with chickpea flours, in which the fermented one accounted for 75% of the total flour used in the formulation, compared to the control. Interestingly, in our study, C10CF displayed a 7-fold increase in TAC compared to its ingredient (CF), suggesting that baking played a major role in the release and/or the formation of antioxidant compounds such as Maillard reaction products (i.e., Amadori compounds, hydroxymethylfurfural, furfural, aldehydes, aldols and melanoidins).40,41 Specifically, thermal treatment above 150 °C are well known to promote the formation of high-molecular-weight melanoidins which exhibit radical-scavenging activity, thereby contributing to overall antioxidant capacity of bakery products.42 In addition, Maillard reaction products have also been reported to interact with dietary fiber and cereal-derived polyphenols (e.g., ferulic acid), forming so-called “Maillardized dietary fiber”, which may further enhance the antioxidant activity in the final product.43 In FF-enriched crackers, C25FF showed approximately twice the TAC of C10FF, indicating a dose-dependent effect of fermented flour addition. Conversely, in CF-enriched crackers, C10CF exhibited a higher TAC than C25CF. This discrepancy may not be related directly to total polyphenol content, which was higher in both C10FF and C25FF compared to CF crackers, but rather to differences in polyphenol profile and their interactions with each other and with free radicals and/or to the release of bioactive peptides with antioxidant potential.44,45 Moreover, in protein-rich matrices, such as crackers containing 25% chickpea flour, high-molecular-weight melanoidins may form and interact with chickpea-derived polyphenols, potentially limiting their contribution to the measured antioxidant activity compared to less enriched formulations, such as C10FF.42,46 As with the flours, in vitro digestion led to a reduction in the soluble antioxidant activity of CW and C10CF bioaccessible fractions, by 2.5- and 1.2-fold, respectively, compared to their undigested forms (Fig. 1A, blue bars). In contrast, the TAC of C25CF, C10FF, and C25FF increased post-digestion by 2.9-, 1.4-, and 1.5-fold, respectively. In CW, in vitro digestion appeared to induce a reduction and/or transformation of antioxidant compounds associated with the cereal matrix, likely driven by digestive factors such as pH variations and interactions with other matrix components, including dietary fiber, as previously reported for cereal-based products.16,43 This result correlates with the modest antioxidant activity retained in the undigested residue fraction of CW.
In contrast, in chickpea-enriched and fermented formulations, digestion is consistent with an enhanced release of polyphenols from the legume–cereal matrix, leading to increased antioxidant activity in the bioaccessible fraction. These differences indicate that matrix structure and composition together with changes in formulations involving more complex matrices (chickpea) and the impact of thermal treatments, are key factors influencing the release of antioxidant compounds during digestion.18 Consistently, the residual fractions from digested crackers exhibited a promising radical scavenging activity (Fig. 1B). Notably, C10CF retained the highest TAC in the residual fraction, approximately 1.8 times greater than the other formulations, followed by C25FF and CW, which showed ∼1.2-fold higher values compared to the remaining samples. The high antioxidant activity in the residual fraction may result from the stabilization of polyphenols during digestion, potentially through interactions with proteins and peptides that entrap them within the insoluble matrix. Although not absorbed in the upper gastrointestinal tract, these compounds may reach the colon, where they can exert health benefits through interactions with the gut microbiota.27 In comparison with other fermented legume-based systems, the antioxidant activity of FF-enriched crackers was within the range of DPPH radical-scavenging activities reported for fermented legume beverages and non-baked matrices, such as legume-based sourdoughs and fermented soybean, lupin, pea, and bean products.41,47,48 For instance, Vurro et al.41 reported that focaccia bread enriched with pea-based sourdough at 40% exhibited DPPH radical-scavenging activity comparable in magnitude to that observed in the present FF-enriched cracker formulations with value below 1 µmol eq. Trolox per g samples. Taken together, these results indicate that fermented chickpea flour can contribute to measurable antioxidant activity in complex baked matrices, even at relatively low levels of inclusion.
3.2 Polyphenol bioaccessibility
In the present study, six phenolic compounds were identified in the flours, whereas four and nine of them were detected in the CW and cracker formulated with the innovative ingredients, respectively (Table 2).| Compound | CF | FF | CW | C10CF | C25CF | C10FF | C25FF |
|---|---|---|---|---|---|---|---|
| CF: cooked chickpea flour; FF: fermented cooked chickpea flour; CW: 100% wheat-based cracker; C10CF: 10% cooked chickpea flour-enriched cracker; C25CF: 25% cooked chickpea flour-enriched cracker; C10FF: 10% fermented cooked chickpea flour-enriched cracker; C25FF: 25% fermented cooked chickpea flour-enriched cracker. Data are shown as mean ± SD. Different letters indicate differences among the samples before digestion within the same row for each compound, as assessed by One-way ANOVA and Tukey's post hoc (p < 0.05). | |||||||
| p-Hydroxybenzoic acid | 6.8 ± 0.32b | 11.1 ± 0.6a | 2.8 ± 0.02d,e | 3.1 ± 0.1d,e | 3.4 ± 0.1d | 2.4 ± 0.03e | 4.8 ± 0.2c |
| Protocatechuic 4-O-glucoside | 27.3 ± 0.5c | 46.0 ± 0.4a | — | 9.7 ± 0.01f | 21.6 ± 0.3d | 10.9 ± 0.2e | 33.7 ± 0.61b |
| 4-Hydroxyphenyl acetic acid | 9.8 ± 0.2b | 21.1 ± 0.2a | — | 2.0 ± 0.2d | 2.9 ± 0.02c | 1.7 ± 0.1d | 2.8 ± 0.20c |
| Sinapoyl D-glucoside | 43.4 ± 0.8a | 41.2 ± 1b | — | 6.3 ± 0.1d | 8.4 ± 0.1c | 1.2 ± 0.03e | 5.44 ± 0.02d |
| Kampferol 3-O-glucoside | 3.9 ± 0.01b | 5.7 ± 0.05a | — | 1.9 ± 0.1e | 2.2 ± 0.1d,e | 2.1 ± 0.01d | 2.82 ± 0.01c |
| Pyrogallol | — | 123.5 ± 1.1a | — | — | — | 6.5 ± 0.04c | 11.6 ± 0.04b |
| Sinapic acid | — | — | 2.4 ± 0.1a | 1.6 ± 0.01b | 1.0 ± 0.1c | 0.7 ± 0.1d | 0.6 ± 0.05d |
| Ferulic acid | — | — | 5.8 ± 0.4a | 3.0 ± 0.04b | 2.4 ± 0.1c | 2.4 ± 0.02c | 1.6 ± 0.00d |
| Vanillic acid | — | — | 1.2 ± 0.1a | — | — | — | — |
| Total | 91.2 ± 0.8b | 248.6 ± 0.9a | 12.2 ± 0.2f | 27.7 ± 0.5e | 41.8 ± 0.3d | 27.8 ± 0.3e | 63.3 ± 0.6c |
The fermentation of chickpea flour increased the polyphenol content by approximately 2.7 times. Similarly, Sáez et al.24 and De Pasquale et al.36 reported that the solid-state fermentation of chickpea flours increased the polyphenol content by 2-fold compared to the control flours. In chickpea seeds, phenolic compounds are mainly in bound forms; therefore, the enzymes expressed by LABs (i.e., esterases, phenolic acid decarboxylases, and glycosidases) may have caused the breakdown and their release from the plant cell walls.20,49 Compared to the non-fermented flour, FF contained double the amount of p-hydroxybenzoic acid, 4-hydroxyphenyl acetic acid, kaempferol 3-O-glucoside, and protocatechuic 4-O-glucoside. Xu et al.50 reported that the glucoside of protocatechuic acid is one of the main phenolic compounds released from the cell wall matrix during chickpea germination and contributed to their higher antioxidant activity compared to non-germinated chickpeas. Conversely, synapoyl D-glucoside was found more abundant in CF flour and its derived-products. During fermentation new bioactive molecules can be produced by LABs. Indeed, pyrogallol was the predominant phenolic in FF but was absent in CF. This is likely due to the bioconversion of gallic acid, following tannin hydrolysis and subsequent decarboxylation mediated by LABs as previously reported.20
Consistent with the flour composition, the predominant phenolic compounds identified in the crackers were glucosides of sinapic acid and protocatechuic acid. The novel prototypes showed, on average, a 3-fold higher total polyphenol content compared to the conventional cracker (CW), with C25FF and C25CF exhibiting the highest concentrations, 1.8-fold and 1.2-fold greater than the other formulations, respectively. These findings suggest that the percentage of chickpea flour enrichment, particularly with FF, plays a critical role in enhancing the functional properties of the final product. As expected, crackers formulated with a higher inclusion level of chickpea flour (25%), both fermented and non-fermented, contained significantly more polyphenols than those enriched at 10%. However, in the undigested samples, the increase in total polyphenol content did not mirror the respective measured antioxidant activity. This observation aligns with previous reports indicating that the antioxidant capacity of plant-based foods cannot be attributed solely to total phenolic content, as synergistic interactions among polyphenols, and with other bioactive compounds, may influence the overall activity through different modes of interaction with the DPPH radical.51,52 Additionally, interactions between phenolic compounds and the food matrix can modulate their bioactivity. Within complex food systems, polyphenols may bind with dietary fibers, proteins, or carbohydrates, potentially enhancing or diminishing their antioxidant potential depending on the nature and strength of these interactions.53 The influence of matrix effects is also compound-specific, as shown by Sęczyk et al.,54 who reported a reduction in antioxidant activity in white bean–fortified pasta due to interactions between polyphenols and macromolecules in the pasta matrix. In the present study, no significant difference in total polyphenol content was observed between C10FF and C10CF (p > 0.05), indicating that fermentation at this lower inclusion level did not markedly affect overall phenolic release from the matrix. At lower inclusion levels, interactions between polyphenols and the wheat-based matrix may have limited phenolic release, thereby attenuating the differences between fermented and non-fermented flours.43,55
As shown in Table 3, in vitro digestion resulted in 1.3- to 4.1-fold increase in total polyphenols compared with the undigested products. The greatest release was observed for C10FF, followed by C25FF and FF, with increases of approximately 4.0-, 2.4-, and 1.9-fold, respectively. After digestion, protocatechuic and sinapic acid glucosides remained the predominant phenolic compounds, along with pyrogallol, whose bioaccessibility in FF-enriched crackers exceeded 100%. This effect may be explained by the release of additional phenolic compounds previously entrapped within the food matrix during digestion, driven by the physicochemical conditions of the gastrointestinal process, including pH changes and digestive enzyme activity.56 Inclusion of FF at both 10% and 25% substitution levels resulted in up to a twofold increase in polyphenol release during digestion compared with the corresponding unfermented formulations. This enhanced bioaccessibility may be attributed to the lower pH of the fermented matrix, resulting from acidification during fermentation which can promote disruption of complex matrix structures and facilitate the release and solubilization of bound polyphenols.57 Consistent with pre-digestion results, the conventional wheat-based cracker exhibited the lowest total polyphenol content in the bioaccessible fraction, further highlighting the functional advantage conferred by FF enrichment.
| Compound | CF | FF | CW | C10CF | C25CF | C10FF | C25FF |
|---|---|---|---|---|---|---|---|
| CF: cooked chickpea flour; FF: fermented cooked chickpea flour; CW: 100% wheat-based cracker; C10CF: 10% cooked chickpea flour-enriched cracker; C25CF: 25% cooked chickpea flour-enriched cracker; C10FF: 10% fermented cooked chickpea flour-enriched cracker; C25FF: 25% fermented cooked chickpea flour-enriched cracker. Data are shown as mean ± SD. Different letters indicate differences among the bioaccessible fractions after digestion within the same row for each compound, as assessed by One-way ANOVA and Tukey's post hoc (p < 0.05). | |||||||
| p-Hydroxybenzoic acid | 7.4 ± 0.4c | 30.8 ± 0.5a | 4.4 ± 0.1e | 7.6 ± 0.6c | 9.0 ± 0.1b | 6 ± 0.2d | 7.5 ± 0.06c |
| Protocatechuic 4-O-glucoside | 42.5 ± 0.3c | 200.2 ± 2a | — | 11.7 ± 0.1f | 30.1 ± 0.2d | 15.9 ± 0.2e | 53.1 ± 0.08b |
| 4-Hydroxyphenyl acetic acid | 21.5 ± 0.4b | 33.5 ± 0.42a | — | 2.7 ± 0.2e | 6 ± 0.04d | 2.6 ± 0.2e | 10.6 ± 0.3c |
| Sinapoyl D-glucoside | 61.1 ± 0.6b | 64.0 ± 0.2a | — | 7.2 ± 0.3e | 17.8 ± 0.03c | 6.5 ± 0.4e | 14.8 ± 0.9d |
| Kampferol 3-O-glucoside | 4.6 ± 0.04b | 7.5 ± 0.2a | — | 2.9 ± 0.2c | 3.2 ± 0.14c | 2.4 ± 0.2d | 4.4 ± 0.3b |
| Pyrogallol | — | 144.0 ± 0.04a | — | — | — | 76.9 ± 2.0b | 61.4 ± 4.4c |
| Sinapic acid | — | — | 1.6 ± 0.04a | 0.6 ± 0.02d | 0.5 ± 0.01d | 1.1 ± 0.00b | 0.9 ± 0.04c |
| Ferulic acid | — | — | 10.3 ± 0.2a | 5 ± 0.1b | 3.6 ± 0.00d | 4.2 ± 0.10c | 2.1 ± 0.02e |
| Vanillic acid | — | — | 1.1 ± 0.02a | — | — | — | — |
| Total | 137.2 ± 0.5c | 480.0 ± 2.5a | 17.4 ± 0.2f | 37.8 ± 0.2e | 70.1 ± 0.3d | 115.5 ± 2.6c | 154.0 ± 4b |
3.3 Potential antidiabetic effect in vitro: enzyme inhibition
The bioaccessible fractions were tested for the inhibitory activity towards the digestive enzymes α-amylase, α-glucosidase, and DPP-IV (Table 4).| Sample | α-Amylase | α-Glucosidase | DPP-IV |
|---|---|---|---|
| CF: cooked chickpea flour; FF: fermented cooked chickpea flour; CW: 100% wheat-based cracker; C10CF: 10% cooked chickpea flour-enriched cracker; C25CF: 25% cooked chickpea flour-enriched cracker; C10FF: 10% fermented cooked chickpea flour-enriched cracker; C25FF: 25% fermented cooked chickpea flour-enriched cracker. Data are shown as % of inhibition (mean ± SD). Different letters indicate differences among the bioaccessible fractions within the same column for each digestive enzyme assessed by One-way ANOVA and Tukey's post hoc (p < 0.05). | |||
| CF | 30.2 ± 1.6d | 6.1 ± 0.6b | 20.8 ± 0.08a |
| FF | 46.6 ± 0.2b | 8.4 ± 0.3a | 19.4 ± 0.2b |
| CW | — | — | 3.8 ± 0.3e |
| C10CF | 49.2 ± 1.1a | 3.4 ± 0.2c | 11.8 ± 0.1d |
| C25CF | 32.8 ± 0.3c | 4.1 ± 0.5c | 11.7 ± 0.1c |
| C10FF | 50.2 ± 0.5a | 4.0 ± 0.3c | 12.0 ± 0.1d |
| C25FF | 45.4 ± 0.1b | 8.3 ± 0.8a,b | 12.2 ± 0.1d |
All bioaccessible fractions from innovative crackers showed higher enzyme inhibition than control cracker, which was not effective in reducing the activity of α-amylase and α-glucosidase. The inhibition rate of α-amylase ranged from 30% to 50%. Similarly, Arnal et al.58 reported an inhibition rate of around 60% for the bioaccessible fractions of cooked chickpeas, whereas values of up to 30% were obtained for fermented chickpea-based beverage by Zhu et al.59 Interestingly, the 25% enrichment with both FF and CF led to lower α-amylase inhibitory activity compared with 10% enrichment, with the overall ranking being C10FF = C10CF > C25FF > C25CF. At higher levels of chickpea flour incorporation, the increased contents of starch, protein, polysaccharide, and dietary fiber may limit the release or accessibility of inhibitory compounds during digestion or promote non-specific interactions that reduce effective enzyme inhibition. Indeed, polyphenols are known to interact with proteins and soluble polysaccharides both before and after reaching the small intestine, thereby reducing their availability to interact with digestive enzymes and attenuating α-amylase inhibition.60 In addition, processing-induced compounds such as melanoidins may contribute to enzyme inhibition in a non-linear manner, partially masking dose-dependent effects attributable to polyphenols alone.61 Studies on fermented baked products, including sourdough breads, generally report α-amylase inhibition values below 20% following in vitro digestion.62 In this context, the α-amylase inhibition observed for the innovative crackers in the present study (approximately 30–50%), at both low and high inclusion levels, exceeds that commonly reported for fermented cereal-based baked products and may therefore be functionally relevant in modulating starch digestion. In contrast, all samples exhibited relatively weak α-glucosidase inhibitory activity, with values ranging from 3% to 8%. The highest inhibition was observed for FF (approximately 8%), followed by C25FF and CF, while no significant differences were detected among the other enriched crackers (p > 0.05). The slightly higher α-glucosidase inhibitory activity of FF and C25FF compared with their unfermented counterparts may be attributed to increased levels of glycosylated phenolic compounds, including glucosides of protocatechuic and sinapic acids, as well as kaempferol glucoside. This interpretation is consistent with the findings of Zhu et al.,59 who reported greater α-glucosidase inhibition for glycosylated polyphenol fractions than for free polyphenols in fermented chickpea-based beverages.
Among the tested flours, CF exhibited the highest DPP-IV inhibitory activity (21%), followed by FF (19%), with the difference being small but statistically significant. This result is consistent with our previous characterization of chickpea flours fermented with different LAB strains,20 in which unfermented flour contained a higher abundance of peptides with predicted DPP-IV inhibitory potential than most fermented counterparts. Although LAB fermentation is often reported to enhance the release of DPP-IV inhibitory peptides from legume substrates, the higher activity observed for CF may reflect more efficient peptide release during simulated gastrointestinal digestion or the effects of processing steps, such as mixing and thermal treatment, which can promote protein denaturation and the generation of short bioactive peptides.63–65 In enriched crackers, DPP-IV inhibitory activity was, on average, 3.2-fold higher than in the control wheat-based cracker (CW), with C25CF showing the strongest effect. These findings align with previous studies by Lammi et al.66 and Bollati et al.,67 which reported DPP-IV inhibition values ranging from 8% to 82% for legume protein hydrolysates, particularly from soybeans and peas. Chickpea proteins are especially rich in hydrophobic amino acids (approximately 35% of total residues), a feature that enhances their potential as DPP-IV inhibitors, given the enzyme's preference for peptide bonds adjacent to hydrophobic residues such as proline and alanine.68 Overall, DPP-IV inhibition is nutritionally relevant because it contributes to the preservation of incretin hormones, including glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1), which play a key role in postprandial glucose regulation. Collectively, these results support the potential of chickpea-based crackers to modestly modulate enzymes involved in postprandial glucose metabolism. Nevertheless, further in vivo and clinical studies are required to determine whether these in vitro effects translate into physiologically meaningful benefits relevant to glycaemic control and diabetes-related metabolic outcomes.
3.4 Cytotoxicity effect of bioaccessible fractions on Caco-2 cells
Caco-2 cells were exposed to both undiluted and serially diluted bioaccessible fractions to evaluate their potential cytotoxicity based on the effect on cellular viability. As shown in Fig. S2, neither diluted nor undiluted digests had cytotoxic effect on Caco-2 cells, with cell viability ranging from 68% to 197%. After 24 h and 48 h of exposure, cell viability was generally homogenous, except for FF, C10CF, and C25CF, which on average showed increased viability after 48 h compared to 24 h, by 1.1-, 1.4-, and 1.7-fold, respectively. Based on these results, undiluted bioaccessible fractions were selected to evaluate polyphenol bioavailability and their impact on Caco-2 cell layer permeability.3.5 Transport of polyphenols in Caco-2 cell line
To study the transport of polyphenols and their potential uptake, the bioaccessible fractions were incubated on Caco-2 cells for 4 h. Their concentrations (ng mL−1) at different transport time points, along with their overall bioavailability, are shown in Fig. 2A, B and C, respectively. As depicted in the heatmap, distinct transport patterns over time (rate) were observed among the various samples, likely determined both by the different matrices (ingredient vs. cracker) and by the specific phenolic compound profiles. CF exhibited peaks of total permeated polyphenols after 1 h and 3 h, while the fermented counterpart showed a gradual release over time with a maximum peak at 3 h. At the end of the transport (4 h), the FF had the highest amount of polyphenols permeated compared to CF. Among the monitored compounds, only p-hydroxybenzoic acid and pyrogallol were consistently detected across all time points aligning with previous findings.69Noteworthy, the majority of polyphenols were still present in the apical compartment after 4 h.
The presence in the apical compartment of different polyphenols, along with other compounds yet after 4 h, suggest a saturation of specific transporters in the cell monolayer, thereby hindering or preventing permeation to the basolateral side.70 The CF showed the highest bioavailability of p-hydroxybenzoic acid and sinapoyl D-glucoside, with values of 79% and 44%, respectively (Fig. 2A). Conversely, the FF exhibited higher bioavailability of 4-hydroxyphenylacetic acid (62%) compared to CF and contained pyrogallol, which was not detected in the unfermented counterpart. Interestingly, pyrogallol produced through the microbial fermentation, exhibited a bioavailability of approximately 65%, with the permeated concentration reaching a plateau between 2 h and 3 h. This finding is particularly relevant, as pyrogallol is known to reinforce the mucus barrier and attenuate gliadin-induced cytotoxicity and inflammation, in previous in vitro studies.71 Among the two ingredients, no significant differences were observed for protocatechuic 4-O-glucoside, showing the highest overall bioavailability among the phenolic compounds. Concerning the crackers, only 3 polyphenols out of 9 were detected following the transport in the basolateral compartment, with differences in abundance depending on both the inclusion level and type of flour (Fig. 2B and C).
Interestingly, for both fermented and unfermented chickpea flour enriched crackers, the predominant permeated polyphenols were the glucosides of protocatechuic and sinapic acids.
This observation is consistent with Guo et al.,72 who showed that lentil-derived polyphenols with a higher degree of glycosylation can permeate the Caco-2 monolayer via active transport mediated by the sodium-dependent glucose transporter 1 (SGLT1) and glucose transporter 2 (GLUT2), both of which are expressed in Caco-2 cells.
These transporters may actively aid in the absorption of mono-glucoside phenolics because of the preferential transportation of glucose/galactose moieties. Consequently, mono-glucoside phenolics have a faster and higher absorption rate than other phenolics lacking active transport and are regarded as competitive substrates for glucose transport by SGLT1 at the same time.73 Furthermore, aglycone polyphenols may interact with glycosidases expressed by Caco-2 cells, thereby potentially reducing their deglycosylation activity.74 However, further studies should investigate the specific transport mechanisms of these polyphenols. Consistent with what was observed in the bioaccessible fractions following digestion, the crackers formulated with FF, contained a higher amount of bioavailable polyphenols ranging from 40–80% compared to the wheat-based control (17%). Thus, incorporating the innovative ingredients to crackers led to a significant enhancement of the nutritional value of the final products, such effect was more pronounced in the prototypes made with the innovative FF. Indeed, at both inclusion levels of FF, the bioavailable fractions of the crackers showed appreciable amount of pyrogallol, whose concentration in the basolateral compartment ranged from 2–5 ng mL−1 and 8–10 ng mL−1 for C10FF and C25FF, respectively, showing a decreasing trend from 1 h to 4 h. Unlike the CF-enriched prototypes, the polyphenols from FF-based crackers showed a gradual permeation pattern over time, with a peak concentration after 4 h (7–32 ng mL−1). Specifically, the C25FF showed a gradual but sustained release of polyphenols over time probably due to the more complex matrix that slowed diffusion while prolonging availability. In general, the release of polyphenols was more abundant in prototypes with higher amount of innovative FF, suggesting that the transport kinetics and efficiency are matrix dependent.75 On the other hand, the crackers C10CF and C25CF, showed the highest bioavailability of sinapoyl D-glucoside (52% and 65%, respectively), mirroring the CF ingredient. Protocatechuic 4-O-glucoside was only found in crackers formulated with the highest inclusion levels (C25CF and C25FF), suggesting that, since it was not detected in the apical compartment after 4 h, enzymatic degradation or metabolic conversion may have occurred via enzymes expressed by Caco-2 cells, such as Phase II conjugation enzymes.76 The same metabolic fate may be hypothesized for 4-hydroxyphenylacetic acid, which was not identified in neither the apical nor basolateral compartments. Moreover, it may be hypothesized that following fermentation and/or digestion the breakdown of chemical bonds occurred along with the formation of new aggregates (dietary fiber–polyphenol, melanoidin–polyphenol, starch-polyphenols, or peptides–polyphenol), which may have limited the availability of polyphenols for cellular absorption.77–79 Such complexes may act as carriers, allowing polyphenols to reach the large intestine intact, where they may modulate gut microbial composition and promote the growth of beneficial bacteria such as Bifidobacterium and Lactobacillus.80 This hypothesis is also supported by the residual TAC observed in the crackers following digestion. Overall, this dual protective and prebiotic role extends the health potential of phenolic-rich matrices beyond their immediate bioavailability in the small intestine. Thus, these results demonstrate that the more complex food matrix can modulate polyphenol bioavailability beyond the structural characteristics of individual compounds.69
3.5 trans-Epithelial electrical resistance and Caco-2 barrier integrity
To assess whether the transport of polyphenols through the monolayer affected the permeability and therefore the integrity of the monolayer, TEER was measured at 0 h, 2 h, and 4 h. A higher TEER value corresponds to lower permeability, and vice versa; therefore, its measurements can provide insights into the modulation of tight junctions’ integrity.81The permeability-time curves in Fig. 3 show that the TEER values changed over time from baseline (0 h), ranging between 57% and 124%. After 2 h of exposure, cells treated with FF exhibited a greater reduction in TERR than those treated with CF, suggesting an increase in epithelial permeability. This effect became more pronounced after 4 h and may reflect the progressive permeation of polyphenols across the monolayer, with a maximum detected at 3 h. Consistently, a statistically significant inverse Pearson correlation was observed between the amount of permeated polyphenols and the TEER values over time (p < 0.05, r = −0.47).
These findings agree with previous studies reporting that specific flavonoids and flavan-3-ols readily diffuse across Caco-2 monolayers and increase paracellular permeability.82,83 Although sustained increases in epithelial permeability may theoretically facilitate the transport of both beneficial and potentially harmful compounds, the TEER variations observed in this study are indicative of a transient and functional modulation of tight junction dynamics rather than epithelial damage. This effect is likely associated with the action of phenolic compounds present in FF as previously described.84,85 Indeed, the modulation of tight junction proteins by polyphenols is considered to be temporary and functional rather than cytotoxic, which is consistent with the absence of cytotoxic effects observed in our experiments. The physiological activity of flavonoids has been linked to their ability to modulate intracellular signalling pathways and enzyme activities, thereby influencing tight junction dynamics and epithelial barrier function.86 In contrast, crackers exerted a milder effect on epithelial permeability. TEER values for C10CF and C10FF remained relatively stable over time, maintaining approximately 95% and 80% of their baseline values, respectively. Accordingly, these treatments induced only limited modulation of epithelial permeability, with no significant differences compared with the control (red dashed line; p > 0.05).
4. Conclusions
This study demonstrated that partially replacing wheat flour with cooked chickpea flour, particularly at a 25% inclusion level, either in its native or Lent. diolivorans 13-4A-fermented form, results in innovative crackers with enhanced nutritional properties. Specifically, the use of fermented cooked chickpea flour significantly increased total polyphenol content, improved polyphenol bioaccessibility, and conferred moderate inhibitory effects on digestive enzymes involved in glucose metabolism.Notably, pyrogallol emerged as a distinctive polyphenol in the fermented flour and its corresponding cracker formulations, highlighting the potential of fermentation to modify and enrich the polyphenolic profile of legume-based products. Alongside synapoyl D-glucoside and protocatechuic 4-O-glucoside, pyrogallol was also among the most bioaccessible and bioavailable compounds following digestion, reinforcing the functional benefits of fermentation.
Overall, these findings support the application of Lent. diolivorans 13-4A-fermented chickpea flour as a promising functional ingredient for the development of health-promoting baked goods. Further in vivo studies are warranted to confirm the physiological relevance and stability of the observed effects.
Author contributions
Manuela Flavia Chiacchio: investigation, writing – original draft, formal analysis, visualization. Silvia Tagliamonte: investigation, writing – review & editing, visualization. Angela Pazzanese: investigation. Giuseppe Blaiotta: conceptualization, resources, review & editing. Cristina Juan: conceptualization, resources, funding acquisition, review & editing. Ana Juan-Garcìa: conceptualization, resources, funding acquisition, review & editing. Paola Vitaglione: conceptualization, supervision, project administration, funding acquisition, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo05351j.Acknowledgements
National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.3 – Call for tender No. 341 of 15 March 2022 of Italian Ministry of University and Research funded by the European Union –Next Generation EU; Project code PE00000003, Concession Decree No. 1550 of 11 October 2022, Project title “ON Foods – Research and innovation network on food and nutrition Sustainability, Safety and Security-Working ON Foods”. Conselleria d'Educació, Universitats I Ocupació from Generalitat Valenciana project CIAICO2022/199.References
- Food technology magazine, https://www.ift.org/~/media/food%20technology/pdf/2018/04/0418_feat1_topten.pdf, (accessed November 2025).
- A. Bresciani, F. Vanara, E. Pagliarini, M. Locatelli, C. Proserpio, F. Travaglia and A. Marti, Effect of enrichment of rice snacks with pulse seed coats on phenolic compound, content product features and consumer hedonic response, Food Chem., 2023, 398, 133936 CrossRef CAS PubMed.
- P. Binou, A. E. Yanni and V. T. Karathanos, Physical properties, sensory acceptance, postprandial glycemic response, and satiety of cereal-based foods enriched with legume flours: a review, Crit. Rev. Food Sci. Nutr., 2022, 62, 2722–2740 CrossRef CAS PubMed.
- J. Hughes, E. Pearson and S. Grafenauer, Legumes—a comprehensive exploration of global food-based dietary guidelines and consumption, Nutrients, 2022, 14, 3080 CrossRef CAS PubMed.
- F. Boukid, E. Zannini, E. Carini and E. Vittadini, Pulses for bread fortification: a necessity or a choice?, Trends Food Sci. Technol., 2019, 88, 416–428 CrossRef CAS.
- G. Giuberti and A. Gallo, Reducing the glycaemic index and increasing the slowly digestible starch content in gluten-free cereal-based foods: a review, Int. J. Food Sci. Technol., 2018, 53, 50–60 CrossRef CAS.
- M. A. Augustin, J. Y. Chen and J. H. Ye, Processing to improve the sustainability of chickpea as a functional food ingredient, J. Sci. Food Agric., 2024, 104, 8397–8413 CrossRef CAS PubMed.
- A. Pasqualone, D. De Angelis, G. Squeo, G. Difonzo, F. Caponio and C. Summo, The effect of the addition of Apulian black chickpea flour on the nutritional and qualitative properties of durum wheat-based bakery products, Foods, 2019, 8, 504 CrossRef CAS PubMed.
- L. M. Cunha, S. C. Fonseca, R. C. Lima, J. Loureiro, A. S. Pinto, M. C. Vaz Patto and C. Brites, Consumer-driven improvement of maize bread formulations with legume fortification, Foods, 2019, 8, 235 CrossRef CAS PubMed.
- D. Guardado-Félix, M. A. Lazo-Vélez, E. Pérez-Carrillo, D. E. Panata-Saquicili and S. O. Serna-Saldívar, Effect of partial replacement of wheat flour with sprouted chickpea flours with or without selenium on physicochemical, sensory, antioxidant and protein quality of yeast-leavened breads, LWT, 2020, 129, 109517 CrossRef.
- S. Shaabani, M. S. Yarmand, H. Kiani and Z. Emam-Djomeh, The effect of chickpea protein isolate in combination with transglutaminase and xanthan on the physical and rheological characteristics of gluten free muffins and batter based on millet flour, LWT, 2018, 90, 362–372 CrossRef CAS.
- D. E. Garcia-Valle, L. A. Bello-Pérez, E. Agama-Acevedo and J. Alvarez-Ramirez, Structural characteristics and in vitro starch digestibility of pasta made with durum wheat semolina and chickpea flour, LWT, 2021, 145, 111347 CrossRef CAS.
- A. Saint-Eve, P. Granda, G. Legay, G. Cuvelier and J. Delarue, Consumer acceptance and sensory drivers of liking for high plant protein snacks, J. Sci. Food Agric., 2019, 99, 3983–3991 CrossRef CAS PubMed.
- J. A. Curiel, R. Coda, I. Centomani, C. Summo, M. Gobbetti and C. G. Rizzello, Exploitation of the nutritional and functional characteristics of traditional Italian legumes: The potential of sourdough fermentation, Int. J. Food Microbiol., 2015, 196, 51–61 CrossRef CAS PubMed.
- C. G. Rizzello, R. Coda and M. Gobbetti, Use of sourdough fermentation and nonwheat flours for enhancing nutritional and healthy properties of wheat-based foods, in Fermented foods in health and disease prevention, Academic Press, 2017, Chapter 18, pp. 433–452 Search PubMed.
- F. Shahidi and Y. Pan, Influence of food matrix and food processing on the chemical interaction and bioaccessibility of dietary phytochemicals: a review, Crit. Rev. Food Sci. Nutr., 2022, 62, 6421–6445 CrossRef CAS PubMed.
- G. Catalkaya, K. Venema, L. Lucini, G. Rocchetti, D. Delmas, M. Daglia and E. Capanoglu, Interaction of dietary polyphenols and gut microbiota: microbial metabolism of polyphenols, influence on the gut microbiota, and implications on host health, Food Front., 2020, 1, 109–133 CrossRef.
- C. Dima, E. Assadpour, S. Dima and S. M. Jafari, Bioavailability of nutraceuticals: role of the food matrix, processing conditions, the gastrointestinal tract, and nanodelivery systems, Compr. Rev. Food Sci. Food Saf., 2020, 19, 954–994 CrossRef CAS PubMed.
- Z. Kohajdová, J. Karovicova and M. Magala, Utilisation of chickpea flour for crackers production, Acta Chim. Slovaca, 2011, 4, 98–107 Search PubMed.
- M. F. Chiacchio, S. Tagliamonte, A. Pazzanese, P. Vitaglione and G. Blaiotta, Lactic acid fermentation improves nutritional and functional properties of chickpea flours, Food Res. Int., 2025, 115899 CrossRef CAS PubMed.
- S. Tiwari, D. Kavitake, P. B. Devi and P. H. Shetty, Bacterial exopolysaccharides for improvement of technological, functional and rheological properties of yoghurt, Int. J. Biol. Macromol., 2021, 183, 1585–1595 CrossRef CAS PubMed.
- M. P. Gangola, B. R. Ramadoss, S. Jaiswal, H. Fabek, M. Tulbek, G. H. Anderson and R. N. Chibbar, Nutritional composition and in vitro starch digestibility of crackers supplemented with faba bean whole flour, starch concentrate, protein concentrate and protein isolate, Foods, 2022, 11, 645 CrossRef CAS PubMed.
- N. Grasso, N. L. Lynch, E. K. Arendt and J. A. O'Mahony, Chickpea protein ingredients: a review of composition, functionality, and applications, Compr. Rev. Food Sci. Food Saf., 2022, 21, 435–452 CrossRef CAS PubMed.
- G. D. Sáez, C. Sabater, A. Fara and G. Zárate, Fermentation of chickpea flour with selected lactic acid bacteria for improving its nutritional and functional properties, J. Appl. Microbiol., 2022, 133, 181–199 CrossRef PubMed.
- European Commission. Regulation (EC) No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods, https://data.europa.eu/eli/reg/2006/1924/oj, (accessed on November 2025).
- A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assunção and S. Ballance, et al., INFOGEST static in vitro simulation of gastrointestinal food digestion, Nat. Protoc., 2019, 14, 991–1014 CrossRef CAS PubMed.
- A. Hamzalioglu, S. Tagliamonte, V. Gökmen and P. Vitaglione, Casein–phenol interactions occur during digestion and affect bioactive peptide and phenol bioaccessibility, Food Funct., 2023, 14, 9457–9469 RSC.
- P. Llorens, M. F. Chiacchio, S. Tagliamonte, A. Juan-García, N. Pallarés and J. C. Moltó, et al., Potential bioaccessibility and bioavailability of polyphenols… of tiger nut beverage…, Food Funct., 2024, 15, 8143–8152 RSC.
- H. You, T. Wu, W. Wang, Y. Li, X. Liu and L. Ding, Preparation and identification of dipeptidyl peptidase IV inhibitory peptides from quinoa protein, Food Res. Int., 2022, 156, 111176 CrossRef CAS PubMed.
- A. Juan-García, D. Montesano, J. Mañes and C. Juan, Cytoprotective effects of carotenoid-rich extract from Lycium barbarum L. on beauvericin-induced cytotoxicity in Caco-2 cells, Food Chem. Toxicol., 2019, 133, 110798 CrossRef PubMed.
- A. Bocsik, I. Gróf, L. Kiss, F. Ötvös, O. Zsíros and L. Daruka, et al., Dual action of the PN159/KLAL/MAP peptide: Increase of drug penetration across caco-2 intestinal barrier model by modulation of tight junctions and plasma membrane permeability, Pharmaceutics, 2019, 11, 73 CrossRef CAS PubMed.
- Z. Gu, R. Eils and M. Schlesner, Complex heatmaps reveal patterns and correlations in multidimensional genomic data, Bioinformatics, 2016, 32, 2847–2849 CrossRef CAS PubMed.
- J. Krooneman, F. Faber, A. C. Alderkamp, S. O. Elferink, F. Driehuis and I. Cleenwerck, et al., Lactobacillus diolivorans sp. nov., a 1, 2-propanediol-degrading bacterium isolated from aerobically stable maize silage, Int. J. Syst. Evol. Microbiol., 2002, 52, 639–646 CrossRef CAS PubMed.
- J. Samelis and A. Kakouri, Microbiological characterization of Greek Galotyri cheese PDO products relative to fresh or ripened marketing, Fermentation, 2022, 8, 492 CrossRef CAS.
- K. Skrzypczak, D. Teterycz, W. Gustaw, D. Domagała, P. Mielczarek and J. Kasprzyk-Pochopień, The possibility of using Lactobacillus plantarum, 299v to reinforce the bioactive properties of legume-derived beverages, Appl. Sci., 2024, 14, 5187 CrossRef CAS.
- I. De Pasquale, E. Pontonio, M. Gobbetti and C. G. Rizzello, Nutritional and functional effects of lactic acid bacteria fermentation on gelatinized legume flours, Int. J. Food Microbiol., 2020, 316, 108426 CrossRef CAS PubMed.
- S. Zhou, Y. Wang, Q. Hu, J. Li, J. Chen and X. Liu, Enhancement of nutritional quality of chickpea flour by solid-state fermentation for improvement of in vitro antioxidant activity and protein digestibility, Food Chem., 2025, 468, 142418 CrossRef CAS PubMed.
- M. Emkani, B. Oliete and R. Saurel, Effect of lactic acid fermentation on legume protein properties: a review, Fermentation, 2022, 8, 244 CrossRef CAS.
- S. Garrido-Galand, A. Asensio-Grau, J. Calvo-Lerma, A. Heredia and A. Andrés, The potential of fermentation on nutritional and technological improvement of cereal and legume flours: a review, Food Res. Int., 2021, 145, 110398 CrossRef CAS PubMed.
- D. Virág, A. Kiss, P. Forgó, C. Csutorás and S. Molnár, Study on Maillard-reaction driven transformations and increase of antioxidant activity in lysine fortified biscuits, Microchem. J., 2013, 107, 172–177 CrossRef.
- F. Vurro, M. Santamaria, C. Summo, A. Pasqualone and C. M. Rosell, Exploring the functional potential of pea-based sourdough in traditional durum wheat focaccia: role in enhancing bioactive compounds, in vitro antioxidant activity, in vitro digestibility and aroma, J. Funct. Foods, 2024, 123, 106607 CrossRef CAS.
- M. Patrignani, L. Del Sol González-Forte, A. A. P. Pierri, J. M. Awika and P. A. Conforti, Interaction between Maillard reaction products and phenolics in brewer’s spent grain: structural and antioxidant insights, Food Res. Int., 2025, 117643 CrossRef CAS PubMed.
- J. Pérez-Jiménez, M. E. Díaz-Rubio, M. Mesías, F. J. Morales and F. Saura-Calixto, Evidence for the formation of maillardized insoluble dietary fiber in bread: a specific kind of dietary fiber in thermally processed foods, Food Res. Int., 2014, 55, 391–396 CrossRef.
- M. Verni, V. Verardo and C. G. Rizzello, How fermentation affects the antioxidant properties of cereals and legumes, Foods, 2019, 8, 362 CrossRef CAS PubMed.
- Y. Xiao, L. Huang, Y. Chen, S. Zhang, X. Rui and M. Dong, Comparative study of the effects of fermented and non-fermented chickpea flour addition on quality and antioxidant properties of wheat bread, CyTA J. Food, 2016, 14, 621–631 CrossRef CAS.
- S. Shaheen, M. Shorbagi, J. M. Lorenzo and M. A. Farag, Dissecting dietary melanoidins: formation mechanisms, gut interactions and functional properties, Crit. Rev. Food Sci. Nutr., 2022, 62(32), 8954–8971 CrossRef CAS PubMed.
- J. A. Marazza, M. A. Nazareno, G. S. de Giori and M. S. Garro, Enhancement of the antioxidant capacity of soymilk by fermentation with Lactobacillus rhamnosus, J. Funct. Foods, 2012, 4, 594–601 CrossRef CAS.
- J. A. Curiel, R. Coda, I. Centomani, C. Summo, M. Gobbetti and C. G. Rizzello, Exploitation of the nutritional and functional characteristics of traditional Italian legumes: the potential of sourdough fermentation, Int. J. Food Microbiol., 2015, 196, 51–61 CrossRef CAS PubMed.
- A. C. de Camargo, A. C. Concepción Alvarez, M. F. Arias-Santé, J. E. Oyarzún, M. E. Andia and S. Uribe, et al., Soluble free, esterified and insoluble-bound phenolic antioxidants from chickpeas prevent cytotoxicity in human hepatoma HuH-7 cells induced by peroxyl radicals, Antioxidants, 2022, 11, 1139 CrossRef CAS PubMed.
- M. Xu, Z. Jin, J. B. Ohm, P. Schwarz, J. Rao and B. Chen, Improvement of the antioxidative activity of soluble phenolic compounds in chickpea by germination, J. Agric. Food Chem., 2018, 66, 6179–6187 CrossRef CAS PubMed.
- S. J. Hur, S. Y. Lee, Y. C. Kim, I. Choi and G. B. Kim, Effect of fermentation on the antioxidant activity in plant-based foods, Food Chem., 2014, 160, 346–356 CrossRef CAS PubMed.
- S. Ketnawa, F. C. Reginio Jr, S. Thuengtung and Y. Ogawa, Changes in bioactive compounds and antioxidant activity of plant-based foods by gastrointestinal digestion: a review, Crit. Rev. Food Sci. Nutr., 2022, 62, 4684–4705 CrossRef CAS PubMed.
- Y. Feng, C. Jin, S. Lv, H. Zhang, F. Ren and J. Wang, Molecular mechanisms and applications of polyphenol-protein complexes with antioxidant properties: a review, Antioxidants, 2023, 12, 1577 CrossRef CAS PubMed.
- Ł. Sęczyk, U. Gawlik-Dziki and M. Świeca, Influence of phenolic-food matrix interactions on in vitro bioaccessibility of selected phenolic compounds and nutrients digestibility in fortified white bean paste, Antioxidants, 2021, 10, 1825 CrossRef PubMed.
- E. Nemli, G. Ozkan, B. Gultekin Subasi, H. Cavdar, J. M. Lorenzo, C. Zhao and E. Capanoglu, Interactions between proteins and phenolics: effects of food processing on the content and digestibility of phenolic compounds, J. Sci. Food Agric., 2024, 104, 2535–2550 CrossRef CAS PubMed.
- G. Quintana, V. Spínola, G. N. Martins, E. Gerbino, A. Gómez-Zavaglia and P. C. Castilho, Release of health-related compounds during in vitro gastro-intestinal digestion of okara and okara fermented with Lactobacillus plantarum, J. Food Sci. Technol., 2020, 57, 1061–1070 CrossRef CAS PubMed.
- J. A. Adebo, P. B. Njobeh, S. Gbashi, A. B. Oyedeji, O. M. Ogundele, S. A. Oyeyinka and O. A. Adebo, Fermentation of cereals and legumes: impact on nutritional constituents and nutrient bioavailability, Fermentation, 2022, 8, 63 CrossRef CAS.
- M. Arnal, M. Gallego, P. Talens and L. Mora, Peptidomic profile and α-glucosidase inhibitory activity of cooked and gastrointestinal digested legumes, LWT, 2024, 201, 116283 CrossRef CAS.
- M. X. Zhu, C. Xu and Y. P. Hao, Lactobacillus fermentation improved the nutrient value and hypoglycemic activity of chickpea milk, Emir. J. Food Agric., 2023, 35, 242–250 Search PubMed.
- L. Sun and M. Miao, Dietary polyphenols modulate starch digestion and glycaemic level: a review, Crit. Rev. Food Sci. Nutr., 2020, 60, 541–555 CrossRef CAS PubMed.
- Z. Wang, C. Gao, Q. Chen, K. Wang and B. Fan, A comprehensive perspective on melanoidins: definitions, polymerization, chromogenic mechanism, cross-linking and bioactivity, Food Chem. Adv., 2025, 9, 101181 CrossRef.
- A. Diowksz, A. Malik, A. Jaśniewska and J. Leszczyńska, The inhibition of amylase and ACE enzyme and the reduction of immunoreactivity of sourdough bread, Foods, 2020, 9, 656 CrossRef CAS PubMed.
- M. F. Quintero-Soto, J. Chávez-Ontiveros, J. A. Garzón-Tiznado, N. Y. Salazar-Salas, K. V. Pineda-Hidalgo, F. Delgado-Vargas and J. A. López-Valenzuela, Characterization of peptides with antioxidant activity and antidiabetic potential obtained from chickpea (Cicer arietinum L.) protein hydrolyzates, J. Food Sci., 2021, 86, 2962–2977 CrossRef CAS PubMed.
- R. Indrati, Bioactive peptides from legumes and their bioavailability, in Legumes Research – Volume 2, IntechOpen, 2021 Search PubMed.
- N. Begum, Q. U. Khan, L. G. Liu, W. Li, D. Liu and I. U. Haq, Nutritional composition, health benefits and bio-active compounds of chickpea (Cicer arietinum L.), Front. Nutr., 2023, 10, 1218468 CrossRef PubMed.
- C. Lammi, C. Bollati, S. Ferruzza, G. Ranaldi, Y. Sambuy and A. Arnoldi, Soybean- and lupin-derived peptides inhibit DPP-IV activity on in situ human intestinal Caco-2 cells and ex vivo human serum, Nutrients, 2018, 10, 1082 CrossRef PubMed.
- C. Bollati, R. Xu, G. Boschin, M. Bartolomei, F. Rivardo and J. Li, et al., Integrated evaluation of the multifunctional DPP-IV and ACE inhibitory effect of soybean and pea protein hydrolysates, Nutrients, 2022, 14, 2379 CrossRef CAS PubMed.
- A. B. Nongonierma and R. J. FitzGerald, Features of dipeptidyl peptidase IV (DPP-IV) inhibitory peptides from dietary proteins, J. Food Biochem., 2019, 43, e12451 CrossRef PubMed.
- G. Hithamani, D. Kizhakayil and K. Srinivasan, Uptake of phenolic compounds from plant foods in human intestinal Caco-2 cells, J. Biosci., 2017, 42, 603–611 CrossRef CAS PubMed.
- E. Elejalde, R. M. Alonso, M. C. Villarán, L. Díez-Gutiérrez, M. Chávarri and I. López-de-Armentia, Exploring the bioavailability of red grape skin extract polyphenols: a Caco-2 cell model study, Foods, 2025, 14, 2253 CrossRef CAS PubMed.
- G. Feng, K. Han, Q. Yang, W. Feng, J. Guo, J. Wang and X. Yang, Interaction of pyrogallol-containing polyphenols with mucin reinforces intestinal mucus barrier properties, J. Agric. Food Chem., 2022, 70, 9536–9546 CrossRef CAS PubMed.
- F. Guo, L. Peng, H. Xiong, R. Tsao, H. Zhang, L. Jiang and Y. Sun, Bioaccessibility and transport of lentil hull polyphenols in vitro, and their bioavailability and metabolism in rats, Food Res. Int., 2023, 167, 112634 CrossRef CAS PubMed.
- F. Shahidi and H. Peng, Bioaccessibility and bioavailability of phenolic compounds, J. Food Bioact., 2018, 4, 11–68 CrossRef.
- Y. Wang, S. Lan, L. Zhang, Y. Li, Z. Deng and X. Ye, et al., Mucus barrier weakens the inhibitory activity of pyrogallol-based polyphenols against α-glucosidase, Mol. Nutr. Food Res., 2025, 69, e202400838 CrossRef CAS PubMed.
- B. Ed Nignpense, N. Francis, C. Blanchard and A. B. Santhakumar, Effect of gastrointestinal digestion on the stability, antioxidant activity, and Caco-2 cellular transport of pigmented grain polyphenols, J. Food Sci., 2024, 89, 2701–2715 CrossRef CAS PubMed.
- R. B. Van Breemen and Y. Li, Caco-2 cell permeability assays to measure drug absorption, Expert Opin. Drug Metab. Toxicol., 2005, 1, 175–185 CrossRef CAS PubMed.
- G. Rocchetti, R. P. Gregorio, J. M. Lorenzo, F. J. Barba, P. G. Oliveira and M. A. Prieto, et al., Functional implications of bound phenolic compounds and phenolics–food interaction: a review, Compr. Rev. Food Sci. Food Saf., 2022, 21, 811–842 CrossRef CAS PubMed.
- F. Shahidi and C. S. Dissanayaka, Phenolic-protein interactions: insight from in-silico analyses–a review, Food Prod., Process. Nutr., 2023, 5, 2 CrossRef CAS.
- M. Cai, J. Feng, J. Wang, P. Chen, Z. Ge and W. Liu, et al., Characterization of various noncovalent polyphenol–starch complexes and their prebiotic activities during in vitro digestion and fermentation, J. Agric. Food Chem., 2024, 72, 2250–2262 CrossRef CAS PubMed.
- Y. Wu, Y. Liu, Y. Jia, H. Zhang and F. Ren, Formation and application of starch–polyphenol complexes: influencing factors and rapid screening based on chemometrics, Foods, 2024, 13, 1557 CrossRef CAS PubMed.
- Y. Ohno, T. Naganuma, T. Ogawa and K. Muramoto, Effect of lectins on the transport of food factors in Caco-2 cell monolayers, J. Agric. Food Chem., 2006, 54, 548–553 CrossRef CAS PubMed.
- M. N. Chabane, A. A. Ahmad, J. Peluso, C. D. Muller and G. Ubeaud-Séquier, Quercetin and naringenin transport across human intestinal Caco-2 cells, J. Pharm. Pharmacol., 2009, 61, 1473–1483 CrossRef CAS PubMed.
- T. Suzuki and H. Hara, Role of flavonoids in intestinal tight junction regulation, J. Nutr. Biochem., 2011, 22, 401–408 CrossRef CAS PubMed.
- T. Mukherjee, E. Squillante, M. Gillespie and J. Shao, Transepithelial electrical resistance is not a reliable measurement of the Caco-2 monolayer integrity in Transwell, Drug Delivery, 2004, 11, 11–18 CrossRef CAS PubMed.
- H. Sun, E. C. Chow, S. Liu, Y. Du and K. S. Pang, The Caco-2 cell monolayer: usefulness and limitations, Expert Opin. Drug Metab. Toxicol., 2008, 4, 395–411 CrossRef CAS PubMed.
- S. Noda, S. Tanabe and T. Suzuki, Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells, J. Agric. Food Chem., 2012, 60, 4628–4633 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |