Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Prune (dried plum) consumption does not reduce colonic tumor formation but drives beneficial changes in the gut microbiome of rats
Minghong
Jiang
 a,
Andres
Gomez
b,
Davis M.
Seelig
c and
Daniel D.
Gallaher
a,
Andres
Gomez
b,
Davis M.
Seelig
c and
Daniel D.
Gallaher
 *a
*a
aDepartment of Food Science and Nutrition, University of Minnesota, St. Paul, MN 55108, USA. E-mail: dgallahe@umn.edu
bDepartment of Animal Science, University of Minnesota, St. Paul, MN 55108, USA
cDepartment of Clinical Pathology, Veterinary Clinical Sciences, University of Minnesota, St. Paul, MN 55108, USA
First published on 20th March 2026
Abstract
Previous research has highlighted the potential benefits of prune consumption, including a changed gut microbiome composition and a reduction in colon cancer risk factors. This study investigated whether prune consumption reduced colon tumor development and led to positive changes in the large intestinal microbiome in a chemically induced colon cancer model in rats. Male Wistar rats were fed one of three diets: 5% (by energy) prune, 10% prune, or a prune-free control. Rats were fed the diets for 32 weeks. Rats received weekly injections of 1,2-dimethylhydrazine for 15 weeks to induce colon tumorigenesis. Colonic tumor number or size did not differ among the diet groups. However, there was a trend toward fewer small intestinal tumors in the 10% prune diet group (p < 0.1). Groups fed prune had heavier cecum tissue, indicating greater large intestinal fermentation. The prune diets increased taxonomic richness and altered bacterial species composition. Specifically, prune consumption was associated with increased abundance of Methanosphaera genus and taxa from the Lachnospiraceae family, such as Blautia and Coprococcus. Prune diets also increased total cecal SCFA amount, notably butyrate. However, 24 hour fecal excretion of p-cresol, indole, and total bile acids did not differ significantly among the groups. While prune consumption did not show a significant reduction in colonic tumor formation, potential benefits were noted in a trend towards reducing small intestine tumors, increasing large intestinal fermentation and SCFA production, and increasing microbial richness, suggesting prune consumption may provide other health benefits.
1. Introduction
Colorectal cancer (CRC) is the third leading cause of cancer-related mortality in the United States and the second globally.1,2 The incidence of CRC has increased rapidly in countries that have adopted a Westernized diet. The Western diet, characterized by the consumption of large amounts of red and processed meat, refined grains, and sugar, coupled with low intake of whole grains, fruits, and vegetables has been associated with dysbiosis of the gut microbiome and inflammation in the gut, thus contributing to the development of CRC.3 Due to advancements in treatment and screening, there has been a 57% decline in overall CRC mortality in the U.S. from 1970 to 2020. However, there is a concerning recent trend of an upward trajectory in CRC cases in individuals under 50 years old, accompanied by a significant shift towards advanced disease stages.Fruit intake has been inversely associated with CRC risk, but most epidemiological studies report only total fruit intake rather than specific types, making it difficult to assess the chemopreventive potential of individual fruits.4,5 Prunes, recognized as a functional food, offer various health benefits, including alleviating constipation, lowering cholesterol, and improving bone density. Derived from the dried fruit of Prunus domestica, prunes are rich in dietary fiber, sorbitol, and phytochemicals such as polyphenols and carotenoids.6 However, compared to more commonly studied fruits like citrus and apples, prunes (dried plums) are less frequently consumed and even less studied, leaving their chemopreventive effects largely unknown.5
Yet there is evidence that prune consumption may reduce colon cancer risk. In carcinogen-treated rats, feeding a plum beverage reduced inflammation and colonic pre-neoplastic lesions known as aberrant crypt foci (ACF), which have been commonly used as an early biomarker of colon carcinogenesis. Polyphenols extracted from prunes have been shown to exhibit strong antioxidant and anti-inflammatory properties,7,8 reducing two established hallmarks of cancer.9 In cancer cell lines, prune extracts or isolated polyphenols induced apoptosis and suppressed cell proliferation,10,11 indicating potential chemopreventive effects against colonic tumor formation. However, feeding a prune powder in another study did not show a reduction in ACF, although changes in other colorectal cancer risk factors, including reduced total and secondary bile acids, were observed.12,13 Due to these inconsistent findings, it is uncertain whether prunes or prune products will reduce the risk of colon cancer.
There is also growing interest in the role of the gut microbiome in the development of CRC. CRC patients have lower microbial diversity and different fecal microbial population patterns compared to healthy individuals.14 Prunes exert beneficial effects on the gut ecosystem, likely due to its being a rich source of fiber, with 40–80% of the fiber being pectin, and a high concentration of insoluble fibers, polyphenols, and sorbitol.6 These nutrients undergo fermentation by the gut microbiota in the large intestine, leading to increased production of short chain fatty acids (SCFAs), including butyrate, which is an important energy source for the colonic epithelium. Butyrate has also been found to reduce CRC risk.15,16 Recent research involving prune supplementation in human studies revealed significant shifts in microbial composition in prune consumption groups.17,18 Studies found that prune consumption increased bacterial populations typically regarded as beneficial, such as Bifidobacterium and Lactobacillus, while reducing harmful bacteria such as Escherichia coli and Clostridium perfringens (C. perfringens).19,20 The increased abundance of the Lachnospiraceae family was correlated with a reduced plasma concentration of the inflammatory cytokine IL-1β, suggesting a positive effect of prune consumption in modulating the gut microbiome and reducing systemic inflammation.17
These observations support the chemopreventive potential of prune consumption. Therefore, we hypothesized that diets containing prunes would reduce dimethylhydrazine-induced colonic tumors in rats. Furthermore, given the role of the gut microbiome in modulating colon cancer risk,21 we analyzed bacterial abundance in the large intestine as well as determined bacterial metabolites implicated in colon cancer risk, including short chain fatty acids, bile acids, and markers of inflammation.
2. Materials and methods
2.1. Animals and diet
Eighty-one male Wistar rats, initially weighing between 100 and 170 g, were purchased from Envigo (Indianapolis, IN). The rats were housed in pairs and maintained under a 12 hour light/dark cycle, with ad libitum access to food and water. All animal handling procedures were conducted in accordance with the guidelines set by the National Institutes of Health and met the institutional standards for animal care. The experimental protocol was approved by the University of Minnesota Animal Care and Use Committee (protocol #2012-38704A).The prune product (hereafter referred to as prune) was provided by Sunsweet Growers Inc. (Yuba City, CA). It was prepared by blending 75% prune concentrate with 25–30% prune paste by weight. The prune concentrate was produced by removing pulp and pits, followed by vacuum evaporation until reaching 68° Brix, while the prune paste was made from steamed plums adjusted to 22–25% moisture. The prune used in this study had a moisture content of approximately 29.8%. The primary compositional difference between the prune and whole prunes was the fiber content. The prune contained more soluble fiber (6.4 g vs. 2.1 g per 100 g) and less insoluble fiber (1.1 g vs. 4.1 g per 100 g) compared with whole prune but was otherwise similar. The macronutrient composition of the prune, whole prunes, prune juice, and fresh plums is shown in Table S1.
All diets were formulated to provide equal macronutrient and total energy levels (Table 1). The pectin concentrations in the 5% and 10% prune diets were 0.28% and 0.56%, respectively, while sorbitol concentrations were 1.15% and 2.30%, respectively. The basal diet contained cellulose as the sole fiber source.
| Basal | 5% prune | 10% prune | |
|---|---|---|---|
| Ingredients (g kg −1 ) | |||
| Dried prune puree | 0 | 76 | 154 |
| Sucrose | 100 | 73 | 66 |
| Maltodextrin | 132 | 132 | 132 |
| Cornstarch | 368 | 368 | 368 |
| Casein | 200 | 198 | 196 |
| Soybean oil | 70 | 70 | 70 |
| Cellulose | 80 | 73 | 66 |
| AIN-93G mineral mix | 35 | 35 | 35 |
| AIN-93G vitamin mix | 10 | 10 | 10 |
| L-Cystine | 3 | 3 | 3 |
| Choline bitartrate | 2.5 | 2.5 | 2.5 |
| TBHQ | 0.014 | 0.014 | 0.014 |
| Macronutrient composition (% of total energy) | |||
| Carbohydrate (% of energy) | 62.6 | 62.6 | 62.6 |
| Fat (% of energy) | 16.5 | 16.4 | 16.4 |
| Protein (% of energy) | 20.9 | 20.9 | 20.9 |
| Prune pectin and sorbitol (% of energy) | 0 | 1.4 | 2.8 |
| Total energy (kcal g−1) | 3.8 | 3.7 | 3.5 |
The prune was incorporated into the AIN-93G diet21 at concentrations providing 5% and 10% of total energy. The prune was thoroughly mixed with other dry ingredients and soybean oil using a commercial mixer to achieve a uniform texture. The diets were fed as a powder and stored at 4 °C.
2.2. Experimental design
Rats were randomly assigned to one of the three diets. After a two-week adaptation period to their respective diets, the rats were administered a weekly subcutaneous injection of dimethylhydrazine (20 mg per kg body weight) for 15 consecutive weeks. Following the completion of all injections, the rats were placed in metabolic cages for 24 hours to collect urine and fecal samples, which were subsequently stored at −80 °C. The rats continued to be fed their respective diets for an additional 15 weeks until the end of the feeding trial. Throughout the study, food intake and body weight were recorded weekly.At the end of the 32 week period, the rats were fasted overnight and then anesthetized the next morning using isoflurane. Blood was collected through cardiocentesis and the liver, epididymal fat pad, and cecum were excised, rinsed of adhering blood, and weighed. Cecum contents were collected, and the empty cecum was weighed. Tissues were rapidly frozen in liquid nitrogen before being stored at −80 °C. Serum was separated and stored at −20 °C for subsequent analysis.
The colon was dissected from the anus to the cecum and opened longitudinally, and colon pellets were collected and snap-frozen in liquid nitrogen before storage at −80 °C. Tumors were counted and tumor volume calculated using the formula V = length × width × diameter × π/6, where length is the longest dimension, width is the middle dimension, perpendicular to length, and diameter is the smallest dimension, perpendicular to both length and width.22 Following tumor excision, each tumor with a segment of normal colonic interstitial tissue was fixed in 10% neutral buffered formalin for 72 hours and then transferred to 70% ethanol for histopathological analysis. The remaining colon tissue was placed on an ice-cold metal tray, and the mucosa was carefully scraped off using a glass slide. The resulting mucosal scraps were snap-frozen in liquid nitrogen and stored at −80 °C for subsequent measurement of inflammatory markers.
2.3. Tumor histopathologic analysis
The formalin-fixed colon tissue underwent a standard processing procedure, involving dehydration through a series of graded alcohols, followed by embedding in paraffin wax. Subsequently, 4 µm tissue sections were cut and stained with hematoxylin and eosin. The stained tissue sections were examined by a board-certified veterinary pathologist according to established consensus guidelines on the pathology of rodent models of intestinal cancer.23,24 The pathologist was blinded to treatment groups during the assessment of the proliferative lesions. Lesions were categorized as hyperplasia, adenoma, intramucosal adenocarcinoma, or adenocarcinoma, based on specific morphological criteria and cellular characteristics observed in the tissue sections.23,252.4. Analysis of fecal p-cresol and indole
Fecal pellets collected during housing in the metabolic cages were freeze-dried and ground into a fine powder. Five mL of methanol and 100 μl of internal standard (300 μg mL−1 4-ethylphenol in ethanol) were added to duplicate samples of 0.5 g of fecal powder, and the samples were mixed and refluxed at 100 °C for 15 minutes. After cooling, the samples were centrifuged at 2000g for 5 minutes, and the supernatant was carefully decanted into a new tube. The extracted samples were then resuspended in 5 mL of methanol, refluxed again, and centrifuged as previously described. The sample was further washed with 0.5 mL of methanol and centrifuged once more. The pooled supernatants were dried under nitrogen gas and reconstituted with 0.3 mL of methanol and filtered through a 0.45 μm syringe filter. Quantification of p-cresol and indole was performed using HPLC with fluorescence detection, following a modified version of the method described by King et al.26 Separation was achieved using a C18 column with a flow rate of 1 mL min−1. The mobile phase consisted of a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 water/acetonitrile solution adjusted to pH 3.2 with glacial acetic acid.
3 water/acetonitrile solution adjusted to pH 3.2 with glacial acetic acid.
2.5. Analysis of total fecal bile acids and cecal SCFAs
Analysis of total bile acids was conducted on ground fecal samples collected during housing in the metabolic cages. The bile acids were extracted from dried fecal powder and partially purified using solid-phase extraction columns, as described by Locket and Gallaher.27 Bile acids were assayed enzymatically using 3α-hydroxysteroid dehydrogenase.28 SCFAs were extracted from cecal contents and quantified using gas chromatography, as described by Carlson et al.292.6. Determination of inflammatory markers
Colon mucosal scrapings were homogenized in 1 mL of phosphate-buffered saline solution (pH 7.4) containing protease and phosphatase inhibitors (Roche, Switzerland). After centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g at 4 °C for 5 minutes, supernatants were diluted 1
000g at 4 °C for 5 minutes, supernatants were diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 with PBS, proteins separated on a 10% SDS-PAGE gel, and the bands transferred to nitrocellulose membranes.
3 with PBS, proteins separated on a 10% SDS-PAGE gel, and the bands transferred to nitrocellulose membranes.
The membranes were blocked with 5% non-fat dry milk solution in TBST (0.1% Tween-20) and incubated with primary antibodies overnight at 4 °C. The membranes were incubated with secondary antibodies for an hour at room temperature. Protein bands were visualized using Super-Signal West Atto Ultimate Sensitivity Chemiluminescent Substrate (Thermo Fisher Scientific) and scanned using the Licor Odyssey imaging system.
The primary antibodies used in this study included NFκB, p-NFκB p65 (Ser536), β-catenin, and β-actin (Cell Signaling Technology, Beverly, MA). Serum calprotectin levels were determined using an ELISA assay following the manufacturer's guidelines (S100A8/S100A9, Immundiagnostik AG, Bensheim, Germany).
2.7. DNA extraction and 16S rRNA gene sequencing
The colonic pellets were mechanically disrupted by bead beating for 3 minutes at 6 m s−1, and microbial DNA was extracted using the QIAamp Fast DNA Stool Mini Kit (QIAGEN, Germantown, USA), following the manufacturer's instructions. The 16S rRNA bacterial gene (V4 region) was sequenced at the University of Minnesota Genomics Center (UMGC), on the Illumina MiSeq sequencing platform, using an optimized method for amplicon-based library preparation.302.8. Processing of sequencing data
Amplicon sequence variants (ASVs) were identified from raw Illumina sequences using a combination of Cutadapt, BBMap, and the DADA2 plugin within QIIME 2.31–33 Briefly, adapter sequences, barcodes, primers, and low-quality sequences were eliminated from the pair-end reads using Cutadapt with default parameters. Subsequently, the pair-end reads were merged using the BBMap merger function, and singleton sequences were discarded. The resulting quality-filtered data were then imported into QIIME2 (version 2022.8) and processed through the DADA2 pipeline, where sequences underwent demultiplexing, quality filtering, and denoising. Taxonomic analysis of the ASVs was performed using the Greengenes 13_8 database.34 Following preprocessing, approximately 70% of the reads were retained, yielding an average of 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 reads per sample (range: 3315 to 34
000 reads per sample (range: 3315 to 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 582).
582).
The ASV table generated from these steps was then imported into R Studio for further analysis. Bray–Curtis beta-diversity and Shannon alpha-diversity were calculated using the Vegan package in R.35 Additionally, indicator species analysis was used to identify ASVs associated with each dietary group by calculating an indicator value (IndVal), the product of an ASV's frequency and abundance within a group. IndVal ranges from 0 to 1, with 1 indicating the strongest association within a specific group.36 IndVal analysis was conducted at both the genus and species levels and significance was assessed using the Kruskal–Wallis test.36
2.9. Statistical analysis
Statistically significant differences among the groups were inspected using the analysis of variance (ANOVA) test for numerical variables using Statistical Analysis Software (SAS, version 9.4), with final body weight used as a covariate for liver and fat pad weight analyses. Given the unequal variance observed in tumor data, according to the Shapiro–Wilk test, the data were ranked and subsequently assessed using the ANOVA test within the SAS software. Duncan's multiple range test was used to identify statistically significant differences among groups. Comparisons of tumor incidence among groups were conducted using the chi-square test.Microbial community differences were evaluated using PERMANOVA, which tests significant differences in group centroids, and ANOSIM (Analysis of Similarities), which assesses whether between-group dissimilarities exceed within-group dissimilarities. Both were performed with 999 permutations on Bray–Curtis distances using the vegan R package.35 Alpha diversity indices, richness, and individual taxa relative abundances were tested for normality using the Shapiro–Wilk test. Based on the results, group differences were assessed using one-way ANOVA for normally distributed data or the Kruskal–Wallis test for non-normal data. Following this, pairwise comparisons were performed using Tukey's HSD post hoc test or Dunn's test to identify specific group differences.
Associations between individual taxa and SCFAs were determined by calculating Spearman's correlation coefficients. The top five taxa strongly correlated with SCFAs underwent further analysis using a multiple regression model to explore the association between each taxon, serving as a predictor, and each individual SCFA. The multiple regression model handles multicollinearity of independent variables, providing a more reliable estimate of the association between taxa and SCFAs. Taxa exhibiting a non-significant association with specific SCFAs were excluded from the multiple regression model. Before conducting the multiple regression analysis, normality and multicollinearity assumptions were thoroughly assessed, which included checking normality using Q–Q plots and evaluating multicollinearity through a correlation plot of all independent variables. These steps were essential to ensure the reliability and validity of the subsequent statistical analyses.
3. Results
3.1. Tissue weight, energy intake, and weight gain
Throughout the study duration, the weekly and final body weights of rats showed no significant differences among the groups, indicating that prune consumption did not influence body weight. Similarly, energy intake remained equivalent across all groups throughout the study period. Differences were observed in liver and epididymal fat pad weights among the groups. The group consuming the 5% prune diet exhibited significantly lower liver and epididymal fat pad weights compared to the basal group (p = 0.05 and p = 0.04, respectively, Table 2), despite no significant differences in overall body weight. Empty cecum weight, an indicator of large intestinal fermentation, was significantly greater in the prune diet groups (p = 0.01). Furthermore, the cecal content weight was greater in the 5% prune diet group compared to the basal group (p = 0.04), indicating greater large intestinal contents due to prune consumption.| Diet | |||
|---|---|---|---|
| Basal | 5% prune | 10% prune | |
| Values are mean ± SE; n = 29 (basal), 25 (5% prune), and 21 (10% prune); values not sharing a common superscript are statistically different, (P < 0.05). Liver and fat pad weights were analyzed using body weight as a covariate, as body weight was a significant covariate for both (P < 0.0001 and P = 0.002, respectively). | |||
| Final body weight (g) | 613 ± 18 | 575 ± 18 | 608 ± 18 |
| Energy intake (kcal d−1) | 89.56 ± 3.47 | 91.15 ± 3.32 | 89.28 ± 3.52 |
| Liver weight (g) | 16.0 ± 0.81* | 14.8 ± 0.69† | 15.40 ± 0.65*† |
| Epididymal fat pad weight (g) | 7.10 ± 0.49* | 5.60 ± 0.37† | 6.60 ± 0.43*† |
| Cecum weight (g) | 0.58 ± 0.02* | 0.80 ± 0.08† | 0.75 ± 0.03† |
| Cecum content weight (g) | 2.13 ± 0.16* | 4.71 ± 1.23† | 3.22 ± 0.14*† |
| Fecal weight (g per 24 h) | 1.66 ± 0.31 | 1.49 ± 0.11 | 1.71 ± 0.20 |
3.2. Tumor number and volume
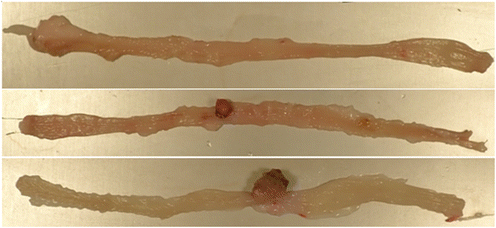
The physical appearance of the colon segments varied, as shown in Fig. 1. The healthy (i.e., uninvolved) colon segments exhibited a smooth and uniform appearance, lacking any visible irregularities or protrusions. Adenomas, early-stage tumor growths, were identifiable by their smooth and round polyp-like formations along the inner lining of the colon. Adenocarcinoma, indicative of advanced and malignant growth, presented as irregular masses with an uneven surface. Additionally, the colon segment lining displayed a pale appearance, potentially signaling pathological changes or inflammation within the tissue.No significant differences were observed in the count or volume of non-cancerous lesions, colon adenocarcinoma, total colonic tumors, or total GI tumors across the studied groups. However, both the 5% and 10% prune diets displayed a trend towards fewer small intestine tumors (p = 0.1) (Table 3). Moreover, the 10% prune group also had a tendency toward a lower volume of small intestine tumors compared to the basal diet group (p < 0.1). Finally, the incidence of tumors, defined as the percentage of rats with tumors, did not significantly differ among the experimental groups.
| Diet group | |||
|---|---|---|---|
| Basal | 5% prune | 10% prune | |
| Tumor count and volume are presented as ranked means ± SE; n = 29 (basal), 25 (5% prune), and 24 (10% prune). Tumor incidence is expressed as the percentage of animals with tumors in each diet group. Values not sharing a common superscript indicate a trend toward significance (P < 0.1) based on the chi-square test. | |||
| Count | |||
| Small intestine adenocarcinomas | 44.2 ± 3.6* | 37.1 ± 2.6† | 36.3 ± 2.7† |
| Colonic non-cancerous lesions | 40.3 ± 3.8 | 39.9 ± 4.5 | 38.1 ± 3.9 |
| Colonic adenomas | 40.3 ± 3.1 | 41.8 ± 3.6 | 36.1 ± 2.8 |
| Colonic adenocarcinomas | 39.3 ± 3.8 | 37.5 ± 4.8 | 41.8 ± 4.3 |
| Total colonic tumors | 39.3 ± 3.8 | 38.9 ± 4.9 | 40.3 ± 3.8 |
| Total GI tumors | 44.3 ± 3.8 | 35.4 ± 4.7 | 37.9 ± 4.6 |
| Volume (mm 3 ) | |||
| Small intestine adenocarcinoma | 38.9 ± 3.3* | 33.4 ± 3.0*† | 31.9 ± 2.7† |
| Colonic non-cancerous lesions | 39.9 ± 3.8 | 39.5 ± 4.4 | 38.9 ± 4.2 |
| Colonic adenoma | 35.5 ± 3.0 | 38.0 ± 3.7 | 31.5 ± 2.8 |
| Colonic adenocarcinoma | 34.0 ± 4.1 | 33.5 ± 4.1 | 37.6 ± 4.4 |
| Total colonic tumor | 34.5 ± 4.0 | 34.7 ± 3.9 | 35.9 ± 4.7 |
| Total GI tumor | 36.1 ± 4.2 | 33.0 ± 3.8 | 35.1 ± 4.2 |
| Incidence (%) | |||
| Small intestine tumor | 31 | 16 | 17 |
| Colonic adenoma | 24 | 28 | 13 |
| Colonic adenocarcinoma | 76 | 72 | 87 |
| Colonic tumor | 86 | 80 | 92 |
| Total GI tumor | 90 | 92 | 84 |
3.3. Fecal and cecal metabolites
Analysis of cecal content SCFAs revealed significant increases in butyrate (P < 1 × 10−4) and total SCFA amount (p = 0.01) per cecum in both the 5% prune and 10% prune diets (Fig. 2). Butyrate increased linearly with the dietary concentration of prune (R2 = 0.98). Animals fed the 5% prune diet had the highest amount of cecal propionate (p = 0.03) and the branched short chain fatty acid isovalerate (p = 0.049) compared to other dietary groups. Additionally, there was a trend towards higher acetate and valerate amounts (p = 0.1) in the 5% prune diet. Across all diet groups, acetate was present in the largest amount among all SCFAs.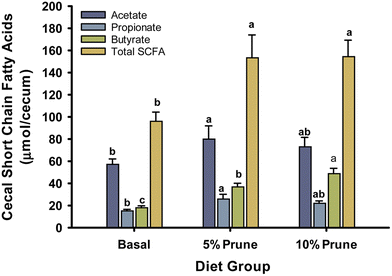 | ||
| Fig. 2 Amount of short chain fatty acids in cecal contents. Values are mean ± SE. Values not sharing a common letter are statistically different, P < 0.05. | ||
In contrast, the analysis of total fecal bile acids did not reveal any statistically significant differences among the groups (p = 0.44) (Table 4). There were no significant differences among the groups in the fecal concentration of p-cresol or indole (Table 4). These findings suggest that while prune intake influenced SCFA levels, particularly butyrate and propionate, it did not significantly impact fecal bile acids or metabolites of aromatic amino acids in this study.
| Diet | |||
|---|---|---|---|
| Basal | 5% prune | 10% prune | |
| Values are expressed as mean ± SE. Sample sizes were as follows: p-cresol and indole, n = 16 (basal), 13 (5% prune), and 18 (10% prune); bile acids, n = 11 (basal), 14 (5% prune), and 12 (10% prune). | |||
| p-Cresol (µg d−1) | 7.94 ± 1.59 | 6.24 ± 1.02 | 7.99 ± 1.23 |
| Indole (µg d−1) | 0.53 ± 0.21 | 0.27 ± 0.08 | 0.62 ± 0.20 |
| Total bile acids (μmol d−1) | 63.97 ± 12.06 | 50.21 ± 9.18 | 59.60 ± 10.84 |
3.4. Inflammatory markers
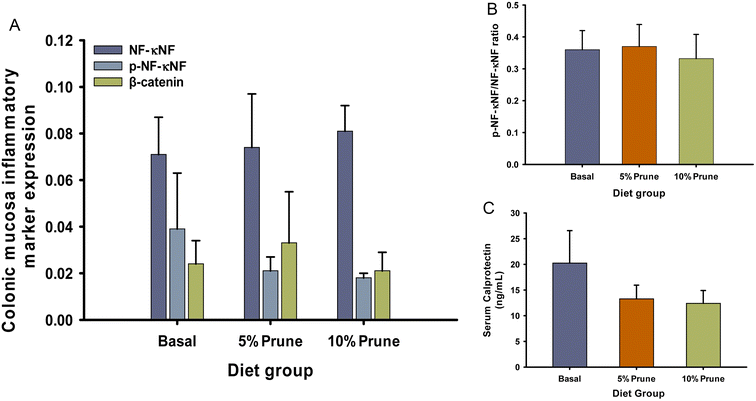
The inflammatory markers NFκB, p-NFκB, the p-NFκB/NFκB ratio, and β-catenin were analyzed in non-involved colon tissue by western blotting. There were no statistically significant differences among the diets for any marker (Fig. 3A and B). Serum calprotectin, a well-known inflammatory marker utilized for diagnosing inflammatory bowel diseases,37 did not differ among the dietary groups (Fig. 3C).3.5. Changes in the gut microbiome
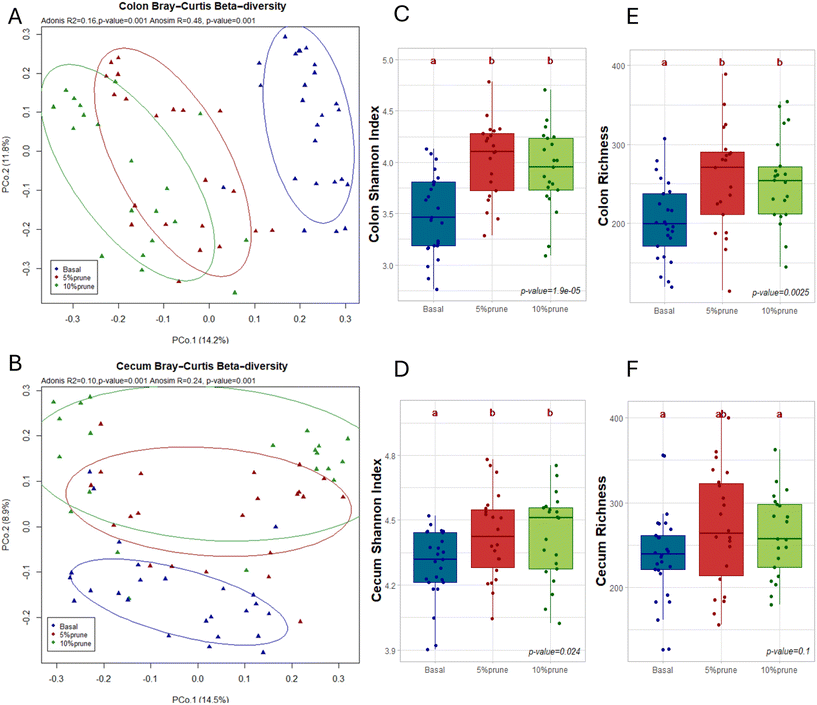
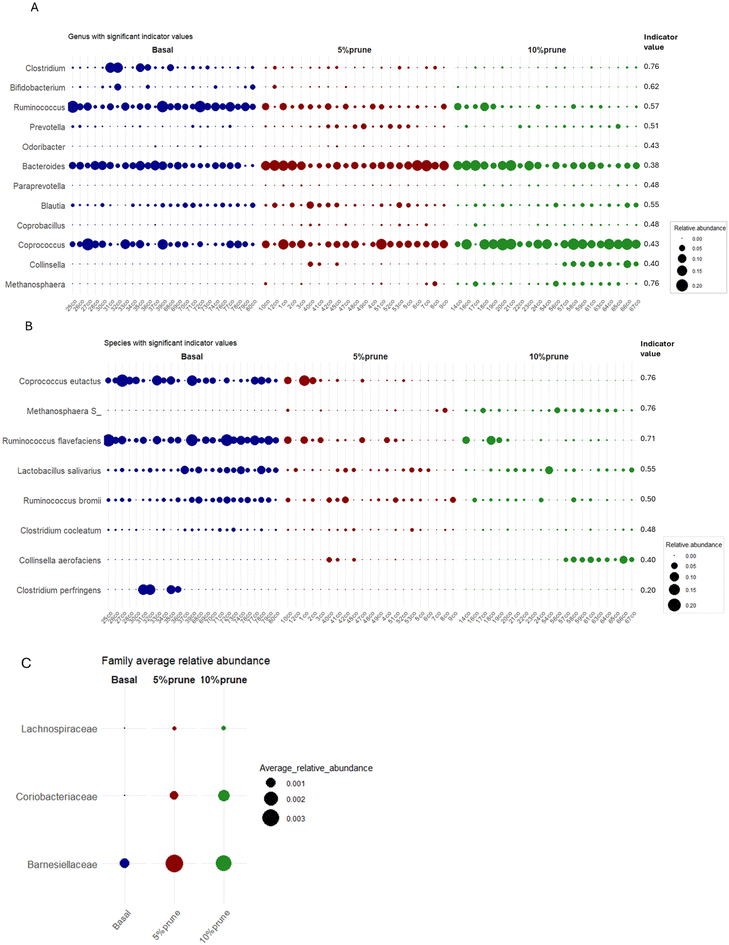
The Bray–Curtis dissimilarity index shows a clear separation in microbiome composition between the prune-containing diets and the basal diet in both cecum and colon contents (Fig. 4A and B). PERMANOVA analysis confirmed significant group differences (colon: R2 = 0.16, p = 0.001), indicating that 16% of the variation in the colonic microbiota was explained by diet. ANOSIM analysis further supported a moderate separation (R = 0.48, p = 0.001). In contrast, the cecal microbiome showed a smaller dietary effect (R2 = 0.10, p = 0.001), suggesting that prune consumption may have had a slightly more pronounced impact on the colonic microbial community structure. Alpha diversity was assessed using the Shannon index and richness. The Shannon index was significantly higher in both prune groups compared with the basal group (colon: p = 1.9 × 10−5; cecum: p = 0.024; Fig. 4C and D), indicating greater microbial diversity and evenness of species. Similarly, richness increased in the colon (p = 0.0025; Fig. 4E), reflecting an overall greater bacterial diversity with prune supplementation.Indicator species analysis identified taxa differentiating the dietary groups at both genus and species levels (Fig. 5A and B). Prune consumption increased the relative abundance of Prevotella, Paraprevotella, Coprococcus, Coprobacillus, Methanosphaera, and Collinsella, while reducing Clostridium, Ruminococcus, and Bifidobacterium. At the species level, Lactobacillus salivarius, Ruminococcus flavefaciens, Ruminococcus bromii, Coprococcus eutactus and C. perfringens decreased with prune intake, whereas Collinsella aerofaciens increased. Blautia was enriched only in the 5% prune group, suggesting a nonlinear response to prune in the diet. At the family level, Coriobacteriaceae, Lachnospiraceae, and Barnesiellaceae were more abundant in prune-fed rats, with the largest increase observed for Barnesiellaceae in the 5% prune group (Fig. 5C).
Associations between microbial taxa and cecal SCFAs are summarized in Table 5. Methanosphaera showed consistent positive correlations with all SCFAs, including acetate, butyrate, valerate, and total SCFAs, suggesting a broad role in SCFA metabolism. Prevotella exhibited the strongest correlation with butyrate, while Coriobacteriaceae, Blautia, and Paraprevotella were positively associated with both butyrate and propionate. Multiple regression analysis further identified Methanosphaera as a key predictor for SCFA levels and confirmed significant associations between Coriobacteriaceae and Prevotella with butyrate levels (Table 6).
| Acetate | Butyrate | Propionate | Valerate | Total SCFAs | |
|---|---|---|---|---|---|
| For each SCFA and the total SCFA, the top five taxa with the strongest correlations are listed. Values are presented as Spearman's correlation coefficients (ρ) along with the corresponding p-values in parentheses. ρ = 0.1–0.4, weak positive correlation; 0.4–0.7, moderate positive correlation; >0.7, strong positive correlation. | |||||
| g_Methanosphaera | 0.42 (5.8 × 10−4) | 0.53 (1.2 × 10−5) | 0.32 (1.0 × 10−2) | 0.44 (3.1 × 10−4) | 0.52 (1.2 × 10−5) |
| g_Blautia | 0.37 (3.0 × 10−3) | 0.50 (4.1 × 10−5) | 0.45 (2.0 × 10−4) | ||
| g_Ruminococcus | 0.37 (3.2 × 10−6) | 0.31 (1.4 × 10−2) | |||
| f_Barnesiellaceae | 0.33 (7.8 × 10−3) | 0.28 (2.9 × 10−2) | 0.37 (3.2 × 10−3) | ||
| g_Christensenella | 0.33 (8.4 × 10−3) | ||||
| f_Victivallaceae | 0.34 (6.5 × 10−3) | ||||
| f_Coriobacteriaceae | 0.66 (6.7 × 10−9) | 0.42 (7.1 × 10−4) | 0.25 (5.0 × 10−2) | 0.41 (1.0 × 10−3) | |
| g_Prevotella | 0.71 (1.2 × 10−10) | 0.41 (9.3 × 10−4) | 0.35 (5.7 × 10−3) | 0.48 (9.5 × 10−5) | |
| g_Paraprevotella | 0.46 (1.7 × 10−4) | 0.30 (1.8 × 10−3) | |||
| Acetate | Butyrate | Propionate | Valerate | Total SCFAs | |
|---|---|---|---|---|---|
| Values are presented as standardized regression coefficients (β) with their respective p-values in parentheses. | |||||
| g_Methanosphaera | 0.47 (2.7 × 10−6) | 0.73 (1.5 × 108) | 0.55 (3.1 × 10−7) | 0.63 (9.8 × 10−8) | |
| g_Ruminococcus | 0.54 (3.2 × 10−6) | 0.42 (1.6 × 10−5) | |||
| f_Coriobacteriaceae | 0.45 (7.1 × 10−3) | ||||
| g_Prevotella | 0.33 (9.8 × 10−3) | ||||
| Adjusted R2 | 0.57 (4.2 × 10−10) | 0.46 (1.5 × 10−7) | 0.50 (2.0 × 10−8) | 0.57 (3.0 × 10−10) | 0.46 (1.4 × 10−7) |
4. Discussion
In the present study, we incorporated a prune product (prune) that was substantially similar to whole prunes into the diets of carcinogen-treated rats to evaluate its effect on tumor formation. Our hypothesis was that the prune, as a food high in fermentable fibers, sorbitol, and polyphenolics, would decrease tumor development. However, prune consumption did not reduce colonic tumors. We also carried out both microbiome and metabolite analyses of large intestinal contents to assess changes in the microbial composition and bacterial metabolites. Prune feeding resulted in substantial changes to the microbiome but did not affect daily fecal bacterial metabolite excretion.Previous cohort studies have reported that high dietary fiber intake, particularly from cereals and fruits, is associated with reduced colorectal adenomas and distal colon cancer.38 However, in the present study, all three diets were matched for total fiber content, which was primarily cellulose in all three diets (80, 73, and 66 g kg−1 in the basal, 5%, and 10% prune diets, respectively), a fiber that is highly resistant to fermentation. Pectin, a highly fermentable soluble fiber, and sorbitol, a highly fermentable sugar alcohol, were present only in the prune diets. Consequently, this study represents a comparison not of differences in the dietary concentration of dietary fiber but of the type of fiber – non-fermentable versus fermentable.
It has sometimes been assumed that feeding highly fermentable fibers would reduce colon cancer risk, largely based on the belief that short chain fatty acids, particularly butyrate, produced by large intestinal fermentation, are protective against colon cancer. However, feeding isolated pectin to carcinogen-treated rats has resulted in either no difference39–43 or increases44–47 in tumorigenesis compared to the cellulose control, with only two studies reporting reduced tumor number.48,49 Feeding of other types of highly fermentable fiber such guar gum resulted in higher tumor number in carcinogen-treated rats50 as well. Finally, in carcinogen-treated mice, feeding inulin, a highly fermentable fiber, resulted in greatly increased tumor incidence compared with cellulose feeding.51 Thus, in most studies, feeding a highly fermentable purified fiber did not decrease tumor development and often increased it. Our finding that prune, a rich source of fermentable materials, in the form of pectin and sorbitol, did not reduce tumor number is consistent with these studies.
Prunes are a rich source of phenolic compounds, primarily chlorogenic and neochlorogenic acids.52 Certain phenolic compounds, such as ferulic acid53 and curcumin,54 reduce tumor incidence in carcinogen-treated rats. Although no reduction in tumor incidence has been found with feeding chlorogenic acid,55,56 reductions in tumor multiplicity55 and in colonic precancerous lesions (aberrant crypt foci, ACF)57 have been reported. Chlorogenic acid upregulates the antioxidant enzyme glutathione S-transferase and suppresses ROS-mediated NFκB, AP-1 and MAPK activation,58 suggesting a molecular mechanism for chemoprevention. In our study, however, no increase in NFκB was noted with prune feeding. It may be that the quantity of chlorogenic acid consumed by rats fed prune was too little to reduce carcinogenesis.
A difference in phenolic acid content may explain the difference observed between two previous studies examining prune- or plum-beverage on colonic precancerous lesions in rats. Banerjee et al.12 reported a reduction in ACF in carcinogen-treated rats administered plum beverage, whereas Yang et al.13 found no reduction in ACF with prune powder-containing diets. The two prune treatments differed substantially in their phenolic acid content. The concentration of neochlorogenic and chlorogenic acids in the plum beverage used by Banerjee et al. was approximately ten times higher than those in the prune powder used by Yang et al. In the latter study, prunes were ground and dried at 120–148 °C to produce a powder. Such a heat treatment would have oxidized much of the phenolic compounds present, as has been shown for red grape pomace dried at temperatures between 100 and 140 °C.59 In addition, the plum beverage and prune powder differed in their pectin contents. Although neither study disclosed exact pectin concentrations, treatment of the plum beverage with pectinase by Banerjee et al. would have markedly reduced its pectin content, whereas the prune powder used by Yang et al. retained the full complement of pectin. Thus, the divergent results could be due to the difference in pectin or phenolic acid content of the prune products used in the two studies.
High butyrate concentrations have long been suggested to reduce colon cancer risk, based in part on cell culture studies demonstrating its ability to inhibit histone deacetylase activity.60,61 We demonstrated that feeding prune greatly increased total cecal SCFAs and increased butyrate amount linearly with the dietary concentration of prune (total SCFA and butyrate, R2 = 0.44 and 0.73, respectively). Our results are consistent with earlier studies demonstrating that fermentable fibers significantly increase fecal butyrate and propionate levels.62,63 As discussed above, these diets are generally not effective in reducing tumorigenesis. It is possible that the failure of high large intestinal butyrate to reduce colon cancer risk in in vivo studies could be due to experimental design differences among studies. However, it may be that butyrate produced by fermentation in the large intestine is not chemopreventive. Whether this might be due to insufficient butyrate production, the location in the large intestine where it is produced, or simply that butyrate is not chemopreventive is unclear. Furthermore, animal studies examining the chemopreventive effect of administered butyrate have not clarified the situation, as they are conflicting. For example, oral administration of butyrate to carcinogen-treated rats increased fecal butyrate levels but did not reduce ACF or colonic tumors.64,65 In contrast, Tian et al. reported that oral administration of a SCFA mixture significantly increased cecal SCFAs and reduced colonic tumors in mice given DSS.66 The cecal butyrate concentration (26 µmol g−1) achieved through oral SCFA administration was substantially higher than the concentration observed in our 10% prune diet group (14 µmol g−1). It may be that a threshold concentration of butyrate must be achieved to exert protective effects in the colon, a threshold that is difficult to achieve by dietary means. The concept of a threshold is supported by the finding that low doses of butyrate (0.5 and 1.0 mM) stimulated growth of HCT116 colon carcinoma cells, while higher doses (2.0 and 5.0 mM) had an inhibitory effect on growth.61 Thus, whether increases in large intestinal butyrate produced through dietary means can be chemopreventive remains an open question.
Although pectin feeding has rarely been found to reduce large intestinal tumors, it has been shown to decrease small intestinal tumors in DMH-treated rats in two previous studies.39,67 This observation is consistent with our finding of a trend toward fewer small intestinal tumors in both prune diet groups. Prune pectin is composed primarily of rhamnogalacturonans and type I arabinogalactans. Oral administration of rhamnogalacturonans has been reported to significantly increase the expression of MUC1 and the number of mucin-producing goblet cells in the colons of DSS-treated mice.68 Increased MUC1 expression attenuated inflammatory responses by dampening NF-κB activation.69 Thus, the role of pectin in tumorigenesis appears to be complex. It has shown potential colon tumor-promoting effects, yet it has demonstrated protective effects against small intestinal tumor development. Understanding the mechanisms underlying these contrasting effects and identifying the physiological conditions under which pectin exerts protective versus promotive influences on intestinal tumorigenesis would be most helpful in clarifying the role of fermentable dietary fiber in carcinogenesis.
Prunes contain fiber, polyphenols, and sorbitol, all of which are known to be fermentable in the large intestine. Empty cecum weight, a well-established marker of fermentation,70 was significantly increased in both prune-supplemented diets compared to the fiber (cellulose)-matched basal diet, confirming the greater degree of fermentation with these diets. A higher cecal content mass accompanied this greater cecal fermentation. However, daily fecal weight did not differ among the groups, indicating that the non-fermentable cellulose fiber was primarily responsible for influencing fecal mass.
Another noteworthy finding was the decrease in fat pad weight in animals fed 5% prune, and a trend towards a decrease in the 10% prune group. Since fat pad weight is highly correlated with whole body adiposity,71 this indicates that long-term feeding of prune reduces body fat. This is most likely due to the small intestinal viscosity produced by the pectin in the prune, as viscosity appears to be the characteristic of dietary fiber responsible for reduction in body fat.72
Analysis of the large intestinal microbiome indicated that prune feeding promotes greater fermentation and bacterial richness relative to the basal diet. At the genus level, prune feeding led to increases in Prevotella, Paraprevotella, Collinsella, Blautia, and Coprococcus. Simpson et al. reported that greater abundance of Blautia was observed in the 50 g d−1 prune-fed group rather than the 100 g d−1 group in post-menopausal women. They also reported an inverse correlation between Blautia and total urinary phenolics, suggesting Blautia may be involved in metabolism of polyphenols.17
g_Blautia, g_Prevotella, g_Paraprevotella, and f_Coriobacteriaceae abundance was positively correlated with cecal butyrate amounts. While g_Prevotella and g_Blautia are not classified as primary butyrate producers, they may contribute indirectly to butyrate production through cross-feeding mechanisms, by providing precursor substrates such as succinate or acetate.73,74 In addition, g_Prevotella and g_Paraprevotella are known to ferment dietary glycans and plant-derived polysaccharides, which are abundant in prunes.75 f_Coriobacteriaceae have also been identified as butyrate producers, based on the presence of the gene for butyrate kinase (buk), a marker gene for butyrate production.76 In mice fed either a carbohydrate fraction or a polyphenol fraction from prunes, only the carbohydrate fraction increased the abundance of f_Coriobacteriaceae.77 These findings suggest that prune feeding enhances butyrate production through fiber fermentation, not polyphenol metabolism.
We found that prune feeding reduces the abundance of the probiotic genus Bifidobacterium. A similar reduction of Bifidobacterium was observed in mice fed a 25% prune diet.77 However, findings from human studies are inconsistent. Lever et al.19 reported a trend towards an increase in Bifidobacteria abundance in humans fed prunes for 9 weeks, whereas Simpson et al.17,18 found no difference in abundance in humans consuming prunes for 12 months.
A novel finding from our microbiome analysis was the greater abundance of g_Methanosphaera in the prune diets relative to the basal diet. This abundance was positively correlated with all SCFAs, showing the strongest correlation with butyrate (R = 0.53). Methanosphaera is an archaeal genus characterized by its ability to utilize hydrogen to reduce various organic compounds to methane.78 Methane's oxidation byproduct, formaldehyde, is considered a genotoxic compound. The relationship between methanogens and colon cancer risk has become an area of growing interest. For instance, one study reported a greater abundance of methanogens in colon biopsy samples from CRC patients.79 Similarly, a multi-cohort study found increasing abundance of Methanobacteriota in fecal samples from healthy controls to patients with adenomas to patients with CRC.80 The role of diet, particularly a diet high in fermentable constituents, in influencing abundance of methanogens in the large intestine, and its relation to chemoprevention would seem to warrant further study.
Epidemiological studies report both elevated primary and secondary bile acid concentrations in colon cancer patients, particularly cholic acid, deoxycholic acid, and lithocholic acid.81–83 Tinker et al. reported that in humans consuming a prune supplement for 4 weeks, total fecal bile acid concentration showed a trend toward reduction, with a significant reduction in secondary bile acids, relative to a grape juice control group.84 Similarly, Yang et al. found that rats fed a 5% prune powder diet had significantly reduced total fecal bile acid concentrations compared to both basal and fiber-matched control groups, suggesting that the type of fiber in prunes contributes to this effect.13 In the present study, fecal bile acid concentration did not differ among the diets. Since the fecal weight did not differ among the diets, no doubt due to non-fermentable cellulose being the major dietary fiber in all diets, this suggests that prune did not alter the bile acid pool or bile acid reabsorption from the intestinal tract.
Dietary fiber and undigested protein represent the major macronutrients entering the large intestine. Fermentation of fiber (saccharolytic fermentation) produces metabolites considered neutral or beneficial, such as short chain fatty acids and fermentation gases, whereas protein fermentation (proteolytic fermentation) produces metabolites that may be detrimental, such as branch chain fatty acids and derivatives of aromatic amino acids, such as phenols and indoles. For example, proteolytic byproducts such as p-cresol and phenol are often genotoxic.85 However, in some cases, proteolytic fermentation products may be beneficial, as indole enhances epithelial barrier function, decreases pathogen colonization, and attenuates NF-κB activation in HCT-8 cells.86 Since increasing saccharolytic fermentation has been found to reduce the products of proteolytic fermentation,87 we determined the fecal excretion of p-cresol and indole. Neither prune-containing diet reduced these products of protein fermentation, suggesting that prune supplementation did not reduce proteolytic fermentation.
Colonic inflammation is well established to increase colon cancer risk.3 NF-κB, a pro-inflammatory transcription factor regulating the synthesis of inflammatory cytokines, was determined to assess colon tissue inflammation. Banerjee et al. previously reported reduced colonic mucosal NF-κB levels in rats fed a plum beverage.12 However, prune feeding in the present study had no effect on NF-κB. The unchanged NF-κB levels observed here may be due to the absence of any dietary component that would induce intestinal inflammation (e.g., high fat or high sugar), resulting in a very low baseline level of colonic inflammation. Furthermore, we observed no reduction in serum calprotectin, an established marker of inflammation. Whether prune consumption can ameliorate intestinal inflammation should therefore be examined under conditions where some degree of inflammatory stress is induced.
Finally, we examined β-catenin, a key regulator in the Wnt signaling pathway that controls cell proliferation and differentiation. Activation of β-catenin, the central regulator of the canonical Wnt pathway, results in enhanced transcription of factors required for normal intestinal function. However, hyperactivation of Wnt/β-catenin signaling is associated with progression to colon cancer. Previous studies have shown that fruit-derived polyphenols, such as stilbenes, pterostilbene and resveratrol, can reduce β-catenin expression.88 However, in our study, no significant differences in β-catenin protein expression were observed among dietary groups, suggesting that prune consumption did not modulate the Wnt/β-catenin signaling pathway.
5. Conclusion
Our study found that prune consumption does not significantly induce or reduce tumor formation in the colon, although a trend towards reducing small intestine tumors was observed. Prune consumption, however, induced significant shifts in the gut microbiome, particularly enriching bacteria from families such as Prevotellaceae and Lachnospiraceae, while reducing the abundance of potentially pathogenic species such as C. perfringens. The strong correlation observed between cecal butyrate levels and specific bacterial genera highlights the butyrogenic effect of prune consumption. However, our findings raise the question of whether the amount of SCFAs, particularly butyrate, that can be produced from dietary sources containing fermentable components at levels achievable in the human diet would be sufficient to reduce colon cancer risk. Therefore, understanding whether there is a threshold concentration of butyrate necessary for colon cancer prevention will be crucial for developing effective dietary strategies. Moreover, future studies should evaluate the potential chemoprotective effects of prunes under conditions of pre-existing intestinal inflammation, as it may be that a chemopreventive effect of prunes will only become apparent in the presence of an inflamed colon. This information will be valuable for developing therapeutic strategies for patients with pre-existing colonic inflammation.Author contributions
Conceptualization: D. D. G. and A. G.; experiments and formal analysis: M. J. and D. M. S.; methodology and data curation: M. J., A. G., and D. D. G.; funding acquisition: D. D. G. and A. G.; draft preparation: M. J. and A. G.; finalizing the manuscript: D. D. G.; project administration and supervision: D. D. G. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
D. D. G. is a member of the Nutrition Advisory Panel of the California Prune Board. No other authors have a conflict of interest.Abbreviations
| ACF | Aberrant crypt foci |
| AIN-93G | American Institute of Nutrition-93G |
| ASV | Amplicon sequence variant |
| CRC | Colorectal cancer |
| DSS | Dextran sulfate sodium |
| IndVal | Indicator value |
| NF-κB | Nuclear factor kappa B |
| SCFA | Short chain fatty acid |
Data availability
Microbiome-related data files may be accessed at https://hdl.handle.net/11299/166578.Supplementary information (SI) describing the composition of the dried prune product, as well as the composition of dried prunes, prune juice, and fresh plums, is available. See DOI: https://doi.org/10.1039/d5fo03398e.
Acknowledgements
This work was supported by funding from the California Prune Board to D. D. G. and A. G.References
- R. L. Siegel, N. S. Wagle, A. Cercek, R. A. Smith and A. Jemal, Colorectal cancer statistics, 2023, Ca-Cancer J. Clin., 2023, 73, 233–254 CrossRef PubMed.
- WHO, United State COVID case count, https://covid19.who.int/region/amro/country/us, (accessed 5/24/23, 2023).
- M. Schmitt and F. R. Greten, The inflammatory pathogenesis of colorectal cancer, Nat. Rev. Immunol., 2021, 21, 653–667 CrossRef CAS PubMed.
- F. J. van Duijnhoven, H. B. Bueno-De-Mesquita, P. Ferrari, M. Jenab, H. C. Boshuizen, M. M. Ros, C. Casagrande, A. Tjonneland, A. Olsen, K. Overvad, O. Thorlacius-Ussing, F. Clavel-Chapelon, M. C. Boutron-Ruault, S. Morois, R. Kaaks, J. Linseisen, H. Boeing, U. Nothlings, A. Trichopoulou, D. Trichopoulos, G. Misirli, D. Palli, S. Sieri, S. Panico, R. Tumino, P. Vineis, P. H. Peeters, C. H. van Gils, M. C. Ocke, E. Lund, D. Engeset, G. Skeie, L. R. Suarez, C. A. Gonzalez, M. J. Sanchez, M. Dorronsoro, C. Navarro, A. Barricarte, G. Berglund, J. Manjer, G. Hallmans, R. Palmqvist, S. A. Bingham, K. T. Khaw, T. J. Key, N. E. Allen, P. Boffetta, N. Slimani, S. Rinaldi, V. Gallo, T. Norat and E. Riboli, Fruit, vegetables, and colorectal cancer risk: the European Prospective Investigation into Cancer and Nutrition, Am. J. Clin. Nutr., 2009, 89, 1441–1452 CrossRef CAS PubMed.
- Z. Y. Wu, J. L. Chen, H. Li, K. Su and Y. W. Han, Different types of fruit intake and colorectal cancer risk: A meta-analysis of observational studies, World J. Gastroenterol., 2023, 29, 2679–2700 CrossRef CAS PubMed.
- M. Stacewicz-Sapuntzakis, P. E. Bowen, E. A. Hussain, B. I. Damayanti-Wood and N. R. Farnsworth, Chemical composition and potential health effects of prunes: a functional food?, Crit. Rev. Food Sci. Nutr., 2001, 41, 251–286 CrossRef CAS PubMed.
- S. Hooshmand, A. Kumar, J. Y. Zhang, S. A. Johnson, S. C. Chai and B. H. Arjmandi, Evidence for anti-inflammatory and antioxidative properties of dried plum polyphenols in macrophage RAW 264.7 cells, Food Funct., 2015, 6, 1719–1725 RSC.
- M. Vizzotto, W. Porter, D. Byrne and L. Cisneros-Zevallos, Polyphenols of selected peach and plum genotypes reduce cell viability and inhibit proliferation of breast cancer cells while not affecting normal cells, Food Chem., 2014, 164, 363–370 CrossRef CAS PubMed.
- D. Hanahan and R. A. Weinberg, Hallmarks of cancer: the next generation, Cell, 2011, 144, 646–674 CrossRef CAS PubMed.
- M. A. Lea, C. Ibeh, C. desBordes, M. Vizzotto, L. Cisneros-Zevallos, D. H. Byrne, W. R. Okie and M. P. Moyer, Inhibition of growth and induction of differentiation of colon cancer cells by peach and plum phenolic compounds, Anticancer Res., 2008, 28, 2067–2076 CAS.
- T. Fujii, T. Ikami, J. W. Xu and K. Ikeda, Prune extract (Prunus domestica L.) suppresses the proliferation and induces the apoptosis of human colon carcinoma Caco-2, J. Nutr. Sci. Vitaminol., 2006, 52, 389–391 CrossRef CAS PubMed.
- N. Banerjee, H. Kim, S. T. Talcott, N. D. Turner, D. H. Byrne and S. U. Mertens-Talcott, Plum polyphenols inhibit colorectal aberrant crypt foci formation in rats: potential role of the miR-143/protein kinase B/mammalian target of rapamycin axis, Nutr. Res., 2016, 36, 1105–1113 CrossRef CAS PubMed.
- Y. Yang and D. D. Gallaher, Effect of dried plums on colon cancer risk factors in rats, Nutr. Cancer, 2005, 53, 117–125 CrossRef CAS PubMed.
- J. Ahn, R. Sinha, Z. Pei, C. Dominianni, J. Wu, J. Shi, J. J. Goedert, R. B. Hayes and L. Yang, Human gut microbiome and risk for colorectal cancer, J. Natl. Cancer Inst., 2013, 105, 1907–1911 CrossRef CAS PubMed.
- N. Larsen, C. B. de Souza, L. Krych, T. B. Cahú, M. Wiese, W. Kot, K. M. Hansen, A. Blennow, K. Venema and L. Jespersen, Potential of Pectins to Beneficially Modulate the Gut Microbiota Depends on Their Structural Properties, Front. Microbiol., 2019, 10, 223 CrossRef PubMed.
- M. G. Xie, Y. Q. Fei, Y. Wang, W. Y. Wang and Z. Wang, Chlorogenic Acid Alleviates Colon Mucosal Damage Induced by a High-Fat Diet via Gut Microflora Adjustment to Increase Short-Chain Fatty Acid Accumulation in Rats, Oxid. Med. Cell. Longevity, 2021, 2021, 3456542 CrossRef PubMed.
- A. M. R. Simpson, M. J. De Souza, J. Damani, C. Rogers, N. I. Williams, C. Weaver, M. G. Ferruzzi, S. Chadwick-Corbin and C. H. Nakatsu, Prune supplementation for 12 months alters the gut microbiome in postmenopausal women, Food Funct., 2022, 13, 12316–12329 RSC.
- B. J. Smith, B. Hatter, K. Washburn, J. Graef-Downard, B. A. Ojo, G. D. El-Rassi, R. H. Cichewicz, M. Payton and E. A. Lucas, Dried Plum's Polyphenolic Compounds and Carbohydrates Contribute to Its Osteoprotective Effects and Exhibit Prebiotic Activity in Estrogen Deficient C57BL/6 Mice, Nutrients, 2022, 14 Search PubMed.
- E. Lever, S. M. Scott, P. Louis, P. W. Emery and K. Whelan, The effect of prunes on stool output, gut transit time and gastrointestinal microbiota: A randomised controlled trial, Clin. Nutr., 2019, 38, 165–173 CrossRef PubMed.
- H. F. Chiu, Y. C. Huang, Y. Y. Lu, Y. C. Han, Y. C. Shen, O. Golovinskaia, K. Venkatakrishnan and C. K. Wang, Regulatory/modulatory effect of prune essence concentrate on intestinal function and blood lipids, Pharm. Biol., 2017, 55, 974–979 CrossRef CAS PubMed.
- M. Song, A. T. Chan and J. Sun, Influence of the Gut Microbiome, Diet, and Environment on Risk of Colorectal Cancer, Gastroenterology, 2020, 158, 322–340 CrossRef CAS PubMed.
- J. A. Tayek, N. W. Istfan, C. T. Jones, K. J. Hamawy, B. R. Bistrian and G. L. Blackburn, Influence of the Walker 256 carcinosarcoma on muscle, tumor, and whole-body protein synthesis and growth rate in the cancer-bearing rat, Cancer Res., 1986, 46, 5649–5654 CAS.
- G. P. Boivin, K. Washington, K. Yang, J. M. Ward, T. P. Pretlow, R. Russell, D. G. Besselsen, V. L. Godfrey, T. Doetschman, W. F. Dove, H. C. Pitot, R. B. Halberg, S. H. Itzkowitz, J. Groden and R. J. Coffey, Pathology of mouse models of intestinal cancer: consensus report and recommendations, Gastroenterology, 2003, 124, 762–777 CrossRef PubMed.
- J. M. Ward and P. M. Treuting, Rodent intestinal epithelial carcinogenesis: pathology and preclinical models, Toxicol. Pathol., 2014, 42, 148–161 CrossRef CAS PubMed.
- M. K. Washington, A. E. Powell, R. Sullivan, J. P. Sundberg, N. Wright, R. J. Coffey and W. F. Dove, Pathology of rodent models of intestinal cancer: progress report and recommendations, Gastroenterology, 2013, 144, 705–717 CrossRef PubMed.
- R. A. King, B. L. May, D. A. Davies and A. R. Bird, Measurement of phenol and p-cresol in urine and feces using vacuum microdistillation and high-performance liquid chromatography, Anal. Biochem., 2009, 384, 27–33 CrossRef CAS PubMed.
- P. L. Locket and D. D. Gallaher, An improved procedure for bile acid extraction and purification and tissue distribution in the rat, Lipids, 1989, 24, 221–223 CrossRef CAS PubMed.
- M. J. Sheltawy and M. S. Losowsky, Determination of faecal bile acids by an enzymic method, Clin. Chim. Acta, 1975, 64, 127–132 CrossRef CAS PubMed.
- J. Carlson, A. Hospattankar, P. Deng, K. Swanson and J. Slavin, Prebiotic effects and fermentation kinetics of wheat dextrin and partially hydrolyzed guar gum in an in vitro batch fermentation system, Foods, 2015, 4, 349–358 CrossRef CAS PubMed.
- D. M. Gohl, P. Vangay, J. Garbe, A. MacLean, A. Hauge, A. Becker, T. J. Gould, J. B. Clayton, T. J. Johnson, R. Hunter, D. Knights and K. B. Beckman, Systematic improvement of amplicon marker gene methods for increased accuracy in microbiome studies, Nat. Biotechnol., 2016, 34, 942–949 CrossRef CAS PubMed.
- A. Kechin, U. Boyarskikh, A. Kel and M. Filipenko, cutPrimers: A New Tool for Accurate Cutting of Primers from Reads of Targeted Next Generation Sequencing, J. Comput. Biol., 2017, 24, 1138–1143 CrossRef CAS PubMed.
- B. J. Callahan, P. J. McMurdie, M. J. Rosen, A. W. Han, A. J. Johnson and S. P. Holmes, DADA2: High-resolution sample inference from Illumina amplicon data, Nat. Methods, 2016, 13, 581–583 CrossRef CAS PubMed.
- B. Bushnell, J. Rood and E. Singer, BBMerge - Accurate paired shotgun read merging via overlap, PLoS One, 2017, 12, e0185056 CrossRef PubMed.
- N. A. Bokulich, B. D. Kaehler, J. R. Rideout, M. Dillon, E. Bolyen, R. Knight, G. A. Huttley and J. Gregory Caporaso, Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2's q2-feature-classifier plugin, Microbiome, 2018, 6, 90 CrossRef PubMed.
- P. Dixon, VEGAN, a package of R functions for community ecology, J. Veg. Sci., 2003, 14, 927–930 CrossRef.
- M. Dufrene and P. Legendre, Species assemblages and indicator species: The need for a flexible asymmetrical approach, Ecol. Monogr., 1997, 67, 345–366 Search PubMed.
- W. G. W. Pathirana, S. P. Chubb, M. J. Gillett and S. D. Vasikaran, Faecal Calprotectin, Clin. Biochem. Rev., 2018, 39, 77–90 Search PubMed.
- A. T. Kunzmann, H. G. Coleman, W. Y. Huang, C. M. Kitahara, M. M. Cantwell and S. I. Berndt, Dietary fiber intake and risk of colorectal cancer and incident and recurrent adenoma in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial, Am. J. Clin. Nutr., 2015, 102, 881–890 CrossRef CAS PubMed.
- L. R. Jacobs and J. R. Lupton, Relationship between colonic luminal pH, cell proliferation, and colon carcinogenesis in 1,2-dimethylhydrazine treated rats fed high fiber diets, Cancer Res., 1986, 46, 1727–1734 CAS.
- C. Avivi-Green, Z. Madar and B. Schwartz, Pectin-enriched diet affects distribution and expression of apoptosis-cascade proteins in colonic crypts of dimethylhydrazine-treated rats, Int. J. Mol. Med., 2000, 6, 689–698 CAS.
- O. Kazuko, N. Seiko, T. Satomi, I. Kikuji, M. Tomotari, I. Toshio, H. Kyoji and N. Tatsuji, Inhibitory effect of apple pectin and culture condensate of bifidobacterium longum on colorectal tumors induced by l, 2-dimethylhydrazine in transgenic mice harboring human prototype c-ha-ras genes, Exp. Anim., 2000, 49, 305–307 CrossRef PubMed.
- G. Jacobasch, G. Dongowski, S. Florian, K. Muller-Schmehl, B. Raab and D. Schmiedl, Pectin does not inhibit intestinal carcinogenesis in APC-deficient Min/+ mice, J. Agric. Food Chem., 2008, 56, 1501–1510 CrossRef CAS PubMed.
- A. Ferreira-Lazarte, J. Fernandez, P. Gallego-Lobillo, C. J. Villar, F. Lombo, F. J. Moreno and M. Villamiel, Behaviour of citrus pectin and modified citrus pectin in an azoxymethane/dextran sodium sulfate (AOM/DSS)-induced rat colorectal carcinogenesis model, Int. J. Biol. Macromol., 2021, 167, 1349–1360 CrossRef CAS PubMed.
- H. G. Bauer, N. G. Asp, R. Oste, A. Dahlqvist and P. E. Fredlund, Effect of dietary fiber on the induction of colorectal tumors and fecal beta-glucuronidase activity in the rat, Cancer Res., 1979, 39, 3752–3756 CAS.
- H. J. Freeman, G. A. Spiller and Y. S. Kim, A double-blind study on the effects of differing purified cellulose and pectin fiber diets on 1,2-dimethylhydrazine-induced rat colonic neoplasia, Cancer Res., 1980, 40, 2661–2665 CAS.
- H. J. Freeman, Effects of differing purified cellulose, pectin, and hemicellulose fiber diets on fecal enzymes in 1,2-dimethylhydrazine-induced rat colon carcinogenesis, Cancer Res., 1986, 46, 5529–5532 CAS.
- H. G. Bauer, N. G. Asp, A. Dahlqvist, P. E. Fredlund, M. Nyman and R. Oste, Effect of two kinds of pectin and guar gum on 1,2-dimethylhydrazine initiation of colon tumors and on fecal beta-glucuronidase activity in the rat, Cancer Res., 1981, 41, 2518–2523 CAS.
- K. Tazawa, H. Okami, I. Yamashita, Y. Ohnishi, K. Kobashi and M. Fujimaki, Anticarcinogenic action of apple pectin on fecal enzyme activities and mucosal or portal prostaglandin E2 levels in experimental rat colon carcinogenesis, J. Exp. Clin. Cancer Res., 1997, 16, 33–38 CAS.
- K. Watanabe, B. S. Reddy, J. H. Weisburger and D. Kritchevsky, Effect of dietary alfalfa, pectin, and wheat bran on azoxymethane-or methylnitrosourea-induced colon carcinogenesis in F344 rats, J. Natl. Cancer Inst., 1979, 63, 141–145 CAS.
- A. McIntyre, P. R. Gibson and G. P. Young, Butyrate production from dietary fibre and protection against large bowel cancer in a rat model, Gut, 1993, 34, 386–391 CrossRef CAS PubMed.
- J. Yang, H. Wei, Y. F. Lin, E. S. H. Chu, Y. F. Zhou, H. Y. Gou, S. Guo, H. C. H. Lau, A. H. K. Cheung, H. R. Chen, K. F. To, J. J. Y. Sung, Y. Wang and J. Yu, High soluble fiber promotes colorectal tumorigenesis through modulating gut microbiota and metabolites in mice, Gastroenterology, 2024, 166, 323–337 e7 CrossRef CAS PubMed.
- S. K. Gill, E. Lever, P. W. Emery and K. Whelan, Nutrient, fibre, sorbitol and chlorogenic acid content of prunes (Prunus domestica): an updated analysis and comparison of different countries of origin and database values, Int. J. Food Sci. Nutr., 2019, 70, 924–931 CrossRef PubMed.
- K. Kawabata, T. Yamamoto, A. Hara, M. Shimizu, Y. Yamada, K. Matsunaga, T. Tanaka and H. Mori, Modifying effects of ferulic acid on azoxymethane-induced colon carcinogenesis in F344 rats, Cancer Lett., 2000, 157, 15–21 CrossRef CAS PubMed.
- C. V. Rao, A. Rivenson, B. Simi and B. S. Reddy, Chemoprevention of colon carcinogenesis by dietary curcumin, a naturally occurring plant phenolic compound, Cancer Res., 1995, 55, 259–266 CAS.
- K. Matsunaga, M. Katayama, K. Sakata, T. Kuno, K. Yoshida, Y. Yamada, Y. Hirose, N. Yoshimi and H. Mori, Inhibitory Effects of Chlorogenic Acid on Azoxymethane-induced Colon Carcinogenesis in Male F344 Rats, Asian Pac. J. Cancer Prev., 2002, 3, 163–166 Search PubMed.
- H. J. Park, S. R. Davis, H. Y. Liang, D. W. Rosenberg and R. S. Bruno, Chlorogenic acid differentially alters hepatic and small intestinal thiol redox status without protecting against azoxymethane-induced colon carcinogenesis in mice, Nutr. Cancer, 2010, 62, 362–370 CrossRef CAS PubMed.
- Y. Morishita, N. Yoshimi, K. Kawabata, K. Matsunaga, S. Sugie, T. Tanaka and H. Mori, Regressive effects of various chemopreventive agents on azoxymethane-induced aberrant crypt foci in the rat colon, Jpn. J. Cancer Res., 1997, 88, 815–820 CrossRef CAS PubMed.
- R. Feng, Y. Lu, L. L. Bowman, Y. Qian, V. Castranova and M. Ding, Inhibition of activator protein-1, NF-kappaB, and MAPKs and induction of phase 2 detoxifying enzyme activity by chlorogenic acid, J. Biol. Chem., 2005, 280, 27888–27895 CrossRef CAS PubMed.
- J. A. Larrauri, P. Ruperez and F. SauraCalixto, Effect of drying temperature on the stability of polyphenols and antioxidant activity of red grape pomace peels, J. Agric. Food Chem., 1997, 45, 1390–1393 CrossRef CAS.
- P. Louis, G. L. Hold and H. J. Flint, The gut microbiota, bacterial metabolites and colorectal cancer, Nat. Rev. Microbiol., 2014, 12, 661–672 CrossRef CAS PubMed.
- D. R. Donohoe, L. B. Collins, A. Wali, R. Bigler, W. Sun and S. J. Bultman, The Warburg effect dictates the mechanism of butyrate-mediated histone acetylation and cell proliferation, Mol. Cell, 2012, 48, 612–626 CrossRef CAS PubMed.
- K. Weitkunat, S. Schumann, K. J. Petzke, M. Blaut, G. Loh and S. Klaus, Effects of dietary inulin on bacterial growth, short-chain fatty acid production and hepatic lipid metabolism in gnotobiotic mice, J. Nutr. Biochem., 2015, 26, 929–937 CrossRef CAS PubMed.
- D. P. Belobrajdic, S. A. Jobling, M. K. Morell, S. Taketa and A. R. Bird, Wholegrain barley β-glucan fermentation does not improve glucose tolerance in rats fed a high-fat diet, Nutr. Res., 2015, 35, 162–168 CrossRef CAS PubMed.
- G. Caderni, C. Luceri, C. De Filippo, M. Salvadori, A. Giannini, L. Tessitore and P. Dolara, Slow-release pellets of sodium butyrate do not modify azoxymethane (AOM)-induced intestinal carcinogenesis in F344 rats, Carcinogenesis, 2001, 22, 525–527 CrossRef CAS PubMed.
- G. Caderni, C. Luceri, L. Lancioni, L. Tessitore and P. Dolara, Slow-release pellets of sodium butyrate increase apoptosis in the colon of rats treated with azoxymethane, without affecting aberrant crypt foci and colonic proliferation, Nutr. Cancer, 1998, 30, 175–181 CrossRef CAS PubMed.
- Y. Tian, Q. Xu, L. Sun, Y. Ye and G. Ji, Short-chain fatty acids administration is protective in colitis-associated colorectal cancer development, J. Nutr. Biochem., 2018, 57, 103–109 CrossRef CAS PubMed.
- B. S. Reddy and H. Mori, Effect of dietary wheat bran and dehydrated citrus fiber on 3,2′-dimethyl-4-aminobiphenyl-induced intestinal carcinogenesis in F344 rats, Carcinogenesis, 1981, 2, 21–25 CrossRef CAS PubMed.
- D. Maria-Ferreira, A. M. Nascimento, T. R. Cipriani, A. P. Santana-Filho, P. D. S. Watanabe, D. M. G. Sant Ana, F. B. Luciano, K. C. P. Bocate, R. M. van den Wijngaard, M. F. P. Werner and C. H. Baggio, Rhamnogalacturonan, a chemically-defined polysaccharide, improves intestinal barrier function in DSS-induced colitis in mice and human Caco-2 cells, Sci. Rep., 2018, 8, 12261 CrossRef PubMed.
- Y. H. Sheng, S. Triyana, R. Wang, I. Das, K. Gerloff, T. H. Florin, P. Sutton and M. A. McGuckin, MUC1 and MUC13 differentially regulate epithelial inflammation in response to inflammatory and infectious stimuli, Mucosal Immunol., 2013, 6, 557–568 CrossRef CAS PubMed.
- I. T. Johnson and J. M. Gee, Gastrointestinal adaptation in response to soluble non-available polysaccharides in the rat, Br. J. Nutr., 1986, 55, 497–505 CrossRef CAS PubMed.
- S. Judex, Y. K. Luu, E. Ozcivici, B. Adler, S. Lublinsky and C. T. Rubin, Quantification of adiposity in small rodents using micro-CT, Methods, 2010, 50, 14–19 CrossRef CAS PubMed.
- D. A. Brockman, X. Chen and D. D. Gallaher, High-viscosity dietary fibers reduce adiposity and decrease hepatic steatosis in rats fed a high-fat diet, J. Nutr., 2014, 144, 1415–1422 CrossRef CAS PubMed.
- J. Ou, F. Carbonero, E. G. Zoetendal, J. P. DeLany, M. Wang, K. Newton, H. R. Gaskins and S. J. O'Keefe, Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans, Am. J. Clin. Nutr., 2013, 98, 111–120 CrossRef CAS PubMed.
- S. Esquivel-Elizondo, Z. E. Ilhan, E. I. Garcia-Pena and R. Krajmalnik-Brown, Insights into butyrate production in a controlled fermentation system via gene predictions, mSystems, 2017, 2 DOI:10.1128/msystems.00051-17.
- E. J. C. Galvez, A. Iljazovic, L. Amend, T. R. Lesker, T. Renault, S. Thiemann, L. Hao, U. Roy, A. Gronow, E. Charpentier and T. Strowig, Distinct Polysaccharide Utilization Determines Interspecies Competition between Intestinal Prevotella spp, Cell Host Microbe, 2020, 28, 838–852 CrossRef CAS PubMed.
- X. Meng and Q. Shu, Novel primers to identify a wider diversity of butyrate-producing bacteria, World J. Microbiol. Biotechnol., 2024, 40, 76 CrossRef CAS PubMed.
- B. J. Smith, B. Hatter, K. Washburn, J. Graef-Downard, B. A. Ojo, G. D. El-Rassi, R. H. Cichewicz, M. Payton and E. A. Lucas, Dried Plum's Polyphenolic Compounds and Carbohydrates Contribute to Its Osteoprotective Effects and Exhibit Prebiotic Activity in Estrogen Deficient C57BL/6 Mice, Nutrients, 2022, 14, 1685 CrossRef CAS PubMed.
- N. Gaci, G. Borrel, W. Tottey, P. W. O'Toole and J. F. Brugere, Archaea and the human gut: new beginning of an old story, World J. Gastroenterol., 2014, 20, 16062–16078 CrossRef CAS PubMed.
- H. Abdi, D. M. Kordi-Tamandani, M. Lagzian and A. Bakhshipour, Archaeome in colorectal cancer: High abundance of methanogenic Archaea in colorectal cancer patients, Int. J. Cancer Manag., 2022, 15, e117843 Search PubMed.
- T. Li, O. O. Coker, Y. Sun, S. Li, C. Liu, Y. Lin, S. H. Wong, Y. Miao, J. J. Y. Sung and J. Yu, Multi-Cohort Analysis Reveals Altered Archaea in Colorectal Cancer Fecal Samples Across Populations, Gastroenterology, 2025, 168, 525–538 CrossRef CAS PubMed.
- M. J. Hill, Bile flow and colon cancer, Mutat. Res., 1990, 238, 313–320 CAS.
- B. S. Reddy and E. L. Wynder, Large-bowel carcinogenesis: fecal constituents of populations with diverse incidence rates of colon cancer, J. Natl. Cancer Inst., 1973, 50, 1437–1442 CrossRef CAS PubMed.
- O. M. Jensen, R. MacLennan and J. Wahrendorf, Diet, bowel function, fecal characteristics, and large bowel cancer in Denmark and Finland, Nutr. Cancer, 1982, 4, 5–19 CrossRef CAS PubMed.
- L. F. Tinker, B. O. Schneeman, P. A. Davis, D. D. Gallaher and C. R. Waggoner, Consumption of prunes as a source of dietary fiber in men with mild hypercholesterolemia, Am. J. Clin. Nutr., 1991, 53, 1259–1265 CrossRef CAS PubMed.
- E. A. Al Hinai, P. Kullamethee, I. R. Rowland, J. Swann, G. E. Walton and D. M. Commane, Modelling the role of microbial p-cresol in colorectal genotoxicity, Gut Microbes, 2019, 10, 398–411 CrossRef CAS PubMed.
- T. Bansal, R. C. Alaniz, T. K. Wood and A. Jayaraman, The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 228–233 CrossRef CAS PubMed.
- A. Birkett, J. Muir, J. Phillips, G. Jones and K. O'Dea, Resistant starch lowers fecal concentrations of ammonia and phenols in humans, Am. J. Clin. Nutr., 1996, 63, 766–772 CrossRef CAS PubMed.
- S. Paul, A. J. DeCastro, H. J. Lee, A. K. Smolarek, J. Y. So, B. Simi, C. X. Wang, R. Zhou, A. M. Rimando and N. Suh, Dietary intake of pterostilbene, a constituent of blueberries, inhibits the beta-catenin/p65 downstream signaling pathway and colon carcinogenesis in rats, Carcinogenesis, 2010, 31, 1272–1278 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |