Open Access Article
Open Access ArticleAssociation between mushroom consumption and digit span performance among middle-aged and older community-dwelling Japanese: the NILS-LSA project
Shu
Zhang
 a,
Djibril M.
Ba
a,
Djibril M.
Ba
 b,
Yukiko
Nishita
b,
Yukiko
Nishita
 a,
Chikako
Tange
a,
Chikako
Tange
 a,
Junjia
Zhu
a,
Junjia
Zhu
 b,
Rei
Otsuka
b,
Rei
Otsuka
 *a and
Joshua
Muscat
*a and
Joshua
Muscat
 b
b
aDepartment of Epidemiology of Aging, Research Institute, National Center for Geriatrics and Gerontology, Obu, Japan. Tel: +81-562-46-2311E-mail: otsuka@ncgg.go.jp
bPenn State College Medicine, Department of Public Health Sciences, Hershey, USA
First published on 4th March 2026
Abstract
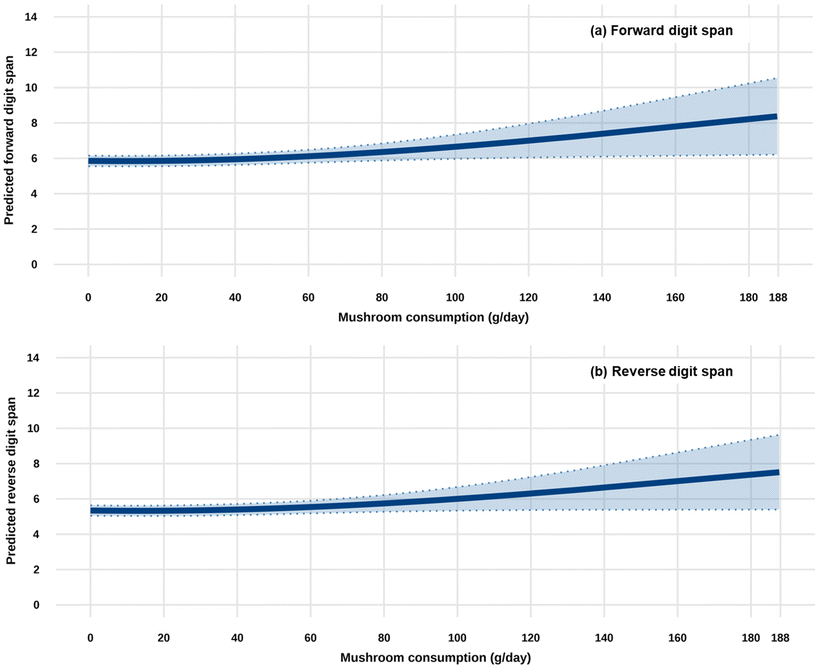
Cognitive decline, particularly in short-term and working memory, is a major concern in aging populations. Mushrooms contain bioactive compounds that may support brain health, but the association between mushroom consumption and specific memory functions remains unclear. This study investigated the association between mushroom intake and digit span performance among 3162 Japanese adults (mean age 57.9 ± 13.1 years; mean follow-up duration 10.5 ± 6.1 years) from the National Institute for Longevity Sciences-Longitudinal Study of Aging (2002–2022). Mushroom consumption (g/day) at baseline was assessed using 3-day dietary records. Short-term and working memory, which are fundamental components of general cognitive ability, were evaluated using forward and reverse digit span scores (range: 0–14 points for each) from the Wechsler Adult Intelligence Scale–Revised. Mixed-effects models with splines, adjusted for demographic, dietary, health, and socioeconomic factors, revealed significant non-linear associations: higher mushroom consumption was linked to better performance on forward digit span (full-adjusted spline terms β1 = 1.192, p = 0.0117; β2 = 2.549, p = 0.0235) and reverse digit span (full-adjusted spline terms β1 = 0.974, p = 0.0352; β2 = 2.209, p = 0.0447) tests, with benefits most pronounced at moderate to high intake levels. These findings suggest that regular mushroom consumption, measured in grams per day, may enhance short-term and working memory in middle-aged and older adults, highlighting a potentially simple dietary approach to support cognitive function during aging.
Introduction
As societies worldwide face rapid aging, the preservation of cognitive function has become a critical public health priority. Short-term memory, which represents one of the most fundamental cognitive processes underlying human cognition and daily functioning, involves the temporary storage of information and is a core component of the working memory system.1 Working memory, the broader cognitive system that encompasses short-term memory along with the ability to manipulate stored information, is crucial for daily activities and independent living.2,3 The challenge lies in the fact that these critical memory systems are significantly affected by normal aging,4 making them important targets for intervention strategies.Recent research has highlighted the potential role of dietary factors in cognitive health. Mushrooms are traditionally common dietary items in many parts of Asia, including Japan, whereas their consumption was lower in Western populations. For example, in 2019, the per capita consumption of fresh mushrooms in Japan and the United States was 6.2 kg (ref. 5) and 1.7 kg,6 respectively. Edible mushrooms represent a unique functional food characterized by their rich concentration of bioactive compounds rarely found elsewhere in the human diet,7–9 including but not limited to powerful antioxidants, anti-inflammatory agents, neuroprotective substances, and gut-brain axis modulators (such as ergothioneine, phenolic compounds, hericenones, erinacines, β-glucans, etc.).
Epidemiological and clinical investigations have increasingly explored the neuroprotective effects of mushroom consumption. Notably, clinical trials involving specific species, such as Hericium erinaceus, have demonstrated significant cognitive improvements among individuals with mild impairment.10 While cross-sectional findings link higher intake to a reduced risk of mild cognitive impairment11 and enhanced performance in specific cognitive tasks,12 longitudinal evidence remains predominantly focused on late-stage outcomes such as incident dementia.13 However, while these findings are promising, evidence remains limited regarding the long-term changes of specific memory domains—such as short-term and working memory—especially when utilizing precise dietary assessment methods like 3-day food records. The objective of this study was to fill this gap by examining whether higher mushroom consumption is linked to better performance on short-term and working memory tasks in real-world populations using data from a well-established longitudinal cohort study. In this study, these specific memory functions were measured using the digit span test, a standardized component of the Wechsler Adult Intelligence Scale.14,15 Widely considered representative of memory tasks, the digit span test provides a reliable measure that correlates significantly with cognitive ability in the domains of short-term and working memory.16,17
Methods
Study cohort and participants
Data for this study originated from the National Institute for Longevity Sciences-Longitudinal Study of Aging (NILS-LSA) project. NILS-LSA represents a Japanese community-based longitudinal cohort investigation examining natural aging processes and age-associated pathologies. Study participants were selected through age- and sex-stratified random sampling from residential areas surrounding the research institute, specifically from Obu City and Higashiura Town located in Aichi Prefecture, Japan. The initial wave of NILS-LSA surveys occurred between November 1997 and April 2000, encompassing 2267 individuals aged 40–79 years. These individuals underwent biennial follow-up evaluations, with new randomly selected, age- and sex-matched participants replacing those (aged ≤79 years) unable to continue participation in subsequent investigations. Additionally, new 40-year-old participants were enrolled annually. Beginning in October 2013, follow-up investigations were initiated among all participants from the first through seventh survey waves. These comprised the eighth survey wave (October 2013 to February 2016), the ninth survey wave (October 2018 to February 2022), and the currently ongoing tenth survey wave (commenced February 2023). Details of the NILS-LSA have been reported previously.18Participants for this study were selected from the third (May 2002 to May 2004) to the ninth (October 2018 to February 2022) waves of the NILS-LSA, as data on short-term and working memory tasks were available for these periods. Of the initial 3385 participants, we excluded participants with missing baseline (the first participation) data for mushroom consumption (n = 160) or digit span performance (n = 15); those with missing information on (n = 1) or a self-reported history of (n = 5) dementia; and those with missing baseline data for relevant covariates (n = 42). The final analytical sample comprised 3162 participants (49.9% male), ranging in age from 40 to 85 (mean 57.9 ± 13.1) years. The mean follow-up duration was 10.5 ± 6.1 years.
Mushroom consumption assessment (at baseline)
Food consumption, including mushrooms, was assessed using 3-day dietary records completed over 3 continuous days (2 weekdays and 1 weekend day). Most participants filled out these records at home and returned them within 1 month. Food portions were either weighed separately using a 1 kg kitchen scale (Sekisui Jushi, Tokyo, Japan) before cooking or estimated by portion size. To supplement the written records, participants photographed their meals before and after eating using disposable cameras (27 shots; Fuji Film, Tokyo, Japan). Dietitians utilized these photographs to address missing information and contacted participants by telephone when discrepancies arose or additional clarification was needed.Cognitive assessment
Digit span performance was assessed using the digit span test from the Japanese Wechsler Adult Intelligence Scale-Revised (JWAIS-R),14,15 which evaluates short-term and working memory function. This test comprises two components designed to evaluate different aspects of memory function. The forward digit span measures short-term memory capacity, requiring participants to repeat number sequences in the same order as presented (e.g., hearing “6-3-8-2” and responding “6-3-8-2”). The reverse digit span assesses working memory, where participants must repeat number sequences in reverse order (e.g., hearing “3-7-4-1” and responding “1-4-7-3”). Scoring was based on correct responses, with one point awarded for accuracy and zero points for incorrect answers. The forward digit span started with sequences of 3 digits, and the reverse digit span started with sequences of 2 digits, with both progressively increased in length, with two trials at each sequence length. Testing continued until the participant failed both trials at a given sequence length, with a maximum possible score of 14 points for each test component.Covariates
In the baseline survey, body mass index (BMI; kg/m2; continuous) was computed as weight (kg) divided by height (m) squared using digital scale measurements. A self-administered questionnaire captured medical history including hypertension, heart disease, dyslipidemia, and diabetes (yes or no for each condition), along with smoking status (never, former, or current) and educational attainment (years; continuous). Physical activity over 24 hours was quantified using Metabolic Equivalent of Task (MET) scores (METs-h per day; continuous), obtained through participant interviews conducted by trained personnel using a semi-quantitative assessment approach.19 Depressive symptoms were evaluated using the self-reported Center for Epidemiologic Studies Depression (CES-D) Scale,20,21 with scores categorized as ≤15 (normal status) or ≥16 (presence of relevant symptoms). Dietary assessments from 3-day records provided consumption of seafood, vegetables, and fruit (g/day; continuous) and estimates of total energy intake (kcal/day; continuous; calculated based on the Standard Tables of Foods Composition 2010 in Japan and other established sources22,23). Genomic DNA was extracted from the peripheral blood lymphocytes using standard procedures. APOE genotypes were determined using polymerase chain reaction amplification.24Statistical analysis
Given the potential sex differences in dietary patterns, including mushroom consumption, and cognitive performance characteristics, baseline characteristics were compared between male and female participants using the Wilcoxon rank sum test for continuous variables and Pearson's chi-squared test for categorical variables.Mixed-effects models with natural splines25 were employed to estimate the associations between mushroom consumption and digit span performance in the total population and stratified by sex. Linear mixed-effects models were selected as the primary analytical approach to account for the inherent correlation between repeated measurements from the same participant over multiple survey waves and to accommodate missing data, thereby maximizing the use of longitudinal information. The crude models were adjusted for baseline information including centered age (years; centered at mean value), square of centered age, sex, APOE genotype (APOE-ε4 carriers: 2/4, 3/4, 4/4, or APOE-ε4 noncarriers: 2/2, 2/3, 3/3), follow-up time (years), and survey seasons (Spring: March to May, Summer: June to August, Autumn: September to November, Winter: December to February of the following year). Age was centered prior to creating the quadratic term to reduce multicollinearity between the linear and squared age variables. The full models were further adjusted for baseline information on energy intake (kcal/day; sex-specific tertiles); consumption of seafood, vegetables, and fruit (g/day; sex-specific tertiles), which were included due to their documented associations with cognitive performance and thus serve as proxies for overall dietary quality;26 BMI (kg/m2; <18.5, 18.5–<25.0, or ≥25.0); smoking status (current, former, or never); total physical activity (METs-h per day; centered at mean value); education level (≤9, 10–12, or ≥13 years), utilized as a robust proxy for socioeconomic status27 to avoid potential multicollinearity and model overfitting associated with multiple socioeconomic indicators; medical history (yes or no for hypertension, heart disease, dyslipidemia, and diabetes); and depressive symptoms (CES-D scale score <16 or ≥16).
Model selection for the optimal number of spline degrees of freedom (df) was conducted by comparing models with 1 to 3 df using Akaike Information Criterion (AIC), Bayesian Information Criterion (BIC), and log-likelihood values. Additionally, models with and without interaction terms between mushroom consumption and age were compared. As models without interaction terms consistently yielded lower AIC and BIC values, Model 2 (df = 2) was selected as providing the optimal balance between goodness of fit and model complexity for both memory components (SI Table S1). Natural splines were utilized instead of traditional linear terms to allow for the detection of potential non-linear dose–response relationships (e.g., threshold or saturation effects), which are biologically plausible in nutritional research and have been widely applied in studies evaluating food components and health outcomes.28–30 For both forward digit span (short-term memory) and reverse digit span (working memory), the final selected models employed natural splines with 2 df, corresponding to one internal knot. Knot placement followed the default quantile-based positioning implemented in the ns() function of the R splines package, which places internal knots at equally spaced quantiles of the predictor variable distribution (approximately at the median of mushroom consumption in this study). According to the characteristics summarized in SI Table S1 footnotes c–e, the model with 1 df generally captures only a linear or slightly curved trend, while the model with 3 df introduces a more complex, highly flexible curve that may capture local variations but risks overfitting. In contrast, the 2-df spline represents an intermediate level of flexibility—able to model modest curvature without unnecessary complexity—thus providing a parsimonious yet adequately responsive fit to the observed data.
Sensitivity analyses were conducted to assess the potential influence of extreme values on the observed associations. Given the right-skewed distribution of mushroom consumption, values exceeding the 99th percentile (58.4 g/day) were winsorized to this threshold value, and the primary mixed-effects models were refitted to examine whether the associations remained consistent.
All statistical analyses were two-sided, with a p < 0.05 considered significant. Statistical analyses were conducted using R software (version 4.5.1; R Foundation for Statistical Computing, Vienna, Austria) and RStudio (version 2025.05.1; RStudio Inc., Boston, MA, USA).
Results
Participant characteristics
Baseline characteristics differed significantly between male and female participants (Table 1). The proportion of participants with zero mushroom consumption was similar between males and females. Among mushroom consumers, consumption levels were comparable between sexes. Males had significantly higher body mass index, higher prevalence of diabetes, and substantially higher smoking rates compared to females. In contrast, females showed higher prevalence of dyslipidemia, were more likely to have lower educational attainment, and also reported higher total physical activity levels. Dietary patterns also differed by sex. Males had significantly higher energy intake, greater seafood consumption, and higher vegetable consumption. Conversely, females consumed more fruit.| All participants (n = 3162) | Male (n = 1578) | Female (n = 1584) | p-Valuea | |
|---|---|---|---|---|
| a Estimated using Wilcoxon rank sum test for continuous variables and Pearson's Chi-squared test for categorical variables. | ||||
| Participants with 0 mushroom consumption (g/day), % | 20.5 | 21.3 | 19.8 | 0.286 |
| Mushroom consumption (g/day; participants with 0 consumption were excluded), mean (SD) | 15.3 (13.8) | 15.1 (13.8) | 15.4 (13.8) | 0.369 |
| Age (years), mean (SD) | 57.9 (13.1) | 57.8 (13.0) | 58.0 (13.2) | 0.745 |
| Body mass index (kg/m2), mean (SD) | 22.8 (3.1) | 23.2 (2.9) | 22.5 (3.3) | <0.001 |
| Medical history (present), % | ||||
| Hypertension | 25.3 | 25.7 | 24.8 | 0.553 |
| Heart disease | 4.9 | 5.2 | 4.5 | 0.395 |
| Dyslipidemia | 17.3 | 15.6 | 19.1 | 0.010 |
| Diabetes | 7.0 | 8.7 | 5.3 | <0.001 |
| Current smoker, % | 17.5 | 29.0 | 6.1 | <0.001 |
| Total physical activity (METs-h per day), mean (SD) | 32.4 (3.6) | 31.9 (4.2) | 32.9 (2.8) | <0.001 |
| Education ≤9 years, % | 21.2 | 19.3 | 23.0 | 0.009 |
| Depressive symptoms (present), % | 12.5 | 11.4 | 13.6 | 0.065 |
| Energy intake (kcal/day), mean (SD) | 2058.0 (420.3) | 2254.1 (416.4) | 1862.7 (321.7) | <0.001 |
| Seafood consumption (g/day), mean (SD) | 92.9 (53.9) | 104.6 (58.8) | 81.3 (45.7) | <0.001 |
| Vegetables consumption (g/day), mean (SD) | 277.5 (121.7) | 285.1 (127.4) | 269.9 (115.2) | 0.002 |
| Fruit consumption (g/day), mean (SD) | 133.0 (118.2) | 119.4 (121.3) | 146.5 (113.5) | <0.001 |
Association between mushroom consumption and digit span performance
Table 2 and Fig. 1 demonstrate the longitudinal association between mushroom consumption and digit span performance. With increasing mushroom consumption, both forward and reverse digit span scores exhibited an upward trend. Notably, this positive association was statistically significant for forward digit span scores (p for overall trend = 0.0403; full-adjusted spline terms β1 = 1.192, p = 0.0117; β2 = 2.549, p = 0.0235). For reverse digit span scores, although the overall association was marginally significant (p for overall trend = 0.0995), the individual spline terms remained significant, suggesting a consistent positive non-linear trend (full-adjusted spline terms β1 = 0.974, p = 0.0352; β2 = 2.209, p = 0.0447). In spline models, β coefficients do not represent simple linear effects; rather, they describe the contribution of each basis function. The first term generally reflects the overall trend, while the second captures curvature, indicating a modestly non-linear positive association between mushroom consumption and cognitive performance. The improvement in digit span scores was modest at low to moderate mushroom consumption levels, whereas the enhancement became more pronounced at moderate to high consumption levels.| Forward digit-span | Reverse digit-span | |||||
|---|---|---|---|---|---|---|
| β value | Std. error | p-Value | β value | Std. error | p-Value | |
| a Analyzed by mixed-effects models with natural splines. b Adjusted for baseline information including centered age (years; centered at mean value), square of centered age, sex, APOE genotype (APOE-ε4 carriers: 2/4, 3/4, 4/4, or APOE-ε4 noncarriers: 2/2, 2/3, 3/3), follow-up time (years), and survey seasons (Spring: March to May, Summer: June to August, Autumn: September to November, Winter: December to February of the following year). c Adjusted for baseline information including centered age (years; centered at mean value), square of centered age, sex, APOE genotype (APOE-ε4 carriers: 2/4, 3/4, 4/4, or APOE-ε4 noncarriers: 2/2, 2/3, 3/3), follow-up time (years), survey seasons (Spring: March to May, Summer: June to August, Autumn: September to November, Winter: December to February of the following year), energy intake (kcal/day; sex-specific tertiles), consumption of seafood, vegetables, and fruit (g/day; sex-specific tertiles), body mass index (kg/m2; <18.5, 18.5–<25.0, or ≥25.0), smoking status (current, former, or never), total physical activity (METs-h per day; centered at mean value), education level (≤9, 10–12, or ≥13 years), medical history (yes or no for hypertension, heart disease, dyslipidemia, and diabetes), and depressive symptoms (CES-D scale score <16 or ≥16). d Often resembles a linear function or a slightly curved line. Captures the overall trend in the data. e Typically has a more pronounced curvature. Often resembles a quadratic or cubic function. | ||||||
| Crude model | ||||||
| Natural spline (mushroom consumption, df = 2) (basis function 1)b,d | 1.244 | 0.479 | 0.0099 | 1.107 | 0.464 | 0.0170 |
| Natural spline (mushroom consumption, df = 2) (basis function 2)b,e | 2.175 | 1.156 | 0.0600 | 2.055 | 1.118 | 0.0663 |
| For overall trend | 0.0277 | 0.0535 | ||||
| Full adjusted model | ||||||
| Natural spline (mushroom consumption, df = 2) (basis function 1)c,d | 1.192 | 0.473 | 0.0117 | 0.974 | 0.462 | 0.0352 |
| Natural spline (mushroom consumption, df = 2) (basis function 2)c,e | 2.549 | 1.125 | 0.0235 | 2.209 | 1.100 | 0.0447 |
| For overall trend | 0.0403 | 0.0995 | ||||
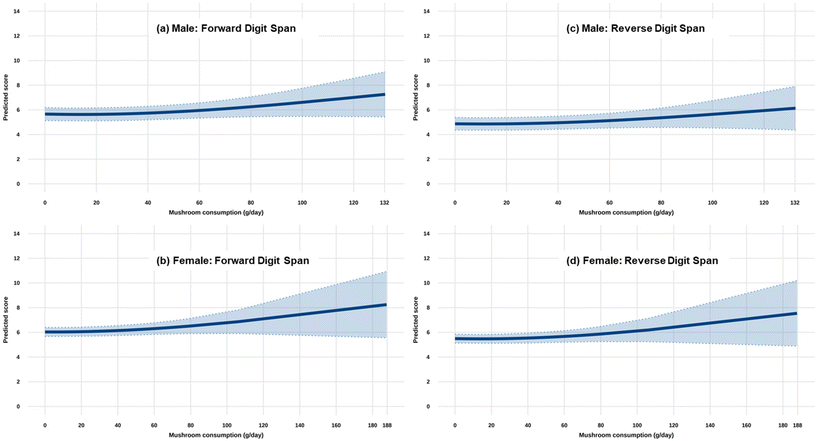
Sex-stratified analyses revealed consistent patterns generally mirroring the main findings, with both males and females demonstrating positive trends between mushroom consumption and digit span performance (Fig. 2). However, these associations did not reach statistical significance in the stratified analyses (all p-values for overall trend >0.05; Table 3). Specifically, both forward and reverse digit span performance showed consistent positive associations (positive β values) with mushroom consumption in both sexes. The lack of statistical significance in these subgroups, despite effect sizes (coefficients) being comparable to or even larger than those in the overall analysis, is likely attributable to the reduced statistical power and increased standard errors resulting from the smaller sample sizes after stratification.
| Forward digit-span | Reverse digit-span | |||||
|---|---|---|---|---|---|---|
| β value | Std. error | p-Value | β value | Std. error | p-Value | |
| a Analyzed by mixed-effects models with natural splines. b Adjusted for baseline information including centered age (years; centered at mean value), square of centered age, APOE genotype (APOE-ε4 carriers: 2/4, 3/4, 4/4, or APOE-ε4 noncarriers: 2/2, 2/3, 3/3), follow-up time (years), survey seasons (Spring: March to May, Summer: June to August, Autumn: September to November, Winter: December to February of the following year), energy intake (kcal/day; sex-specific tertiles), consumption of seafood, vegetables, and fruit (g/day; sex-specific tertiles), body mass index (kg/m2; <18.5, 18.5–<25.0, or ≥25.0), smoking status (current, former, or never), total physical activity (METs-h per day; centered at mean value), education level (≤9, 10–12, or ≥13 years), medical history (yes or no for hypertension, heart disease, dyslipidemia, and diabetes), and depressive symptoms (CES-D scale score <16 or ≥16). c Often resembles a linear function or a slightly curved line. Captures the overall trend in the data. d Typically has a more pronounced curvature. Often resembles a quadratic or cubic function. | ||||||
| Male (n = 1578) | ||||||
| Natural spline (mushroom consumption, df = 2) (basis function 1)c | 0.649 | 0.403 | 0.1081 | 0.562 | 0.391 | 0.1508 |
| Natural spline (mushroom consumption, df = 2) (basis function 2)d | 1.650 | 0.929 | 0.0760 | 1.290 | 0.900 | 0.1520 |
| For overall trend | 0.1801 | 0.2958 | ||||
| Female (n = 1584) | β value | Std. error | p-Value | β value | Std. error | p-Value |
| Natural spline (mushroom consumption, df = 2) (basis function 1)c | 1.121 | 0.614 | 0.0680 | 0.900 | 0.607 | 0.1387 |
| Natural spline (mushroom consumption, df = 2) (basis function 2)d | 2.216 | 1.393 | 0.1120 | 2.088 | 1.377 | 0.1295 |
| For overall trend | 0.1865 | 0.2907 | ||||
Sensitivity analysis with winsorized mushroom consumption values
To assess the potential influence of extreme values on the observed associations, we conducted sensitivity analyses by winsorizing mushroom consumption values exceeding the 99th percentile (58.4 g/day) to this threshold (Table 4). The results demonstrated patterns consistent with the primary analyses, although the statistical significance was slightly attenuated due to the restricted exposure range. For forward digit span, the overall association was marginally significant (p for overall trend = 0.0511). However, the second spline term remained statistically significant (β2 = 0.470, p = 0.0149), while the first term was not (β1 = 0.056, p = 0.7167), reinforcing the presence of a non-linear positive curvature where beneficial effects are more pronounced at moderate-to-high intake levels. For reverse digit span, the association did not reach statistical significance in this sensitivity analysis (p for overall trend = 0.263), likely due to the reduced variation in high-intake data, though the coefficients remained positive (full-adjusted spline terms β1 = 0.040, p = 0.7905; β2 = 0.309, p = 0.1023).| Forward digit-span | Reverse digit-span | |||||
|---|---|---|---|---|---|---|
| β value | Std. error | p-Value | β value | Std. error | p-Value | |
| a Analyzed by mixed-effects models with natural splines. b Adjusted for baseline information including centered age (years; centered at mean value), square of centered age, APOE genotype (APOE-ε4 carriers: 2/4, 3/4, 4/4, or APOE-ε4 noncarriers: 2/2, 2/3, 3/3), follow-up time (years), survey seasons (Spring: March to May, Summer: June to August, Autumn: September to November, Winter: December to February of the following year), energy intake (kcal/day; sex-specific tertiles), consumption of seafood, vegetables, and fruit (g/day; sex-specific tertiles), body mass index (kg/m2; <18.5, 18.5–<25.0, or ≥25.0), smoking status (current, former, or never), total physical activity (METs-h per day; centered at mean value), education level (≤9, 10–12, or ≥13 years), medical history (yes or no for hypertension, heart disease, dyslipidemia, and diabetes), and depressive symptoms (CES-D scale score <16 or ≥16). c Often resembles a linear function or a slightly curved line. Captures the overall trend in the data. d Typically has a more pronounced curvature. Often resembles a quadratic or cubic function. | ||||||
| Natural spline (mushroom consumption, df = 2) (basis function 1) c | 0.056 | 0.153 | 0.7167 | 0.040 | 0.150 | 0.7905 |
| Natural spline (mushroom consumption, df = 2) (basis function 2) d | 0.470 | 0.193 | 0.0149 | 0.309 | 0.189 | 0.1023 |
| For overall trend | 0.0511 | 0.2629 | ||||
Discussion
The present study investigated the longitudinal association between mushroom consumption and digit span performance over a mean follow-up period of 10.5 years among community-dwelling middle-aged and older Japanese adults. The results showed that mushroom consumption significantly predicted better forward and reverse digit span scores through a non-linear association.The observed nonlinear associations between mushroom consumption and digit span performance could potentially be attributed to several hypothesized neurobiological mechanisms involving mushroom-derived bioactive compounds. For instance, animal studies have suggested that erinacines may stimulate nerve growth factor synthesis, particularly benefiting hippocampal regions essential for memory processing.7 Additionally, ergothioneine, which accumulates in brain tissues,31,32 is thought to potentially protect neurons against oxidative damage,33 thereby potentially supporting working memory.34 Similarly, the modulation of gut-brain axis by β-glucans is hypothesized to contribute to cognition enhancement and the delaying neurodegeneration.9 These and other bioactive compounds may support short-term memory and working memory, which rely heavily on prefrontal cortex function and cholinergic neurotransmission,35,36 and are sensitive to oxidative stress.37 However, it is essential to emphasize that these remain potential mechanisms derived primarily from preclinical evidence and cannot be directly verified within the framework of the current study.
The lack of statistical significance in sex-stratified analyses, despite consistent effect directions and magnitudes, can be primarily attributed to reduced statistical power when the sample was divided by sex (males: n = 1578; females: n = 1584). This power reduction was compounded by increased standard errors, particularly in females where standard errors increased by approximately 30% compared to the overall model. However, it is noteworthy that there was little heterogeneity between the sex-specific findings. Both males and females demonstrated consistent positive associations (positive β values) with mushroom consumption. This high degree of consistency reinforces the validity of our overall findings, suggesting that the observed cognitive benefits of mushroom intake are likely applicable to both Japanese men and women within this age range.
The sensitivity analyses, which capped mushroom consumption values at the 99th percentile (58.4 g/day), showed attenuated but consistent associations compared to the primary analyses. The non-linear association between mushroom consumption and forward digit span performance remained statistically significant, while the association with reverse digit span showed a similar pattern but was marginally significant. Only 32 participants (1.0%) had mushroom consumption exceeding this threshold, and the attenuation in winsorized analyses suggests these high consumers may have contributed meaningfully to the observed associations.
Importantly, the wide variability in mushroom consumption observed in our cohort is aligns with national data. According to the National Health and Nutrition Survey in Japan,38 mushroom consumption among adults aged 40 years and older is highly variable; statistical reports show that the standard deviations (20.5–28.9 g/day) exceed the mean intake values (11.9–16.6 g/day),39 indicating a wide distribution with a subset of the population consuming significantly higher amounts than the average. This suggests that the presence of high consumers is a normal characteristic of the Japanese population rather than a data anomaly. Whether the observed attenuation in sensitivity analyses reflects a dose–response benefit at very high intake levels or is driven by extreme values requires further investigation in larger samples with adequate representation across the full range of mushroom consumption.
Our findings complement the earlier Ohsaki Cohort 2006 Study, which reported that frequent mushroom consumption (≥3 times per week) was associated with a significantly lower risk of incident dementia among Japanese adults aged 65 years and older.13 In contrast, the present NILS-LSA analysis—which includes middle-aged adults from age 40—extends this evidence to an earlier phase of cognitive aging. A key rationale for focusing on mid-term memory functions (digit span), rather than dementia incidence, is that identifying dietary associations at this stage is critical for developing early intervention and prevention strategies before the onset of clinical dementia. Furthermore, compared with the food-frequency questionnaire utilized in the Ohsaki study, the NILS-LSA employed 3-day dietary records, thereby reducing potential recall bias and enhancing the precision of dietary exposure assessment. Together, these findings suggest that mushroom consumption may support the entire continuum of cognitive aging, from maintaining early memory performance to preventing overt dementia.
The study benefits from several methodological strengths, including its longitudinal design, comprehensive covariate adjustment, and use of validated cognitive assessments. However, several limitations should be acknowledged in interpreting these findings. First and foremost, as this is an observational study, we cannot establish a definitive causal relationship between mushroom consumption and cognitive performance, nor can we verify the specific biological mechanisms mentioned above. Although this study employed a longitudinal design, the temporal association between changes in mushroom consumption and cognitive performance requires further investigation, as dietary habits may vary over the follow-up period. Second, mushroom consumption was assessed using 3-day dietary records at baseline. While dietary habits among middle-aged and older adults tend to remain relatively stable over time, these short-term records may not fully capture long-term dietary variability or changes related to fluctuations in health status and seasonal availability. It is well-recognized in nutritional epidemiology that such measurement error and within-person variability in dietary assessment can lead to regression dilution bias,40 which typically attenuates observed associations toward the null. Consequently, the correlations reported in this study might represent conservative estimates of the true long-term effects of mushroom consumption on cognitive performance. Third, the study population consisted primarily of middle-aged and older Japanese adults residing in Aichi Prefecture. While the relative homogeneity of this regional cohort helps minimize potential confounding from broad ethnic and cultural variations, it restricts the generalizability of our findings to other populations. The external validity of these results may be influenced by specific Japanese dietary patterns, characterized by significantly higher baseline intake of mushrooms and seafood compared to many Western populations. For example, mushroom consumption in Japan is reported to be nearly four times higher than that in the United States.5,6 Consequently, the non-linear dose–response relationship observed in this study—where benefits were more pronounced at moderate to high intake levels—might not be directly translatable to populations with lower baseline exposure or different nutritional backgrounds. Furthermore, potential ethnic differences in genetic factors, such as the frequency of APOE ε4 alleles,41,42 could also moderate the observed associations. Future research in more diverse ethnic and geographic cohorts is necessary to confirm whether these dietary benefits are consistent across different global populations. Fourth, the present study did not differentiate between individual mushroom species, such as shiitake, maitake, or lion's mane. Edible mushrooms are known to contain varying concentrations of bioactive compounds, including but not limited to ergothioneine, hericenones, and β-glucans.43 While total mushroom intake serves as a broad proxy for exposure to these neuroprotective agents, the lack of species-specific data limits our ability to identify which mushrooms provide the most significant cognitive benefits or to provide targeted dietary recommendations. Future research is warranted to elucidate the effects of specific mushroom species on memory trajectories. Finally, the sex-stratified analyses lacked sufficient statistical power to detect meaningful differences between males and females. While the observed effect sizes (β coefficients) were consistent with the total population, the increased standard errors in subgroups—especially among females—resulted in non-significant p-values. We did not perform post-hoc power calculations as they are increasingly recognized as an inappropriate method for interpreting observed findings.44,45 Instead, our results provide essential effect size estimates that can be utilized by future research to conduct formal prospective power calculations and determine the sample sizes required to confirm potential sex-specific associations.
Our findings suggest that regularly including mushrooms in the diet might help maintain the type of memory that is crucial for independent living as we age. The non-linear dose–response association indicates that moderate to higher consumption levels may be necessary to achieve meaningful cognitive benefits. Given that mushrooms are widely available, affordable, and generally well-tolerated foods, they may represent a practical intervention strategy for supporting cognitive health during aging.
Conclusions
This longitudinal study identifies a positive, nonlinear association between mushroom consumption and cognitive performance in middle-aged and older Japanese adults, suggesting that moderate to high mushroom consumption may support the maintenance of short-term and working memory during aging. These findings highlight a potential dietary strategy for promoting cognitive health; however, they should be interpreted with caution. Further longitudinal research is needed to determine optimal consumption levels, examine potential sex-specific effects with adequately powered samples, and elucidate the neurobiological mechanisms underlying these associations. Furthermore, given the distinct bioactive profiles across various edible mushroom species, future investigations into species-specific effects are essential for developing refined, evidence-based dietary guidelines for the preservation of cognitive function.Author contributions
Shu Zhang: formal analysis, methodology, funding acquisition, writing – original draft. Djibril M. Ba: funding acquisition, writing – review & editing. Yukiko Nishita: investigation, data curation, writing – review & editing. Chikako Tange: investigation, data curation, writing – review & editing. Junjia Zhu: writing – review & editing. Rei Otsuka: conceptualization, methodology, investigation, funding acquisition, supervision, writing – review & editing. Joshua Muscat: funding acquisition, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets analyzed in the current study are not publicly available for privacy reasons, but are available from the corresponding author upon reasonable request. The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo04625d.Acknowledgements
This work was partially supported by the National Institutes of Health (NIH), U.S.A. (NIH Grant, grant number R21AG085006) and the National Center for Geriatrics and Gerontology, Japan (grant number 21-18, 24-10). The funding sources had no role in the study design, data collection, analysis, interpretation, writing of the report, or decision to submit the paper. We thank the staff of NILS-LSA project for their contributions and efforts in conducting the survey. Members of the National Institute for Longevity Sciences-Longitudinal Study of Aging (NILS-LSA) are listed in https://www.ncgg.go.jp/cgss/department/ep/index.html.References
- A. D. Baddeley, G. J. Hitch and R. J. Allen, From short-term store to multicomponent working memory: The role of the modal model, Mem. Cogn., 2019, 47, 575–588 CrossRef PubMed.
- K. L. Bopp and P. Verhaeghen, Aging and verbal memory span: A meta-analysis, J. Gerontol. B Psychol. Sci. Soc. Sci., 2005, 60, P223–P233 CrossRef PubMed.
- C. Pliatsikas, J. Veríssimo, L. Babcock, M. Y. Pullman, D. A. Glei, M. Weinstein, N. Goldman and M. T. Ullman, Working memory in older adults declines with age, but is modulated by sex and education, Q. J. Exp. Psychol., 2019, 72, 1308–1327 CrossRef PubMed.
- D. C. Park, G. Lautenschlager, T. Hedden, N. S. Davidson, A. D. Smith and P. K. Smith, Models of visuospatial and verbal memory across the adult life span, Psychol. Aging, 2002, 17, 299–320 Search PubMed.
- National Institutes of Biomedical Innovation, Annual trends in food group intake: Mushrooms (mean and standard deviation) by sex and age group, https://www.nibn.go.jp/eiken/kenkounippon21/eiyouchousa/keinen_henka_syokuhin.html, (accessed 20 January, 2026).
- SR Department, Per capita consumption of fresh mushrooms in the United States from 2010 to 2024 (in pounds), https://www.statista.com/statistics/257314/per-capita-consumption-of-fresh-mushrooms-in-the-us/?srsltid=AfmBOorU9Cy4-ORLavZ0haXVLqObiFXnRyH8H2ZlPzu7d_hRuIefqJIR, (accessed 20 January, 2026).
- I. C. Li, L. Y. Lee, T. T. Tzeng, W. P. Chen, Y. P. Chen, Y. J. Shiao and C. C. Chen, Neurohealth properties of Hericium erinaceus mycelia enriched with erinacines, Behav. Neurol., 2018, 2018, 5802634 Search PubMed.
- M. D. Kalaras, J. P. Richie, A. Calcagnotto and R. B. Beelman, Mushrooms: A rich source of the antioxidants ergothioneine and glutathione, Food Chem., 2017, 233, 429–433 CrossRef CAS PubMed.
- M. Hu, P. Zhang, R. Wang, M. Zhou, N. Pang, X. Cui, X. Ge, X. Liu, X. F. Huang and Y. Yu, Three different types of β-glucans enhance cognition: The role of the gut-brain axis, Front. Nutr., 2022, 9, 848930 CrossRef PubMed.
- K. Mori, S. Inatomi, K. Ouchi, Y. Azumi and T. Tuchida, Improving effects of the mushroom Yamabushitake (Hericium erinaceus) on mild cognitive impairment: A double-blind placebo-controlled clinical trial, Phytother. Res., 2009, 23, 367–372 CrossRef CAS PubMed.
- L. Feng, I. K. M. Cheah, M. M. X. Ng, J. Li, S. M. Chan, S. L. Lim, R. Mahendran, E. H. Kua and B. Halliwell, The association between mushroom consumption and mild cognitive impairment: A community-based cross-sectional study in Singapore, J. Alzheimer's Dis., 2019, 68, 197–203 CAS.
- S. Cha, L. Bell and C. M. Williams, The relationship between mushroom intake and cognitive performance: An epidemiological study in the European investigation of cancer-Norfolk cohort (EPIC-Norfolk), Nutrients, 2024, 16, 353 CrossRef PubMed.
- S. Zhang, Y. Tomata, K. Sugiyama, Y. Sugawara and I. Tsuji, Mushroom consumption and incident dementia in elderly Japanese: The Ohsaki cohort 2006 study, J. Am. Geriatr. Soc., 2017, 65, 1462–1469 CrossRef PubMed.
- D. Wechsler, The measurement of adult intelligence, J. Nerv. Ment. Dis., 1940, 91, 548 CrossRef.
- K. Fujita, H. Maekawa, H. Dairoku and K. Yamanaka, in Japanese Wechsler Adult Intelligence Scale, Nihon Bunka Kagakusha, Tokyo, 2006 Search PubMed.
- N. Seraji-Bzorgzad, H. Paulson and J. Heidebrink, Neurologic examination in the elderly, Handb. Clin. Neurol., 2019, 167, 73–88 Search PubMed.
- C. M. Cullum, in Comprehensive Clinical Psychology, ed. A. S. Bellack and M. Hersen, Elsevier, Oxford, 1998, pp. 303–347. DOI:10.1016/B0080-4270(73)00227-3.
- H. Shimokata, F. Ando and N. Niino, A new comprehensive study on aging—The National Institute for Longevity Sciences, Longitudinal Study of Aging (NILS-LSA), J. Epidemiol., 2000, 10(Supplement), S1–S9 CAS.
- R. Kozakai, F. Ando, H. Y. Kim, T. Rantanen and H. Shimokata, Regular exercise history as a predictor of exercise in community-dwelling older Japanese people, J. Phys. Fitness Sports Med., 2012, 1, 167–174 CrossRef.
- S. Shima, New self-rating scale for depression, Seisin Igaku, 1985, 27, 717–723 Search PubMed.
- L. S. Radloff, The CES-D scale: A self-report depression scale for research in the general population, Appl. Psychol. Meas., 1977, 1, 385–401 CrossRef.
- T. Imai, S. Sakai, K. Mori, F. Ando, N. Niino and H. Shimokata, Nutritional assessments of 3-day dietary records in National Institute for Longevity Sciences–Longitudinal Study of Aging (NILS-LSA), J. Epidemiol., 2000, 10(Supplement), S70–S76 CrossRef CAS PubMed.
- Ministry of Education, Science and Technology, The Council for Science and Technology, culture, sports, science and technology, standard tables of food composition in Japan, Report of the Subdivision on Resources, 2010 Search PubMed , https://www.mext.go.jp/b_menu/shingi/gijyutu/gijyutu3/houkoku/1298713.htm (accessed 15 June, 2018).
- H. Shimokata, Y. Yamada, M. Nakagawa, R. Okubo, T. Saido, A. Funakoshi, K. Miyasaka, S. Ohta, G. Tsujimoto, M. Tanaka, F. Ando and N. Niino, Distribution of geriatric disease-related genotypes in the National Institute for Longevity Sciences, Longitudinal Study of Aging (NILS-LSA), J. Epidemiol., 2000, 10(Supplement), S46–S55 CrossRef CAS PubMed.
- R. Otsuka, S. Zhang, R. Kozakai, C. Tange, S. Kubota, K. Furuya, F. Ando, H. Shimokata, Y. Nishita and H. Arai, Skin tactile perception is associated with longitudinal gait performance in middle-aged and older Japanese community dwellers, J. Frailty Aging, 2025, 14, 100006 CrossRef PubMed.
- S. Puri, M. Shaheen and B. Grover, Nutrition and cognitive health: A life course approach, Front. Public Health, 2023, 11, 1023907 CrossRef PubMed.
- S. Araki, Education and multidimensional inequalities in contemporary Japan and beyond: A call for longitudinal and comparative studies, Sociol. Compass, 2025, 19, e70072 CrossRef.
- L. Desquilbet and F. Mariotti, Dose-response analyses using restricted cubic spline functions in public health research, Stat. Med., 2010, 29, 1037–1057 CrossRef PubMed.
- R. Y. Liu and E. Liu, Longitudinal association of coffee and tea consumption with bone mineral density in older women: A 10-year repeated-measures analysis in the study of osteoporotic fractures, Nutrients, 2025, 17, 3660 CrossRef PubMed.
- A. Bechthold, H. Boeing, C. Schwedhelm, G. Hoffmann, S. Knüppel, K. Iqbal, S. De Henauw, N. Michels, B. Devleesschauwer, S. Schlesinger and L. Schwingshackl, Food groups and risk of coronary heart disease, stroke and heart failure: A systematic review and dose-response meta-analysis of prospective studies, Crit. Rev. Food Sci. Nutr., 2019, 59, 1071–1090 CrossRef CAS PubMed.
- R. M. Y. Tang, I. K.-M. Cheah, T. S. K. Yew and B. Halliwell, Distribution and accumulation of dietary ergothioneine and its metabolites in mouse tissues, Sci. Rep., 2018, 8, 1601 CrossRef PubMed.
- N. Nakamichi, T. Taguchi, H. Hosotani, T. Wakayama, T. Shimizu, T. Sugiura, S. Iseki and Y. Kato, Functional expression of carnitine/organic cation transporter OCTN1 in mouse brain neurons: Possible involvement in neuronal differentiation, Neurochem. Int., 2012, 61, 1121–1132 CrossRef CAS PubMed.
- T. Ishimoto and Y. Kato, Ergothioneine in the brain, FEBS Lett., 2022, 596, 1290–1298 CrossRef CAS PubMed.
- N. Watanabe, Effect of ergothioneine on the cognitive function improvement in healthy volunteers and mild cognitive impairment subjects–A randomized, double–blind, parallel–group comparison study, Jpn. Pharmacol. Ther., 2020, 48, 685 CAS.
- M. L. Furey, P. Pietrini and J. V. Haxby, Cholinergic enhancement and increased selectivity of perceptual processing during working memory, Science, 2000, 290, 2315–2319 CrossRef CAS PubMed.
- P. L. Croxson, D. A. Kyriazis and M. G. Baxter, Cholinergic modulation of a specific memory function of prefrontal cortex, Nat. Neurosci., 2011, 14, 1510–1512 CrossRef CAS PubMed.
- Y. Hayashi, M. Yoshida, M. Yamato, T. Ide, Z. Wu, M. Ochi-Shindou, T. Kanki, D. Kang, K. Sunagawa, H. Tsutsui and H. Nakanishi, Reverse of age-dependent memory impairment and mitochondrial DNA damage in microglia by an overexpression of human mitochondrial transcription factor a in mice, J. Neurosci., 2008, 28, 8624–8634 CrossRef CAS PubMed.
- C. Okada and H. Takimoto, The national health and nutrition survey in Japan: 75 years of history, Eiyougakuzashi, 2020, 78(Supplement), S5–S15 CrossRef.
- Ministry of Health and Welfare Labour, The National Health and Nutrition Survey inJapan, 2023, Ministry of Health, Labour and Welfare, Japan, 2025 Search PubMed.
- J. A. Hutcheon, A. Chiolero and J. A. Hanley, Random measurement error and regression dilution bias, Br. Med. J., 2010, 340, c2289 CrossRef PubMed.
- J. Perales-Puchalt, C. O. Mansel and O. J. Veatch, Variation in APOE ε4 prevalence and brain health associations by European descent in white all of us participants, medRxiv, 2025, 7 DOI:10.1101/2025.07.16.25331679.
- A. Nishimura, H. Nonomura, S. Tanaka, M. Yoshida, Y. Maruyama, Y. Aritomi, A. M. Saunders, D. K. Burns, M. W. Lutz, G. Runyan, E. Lai, K. Budur and A. D. Roses, Characterization of APOE and TOMM40 allele frequencies in the Japanese population, Alzheimers Dement, 2017, 3, 524–530 Search PubMed.
- A. Singh, R. K. Saini, A. Kumar, P. Chawla and R. Kaushik, Mushrooms as nutritional powerhouses: A review of their bioactive compounds, health benefits, and value-added products, Foods, 2025, 14, 741 CrossRef CAS PubMed.
- A. Gelman, Don't calculate post-hoc power using observed estimate of effect size, Ann. Surg., 2019, 269, e9–e10 CrossRef PubMed.
- M. G. Heckman, J. M. Davis and C. S. Crowson, Post hoc power calculations: An inappropriate method for interpreting the findings of a research study, J. Rheumatol., 2022, 49, 867–870 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |