Pro-inflammatory and pro-oxidant diets and CKD risk across cardiovascular–kidney–metabolic syndrome stages: a multi-omics mediation analysis
Yiwei
Zhang
,
Yu
Huang
,
Xiaoqin
Gan
,
Sisi
Yang
,
Yanjun
Zhang
,
Yiting
Wu
,
Ziliang
Ye
,
Xianglian
Cai
,
Dan
Chen
,
Xiaolong
Liang
,
Xianhui
Qin
 * and
Yuanyuan
Zhang
*
* and
Yuanyuan
Zhang
*
Division of Nephrology, Nanfang Hospital, Southern Medical University; National Clinical Research Center for Kidney Disease; State Key Laboratory of Multi-organ Injury Prevention and Treatment; Guangdong Provincial Institute of Nephrology; Guangdong Provincial Key Laboratory of Renal Failure Research, Guangzhou 510515, China. E-mail: pharmaqin@126.com; doctoryyzhang@126.com; Fax: +86-20-87281713; Tel: +86-20-61641591
First published on 27th November 2025
Abstract
Background & Purpose: While inflammation and oxidative stress contribute to renal injury, evidence linking dietary inflammatory/antioxidant potential to chronic kidney disease (CKD) incidence remains limited. We investigated associations of the Energy-adjusted Dietary Inflammatory Index (E-DII) and the Composite Dietary Antioxidant Index (CDAI) with CKD risk across Cardiovascular–Kidney–Metabolic (CKM) stages, and elucidated underlying mechanisms through multi-omics profiling. Methods: in this prospective analysis of 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 493 UK Biobank participants, dietary indices were derived from 24 hour recalls. Incident CKD was ascertained through health records. Cox models assessed associations, stratified by CKM stages (0–4), with mediation analyses incorporating proteomic (n = 18
493 UK Biobank participants, dietary indices were derived from 24 hour recalls. Incident CKD was ascertained through health records. Cox models assessed associations, stratified by CKM stages (0–4), with mediation analyses incorporating proteomic (n = 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 836) and metabolomic (n = 93
836) and metabolomic (n = 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 416) profiles. All analyses were adjusted for sociodemographic, lifestyle, and clinical factors, and genetic risk scores. Results: during a median 13.2 year follow-up (5799 incident CKD cases), both anti-inflammatory (E-DII < 0; adjusted HR, 0.88, 95% CI: 0.83–0.93) and antioxidant (CDAI ≥ 0; adjusted HR, 0.91; 95% CI: 0.86–0.97) diets were found to exhibit protective effects against CKD versus pro-inflammatory and pro-oxidant diets. Effects were most pronounced in early CKM stages (stages 0–2 vs. 3–4; both P-interactions < 0.05). Combined anti-inflammatory and antioxidant diets conferred maximal protection (adjusted HR, 0.85, 95% CI: 0.80–0.91), corresponding to a 2.5 year delay in CKD onset. Mechanistically, proteomic (56.9%) and metabolomic (60.6%) signatures mediated the E-DII-CKD association, predominantly through the degree of unsaturation (38.7%), FSTL3 (34.8%), and SPON2 (23.2%). Conclusions: pro-inflammatory and pro-oxidant diets synergistically increase CKD risk, particularly in the early CKM stages, while anti-inflammatory/antioxidant diets confer protection via multi-omics pathways linked to lipid metabolism and the extracellular matrix. These findings advocate for CKM stage-specific dietary interventions and integration of multi-omics biomarkers into CKD prevention.
416) profiles. All analyses were adjusted for sociodemographic, lifestyle, and clinical factors, and genetic risk scores. Results: during a median 13.2 year follow-up (5799 incident CKD cases), both anti-inflammatory (E-DII < 0; adjusted HR, 0.88, 95% CI: 0.83–0.93) and antioxidant (CDAI ≥ 0; adjusted HR, 0.91; 95% CI: 0.86–0.97) diets were found to exhibit protective effects against CKD versus pro-inflammatory and pro-oxidant diets. Effects were most pronounced in early CKM stages (stages 0–2 vs. 3–4; both P-interactions < 0.05). Combined anti-inflammatory and antioxidant diets conferred maximal protection (adjusted HR, 0.85, 95% CI: 0.80–0.91), corresponding to a 2.5 year delay in CKD onset. Mechanistically, proteomic (56.9%) and metabolomic (60.6%) signatures mediated the E-DII-CKD association, predominantly through the degree of unsaturation (38.7%), FSTL3 (34.8%), and SPON2 (23.2%). Conclusions: pro-inflammatory and pro-oxidant diets synergistically increase CKD risk, particularly in the early CKM stages, while anti-inflammatory/antioxidant diets confer protection via multi-omics pathways linked to lipid metabolism and the extracellular matrix. These findings advocate for CKM stage-specific dietary interventions and integration of multi-omics biomarkers into CKD prevention.
Introduction
Chronic kidney disease (CKD) represents a significant global health challenge, contributing substantially to end-stage renal disease, cardiovascular morbidity, and premature mortality.1,2 Identifying modifiable dietary risk factors has thus become a crucial strategy for CKD prevention and management.The pathogenesis of CKD involves two key interrelated pathways: chronic inflammation and oxidative stress.3 Emerging evidence suggests that dietary patterns may significantly influence these pathways, with anti-inflammatory diets demonstrating renal protective effects4,5 and pro-inflammatory diets exacerbating renal injury.6 The Dietary Inflammatory Index (DII), a validated tool for assessing diet-associated inflammation,7–9 has shown associations with renal function decline in elderly women (aged >70 years).10 However, this study has several limitations, including its restriction to older females, the absence of a standardized CKD definition, and inadequate adjustment for key confounders such as hypertension, dyslipidemia, estimated glomerular filtration rate (eGFR), urine albumin to creatinine ratio (UACR), and genetic predisposition.
Oxidative stress similarly plays a fundamental role in CKD incidence.11 The Composite Dietary Antioxidant Index (CDAI), which quantifies total antioxidant intake,12–14 has demonstrated an inverse association with CKD prevalence in cross-sectional studies.15 Nevertheless, longitudinal data and mechanistic insights remain notably lacking.
The Cardiovascular–Kidney–Metabolic (CKM) syndrome framework16 provides a clinically relevant approach for CKD risk stratification, integrating metabolic dysfunction, kidney injury, and cardiovascular outcomes. This model enables targeted evaluation of dietary influences across disease stages.
We hypothesize that pro-inflammatory and pro-oxidant diets increase CKD risk through three principal mechanisms: (1) obesity-mediated metabolic dysregulation;17,18 (2) sustained low-grade systemic inflammation;19–21 and (3) alterations in proteomic and metabolomic signatures associated with pro-inflammatory and pro-oxidant diets.
To address these research gaps, we will utilize UK Biobank data to: (1) examine both independent and joint associations between Energy-adjusted DII (E-DII) and CDAI with incident CKD; (2) evaluate stage-specific effects across the CKM continuum; (3) identify multi-omics signatures for E-DII and CDAI; and (4) elucidate the mediating roles of obesity metrics (e.g., BMI [body mass index]), systemic inflammatory markers (e.g., CRP [C-reactive protein]), and multi-omics profiles.
Methods
Data source and study population
The UK Biobank is a population-based prospective cohort comprising 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 UK residents aged 37–73 years recruited between 2006 and 2010.22,23 The study was approved by the North West Research Ethics Committee (Ref: 11/NW/0382) and all participants provided written informed consent. At baseline, participants completed detailed health and lifestyle questionnaires, underwent standardized physical measurements, and provided biological samples for biomarker analysis.
000 UK residents aged 37–73 years recruited between 2006 and 2010.22,23 The study was approved by the North West Research Ethics Committee (Ref: 11/NW/0382) and all participants provided written informed consent. At baseline, participants completed detailed health and lifestyle questionnaires, underwent standardized physical measurements, and provided biological samples for biomarker analysis.
From 210![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 883 participants with at least one complete 24-hour dietary recall data, we excluded those with: (1) baseline CKD (eGFR < 60 mL min−1 per 1.73 m2, UACR ≥ 30 mg g−1, or physician-diagnosed CKD [n = 29
883 participants with at least one complete 24-hour dietary recall data, we excluded those with: (1) baseline CKD (eGFR < 60 mL min−1 per 1.73 m2, UACR ≥ 30 mg g−1, or physician-diagnosed CKD [n = 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 662]); and (2) implausible energy intake [n = 1
662]); and (2) implausible energy intake [n = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 728], yielding 179
728], yielding 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 493 participants for primary analysis. Multi-omics analyses included subsets with complete metabolomic (n = 93
493 participants for primary analysis. Multi-omics analyses included subsets with complete metabolomic (n = 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 416) and proteomic (n = 18
416) and proteomic (n = 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 836) data (Fig. S1).
836) data (Fig. S1).
Ascertainment of E-DII and CDAI
In the UK Biobank, dietary data were collected via the validated Oxford WebQ (online 24 hour recall system administered up to five times between 2009 and 2012 to account for seasonal variation).1 The reliability and validity of this questionnaire have been previously described.24The E-DII was derived from 29 dietary components, each weighted (range: ±1; positive values indicating pro-inflammatory effects, negative values indicating anti-inflammatory effects) based on established inflammatory associations with six biomarkers (IL-1β, IL-4, IL-6, IL-10, TNF-α [Tumour necrosis factor-alpha], and CRP) (Table S1).7,8 Global reference data enabled energy-adjusted Z-score standardization (per 1000 kcal day−1) and weighted summation. The CDAI was calculated by standardizing intake deviations of six micronutrients (selenium, zinc, vitamins A/C/E, and carotenoids) relative to means.14 Both indices were dichotomized at zero, with E-DII < 0/CDAI ≥ 0 representing anti-inflammatory/antioxidant diets and E-DII ≥ 0/CDAI < 0 indicating pro-inflammatory/pro-oxidant diets.
Plasma multi-omics biomarkers profiling
Among the 2923 proteins, 12 with a missing rate >20% were excluded, resulting in 2911 proteins for analysis. For the remaining proteins, missing values were imputed using the mean.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 UK Biobank participants using a targeted NMR-based platform (Nightingale Health Ltd).27 Samples were randomly selected from the full cohort. The platform simultaneously quantified 249 metabolic measures, including 168 absolute metabolites (e.g., fatty acids, amino acids, lipids, and lipoproteins) and 81 derived ratios. All procedures followed standardized operating protocols, as described in prior publications.28
000 UK Biobank participants using a targeted NMR-based platform (Nightingale Health Ltd).27 Samples were randomly selected from the full cohort. The platform simultaneously quantified 249 metabolic measures, including 168 absolute metabolites (e.g., fatty acids, amino acids, lipids, and lipoproteins) and 81 derived ratios. All procedures followed standardized operating protocols, as described in prior publications.28
In this study, the 168 absolute metabolite concentrations were used. Data were natural-log-transformed (ln[x + 1]) and Z-standardized to improve normality and remove unit effects.29
Ascertainment of covariates and mediators
Covariates included age, sex, race, education, employment, Townsend Deprivation Index (TDI), smoking status, alcohol consumption status, optimal physical activity, history of diseases (including hypertension, diabetes, dyslipidemia, cardiovascular disease [CVD], and inflammatory diseases), total energy intake, eGFR, UACR, and genetic risk score (GRS) of CKD.30,31Potential mediators comprised obesity metrics (BMI and WC [waist circumference]) and inflammatory markers (CRP and INFLA [low-grade inflammation] score), with the latter incorporating four systemic inflammation components (CRP, WBC [white blood cell], PLT [platelet], and NLR [neutrophil-to-lymphocyte ratio]). The INFLA-score was calculated by assigning values from +1 to +4 (highest deciles, 7th to 10th), −4 to −1 (lowest deciles, 1st to 4th), and 0 (middle deciles, 5th to 6th), yielding a total range of −16 to +16 where higher scores indicate greater low-grade inflammation.32 Variable definitions are provided in the SI.
Study outcomes
The study outcome was incident CKD, identified using ICD-10 code N18 from linked primary care, hospital admissions, and mortality records through validated algorithms. The follow-up duration spanned from enrollment until the earliest of: CKD diagnosis, death, loss to follow-up, or end of follow-up.Ascertainment of genetic predisposition of CKD
Detailed information about genotyping, imputation and quality control in the UK Biobank study has been provided previously.33 We calculated a weighted genetic risk score (GRS) for eGFR using 263 established SNPs,34 where higher scores indicated lower CKD risk. Participants were stratified into tertiles of genetic predisposition: high (tertile 1), medium (tertile 2), or low (tertile 3) risk.CKM syndrome staging
Participants were classified into CKM syndrome stages: stage 0 (absence of CKM risk factors), stage 1 (excess or dysfunctional adiposity), stage 2 (metabolic risk factors/moderate-severe CKD), stage 3 (high-risk CKD/CVD risk), and stage 4 (established CVD).35 All baseline CKD cases were excluded from analysis. Staging criteria were adapted for UK Biobank data (SI).Statistical analysis
Baseline characteristics were summarized as means (standard deviations [SDs]) for continuous variables and counts (%) for categorical variables, stratified by E-DII or CDAI categories. Differences across categories were assessed using ANOVA for continuous variables and chi-square tests for categorical variables.Missing covariate data were imputed using the mode for categorical variables and the mean for continuous variables when the missing rate was <1%.36 For variables with ≥1% missing data, a separate ‘unknown/missing’ category was created (Table S2). Potential mediators were excluded from adjusted models to avoid overadjustment.37 Laplace regression was used to estimate the percentile differences in time (years) to CKD onset as a function of dietary status.
Five sensitivity analyses were performed: (1) stratified analyses with likelihood ratio tests to evaluate potential effect modification; (2) replication of analyses using the non-energy-adjusted DII; (3) exclusion of participants with <5 years of follow-up to address reverse causation; (4) additional adjustment for vitamin/mineral supplements; and (5) complete-case analysis excluding individuals with any missing covariates.
All statistical tests were two-tailed with a significance threshold of P < 0.05. For multi-omics analyses, Bonferroni correction was applied during biomarker identification, and FDR correction was used in subsequent mediation analyses. All analyses were conducted using R software (version 4.1.1; R Foundation for Statistical Computing).
Results
Baseline characteristics of study participants
The participants had a mean age of 55.9 ± 7.9 years, were predominantly White (96.0%), and included 45.4% males. The average number of completed dietary recalls per participant was 2.2, with the detailed distribution presented in Fig. S2. The median (Interquartile Range, IQR) scores were −0.54 (−1.81–0.67) for E-DII and −0.41 (−2.49–1.98) for CDAI.Participants with anti-inflammatory diets (vs. pro-inflammatory diets) tended to be older, female, non-smokers, and more physically active. They also had higher UACR, education levels, and dyslipidemia prevalence, but lower eGFR, TDI, inflammatory disease prevalence, and energy intake (Table 1). Those with antioxidant diets (vs. pro-oxidant diets) were older, more often male, non-smokers, White, and more physically active. They also exhibited higher education levels, hypertension prevalence, energy intake, UACR, and eGFR, along with lower TDI (Table 1).
| Total | Dietary inflammatory potential (E-DII) | Dietary oxidant potential (CDAI) | |||||
|---|---|---|---|---|---|---|---|
| Pro-inflammatory | Anti-inflammatory | P value | Pro-oxidant | Antioxidant | P value | ||
| a Stratified by E-DII [anti-inflammatory (E-DII < 0) vs. pro-inflammatory] and CDAI [antioxidant (CDAI ≥ 0) vs. pro-oxidant]. Variables are presented as mean (SD) or n (%). Abbreviations: CDAI, composite dietary antioxidant index; E-DII, energy-adjusted dietary inflammatory index. | |||||||
| N | 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 493 493 |
69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116 116 |
110![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 377 377 |
98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 439 439 |
81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 054 054 |
||
| Age, years | 55.9 (7.9) | 54.8 (8.1) | 56.7 (7.7) | <0.001 | 55.6 (7.9) | 56.3 (7.9) | <0.001 |
| Male, N (%) | 81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 502 (45.4) 502 (45.4) |
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 419 (54.1) 419 (54.1) |
44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 083 (39.9) 083 (39.9) |
<0.001 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 632 (43.3) 632 (43.3) |
38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 870 (48.0) 870 (48.0) |
<0.001 |
| White, N (%) | 171![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 777 (96.0) 777 (96.0) |
65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 725 (95.5) 725 (95.5) |
106![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 052 (96.4) 052 (96.4) |
<0.001 | 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 881 (95.7) 881 (95.7) |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 896 (96.4) 896 (96.4) |
<0.001 |
| Townsend deprivation index | −1.61 (2.9) | −1.4 (3.0) | −1.7 (2.8) | <0.001 | −1.5 (2.9) | −1.7 (2.8) | <0.001 |
| College or university degree, N (%) | 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 633 (53.5) 633 (53.5) |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 912 (50.8) 912 (50.8) |
60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 721 (55.3) 721 (55.3) |
<0.001 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 221 (51.3) 221 (51.3) |
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 412 (56.2) 412 (56.2) |
<0.001 |
| Employment, N (%) | 168![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 605 (94.6) 605 (94.6) |
64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 471 (94.0) 471 (94.0) |
104![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 134 (95.1) 134 (95.1) |
<0.001 | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 331 (94.5) 331 (94.5) |
76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 274 (94.8) 274 (94.8) |
0.006 |
| Optimal physical activity, N (%) | 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 005 (56.7) 005 (56.7) |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 270 (53.2) 270 (53.2) |
62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 735 (58.9) 735 (58.9) |
<0.001 | 51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 048 (54.1) 048 (54.1) |
46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 957 (59.8) 957 (59.8) |
<0.001 |
| Smoking status, N (%) | <0.001 | <0.001 | |||||
| Never | 101![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 842 (56.9) 842 (56.9) |
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 443 (54.3) 443 (54.3) |
64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 399 (58.5) 399 (58.5) |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 206 (56.2) 206 (56.2) |
46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 636 (57.7) 636 (57.7) |
||
| Former | 63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 270 (35.3) 270 (35.3) |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 010 (34.8) 010 (34.8) |
39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 260 (35.7) 260 (35.7) |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 518 (35.2) 518 (35.2) |
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 752 (35.5) 752 (35.5) |
||
| Current | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 937 (7.8) 937 (7.8) |
7481 (10.9) | 6456 (5.9) | 8435 (8.6) | 5502 (6.8) | ||
| Alcohol consumption status, N (%) | <0.001 | <0.001 | |||||
| <1 time per week | 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 681 (26.6) 681 (26.6) |
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 901 (27.4) 901 (27.4) |
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 780 (26.1) 780 (26.1) |
26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 637 (27.1) 637 (27.1) |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 044 (26.0) 044 (26.0) |
||
| 1–2 times per week | 44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 807 (25.0) 807 (25.0) |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 555 (24.0) 555 (24.0) |
28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 252 (25.6) 252 (25.6) |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 538 (24.9) 538 (24.9) |
20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 269 (25.0) 269 (25.0) |
||
| 3–4 times per week | 45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 626 (25.4) 626 (25.4) |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 (23.9) 500 (23.9) |
29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 126 (26.4) 126 (26.4) |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 451 (24.9) 451 (24.9) |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 175 (26.1) 175 (26.1) |
||
| >4 times per week | 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 258 (23.0) 258 (23.0) |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 093 (24.8) 093 (24.8) |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 165 (21.9) 165 (21.9) |
22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 735 (23.1) 735 (23.1) |
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 523 (22.9) 523 (22.9) |
||
| Energy, kcal | 2052.7 (555.3) | 2167.0 (593.5) | 1981.1 (517.3) | <0.001 | 1800.8 (436.1) | 2358.6 (530.3) | <0.001 |
| Hypertension, N (%) | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 573 (50.6) 573 (50.6) |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 742 (50.4) 742 (50.4) |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 831 (50.7) 831 (50.7) |
0.254 | 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 154 (50.1) 154 (50.1) |
41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 419 (51.3) 419 (51.3) |
<0.001 |
| Diabetes, N (%) | 7507 (4.2) | 2870 (4.2) | 4637 (4.2) | 0.625 | 4127 (4.2) | 3380 (4.2) | 0.823 |
| Dyslipidemia, N (%) | 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 671 (15.4) 671 (15.4) |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 243 (14.8) 243 (14.8) |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 428 (15.8) 428 (15.8) |
<0.001 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 229 (15.5) 229 (15.5) |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 442 (15.4) 442 (15.4) |
0.487 |
| Cardiovascular disease, N (%) | 8828 (4.9) | 3327 (4.8) | 5501 (5.0) | 0.108 | 4867 (5.0) | 3961 (4.9) | 0.579 |
| Inflammatory diseases, N (%) | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 055 (14.0) 055 (14.0) |
9919 (14.4) | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 136 (13.7) 136 (13.7) |
<0.001 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 656 (13.9) 656 (13.9) |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 399 (14.1) 399 (14.1) |
0.254 |
| Estimated glomerular filtration rate, ml min−1 per 1.73 m2 | 95.7 (11.6) | 96.1 (11.8) | 95.4 (11.5) | <0.001 | 95.6 (11.7) | 95.8 (11.5) | <0.001 |
| Urine albumin to creatinine ratio, mg g−1 | 9.1 (5.7) | 8.3 (5.4) | 9.6 (5.8) | <0.001 | 9.0 (5.7) | 9.2 (5.7) | <0.001 |
The proteomic and metabolomic subcohorts demonstrated characteristics closely aligned with the full analytical sample (Table S3).
Association of E-DII and CDAI with the incident CKD risk
During a median 13.2 year follow-up in the UK Biobank study, 5799 incident CKD cases (3.2%) were identified. Higher E-DII scores (greater pro-inflammatory potential) showed a positive association with CKD incidence (per SD increment; adjusted HR, 1.09, 95% CI: 1.06–1.12), with anti-inflammatory diets (E-DII ≤0) exhibiting lower CKD risk (adjusted HR, 0.88; 95% CI: 0.83–0.93) compared to pro-inflammatory diets (E-DII >0). Similarly, higher CADI scores (greater antioxidant potential) demonstrated an inverse association with CKD risk (per SD increment; adjusted HR, 0.94; 95% CI: 0.91–0.98), and antioxidant diets (CADI >0) were associated with reduced CKD risk (adjusted HR, 0.91; 95% CI: 0.86–0.97) relative to pro-oxidant diets (CADI ≤0) (Table 2 and Fig. 1A, B).| N | Cases (%) | Model 1a | Model 2b | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |||
| a Model 1: adjusted for age, sex, race, education, employment, and Townsend deprivation index. b Model 2: adjusted for covariates in Model 1, plus smoking status, alcohol consumption status, optimal physical activity, hypertension, diabetes, dyslipidemia, cardiovascular disease, inflammatory diseases, total energy intake, estimated glomerular filtration rate, urine albumin to creatinine ratio, and genetic risk score of chronic kidney disease. Abbreviations: CDAI, composite dietary antioxidant index; E-DII, energy-adjusted dietary inflammatory index. | ||||||
| Dietary inflammatory potential (E-DII) | ||||||
| Per SD increment | 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 493 493 |
5799 (3.2) | 1.12 (1.09, 1.15) | <0.001 | 1.09 (1.06, 1.12) | <0.001 |
| Categories | ||||||
| Pro-inflammatory (E-DII ≥ 0) | 69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116 116 |
2306 (3.3) | Ref | Ref | ||
| Anti-inflammatory (E-DII < 0) | 110![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 377 377 |
3493 (3.2) | 0.84 (0.79, 0.88) | <0.001 | 0.88 (0.83, 0.93) | <0.001 |
| Dietary oxidant potential (CDAI) | ||||||
| Per SD increment | 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 493 493 |
5799 (3.2) | 0.94 (0.91, 0.96) | <0.001 | 0.94 (0.91, 0.98) | 0.001 |
| Categories | ||||||
| Pro-oxidant (CDAI < 0) | 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 439 439 |
3287 (3.3) | Ref | Ref | ||
| Antioxidant (CDAI ≥ 0) | 81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 054 054 |
2512 (3.1) | 0.88 (0.83, 0.93) | <0.001 | 0.91 (0.86, 0.97) | 0.003 |
| Joint effects of dietary inflammatory and oxidant potential | ||||||
| Pro-inflammatory + pro-oxidant | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 109 109 |
1709 (3.4) | Ref | Ref | ||
| Pro-inflammatory + antioxidant | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 007 007 |
597 (3.1) | 0.92 (0.84, 1.01) | 0.072 | 0.93 (0.84, 1.04) | 0.198 |
| Anti-inflammatory + pro-oxidant | 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 330 330 |
1578 (3.3) | 0.86 (0.80, 0.92) | <0.001 | 0.88 (0.82, 0.94) | <0.001 |
| Anti-inflammatory + antioxidant | 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 047 047 |
1915 (3.1) | 0.79 (0.74, 0.84) | <0.001 | 0.85 (0.80, 0.91) | <0.001 |
In multivariable Laplace regression analysis, compared to pro-inflammatory/pro-oxidant dietary patterns, the anti-inflammatory with pro-oxidant pattern was associated with CKD onset being delayed by 1.7 years, while the combined anti-inflammatory and antioxidant pattern showed a 2.5 year delay in CKD incidence (Fig. 1D).
Multi-omics signatures for E-DII and CDAI
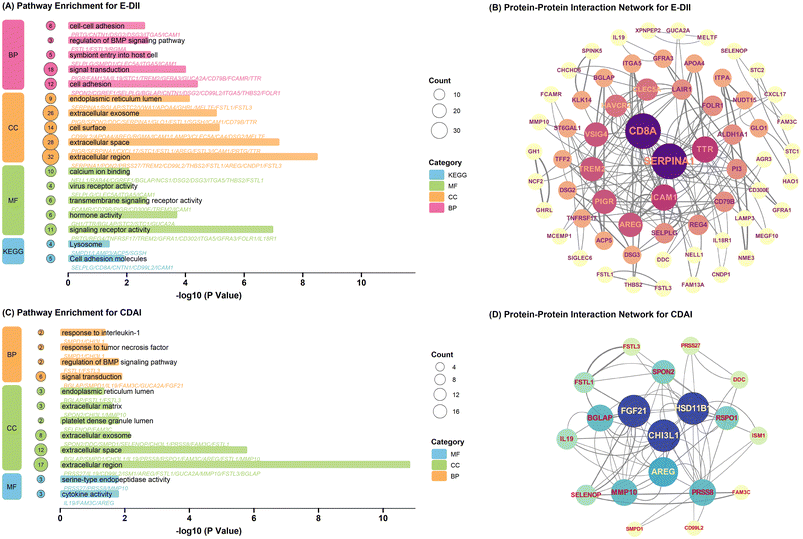
Our integrated multi-omics analysis revealed distinct signatures for E-DII (92 proteins/37 metabolites) and CDAI (23 proteins/15 metabolites) through multivariable linear and LASSO regression. The most significant E-DII-associated biomarkers included DDC (aromatic-L-amino-acid decarboxylase; β = −0.111), FSTL3 (follistatin-related protein 3; β = 0.093), and GUCA2A (guanylin; β = −0.091) for proteins, and degree of unsaturation (β = −0.187), average diameter for LDL particles (β = −0.054), and triglycerides in small HDL (β = 0.052) for metabolites. For CDAI, top associations were BGLAP (osteocalcin; β = −0.122), ISM1 (isthmin-1; β = −0.090), and FSTL3 (β = −0.088) among proteins, and omega-3 fatty acids (β = 0.157), valine (β = 0.069), and saturated fatty acids (β = −0.037) among metabolites, with FSTL3 emerging as a shared significant protein in both analyses (Tables S5–12).CDAI demonstrated enrichment in 12 pathways, with significant involvement in signal transduction and extracellular regions, indicating its impact on cellular communication and tissue structure (Fig. 2C and Table S14). PPI network analysis identified CHI3L1 as a central hub protein in the CDAI-associated protein interactions (Fig. 2D and Table S16).
Functional enrichment analysis identified shared and distinct biological pathways between E-DII and CDAI. Both indices showed significant enrichment in signal transduction and extracellular components, indicating common roles in cellular communication and tissue structure. Notably, they shared enrichment in the extracellular space and exosomes, suggesting involvement in cell interactions and protein transport. However, E-DII displayed unique enrichments in cell adhesion, T cell activation, plasma membrane components, and extracellular matrix, highlighting its broader role in immune modulation and cellular signaling (Fig. 2A and B).
Association of multi-omics signatures and incident CKD Risk
The proteomic (per SD increment; adjusted HR, 1.24, 95% CI: 1.15–1.35) and metabolomic (per SD increment; adjusted HR, 1.22, 95% CI: 1.17–1.26) signature scores for E-DII were significantly associated with an increased risk of incident CKD (Fig. S4A and B). In contrast, the proteomic (per SD increment; adjusted HR, 0.78, 95% CI: 0.72–0.84) and metabolomic (per SD increment; adjusted HR, 0.84, 95% CI: 0.81–0.87) signature scores for CDAI were significantly associated with a decreased risk of CKD (Fig. S4C and D).Mediation analyses
| Mediators | Proportion mediated (%) (95% CI) | P value (FDR-corrected) |
|---|---|---|
| Abbreviations: BMI, body mass index; CDAI, composite dietary antioxidant index; CRP, C-reactive protein; E-DII, energy-adjusted dietary inflammatory index; INFLA-score, low-grade inflammation score; WC, waist circumference. | ||
| Dietary inflammatory potential (E-DII) | ||
| Obesity markers | ||
| BMI | 10.25 (7.14, 15.66) | <0.001 |
| WC | 13.96 (9.82, 20.73) | <0.001 |
| Inflammatory biomarkers | ||
| CRP | 4.67 (3.27, 7.25) | <0.001 |
| INFLA-score | 5.93 (3.13, 9.97) | <0.001 |
| Dietary oxidant potential (CDAI) | ||
| Obesity markers | ||
| WC | 4.74 (2.29, 12.43) | <0.001 |
| Inflammatory biomarkers | ||
| CRP | 4.57 (3.21, 7.19) | <0.001 |
| INFLA-score | 5.20 (2.48, 16.44) | <0.001 |
Discussion
This comprehensive investigation provides strong evidence that both pro-inflammatory and pro-oxidant dietary patterns are independently and synergistically associated with increased CKD risk. Our findings advance current understanding in four key aspects: (1) establishing robust longitudinal associations between dietary inflammatory/oxidative potential and CKD incidence; (2) revealing stage-specific effects across the CKM syndrome continuum; (3) uncovering novel mechanistic pathways through integrated multi-omics profiling; and (4) identifying obesity-related metabolic dysregulation, systemic inflammation, and multi-omics signatures as key mediators. These findings have immediate clinical relevance for developing targeted nutritional interventions in CKD prevention and management.Dietary patterns and CKD risk
Our findings establish clear associations between dietary patterns and CKD risk, with anti-inflammatory diets showing a 12% risk reduction and antioxidant diets demonstrating a 9% protective effect compared to their pro-inflammatory/pro-oxidant counterparts. The most significant protection emerged from combined anti-inflammatory and antioxidant dietary patterns, which reduced CKD risk by 15% and delayed disease onset by 2.5 years – an effect size comparable to that of first-line pharmacological interventions.39 These results suggest complementary renal protective mechanisms: anti-inflammatory components may reduce glomerular injury by attenuating inflammation and endothelial dysfunction,40 while antioxidants likely protect tubules by neutralizing oxidative stress.41 These findings support incorporating dietary assessments into CKD risk stratification, emphasizing combined anti-inflammatory/antioxidant nutritional counseling and developing targeted dietary interventions that address both pathways.Stage-specific effects across the CKM spectrum
Our study reveals important temporal and pathophysiological patterns in dietary protection against CKD. The significantly stronger protective effects of anti-inflammatory and antioxidant diets in early CKM stages highlight two key insights: first, that nutritional interventions have their greatest impact during the initial phases of disease development; and second, that the window of maximal dietary responsiveness may close as cardiorenal dysfunction progresses. This stage-dependent efficacy was particularly pronounced for anti-inflammatory diets in participants with UACR ≥ 10 mg g−1, suggesting that early renal microvascular changes – marked by subclinical albuminuria – represent a critical period when inflammatory pathways are most amenable to dietary modulation.These results underscore the importance of implementing targeted dietary strategies based on individual CKM staging and albuminuria status, rather than a one-size-fits-all approach. The differential benefits observed across disease stages support a precision nutrition approach for CKD prevention, with optimal efficacy likely achieved through early intervention. Future studies should investigate whether dietary modifications can meaningfully alter the natural history of early CKM progression.
Mechanistic insights from multi-omics mediation
Our multi-omics analyses provide novel mechanistic insights into how dietary inflammatory and oxidative potential influence CKD risk. We identified distinct proteomic (92 proteins for E-DII, 23 for CDAI) and metabolomic (37 metabolites for E-DII, 15 for CDAI) signatures, with FSTL3 emerging as a key shared protein across both dietary patterns. Notably, E-DII-associated signatures exhibited robust associations with CKD risk, mediating over 50% of the total effect through proteomic (56.92%) and metabolomic (60.61%) pathways. It is important to note that these proportions, derived from separate models, do not represent simple additive effects but rather reflect overlapping biological pathways, as proteins and metabolites function within tightly integrated networks.42 The top mediators—degree of unsaturation (38.70%), FSTL3 (34.81%), and SPON2 (23.19%)—underscore the critical roles of lipid metabolism (reflected in fatty acid profiles) and extracellular matrix regulation (via FSTL3 and SPON2) in diet–renal disease interactions.43–45 Functional enrichment analyses further elucidated these mechanisms, revealing that E-DII-associated proteins were heavily involved in extracellular processes, with SERPINA1 (a key regulator of protease-antiprotease balance) acting as a central hub in protein–protein interaction networks. These findings suggest that pro-inflammatory diets may drive renal damage through dysregulated tissue remodeling and impaired extracellular matrix homeostasis.46Interestingly, while both E-DII and CDAI demonstrated phenotypic mediation through obesity (BMI, waist circumference) and systemic inflammation (CRP, INFLA-score), only E-DII exhibited significant multi-omics mediation. This distinction may explain the stronger association between pro-inflammatory diets and CKD risk compared to pro-oxidant diets. The lack of multi-omics mediation for CDAI could reflect either fundamental biological differences in how antioxidant nutrients modulate renal pathways (e.g., via direct redox regulation rather than proteomic/metabolomic remodeling) or methodological limitations in capturing subtle antioxidant effects or potential limitations in statistical power due to sample size constraints. The pathway analyses further support this dichotomy, with E-DII signatures predominantly enriched in immune-related processes, while CDAI signatures were more closely tied to metabolic regulation. These results refine our understanding of diet–CKD pathophysiology and nominate specific molecular targets (e.g., FSTL3, fatty acid composition) for future dietary interventions aimed at mitigating renal risk.
Clinical translation and implementation
The study has several immediate applications for clinical practice and public health: first, the E-DII and the CDAI provide practical tools for assessing dietary risks during routine CKD screening. Second, our CKM stage-specific results enable personalized dietary recommendations, potentially through decision-support systems integrated with electronic health records. Third, the identified biomarkers (particularly fatty acid profiles and FSTL3) could serve as objective monitoring tools in dietary intervention trials. From a public health perspective, these findings strengthen the evidence base for population-level strategies to promote anti-inflammatory/antioxidant diets, such as front-of-package labeling reform or institutional meal programs targeting high-risk groups.Limitations
Several limitations warrant consideration: residual confounding from unmeasured lifestyle factors, potential measurement error in dietary assessments, and limited ethnic diversity in the UK Biobank. Furthermore, both proteomic and metabolomic data were available only for subsets of the full cohort, which may affect the generalizability of our findings and reduce statistical power in the corresponding mediation analyses. Additionally, the mediation analysis, which examined proteomic and metabolomic signatures in separate models, may have led to an overestimation of their combined mediating effect due to inherent correlations between these omics layers. The observational nature precludes causal inferences, although the consistent mediation proportions and biological plausibility support potential causality.Conclusion
This study demonstrates that pro-inflammatory and pro-oxidant dietary patterns independently and synergistically increase CKD risk, whereas anti-inflammatory and antioxidant diets offer significant protection—particularly in early-stage CKM. Multi-omics analyses revealed key mechanistic pathways involving lipid metabolism, extracellular matrix regulation, and systemic inflammation, with FSTL3 and fatty acid profiles as critical mediators. These findings underscore the importance of implementing early dietary interventions and personalizing nutritional strategies according to disease stage for effective CKD prevention and management.Author contributions
Yiwei Zhang, Xianhui Qin, and Yuanyuan Zhang conceived and designed the study; Yiwei Zhang conducted the study; Yiwei Zhang and Yu Huang contributed to statistical analysis; Yiwei Zhang, Xianhui Qin, and Yuanyuan Zhang drafted the manuscript; Xianhui Qin and Yuanyuan Zhang provided critical supervision. All authors reviewed/edited the manuscript for important intellectual content and read and approved the final manuscript.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
The data are available at the UK Biobank (https://www.ukbiobank.ac.uk), and the analytical methods and study materials that support the findings of this study will be made available from the corresponding authors on request and can be accessed with a reasonable request.The Supplementary Information (SI) file for this article contains detailed supplementary methods, tables, and figures. See DOI: https://doi.org/10.1039/d5fo03952e.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (82570914, 81973133, 82030022 and 82330020), Key Technologies R&D Program of Guangdong Province (2023B1111030004), Guangdong Provincial Clinical Research Center for Kidney Disease (2020B1111170013), the Program of Introducing Talents of Discipline to Universities, 111 Plan (D18005), President Foundation of Nanfang Hospital, Southern Medical University (2024B029) and the Development and Reform Commission of Shenzhen Municipality (XMHT20220104055). This research has been conducted using the UK Biobank Resource under Application Number 73201. The authors would like to thank the UK Biobank participants.References
- B. Bikbov, C. A. Purcell, A. S. Levey, M. Smith, A. Abdoli and M. Abebe, et al., Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017, Lancet, 2020, 395(10225), 709–733 CrossRef PubMed.
- J. S. Lees, C. E. Welsh, C. A. Celis-Morales, D. Mackay, J. Lewsey, S. R. Gray, D. M. Lyall, J. G. Cleland, J. M. R. Gill, P. S. Jhund, J. Pell, N. Sattar, P. Welsh and P. B. Mark, Glomerular filtration rate by differing measures, albuminuria and prediction of cardiovascular disease, mortality and end-stage kidney disease, Nat. Med., 2019, 25(11), 1753–1760 CrossRef CAS PubMed.
- K. Kalantar-Zadeh, T. H. Jafar, D. Nitsch, B. L. Neuen and V. Perkovic, Chronic kidney disease, Lancet, 2021, 398(10302), 786–802 CrossRef CAS PubMed.
- M. Liu, Z. Ye, Q. Wu, S. Yang, Y. Zhang, C. Zhou, P. He, Y. Zhang, J. Nie, M. Liang, F. F. Hou and X. Qin, Folate intake and incident chronic kidney disease: A 30-year follow-up study from young adulthood to midlife, Am. J. Clin. Nutr., 2022, 116(2), 599–607 CrossRef CAS PubMed.
- C. Zhou, P. He, Z. Ye, Y. Zhang, Y. Zhang, S. Yang, Q. Wu, M. Liu, J. Nie and X. Qin, Erratum. Relationships of serum 25-hydroxyvitamin D concentrations, diabetes, genetic susceptibility, and new-onset chronic kidney disease, Diabetes Care, 2023, 46(8), 1566 CrossRef CAS PubMed.
- E. Lopez-Garcia, M. B. Schulze, T. T. Fung, J. B. Meigs, N. Rifai, J. E. Manson and F. B. Hu, Major dietary patterns are related to plasma concentrations of markers of inflammation and endothelial dysfunction, Am. J. Clin. Nutr., 2004, 80(4), 1029–1035 Search PubMed.
- N. Shivappa, S. E. Steck, T. G. Hurley, J. R. Hussey and J. R. Hébert, Designing and developing a literature-derived, population-based dietary inflammatory index, Public Health Nutr., 2014, 17(8), 1689–1696 Search PubMed.
- J. R. Hébert, N. Shivappa, M. D. Wirth, J. R. Hussey and T. G. Hurley, Perspective: The Dietary Inflammatory Index (DII)—Lessons learned, improvements made, and future directions, Adv. Nutr., 2019, 10(2), 185–195 CrossRef PubMed.
- N. Shivappa, J. R. Hebert, A. Marcos, L. Diaz, S. Gomez, E. Nova, N. Michels, A. Arouca, E. González-Gil, G. Frederic, M. González-Gross, M. J. Castillo, Y. Manios, M. Kersting, M. J. Gunter, S. D. Henauw, K. Antonios, K. Widhalm, D. Molnar, L. Moreno and I. Huybrechts, Association between dietary inflammatory index and inflammatory markers in the HELENA study, Mol. Nutr. Food Res., 2017, 61(6), 1600707 CrossRef PubMed.
- N. P. Bondonno, L. C. Blekkenhorst, A. L. Bird, J. R. Lewis, J. M. Hodgson, N. Shivappa, J. R. Hébert, R. J. Woodman, G. Wong, D. A. Kerr, W. H. Lim and R. L. Prince, Dietary inflammatory index and the aging kidney in older women: A 10-year prospective cohort study, Eur. J. Nutr., 2020, 59(7), 3201–3211 CrossRef CAS PubMed.
- D. M. Okamura and S. Pennathur, The balance of powers: Redox regulation of fibrogenic pathways in kidney injury, Redox Biol., 2015, 6, 495–504 CrossRef CAS PubMed.
- Y. Zhang, S. Yang, M. Liu, Q. Wu, Z. Ye, C. Zhou, P. He, Y. Zhang, X. Gan and X. Qin, Dietary vitamin E and tocopherol isoforms and incident chronic kidney disease: A 30-y follow-up study from young adulthood to midlife, Free Radicals Biol. Med., 2022, 190, 284–291 CrossRef CAS PubMed.
- Y. S. Joo, H. W. Kim, S. Lee, K. H. Nam, H. R. Yun, J. H. Jhee, S. H. Han, T. H. Yoo, S. W. Kang and J. T. Park, Dietary zinc intake and incident chronic kidney disease, Clin. Nutr., 2021, 40(3), 1039–1045 Search PubMed.
- M. E. Wright, S. T. Mayne, R. Z. Stolzenberg-Solomon, Z. Li, P. Pietinen, P. R. Taylor, J. Virtamo and D. Albanes, Development of a comprehensive dietary antioxidant index and application to lung cancer risk in a cohort of male smokers, Am. J. Epidemiol., 2004, 160(1), 68–76 CrossRef PubMed.
- M. Wang, Z. H. Huang, Y. H. Zhu, P. He and Q. L. Fan, Association between the composite dietary antioxidant index and chronic kidney disease: evidence from NHANES 2011–2018, Food Funct., 2023, 14(20), 9279–9286 RSC.
- C. E. Ndumele, J. Rangaswami, S. L. Chow, I. J. Neeland, K. R. Tuttle, S. S. Khan, J. Coresh, R. O. Mathew, C. M. Baker-Smith, M. R. Carnethon, J. P. Despres, J. E. Ho, J. J. Joseph, W. N. Kernan, A. Khera, M. N. Kosiborod, C. L. Lekavich, E. F. Lewis, K. B. Lo, B. Ozkan, L. P. Palaniappan, S. S. Patel, M. J. Pencina, T. M. Powell-Wiley, L. S. Sperling, S. S. Virani, J. T. Wright, R. R. Singh and M. S. V. Elkind, Cardiovascular–kidney–metabolic health: A Presidential Advisory from the American Heart Association, Circulation, 2023, 148(20), 1606–1635 CrossRef PubMed.
- R. Hariharan, E. N. Odjidja, D. Scott, N. Shivappa, J. R. Hébert, A. Hodge and B. de Courten, The dietary inflammatory index, obesity, type 2 diabetes, and cardiovascular risk factors and diseases, Obes. Rev., 2022, 23(1), e13349 CrossRef CAS PubMed.
- X. Qin, Y. Zhang, Y. Cai, M. He, L. Sun, J. Fu, J. Li, B. Wang, H. Xing, G. Tang, X. Wang, X. Xu, X. Xu and Y. Huo, Prevalence of obesity, abdominal obesity and associated factors in hypertensive adults aged 45–75 years, Clin. Nutr., 2013, 32(3), 361–367 CrossRef PubMed.
- D. Furman, J. Campisi, E. Verdin, P. Carrera-Bastos, S. Targ, C. Franceschi, L. Ferrucci, D. W. Gilroy, A. Fasano, G. W. Miller, A. H. Miller, A. Mantovani, C. M. Weyand, N. Barzilai, J. J. Goronzy, T. A. Rando, R. B. Effros, A. Lucia, N. Kleinstreuer and G. M. Slavich, Chronic inflammation in the etiology of disease across the life span, Nat. Med., 2019, 25(12), 1822–1832 CrossRef CAS PubMed.
- J. Iqbal, H. Wu, M. A. Nawaz, H. Jiang, S. Xu, B. Huang, L. Li, J. Cai and H. Zhou, Risk of incident chronic kidney disease in metabolically healthy obesity and metabolically unhealthy normal weight: A systematic review and meta-analysis, Obes. Rev., 2024, 25(2), e13656 CrossRef CAS PubMed.
- J. D. Imig and M. J. Ryan, Immune and inflammatory role in renal disease, Compr. Physiol., 2013, 3(2), 957–976 CrossRef PubMed.
- C. Sudlow, J. Gallacher, N. Allen, V. Beral, P. Burton, J. Danesh, P. Downey, P. Elliott, J. Green, M. Landray, B. Liu, P. Matthews, G. Ong, J. Pell, A. Silman, A. Young, T. Sprosen, T. Peakman and R. Collins, UK Biobank: An open access resource for identifying the causes of a wide range of complex diseases of middle and old age, PLoS Med., 2015, 12(3), e1001779 CrossRef PubMed.
- M. Liu, X. Gan, Z. Ye, Y. Zhang, P. He, C. Zhou, S. Yang, Y. Zhang and X. Qin, Association of accelerometer-measured physical activity intensity, sedentary time, and exercise time with incident Parkinson's disease, NPJ Digit. Med., 2023, 6(1), 224 CrossRef PubMed.
- B. Liu, H. Young, F. L. Crowe, V. S. Benson, E. A. Spencer, T. J. Key, P. N. Appleby and V. Beral, Development and evaluation of the Oxford WebQ, a low-cost, web-based method for assessment of previous 24 h dietary intakes in large-scale prospective studies, Public Health Nutr., 2011, 14(11), 1998–2005 CrossRef PubMed.
- B. B. Sun, J. Chiou, M. Traylor, C. Benner, Y. H. Hsu, T. G. Richardson, P. Surendran, A. Mahajan, C. Robins, S. G. Vasquez-Grinnell, L. Hou, E. M. Kvikstad, O. S. Burren, J. Davitte, K. L. Ferber, C. E. Gillies, Å.K Hedman, S. Hu, T. Lin, R. Mikkilineni, R. K. Pendergrass, C. Pickering, B. Prins, D. Baird, C. Y. Chen, L. D. Ward, A. M. Deaton, S. Welsh, C. M. Willis, N. Lehner, M. Arnold, M. A. Wörheide, K. Suhre, G. Kastenmüller, A. Sethi, M. Cule, A. Raj, H. M. Kang, L. Burkitt-Gray, E. Melamud, M. H. Black, E. B. Fauman, J. M. M. Howson, M. I. McCarthy, P. Nioi, S. Petrovski, R. A. Scott, E. N. Smith, S. Szalma, D. M. Waterworth, L. J. Mitnaul, J. D. Szustakowski, B. W. Gibson, M. R. Miller and C. D. Whelan, Plasma proteomic associations with genetics and health in the UK Biobank, Nature, 2023, 622(7982), 329–338 Search PubMed.
- Z. Ye, Y. Zhang, Y. Zhang, S. Yang, P. He, M. Liu, C. Zhou, X. Gan, Y. Huang, H. Xiang, F. F. Hou and X. Qin, Large-scale proteomics improve prediction of chronic kidney disease in people with diabetes, Diabetes Care, 2024, 47(10), 1757–1763 CrossRef CAS PubMed.
- H. Xiang, X. Gan, Y. Zhang, Y. Zhang, Z. Ye, S. Yang, Y. Huang, Y. Wu, Y. Zhang and X. Qin, Association of social isolation and plasma metabolites with the risk of venous thromboembolism, Arterioscler., Thromb., Vasc. Biol., 2025, 45(2), 332–340 CrossRef CAS PubMed.
- Z. Yao, X. Jia, Z. Chen, T. Zhang, X. Li, L. Zhang, F. Chen, J. Zhang, Z. Zhang, Z. Liu and Z. Chen, Dietary patterns, metabolomics and frailty in a large cohort of 120000 participants, Food Funct., 2024, 15(6), 3174–3185 Search PubMed.
- P. Würtz, A. J. Kangas, P. Soininen, D. A. Lawlor, G. D. Smith and M. Ala-Korpela, Quantitative serum nuclear magnetic resonance metabolomics in large-scale epidemiology: A primer on -omic technologies, Am. J. Epidemiol., 2017, 186(9), 1084–1096 CrossRef PubMed.
- X. Qin, J. Li, Y. Cui, Z. Liu, Z. Zhao, J. Ge, D. Guan, J. Hu, Y. Wang, F. Zhang, X. Xu, X. Wang, X. Xu and Y. Huo, Effect of folic acid intervention on the change of serum folate level in hypertensive Chinese adults, Pharmacogenet. Genomics, 2012, 22(6), 421–428 CrossRef CAS PubMed.
- P. He, H. Li, C. Liu, M. Liu, Z. Zhang, Y. Zhang, C. Zhou, Q. Li, Z. Ye, Q. Wu, J. Jiang, G. Wang, M. Liang, J. Nie, F. F. Hou and X. Qin, U-shaped association between dietary copper intake and new-onset hypertension, Clin. Nutr., 2022, 41(2), 536–542 Search PubMed.
- H. Shi, L. J. S. Schweren, R. ter Horst, M. Bloemendaal, D. van Rooij, A. A. Vasquez, C. A. Hartman and J. K. Buitelaar, Low-grade inflammation as mediator between diet and behavioral disinhibition: A UK Biobank study, Brain, Behav., Immun., 2022, 106, 100–110 CrossRef CAS PubMed.
- A. V. Khera, C. A. Emdin, I. Drake, P. Natarajan, A. G. Bick, N. R. Cook, D. I. Chasman, U. Baber, R. Mehran, D. J. Rader, V. Fuster, E. Boerwinkle, O. Melander, M. Orho-Melander, P. M. Ridker and S. Kathiresan, Genetic risk, adherence to a healthy lifestyle, and coronary disease, N. Engl. J. Med., 2016, 375(24), 2349–2358 CrossRef CAS PubMed.
- H. Zhang, B. Wang, C. Chen, Y. Sun, J. Chen, X. Tan, F. Xia, J. Zhang, Y. Lu and N. Wang, Sleep patterns, genetic susceptibility, and incident chronic kidney disease: A prospective study of 370671 participants, Front. Neurosci., 2022, 16, 725478 CrossRef PubMed.
- R. Aggarwal, J. W. Ostrominski and M. Vaduganathan, Prevalence of cardiovascular–kidney–metabolic syndrome stages in US adults, 2011–2020, JAMA, 2024, 331(21), 1858–1860 CrossRef CAS PubMed.
- Y. Huang, Y. Zhang, Y. Zhang, H. Xiang, Z. Ye, S. Yang, X. Gan, Y. Wu, Y. Zhang and X. Qin, Hearing impairment, psychological distress, and incident heart failure: A prospective cohort study, Heart, 2025, 111(18), 877–883 CrossRef PubMed.
- L. Richiardi, R. Bellocco and D. Zugna, Mediation analysis in epidemiology: Methods, interpretation and bias, Int. J. Epidemiol., 2013, 42(5), 1511–1519 CrossRef PubMed.
- S. Yang, Z. Ye, X. Gan, Y. Zhang, Y. Huang, H. Xiang, Y. Wu, Y. Zhang, Y. Zhang, F. F. Hou and X. Qin, Plasma metabolites mediate the association of total and specific fruit intake with incident chronic kidney disease risk, Food Funct., 2025, 16(16), 6612–6621 RSC.
- P. Ruggenenti, A. Perna, G. Gherardi, G. Garini, C. Zoccali, M. Salvadori, F. Scolari, F. P. Schena and G. Remuzzi, Renoprotective properties of ACE-inhibition in non-diabetic nephropathies with non-nephrotic proteinuria, Lancet, 1999, 354(9176), 359–364 Search PubMed.
- N. Jourde-Chiche, F. Fakhouri, L. Dou, J. Bellien, S. Burtey, M. Frimat, P. A. Jarrot, G. Kaplanski, M. L. Quintrec, V. Pernin, C. Rigothier, M. Sallée, V. Fremeaux-Bacchi, D. Guerrot and L. T. Roumenina, Endothelium structure and function in kidney health and disease, Nat. Rev. Nephrol., 2019, 15(2), 87–108 CrossRef PubMed.
- Q. Yuan, B. Tang and C. Zhang, Signaling pathways of chronic kidney diseases, implications for therapeutics, Signal Transduction Targeted Ther., 2022, 7(1), 182 CrossRef CAS PubMed.
- K. G. Hicks, A. A. Cluntun, H. L. Schubert, S. R. Hackett, J. A. Berg, P. G. Leonard, M. A. A. Aleixo, Y. Zhou, A. J. Bott, S. R. Salvatore, F. Chang, A. Blevins, P. Barta, S. Tilley, A. Leifer, A. Guzman, A. Arok, S. Fogarty, J. M. Winter, H. C. Ahn, K. N. Allen, S. Block, I. A. Cardoso, J. Ding, I. Dreveny, W. C. Gasper, Q. Ho, A. Matsuura, M. J. Palladino, S. Prajapati, P. Sun, K. Tittmann, D. R. Tolan, J. Unterlass, A. P. VanDemark, M. G. Vander Heiden, B. A. Webb, C. H. Yun, P. Zhao, B. Wang, F. J. Schopfer, C. P. Hill, M. C. Nonato, F. L. Muller, J. E. Cox and J. Rutter, Protein–metabolite interactomics of carbohydrate metabolism reveal regulation of lactate dehydrogenase, Science, 2023, 379(6636), 996–1003 CrossRef CAS PubMed.
- S. Kralisch, A. Hoffmann, N. Klöting, A. Bachmann, J. Kratzsch, J. U. Stolzenburg, A. Dietel, J. Beige, M. Anders, I. Bast, M. Blüher, M. Z. Zhang, R. C. Harris, M. Stumvoll, M. Fasshauer and T. Ebert, FSTL3 is increased in renal dysfunction, Nephrol., Dial., Transplant., 2017, 32(10), 1637–1644 CrossRef CAS PubMed.
- C. Huang, R. Ou, X. Chen, Y. Zhang, J. Li, Y. Liang, X. Zhu, L. Liu, M. Li, D. Lin, J. Qiu, G. Liu, L. Zhang, Y. Wu, H. Tang, Y. Liu, L. Liang, Y. Ding and W. Liao, Tumor cell-derived SPON2 promotes M2-polarized tumor-associated macrophage infiltration and cancer progression by activating PYK2 in CRC, J. Exp. Clin. Cancer Res., 2021, 40(1), 304 CrossRef CAS PubMed.
- J. J. Moreno and M. T. Mitjavila, The degree of unsaturation of dietary fatty acids and the development of atherosclerosis (review), J. Nutr. Biochem., 2003, 14(4), 182–195 CrossRef CAS PubMed.
- M. Ondrussek-Sekac, D. Navas-Carrillo and E. Orenes-Piñero, Intestinal microbiota alterations in chronic kidney disease and the influence of dietary components, Crit. Rev. Food Sci. Nutr., 2021, 61(9), 1490–1502 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |