Camel milk exosomes alleviate hyperglycemia by regulating the hepatic mitochondrial complex I activity, hepatic metabolome, and gut microbiota in high-fat diet (HFD) and streptozotocin (STZ)-induced diabetic mice
Bin
Yang†
ab,
Ling
Liu†
a,
Jingjing
Wang
a,
Shifeng
Du
a,
Hao
Liang
a and
Demtu
Er
 *a
*a
aInner Mongolia Key Laboratory of Basic Veterinary Science, College of Veterinary Medicine, Inner Mongolia Agricultural University, Hohhot, 010011, China. E-mail: eedmt@imau.edu.cn
bKey Laboratory of Animal Embryo and Development Engineering of Universities of Higher Learning, Inner Mongolia Agricultural University, Hohhot 010011, China
First published on 26th November 2025
Abstract
The role of camel milk exosomes (CM-exos) in regulating glucose metabolism in hepatocytes has been demonstrated; however, their therapeutic effects and mechanisms in diabetic mice remain unclear. Hence, this study aimed to investigate the hypoglycemic effect of CM-exos on diabetic mice and unravel the underlying mechanism. The findings showed that CM-exos significantly reduced blood glucose levels, improved lipid metabolism, attenuated liver injury, increased liver glycogen content, and inhibited hepatic gluconeogenesis in diabetic mice. Furthermore, CM-exos restored the activity of mitochondrial complex I in the liver and regulated the liver metabolite levels and gut microbiota composition. In conclusion, the findings provide new evidence that CM-exos may be used as a promising functional food for the intervention of diabetes.
1. Introduction
Diabetes is a global epidemic; a substantial portion of the world's population is currently affected by the disease, and current trends indicate that the incidence will continue to rise. The global prevalence of the disease has been estimated to increase from 10.5% (536.6 million people) in 2021 to 12.2% (783.2 million people) in 2045.1 Conventional antidiabetic drugs include sulfonylureas, biguanides, peroxisome proliferator-activated receptor-γ agonists, and α-glucosidase inhibitors. However, these drugs, when administered singly or in combination, can cause side effects such as severe hypoglycemia, weight gain, low efficacy, lack of target specificity, and solubility and permeability issues.2 Therefore, developing natural hypoglycemic compounds with low side effects and high effectiveness has attracted research attention.3 In this context, foodborne bioactive substances have broad application prospects.4Camel milk (CM), a natural and safe product, has a therapeutic effect on reducing blood glucose levels.5 The anti-diabetic effect of CM is attributed to its content of insulin, insulin-like proteins, and endogenous peptides with dipeptidyl peptidase IV (DPP-IV) inhibitory activity.6,7 Several studies have reported that consuming CM reduces blood glucose levels in patients with diabetes.8 Many bioactive peptides derived from CM have recently been identified and found to target certain key molecular pathways involved in glucose homeostasis.9 Although CM-derived bioactive peptides are a new idea for developing novel antidiabetic drugs, such as peptide drugs, CM-derived bioactive peptides face the challenge of low oral bioavailability owing to their susceptibility to degradation in the gastrointestinal tract.10
Milk (including CM) is rich in exosomes. As natural extracellular nanocarriers, milk exosomes contain bioactive substances, such as proteins, DNA, and microRNAs. The phospholipid bilayer of exosomes protects these molecules and thus prevents miRNAs in exosomes from being degraded in the gastrointestinal tract.11 Although the main active substances in camel milk exosomes (CM-exos) that contribute to lowering blood glucose have not yet been fully identified, current research suggests that proteins, lipids, and miRNAs in CM-exos may play a key role.12,13 CM-exos have been reported to alleviate cancer-related oxidative stress, immunotoxicity, and diabetic nephropathy.11,14 However, the therapeutic effects of CM-exos on diabetes and the underlying mechanisms are yet to be elucidated.
Hepatic glucose production is the leading cause of the increase in fasting blood glucose (FBG) level in patients with diabetes.15 Therefore, regulating hepatic glucose production is essential to develop therapeutic strategies for diabetes.16 Metformin alleviates hyperglycemia primarily by inhibiting hepatic gluconeogenesis via a mechanism that involves the inhibition of mitochondrial complex I, resulting in the activation of adenosine monophosphate-activated protein kinase (AMPK). Activated AMPK subsequently downregulates the expression of key gluconeogenic enzymes, leading to reduced hepatic glucose production.15 This study focuses on the AMPK pathway, based on two lines of prior evidence. First, RNA sequencing of liver tissue from diabetic mice treated with CM-exos revealed a significant downregulation of genes enriched in the “oxidative phosphorylation” pathway. As this process is central to ATP production, its suppression likely reduces the intracellular ATP/AMP ratio—a key trigger for AMPK activation.17 Second, and in direct support of this, our independent in vitro experiments confirmed that CM-exos directly induce AMPK phosphorylation.18 This coherent evidence, from in vivo observation to in vitro validation, justifies targeting the AMPK pathway as the focal point for our mechanistic investigation.
Diabetes is also a metabolic disorder, and metabolomics is helpful for understanding the pathophysiological mechanisms of diabetes, predicting individuals at risk of diabetes, and developing treatments for the disease. STZ-induced type 1 diabetic rats showed metabolite changes in multiple organs.19 Therefore, metabolomics must be applied to explore the effects of alterations in CM-exos on the liver metabolic profile of patients with diabetes. The gut microbiota may contribute to the metabolic health of the human host. Gut microbiota dysbiosis may influence the onset and progression of type 1 diabetes (T1D).20 Studies have shown that diet can alter the gut microbiota composition and influence the development of T1D.21 CM polypeptides can reverse the dysbiosis of the intestinal microbiota in T2D mice by changing the relative expression abundance of certain flora.22 Studies have observed that CM alleviates colitis and acute alcoholic liver disease by modulating the gut microbiota.23,24 Therefore, the gut microbiota may be a potential therapeutic target for CM-exos to resist hyperglycemia in diabetes.
This study provides new insights into the potential of CM-exos in alleviating hyperglycemia in diabetes. By integrating data from multiple systems (mitochondrial function, liver metabolism, and gut microbiota), this research may reveal the complete mechanism of action, paving the way for developing CM-exos-based adjuvant therapies for diabetes.
2. Materials and methods
2.1. Isolation and identification of CM-exos
The milk of the Bactrian camel in mid-lactation was collected from Ejin Banner, Inner Mongolia. The collected CM was transported to the laboratory in ice boxes. CM-exos were isolated via ultracentrifugation as previously described, with some modifications.20 CM was centrifuged at 8000g and 4 °C for 30 min to remove fat, casein, and cell debris. The supernatant was centrifuged at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g and 4 °C for 1 h to remove the remaining fat and cell debris. The skim milk supernatant was ultracentrifuged at 120
000g and 4 °C for 1 h to remove the remaining fat and cell debris. The skim milk supernatant was ultracentrifuged at 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g and 4 °C for 120 min to eliminate the supernatant and obtain the exosome precipitate. The exosome particles were suspended in phosphate-buffered saline to obtain a homogeneous suspension. The CM-exos were filtered using a 0.22 µm filter and then stored in a refrigerator at −80 °C. The total protein concentration of CM-exos was determined using the bicinchoninic acid (BCA) method. The exosomes were fixed in 2.5% glutaraldehyde at 20 °C for 1 h, stained with 2% phosphotungstic acid, and examined using transmission electron microscopy (TEM) (Talos F200C G2, Thermo Fisher Scientific). The sizes of CM-exos were analyzed by nanoparticle tracking analysis (NTA) using ZetaView (Particle Metrix, Munich, Germany). The membrane protein marker cluster of differentiation 81 (CD81) and the tumor susceptibility gene 101 (TSG101) of CM-exos were detected using western blotting (WB).
000g and 4 °C for 120 min to eliminate the supernatant and obtain the exosome precipitate. The exosome particles were suspended in phosphate-buffered saline to obtain a homogeneous suspension. The CM-exos were filtered using a 0.22 µm filter and then stored in a refrigerator at −80 °C. The total protein concentration of CM-exos was determined using the bicinchoninic acid (BCA) method. The exosomes were fixed in 2.5% glutaraldehyde at 20 °C for 1 h, stained with 2% phosphotungstic acid, and examined using transmission electron microscopy (TEM) (Talos F200C G2, Thermo Fisher Scientific). The sizes of CM-exos were analyzed by nanoparticle tracking analysis (NTA) using ZetaView (Particle Metrix, Munich, Germany). The membrane protein marker cluster of differentiation 81 (CD81) and the tumor susceptibility gene 101 (TSG101) of CM-exos were detected using western blotting (WB).
2.2. Animals and experimental design
Five-week-old male C57BL/6J mice were purchased from SKBEX Biology (Henan, China). All animal experiments were approved by the Experimental Animal Welfare and Ethics Committee of Inner Mongolia Agricultural University (no. NND2021094). The mice were kept under 22 °C ± 1 °C temperature, 50% ± 5% humidity, 12 h day/night cycle, and ad libitum feeding conditions. The mice were randomly categorized into five groups (n = 8, 4 mice per cage). The mice in the normal control (NC) group were fed a normal diet. The other groups were fed a high-fat diet (HFD) (D12492; 20% carbohydrate, 20% protein, and 60% fat). After 4 weeks of feeding, mice in the HFD groups were intraperitoneally injected with 40 mg kg−1 streptozotocin (STZ, Yeasen Biotechnology, Shanghai) dissolved in 0.1 mol L−1 citric acid–sodium citrate buffer (pH = 4.4) for five consecutive days to establish diabetic mouse models. Mice in the NC group were intraperitoneally injected with an equal volume of 0.1 mol L−1 citric acid–sodium citrate buffer (pH = 4.4). After 7 days, fasting blood glucose (FBG) was measured from tail vein blood samples in mice after a 12-hour fast, using a glucometer (Roche, Basel, Switzerland). FBG > 11.1 mmol L−1 confirmed the establishment of the diabetic mouse model. Subsequently, 32 diabetic mice were randomly categorized into four groups. The diabetic control (DC) group was given saline by gavage. The metformin (Met) group was treated with 250 mg kg−1 metformin by gavage every 2 days for 5 weeks. The intragastric administration of CM-exos (ig-exo/ig_exo) group was given 2.5 mg kg−1 CM-exos by gavage every 2 days for 5 weeks. The intraperitoneal injection of CM-exos (ip-exo) group received an intraperitoneal injection of 2.5 mg kg−1 CM-exos every 2 days for 5 weeks. Body weight (BW) and FBG were measured weekly in all mice. At the end of the experiment, all mice were sacrificed after 12 h of fasting. Blood samples and liver tissue were stored at −80 °C. A portion of the liver tissue was immersed in 10% formalin for hematoxylin and eosin (HE) staining, and another portion was fixed in Carnoy's fixative solution for Periodic Acid–Schiff (PAS) staining. Moreover, some liver tissue and colon contents were stored in liquid nitrogen for liver metabolomic and intestinal microbiome analyses.2.3. Oral glucose tolerance test (OGTT) and pyruvate tolerance test (PTT)
The OGTT and PTT were performed in diabetic mice treated with metformin or CM-exos at the 5th week. All mice were treated with gavage of glucose (2 g kg−1) or an intraperitoneal injection of pyruvate (2 g kg−1) for the OGTT or PTT, respectively. For the OGTT and PTT, mice were fasted for 16 h, and blood glucose measured immediately before glucose or pyruvate administration (0 min) was defined as the FBG level. Subsequently, FBG was measured continuously at 0, 30, 60, 90, and 120 min using a glucometer. The areas under the curve (AUC) were calculated for the OGTT and PTT.2.4. Measurement of fasting insulin and serum lipids
The fasting insulin (FINS) level in serum was measured using an ELISA kit (mmbio, Jiangsu, China). Homeostatic model assessment of insulin resistance (HOMA-IR) was calculated by using the following equation: HOMA-IR = (FBG (mmol L−1) × FINS (mIU L−1))/22.5.The contents of triglycerides (TGs), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C) in the serum were determined using an appropriate kit (Nanjing Jiancheng Bioengineering Institute, Jiangsu, China).
2.5. Histopathological examination and PAS staining
The liver tissues were collected after euthanizing each group of mice and fixed in 10% formalin for HE staining and Carnoy's fixative solution for PAS staining overnight. The liver tissues were dehydrated with an alcohol gradient and embedded in paraffin. The liver tissues were cut into 5 µm sections and stained with HE and PAS. Images were captured using the ZEN 2.3 system (ZEISS, Oberkochen, Germany). Optical densities of the PAS-stained sections were analyzed using the Image-Pro Plus 6.0 software.2.6. Determination of the mitochondrial complex I activity and reactive oxygen species (ROS) content in the liver tissue
The mitochondrial complex I activities of fresh liver tissue were determined using commercial kits (Solarbio, Beijing, China) according to the manufacturer's instructions. Briefly, the complex I activity was assayed by monitoring the decrease in nicotinamide adenine dinucleotide (NADH) at 340 nm for 2 min. The ROS content of fresh liver tissue was quantified using the ROS content detection kit (Mlbio, Shanghai, China).2.7. Detection of the mRNA level of NADH dehydrogenase (ubiquinone) Fe–S protein 8 (NDUFS8) in the liver tissue
Total RNA was isolated from the liver tissue using the RNAiso Plus reagent (Takara). Total RNA was reverse transcribed using the PrimeScript RT reagent kit with a gDNA Eraser (Takara) according to the manufacturer's instructions. Quantitative real-time PCR (qRT-PCR) was performed with the iTaq Universal SYBY Green Supermix (BIO-RAD) using the ABI7500 Real-Time PCR System (Thermo Fisher Scientific). All values were calculated using the 2−ΔΔCt method and expressed as the change relative to β-actin mRNA expression. The sequences of primers were as follows: NDUFS8-F:GTGGCGGCAACGTACAAGTAT, NDUFS8-R:GAATCCGAGCTGCATTGTCAG; β-actin-F:CTACCTCATGAAGATCCTGACC, β-actin-R:CACAGCTTCTCTTTGATGTCAC.2.8. Measurement of mitochondrial membrane potential (MMP)
To further validate the effect of CM-exos on hepatic mitochondrial function, the MMP of the mouse hepatocyte line AML12 was measured using a JC-1 assay kit (Solarbio, Beijing). Briefly, after treating AML12 cells with metformin (5 mM), rotenone (0.5 µM), and CM-exos (6 ng µL−1), respectively, for 24 h, the cells were incubated with JC-1 at 37 °C for 20 min and washed twice with a staining buffer. Fluorescence intensity was detected using an LSM800 laser scanning confocal microscope (Zeiss, Germany). The ratio of red to green fluorescence reflects the changes in MMP.2.9. Determination of the ATP, ADP, and AMP levels in the liver and serum of mice
The ATP levels in the liver tissue were determined using the ATP content detection kit (Solarbio, Beijing, China). The ATP levels in the serum were measured using ELISA kits (mmbio, Jiangsu, China). The ADP and AMP levels in the mouse liver and serum were measured using ELISA kits (mmbio, Jiangsu, China).2.10. WB
Liver tissues were lysed with radio-immunoprecipitation assay buffer containing phenylmethanesulfonyl fluoride, protease inhibitors (Sigma), and phosphatase inhibitors (Sigma). The protein samples were extracted, and the concentration of each sample was determined using the BCA protein assay kit (Beyotime, Shanghai, China). After adding a loading buffer, the protein samples were denatured at 100 °C for 5 min. Equal-mass protein samples were separated using sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes (Millipore). After blocking with a blocking buffer (Beyotime, Shanghai, China) for 2 h at room temperature, the membranes were incubated with primary antibodies against AMPK (2532, Cell Signaling Technology), phosphorylated AMPK (p-AMPK) (2535, Cell Signaling Technology), phosphoenolpyruvate carboxykinase (PEPCK) (ab70358, Abcam), glucose-6-phosphatase (G6PC) (PA5-42541, Invitrogen), β-actin (GB15003, Servicebio), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH, GB15004, Servicebio) overnight at 4 °C. After rinsing three times with Tris-buffered saline with Tween 20 (TBST), the membrane was incubated with horseradish peroxidase–conjugated secondary antibodies for 1 h at room temperature. The membranes were rinsed three times with TBST and incubated in an enhanced chemiluminescence ultrasensitive luminescent solution for 3 min at room temperature before the signals were captured using a chemiluminescence imaging system (Shenhua Technology, Hangzhou, China). The densities of the protein bands were analyzed using the ImageJ software.2.11. Liver metabolomic analysis
Nontargeted metabolomic analysis of the liver tissue was performed using ultrahigh performance liquid chromatography and mass spectrometry (UHPLC–MS/MS). The metabolites were initially extracted, and 100 mg of the liver tissue was extracted with 500 µL of 80% methanol in water. The samples were incubated on ice for 5 min and then centrifuged at 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g and 4 °C for 20 min. A portion of the supernatant was diluted to a final concentration containing 53% methanol with water. After centrifugation at 15
000g and 4 °C for 20 min. A portion of the supernatant was diluted to a final concentration containing 53% methanol with water. After centrifugation at 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 20 min at 4 °C, the supernatant was collected and injected for liquid chromatography–mass spectrometry analysis.
000g for 20 min at 4 °C, the supernatant was collected and injected for liquid chromatography–mass spectrometry analysis.
UHPLC–MS/MS analyses were performed using a Vanquish UHPLC system (Thermo Fisher Scientific, Germany) coupled with an Orbitrap Q Exactive™ HF mass spectrometer (Thermo Fisher Scientific, Germany) in Novogene Co., Ltd (Beijing, China). Samples were injected onto an ACQUITY UPLC BEH amide column (100 × 2.1 mm, 1.7 μm) using a 12 min linear gradient at a flow rate of 0.2 mL min−1. The eluents for the positive polarity mode were eluent A (0.1% formic acid (FA) in 95% acetonitrile (ACN), 10 mM ammonium acetate) and eluent B (0.1% FA in 50% ACN, 10 mM ammonium acetate). The eluents for the negative polarity mode were eluent A (95% ACN, 10 mM ammonium acetate) and eluent B (50% ACN, 10 mM ammonium acetate). The solvent gradient was set as follows: 2% B, 1.5 min; 2%–100% B, 7 min; 100% B, 9 min; 100%–2% B, 9.1 min; 2% B, 12 min. The Q Exactive™ HF mass spectrometer was operated in the positive/negative polarity mode with a spray voltage of 3.5 kV, a capillary temperature of 320 °C, a sheath gas flow rate of 35 arb, an aux gas flow rate of 10 arb, an S-lens RF level of 60, and an aux gas heater temperature of 350 °C.
These metabolites were annotated using the KEGG (https://www.genome.jp/kegg/pathway.html), HMDB (https://hmdb.ca/metabolites), and LIPIDMaps (https://www.lipidmaps.org/) databases. Principal component analysis (PCA) was performed using metaX (a flexible and comprehensive software for processing metabolomic data). Univariate analysis (t-test) was applied to calculate the statistical significance (P-value). Differential metabolites were screened using a threshold of VIP > 1.0, FC > 1.2, or FC < 0.833, with P < 0.05. The correlation analysis between the liver metabolites and the biochemical parameters was performed using Spearman's correlation in IBM SPSS Statistics 19.
2.12. Gut microbiome analysis
Sequencing libraries were generated, and indexes were added to attribute sequences to each sample. Briefly, polymerase chain reaction (PCR) amplification of the targeted regions was performed using specific primers connected to barcodes. The PCR products of appropriate sizes were selected using 2% agarose gel electrophoresis. Similar amounts of PCR products from each sample were pooled, end-repaired, A-tailed, and further ligated with Illumina adapters. Libraries were sequenced on a paired-end Illumina platform to generate paired-end raw reads. Subsequently, the library quality was evaluated using quantitative PCR. Quantified libraries were pooled and sequenced according to the effective library concentration and the amount required.Paired-end reads were assigned to samples based on their unique barcodes and truncated by cutting off the barcode and primer sequence. Paired-end reads were merged using FLASH (https://ccb.jhu.edu/software/FLASH/). Quality filtering on the raw tags was performed using the fastp (version 0.23.1) software to obtain high-quality clean tags. Chimera sequences were removed using the Vsearch software (V2.16.0).
The Chao1 index was used to evaluate community richness, and the Shannon and Simpson indexes were used to assess community diversity. Principal coordinate analysis (PCoA) and nonmetric multidimensional scaling (NMDS) were used to find differences between groups in the gut microbiota community. Linear discriminant analysis (LDA) effect size (LEfSe) was employed to identify biomarkers (LDA score > 4). The correlation analysis between the gut microbiota and the biochemical parameters was performed using Spearman's correlation in IBM SPSS Statistics 19.
2.13. Statistical analysis
All analyses were performed in three technical replicates. One-way analysis of variance (ANOVA) was applied to determine significant differences among the groups using GraphPad Prism 8 (GraphPad Software, USA). Multiple comparisons of all groups were performed using Tukey's multiple comparison test. Statistical analyses for correlation were performed using Spearman's correlation via IBM SPSS Statistics 19. The data were presented as mean ± standard error of the mean (SEM). Differences of p < 0.05 were considered statistically significant. To elucidate the interplay between the gut microbiota and the liver metabolites, a Sankey diagram was constructed.3. Results
3.1. Identification of CM-exos
After isolating CM-exos via ultracentrifugation, TEM revealed that their morphology was round (Fig. 1A). The NTA results showed that the size of CM-exos ranged from 30 to 180 nm (Fig. 1B). WB identification revealed the presence of the exosome markers TSG101 and CD81 (Fig. 1C).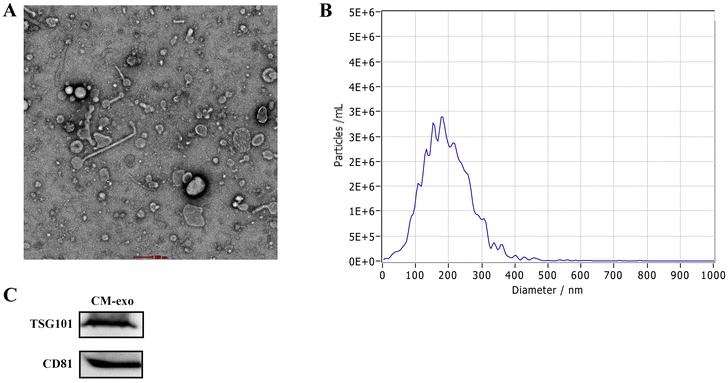 | ||
| Fig. 1 Identification of the isolated CM-exos. (A) TEM image of CM-exos; (B) size distribution of CM-exos determined by NTA; and (C) WB showed the presence of TSG101 and CD81 of the CM-exos protein. | ||
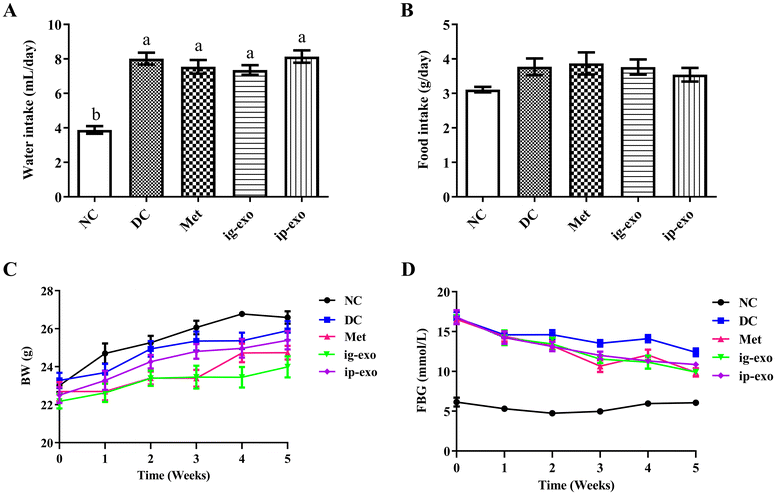
3.2. Effects of CM-exos on water intake, food intake, BW, and FBG in diabetic mice
Significant differences in water or food intake were not observed in the Met (P = 0.842; P = 0.998), ig-exo (P = 0.607; P > 0.999), and ip-exo (P = 0.999; P = 0.953) groups compared with the DC group (Fig. 2A and B). The effect of CM-exos on BW in diabetic mice was observed over a period of 5 weeks. At the 4th and 5th weeks, the BW of mice in the DC group was lower than that of the NC group but not significantly (P = 0.183; P = 0.821). Compared with the DC group, the BW of mice in the Met (P = 0.047; P = 0.040) and ig-exo (P = 0.045; P = 0.0499) groups was significantly decreased at the 2nd and 3rd weeks, whereas there was no significant difference in the ip-exo group (P = 0.709; P = 0.919). At the 4th and 5th weeks, the ig-exo group showed significant weight loss (P = 0.031; P = 0.040), but the weight loss in the Met (P = 0.845; P = 0.385) and ip-exo (P = 0.968; P = 0.930) groups was not significantly different from that in the DC group (Fig. 2C and Table S1). This result suggested that gavage of Met or CM-exos reduced BW in diabetic mice.At week 2, the FBG level began to decrease in the Met group, but it was not significant compared with the DC group (P = 0.432). FBG declined to 10.66 ± 0.74 mmol L−1 at week 3 (P = 0.002) and to 9.88 ± 0.51 at week 5 (P = 0.001), both of which were significant compared with the DC group. The FBG line of the ig-exo group showed a gradual decline from the 1st week to the 5th week, and the average FBG decreased to 11.15 ± 0.82 mmol L−1 at week 4 (P = 0.003) and to 9.91 ± 0.33 mmol L−1 at week 5 (P = 0.001). FBG in the ip-exo group reduced from 16.81 ± 0.87 mmol L−1 at week 1 to 11.31 ± 0.14 mmol L−1 at week 4 (P = 0.006) (Fig. 2D and Table S2). These results indicated that both intragastric administration and intraperitoneal injection of CM-exos could resist hyperglycemia in diabetic mice.
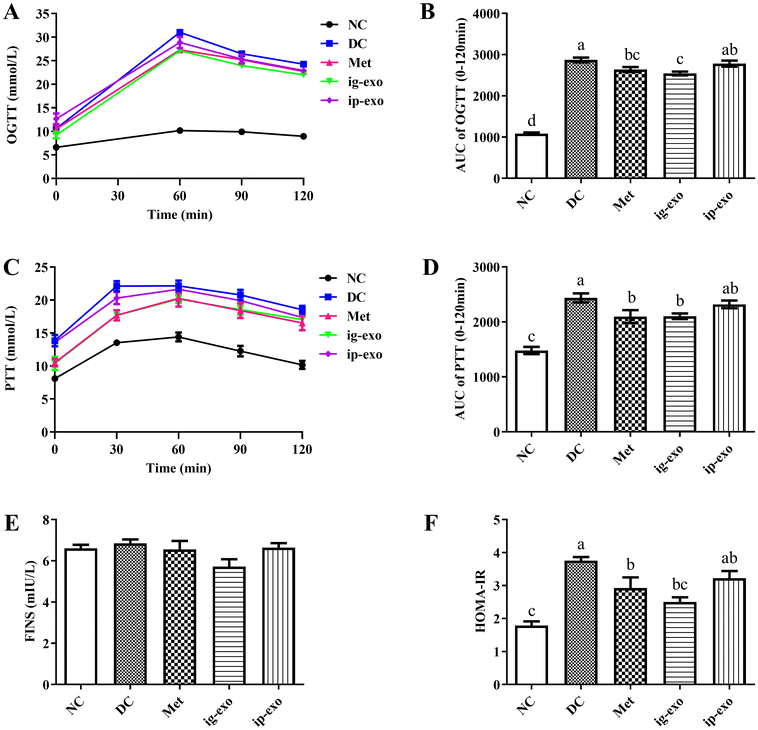
3.3. Effects of CM-exos on the OGTT, PTT, FINS, and insulin resistance
In the OGTT, at 30 min, the blood glucose levels of mice in the DC, Met, ig-exo and ig-exo groups were >33.3 mmol L−1, which was beyond the detection range of the glucometer (detection range: 0.6–33.3 mmol L−1); therefore, the blood glucose value could not be displayed. This experiment recorded blood glucose values at 0, 60, 90, and 120 min. At 60, 90, and 120 min, the blood glucose values in the ig-exo group were significantly lower than those in the DC group (P = 0.004; P = 0.045; P = 0.025). At 60 min, the blood glucose value in the Met group was significantly lower than that in the DC group (P = 0.006) (Fig. 3A and Table S3). The AUC of the OGTT showed that the values of the Met (P = 0.036) and ig-exo (P = 0.001) groups were significantly lower than those of the DC group (Fig. 3B). These results signified that the oral administration of CM-exos exerted a positive effect against diabetes.The peak value was reached at 60 min after the intraperitoneal injection of pyruvate in each group of mice. At 30 min, blood glucose values were significantly lower in the Met (P = 0.002) and ig-exo (P = 0.002) groups than in the DC group (Fig. 3C and Table S4). The AUC of the PTT showed that the values of these two groups were significantly lower than those of the DC group (P = 0.037; P = 0.043) (Fig. 3D). These observations showed that the gastric administration of CM-exos exerted an inhibitory effect on gluconeogenesis in diabetic mice.
No significant differences in FINS levels were found among the groups (P = 0.077) (Fig. 3E). Compared with the NC group, the HOMA-IR was significantly increased in the DC group (P < 0.0001). The HOMA-IR was significantly decreased in the Met (P = 0.041) and ig-exo (P = 0.001) groups compared to the DC group (Fig. 3F). These results indicated that oral administration of CM-exos significantly reduced insulin resistance in diabetic mice.
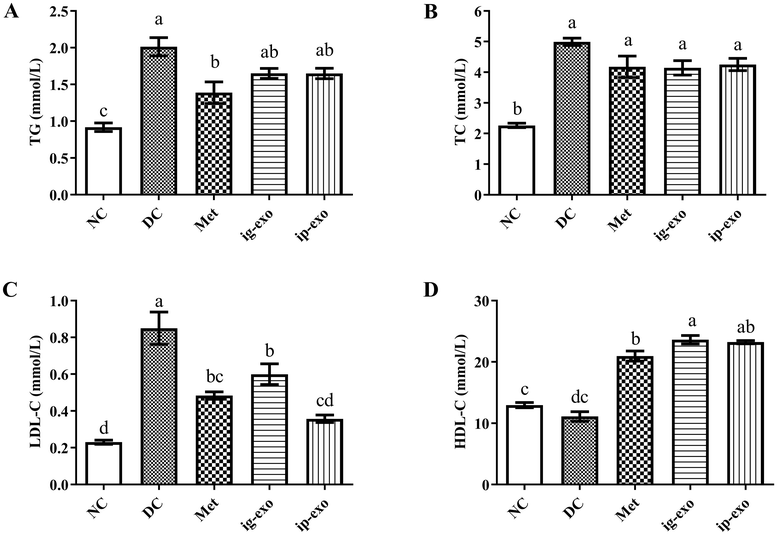
3.4. Effects of CM-exos on serum lipids
Compared with the NC group, TG (P < 0.0001), TC (P < 0.0001), and LDL-C (P < 0.0001) were significantly increased in the DC group. Compared with the DC group, TG in the Met (P = 0.001) group was significantly decreased and TG in the ig-exo (P = 0.098) and ip-exo (P = 0.096) groups was reduced but not significantly (Fig. 4A). TC in the Met (P = 0.084), ig-exo (P = 0.065), and ip-exo (P = 0.139) groups was lowered but not significantly (Fig. 4B). LDL-C in the Met (P < 0.0001), ig-exo (P = 0.007), and ip-exo (P < 0.0001) groups was significantly decreased (Fig. 4C). HDL-C in the Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups was significantly increased (Fig. 4D). These results showed that the intragastric administration and intraperitoneal injection of CM-exos modulated lipid metabolism in diabetic mice.3.5. Effects of CM-exos on liver histopathology and hepatic glycogen
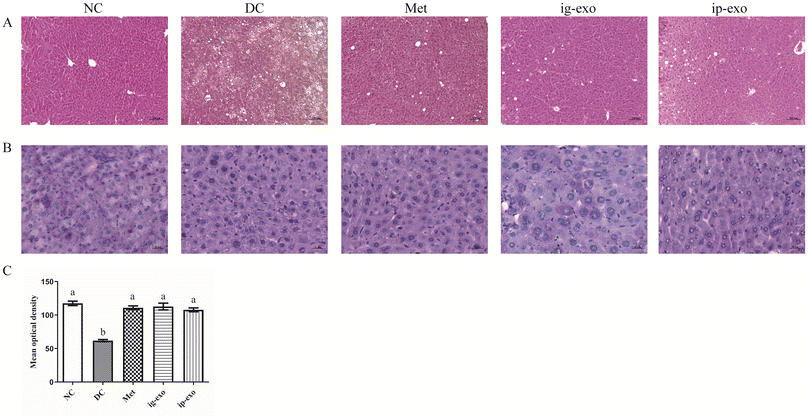
Fat vacuoles were not present in the hepatocytes of the NC group, but many hepatocytes in the DC group contained fat vacuoles of varying sizes. The number of fat vacuoles in the hepatocytes was reduced in the Met, ig-exo, and ip-exo groups compared with the DC group (Fig. 5A). These results indicated that the intragastric administration and intraperitoneal injection of CM-exos could alleviate hepatic steatosis in diabetic mice.The liver glycogen content in the NC (P < 0.0001), Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups was significantly higher than that in the DC group (Fig. 5B and C), which signified that the intragastric administration and intraperitoneal injection of CM-exos could enhance the liver glycogen content.
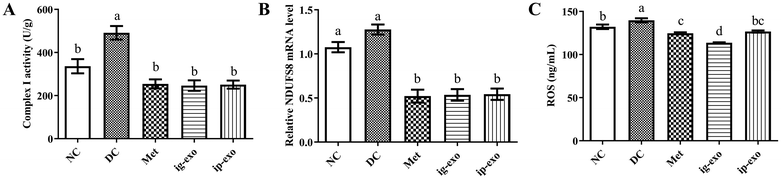
3.6. Effects of CM-exos on the mitochondrial complex I activity, NDUFS8 mRNA level, and ROS level in the liver tissue
The complex I activity of mice in each group was estimated to investigate the effect of CM-exos on the activity of mitochondrial complex I in the liver tissue. The findings indicated that the mitochondrial complex I activity was significantly increased in the DC (P = 0.002) group compared with the NC group. However, the activities of the Met (P = 0.201), ig-exo (P = 0.134), and ip-exo (P = 0.168) groups did not differ significantly from those of the NC group. Compared with the DC group, the activities of the Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups were significantly decreased (Fig. 6A). These results showed that the intragastric administration and intraperitoneal injection of CM-exos could inhibit the activity of mitochondrial complex I in the liver tissue.The mRNA level of NDUFS8, one of the core subunits of mitochondrial complex I, was detected. The results showed that compared with the NC group, the DC group exhibited a slight increase, but the difference was not significant (P = 0.201). The Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups showed a significant decrease. Compared with the DC group, the Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups also showed a significant reduction (Fig. 6B).
The ROS content in the liver tissues of each group revealed that it was significantly increased in the DC group compared with the NC group (P = 0.035). Compared with the DC group, the ROS content was significantly decreased in the Met (P < 0.0001), ig-exo (P < 0.0001), and ip-exo (P < 0.0001) groups (Fig. 6C).
In JC-1 staining, red fluorescence indicates aggregates, reflecting high MMP, while green fluorescence represents monomers, indicating low MMP. The results showed that metformin (P < 0.0001), rotenone (P < 0.0001), and CM-exos (P < 0.0001) all reduced the red to green fluorescence ratio, suggesting a decrease in MMP in each case (Fig. S1).
3.7. Effects of CM-exos on the ATP, ADP, and AMP levels in the liver and serum of mice
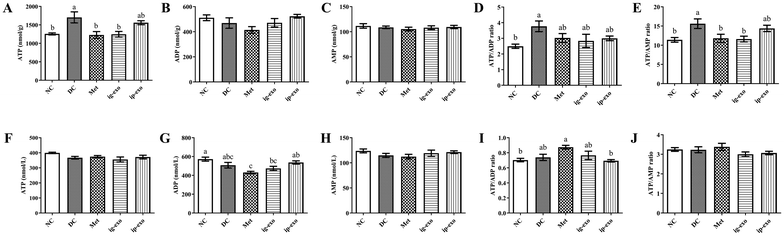
The results of the ATP level in the liver tissues of mice in each group revealed that it was significantly elevated in the DC group compared with the NC group (P = 0.010), whereas its level in the Met (P = 0.999), ig-exo (P > 0.9999), and ip-exo (P = 0.140) groups did not differ significantly from the NC group. Compared with the DC group, the ATP level was significantly decreased in the Met (P = 0.005) and ig-exo (P = 0.008) groups; however, there was no significant difference in the ip-exo group (P = 0.786) (Fig. 7A). No significant differences were observed in the hepatic ADP (P = 0.100) and AMP (P = 0.814) levels across the experimental groups (Fig. 7B and C). Analysis of the liver ATP/ADP ratio revealed a significant increase in the DC group compared to the NC group (P = 0.027). Compared to the DC group, the Met (P = 0.377), ig-exo (P = 0.173), and ip-exo (P = 0.344) groups showed reductions, though these were not statistically significant (Fig. 7D). Analysis of the liver ATP/AMP ratio indicated that its statistical trend was consistent with the content of liver ATP (Fig. 7E).The measurement of serum ATP levels in each group of mice showed no significant differences (P = 0.092) (Fig. 7F). Serum ADP analysis revealed significantly lower levels in the Met (P = 0.0004) and ig-exo (P = 0.021) groups compared to the NC controls, while no significant differences were observed between the DC group and the Met (P = 0.104), ig-exo (P = 0.791), and ip-exo (P = 0.872) groups (Fig. 7G). Analysis of serum AMP levels in mice showed no statistically significant differences among all experimental groups (P = 0.373) (Fig. 7H). Analysis of the serum ATP/ADP ratio showed a significant increase in the Met group compared to the NC group (P = 0.014) (Fig. 7I). Analysis of the serum ATP/AMP ratio showed no significant differences among the groups (P = 0.282) (Fig. 7J).
3.8. Effects of CM-exos on the expression of p-AMPK, PEPCK, and G6PC proteins in the liver tissue
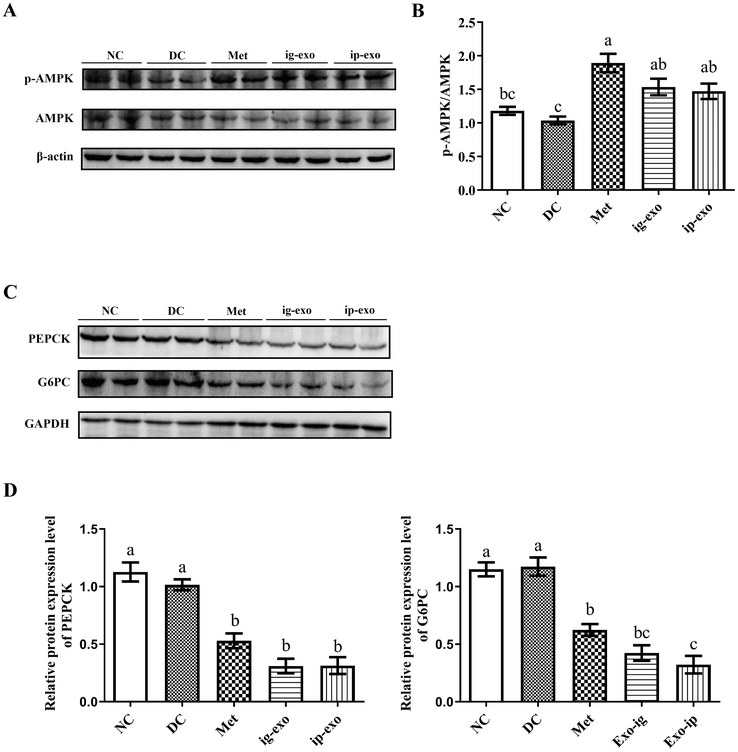
To determine whether CM-exos could activate AMPK, the protein expression levels of AMPK and p-AMPK were detected in the liver tissues of mice in each group. The results showed that compared with the NC group, the p-AMPK/AMPK ratio was decreased in the DC group but not significantly (P = 0.867). Compared with the DC group, the ratio was significantly increased in the Met (P < 0.0001), ig-exo (P = 0.019), and ip-exo (P = 0.049) groups (p < 0.05) (Fig. 8A and B). These findings established that gavage and intraperitoneal injection of CM-exos could activate AMPK.To evaluate whether CM-exos could inhibit the protein expression levels of PEPCK and G6PC, key enzymes of gluconeogenesis, the protein expression levels of PEPCK and G6PC were detected in the liver tissues of mice in each group. The findings signified that the protein expression levels of PEPCK and G6PC in the Met (P = 0.0002; P < 0.0001), ig-exo (P < 0.0001; P < 0.0001), and ip-exo (P < 0.0001; P < 0.0001) groups were significantly decreased compared with the DC group. Nonetheless, there was no significant difference between the NC and DC groups (P = 0.764; P = 0.999) (Fig. 8C and D). These observations showed that the intragastric administration and intraperitoneal injection of CM-exos could inhibit the protein expression levels of PEPCK and G6PC.
3.9. Effects of CM-exos on metabolites in the liver tissue
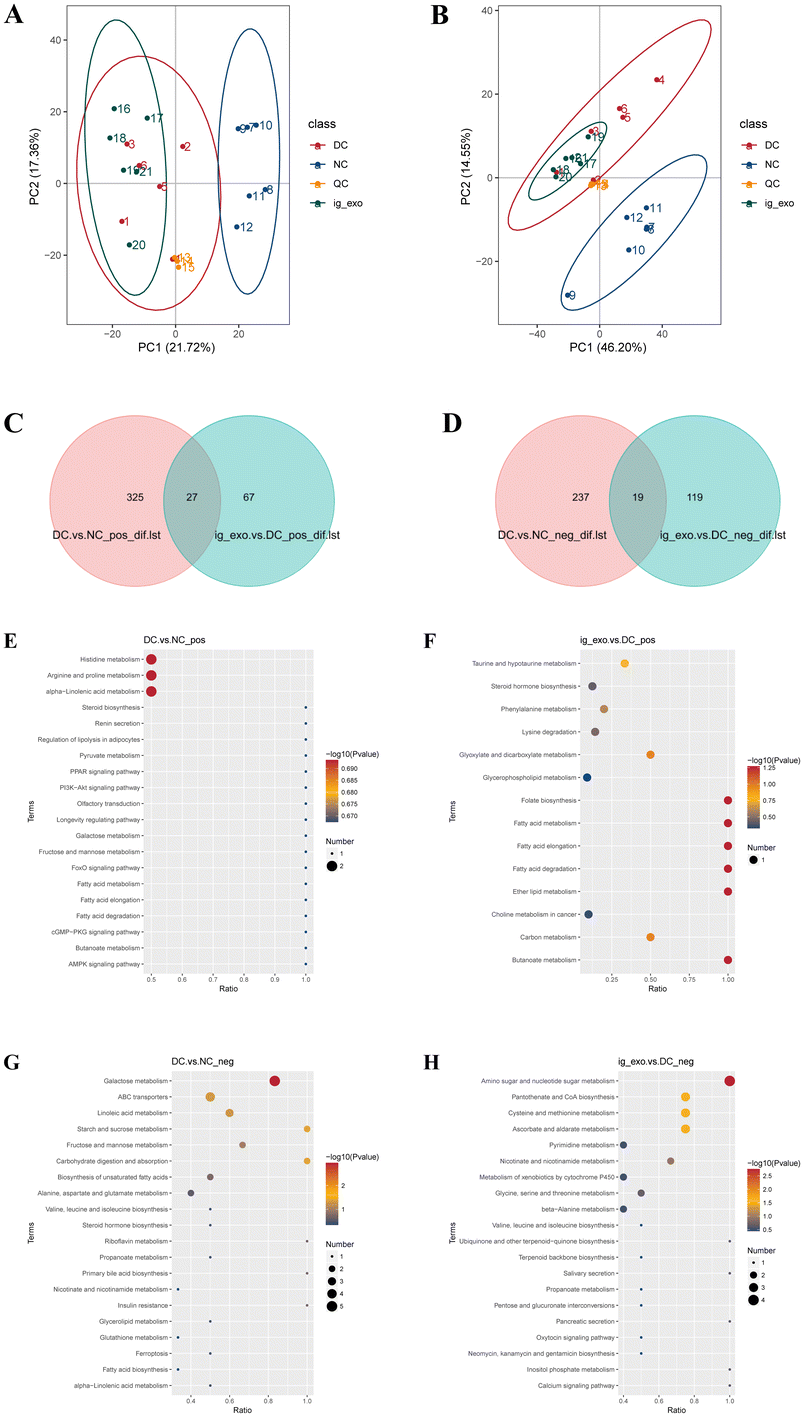
The differences in liver metabolites among the NC, DC, and ig-exo groups were examined using nontargeted metabolomics. The PCA plots showed a significant separation between the NC and DC groups in both positive and negative modes, indicating a considerable difference in metabolites between these two groups (Fig. 9A and B). Differential metabolites were screened using a threshold of VIP > 1.0, FC > 1.2 or FC < 0.833, with P < 0.05. A total of 94 (in the positive mode) and 138 (in the negative mode) differential metabolites were identified between the ig-exo and DC groups. The Venn diagram showed 27 (in the positive mode) and 19 (in the negative mode) overlapping differential metabolites in the DC vs. NC and ig-exo vs. DC comparisons (Fig. 9C and D) (Table S5 and S6).Pathway enrichment analysis of the differential metabolites was performed to identify the main biochemical metabolic pathways involved in them. In the positive mode, differential metabolites in the DC vs. NC group were enriched in pathways such as arginine and proline metabolism, histidine metabolism, and alpha-linolenic acid metabolism (Fig. 9E). Differential metabolites in the ig-exo vs. DC group were enriched in pathways such as fatty acid metabolism, elongation, and degradation (Fig. 9F).
In the negative mode, differential metabolites in the DC vs. NC group were significantly enriched in pathways such as galactose metabolism (P = 0.001), starch and sucrose metabolism (P = 0.038), carbohydrate digestion and absorption (P = 0.038), and ABC transporters (P = 0.049) (Fig. 9G and Table 1). Differential metabolites in the ig-exo vs. DC group were enriched in amino sugar and nucleotide sugar metabolism (P = 0.002), ascorbate and aldarate metabolism (P = 0.030), cysteine and methionine metabolism (P = 0.030), and pantothenate and CoA biosynthesis (P = 0.030) (Fig. 9H and Table 2).
| Pathway | P-value | Compound name |
|---|---|---|
| Galactose metabolism | 0.0012 | 3-Beta-D-galactosyl-sn-glycerol; galactinol; galactose-1-phosphate; stachyose; D-fructose |
| Starch and sucrose metabolism | 0.0382 | Trehalose; D-fructose |
| Carbohydrate digestion and absorption | 0.0382 | Maltotriose; D-fructose |
| ABC transporters | 0.0495 | D-Allose; maltotriose; trehalose; D-fructose |
| Pathway | P-value | Compound name |
|---|---|---|
| Amino sugar and nucleotide sugar metabolism | 0.0017 | N-Acetyl-alpha-D-glucosamine-1-phosphate; D-glucuronic acid; galactose-1-phosphate; N-glycolylneuraminic acid |
| Ascorbate and aldarate metabolism | 0.0304 | D-Glucuronic acid; gulonolactone; threonic acid |
| Cysteine and methionine metabolism | 0.0304 | N-Formylmethionine; 2-ketobutyric acid; L-aspartic acid |
| Pantothenate and CoA biosynthesis | 0.0304 | Pantetheine 4′-phosphate; vitamin B5; L-aspartic acid |
3.10. Effects of CM-exos on the gut microbiome
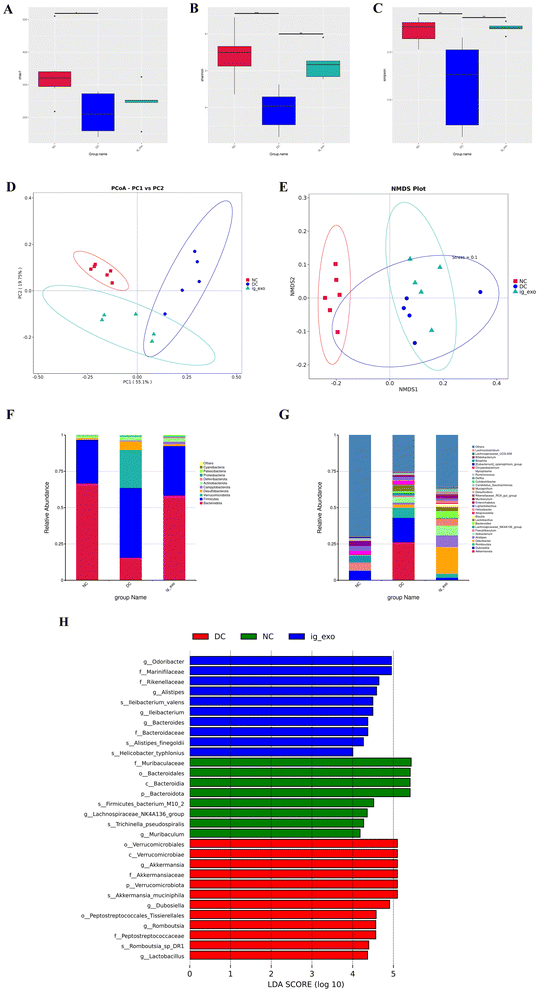
α-Diversity analysis revealed that the Chao1 index was significantly higher in the NC group than in the DC group (P = 0.014), higher in the ig-exo group than in the DC group but not significant (P = 0.484), and higher in the NC group than in the ig-exo group but not significant (P = 0.058) (Fig. 10A). Upon comparing the Shannon indexes of the three groups, it was seen that the NC group had a significantly higher value than the DC group (P = 0.001), the ig-exo group exhibited a significantly higher value than the DC group (P = 0.004), and the NC group displayed a higher value than the ig-exo group but not significant (P = 0.511) (Fig. 10B). Upon comparing the Simpson indexes of the three groups, it was seen that the value of the NC group was significantly higher than that of the DC group (P = 0.004), the value of the ig-exo group was significantly higher than that of the DC group (P = 0.003), and the value of the NC group was higher than that of the ig-exo group but not significant (P = 0.805) (Fig. 10C).The β-diversity of the colonic microbiome in the NC, DC, and ig-exo groups was analyzed. The PCoA and NMDS analyses revealed a clear separation among the NC, DC, and ig-exo groups (Fig. 10D and E), indicating that the gastric administration of CM-exos substantially altered the intestinal microbiota structure in diabetic mice.
Relative abundances at the top 10 phylum levels and top 30 genus levels in the three groups of mice were identified (Fig. 10F and G). LEfSe analysis was performed to determine the main biomarkers (LDA score > 4) (Fig. 10H). The results showed that the main biomarkers in the NC group were Bacteroidota at the phylum level and Lachnospiraceae_NK4A136_group and Muribaculum at the genus level. The key biomarkers in the DC group were Verrucomicrobiota at the phylum level and Akkermansia, Dubosiella, Romboutsia, and Lactobacillus at the genus level. The major biomarkers in the ig-exo group were Odoribacter, Alistipes, Ileibacterium, and Bacteroides at the genus level.
At the phylum level, the relative abundance of Firmicutes, Verrucomicrobiota, and Desulfobacterota was higher in the DC group than in the NC and ig-exo groups, whereas the relative abundance of Bacteroidota and Patescibacteria was lower in the DC group than in the NC and ig-exo groups. The Firmicutes/Bacteroidota ratio was higher in the DC group than in the NC and ig-exo groups. This result indicated that the intragastric administration of CM-exos reversed the trend of flora abundance in these five phyla (Fig. 11). At the genus level, Akkermansia, Dubosiella, and Romboutsia were more abundant in the DC group than in the NC and ig-exo groups. Blautia, Candidatus_Saccharimonas, Bilophila, and Lachnospiraceae_UCG-006 were less abundant in the DC group than in the NC and ig-exo groups (Fig. 11). These findings signified that the intragastric administration of CM-exos reversed the trend of flora abundance in these seven genera.
 | ||
| Fig. 11 Differences in the relative abundances of the gut microbiota. The letters with different superscripts indicate significant differences (p < 0.05) between each group. | ||
3.11. Correlation analysis of the liver metabolites, gut microbiota, and biochemical parameters
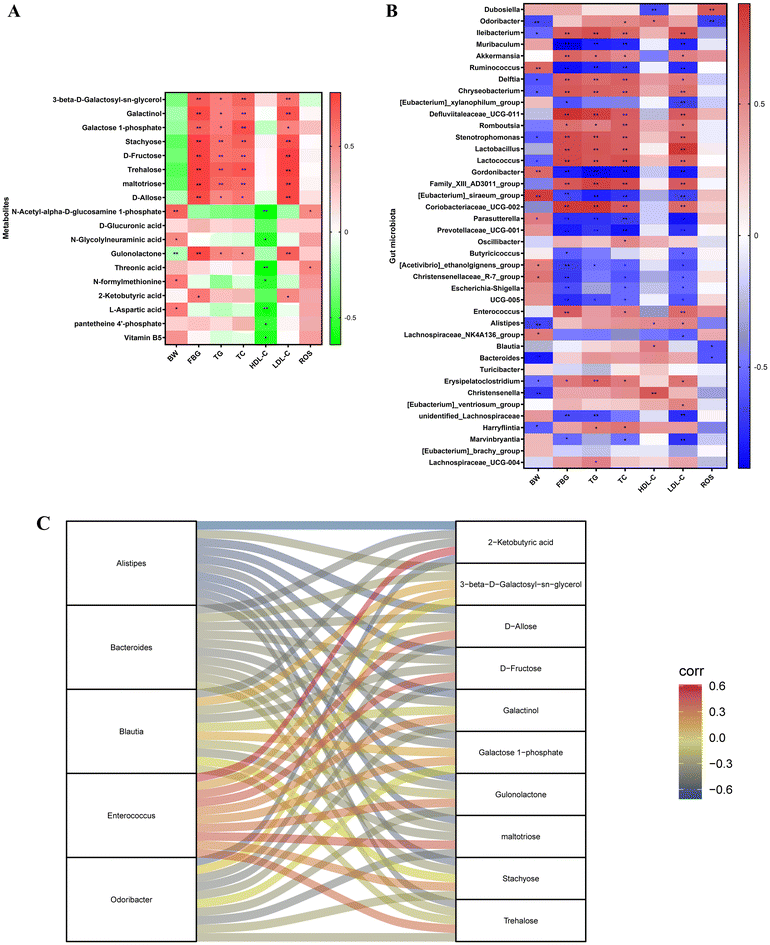
The correlation analysis between the liver metabolites and the biochemical parameters revealed that 3-beta-D-galactosyl-sn-glycerol, galactinol, galactose-1-phosphate, stachyose, D-fructose, trehalose, maltotriose, D-allose, gulonolactone, and 2-ketobutyric acid were positively correlated with FBG, TG, TC, and LDL-C (Fig. 12A). Furthermore, these metabolites were consistently higher in the DC group than in the NC and ig-exo groups (Fig. S2). 3-Beta-D-galactosyl-sn-glycerol, galactinol, galactose-1-phosphate, stachyose, D-fructose, trehalose, maltotriose, D-allose and gulonolactone were negatively correlated with BW. Galactinol, galactose-1-phosphate, D-allose, N-acetyl-alpha-D-glucosamine-1-phosphate, D-glucuronic acid, N-glycolylneuraminic acid, gulonolactone, threonic acid, N-formylmethionine, 2-ketobutyric acid, L-aspartic acid, pantetheine 4′-phosphate and vitamin B5 were negatively correlated with HDL-C (Fig. 12A).The correlation analysis between the gut microbiota and the biochemical parameters showed that Ileibacterium, Akkermansia, Delftia, Chryseobacterium, Defluviitaleaceae_UCG-011, Romboutsia, Stenotrophomonas, Lactobacillus, Lactococcus, Family_XIII_AD3011_group, Coriobacteriaceae_UCG-002, Enterococcus, and Erysipelatoclostridium were positively correlated with FBG, TG, TC, and LDL-C. Muribaculum, Ruminococcus, [Eubacterium]_xylanophilum_group, Gordonibacter, [Eubacterium]_siraeum_group, Parasutterella, Prevotellaceae_UCG-001, Butyricicoccus, [Acetivibrio]_ethanolgignens_group, Christensenellaceae_R-7_group, Escherichia-Shigella, UCG-005, unidentified_Lachnospiraceae, and Marvinbryantia were negatively correlated with FBG, TG, TC, and LDL-C (Fig. 12B).
This study employed Sankey diagrams (Fig. 12C) to visualize and systematically analyze the correlation network between the key intestinal bacterial genera and hepatic metabolites in diabetic mice following CM-exos intervention. The results revealed a range of significant and potentially biologically relevant associations between specific gut microbes and liver metabolites. Among all the correlations analyzed, the genus Alistipes showed the strongest negative correlation with the hepatic metabolite 2-ketobutyric acid (r = −0.709, P = 0.022), suggesting that an increase in its abundance may be closely linked to reduced hepatic levels of 2-ketobutyric acid under CM-exos intervention. Furthermore, the genus Alistipes displayed a broad pattern of negative correlations with several carbohydrate metabolites, including galactitol, stachyose, D-fructose, trehalose, D-allose, and gulonolactone. Similarly, the genus Blautia showed a consistent negative correlation trend, particularly with respect to 2-ketobutyric acid, D-fructose, and maltotriose. It is also noteworthy that Odoribacter was moderately negatively correlated with both D-fructose and maltotriose (r = −0.491 for both), while the genus Bacteroides displayed a widespread tendency for moderate negative correlations with a spectrum of metabolites.
4. Discussion
Although CM has long been proven effective in treating diabetes, few reports are available on managing the condition with CM-exos.25 In recent years, CM-exos have shown great potential in cancer and diabetic nephropathy therapy, and immune stimulation.26 A high FBG level is the key indicator of diabetes. In this study, T1D was successfully induced in mice using an HFD combined with intraperitoneal injection of STZ. High FBG levels in diabetic mice persisted for 5 weeks. This modeling method was similar to that reported in previous studies.27 Administering CM-exos by gavage and intraperitoneal injection decreased the FBG level significantly at the 4th week, showing that CM-exos exerted an antihyperglycemic role, but rather gradually. The animal model in this study showed hyperglycemic characteristics of T1D, but the absence of significant weight loss in the DC group was likely attributable to the relatively short post-modeling observation period and the ongoing HFD, which supplied excess energy that partly offset catabolic processes. This metabolic profile suggested that the model represented an early stage of T1D. Gavage of metformin and CM-exos resulted in weight loss in T1D mice. The reason may be the activation of AMPK, which promotes fat metabolism and further reduces BW.28Compared with the Met and ip-exo groups, the OGTT levels in the ig-exo group were significantly decreased at 60, 90, and 120 min, showing that the intragastric administration of CM-exos exerted a more pronounced antihyperglycemic effect than the intragastric administration of metformin and intraperitoneal injection of CM-exos. Similarly, the OGTT of diabetic rats gavaged with CM revealed a sustained and significant improvement in FBG levels.29 The PTT is the method for determining gluconeogenesis.30 After the gastric administration of metformin and CM-exos, the PTT results showed a decrease in blood glucose levels at 30 min, implying inhibition of hepatic gluconeogenesis in diabetic mice. The decreased expression of PEPCK and G6PC proteins, key enzymes in hepatic gluconeogenesis, supported this finding.
Diabetes is often accompanied by dyslipidemia, resulting in alterations in plasma TC, TG, LDL-C and HDL-C.31 The TG and TC levels were significantly elevated in diabetic mice in this study. These observations agree with the dyslipidemia reported previously in T1D mice.32 The intragastric or intraperitoneal injection of CM-exos did not considerably reduce the levels of TC and TG. Combined with a systematic review of the effect of CM on blood lipids in patients with diabetes,33 the reason may be the short duration of intervention with CM-exos in diabetic mice. The steatosis in the liver of diabetic mice in this study is consistent with previous investigations.34 Gavage or intraperitoneal injection of CM-exos reduced liver steatosis in diabetic mice to different degrees, which agrees with the finding that CM peptides decreased the number of vacuoles in the liver.22 The liver is the main organ for glycogen storage, and diabetes leads to reduced glycogen synthesis.35,36 This study showed that treatment with CM-exos could restore liver glycogen content and sustain glucose homeostasis in diabetic mice.
Alterations in mitochondrial complex I activity may influence ATP levels.37 Accordingly, this study quantified the concentrations of ATP, ADP, and AMP in both liver tissue and serum. While AMP levels showed no significant change, CM-exos treatment resulted in a reduced ATP/AMP ratio in the liver. Since a decreased ATP/AMP ratio is known to activate AMPK,17 the activity of AMPK was also assessed in this investigation. Our previous research showed that CM-exos activated AMPK and reduced the protein expression levels of PEPCK and G6PC by inhibiting mitochondrial complex I in mouse hepatocytes.18 These results were confirmed using a diabetic mouse model. Hepatic mitochondrial complex I-dependent oxidative phosphorylation was significantly elevated in T1D mice during the early stages of the disease. Cellular experiments have shown that inhibiting hepatic mitochondrial complex I may reduce gluconeogenesis.38 The study concluded that CM-exos may exert a preventive effect against T1D in its early stages by inhibiting hepatic mitochondrial complex I. The results of this study indicate that CM-exos and metformin exhibit similar hypoglycemic mechanisms,39 inhibiting mitochondrial complex I, activating AMPK, and preventing hepatic gluconeogenesis. Mitochondrial complexes I and III are considered the main sites of ROS production.40 Therefore, ROS reduction in the liver after CM-exos intervention in the present study could be attributed to the inhibition of mitochondrial complex I. CM has been reported to improve glucose homeostasis in diabetic rats by activating hepatic AMPK.41 The alleviation of hepatic steatosis by CM protein hydrolysates in rats was associated with increased hepatic AMPK activity.42 These studies support the results of the present study in which CM-exos activated hepatic AMPK in diabetic mice. Building on our previous work, the present findings provide further evidence that CM-exos alleviate diabetic liver injury through the AMPK pathway. Our initial hypothesis originated from the hepatic transcriptome data showing that CM-exos treatment downregulated oxidative phosphorylation genes, thereby implying energy stress and potential AMPK activation. Subsequent in vitro experiments directly confirmed this in vivo-derived hypothesis. Therefore, although diabetic signaling is complex, we have defined AMPK as the core mechanistic target in this study, justified by our specific and sequential experimental evidence.
To elucidate the central metabolites in the key metabolic pathways, a systematic screening of differential metabolites was performed. We first identified four pathways that were significantly intervened by CM-exos. Subsequently, within the pathway most relevant to hepatic glucose dysregulation, we focused on two key metabolites: galactose-1-phosphate and 2-ketobutyric acid. This study showed that treatment with CM-exos markedly decreased the levels of galactose-1-phosphate and 2-ketobutyric acid in the livers of diabetic mice, implying that the improvement in glycemic stability may be attributed to the suppression of excessive hepatic gluconeogenesis. Galactose-1-phosphate, an intermediate in the Leloir pathway, is ultimately converted to glucose-6-phosphate (G6P), a direct precursor of gluconeogenesis.43 The observed reduction in its level suggested an enhanced flux through the Leloir pathway, accompanied by diminished intermediate accumulation, which likely led to decreased net formation of galactose-derived G6P, thereby reducing the substrate availability for gluconeogenesis. 2-Ketobutyric acid, a metabolite derived from the catabolism of glucogenic amino acids, serves as a precursor for oxaloacetate—a key anaplerotic substrate that initiates gluconeogenesis.44 The lowered level of 2-ketobutyrate indicated that CM-exos inhibited the excessive breakdown of glucogenic amino acids, consequently restricting carbon flux into the gluconeogenic pathway at an upstream stage and attenuating hepatic glucose output from the source. Additionally, two potential functional molecules with reported glucose-lowering efficacy were identified: artemether (Table S5) and antcin K (Table S6). Literature reports support that artemether lowers FBG and lipids in diabetic mice by inhibiting gluconeogenesis and fatty acid synthesis via activation of the hepatic AMPK pathway.45 Similarly, antcin K is known to improve glycemia and insulin resistance by suppressing gluconeogenesis and reducing liver fat deposition through AMPK pathway activation.46
In this study, correlation analysis revealed that D-fructose and 2-ketobutyric acid were positively associated with FBG, TG, TC, and LDL-C. D-Fructose, which is primarily metabolized in the liver, serves as a key substrate for intrahepatic triglyceride synthesis. Excessive intake of D-fructose can lead to hypertriglyceridemia, oxidative stress, insulin resistance, gut dysbiosis, and non-alcoholic fatty liver disease (NAFLD).47 The reduction of hepatic fructose levels by CM-exos directly diminishes the substrate available for de novo lipogenesis, indicating their therapeutic potential in the prevention and treatment of NAFLD. Our previous studies have identified several miRNAs enriched in CM-exos, such as miR-148a, let-7i, and miR-200a.48 Of these, miR-200a is well established for its potent antioxidant properties.49 We thus propose that miR-200a delivered by CM-exos improves the hepatic microenvironment by mitigating oxidative stress, thereby restoring normal hepatocyte function. This improvement subsequently enhances the liver's ability to metabolize fructose, thereby reducing fructose accumulation. 2-Ketobutyric acid is involved in the metabolism of various amino acids. It is one of the degradation products of threonine and can be converted into an intermediate of the citric acid cycle, thereby participating in the cycle itself.50 Plasma levels of 2-ketobutyric acid showed a significant positive correlation with both impaired glucose tolerance and elevated fasting glucose.51 Elevated levels of 2-ketobutyric acid have been associated with disruptions in both energy and amino acid metabolism during the early stages of diabetes.52 These findings indicate that CM-exos contribute to the restoration of hepatic homeostasis in amino acid and energy metabolism. Alterations in pantothenate and CoA levels regulate mitochondrial energy metabolism and are closely linked to the progression of diabetic retinopathy.53,54 Pantetheine 4′-phosphate serves as a vital cofactor for the key proteins involved in diverse metabolic pathways in the cytosol and mitochondria, such as fatty acid biosynthesis and mitochondrial respiratory complex I assembly.55 In this study, the level of pantetheine 4′-phosphate enriched in pantothenate and the biosynthesis of CoA were lower in the ig-exo group than in the DC group. Therefore, we speculate that the reduction in this essential cofactor may contribute to the observed inhibition of hepatic mitochondrial complex I.
Milk-derived exosomal miRNAs, such as miR-200a and let-7a, can regulate the expression of the key genes involved in intestinal barrier integrity and immune homeostasis. This regulatory effect is significantly correlated with changes in the composition of the gut microbiota.13 The microbiota structure was altered in diabetic mice after gavage with CM-exos. The increased abundance of Firmicutes and the decreased abundance of Bacteroidota in diabetic mice observed in the present investigation have been confirmed by another study.56 The gavage of CM-exos reversed the abundance of these two bacteria, suggesting that the hypoglycemic effect of CM-exos is mediated through the regulation of the intestinal microbiota composition in diabetic mice. Desulfobacterota can reduce sulfate and other sulfur compounds. The bacteria might exacerbate abnormal energy metabolism.57 Desulfobacterota is a pathogenic bacterium known for its toxicity to the intestinal epithelium and is implicated in the development of gastrointestinal disorders.58
At the genus level, the increase in Akkermansia, Dubosiella, and Romboutsia and the decrease in Blautia, Candidatus_Saccharimonas, Bilophila, and Lachnospiraceae_UCG-006 were reversed by the intragastric administration of CM-exos. High-fat diet feeding in mice induces an increase in intestinal mucus layer thickness and upregulates the expression of mucin genes (such as Muc2).59Akkermansia is highly abundant in mucin-enriched regions of the gut and is positively correlated with mucin concentration. Akkermansia may potentially increase the risk of enteropathogenic infections and inflammation.60 In this study, the relative abundance of Akkermansia in the DC group of mice reached a peak value of 50.1%. These findings indicated that the high-fat diet likely promoted excessive secretion of aberrant mucins by goblet cells. Since Akkermansia thrives almost exclusively on mucin as its nutrient source, this altered mucin availability may have directly fueled its rapid proliferation, ultimately leading to a dominant presence within the gut microbial community. CM-exos decreased the relative abundance of Akkermansia in the colon of diabetic mice induced by STZ and a high-fat diet. These results implied that CM-exos contributed to the restoration of a severely dysbiotic state, thereby promoting a return to a healthy and balanced gut environment. Dubosiella is linked to lipid metabolism and has exhibited opposing abundance trends in different studies. Certain studies have observed that it was higher in the intestines of HFD/STZ-induced diabetic mice than in normal mice, which was reversed in the treatment group.61,62 Other studies have reported contradictory findings.63,64 Both Romboutsia and Dubosiella belong to the phylum Firmicutes. Studies on the intestinal microbiota in HFD/STZ-induced diabetic mice showed inconsistencies in the abundance of Romboutsia. A study reported that increased Romboutsia abundance in HFD/STZ-induced diabetic mice was reduced by ginseng-derived γ-aminobutyric acid–fructosyl-glucose,65 a result similar to the present study. However, another investigation found that Romboutsia abundance in HFD/STZ-induced diabetic mice was lower than that in normal mice.58Blautia was identified as a genus that produces abundant short-chain fatty acids (SCFAs).66 The decrease in Blautia abundance is one of the key characteristics of diabetes-associated dysbiosis.67 Oral administration of Blautia inhibited BW gain, lipid accumulation, and glucose level increase in a HFD-induced T2D mouse model.68 In this research, the relative abundance of Blautia was much higher in mice gavaged with CM-exos than in normal and diabetic mice. Lachnospiraceae_UCG-006 belongs to the Lachnospiraceae family, which can convert polysaccharides into SCFAs.69 The relative abundance of Lachnospiraceae_UCG-006 in the stool of patients with T2D was significantly lower than that in healthy individuals.70 Statin and tocotrienol supplementation enhanced the abundance of Lachnospiraceae_UCG-006 in obese mice.71 The above finding is comparable to this study.
The genera Ruminococcus, [Eubacterium]_xylanophilum_group, and Muribaculum were negatively correlated with the FBG, TG, TC, and LDL-C levels. Their relative abundances were lower in the DC group than in both the NC group and the ig-exo group. All three genera are known to produce SCFAs,56,72,73 which serve as the primary energy source for colonocytes, enhance intestinal barrier integrity, and mitigate conditions such as “leaky gut” and endotoxemia. Furthermore, SCFAs entering the systemic circulation contribute to the regulation of glucose metabolism by stimulating glucagon-like peptide-1 (GLP-1) secretion, suppressing hepatic glucose production, and improving insulin sensitivity.74
To further elucidate the gut–liver axis communication under CM-exos intervention, we performed a correlation network analysis complemented by a Sankey diagram, which clearly illustrates the potential associations linking the gut microbiota and the liver metabolites. Regarding the origin of the metabolites, it is important to clarify that the hepatic metabolites identified in our study (such as 2-ketobutyric acid and various sugars) are primarily host-derived intermediates of internal metabolic pathways, rather than direct products of gut microbial metabolism. However, their levels in the liver can be significantly modulated by the gut microbiota through indirect mechanisms. Our analysis revealed that the most robust association was a significant negative correlation between the genus Alistipes and hepatic 2-ketobutyric acid. We proposed a potential mechanistic pathway: CM-exos may promote the growth of Alistipes in the gut. This shift in the microbial community then contributes, through mechanisms yet to be fully elucidated, to the improved hepatic metabolic homeostasis, leading to a reduction in 2-ketobutyric acid. This reduction is crucial as 2-ketobutyric acid, a key intermediate in threonine and methionine metabolism, accumulates under diabetic conditions and is often linked to oxidative stress and mitochondrial dysfunction.52 Thus, the Alistipes–2-ketobutyric acid axis represents a novel potential mechanism through which CM-exos alleviate hepatic metabolic stress. This perspective is further supported by the broad negative correlations between Alistipes and various carbohydrates, indicating its potential role in enhancing systemic carbohydrate utilization and reducing the hepatic sugar load. Similarly, the consistent negative correlation pattern exhibited by the genus Blautia with hepatic sugars, though not statistically significant in our cohort, suggests another plausible pathway. Blautia is a notable producer of SCFAs, particularly butyrate.75 We hypothesize that CM-exos intervention might enrich Blautia, thereby increasing butyrate production. This butyrate can then reach the liver via the portal circulation, where it may enhance hepatocyte sugar uptake and oxidation, or inhibit gluconeogenesis. This would logically result in the decreased hepatic levels of sugar intermediates we observed, ultimately contributing to improved insulin sensitivity and metabolic health. In summary, our findings delineate a complex interaction network along the gut–liver axis. We provide evidence that CM-exos intervention is associated with specific alterations in the gut microbiota, which are in turn correlated with beneficial shifts in host hepatic metabolism. The associations centered on Alistipes and Blautia offer compelling, albeit correlative, insights into how CM-exos might exert their anti-diabetic effects by modulating the gut–liver axis, highlighting its potential as a target for nutritional interventions.
While our study provides compelling evidence that CM-exos alleviate hyperglycemia through integrated modulation of the gut microbiota, liver metabolome, and mitochondrial function, several limitations should be acknowledged. First, although strong correlations were observed, direct causal evidence linking the altered gut microbiota to downstream hepatic effects remains lacking. Future studies employing fecal microbiota transplantation will be essential to establish causality. Second, the specific bioactive components within CM-exos responsible for the observed benefits have not been identified. Comprehensive omics profiling followed by functional validation of candidate miRNAs or proteins is warranted. Third, the molecular basis for the downregulation of mitochondrial complex I activity, though interpreted as an adaptive response, requires further elucidation at the level of subunit expression, complex assembly, or post-translational modifications. Despite these limitations, our findings reveal a novel and intricate mechanism through which CM-exos exert anti-diabetic effects, positioning them as a promising multi-target therapeutic candidate worthy of further investigation.
5. Conclusion
Our study shows that orally administered CM-exos ameliorate hyperglycemia in diabetic mice through a coordinated gut–liver axis mechanism. We propose that the primary site of action for oral CM-exos is the intestinal microenvironment, where they reshape the gut microbiota, notably reducing the Firmicutes/Bacteroidota ratio. This microbial remodeling is postulated to modulate the production and systemic delivery of key metabolites to the liver. Within the liver, these metabolite alterations—specifically the reduction in galactose-1-phosphate, 2-ketobutyric acid, and D-fructose—initiate a metabolic reprogramming. This reprogramming is characterized by an adaptive downregulation of mitochondrial complex I activity, which likely serves to attenuate excessive ROS generation under diabetic stress. Concurrently, we observed activation of the energy sensor AMPK. The coordinated action of reduced metabolic pressure (from metabolite shifts) and enhanced AMPK signaling subsequently suppresses hepatic gluconeogenesis and promotes glycogen storage, ultimately restoring systemic glucose homeostasis. In contrast, intraperitoneally administered CM-exos, which may bypass the gut, primarily act directly on the liver to suppress gluconeogenesis, resulting in a more limited glycemic improvement. Collectively, our findings delineate a novel mechanism whereby oral CM-exos integrate gut microbiota signaling with hepatic metabolic and energetic regulation, providing a comprehensive theoretical foundation for their use as a multi-targeted antidiabetic agent.Author contributions
Bin Yang: conceptualization, investigation, software, and writing – original draft. Ling Liu: investigation, methodology and data curation. Jingjing Wang, Shifeng Du and Hao Liang: investigation and supervision. Demtu Er: resources and writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Abbreviations
| CM | Camel milk |
| DPP-IV | Dipeptidyl peptidase IV |
| CM-exos | Camel milk exosomes |
| FBG | Fasting blood glucose |
| AMPK | Adenosine monophosphate-activated protein kinase |
| T1D | Type 1 diabetes |
| BCA | Bicinchoninic acid |
| TEM | Transmission electron microscopy |
| NTA | Nanoparticle tracking analysis |
| CD81 | Cluster of differentiation 81 |
| TSG101 | Tumor susceptibility gene 101 |
| WB | Western blotting |
| NC | Normal control |
| HFD | High-fat diet |
| STZ | Streptozotocin |
| DC | Diabetic control |
| Met | Metformin |
| ig-exo/ig_exo | Intragastric administration of CM-exos |
| ip-exo | Intraperitoneal injection of CM-exos |
| BW | Body weight |
| HE | Hematoxylin and eosin |
| PAS | Periodic acid–Schiff |
| OGTT | Oral glucose tolerance test |
| PTT | Pyruvate tolerance test |
| AUC | Area under the curve |
| FINS | Insulin |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
| TG | Triglyceride |
| TC | Total cholesterol |
| LDL-C | Low-density lipoprotein cholesterol |
| HDL-C | High-density lipoprotein cholesterol |
| ROS | Reactive oxygen species |
| NADH | Nicotinamide adenine dinucleotide |
| NDUFS8 | NADH dehydrogenase (ubiquinone) Fe–S protein 8 |
| qRT-PCR | Quantitative real-time PCR |
| MMP | Mitochondrial membrane potential |
| PEPCK | Phosphoenolpyruvate carboxykinase |
| G6PC | Glucose-6-phosphatase |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| TBST | Tris-buffered saline with Tween 20 |
| UHPLC-MS/MS | Ultrahigh performance liquid chromatography and mass spectrometry |
| FA | Formic acid |
| ACN | Acetonitrile |
| PCA | Principal component analysis |
| PCR | Polymerase chain reaction |
| PCoA | Principal coordinate analysis |
| NMDS | Nonmetric multidimensional scaling |
| LDA | Linear discriminant analysis |
| LEfSe | Linear discriminant analysis effect size |
| ANOVA | Analysis of variance |
| SEM | Standard error of the mean |
| G6P | Glucose-6-phosphate |
| NAFLD | Non-alcoholic fatty liver disease |
| SCFAs | Short-chain fatty acids |
| GLP-1 | Glucagon-like peptide-1 |
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information (SI). Supplementary information is available. Fig. S1: MMP was detected using the JC-1 assay. (A) JC-1 assay of AML12 cells in each indicated group. Scale bar, 20 µm. (B) Quantification of MMP by the red/green fluorescence ratio in the JC-1 assay (n = 5). Fig. S2: Box plots of differential metabolites. Table S5: The overlapping differential metabolites in DC vs. NC and ig-exo vs. DC comparisons in the positive mode. Table S6. The overlapping differential metabolites in DC vs. NC and ig-exo vs. DC comparisons in the negative mode. See DOI: https://doi.org/10.1039/d5fo02492g.Acknowledgements
This work was supported by the Program for Improving the Scientific Research Ability of Youth Teachers of Inner Mongolia Agricultural University (Grant No. BR230157), the Plan (Science and Technology Cooperation) Project of 2023 Autonomous Region Major Research and Development and Achievements Transformation, Science and Technology Agency of Inner Mongolia Autonomous Region (Grant No. 2023KJHZ0003), the Open Project of Key Laboratory of Animal Embryo and Development Engineering of Universities of Higher Learning (DWPT-01) and the Natural Science Foundation Project of Inner Mongolia Autonomous Region (Grant No. 2023LHMS03054).References
- K. Chatzianagnostou, M. Gaggini, A. S. Florentin, L. Simonini and C. Vassalle, New Molecules in Type 2 Diabetes: Advancements, Challenges and Future Directions, Int. J. Mol. Sci., 2024, 25, 6218 CrossRef CAS PubMed.
- S. Padhi, A. K. Nayak and A. Behera, Type II diabetes mellitus: a review on recent drug based therapeutics, Biomed. Pharmacother., 2020, 131, 110708 CrossRef CAS PubMed.
- S. Kumar, A. Mittal and D. Babu, Herbal Medicines for Diabetes Management and its Secondary Complications, Curr. Diabetes Rev., 2021, 17, 437–456 CAS.
- A. W. K. Yeung, N. T. Tzvetkov, A. Durazzo, M. Lucarini, E. B. Souto, A. Santini, R. Y. Gan, A. Jozwik, W. Grzybek, J. O. Horbanczuk, A. Mocan, J. Echeverria, D. Wang and A. G. Atanasov, Natural products in diabetes research: quantitative literature analysis, Nat. Prod. Res., 2021, 35, 5813–5827 CrossRef CAS PubMed.
- H. Hussain, F. H. Wattoo, M. H. S. Wattoo, M. Gulfraz, T. Masud, I. Shah, S. Ali and S. E. Alavi, Camel milk as an alternative treatment regimen for diabetes therapy, Food Sci. Nutr., 2021, 9, 1347–1356 CrossRef CAS PubMed.
- W. E. Alhassani, Camel milk: Nutritional composition, therapeutic properties, and benefits for human health, Open Vet. J., 2024, 14, 3164–3180 CrossRef PubMed.
- S. Seyiti, A. Kelimu and G. Yusufu, Bactrian Camel Milk: Chemical Composition, Bioactivities, Processing Techniques, and Economic Potential in China, Molecules, 2024, 29, 4680 CrossRef CAS PubMed.
- S. F. Al Nohair, Medical benefits of camel's milk: A comprehensive review, J. Pak. Med. Assoc., 2021, 71, 933–937 Search PubMed.
- M. A. Ayoub, P. G. Yap, P. Mudgil, F. B. Khan, I. Anwar, K. Muhammad, C. Y. Gan and S. Maqsood, Invited review: Camel milk-derived bioactive peptides and diabetes-Molecular view and perspectives, J. Dairy Sci., 2024, 107, 649–668 CrossRef CAS PubMed.
- Y. Li, W. Zhang, R. Zhao and X. Zhang, Advances in oral peptide drug nanoparticles for diabetes mellitus treatment, Bioact. Mater., 2022, 15, 392–408 CAS.
- M. Rashidi, S. Bijari, A. H. Khazaei, F. Shojaei-Ghahrizjani and L. Rezakhani, The role of milk-derived exosomes in the treatment of diseases, Front. Genet., 2022, 13, 1009338 CrossRef CAS PubMed.
- X. Feng, X. Chen, X. Zheng, H. Zhu, Q. Qi, S. Liu, H. Zhang and J. Che, Latest Trend of Milk Derived Exosomes: Cargos, Functions, and Applications, Front. Nutr., 2021, 8, 747294 CrossRef PubMed.
- M. Jablonska, T. Sawicki, J. Zulewska, K. Staniewska, A. Lobacz and K. E. Przybylowicz, The Role of Bovine Milk-Derived Exosomes in Human Health and Disease, Molecules, 2024, 29, 5835 CrossRef CAS PubMed.
- A. M. Shaban, M. Raslan, S. H. Qahl, K. Elsayed, M. S. Abdelhameed, A. A. A. Oyouni, O. M. Al-Amer, O. Hammouda and M. A. El-Magd, Ameliorative Effects of Camel Milk and Its Exosomes on Diabetic Nephropathy in Rats, Membranes, 2022, 12, 1060 CrossRef CAS PubMed.
- M. C. Petersen, D. F. Vatner and G. I. Shulman, Regulation of hepatic glucose metabolism in health and disease, Nat. Rev. Endocrinol., 2017, 13, 572–587 CrossRef CAS PubMed.
- X. Zhang, S. Yang, J. Chen and Z. Su, Unraveling the Regulation of Hepatic Gluconeogenesis, Front. Endocrinol., 2018, 9, 802 CrossRef.
- Y. Yan, X. E. Zhou, H. E. Xu and K. Melcher, Structure and Physiological Regulation of AMPK, Int. J. Mol. Sci., 2018, 19, 3534 CrossRef PubMed.
- B. Yang, S. Du, L. Liu, J. Wang and D. Er, Camel milk exosomes regulate glucose metabolism by inhibiting mitochondrial complex I in hepatocytes, BMC Vet. Res., 2025, 21, 85 CrossRef CAS PubMed.
- Q. Si, J. Guo, X. Yang, Y. Guo, L. Wu, D. Xie and P. Jiang, Systematic assessment of streptozotocin-induced diabetic metabolic alterations in rats using metabolomics, Front. Endocrinol., 2023, 14, 1107162 CrossRef.
- F. Del Chierico, N. Rapini, A. Deodati, M. C. Matteoli, S. Cianfarani and L. Putignani, Pathophysiology of Type 1 Diabetes and Gut Microbiota Role, Int. J. Mol. Sci., 2022, 23, 14650 CrossRef CAS PubMed.
- H. Han, Y. Li, J. Fang, G. Liu, J. Yin, T. Li and Y. Yin, Gut Microbiota and Type 1 Diabetes, Int. J. Mol. Sci., 2018, 19, 995 CrossRef PubMed.
- Y. Zhang, J. Wang, W. Ge, Y. Song, R. He, Z. Wang and L. Zhao, Camel milk peptides alleviate hyperglycemia by regulating gut microbiota and metabolites in type 2 diabetic mice, Food Res. Int., 2023, 173, 113278 CrossRef CAS PubMed.
- J. He, K. Guo, Q. Chen, Y. Wang and Jirimutu, Camel milk modulates the gut microbiota and has anti-inflammatory effects in a mouse model of colitis, J. Dairy Sci., 2022, 105, 3782–3793 CrossRef CAS PubMed.
- L. Ming, X. Qiao, L. Yi, D. Siren, J. He, L. Hai, F. Guo, Y. Xiao and R. Ji, Camel milk modulates ethanol-induced changes in the gut microbiome and transcriptome in a mouse model of acute alcoholic liver disease, J. Dairy Sci., 2020, 103, 3937–3949 CrossRef CAS PubMed.
- F. B. Khan, M. A. Ansari, S. Uddin, A. R. Palakott, I. Anwar, A. Almatroudi, M. N. Alomary, F. Alrumaihi, F. F. Aba Alkhayl, S. Alghamdi, K. Muhammad, C. Y. Huang, J. R. Daddam, H. Khan, S. Maqsood and M. A. Ayoub, Prospective Role of Bioactive Molecules and Exosomes in the Therapeutic Potential of Camel Milk against Human Diseases: An Updated Perspective, Life, 2022, 12, 990 CrossRef CAS PubMed.
- A. A. Badawy, S. M. El-Hofey, A. M. Shaban, S. E. Orif, Y. Uyanikgil and M. A. El-Magd, Camel milk extracellular vesicles/exosomes: a fascinating frontier in isolation and therapeutic potential, Food Funct., 2025, 16, 344–365 RSC.
- B. L. Furman, Streptozotocin-Induced Diabetic Models in Mice and Rats, Curr. Protoc., 2021, 1, e78 CrossRef CAS PubMed.
- R. Bao, Y. Meng, H. Zhang, C. Yang, W. Li, C. Zhang, J. Zhang, R. Sun, Z. Li, W. Jiang, H. X. Yuan and Y. Dang, Elaiophylin reduces body weight and lowers glucose levels in obese mice by activating AMPK, Cell Death Dis., 2021, 12, 972 CrossRef CAS PubMed.
- S. Meena, Y. S. Rajput, A. K. Pandey, R. Sharma and R. Singh, Camel milk ameliorates hyperglycaemia and oxidative damage in type-1 diabetic experimental rats, J. Dairy Res., 2016, 83, 412–419 CrossRef CAS PubMed.
- M. Gan, X. Chen, Z. Chen, L. Chen, S. Zhang, Y. Zhao, L. Niu, X. Li, L. Shen and L. Zhu, Genistein Alleviates High-Fat Diet-Induced Obesity by Inhibiting the Process of Gluconeogenesis in Mice, Nutrients, 2022, 14, 1551 CrossRef CAS PubMed.
- D. Khedr, M. Hafez, J. Lumpuy-Castillo, S. Emam, A. Abdel-Massih, F. Elmougy, R. Elkaffas, I. Mahillo-Fernandez, O. Lorenzo and N. Musa, Lipid Biomarkers as Predictors of Diastolic Dysfunction in Diabetes with Poor Glycemic Control, Int. J. Mol. Sci., 2020, 21, 5079 CrossRef CAS PubMed.
- C. H. Lin, Y. H. Kuo and C. C. Shih, Antidiabetic and hypolipidemic activities of eburicoic acid, a triterpenoid compound from Antrodia camphorata, by regulation of Akt phosphorylation, gluconeogenesis, and PPARalpha in streptozotocin-induced diabetic mice, RSC Adv., 2018, 8, 20462–20476 RSC.
- N. Khalid, D. N. Abdelrahim, N. Hanach, R. AlKurd, M. Khan, L. Mahrous, H. Radwan, F. Naja, M. Madkour, K. Obaideen, H. Khraiwesh and M. Faris, Effect of camel milk on lipid profile among patients with diabetes: a systematic review, meta-analysis, and meta-regression of randomized controlled trials, BMC Complementary Med. Ther., 2023, 23, 438 CrossRef CAS.
- W. Meng and L. Li, ZHX2 inhibits diabetes-induced liver injury and ferroptosis by epigenetic silence of YTHDF2, Nutr. Diabetes, 2025, 15, 6 CrossRef CAS PubMed.
- G. S. T. Soon and M. Torbenson, The Liver and Glycogen: In Sickness and in Health, Int. J. Mol. Sci., 2023, 24, 6133 CrossRef CAS PubMed.
- S. Murakami, K. Funahashi, N. Tamagawa, M. Ning and T. Ito, Taurine Ameliorates Streptozotocin-Induced Diabetes by Modulating Hepatic Glucose Metabolism and Oxidative Stress in Mice, Metabolites, 2022, 12, 524 CrossRef CAS PubMed.
- E. Trushina, S. Trushin and M. F. Hasan, Mitochondrial complex I as a therapeutic target for Alzheimer's disease, Acta Pharm. Sin. B, 2022, 12, 483–495 CrossRef CAS PubMed.
- M. Alimujiang, X. Y. Yu, M. Y. Yu, W. L. Hou, Z. H. Yan, Y. Yang, Y. Q. Bao and J. Yin, Enhanced liver but not muscle OXPHOS in diabetes and reduced glucose output by complex I inhibition, J. Cell. Mol. Med., 2020, 24, 5758–5771 CrossRef CAS PubMed.
- T. E. LaMoia and G. I. Shulman, Cellular and Molecular Mechanisms of Metformin Action, Endocr. Rev., 2021, 42, 77–96 CrossRef PubMed.
- R. Hosoi, Y. Fujii, O. Hiroyuki, M. Shukuri, S. Nishiyama, M. Kanazawa, K. Todoroki, Y. Arano, T. Sakai, H. Tsukada and O. Inoue, Evaluation of intracellular processes in quinolinic acid-induced brain damage by imaging reactive oxygen species generation and mitochondrial complex I activity, EJNMMI Res., 2021, 11, 99 CrossRef CAS.
- B. Han, L. Zhang, Y. Hou, J. Zhong, K. Hettinga and P. Zhou, Phosphoproteomics reveals that camel and goat milk improve glucose homeostasis in HDF/STZ-induced diabetic rats through activation of hepatic AMPK and GSK3-GYS axis, Food Res. Int., 2022, 157, 111254 CrossRef CAS PubMed.
- M. A. Alshuniaber, G. M. Alshammari, S. M. Eleawa, A. E. A. Yagoub, A. S. Al-Khalifah, M. H. Alhussain, L. N. Al-Harbi and M. A. Yahya, Camel milk protein hydrosylate alleviates hepatic steatosis and hypertension in high fructose-fed rats, Pharm. Biol., 2022, 60, 1137–1147 CrossRef CAS PubMed.
- A. I. Coelho, M. E. Rubio-Gozalbo, J. B. Vicente and I. Rivera, Sweet and sour: an update on classic galactosemia, J. Inherited Metab. Dis., 2017, 40, 325–342 CrossRef CAS PubMed.
- M. Zhang, X. W. Yu, Y. Xu, P. Jouhten, G. V. T. Swapna, R. W. Glaser, J. F. Hunt, G. T. Montelione, H. Maaheimo and T. Szyperski, (13) C metabolic flux profiling of Pichia pastoris grown in aerobic batch cultures on glucose revealed high relative anabolic use of TCA cycle and limited incorporation of provided precursors of branched-chain amino acids, FEBS J., 2017, 284, 3100–3113 CrossRef CAS PubMed.
- X. Bai, R. Pei, W. Lei, M. Zhao, J. Zhang, L. Tian and J. Shang, Antidiabetic Effect of Artemether in Db/Db Mice Involves Regulation of AMPK and PI3K/Akt Pathways, Front. Endocrinol., 2020, 11, 568864 CrossRef PubMed.
- Y. H. Kuo, C. H. Lin, C. C. Shih and C. S. Yang, Antcin K, a Triterpenoid Compound from Antrodia camphorata, Displays Antidiabetic and Antihyperlipidemic Effects via Glucose Transporter 4 and AMP-Activated Protein Kinase Phosphorylation in Muscles, J. Evidence-Based Complementary Altern. Med., 2016, 2016, 4867092 CrossRef PubMed.
- P. Muriel, P. Lopez-Sanchez and E. Ramos-Tovar, Fructose and the Liver, Int. J. Mol. Sci., 2021, 22, 6969 CrossRef CAS PubMed.
- R. Wu, C. Yue, Y. Xie, Y. Fu, X. Yu, Z. Li and B. Yang, Sequencing and analysis of micro RNAs in camel milk exosomes, Acta Vet. Hung., 2023, 82–85 CAS.
- S. Peng, L. Shen, X. Yu, J. Wu, L. Zha, Y. Xia and H. Luo, miR-200a attenuated oxidative stress, inflammation, and apoptosis in dextran sulfate sodium-induced colitis through activation of Nrf2, Front. Immunol., 2023, 14, 1196065 CrossRef CAS PubMed.
- S. Li, C. Shi, H. Wu, H. Yan, M. Xia, H. Jiao, Y. He, M. Zhong, W. Lou, X. Gao, H. Bian and X. Chang, Longitudinal changes of serum metabolomic profile after laparoscopic sleeve gastrectomy in obesity, Endocr. Connect., 2024, 13, e240292 CAS.
- J. Cobb, A. Eckhart, A. Motsinger-Reif, B. Carr, L. Groop and E. Ferrannini, alpha-Hydroxybutyric Acid Is a Selective Metabolite Biomarker of Impaired Glucose Tolerance, Diabetes Care, 2016, 39, 988–995 CrossRef CAS PubMed.
- H. M. Bergman, L. Lindfors, F. Palm, J. Kihlberg and I. Lanekoff, Metabolite aberrations in early diabetes detected in rat kidney using mass spectrometry imaging, Anal. Bioanal. Chem., 2019, 411, 2809–2816 CrossRef CAS PubMed.
- Z. Wang, J. Tang, E. Jin, Y. Zhong, L. Zhang, X. Han, J. Liu, Y. Cheng, J. Hou, X. Shi, H. Qi, T. Qian, L. Yuan, X. Hou, H. Yin, J. Liang, M. Zhao, L. Huang and J. Qu, Serum Untargeted Metabolomics Reveal Potential Biomarkers of Progression of Diabetic Retinopathy in Asians, Front. Mol. Biosci., 2022, 9, 871291 CrossRef CAS PubMed.
- H. Wang, S. Li, C. Wang, Y. Wang, J. Fang and K. Liu, Plasma and Vitreous Metabolomics Profiling of Proliferative Diabetic Retinopathy, Invest. Ophthalmol. Visual Sci., 2022, 63, 17 CrossRef CAS PubMed.
- M. Alvarez-Cordoba, M. Talaveron-Rey, I. Villalon-Garcia, S. Povea-Cabello, J. M. Suarez-Rivero, A. Suarez-Carrillo, M. Munuera-Cabeza, J. J. Salas and J. A. Sanchez-Alcazar, Down regulation of the expression of mitochondrial phosphopantetheinyl-proteins in pantothenate kinase-associated neurodegeneration: pathophysiological consequences and therapeutic perspectives, Orphanet J. Rare Dis., 2021, 16, 201 CrossRef PubMed.
- Q. Yang, L. Deng, C. Feng and J. Wen, Comparing the effects of empagliflozin and liraglutide on lipid metabolism and intestinal microflora in diabetic mice, PeerJ, 2024, 12, e17055 CrossRef PubMed.
- J. Bai, Z. Wan, Y. Zhang, T. Wang, Y. Xue and Q. Peng, Composition and diversity of gut microbiota in diabetic retinopathy, Front. Microbiol., 2022, 13, 926926 CrossRef PubMed.
- X. Peng, Y. Wei, D. Gong and G. Zhang, Modulatory Role of Hesperetin-Copper(II) on Gut Microbiota in Type 2 Diabetes Mellitus Mice, Foods, 2025, 14, 2390 CrossRef CAS.
- A. Bourragat, Q. Escoula, S. Bellenger, O. Zemb, M. Beaumont, K. Chaumonnot, J. P. Farine, E. Jacotot, A. Bonnotte, L. Avoscan, J. Lherminier, K. Luo, M. Narce and J. Bellenger, The transplantation of the gut microbiome of fat-1 mice protects against colonic mucus layer disruption and endoplasmic reticulum stress induced by high fat diet, Gut Microbes, 2024, 16, 2356270 CrossRef PubMed.
- Y. Luo, C. Lan, H. Li, Q. Ouyang, F. Kong, A. Wu, Z. Ren, G. Tian, J. Cai, B. Yu, J. He and A. G. Wright, Rational consideration of Akkermansia muciniphila targeting intestinal health: advantages and challenges, NPJ Biofilms Microbiomes, 2022, 8, 81 CrossRef PubMed.
- Q. Ma, R. Zhai, X. Xie, T. Chen, Z. Zhang, H. Liu, C. Nie, X. Yuan, A. Tu, B. Tian, M. Zhang, Z. Chen and J. Li, Hypoglycemic Effects of Lycium barbarum Polysaccharide in Type 2 Diabetes Mellitus Mice via Modulating Gut Microbiota, Front. Nutr., 2022, 9, 916271 CrossRef PubMed.
- Y. Xie, X. Li, Q. Meng, J. Li, X. Wang, L. Zhu and W. Wang, Interplay between gut microbiota and tryptophan metabolism in type 2 diabetic mice treated with metformin, Microbiol. Spectrum, 2024, 12, e0029124 CrossRef.
- Z. R. Huang, Q. Z. Huang, K. W. Chen, Z. F. Huang, Y. Liu, R. B. Jia and B. Liu, Sanghuangporus vaninii fruit body polysaccharide alleviates hyperglycemia and hyperlipidemia via modulating intestinal microflora in type 2 diabetic mice, Front. Nutr., 2022, 9, 1013466 CrossRef PubMed.
- N. Liu, M. Chen, J. Song, Y. Zhao, P. Gong and X. Chen, Effects of Auricularia auricula Polysaccharides on Gut Microbiota Composition in Type 2 Diabetic Mice, Molecules, 2022, 27, 6061 CrossRef CAS PubMed.
- M. H. Qi, H. Y. Zhang, Y. Y. Hou, I. S. Nguepi Tsopmejio, W. Liu, W. G. Chang, C. Chen, Z. Wang and W. Li, Ginseng-derived GABAFG ameliorates type 2 diabetes mellitus by modulating autophagy-lysosome pathway and gut microbiota, J. Adv. Res., 2025, 465–480 CrossRef CAS PubMed.
- Y. E. Martinez-Lopez, D. Neri-Rosario, D. A. Esquivel-Hernandez, C. Padron-Manrique, A. Vazquez-Jimenez, J. P. Sanchez-Castaneda, D. Giron-Villalobos, C. Mendoza-Ortiz, M. L. Reyes-Escogido, M. L. Evia-Viscarra, A. Aguilar-Garcia, O. Resendis-Antonio and R. Guardado-Mendoza, Effect of metformin and metformin/linagliptin on gut microbiota in patients with prediabetes, Sci. Rep., 2024, 14, 9678 CrossRef.
- N. Chu, J. C. N. Chan and E. Chow, Pharmacomicrobiomics in Western Medicine and Traditional Chinese Medicine in Type 2 Diabetes, Front. Endocrinol., 2022, 13, 857090 CrossRef PubMed.
- Q. Guo, Z. Gao, L. Zhao, H. Wang, Z. Luo, D. Vandeputte, L. He, M. Li, S. Di, Y. Liu, J. Hou, X. Jiang, H. Zhu and X. Tong, Multiomics Analyses With Stool-Type Stratification in Patient Cohorts and Blautia Identification as a Potential Bacterial Modulator in Type 2 Diabetes Mellitus, Diabetes, 2024, 73, 511–527 CrossRef CAS PubMed.
- P. Li, T. Tong, Y. Wu, X. Zhou, M. Zhang, J. Liu, Y. She, Z. Li and Y. Li, The Synergism of Human Lactobacillaceae and Inulin Decrease Hyperglycemia via Regulating the Composition of Gut Microbiota and Metabolic Profiles in db/db Mice, J. Microbiol. Biotechnol., 2023, 33, 1657–1670 CrossRef CAS PubMed.
- J. Siptroth, O. Moskalenko, C. Krumbiegel, J. Ackermann, I. Koch and H. Pospisil, Variation of butyrate production in the gut microbiome in type 2 diabetes patients, Int. Microbiol., 2023, 26, 601–610 CrossRef CAS PubMed.
- C. L. Shen, U. D. Wankhade, K. Shankar, R. S. Najjar, R. G. Feresin, M. M. Elmassry, J. M. Dufour, G. Kaur, S. V. Chintapalli, B. D. Piccolo, D. M. Dunn and J. J. Cao, Effects of Statin and Annatto-extracted Tocotrienol Supplementation on Glucose Homeostasis, Bone Microstructure, and Gut Microbiota Composition in Obese Mice, In Vivo, 2024, 38, 1557–1570 CrossRef CAS PubMed.
- L. Li, Z. Meng, Y. Huang, L. Xu, Q. Chen, D. Qiao and X. Yue, Chronic Sleep Deprivation Causes Anxiety, Depression and Impaired Gut Barrier in Female Mice-Correlation Analysis from Fecal Microbiome and Metabolome, Biomedicines, 2024, 12, 2654 CrossRef CAS PubMed.
- J. Hu, H. Xie, N. Lin and Y. Yang, Penthorum chinense Pursh improves type 2 diabetes mellitus via modulating gut microbiota in db/db mice, BMC Complementary Med. Ther., 2023, 23, 314 CrossRef CAS PubMed.
- P. Portincasa, L. Bonfrate, M. Vacca, M. De Angelis, I. Farella, E. Lanza, M. Khalil, D. Q. Wang, M. Sperandio and A. Di Ciaula, Gut Microbiota and Short Chain Fatty Acids: Implications in Glucose Homeostasis, Int. J. Mol. Sci., 2022, 23, 1105 CrossRef CAS PubMed.
- D. Zhou, Q. Pan, F. Z. Xin, R. N. Zhang, C. X. He, G. Y. Chen, C. Liu, Y. W. Chen and J. G. Fan, Sodium butyrate attenuates high-fat diet-induced steatohepatitis in mice by improving gut microbiota and gastrointestinal barrier, World J. Gastroenterol., 2017, 23, 60–75 CrossRef CAS PubMed.
Footnote |
| † These authors are co-first authors. |
| This journal is © The Royal Society of Chemistry 2026 |