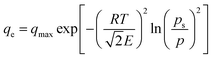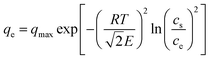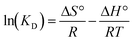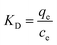Comment on “Shallow Shell SSTA63 resin: a rapid approach to remediation of hazardous nitrate” by E. Çendik, M. Saygı, Y. K. Recepoğlu and Ö. Arar, Environ. Sci.: Water Res. Technol., 2024, 10, 2765
Received
1st December 2024
, Accepted 27th October 2025
First published on 29th October 2025
Abstract
This communication highlights several modeling issues in the paper referenced in the title, including: (1) the improper application of the pseudo-first-order kinetic model, (2) the use of incorrect formulations of the Temkin and Dubinin–Radushkevich isotherm models, and (3) methodological errors in the calculation of thermodynamic parameters. Taken together, these issues call into question the scientific rigor of the study and emphasize the importance of adopting sound modeling practices in sorption research.
Water impact
This comment highlights modeling deficiencies identified in a recent paper on water contaminant sorption. A more rigorous approach to modeling sorption phenomena is essential for ensuring the reliable interpretation of experimental findings. Such improvements will advance the development of efficient sorption processes for water contaminant removal, where precise predictions of sorption behavior are fundamental to success.
|
1. Introduction
In their recent paper, Çendik et al.1 investigated the removal of nitrate ions from water using the commercial anion exchange resin Purolite Shallow Shell™ SSTA63. They analyzed kinetic and equilibrium data from batch experiments, applying the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models, along with the Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich (D–R) isotherms for equilibrium modeling. Thermodynamic parameters were also estimated using data collected at different temperatures. The purpose of this communication is to highlight several important issues in the formulation and application of some of these models. Addressing these points would enhance the scientific reliability of the study's findings. We hope this feedback will be helpful not only to the authors but also to others working with sorption models.
2. Discussion
This section outlines several issues identified in the modeling approaches used in the study. Specifically, concerns were noted with: (1) the PFO kinetic model, (2) the Freundlich isotherm, (3) the Temkin isotherm, (4) the D–R isotherm, and (5) the thermodynamic analysis.
2.1. The PFO kinetic model
The authors applied the linearized form of the PFO kinetic model,2 as given in eqn (1), where qe is the equilibrium solid phase concentration, qt is the solid phase concentration at time t, and k1 is the rate constant. They obtained a poor model fit, as indicated by an R2 value of only 0.623 (Table 1). This poor performance is further illustrated in Fig. 1(a), which compares the experimental kinetic profile with the predictions from the nonlinear form of the PFO model (eqn (2)), using the qe and k1 values reported by the authors (Table 1). The qt-versus-t data in Fig. 1(a) were obtained by converting the original liquid phase concentration-versus-t data using the mass balance equation for batch sorption, based on an adsorbent dosage of 4 g L−1 and an initial nitrate concentration of 100 mg L−1.| | | ln(qe − qt) = ln(qe) − k1t | (1) |
| | | qt = qe[1 − exp(−k1t)] | (2) |
Table 1 Parameter estimates obtained by fitting the PFO model to the sorption data of nitrate ions on Purolite SSTA63 resin
| Source |
Eqn |
k
1 (min−1) |
q
e (mg g−1) |
R
2 (—) |
| Çendik et al.1 |
(1) |
0.097 |
0.3 |
0.623 |
| Present study |
(2) |
0.688 |
24.17 |
1.000 |
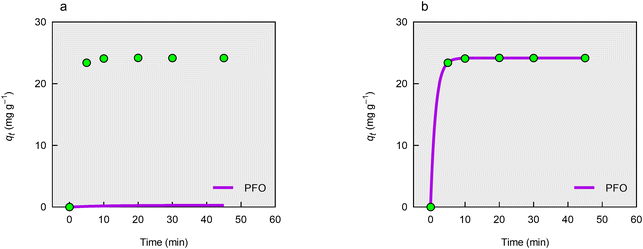 |
| | Fig. 1 Sorption kinetics of nitrate ions on Purolite SSTA63 resin. (a) Kinetic profile predicted by the PFO model using the parameter estimates reported by Çendik et al.1 (b) Kinetic profile predicted by the PFO model using the parameter estimates obtained in this work. | |
In contrast to the linearized form, Fig. 1(b) shows that the nonlinear version of the PFO model (eqn (2)) provides an excellent fit, with an R2 value of 1 (Table 1). The poor performance of the linearized model likely stems from the distribution of the kinetic data. As shown in Fig. 1, most data points were collected near equilibrium, with the dynamic uptake phase largely unsampled. This imbalance limits the linearized model's ability to yield a reliable fit. While the nonlinear model appears highly accurate, this does not necessarily translate to meaningful kinetic insight. Without sufficient data from the early, rapidly changing stage of sorption, even a perfect fit can result in unreliable and misleading parameter estimates.
The following recommendations can help improve the reliability and interpretability of results obtained from the PFO model:
1. Collect sufficient data in the early, ascending portion of the kinetic curve. This initial phase captures the system's transient behavior, where sorption rates are highest and the most valuable kinetic information is available. If this region is under-sampled, the model fit may become dominated by equilibrium data, potentially leading to misleading conclusions about the system's kinetics.
2. Use the nonlinear form of the PFO model (eqn (2)) for data fitting. The nonlinear form preserves the original structure of the model and avoids distortions caused by linearization. Linearized forms often yield biased or unreliable parameter estimates due to uneven data weighting and increased sensitivity to particular data points. Nonlinear regression provides a more statistically sound and robust method for extracting meaningful kinetic parameters.
2.2. The Freundlich isotherm
Eqn (3) presents the Freundlich isotherm3 as used by Çendik et al., where ce is the equilibrium concentration in the fluid phase, and KF and n are adjustable parameters. While the authors reported parameter units for their other isotherm models, they omitted the units for KF. When qe and ce are expressed in mg g−1 and mg L−1, respectively, the correct units for KF are (mg g−1)(L mg−1)1/n, or equivalently, mg1−1/n L1/n g−1. This point is important, as confusion about the correct units of KF has been previously noted in the literature.4
2.3. The Temkin isotherm
The Temkin isotherm used by the authors is given in eqn (4), where R is the universal gas constant, T is the absolute temperature, and AT and bT are adjustable parameters. However, this equation is dimensionally inconsistent: the left-hand side has units of mg g−1, while the right-hand side is unitless. Despite this issue, this misformulated version of the Temkin isotherm is commonly used in sorption studies.4–7| | 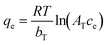 | (4) |
The correct form is shown in eqn (5), where qmax denotes the maximum sorption capacity.8,9 The adjustable parameters in eqn (5) are qmax/bT (not bT alone) and AT. However, the authors reported AT using both L mol−1 and L mg−1, introducing inconsistency and potential confusion. Since ce is consistently expressed in mg L−1 throughout the article, AT should likewise be reported in L mg−1 to ensure dimensional consistency.
| | 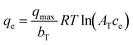 | (5) |
The incorrectly formulated Temkin isotherm (eqn (4)) has been repeatedly presented in several review articles.10,11 These reviews seem to have contributed to the spread of this erroneous form, as researchers, particularly those new to the field, often regard them as authoritative. This situation highlights the need to critically evaluate any isotherm model before using it in research. Researchers should verify both the validity and correct formulation of sorption models by consulting original publications or authoritative texts on sorption theory.12,13 Taking this careful approach not only helps prevent the perpetuation of modeling errors but also strengthens the reliability and scientific integrity of sorption studies.
2.4. The D–R isotherm
The D–R isotherm used by the authors is shown in eqn (6), where E represents the characteristic energy of sorption. However, like the Temkin isotherm in eqn (4), this formulation is dimensionally inconsistent. Specifically, adding the number 1 to 1/ce (which has units of L mg−1) is both mathematically and physically invalid. It is concerning that this flawed form remains widely used in the literature.4–6| | 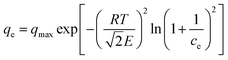 | (6) |
The D–R isotherm was originally developed for gas phase sorption, as expressed in eqn (7), where p and ps are the equilibrium and saturation vapor pressures, respectively.14 For liquid phase sorption, the corresponding form is given by eqn (8), obtained by substituting p and ps in eqn (7) with ce and cs (the solubility limit of the solute).15,16 The 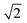 factor introduced by Hobson17 is optional. Although eqn (8) is dimensionally consistent, it is rarely used in water contaminant studies, likely because accurately determining cs is experimentally challenging, as it can vary with pH, temperature, and other factors.
factor introduced by Hobson17 is optional. Although eqn (8) is dimensionally consistent, it is rarely used in water contaminant studies, likely because accurately determining cs is experimentally challenging, as it can vary with pH, temperature, and other factors.
| | 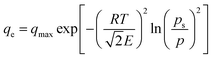 | (7) |
| | 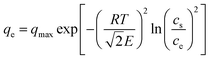 | (8) |
As with the misformulated Temkin isotherm, the dimensionally inconsistent form of the D–R isotherm (eqn (6)) has also appeared in several review articles.10,11 This highlights a broader issue in the field, where reviews, often regarded as authoritative, can inadvertently propagate errors. Researchers should treat isotherm models presented in reviews with caution, verifying their mathematical forms and underlying assumptions against the original sources. This careful validation not only ensures proper model application but also enhances the overall rigor of sorption modeling.
2.5. Thermodynamic calculations
The authors estimated the standard Gibbs energy of sorption (ΔG°), standard enthalpy (ΔH°), and standard entropy (ΔS°) using eqn (9)–(11). In these expressions, KD represents the distribution coefficient. However, KD is not appropriate for use in eqn (10), as it has units of L g−1 when qe and ce are expressed in mg g−1 and mg L−1, respectively. Since the argument of the logarithmic term in eqn (10) must be dimensionless, the correct form involves ln(K°), where K° is the dimensionless thermodynamic equilibrium constant.18| | 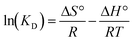 | (10) |
| | 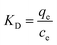 | (11) |
A suitable approach for calculating K° is provided in eqn (12), where bL is the Langmuir equilibrium constant from the expression qe = qmaxbLce/(1 + bLce), MW is the molecular weight of the solute, and cref is the reference concentration, typically taken as 1 mol L−1.19 To ensure K° is dimensionless, bL should be expressed in L mg−1 and MW in mg mol−1.
Conclusions
The issues discussed herein are not confined to the work of Çendik et al.; rather, they are prevalent across the broader literature on the sorption of water contaminants. Misapplications of kinetic and isotherm models, as well as inconsistencies in thermodynamic calculations, can inadvertently compromise the scientific rigor of sorption research. This communication aims to provide constructive feedback, encouraging more careful application of these models. It is hoped that these observations will benefit not only the authors of the study but also others in the field, promoting more accurate and reliable research within the domain of water science and technology.
Author contributions
Khim Hoong Chu: conceptualization, writing – original draft, writing – review & editing.
Conflicts of interest
There are no conflicts of interest to declare.
Data availability
The data analyzed in this comment are available in the publication by Çendik et al.1
References
- E. Çendik, M. Saygı, Y. K. Recepoğlu and Ö. Arar, Shallow Shell SSTA63 resin: A rapid approach to remediation of hazardous nitrate, Environ. Sci.: Water Res. Technol., 2024, 10, 2765–2775 RSC.
- S. Lagergren, Zur Theorie der sogenannten Adsorption gelöster Stoffe, Bihang till Kungliga Svenska Vetenskapsakademiens Handlingar, 1898, 24, 1–39 Search PubMed.
- H. Freundlich, Über die Adsorption in Lösungen, Z. Phys. Chem., 1907, 57U, 385–470 CrossRef.
- J.-C. Bollinger, K. H. Chu and S. Salvestrini, Comments concerning papers about cationic dyes sorption, published by Saeed et al. (2023), Tan et al. (2023) and Yıldız et al. (2023), Sep. Sci. Technol., 2024, 59, 921–928 CrossRef CAS.
- K. H. Chu, M. A. Hashim, G. Hayder and J.-C. Bollinger, Comment on “Highly efficient and sustainable cationic polyvinyl chloride nanofibrous membranes for removal of E. coli and Cr (VI): Filtration and adsorption”, Chem. Eng. J., 2024, 484, 149588 CrossRef CAS.
- K. H. Chu, M. A. Hashim, G. Hayder and J.-C. Bollinger, Rectifying inaccuracies in adsorption modeling. Comment on “amino-and sulfur-containing POSS for highly efficient and selective extraction of platinum group elements from acidic solution”, J. Cleaner Prod., 2024, 464, 142801 CrossRef CAS.
- K. H. Chu, M. A. Hashim and G. Hayder, Comment on “Algal mediated intervention for the retrieval of emerging pollutants from aqueous media”, J. Hazard. Mater., 2024, 465, 133370 CrossRef CAS PubMed.
- M. I. Temkin, The kinetics of some industrial heterogeneous catalytic reactions, Adv. Catal., 1979, 28, 173–291 CAS.
- K. H. Chu, Revisiting the Temkin isotherm: Dimensional inconsistency and approximate forms, Ind. Eng. Chem. Res., 2021, 60, 13140–13147 CrossRef CAS.
- K. Y. Foo and B. H. Hameed, Insights into the modeling of adsorption isotherm systems, Chem. Eng. J., 2010, 156, 2–10 CrossRef CAS.
- M. A. Al-Ghouti and D. A. Da'ana, Guidelines for the use and interpretation of adsorption isotherm models: A review, J. Hazard. Mater., 2020, 393, 122383 CrossRef CAS PubMed.
-
D. M. Ruthven, Principles of Adsorption and Adsorption Processes, John Wiley & Sons, New York, 1984 Search PubMed.
-
D. D. Do, Adsorption Analysis: Equilibria and Kinetics, Imperial College Press, London, 1998 Search PubMed.
- M. M. Dubinin, Fundamentals of the theory of adsorption in micropores of carbon adsorbents: Characteristics of their adsorption properties and microporous structures, Carbon, 1989, 27, 457–467 CrossRef CAS.
- A. Seidel, E. Tzscheutschler, K.-H. Radeke and D. Gelbin, Adsorption equilibria of aqueous phenol and indol solutions on activated carbons, Chem. Eng. Sci., 1985, 40, 215–222 CrossRef CAS.
- K. H. Chu, M. A. Hashim, G. Hayder and J.-C. Bollinger, Comparative evaluation of the Dubinin–Radushkevich isotherm and its variants, Ind. Eng. Chem. Res., 2024, 63, 15002–15011 CrossRef CAS.
- J. P. Hobson, Physical adsorption isotherms extending from ultrahigh vacuum to vapor pressure, J. Phys. Chem., 1969, 73, 2720–2727 CrossRef CAS.
-
P. Atkins, J. De Paula and J. Keeler, Atkins' Physical Chemistry, Oxford University Press, Oxford, 12th edn, 2023 Search PubMed.
- F.-D. Kopinke, Comment on “Adsorption of uranium (VI) complexes with polymer-based spherical activated carbon”, published by Y.-A. Boussouga et al. [Water Research 249 (2024) 120825], Water Res., 2024, 261, 122031 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  *
*

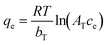
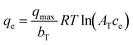
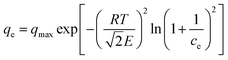
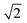 factor introduced by Hobson17 is optional. Although eqn (8) is dimensionally consistent, it is rarely used in water contaminant studies, likely because accurately determining cs is experimentally challenging, as it can vary with pH, temperature, and other factors.
factor introduced by Hobson17 is optional. Although eqn (8) is dimensionally consistent, it is rarely used in water contaminant studies, likely because accurately determining cs is experimentally challenging, as it can vary with pH, temperature, and other factors.