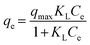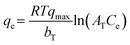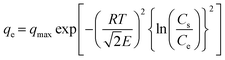Reply to the ‘Comment on “Shallow Shell SSTA63 resin: a rapid approach to remediation of hazardous nitrate”’ by K. H. Chu, Environ. Sci.: Water Res. Technol., 2026, 12, DOI: 10.1039/D4EW00976B
Received
13th June 2025
, Accepted 27th October 2025
First published on 3rd December 2025
Abstract
Corrections are provided for “Shallow Shell SSTA63 resin: a rapid approach to remediation of hazardous nitrate” (Çendik et al., Environ. Sci.: Water Res. Technol., 2024, 10, 2765–2775, https://doi.org/10.1039/D4EW00584H) in response to the comment by K. H. Chu (Environ. Sci.: Water Res. Technol., 2026, 12, https://doi.org/10.1039/D4EW00976B).
Water impact
Our study highlights Purolite Shallow Shell™ SSTA63 resin as a highly effective solution for mitigating nitrate ion (NO3−) contamination in water. Utilizing advanced anion technology that strategically deactivates the gel bead center, the resin shows rapid and efficient removal within 10 minutes and robust regeneration capabilities with hydrochloric acid. Minimal interference from coexisting ions underscores its reliability in diverse environmental conditions, suggesting broad applicability in water treatment and environmental remediation efforts.
|
1. Introduction
In response to the comment by K. H. Chu (https://doi.org/10.1039/D4EW00976B), the authors would like to correct some errors in their original article (https://doi.org/10.1039/D4EW00584H).
The authors regret that there are a few errors in the equation of the Temkin and D–R isotherm models, as well as in calculating the thermodynamic parameters.
3.4. Sorption isotherms
In the initial version of our paper, the Temkin isotherm model was presented using the following equations:| | qe = B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(ATCe) ln(ATCe) | (1) |
However, in these equations, the parameter qmax was inadvertently omitted. The correct form of the Temkin isotherm model1 is shown in eqn (3):| | 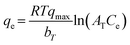 | (3) |
In the Temkin model, qe (mg g−1) represents the resin capacity at equilibrium and qmax (mg g−1) is the maximum capacity of the resin, while R is the universal gas constant (J mol−1 K−1), T is the absolute temperature (K), and bT (J mol−1) is the Temkin constant related to the heat of adsorption.
The calculations for the Dubinin–Radushkevich (D–R) model were originally presented using the following equations:
| | qe = qmax![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−βε2) exp(−βε2) | (4) |
| | 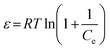 | (5) |
| | 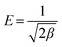 | (6) |
However, in
eqn (5), the term (1 + 1/
Ce) was incorrectly given. The correct expression should be
Cs/
Ce.
The correct form of the D–R model2 is shown in eqn (7).
| | 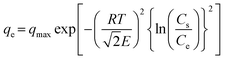 | (7) |
In this equation,
Cs is the solute solubility of NaNO
3, which is 11.327 mol kg
−1 water at 30 °C.
3 This value was converted to 699
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
476 mg NO
3− per L by calculating mol L
−1 using the density of water at 30 °C (0.99565 g cm
−3).
4E (kJ mol
−1) is the mean free energy of adsorption per mole of solute.
The calculated qmax values from the Temkin model were significantly lower than the experimental findings (Table 1). This discrepancy may indicate that the Temkin model is not suitable for high solute concentration.1 In contrast, the DR isotherm yielded a higher qmax value, which could be attributed to the high Cs value.
Table 1 Isotherm models with associated parameters and values for the sorption of NO3− on Purolite SSTA63 anion exchange resin
| Isotherm model |
Nonlinear equations |
Parameters |
Values |
| Langmuir |
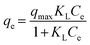
|
q
max (mg g−1) |
53.65 |
|
K
L (L mg−1) |
0.069 |
|
R
2
|
0.99 |
| Freundlich |
q
e = KFC1/ne |
K
F (mg g−1)(L mg−1)1/n |
6.38 |
|
n
|
2.38 |
|
R
2
|
0.95 |
| Temkin |
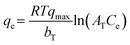
|
b
T (J mol−1) |
8.88 |
|
A
T (L mg−1) |
1.26 |
|
q
max (mg g−1) |
0.03 |
|
R
2
|
0.92 |
| D–R |
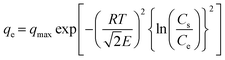
|
E (kJ mol−1) |
12.05 |
|
q
max (mg g−1) |
205.08 |
|
R
2
|
0.96 |
3.5. Sorption thermodynamics
In the initial version of the paper, the thermodynamic calculations were performed using the following eqn (8):However, in the corrected form, the distribution coefficient (KD) has been replaced with the equilibrium constant (K0e), as shown in eqn (9).
The nature and thermodynamic feasibility of the sorption process were assessed by analyzing the thermodynamic constants, including the standard free energy (ΔG°, kJ mol−1), standard enthalpy (ΔH°, kJ mol−1), and standard entropy (ΔS°, kJ mol−1 K−1), using eqn (9)–(12):5
| | 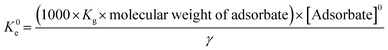 | (9) |
| | ΔG° = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(K0e) ln(K0e) | (10) |
| | 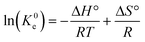 | (12) |
K0e is the thermodynamic equilibrium constant,
γ is the coefficient of activity, [Adsorbate]
0 is the standard concentration of the adsorbate (1 mol L
−1),
Kg is the Langmuir equilibrium constant (obtained from sorption isotherm tests), and Δ
G° is the Gibbs free energy change (kJ mol
−1). Δ
H° (kJ mol
−1) and Δ
S° (J mol
−1 K
−1) are the enthalpy change and entropy change, respectively. For this calculation, the adsorbate solution is significantly diluted; therefore, one can consider the activity coefficient as unitary.
6
Fig. 1 is the plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0evs. 1/T used to estimate the ΔH° and ΔS° values for the sorption of NO3− using Purolite SSTA63 resin from the slope and intercept, respectively. The summarized values of the parameters are presented in Table 2. The sorption process exhibits spontaneity, as evidenced by the negative ΔG° value at all studied temperatures. The negative ΔH° value (−15.3 kJ mol−1) indicates an exothermic interaction between NO3− and the resin. Furthermore, the positive ΔS° value (+19.1 J mol−1 K−1) indicates the affinity of the sorbent for NO3− as randomness increased in the sorption process.
K0evs. 1/T used to estimate the ΔH° and ΔS° values for the sorption of NO3− using Purolite SSTA63 resin from the slope and intercept, respectively. The summarized values of the parameters are presented in Table 2. The sorption process exhibits spontaneity, as evidenced by the negative ΔG° value at all studied temperatures. The negative ΔH° value (−15.3 kJ mol−1) indicates an exothermic interaction between NO3− and the resin. Furthermore, the positive ΔS° value (+19.1 J mol−1 K−1) indicates the affinity of the sorbent for NO3− as randomness increased in the sorption process.
 |
| | Fig. 1 ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0evs. 1/T plot for sorption thermodynamics of NO3− onto Purolite SSTA63 anion exchange resin. K0evs. 1/T plot for sorption thermodynamics of NO3− onto Purolite SSTA63 anion exchange resin. | |
Table 2 Thermodynamic parameters for the sorption of NO3− onto Purolite SSTA63 anion exchange resin
| Temperature (°C) |
ΔG° (kJmol−1) |
ΔH° (kJ mol−1) |
ΔS° (J mol−1 K−1) |
| 15 |
−20.8 |
−15.3 |
+19.1 |
| 20 |
−20.9 |
| 30 |
−21.1 |
The corrections made to the Temkin and Dubinin–Radushkevich isotherm models, as well as to the thermodynamic parameters, provide a more accurate description of the sorption process. Although the corrected equations lead to changes in the calculated parameter values, they do not affect the overall interpretation of the sorption behavior. The main conclusions regarding the efficiency of NO3− removal by Purolite Shallow Shell™ SSTA63 anion exchange resin and the underlying sorption mechanism remain valid.
Author contributions
Elif Çendik: funding acquisition, investigation. Mügenur Saygı: investigation. Yaşar Kemal Recepoğlu: writing – review & editing, conceptualization, visualization. Özgür Arar: writing – review &editing, supervision, resources, investigation.
Conflicts of interest
There are no conflicts to declare.
Data availability
Data will be available upon request.
Acknowledgements
We thank Purolite Int. Co. (Türkiye) for providing ion-exchange resin samples.
References
- K. H. Chu, Revisiting the Temkin isotherm: dimensional inconsistency and approximate forms, Ind. Eng. Chem. Res., 2021, 60, 13140–13147 CrossRef CAS.
- K. H. Chu, M. A. Hashim, G. Hayder and J.-C. Bollinger, Comparative Evaluation of the Dubinin–Radushkevich Isotherm and Its Variants, Ind. Eng. Chem. Res., 2024, 63, 15002–15011 CrossRef CAS.
- D. G. Archer, Thermodynamic properties of the NaNO3+ H2O system, J. Phys. Chem. Ref. Data, 2000, 29, 1141–1156 CrossRef CAS.
-
W. M. Haynes, CRC handbook of chemistry and physics, CRC press, 2014 Search PubMed.
- E. C. Lima, A. Hosseini-Bandegharaei, J. C. Moreno-Piraján and I. Anastopoulos, A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van't Hoof equation for calculation of thermodynamic parameters of adsorption, J. Mol. Liq., 2019, 273, 425–434 CrossRef CAS.
- I. Anastopoulos, I. Pashalidis, A. Hosseini-Bandegharaei, D. A. Giannakoudakis, A. Robalds, M. Usman, L. B. Escudero, Y. Zhou, J. C. Colmenares and A. Núñez-Delgado, Agricultural biomass/waste as adsorbents for toxic metal decontamination of aqueous solutions, J. Mol. Liq., 2019, 295, 111684 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  b and
Özgür
Arar
b and
Özgür
Arar
 *a
*a
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(ATCe)
ln(ATCe)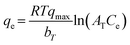
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−βε2)
exp(−βε2)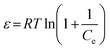
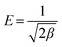
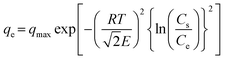
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 476 mg NO3− per L by calculating mol L−1 using the density of water at 30 °C (0.99565 g cm−3).4E (kJ mol−1) is the mean free energy of adsorption per mole of solute.
476 mg NO3− per L by calculating mol L−1 using the density of water at 30 °C (0.99565 g cm−3).4E (kJ mol−1) is the mean free energy of adsorption per mole of solute.
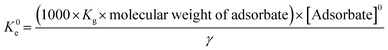
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(K0e)
ln(K0e)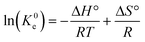
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0evs. 1/T used to estimate the ΔH° and ΔS° values for the sorption of NO3− using Purolite SSTA63 resin from the slope and intercept, respectively. The summarized values of the parameters are presented in Table 2. The sorption process exhibits spontaneity, as evidenced by the negative ΔG° value at all studied temperatures. The negative ΔH° value (−15.3 kJ mol−1) indicates an exothermic interaction between NO3− and the resin. Furthermore, the positive ΔS° value (+19.1 J mol−1 K−1) indicates the affinity of the sorbent for NO3− as randomness increased in the sorption process.
K0evs. 1/T used to estimate the ΔH° and ΔS° values for the sorption of NO3− using Purolite SSTA63 resin from the slope and intercept, respectively. The summarized values of the parameters are presented in Table 2. The sorption process exhibits spontaneity, as evidenced by the negative ΔG° value at all studied temperatures. The negative ΔH° value (−15.3 kJ mol−1) indicates an exothermic interaction between NO3− and the resin. Furthermore, the positive ΔS° value (+19.1 J mol−1 K−1) indicates the affinity of the sorbent for NO3− as randomness increased in the sorption process.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K0evs. 1/T plot for sorption thermodynamics of NO3− onto Purolite SSTA63 anion exchange resin.
K0evs. 1/T plot for sorption thermodynamics of NO3− onto Purolite SSTA63 anion exchange resin.