Molecular-scale investigation of Cu(II) interactions with synthetic and natural zeolites during removal and recovery
Case M.
van Genuchten
 *,
Kaifeng
Wang
,
Claus
Kjøller
and
Knud
Dideriksen
*,
Kaifeng
Wang
,
Claus
Kjøller
and
Knud
Dideriksen

Department of Geochemistry, Geological Survey of Denmark and Greenland (GEUS), Øster Voldgade 10, Copenhagen, Denmark. E-mail: cvg@geus.dk
First published on 16th December 2025
Abstract
Copper (Cu) is simultaneously an environmental pollutant present in industrially relevant waters, including geothermal fluids, and a strategic raw material (SRM) in the European Union, which highlights the value of technologies that couple its removal and recovery. In this work, we investigated the uptake and subsequent recovery of Cu(II) using two zeolites with distinct structures: synthetic faujasite and natural clinoptilolite. Batch Cu(II) adsorption isotherms (0.001 to >1 mM Cu(II)), kinetic Cu(II) uptake measurements, and acidic zeolite regeneration experiments were combined with molecular-scale solid-phase characterization by synchrotron-based X-ray diffraction and Cu K-edge X-ray absorption spectroscopy. Our results revealed significant differences in Cu(II) uptake, extractability and solid-phase speciation depending on zeolite structure. With respect to Cu(II) uptake, synthetic faujasite outperformed natural clinoptilolite, removing more Cu(II) per zeolite mass with faster uptake kinetics. The characterization data indicated synthetic faujasite removed Cu(II) primarily via monomeric adsorption (i.e., outer- and inner-sphere complexes), whereas Cu-loaded natural clinoptilolite contained a mixture of monomeric and polymeric Cu (i.e., Cu–Cu bonding was detected). Multiple acidic regeneration cycles of synthetic faujasite was highly effective (>95% Cu(II) extracted) using 0.01 M HCl, with higher HCl concentrations destabilizing the faujasite structure. By contrast, 0.1 M HCl was required to extract Cu(II) efficiently from natural clinoptilolite, with minimal impact on zeolite structure. Taken together, these macroscopic and molecular-scale results provide critical information to optimize the deployment of zeolite-based filters for holistic Cu(II) removal and recovery from aqueous solution.
Water impactThis study demonstrates how zeolite structure controls copper removal and recovery from water. Copper removal and recovery performance are linked with molecular-scale mechanistic data, resulting in proposed improvements to the design of reusable filters that can both clean industrial wastewater and recover copper as a valuable resource. |
1. Introduction
The presence of aqueous divalent copper, Cu(II), in water sources is both an environmental issue due to its aquatic ecotoxicity1 and a growing concern for many industries.2 A pertinent example of the industrial challenges arising from Cu(II) is found in geothermal energy production, which is a sector that is increasingly relied on to transition from fossil fuels.3 High salinity geothermal fluids can contain relatively high concentrations of aqueous Cu(II). Upon production, the Cu(II) can be reduced to particulate Cu(0) metal through redox reaction with iron (Fe(0)) metal in well liners, leading to well clogging.4,5 Therefore, Cu(II) removal from geothermal fluids can help eliminate major inefficiencies in geothermal energy production due to Cu-bearing scale formation and galvanic corrosion. In addition to these industrial challenges, aqueous Cu(II) also represents an opportunity for resource recovery. The EU has classified Cu as a strategic raw material (SRM) due to its critical importance to many key sectors, including defence, energy, mobility and aerospace.6 Therefore, the removal of problematic aqueous Cu(II) from targeted water sources would not only have industrial and environmental benefits, but can also be coordinated with Cu recovery in holistic systems. Such an approach that couples Cu(II) treatment with recovery is also consistent the Critical Raw Materials Act adopted by the EU in 2024, which sets benchmarks for the domestic production of SRMs derived from secondary sources, such as wastewater.7The application of mineral adsorbents that can be regenerated is a promising approach for Cu(II) removal and recovery, with zeolite minerals having particularly relevant properties. Zeolites include a wide range synthetic and natural aluminosilicate solids that have a microporous structure and variable composition,8 with the general formula [(M1)a(M2)d(Al(a+2d))Sin−(a+2d)O2n]·mH2O. Here, M1 and M2 represent monovalent and divalent cations, respectively, which compensate for the negative charge of the Al–Si framework. Zeolites have a high adsorption capacity because M1 and M2 cations can undergo ion exchange and because of abundant external and internal surfaces, which are terminated by silica (SiO4) and alumina (AlO4) tetrahedra. In addition, zeolites typically have low points of zero charge (PZCs < 3), leading to net negative surface charges even at the mildly acidic pH levels of geothermal fluid.9 These unique surface properties make synthetic and natural zeolites excellent candidates for metal cation removal from various water sources, including geothermal fluid.
Despite the potential of zeolites to be deployed for Cu(II) treatment and recovery, key fundamental questions regarding Cu(II) adsorption and extraction from zeolites remain, particularly related to solid-phase Cu(II) speciation and its relationship with zeolite structure. For example, the formation of both outer-sphere Cu(II) complexes (i.e., Cu adsorption through H-bonding and electrostatic interactions) and inner-sphere Cu(II) complexes (i.e., Cu linked directly to zeolite surfaces) on clinoptilolite have been reported, with the fraction of inner-sphere complexes increasing with Cu(II) loading.10 However, the presence of both inner-sphere Cu(II) complexes and multinuclear Cu–Cu polymeric species has also been detected for Cu-exchanged faujasite using Cu K-edge EXAFS spectroscopy.11 Evidently, Cu(II) can be taken up by zeolites via several mechanisms (i.e., outer-sphere and inner-sphere complexes, as well as polymeric Cu clusters) depending on zeolite structure and chemical conditions. Knowledge of the Cu(II) uptake mode is important partly because each Cu(II) species likely exhibits different desorption properties depending on the recovery method.
One recovery method often employed to regenerate mineral adsorbents and extract metal cations is by washing the adsorbent with acid to decrease pH. This process promotes H+ substitution for the adsorbed cation and, if pH decreases to below the PZC, leads to increased electrostatic repulsion between the cation and particle surface.12,13 Acidic regeneration of Cu(II)-bearing zeolites would thus yield a Cu(II)-rich extraction solution that can be subsequently refined via controlled electrochemical Cu(II) deposition to form valuable Cu(0) end-products.14 Effective acidic extraction of Cu(II) from natural clinoptilolite has been reported in previous studies, with the fraction of desorbed Cu(II) increasing with decreasing pH.15–18 However, this previous work was limited to a relatively narrow range of acid concentrations with little attention given to changes in the structure and ensuing adsorption reactivity of the zeolite. Although decreasing solution pH can regenerate adsorbents, acidic conditions can also alter the structure of the adsorbent material, modifying metal adsorption reactivity. For example, repeated exposures to widely varied pH have been shown to increase zeolite weathering, thus altering external and internal surface area.19 Similar hypothetical structural modifications induced by acidic Cu(II) recovery might result in an undesirable decrease in subsequent Cu(II) uptake, limiting zeolite reuse. However, the relationship between acidic regeneration, zeolite structure and changes to Cu(II) adsorption reactivity has not been investigated systematically.
In this work, we compared the removal and recovery of aqueous Cu(II) using two zeolites, synthetic faujasite and natural clinoptilolite. These zeolites were selected to encompass a variety of zeolite structure, composition and impurity content, as well as zeolite source. Specifically, we investigated Cu(II) uptake by synthetic faujasite and natural clinoptilolite across several variables, including pH, time and co-occurring NaCl and CaCl2 concentrations that approach the salinities of some geothermal fluids. In addition, the recovery of adsorbed Cu(II) and alterations to zeolite structural properties and Cu(II) adsorption reactivity were tracked during successive cycles of acidic regeneration using multiple HCl concentrations. Molecular-scale X-ray characterization techniques, including Cu K-edge EXAFS spectroscopy and synchrotron X-ray diffraction (XRD), were applied to identify the solid-phase Cu(II) speciation and zeolite structure of the end-products of the adsorption and regeneration experiments. Our study reveals key differences in Cu(II) uptake and extraction depending on zeolite structure, which can be used to inform the design of holistic zeolite-based treatment systems.
2. Materials and methods
2.1 Materials
Stock solutions of reagent grade Cu(SO4)2·4H2O, CaCl2 and NaCl were prepared with 18 MΩ cm deionized (DI) water. The stock solutions were adjusted with HNO3 or NaOH to pH 5 prior to use in experiments. Two different zeolites were used in this study: natural clinoptilolite and synthetic faujasite. The natural zeolite was derived from the Nizny Hrabovec quarry site in eastern Slovakia. This sample was obtained from the Zeocem company and sieved to a particle size of <20 μm prior to use. The material has been characterized previously,20 but additional X-ray diffraction (XRD) and scanning electron microscopy (SEM–EDS) data are given in the SI. We refer to this zeolite as natural clinoptilolite, but note that the material contains traces of quartz and carbonate-containing phases (3.8 ± 0.8% C; Fig. S1). The specific surface area of the natural clinoptilolite was determined by the Brunauer–Emmett–Teller (BET) method to be 30–40 m2 g−1. The dominant micropores of clinoptilolite have been reported to be approximately 0.4 nm in size, while the main pore size in the mesopore range is centered at 2.16 nm.21 The second zeolite was synthetic faujasite, zeolite Y–Na, which was purchased from Merck and used directly. Synthesized faujasite exhibits a much higher BET specific surface area (>300 m2 g−1) and a well-defined porous structure consisting of micropores with average diameters in the range of 1.1–1.5 nm, together with mesopores having a characteristic pore diameter of about 8.5 nm.22 Additional structural data (XRD and SEM–EDS) for this zeolite are given in the SI.2.2 Batch adsorption isotherms
Adsorption isotherms were performed by adding defined volumes of Cu(II) and NaCl stock solutions to faujasite or clinoptilolite suspensions, adjusting pH to 5 or 6 with dilute HCl or NaOH, and adding DI water to yield the desired initial conditions (typically from 0.001 to 3 mM Cu(II), 0.1 M NaCl, 5 g L−1 zeolites, 40 mL total volume). Saturation indices for Cu-bearing solids in these conditions were calculated by PHREEQC,23 with details of these calculations given in Section S1 of the SI. We selected a zeolite solids concentration of 5 g L−1 to ensure adequate mass was available for subsequent synchrotron characterization. After adding all components, the suspensions were sealed in air-tight reactors and mixed end-over-end for 14 d. Variations in pH during the experiment, which were most pronounced at early stages of the reaction (<1 d), were controlled by opening the reactors, measuring pH and adjusting (if needed) by manual additions of dilute HCl or NaOH. Following the 14 d reaction, the suspension was centrifuged at 4000 rpm for 20 min. An aliquot of the supernatant solution of the centrifuged suspension was passed through 0.2 μm syringe filters and immediately acidified with HNO3 for measurements of dissolved Cu, Al, Si, Ca, Mg and other ions by inductively-coupled plasma optical emission spectrometry (ICP-OES; Perkin Elmer Avio 550 Max). From selected results of the adsorption experiments, we optimized a model for cation exchange in PHREEQC, the details of which are given in Fig. S2 and Section S1 of the SI. The solids remaining after centrifugation were reserved for synchrotron-based characterization. We report the uptake of Cu(II) in μmol g−1, which represents the difference between the initial and final dissolved Cu(II) concentration normalized by the zeolite concentration. For the batch adsorption isotherms, as well as all subsequent experiments, data are given as the average and standard deviation of replicate experiments, with solid-phase characterization performed on one of the replicates.2.3 Adsorption kinetics and impact of ionic composition
For a subset of adsorption conditions, a series of experiments were performed to evaluate adsorption kinetics and the impact of solution composition on Cu(II) uptake. We selected the initial conditions of 0.1 mM Cu(II), pH 6, 0.1 M NaCl and 5 g L−1 zeolites for the kinetic experiments because the adsorption isotherms showed that pH adjustment was minimal for these conditions, which facilitates sample collection at very short timescales. Sample collection procedures matched the adsorption isotherms with a few exceptions. To facilitate sampling at early time points (<1 min), the suspensions were sealed and mixed by hand immediately after adding Cu(II) and were not centrifuged prior to syringe filtration. The reported sampling times refer to the time at which aliquots were removed from the suspension for filtration, though filtering required only a few seconds.The impact of solution composition, particularly the concentrations of NaCl and a common competing bivalent cation in geothermal fluid, CaCl2, was investigated using initial conditions of 0.1 mM Cu(II), pH 6, and 5 g L−1 of zeolites. The NaCl concentration was systematically varied in separate experiments from 0.01 to 1.0 M, whereas the CaCl2 concentration ranged from 0.01 to 0.03 M (the CaCl2 experiments also contained 0.1 M NaCl). These NaCl and CaCl2 concentrations were selected to span the Na/Cu (10000) and Ca/Cu (300) mol ratios typical of geothermal fluid. The pH of these experiments was monitored and adjusted, if needed, and the reaction time was 1 d.
2.4 Cu(II) extraction and Cu(II) uptake over consecutive acidic zeolite regeneration cycles
The recovery of Cu(II) from the synthetic and natural zeolites was investigated over three consecutive cycles of Cu(II) uptake and acidic extraction using either 0.01 or 0.1 M HCl (experimental design given in Fig. S3). For these experiments, the initial solids were generated using similar protocols as the adsorption isotherms (0.6 mM Cu(II), 0.1 M NaCl, pH 6, 5 g L−1 zeolites, 1 d reaction time). Following centrifugation of the initial suspension, the Cu(II)-loaded zeolites were resuspended in 40 mL of the acidic extraction solution, yielding 5 g L−1 zeolite, and mixed end-over-end for 30 min, which mimics the rapid washing of zeolite-based filters expected for on-site regeneration. Next, the acidic zeolite suspension was centrifuged, with an aliquot of the supernatant passed through 0.2 μm filters for ICP-OES measurements of extracted Cu(II) (and other ions). The regenerated zeolites were then resuspended in 40 mL of solution with identical composition as the initial sample (0.6 mM Cu(II), pH 6, 0.1 M NaCl), mixed end-over-end for 1 d, and centrifuged again for measurements of Cu(II) uptake. This cycle of Cu(II) uptake and subsequent acidic Cu(II) extraction was repeated three times using the same solids, with ICP-OES measurements of the filtered supernatant solution performed at each step and the final solids retained for solid-phase characterization. Notably, after each centrifugation step, the turbidity was measured to ensure zeolite solids were not discarded with the supernatant solution. If the turbidity exceeded 10 NTU for any supernatant solution, which was the case for the regeneration of synthetic faujasite with 0.1 M HCl, the experiment was terminated.2.5 Synchrotron-based X-ray characterization
Synchrotron X-ray diffraction (XRD) data were collected at the Balder beam line for a subset of zeolite samples, with the specific objective to investigate the impact of acidic zeolite regeneration on crystal structure. The Balder end-station is fashioned with multimodal XAS–XRD capabilities, whereby XRD data can be collected on identical samples as XAS data with minimal down time and no sample reconfiguration. The XRD data were collected using a Dectris 2-D Eiger 1 M detector that was mounted to a robot arm to facilitate rapid positioning during the experiment. The X-ray energy for XRD data collection was 10 keV and data collection time was <5 s per sample. Calibration and integration of raw images to plots of intensity versus scattering vector were performed using custom python script available at the beam line. Data are reported with the x-axis in Q-space. Additional synchrotron XRD data for a subset of Cu(II) adsorption samples were also collected at the European Synchrotron Radiation Facility (ESRF; Grenoble, FR). Details on the collection and processing of these data are described in Section S2 of the SI.
3. Results and discussion
3.1 Adsorption isotherms
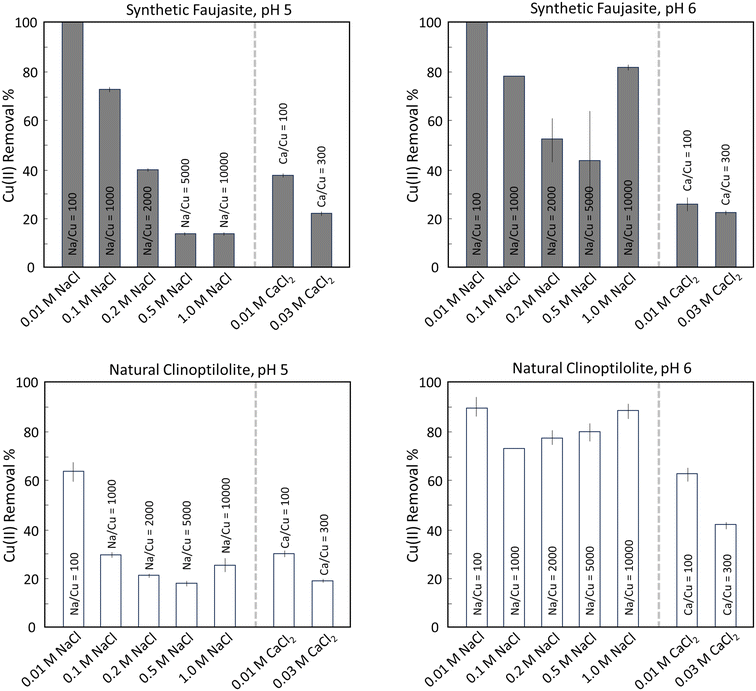
Fig. 1 shows the Cu adsorption isotherms for synthetic faujasite and natural clinoptilolite (solids concentration of 5 g L−1) at pH 5 and 6. For pH 5 data, we have fitted Langmuir isotherms to the experimental uptake data. The Cu(II) adsorption isotherms indicated several trends in Cu(II) interactions with the synthetic and natural zeolites. The most pronounced trend was that Cu(II) uptake was substantially higher for synthetic faujasite than natural clinoptilolite when normalized by mass of zeolite added, especially at pH 5. For example, at the highest initial Cu(II) concentration of 3 mM for experiments at pH 5, the solid-phase Cu(II) content for faujasite reached nearly 300 μmol g−1, which was ∼5 times higher than Cu(II) uptake by clinoptilolite in identical conditions. Similarly, the fitted maximum adsorption (Gmax) and the adsorption equilibrium constant (Kads) are 3–4 times higher for the faujasite data (Gmax = 4.7 × 102; Kads = 1.0 × 10−3) compared to the clinoptilolite data (Gmax = 1.3 × 102; Kads = 3.2 × 10−4). This enhanced Cu(II) uptake for faujasite is consistent with its higher specific surface area (SSA; >300 m2 g−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) compared to natural clinoptilolite (30–40 m2 g−1). The uptake of Cu(II) was also found to increase with pH for both zeolites, which is consistent with a decrease in net particle surface charge at elevated pH, but the impact of pH on Cu(II) adsorption was lower for faujasite than for clinoptilolite. For example, at equivalent initial Cu(II) concentrations of 0.3 mM, Cu(II) uptake by clinoptilolite at pH 6 was 39 μmol g−1 (>60% Cu(II) removal), which was more than 3 times higher than the 12 μmol g−1 measured at pH 5 (<25% Cu(II) removal). Finally, it is important to note here that the aqueous Cu(II) concentrations for the pH 6 adsorption isotherms often exceeded the calculated solubility for atacamite (Cu2Cl(OH)3), which is the least soluble Cu(II) compound in these conditions identified by thermodynamic calculations (Section S1). Thus, the highest initial Cu(II) concentrations are only plotted in Fig. 1 for the pH 5 experiments, where calculated atacamite solubility is larger (∼1.6 mM).
30) compared to natural clinoptilolite (30–40 m2 g−1). The uptake of Cu(II) was also found to increase with pH for both zeolites, which is consistent with a decrease in net particle surface charge at elevated pH, but the impact of pH on Cu(II) adsorption was lower for faujasite than for clinoptilolite. For example, at equivalent initial Cu(II) concentrations of 0.3 mM, Cu(II) uptake by clinoptilolite at pH 6 was 39 μmol g−1 (>60% Cu(II) removal), which was more than 3 times higher than the 12 μmol g−1 measured at pH 5 (<25% Cu(II) removal). Finally, it is important to note here that the aqueous Cu(II) concentrations for the pH 6 adsorption isotherms often exceeded the calculated solubility for atacamite (Cu2Cl(OH)3), which is the least soluble Cu(II) compound in these conditions identified by thermodynamic calculations (Section S1). Thus, the highest initial Cu(II) concentrations are only plotted in Fig. 1 for the pH 5 experiments, where calculated atacamite solubility is larger (∼1.6 mM).
3.2 Kinetics of Cu(II) adsorption
The kinetics of Cu(II) adsorption to synthetic faujasite and natural clinoptilolite are shown in Fig. 2. For synthetic faujasite, Cu(II) was taken up rapidly, with over 70% of the 0.1 mM initial aqueous Cu(II) removed in <5 min. The uptake of Cu(II) did not increase substantially in the next 4 hours of reaction, remaining below 80%. The removal of Cu(II) by natural clinoptilolite increased gradually with reaction time and was generally slower compared to synthetic faujasite. At the shortest timescales (i.e., <1 h), Cu(II) uptake increased continuously from nearly 45% to ∼60%, which contrasts the behaviour of rapid Cu(II) uptake by synthetic faujasite over this timescale. The removal of Cu(II) remained near 60% beyond 1 hour of reaction for clinoptilolite, which was comparatively less than the 70% Cu(II) removal with faujasite over the same timescale.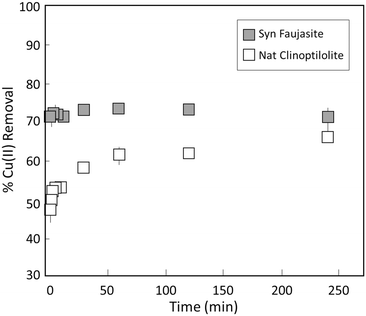 | ||
| Fig. 2 Kinetics of Cu(II) uptake by synthetic faujasite (grey squares) and natural clinoptilolite (white squares). Experiments were performed using 0.1 mM Cu(II) at pH 6. | ||
3.3 Impact of ionic composition on Cu(II) uptake
For synthetic faujasite, increasing the concentrations of NaCl (0.01 to 1.0 M) and CaCl2 (0.01 to 0.03 M) significantly decreased Cu(II) uptake, especially at the lower pH of 5 (Fig. 3). For example, at the lowest NaCl concentration of 0.01 M, essentially complete Cu(II) removal was measured at both pH 5 and pH 6 using synthetic faujasite. However, increasing the NaCl concentration to 1.0 M resulted in a systematic decrease in Cu(II) uptake at pH 5, from >95% (0.01 M) to <15% at 0.5 and 1.0 M. A similar decrease in Cu(II) uptake was observed at pH 6 with increasing NaCl concentrations up to 0.5 M, but further increasing the NaCl concentration to 1.0 M resulted in higher Cu(II) removal, which is attributed to the formation of Cu-bearing chloride solids.31,32 In fact, solutions are predicted to be close to saturation with respect to atacamite for NaCl concentrations >0.2 M in the pH 6 experiments, suggesting that the observed differences between pH 5 and 6 experiments may reflect atacamite precipitation.33 The presence of high CaCl2 concentrations, which is common in geothermal brines, also decreased the Cu(II) removal efficiency of synthetic faujasite at both pH 5 and 6. For example, whereas >95% Cu(II) was removed in the absence of CaCl2 at both pH 5 and 6, <25% was removed in the presence of 0.03 M CaCl2 at both pH values.For natural clinoptilolite, the trends in Cu(II) removal in the presence of NaCl and CaCl2 were similar to those observed for synthetic faujasite. However, the effect of co-occurring NaCl and CaCl2 was less pronounced. The most effective Cu(II) uptake was measured at the lowest NaCl concentration of 0.01 M at both pH 5 (63% removal) and pH 6 (>85% removal), with increasing NaCl concentrations generally resulting in lower Cu(II) uptake (e.g., <20% removal with 0.5 M NaCl at pH 5). The increase in Cu(II) removal at the highest NaCl concentrations that was attributed to the formation of atacamite in the faujasite experiments occurred at lower NaCl concentrations (0.2, 0.5 and 1.0 M NaCl) at pH 6 in the clinoptilolite experiments. The impact of CaCl2 on Cu(II) uptake by natural clinoptilolite was also different than observed for synthetic faujasite at pH 6. The highest CaCl2 concentration of 0.03 M only decreased Cu(II) removal to 40% for natural clinoptilolite at pH 6, whereas the same CaCl2 concentration decreased Cu(II) removal to <20% for synthetic faujasite at pH 6. The less pronounced impact of CaCl2 on the reactivity of natural zeolite compared to synthetic zeolite might be due to a lower affinity of Ca to bind to natural clinoptilolite. Taken together, the results of the NaCl and CaCl2 impact experiments suggest that while Cu(II) uptake to synthetic faujasite is more favourable than clinoptilolite in solutions containing low levels of NaCl and CaCl2, there are some aqueous conditions in which natural clinoptilolite outperforms faujasite, particularly in the presence of CaCl2.
3.4 Acidic regeneration of Cu(II)-loaded synthetic and natural zeolites
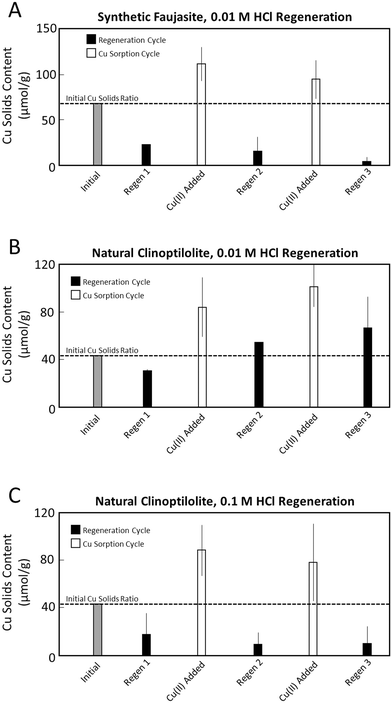
For synthetic faujasite, 0.01 M HCl effectively released adsorbed Cu(II) over several cycles (Fig. 4). The first 0.01 M HCl regeneration cycle decreased the solid-phase Cu(II) content from 63 to ∼25 μmol g−1. Following this first cycle, the Cu(II) adsorption reactivity of faujasite was not impacted negatively, with the Cu(II) content of the solids increasing to 112 μmol g−1 after reaction with 0.6 mM Cu(II). This trend continued over three 0.01 M HCl regeneration cycles, leading to consistently effective Cu(II) uptake and nearly complete Cu(II) release following the third 0.01 M HCl extraction (Fig. 4). Concomitant with effective Cu(II) release from synthetic faujasite using 0.01 M HCl, a small fraction of Al was also released from the solids (∼3% of the total Al added as faujasite; Fig. S4). This Al release is attributed to the low pH (∼2) of the zeolite suspension following addition of 0.01 M HCl leading to some zeolite dissolution. Synchrotron XRD data of the initial and final faujasite solids following 0.01 M HCl regeneration were similar, indicating minimal solid-phase transformation (Fig. S5).The results of Cu(II) release from synthetic faujasite using 0.1 M HCl contrasted starkly with those using 0.01 M HCl. The addition of 0.1 M HCl to Cu(II)-loaded faujasite resulted in solids that could not be centrifuged (supernatant turbidity >10 NTU), the dissolution of >90% of Al contained in the faujasite structure and loss of the zeolite structural framework (Fig. S5). These results imply that synthetic faujasite was not stable at this higher HCl concentration, thus subsequent regeneration experiments were not performed.
Acidic regeneration of Cu(II)-loaded natural clinoptilolite with 0.01 M HCl was somewhat effective over three regeneration cycles, though not as effective as with faujasite. The addition 0.01 M HCl decreased the Cu(II) content of clinoptilolite from 43 to 30 μmol g−1 after one cycle (Fig. 4). Similar to the faujasite series, the Cu(II) adsorption reactivity of clinoptilolite did not decrease following acidic regeneration, with the Cu(II) content increasing to 85 and 102 μmol g−1 after subsequent reactions with 0.6 mM Cu(II). However, repeated cycles of 0.01 M HCl regeneration were not able to release Cu(II) from clinoptilolite to the same extent that was observed for faujasite. The final Cu(II) content of the natural clinoptilolite solids after three 0.01 M HCl regeneration cycles was 68 μmol g−1, which was more than 50% higher than the initial Cu-loaded sample before regeneration. Consistent with the moderate effectiveness of 0.01 M HCl, only 6% of Al present in the initial clinoptilolite solids was released during all three regeneration steps (Fig. S4) and synchrotron XRD data for the initial and regenerated clinoptilolite solids were similar. However, the aqueous solution generated by 0.01 M HCl regeneration had a higher pH (2.4–2.5) compared to the analogous faujasite experiments and also contained tens of μM Ca(II) (Fig. S4). These observations are consistent with the dissolution of Ca-carbonate mineral impurities in the natural clinoptilolite and can explain the lower effectiveness of 0.01 M HCl for clinoptilolite regeneration.
Whereas the faujasite structure was not stable in 0.1 M HCl (Fig. S5), the regeneration of clinoptilolite at the higher HCl concentration of 0.1 M was highly effective. After one cycle of 0.1 M HCl regeneration, the Cu(II) content decreased from 43 to 17 μmol g−1, then increased to 88 μmol g−1 following the next round of reaction with 0.6 mM Cu(II). Most notably, the final Cu(II) content of natural clinoptilolite following three 0.1 M regeneration cycles was only 10 μmol g−1, indicating sustained uptake and release of Cu(II) in these conditions. Similar to experiments at the lower HCl concentration of 0.01 M, some aqueous Al and Ca were released from the solid phase (Fig. S4), but XRD data indicated that the clinoptilolite structural framework was largely preserved (Fig. S5). The enhanced stability of natural clinoptilolite compared to synthetic faujasite in the 0.1 M HCl regeneration solution is most likely due to the role of mineral impurities in the natural sample that limited H+ attack of the zeolite structure by consuming H+ in side reactions, as well as the smaller specific surface area, which would decrease dissolution rates.
3.5 Principal component analysis of the EXAFS dataset and target transformations
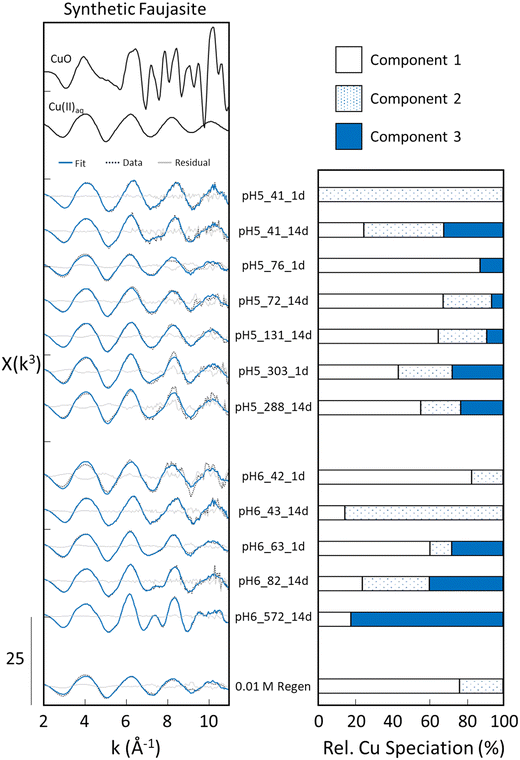
Principal component analysis of the entire EXAFS dataset, which included synthetic and natural zeolite adsorption and regeneration samples, yielded a minimum in the indicator function (IND) with only three independent (principal) components (Fig. S6). This result implies that the major variance across the entire EXAFS dataset can be reproduced using only three unique Cu(II) species (i.e., principal components). Therefore, linear combinations of three components were used to reconstruct all experimental EXAFS spectra, with the three corresponding endmember spectra extracted by iterative target transformation (ITT) analysis. Shell-by-shell fits of the extracted endmember spectra were performed to determine the Cu(II) bonding environment represented by each endmember spectra (Fig. S7), the results of which are described below and summarized in Table S3.Component 1 consisted of a largely periodic and smooth EXAFS oscillation that peaked in intensity near k of 6 Å−1 and continuously decreased in amplitude out to k of 11 Å−1. The Fourier-transformed EXAFS spectrum of component 1 was characterized by a single major first-shell peak that was fit with a Cu–O pair with coordination number (CNCu–O) of 4.3 ± 0.2 and interatomic distance (RCu–O) of 1.97 ± 0.01 Å. These first-shell fitting parameters match those reported in previous Cu K-edge EXAFS studies of aqueous Cu(II) and Cu(II) (hydr)oxides and are consistent with Cu(II) in disordered octahedral coordination due to Jahn–Teller distortion.11 In addition, a second peak of much lower amplitude in the Fourier transform (arrow in Fig. S7) was visible above the noise and was fit with a geometrically-constrained Cu–O–O multiple scattering (MS) path with RCu–O–O = 4.04 ± 0.03 Å, in agreement with previous EXAFS fits of Cu-bearing materials.29 The shell-by-shell fitting results for component 1 were identical to those of aqueous Cu(II) (Table S3), consistent with the similar shapes of the two EXAFS spectra. Therefore, we conclude that component 1 represents a local Cu coordination environment that matches aqueous Cu(II), which we interpret as Cu(II) uptake via outer-sphere complexation, whereby Cu(II) ions remain fully solvated and bind to zeolite surfaces through H-bonding or electrostatic interaction.
Component 2 exhibited EXAFS features that resembled component 1, but some key differences were apparent, especially the higher amplitude of the third and fourth oscillations near k of 8.2 and 10.2 Å−1. The Fourier transform of component 2 was characterized by an intense first-shell peak and second-shell peak with lower amplitude, similar to component 1 (Fig. S7). However, the second-shell peak of component 2 exhibited a slightly higher amplitude than that of component 1, suggesting the presence of next-nearest-neighbour backscattering atoms. Fits of the first-shell peak of the Fourier transform returned similar fitting parameters as those of component 1, including CNCu–O = 3.8 ± 0.3 and RCu–O = 1.94 ± 0.01 Å, consistent with Jahn–Teller distorted CuO6 octahedra.34 However, in contrast to component 1, fits of the second-shell peak required contributions from both Cu–O–O MS paths (RCu–O–O = 3.96 ± 0.05 Å) and a Cu–Al/Si path with fit-derived CNCu–Al/Si = 0.8 ± 0.5 and RCu–Al/Si = 2.82 ± 0.05 Å. Due to the similarity in atomic number between Al and Si, it is difficult to reliably distinguish these two backscattering atoms with EXAFS spectroscopy. Nevertheless, the fitting parameter for this Cu–Al/Si path are identical within standard errors to previous EXAFS investigations of Cu(II) bound in inner-sphere complexes to surface Al polyhedra of Al-bearing minerals, including zeolites.16,29 Therefore, we conclude that component 2 represents Cu(II) bound to the zeolite surface in an inner-sphere complex, most likely via edge-sharing mononuclear linkages between CuO6 and Al/Si polyhedra, which would have a theoretical CNCu–Al = 1 and relatively short RCu–Al/Si compared to corner-sharing complexes. We note here that this Cu bonding environment does not, by itself, indicate whether Cu is bound to internal or external surfaces of the zeolite. Finally, it is important to recognize that the distinction between component 1 and component 2 was largely based on only modest changes in second-shell peak intensity, thus to avoid over interpreting these differences, we will primarily refer to both component 1 (outer-sphere complexes) and component 2 (inner-sphere complexes) together as monomeric Cu in the remainder of this work.
In contrast to components 1 and 2, component 3 exhibited EXAFS features that indicated pronounced interferences from multiple backscattered waves, consistent with strong contributions from nearest- and next-nearest neighbour atomic shells. The Fourier transform of component 3 displayed a first-shell peak with similar amplitude and position as components 1 and 2, which was fit with a Cu–O atomic pair indicative of distorted CuO6 octahedra (CN = 3.5 ± 0.4 and R = 1.97 ± 0.01 Å). However, the second-shell peak of component 3 had much higher amplitude than the other components and contained a clear shoulder at longer R, indicating the presence of multiple next-nearest neighbour atomic pairs. The best fit of the second-shell peak of component 3 was achieved with two Cu–Cu paths, positioned at 3.04 ± 0.01 Å (CNCu–Cu1 = 1.9 ± 0.3) and 3.45 ± 0.01 Å (CNCu–Cu2 = 1.4 ± 0.3). These fit-derived RCu–Cu values are a good match to the theoretical RCu–Cu values expected for edge-sharing and corner-sharing CuO6 octahedra based on the crystal structures of tenorite (RCu–Cu1 = 2.9 to 3.1 Å, RCu–Cu2 = 3.43 Å)35 and atacamite (RCu–Cu1 = 3.0 to 3.1 Å, RCu–Cu2 = 3.37 to 3.43 Å).36 However, the fit-derived CNs for the Cu–Cu paths were significantly lower than the theoretical CNs for crystalline Cu-bearing minerals (e.g., CNCu–Cu1 = 8 for tenorite). Therefore, we interpret the fits of component 3 as polymeric Cu, which consists of Cu–Cu clusters with lower degrees of Cu–Cu polymerization than crystalline Cu-bearing minerals.
3.6 Cu K-edge EXAFS analysis of synthetic faujasite samples
Fig. 5 shows the experimental Cu K-edge EXAFS spectra of the synthetic faujasite series, including adsorption and regeneration samples, plotted beneath spectra of Cu-bearing reference materials. This figure also shows bar charts of the LCF-derived relative concentrations of each component for all samples, with the sample names reflecting the pH, solid-phase Cu(II) content and reaction time. Comparing the EXAFS analysis across the entire faujasite series reveals a key result: monomeric Cu was the dominant form of Cu(II) detected in all faujasite samples. The adsorption samples generated at pH 5 were all found to contain >60% monomeric Cu, regardless of Cu(II) surface loading and reaction time. For example, the sample with the lowest Cu(II) loading of 41 μmol g−1 and reaction time of 1 d (pH5_41_1d) contained 0% polymeric Cu, whereas that with the highest Cu(II) loading (pH5_303_1d) contained <30% polymeric Cu. However, no clear and systematic trend in the fraction of polymeric Cu with reaction time or Cu(II) loading was identified by the EXAFS analysis of the pH 5 adsorption samples. The adsorption samples generated at pH 6 also generally contained <40% polymeric Cu, although a trend in increasing polymeric Cu content with increasing surface loading was evident. For example, the fit-derived polymeric Cu fraction increased from 0% for the solids with the lowest Cu(II) loading (pH6_42_1d) to nearly 40% for the pH6_82_14d sample. Consistent with the minor polymeric Cu(II) content of the adsorption samples, the faujasite solids remaining after three cycles of 0.01 M HCl regeneration were also found to consist of 100% monomeric Cu, which contrasts the >20% polymeric Cu detected in the initial sample before regeneration (pH6_63_1d). The exception to the predominance of monomeric Cu in the faujasite series was the sample containing the highest Cu(II) loading at pH 6. This sample was generated in experimental conditions that are most likely to lead to Cu-bearing solids due to supersaturation, with XRD analysis suggesting this sample contained a minor fraction of atacamite (Fig. S8).3.7 Cu K-edge EXAFS analysis of natural clinoptilolite samples
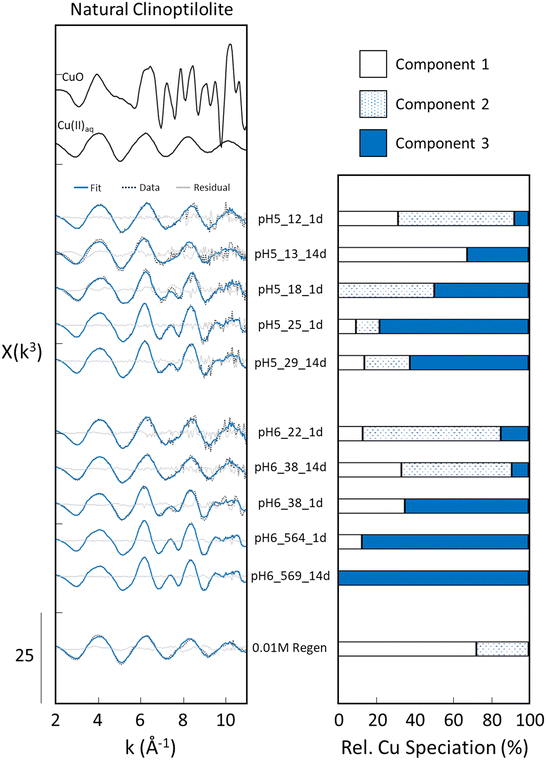
Fig. 6 displays the EXAFS analysis of the natural clinoptilolite samples, including the experimental spectra, LCF output and bar charts of the relative concentrations of each component. A major difference in solid-phase Cu speciation between the natural clinoptilolite and synthetic faujasite series was the detection of substantially higher fractions of polymeric Cu in the clinoptilolite samples. Whereas the EXAFS analysis found no evidence for polymeric Cu in several faujasite samples, polymeric Cu was detected in every clinoptilolite adsorption sample at both pH 5 and 6, despite the generally lower solid-phase Cu(II) loading of the clinoptilolite series compared to faujasite. In addition, a more systematic trend in increasing fraction of polymeric Cu with increasing Cu(II) loading was found for the clinoptilolite series than faujasite. For example, polymeric Cu in the pH 5 adsorption samples increased continuously from 7% at the lowest Cu(II) loading (pH5_12_1d) to >60–70% at the highest Cu(II) loadings (pH5_25_1d, pH5_29_14d). The detection of polymeric Cu in the pH 5 conditions is particularly striking given that the solution conditions at pH 5 are largely undersaturated with respect to Cu-bearing minerals. Thus, the polymeric Cu detected in such samples likely reflects the formation of a short-ranged cluster of Cu–Cu polymers rather than the precipitation of a Cu-bearing mineral. Despite the solution conditions at pH 5 being largely undersaturated with respect to Cu-bearing minerals. A similar trend in increased polymeric Cu fraction with increasing surface loading was also observed for the pH 6 series, with the samples with the highest Cu(II) loading containing the highest fraction of polymeric Cu. Finally, despite the general differences in solid-phase Cu speciation between the clinoptilolite and faujasite adsorption samples, the 0.01 M HCl regenerated clinoptilolite sample was found to contain 100% monomeric Cu (73% outer-sphere Cu, 27% inner-sphere Cu), which closely resembled the Cu speciation of the 0.01 M HCl regenerated faujasite sample (76% outer-sphere Cu, 24% inner-sphere Cu).3.8 Differences in Cu(II) adsorption reactivity of synthetic and natural zeolites
Combining all macroscopic and molecular-scale data for both zeolites reveals distinct Cu(II) interactions between synthetic and natural zeolites. The most striking differences between the two solids with respect to Cu(II) adsorption reactivity were the significantly higher Cu(II) uptake efficiency for synthetic faujasite compared to clinoptilolite and the greater predominance of monomeric Cu associated with faujasite relative to clinoptilolite. Although both zeolites exhibited systematic increases in Cu(II) uptake with pH, the faujasite samples removed several times more Cu(II) for a given pH and initial Cu(II) concentration compared to natural clinoptilolite. The most likely explanation for the enhanced Cu(II) uptake by synthetic faujasite is the substantially higher specific surface area of faujasite compared to clinoptilolite, which would result in more external and internal surface available for Cu(II) uptake. This difference in specific surface area and particle size might also play a role in the lower fractions of polymeric Cu detected by EXAFS spectroscopy. A scenario can be envisioned wherein, for a given Cu(II) loading, Cu that is distributed across several times larger surface area (for adsorption) or volume (for absorption) is less likely to form Cu–Cu polymers. However, the higher fraction of polymeric Cu for the natural zeolites might also be related to differences in the presence of mineral impurities in the two types of zeolites. In particular, the presence of carbonate minerals in the natural clinoptilolite samples might lead to localized microenvironments of alkaline pH due to carbonate dissolution during exposure to aqueous solutions,37 which would favour polymeric Cu formation. This pH-related effect of carbonate mineral impurities in the natural clinoptilolite samples comes in addition to the implied role of carbonates in resisting pH changes of clinoptilolite suspensions during HCl regeneration (Fig. S4).Although the Cu EXAFS analysis indicated the entire dataset, including adsorption and regeneration samples for both zeolites, could be reproduced using components representing outer-sphere, inner-sphere and polymeric Cu, the fractional distribution of these Cu(II) species across external and internal surfaces remains an interesting unresolved question. Considering the macroscopic and molecular-scale data, an argument can be made that outer-sphere Cu(II) adsorption within tunnel sites of faujasite is a likely scenario due to 1) the large specific surface area of these solids, 2) the predominance of monomeric Cu and 3) the theoretically larger internal tunnels of faujasite compared to clinoptilolite based on their crystal structures, which might facilitate Cu(II) entry into the cavities.38,39 However, based on tunnel geometry considerations, polymeric Cu can also occur within tunnels of both zeolites. For example, the shell-by-shell fit results (i.e., fit-derived CN and R values) for component 3 are consistent with a roughly 0.5–0.6 nm by 1.0–1.2 nm cluster of Cu(II) atoms (Fig. S9), which would fit within the cavities of both zeolites. Therefore, our dataset cannot exclude the formation of polymeric Cu in internal surfaces. However, it is important to note that the HCl regeneration steps released polymeric Cu preferentially, suggesting that polymeric Cu species were more accessible to H+-induced dissolution and/or more weakly associated to the zeolite surface than monomeric Cu, which might be explained by polymeric Cu being preferentially associated to external surfaces.
3.9 Implications for the deployment of zeolite-based filters for Cu(II) removal and recovery
Our study uncovered several key aspects of Cu(II) adsorption and extraction from zeolites that can be used to inform the deployment of zeolite-based filter media for holistic Cu(II) treatment and recovery. First, synthetic faujasite exhibited more rapid Cu(II) removal kinetics and several times greater Cu(II) uptake efficiency compared to natural clinoptilolite, which are two critical parameters that improve filter performance in field application. Furthermore, weaker acid concentrations were able to regenerate faujasite, with nearly complete release of Cu(II) and minimal impacts to subsequent Cu(II) adsorption reactivity using 0.01 M HCl. Indeed, the efficiency of Cu(II) recovery normalized by HCl dosage (i.e., Cu(II) recovered/H+ dosed) for faujasite using 0.01 M HCl (0.038 mmol Cu(II)/mmol H+) was higher than any other regeneration experiment (0.011 mmol Cu(II)/mmol H+ for clinoptilolite using 0.01 M HCl; 0.003 mmol Cu(II)/mmol H+ for clinoptilolite using 0.1 M HCl). The lower HCl concentration for faujasite regeneration compared to clinoptilolite is an advantage because the use of concentrated acids can require more complex safety precautions and necessitate changes in water distribution infrastructure (e.g., higher qualities of steel or other often expensive pipe materials). While these key parameters favour the application of synthetic faujasite in filter media, our study also indicated several practical benefits of natural clinoptilolite. First, the impact of CaCl2 on Cu(II) uptake was less pronounced with clinoptilolite compared to faujasite, which is a property of clinoptilolite that should not be ignored due to the typically high Ca concentration of geothermal fluid. Second, the clinoptilolite sample was more resistant to pH changes during acidic regeneration and the structure of the zeolite was stable at the higher HCl concentration of 0.1 M. This enhanced stability at higher HCl concentrations, which is attributed partly to the presence of carbonate mineral impurities that help buffer pH, could be an advantage in cases where lower HCl concentrations are not effective at releasing adsorbed cations and regenerating the zeolite, which can be relevant for ions that adsorb to zeolites more strongly than Cu(II), such as Pb(II). However, the most obvious benefit of the natural clinoptilolite zeolite is its relative ease of procurement. Natural clinoptilolite can be obtained directly from zeolite mineral quarries without the need for costly or complicated syntheses, which can dramatically decrease the environmental impact and economic costs of clinoptilolite-based filter media compared to faujasite, especially if deployed at large scales. Although not part of the current study, a comparison of the requisite resources and costs associated with obtaining each zeolite must be combined with Cu(II) removal and regeneration efficiencies established here to best inform the selection of zeolite for field deployment.Conclusions
Our study evaluated the performance of two structurally distinct zeolites for holistic Cu(II) removal and recovery in the context of treatment of geothermal fluid. Our results uncovered key differences between synthetic faujasite and natural clinoptilolite, which has direct consequences for their deployment in zeolite-based filters. The key conclusions from this work are as follows:• Synthetic faujasite outperformed natural clinoptilolite with respect to Cu(II) removal in most conditions (e.g., more rapid uptake and higher Cu(II) removal per mass of zeolite).
• Cu(II) uptake by natural clinoptilolite was impacted less by co-occurring aqueous Ca, a common competing cation found in geothermal fluid, than synthetic faujasite.
• Regeneration with 0.01 M HCl was highly effective for Cu(II) recovery from synthetic faujasite, whereas 0.1 M HCl was required for clinoptilolite.
• Solid-phase Cu speciation was dominated by monomeric Cu for synthetic faujasite, whereas polymeric Cu was the main Cu(II) species taken up by natural clinoptilolite.
• Selection of zeolite-based filters for holistic Cu(II) removal and recovery must consider the reactivity of the filter media over extended regeneration cycles, in addition to the environmental and economic costs of obtaining the filter materials.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information is available. See DOI: https://doi.org/10.1039/d5ew00972c.
Acknowledgements
This work was part of the Danish sub-project under the European PERFORM II project. Funding for the trans-European project was obtained from GEOTHERMICA & JPP-SES, the European Commission and the National Funding Agencies in Denmark (EUDP), Germany (PtJ), and the Netherlands (RVO). Christina Rosenberg Lynge and Pernille Stockmarr are acknowledged for assistance with adsorption experiments and analytical measurements. Kajsa Sigfridsson Clauss and Mahesh Ramakrishnan at MAX IV are thanked for support during XAS and XRD data collection. We acknowledge MAX IV Laboratory for time on the Balder beam line under Proposal 20221096. Research conducted at MAX IV, a Swedish national user facility, is supported by the Swedish Research council under contract 2018-07152, the Swedish Governmental Agency for Innovation Systems under contract 2018-04969, and Formas under contract 2019-02496. We acknowledge DESY (Hamburg, Germany), a member of the Helmholtz Association HGF, for the provision of experimental facilities. Parts of this research were carried out at PETRA III and we would like to thank Edmund Welter for assistance in using the P.65 beam line. Finally, we acknowledge the European Synchrotron Radiation Facility (ESRF) for provision of synchrotron radiation facilities and Momentum Transfer for facilitating the measurements. Jakub Drnec is thanked for assistance and support in using beam line ID31. The measurement setup was developed with funding from the European Union's Horizon 2020 research and innovation program under the STREAMLINE project (grant agreement ID 870313).References
- M. Rehman, L. Liu, Q. Wang, M. H. Saleem, S. Bashir and S. Ullah, et al. Copper environmental toxicology, recent advances, and future outlook: a review, Environ. Sci. Pollut. Res., 2019, 26(18), 18003–18016 CrossRef CAS.
- S. A. Al-Saydeh, M. H. El-Naas and S. J. Zaidi, Copper removal from industrial wastewater: A comprehensive review, J. Ind. Eng. Chem., 2017, 56, 35–44, DOI:10.1016/j.jiec.2017.07.026.
- R. Meirbekova, D. Bonciani, D. I. Olafsson, A. Korucan, P. Derin-Güre and V. Harcouët-Menou, et al. Opportunities and Challenges of Geothermal Energy: A Comparative Analysis of Three European Cases—Belgium, Iceland, and Italy, Energies, 2024, 17(16), 4134 Search PubMed.
- S. Regenspurg, E. Feldbusch, J. Byrne, F. Deon, D. L. Driba and J. Henninges, et al. Mineral precipitation during production of geothermal fluid from a Permian Rotliegend reservoir, Geothermics, 2015, 54, 122–135, DOI:10.1016/j.geothermics.2015.01.003.
- S. Regenspurg, I. Geigenmüller, H. Milsch and M. Kühn, Copper precipitation as consequence of steel corrosion in a flow-through experiment mimicking a geothermal production well, Geotherm. Energy, 2017, 5(1), 1–15 Search PubMed.
- European Commission, Study on the Critical Raw Materials for the EU, 2023 Search PubMed.
- Council of the European Union, Regulation of the European Parliament and of the Council establishing a framework for ensuring a secure and sustainable supply of critical raw materials, 2024 Search PubMed.
- M. Moshoeshoe, M. Silas Nadiye-Tabbiruka and V. Obuseng, A Review of the Chemistry, Structure, Properties and Applications of Zeolites, Am. J. Mater. Sci., 2017, 2017(5), 196–221 Search PubMed , Available from: http://journal.sapub.org/materials.
- J. Cieśla, W. Franus, M. Franus, K. Kedziora, J. Gluszczyk and J. Szerement, et al., Environmental-friendly modifications of zeolite to increase its sorption and anion exchange properties, physicochemical studies of the modified materials, Materials, 2019, 12(19), 3213 CrossRef.
- M. Doula, A. Ioannou and A. Dimirkou, Copper adsorption and Si, Al, Ca, Mg, and Na release from clinoptilolite, J. Colloid Interface Sci., 2002, 245(2), 237–250 CrossRef CAS PubMed.
- V. L. Sushkevich, O. V. Safonova, D. Palagin, M. A. Newton and J. A. van Bokhoven, Structure of copper sites in zeolites examined by Fourier and wavelet transform analysis of EXAFS, Chem. Sci., 2020, 11(20), 5299–5312 RSC.
- G. E. Brown, A. L. Foster and J. D. Ostergren, Mineral surfaces and bioavailability of heavy metals: A molecular-scale perspective, Proc. Natl. Acad. Sci. U. S. A., 1999, 96(7), 3388–3395 CrossRef CAS.
- M. F. Schultz, M. M. Benjamin and J. F. Ferguson, Adsorption and desorption of metals on ferrihydrite – Reversibility of the rection and sorption properties of the regenerated solid, Environ. Sci. Technol., 1987, 21(9), 863–869 CrossRef CAS.
- C. Nila and I. González, The role of pH and Cu(II) concentration in the electrodeposition of Cu(II) in NH4Cl solutions, J. Electroanal. Chem., 1996, 401(1–2), 171–182 CrossRef.
- S. Tahervand and M. Jalali, Sorption and desorption of potentially toxic metals (Cd, Cu, Ni and Zn) by soil amended with bentonite, calcite and zeolite as a function of pH, J. Geochem. Explor., 2017, 181, 148–159, DOI:10.1016/j.gexplo.2017.07.005.
- N. Z. Logar, I. Arčon, J. Kovač and M. Popova, Removal of Copper from Aqueous Solutions with Zeolites and Possible Treatment of Exhaust Materials, Chem. Ing. Tech., 2021, 93(6), 941–948 CrossRef CAS.
- A. F. Ankrah, B. Tokay and C. E. Snape, Regenerability of fly-ash derived zeolite NaP1: Evaluation via copper recovery, Eng. Rep., 2023, 5(4), 1–12 Search PubMed.
- E. Svobodová, Z. Tišler, K. Peroutková, K. Strejcová, J. Abrham and J. Šimek, et al. Adsorption of Cu(II) and Ni(II) from Aqueous Solutions Using Synthesized Alkali-Activated Foamed Zeolite Adsorbent: Isotherm, Kinetic, and Regeneration Study, Molecules, 2024, 29(10), 2357 CrossRef PubMed.
- S. Shevade and R. G. Ford, Use of synthetic zeolites for arsenate removal from pollutant water, Water Res., 2004, 38(14–15), 3197–3204 CrossRef CAS.
- S. Regenspurg, E. Feldbusch, J. Byrne, F. Eichinger, A. Henkel and J. Iannotta, et al. Prevention of Scaling by Quick Removal of Metals from the Brine via Adsorption, in World Geothermal Congress 2020, Reykjavik, Iceland, 2020, pp. 1–7 Search PubMed.
- N. Mansouri, N. Rikhtegar, H. A. Panahi, F. Atabi and B. K. Shahraki, Porosity, Characterization and Structural Properties of Natural Zeolite–Clinoptilolite – as a Sorbent, Environ. Prot. Eng., 2013, 39(1), 139 CAS.
- D. Reinoso, M. Adrover and M. Pedernera, Green synthesis of nanocrystalline faujasite zeolite, Ultrason. Sonochem., 2018, 42(November 2017), 303–339, DOI:10.1016/j.ultsonch.2017.11.034.
- D. L. Parkhurst and C. A. J. Appelo, User's guide to PHREEQC (Version 2) – A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical equations, US Department of the Interior USGS, Denver, Colorado, Water-Resources Investigations Report 99–4259, 1999 Search PubMed.
- S. Webb, SIXPACK: a graphical user interface for XAS analysis using IFEFFIT, Phys. Scr., 2005, T115, 1011–1014 CrossRef CAS.
- C. M. van Genuchten, S. E. A. Addy, J. Pena and A. J. Gadgil, Removing arsenic from synthetic groundwater with iron electrocoagulation: An Fe and As K-edge EXAFS study, Environ. Sci. Technol., 2012, 46(2), 986–994 CrossRef CAS.
- A. Rossberg, T. Reich and G. Bernhard, Complexation of uranium(VI) with protocatechuic acid – application of iterative transformation factor analysis to EXAFS spectroscopy, Anal. Bioanal. Chem., 2003, 376(5), 631–638 CrossRef CAS.
- C. M. van Genuchten, T. Behrends, P. Kraal, S. L. S. Stipp and K. Dideriksen, Controls on the formation of Fe(II,III) (hydr)oxides by Fe(0) electrolysis, Electrochim. Acta, 2018, 286, 324–338 CrossRef CAS.
- C. Lenz, T. Behrends, T. Jilbert, M. Silveira and C. P. Slomp, Redox-dependent changes in manganese speciation in Baltic Sea sediments from the Holocene Thermal Maximum: An EXAFS, XANES and LA-ICP-MS study, Chem. Geol., 2014, 370, 49–57 CrossRef CAS.
- S. F. Cheah, G. E. Brown and G. A. Parks, XAFS study of Cu model compounds and Cu2+ sorption products on amorphous SiO2, γ-Al2O3, and anatase, Am. Mineral., 2000, 85(1), 118–132 CrossRef CAS.
- W. Song, G. Li, V. H. Grassian and S. C. Larsen, Development of improved materials for environmental applications: Nanocrystalline NaY zeolites, Environ. Sci. Technol., 2005, 39(5), 1214–1220 CrossRef CAS.
- A. M. Pollard, R. G. Thomas and P. A. Williams, Synthesis and stabilities of the basic copper (II) chlorides atacamite, paratacamite and botallackite, Mineral. Mag., 1989, 53(December), 557–563 CrossRef CAS.
- P. Lopesino, J. Alcantara, D. De La Fuente, B. Chico, J. A. Jimenez and M. Morcillo, Corrosion of Copper in Unpolluted Chloride-Rich Atmospheres, Metals, 2018, 8, 866 CrossRef CAS.
- S. G. Le Roux, J. A. Miller, A. J. Dunford and C. E. Clarke, The dissolution kinetics of atacamite in the acid range and the stability of atacamite containing soils from Namaqualand, South Africa, Appl. Geochem., 2016, 64, 22–29, DOI:10.1016/j.apgeochem.2015.09.003.
- I. A. Pankin, A. Martini, K. A. Lomachenko, A. V. Soldatov, S. Bordiga and E. Borfecchia, Identifying Cu-oxo species in Cu-zeolites by XAS: A theoretical survey by DFT-assisted XANES simulation and EXAFS wavelet transform, Catal. Today, 2020, 345, 125–135, DOI:10.1016/j.cattod.2019.09.032.
- S. Åsbrink and L. J. Norrby, A refinement of the crystal structure of copper(II) oxide with a discussion of some exceptional e.s.d.'s, Acta Crystallogr., Sect. B, 1970, 26(1), 8–15 CrossRef.
- X. G. Zheng, T. Mori, K. Nishiyama, W. Higemoto, H. Yamada and K. Nishikubo, et al. Antiferromagnetic transitions in polymorphous minerals of the natural cuprates atacamite and botallackite Cu2Cl(OH)3, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71(17), 1–8 CrossRef.
- K. Biçe, T. Myers Stewart, G. G. Waldbusser and C. Meile, The effect of carbonate mineral additions on biogeochemical conditions in surface sediments and benthic – pelagic exchange fluxes, Biogeosciences, 2025, 641–657 CrossRef.
- J. R. Smyth, A. T. Spaid and D. L. Bish, Crystal structures of a natural and a Cs-exchanged clinoptilolite, Am. Mineral., 1990, 75(5–6), 522–528 CAS.
- W. H. Baur, On the cation and water positions in faujasite, Am. Mineral., 1964, 49(May–June), 697–704 CAS.
| This journal is © The Royal Society of Chemistry 2026 |