Influence of physicochemical factors on the transport behavior of polyethylene nanoplastics in black soil and brown earth
Received
12th September 2025
, Accepted 12th November 2025
First published on 12th November 2025
Abstract
Polyethylene nanoplastics (PENPs) accumulate in soil, presenting significant environmental risks. However, research on the transport behavior of PENPs in soils remains limited. In this study, we employed saturated column experiments to systematically examine the transport of PENPs in two contrasting soil types, black soil (BS) and brown earth (BE), while evaluating the critical effects of ionic strength (IS), pH, and flow velocity. The results indicated that PENP mobility increased with higher flow velocity (1–5 mL min−1), lower IS (1–10 mM NaCl/CaCl2), and higher pH (5–9) in both soils. Notably, PENPs exhibited significantly greater transport capacity in BS compared to BE. Under identical IS and pH conditions, BS exhibited higher surface negative charges and more negative zeta potential than BE. DLVO theory calculations confirmed stronger electrostatic repulsion between the negatively charged PENPs and BS, resulting in significantly enhanced transport capacity and reduced deposition compared to BE. These findings demonstrate that soil surface charge critically governs PENP transport and provide a mechanistic foundation for assessing the mobility and ecological risks of nanoplastics in soils with distinct physicochemical properties.
Environmental significance
Polyethylene nanoplastics enter soil systems via multiple pathways, including the use of agricultural plastic mulch, sewage irrigation, and atmospheric deposition. These particles accumulate substantially in soils, presenting considerable environmental risks. This study examines the transport behavior of polyethylene nanoplastics in two distinct soil types and identifies key factors influencing their mobility, highlighting the crucial regulatory role of soil surface charge in controlling nanoplastic movement. The results offer a scientific basis for assessing the migration patterns and ecological risks of nanoplastics across diverse soil environments.
|
1. Introduction
Since the 1950s, plastics have been widely used in packaging, healthcare, agriculture, and other industries, with annual production continuously rising. Global plastic production is projected to reach 33 billion metric tons annually by 2050.1 Currently, approximately 360 million tons of microplastics are generated worldwide each year, increasing at a steady rate of 3%.2 These plastic wastes enter the environment through various pathways. Owing to extensive consumption and release, plastic debris of various sizes has been detected across diverse environmental media.3 Once released, these plastics undergo further fragmentation into smaller particles via microbial degradation, UV irradiation, and other processes.4,5 Plastic fibers, fragments, and granules with particle sizes <5 mm are generally defined as microplastics,6 while those <1 µm are termed “nanoplastics”.7 Compared to microplastics, nanoplastics exhibit smaller particle sizes and larger specific surface areas, posing greater environmental risks.8 As both the source and a major sink of plastics, terrestrial systems receive 4 to 23 times more microplastics annually than marine environments.9,10 Key accumulation pathways in soils include agricultural mulch films,11 sludge and organic fertilizers,12,13 wastewater irrigation, atmospheric deposition,13 and landfill leachate infiltration.14,15
Recent studies have shown that microplastics and their adsorbed contaminants can be taken up by plant roots, entering and accumulating in food chains. This process not only threatens human health but also impacts the growth and reproduction of soil fauna.16 In terrestrial environments, microplastics migrate downward under various factors, eventually infiltrating groundwater systems. This phenomenon poses dual risks: compromising drinking water safety and serving as a critical pathway for microplastic dispersion to other ecosystems.17 Understanding the transport characteristics of microplastics in soils is therefore essential for elucidating their environmental fate and contamination patterns. Furthermore, compared to microplastics, nanoplastics have significantly larger specific surface areas, enhancing their adsorption capacities for environmental pollutants. These nano-sized particles act as efficient vectors for contaminant transport, amplifying their hazardous impacts on human health and ecological systems.18–20 As emerging contaminants to soil biota and ecosystems globally, nanoplastics have attracted worldwide attention due to their unique environmental behaviors and potential risks.21
To date, numerous studies have focused on the transport of polystyrene (PS) in porous media,22 revealing that their transport behavior is governed by multiple factors, including porous medium properties,23 microplastic/nanoplastic characteristics,24,25 and hydrodynamic conditions.26 However, research remains limited regarding soil contamination by high-production-volume plastics like polyethylene (PE) and polypropylene (PP), with only recent reports documenting nanoplastic-induced soil ecosystem hazards.27 Existing studies demonstrate that during the transport of nanoplastics in soils, interactions with soil mineral particles, organic matter, etc., may affect their migration behavior.28 Furthermore, the small size of nanoplastics enables uptake by soil microorganisms,29 which subsequently act as biological vectors facilitating deep soil penetration. The complex soil matrix, combined with the size, surface smoothness, and mobility of nanoplastics, profoundly influences their transport characteristics.30 To systematically investigate the soil-dependent transport of PENPs in environmentally relevant contexts, we selected the black soil and brown earth from farmlands of Heilongjiang and Jiangsu, China. These regions represent two of China's most important agricultural zones, yet they possess fundamentally different soil characteristics that govern colloidal interactions.31 BS is typically characterized by higher organic matter content, a near-neutral pH, and a permanent negative charge dominated by 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals. In contrast, BE generally possesses lower organic matter content, is more acidic, and its variable charge is influenced by 1
1-type clay minerals. In contrast, BE generally possesses lower organic matter content, is more acidic, and its variable charge is influenced by 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals and iron/aluminum oxides. The fundamental differences in charge properties between these soil types are well-established in soil colloid chemistry.32 We hypothesize that these stark contrasts, particularly in surface charge density and solution chemistry, will lead to profoundly different electrostatic interactions with negatively charged PENPs. This comparison not only deciphers the key soil properties controlling nanoplastic mobility but also provides a mechanistic basis for understanding the fate of PENPs across a wide range of environmental conditions. PENPs have emerged as critical environmental contaminant vectors due to their unique physicochemical properties. These ultra-small particles possess exceptionally high specific surface areas and pronounced hydrophobicity, endowing them with remarkable adsorption capacities. PENPs efficiently concentrate heavy metal ions and organic pollutants, functioning as “pollutant shuttles” that amplify contaminant dispersion and ecological risks.33–35 Consequently, elucidating the transport mechanisms of nanoplastics in soils is imperative for accurately assessing their environmental fate and predicting associated risks.
1-type clay minerals and iron/aluminum oxides. The fundamental differences in charge properties between these soil types are well-established in soil colloid chemistry.32 We hypothesize that these stark contrasts, particularly in surface charge density and solution chemistry, will lead to profoundly different electrostatic interactions with negatively charged PENPs. This comparison not only deciphers the key soil properties controlling nanoplastic mobility but also provides a mechanistic basis for understanding the fate of PENPs across a wide range of environmental conditions. PENPs have emerged as critical environmental contaminant vectors due to their unique physicochemical properties. These ultra-small particles possess exceptionally high specific surface areas and pronounced hydrophobicity, endowing them with remarkable adsorption capacities. PENPs efficiently concentrate heavy metal ions and organic pollutants, functioning as “pollutant shuttles” that amplify contaminant dispersion and ecological risks.33–35 Consequently, elucidating the transport mechanisms of nanoplastics in soils is imperative for accurately assessing their environmental fate and predicting associated risks.
The primary objective of this study is to elucidate the vertical transport mechanisms of PENPs in two contrasting soil types. The selected soils exhibit distinct physicochemical properties representative of different regional ecosystems, while polyethylene was chosen as a model polymer due to its ubiquitous presence in packaging, plastic bags, and containers. Ionic strength, pH, and flow rate are typical factors influencing the migration of microplastics/nanoplastics and are often used to study their transport behavior.36 A saturated soil column was used as the experimental equipment to study the migration characteristics of PENPs in soil. The specific objectives are as follows: (1) to explore the effect of ionic strength on the transport of PENPs in soil; (2) to investigate the effect of pH on the transport of PENPs in soil; (3) to explore the effect of water flow velocity on the transport of PENPs in soil; (4) to reveal the potential mechanisms of migration and retention of PENPs in soil, providing a reference for studying their migration patterns.
2. Materials and methods
2.1. PENP suspensions
Polyethylene powder was purchased from Qihong Plastic Co., Ltd (Dongguan, China), with a certified average particle size of 100 nm (SD ± 5 nm) and density of 0.93 g cm−3. All other chemical reagents used in this study were of analytical grade. A stock suspension of PENPs was prepared by dispersing 250 mg of powder in deionized water and diluting to 500 mL in a volumetric flask, yielding a concentration of 500 mg L−1. The suspension was sonicated for 30 minutes using an ultrasonic cleaner (KQ-500DE, Kunshan Ultrasonic Instruments, China) to ensure stability. The ionic strength of the suspension was adjusted using background electrolytes (NaCl and CaCl2), and the PENP suspension was further diluted to 80 mg L−1 for experimental use. The stock suspension was stored at 4 °C in the dark. The relatively high concentration of PENPs was chosen to better elucidate the interactions between PENPs and soil media under various conditions. The zeta potential and particle size of PENPs and soil particles in solutions with varying ionic strengths were characterized using a nanoparticle size analyzer (ZEN3690, Malvern Instruments Ltd, UK) (Table 1). Morphological analysis of PENPs was performed by Scanning Electron Microscopy (SEM). Surface functional groups of PENPs were determined by using a Fourier transform infrared (FTIR) spectrometer.
Table 1 Summary of experimental conditions and physiochemical information for PENP transport experiments conducted in two soils
| Porous medium |
Salt composition |
IS (mM) |
pH |
Zeta potentials (mV) |
DLS diameter of PENPs (nm) |
PENPs–soil |
| PENPs |
Soil |
Φ
max (KT) |
Φ
min2 (KT) |
| Black soil |
NaCl |
1 |
7 |
−15.00 ± 1.33 |
−26.30 ± 0.56 |
206 ± 27.57 |
39.21 |
−0.0058 |
| NaCl |
10 |
7 |
−9.18 ± 0.53 |
−15.10 ± 0.42 |
221 ± 35.53 |
8.27 |
−0.1254 |
| CaCl2 |
1 |
5 |
−8.49 ± 0.67 |
−20.80 ± 0.31 |
224 ± 13.58 |
18.56 |
−0.0074 |
| CaCl2 |
1 |
7 |
−18.20 ± 2.73 |
−10.80 ± 0.15 |
191 ± 28.92 |
19.68 |
−0.0063 |
| CaCl2 |
1 |
9 |
−20.00 ± 0.62 |
−31.40 ± 0.57 |
152 ± 10.82 |
51.17 |
−0.0039 |
| CaCl2 |
10 |
7 |
−15.60 ± 1.36 |
−8.83 ± 0.82 |
217 ± 15.54 |
9.45 |
−0.1200 |
| Brown earth |
NaCl |
1 |
7 |
— |
−20.20 ± 0.45 |
— |
28.65 |
−0.0176 |
| NaCl |
10 |
7 |
— |
−17.30 ± 0.90 |
— |
3.28 |
−0.4054 |
| CaCl2 |
1 |
5 |
— |
−3.16 ± 0.12 |
— |
0.39 |
−0.0452 |
| CaCl2 |
1 |
7 |
— |
−7.89 ± 0.25 |
— |
9.05 |
−0.0197 |
| CaCl2 |
1 |
9 |
— |
−20.90 ± 0.19 |
— |
32.16 |
−0.0120 |
| CaCl2 |
10 |
7 |
— |
−7.25 ± 0.70 |
— |
1.21 |
−0.4577 |
2.2. Porous media
Two contrasting soil types were collected from non-agricultural areas in China: black soil (BS) from Heilongjiang Province and brown earth (BE) from Jiangsu Province. To minimize microplastic contamination, soil samples were obtained from depths >150 cm using a single-point sampling method. The soils were air-dried indoors, crushed, and sieved to <2 mm for subsequent experiments. Soil pH was determined using a pH meter at a soil-to-water ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 (w/v).
2.5 (w/v).
2.3. Column experiments
The transport experiments were conducted in acrylic columns (2 cm inner diameter, 15 cm length) with 100-mesh nylon membranes fixed at both ends to ensure uniform flow distribution and prevent soil loss. After adding a small amount of deionized water, black soil or brown earth was incrementally packed into the columns. Each soil addition was followed by tapping with a rubber hammer and stirring with a glass rod to minimize stratification and air entrapment, ensuring homogeneous packing.37 Soil porosity was determined gravimetrically (Table 2). Prior to experiments, columns were flushed with deionized water at 1 mL min−1 using a peristaltic pump (BT100-1L, Shanghai Qingpu Huxi Instrument Factory, China) until the effluent was free of visible impurities. Non-reactive tracer tests (1 mM NaNO3) were performed to characterize flow properties. Symmetric breakthrough curves of NO3− indicated uniform soil distribution and optimal packing with negligible preferential flow. Dispersion coefficients were derived by fitting the NO3− breakthrough curves with HYDRUS-1D.
Table 2 Physical and chemical properties of the two soils
| Soils |
Particle size distribution |
Porosity |
pH |
SOM (g kg−1) |
| Clay (%) |
Silt (%) |
Sand (%) |
| Black soil |
24 |
55 |
18 |
0.41 |
6.06 |
32.78 |
| Brown earth |
20 |
61 |
22 |
0.45 |
5.63 |
12.56 |
Soil columns were pre-equilibrated by flushing with deionized water for 12 h, followed by replacement with the desired background electrolyte solution to stabilize experimental conditions. Subsequently, 3 pore volumes (PV) of PENP suspensions were pumped through the columns under varying conditions of ionic strength (IS: 1 mM and 10 mM NaCl/CaCl2) and pH (5, 7, and 9), followed by flushing with 3 PV of PENP-free background solution until PENPs were no longer detected in the effluent. Parallel column experiments were conducted under varying flow rates following the same procedure. Effluent from the column outlet was continuously collected in test tubes using graduated cylinders. PENP concentrations were determined by turbidimetric analysis at a wavelength of 350 nm.38 During injection, the PENP suspension was continuously stirred. All column experiments were performed in duplicate.
2.4. Penetration curve plotting
The breakthrough curves (BTCs) of PENP colloids collected at the column outlet were analyzed using the method of moments. BTCs were generated by plotting the relative concentration of PENPs in the column effluent against pore volumes. The recovery rate of PENPs (Meff) was calculated from the zeroth moment of the breakthrough curves:39| | 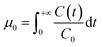 | (1) |
where µ0 is the zeroth moment of the breakthrough curve; C(t) is the concentration of microplastics over time; C0 is the initial concentration of nanoplastics. The recovery rate is defined as the ratio of the mass of PENP colloids recovered at the outlet of the packed column to the mass injected at the inlet of the packed column. The expression is as follows:| | 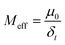 | (2) |
where δt is the injection duration of PENPs into the packed column (min).
2.5. DLVO interaction forces
The Derjaguin–Landau–Verwey–Overbeek (DLVO) theory is used to explain the transport and deposition behavior of PENPs in porous media under different experimental conditions.40,41 The DLVO theory is a core theory in colloid chemistry. By analyzing the interparticle forces between colloidal particles, it explains the stability of colloids and their flocculation phenomena, thereby elucidating the interparticle interaction relationships. Derjaguin–Landau–Verwey–Overbeek (DLVO) theory was used to calculate the PENP–PENP and PENP–soil interaction energies (ΦTOTS(h)) as the sum of van der Waals (ΦvdWS(h)) and electrostatic double layer (ΦEDLS(h)):| | | ΦTOTS(h) = ΦvdWS(h) + ΦEDLS(h) | (3) |
Interaction energies for PENP–PENP nanoparticles were determined by assuming sphere–sphere geometry and the following equations were utilized:
| | 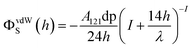 | (4) |
| | ΦEDLS(h) = πε0εrdpΨP2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln[1 + exp(−kh)] ln[1 + exp(−kh)] | (5) |
| | 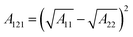 | (6) |
where
dp is the diameter of the PENP nanoparticle (nm),
h is the separation distance between PENP and PENP or PENP and soil (nm),
λ usually denotes a characteristic wavelength of 100 nm,
A121is the Hamaker constant for the PENP–water–soil,
A11is the Hamaker constant for PENPs (7.6 × 10
−20 J),
A22is the Hamaker constant for water (3.7 × 10
−20 J),
ε0 is the permittivity in vacuum (8.854 × 10
−12 C/V/M),
εr is the dielectric constant (78.5), and
ΨP is the surface potential of the PENPs, and
k is the inverse Debye length (m
−1).
Interaction energies between PENPs and soil particles were developed assuming a sphere-plate geometry according to the following equations:
| | 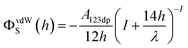 | (7) |
| | 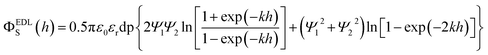 | (8) |
| | 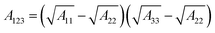 | (9) |
| |  | (10) |
where
A33 is the Hamaker constant for the black soil (5.16 × 10
−20 J) and brown earth (7.5 × 10
−20 J),
Ψ1 is the surface potential of the PENPs,
Ψ2 is the surface potential of the soil,
NA is Avogadro's constant (6.02 × 10
23 mol
−1),
e is the electron charge (−1.60 × 10
−19 C),
I is ionic strength of electrolytes (mol m
−3),
kB is the Boltzmann constant (1.38 × 10
−23 J/K), and
T is the temperature in Kelvin (298 K).
2.6. PENP transport model
The two-site kinetic retention model42,43 was employed to simulate the transport of PENPs in saturated porous media:| | 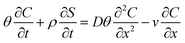 | (11) |
| | 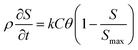 | (12) |
where θ is the porosity; ρ is the bulk density of soil (g cm−3); D is the dispersion coefficient (cm2 min−1); v is the Darcy velocity (cm min−1); k is the deposition coefficient of PENPs (min−1); Smax is the maximum deposition concentration of PENPs on soil surfaces (mg g−1); t is the time (min); x is the flow direction along the travel distance; and C is the concentration of PENPs in solution (mg L−1).
The model was first applied to simulate the breakthrough curve (BTC) of the tracer to determine the hydrodynamic dispersion coefficient (D), followed by inverse modeling of PENP transport experiments to derive the kinetic parameter (k).
3. Results and discussion
3.1. Characterization of PENPs
The surface morphology of PENPs was observed using scanning electron microscopy (Fig. 1a). The particles appear as irregularly shaped spheres with a rough, wrinkled surface texture, covered with irregular cracks and pores of varying sizes. The Fourier transform infrared spectra of PENPs are shown in Fig. 1b. The peaks at 2914 and 2849 cm−1 in the infrared spectrum of PENPs correspond to the asymmetric stretching vibration and stretching vibration of the methylene group (–CH2–), respectively. The peak at 1468 cm−1 originates from the bending vibration induced by the methylene group (–CH2–), and the peak at 717 cm−1 corresponds to the in-plane deformation vibration of the –(CH2) n-group.44
 |
| | Fig. 1 The results obtained by scanning electron microscopy (SEM) and Fourier transform infrared (FTIR) spectroscopy: (a) SEM of PENPs; (b) FTIR of PENPs. | |
3.2. PENP suspension stability
The stability of PENP concentrations under varying solution chemistry conditions (NaCl: 1–10 mM; CaCl2: 1–10 mM) is shown in Fig. 2. During the 60-minute injection period of the suspension into the packed column (the PENP introduction phase in this experiment), the relative concentrations of PENPs remained stable above 0.93 in both NaCl and CaCl2 solutions throughout the transport experiments. This demonstrates excellent stability and indicates that aggregation effects had negligible influence on subsequent transport studies. As shown in Table 1, PENPs retained negative surface charges under all tested conditions (NaCl: −15.0 ± 1.33 mV to −9.18 ± 0.53 mV and CaCl2: −18.20 ± 2.73 mV to −15.60 ± 1.36 mV). The consistently negative zeta potentials resulted in high energy barriers between PENPs, with the exceptional suspension stability attributed to strong electrostatic repulsion among particles.45 Additionally, a magnetic stirrer was employed throughout the experiments to ensure homogeneous dispersion and prevent aggregation.46
 |
| | Fig. 2 Stability of PENP influent suspensions versus time at varying ionic strength solutions of NaCl and CaCl2. | |
3.3. Effects of different factors on the transport of MPs in soil
3.3.1. Effects of IS.
The interaction energies between PENPs and soil particle surfaces under varying ionic strengths were calculated using DLVO theory (Table 1 and Fig. 3). The results indicate that at low ionic strength, strong electrostatic repulsion between PENPs and soil particles enhances PENP mobility. According to DLVO theory, higher ionic strength can significantly influence the physicochemical surface properties (e.g., zeta potential) and stability of colloids.47 The absolute values of zeta potential of both PENPs and soil decrease with rising ionic strength, leading to reduced repulsive forces and thereby promoting the deposition of PENPs onto soil.48 As shown in Fig. 3a and b, the maximum energy barrier (Φmax) between PENPs and soil decreases with increasing solution ionic strength, accompanied by a corresponding reduction in the secondary energy well (Φmin2). These results indicate that elevated ionic strength attenuates the repulsive forces, thereby facilitating the adsorption of PENPs onto soil surfaces and consequently reducing their mobility. The impact of ionic strength on PENP recovery rates is presented in Table 3. Recovery rates decreased with increasing ionic strength. Specifically, increasing NaCl concentration from 1 mM to 10 mM reduced PENP recovery rates in black soil and brown soil from 77.74% to 69.76% and from 63.76% to 47.11%, respectively. Similarly, for CaCl2 solutions, increasing ionic strength from 1 mM to 10 mM decreased recovery rates from 68.26% to 57.78% (BS) and from 51.74% to 32.92% (BE).
 |
| | Fig. 3 The DLVO interaction energies between PENPs and black soil/brown soil as a function of separation distance under different conditions ((a) and (b): ionic strength; (c) and (d): pH). | |
Table 3 Recovery rates of PENPs and simulation parameters in saturated porous media under different experimental conditions
| Porous medium |
Salt composition |
IS (mM) |
pH |
Flow rate (ml min−1) |
M
eff (%) |
K (min−1) |
R
2
|
| Black soil |
NaCl |
1 |
7 |
1 |
77.74 |
0.0057 |
0.95 |
| NaCl |
10 |
7 |
1 |
69.76 |
0.0160 |
0.91 |
| CaCl2 |
1 |
7 |
1 |
68.12 |
0.0181 |
0.97 |
| CaCl2 |
10 |
7 |
1 |
57.78 |
0.0263 |
0.90 |
| CaCl2 |
1 |
5 |
1 |
53.09 |
0.0308 |
0.94 |
| CaCl2 |
1 |
9 |
1 |
83.25 |
0.0093 |
0.91 |
| CaCl2 |
1 |
7 |
5 |
88.25 |
0.1100 |
0.92 |
| Brown earth |
NaCl |
1 |
7 |
1 |
63.76 |
0.0182 |
0.97 |
| NaCl |
10 |
7 |
1 |
47.11 |
0.0336 |
0.88 |
| CaCl2 |
1 |
7 |
1 |
52.68 |
0.0301 |
0.95 |
| CaCl2 |
10 |
7 |
1 |
32.92 |
0.0573 |
0.91 |
| CaCl2 |
1 |
5 |
1 |
42.63 |
0.0418 |
0.91 |
| CaCl2 |
1 |
9 |
1 |
65.41 |
0.0201 |
0.95 |
| CaCl2 |
1 |
7 |
5 |
62.80 |
0.2326 |
0.92 |
As illustrated in Fig. 4a and b, ionic strength significantly influenced the transport of PENPs in soil columns. In black soil, the maximum relative concentration (C/C0) of the breakthrough curve decreased from 89.97% at 1 mM NaCl to 80.79% at 10 mM NaCl. Similarly, in brown soil, the maximum C/C0 declined from 71.60% to 57.81% under the same ionic strength increase. A consistent trend was observed in CaCl2 solutions, where higher ionic strength reduced PENP mobility. These results demonstrate that PENP transport in both black and brown soils is inversely correlated with ionic strength, consistent with previous findings.49 Notably, CaCl2 exhibited a stronger suppression effect on PENP mobility than NaCl, particularly at higher concentrations. This discrepancy arises from the smaller hydrated radius of Ca2+ compared to Na+, which enhances charge screening efficiency while minimizing steric hindrance, thereby promoting particle deposition and reducing mobility.50
 |
| | Fig. 4 The BTCs of PENPs in black soil (BS) and brown earth (BE) under different solution conditions ((a) and (b): ionic strength, at fixed pH 7 and flow rate 1 mL min−1; (c) and (d): pH, at fixed ionic strength 1 mM and flow rate 1 mL min−1). | |
The breakthrough curves were fitted using a two-site kinetic retention model, effectively simulating PENP transport under varying ionic strengths. As the ionic strength of NaCl increased from 0.1 mM to 10 mM, the fitted k values increased from 0.0057 min−1 to 0.0160 min−1 in black soil and from 0.0182 min−1 to 0.0336 min−1 in brown soil (Table 3). These results indicate enhanced deposition rates and consequently reduced mobility of PENPs with increasing ionic strength. The breakthrough curve results align well with DLVO theory predictions, where ionic strength modulates electrostatic repulsion between PENPs and soil particles by compressing the electric double layer. At lower ionic strengths, the thicker diffuse double layer results in stronger electrostatic repulsion, facilitating PENP transport. As ionic strength increases, compression of the diffuse double layer reduces electrostatic repulsion while van der Waals attraction becomes dominant, promoting PENP deposition on soil surfaces and consequently decreasing both mobility and recovery rates. A comparative analysis revealed significantly higher PENP mobility and lower deposition in black soil than in brown soil at equivalent ionic strengths. This discrepancy is attributed to differences in soil physicochemical properties (Table 2). The significantly higher soil organic matter (SOM) content, coupled with its higher pH in black soil, collectively contributes to a greater negative surface charge density, thereby inducing stronger electrostatic repulsion against the negatively charged PENPs, which is supported by our zeta potential measurements and DLVO calculations. This enhanced electrostatic barrier effectively reduces PENP deposition in black soil. Furthermore, the significantly higher SOM and clay content of black soil endow it with a substantially higher CEC compared to brown earth.51 This elevated CEC directly translates to a greater capacity for maintaining negative surface charges, thereby sustaining stronger electrostatic repulsion against PENPs. Second, the particle size distribution plays a synergistic role. Regarding particle size distribution, while the porosity and sand/silt/clay fractions are comparable (Table 2), the specific mineralogy associated with the clay fraction plays a critical role. BS is dominated by 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals which carry a permanent negative charge. In contrast, the clay fraction in brown earth is more likely composed of 1
1-type clay minerals which carry a permanent negative charge. In contrast, the clay fraction in brown earth is more likely composed of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type minerals, which exhibit pH-dependent charge and can even possess positive charges at the observed pH, providing favorable sites for PENP attachment. Therefore, the superior mobility of PENPs in black soil is attributed to the synergistic effect of its favorable chemical conditions, whereas in brown earth, weaker electrostatic repulsion and the presence of attractive sites on the 1
1-type minerals, which exhibit pH-dependent charge and can even possess positive charges at the observed pH, providing favorable sites for PENP attachment. Therefore, the superior mobility of PENPs in black soil is attributed to the synergistic effect of its favorable chemical conditions, whereas in brown earth, weaker electrostatic repulsion and the presence of attractive sites on the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 clay surface act in concert to enhance retention. Collectively, these findings underscore the critical role of ionic strength in governing PENP transport in soil environments, with black soil consistently exhibiting greater PENP mobility across all tested conditions.
1 clay surface act in concert to enhance retention. Collectively, these findings underscore the critical role of ionic strength in governing PENP transport in soil environments, with black soil consistently exhibiting greater PENP mobility across all tested conditions.
3.3.2. Effects of pH.
As shown in Table 3, increasing solution pH from 5 to 9 significantly enhanced PENP recovery rates in both black and brown soils at 1 mM ionic strength. Specifically, recovery rates increased from 53.09% to 83.25% in black soil and from 42.63% to 65.41% in brown soil. This trend aligns with DLVO theory predictions, where higher pH values result in more negative zeta potentials for both PENPs and soil particles. The increased surface charge density thickens the electric double layer (EDL), strengthening electrostatic repulsion and reducing attractive forces between particles. Consequently, the electrostatic repulsion between PENPs and soil particles is strengthened, while attractive forces are reduced, thereby improving the stability of PENPs and inhibiting their deposition on soil surfaces. These effects collectively contribute to the observed increase in recovery rates. Nevertheless, the extent of this enhancement is soil-specific and governed by intrinsic soil properties. The more pronounced increase in recovery rate in BS can be attributed to its greater buffering capacity and higher permanent charge density derived from its 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals and SOM, which sustain strong negative charges across the tested pH range. The lower SOM and prevalence of variable-charge minerals in BE limit the development of negative charge as pH rises. The point of zero charge for these oxides often falls within the tested pH range, meaning that parts of the BE surface may have transitioned from attractive to repulsive, but the overall charge density remains lower than in BS, resulting in a comparatively weaker electrostatic response and lower recovery. Furthermore, an elevated pH promotes the deprotonation of functional groups on PENP surfaces.52 This deprotonation reaction enhances the hydrophilicity and colloidal stability of PENPs in aqueous solutions, further suppressing their deposition in porous media. Analysis of the interaction energy profiles (Fig. 3c and d) demonstrates that higher solution pH increases the zeta potential, elevates the maximum energy barrier (Φmax), and reduces the depth of the secondary energy well (Φmin2). These changes significantly decrease colloidal retention. Therefore, increased pH favors PENP transport through soil columns.53
1-type clay minerals and SOM, which sustain strong negative charges across the tested pH range. The lower SOM and prevalence of variable-charge minerals in BE limit the development of negative charge as pH rises. The point of zero charge for these oxides often falls within the tested pH range, meaning that parts of the BE surface may have transitioned from attractive to repulsive, but the overall charge density remains lower than in BS, resulting in a comparatively weaker electrostatic response and lower recovery. Furthermore, an elevated pH promotes the deprotonation of functional groups on PENP surfaces.52 This deprotonation reaction enhances the hydrophilicity and colloidal stability of PENPs in aqueous solutions, further suppressing their deposition in porous media. Analysis of the interaction energy profiles (Fig. 3c and d) demonstrates that higher solution pH increases the zeta potential, elevates the maximum energy barrier (Φmax), and reduces the depth of the secondary energy well (Φmin2). These changes significantly decrease colloidal retention. Therefore, increased pH favors PENP transport through soil columns.53
The breakthrough curves (Fig. 4c and d) demonstrate a clear pH-dependent enhancement in PENP mobility. In black soil, the maximum relative concentration (C/C0) increased from 63.31% (pH 5) to 92.53% (pH 9), while in brown soil, it rose from 51.96% (pH 5) to 69.92% (pH 9). The fitted kinetic retention model parameters (Table 3) further support these trends, with k values decreasing from 0.0308 to 0.0093 min−1 (BS) and from 0.0418 to 0.0201 min−1 (BE) as pH increased from 5 to 9. All R2 values exceeded 0.90, indicating excellent model performance. At lower pH, the negative charges on the surface of PENPs and soil particles were neutralized by H+, which resulted in a reduction of the electrostatic repulsion between PENPs and the soil, enhancing the effect of mutual aggregation, and thus leading to the deposition of PENPs on the soil surface.54
Therefore, these results collectively demonstrate that the enhanced electrostatic repulsion between PENPs and soil surfaces at higher pH values plays a dominant role in promoting PENP mobility.
3.3.3. Effects of flow rates.
The breakthrough curves of PENPs at different flow rates are shown in Fig. 5. Flow rate significantly influenced the transport of PENPs in porous media. A notable decrease in PENP deposition was observed with increasing flow rate. Table 3 shows that when the flow rate was increased from 1 mL min−1 to 5 mL min−1, the concentration of PENPs in the effluent increased consequently, and the recovery of PE also increased from 74.51% and 51.42% to 88.25% and 62.80% in black soil and brown soil columns, respectively, due to the increase of flow rate. The transport behavior of PENPs exhibited significant flow rate dependence. Different flow rate conditions affect the drag force exerted by water flow on PENPs. At a flow rate of 1 mL min−1, the retention time of colloids at specific locations on the porous medium surface increases, leading to enhanced deposition and stronger attachment between PENPs and the soil due to the secondary energy minimum effect. When the flow rate increases to 5 mL min−1, the drag force may cause the PENPs attached to the soil surface to be released back into the solution, thereby promoting PENP transport and resulting in higher recovery rates.
 |
| | Fig. 5 The BTCs of PENPs in black soil (BS) and brown earth (BE) at different flow rates (at a fixed ionic strength of 1 mM and pH 7). | |
Chun-Han Ko and Menachem Elimelech55 proposed the shadow effect theory, suggesting that surface roughness of sand grains creates significant shadow zones along surface protrusions, which influence colloidal particle deposition. At higher flow rates (5 mL min−1), the increased hydrodynamic drag forces make PENPs less likely to deposit on soil surfaces, while the shadow zones formed on soil surfaces under high flow conditions also hinder PENP deposition. This results in enhanced transport capacity and increased breakthrough rates in soil columns. These findings demonstrate that the mobility of PENPs increases with flow rate, which is consistent with the transport behavior of most flow-affected engineered nanoparticles (ENPs).56
4. Conclusion
This study investigated the transport behavior and underlying mechanisms of PENPs in two distinct soil types under various physicochemical conditions through column experiments, DLVO theory calculations, and model fitting. The results demonstrate that PENPs exhibit greater mobility in black soil than in brown soil under identical physicochemical conditions. This discrepancy is directly linked to the higher pH of black soil and is further supported by its significantly higher organic matter content. These properties elevate the soil's negative surface charge density, thereby enhancing electrostatic repulsion between PENPs and soil particles and reducing deposition. Furthermore, PENP mobility in soils increases with decreasing electrolyte concentration and valence, and increasing pH and flow rate. Beyond the factors investigated in this study, particle size is another critical parameter influencing transport dynamics through mechanisms such as diffusion and straining, which should be systematically investigated in future work. In summary, our findings establish how intrinsic soil properties and solution properties govern PENP transport. This mechanistic understanding, derived under defined laboratory conditions, provides a foundational mechanistic understanding for predicting PENP behavior in more complex settings.
Author contributions
Qiong Liu: writing – review & editing, writing – original draft, visualization, validation, investigation, formal analysis, data curation. Ying Liu: writing – review & editing, validation, visualization, investigation, data curation. Xiaobing Wang: writing – review & editing, supervision, validation, funding acquisition, conceptualization. Jingyi Li: writing – review & editing, validation, data curation. Guangzhen Pang: investigation, data curation. Ke Feng: supervision, funding acquisition, conceptualization. Xiaoli Wang (corresponding author): writing – review & editing, writing – original draft, supervision, validation, funding acquisition, conceptualization.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
All data used in the preparation of this manuscript are included.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 41471236), the Agricultural Science and Technology Innovation Fund of Jiangsu Province (Grant No. CX (20) 3082), and the Postgraduate Research & Practice Innovation Program of Jiangsu Province (Grant No. SJCX24_2266).
References
- M. D. Sharma, A. I. Elanjickal, J. S. Mankar and R. J. Krupadam, Assessment of cancer risk of microplastics enriched with polycyclic aromatic hydrocarbons, J. Hazard. Mater., 2020, 398, 122994 CrossRef CAS.
- C. E. Enyoh, A. W. Verla, E. N. Verla, F. C. Ibe and C. E. Amaobi, Airborne microplastics: a review study on method for analysis, occurrence, movement and risks, Environ. Monit. Assess., 2019, 191, 668 CrossRef CAS PubMed.
- K. Pabortsava and R. S. Lampitt, High concentrations of plastic hidden beneath the surface of the Atlantic Ocean, Nat. Commun., 2020, 11, 4073 CrossRef CAS PubMed.
- Z. Li, C. Feng, Y. Wu and X. Guo, Impacts of nanoplastics on bivalve: Fluorescence tracing of organ accumulation, oxidative stress and damage, J. Hazard. Mater., 2020, 392, 122418 CrossRef CAS PubMed.
- M. Padervand, E. Lichtfouse, D. Robert and C. Wang, Removal of microplastics from the environment. A review, Environ. Chem. Lett., 2020, 18, 807–828 CrossRef CAS.
- K. Zhang, H. Shi, J. Peng, Y. Wang, X. Xiong, C. Wu and P. K. S. Lam, Microplastic pollution in China's inland water systems: A review of findings, methods, characteristics, effects, and management, Sci. Total Environ., 2018, 630, 1641–1653 CrossRef CAS PubMed.
- M. Cole and T. S. Galloway, Ingestion of nanoplastics and microplastics by pacific oyster larvae, Environ. Sci. Technol., 2015, 49, 14625–14632 CrossRef CAS PubMed.
- O. S. Alimi, J. Farner Budarz, L. M. Hernandez and N. Tufenkji, Microplastics and nanoplastics in aquatic environments: Aggregation, deposition, and enhanced contaminant transport, Environ. Sci. Technol., 2018, 52, 1704–1724 CrossRef CAS PubMed.
- A. A. Horton, A. Walton, D. J. Spurgeon, E. Lahive and C. Svendsen, Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities, Sci. Total Environ., 2017, 586, 127–141 CrossRef CAS PubMed.
- A. A. d. S. Machado, C. W. Lau, J. Till, W. Kloas, A. Lehmann, R. Becker and M. C. Rillig, Impacts of microplastics on the soil biophysical environment, Environ. Sci. Technol., 2018, 52, 9656–9665 CrossRef PubMed.
- D. He, Y. Luo, S. Lu, M. Liu, Y. Song and L. Lei, Microplastics in soils: Analytical methods, pollution characteristics and ecological risks, TrAC, Trends Anal. Chem., 2018, 109, 163–172 CrossRef CAS.
- J. Sun, X. Dai, Q. Wang, M. C. M. van Loosdrecht and B.-J. Ni, Microplastics in wastewater treatment plants: Detection, occurrence and removal, Water Res., 2019, 152, 21–37 CrossRef CAS PubMed.
- L. Yongming, Z. Qian, Z. Haibo, P. Xiangliang, T. Chen, L. Lianzhen and Y. Jie, Pay attention to research on microplastic pollution in soil for prevention of ecological and food chain risks, Bull. Chin. Acad. Sci., 2018, 33, 1021–1030 Search PubMed.
- P. He, L. Chen, L. Shao, H. Zhang and F. Lu, Municipal solid waste (MSW) landfill: A source of microplastics? - Evidence of microplastics in landfill leachate, Water Res., 2019, 159, 38–45 CrossRef CAS.
- D. L. Jones, K. L. Williamson and A. G. Owen, Phytoremediation of landfill leachate, Waste Manage., 2006, 26, 825–837 CrossRef CAS PubMed.
- R. Qi, D. L. Jones, Z. Li, Q. Liu and C. Yan, Behavior of microplastics and plastic film residues in the soil environment: A critical review, Sci. Total Environ., 2020, 703, 134722 CrossRef CAS PubMed.
-
M.-H. Yu and S.-Q. Yu, Introduction to Unified Strength Theory, CRC Press, 2019 Search PubMed.
- J. P. da Costa, P. S. M. Santos, A. C. Duarte and T. Rocha-Santos, (Nano)plastics in the environment - Sources, fates and effects, Sci. Total Environ., 2016, 566, 15–26 CrossRef.
- X. Guo, G. Hu, X. Fan and H. Jia, Sorption properties of cadmium on microplastics: The common practice experiment and a two-dimensional correlation spectroscopic study, Ecotoxicol. Environ. Saf., 2020, 190, 110118 CrossRef CAS PubMed.
- S. E. Nelms, T. S. Galloway, B. J. Godley, D. S. Jarvis and P. K. Lindeque, Investigating microplastic trophic transfer in marine top predators, Environ. Pollut., 2018, 238, 999–1007 CrossRef CAS PubMed.
- T. Atugoda, H. Piyumali, H. Wijesekara, C. Sonne, S. S. Lam, K. Mahatantila and M. Vithanage, Nanoplastic occurrence, transformation and toxicity: a review, Environ. Chem. Lett., 2023, 21, 363–381 CrossRef CAS.
- X. Ye, Z. Cheng, M. Wu, Y. Hao, B. X. Hu, C. Mo and G. Lu, Investigating transport kinetics of polystyrene nanoplastics in saturated porous media, Ecotoxicol. Environ. Saf., 2022, 241, 113820 CrossRef CAS PubMed.
- L. He, H. Rong, D. Wu, M. Li, C. Wang and M. Tong, Influence of biofilm on the transport and deposition behaviors of nano- and micro-plastic particles in quartz sand, Water Res., 2020, 178, 115808 CrossRef CAS PubMed.
- Y. Jiang, X. Yin, X. Xi, D. Guan, H. Sun and N. Wang, Effect of surfactants on the transport of polyethylene and polypropylene microplastics in porous media, Water Res., 2021, 196, 117016 CrossRef CAS PubMed.
- C. Rieckhof, V. Martinez-Hernandez, E. Holzbecher and R. Meffe, Effect of particle size on the transport of polystyrene micro- and nanoplastic particles through quartz sand under unsaturated conditions, Environ. Pollut., 2024, 363, 125193 CrossRef CAS PubMed.
- X. Ling, Z. Yan and G. Lu, Vertical transport and retention behavior of polystyrene nanoplastics in simulated hyporheic zone, Water Res., 2022, 219, 118609 CrossRef CAS PubMed.
- D. He, X. Zhang and J. Hu, Methods for separating microplastics from complex solid matrices: Comparative analysis, J. Hazard. Mater., 2021, 409, 124640 CrossRef CAS PubMed.
- D. Shaniv, I. Dror and B. Berkowitz, Effects of particle size and surface chemistry on plastic nanoparticle transport in saturated natural porous media, Chemosphere, 2021, 262, 127854 CrossRef CAS PubMed.
- H. Bouwmeester, P. C. H. Hollman and R. J. B. Peters, Potential health impact of environmentally released micro- and nanoplastics in the human food production chain: Experiences from nanotoxicology, Environ. Sci. Technol., 2015, 49, 8932–8947 CrossRef CAS PubMed.
- Z. Song, X. Yang, F. Chen, F. Zhao, Y. Zhao, L. Ruan and Y. Yang,
et al., Fate and transport of nanoplastics in complex natural aquifer media: Effect of particle size and surface functionalization, Sci. Total Environ., 2019, 669, 120–128 CrossRef CAS PubMed.
-
Z. T. Gong, G. L. Zhang and Z. C. Chen, Development of Soil Classification in China, in Soil Classification, CRC Press, 2002, 101–126 Search PubMed.
-
Z. D. L. Sparks, B. Singh and M. G. Siebecker, Environmental Soil Chemistry, Elsevier, 2022 Search PubMed.
- A. A. Koelmans, A. Bakir, G. A. Burton and C. R. Janssen, Microplastic as a vector for chemicals in the aquatic environment: critical review and model-supported reinterpretation of empirical studies, Environ. Sci. Technol., 2016, 50, 3315–3326 CrossRef CAS PubMed.
- Y. Luo, Y. Zhang, Y. Xu, X. Guo and L. Zhu, Distribution characteristics and mechanism of microplastics mediated by soil physicochemical properties, Sci. Total Environ., 2020, 726, 138389 CrossRef CAS PubMed.
- W. Wang and J. Wang, Comparative evaluation of sorption kinetics and isotherms of pyrene onto microplastics, Chemosphere, 2018, 193, 567–573 CrossRef CAS PubMed.
- S. Dong, J. Xia, L. Sheng, W. Wang, H. Liu and B. Gao, Transport characteristics
of fragmental polyethylene glycol terephthalate (PET) microplastics in porous media under various chemical conditions, Chemosphere, 2021, 276, 130214 CrossRef CAS PubMed.
- H. Sun, B. Gao, Y. Tian, X. Yin, C. Yu, Y. Wang and L. Q. Ma, Kaolinite and lead in saturated porous media: Facilitated and impeded transport, J. Environ. Eng., 2010, 136, 1305–1308 CrossRef CAS.
- X. Yin, B. Gao, L. Q. Ma, U. K. Saha, H. Sun and G. Wang, Colloid-facilitated Pb transport in two shooting-range soils in Florida, J. Hazard. Mater., 2010, 177, 620–625 CrossRef CAS PubMed.
- H. Bai, N. Cochet, A. Drelich, A. Pauss and E. Lamy, Comparison of transport between two bacteria in saturated porous media with distinct pore size distribution, RSC Adv., 2016, 6, 14602–14614 RSC.
- B. V. Derjaguin, Theory of the stability of strongly charged lyophobic sol and of the adhesion of strongly charged particles in solutions of electrolytes, Acta Physicochim. USSR, 1941, 14, 633 Search PubMed.
-
E. J. W. Verwey and J. T. G. Overbeek, Theory of the Stability of Lyophobic Colloids, Elsevier, 1955 Search PubMed.
- S. Dong, Y. Sun, B. Gao, X. Shi, H. Xu, J. Wu and J. Wu, Retention and transport of graphene oxide in water-saturated limestone media, Chemosphere, 2017, 180, 506–512 CrossRef CAS PubMed.
- M. Wu, H. Cao, G. Lu, B. X. Hu, Z. Cheng, C. Mo and J. Wu,
et al., Effects of nanometer alumina and humic acid on the retention and transport of hexavalent chromium in porous media, Ecotoxicol. Environ. Saf., 2021, 228, 113005 CrossRef CAS PubMed.
- A. Strålin and T. Hjertberg, FTIR Study on Interfacial Interactions Between Hydrated Aluminium and Polar Groups in Ethylene Copolymers, Surf. Interface Anal., 1993, 20, 337–340 CrossRef.
- X. Wu, X. Lyu, Z. Li, B. Gao, X. Zeng, J. Wu and Y. Sun, Transport of polystyrene nanoplastics in natural soils: Effect of soil properties, ionic strength and cation type, Sci. Total Environ., 2020, 707, 136065 CrossRef CAS PubMed.
- J. Hou, X. Xu, L. Lan, L. Miao, Y. Xu, G. You and Z. Liu, Transport behavior of micro polyethylene particles in saturated quartz sand: Impacts of input concentration and physicochemical factors, Environ. Pollut., 2020, 263, 114499 CrossRef CAS PubMed.
- Z. Dong, Y. Qiu, W. Zhang, Z. Yang and L. Wei, Size-dependent transport and retention of micron-sized plastic spheres in natural sand saturated with seawater, Water Res., 2018, 143, 518–526 CrossRef CAS PubMed.
- X. Xi, L. Wang, T. Zhou, J. Yin, H. Sun, X. Yin and N. Wang, Effects of physicochemical factors on the transport of aged polystyrene nanoparticles in saturated porous media, Chemosphere, 2022, 289, 133239 CrossRef CAS PubMed.
- S. Li, H. Liu, R. Gao, A. Abdurahman, J. Dai and F. Zeng, Aggregation kinetics of microplastics in aquatic environment: Complex roles of electrolytes, pH, and natural organic matter, Environ. Pollut., 2018, 237, 126–132 CrossRef CAS PubMed.
- T. Xia, Y. Qi, J. Liu, Z. Qi, W. Chen and M. R. Wiesner, Cation-inhibited transport of graphene oxide nanomaterials in saturated porous media: The Hofmeister effects, Environ. Sci. Technol., 2017, 51, 828–837 CrossRef CAS PubMed.
-
N. C. Brady and R. R. Weil, The Nature and Properties of Soils, Pearson Education, Columbus, 2016, pp. 328–372 Search PubMed.
- M. Parvinzadeh, S. Moradian, A. Rashidi and M.-E. Yazdanshenas, Surface characterization of polyethylene terephthalate/silica nanocomposites, Appl. Surf. Sci., 2010, 256, 2792–2802 CrossRef CAS.
- V. L. Morales, W. Zhang, B. Gao, L. W. Lion, J. J. Bisogni Jr, B. A. McDonough and T. S. Steenhuis, Impact of dissolved organic matter on colloid transport in the vadose zone: Deterministic approximation of transport deposition coefficients from polymeric coating characteristics, Water Res., 2011, 45, 1691–1701 CrossRef CAS PubMed.
- J. L. Renaux, M. Thomas, T. Crost, N. Loughraieb and G. Vantard, Activation of the kallikrein-kinin system in hemodialysis: Role of membrane electronegativity, blood dilution, and pH, Kidney Int., 1999, 55, 1097–1103 CrossRef CAS PubMed.
- C. H. Ko and M. Elimelech, The “shadow effect” in colloid transport and deposition dynamics in granular porous media: Measurements and mechanisms, Environ. Sci. Technol., 2000, 34, 3681–3689 CrossRef CAS.
- X. Liu, D. M. O'Carroll, E. J. Petersen, Q. Huang and C. L. Anderson, Mobility of multiwalled carbon nanotubes in porous media, Environ. Sci. Technol., 2009, 43, 8153–8158 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  a,
Ying
Liu
a,
Xiaobing
Wang
ab,
Jingyi
Li
a,
Guangzhen
Pang
a,
Ke
Feng
ab and
Xiaoli
Wang
*ab
a,
Ying
Liu
a,
Xiaobing
Wang
ab,
Jingyi
Li
a,
Guangzhen
Pang
a,
Ke
Feng
ab and
Xiaoli
Wang
*ab
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals. In contrast, BE generally possesses lower organic matter content, is more acidic, and its variable charge is influenced by 1
1-type clay minerals. In contrast, BE generally possesses lower organic matter content, is more acidic, and its variable charge is influenced by 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals and iron/aluminum oxides. The fundamental differences in charge properties between these soil types are well-established in soil colloid chemistry.32 We hypothesize that these stark contrasts, particularly in surface charge density and solution chemistry, will lead to profoundly different electrostatic interactions with negatively charged PENPs. This comparison not only deciphers the key soil properties controlling nanoplastic mobility but also provides a mechanistic basis for understanding the fate of PENPs across a wide range of environmental conditions. PENPs have emerged as critical environmental contaminant vectors due to their unique physicochemical properties. These ultra-small particles possess exceptionally high specific surface areas and pronounced hydrophobicity, endowing them with remarkable adsorption capacities. PENPs efficiently concentrate heavy metal ions and organic pollutants, functioning as “pollutant shuttles” that amplify contaminant dispersion and ecological risks.33–35 Consequently, elucidating the transport mechanisms of nanoplastics in soils is imperative for accurately assessing their environmental fate and predicting associated risks.
1-type clay minerals and iron/aluminum oxides. The fundamental differences in charge properties between these soil types are well-established in soil colloid chemistry.32 We hypothesize that these stark contrasts, particularly in surface charge density and solution chemistry, will lead to profoundly different electrostatic interactions with negatively charged PENPs. This comparison not only deciphers the key soil properties controlling nanoplastic mobility but also provides a mechanistic basis for understanding the fate of PENPs across a wide range of environmental conditions. PENPs have emerged as critical environmental contaminant vectors due to their unique physicochemical properties. These ultra-small particles possess exceptionally high specific surface areas and pronounced hydrophobicity, endowing them with remarkable adsorption capacities. PENPs efficiently concentrate heavy metal ions and organic pollutants, functioning as “pollutant shuttles” that amplify contaminant dispersion and ecological risks.33–35 Consequently, elucidating the transport mechanisms of nanoplastics in soils is imperative for accurately assessing their environmental fate and predicting associated risks.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 (w/v).
2.5 (w/v).
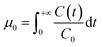
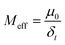
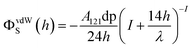
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln[1 + exp(−kh)]
ln[1 + exp(−kh)]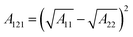
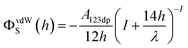
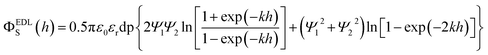
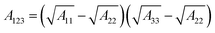

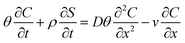
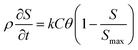



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals which carry a permanent negative charge. In contrast, the clay fraction in brown earth is more likely composed of 1
1-type clay minerals which carry a permanent negative charge. In contrast, the clay fraction in brown earth is more likely composed of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type minerals, which exhibit pH-dependent charge and can even possess positive charges at the observed pH, providing favorable sites for PENP attachment. Therefore, the superior mobility of PENPs in black soil is attributed to the synergistic effect of its favorable chemical conditions, whereas in brown earth, weaker electrostatic repulsion and the presence of attractive sites on the 1
1-type minerals, which exhibit pH-dependent charge and can even possess positive charges at the observed pH, providing favorable sites for PENP attachment. Therefore, the superior mobility of PENPs in black soil is attributed to the synergistic effect of its favorable chemical conditions, whereas in brown earth, weaker electrostatic repulsion and the presence of attractive sites on the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 clay surface act in concert to enhance retention. Collectively, these findings underscore the critical role of ionic strength in governing PENP transport in soil environments, with black soil consistently exhibiting greater PENP mobility across all tested conditions.
1 clay surface act in concert to enhance retention. Collectively, these findings underscore the critical role of ionic strength in governing PENP transport in soil environments, with black soil consistently exhibiting greater PENP mobility across all tested conditions.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-type clay minerals and SOM, which sustain strong negative charges across the tested pH range. The lower SOM and prevalence of variable-charge minerals in BE limit the development of negative charge as pH rises. The point of zero charge for these oxides often falls within the tested pH range, meaning that parts of the BE surface may have transitioned from attractive to repulsive, but the overall charge density remains lower than in BS, resulting in a comparatively weaker electrostatic response and lower recovery. Furthermore, an elevated pH promotes the deprotonation of functional groups on PENP surfaces.52 This deprotonation reaction enhances the hydrophilicity and colloidal stability of PENPs in aqueous solutions, further suppressing their deposition in porous media. Analysis of the interaction energy profiles (Fig. 3c and d) demonstrates that higher solution pH increases the zeta potential, elevates the maximum energy barrier (Φmax), and reduces the depth of the secondary energy well (Φmin2). These changes significantly decrease colloidal retention. Therefore, increased pH favors PENP transport through soil columns.53
1-type clay minerals and SOM, which sustain strong negative charges across the tested pH range. The lower SOM and prevalence of variable-charge minerals in BE limit the development of negative charge as pH rises. The point of zero charge for these oxides often falls within the tested pH range, meaning that parts of the BE surface may have transitioned from attractive to repulsive, but the overall charge density remains lower than in BS, resulting in a comparatively weaker electrostatic response and lower recovery. Furthermore, an elevated pH promotes the deprotonation of functional groups on PENP surfaces.52 This deprotonation reaction enhances the hydrophilicity and colloidal stability of PENPs in aqueous solutions, further suppressing their deposition in porous media. Analysis of the interaction energy profiles (Fig. 3c and d) demonstrates that higher solution pH increases the zeta potential, elevates the maximum energy barrier (Φmax), and reduces the depth of the secondary energy well (Φmin2). These changes significantly decrease colloidal retention. Therefore, increased pH favors PENP transport through soil columns.53


