Atmospheric oxidation of hydroxymethyl methyl α-lactone (HMML), initiated by OH radicals and Cl atoms
Samsung Raja
Daimari
 ,
Rabu Ranjan
Changmai
,
Rabu Ranjan
Changmai
 and
Manabendra
Sarma
and
Manabendra
Sarma
 *
*
Department of Chemistry, Indian Institute of Technology, Guwahati, Assam 781039, India. E-mail: msarma@iitg.ac.in
First published on 20th November 2025
Abstract
Isoprene, the most predominant biogenically emitted volatile organic compound (VOC) in the atmosphere, undergoes oxidation to yield hydroxymethyl methyl α-lactone (HMML) in the presence of NO. While the particle-phase chemistry of HMML has been explored to some extent, its gas-phase chemistry remains unexplored. In this study, we have performed an extensive computational investigation involving the thermodynamics and kinetics of the oxidation reaction of HMML in the gas phase, driven by hydroxy radicals (OH) and chlorine (Cl) atoms. The oxidation of HMML proceeds via hydrogen atom abstraction reactions, all of which are exothermic in nature. The potential energy profile diagram representing all possible reaction routes has been constructed using CCSD(T)//M06-2X/6-311++G(d,p) level of theory. The rate coefficients of all the reaction pathways were estimated using the variational transition state theory (VTST), corrected with a tunneling factor, across the 200 to 400 K temperature range at the M06-2X/6-311++G(d,p) level of theory. At 298 K, the atmospheric lifetime of HMML was determined to be 9.6 days in the marine boundary layer (MBL) and 62.5 days in the normal tropospheric conditions. Furthermore, the degradation study of the product radicals revealed various end products with much higher reactivity and shorter lifetimes, such as diketones, α, β-unsaturated carboxylic acids, formic acid, pyruvic acid, methylglyoxal, and dioxopropanoic acid.
Environmental significanceThis work emphasizes the gas phase oxidation of hydroxymethyl methyl α-lactone (HMML), a crucial byproduct of isoprene oxidation under high NOx conditions. While the particle-phase chemistry of HMML has been relatively well studied, its gas-phase oxidation remains largely unexplored. We have addressed this critical knowledge gap through high-level quantum mechanical calculations and kinetic studies. Our results show that HMML readily reacts with OH radicals and Cl atoms, resulting in a short atmospheric lifetime of 9.6 days under marine boundary layer (MBL) conditions. These findings reveal that gas phase oxidation of HMML can compete with heterogeneous uptake and therefore, should be considered in atmospheric modelling. The oxidation of the product radicals formed from the H-abstraction of HMML can result in highly reactive intermediates such as diketones, α,β-unsaturated aldehydes, pyruvic acid, methylglyoxal, formic acid, and dioxopropanoic acid, which can further contribute to secondary organic aerosol (SOA) formation. Our study improves the understanding of the gasphase oxidation of secondary volatile organic compounds and highlights the broader effect of biogenically emitted species undergoing chemical reactions in the atmosphere. |
1 Introduction
Isoprene (2-methyl-1,3-butadiene) is one of the major biogenically produced volatile organic compounds (VOCs) in the atmosphere. It is estimated that 500 to 750 Tg of isoprene are released into the air each year.1,2 Due to the high reactivity of Isoprene, it undergoes oxidation with various atmospheric oxidants such as hydroxyl radicals (OH), chlorine (Cl) atoms, nitrate radicals (NO3), and ozone (O3), among which the primary daytime sink is the reaction with OH radicals. Isoprene is a primary source of secondary organic aerosols (SOA), ground-level ozone, and other trace organic chemicals due to its high reactivity and abundance.3–6In the atmosphere, depending upon the conditions (under low or high NOx conditions), isoprene primarily reacts with OH radicals during daytime and forms various end products. Under low NOx conditions, isoprene undergoes oxidation to form isoprene epoxydiols (IEPOX), a known precursor to SOA.7–10 While under high NOx conditions, isoprene oxidation follows a different pathway, forming methacrolein (MACR), which oxidizes to form methacryloyl peroxynitrate (MPAN). Further, MPAN undergoes OH radical-initiated oxidation to produce hydroxymethyl methyl-α-lactone (HMML).11–13 The oxidation of MPAN by hydroxyl radicals in high NOx conditions can yield as much as 75% HMML + NO3.13
HMML is a highly reactive three-member lactone ring with hydroxymethyl and methyl substituents attached directly to the ring. Once formed in the atmosphere, HMML can undergo hydrolysis or remain in the gas phase and react with various oxidants such as OH radicals and Cl atoms. Experimental and modelling studies have shown that HMML can undergo heterogeneous uptake in the particle phase under both aqueous and acidic conditions, forming relatively lower volatile compounds like 2-methylglyceric acid (2-MGA), 2-methyltetrols (2-MT), and 2-methylglyceric acid organosulfates (2-MGAOS).14–17
Although the particle-phase chemistry of HMML has been well documented, there has been no direct study of the gas-phase chemistry of HMML. The interaction of HMML with atmospheric oxidants such as hydroxy radicals and chlorine atoms could result in significant gas-phase pathway loss, particularly in dry conditions. The gas-phase oxidation of HMML could lead to smaller fragmented molecules, such as hydroxy acetone, with higher volatility.18 The gas-phase reaction of HMML with OH radicals is particularly important because OH radicals are the primary oxidant in the tropospheric oxidation reaction. OH radicals are the primary sink for most volatile organic compounds in the troposphere, affecting their lifetime and product formation.6,19,20 Likewise, Cl atoms, despite being lower in concentration than OH radicals in the atmosphere, can play a crucial role in the oxidation of VOCs near the marine boundary layer (MBL). In the coastal and marine environment, the concentration of Cl atoms becomes much more significant, acting as a major sink for VOCs and influencing the halogen cycle.21–23
This work presents the first comprehensive study of gas-phase oxidation of HMML's initiated by OH radicals and Cl atoms, addressing a critical gap in the literature where previous studies focus only on particle-phase reactions.14–17 The gas-phase oxidation of HMML may present a competitive loss pathway, diverting it from heterogeneous uptake. Therefore, it is essential to understand the kinetics and mechanism of the reaction of HMML with OH radicals and Cl atoms to determine its atmospheric fate, contribution to SOA formation, atmospheric lifetime, and the broader implications for air quality. Thus, we have used high-level quantum mechanical calculations, CCSD(T) and M06-2X, to calculate thermochemistry of the reaction pathways and calculated rate coefficients with variational transition state theory (VTST) and tunneling corrections to provide accurate kinetic data across an atmospheric-relevant temperature range of 200–400 K. The rate coefficients of each reaction pathway, along with their atmospheric lifetimes, were calculated within the investigated temperature range. In addition, the most plausible degradation pathways of the product radicals formed from the initial reaction have also been investigated, identifying highly reactive and atmospherically relevant products. We believe that this study enhances the understanding of the fate of HMML in atmospheric oxidation, emphasising its impact on atmospheric chemistry and environmental processes.
2 Computational details
In this investigation, the Gaussian 16 software program was used to perform all of the quantum mechanical calculations related to the gas phase oxidation of HMML.24,25 All the geometry of the compounds, including reactants, pre-reaction complexes (CRs), transition states (TSs), post-reaction complexes (PRC), and products, were optimised using M06-2X meta-hybrid density functional26,27 combined with Pople's basis set 6-311++G(d,p).28,29 The harmonic vibrational frequency calculations were performed in order to describe the characteristics of the stationary point that was obtained. The presence of only positive frequencies indicated stable minima, while the presence of one imaginary frequency indicated transition states. A vibrational scaling factor of 0.98 was used to at the M06-2X/6-311++G(d,p) level of theory to correct the overestimated vibrational frequencies.30,31 Further, intrinsic reaction coordinate (IRC)32,33 calculations were carried out to validate the connectivity of the identified transition states between the respective reactants and products. The M06-2X functional in conjunction with 6-311++G(d,p) basis was selected for this study because of its reliability and accuracy in studying the thermochemistry of atmospheric oxidation reactions as reported by previous studies in the literature.27,34–38In order to enhance computational accuracy, the energies of all the geometries obtained from the M06-2X/6-311++G(d,p) related to the oxidation reaction of HMML were refined via single-point calculations using the coupled cluster method with singles, doubles and perturbative triple excitation [CCSD(T)]39,40 level of theory in combination with the same basis set of 6-311++G(d,p). To assess whether the obtained geometries exhibit multireference character, T1-diagnostic values were evaluated. For the single-reference calculation, all of the values were determined to be below 0.02 and 0.045 for closed and open shell systems, respectively, indicating their reliability.41 The Cartesian coordinates and vibrational frequencies of the transition states, as well as T1-diagnostic, the energies, and corrected electronic energies for all stationary points, are presented in Tables S1–S4 of the SI.
3 Results and discussion
The structure of HMML has a hydroxy methyl substituent bonded to the α-lactone ring, which can rotate and have more than one conformation existing in thermal equilibrium. To identify the stable minima, we conducted a potential energy (PE) scan by simultaneously rotating two dihedral angles: the O–C–C–O torsion connecting the α-lactone ring to the hydroxymethyl group, and the C–C–O–H torsion of the hydroxyl group. From the PE scan, shown in Fig. S1, we found multiple minima, among which two have the lowest energy (separated by 0.34 kcal mol−1) and can exist in thermal equilibrium. Therefore, we have carried out our further studies using these two minima. The optimized structures of HMML-I (S-I), the most stable conformer, and HMML-II (S-II) are shown in Fig. 1. | ||
| Fig. 1 Optimized structure of (a) HMML-I (S-I) and (b) HMML-II (S-II) at the M06-2X/6-311++G(d,p) level of theory. | ||
3.1 Oxidation reaction pathways of S-I with OH radicals and Cl atoms
The oxidation reaction of S-I with OH radicals and Cl atoms occurs mainly through the H-abstraction reaction; no direct addition to the α-lactone ring could be found. From Fig. 2, we can see five different abstractable hydrogens, three associated with the methyl group and two with the hydroxymethyl group. From Fig. 2, it can be inferred that all H-abstraction reactions for both S-I + ˙OH and S-I + Cl are feasible and exothermic.The hydrogen atom abstraction from the methyl group (R1-Ia/Ib to R3-Ia/Ib) proceeds via three transition states, TS1-Ia/Ib to TS3-Ia/Ib, leading to the product radical P1 along with byproducts H2O and HCl. Moreover, the two hydrogen atoms are abstracted from the hydroxymethyl group (R4-Ia/Ib and R5-Ia/Ib), leading to the formation of product radical P2, also producing H2O and HCl, via the formation of transition states TS4-Ia/Ib and TS5-Ia/Ib.All the reaction channels R1-Ia/Ib to R5-Ia/Ib result in the ring opening of the α-lactone. Furthermore, in the case of R4-Ia/Ib and R5-Ia/Ib, an additional intramolecular hydrogen atom is transferred from the hydroxymethyl group's O–H to the α-lactone group's C–O, resulting in a comparatively more stable product radical P2.
We have also constructed the potential energy (PE) profile for each reaction pathway involved in the S-I + ˙OH and S-I + Cl reactions. The PE profiles were constructed using the energies obtained from single-point calculations at the CCSD(T)//M06-2X/6-311++G(d,p) level of theory shown in Fig. 3. Moreover, due to spin–orbit coupling (SOC) effects, the electronic levels of Cl atoms are split into two states: 2P3/2 (ground state, degeneracy = 4) and 2P1/2 (excited state, degeneracy = 2), which are separated by 882.36 cm−1. The ground state lies 0.001338 hartree or 0.84 kcal mol−1 below the spin-averaged state. Thus, a correction of 0.84 kcal mol−1 has been used to construct the PE of HMML + Cl in order to get the correct PE energy profile.42,43
From the depicted PE profiles, we can see that all the reaction pathways involve the creation of loosely bound pre-reaction complexes (CRs), which are more stable than the separated individual reactants due to weak van der Waals interactions. The optimized geometries involved in these reactions are shown in Fig. S2.
Three hydrogen atoms are abstracted from the methyl group (R1-Ia to R3-Ia) with OH radicals, occurring via three different, distinctive transition states; TS1-Ia to TS3-Ia, leading to the formation of product radical P1 along with H2O. In contrast, for the S-I reaction with the Cl atoms, the hydrogen abstraction reaction pathways, R1-Ib and R3-Ib, share the same transition states, TS1-Ib and TS3-Ib. Meanwhile, the reaction pathway R2-Ib follows a different route via the formation of transition state TS2-Ib.
Additionally, the other two hydrogen abstraction reaction pathways (from the hydroxymethyl group), R4-Ia/Ib and R5-Ia/Ib, are characterized by the formation of distinctive transition states TS4-Ia/Ib and TS5-Ia/Ib, respectively. Moreover, from Fig. 3, we can see that the reaction pathway R5-Ia/Ib involves the transition states with the lowest energy due to the relatively lower bond dissociation energy of the C–H bond of the hydroxymethyl group, suggesting it to be the major reaction pathway. On the other hand, the reaction pathways R1-Ia/Ib have the highest energy barriers for both S-I + ˙OH and S-I + Cl.
3.2 Oxidation reaction pathways of S-II with OH radicals and Cl atoms
Similar to the S-I reaction with OH radicals and Cl atoms, the oxidation reaction of S-II occurs via five different hydrogen abstraction reactions; three associated with the methyl group and two with the hydroxymethyl group. All the reactions involved in S-II + ˙OH and S-II + Cl are feasible and exothermic in nature, as shown in Fig. 4.The reaction pathways R1-IIa/IIb to R3-IIa/IIb occur through the formation of transition states TS1-IIa/IIb to TS3-IIa/IIb, leading to product radical P3, along with H2O and HCl. Unlike the oxidation reaction of S-I, the reaction pathways R4-IIa and R5-IIa/IIb do not undergo intramolecular hydrogen atom shift from the O–H of the hydroxymethyl group to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O of the α-lactone group. Specifically, for the R4-IIa reaction pathway, it is primarily due to the weak hydrogen bonding linking the hydroxy radical and the α-lactone's oxygen atom, as well as the hydrogen bonding between the leaving group H2O with the O–H of the hydroxy group (TS4-IIa and PC4-IIa in Fig. S2), which prevents intramolecular hydrogen transfer. In the case of R5-IIa/IIb, it is due to the orientation of the transition state TS5-IIa/IIb (shown in Fig. S2), where the O–H group is on the opposite side of the C
O of the α-lactone group. Specifically, for the R4-IIa reaction pathway, it is primarily due to the weak hydrogen bonding linking the hydroxy radical and the α-lactone's oxygen atom, as well as the hydrogen bonding between the leaving group H2O with the O–H of the hydroxy group (TS4-IIa and PC4-IIa in Fig. S2), which prevents intramolecular hydrogen transfer. In the case of R5-IIa/IIb, it is due to the orientation of the transition state TS5-IIa/IIb (shown in Fig. S2), where the O–H group is on the opposite side of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, thereby restricting hydrogen atom transfer. The reaction pathways R4-IIa and R5-IIa/IIb lead to product radical P4.
O, thereby restricting hydrogen atom transfer. The reaction pathways R4-IIa and R5-IIa/IIb lead to product radical P4.
The same is not the case for the R4-IIb reaction pathway, where there is intramolecular hydrogen transfer. This is primarily because there is no hydrogen bonding between the Cl atoms and the α-lactone ring (TS4-IIb and PC4-IIb in Fig. S2), thereby leading to the formation of the product radical P2.
The PE profiles for each of the reaction pathways involved in the S-II + ˙OH and S-II + Cl are represented in Fig. 5, constructed using the energies obtained from single-point calculations at the CCSD(T)//M06-2X/6-311++G(d,p) level of theory. Similar to S-I + Cl, the PE for S-II + Cl was also corrected for spin–orbit coupling.
Each reaction pathway initiates with the formation of pre-reaction complexes (CRs). These reactions involve three hydrogen atoms' abstraction from the methyl group by OH radicals via three distinct transition states, TS1-IIa to TS3-IIb, resulting in product radical P3. In contrast, similar to S-I + Cl, the reactions involving pathways R1-IIb and R3-IIb share the same transition states TS1-IIb and TS3-IIb, while pathway R2-IIb proceeds via transition state TS2-IIb, forming product radical P3. The hydrogen abstraction from the hydroxymethyl group yields two pathways, R4-IIa/IIb and R5-IIa/IIb, each characterized by unique transition states (TS4-IIa/IIb and TS5-IIa/IIb). Notably, the R4-IIa reaction pathway has the lowest energy barrier for S-II + ˙OH, while for S-II + Cl, the R5-IIb reaction pathway has the lowest energy barrier.
3.3 Kinetics study
The rate coefficients of the oxidation pathways were computed using the variational transition state theory (VTST)44,45 along with Eckart46,47 in the KisthelP software.48 The Eckart tunneling, despite its limitations compared to multidimensional tunneling, still offers reasonably accurate kinetics results and is still being widely used.49–51 The VTST corrected with the tunneling factor incorporated in KisthelP software is shown below in eqns. 1 and 2.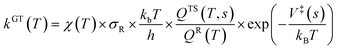 | (1) |
| kVTST = minskGT(T,s) | (2) |
The contributions of both conformers, S-I and S-II, were included in the calculation of the total rate coefficient for the reaction of HMML with OH radicals and Cl atoms. Thus, we calculated the population distribution of these conformers across 200–400 K temperature range and weighted their overall contribution accordingly. The Boltzmann distribution expressed as a function of Gibbs free energy is represented in eqn (3).52
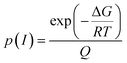 | (3) |
| kn = kS-I(n) × p(S-I) + kS-II(n) × p(S-II) | (4) |
The overall individual and total rate coefficients (S-I + S-II) for the HMML + ˙OH are given in Table 1. From Table 1, we can see that as the temperature increases, the rate coefficient decreases, indicating negative temperature dependency. The negative temperature dependency of the rate coefficient is a relatively common occurrence in atmospheric oxidation reactions due to low-lying transition states and the formation of pre-reaction complexes, which are thermodynamically favoured at low temperatures.53
| Temp (K) | k 1 | k 2 | k 3 | k 4 | k 5 | k overall |
|---|---|---|---|---|---|---|
| 200 | 3.35 × 10−16 | 1.04 × 10−14 | 2.42 × 10−16 | 7.18 × 10−15 | 1.45 × 10−13 | 1.64 × 10−13 |
| 220 | 4.09 × 10−16 | 1.17 × 10−14 | 3.01 × 10−16 | 8.04 × 10−15 | 1.05 × 10−13 | 1.25 × 10−13 |
| 240 | 4.93 × 10−16 | 1.30 × 10−14 | 3.68 × 10−16 | 8.87 × 10−15 | 7.93 × 10−14 | 1.02 × 10−13 |
| 260 | 5.85 × 10−16 | 1.43 × 10−14 | 4.43 × 10−16 | 9.69 × 10−15 | 6.25 × 10−14 | 8.75 × 10−14 |
| 280 | 6.84 × 10−16 | 1.55 × 10−14 | 5.25 × 10−16 | 1.05 × 10−14 | 5.08 × 10−14 | 7.80 × 10−14 |
| 298 | 7.80 × 10−16 | 1.64 × 10−14 | 6.04 × 10−16 | 1.12 × 10−14 | 4.32 × 10−14 | 7.22 × 10−14 |
| 320 | 9.05 × 10−16 | 1.77 × 10−14 | 7.08 × 10−16 | 1.21 × 10−14 | 3.63 × 10−14 | 6.76 × 10−14 |
| 340 | 1.02 × 10−15 | 1.89 × 10−14 | 8.09 × 10−16 | 1.27 × 10−14 | 3.16 × 10−14 | 6.50 × 10−14 |
| 360 | 1.15 × 10−15 | 2.01 × 10−14 | 9.16 × 10−16 | 1.34 × 10−14 | 2.79 × 10−14 | 6.35 × 10−14 |
| 380 | 1.28 × 10−15 | 2.14 × 10−14 | 1.03 × 10−15 | 1.41 × 10−14 | 2.51 × 10−14 | 6.29 × 10−14 |
| 400 | 1.42 × 10−15 | 2.26 × 10−14 | 1.14 × 10−15 | 1.48 × 10−14 | 2.28 × 10−14 | 6.27 × 10−14 |
The rate coefficient obtained at 298 K, 7.22 × 10−14 cm3 per molecule per s, is well within the trend of the experimental and computational study done by Barnes et al.54 They carried out an oxidation reaction study of five lactones, namely: β-butyrolactone, γ-butyrolactone, γ-valeroalactone, α-methyl-γ-butyrolactone, and δ-valero-lactone, with OH radicals. Their study concluded that the rate coefficient decreases with the decrease in size of the lactone ring. They found that the rate coefficient at 298 K decreases from (5.24 ± 0.63) × 10−12 cm3 per molecule per s for δ-valero-lactones, to (9.7 ± 0.26) × 10−13 cm3 per molecule per s for β-butyrolactone. The rate coefficient of HMML (three-membered lactone) with OH radicals obtained in our calculation is the lowest among the five lactones studied by Barnes et al.,54 thus agreeing with their trend.
The branching ratios calculation (shown in Fig. S3), determined by dividing the individual rate coefficients by the overall rate coefficient, shows that at the lower temperature, the reaction pathway R5-Ia/IIa is the major oxidation pathway; however, as the temperature rises, this reaction pathway becomes less dominant and the contribution of R2-Ia/IIa and R4-Ia/IIa increases. Initially, due to the low energy barrier for the R5-Ia reaction pathway (see Fig. 3), the contribution from this pathway dominates. As the temperature increases, the higher reaction barriers for the R4-Ia/IIa and R2-Ia/IIa pathways are eventually surpassed, leading to an increased contribution from these alternative pathways. However, the overall rate coefficient remains negative temperature dependent up to 400 K due to a larger contribution from the R5-Ia reaction pathway. The negative temperature dependence of the overall rate coefficient may be altered at higher temperature ranges, but the study is not relevant to this case, as it is unlikely that HMML can reach the thermosphere where the temperature can exceed 400 K.
The overall individual and total rate coefficients (S-I + S-II) for the reaction of HMML with Cl atoms are presented in Table 2. The rate coefficient obtained at 298 K is 1.13 × 10−11 cm3 per molecule per s.
| Temp (K) | k 1 | k 2 | k 3 | k 4 | k 5 | k overall |
|---|---|---|---|---|---|---|
| 200 | 2.50 × 10−15 | 5.01 × 10−14 | 2.50 × 10−15 | 9.62 × 10−14 | 3.52 × 10−11 | 3.54 × 10−11 |
| 220 | 3.62 × 10−15 | 6.58 × 10−14 | 3.62 × 10−15 | 1.17 × 10−13 | 2.49 × 10−11 | 2.51 × 10−11 |
| 240 | 5.04 × 10−15 | 8.46 × 10−14 | 5.04 × 10−15 | 1.37 × 10−13 | 1.89 × 10−11 | 1.91 × 10−11 |
| 260 | 6.80 × 10−15 | 1.04 × 10−13 | 6.80 × 10−15 | 1.58 × 10−13 | 1.51 × 10−11 | 1.53 × 10−11 |
| 280 | 8.93 × 10−15 | 1.25 × 10−13 | 8.93 × 10−15 | 1.79 × 10−13 | 1.26 × 10−11 | 1.29 × 10−11 |
| 298 | 1.12 × 10−14 | 1.45 × 10−13 | 1.12 × 10−14 | 1.99 × 10−13 | 1.10 × 10−11 | 1.13 × 10−11 |
| 320 | 1.44 × 10−14 | 1.74 × 10−13 | 1.44 × 10−14 | 2.23 × 10−13 | 9.59 × 10−12 | 9.92 × 10−12 |
| 340 | 1.78 × 10−14 | 2.03 × 10−13 | 1.78 × 10−14 | 2.46 × 10−13 | 8.64 × 10−12 | 9.02 × 10−12 |
| 360 | 2.17 × 10−14 | 2.36 × 10−13 | 2.17 × 10−14 | 2.70 × 10−13 | 7.93 × 10−12 | 8.35 × 10−12 |
| 380 | 2.61 × 10−14 | 2.73 × 10−13 | 2.61 × 10−14 | 2.94 × 10−13 | 7.37 × 10−12 | 7.85 × 10−12 |
| 400 | 3.10 × 10−14 | 3.14 × 10−13 | 3.10 × 10−14 | 3.18 × 10−13 | 6.94 × 10−12 | 7.47 × 10−12 |
Similarly to HMML + ˙OH, the total rate coefficient is inversely related to the temperature with a major contribution from the reaction pathway R5-Ib/IIb. The rate coefficient obtained for the reaction of HMML with Cl is found to be much higher than with OH radicals throughout the investigated temperature range. This trend aligns well with studies reported in the literature, where Cl-initiated oxidation reactions of VOCs or other organic compounds are generally reported to have higher rate coefficients.55–57 The overall total rate coefficient of HMML + ˙OH and HMML + Cl across the temperature range are depicted in Fig. 6.
 | ||
| Fig. 6 Computed overall total rate coefficients for the HMML + ˙OH and HMML + Cl reactions in the 200–400 K temperature range at the M06-2X/6-311++G(d,p) level of theory. | ||
In contrast to the oxidation of lactones with OH radicals, there are very few works reported in the literature for the reaction of lactones with Cl atoms. According to the studies reported by Baptista et al.58 and Ausmeel et al.59 the rate coefficient of the reaction of γ-lactones (five-membered ring) with Cl atoms varies from 10−11 to 10−10 cm3 per molecule per s at 298 K, based on the nature of side chain attached to the ring. The calculated rate coefficient in our study, 1.13 × 10−11 m3 molecule−1 s−1 at 298 K, also closely aligns with the reported rate coefficient. The correlation of ring size in lactones and the rate coefficient for the reaction with Cl atoms has not been studied. However, the ring attached with a longer side chain has been reported to have a higher rate coefficient due to more reactive sites.58
The branching ratio calculation (see Fig. S4) shows that the reaction pathway R5-Ib/IIb is the major pathway throughout the studied temperature range. This is primarily because of the very low energy barrier (see Fig. 3) of the reaction pathway R5-Ib and the lower bond dissociation energy of the C–H bond in the hydroxymethyl group, due to which it dominates the overall rate coefficient. The contribution of R5-Ib/IIb follows a decreasing trend as temperature rises, but it is negligible in comparison to the decrease in R5-Ia/IIa of S-I/II + ˙OH, while the contributions of R2-Ib/IIb and R4-Ib/IIb increase slightly.
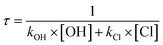 | (5) |
The estimated global abundances of OH radicals and Cl atoms are 1.0 × 106 and 1.0 × 104 molecules cm−3, respectively.60–63 OH radicals are the primary scavenger of VOCs in the atmosphere due to their abundance. However, near the marine boundary layer (MBL), the concentration of Cl atoms can reach up to 105 molecules cm−3.21,64,65
The estimated atmospheric lifetime of HMML at 298 K is 62.5 days under normal tropospheric conditions, while if we consider MBL conditions, the lifetime can be as low as 9.6 days. The estimated lifetimes of HMML within the investigated temperature range are provided in the ESI as Table S8.
Fig. 7–9 depict the plausible degradation paths of product radicals P1, P2, and P4, respectively, along with their reaction barrier, enthalpy and Gibbs free energy changes.
 | ||
| Fig. 7 Reaction fate of the product radical P1. All energies (in kcal mol−1) were computed at the M06-2X/6-311++G(d,p) level of theory. | ||
 | ||
| Fig. 8 Reaction fate of the product radical P2. All energies (in kcal mol−1) were computed at the M06-2X/6-311++G(d,p) level of theory. | ||
 | ||
| Fig. 9 Reaction fate of the product radical P4. All energies (in kcal mol−1) were computed at the M06-2X/6-311++G(d,p) level of theory. | ||
From Fig. 7, we can see that the product radical P1 can undergo reaction in two different ways: (1) P1 may undergo a reaction with hydroperoxyl radical (˙OOH), resulting in the formation of D1P1 along with the release of O2. This reaction has a negative barrier relative to starting reactants because it proceeds via the formation of a low-energy pre-reaction complex, which then proceeds via the formation of transition state TS1P1. (2) Alternatively, P1 can undergo homolytic cleavage of the C–C bond to form D2P1 along with CO2. The product radical D2P1 can further react in two ways. First, D2P1 can transform into a relatively more stable product radical, D3P1, via intramolecular hydrogen atom transfer (HAT). Second, D2P1 can rapidly react with O2 in a barrierless process to form peroxy radical D4P1. The HAT reaction has a high reaction barrier of 34.1 kcal mol; therefore, the reaction pathway involving O2 addition will dominate the reaction, thereby forming D4P1 as the major product. The peroxy radical, D4P1, can then react with NO to form D5P1 and NO2 in a barrierless process. Additionally, product radical D5P1 can undergo homolytic C–C bond cleavage to form a ketene, D6P1, along with a hydroxymethyl radical, or D5P1 can undergo O2 addition to form a peroxy radical, D7P1. The C–C bond cleavage has a high reaction barrier and is not feasible; thus, D7P1 will be the major product. D7P1 further degrades to alkoxy radical D8P1 in the presence of NO, and the alkoxy radical D8P1 subsequently reacts to form a diketone, D9P1, via hydrogen abstraction reaction in the presence of O2.
Furthermore, as shown in Fig. 8, the product radical P2 can react with O2via an addition reaction to give a peroxy radical D1P2, which reacts with NO to produce D2P2 along with the release of NO2. The alkoxy radical D2P2 can participate in homolytic C–C bond breaking via three different ways: (1) The carboxylic acid group's C–C bond cleavage to produce the diketone D2aP2. (2) The carbonyl group's C–C bond cleavage to produce the pyruvic acid D2bP2. (3) The methyl group's C–C bond cleavage creates the dioxopropanoic acid D2cP2. Among these three pathways, the third pathway has the least possibility of occurring, as the reaction is endothermic and not feasible in nature. The first and second pathways are both feasible in nature; however, the second pathway is also exothermic in nature, as illustrated in Fig. 8
Similar to the degradation of P1, the product radical P4, as shown in Fig. 9, can react in two possible ways. In the first, it can react with hydroperoxyl to form D1P4 and release O2, while in the second pathway, it can participate in C–C bond scission to form D2P4 along with CO2. The D2P4 product radical can further undergo intramolecular HAT to form D3P4 or react with O2 to form peroxy radical D4P4. The first pathway of HAT is less likely to occur due to a very high reaction barrier, as can be seen from Fig. 9. Thus, D4P4 will be formed and subsequently react to form D5P4 with the release of NO2 in the presence of NO. The product radical D5P4 can undergo C–C bond cleavage; however, this process is not feasible and has a high reaction barrier. Therefore, D5P4 reacts with O2 to form D7P4. Lastly, the peroxy radical D7P4 can react with NO via a barrierless process to form formic acid, D8P4, or may undergo intramolecular rearrangement to form D9P4 with the release of ˙OOH.
Our study shows that HMML can react efficiently with OH radicals and Cl atoms in the atmosphere. This indicates that the gas-phase oxidation of HMML can compete with or even exceed heterogeneous uptake in marine and coastal regions. This potential shift in the fate of HMML can alter the distribution of isoprene-derived oxidation products, influencing both VOCs and SOAs formation. While isoprene oxidation via IEPOX under low-NOx conditions is found to be a significant contributor to SOA production, the HMML pathway represents a significant complementary pathway, as found in our degradation study of the product radicals initially formed from the hydrogen abstraction reaction. The end products obtained from the degradation study of P1, P2, and P4 are highly reactive compounds such as diketones, α, β-unsaturated carboxylic acids, formic acid, pyruvic acid, methylglyoxal, and dioxopropanoic acid. These products are known precursors of SOA by partitioning into the particle phase or can undergo further reactions with various oxidants at much higher rate coefficients and shorter lifetimes than HMML, as reported in the literature.66–70 Thus, the inclusion of HMML oxidation in atmospheric modelling can improve the prediction of SOA formation, especially in marine boundary layers, where it can have a short lifetime.
4 Conclusion
We have carried out detailed thermodynamics and kinetics studies of the gas phase oxidation reaction of HMML with OH radicals and Cl atoms at the M06-2X/6-311++G(d,p) level of theory. The oxidation reaction of HMML proceeds via hydrogen atom abstraction reactions. All the oxidation reaction routes involving hydrogen abstractions are exothermic and feasible. The potential energy diagram constructed at the CCSD(T)//M06-2X/6-311++G(d,p) level showed that hydrogen abstraction from the hydroxymethyl group of HMML exhibits a comparatively low reaction barrier, attributed to the lower dissociation enthalpy.The computed rate coefficients for the reactions of HMML with hydroxy radicals and chlorine atoms at 298 K were determined to be 7.22 × 10−14 and 1.13 × 10−11 cm3 per molecule per s, respectively, aligning closely with the previously reported experimental trends obtained by Barnes et al.,54 Baptista et al.,58 and Ausmeel et al.59 Based on the rate coefficients obtained in this study, the atmospheric lifetime of HMML is estimated to be 62.5 days under normal tropospheric conditions, or 9.6 days under MBL conditions. This suggests that the gas oxidation reaction of HMML can be a significant loss pathway competing with heterogeneous uptake, especially under MBL conditions.
Additionally, we also conducted an analysis on the subsequent reaction of the product radicals formed from the initial oxidation of HMML. The study resulted in various end products with higher reactivity and shorter atmospheric lifetimes. Thus, our study provides a thorough analysis of HMML's fate in the atmosphere and should help in more accurate atmospheric modelling, as the gas-phase oxidation of HMML can be a competitive loss process in addition to the heterogeneous uptake.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: the SI contains vibrational frequencies and Cartesian coordinates of transition states and the energies of all the species involved in the reaction of HMML with OH radicals and Cl atoms. It also contains single-point energy corrected electronic energies, the T1 diagnostic values, individual rate coefficients of HMML-I (S-I) and HMML-II (S-II), and population distribution of S-I and S-II across the studied temperature range of 200–400 K. Moreover, Cartesian coordinates and vibrational frequencies of all the transition states involved in the degradation of product radicals, along with their energies, are also included. Further, figures for the PE scans, branching ratios, and some of the optimized structures have been given. See DOI: https://doi.org/10.1039/d5em00635j.Acknowledgements
The research team sincerely acknowledges the use of PARAM-ISHAN and PARAM-Kamrupa, the high-performance computing facilities at IIT Guwahati. They are also thankful to the Department of Chemistry, IIT Guwahati, for providing essential infrastructural support. S. R. D. and R. R. C. acknowledge the CSIR, India (Registration No. 09/0731(15684)/2022-EMR-I), and MHRD, India, respectively, for financial support.References
- A. B. Guenther, X. Jiang, C. L. Heald, T. Sakulyanontvittaya, T. Duhl, L. K. Emmons and X. Wang, The Model of Emissions of Gases and Aerosols from Nature version 2.1 (MEGAN2.1): an extended and updated framework for modeling biogenic emissions, Geosci. Model Dev., 2012, 5, 1471–1492 CrossRef.
- A. Guenther, T. Karl, P. Harley, C. Wiedinmyer, P. I. Palmer and C. Geron, Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature), Atmos. Chem. Phys., 2006, 6, 3181–3210 CrossRef CAS.
- A. G. Carlton, C. Wiedinmyer and J. H. Kroll, A review of Secondary Organic Aerosol (SOA) formation from isoprene, Atmos. Chem. Phys., 2009, 9, 4987–5005 CrossRef CAS.
- R. Atkinson, Atmospheric chemistry of VOCs and NOx, Atmos. Environ., 2000, 34, 2063–2101 CrossRef CAS.
- P. O. Wennberg, K. H. Bates, J. D. Crounse, L. G. Dodson, R. C. McVay, L. A. Mertens, T. B. Nguyen, E. Praske, R. H. Schwantes, M. D. Smarte, J. M. St Clair, A. P. Teng, X. Zhang and J. H. Seinfeld, Gas-Phase Reactions of Isoprene and Its Major Oxidation Products, Chem. Rev., 2018, 118, 3337–3390 CrossRef CAS PubMed.
- R. Atkinson and J. Arey, Atmospheric Degradation of Volatile Organic Compounds, Chem. Rev., 2003, 103, 4605–4638 CrossRef CAS PubMed.
- F. Paulot, J. D. Crounse, H. G. Kjaergaard, A. Kürten, J. M. S. Clair, J. H. Seinfeld and P. O. Wennberg, Unexpected Epoxide Formation in the Gas-Phase Photooxidation of Isoprene, Science, 2009, 325, 730–733 CrossRef CAS.
- J. D. Surratt, A. W. H. Chan, N. C. Eddingsaas, M. Chan, C. L. Loza, A. J. Kwan, S. P. Hersey, R. C. Flagan, P. O. Wennberg and J. H. Seinfeld, Reactive intermediates revealed in secondary organic aerosol formation from isoprene, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 6640–6645 CrossRef CAS PubMed.
- Y.-H. Lin, Z. Zhang, K. S. Docherty, H. Zhang, S. H. Budisulistiorini, C. L. Rubitschun, S. L. Shaw, E. M. Knipping, E. S. Edgerton, T. E. Kleindienst, A. Gold and J. D. Surratt, Isoprene Epoxydiols as Precursors to Secondary Organic Aerosol Formation: Acid-Catalyzed Reactive Uptake Studies with Authentic Compounds, Environ. Sci. Technol., 2012, 46, 250–258 CrossRef CAS.
- M. Karl, K. Tsigaridis, E. Vignati and F. Dentener, Formation of secondary organic aerosol from isoprene oxidation over Europe, Atmos. Chem. Phys., 2009, 9, 7003–7030 CrossRef CAS.
- Y.-H. Lin, H. Zhang, H. O. T. Pye, Z. Zhang, W. J. Marth, S. Park, M. Arashiro, T. Cui, S. H. Budisulistiorini, K. G. Sexton, W. Vizuete, Y. Xie, D. J. Luecken, I. R. Piletic, E. O. Edney, L. J. Bartolotti, A. Gold and J. D. Surratt, Epoxide as a precursor to secondary organic aerosol formation from isoprene photooxidation in the presence of nitrogen oxides, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 6718–6723 CrossRef CAS.
- H. G. Kjaergaard, H. C. Knap, K. B. Ørnsø, S. Jørgensen, J. D. Crounse, F. Paulot and P. O. Wennberg, Atmospheric Fate of Methacrolein. 2. Formation of Lactone and Implications for Organic Aerosol Production, J. Phys. Chem. A, 2012, 116, 5763–5768 CrossRef CAS PubMed.
- T. B. Nguyen, K. H. Bates, J. D. Crounse, R. H. Schwantes, X. Zhang, H. G. Kjaergaard, J. D. Surratt, P. Lin, A. Laskin, J. H. Seinfeld and P. O. Wennberg, Mechanism of the hydroxyl radical oxidation of methacryloyl peroxynitrate (MPAN) and its pathway toward secondary organic aerosol formation in the atmosphere, Phys. Chem. Chem. Phys., 2015, 17, 17914–17926 RSC.
- K. Nestorowicz, M. Jaoui, K. J. Rudzinski, M. Lewandowski, T. E. Kleindienst, G. Spólnik, W. Danikiewicz and R. Szmigielski, Chemical composition of isoprene SOA under acidic and non-acidic conditions: effect of relative humidity, Atmos. Chem. Phys., 2018, 18, 18101–18121 CrossRef CAS PubMed.
- M. Qin, X. Wang, Y. Hu, X. Ding, Y. Song, M. Li, P. Vasilakos, A. Nenes and A. G. Russell, Simulating Biogenic Secondary Organic Aerosol During Summertime in China, J. Geophys. Res. Atmos., 2018, 123, 11100–11119 Search PubMed.
- H. O. T. Pye, R. W. Pinder, I. R. Piletic, Y. Xie, S. L. Capps, Y.-H. Lin, J. D. Surratt, Z. Zhang, A. Gold, D. J. Luecken, W. T. Hutzell, M. Jaoui, J. H. Offenberg, T. E. Kleindienst, M. Lewandowski and E. O. Edney, Epoxide Pathways Improve Model Predictions of Isoprene Markers and Reveal Key Role of Acidity in Aerosol Formation, Environ. Sci. Technol., 2013, 47, 11056–11064 CrossRef CAS PubMed.
- Y.-Q. Zhang, X. Ding, Q.-F. He, T.-X. Wen, J.-Q. Wang, K. Yang, H. Jiang, Q. Cheng, P. Liu, Z.-R. Wang, Y.-F. He, W.-W. Hu, Q.-Y. Wang, J.-Y. Xin, Y.-S. Wang and X.-M. Wang, Observational Insights into Isoprene Secondary Organic Aerosol Formation through the Epoxide Pathway at Three Urban Sites from Northern to Southern China, Environ. Sci. Technol., 2022, 56, 4795–4805 CrossRef CAS PubMed.
- R. H. Schwantes, S. M. Charan, K. H. Bates, Y. Huang, T. B. Nguyen, H. Mai, W. Kong, R. C. Flagan and J. H. Seinfeld, Low-volatility compounds contribute significantly to isoprene secondary organic aerosol (SOA) under high-NOx conditions, Atmos. Chem. Phys., 2019, 19, 7255–7278 CrossRef CAS.
- D. Stone, L. K. Whalley and D. E. Heard, Tropospheric OH and HO2 radicals: field measurements and model comparisons, Chem. Soc. Rev., 2012, 41, 6348–6404 RSC.
- D. F. Zhao, M. Kaminski, P. Schlag, H. Fuchs, I.-H. Acir, B. Bohn, R. Häseler, A. Kiendler-Scharr, F. Rohrer, R. Tillmann, M. J. Wang, R. Wegener, J. Wildt, A. Wahner and T. F. Mentel, Secondary organic aerosol formation from hydroxyl radical oxidation and ozonolysis of monoterpenes, Atmos. Chem. Phys., 2015, 15, 991–1012 CrossRef.
- S. Pechtl and R. von Glasow, Reactive chlorine in the marine boundary layer in the outflow of polluted continental air: A model study, Geophys. Res. Lett., 2007, 34, 1–5 CrossRef.
- X. Wang, D. J. Jacob, W. Downs, S. Zhai, L. Zhu, V. Shah, C. D. Holmes, T. Sherwen, B. Alexander, M. J. Evans, S. D. Eastham, J. A. Neuman, P. R. Veres, T. K. Koenig, R. Volkamer, L. G. Huey, T. J. Bannan, C. J. Percival, B. H. Lee and J. A. Thornton, Global tropospheric halogen (Cl, Br, I) chemistry and its impact on oxidants, Atmos. Chem. Phys., 2021, 21, 13973–13996 CrossRef CAS.
- W. C. Keene, J. Stutz, A. A. P. Pszenny, J. R. Maben, E. V. Fischer, A. M. Smith, R. von Glasow, S. Pechtl, B. C. Sive and R. K. Varner, Inorganic chlorine and bromine in coastal New England air during summer, J. Geophys. Res. Atmos., 2007, 112, D10S12 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16 Revision C.01, Gaussian Inc. Wallingford CT, 2016 Search PubMed.
- R. Dennington, T. A. Keith and J. M. Millam, GaussView Version 6, Semichem Inc. Shawnee Mission KS, 2019 Search PubMed.
- Y. Zhao and D. G. Truhlar, The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed.
- Y. Zhao and D. G. Truhlar, Density functionals with broad applicability in chemistry, Acc. Chem. Res., 2008, 41, 157–167 CrossRef CAS PubMed.
- J. A. Pople, M. Head-Gordon and K. Raghavachari, Quadratic configuration interaction. A general technique for determining electron correlation energies, J. Chem. Phys., 1987, 87, 5968–5975 CrossRef CAS.
- R. Krishnan, J. S. Binkley, R. Seeger and J. A. Pople, Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions, J. Chem. Phys., 1980, 72, 650–654 CrossRef CAS.
- A. Shiroudi, T. Kurtén and J. Czub, Mechanistic insights and atmospheric implications of the degradation reaction of 3-methoxy-1-propanol by reaction with hydroxyl radicals and identification of the end products in the presence of O2/NO, Sci. Rep., 2025, 15, 1038 CrossRef CAS.
- S. Kanchanakungwankul, J. L. Bao, J. Zheng, I. M. Alecu, B. J. Lynch, Y. Zhao and D. G. Truhlar, Database of Frequency Scale Factors for Electronic Model Chemistries-Version 5, 2021 Search PubMed.
- H. P. Hratchian and H. B. Schlegel, Accurate reaction paths using a Hessian based predictor–corrector integrator, J. Chem. Phys., 2004, 120, 9918–9924 CrossRef CAS PubMed.
- H. Hratchian and H. Schlegel, Using Hessian updating to increase the efficiency of a Hessian based predictor-corrector reaction path following method, J. Chem. Theory Comput., 2005, 1, 61–69 CrossRef CAS PubMed.
- D. Mandal, C. Sahu, S. Bagchi and A. K. Das, Kinetics and mechanism of the tropospheric oxidation of vinyl acetate initiated by OH radical: a theoretical study, J. Phys. Chem. A, 2013, 117, 3739–3750 CrossRef CAS PubMed.
- R. R. Changmai, S. R. Daimari and M. Sarma, Unveiling the Atmospheric Oxidation of Hexafluoroisobutylene,(CF3)2C =CH2, with Cl Atom, NO3 Radical, and O3 Molecule, J. Phys. Chem. A, 2025, 129, 3906–3920 CrossRef CAS PubMed.
- S. Pan and L. Wang, Atmospheric Oxidation Mechanism of m-Xylene Initiated by OH Radical, J. Phys. Chem. A, 2014, 118, 10778–10787 CrossRef CAS.
- Y. Yu, L. Pan, Q. Sun and J. Wang, The mechanism and kinetics of the atmospheric oxidation of CF 3 (CF 2) 2 CH double bond, length as m-dash CH 2 (HFC-1447fz) by hydroxyl radicals: ab initio investigation, Phys. Chem. Chem. Phys., 2024, 26, 10989–10997 RSC.
- P. Rutto, E. Ubana, T. M. Selby and F. Goulay, Kinetic Study of OH Radical Reactions with Cyclopentenone Derivatives, J. Phys. Chem. A, 2024, 128, 8209–8219 CrossRef CAS PubMed.
- J. Noga and R. J. Bartlett, The full CCSDT model for molecular electronic structure, J. Chem. Phys., 1987, 86, 7041–7050 CrossRef CAS.
- R. O. Ramabhadran and K. Raghavachari, Extrapolation to the gold-standard in quantum chemistry: computationally efficient and accurate CCSD (T) energies for large molecules using an automated thermochemical hierarchy, J. Chem. Theory Comput., 2013, 9, 3986–3994 CrossRef CAS PubMed.
- T. J. Lee and P. R. Taylor, A diagnostic for determining the quality of single-reference electron correlation methods, Int. J. Quantum Chem., 1989, 36, 199–207 CrossRef.
- M. W. Chase Jr, JANAF thermochemical tables, J. Phys. Chem. Ref. Data, 1985, 14(Suppl. 1), 718 Search PubMed.
- R. D. Johnson, NIST Computational Chemistry Comparison and Benchmark Database (CCCBDB): Electronic Spin–Orbit Splittings, National Institute of Standards and Technology, 2025, https://cccbdb.nist.gov/elecspinx.asp, Standard Reference Database Number 101, accessed: 2025-10-07 Search PubMed.
- D. G. Truhlar and B. C. Garrett, Variational transition-state theory, Acc. Chem. Res., 1980, 13, 440–448 CrossRef CAS.
- J. L. Bao and D. G. Truhlar, Variational transition state theory: theoretical framework and recent developments, Chem. Soc. Rev., 2017, 46, 7548–7596 RSC.
- C. Eckart, The penetration of a potential barrier by electrons, Phys. Rev., 1930, 35, 1303–1310 CrossRef CAS.
- H. S. Johnston and J. Heicklen, Tunnelling corrections for unsymmetrical Eckart potential energy barriers, J. Phys. Chem., 1962, 66, 532–533 CrossRef.
- S. Canneaux, F. Bohr and E. Henon, KiSThelP: a program to predict thermodynamic properties and rate constants from quantum chemistry results, J. Comput. Chem., 2014, 35, 82–93 CrossRef CAS.
- P. Seal, S. Barua, S. Iyer, A. Kumar and M. Rissanen, A systematic study on the kinetics of H-shift reactions in pristine acyl peroxy radicals, Phys. Chem. Chem. Phys., 2023, 25, 28205–28212 RSC.
- N. Tsona Tchinda, X. Lv, S. N. Tasheh, J. N. Ghogomu and L. Du, Atmospheric fate of organosulfates through gas-phase and aqueous-phase reactions with hydroxyl radicals: implications for inorganic sulfate formation, Atmos. Chem. Phys., 2025, 25, 8575–8590 CrossRef.
- T. V.-T. Mai, O. M. Na, T. Pham and L. K. Huynh, Hydrogen abstraction reactions by NH2 radicals from C3-C6 Cycloalkanes: A theoretical study, Chem. Phys. Lett., 2024, 856, 141658–141663 CrossRef.
- D. A. McQuarrie, Statistical Mechanics, Harper Collins, 1976 Search PubMed.
- D. Singleton and R. Cvetanovic, Temperature dependence of the reaction of oxygen atoms with olefins, J. Am. Chem. Soc., 1976, 98, 6812–6819 CrossRef CAS.
- I. Barnes, S. Kirschbaum and J. M. Simmie, Combined experimental and theoretical study of the reactivity of γ-butyro-and related lactones, with the OH radical at room temperature, J. Phys. Chem. A, 2014, 118, 5013–5019 CrossRef CAS PubMed.
- I. Barnes, P. Wiesen and M. Gallus, Kinetics and mechanism of the OH-radical and Cl-atom oxidation of propylene carbonate, RSC Adv., 2016, 6, 98234–98242 RSC.
- P. M. Edwards and C. J. Young, Primary Radical Effectiveness: Do the Different Chemical Reactivities of Hydroxyl and Chlorine Radicals Matter for Tropospheric Oxidation?, ACS EST Air, 2024, 1, 780–788 CrossRef PubMed.
- V. F. Andersen, K. B. Ørnsø, S. Jørgensen, O. J. Nielsen and M. S. Johnson, Atmospheric chemistry of ethyl propionate, J. Phys. Chem. A, 2012, 116, 5164–5179 CrossRef CAS PubMed.
- A. Baptista, R. G. Gibilisco, P. Wiesen and M. A. Teruel, FTIR kinetic study of the reactions of γ-caprolactone and γ-heptalactone initiated by Cl and OH radicals at 298 K and atmospheric pressure, Chem. Phys. Lett., 2021, 765, 138313–138320 CrossRef CAS.
- S. Ausmeel, C. Andersen, O. J. Nielsen, F. F. Østerstrøm, M. S. Johnson and E. J. Nilsson, Reactions of three lactones with Cl, OD, and O3: atmospheric impact and trends in furan reactivity, J. Phys. Chem. A, 2017, 121, 4123–4131 CrossRef CAS PubMed.
- M. Krol, P. J. van Leeuwen and J. Lelieveld, Global OH trend inferred from methylchloroform measurements, J. Geophys. Res. Atmos., 1998, 103, 10697–10711 CrossRef CAS.
- U. Platt, W. Allan and D. Lowe, Hemispheric average Cl atom concentration from 13 C/12 C ratios in atmospheric methane, Atmos. Chem. Phys., 2004, 4, 2393–2399 CrossRef CAS.
- R. R. Changmai and M. Sarma, Tropospheric Oxidation of Dichlorotetrafluoropropene CF3CF=CCl2 Initiated by OH Radical: Reaction Mechanisms, Kinetic Studies, and Atmospheric Implications, ACS Earth Space Chem., 2022, 6, 1782–1792 CrossRef CAS.
- R. R. Changmai, S. R. Daimari, A. K. Yadav and M. Sarma, Atmospheric Oxidation Pathways of CF3CH2CFCl2 (HCFC-234fb) with OH-radicals and Cl-atoms: Insights into the Mechanism, Thermodynamics, and Kinetics, Phys. Chem. Chem. Phys., 2024, 26, 23363–23371 RSC.
- C.-T. Chang, T.-H. Liu and F.-T. Jeng, Atmospheric concentrations of the Cl atom, CIO radical, and HO radical in the coastal marine boundary layer, Environ. Res., 2004, 94, 67–74 CrossRef CAS PubMed.
- V. Wagner, R. Von Glasow, H. Fischer and P. Crutzen, Are CH2O measurements in the marine boundary layer suitable for testing the current understanding of CH4 photooxidation?: A model study, J. Geophys. Res. Atmos., 2002, 107, ACH–3 Search PubMed.
- R. Talukdar, L. Zhu, K. Feierabend and J. Burkholder, Rate coefficients for the reaction of methylglyoxal (CH3COCHO) with OH and NO3 and glyoxal (HCO)2 with NO3, Atmos. Chem. Phys., 2011, 11, 10837–10851 CrossRef CAS.
- F. Berg, A. Novelli, R. Dubus, A. Hofzumahaus, F. Holland, A. Wahner and H. Fuchs, Temperature-dependent rate coefficients for the reactions of OH radicals with selected alkanes, aromatic compounds, and monoterpenes, Atmos. Chem. Phys., 2024, 24, 13715–13731 CrossRef CAS.
- M. E. Jenkin, R. Valorso, B. Aumont, A. R. Rickard and T. J. Wallington, Estimation of rate coefficients and branching ratios for gas-phase reactions of OH with aliphatic organic compounds for use in automated mechanism construction, Atmos. Chem. Phys., 2018, 18, 9297–9328 CrossRef CAS.
- A. Mellouki, T. J. Wallington and J. Chen, Atmospheric chemistry of oxygenated volatile organic compounds: impacts on air quality and climate, Chem. Rev., 2015, 115, 3984–4014 CrossRef CAS PubMed.
- T.-M. Fu, D. J. Jacob, F. Wittrock, J. P. Burrows, M. Vrekoussis and D. K. Henze, Global budgets of atmospheric glyoxal and methylglyoxal, and implications for formation of secondary organic aerosols, J. Geophys. Res. Atmos., 2008, 113, D15303 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




