Reduction and adsorption capacities of soils for Cr(VI) and quantitative contributions of key influencing factors
Ze
Zhang
,
Libing
Zheng
,
Xinru
Zhai
,
Shi-Wei
Li
and
Helian
Li
 *
*
School of Water Conservancy and Environment, University of Jinan, Jinan 250022, China. E-mail: chm_lihl@ujn.edu.cn
First published on 9th December 2025
Abstract
The saturated sorption capacity of Cr(VI) in soil is determined by reduction and adsorption processes. Red soil, paddy soil, black soil, and fluvo-aquic soil were selected for repeated sorption experiments in this study to determine Cr(VI) saturated sorption capacity as well as reduction/adsorption capacities and the contributions of key factors were analyzed through regulating soil properties. The results showed that the saturated sorption capacities of the four soils were 972, 589, 551, and 76 mg kg−1. Per pH unit increase, saturated sorption capacity decreased in the order: red soil (142 mg kg−1) > paddy soil (134 mg kg−1) > black soil (132 mg kg−1) > fluvo-aquic soil (25 mg kg−1). Specifically, red soil showed a significantly greater drop in adsorption capacity (50 mg kg−1) than the other three soils; paddy soil and black soil had over 27% decrease in reduction capacity, while fluvo-aquic soil exhibited no significant change. Organic matter removal decreased reduction capacities by over 60% but increased adsorption capacities by over 20%. Removing Fe and Al oxides significantly reduced adsorption capacities by over 50%; Mn oxide removal had minor impact. Correlation analysis and random forest modelling identified pH as the primary factor influencing soil Cr(VI) sorption, contributing 27.8%, 45.4%, and 28.0% respectively to saturated sorption capacity, reduction capacity, and adsorption capacity. Adsorption capacity was mainly affected by Fe, Al and Mn oxides, collectively contributing 70.5%, while organic matter mainly affected reduction capacity, contributing 26.6%. This initial quantitative analysis provides new insights into the fate of Cr(VI) in soil environments.
Environmental significanceSorption is a key process in mitigating the migration, toxicity, and bioavailability of hexavalent chromium [Cr(VI)] in soils. Sorbed Cr(III) as the reduction product of Cr(VI) showed a great difference in bioavailability from sorbed Cr(VI). Therefore, it is essential to distinguish the reduction and adsorption processes for the scientific evaluation of environmental risks of Cr(VI). This study is the first to determine the saturated sorption capacity, as well as the reduction and adsorption capacities, of Cr(VI) in soils, and quantitatively analyze the contributions of key soil physicochemical factors. This initial quantitative analysis provides new insights into the fate of Cr(VI) in soil environments. |
1. Introduction
The accumulation of chromium in soils is influenced by anthropogenic industrial activities, as well as climate and topographical factors.1–3 Chromium is a major heavy metal contaminant in soil environments and primarily exists in two valence states: Cr(III) and Cr(VI).4 Cr(III) functions as an essential trace element for humans and animals; in contrast, Cr(VI) is listed as a priority chemical in China due to its genotoxicity and carcinogenicity. In soils, Cr(VI) predominantly occurs as oxyanion species, including CrO42−, HCrO4−, and Cr2O72−.5 These chemical forms confer high mobility to Cr(VI) compared to Cr(III), thereby increasing its potential risks to crop growth and the safety of agricultural products. Consequently, the fate and species transformation of Cr(VI) in soils critically affect its ecotoxicity.When Cr(VI) is introduced into soil, sorption mechanisms—including electrostatic adsorption, surface complexation, and adsorption-coupled reduction—are key processes governing its mobility, toxicity, and bioavailability.6,7 Although numerous previous studies have measured Cr(VI) sorption capacities in various soils (SI, Table S1), they have typically focused on equilibrium sorption capacities at specific initial Cr(VI) concentrations,8 which does not ensure the attainment of sorption saturation. Furthermore, Cr(VI) can either be adsorbed directly in its original valence state or transformed into Cr(III) via reduction prior to adsorption. Studies have attempted to determine the reduction amount of Cr(VI) through batch experiments conducted in a phosphate buffer solution, thereby eliminating its direct adsorption onto the soil. However, in the absence of a buffer solution, both the direct adsorption of Cr(VI) and the adsorption of Cr(III) (formed after Cr(VI) reduced) take place. To our knowledge, only a few studies have systematically distinguished between these adsorption and reduction processes. For instance, James and Bartlett9 added a phosphate buffer solution to the soil–Cr(VI) equilibrium system to extract the adsorbed Cr(VI). The portion that could not be extracted corresponded to the amount of Cr(VI) reduced to Cr(III), which was then used to determine the soil’s effective reduction capacity. In this context, the soil’s ability to reduce Cr(VI) to Cr(III) is defined as “reduction capacity”, while the adsorption capacity resulting from the direct adsorption of Cr(VI) (a combination of electrostatic and specific adsorption mechanisms) is designated as adsorption capacity. Given the marked difference in toxicity between Cr(VI) and Cr(III), the environmental risks associated with Cr(VI) contamination in soils are primarily determined by the relative proportions of these two capacities.
The reduction capacity of soil for Cr(VI) is primarily determined by the content of organic matter and reducing substances such as iron and sulfur compounds. Under aerobic conditions, organic matter and divalent iron minerals serve as the principal reductants for Cr(VI), whereas under anoxic conditions, the reduction process is mainly mediated by Fe2+, solid FeS/FeS2, and reduced sulfur species.10 Additionally, the reduction capacity is influenced by pH, with lower pH conditions facilitating the reduction of Cr(VI).11 Regarding adsorption capacity, minerals such as goethite, ferrihydrite, and aluminum oxides are the predominant components influencing Cr(VI) adsorption.12,13 The adsorption of Cr(VI) by iron and aluminum oxides generally decreases with increasing pH.14 Furthermore, soil organic matter and competing anions, including phosphates, sulfates, and carbonates, inhibit Cr(VI) adsorption by competing for adsorption sites.12 Although the key factors influencing Cr(VI) reduction and adsorption in soil have been identified, the specific contribution of each factor remains unclear.
To address the above-mentioned knowledge gaps, in this study, four soils with contrasting sorption capacities (i.e. red soil, paddy soil, black soil, and fluvo-aquic soil) were selected to conduct repeated sorption experiments. The main aims were to (1) determine the saturated sorption capacity of Cr(VI) in different soils, (2) assess the relative contributions of reduction and adsorption processes to the saturated sorption capacity of Cr(VI), and (3) quantitatively analyze the contributions of key soil physicochemical factors to the saturated sorption capacity, as well as to the reduction and adsorption capacities of Cr(VI).
2. Materials and methods
2.1 Soil sampling and characterization
Based on the results of the preliminary experiment (not published yet), four soils exhibiting significant differences in Cr(VI) sorption capacity—red soil, paddy soil, black soil, and fluvo-aquic soil—were selected as tested soils. All soil samples were taken from the surface layer of farmland (0–20 cm). The soil samples were air-dried at room temperature, and plant roots, stones, and other impurities were removed. Subsequently, the samples were ground and passed through a 2 mm sieve prior to further analysis.Soil acidity was determined using a pH meter (Star A211, Thermo, USA) in a 0.01 M KCl solution at a solid–solution ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 (w
2.5 (w![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v). Soil organic matter content was measured by the potassium dichromate oxidation-spectrophotometry method.15 Cation exchange capacity was assessed using the hexamminecobalt trichloride method.16 Soil mechanical composition was analyzed with a soil hydrometer.17 The sodium bisulfite-sodium citrate-sodium bicarbonate (DCB) method was used to extract free Fe and Al oxides from the soil. The concentrations of Fe and Al in the extracts were then determined using phenanthroline spectrophotometry,18 while free Mn oxides were quantified by titration with ammonium ferrous sulfate.19 Total Cr content in the soil was measured using a flame atomic absorption spectrophotometer (AA-7000F/G, Shimadzu, Japan) following microwave digestion.
v). Soil organic matter content was measured by the potassium dichromate oxidation-spectrophotometry method.15 Cation exchange capacity was assessed using the hexamminecobalt trichloride method.16 Soil mechanical composition was analyzed with a soil hydrometer.17 The sodium bisulfite-sodium citrate-sodium bicarbonate (DCB) method was used to extract free Fe and Al oxides from the soil. The concentrations of Fe and Al in the extracts were then determined using phenanthroline spectrophotometry,18 while free Mn oxides were quantified by titration with ammonium ferrous sulfate.19 Total Cr content in the soil was measured using a flame atomic absorption spectrophotometer (AA-7000F/G, Shimadzu, Japan) following microwave digestion.
2.2 Modification of soil physicochemical properties
Soil pH was adjusted by amendment with MgCO3.20 First, 20 g of test soil was weighed, and 30, 90, and 120 mg of MgCO3 were added and thoroughly mixed. Subsequently, 4 g subsamples from each treatment were weighed, and 0.01 M KCl solution was added at a soil-to-water ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 (m
2.5 (m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v). The mixtures were shaken for 30 minutes, allowed to stand until the supernatant clarified, and then the soil pH was measured. The remaining treated soil samples were sealed and stored at room temperature for subsequent use.
v). The mixtures were shaken for 30 minutes, allowed to stand until the supernatant clarified, and then the soil pH was measured. The remaining treated soil samples were sealed and stored at room temperature for subsequent use.
The removal of organic matter from soils followed the method of Mikutta et al.21 Briefly, 30 g of soil was weighed into a beaker and moistened with a small amount of deionized water, and then 20 mL of 30% H2O2 was added. The mixture was continuously heated and stirred in a thermostatic water bath at 80 °C until the reaction reached stability. The treated sample was then transferred to a centrifuge tube and centrifuged at 4000 rpm for 10 minutes. The supernatant was discarded, and the residue was washed three times with deionized water. Finally, the washed sample was freeze-dried for 48 hours and stored in a sealed container for later use.
The method described by Ke et al. was used to remove Fe and Al oxides from soils.22 Briefly, 5 g of soil was weighed into a 50 mL centrifuge tube, followed by the sequential addition of 20 mL of 0.3 M sodium citrate solution, 2.5 mL of 1 M sodium bicarbonate solution, and 1 g of sodium dithionite powder. The mixture was thoroughly shaken to ensure homogeneity, and then placed in a constant-temperature water bath at 80 °C for 15 minutes. After cooling to room temperature, the sample was centrifuged at 4000 rpm for 10 minutes, and the supernatant was discarded. The residue was then rinsed once with cold 0.01 M CaCl2–0.1 M NaCl solution, followed by three rinses with deionized water. Finally, the washed sample was freeze-dried for 48 hours and stored in a sealed container for later use.
The method described by Neaman et al. was used to remove Mn oxides from soils.23 Briefly, 5 g of soil was weighed into a 50 mL centrifuge tube, and 40 mL of a mixed solution containing 0.1 M NH2OH·HCl and 0.01 M HCl was added. The mixture was thoroughly shaken to ensure homogeneity, then placed in a water bath at a constant temperature of 50 °C and kept in the dark for 2 hours until the reaction reached stability. The sample was subsequently centrifuged at 4000 rpm for 10 minutes, and the supernatant was discarded. The residue was rinsed three times with deionized water and then centrifuged. Finally, the washed sample was freeze-dried for 48 hours and stored in a sealed container for later use.
2.3 Determination of saturated sorption, adsorption, and reduction capacities of soils for Cr(VI)
The saturated sorption capacity was determined using a method modified from Jardine et al.24 Briefly, 3 g of tested soil and 30 mL of Cr(VI) solution at a specified concentration were sequentially added to a 50 mL centrifuge tube. To mitigate interference arising from variations in the soil's background electrolytes, all solutions were amended with 0.01 M KCl to maintain a constant ionic strength. Given the substantial differences in Cr(VI) sorption capacity among the four soils, using excessively high initial concentrations for those with low sorption capacity would undermine the accuracy of Cr(VI) determination. Based on preliminary experimental results, the initial Cr(VI) concentration was set at 200 mg L−1 for red soil, paddy soil, and black soil, and at 50 mg L−1 for fluvo-aquic soil. The samples were incubated horizontally at a constant temperature (25 ± 1 °C) and shaken at 180 rpm for 48 hours. After then, the samples were centrifuged at 4000 rpm for 10 minutes. The supernatant was filtered through a 0.45 µm membrane (Jinteng, China), and the concentration of Cr(VI) was determined using the diphenylcarbazide spectrophotometric method, with measurements performed using a UV-visible spectrophotometer (UV-6100, Metash, China). The Cr(VI) sorption capacity of the soil was calculated from the difference in Cr(VI) concentration in the supernatant before and after reaction. Each soil sample was then rinsed three times with deionized water, and fresh Cr(VI) solution was added for the next cycle of sorption. This process was repeated ten times to ensure that the adsorption saturation is achieved, with each test performed in duplicate. The total sorption capacity was calculated as the sum over ten cycles. This approach was designed to determine the saturated sorption capacity, in contrast to the equilibrium sorption capacity derived from classical adsorption models. After all sorption reactions were complete, the soil samples were freeze-dried for 48 hours and subsequently used for desorption experiments as described below.Desorption experiments were performed to release the adsorbed Cr(VI). Two grams of freeze-dried soil were weighed into a centrifuge tube, and 20 mL of 0.01 M KH2PO4–K2HPO4 mixture was added for its efficacy in displacing Cr(VI) through competitive ligand exchange on oxide surfaces.25 The sample was placed horizontally in an incubator at a constant temperature (25 ± 1 °C) and shaken at 180 rpm for 2 hours to reach equilibrium. After then, the sample was centrifuged at 4000 rpm for 10 minutes, and the supernatant was filtered through a 0.45 µm membrane. The Cr(VI) concentration in the supernatant was determined as described above, and the amount of Cr(VI) desorbed was calculated accordingly. The soil residue was then rinsed three times with deionized water, and the desorption process was repeated as described above. This procedure was conducted three times to approximate full desorption of Cr(VI), with each set of experiments performed in duplicate. The total desorbed amount was calculated as the sum of the Cr(VI) released across the three cycles of desorption.
Statistical analysis results indicated that the amounts of both sorption capacity and desorption capacity showed no significant increase (p > 0.05) between the last two sorption or desorption cycles. This suggests that sorption had reached saturation and desorption was complete. At this stage, the total amount of sorption capacity was defined as the saturated sorption capacity, while the total amount of Cr(VI) released during the desorption experiment was considered as the adsorption capacity. The reduction capacity was then calculated by subtracting the adsorption capacity from the saturated sorption capacity.
2.4 Data processing and statistical analysis
Data processing and statistical analysis were performed using SPSS 26.0. Pearson's correlation analysis (two-tailed, p < 0.05) was used to assess relationships between saturated sorption capacity, reduction capacity, adsorption capacity and soil physicochemical properties. One-way ANOVA followed by Duncan's multiple range tests was used to evaluate significant differences among treatments. Random forest models were constructed in R software, with saturated sorption capacity, adsorption capacity, and reduction capacity as the dependent variables and soil physicochemical properties as the independent variables; the number of decision trees was set to 1000 to quantify the variable importance. Data visualization was performed using Origin 2021.3. Results and discussion
3.1 Soil physicochemical properties
The basic physicochemical properties of the four tested soils are presented in Table 1. Soil pH ranged from 4.32 in red soil to 7.66 in fluvo-aquic soil. Red soil exhibited the highest contents of free Fe oxides (23.04 g kg−1) and Al oxides (3.77 g kg−1), suggesting an abundance of adsorption sites which may enhance its adsorption capacity for Cr(VI). Black soil had the highest organic matter content (54.71 g kg−1), followed by paddy soil (33.43 g kg−1); both were significantly higher than those in the other two soils, indicating a higher reduction potential. In addition, the initial concentrations of Mn oxides were low in all four soils. The Cr contents in all soils were below the screening threshold specified in the risk control standard for soil contamination of agricultural land.26| Soil type | Sampling site | Soil texture | Soil pH | Organic matter (g kg−1) | CEC (cmol kg−1) | Fe oxide (g kg−1) | Al oxide (g kg−1) | Mn oxide (g kg−1) | Total Cr (mg kg−1) |
|---|---|---|---|---|---|---|---|---|---|
| Red soil | Yingtan, Jiangxi, China | Clay | 4.32 | 10.11 | 12.07 | 23.04 | 3.77 | 0.16 | 83.07 |
| Paddy soil | Jiaxing, Zhejiang, China | Silty loam | 5.38 | 33.43 | 20.42 | 8.07 | 0.69 | 0.13 | 75.50 |
| Black soil | Hailun, Heilongjiang, China | Silty clay loam | 5.53 | 54.71 | 33.41 | 7.42 | 1.67 | 0.52 | 52.78 |
| Fluvo-aquic soil | Baoding, Hebei, China | Silty loam | 7.66 | 7.90 | 9.50 | 5.97 | 0.56 | 0.18 | 54.46 |
3.2 Saturated sorption, adsorption, and reduction capacities of soils for Cr(VI)
Fig. 1 presents the sorption capacity, desorption capacity, and the contributions of adsorption and reduction to the saturated sorption capacity of Cr(VI) in the four soils. As shown in Fig. 1(a), significant differences in the initial Cr(VI) sorption capacity were observed among the soils, with the order of red soil > paddy soil > black soil > fluvo-aquic soil. As the reaction proceeded, red soil exhibited the highest sorption capacity, reaching a saturated sorption capacity of 972 mg kg−1 and displaying a significantly greater increasing trend compared to the other three soils. This strong sorption potential of red soil can be attributed to its low pH and high contents of Fe and Al oxides. On one hand, an acidic environment can enhance the redox potential27 and increase proton concentrations,28 which synergistically promote Cr(VI) reduction. On the other hand, the adsorption capacity of Fe and Al oxides for Cr(VI) is enhanced under lower pH conditions due to changes in their surface complexation behavior: when pH > 7, outer-sphere complexes predominate; whereas at pH < 6, binuclear monoprotonated inner-sphere complexes are dominant, which greatly facilitates the adsorption and immobilization of Cr(VI).13 Additionally, the sorption processes in paddy soil and black soil, both of which are rich in organic matter, were more dependent on reduction reactions. The continuous consumption of organic matter during these reactions led to fluctuations in reduction capacity, which resulted in lower sorption capacities and slower sorption rates in these soils compared to red soil, with their saturated sorption capacities being 589 mg kg−1 and 551 mg kg−1, respectively. In contrast, the fluvo-aquic soil exhibited the lowest sorption capacities, with a saturated sorption capacity of only 76 mg kg−1. This limited sorption potential is closely associated with its high pH, as alkaline conditions reduce the abundance of active sites on soil particle surfaces and significantly decrease the reactivity of Cr(VI),13 thereby inhibiting the sorption process. | ||
| Fig. 1 Sorption capacity (a), desorption capacity (b), and the contributions of adsorption and reduction to the saturated sorption capacity (c) of Cr(VI) across four soil types. | ||
It is noteworthy that although the initial and final Cr(VI) sorption capacities in black soil and paddy soil were comparable, a marked difference was observed during the intermediate stage. This phenomenon can be attributed to the dual role of soil organic matter. Specifically, in the early stage of the reaction, carboxyl and phenolic groups in the organic matter preferentially occupied the active sites on Fe and Al oxides,29 which inhibited Cr(VI) adsorption. As a result, at this stage, black soil with a higher organic matter content exhibited a lower sorption capacity than paddy soil. However, organic matter does not solely inhibit adsorption; it also contains reducing groups such as phenolic and hydroxyl groups, which can reduce a portion of Cr(VI) to Cr(III).30 In the early stages of the reaction, kinetic limitations restricted the initial formation of reduction products, thereby limiting their contribution to the overall removal of Cr(VI). As the reaction proceeded, continuous reduction reactions generated increasing amounts of Cr(III), which readily forms coordination compounds or hydroxide precipitates with clay minerals and humus.31 Meanwhile, the reduction of Cr(VI) by organic matter consumes competing adsorptive functional groups, thereby gradually liberating the active sites on Fe and Al oxides that were previously occupied. Consequently, the Cr(VI) adsorption capacity increased progressively over time. In paddy soil, owing to its relatively lower organic matter content, Cr(VI) adsorption was initially dominated by Fe and Al oxides, leading to a more rapid rise in sorption capacity at the outset. In contrast, black soil exhibited an initial inhibitory effect due to its higher organic content; subsequently, enhanced reduction and the delayed release of adsorption sites resulted in a sustained rise in sorption capacity during the middle and later stages, ultimately reaching levels comparable to those in paddy soil. This dynamic demonstrates the dual role of organic matter in both inhibiting and enhancing Cr(VI) fixation in soil.
The data presented in Fig. 1(b) and (c) reveal that the four soils exhibited distinctly different capacities for adsorbing and reducing Cr(VI). Red soil showed an adsorption capacity of 524 mg kg−1 (54%) and a reduction capacity of 447 mg kg−1 (46%), reflecting the coupling of adsorption and reduction processes. Paddy soil and black soil behaved similarly: paddy soil had an adsorption capacity of 194 mg kg−1 (33%) and a reduction capacity of 394 mg kg−1 (67%), while black soil exhibited 147 mg kg−1 (27%) for adsorption and 403 mg kg−1 (73%) for reduction. The proportion of reduction capacity in both soils was significantly higher than that in the other two soil types, indicating that Cr(VI) immobilization in soils with high organic matter is predominantly driven by reduction processes. In contrast, fluvo-aquic soil displayed the lowest adsorption and reduction capacities (29 mg kg−1 and 47 mg kg−1, respectively), further confirming that soil reactivity toward Cr(VI) decreases markedly under alkaline conditions.13
According to the relative contributions of adsorption and reduction processes, the four soils in this study were categorized into distinct types.32 Red soil was identified as a typical “adsorptive-reductive soil” due to its high adsorption and reduction capacities, as well as the balanced contributions of these two processes to the overall Cr(VI) retention. Both paddy soil and black soil were classified as “reductive soils” because their reduction capacity was predominant over adsorption. Fluvo-aquic soil, characterized by an extremely low saturated sorption capacity and minimal adsorption and reduction capabilities, was defined as a “weakly reactive soil.” This classification reflects the functional differences among soil types with respect to Cr(VI) immobilization mechanisms.
In summary, soil physicochemical properties play a crucial role in governing Cr(VI) sorption processes. However, the foregoing analysis has not elucidated the specific contributions of individual physicochemical factors to Cr(VI) reduction and adsorption capacities. To further evaluate the effects of specific factors—namely, pH, organic matter content, and types of oxides—on the reduction and adsorption processes, single-factor batch experiments were conducted.
3.3 Contribution of key factors to adsorption and reduction capacities
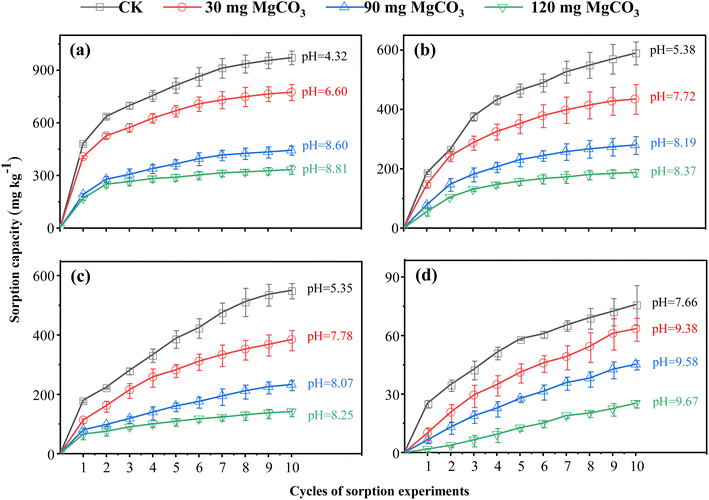
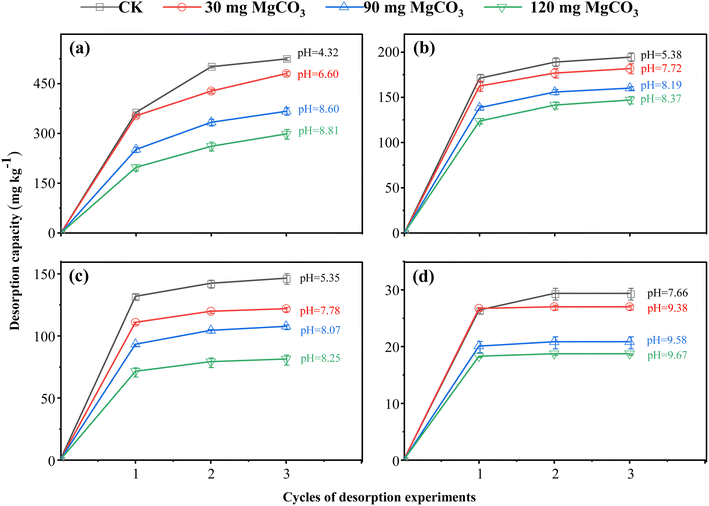
To investigate the effect of pH on the Cr(VI) sorption capacity, reduction capacity, and adsorption capacity, the pH of the four soils was adjusted by adding 30, 90, or 120 mg of MgCO3 to each sample. Following pH adjustment, adsorption and desorption experiments were conducted. The corresponding results are presented in Fig. 2 (adsorption), Fig. 3 (desorption), and Fig. 4 (percentage contributions of reduction and adsorption capacities).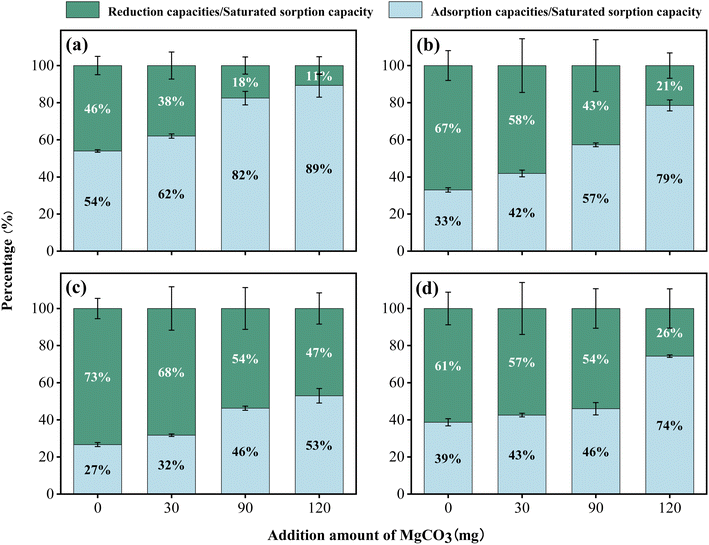 | ||
| Fig. 4 Effect of pH on the proportions of reduction and adsorption capacities across four soil types. (a) Red soil; (b) paddy soil; (c) black soil; and (d) fluvo-aquic soil. | ||
The data indicated that with increasing pH, both the saturated sorption capacity and adsorption rate of Cr(VI) in the four soils decreased significantly, though the magnitude of these declines varied across soil types. Specifically, for red soil, each 1-unit increase in pH led to a reduction of 159 mg kg−1 in saturated sorption capacity, which comprised a 103 mg kg−1 decrease in reduction capacity and a 56 mg kg−1 decrease in adsorption capacity. Notably, the decline in the adsorption capacity for red soil was significantly greater than that of the other three soils. Mechanistically, pH impacts Cr(VI) adsorption primarily by altering the speciation of chromium and the net surface charge of soil particles.33 The zeta potential of soil colloids decreases as the pH of the surrounding solution increases,34 which is primarily due to the deprotonation of acidic functional groups (such as hydroxyl and carboxyl groups) on the soil surface, as well as the deprotonation of hydroxyl groups on Fe/Al oxide surfaces. This process reduces the density of positive charges while increasing that of negative charges.35 At lower pH levels, the soil surface has a higher density of positive charges, enabling it to strongly adsorb Cr(VI) in its anionic form through electrostatic interactions. As pH increases, the number of positive surface charges decreases while negative charges increase, enhancing electrostatic repulsion between the negatively charged Cr(VI) anions and the soil surface, thereby significantly reducing the adsorption capacity.36 At lower pH levels, Cr(VI) predominantly exists as HCrO4−; whereas as pH increases, CrO42− becomes the dominant species.37 Notably, the adsorption affinity of Fe and Al oxides for CrO42− is markedly weaker than that for HCrO4−. Concurrently, OH− ions compete with CrO42− for available adsorption sites, further inhibiting Cr(VI) adsorption.33,38 Paddy soil and black soil were particularly sensitive to pH changes. In paddy soil, each 1-unit increase in pH led to a 134 mg kg−1 reduction in saturated sorption capacity, with reductions of 118 mg kg−1 in reduction capacity and 16 mg kg−1 in adsorption capacity. For black soil, the corresponding decreases were 132 mg kg−1, 114 mg kg−1, and 22 mg kg−1, respectively. The decreases in saturated sorption capacities for both soils exceeded 22% of their initial values, while their reduction capacities declined by more than 27%. This pronounced sensitivity is mainly attributed to the decrease in proton concentration with increasing pH, which directly retards proton-dependent reduction reactions,38 thus substantially diminishing the overall Cr(VI) sorption capacity of the soils. In contrast, fluvo-aquic soil exhibited much lower sensitivity to pH changes; a 1-unit increase in pH reduced its saturated sorption capacity by only 25 mg kg−1, consisting of a 20 mg kg−1 decrease in reduction capacity and a 5 mg kg−1 decrease in adsorption capacity. This can be ascribed to its inherently low saturated sorption capacity, further illustrating the inhibitory role of alkaline conditions in Cr(VI) transformation.
Further analysis indicates that as pH increases, the reduction capacity decreases by more than 85%, with its contribution to the saturated sorption capacity also declining by over 30%. Notably, while the adsorption capacity itself exhibits a significant downward trend (decreasing by 25% to 65%), its relative contribution to the saturated sorption capacity increases by more than 25%. This discrepancy demonstrates that pH exerts a far stronger inhibitory effect on the reduction process than on the adsorption process. Although increasing pH inhibits both processes, the effect is particularly pronounced for reduction reactions, given their high dependence on proton availability.
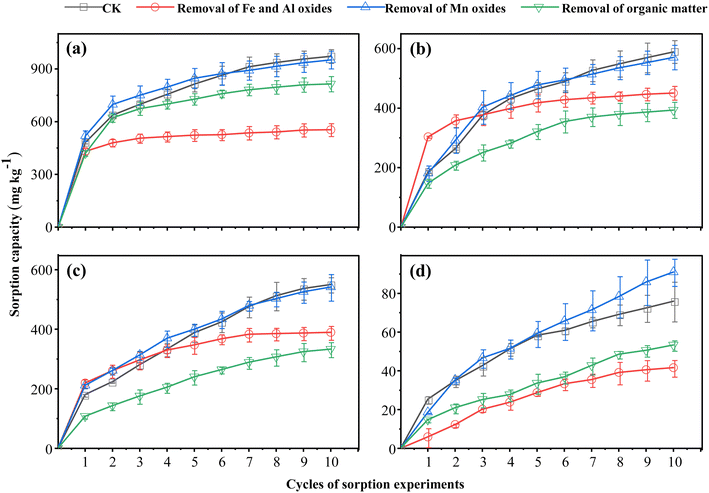
Fig. 5–7 respectively illustrate the sorption capacity, desorption capacity, and the contributions of adsorption and reduction to the saturated sorption capacity of Cr(VI) in the four soils after the removal of oxides and organic matter. The results indicate that the Cr(VI) saturated sorption capacity of all four soils decreased to varying degrees following organic matter removal, with the order of adsorption capacity remaining red soil > paddy soil > black soil > fluvo-aquic soil, and a significant negative correlation with pH was maintained. In terms of reduction capacity, all four soils exhibited decreases of over 60% following organic matter removal, and the proportion of reduction capacity decreased by more than 20%. Notably, in soils with high organic matter content (black soil and paddy soil), the reduction capacity and its proportion decreased by over 30%, rendering adsorption the dominant mechanism for Cr(VI) sorption. These findings suggest that organic matter is a key factor affecting Cr(VI) reduction in soil. Importantly, the reduction capacity share of red soil decreased to a lesser extent, likely due to its lower pH; under acidic conditions, even in the absence of organic matter, Cr(VI) can still undergo partial reduction through alternative pathways such as soil mineral-mediated reduction.39 Conversely, the Cr(VI) adsorption capacity of soils was generally enhanced by 20% to 40% after the removal of organic matter. This is because the positively charged sites on the surface of soil oxide colloids—originally masked and consumed by binding to negatively charged organic ions29—became available again following organic matter removal, thereby enhancing Cr(VI) adsorption capacity. Overall, the removal of organic matter had a considerable impact on the soil's ability to reduce and adsorb Cr(VI). Differences observed among soils with varying organic matter contents further confirm that organic matter is critical for Cr(VI) reduction in soils, and that acidic conditions can partially compensate for the impairment of reduction processes caused by organic matter deficiency.
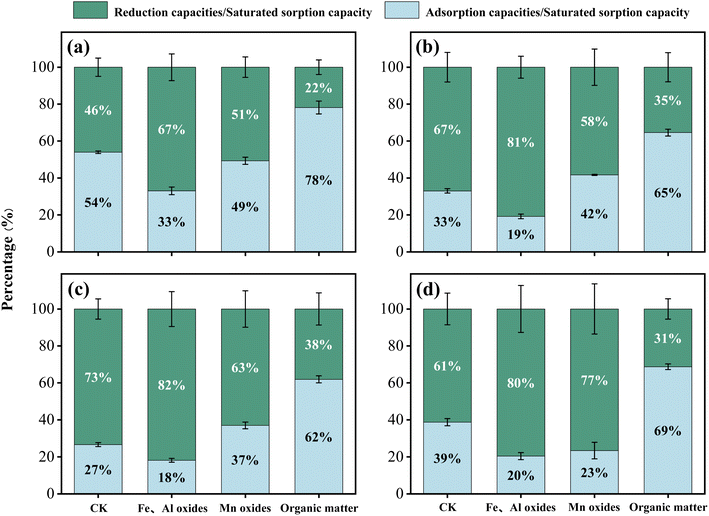 | ||
| Fig. 7 Effects of oxides and organic matter on the proportions of reduction and adsorption capacities. (a) Red soil; (b) paddy soil; (c) black soil; and (d) fluvo-aquic soil. | ||
Following the removal of Fe and Al oxides, the Cr(VI) adsorption capacity of all four soils decreased by more than 50%, indicating that Fe and Al oxides are the primary contributors to Cr(VI) adsorption in soils. Cr(VI) adsorption onto these oxides occurs primarily via two mechanisms: inner-sphere complexation (through monodentate and bidentate coordination) and outer-sphere complexation (via electrostatic interactions).40–42 The removal of Fe and Al oxides markedly reduces the number of available adsorption sites, thereby substantially diminishing the soils' Cr(VI) adsorption capacity. Interestingly, the decrease in the relative proportion of adsorption capacity to saturated sorption capacity following Fe and Al oxide removal was not significant (9–21%) compared to changes induced by modifications of other soil physicochemical properties. This observation may be attributed to two reasons. First, the inherent relative proportion of adsorption capacity is comparatively low, thus limiting the magnitude of change. Second, although Fe and Al oxides serve as the principal carriers for Cr(VI) adsorption in soils, their adsorption behaviors and complexation patterns are strongly pH-dependent. pH variations modulate both electrostatic and specific adsorption by altering the surface charge characteristics of the oxides and the chemical speciation of Cr(VI).13 These combined effects ultimately impact the overall Cr(VI) adsorption capacity of soils.
Previous studies have demonstrated that Mn oxides serve as the main oxidizing agents in the transformation of Cr(III) to Cr(VI) in soils,43 while their surface functional groups can also improve Cr(VI) adsorption through coordination mechanisms.44 Additionally, Mn-incorporated ferrihydrite has been shown to synergistically enhance Cr(VI) adsorption,45 justifying the inclusion of Mn oxides as a key factor in this study. However, our experimental data revealed no consistent changes in the measured parameters following the removal of Mn oxides from the soils. This is likely due to the low initial content of Mn oxides in the soils, which limited their influence on the overall sorption process. Interestingly, a transient increase in Cr(VI) sorption capacity was observed in soils with Mn oxides removed—particularly during the initial and middle stages of the reaction—compared to the control group. This phenomenon can be explained by redox kinetics: Mn oxides, being strongly oxidizing, rapidly oxidize Cr(III) to Cr(VI) in the early stages of the reaction while being reduced themselves in the process.46 Given their low abundance in the four soils examined, however, their oxidizing capacity was quickly depleted, resulting in a negligible overall increase in Cr(VI) concentration within the system. Consequently, the removal of Mn oxides enabled the soil's reducing components—previously inhibited by the presence of Mn oxides—to more efficiently reduce Cr(VI) to Cr(III).
The above results demonstrate that pH, Fe oxides, Al oxides, Mn oxides, and organic matter each exert varying degrees of influence on Cr(VI) sorption processes in soils. To further clarify the primary factors governing reduction and adsorption capacities, more comprehensive processing and analysis of the experimental data are required.
3.4 Key factors influencing soil Cr(VI) adsorption capacity
Pearson correlation analysis was conducted to quantify the strength and statistical significance of linear relationships among saturated sorption capacity, reduction capacity, adsorption capacity, and various soil physicochemical properties. The corresponding results are presented in the correlation heatmap (Fig. 8). Analysis of these capacity-related indicators revealed that saturated sorption capacity was strongly positively correlated with both reduction capacity (R = 0.840, p < 0.01) and adsorption capacity (R = 0.870, p < 0.01), indicating that both reduction and adsorption processes contribute substantially to the overall saturated sorption capacity. Moreover, adsorption capacity displayed a significant positive correlation with reduction capacity (R = 0.460, p < 0.05), implying that these two processes can interact under certain conditions and jointly affect the environmental fate of Cr(VI) in soils. | ||
| Fig. 8 Correlation heatmap among saturated sorption capacity, reduction capacity, adsorption capacity, and soil physicochemical properties. | ||
With respect to soil physicochemical properties, soil pH showed a strong negative correlation with saturated sorption capacity (R = −0.787, p < 0.01), reduction capacity (R = −0.808, p < 0.01), and adsorption capacity (R = −0.550, p < 0.01). These findings are in agreement with previous studies, further underscoring the critical role of pH in regulating sorption processes. The contents of Fe and Al oxides exhibited significant positive correlations with saturated sorption capacity (Fe: R = 0.860, p < 0.01; Al: R = 0.839, p < 0.01) and adsorption capacity (Fe: R = 0.613, p < 0.01; Al: R = 0.636, p < 0.01), whereas their correlations with reduction capacity were relatively weak (Fe: R = 0.150; Al: R = 0.220). These observations indicate that Fe and Al oxides enhance Cr(VI) retention in soils mainly by promoting adsorption rather than facilitating reduction. Although the correlations between organic matter content and adsorption capacity (R = −0.250), organic matter content and reduction capacity (R = 0.340), as well as between Mn oxide content and adsorption capacity (R = −0.240), were not statistically significant, experimental observations suggest that the potential effects of these components on Cr(VI) behavior should not be overlooked.
To further quantify the relative contributions of key factors to Cr(VI) saturated sorption capacity, reduction capacity, and adsorption capacity, a random forest model was constructed, leveraging the robust ensemble learning capabilities inherent to this algorithm (Fig. 9). The results showed that, for saturated sorption capacity, the most influential factors by order of importance were pH (27.8%), Al oxide (21.5%), Fe oxide (19.7%), organic matter (15.7%), and Mn oxide (15.3%). For reduction capacity, pH (45.4%) and organic matter (26.6%) emerged as the primary drivers, collectively accounting for 72% of the overall contribution. With regard to adsorption capacity, Fe, Al, and Mn oxides were identified as the main contributors, with relative importance values of 28.0%, 24.0%, and 18.5% respectively, totaling 70.5%.
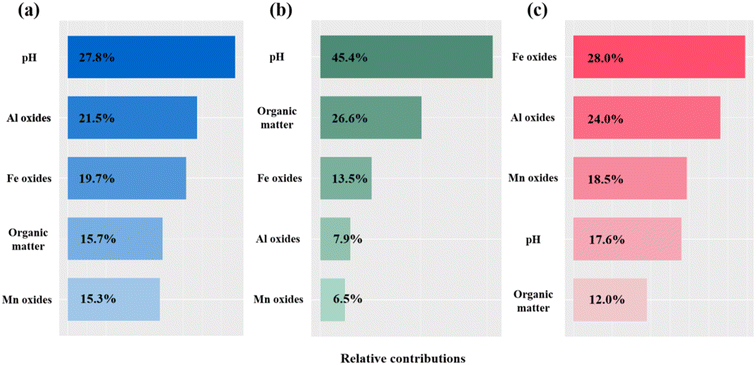 | ||
| Fig. 9 Relative contributions of key factors to saturated sorption capacity (a), reduction capacity (b), and adsorption capacity (c). | ||
Overall, the analysis highlighted that pH was the most influential factor governing Cr(VI) sorption processes in soils. Furthermore, Fe, Al, and Mn oxides predominantly contributed to Cr(VI) immobilization by enhancing adsorption mechanisms, whereas organic matter played a major role in facilitating reduction processes. These findings are largely consistent with the preceding analyses.
4. Conclusion
This study quantified the saturated sorption, adsorption, and reduction capacities of Cr(VI) in four soils along with the contributions of soil pH, organic matter, and Fe/Al/Mn oxides. Red soil had the highest saturated sorption capacity (972 mg kg−1), followed by black soil (551 mg kg−1), paddy soil (451 mg kg−1), and fluvo-aquic soil (76 mg kg−1). Increasing pH exerted a stronger inhibitory effect on the reduction process than on adsorption, especially in paddy soil and black soil, where the reduction capacity decreased by more than 27%. Organic matter removal decreased the reduction capacity by more than 65% while concurrently increasing the adsorption capacity by 20% to 40%. The removal of Fe and Al oxides resulted in a decline in adsorption capacity of over 50%. By contrast, the removal of Mn oxides had only a minor influence on the sorption process. Soil pH accounted for 27.8%, 45.4%, and 28.0% of the saturated sorption capacity, reduction capacity, and adsorption capacity, respectively. Fe and Al oxides collectively contributed more than 50% to the adsorption capacity, and organic matter contributed 26.6% to the reduction capacity. Based on the above findings, the relationship between key soil properties and Cr(VI) adsorption/reduction capacities has been clarified. This relationship can be used to accurately predict the stabilization potential of Cr(VI) in site-specific soils, thereby aiding in the identification of areas with higher migration risks. Furthermore, these insights not only provide a scientific basis for establishing differentiated risk assessment frameworks but also offer a reference for refining soil environmental quality standards.Author contributions
Ze Zhang: investigation, methodology, writing – original draft. Libing Zheng: investigation. Xinru Zhai: methodology, investigation. Shi-Wei Li: formal analysis. Helian Li: conceptualization, funding acquisition, supervision, writing – review and editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data will be made available on request.Supplementary information (SI): Table S1, which summarizes existing studies on Cr(VI) sorption in soils. See DOI: https://doi.org/10.1039/d5em00633c.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (42377019), the Shandong Provincial Natural Science Foundation, China (ZR2023MD026), and the Development Plan of Youth Innovation Team in Colleges and Universities of Shandong Province (2022KJ099).References
- Z. Yang, X. Zhang, Z. Jiang, Q. Li, P. Huang, C. Zheng, Q. Liao and W. Yang, Reductive materials for remediation of hexavalent chromium contaminated soil – A review, Sci. Total Environ., 2021, 773, 145654 CrossRef PubMed.
- M. Ao, X. Chen, T. Deng, S. Sun, Y. Tang, J. L. Morel, R. Qiu and S. Wang, Chromium biogeochemical behaviour in soil-plant systems and remediation strategies: A critical review, J. Hazard. Mater., 2022, 424, 127233 CrossRef PubMed.
- D. Hou, X. Jia, L. Wang, S. P. McGrath, Y. G. Zhu, Q. Hu, F. J. Zhao, M. S. Bank, D. O'Connor and J. Nriagu, Global soil pollution by toxic metals threatens agriculture and human health, Science, 2025, 388(6744), 316–321 CrossRef PubMed.
- X. Liu, X. Chen, X. Zhang, H. Guo, C. Zhang, X. Zang and B. Li, Quantifying the influence of soil factors on the migration of chromium (VI), Process Saf. Environ., 2021, 155, 32–40 CrossRef.
- U. Zulfiqar, F. U. Haider, M. Ahmad, S. Hussain, M. F. Maqsood, M. Ishfaq, B. Shahzad, M. M. Waqas, B. Ali, M. N. Tayyab, S. A. Ahmad, I. Khan and S. M. Eldin, Chromium toxicity, speciation, and remediation strategies in soil-plant interface: A critical review, Front. Plant Sci., 2023, 13, 1081624 CrossRef PubMed.
- M. A. Islam, M. J. Angove, D. W. Morton, B. K. Pramanik and M. R. Awual, A mechanistic approach of chromium (VI) adsorption onto manganese oxides and boehmite, J. Environ. Chem. Eng., 2020, 8, 103515 CrossRef.
- D. Park, S. R. Lim, Y. S. Yun and J. M. Park, Reliable evidences that the removal mechanism of hexavalent chromium by natural biomaterials is adsorption-coupled reduction, Chemosphere, 2007, 70, 298–305 CrossRef PubMed.
- Y. Yang, Y. Peng, Z. Yang, P. Cheng, F. Li, M. Wang and T. Liu, The kinetics of aging and reducing processes of Cr(VI) in two soils, Bull. Environ. Contam. Toxicol., 2019, 103, 82–89 CrossRef PubMed.
- B. R. James and R. J. Bartlett, Behavior of chromium in soils: VII. Adsorption and reduction of hexavalent forms, J. Environ. Qual., 1983, 12(2), 177–181 CrossRef.
- A. R. Wadhawan, A. T. Stone and E. J. Bouwer, Biogeochemical controls on hexavalent chromium formation in estuarine sediments, Environ. Sci. Technol., 2013, 47, 8220–8228 Search PubMed.
- G. Choppala, N. Bolan and B. Seshadri, Chemodynamics of chromium reduction in soils: Implications to bioavailability, J. Hazard. Mater., 2013, 261, 718–724 CrossRef PubMed.
- X. Gu, J. Xie, X. Wang and L. J. Evans, A simple model to predict chromate partitioning in selected soils from China, J. Hazard. Mater., 2017, 322, 421–429 CrossRef PubMed.
- C. Pérez, J. Antelo, S. Fiol and F. Arce, Modeling oxyanion adsorption on ferralic soil, part 2: Chromate, selenate, molybdate, and arsenate adsorption, Environ. Toxicol. Chem., 2014, 33(10), 2217–2224 CrossRef PubMed.
- J. M. Tinjum, C. H. Benson and T. B. Edil, Mobilization of Cr(VI) from chromite ore processing residue through acid treatment, Sci. Total Environ., 2008, 391, 13–25 CrossRef CAS.
- S. Liu, C. Miao, S. Yao, H. Ding and K. Zhang, Soil stabilization/solidification (S/S) agent---water-soluble thiourea formaldehyde (WTF) resin: Mechanism and performance with cadmium (II), Environ. Pollut., 2021, 272, 116025 CrossRef CAS PubMed.
- J. Hadi, C. Tournassat and C. Lerouge, Pitfalls in using the hexaamminecobalt method for cation exchange capacity measurements on clay minerals and clay-rocks: Redox interferences between the cationic dye and the sample, Appl. Clay Sci., 2016, 119, 393–400 CrossRef CAS.
- G. J. Bouyoucos, Hydrometer method improved for making particle size analyses of soils, Agron. J., 1962, 54(5), 464–465 CrossRef.
- O. P. Mehra and M. L. Jackson, Iron oxide removal from soils and clays by a dithionite–citrate system buffered with sodium bicarbonate, Clays Clay Miner., 2013, 317–327 Search PubMed.
- C. M. Chon, J. G. Kim, G. H. Lee and T. H. Kim, Influence of extractable soil manganese on oxidation capacity of different soils in Korea, Environ. Geol., 2008, 55(4), 763–773 CrossRef CAS.
- S. Staunton and R. Naidu, Buffering capacity, in Encyclopedia of Soils in the Environment, ed. M. J. Goss, and M. Oliver, Academic Press, Oxford, 2nd edn, 2023, pp. 15–21 Search PubMed.
- R. Mikutta, M. Kleber, K. Kaiser and R. Jahn, Soil Sci. Soc. Am. J., 2005, 69(1), 120–135 CrossRef CAS.
- Q. Ke, J. Ren, K. Feng, Z. Zhang, W. Huang, X. Xu, L. Zhao, H. Qiu and X. Cao, Crucial roles of soil inherent Fe-bearing minerals in enhanced Cr(VI) reduction by biochar: The electronegativity neutralization and electron transfer mediation, Environ. Pollut., 2024, 350, 124014 CrossRef CAS PubMed.
- A. Neaman, F. Mouélé, F. Trolard and G. Bourrié, Improved methods for selective dissolution of Mn oxides: Applications for studying trace element associations, Appl. Geochem., 2004, 19(6), 973–979 CrossRef CAS.
- P. M. Jardine, M. A. Stewart, M. O. Barnett, N. T. Basta, S. C. Brooks, S. Fendorf and T. L. Mehlhorn, Influence of soil geochemical and physical properties on chromium(VI) sorption and bioaccessibility, Environ. Sci. Technol., 2013, 47, 11241–11248 CrossRef CAS.
- R. Bartlett and B. James, Behavior of chromium in soils: III. Oxidation, J. Environ. Qual., 1979, 8(1), 31–35 CrossRef CAS.
- MEPPRC (Ministry of Ecology and Environment of the People’s Republic of China), SAMRPRC (State Administration for Market Regulation of the People’s Republic of China), Soil environmental quality standard: risk control standard for soil contamination of agricultural land (Trial), 2018, Available at: https://www.mee.gov.cn/ywgz/fgbz/bz/bzwb/trhj/201807/W020190626595212456114.pdf.
- B. Kumari and S. Dutta, Integrating starch encapsulated nanoscale zero-valent iron for better chromium removal performance, J. Water Process Eng., 2020, 37, 101370 CrossRef.
- Y. Pang, L. Kong, D. Chen and G. Yuvaraja, Rapid Cr(VI) reduction in aqueous solution using a novel microwave-based treatment with MoS2-MnFe2O4 composite, Appl. Surf. Sci., 2019, 471, 408–416 CrossRef CAS.
- E. F. Covelo, M. L. Andrade and F. A. Vega, Heavy metal adsorption by humic umbrisols: selectivity sequences and competitive sorption kinetics, J. Colloid Interface Sci., 2004, 280, 1–8 CrossRef CAS.
- S. T. Aldmour, I. T. Burke, A. W. Bray, D. L. Baker, A. B. Ross, F. L. Gill, G. Cibin, M. E. Ries and D. I. Stewart, Abiotic reduction of Cr(VI) by humic acids derived from peat and lignite: kinetics and removal mechanism, Environ. Sci. Pollut. Res., 2019, 26(5), 4717–4729 CrossRef CAS.
- Y. Xing, Y. Zheng and X. Wang, Integrated strategies for effective remediation of chromium-contaminated soils: Advancements, challenges, and sustainability implications, Environ. Adv., 2025, 19, 100614 CrossRef CAS.
- F. Yan, PhD Thesis, Northwest A & F University, 2011 Search PubMed.
- A. Gaffer, A. A. Al Kahlawy and D. Aman, Magnetic zeolite-natural polymer composite for adsorption of chromium (VI), Egypt. J. Pet., 2017, 26, 995–999 CrossRef.
- H. Hua, Z. Zhao, R. Xu, E. Chang, D. Fang, Y. Dong, Z. Hong, R. Shi and J. Jiang, Effect of ferrolysis and organic matter accumulation on chromate adsorption characteristics of an oxisol-derived paddy soil, Sci. Total Environ., 2020, 744, 140868 CrossRef CAS.
- Y. Liu, Z. Q. Zhu, X. S. He, C. Yang, Y. Q. Du, Y.-D. Huang, P. Su, S. Wang, X. X. Zheng and Y. J. Xue, Mechanisms of rice straw biochar effects on phosphorus sorption characteristics of acid upland red soils, Chemosphere, 2018, 207, 267–277 CrossRef CAS.
- D. A. Gkika, A. K. Tolkou, I. A. Katsoyiannis and G. Z. Kyzas, The adsorption-desorption-regeneration pathway to a circular economy: The role of waste-derived adsorbents on chromium removal. Separation Purif, Technol, 2025, 368, 132996 CAS.
- S. Mallick, S. S. Dash and K. M. Parida, Adsorption of hexavalent chromium on manganese nodule leached residue obtained from NH3–SO2 leaching, J. Colloid Interface Sci., 2006, 297, 419–425 CrossRef CAS.
- A. Kunhikrishnan, R. Thangarajan, N. S. Bolan, Y. Xu, S. Mandal, D. B. Gleeson, B. Seshadri, M. Zaman, L. Barton, C. Tang, J. Luo, R. Dalal, W. Ding, M. B. Kirkham and R. Naidu, Chapter one - Functional relationships of soil acidification, liming, and greenhouse gas flux, in Advances in Agronomy, D. L. Sparks, Academic Press., San Diego, CA, 2016, pp. 1–71 Search PubMed.
- Y. H. Fseha, J. O. Eniola, B. Sizirici, S. Stephen, I. Yildiz, A. Khaleel and A. Adamson, Application of natural earth-based materials as adsorbents for the treatment of chromium (VI)-contaminated tannery wastewater: Box-Behnken and fixed-bed column optimization, Sustain. Chem. Environ., 2024, 7, 100127 CrossRef.
- C. P. Johnston and M. Chrysochoou, Mechanisms of chromate adsorption on boehmite, J. Hazard. Mater., 2015, 281, 56–63 CrossRef CAS.
- Y. Li, J. Lin, Y. Wu, S. Jiang, C. Huo, T. Liu, Y. Yang and Y. Ma, Transformation of exogenous hexavalent chromium in soil: Factors and modelling, J. Hazard. Mater., 2024, 480, 135799 CrossRef CAS PubMed.
- V. Veselská, R. Fajgar, S. Číhalová, R. M. Bolanz, J. Göttlicher, R. Steininger, J. A. Siddique and M. Komárek, Chromate adsorption on selected soil minerals: Surface complexation modeling coupled with spectroscopic investigation, J. Hazard. Mater., 2016, 318, 433–442 CrossRef.
- S. Namgung, M. J. Kwon, N. P. Qafoku and G. Lee, Cr(OH)3(s) oxidation induced by surface catalyzed Mn(II) oxidation, Environ. Sci. Technol., 2014, 48, 10760–10768 CrossRef CAS PubMed.
- M. Ao, S. Sun, T. Deng, J. Li, T. Liu, Y. Tang, S. Wang and R. Qiu, Interaction between chromite and Mn(II/IV) under anoxic, oxic and anoxic-oxic conditions: Dissolution, oxidation and pH dependence, J. Environ. Manag., 2024, 349, 119475 CrossRef CAS.
- C. Liang, F. Fu and B. Tang, Mn-incorporated ferrihydrite for Cr(VI) immobilization: Adsorption behavior and the fate of Cr(VI) during aging, J. Hazard. Mater., 2021, 417, 126073 CrossRef CAS.
- B. Dhal, H. N. Thatoi, N. N. Das and B. D. Pandey, Chemical and microbial remediation of hexavalent chromium from contaminated soil and mining/metallurgical solid waste: A review, J. Hazard. Mater., 2013, 250–251, 272–291 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |