Process-based VOC emission characteristics, environmental impact and health risk assessment in typical breweries in Beijing
Huan
Zhao
a,
Hongna
Ren
a,
Hailin
Wang
b,
Yiwen
Wang
a,
Yiming
Lu
a,
Jie
Cheng
a,
Guoxia
Jiang
*a and
Zhengping
Hao
 *a
*a
aNational Engineering Laboratory for VOCs Pollution Control Material & Technology, Research Center for Environmental Material and Pollution Control Technology, University of Chinese Academy of Sciences, Beijing 101408, China. E-mail: zphao@ucas.ac.cn; jiangguoxia@ucas.ac.cn; Tel: +86-10-62923564 Tel: +010-69672977
bBeijing Key Laboratory for Urban Atmospheric VOCs Pollution Control and Technology Application, Beijing Municipal Research Institute of Environmental Protection, Beijing 100037, China
First published on 22nd December 2025
Abstract
Brewing is an understudied but influential source of VOC emissions in the food manufacturing industry. In this study, we conducted a first comprehensive analysis of process-based VOC concentration characteristics, ozone formation potential (OFP), secondary organic aerosol formation potential (SOAFP) and health risks in two typical breweries in Beijing that use malted barley, hops, water, and yeast. In Brewery A, 35 to 53 distinct VOC species were detected, with total mass concentrations ranging from 148.17 ± 18.64 µg m−3 to 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 225.91 ± 1912.51 µg m−3. Brewery B demonstrated comparable patterns, with 28 to 49 species detected at concentrations between 104.49 ± 8.48 µg m−3 and 10
225.91 ± 1912.51 µg m−3. Brewery B demonstrated comparable patterns, with 28 to 49 species detected at concentrations between 104.49 ± 8.48 µg m−3 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368.87 ± 879.47 µg m−3. Process-stage analysis identified boiling and fermentation stages as the key stages with the highest VOC concentrations, dominated by oxygenated VOCs (OVOCs) such as acetaldehyde, acetone, ethyl acetate, and 2-butanone, as well as the alkane isobutane. Atmospheric dispersion modeling (AERMOD) indicated negligible public health risks from organized stack emissions. In contrast, occupational health assessment revealed significant risks for workers from fugitive emissions, with the cumulative hazard index (HI) far exceeding the threshold. The OFP and SOAFP results, representing the secondary pollutant formation potential of the source mixtures, highlighted OVOCs and aromatics as priority control species for mitigating the secondary pollution potential. The findings demonstrate that VOC control strategies must be differentiated, with large-scale breweries prioritizing organized emissions, while small breweries urgently need to control fugitive emissions. This study aims to promote the implementation of VOC regulations and occupational health protection strategies within the brewing industry.
368.87 ± 879.47 µg m−3. Process-stage analysis identified boiling and fermentation stages as the key stages with the highest VOC concentrations, dominated by oxygenated VOCs (OVOCs) such as acetaldehyde, acetone, ethyl acetate, and 2-butanone, as well as the alkane isobutane. Atmospheric dispersion modeling (AERMOD) indicated negligible public health risks from organized stack emissions. In contrast, occupational health assessment revealed significant risks for workers from fugitive emissions, with the cumulative hazard index (HI) far exceeding the threshold. The OFP and SOAFP results, representing the secondary pollutant formation potential of the source mixtures, highlighted OVOCs and aromatics as priority control species for mitigating the secondary pollution potential. The findings demonstrate that VOC control strategies must be differentiated, with large-scale breweries prioritizing organized emissions, while small breweries urgently need to control fugitive emissions. This study aims to promote the implementation of VOC regulations and occupational health protection strategies within the brewing industry.
Environmental significanceVolatile organic compounds (VOCs) from food and beverage manufacturing represent significant yet inadequately characterized sources of atmospheric oxidants and particulate matter. This investigation provides the first process-based analysis of brewery emissions, with boiling and fermentation identified as critical sources characterized by OVOCs. The study reveals substantially different risk profiles between emission types, with organized releases showing minimal community impact compared to the substantial occupational hazards created by fugitive emissions. Additionally, aromatic hydrocarbons display disproportionate contributions to secondary organic aerosol formation relative to their minor mass fractions. These findings establish a scientific foundation for developing differentiated control strategies that concurrently address secondary pollution formation and workplace health risks, thereby facilitating precision environmental management in food and beverage industry operations. |
1. Introduction
Volatile Organic Compounds (VOCs) are crucial precursors of PM2.5 and O3 that participate in photochemical reactions that lead to photochemical smog and secondary organic aerosol (SOA) pollution, which are the main contributors to haze pollution.1,2 Moreover, exposure to VOCs can induce cancer and other diseases, with long-term exposure to high concentrations having the most significant effects on the respiratory tract and lungs,3 thereby posing a major threat to human health. Numerous previous studies have shown that industrial sources are the dominant anthropogenic contributors to VOC emissions in China.4,5 Consequently, understanding the sources and emission characteristics of VOCs is critically important for the implementation of effective control strategies.Research findings consistently identify industrial sources as the primary contributors to VOC emissions. Industrial emissions stand out due to their multifaceted origins, high intensity, complex composition, and elevated concentrations.6 In the past few years, Chinese scholars have paid much attention to quantifying and characterizing VOC emissions from specific industries, and previous studies have focused on large scale and heavily polluting industries such as the coking and coating industries. However, only limited studies have been conducted on the emission characterization of VOCs from other light industrial sources, such as the food manufacturing industry and the chemical materials industry.7 In the 21st century, China's food industry has experienced rapid and steady growth,8,9 leading to increased VOC emissions from enterprises. These emissions predominantly consist of malodorous releases and organic by-products of fermentation.10 The food industry occupies a prominent position among the sources of VOC emissions, after manufacturing, pharmaceuticals, and transportation.11–13 While current research has primarily focused on odorous emissions and organic by-products in the food industry, there is a noticeable dearth of studies examining VOCs at different process stages.14 Therefore, addressing VOC emissions from the food industry necessitates a more comprehensive understanding of the specific stages involved in food processing.
Currently, there is almost no research concerning the detection and analysis of breweries. Based on previous research, odor gases are released during the production of beer,15,16 which involves processes such as malting, saccharification, fermentation, boiling, filtration, filling.17 A general idea of the type of pollutant can be obtained, but it is not sufficiently clear and the range of concentrations is not clear. Previous studies related to the brewing industry have focused on the characterization of odorants of beer itself and the pollution characteristics of beer wastewater and have paid more attention to individual facilities or generic industry average emission values, without explicitly examining the impacts of different scales and technologies on VOC emissions. There is a lack of research on VOC emission characteristics and health risks at different process stages, so studying the emission characteristics of VOCs at different scales is essential for developing more targeted control strategies.
In this study, VOC emissions from organized and fugitive sources of the brewing process were analyzed by comparing two breweries with the same core technologies but different scales. The study aimed to characterize VOC emission characteristics, quantify their atmospheric impacts through OFP and SOA generation assessments, model dispersion patterns using AERMOD simulations, and evaluate health risks following the U.S. Environmental Protection Agency (U.S. EPA) risk assessment method. The key objectives included: (1) comparative analysis of organized versus fugitive emission characteristics in brewing processes; (2) mechanistic evaluation of VOC-driven photochemical ozone and SOA formation; (3) quantifying comprehensive health risks for both non-carcinogenic and carcinogenic risks and analyzing the dispersion of organized VOCs using the AERMOD model; (4) comparing breweries of different scales and focusing on their own differences to determine the factors affecting the emission characteristics of VOCs. The results of the study advance the scientific understanding of pollution characteristics and health risks in the brewing industry, and offer differentiated emission reduction pathways for breweries of varying scales, effectively circumventing the limitations of "one-size-fits-all" environmental regulatory approaches.
2. Methodology
2.1. Sampling sites and VOC sampling
In this study, two breweries with different scales of production and technological levels were selected in Beijing, China, designated as Brewery A and Brewery B, respectively. Both breweries primarily produce lager beer, using malted barley, hops, water, and yeast as the main raw materials. The location is shown in Fig. S1. The differences between Brewery A and Brewery B in terms of production scale, process details, and equipment types are summarized in Table S1. Brewery A employs fully automated production lines, enclosed fermentation systems, and regenerative thermal oxidation (RTO) for exhaust gas treatment. Brewery B retains semi-automated equipment, open fermentation processes, and activated carbon adsorption for waste gas treatment. Involving different company scales, the rated capacities of Brewery A and Brewery B are 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 10
000 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons per year, respectively. During the sampling period, both breweries were operating at or near their full rated capacity. By comparing the two breweries, it is possible to compare the impact of the production mode and treatment technology of the enterprises on VOC emissions.
000 tons per year, respectively. During the sampling period, both breweries were operating at or near their full rated capacity. By comparing the two breweries, it is possible to compare the impact of the production mode and treatment technology of the enterprises on VOC emissions.
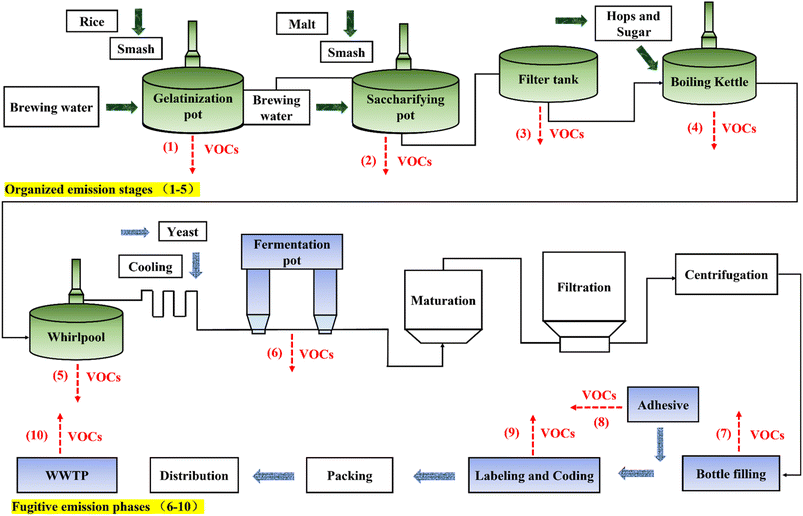
The sampling period was June–July 2022, and the sampling cycle included two complete production batches, mainly focusing on the peak season. A total of 44 samples were collected during each sampling period. The typical beer production process and VOC emission points are shown in Fig. 1. The process and pollution emission links were carefully investigated to select appropriate sampling sites for VOCs. In addition, background monitoring points were set up 500 meters upwind and downwind of the breweries. The organized emission sampling points included the stages of gelatinization, saccharification, filtration, boiling and sedimentation. The fugitive sampling points include five stages of fermentation, filling, labeling and coding, adhesives and emission of VOCs from the biobox at the sewage station. The selected sample sites covered almost all target factories' mission units. During the sampling period, the production status and processes of the breweries operated normally, so the sampling was well representative.
Air samples were taken using 3.2 L fused silica stainless steel canisters (Entech Instruments, Inc., Simi Valley, CA, USA). The containers should be pre-cleaned three times with high purity nitrogen and evacuated to <10 Pa.18,19 Samples were collected using polytetrafluoroethylene (Teflon) tubing connected from the particulate filter to the SUMMA canister.20 A flow restrictor valve was connected to the inlet of the canister to control the flow rate to 20 mL min−1, and the sampling time was 120 min. Before sampling, a portable photoionization detector (PID, ppbRAE 3000, RAE Systems, San Jose, CA, USA) was used to make initial measurements of VOC concentrations at each sampling site. Fugitive emission samples were collected in the workshop at a height of 1.5 m above the ground, positioned within 1–2 m of the primary emission point for each process stage to capture representative process-specific concentrations. Workshop air samples were collected at a height of 1.5 meters above the ground near primary production units, The canisters were immediately sent to the laboratory and analyzed within 15 days.21
2.2. Sample analysis and quality assurance
Samples need to be diluted with high purity nitrogen prior to detection, and the dilution ratio was determined by the PID value of each sample. The analysis was performed using a gas chromatograph equipped with a mass selective detector and a flame ionization detector (7890B-5977B GC-MSD; Agilent Technologies Inc., USA) and coupled with a pre-concentrator system (model 7200, Entech Instruments Inc., USA).22 The system incorporated cryogenic preconcentration to eliminate water and carbon dioxide interference. Given the analytical challenge of separating C2–C12 compounds, we implemented a dual-column strategy. For light hydrocarbon separation, including ethane, ethene, acetylene, propane, and propene, we utilized a Plot Q column with FID detection following established techniques.23 The remaining 112 compounds were separated using a DB-1 column with MS detection. The initial temperatures were held at 5 °C for 6 min, increased at a rate of 5 °C min−1 to 200 °C, and kept for 8 minutes until all target compounds came out finally.24The standard gas used for measurement included the Photochemical Assessment Monitoring Stations (PAMs) standard mixture (56 NMHCs, Linde Spectra Gases, Inc., USA) and TO-15 standard mixture (65 compounds, Linde Spectra Gases, Inc., USA).25,26 Based on these standard gases, calibration curves were calculated for each target VOC species using a serial concentration gradient (7 levels: 0.5, 1.0, 2.0, 5.0, 10.0, 15, and 20 ppb). The correlation coefficients (R2) of calibration curves were more than 0.99. Bromochloromethane, 1,2-difluorobenzene, chlorobenzene-d5, and 1,4-difluorobenzene of known concentrations were used as internal standards for the calibration of the system. The measurement accuracy of each species was within 5%. In addition, the daily calibration results of the measuring system were consistent, indicating that the system was stable.
For the quality assurance (QA) and quality control (QC), no detectable VOCs were present when ten laboratory blank samples were processed using the same method as the real samples. No discernible contamination was found in the field blanks. The total recovery efficiencies of VOCs ranged from 76% to 128%, and the variance coefficient was <20%. The detection limits were 0.17–2.57 µg m−3. The relative correlation for the standard curve was in the range of 0.992–0.999.
2.3. Estimation of the secondary pollution formation potential
In this study, both the OFP and SOAFP were calculated based on VOC mass concentrations. This approach aims to evaluate the intrinsic chemical reactivity and aerosol formation potential of VOCs at each process stage. The types of measured VOCs, chemical categories, MIR values, and SOAP values are shown in Table S2. It is crucial to note that these values cannot be directly equated to the absolute mass of ozone or SOA ultimately formed in the atmosphere. The latter depends on total emission rates, subsequent atmospheric dispersion and chemistry, which are affected by factors beyond the scope of this study, including emission transport efficiency, concentrations of nitrogen oxides, and aerosol concentration levels.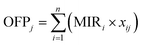 | (1) |
In this study, the contributions of VOC species to SOA were assessed based on Derwent's SOAP coefficient method.31 The SOAP index quantifies the relative SOA formation capacity of organic compounds normalized to toluene, where toluene's SOAP value is standardized at 100 under equivalent mass emission conditions. Toluene was selected as the reference compound due to its emission characteristics and its widespread recognition as a predominant anthropogenic SOA precursor.32,33 The methodology for calculating SOAP is shown in eqn (2).
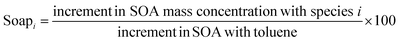 | (2) |
| SOAFPi = VOCi × SOAPi × FACtoluene | (3) |
2.4. Atmospheric diffusion simulation and health risk assessment
In this study, the assessment of health risks adopts a combined approach of the U.S. EPA Inhalation Risk Assessment Model and the AERMOD atmospheric dispersion model.| Q = C × F | (4) |
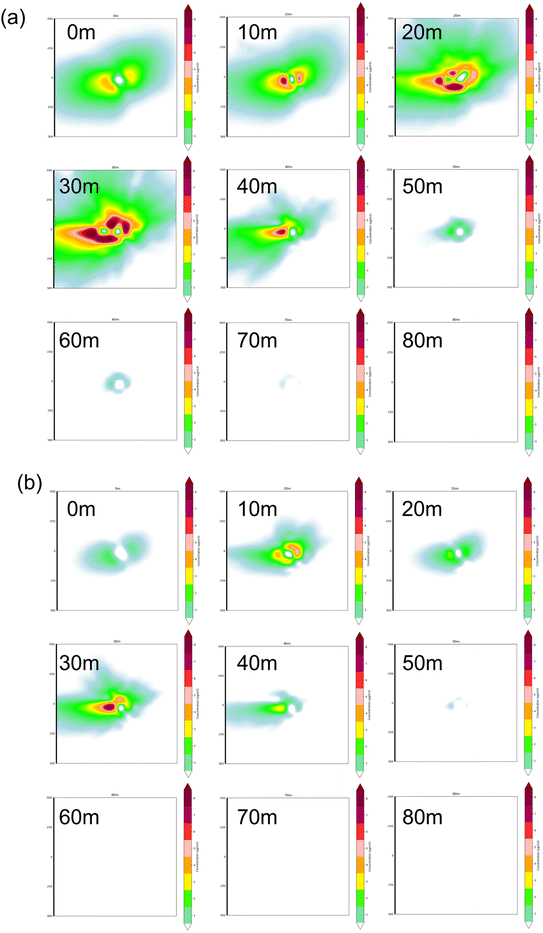
The simulation utilized hourly surface meteorological data (wind speed, wind direction, temperature, cloud cover, etc.) from a nearby national weather station throughout 2022. Covering a 500 m × 500 m area centered on the brewery with a receptor spacing of 10 m, the analysis examined vertical dispersion processes from 0 to 80 m. AERMOD input parameters are shown in Table S3.
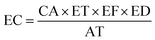 | (5) |
Inhalation risk assessment involves both non-carcinogenic risk and carcinogenic risk,36,37 expressed, respectively, as the Hazard Quotient (HQ) and the Lifetime Cancer Risk (LCR). The Hazard Index (HI) is the sum of individual HQs for various pollutants. HQ is classified into five risk levels, the HQ risk level is divided into five levels, with HQ <0.1 indicating negligible risk, 0.1 < HQ < 0.5 indicating low risk, 0.5 < HQ < 1 indicating moderate risk, 1 < HQ < 2 indicating high risk, and HQ ≥2.0 indicating extremely high risk. Carcinogenic risk is divided into four levels, with compounds greater than 10−4, between 10−5 and 10−4, between 10−6 and 10−5, and below 10−6 considered as "definite," "probable," "possible," and "negligible risk," respectively. The calculation formulas are (5–7):
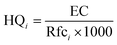 | (6) |
| HI = ∑HQi | (7) |
| LCRi = EC × IURi | (8) |
3. Results and analysis
3.1. Overview of VOC concentrations
Quantifying VOC concentrations and emission contributions across production stages enables the systematic identification of dominant species and distribution characteristics. The 116 analyzed VOCs were classified into six primary groups based on their functional groups: alkanes, halogenated hydrocarbons, alkenes, oxygenated hydrocarbons, aromatic hydrocarbons, and other species including CS2 and acetylene.38 The concentrations of VOCs at different stages of the breweries and the contribution rates of various components are illustrated in Fig. 2.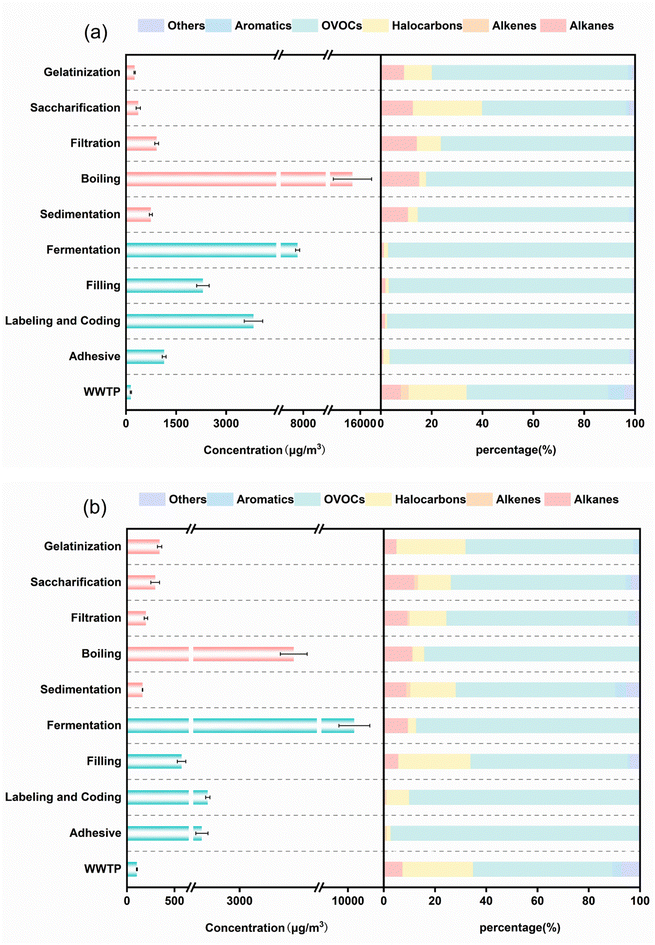 | ||
| Fig. 2 The concentrations of VOCs and the percentage composition of each component by mass at different stages in Brewery A (a) and Brewery B (b). | ||
The organized emission stages include five processes of gelatinization, saccharification, filtration, boiling, and sedimentation, with significant variations observed across stages. Brewery A detected 35, 38, 47, 53, and 37 VOC species across these stages, while Brewery B demonstrated 28, 42, 28, 48, and 38 types, respectively. From Fig. 2, it can be observed that both breweries displayed peak VOC concentrations during the boiling stage, with Brewery A registering 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 225.91 ± 1912.51 µg m−3 and Brewery B 4752.01 ± 432.41 µg m−3. The high temperature during the boiling stage accelerates the escape of VOCs, and the added hops contain a large quantity of volatile terpenes, which are highly volatile compounds at high temperatures. OVOCs dominated emissions across all organized production stages, accounting for 56.6–83.1% of total VOCs in Brewery A and 62.3–83.6% in Brewery B.
225.91 ± 1912.51 µg m−3 and Brewery B 4752.01 ± 432.41 µg m−3. The high temperature during the boiling stage accelerates the escape of VOCs, and the added hops contain a large quantity of volatile terpenes, which are highly volatile compounds at high temperatures. OVOCs dominated emissions across all organized production stages, accounting for 56.6–83.1% of total VOCs in Brewery A and 62.3–83.6% in Brewery B.
The proportional contributions of the top ten VOC species in both organized and fugitive emission phases for each brewery are illustrated in Fig. S2–S5. In Brewery A, the priority pollutants include acetaldehyde, acetone, isobutane, ethyl acetate, and 2,3-dimethylbutane, with concentrations of 9913.76 ± 257.21 µg m−3, 2662.36 ± 98.11 µg m−3, 1922.89 ± 66.64 µg m−3, 639.84 ± 19.62 µg m−3 and 581.90 ± 14.31 µg m−3, respectively. In contrast, the characteristic species in Brewery B are acetaldehyde, ethyl acetate, 2-butanone, isobutane, and 1,1-dichloroethene, with concentrations of 2729.79 ± 80.27 µg m−3, 1134.16 ± 24.54 µg m−3, 513.70 ± 18.04 µg m−3, 253.74 ± 12.21 µg m−3, and 189.19 ± 6.39 µg m−3, respectively.
Fugitive emissions predominantly occur during non-enclosed operational phases, exhibiting irregular release patterns and intermittent temporal characteristics. The stages of fugitive emissions include five sources identified as fermentation units, filling stations, inkjet labeling operations, adhesive volatilization, and biological treatment tanks in wastewater facilities. Brewery A demonstrated VOC species counts of 44, 43, 40, 40, and 38 across these stages, while Brewery B exhibited 49, 39, 40, 35, and 36. The fermentation stage emerged as the dominant emission source, displaying both the highest VOC diversity and concentration levels due to elevated airflow rates, complex substrate compositions, and abundant byproduct generation. OVOCs and halogenated hydrocarbons constituted the primary components of fugitive emissions. In Brewery A, OVOCs accounted for 55.7–97.1% of total emissions, with ethyl acetate reaching 7837.77 ± 153.22 µg m−3 during fermentation. Acetaldehyde and 2-butanone dominated inkjet labeling emissions at 705.39 ± 21.14 µg m−3 and 2259.19 ± 44.63 µg m−3, respectively. Filling stages primarily released ethyl acetate, acetaldehyde, and 2-butanone. Brewery B showed OVOC concentrations ranging from 54.5% to 96.4%, accompanied by halogenated hydrocarbons from 1.6% to 28.1%. Fermentation emissions were dominated by acetaldehyde and ethyl acetate, collectively representing 97.0% of total OVOCs. The filling stage primarily released acetaldehyde, 1,2-dichloropropane, and ethyl acetate, while 2-butanone exhibited peak concentrations during inkjet labeling operations. The specific characteristic pollutants for fugitive emissions can be observed in Fig. S3 and S5.
The analysis results show that the overall emission characteristics of VOCs from the two breweries are similar, with OVOCs dominating and the key emission stages being boiling and fermentation. At the concentration level, Brewery A emissions are dominated by organized emissions, while brewery B is dominated by fugitive emissions. The reason for this is that Brewery A has a high capacity and a high degree of process confinement, so the organized emissions are concentrated, whereas Brewery B has a high degree of equipment openness, so there is a high percentage of fugitive emissions. Essentially, it is the difference in the degree of industrialization and traditional techniques. It is confirmed that the difference in VOC emissions from breweries are determined by a combination of production scale and process technology. The consistency in the dominant VOC species across both breweries, despite their scale and technological differences, points to their role as characteristic pollutants for the brewing industry. The prevalence of acetaldehyde and ethyl acetate is a direct signature of yeast metabolism during fermentation, while isobutane is likely a characteristic volatile released from hops during the high-temperature boiling process. Therefore, these compounds serve as key chemical indicators for brewery emissions.
3.2. Secondary pollution formation potential results
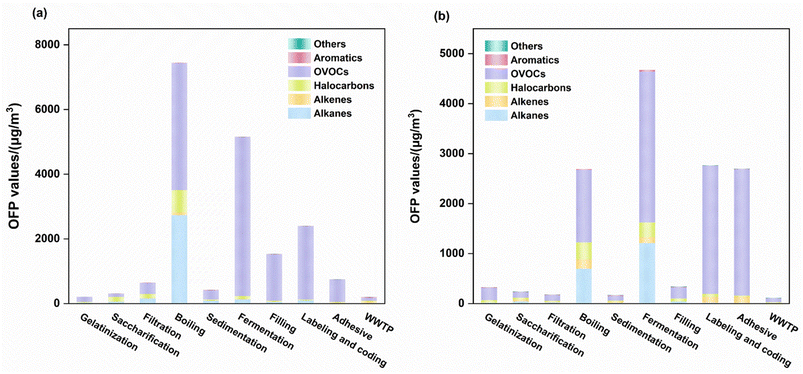
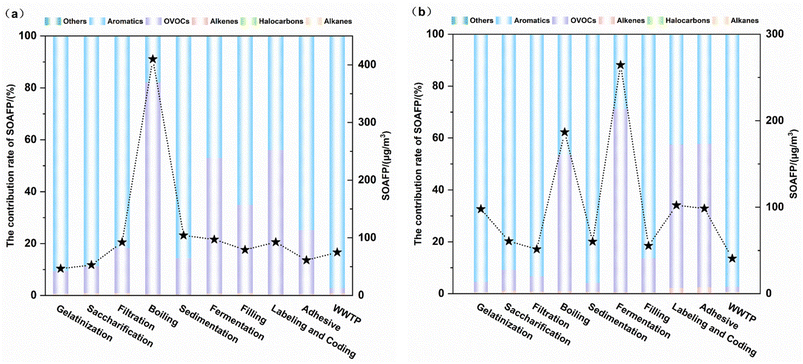
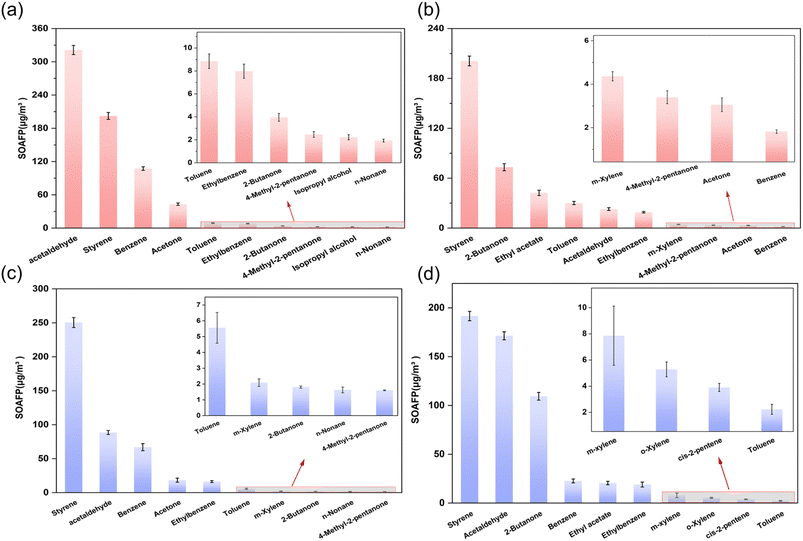
OFP analysis identified distinct key reactive species contributions across emission types. During the organized emission stages in Brewery A, six compounds, including isobutane, ethyl acetate, tetrahydrofuran, acetone, 1,1-dichloroethylene, and 2,3-dimethylbutane, collectively accounted for 89.0% of the total OFP. Fugitive emissions demonstrated higher OFP dominance from ethyl acetate, 2-butanone, tetrahydrofuran, isobutane, methyl methacrylate, and 2-hexanone, contributing 93.6% of the total potential. Brewery B demonstrated different patterns with five major compounds in organized emissions, such as isobutane, ethyl acetate, tetrahydrofuran, acetone, and 1,1-dichloroethylene, driving 68.75% of the OFP. Fugitive phase emissions involved six compounds comprising 2-butanone, ethyl acetate, isobutane, tetrahydrofuran, 1,1-dichloroethylene, and cis-2-pentene, representing 63.7% of the total potential. From Fig. 3, it is evident that fugitive emissions exhibit significantly higher OFP than organized emission sources in Brewery B, which is consistent with the concentration characteristics.
This finding emphasizes the urgency of managing fugitive emission stages of VOCs in traditional semi-automated breweries. Analytical results reveal a notable discrepancy between the ten species exhibiting peak OFP values and those with maximum VOC concentrations identified in this investigation. These results indicate that VOC-induced ozone generation depends on both ambient concentration levels and compound-specific MIR coefficients. These findings highlight that reducing OVOCs from these key processes should be a priority, as they constitute the chemical mixtures with the highest intrinsic OFP per unit volume.
3.3. Exposure, dispersion simulation, and health risk assessment
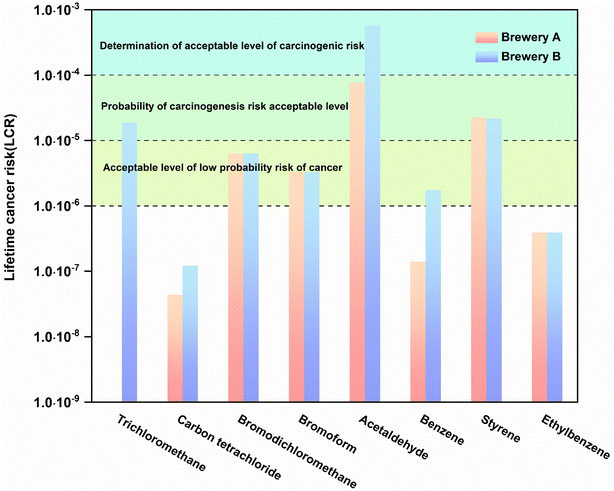
Carcinogenic risk: the Lifetime Cancer Risk (LCR) for all individual species was below 10−6. The HQ and LCR values were used to assess the health risk of fugitive emissions to workers. For individual species, at Brewery A, acrolein and acetaldehyde had HQ values of 33.67 and 3.88, respectively, while the rest of the substances had negligible HQ values of less than 0.1 and an HI of 37.55. At Brewery B, the substances posing a non-carcinogenic risk were 1,2-dichloropropane, acrolein, and acetaldehyde, with HQs of 3.42, 25.54, and 29.06, respectively, and an HI of 58.02. The HI was 58.02. Overall, inhalation of VOCs by workers during working hours posed a non-cancer risk. For the fugitive phases, an inhalation risk assessment model was employed to calculate the risk of VOC exposure during working hours for workers. Data for non-carcinogenic risk-related substances are presented in Table 1. The HI values for both breweries are greater than 1, indicating non-carcinogenic risks. As for cancer risk, carcinogenic risk data for relevant substances are shown in Table S2. From Fig. 7, it can be observed that, except for dichloromethane, carbon tetrachloride, and ethylbenzene, the LCR for other substances is greater than 10−6. In other words, exposure to VOCs in the workplace will pose a clear risk of chronic cancer for workers. Brewery B has a greater cancer risk than Brewery A, and the health risk of the open traditional technique is seriously underestimated.
| Species | RFC(mg m−3) | HQ | |
|---|---|---|---|
| Brewery A | Brewery B | ||
| Chloromethane | 9.00 × 10−2 | 1.45 × 10−3 | 1.54 × 10−3 |
| Dichloromethane | 6.00 × 10−1 | 1.75 × 10−4 | — |
| 1,1-Dichloroethene | 2.00 × 10−1 | 9.52 × 10−3 | 3.78 × 10−2 |
| Carbon tetrachloride | 1.00 × 10−1 | 7.21 × 10−5 | 2.02 × 10−4 |
| 1,2-Dichloropropane | 4.00 × 10−3 | 3.09 × 10−2 | 3.42 × 10 |
| Acrolein | 2.00 × 10−5 | 3.37 × 101 | 2.55 × 101 |
| Acetaldehyde | 9.00 × 10−3 | 3.88 × 10 | 2.91 × 101 |
| 4-Methyl-2-pentanone | 3.00 | 1.73 × 10−3 | 6.83 × 10−4 |
| 2-Hexanone | 3.00 × 10−2 | 6.46 × 10−2 | 6.48 × 10−2 |
| Propionaldehyde | 8.00 × 10−3 | 4.64 × 10−2 | 1.45 × 10−2 |
| Ethyl acetate | 2.00 × 10−1 | 3.99 × 10−3 | 3.08 × 10−3 |
| Methyl tert-butyl ether | 3.00 × 10 | 2.53 × 10−5 | — |
| 2-Butanone | 5.00 × 10 | 2.23 × 10−2 | 3.34 × 10−2 |
| Tetrahydrofuran | 2.00 × 10 | 3.96 × 10−3 | 3.63 × 10−3 |
| 1,4-Dioxane | 3.00 × 10−2 | 1.39 × 10−2 | 1.26 × 10−2 |
| Methyl methacrylate | 7.00 × 10−1 | 6.52 × 10−4 | 6.43 × 10−4 |
| m-Xylene | 1.00 × 10−1 | 4.73 × 10−4 | 8.51 × 10−4 |
| o-Xylene | 1.00 × 10−1 | — | 5.11 × 10−4 |
| Benzene | 3.00 × 10−2 | 5.99 × 10−4 | 7.45 × 10−3 |
| Toluene | 5.00 × 10 | 5.51 × 10−5 | 4.08 × 10−6 |
| Ethylbenzene | 1.00 × 10 | 1.57 × 10−4 | 1.56 × 10−4 |
| Styrene | 1.00 × 10 | 8.67 × 10−4 | 8.26 × 10−4 |
| Carbon disulfide | 7.00 × 10−1 | 3.60 × 10−3 | 2.61 × 10−3 |
| HI | — | 37.75478 | 58.20087 |
3.4. Recommendations and strategies for VOC control
Summarizing the analysis above, this study proposes recommendations and strategies for VOC control in two different models of breweries. For Brewery A, the following integrated strategies are proposed to enhance VOC control. (1) It is recommended to implement process optimization measures, such as introducing low-temperature yeast strains to suppress acetaldehyde formation at the source, as acetaldehyde is the primary contributor to the OFP. This strategy targets a key reactive VOC and is expected to reduce the OFP of the fermentation emissions. This preventive approach aligns with the priority of cleaner production and achieves synergistic carbon reduction by lowering the energy demand for subsequent air pollution control devices. (2) Upgraded abatement technology by adding a palladium-based catalyst bed (Pd/Al2O3) in the rear section of the RTO to enhance the oxidation efficiency of acetaldehyde. (3) Deploy an online monitoring system for VOCs to dynamically track key species and auto-adjust RTO operational parameters. For fugitive emissions, adopt a closed fermentation tank process to reduce the escape of VOCs, which is a foundational measure for advancing green and low-carbon development in industry. The comprehensive implementation of these strategies not only enables breweries to comply with VOC emission regulations but also advances corporate sustainability and social responsibility initiatives, contributing to the national strategic goals of Beautiful China and Carbon Peaking and Carbon Neutrality.The comparative analysis of the two breweries demonstrates that Brewery A requires a primary focus on capacity-linked gross emission control, whereas Brewery B necessitates targeted management of fugitive process sources emitting highly reactive VOCs. This empirical evidence provides a scientific foundation for implementing differentiated regulatory frameworks within China's environmental policy, challenging the conventional one-size-fits-all approach and advocating for technology-informed, precision governance in the food industry.
3.5. Limitations
This study has several limitations that should be considered when interpreting the results. The most significant limitation is the inability to convert the measured VOC concentrations into emission rates due to a lack of air exchange rate or exhaust flow rate data. Therefore, the environmental impact potentials are presented as intrinsic properties of the source gas mixture and not as absolute contributions to the regional atmosphere. Future studies should incorporate direct emission rate measurements to accurately quantify the brewery's contribution to ambient air quality.4. Conclusion
This study presents the first process-based analysis of VOC concentration profiles, environmental impacts and health risks in breweries of contrasting scales and technologies. The two breweries exhibited highly similar VOC species and key emission processes, with boiling and fermentation phases being identified as the dominant sources of high concentration and chemically reactive VOC mixtures. OVOCs such as acetaldehyde, ethyl acetate and ketones were the predominant groups across all stages. The assessment of chemical reactivity revealed that OVOCs possess the highest OFP per unit volume. Although aromatic hydrocarbons account for 0.19–0.70% of the mass concentration of TVOCs, they contribute as much as 44.89–75.31% to the SOAFP. This indicates that reducing OVOCs is the most effective approach to lowering the OFP, while targeting aromatic compounds is key to reducing the SOAFP. The health risk assessment yielded sharply different conclusions for workers versus the general public. AERMOD dispersion simulation results indicated negligible impacts from organized stack emissions on surrounding communities, with both carcinogenic and non-carcinogenic risks falling below thresholds of concern. However, the assessment based on direct in-plant measurements revealed significant health risks. Workers are exposed to fugitive emissions that far exceed acceptable levels for both non-carcinogenic and carcinogenic effects, with acrolein, acetaldehyde, and 1,2-dichloropropane identified as the primary drivers of risk. Consequently, VOC control strategies must be differentiated. For large modern facilities, the primary task is managing the total mass and reactivity control of concentrated organized emissions. For smaller traditional facilities, the immediate priority is controlling fugitive emissions to protect worker health. The study provides a scientific basis for developing VOC-targeted risk management strategies that prioritize the most significant and actionable exposures.Author contributions
Huan Zhao: conceptualization, investigation, writing – original draft. Hongna Ren: investigation, visualization. Hailin Wang: formal analysis, software. Yiwen Wang: investigation. Yiming Lu: data curation. Jie Cheng: methodology, writing – review & editing, acquisition. Guoxia Jiang: writing –review & editing, investigation. Zhengping Hao: supervision.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The datasets generated during this study, including raw GC-MS chromatograms, AERMOD input parameters, and health risk calculations, are available from the corresponding authors upon reasonable request.Supplementary information (SI): geolocation data of breweries due to confidentiality agreements with participating enterprises, process-specific emission factors, SOAP/OFP coefficients, and supplementary tables (S1–S6). See DOI: https://doi.org/10.1039/d5em00567a.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (52370123) and the Fundamental Research Funds for the Central Universities.References
- W. Wu, B. Zhao, S. Wang and J. Hao, Ozone and secondary organic aerosol formation potential from anthropogenic volatile organic compounds emissions in China, J. Environ. Sci., 2017, 53, 224–237 CrossRef CAS.
- E. Nie, G. Zheng, Z. Shao, J. Yang and T. Chen, Emission characteristics and health risk assessment of volatile organic compounds produced during municipal solid waste composting, Waste Manag., 2018, 79, 188–195 CrossRef CAS PubMed.
- Y. S. Huang and C. C. Hsieh, Ambient volatile organic compound presence in the highly urbanized city: source apportionment and emission position, Atmos. Environ., 2019, 206, 45–59 CrossRef CAS.
- X. Liang, X. Chen, J. Zhang, T. Shi, X. Sun and L. Fan, et al., Reactivity-based industrial volatile organic compounds emission inventory and its implications for ozone control strategies in China, Atmos. Environ., 2017, 162, 115–126 CrossRef CAS.
- C. Hsu, H. Chiang, R. Shie, C. Ku, T. Lin and M. Chen, et al., Ambient VOCs in residential areas near a large-scale petrochemical complex: Spatiotemporal variation, source apportionment and health risk, Environ. Pollut., 2018, 240, 95–104 CrossRef CAS.
- H. Xie, W. Gao, W. Zhao, Y. Han, Y. Gao and B. Liu, et al., Source profile study of VOCs unorganized emissions from typical aromatic devices in petrochemical industry, Sci. Total Environ., 2023, 889 Search PubMed.
- Z. Liang, Y. Yu, B. Sun, Q. Yao, X. Lin and Y. Wang, et al., The underappreciated role of fugitive VOCs in ozone formation and health risk assessment emitted from seven typical industries in China, J. Environ. Sci., 2024, 136, 647–657 CrossRef CAS.
- W. Xu, Y. Kuang, Y. Bian, L. Liu, F. Li and Y. Wang, et al., Current Challenges in Visibility Improvement in Southern China, Environ. Sci. Technol. Lett., 2020, 7(6), 395–401 CrossRef CAS.
- M. Wen, W. Deng, J. Huang, S. Zhang, Q. Lin and C. Wang, et al., Atmospheric VOCs in an industrial coking facility and the surrounding area: Characteristics, spatial distribution and source apportionment, J. Environ. Sci., 2024, 138, 660–670 CrossRef CAS.
- Z. Q. Gao, G. J. Hu, H. Wang and B. Q. Zhu, Characterization and assessment of volatile organic compounds (VOCs) emissions from the typical food manufactures in Jiangsu province, China, Atmos. Pollut. Res., 2019, 10(2), 571–579 CrossRef CAS.
- B. Lin and X. Lei, Carbon emissions reduction in China's food industry, Energy Policy, 2015, 86, 483–492 CrossRef CAS.
- L. Wang, Z. Xiang, S. Stevanovic, Z. Ristovski, F. Salimi and J. Gao, et al., Role of Chinese cooking emissions on ambient air quality and human health, Sci. Total Environ., 2017, 589, 173–181 CrossRef CAS PubMed.
- G. Zhang, S. Li, G. Pan, L. Zhu and Y. Sun, Determination and formation mechanism of precedence-controlled VOCs pollutants in chemical plant, China Environ. Sci., 2019, 39(4), 1380–1389 CAS.
- N. R. Passant, S. J. Richardson, R. P. J. Swannell, N. Gibson, M. J. Woodfield and J. P. van der Lugt, et al., Emissions of volatile organic compounds (VOCs) from the food and drink industries of the European community, Atmos. Environ. Part A Gen. Top., 1993, 27(16), 2555–2566 CrossRef.
- A. Mellouki, T. J. Wallington and J. Chen, Atmospheric Chemistry of Oxygenated Volatile Organic Compounds: Impacts on Air Quality and Climate, Chem. Rev., 2015, 115(10), 3984–4014 CrossRef CAS.
- Z. Ni, J. Liu, M. Song, X. Wang, L. Ren and X. Kong, Characterization of odorous charge and photochemical reactivity of VOC emissions from a full-scale food waste treatment plant in China, J. Environ. Sci., 2015, 29, 34–44 CrossRef CAS.
- N. B. Gibson, G. T. Costigan, R. P. J. Swannell and M. J. Woodfield, Volatile organic compound (VOC) emissions during malting and beer manufacture, Atmos. Environ., 1995, 29(19), 2661–2672 CrossRef CAS.
- H. Wang, R. Hao, L. Fang, L. Nie, Z. Zhang and Z. Hao, Study on emissions of volatile organic compounds from a typical coking chemical plant in China, Sci. Total Environ., 2021, 752 Search PubMed.
- R. Wang, X. Wang, S. Cheng, K. Wang, L. Cheng and J. Zhu, et al., Emission characteristics and reactivity of volatile organic compounds from typical high-energy-consuming industries in North China, Sci. Total Environ., 2022, 809, 151134 CrossRef CAS.
- G. You, H. Liu, R. Sun, Z. Jin, J. Li and X. Zhou, et al., Characterizing VOCs emissions of five packaging and printing enterprises in China and the emission reduction potential of this industry, J. Clean. Prod., 2023, 420, 138445 CrossRef CAS.
- C. Wang, W. Wang, W. Deng, S. Zhang, S. Shao and M. Wen, et al., Distribution characteristics, air-water exchange, ozone formation potential and health risk assessments of VOCs emitted from typical coking wastewater treatment process, Sci. Total Environ., 2023, 862, 160845 CrossRef CAS PubMed.
- G. Li, W. Wei, X. Shao, L. Nie, H. Wang and X. Yan, et al., A comprehensive classification method for VOC emission sources to tackle air pollution based on VOC species reactivity and emission amounts, J. Environ. Sci., 2018, 67, 78–88 CrossRef CAS.
- Z. Gao, X. Zhang, W. Zhao, X. Zhang, Z. Xue and J. Du, Characteristic Analysis of VOCs Emitted from a Typical Coking Plant, Res. Environ. Sci., 2019, 32(9), 1540–1545 CAS.
- J. Shi, H. Deng, Z. Bai, S. Kong, X. Wang and J. Hao, et al., Emission and profile characteristic of volatile organic compounds emitted from coke production, iron smelt, heating station and power plant in Liaoning Province, China, Sci. Total Environ., 2015, 515, 101–108 CrossRef PubMed.
- G. Wang, S. Cheng, W. Wei, Y. Zhou, S. Yao and H. Zhang, Characteristics and source apportionment of VOCs in the suburban area of Beijing, China, Atmos. Pollut. Res., 2016, 7(4), 711–724 CrossRef.
- C. M. Da Silva, B. Siciliano, G. Dantas and G. Arbilla, An improvement of method TO-15A, aided by heart-cutting multidimensional gas chromatography, for the analysis of C2-C12 hydrocarbons in atmospheric samples, Microchem. J., 2022, 183, 108008 CrossRef CAS.
- J. Zheng, M. Shao, W. Che, L. Zhang, L. Zhong and Y. Zhang, et al., Speciated VOC Emission Inventory and Spatial Patterns of Ozone Formation Potential in the Pearl River Delta, China, Environ. Sci. Technol., 2009, 43(22), 8580–8586 CrossRef CAS PubMed.
- A. Kumar, D. Singh, K. Kumar, B. B. Singh and V. K. Jain, Distribution of VOCs in urban and rural atmospheres of subtropical India: Temporal variation, source attribution, ratios, OFP and risk assessment, Sci. Total Environ., 2018, 613–614, 492–501 CrossRef CAS.
- W. P. L. Carter, Development of Ozone Reactivity Scales for Volatile Organic Compounds, Air Waste, 1994, 44(7), 881–899 CrossRef CAS.
- W. P. L. Carter, Development of the SAPRC-07 chemical mechanism, Atmos. Environ., 2010, 44(40), 5324–5335 CrossRef CAS.
- R. G. Derwent, M. E. Jenkin, S. R. Utembe, D. E. Shallcross, T. P. Murrells and N. R. Passant, Secondary organic aerosol formation from a large number of reactive man-made organic compounds, Sci. Total Environ., 2010, 408(16), 3374–3381 CrossRef CAS PubMed.
- M. Kanakidou, J. H. Seinfeld, S. N. Pandis, I. Barnes, F. J. Dentener and M. C. Facchini, et al., Organic aerosol and global climate modelling: a review, Atmos. Chem. Phys., 2005, 5(4), 1053–1123 CrossRef CAS.
- D. Johnson, S. R. Utembe and M. E. Jenkin, Simulating the detailed chemical composition of secondary organic aerosol formed on a regional scale during the TORCH 2003 campaign in the southern UK, Atmos. Chem. Phys., 2006, 6(2), 419–431 CrossRef CAS.
- D. GROSJEAN, INSITU ORGANIC AEROSOL FORMATION DURING A SMOG EPISODE - ESTIMATED PRODUCTION AND CHEMICAL FUNCTIONALITY, Atmos. Environ. Part A Gen. Top., 1992, 26(6), 953–963 CrossRef.
- L. Hui, X. Liu, Q. Tan, M. Feng, J. An and Y. Qu, et al., Characteristics, source apportionment and contribution of VOCs to ozone formation in Wuhan, Central China, Atmos. Environ., 2018, 192, 55–71 CrossRef CAS.
- H. Zhang, H. Li, Q. Zhang, Y. Zhang, W. Zhang and X. Wang, et al., Atmospheric Volatile Organic Compounds in a Typical Urban Area of Beijing: Pollution Characterization, Health Risk Assessment and Source Apportionment, Atmosphere, 2017, 8(3), 61 CrossRef.
- H. Zhang, Y. Ji, Z. Wu, L. Peng, J. Bao and Z. Peng, et al., Atmospheric volatile halogenated hydrocarbons in air pollution episodes in an urban area of Beijing: Characterization, health risk assessment and sources apportionment, Sci. Total Environ., 2022, 806, 150283 CrossRef CAS PubMed.
- X. Huang, D. Han, J. Cheng, X. Chen, Y. Zhou and H. Liao, et al., Characteristics and health risk assessment of volatile organic compounds (VOCs) in restaurants in Shanghai, Environ. Sci. Pollut. Res., 2020, 27(1), 490–499 CrossRef CAS.
- L. Li, J. Y. An, Y. Y. Shi, M. Zhou, R. S. Yan and C. Huang, et al., Source apportionment of surface ozone in the Yangtze River Delta, China in the summer of 2013, Atmos. Environ., 2016, 144, 194–207 CrossRef CAS.
- L. Hui, X. Liu, Q. Tan, M. Feng, J. An and Y. Qu, et al., VOC characteristics, chemical reactivity and sources in urban Wuhan, central China, Atmos. Environ., 2020, 224, 117340 CrossRef CAS.
- C. Li, Y. Liu, B. Cheng, Y. Zhang, X. Liu and Y. Qu, et al., A comprehensive investigation on volatile organic compounds (VOCs) in 2018 in Beijing, China: Characteristics, sources and behaviours in response to O3 formation, Sci. Total Environ., 2022, 806, 150247 CrossRef CAS.
- Q. Wang, S. Li, M. Dong, W. Li, X. Gao and R. Ye, et al., VOCs emission characteristics and priority control analysis based on VOCs emission inventories and ozone formation potentials in Zhoushan, Atmos. Environ., 2018, 182, 234–241 CrossRef CAS.
- Q. Li, G. Su, C. Li, M. Wang, L. Tan and L. Gao, et al., Emission profiles, ozone formation potential and health-risk assessment of volatile organic compounds in rubber footwear industries in China, J. Hazard Mater., 2019, 375, 52–60 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |