Regulation of ammonium loss under contrasting upwelling conditions: sensitivity of Feammox to environmental drivers
Guillermo
Samperio-Ramos
 *a,
Oscar
Hernández-Sánchez
a,
Jorge A.
Velásquez-Aristizábal
*a,
Oscar
Hernández-Sánchez
a,
Jorge A.
Velásquez-Aristizábal
 ab,
Víctor F.
Camacho-Ibar
a,
Silvia
Pajares
c,
Aaron
Gutiérrez
a,
Ariadna
Aldrich
a and
Francisco J.
Cervantes
ab,
Víctor F.
Camacho-Ibar
a,
Silvia
Pajares
c,
Aaron
Gutiérrez
a,
Ariadna
Aldrich
a and
Francisco J.
Cervantes
 d
d
aNutrient Cycling in Marine Ecosystems (CiNEMa) Research Group, Instituto de Investigaciones Oceanológicas, Universidad Autónoma de Baja California, 22860 Ensenada, Mexico. E-mail: guillermo.samperio@uabc.edu.mx; oskrgabo@gmail.com; jorge.velasquez36@uabc.edu.mx; velasquez.jorge@uabc.edu.mx; vcamacho@uabc.edu.mx; aaron.gutierrez@uabc.edu.mx; ariadna.aldrich@uabc.edu.mx
bDepartamento de Oceanografía Física, Centro de Investigación Científica y de Educación Superior de Ensenada, 22860 Ensenada, Mexico
cUnidad Académica de Ecología y Biodiversidad Acuática, Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México, 04510 Ciudad de México, Mexico. E-mail: spajares@cmarl.unam.mx
dLaboratory for Research on Advanced Processes for Water Treatment, Engineering Institute, Campus Juriquilla, Universidad Nacional Autónoma de México, 76230 Querétaro, Mexico. E-mail: fcervantesc@iingen.unam.mx
First published on 19th November 2025
Abstract
Understanding the microbial processes involved in ammonium loss in highly productive marine systems is crucial for reconstructing the global nitrogen cycle. This study is the first to examine the anaerobic ammonium oxidation coupled with dissimilatory Fe(III) reduction (Feammox) and the abundance of iron-reducing bacteria (IRB) in a seagrass-dominated coastal lagoon exposed to two contrasting upwelling conditions. Potential Feammox rates varied from 6.0 to 39.2 mg N per m2 per day and were positively correlated with the abundance of IRB (Acidomicrobiaceae A6 spp. and Geobacteraceae spp.), suggesting that IRB mediated the Feammox process. The limited impact of near-mouth productivity conditions on Feammox activity and IRB was largely inherent to sediment type (eelgrass or adjacent bare bottom) and station-specific, depending on the degree of confinement relative to the nearby ocean. The partial least squares structural equation modeling approach revealed that dissimilatory Fe(III) reduction exerted a direct effect on potential Feammox rates, while upwelling conditions indirectly influenced the process through sediment characteristics. The contribution of Feammox to total ammonium loss exceeded 60% and increased with the distance from the mouth of the lagoon. A minimum of 3.7 ± 0.5 mg N per m2 per day was catalyzed by electron acceptors besides Fe(III), highlighting the co-occurrence of alternative chemoautotrophic pathways in ammonium removal. Furthermore, an average loss of 38.4 ± 6.7 t N per year was attributed to the anaerobic ammonium oxidation processes, accounting for 5.1 ± 1.6% of the annual oceanic N transported into the lagoon. These findings extend our current understanding of N and Fe cycles in coastal environments linked to eastern boundary upwelling systems.
Environmental significanceAnaerobic ammonium oxidation coupled to iron(III) reduction (termed Feammox), with dinitrogen as the end-product, is a recently identified microbial nitrogen removal process across the estuarine–ocean continuum. However, little is known about the pathways of N transformation via Feammox in coastal environments influenced by eastern boundary upwelling systems. These upwelling bays demonstrate unique patterns and processes largely driven by their interaction with the adjacent ocean. Our results show that the impact of near-mouth productivity conditions on Feammox activity is inherent mainly to sediment type and station-specific, with the degree of confinement relative to the adjacent ocean being a determining factor. Overall, the present study highlights the distribution patterns of the Feammox process in an upwelling bay, providing valuable insights for nitrogen management in coastal ecosystems within eastern boundary upwelling regions. |
1. Introduction
Seagrass-dominated environments are among the most productive marine ecosystems, contributing 1–2% to global net primary production despite covering less than 0.2% of the ocean.1 In addition, seagrass meadows provide a wide range of ecosystem services, such as protecting shorelines,2 serving as long-term CO2 sinks,3 and acting as nitrogen (N) biofilters.4 The optimal performance and resilience of seagrasses depend partially on their synergy with rhizosphere microbiome, as microbial activity influences nutrient recycling and uptake.5,6 The deamination of sedimentary organic matter releases ammonium (NH4+), which can be incorporated by below-ground seagrass tissues,7 immobilized as microbial biomass8 removed via microbial-mediated processes,9 or accumulated in interstitial water and subsequently released to the overlying water through diffusive fluxes.10 Indeed, the excessive build-up of endogenous NH4+ in sediments has been found to become an internal driver of coastal eutrophication, which is the main underlying factor for seagrass declines worldwide.11,12 Therefore, a better comprehension of the internal mechanisms regulating the NH4+ inventory in seagrass sediments is required to effectively manage these ecosystems.Microbially-mediated processes driving N-loss in sediments are crucial for attenuating NH4+ loading in coastal seagrass-dominated ecosystems. When oxygen is present, bacterial and archaeal nitrifiers oxidize NH4+ to nitrite and nitrate, which can subsequently diffuse into anoxic zones and be anaerobically reduced to nitrous oxide and dinitrogen (N2) through a stepwise denitrification process.13 Like nitrification–denitrification, anaerobic NH4+ oxidation using nitrite as the terminal electron acceptor (Anammox) is recognized as an important contributor to NH4+ removal in seagrass sediments.10,14 Recent studies have provided insights into novel pathways in the biogeochemical N cycle, identifying alternative electron acceptors beyond nitrite that can react with NH4+. For instance, the simultaneous removal of NH4+ and sulfate, accompanied by the concomitant production of nitrite and sulfur species, has commonly been observed in anaerobic fluidized bed reactors used for wastewater treatment15 and confirmed to occur naturally in marine and groundwater environments.16,17 Meanwhile, anaerobic NH4+ oxidation coupled with dissimilatory Fe(III) reduction, producing N2 (referred to as Feammox), nitrite, or nitrate, has also been recognized across the estuarine–wetland–ocean continuum.17–20 Given that the generation of N2 at seawater pH (∼8.00) is thermodynamically more feasible than anoxic nitrification,21 Feammox likely plays a key role in removing NH4+ in seagrass bed sediments.22 However, the extent to which Feammox contributes to microbially induced NH4+ losses within seagrass-dominated ecosystems remains to be elucidated.
Although the functional genes and enzymes responsible for Feammox remain elusive, identifying iron-reducing bacteria (IRB) enhances our understanding of the Feammox process, as IRB have proposed to catalyze Feammox via extracellular electron transfer.23 Huang and Jaffé24 were the first to show that an isolated Acidimicrobiaceae A6 bacterium is capable of oxidizing NH4+ while reducing Fe(III) under strictly anaerobic conditions. Using RNA-based stable isotope probing combined with illumina sequencing, Li et al.25 recently demonstrated that members of Geobacteraceae spp., Pseudomonadaceae spp., and Shewanellaceae spp. also belong to the Feammox bacteria cluster. However, the prevalence of these well-known IRB appears to be localized to specific marine environments. Whereas several lineages of Pseudomonadaceae and Shewanellaceae typically dominate the IRB community in deep-sea ferromanganese deposits, clades affiliated with the Geobacteraceae family are found in higher proportions near the Fe(III)/Fe(II) redox boundary in estuarine and coastal seagrass-colonized sediments.26
Extensive research has focused on understanding microbial community dynamics and their succession patterns in natural settings.8 Changes in the structure and function of IRB communities have often been associated with temperature, hydrological regimen, and vegetation type, as observed in the abundance and activity of Geobacteraceae in riparian sediments along the Taihu watershed, China.27 It is also known that variations of organic loading can impact the distribution of Feammox in wetland sediments,28 since organic carbon can act as a recyclable electron mediator during dissimilatory Fe(III) reduction.23 Specifically, seagrass meadows are potential sources of organic carbon for IRB because seagrasses release high concentrations of labile organic matter into the sediments surrounding their rhizomes and roots.6 Seagrasses can also facilitate the transfer of organic matter into sediments by promoting the sedimentation of autochthonous and allochthonous organic particles,29 which creates favorable conditions for IRB by elevating oxygen consumption.30 Therefore, the occurrence of Feammox within seagrass-dominated ecosystems may result in spatially heterogeneous patterns, with a synoptic distribution of minimum and maximum values presumably related to water column productivity and vegetation cover.
Coastal embayments linked to eastern boundary upwelling systems (i.e., “upwelling bays”31) are highly productive biomes, where organic carbon production, sedimentation, and microbial respiration pathways link benthic and water-column metabolism.32 However, upwelling bays also exhibit distinct seasonal productivity patterns influenced by surrounding oceanic conditions.31 For instance, offshore-derived phytoplankton can constitute the primary exogenous source of fresh organic matter that fuels benthic anaerobic metabolism in shallow upwelling bays.33,34 Additionally, Caffrey et al.35 proposed a short-term enhancement of dissimilatory Fe(III) reduction in response to increased sediment organic loading accompanying an upwelling event and, thus, it is reasonable to hypothesize that favorable upwelling conditions may support Feammox in upwelling bays.
Although Feammox has been recently documented across a mosaic of coastal and estuarine habitats,18 the influence of upwelling conditions on Feammox activity in seagrass-dominated coastal ecosystems remains unknown. Therefore, this study aimed to investigate the variation of the Feammox process and the abundance of IRB in subtidal sediments across a seagrass-dominated coastal lagoon (Bahía de San Quintín, Mexico) that is subjected to contrasting upwelling conditions. The NH4+ loss and IRB abundance were determined by combining 15N-based isotopic tracing and molecular techniques, respectively. The results of this study elucidate how environmental conditions regulate Feammox activity and the relative contribution of Feammox to overall NH4+ loss across seagrass beds and adjacent bare sediments. This study provides novel insights into the microbially mediated N cycle in coastal environments associated with eastern boundary upwelling systems.
2. Results
2.1 Phenology of upwelling in BSQ
The BEUTI values from January 2020 to December 2021 at 31°N are shown in Fig. 1b. The maximum BEUTI peaks (BEUTImax) recorded over the 30 days preceding the fieldwork were 2.3 mmol m−1 s−1 (September–October 2020) and 19.5 mmol m−1 s−1 (May–June 2021). The average BEUTI values were lower in September–October (0.9 ± 0.1 mmol m−1 s−1) than in May–June (3.8 ± 0.7 mmol m−1 s−1). Additionally, the monthly cumulative BEUTI increased by nearly a factor of 5, from 26.7 mmol m−1 s−1 (September–October 2020) to 113.4 mmol m−1 s−1 (May–June 2021). Consequently, the BEUTI metrics indicated two contrasting coastal upwelling scenarios in the shelf areas neighboring BSQ, with a more intense and sustained vertical nitrate flux in spring (high BEUTI; HB, representing favorable upwelling conditions) than in autumn (low BEUTI; LB, representing relaxed upwelling conditions). Under LB conditions, seawater temperature at the experimental stations varied from 19.7 ± 0.4 °C in station 1 to 22.2 ± 0.3 °C in station 3 (Fig. 1c). The HB values coincided with the lowest recorded temperatures (Fig. 1) in station 1 (16.2 ± 0.8 °C), station 2 (18.9 ± 0.4), and station 3 (21.1 0 ± 0.1 °C). Previous observations and models have also indicated distinctive thermal patterns between upwelling conditions.36,41 Our results also showed a pronounced temperature gradient across BSQ under HB conditions, providing additional evidence that the influence of upwelling is prominent in station 1 (bay inlet) and minimal in station 3 (Fig. 1c) due to the elevated grade of confinement and higher residence times of seawater in the inner sector.362.2 Sediment characteristics
Sediment characteristics of respective sites are summarized in Table S2. The PERMANOVA results indicated that the concentration of NH4+ significantly differed (P < 0.001) between upwelling conditions (r2 = 0.16) and bottom types (r2 = 0.47). A significant difference in NOx− levels was observed between upwelling conditions (P < 0.001; r2 = 0.37) and among stations (P < 0.05; r2 = 0.16). Furthermore, a significant interaction of upwelling-conditions × station was identified (P < 0.001; r2 = 0.18), implying that upwelling influenced the NOx− concentration in stations 1 and 3 across both bottom types (Table S2).A marked spatial gradient in urea and SOC was observed over study area, with station as the most important factor (P < 0.001) affecting the distribution of both parameters, accounting for 44% and 58% of the total variation, respectively. The highest mean SOC concentration was recorded in 3S under HB conditions (22.83 ± 1.27 mg C per g), while the lowest values were found in 1B under LB conditions (3.10 ± 0.06 mg C per g). The C![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N ratio was different between upwelling conditions (P < 0.001; r2 = 0.26) and bottom types (P < 0.05; r2 = 0.09), with significant interaction of both factors (P < 0.05; r2 = 0.08). The C
N ratio was different between upwelling conditions (P < 0.001; r2 = 0.26) and bottom types (P < 0.05; r2 = 0.09), with significant interaction of both factors (P < 0.05; r2 = 0.08). The C![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N ratio in bare sediments was lower under HB conditions, except in station 3. The factors that significantly affected the CCHO concentrations were bottom type (P < 0.001; r2 = 0.47) and its interactions with station (P < 0.001) and upwelling-conditions × station (P < 0.001), which collectively explained 40% of the total variation (Table S3). The effect of upwelling conditions (P < 0.05; r2 = 0.16) resulted in relatively high MPB in 1B (309.2 ± 33.6 µg C per g), with the pairwise post-hoc test revealing a statistically significant difference (P < 0.05) compared to the same station under LB conditions (202.2 ± 36.1 µg C per g).
N ratio in bare sediments was lower under HB conditions, except in station 3. The factors that significantly affected the CCHO concentrations were bottom type (P < 0.001; r2 = 0.47) and its interactions with station (P < 0.001) and upwelling-conditions × station (P < 0.001), which collectively explained 40% of the total variation (Table S3). The effect of upwelling conditions (P < 0.05; r2 = 0.16) resulted in relatively high MPB in 1B (309.2 ± 33.6 µg C per g), with the pairwise post-hoc test revealing a statistically significant difference (P < 0.05) compared to the same station under LB conditions (202.2 ± 36.1 µg C per g).
The concentrations of microbiologically reducible Fe(III) and Mn(IV) spatially varied throughout the coastal lagoon (Table S3) but remained relatively constant (P > 0.05) regarding upwelling conditions and bottom types. The highest concentrations were detected in station 2 (4.63 ± 0.59 mg Fe per g and 0.15 ± 0.02 mg Mn per g) and station 3 (4.61 ± 0.72 mg Fe per g and 0.16 ± 0.03 mg Mn per g). Conversely, the lowest concentrations were measured in 1S (1.43 ± 0.76 mg Fe per g and 0.10 ± 0.01 mg Mn per g). A more comprehensive description of the variability in sediment characteristics is provided in the SI.
2.3 NH4+ loss through Feammox
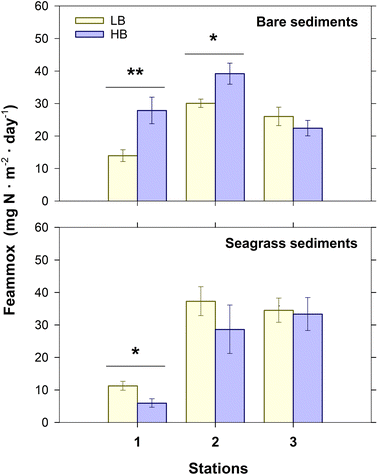
The potential Feammox rates were determined using sediment slurry-based 15N-labeled isotope tracing incubations. The production of 30N2 was detected only in the 15NH4+ treatment, and the addition of 15NH4+ promoted dissimilatory Fe(III) reduction (Fig. S1). The Fe(III) reduction rates in the 15NH4+ treatment ranged from 9.19 to 44.21 g Fe per m2 per day and were significantly higher (t-test; P < 0.05) than those measured in the control (3.94–25.15 g Fe per m2 per day) (Fig. S1). The potential Feammox rates ranged from 5.98 ± 1.30 mg N per m2 per day to 39.19 ± 3.25 mg N per m2 per day, with significant differences among stations (P < 0.001; r2 = 0.42) but not between upwelling conditions or bottom types (P > 0.05). The interactions of upwelling-condition × bottom-type (P < 0.001, r2 = 0.15) and station × bottom-type (P < 0.001, r2 = 0.17) also significantly affected the potential Feammox rates. Similarly, the PERMANOVA (Table S4) revealed that the Fe(III) reduction rates were generally higher (P < 0.001, r2 = 0.34) in stations 2 and 3 than in station 1, although the differences depended upon the bottom type (P < 0.001, r2 = 0.15). The Fe(III) reduction rates were slightly affected by the interactive effect of upwelling-condition × bottom-type (P < 0.05, r2 = 0.05).Specifically, in seagrass sediments, significantly higher Feammox rates (t-test; P < 0.05) were measured in 1S under LB conditions (Table S4) compared to HB conditions. Conversely, Feammox was substantially higher in 1B (Welch F test; P < 0.001) and 2B (Welch F test; P < 0.05) under HB conditions than under LB conditions (Fig. 2). These results suggest that the temporal variability of the Feammox process in BSQ was contingent on the type of bottom and the degree of spatial confinement of sediments relative to the nearby ocean.
Finally, the temporal variability of potential Anammox rates was also quantified in bare and seagrass sediments, in order to assess the relative contributions of Feammox to NH4+ losses across BSQ. As shown in Fig. S4, Anammox ranged from 6.25 ± 2.17 to 11.89 ± 2.85 mg N per m2 per day and from 3.62 ± 0.88 and 7.67 ± 2.85 mg N per m2 per day, in bare and seagrass sediments, respectively (Fig. S2).
2.4 Abundance and distribution of functional IRB genes
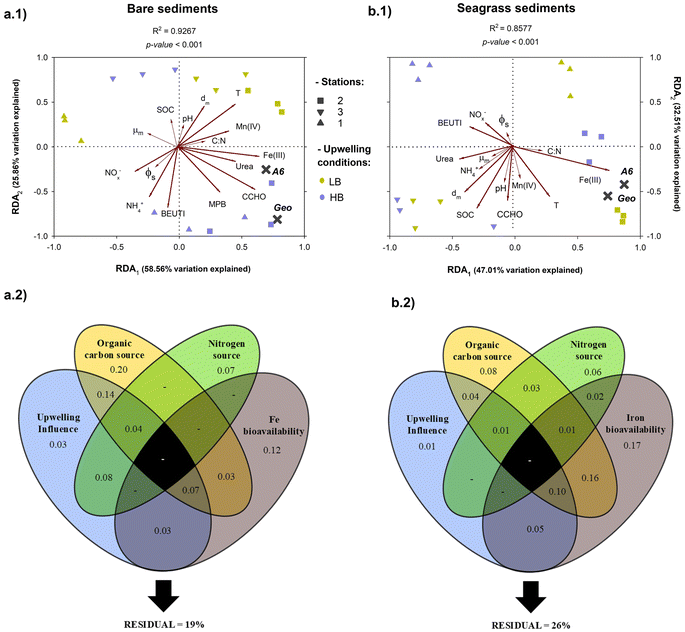
The absolute abundance of the 16S rRNA gene of Geobacteraceae spp. and Acidimicrobiaceae A6 spp. was quantified using qPCR. The average abundance of both IRB ranged from 9.1 ×107 to 6.8 × 108 copies per g for Geobacteraceae and from 5.6 × 105 to 2.8 × 106 copies per g for Acidimicrobiaceae (Fig. S3). The abundance of Geobacteraceae was consistently higher in seagrass sediments than in bare bottoms during both upwelling scenarios. Although the abundance of Geobacteraceae and Acidimicrobiaceae increased in 1B (56% and 23%, respectively) and 2B (20% and 14%) with favorable upwelling conditions, the abundance of both IRB decreased in 3B (29% and 19%) during the transition from LB to HB conditions (Fig. S3). The abundance of both IRB was consistently higher under LB conditions than under HB conditions.In the RDA ordination triplot, which was employed to interpret the influence of environmental characteristics on the abundance distribution of the 16S rRNA gene of IRB in bare (Fig. 3a.1) and seagrass sediments (Fig. 3a.2), axes 1 and 2 accounted for a substantial proportion of the total variance in the data (bare: 84.42%; seagrass sediments: 79.52%). The RDA and corresponding Monte Carlo permutation tests (Table S5) revealed significant correlations (p < 0.05) between IRB distribution and microbiologically reducible Fe(III), urea, CCHO, BEUTI, temperature, station distance from the inlet, and NOx−. Additionally, MPB, NH4+ and microbiologically reducible Mn(IV) were also significant (P < 0.05) environmental factors that partially explained the variance of IRB distribution in bare sediments (Fig. 3a.1). Conversely, the permutation test indicated that SOC significantly affected (p < 0.01) the IRB distribution in seagrass sediments (Table S5).
A VPA, based on the RDA outputs, was employed to elucidate the variation in the abundance distribution of the 16S rRNA gene of IRB independently explained by the combined effects of environmental factors (Fig. 3). These factors were grouped into four categories: upwelling influence (BEUTI, station distance from the inlet, and temperature), organic carbon source (CCHO, SOC, and MPB), N source (NOx−, NH4+, and urea), and Fe bioavailability (microbiologically reducible Fe) (Fig. 3a.2 and b.2). The VPA indicated that the organic carbon source (20%) and the combined effect with the other categories accounted for the greatest significant variation (48%) in the abundance distribution of IRB in bare sediments. Upwelling influence and its combined effects with the other categories were notably higher in bare sediments (39%) than in seagrass sediments (19%). In seagrass sediments, Fe bioavailability, organic carbon source, and the combined effect of both categories contributed 17%, 9%, and 16% of the explained variance, respectively. All environmental factors accounted for less than 1% of 16S rRNA gene structure variation in both bottom types (Fig. 3a.2 and b.2).
2.5 Partial least squares structural equation modeling (PLS-SEM) for Feammox rates: the diagnostic approach
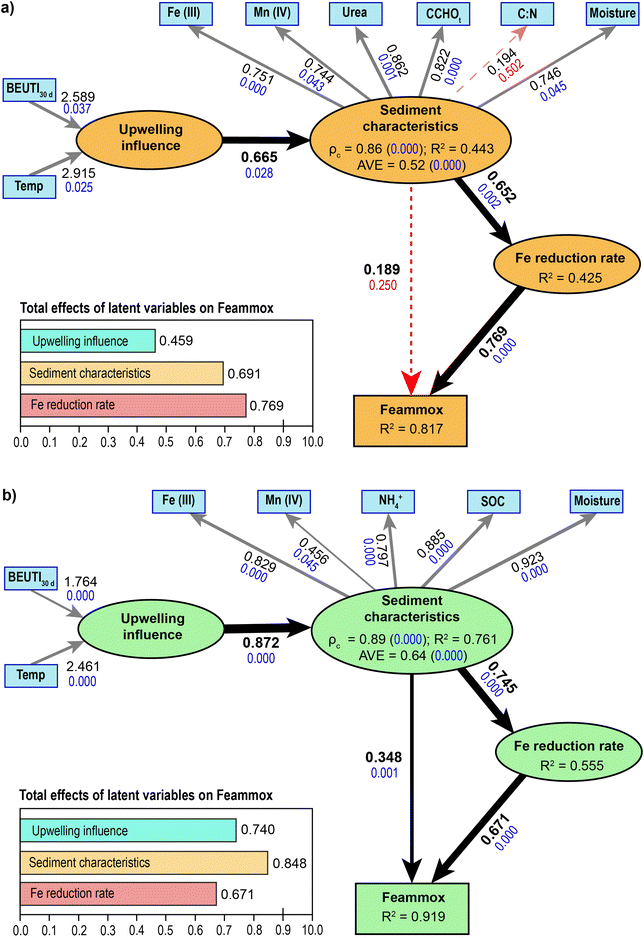
Model diagnostics confirmed the adequacy of both PLS-SEM configurations (Fig. 4). The standardized root mean square residual (SRMR) indicated a good overall fit for the seagrass model (SRMR = 0.08) and a marginally acceptable fit for the bare sediment model (SRMR = 0.11), which is considered reasonable given the inclusion of a formative construct and the predictive orientation of PLS-SEM. For the reflective construct, all indicators exhibited standardized loadings (λ) above 0.746, except for one non-significant indicator (λ = 0.194; p = 0.502) in the bare sediment model, which was retained for theoretical completeness. All other path coefficients were statistically significant (p < 0.05), except for the path from sediment characteristics to Feammox in the bare model, consistent with the results illustrated in Fig. 5. The endogenous constructs showed moderate to high explained variance (R2 ∼ 0.43–0.82 for bare sediments and R2 ∼ 0.56–0.92 for seagrass sediments), indicating robust predictive power. The reliability and convergent validity of the reflective construct (sediment characteristics) demonstrated internal consistency. Composite reliability values of 0.86 for bare and 0.89 for seagrass exceeded the recommended minimum threshold of 0.70. The average variance extracted (AVE) was 0.52 and 0.64 for bare and seagrass models, respectively (both p = 0.000), meeting the criterion of AVE > 0.50 and indicating adequate convergent validity.3. Discussion
3.1 Feammox occurrence in BSQ
Isotopic tracer incubation experiments were conducted to investigate the variation of Feammox activity within a coastal lagoon subjected to contrasting upwelling conditions. Accumulation of 30N2 was exclusively detected in the 15NH4+ treatment, where the addition of 15NH4+ also prompted a significant enhancement in Fe(II) production (Fig. 2). Moreover, a significant positive correlation (P < 0.01) was established between 30N2 production and dissimilatory Fe(III) reduction (Fig. 5). This provides compelling evidence for Fe(III) dependent anaerobic NH4+ oxidation in BSQ.17 During sediment slurry incubations, a limited array of metabolic pathways capable of utilizing NH4+ can be potentially identified. Aerobic nitrification usually emerges as the primary mechanism of NH4+ oxidation in aquatic environments, even under hypoxic conditions.13 However, in this study, slurries were preincubated anaerobically to deplete indigenous oxygen, and strict anaerobic procedures were maintained throughout the 15NH4+ tracing experiments to ensure negligible aerobic nitrification.Under the given conditions, 30N2 accumulation may only be derived directly from Feammox or NOx− produced via anoxic nitrification coupled with Anammox or denitrification.53 The prevailing pH conditions in sediments notably influence the end-product of the coupling of anaerobic NH4+ oxidation with dissimilatory Fe(III) reduction, given that these reactions have been found to consume between 5 protons (Feammox to N2) and 14 protons (anoxic nitrification to generate nitrate) to oxidize 1 mol of NH4+.53 Thus, the substantially lower Gibbs free energy associated with N2 production (−245 kJ mol−1) helps explain the predominance of N2 as the main product observed in numerous studies conducted under neutral or basic conditions.23,28 For instance, Li et al.19 estimated that N2 produced directly from Feammox constituted 79–93% of the total 30N2 accumulated during 15NH4+ treatments with intertidal wetland sediments (pH range: 8.2–8.5). Similarly, Yao et al.54 noted that Feammox directly generated 84–91% of the N2 in eutrophic lake sediments (pH: 7.2–7.6). In our study, sediment pH in BSQ varied between 7.9 and 8.2 (Table S2), and the residual NOx− concentrations measured throughout the slurry incubations remained comparatively low (0.02–0.17 µg N per g) to those in other studies.19,28,54,55 Despite the limited accumulation of NOx− during the isotopic tracing experiments, suggesting that Feammox could be the primary pathway for anaerobic NH4+ oxidation, the possibility of concurrent anoxic nitrification, followed by denitrification and/or conventional Anammox, cannot be completely excluded.53,55 Consequently, further specific studies employing enzymatic inhibitors19,28 are required to elucidate the role of Anammox and denitrification during the anaerobic NH4+ oxidation coupled with Fe(III) reduction in BSQ sediments.
3.2. Links between Fe(III) reduction rates and 30N2 production
The PLS-SEM analysis indicated that dissimilatory Fe(III) reduction sustained the occurrence of Feammox in bare and seagrass sediments (Fig. 4). The slope coefficients relating to the rates of Fe(III) reduction to 30N2 production were not statistically different (P > 0.05) between bare and seagrass sediments, nor between both upwelling scenarios (Fig. 5). Based on the established stoichiometric ratio of 3 mol of Fe(III) reduced for every mol of NH4+ oxidized,52 only a minor proportion of Fe(III) reduction can be attributed to Feammox. Thus, it is reasonable to assume that other electron donors besides NH4+ were available in the examined sediments, implying that surplus Fe(III) reduction is linked to the anaerobic oxidation of other reductants such as organic matter, sulfide, or methane.23 Fe(II) produced from Feammox accounted for 1.8–2.1% of the overall Fe(III) reduction, without differences being detected between bare sediments and seagrass sediments (Fig. 5). These results are comparable to those of Guan et al.28 for mangroves and their adjacent mudflats. We also found that a minimum of 3.7 ± 0.5 mg N per m2 per day (Fig. 5) of anaerobic NH4+ oxidation can be catalyzed by different electron acceptors to Fe(III). Previous tracer experiments with sediments collected in BSQ have provided evidence of Mn(IV) dependent NH4+ oxidation.56 It is also essential to emphasize the potential role of natural organic matter as a sink for NH4+ in BSQ, since it is one of the largest reactive carbon pools in vegetated bottoms.9,57 Similarly, sulfate represents the most prevalent electron acceptor within anoxic marine environments and can also fuel microbial NH4+ oxidation (i.e., Sulfammox reaction) in coastal and estuarine sediments.17 Although Feammox is thermodynamically more favorable than Sulfammox under many geochemical conditions,13,17 the lack of sulfate reduction measurements in this study limits our ability to quantify the trade-offs between both processes. This highlights the need for further research to clarify the feasibility and contributions of potential electron acceptors beyond Fe and nitrite for the anaerobic oxidation of NH4+ in seagrass-dominated coastal ecosystems.3.3 Sensitivity of Feammox and IRB to environmental factors
The paired-samples tests revealed that the potential Feammox rates are influenced by upwelling conditions only at the stations closest to the mouth of BSQ (Fig. 2). Notably, there was an increase of 131% and 24% in Feammox rates at stations 1B and 2B, respectively, when conditions shifted from LB to HB. These findings appear to contrast with the overall results of the PERMANOVA analysis (Table S4), which indicated that upwelling conditions did not significantly affect Feammox rates. However, it is not surprising that an apparent effect of upwelling conditions is observed near the mouth of BSQ, as upwelling bays are transitional waterbodies between terrestrial and marine environments with a gradient of spatial confinement that is influenced by tidal motion and the intrusion of water from the adjacent ocean.31,38 It is evidenced by the residence times of seawater in BSQ, which range from less than two days at station 1, to approximately two weeks at station 3.36 The confinement of station 3, and to a lesser extent of station 2, was also supported by the temperature records (Fig. 1). Consequently, the high variability of Feammox associated with the station factor in the PERMANOVA likely conceals the effect of upwelling-conditions at the inlet sector. In addition, the level of confinement has been shown to shape the ecological niche and functionality of microbial communities inhabiting lagoonal sediments.58 Therefore, the interaction between upwelling intensity (i.e., BEUTI) and confinement (e.g., temperature gradient as proxy) is essential for analyzing how upwelling conditions affected Feammox activity in BSQ (Fig. 4).Our study also found substantial changes in the quantity and quality (C![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N) of sedimentary organic matter in 1B and 2B between both upwelling conditions, indicating that the increased frequency and intensification of upwelling events may influence the sediment characteristics that regulate Feammox activity (Table S2). Northerly wind-driven upwelling conditions (i.e., HB values) enhance the offshore import of labile phytodetritus to coastal systems in eastern boundary upwelling regions.31,59 Stable isotope mixing models have revealed that allochthonous phytoplankton is the primary source of organic matter in the bare flats of the inlet sector of BSQ.33 This influx of organic material may exacerbate oxygen consumption in sediments, creating a favorable anoxic environment for Feammox. Additional support for this claim was provided by Ibarra-Obando et al.34 who found that anaerobic metabolism in unvegetated sediments of bay mouth was partially supported by organic matter of planktonic origin. The VPA results of the present study (Fig. 3a.2) also indicated that up to 15% of the observed variation in the IRB community of bare BSQ sediments was related to the influence of combined effect of upwelling conditions and organic matter availability. In line with our findings, Yao et al.30 provided experimental evidence that phytoplanktonic detritus plays a key role in regulating the abundance and composition of the IRB community, dissimilatory Fe(III) reduction, and Feammox rates. Therefore, as an important biogeochemical factor closely linked to upwelling conditions, the transfer of bioavailable organic matter from the water column to sediments is likely to influence the functional characteristics of the IRB community in bare sediments at the BSQ mouth.
N) of sedimentary organic matter in 1B and 2B between both upwelling conditions, indicating that the increased frequency and intensification of upwelling events may influence the sediment characteristics that regulate Feammox activity (Table S2). Northerly wind-driven upwelling conditions (i.e., HB values) enhance the offshore import of labile phytodetritus to coastal systems in eastern boundary upwelling regions.31,59 Stable isotope mixing models have revealed that allochthonous phytoplankton is the primary source of organic matter in the bare flats of the inlet sector of BSQ.33 This influx of organic material may exacerbate oxygen consumption in sediments, creating a favorable anoxic environment for Feammox. Additional support for this claim was provided by Ibarra-Obando et al.34 who found that anaerobic metabolism in unvegetated sediments of bay mouth was partially supported by organic matter of planktonic origin. The VPA results of the present study (Fig. 3a.2) also indicated that up to 15% of the observed variation in the IRB community of bare BSQ sediments was related to the influence of combined effect of upwelling conditions and organic matter availability. In line with our findings, Yao et al.30 provided experimental evidence that phytoplanktonic detritus plays a key role in regulating the abundance and composition of the IRB community, dissimilatory Fe(III) reduction, and Feammox rates. Therefore, as an important biogeochemical factor closely linked to upwelling conditions, the transfer of bioavailable organic matter from the water column to sediments is likely to influence the functional characteristics of the IRB community in bare sediments at the BSQ mouth.
A significant positive correlation was found between Feammox rates and CCHO content in the bare sediments (r = 0.861, P < 0.001), but not with SOC (r = 0.188, P > 0.05) indicating that the bioavailability of organic matter is a crucial factor influencing Feammox in these sediments (Fig. S4). Furthermore, as shown by the RDA, MPB and CCHO displayed strong relationships with the abundance of Geobacteraceae spp. in 1B and 2B under favorable upwelling conditions (Fig. 3a.1). Notably, MPB have been recognized as a potential source of CCHO in mudflats, as they secrete CCHO-rich polymeric substances that help maintain structural stability of the biofilm.60 This implies that the abundance of Geobacteraceae in the bare sediments near the mouth may be associated with the productivity of MPB, which, in turn, relies on the nutrients supplied by upwelling events. In the present study, Feammox rates in bare sediments were also positively correlated (Fig. S4) with the abundance of IRB (P < 0.001). Although functional Feammox microbes are thought to be chemoautotrophs (e.g., Acidimicrobiaceae) that derive energy from ferric NH4+ oxidation to fix inorganic carbon for growth and biosynthesis, the exopolysaccharides released by MPB could act as an energy source for Geobacteraceae, which coexists and likely cooperates with Acidimicrobiaceae in BSQ.22 Supporting this view, Le et al. and Nguyen et al.61,62 recently demonstrated that Feammox can be associated with heterotrophy in anaerobic digesters, wherein γ-proteobacteria (Pseudomonadaceae) and δ-proteobacteria (Geobacteraceae) were the predominant IRB. These findings also align with the PLS-SEM analysis in the bare sediments of the present study (Fig. 4a), as urea was likely the main N source that indirectly influenced Feammox activity in the bare BSQ sediments, suggesting that NH4+ and organic carbon can be co-removed under Feammox heterotrophic conditions.25,63 Meanwhile, exopolysaccharides have been noted to encourage the extensive aggregation of IRB on Fe-mineral surfaces and to aid in the translocation of the outer membrane porin-cytochrome complexes involved in the Feammox redox network.64,65 Additionally, microalgal exopolymers can store electrochemically active phenolic metabolites, such as flavonoid quinones,66 which can serve as electron shuttles during interfacial electron transfer in Feammox.9 As such, the results of the present study suggest a specific linkage between MPB exopolymers and the Feammox process.
The spatial distribution and temporal variation of Feammox rates in seagrass and bare sediments were significantly correlated (P < 0.05) with changes in the abundance of Acidimicrobiaceae and Geobacteraceae (Fig. S4). These findings suggest IRB influenced Feammox functionality in BSQ.25,62 However, to establish a robust link between specific microbial taxa and Feammox activity, it would be necessary to conduct metatranscriptomic-based analyses and/or investigate functional gene expression of Feammox.
According to the RDA results, microbiologically reducible Fe(III) emerged as the most important environmental factor that influenced the distribution of the microbes involved in Feammox (Fig. 3b.1). This suggests that macrophyte-dominated coastal environments with high levels of bioavailable Fe(III) favor Feammox.18,19 Previous studies have shown that Fe(III) controls the diversity and activity of Feammox bacteria, including Geobacteraceae spp. and Acidimicrobiaceae spp.27,67 The RDA ordination results (Table S5) also indicated that NH4+, temperature, and moisture exerted a highly significant influence (p < 0.001) on the distribution of Feammox bacteria in seagrass sediments. An increase in moisture has been shown to enhance the reactivity of Fe(III) oxides and, by extension, positively affect IRB growth.18 Meanwhile, the influx of upwelled water into BSQ led to lower temperatures than those present under LB conditions, particularly at the stations closer to the mouth of the bay (Fig. 1), as the innermost edges of BSQ are less sensitive to upwelling due to delays in tide propagation and the confinement of this area.36 A decrease in temperature can alter the specific affinity of the membrane transport proteins of IRB and partially inhibit gene replication and transcription.23 The latter is consistent with the results of the VPA, given that up to 8% of the total variation in IRB abundance within seagrass sediments was explained by the interaction between the Fe-bioavailability and the variables belonging to the upwelling-influence category (Fig. 3b.2). The PLS-SEM analysis also identified that changes in seawater temperature associated with upwelling conditions notably affected Feammox activity through the characteristics of seagrass sediments. This can be partially attributed to temperature-dependent abiotic Fe(II) oxidation, which regulates the transient accretion of Fe(III)-(hydr)oxides within the rhizosphere boundary.68,69
As in the vegetated bottoms of mangroves, riparian zones, and eutrophic lakes,27,28,30 the Feammox rates in seagrass sediments were positively related to SOC content (r = 0.624; P < 0.05) (Fig. S4). Eelgrass beds are highly productive, with estimates of net primary production of 2–9 Mg C per ha per year, of which 5–20% is transferred to sediments as an array of organic materials, including plant fragments and root exudates that can be microbiologically processed.70 Rhizodeposits can induce the release of structurally stable Fe from silt minerals into amorphous Fe(III) oxides,5 which more readily interact with the outer membrane protein of IRB to promote electron transfer during the Feammox reaction.23 This mechanism could be particularly relevant in the seagrass sediments from the inner sector of the BSQ (S3), where the contribution of silt-sized particles exceeded 70% (Table S2). Conversely, She et al.55 reported an inhibitory effect of organic matter on Feammox in the rhizosphere of riparian soils from the Taihu watershed based on the idea that labile organic carbon is thermodynamically favored over NH4+ as an electron donor for dissimilatory Fe(III) reduction. Although it can be assumed that most dissimilatory Fe(III) reduction involves the oxidation of SOC, the inhibitory effect of organic matter on Feammox may not fully explain the results of our study. This is because the proportion of Fe(III) reduction related to Feammox did not differ significantly (P > 0.05) between bare sediments and those with seagrass (Fig. 5). In addition, it is known that seagrasses can also mobilize substantial amounts of Fe(III) within their rhizosphere via root-induced local acidification, leading to the protolithic dissolution of Fe(III) minerals.5 Consequently, the apparent efficiency of Feammox observed in the seagrass sediments of BSQ can be ascribed to the sufficient bioavailability of Fe(III) demanded for Fe(III)-catalyzed organic matter respiration, as well as the likely prevalence of alternative metabolic pathways for anaerobic SOC oxidation.
Of note, sulfate-reducing bacteria are abundant in eelgrass sediments, where dissimilatory sulfate reduction is notably stimulated by root exudates.71 Although measurements of sulfate reduction in BSQ are lacking, the spatial pattern of δ34S signatures suggest that this process is a dominant pathway for anaerobic SOC oxidation in the inner sector of BSQ.72 It is also noteworthy that the PLS-SEM analysis of seagrass sediments disclosed that microbiologically reducible Mn(IV) operates as an environmental driver of sediment characteristics, indirectly influencing Feammox rates through Fe(III) reduction (Fig. 4b). Given that amorphous Mn(IV)-bearing minerals can act as preferred electron acceptors during anaerobic SOC degradation due to their high redox potential,73 the bioavailability of Fe(III) for Feammox is likely maximized where labile phase ferromanganese minerals are naturally enriched, namely in the transition and inner sectors of BSQ (Table S2). On the other hand, Feammox exhibited a decline of 47% in 1S between LB and HB conditions (Fig. 2). This was probably due to the stimulation of Fe(III)-catalyzed organic matter respiration by root exudation, as seagrasses release a larger portion of the total amount of fixed carbon to cope with the overflow in their photosynthetic machinery during periods of high productivity.6 This was evidenced by the higher SOC and the lower microbiologically reducible Fe(III) found at S1 under HB conditions as compared to LB conditions (Tables S2 and S3). Therefore, spatial patterns in sediment characteristics, influenced by the degree of confinement, and varying primary productivity, related to upwelling conditions, likely affected the efficiency of Feammox in the seagrass-colonized sediments of BSQ.
3.4 Environmental implications and study limitations
Microbial N cycling processes are essential for understanding the N budget and managing N pollution in estuarine and coastal ecosystems. Anammox and Feammox are recognized as the primary pathways for the anaerobic oxidation of NH4+ to N2 in coastal sediments.10,18,19 Even though Anammox activity was relatively higher under HB conditions (except in 2S and 3S), its contribution to anaerobic NH4+ losses was still ≤40% (Fig. 6). The Feammox contribution increased with the distance from the mouth of the bay. This can be attributed to the constrained activity of Anammox in the inner sectors of BSQ, resulting from constrained N loading from the ocean.38,41 Additionally, Feammox contributed slightly more to NH4+ loss under LB conditions (77–94%) than under HB conditions (61–93%), likely due to the relatively high Anammox rates under upwelling favorable conditions (Fig. 2). The mean contributions of Feammox to NH4+ loss (Fig. 6) in the seagrass sediments (84%) and adjacent bare sediments (88%) of BSQ were comparable to the estimates reported for estuarine wetlands,19 and slightly higher than those observed in riparian soils55 and mangroves.28 | ||
| Fig. 6 Contribution of Anammox and Feammox to NH4+ loss in bare and seagrass sediments from Bahía de San Quintín under high BEUTI (HB) and low BEUTI (LB) conditions. | ||
By extrapolating the potential rates of Feammox and Anammox to the eelgrass coverage at BSQ—obtained from satellite imagery,39—we estimated that removal of NH4+ ranged from 92.6 to 115.2 kg day−1 under LB conditions and from 100.2 to 137.8 kg day−1 under HB conditions. Notably, the gross oceanic imports of reactive N into BSQ can vary between 1.4 and 3.2 t day−1, according to a 3-D coupled physical–biogeochemical model38 that incorporates two contrasting upwelling scenarios and the primary reactive N sources (i.e., NOx−, NH4+, dissolved organic nitrogen, phytoplankton, and zooplankton). These fluxes are analogous to the oceanic inputs into other coastal systems influenced by upwelling along the California Current System74 and suggest that the N loads into BSQ during the upwelling events could exceed those of some land-based inputs in coastal lagoons that experience moderate levels of anthropogenic eutrophication.75 Additionally, the phenology of BEUTI in the southern domain of the California Current System indicates that HB conditions typically encompass from March to July, lasting for 148 ± 26 days.43 According to these approaches, we also estimated an annual loss of NH4+ for BSQ of 38.4 ± 6.7 t, which would be equivalent to 5.1 ± 1.6% of the total external N transported into the system. Consequently, anaerobic NH4+ oxidation processes could play a masked role in the current estimates of total N loss in coastal ecosystems from eastern boundary upwelling regions. However, extrapolating potential rates from two isolated experimental surveys into annual flows introduces uncertainty due to the omission of the intraseasonal variability in upwelling phenology.43 Therefore, further research efforts are required to elucidate how short-term fluctuations in the biogeochemical properties of upwelled waters impact microbial community structure and Feammox activity.67
Whilst potential rates provide relevant insights into environmental drivers controlling individual N cycling processes, their ecological significance must be carefully addressed, as slurry preparation disrupts geochemical zonation in sediments and alters the in situ concentration of electron acceptors present in the interstitial water.76 Therefore, isotope pairing procedures for intact sediments must be developed to accurately determine Feammox rates in seagrass-dominated ecosystems without introducing experimental artifacts.
4. Materials and methods
4.1 Study area and field sampling
Bahía de San Quintín (BSQ) is a well-mixed, shallow lagoon (average depth ∼ 2.5 m) that covers 42 km2 (Fig. 1a) located on the north-western coast of the Baja California peninsula (30.3°N, 116.0°W). Bahía de San Quintín features an inverse salinity gradient, extending from the ocean inlet to the inner bay due to intense evaporation, low precipitation levels, and a lack of runoff. The ocean adjacent to BSQ is strongly influenced by the southern limit of the California Current System. Circulation and water exchange with the California Current System is driven by semi-diurnal tides (amplitude ∼ 1.6 m) and constrained by the morphology of the basin.36 Coastal upwelling in the California Current System is forced primarily by a combination of wind-driven Ekman transport and cross-shore geostrophic flow associated with the pressure gradient around the North Pacific High.37 Upwelling events are considered the primary source of nutrients and phytoplankton for BSQ and tend to be most intense in spring and early summer when north-westerly winds are strongest and most persistent.38Approximately 40% of BSQ is covered by eelgrass (Zostera marina), which forms monospecific submerged and intertidal meadows.39 Eelgrass exhibits a seasonal growth cycle, with maximum biomass in September and October.7 The physiography of the lagoon is characterized by the cineritic volcanic cones surrounding the basin. The sediment composition of BSQ results from alluvial deposits, beach sands, and pyroclastic materials, which result in a notable abundance of Fe-rich oxide minerals.40
Sediment samples were collected in October 2021 and June 2022. Three sampling stations (1, 2, and 3) were established along the subtidal mudflats (Fig. 1a). Stations 1, 2, and 3 were selected based on previous observations of temperature, salinity, and granulometric gradients,36,41 as well as the presence of vegetation cover, to generate a comprehensive understanding of N cycling in the lagoon. Sampling was conducted in the seagrass sediments (1S, 2S, and 3S) and adjacent bare bottoms (1B, 2B, and 3B) of each station, ∼25 m from the edges of the eelgrass beds. At each station, six independent surface sediment samples (0–5 cm) were collected using plexiglass corers and promptly stored in sterile plastic bags in a refrigerated cooler for transport to the laboratory within 4 h. Sediment samples from each site were divided into two subsets. The first subset was preserved at 4 °C for the 15N tracing experiments and to evaluate sediment characteristics. The second subset was preserved at −20 °C for molecular analysis.
4.2 Upwelling phenological parameters
The Bakun Index is typically employed to estimate the cross-shore component of Ekman transport as a proxy for upwelling intensity. However, in the California Current System, upwelled nitrate and Ekman transport may be decoupled due to the subsurface nitrate concentration, which exhibits considerable spatiotemporal variation.42 The Biologically Effective Upwelling Transport Index (BEUTI) is a metric used to estimate the total amount of nitrate transported vertically into (or out of) the surface layer during upwelling (or downwelling) events. This index is based on an observationally derived temperature–latitude–nitrate relationship along the California Current System.43 Consequently, BEUTI metrics were proposed to characterize the seasonal phenology of upwelling in the nearshore waters off BSQ and effectively capture the linkage between bottom-up forcing related to upwelling conditions and biological responses.The BEUTI data (daily, 31°N) were acquired from 2021 to 2022 (https://mjacox.com/upwelling-indices/). Following the methodology of previously derived phenlological indices,42 the cumulative BEUTI was calculated as the sum of daily values beginning 30 days before the field surveys. In addition, submersible HOBO data loggers (Onset Computer Corporation, Bourne, USA) were anchored 10 cm above the sediment surface layer to record seawater temperature hourly for two weeks prior to fieldwork.
4.3 Analysis of sediment characteristics
Sediment moisture was obtained by measuring the weight loss of the sediment before and after drying. The granulometric composition of each sample was determined using a Horiba LA-910 particle analyzer. Sediment bulk density and moisture were determined gravimetrically by weighing 25 cm3 fresh sediments after drying. Sediment pH was measured with a pH meter (OHAUS Starter 2100) in a slurry of sediment and CO2-free artificial seawater (S = 35.5)44 at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (w/v) ratio.18 After extracting the sediments with a 2 M KCl solution, the filtrate was employed to determine NH4+ and nitrate + nitrite (NOx−). The concentration of NH4+ was measured using the optimized indophenol method,45 whereas the analysis of NOx− was performed with an AA3-HR segmented flow autoanalyzer (SEAL Analytical Ltd).46 Chlorophyll in the bare sediments was extracted from approximately 0.5 g of sediment suspended in 10 mL of 90% acetone for 12 h in the dark at 4 °C and quantified spectrophotometrically. Chlorophyll content was converted to carbon equivalents using a conversion factor of 40 µg C per µg Chl and expressed as microphytobenthos biomass (MPB).47 Total sediment organic carbon (SOC) was quantified using an elemental analyzer (Vario Isotope Select). Soluble combined carbohydrates (CCHO) in the sediments were extracted with K2SO4 (0.5 M) and subsequently measured through oxidation with ferricyanide following hydrolysis, which was completed with 1 M HCl at 100 °C for 24 h.48 The mobilizable fractions of Fe(II), Mn(II), and total Fe and Mn were extracted in 10 mL mixtures of 0.5 M HCl and 0.25 M hydroxylamine–HCl, respectively. While Fe(II) was measured following the ferrozine-based colorimetric method with slight modifications,49 Mn(II) was detected as described by Chin et al.50 using a FLAME-S UV-Vis spectrophotometer (Ocean Insight). The quantity of microbiologically reducible Fe(III) and Mn(IV) was determined as the difference between total extractable Fe and Mn and their respective Fe(II) and Mn(II).
3 (w/v) ratio.18 After extracting the sediments with a 2 M KCl solution, the filtrate was employed to determine NH4+ and nitrate + nitrite (NOx−). The concentration of NH4+ was measured using the optimized indophenol method,45 whereas the analysis of NOx− was performed with an AA3-HR segmented flow autoanalyzer (SEAL Analytical Ltd).46 Chlorophyll in the bare sediments was extracted from approximately 0.5 g of sediment suspended in 10 mL of 90% acetone for 12 h in the dark at 4 °C and quantified spectrophotometrically. Chlorophyll content was converted to carbon equivalents using a conversion factor of 40 µg C per µg Chl and expressed as microphytobenthos biomass (MPB).47 Total sediment organic carbon (SOC) was quantified using an elemental analyzer (Vario Isotope Select). Soluble combined carbohydrates (CCHO) in the sediments were extracted with K2SO4 (0.5 M) and subsequently measured through oxidation with ferricyanide following hydrolysis, which was completed with 1 M HCl at 100 °C for 24 h.48 The mobilizable fractions of Fe(II), Mn(II), and total Fe and Mn were extracted in 10 mL mixtures of 0.5 M HCl and 0.25 M hydroxylamine–HCl, respectively. While Fe(II) was measured following the ferrozine-based colorimetric method with slight modifications,49 Mn(II) was detected as described by Chin et al.50 using a FLAME-S UV-Vis spectrophotometer (Ocean Insight). The quantity of microbiologically reducible Fe(III) and Mn(IV) was determined as the difference between total extractable Fe and Mn and their respective Fe(II) and Mn(II).
4.4 Isotopic tracer incubations
The 15N isotopic tracing incubations were performed in a workstation brimmed with high-purity Ar (99.999%) to ensure anaerobic conditions. The experimental procedure was adapted from that of Chen et al.,18 with minor modifications. Briefly, slurries were prepared by adding sterile, sulfate-free artificial seawater at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (w
3 (w![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) and were thoroughly homogenized using a magnetic stirrer. The use of sulfate-free artificial seawater to prepare slurries ensured that the redox properties of the aqueous matrix remain consistent during storage until all experiments are completed, due to the absence of electron acceptors. Each mixture (∼6 g) was transferred into 12 mL vials (Exetainer, Labco Limited, Lampter, UK) and preincubated in the dark for 72 h to deplete background NOx− and oxygen contents. Afterwards, the vials were purged with high-purity Ar for 20 min and separated into two distinct treatments (three replicates each) as follows: (1) control (sterile anoxic artificial seawater) and (2) 15NH4Cl treatment (15N at% >99; Sigma-Aldrich, St. Louis, USA). Each vial contained a final concentration of ∼100 15N µM. All vials were shaken vigorously every 8 h to ensure the treatment solutions were well mixed. The incubations were conducted at temperatures close to in situ levels, and microbial activity was disrupted after 96 h by inoculating with 200 µL of saturated ZnCl2 solution. The accumulation of 30N2 in the headspace was detected using a GasBench + Precon gas concentration system interfaced with a Delta V Plus Isotope-Ratio Mass Spectrometer (IRMS; Thermo-Fisher Scientific, Waltham, USA) in the Stable Isotope Facilities of UC Davis. The potential Feammox rates were conservatively estimated by comparing the accumulation of 30N2 between the 15NH4Cl treatment and the control.27 Rates of dissimilatory Fe(III) reduction were determined by measuring the difference in Fe(II) concentrations throughout the incubation.19 The experimental procedures for measuring the potential Anammox rates are detailed in the SI. Microbial activities were expressed as areal rates of N removal (mg N per m2 per day) using the sediment bulk densities from the study site (1.3–1.8 g cm−3) and depth (5 cm).
v) and were thoroughly homogenized using a magnetic stirrer. The use of sulfate-free artificial seawater to prepare slurries ensured that the redox properties of the aqueous matrix remain consistent during storage until all experiments are completed, due to the absence of electron acceptors. Each mixture (∼6 g) was transferred into 12 mL vials (Exetainer, Labco Limited, Lampter, UK) and preincubated in the dark for 72 h to deplete background NOx− and oxygen contents. Afterwards, the vials were purged with high-purity Ar for 20 min and separated into two distinct treatments (three replicates each) as follows: (1) control (sterile anoxic artificial seawater) and (2) 15NH4Cl treatment (15N at% >99; Sigma-Aldrich, St. Louis, USA). Each vial contained a final concentration of ∼100 15N µM. All vials were shaken vigorously every 8 h to ensure the treatment solutions were well mixed. The incubations were conducted at temperatures close to in situ levels, and microbial activity was disrupted after 96 h by inoculating with 200 µL of saturated ZnCl2 solution. The accumulation of 30N2 in the headspace was detected using a GasBench + Precon gas concentration system interfaced with a Delta V Plus Isotope-Ratio Mass Spectrometer (IRMS; Thermo-Fisher Scientific, Waltham, USA) in the Stable Isotope Facilities of UC Davis. The potential Feammox rates were conservatively estimated by comparing the accumulation of 30N2 between the 15NH4Cl treatment and the control.27 Rates of dissimilatory Fe(III) reduction were determined by measuring the difference in Fe(II) concentrations throughout the incubation.19 The experimental procedures for measuring the potential Anammox rates are detailed in the SI. Microbial activities were expressed as areal rates of N removal (mg N per m2 per day) using the sediment bulk densities from the study site (1.3–1.8 g cm−3) and depth (5 cm).
4.5 Quantitative PCR assays
Genomic DNA was extracted from the sediment samples (0.3 g) using the DNeasy PowerSoil Kit (Qiagen, Hilden, Germany), quantified on a Qubit 4.0 Fluorometer (Invitrogen, Waltham, USA), and diluted to 5 ng µL−1. The abundance of IRB was assessed by measuring the gene copy number of 16S rRNA Geobacteraceae and 16S rRNA Acidomicrobiaceae A6 via qPCR with a StepOnePlus real-time PCR system (Applied Biosystems, Foster City, USA). Reactions were carried out in triplicate in a 20 µL reaction mixture containing 1× SYBR Green PCR Master Mix (Applied Biosystems), primers, 0.2 µg µL−1 of BSA (Thermo Fisher Scientific), and 0.3 ng µL−1 of DNA. Primers and thermal conditions are shown in Table S1. The standard curves consisted of serial 10-fold dilutions of linearized plasmids containing known fragments of the targeted genes. The specificity of the amplified products was confirmed through melt-curve examination and agarose gel electrophoresis. The results with efficiency and correlation coefficients exceeding 90% and 0.99, respectively, were utilized in this study (Table S1).4.6 Statistical analysis
A three-way univariate permutational analysis of variance (PERMANOVA) was employed to evaluate differences in sediment characteristics and Feammox activity across the factors of upwelling conditions, bottom-type, and station. The PERMANOVA was implemented on the ranks of a sample dissimilarity matrix derived from Euclidean distances, utilizing 9999 random permutations to compute the p-values and coefficients of determination. When significant effects were detected in the main test, a pairwise test with Bonferroni correction was carried out. We applied the Student's t-test or, alternatively, the Welch F-test for unequal variances, in order to compare dissimilatory Fe(III) reduction rates between the control and 15NH4+ treatments and to examine the specific effects of contrasting upwelling scenarios on Feammox rates. The assumptions of normality were confirmed using the Shapiro–Wilk test, while the homoscedasticity was tested using Levene's test.Spearman correlation analysis was employed to assess the relationship between Feammox rates and the sediment characteristics, including physicochemical properties and granulometry, as well as the abundance of IRB. A Deming regression analysis51 was applied to examine the relationship between Feammox and Fe(III) reduction rates, as both variables are subject to random measurement and estimation errors. For this analysis, the λ parameter was estimated as the ratio of the sample variances of the two variables plus 10% of that value. Although sample variances are unbiased estimators, this conservative adjustment was introduced to reflect the uncertainty in error variance estimation better when exact values are unavailable. To evaluate the stability and significance of the fitted slope, we generated 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 bootstrap resamples and applied a Deming regression in each iteration. Bias-corrected and accelerated intervals were computed from the bootstrap distribution to account for skewness and bias, yielding robust 95% confidence limits. Slopes whose intervals included zero were deemed statistically indistinguishable from zero. Slope homogeneity was assessed through an F-test.
000 bootstrap resamples and applied a Deming regression in each iteration. Bias-corrected and accelerated intervals were computed from the bootstrap distribution to account for skewness and bias, yielding robust 95% confidence limits. Slopes whose intervals included zero were deemed statistically indistinguishable from zero. Slope homogeneity was assessed through an F-test.
A redundancy analysis (RDA) was conducted to elucidate the association between the abundance distribution of the 16S rRNA gene of IRB and environmental variables. The Monte Carlo permutation method using 999 permutations was used to assess the statistical significance of each variable on the constrained axes. Alongside the RDA, a variation partitioning analysis (VPA) was performed to distinguish the combined effect of environmental factors on the IRB community structure across four explanatory data sets: upwelling influence (BEUTI, station distance from the inlet, and temperature), organic carbon source (CCHO, SOC, and MPB), N source (NOx−, NH4+, and urea), and Fe bioavailability (microbiologically reducible Fe).
Finally, partial least squares structural equation modeling (PLS-SEM) was applied to test the impacts of hydrographic conditions and sediment characteristics on Feammox activity. The selection of PLS-SEM was based on its capacity to model latent constructs with both formative (UI) and reflective (SC) measurement models within a unified structural framework, as well as to estimate complex relationships among observed and latent variables in a prediction-oriented context. The model incorporated one formative construct (upwelling influence), one reflective construct (sediment characteristics), and two single-indicator variables (Feammox activity and iron reduction rates; FeRR). Standard procedures were followed to assess the quality and reliability of the model. The standardized root mean square residual (SRMR) was used as a global goodness-of-fit indicator. Composite reliability and the average variance extracted (AVE) were computed for the reflective construct to evaluate internal consistency and convergent validity, respectively. Bootstrapping with 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 subsamples was utilized to evaluate the statistical significance of path coefficients and model parameters. The model was based on station means (n = 3), and all latent variable indicators satisfied recommended reliability thresholds (composite reliability > 0.7, AVE > 0.5). Internal consistency reliability of the reflective measurement model was assessed using composite reliability (ρc), as proposed by Hair et al.52 Values of ρc ≥ 0.70 indicate acceptable reliability for reflective constructs; in our models, ρc values of 0.86 (bare sediments) and 0.89 (seagrass sediments) confirmed the good internal consistency of the reflective construct sediment characteristics.
000 subsamples was utilized to evaluate the statistical significance of path coefficients and model parameters. The model was based on station means (n = 3), and all latent variable indicators satisfied recommended reliability thresholds (composite reliability > 0.7, AVE > 0.5). Internal consistency reliability of the reflective measurement model was assessed using composite reliability (ρc), as proposed by Hair et al.52 Values of ρc ≥ 0.70 indicate acceptable reliability for reflective constructs; in our models, ρc values of 0.86 (bare sediments) and 0.89 (seagrass sediments) confirmed the good internal consistency of the reflective construct sediment characteristics.
The implementation of statistical procedures was conducted utilizing R software v. 4.3.3 (Development Core Team, 2022), except for the PLS-SEM analysis, which was performed employing XLSTAT software. In both cases, the significance level was set at α = 0.05.
5. Conclusions
Upwelling bays are highly productive marine systems where the microbial oxygen demand in the sediment, development of redox gradients, and availability of diverse electron donors (e.g., organic carbon, and NH4+) favor the processes involved in N loss.32 This study is the first to assess the variability in Feammox activity and its contribution to overall NH4+ loss in a coastal lagoon influenced by contrasting upwelling conditions. Potential Feammox rates ranged from 5.99 ± 1.30 to 39.19 ± 3.25 mg N per m2 per day. Feammox was affected by location-specific environmental factors, primarily regarding the level of confinement in relation to the nearby ocean. The PLS-SEM analysis indicated that sediment characteristics, including moisture, SOC, CCHO, and microbiologically reducible Fe(III) and Mn(IV), directly influenced Feammox activity in seagrass sediments. In the adjacent bare zones, the effect of sediment characteristics was indirect and mediated through dissimilatory Fe(III) reduction. The RDA and VPA results also suggested that organic matter associated with upwelling-driven phytoplankton blooms and MPB may influence the abundance and activity of the IRB community inhabiting the bare sediments near the mouth of BSQ. Although the distribution of Geobacteraceae and Acidimicrobiaceae A6 across lagoonal sediments could be posited as an indication of Feammox functionality, transcript-based analysis and functional marker detection would be necessary to establish direct evidence between specific microbial taxa and the Feammox process.A minimum of 3.7 ± 0.5 mg N per m2 per day in bare sediments was facilitated by electron acceptors besides Fe(III), evidencing the co-occurrence of alternative chemoautotrophic processes in NH4+ removal. The loss of NH4+ through N2 linked to anaerobic NH4+ oxidation varied from 92.6 kg day−1 to 137.8 kg day−1, accounting for 5.1 ± 1.6% of the total oceanic N loading into BSQ, under relaxed and favorable upwelling conditions, respectively. Overall, this study shows the potential importance of Feammox to N removal and provides valuable insight into the mechanisms of sedimentary NH4+ transformations in coastal ecosystems in eastern boundary upwelling regions.
Author contributions
Guillermo Samperio-Ramos: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, supervision, writing – original draft. Oscar Hernández-Sánchez: data curation, formal analysis, software, writing – review & editing. Jorge A. Velásquez-Aristizábal: data curation, formal analysis, software, writing – review & editing. Víctor F. Camacho-Ibar: conceptualization, investigation, supervision, writing – review & editing. Silvia Pajares: data curation, formal analysis, funding acquisition, investigation, methodology, software, supervision, writing – review & editing. Aaron Gutiérrez: formal analysis, validation, writing – review & editing. Ariadna Aldrich-Rodríguez: formal analysis, software, writing – review & editing. Francisco J. Cervantes: conceptualization, investigation, funding acquisition, investigation, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5em00257e.
Acknowledgements
This work was financially supported by the Office for Scientific Research, Technology, and Innovation (SECIHTI) through the Program Frontiers in Science (grant number CF-2019-682328). We thank the members of the CiNEMa-UABC Research Group for their assistance with field sampling and sample processing. We are grateful to Jose Manuel Guzman Calderon, MSc. Anahí Bermúdez, Dr Jose Miguel Sandoval Gil, and personnel from Ostricola Nautilus for their assistance during field sampling. We thank Andrea MacTavish and MSE for English language editing. We also appreciate suggestions from anonymous reviewers. This study uses data from Environmental Research Division of the National Oceanic and Atmospheric Administration (NOAA) and the UC Santa Cruz Ocean Modelling Group.References
- F. Giomi, A. Barausse, A. Steckbauer, D. Daffonchio, C. M. Duarte and M. Fusi, Oxygen dynamics in marine productive ecosystems at ecologically relevant scales, Nat. Geosci., 2023, 16(7), 560–566 CrossRef CAS.
- J. Forrester, N. Leonardi, J. R. Cooper and P. Kumar, Seagrass as a nature-based solution for coastal protection, Ecol. Eng., 2024, 206, 107316 CrossRef.
- P. I. Macreadie, M. D. P. Costa, T. B. Atwood, D. A. Friess, J. J. Kelleway, H. Kennedy, C. E. Lovelock, O. Serrano and C. M. Duarte, Nat. Rev. Earth Environ., 2021, 2, 826–839 CrossRef CAS.
- P. Bonet-Melià, J. M. Sandoval-Gil, G. Samperio-Ramos, M. Vivanco-Bercovich, S. R. Canino-Herrera, R. Durazo, V. F. Camacho-Ibar and A. Alexandre, Mar. Pollut. Bull., 2023, 196, 115651 CrossRef PubMed.
- K. E. Brodersen, K. Koren, M. Moßhammer, P. J. Ralph, M. Kühl and J. Santner, Seagrass-Mediated Phosphorus and Iron Solubilization in Tropical Sediments, Environ. Sci. Technol., 2017, 51, 14155–14163 CrossRef CAS PubMed.
- E. M. Sogin, D. Michellod, H. R. Gruber-Vodicka, P. Bourceau, B. Geier, D. V. Meier, M. Seidel, S. Ahmerkamp, S. Schorn, G. D'Angelo, G. Procaccini, N. Dubilier and M. Liebeke, Sugars dominate the seagrass rhizosphere, Nature Ecology and Evolution, 2022, 6, 866–877 CrossRef PubMed.
- J. M. Sandoval-Gil, M. del Carmen Ávila-López, V. F. Camacho-Ibar, J. M. Hernández-Ayón, J. A. Zertuche-González and A. Cabello-Pasini, Estuaries Coasts, 2019, 42, 731–742 CrossRef CAS.
- J. A. Fuhrman, J. A. Cram and D. M. Needham, Marine microbial community dynamics and their ecological interpretation, Nat. Rev. Microbiol., 2015, 13, 133–146 CrossRef CAS PubMed.
- E. E. Rios-Del Toro, E. I. Valenzuela, J. E. Ramírez, N. E. López-Lozano and F. J. Cervantes, Anaerobic ammonium oxidation linked to microbial reduction of natural organic matter in marine sediments, Environ. Sci. Technol. Lett., 2018, 5, 571–577 CrossRef CAS.
- K. R. Salk, D. V. Erler, B. D. Eyre, N. Carlson-Perret and N. E. Ostrom, Unexpectedly high degree of anammox and DNRA in seagrass sediments: description and application of a revised isotope pairing technique, Geochim. Cosmochim. Acta, 2017, 211, 64–78 CrossRef CAS.
- K. Vopel, C. Del-Río and C. A. Pilditch, Effects of CO2 enrichment on benthic primary production and inorganic nitrogen fluxes in two coastal sediments, Sci. Rep., 2018, 8, 1035 CrossRef PubMed.
- M. P. Turschwell, R. M. Connolly, J. C. Dunic, M. Sievers, C. A. Buelow, R. M. Pearson, V. J. D. Tulloch, I. M. Côté, R. K. F. Unsworth, C. J. Collier and C. J. Brown, Anthropogenic pressures and life history predict trajectories of seagrass meadow extent at a global scale, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2110802118 CrossRef CAS PubMed.
- X. Zhang, B. B. Ward and D. M. Sigman, Global nitrogen cycle: critical enzymes, organisms, and processes for nitrogen budgets and dynamics, Chem. Rev., 2020, 120, 5308–5351 CrossRef CAS PubMed.
- N. Garcias-Bonet, M. Fusi, M. Ali, D. R. Shaw, P. E. Saikaly, D. Daffonchio and C. M. Duarte, High denitrification and anaerobic ammonium oxidation contributes to net nitrogen loss in a seagrass ecosystem in the central Red Sea, Biogeosciences, 2018, 15, 7333–7346 CrossRef CAS.
- L. Y. Liu, G. J. Xie, D. F. Xing, B. F. Liu, J. Ding, G. L. Cao and N. Q. Ren, Sulfate dependent ammonium oxidation: a microbial process linked nitrogen with sulfur cycle and potential application, Environ. Res., 2021, 192, 110282 CrossRef CAS PubMed.
- Y.-Q. He, G.-X. Li, S.-Y.-D. Zhou, R.-F. Li, J.-Y. Zhao, K. Wu, J. Wang, X.-Y. Jia, X.-T. Wu, F. Gao, Y.-Y. Xu and P. Bao, Bacteria Involved in Thiosulfate reduction coupled with anaerobic ammonium oxidation in the critical zone groundwater, ACS Earth Space Chem., 2021, 5, 2142–2151 CrossRef CAS.
- E. E. Rios-Del Toro, E. I. Valenzuela, N. E. López-Lozano, M. G. Cortés-Martínez, M. A. Sánchez-Rodríguez, O. Calvario-Martínez, S. Sánchez-Carrillo and F. J. Cervantes, Anaerobic ammonium oxidation linked to sulfate and ferric iron reduction fuels nitrogen loss in marine sediments, Biodegradation, 2018, 29, 429–442 CrossRef CAS PubMed.
- S. Chen, D. Gao, X. Li, D. Sun, Y. Zheng, H. Dong, X. Liang, G. Yin, P. Han, M. Liu and L. Hou, Nitrogen loss from anaerobic ammonium oxidation coupled to iron(III) reduction activity across estuarine and coastal wetlands of China: spatial variations, controlling factors, and environmental implications, Catena, 2023, 222, 106805 CrossRef CAS.
- X. Li, L. Hou, M. Liu, Y. Zheng, G. Yin, X. Lin, L. Cheng, Y. Li and X. Hu, Evidence of Nitrogen loss from anaerobic ammonium oxidation coupled with ferric iron reduction in an intertidal wetland, Environ. Sci. Technol., 2015, 49, 11560–11568 CrossRef CAS PubMed.
- A. Lai, S. Fan, J. Xue, H. Wang, K. Xie, H. Li, H. Xu, B. Li and Q. L. Wu, Ammonium removal through anaerobic ammonium oxidation coupled to iron(III) reduction along the Yangtze river–estuary continuum, J. Environ. Sci., 2025, 152, 178–187 CrossRef CAS PubMed.
- X. N. Wang, G. X. Sun and Y. G. Zhu, Thermodynamic energy of anaerobic microbial redox reactions couples elemental biogeochemical cycles, J. Soils Sediments, 2017, 17, 2831–2846 CrossRef CAS.
- G. Samperio-Ramos, O. Hernández-Sánchez, V. F. Camacho-Ibar, S. Pajares, A. Gutiérrez, J. M. Sandoval-Gil, M. Reyes, S. De Gyves, S. Balint, A. Oczkowski, S. J. Ponce-Jahen and F. J. Cervantes, Ammonium loss microbiologically mediated by Fe(III) and Mn(IV) reduction along a coastal lagoon system, Chemosphere, 2023, 349, 140933 CrossRef PubMed.
- K. Shi, J. Ju, M. Elsamadony, M. Fujii, J. Liu, J. Qin, Z. Liao and C. Ou, Emerging Feammox technology: mechanisms, biotechnological applications, and future prospects, ACS ES&T Eng., 2024, 4, 2856–2873 Search PubMed.
- S. Huang and P. R. Jaffé, Characterization of incubation experiments and development of an enrichment culture capable of ammonium oxidation under iron-reducing conditions, Biogeosciences, 2015, 12, 769–779 CrossRef.
- H. Li, J. Q. Su, X. R. Yang, G. W. Zhou, S. B. Lassen and Y. G. Zhu, RNA stable isotope probing of potential Feammox population in paddy soil, Environ. Sci. Technol., 2019, 53, 4841–4849 CrossRef CAS PubMed.
- C. Reyes and P. Meister, in Systems Biogeochemistry of Major Marine Biomes, John Wiley & Sons, Ltd, 2022, pp. 41–60 Search PubMed.
- B. Ding, Y. Qin, W. Luo and Z. Li, Spatial and seasonal distributions of Feammox from ecosystem habitats in the Wanshan region of the Taihu watershed, China, Chemosphere, 2020, 239, 124742 CrossRef CAS PubMed.
- Q. S. Guan, W. Z. Cao, M. Wang, G. J. Wu, F. F. Wang, C. Jiang, Y. R. Tao and Y. Gao, Nitrogen loss through anaerobic ammonium oxidation coupled with iron reduction in a mangrove wetland, Eurasian J. Soil Sci., 2018, 69, 732–741 CrossRef CAS.
- T. Tanaya, K. Watanabe, S. Yamamoto, C. Hongo, H. Kayanne and T. Kuwae, Contributions of the direct supply of belowground seagrass detritus and trapping of suspended organic matter to the sedimentary organic carbon stock in seagrass meadows, Biogeosciences, 2018, 15, 4033–4045 CrossRef CAS.
- Z. Yao, L. Yang, N. Song, C. Wang and H. Jiang, Effect of organic matter derived from algae and macrophyte on anaerobic ammonium oxidation coupled to ferric iron reduction in the sediment of a shallow freshwater lake, Environ. Sci. Pollut. Res. Int., 2020, 27, 25899–25907 CrossRef CAS PubMed.
- J. L. Largier, Upwelling bays: how coastal upwelling controls circulation, habitat, and productivity in bays, Annual Review of Marine Science, 2020, 12, 415–447 CrossRef PubMed.
- D. M. Arroyave-Gómez, D. Gallego-Suarez, M. Bartoli and M. Toro-Botero, Spatial and seasonal variability of sedimentary features and nitrogen benthic metabolism in a tropical coastal area (Taganga Bay, Colombia Caribbean) impacted by a sewage outfall, Biogeochemistry, 2020, 150, 85–107 CrossRef.
- J. R. Krause, A. Hinojosa-Corona, A. B. Gray, J. C. Herguera, J. McDonnell, M. V. Schaefer, S. C. Ying and E. B. Watson, Beyond habitat boundaries: organic matter cycling requires a system-wide approach for accurate blue carbon accounting, Limnol. Oceanogr., 2022, 67, S6–S18 CrossRef.
- S. E. Ibarra-Obando, S. V. Smith, M. Poumian-Tapia, V. Camacho-Ibar, J. D. Carriquiry and M. Montes-Hugo, Benthic metabolism in San Quintin Bay, Baja California, Mexico, Mar. Ecol.: Prog. Ser., 2004, 283, 99–112 CrossRef CAS.
- J. M. Caffrey, J. T. Hollibaugh, N. Bano and J. Haskins, Effects of upwelling on short-term variability in microbial and biogeochemical processes in estuarine sediments from Elkhorn Slough, California, USA, Aquat. Microb. Ecol., 2010, 58, 261–271 CrossRef.
- D. Melaku Canu, L. Aveytua-Alcázar, V. F. Camacho-Ibar, S. Querin and C. Solidoro, Hydrodynamic properties of San Quintin Bay, Baja California: merging models and observations, Mar. Pollut. Bull., 2016, 108, 203–214 CrossRef CAS PubMed.
- Y. Quilfen, J. Shutler, J. F. Piolle and E. Autret, Recent trends in the wind-driven California current upwelling system, Remote Sensing of Environment, 2021, 261, 112486 CrossRef.
- L. Aveytua-Alcazar, D. Melaku Canu, V. F. Camacho-Ibar and C. Solidoro, Changes in upwelling regimes in a Mediterranean-type lagoon: a model application, Ecol. Modell., 2020, 418, 108908 CrossRef CAS.
- J. R. Krause, A. Hinojosa-Corona, A. B. Gray and E. B. Watson, Emerging sensor platforms allow for seagrass extent mapping in a turbid estuary and from the meadow to ecosystem scale, Remote Sens., 2021, 13, 3681 CrossRef.
- L. W. Daesslé, G. Rendón-Márquez, V. F. Camacho-Ibar, E. A. Gutiérrez-Galindo, E. Shumilin and E. Ortiz-Campos, Geochemistry of modern sediments from San Quintín coastal lagoon, Baja California: implication for provenance, Rev. Mex. Cienc. Geol., 2009, 26, 117–132 Search PubMed.
- V. F. Camacho-Ibar, J. D. Carriquiry and S. V. Smith, Non-conservative P and N fluxes and net ecosystem production in San Quintin Bay, México, Estuaries, 2003, 26, 1220–1237 CrossRef CAS.
- E. M. Jorgensen, E. L. Hazen, M. G. Jacox, M. Pozo Buil, I. Schroeder and S. J. Bograd, Physical and biogeochemical phenology of coastal upwelling in the California Current System, Geophys. Res. Lett., 2024, 51, e2024GL108194 CrossRef CAS.
- M. G. Jacox, C. A. Edwards, E. L. Hazen and S. J. Bograd, Coastal upwelling revisited: Ekman, Bakun, and improved upwelling indices for the U.S. West Coast, J. Geophys. Res.: Oceans, 2018, 123, 7332–7350 CrossRef.
- F. J. Millero, The pH of estuarine waters, Limnol. Oceanogr., 1986, 31, 839–847 CrossRef CAS.
- J. Ma, P. Li, K. Lin, Z. Chen, N. Chen, K. Liao and D. Yuan, Optimization of a salinity-interference-free indophenol method for the determination of ammonium in natural waters using o-phenylphenol, Talanta, 2018, 179, 608–614 CrossRef CAS PubMed.
- E. Lee-Sánchez, V. F. Camacho-Ibar, J. A. Velásquez-Aristizábal, J. A. Valencia-Gasti and G. Samperio-Ramos, Impacts of mesoscale eddies on the nitrate distribution in the deep-water region of the Gulf of Mexico, Journal of Marine Systems, 2022, 229, 103721 CrossRef.
- A. Pusceddu, A. Dell'Anno, M. Fabiano and R. Danovaro, Quantity and bioavailability of sediment organic matter as signatures of benthic trophic status, Mar. Ecol.: Prog. Ser., 2009, 375, 41–52 CrossRef CAS.
- S. M. Myklestad, E. Skånøy and S. Hestmann, A sensitive and rapid method for analysis of dissolved mono- and polysaccharides in seawater, Mar. Chem., 1997, 56, 279–286 CrossRef CAS.
- G. Samperio-Ramos, J. M. Santana Casiano and M. González Dávila, Effect of ocean warming and acidification on the Fe(II) oxidation rate in oligotrophic and eutrophic natural waters, Biogeochemistry, 2016, 128, 19–34 CrossRef CAS.
- C. S. Chin, K. S. Johnson and K. H. Coale, Spectrophotometric determination of dissolved manganese in natural waters with 1-(2-pyridylazo)-2-naphthol: application to analysis in situ in hydrothermal plumes, Mar. Chem., 1992, 37, 65–82 CrossRef CAS.
- M. Bossé, E. Marland, G. Rhoads, J. A. Sanqui and Z. BeMent, Generalizing R2 for deming regressions, Communications in Statistics - Theory and Methods, 2023, 52, 7731–7743 CrossRef.
- J. F. Hair Jr, G. T. M. Hult, C. M. Ringle and M. Sarstedt, A Primer on Partial Least Squares Structural Equation Modeling (PLS-SEM), Sage Publications, Inc., 3rd edn, 2021 Search PubMed.
- W. H. Yang, K. A. Weber and W. L. Silver, Nitrogen loss from soil through anaerobic ammonium oxidation coupled to iron reduction, Nat. Geosci., 2012, 5, 538–541 CrossRef CAS.
- Z. Yao, F. Wang, C. Wang, H. Xu and H. Jiang, Anaerobic ammonium oxidation coupled to ferric iron reduction in the sediment of a eutrophic lake, Environ. Sci. Pollut. Res. Int., 2019, 26, 15084–15094 CrossRef CAS PubMed.
- Y. She, X. Qi, X. Xin, Y. He, W. Wang and Z. Li, Non-rhizosphere reinforces the contributions of Feammox and anammox to nitrogen loss than rhizosphere in riparian zones, Environ. Res., 2023, 239, 117317 CrossRef CAS PubMed.
- K. A. Avendaño, S. J. Ponce-Jahen, E. I. Valenzuela, S. Pajares, G. Samperio-Ramos, V. F. Camacho-Ibar and F. J. Cervantes, Nitrogen loss in coastal sediments driven by anaerobic ammonium oxidation coupled to microbial reduction of Mn(IV)-oxide, Sci. Total Environ., 2024, 923, 171368 CrossRef PubMed.
- D. R. Shaw, M. Ali, K. P. Katuri, J. A. Gralnick, J. Reimann, R. Mesman, L. van Niftrik, M. S. M. Jetten and P. E. Saikaly, Extracellular electron transfer-dependent anaerobic oxidation of ammonium by anammox bacteria, Nat. Commun., 2020, 11, 2058 CrossRef CAS PubMed.
- B. Aldeguer-Riquelme, E. Rubio-Portillo, J. Álvarez-Rogel, F. Giménez-Casalduero, X. L. Otero, M. D. Belando, J. Bernardeau-Esteller, R. García-Muñoz, A. Forcada, J. M. Ruiz, F. Santos and J. Antón, Factors structuring microbial communities in highly impacted coastal marine sediments (Mar Menor lagoon, SE Spain), Front. Microbiol., 2022, 13, 937683 CrossRef PubMed.
- M. F. Gracia-Escobar, R. Millán-Núñez, E. Valenzuela-Espinoza, A. González-Silvera and E. Santamaría-del-Ángel, Changes in the Composition and Abundance of Phytoplankton in a Coastal Lagoon of Baja California, México, during 2011, Open Journal of Marine Science, 2015, 5, 169–181 CrossRef.
- J. Morelle, P. Claquin and F. Orvain, Evidence for better microphytobenthos dynamics in mixed sand/mud zones than in pure sand or mud intertidal flats (Seine estuary, Normandy, France), PLoS One, 2020, 15, e0237211 CrossRef CAS PubMed.
- C. P. Le, H. T. Nguyen, T. D. Nguyen, Q. H. M. Nguyen, H. T. Pham and H. T. Dinh, Ammonium and organic carbon co-removal under Feammox-coupled-with-heterotrophy condition as an efficient approach for nitrogen treatment, Sci. Rep., 2021, 11, 784 CrossRef CAS PubMed.
- H. T. Nguyen, L. D. Nguyen, C. P. Le, N. D. Hoang and H. T. Dinh, Nitrogen and carbon removal from anaerobic digester effluents with low carbon to nitrogen ratios under Feammox conditions, Bioresour. Technol., 2023, 371, 128585 CrossRef CAS PubMed.
- H. González, C. Rodríguez, M. González, C. González, J. Serrano and E. Leiva, Ammonium and organic carbon co-removal under Feammox heterotrophic conditions at low Fe3+ concentrations, Bioresour. Technol., 2025, 434, 132750 CrossRef PubMed.
- J. B. Rollefson, C. S. Stephen, M. Tien and D. R. Bond, Identification of an extracellular polysaccharide network essential for cytochrome anchoring and biofilm formation in Geobacter sulfurreducens, J. Bacteriol., 2011, 193, 1023–1033 CrossRef CAS PubMed.
- Z. Zhuang, G. Yang and L. Zhuang, Exopolysaccharides matrix affects the process of extracellular electron transfer in electroactive biofilm, Sci. Total Environ., 2021, 806, 150713 CrossRef PubMed.
- M. Rico, P. Santiago-Díaz, G. Samperio-Ramos, M. González-Dávila and J. M. Santana-Casiano, Variations of polyphenols and carbohydrates of Emiliania huxleyi grown under simulated ocean acidification conditions, Biogeosciences, 2024, 21, 4381–4394 CrossRef CAS.
- A. E. Sherman, S. Huang and P. R. Jaffé, Impacts of storm disturbance and the role of the Feammox process in high nutrient riparian sediments, Biogeochemistry, 2023, 165, 113–128 CrossRef CAS.
- Y. Fan, S. Sun and S. He, Iron plaque formation and its effect on key elements cycling in constructed wetlands: functions and outlooks, Water Res., 2023, 235, 119837 CrossRef CAS PubMed.
- G. Samperio-Ramos, M. González-Dávila and J. M. Santana-Casiano, Impact on the Fe redox cycling of organic ligands released by Synechococcus PCC 7002, under different iron fertilization scenarios. Modeling approach, Journal of Marine Systems, 2018, 182, 67–78 CrossRef.
- D. Krause-Jensen, S. Markager and T. Dalsgaard, Benthic and Pelagic Primary Production in Different Nutrient Regimes, Estuaries Coasts, 2012, 35, 527–545 CrossRef CAS.
- N. Soto, G. Winters and G. Antler, The effect of anaerobic remineralization of the seagrass Halophila stipulacea on porewater biogeochemistry in the Gulf of Aqaba, Frontiers in Marine Science, 2023, 10, 1250931 CrossRef.
- E. B. Watson, A. Hinojosa-Corona, J. R. Krause, J. C. Herguera, J. McDonnell, K. R. V. Manríquez, M. E. Gannon and A. B. Gray, Lagoon biogeochemical processing is reflected in spatial patterns of sediment stable isotopic ratios, Journal of Marine Science and Engineering, 2020, 8, 1–19 CrossRef.
- E. L. Trainer, M. Ginder-Vogel and C. K. Remucal, Selective reactivity and oxidation of dissolved organic matter by manganese oxides, Environ. Sci. Technol., 2021, 55, 12084–12094 CrossRef CAS PubMed.
- C. A. Brown and R. J. Ozretich, Coupling between the coastal ocean and Yaquina Bay, Oregon: importance of oceanic inputs relative to other nitrogen sources, Estuaries Coasts, 2009, 32, 219–237 CrossRef CAS.
- P. Tett, L. Gilpin, H. Svendsen, C. P. Erlandsson, U. Larsson, S. Kratzer, E. Fouilland, C. Janzen, J.-Y. Lee, C. Grenz, A. Newton, J. G. Ferreira, T. Fernandes and S. Scory, Eutrophication and some European waters of restricted exchange, Cont. Shelf Res., 2003, 23, 1635–1671 CrossRef.
- E. K. Robertson, M. Bartoli, V. Brüchert, T. Dalsgaard, P. O. J. Hall, D. Hellemann, S. Hietanen, M. Zilius and D. J. Conley, Application of the isotope pairing technique in sediments: use, challenges, and new directions, Limnol. Oceanogr.: Methods, 2019, 17, 112–136 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |