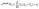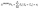Quantitative assessment of Pb sources in urban–rural river sediments based on Pb isotopes and PMF and MixSIAR models
Shanshan
Xi
ac,
Wei
Wang
a,
Lei
Sun
b,
Xing
Chen
 *ac,
Jiamei
Zhang
acd and
Fan
Yu
a
*ac,
Jiamei
Zhang
acd and
Fan
Yu
a
aSchool of Environmental and Energy Engineering, Anhui Jianzhu University, Hefei 230031, Anhui, China. E-mail: 15705610332@163.com
bAnhui Institute of Ecological Civilization, Hefei 230031, Anhui, China
cAnhui Provincial Key Laboratory of Environmental Pollution Control and Resource Reuse, Hefei 230031, Anhui, China
dPollution Control and Resource Utilization in Industrial Parks Joint Laboratory of Anhui Province, Hefei 230031, Anhui, China
First published on 2nd December 2025
Abstract
The complex land use patterns in urban–rural rivers and the presence of diverse point and non-point source pollution pose significant challenges for tracing heavy metal(loid) sources in river sediments. This study employed a combined approach using lead (Pb) stable isotopes, positive matrix factorization (PMF), and a Bayesian mixture model (MixSIAR) to determine the concentrations of Cr, As, Cd, Mn, Cu, Zn, Ni, and Pb along with Pb isotope distribution characteristics in sediments from a typical urban–rural river (Yinghe River). Our investigation enabled the quantitative identification of heavy metal(loid) sources and revealed the contribution patterns of multi-source Pb pollution. The results showed that mean concentrations of all heavy metal(loid)s except Cr and Mn exceeded local soil background values. PMF analysis identified four potential sources: natural sources (19.6%) contributing primarily Cr and Mn; industrial sources (32.1%) associated with Cd, Pb, and Ni; agricultural sources (28.0%) linked to Pb, As, and Zn; and traffic sources (20.3%) related to Cu and Zn. Furthermore, by combining Pb stable isotopes with MixSIAR, the contributions of different Pb pollution sources were quantified as agricultural sources (32.1%), industrial sources (30.5%), traffic sources (27.2%), and natural sources (10.3%). The less-than-10% difference in contribution rates between PMF and MixSIAR for Pb source apportionment demonstrated model reliability. Based on the significant correlation between Pb pollution and land use patterns in the Yinghe River, corresponding pollution prevention strategies were proposed. These findings provide a novel perspective for quantitative source identification of heavy metal(loid) pollution in urban–rural river sediments, offering valuable support for river management and heavy metal(loid) pollution control.
Environmental significanceThis study provides a new perspective for identifying the anthropogenic effects and quantitative sources of Pb pollution in urban–rural rivers, and is expected to be an important basis for guiding the decision-making of urban–rural river management and pollution prevention and control. |
1 Introduction
As the main reservoir of river pollutants, sediments accumulate nutrients, persistent organic pollutants, and heavy metal(loid)s.1–4 These pollutants are mainly derived from urban dust, agricultural runoff, atmospheric deposition, and direct industrial emissions.5 Among them, heavy metal(loid)s have become the core of sediment pollution research due to their high toxicity, persistence, and bioenrichment.6–8 During river transport, the low solubility of heavy metal(loid)s allows them to quickly adsorb to suspended particles and settle to form bottom sediments.9–11 Under certain conditions, heavy metal(loid)s in sediments can be released into water bodies and transmitted through the food chain, posing a threat to human health.12 This issue has attracted wide attention at home and abroad.13–16 Therefore, the accumulation of heavy metal(loid)s in sediments is regarded as an important environmental indicator that can be used to assess regional pollution levels.17–19Traditional source analysis of soil heavy metal(loid) pollution mainly relies on statistical models based on concentration data. For example, multivariate statistical methods (including principal component analysis and cluster analysis), positive matrix factorization (PMF), and absolute factor scores–multiple linear regression (APCS-MLR).20,21 These methods identify pollution source types by analyzing the correlation between elements or performing matrix factorization, and quantify the contribution rate of each pollution source based on statistical assumptions.22 However, in complex pollution environments, traditional methods often struggle to provide accurate and reliable pollution source analysis results. In recent years, Pb isotope tracing technology has gradually become an important tool for soil heavy metal(loid) pollution source analysis due to its unique “fingerprint” characteristics and high accuracy.23 By analyzing the differences in Pb isotope ratios of different pollution sources, this technology can accurately distinguish Pb pollution from different sources and quantitatively assess source contributions.24 Compared with traditional methods, Pb isotope tracing technology has higher anti-interference ability and requires a smaller sampling volume.25
The sources of heavy metal(loid) pollution in urban–rural rivers are extensive and complex, including industrial emissions, agricultural activities, domestic sewage, and traffic-related pollution.26–28 The differences in land use patterns and the intensity of human activities in different regions further aggravate the spatial differentiation of the occurrence forms and migration behaviors of heavy metal(loid)s in the environment.29 As a typical urban–rural river, the Yinghe River is dominated by point-source emissions from mineral mining and metal(loid) processing in the upstream industrial agglomeration areas, domestic sewage discharge and traffic pollution diffusion in the middle reaches of urbanized areas, and non-point source input pressure such as fertilizer and pesticide application in the downstream agricultural areas. The spatial and temporal superposition effects of industrial emissions, agricultural activities, and urban pollution sources, as well as the systematic differences in the intensity of human activities and land use patterns across the urban–rural gradient, lead to significant challenges in the interpretation of pollutant sources.
Given the complex source apportionment context in the Yinghe River, this study employed an integrated methodology to identify and quantify the sources and contributions of Pb pollution, thereby addressing the limitations of single-method approaches. The specific objectives were as follows: (1) to utilize principal component analysis (PCA) and cluster analysis (CA) for processing the concentrations of eight heavy metal(loid)s (Cr, Mn, Zn, Pb, Cu, Ni, Cd, and As) in sediments to qualitatively identify potential pollution sources; (2) to apply the PMF model for preliminary identification and quantification of major pollution sources; and (3) to leverage the high objectivity of Pb isotope tracing to further refine the quantification of source contributions, achieving a more accurate analysis of Pb pollution origins. This study provides a novel framework for identifying anthropogenic influences and quantitatively apportioning Pb sources in urban–rural rivers, offering a scientific basis for informed river management and pollution control strategies.
2 Materials and methods
2.1 Study area
The Shaying River basin is the largest tributary of the Huai River and one of its most polluted tributaries. It originates from Funiu Mountain in western Henan Province, crosses Henan and Anhui provinces, and flows through more than 40 cities and counties such as Pingdingshan, Luohe, Xuchang, Zhoukou and Fuyang, with a total length of 624 km. The Shaying River is called the Yinghe River in Anhui, and its main tributary is the Fenquan River. It starts from Jieshou City in Fuyang, flows through Taihe County, Yingquan County and Yingdong County, and finally merges into the Huai River through the mouth of the Mohe River in Yingshang County.30 Fuyang City is located in the north of Anhui Province, China, in the middle of Jianghuai Plain, with a total land area of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 346 square kilometers and a permanent population of about 2.3 million. The city has a temperate monsoon climate with four distinct seasons, an average annual temperature of 14–16 °C, and an average annual precipitation of 800–1100 mm.31 The land use types along the Yinghe River are rich and diverse, covering planting land, agricultural production land, urban construction and residential land, and other types of land dominated by industrial activities (Fig. 1). At the same time, this area is rich in coal resources, and with its resource advantages, it has become one of the important energy bases in China.32
346 square kilometers and a permanent population of about 2.3 million. The city has a temperate monsoon climate with four distinct seasons, an average annual temperature of 14–16 °C, and an average annual precipitation of 800–1100 mm.31 The land use types along the Yinghe River are rich and diverse, covering planting land, agricultural production land, urban construction and residential land, and other types of land dominated by industrial activities (Fig. 1). At the same time, this area is rich in coal resources, and with its resource advantages, it has become one of the important energy bases in China.32
2.2 Sample collection
As the main river in Fuyang City, the Yinghe River has significant spatial heterogeneity in the area through which its main stream flows. In this study, 27 sediment sampling points (Y1–Y27) were arranged along the main stream of Yinghe River, as shown in Fig. 1. The sample at each sampling point consisted of four to five subsamples at a collection depth of 0–10 cm collected using a Petersen grab. The locations of sampling sites were determined using a handheld global positioning system (GPS). In addition, in order to further study the potential sources of human influence, 13 samples of potential anthropogenic sources were collected from the surrounding area, including 3 samples of coal, 3 samples of automobile exhaust dust, 3 samples of metallurgical dust, 2 samples of compound fertilizer, and 2 samples of livestock manure. Three additional deep soil samples were collected and used as natural origin samples. All samples were stored in clean polyethylene zipper bags after collection and quickly transferred to the laboratory. The samples were air-dried and passed through a 2 mm screen to remove stones and plant debris. Samples were then ground and sieved through a 200-mesh nylon screen, and all samples were stored at 4 °C in the dark before analysis.332.3 Test and analysis
All the analyzed data underwent strict quality assurance and quality control. Instrument calibration was performed daily using reference materials to ensure the accuracy of the analytical results. The certified reference materials GBW07311 (GSD-11) and GBW07366 (GSD-23) were used to validate the precision and accuracy of the analytical method. The recovery rates of the certified reference materials for sediments GBW07311 (GSD-11) and GBW07366 (GSD-23) are presented in Table S1. The detection limits of Pb, Cr, Cu, Ni, Zn, Mn, As and Cd were 1 mg kg−1, 2 mg kg−1, 0.7 mg kg−1, 2 mg kg−1, 5 mg kg−1, 2 mg kg−1, 0.2 mg kg−1 and 0.03 mg kg−1, respectively. In each batch of sample processing, both procedural blanks and instrument blanks were analyzed simultaneously to monitor contamination. The concentrations of all target elements in the blanks were below the method detection limits (MDLs).
Pb isotope ratios were determined using a multi-collector inductively coupled plasma mass spectrometer (MC-ICP-MS, Nu Plasma) at the State Key Laboratory of Environmental Geochemistry, Institute of Geochemistry, Chinese Academy of Sciences, with an external reproducibility (1σ) of ± 0.01%. The internal standard thallium was used to calibrate instrument mass discrimination. Measurement accuracy was checked by analysis of certified reference material (NIST, SRM-981). The measured 206Pb/204Pb and 207Pb/204Pb ratios were 16.9347 ± 0.0007 and 15.4925 ± 0.0004 (n = 20), respectively, which are in good agreement with the values reported in previous studies.35,36
2.4 Data analysis
Basic statistical analyses were conducted using Microsoft Excel 2021 (Microsoft Corp, USA) software. The geo-accumulation index (Igeo) was adopted to evaluate the heavy metal(loid) pollution in sediments. To initially identify the potential sources of Pb pollution in sediments, principal component analysis (PCA) and factor analysis (CA) were performed using SPSS 27.0 (IBM Corp., USA), statistical software for social sciences. The Positive Matrix Factorization model (PMF 5.0, U.S. EPA) was applied to quantify the potential sources and contributions of Pb in the sediments. Pb stable isotope data and physicochemical data were processed using Origin 2022 (OriginLab Corp., Northampton, MA, USA), and relevant graphs were generated. The MixSIAR model (v1.2.4, University of California, Davis, USA) was employed to quantitatively apportion Pb pollution sources. Land use type analysis was conducted using ArcGIS 10.4 (Environmental Systems Research Institute, Redlands, CA, USA).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. Convergence statistics show that all Gelman–Rubin statistics are less than 1.01, indicating that the model met the convergence criteria. A total of 4 pollution sources were incorporated into the model, and 15 sediment samples were used as mixture data. The model expressions are as follows:
000. Convergence statistics show that all Gelman–Rubin statistics are less than 1.01, indicating that the model met the convergence criteria. A total of 4 pollution sources were incorporated into the model, and 15 sediment samples were used as mixture data. The model expressions are as follows:| Sk ∼ N(µk,ωk2) |
| Ck ∼ N(λk,τk2) |
| εk ∼ N(0,σ2) |
| Type of pollution source | 206Pb/207Pb (mean ± SD) | 208Pb/206Pb (mean ± SD) | Number of samples | Data source |
|---|---|---|---|---|
| Industrial source | 1.174 ± 0.008 | 2.110 ± 0.008 | 6 | Metallurgical dust and industrial coal combustion |
| Agricultural source | 1.183 ± 0.004 | 2.090 ± 0.007 | 4 | Livestock manure and compound fertilizer |
| Traffic source | 1.160 ± 0.005 | 2.108 ± 0.004 | 3 | Automobile exhaust deposited particles |
| Natural source | 1.196 ± 0.005 | 2.082 ± 0.003 | 3 | Deep soil in the study area |
| Igeo = log2(Ci/1.5Si) |
3 Results
3.1 Composition of heavy metal(loid)s in sediments
Based on the land use types and spatial distribution characteristics, the main stream of the Yinghe River can be divided into three sections: IM (Y1–Y9) in the upstream section was dominated by industrial mixed land, UM (Y10–Y18) in the middle stream was dominated by urban mixed land, and AM (Y19–Y27) in the downstream section was mainly agricultural mixed land. Table 2 lists the concentrations of 8 elements in the sediments. The results showed that the mean concentrations (range) of Mn, Zn, Cr, Pb, Cu, Ni, As, and Cd were 511.6 (304.3–731.7), 73.79 (57.72–94.72), 63.04 (42.64–89.92), 29.10 (19.56–45.96), 28.76 (21.31–44.42), 28.74 (22.23–41.63), 11.43 (6.35–23.74), and 0.31 (0.20–0.42) mg kg−1, respectively. River sediments are a mixture of surface weathering products in the watershed, so the element concentrations in sediments are closely related to the soil environment of the watershed.39 The Igeo analysis results showed that Cd in all sediment samples was moderately contaminated or unpolluted to moderately contaminated; As in 7 samples was unpolluted to moderately contaminated; Zn in 5 samples was unpolluted to moderately contaminated; Pb in 4 samples was unpolluted to moderately contaminated; Cu and Ni in 2 samples were unpolluted to moderately contaminated; while Mn and Cr in all samples were almost uncontaminated. The order of average Igeo values in all samples was: Cd > Zn > As > Pb > Ni > Cu > Mn > Cr.| Land use types | Mn | Zn | Cr | Pb | Cu | Ni | As | Cd | |
|---|---|---|---|---|---|---|---|---|---|
| a IM, industrial mixed land; UM, urban mixed land; AM, agricultural mixed land. | |||||||||
| IM (Y1∼Y9) | Max | 731.69 | 94.72 | 89.92 | 43.86 | 44.42 | 39.33 | 23.74 | 0.42 |
| Min | 403.89 | 64.52 | 55.22 | 23.56 | 23.12 | 26.03 | 10.64 | 0.24 | |
| Mean | 532.83 | 78.09 | 63.54 | 31.35 | 31.19 | 29.69 | 14.23 | 0.33 | |
| SD | 103.39 | 7.97 | 10.34 | 7.68 | 5.43 | 4.21 | 3.95 | 0.05 | |
| UM (Y10∼Y18) | Max | 78.64 | 75.92 | 78.62 | 45.96 | 34.62 | 34.63 | 13.34 | 0.37 |
| Min | 42.62 | 57.72 | 42.62 | 19.56 | 21.31 | 22.23 | 6.84 | 0.20 | |
| Mean | 490.11 | 69.13 | 62.90 | 28.17 | 26.57 | 27.35 | 10.06 | 0.28 | |
| SD | 72.31 | 5.64 | 9.83 | 8.67 | 3.87 | 2.64 | 1.94 | 0.04 | |
| AM (Y19∼Y27) | Max | 685.39 | 89.72 | 82.22 | 39.66 | 40.12 | 41.63 | 15.74 | 0.40 |
| Min | 304.30 | 66.92 | 47.82 | 22.76 | 23.82 | 25.73 | 6.35 | 0.25 | |
| Mean | 511.86 | 74.17 | 62.68 | 27.79 | 28.54 | 29.19 | 10.04 | 0.32 | |
| SD | 101.05 | 7.24 | 9.03 | 4.75 | 4.99 | 4.58 | 3.32 | 0.05 | |
| Total | Max | 731.69 | 94.72 | 89.92 | 45.96 | 44.42 | 41.63 | 23.74 | 0.42 |
| Min | 304.30 | 57.72 | 42.64 | 19.56 | 21.31 | 22.23 | 6.35 | 0.20 | |
| Mean | 511.60 | 73.79 | 63.04 | 29.10 | 28.76 | 28.74 | 11.43 | 0.31 | |
| SD | 94.94 | 7.82 | 9.75 | 7.13 | 5.17 | 4.02 | 3.74 | 0.05 | |
3.2 PMF results
Four principal components were identified in the sediments through principal component analysis (PCA). The variance explained by each component and its factor contributions to the variables were further assessed using positive matrix factorization (PMF), as shown in Fig. 2.F1 accounted for 19.6% of the four components, mainly including Mn (43.9%) and Cr (41.3%). F2 accounted for 28% of the four components, mainly including As (61.2%), Pb (33.1%) and Zn (24.6%). F3 accounted for 32.1% of the four components, mainly including Cd (48.2%), Ni (37.7%) and Pb (33.6%). F4 accounted for 20.3% of the four components, mainly including Cu (38.8%) and Zn (31.4%).
3.3 Pb isotopic composition
Pb pollution in sediments mainly originates from human activities, including non-ferrous metal(loid) mining and smelting, coal burning, traffic-related sources, and the use of Pb-containing compound fertilizers, livestock manure, and pesticides in agricultural activities.22,40–42 In the present study, three categories of representative anthropogenic source samples were systematically collected from the research area, including livestock manure and compound fertilizers (representing agricultural sources), metallurgical dust and coal combustion residues (representing industrial sources), and automobile exhaust deposition particles (representing traffic sources). Concurrently, deep soil samples from the vicinity of the study area were collected as natural source samples. Pb isotope analyses were subsequently performed on 15 sediment samples, 13 potential anthropogenic source samples, and 3 natural source samples. Fig. 3 shows the relationships between the 206Pb/207Pb and 208Pb/206Pb ratios in sediment samples, as well as the relationships between 1/Pb and the 206Pb/207Pb and 208Pb/206Pb ratios, respectively. | ||
| Fig. 3 The ratios of (a)206Pb/207Pb and 208Pb/206Pb, 1/Pb and (b)206Pb/207Pb, and (c) 208Pb/206Pb in sediments. IM: industrial mixed land; UM: urban mixed land; AM: mixed agricultural land. | ||
In general, natural sources exhibit a relatively higher 206Pb/207Pb ratio.34 The 206Pb/207Pb and 208Pb/206Pb ratios in the deep soil samples from the study area are 1.1965 ± 0.0045 and 2.0815 ± 0.0025, respectively. In comparison, anthropogenic sources show a relatively low 206Pb/207Pb ratio. The 206Pb/207Pb and 208Pb/206Pb ratios of anthropogenic sources are as follows: agricultural sources (1.182 ± 0.004; 2.090 ± 0.007), industrial sources (1.173 ± 0.009; 2.111 ± 0.010), and traffic sources (1.1605 ± 0.0045; 2.1075 ± 0.0035). The lead isotope composition of sediment samples shows significant spatial variations, and such variations are closely related to the land use pattern of the river basin (Fig. 3a). The lead isotope composition (206Pb/207Pb and 208Pb/206Pb) in the upstream industrial area (IM) are 1.1776 ± 0.0030 and 2.1158 ± 0.0040, respectively; those in the midstream urban area (UM) are 1.1628 ± 0.0030 and 2.102 ± 0.0045, respectively; and those in the downstream agricultural area (AM) are 1.1806 ± 0.0025 and 2.09 ± 0.0055, respectively. In this study, the MixSIAR model was used to analyze the lead isotope ratios of 15 sediment samples, and the results were presented in the form of boxplots. In the boxplots, the lower quartile, upper quartile, and median of the box correspond to the values of the MixSIAR model variables at the 25%, 75%, and 50% probability levels, respectively. The lower and upper limits of the whiskers correspond to the values of the model variables at the 5% and 95% probability levels, respectively; the whiskers typically represent the confidence interval, so as to reflect the uncertainty range of the variable values. Additionally, in this study, the mean value from the MixSIAR model run was used to characterize source contribution, as this mean value can serve as a comprehensive estimate of the “most probable values” of the parameters. The source analysis results based on the MixSIAR model showed that the contribution rates of Pb in sediments were as follows: agricultural source (32.1%) > industrial source (30.5%) > traffic source (27.2%) > natural source (10.3%) (Fig. 4a).
 | ||
| Fig. 4 Contribution of Pb in sediments from potential sources based on MixSIAR(a and b) and PMF (a). The mean value represents the source contribution. | ||
4 Discussion
4.1 Assessment of heavy metal(loid) pollution and source contribution
In the upper IM reach, the average Igeo values of Zn and As are greater than 0 and close to the level of moderate contamination, indicating significant enrichment of Zn and As. In the middle UM reach, except for Cd, the average Igeo values of all other elements are less than 0, suggesting almost no heavy metal(loid) contamination. This variation may be related to the effective interception of upstream pollutants by urban sewage treatment facilities and the management of urban point source pollution.44 In the lower AM reach, the average Igeo values of Pb and Cu increase significantly and are both close to the level of moderate contamination, which may be potentially affected by agricultural non-point source pollution. The average Igeo values of Cd in all three reaches are close to or exceed 1, indicating that Cd contamination is severe and affected by multiple factors, including not only industrial waste discharge from the upper reaches but also external pollution inputs from agricultural production and urbanization processes.45
There is a positive correlation between As and Pb. As is an important compound fertilizer and an organic fertilizer component, and is often used together with Pb as a livestock feed additive.49 This mode of use, which is used to promote animal growth, may lead to significantly higher concentrations of arsenic and Pb in livestock manure.50 In addition, Zn, as a common component in agricultural pesticides, contributes about 1200 tons of Zn input to agricultural soils in China each year through pesticide application.51 Considering the large agricultural runoff in the study area, F2 can be judged as an agricultural pollution source.
Cd and Ni were strongly and positively correlated. Numerous studies have shown that Cd and Ni originate from industrial activities, such as perennial mining, including ore smelting, coal consumption, steel production, and metal(loid) processing.52,53 Fuyang City has the largest green recycling and utilization base of regenerated Pb in China, and Pb, Cd and Ni can also accumulate in the environment due to metal(loid) smelting and coal combustion. Therefore, F3 is considered to be of industrial origin.
Cu and Zn are positively correlated, and both Cu and Zn are important components of key parts such as vehicle brake systems, wheel bearing, and transmission systems. Due to the friction generated during vehicle braking, dust containing Cu and Zn enters the surface soil together with dust in the atmosphere.54,55 In addition, modern vehicles are usually supplemented with copper containing lubricants and fuel additives during use. These substances are emitted through the exhaust during vehicle driving, and Zn is also gradually released into the environment through tire friction.56 Therefore, F4 is considered to be the source of traffic.
The weak contribution of natural sources confirmed that human activities were the dominant factor influencing sediment Pb content. The source of Pb in sediments is significantly coupled with the land use pattern in the study area, as shown in Fig. 4b. In the IM section, the contribution of industrial sources to Pb in sediments was the most significant, while the contribution of the other three Pb sources was much lower than that of industrial sources, which was closely related to the spatial distribution of non-ferrous metal(loid) smelting industrial clusters such as recycled Pb industrial parks and coal-fired power plants in this section.58 In the UM section, the contribution of traffic sources to Pb in sediments was the most prominent, while the contribution of natural sources was relatively minimal, which might be attributed to the changes of topography in the process of urbanization and the emission of Pb-containing particles caused by high-intensity traffic activities.59 In the AM section dominated by agricultural activities, the contribution of agricultural sources to Pb in sediments was the most significant, while the contributions of industrial sources and natural sources were similar. It may be related to the application of compound fertilizer in intensive agricultural areas, the long-term accumulation of waste from livestock and poultry breeding, and the input of compound pollution caused by the dense distribution of rural settlements.60,61
4.2 Suggestions for prevention and control of Pb pollution
As shown in Fig. 5, the source apportionment results derived from the MixSIAR model revealed pronounced spatial heterogeneity in the contribution of Pb pollution sources across different land use patterns. This indicates that human activities—specifically industrial, agricultural, and traffic sources—are the primary contributors to Pb pollution, necessitating differentiated control strategies.62 Watershed ecosystem restoration and pollution load control are the core approaches to ensure river water quality and reduce Pb input.38 Among them, industrial wastewater discharge containing Pb and other toxic heavy metal(loid)s should be the key focus of control, as such point source pollution poses direct hazards to water, soil and river ecosystems.63 In high-intensity industrial activity areas represented by the IM section, source reduction should be achieved by strictly formulating pollutant discharge standards and promoting clean production technologies.64 For agricultural-dominated areas represented by the AM section, it is necessary to optimize fertilization patterns, adopt precision fertilization technologies, and promote bio-based fertilizers to reduce non-point source pollution caused by agricultural activities.65 In traffic source-dominated areas represented by the UM section, dust control technologies can be used to prevent Pb-containing dust (such as automobile exhaust particles and tire wear debris) from entering rivers through precipitation runoff.66 Systematic governance must strengthen the collaborative control of multiple sources and establish a Pb pollution prevention and control system based on the spatial heterogeneity of the river basin.5 Conclusions
In this study, PMF was used to analyze the source and contribution of heavy metal(loid) pollution in sediments, and combined with MixSIAR and Pb stable isotope tracing technology, the contribution characteristics of multi-source Pb pollution were systematically revealed. The results indicated that Zn, Pb, Cu, Ni, and As all exhibited varying degrees of contamination in the sediments, while the average concentrations of Cr and Mn remained below the local background values. Four major pollution sources were identified by PMF analysis: industrial, agricultural, traffic, and natural sources. Among them, Cd, Pb and Ni were mainly from industrial sources, Pb, As, and Zn were closely related to agricultural sources, Cu and Zn were closely related to traffic sources, and Cr and Mn were closely related to natural sources. The analysis results based on MixSIAR and Pb isotope tracing technology showed that the difference in contribution rates between the MixSIAR model and PMF model was less than 10%, indicating that the analytical results of the two models were highly consistent, which further enhanced the credibility of source analysis. There was a significant correlation between Pb pollution and land use patterns. According to this, pollution prevention and control strategies were proposed for different land use patterns.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study's findings are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5em00509d.
Acknowledgements
This work was supported by the Anhui Provincial Quality Infrastructure Standardization Special Project (2023MKS16), the Natural Science Research Project of the Anhui Provincial Education Commission (2023AH040040, 2024AH040046), and the Ecological Environment Science and Technology Project of Anhui Province (2024hb011).References
- M. M. Ali, M. L. Ali, M. S. Islam and M. Z. Rahman, Preliminary assessment of heavy metals in water and sediment of Karnaphuli River, Bangladesh, Environ. Nanotechnol., Monit. Manage., 2016, 5, 27–35, DOI:10.1016/j.enmm.2016.01.002.
- M. Ao, T. Deng, S. Sun, M. Li, J. Li, T. Liu, B. Yan, W.-S. Liu, G. Wang, D. Jing, Y. Chao, Y. Tang, R. Qiu and S. Wang, Increasing soil Mn abundance promotes the dissolution and oxidation of Cr (III) and increases the accumulation of Cr in rice grains, Environ. Int., 2023, 175, 107939, DOI:10.1016/j.envint.2023.107939.
- S. Audry, J. Schäfer, G. Blanc and J.-M. Jouanneau, Fifty-year sedimentary record of heavy metal pollution (Cd, Zn, Cu, Pb) in the Lot River reservoirs (France), Environ. Pollut., 2004, 132(3), 413–426, DOI:10.1016/j.envpol.2004.05.025.
- J. Bai, B. Cui, B. Chen, K. Zhang, W. Deng, H. Gao and R. Xiao, Spatial distribution and ecological risk assessment of heavy metals in surface sediments from a typical plateau lake wetland, China, Ecol. Modell., 2011, 222(2), 301–306, DOI:10.1016/j.ecolmodel.2009.12.002.
- A. J.-M. Bory, W. Abouchami, S. J. G. Galer, A. Svensson, J. N. Christensen and P. E. Biscaye, A Chinese imprint in insoluble pollutants recently deposited in central Greenland as indicated by lead isotopes, Environ. Sci. Technol., 2014, 48(3), 1451–1457, DOI:10.1021/es4035655.
- L. Chai, Y. Wang, X. Wang, L. Ma, Z. Cheng, L. Su and M. Liu, Quantitative source apportionment of heavy metals in cultivated soil and associated model uncertainty, Ecotoxicol. Environ. Saf., 2021, 215, 112150, DOI:10.1016/j.ecoenv.2021.112150.
- T. Chen, Q. Chang, J. Liu, J. G. P. W. Clevers and L. Kooistra, Identification of soil heavy metal sources and improvement in spatial mapping based on soil spectral information: A case study in northwest China, Sci. Total Environ., 2016, 565, 155–164, DOI:10.1016/j.scitotenv.2016.04.163.
- H. Cheng and Y. Hu, Lead (Pb) isotopic fingerprinting and its applications in lead pollution studies in China: A review, Environ. Pollut., 2010, 158(5), 1134–1146, DOI:10.1016/j.envpol.2009.12.028.
- F. Derakhshan-Babaei, K. Nosrati, D. Tikhomirov, M. Christl, H. Sadough and M. Egli, Relating the spatial variability of chemical weathering and erosion to geological and topographical zones, Geomorphology, 2020, 363, 107235, DOI:10.1016/j.geomorph.2020.107235.
- T.-T. Ding, S.-L. Du, Z.-Y. Huang, J.-C. Wu and D.-H. Yan, Water quality criteria and ecological risk assessment for ammonia in the Shaying River basin, China, Ecotoxicol. Environ. Saf., 2021, 215, 112141, DOI:10.1016/j.ecoenv.2021.112141.
- X. Gao and P. Li, Concentration and fractionation of trace metals in surface sediments of intertidal Bohai Bay, China, Mar. Pollut. Bull., 2012, 64(8), 1529–1536, DOI:10.1016/j.marpolbul.2012.04.026.
- Q. Guan, F. Wang, C. Xu, N. Pan, J. Lin, R. Zhao, Y. Yang and H. Luo, Source apportionment of heavy metals in agricultural soil based on PMF: A case study in Hexi Corridor, northwest China, Chemosphere, 2018, 193, 189–197, DOI:10.1016/j.chemosphere.2017.10.151.
- S. Guo, Y. Zhang, J. Xiao, Q. Zhang, J. Ling, B. Chang and G. Zhao, Assessment of heavy metal content, distribution, and sources in Nansi Lake sediments, China, Environ. Sci. Pollut. Res., 2021, 28(24), 30929–30942, DOI:10.1007/s11356-021-12729-9.
- L. Han, B. Gao, H. Hao, H. Zhou, J. Lu, J. Sun and K. Sun, Lead contamination in sediments in the past 20 years: A challenge for China, Sci. Total Environ., 2018, 640–641, 746–756, DOI:10.1016/j.scitotenv.2018.05.330.
- C. K. Jain, H. Gupta and G. J. Chakrapani, Enrichment and fractionation of heavy metals in bed sediments of River Narmada, India, Environ. Monit. Assess., 2008, 141(1–3), 35–47, DOI:10.1007/s10661-007-9876-y.
- H.-H. Jiang, L.-M. Cai, H.-H. Wen, G.-C. Hu, L.-G. Chen and J. Luo, An integrated approach to quantifying ecological and human health risks from different sources of soil heavy metals, Sci. Total Environ., 2020, 701, 134466, DOI:10.1016/j.scitotenv.2019.134466.
- X. Ke, S. Gui, H. Huang, H. Zhang, C. Wang and W. Guo, Ecological risk assessment and source identification for heavy metals in surface sediment from the Liaohe River protected area, China, Chemosphere, 2017, 175, 473–481, DOI:10.1016/j.chemosphere.2017.02.029.
- P. Kelderman and A. A. Osman, Effect of redox potential on heavy metal binding forms in polluted canal sediments in Delft (The Netherlands), Water Res., 2007, 41(18), 4251–4261, DOI:10.1016/j.watres.2007.05.058.
- C. Li, M. Sun, C. Song, P. Tao, Y. Yin and M. Shao, Assessment of heavy metal contamination in the sediments of the Shuangtaizi Estuary using multivariate statistical techniques, Soil Sediment Contam., 2017, 26(1), 45–58, DOI:10.1080/15320383.2017.1245710.
- F. Li, J. Huang, G. Zeng, X. Yuan, X. Li, J. Liang, X. Wang and X. Tang, Spatial risk assessment and sources identification of heavy metals in surface sediments from the Dongting Lake, middle China, J. Geochem. Explor., 2013, 132, 75–83, DOI:10.1016/j.gexplo.2013.05.007.
- X. Li, H. Liu, W. Meng, N. Liu, P. Wu and B. Bai, Accumulation and source apportionment of heavy metal(loid)s in agricultural soils based on GIS, SOM and PMF: A case study in superposition areas of geochemical anomalies and zinc smelting, southwest China, Process Saf. Environ. Prot., 2022, 159, 964–977, DOI:10.1016/j.psep.2022.01.072.
- H. Liu, Q. Zhang, Y. Wang, Z. Cai and G. Jiang, Occurrence of polychlorinated dibenzo-p-dioxins, dibenzofurans and biphenyls pollution in sediments from the Haihe River and Dagu Drainage River in Tianjin City, China, Chemosphere, 2007, 68(9), 1772–1778, DOI:10.1016/j.chemosphere.2007.03.061.
- J. Liu, Y. Li, B. Zhang, J. Cao, Z. Cao and J. Domagalski, Ecological risk of heavy metals in sediments of the Luan River source water, Ecotoxicology, 2009, 18(6), 748–758, DOI:10.1007/s10646-009-0345-y.
- J. Liu, P. Yin, B. Chen, F. Gao, H. Song and M. Li, Distribution and contamination assessment of heavy metals in surface sediments of the Luanhe River estuary, northwest of the Bohai Sea, Mar. Pollut. Bull., 2016, 109(1), 633–639, DOI:10.1016/j.marpolbul.2016.05.020.
- M. Liu, J. Chen, X. Sun, Z. Hu and D. Fan, Accumulation and transformation of heavy metals in surface sediments from the Yangtze River estuary to the East China Sea shelf, Environ. Pollut., 2019, 245, 111–121, DOI:10.1016/j.envpol.2018.10.128.
- Y. Liu, Y. Yuan, L. Zhang and T. Du, Exploring the differences of moisture traceability methods based on MixSIAR model under different nitrogen applications of wheat in the arid region of northwest China, Agric. Water Manag., 2024, 294, 108716, DOI:10.1016/j.agwat.2024.108716.
- M. Mariyanto, M. F. Amir, W. Utama, A. M. Hamdan, S. Bijaksana, A. Pratama, R. Yunginger and S. Sudarningsih, Heavy metal contents and magnetic properties of surface sediments in volcanic and tropical environment from Brantas River, Jawa Timur Province, Indonesia, Sci. Total Environ., 2019, 675, 632–641, DOI:10.1016/j.scitotenv.2019.04.244.
- A. Müller, H. Österlund, J. Marsalek and M. Viklander, The pollution conveyed by urban runoff: A review of sources, Sci. Total Environ., 2020, 709, 136125, DOI:10.1016/j.scitotenv.2019.136125.
- B. Parinet, A. Lhote and B. Legube, Principal component analysis: An appropriate tool for water quality evaluation and management—application to a tropical lake system, Ecol. Modell., 2004, 178(3–4), 295–311, DOI:10.1016/j.ecolmodel.2004.03.007.
- E. Resongles, C. Casiot, R. Freydier, L. Dezileau, J. Viers and F. Elbaz-Poulichet, Persisting impact of historical mining activity to metal (Pb, Zn, Cd, Tl, Hg) and metalloid (As, Sb) enrichment in sediments of the Gardon River, southern France, Sci. Total Environ., 2014, 481, 509–521, DOI:10.1016/j.scitotenv.2014.02.078.
- N. Reutova, A. Seleznev, I. Yarmoshenko, M. Chervyakovskaya and M. Streletskaya, Stable lead isotopic ratios as indicator of urban geochemical processes, IOP Conf. Ser. Earth Environ. Sci., 2021, 906(1), 012098, DOI:10.1088/1755-1315/906/1/012098.
- C. Rezza, S. Albanese, R. Ayuso, A. Lima, J. Sorvari and B. De Vivo, Geochemical and Pb isotopic characterization of soil, groundwater, human hair, and corn samples from the Domizio Flegreo and Agro Aversano area (Campania region, Italy), J. Geochem. Explor., 2018, 184, 318–332, DOI:10.1016/j.gexplo.2017.01.007.
- R. Setia, S. S. Dhaliwal, V. Kumar, R. Singh, S. S. Kukal and B. Pateriya, Assessment of environmental and toxicity impacts and potential health hazards of heavy metals pollution of agricultural drainage adjacent to industrial zones in Egypt, Chemosphere, 2020, 318, 137872, DOI:10.1016/j.chemosphere.2023.137872.
- B. H. Shomar, Trace elements in major solid-pesticides used in the Gaza Strip, Chemosphere, 2006, 65(5), 898–905, DOI:10.1016/j.chemosphere.2006.03.004.
- K. P. Singh, D. Mohan, V. K. Singh and A. Malik, Studies on distribution and fractionation of heavy metals in Gomti River sediments—A tributary of the Ganges, India, J. Hydrol., 2005, 312(1–4), 14–27, DOI:10.1016/j.jhydrol.2005.01.021.
- I. D. C. Souza, H. P. Arrivabene, C.-A. Craig, A. J. Midwood, B. Thornton, S. T. Matsumoto, M. Elliott, D. A. Wunderlin, M. V. Monferrán and M. N. Fernandes, Interrogating pollution sources in a mangrove food web using multiple stable isotopes, Sci. Total Environ., 2018, 640–641, 501–511, DOI:10.1016/j.scitotenv.2018.05.302.
- Z. Tao, G. Peng, F. Chen, Q. Guo, R. Wei, J. Liu, Y. Li, H. Jiang, Y. Liao and K. Pan, Isotopic signatures unveil the lead sources and migration in surface mangrove sediments, Sci. Total Environ., 2023, 892, 164765, DOI:10.1016/j.scitotenv.2023.164765.
- M. Thévenot, J.-F. Brunel, F. Brunel, M. Briatte, Y. Desplanques and P. Dufrénoy, Identification of particle emission mechanisms during dry friction with thermomechanical operando monitoring, Wear, 2025, 205974, DOI:10.1016/j.wear.2025.205974.
- M. Tsangas, I. Papamichael, D. Banti, P. Samaras and A. A. Zorpas, LCA of municipal wastewater treatment, Chemosphere, 2023, 341, 139952, DOI:10.1016/j.chemosphere.2023.139952.
- M. Varol, Assessment of heavy metal contamination in sediments of the Tigris River (Turkey) using pollution indices and multivariate statistical techniques, J. Hazard. Mater., 2011, 195, 355–364, DOI:10.1016/j.jhazmat.2011.08.051.
- B. Wang, D. Xia, Y. Yu, J. Jia, Y. Nie and X. Wang, Detecting the sensitivity of magnetic response on different pollution sources – a case study from typical mining cities in northwestern China, Environ. Pollut., 2015, 207, 288–298, DOI:10.1016/j.envpol.2015.08.041.
- L. Wang, S. Gao, X. Yin, X. Yu and L. Luan, Arsenic accumulation, distribution and source analysis of rice in a typical growing area in north China, Ecotoxicol. Environ. Saf., 2019, 167, 429–434, DOI:10.1016/j.ecoenv.2018.10.015.
- S.-L. Wang, X.-R. Xu, Y.-X. Sun, J.-L. Liu and H.-B. Li, Heavy metal pollution in coastal areas of south China: A review, Mar. Pollut. Bull., 2013, 76(1–2), 7–15, DOI:10.1016/j.marpolbul.2013.08.025.
- R. Wei, F. Ge, S. Huang, M. Chen and R. Wang, Occurrence of veterinary antibiotics in animal wastewater and surface water around farms in Jiangsu province, China, Chemosphere, 2011, 82(10), 1408–1414, DOI:10.1016/j.chemosphere.2010.11.067.
- X. Yao, Z. Wang, D. Li, H. Sun, C. Ren, Y. Yu, F. Pei and Y. Li, Distribution, mobilization, risk assessment and source identification of heavy metals and surface sediments of three urban-rural rivers after long-term water pollution treatment, Sci. Total Environ., 2024, 932, 172894, DOI:10.1016/j.scitotenv.2024.172894.
- L. Yu, T. Zheng, X. Zheng, Y. Yuan and R. Yuan, Nitrate source apportionment in groundwater using Bayesian isotope mixing model based on nitrogen isotope fractionation, Sci. Total Environ., 2020, 718, 137242, DOI:10.1016/j.scitotenv.2020.137242.
- C. Zhang, Q. Qiao, J. D. A. Piper and B. Huang, Assessment of heavy metal pollution from a Fe-smelting plant in urban river sediments using environmental magnetic and geochemical methods, Environ. Pollut., 2011, 159(10), 3057–3070, DOI:10.1016/j.envpol.2011.04.006.
- C. Zhang, W. Rao, Z. Wu, F. Zheng, T. Li, C. Li, X. Lei, H. Xie and X. Chu, Anthropogenic impacts and quantitative sources of nitrate in a rural-urban canal using a combined PMF, δ15N/δ18O–NO3-, and MixSIAR approach, Environ. Res., 2024, 251, 118587, DOI:10.1016/j.envres.2024.118587.
- Z. Zhang, Y. Lu, H. Li, Y. Tu, B. Liu and Z. Yang, Assessment of heavy metal contamination, distribution and source identification in the sediments from the Zijiang River, China, Sci. Total Environ., 2018, 645, 235–243, DOI:10.1016/j.scitotenv.2018.07.026.
- K. Zhao, K. Bao, Y. Yan, Y. Zhang, H. Huang, L. Li and W. Jia, Spatial distribution of potentially harmful trace elements and ecological risk assessment in Zhanjiang mangrove wetland, south China, Mar. Pollut. Bull., 2022, 182, 114033, DOI:10.1016/j.marpolbul.2022.114033.
- Z. Zhao, M. Zhang, Y. Chen, Y. Yu, Y. Wang, F. Li, J. Liu, L. Zhang and W. Zhai, Traceability of nitrate polluted hotspots in plain river networks of the Yangtze River delta by nitrogen and oxygen isotopes coupling Bayesian model, Environ. Pollut., 2022, 315, 120438, DOI:10.1016/j.envpol.2022.120438.
- Z.-Q. Zhao, W. Zhang, X.-D. Li, Z. Yang, H.-Y. Zheng, H. Ding, Q.-L. Wang, J. Xiao and P.-Q. Fu, Atmospheric lead in urban Guiyang, southwest China: Isotopic source signatures, Atmos. Environ., 2015, 115, 163–169, DOI:10.1016/j.atmosenv.2015.05.049.
- W. Zhou, R. Yu, F. Guo, C. Shen, Y. Liu and Y. Huang, Source apportionment and risk assessment of soil heavy metals in the Huangshui River basin using a hybrid model, Ecol. Indic., 2024, 160, 111906, DOI:10.1016/j.ecolind.2024.111906.
- A. Zhu, J. Liu, S. Qiao and H. Zhang, Distribution and assessment of heavy metals in surface sediments from the Bohai Sea of China, Mar. Pollut. Bull., 2020, 153, 110901, DOI:10.1016/j.marpolbul.2020.110901.
- Y. B. Guan, C. F. Li, X. M. Cheng, Y. Ba and L. X. Cui, Pollution status and ecological risk assessment of a section of the Shaying River basin, J. Environ. Occup. Med., 2013, 30(11), 825–828, DOI:10.13213/j.cnki.jeom.2013.11.002.
- M. Zhong, Y. Wan, A. Wan, J. Hu, S. An and X. Leng, Characteristics and ecological risk assessment of heavy metal pollution in sediments of the Shaying River basin, Chin. J. Ecol., 2016, 35(7), 1857–1864, DOI:10.13292/j.1000-4890.201607.008.
- G. Müller, Index of geoaccumulation in sediments of the Rhine River, GeoJ., 1969, 2, 108–118 Search PubMed.
- Y. Q. Ji, Using geoaccumulation index to study source profiles of soil dust in China, J. Environ. Sci., 2008, 20(5), 571–578 CrossRef CAS PubMed.
- H. Zhao, Y. Wu, X. Lan, Y. Yang, W. Zhang, S. Xie and L. Wu, Comprehensive assessment of harmful heavy metals in contaminated soil in order to score pollution level, Sci. Rep., 2022, 12, 3552, DOI:10.1038/s41598-022-07602-9.
- R. S. Ahmed, M. E. Abuarab, M. M. Ibrahim, M. Baioumy and A. Mokhtar, Assessment of environmental and toxicity impacts and potential health hazards of heavy metals pollution of agricultural drainage adjacent to industrial zones in Egypt, Chemosphere, 2023, 318, 137872, DOI:10.1016/j.chemosphere.2023.137872.
- M. Anas, W. A. Khattak, W. Sarfraz, N. Khalid, N. Naeem, M. Farid, M. H. Saleem, K. A. Khan, A. Khalid, T. Ahmed and S. Fahad, EDTA-enhanced phytoextraction: Leveraging Ricinus communis for the decontamination of Pb and Cd in agricultural soils, Plant Growth Regul., 2025, 105(1), 215–228, DOI:10.1007/s10725-024-01270-7.
- H. Dong, Y. Xun, M. Yoneda and L. Feng, Risk assessment of complex contaminated sites based on the fractional transformation of metals: Model development and a case study, Sci. Total Environ., 2021, 786, 147509, DOI:10.1016/j.scitotenv.2021.147509.
- Z. Iannuzzi, M. C. Casado-Martinez, T. De Oliveira, D. Lebouil, B. J. D. Ferrari, J. Gasperi and B. Mourier, Tire road wear particles, microplastics and metals in sediment of stormwater detention basins: Co-occurrence and ecological risk assessment, Environ. Int., 2025, 202, 109676, DOI:10.1016/j.envint.2025.109676.
- H. Luo, L. Yang, C. Zhang, X. Xiao and X. Lyu, Early warning of heavy metals contamination in agricultural soils: Spatio-temporal distribution and future trends in the Hexi Corridor, Ecol. Indic., 2024, 160, 111908, DOI:10.1016/j.ecolind.2024.111908.
- Y. Pan, L. Ding, S. Xie, M. Zeng, J. Zhang and H. Peng, Spatiotemporal simulation, early warning, and policy recommendations of the soil heavy metal environmental capacity of the agricultural land in a typical industrial city in China: Case of Zhongshan City, J. Cleaner Prod., 2021, 285, 124849, DOI:10.1016/j.jclepro.2020.124849.
- S. A. Shetaia, M. Li, J. Chen, Y. Wang, M. S. Elhebiry, A. Abdelaal, H. Su, Z. Chen and A. Salem, Comprehensive assessment of remarkable heavy metals contamination in road dust in Greater Cairo: Priority control factors based on source apportionment and risk assessment, J. Environ. Manage., 2025, 394, 127607, DOI:10.1016/j.jenvman.2025.127607.
| This journal is © The Royal Society of Chemistry 2026 |