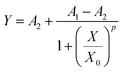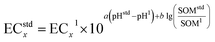Ecological safety thresholds for phenanthrene in Chinese soils: implications for assessing ecological risks to vegetation and for land use
Jiahui
Zhu†
,
Qian
Yang†
,
Jiawei
Wang
,
Xuke
Wang
,
Shuilin
Zhu
and
Xinhua
Zhan
 *
*
College of Resources and Environmental Sciences, Nanjing Agricultural University, Nanjing, Jiangsu Province 210095, People's Republic of China. E-mail: xhzhan@njau.edu.cn; Tel: +86-25-84395210
First published on 12th December 2025
Abstract
Phenanthrene poses carcinogenic, teratogenic and mutagenic risks to plants and soil invertebrates. However, the absence of established soil ecological safety thresholds of phenanthrene has resulted in insufficient evidence in the current risk assessment for soil ecological security. To fill this gap, toxicity data from laboratory experiments and the existing literature, covering 16 plant species, 7 invertebrates, and 3 soil ecological processes, were applied to the species sensitivity distribution approach to determine the soil ecological safety thresholds of phenanthrene across different land types. From experimental results, we found that the effect concentration at 10% values for most plants had a positive correlation with pH, soil organic matter content, cation exchange capacity, and electrical conductivity values. The soil ecological safety thresholds of phenanthrene were estimated to be 7 mg kg−1 for agricultural and forestry land with the hazardous concentration for 5% of the species affected (HC5), 35 mg kg−1 for green spaces and squares with HC20, 95 mg kg−1 for residential land with HC40, and 122 mg kg−1 for commercial and industrial land with HC50, respectively. These findings will serve as a foundation for the ecological risk assessment of phenanthrene on land for different purposes, and are of great significance for ecological species protection.
Environmental significanceEstablishing ecological safety thresholds for PAHs in soil ecosystems is crucial for improving contamination management, conserving biodiversity, maintaining soil health, and supporting environmental protection policies. In our study, we established new ecological safety thresholds for soil PHE, and the ecological safety thresholds of PHE in different land use types were selected: HC5 (agricultural and forestry land, 7 mg kg−1), HC20 (green spaces and squares, 35 mg kg−1), HC40 (residential land, 95 mg kg−1), and HC50 (commercial and industrial land, 122 mg kg−1). This research provides a valuable reference material for further exploration and use by regulators and scientists. |
1 Introduction
Polycyclic aromatic hydrocarbons (PAHs) are a class of complex organic compounds composed of carbon and hydrogen, featuring at least two fused aromatic rings.1,2 Due to their mutagenic, carcinogenic, and highly ecotoxic effects on humans and animals, 16 individual PAHs (∑16PAHs) have been designated as priority control contaminants by the United States Environmental Protection Agency, while the International Agency for Research on Cancer has classified 7 isomers (∑7cPAHs) as mutagenic and carcinogenic pollutants.3 Soil exhibits a strong affinity and capacity to accumulate PAHs, making itself the primary reservoir or sink for PAHs in terrestrial environments.4,5 Phenanthrene (PHE), a typical low-molecular-weight PAH often used as a model compound for studying PAH catabolism, is suspected to be an ultimate carcinogen due to its molecular structure that enables the formation of epoxides.6,7 Present in the environment and characterized by their hydrophobicity and resistance to degradation, PHE poses carcinogenic, teratogenic, and mutagenic risks to plants and soil invertebrates.8–10 Hence, building standards of PHE ecological safety thresholds in soil ecosystems can profoundly enhance the management of PHE contamination, conservation of biodiversity, and preservation of soil health and provide support for environmental protection policies.To date, more than 39 nations have guidance values for soil PAH contamination.11 Despite widespread regulation of PAHs, current soil standards remain limited for PHE from an ecological-protection perspective. For example, the Netherlands Environmental Quality Objectives (EQOs) for PAHs were derived in the 1990s, and are widely cited but predate more recent species sensitivity distribution (SSD) practices and newer ecotoxicity datasets, and the set maximum permissible concentration for PHE is 0.51 mg kg−1.12 Canadian guidelines similarly emphasize carcinogenic benzo[a]pyrene (1.5 mg kg−1) for soil management and comparison across jurisdictions.13 In China, national standards like the Control Values for Soil Pollution at Building Sites (GB 36600-2018) focuses on human-health risk for development land rather than PHE-specific ecological thresholds.14 Collectively, these approaches often under-represent soil-property effects (e.g., soil organic matter, and pH) on PAH bioavailability and lack PHE-targeted ecological benchmarks.
Currently, various statistical methods such as empirical linear regression, correlation analysis, and path analysis can be used to quantitatively analyze the relationships between inherent soil properties and contaminant toxicity.15 Modern SSD methods combine multi-species toxicity data to derive protective hazardous concentrations.16,17 In ecological risk assessment, the protection level is commonly determined by the percentage of safeguarded species indicated by the SSD curve, with the hazardous concentration for 5% of the species affected (HC5) typically used as the threshold to denote the concentration that protects 95% of species.15 Existing studies have primarily relied on traditional endpoints such as survival, development, and growth to construct the SSD for PHE.18 Lots of distribution models such as the log-normal, log-logistic, Burr III and Weibull distribution are used to fit the SSD,19 and the choice of the best-fit model should be selected by using the goodness-of-fit test.
In this study, site-specific ecological safety thresholds for PHE contamination in soil were determined through integrated analysis of multiple online data sources and experimental results. We selected 26 species/soil ecological processes, including 16 plants, 7 invertebrate and 3 soil ecological processes to carry out the ecological safety thresholds (HC5, HC20, HC40 and HC50) by the SSD method. The findings of this study will offer both data and methodological support to governments and environmental protection agencies in developing scientific and rational soil standards.
2 Materials and methods
2.1 Materials and chemicals
Soil samples (0–20 cm) were collected from four different locations: (1) NUS: upland soil from Meishan Family Farm in Meixi Village, Hengxi Town, Yinzhou District, Ningbo City, Zhejiang Province (121.56°, 29.70°). (2) NPS: paddy soil from Mengjiadai, Jia Village, Hengxi Town, Yinzhou District, Ningbo City, Zhejiang Province (121.57°, 29.74°). (3) YUS: upland red soil from Shangzhang Village, Liujiastation Subfarm, Yiyang County, Yingtan City, Jiangxi Province (116.94°, 28.21°). (4) YPS: paddy soil from the same location in Jiangxi Province. All soils were dried and sieved through a 2 mm (10-mesh) sieve to remove debris and ensure uniformity. PHE (purity >97%) was purchased from Fluka Chemical Co. Ltd. Acetone (purity >99.5%) was purchased from Shanghai Lingfeng Chemical Reagent Co., LTD.Wheat (Triticum aestivum L.), rice (Oryza sativa L.), soybean (Glycine max), tomato (Solanum lycopersicum L.), onion (Allium cepa L.), lettuce (Lactuca sativa), oilseed rape (Brassica campestris L.), cucumber (Cucumis sativus L.), brilliant campion (Lychnis fulgens Fisch.), morning glory (Ipomoea nil), clover (Trifolium repens L.), motherwort (Leonurus japonicus) and corn poppy (Papaver rhoeas L.) were used as subjects. These plants were commonly found in the field. All the seeds were purchased from Jiangsu Academy of Agricultural Sciences seed station and seeds selling markets.
2.2 Experiment design
To determine the ecological threshold of phenanthrene, we tested both environmentally relevant low concentrations11,20 and higher levels representative of accidental leaks or local hotspots. In this study, all the soils were spiked with PHE (dissolved in acetone) at concentrations of 0, 50, 100, 500, 1000, 2000 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg kg−1. After incubation for 1 month at a water content of around 70% (w/w) of field capacity, the soils were air-dried, sieved, and mixed again before the toxicity experiment.
000 mg kg−1. After incubation for 1 month at a water content of around 70% (w/w) of field capacity, the soils were air-dried, sieved, and mixed again before the toxicity experiment.
The pot experiment was designed to investigate the toxic effects of PHE-contaminated soil on plants. Following flotation in deionized water to remove hollow seeds, the remaining uniform and healthy plant seeds were selected. After disinfection for 5 min using 3% (v/v) H2O2, the seeds were thoroughly rinsed and soaked in a beaker for 24 hours, followed by germination in an enamel tray lined with absorbent paper moistened with deionized water in darkness at 25 °C in a growth chamber. The experiment was conducted in small plastic pots with an internal diameter of 8.2 cm and a volume of 0.25 L. The inner surface of each pot was lined with aluminum foil to reduce PHE adsorption by the plastic. After preparing soils contaminated with different concentrations of PHE, the soil samples were moistened with deionized water to reach 60% of their water-holding capacity. The soil moisture was adjusted daily by weighing a random sample of pots. Each pot contained 90 g of soil, and 10 seeds were sown per pot for most crops, while larger-seeded crops such as soybeans had 6 seeds sown per pot. Three replicates were performed for each concentration, and the seeds were cultivated for 7 days (post germination to harvest). Cultivation conditions in the growth chamber are as follows: a cycle of 16 h light/8 h darkness, with a daytime temperature of 25 °C, a nighttime temperature of 20 °C, a light intensity of 400 µmol (s m2)−1, and a relative humidity of 75% (Fig. S23).
2.3 Determination of the PHE concentration in soil
In this experiment, concentration dilution is required when measuring the PHE concentration. Then, 0.5 g soil samples were weighed into a 50 mL glass centrifuge tube with 10 mL of the solution (acetone![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) dichloromethane = 1
dichloromethane = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). The sample was ultrasonically extracted for 30 min (with ice cubes) and passed through the column with anhydrous sodium sulfate, and silica gel chromatography was performed. The extraction process was repeated three times. Then, the column was rinsed with 10 mL of a mixture (n-hexane
1, v/v). The sample was ultrasonically extracted for 30 min (with ice cubes) and passed through the column with anhydrous sodium sulfate, and silica gel chromatography was performed. The extraction process was repeated three times. Then, the column was rinsed with 10 mL of a mixture (n-hexane![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) dichloromethane = 1
dichloromethane = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). After the silica gel column dried, the collected extracts and eluent were combined in a 50 mL ground-neck conical flask and dried at 42 °C using a vacuum rotary evaporator. Finally, the residue was redissolved to 1.5 mL chromatographic-grade methanol, filtered through a 0.22 µm membrane, and transferred to a 2 mL sampling vial for further analysis. The recovery rate of PHE using this method was within 88.95–90.88%.
1, v/v). After the silica gel column dried, the collected extracts and eluent were combined in a 50 mL ground-neck conical flask and dried at 42 °C using a vacuum rotary evaporator. Finally, the residue was redissolved to 1.5 mL chromatographic-grade methanol, filtered through a 0.22 µm membrane, and transferred to a 2 mL sampling vial for further analysis. The recovery rate of PHE using this method was within 88.95–90.88%.
The concentration of PHE in soil was determined using a high-performance liquid chromatography (HPLC) system, following the method described by our previous work.8 The HPLC system used was a Thermo Scientific Dionex UltiMate 3000 Series, equipped with an automatic injector (WPS-3000SL), a quaternary analytical pump (LPG-3400SDN), a rapid separation thermostated column compartment (TCC-3000RS), and a variable wavelength detector (VWD-3100). Separation was achieved using a reverse-phase Symmetry C18 column (4.6 × 150 mm, 5 µm particle size). The column temperature was maintained at 30 °C. The mobile phase consisted of methanol and water (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, v/v), with a flow rate of 1.0 mL min−1 and an injection volume of 10 µL. The UV detection wavelength for PHE was set at 254 nm. Analytical standards were run at the start of each series of analyses.
20, v/v), with a flow rate of 1.0 mL min−1 and an injection volume of 10 µL. The UV detection wavelength for PHE was set at 254 nm. Analytical standards were run at the start of each series of analyses.
2.4 Determination of soil water capacity
The ring knife method was used to determine the maximum water-holding capacity of four types of soils.21 A 9 cm diameter filter paper was placed at the bottom of the ring knife and secured with a rubber band. Approximately 20 g soils were weighed into the ring knife, with three replicates for each soil sample. Water was continuously added to keep the soil moist, and after stabilizing for 8 hours to reach a constant weight, the ring knife was removed, and the filter paper was discarded. The soil was then placed in a pre-weighed aluminum box and weighed. It was then placed in an oven at 105 °C for 12 hours until a constant weight was reached, after which it was weighed again.2.5 Determination of soil physicochemical properties
The pH and electrical conductivity (EC) were measured in purified water in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 (w/v) and 1
2.5 (w/v) and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ׃
׃![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (w/v) soil–water suspensions, respectively. SOM was measured by a dichromate method for the <100-mesh fraction. Total inorganic carbon was determined using the high-temperature loss-on-ignition method after the addition of HCl to remove carbonates on the 20-mesh (0.85 mm) fraction (Leco TruMac CNS analyzer, USA). The soil particle size distributions were analyzed using a laser diffraction particle size analyzer (Malvern Mastersizer 2000, UK).
5 (w/v) soil–water suspensions, respectively. SOM was measured by a dichromate method for the <100-mesh fraction. Total inorganic carbon was determined using the high-temperature loss-on-ignition method after the addition of HCl to remove carbonates on the 20-mesh (0.85 mm) fraction (Leco TruMac CNS analyzer, USA). The soil particle size distributions were analyzed using a laser diffraction particle size analyzer (Malvern Mastersizer 2000, UK).
2.6 Plant analysis and dose–response curve fitting
After 7-day culture, all the seedlings were carefully removed from the pots. Then, all the seedlings were rinsed at least three times with deionized water to remove the root surface soil. A vernier caliper was used to measure the root length and stem length, which were recorded. The stem length is defined as the distance from the base of the stem to the midpoint of the leaves, and the root length is defined as the distance from the base of the stem to the longest root (in cm).The total fresh weight of the plant tissues in each pot was measured using an analytical balance. The plant stem length was measured using a ruler. The root length inhibition rate was determined as (root length of the control group − root length of the treatment group)/root length of the control group. The germination index was calculated as follows (germination rate of the treatment group × root length of the treatment group)/(germination rate of the control group × root length of the control group). Each pot contains at least 3 plants in the plant analysis.
The dose–response data were fitted by logistic distribution as follows:
2.7 Screening and normalization of toxicological data for PHE ecotoxicity
The toxicity data of PHE in soil were searched in the Web of Science. Literature collection was employed based on the requirements in Text S1 and Table S1. Eventually, toxicity data of 15 species (or soil microorganism biochemical reaction and enzyme activity) were obtained.Before fitting the distribution curve of toxicity thresholds (preferably using EC10) for different ecological receptors or ecological processes using the Species Sensitivity Distribution (SSD) method, it is necessary to normalize the ecological toxicity effect parameters of the same species or the same variety under different soil conditions to the same soil properties (pH = 6.5, SOM = 20 g kg−1, or CEC = 20 cmol kg−1). The data should be normalized using the following equation:
2.8 SSD construction and HCx value determination
For SSD fitting, we combined our experimental results with toxicity data extracted from published studies, and the aggregated dataset spans multiple reference soil groups—including sandy soil, silt soil, and loam—and sampling locations both within China and internationally. Based on the National Ecological Environment Criteria Calculation Software – Species Sensitivity Distribution Method (EEC-SSD), the SSD curves and HCx values were estimated using logistic distribution. In our study, logistic distribution showed higher goodness of fitting (Table S7).2.9 Statistical analysis
The EEC-SSD software was used for toxicity fitting. Correlation analysis and one-way analysis of variance (ANOVA) followed by Duncan's multiple comparison test were performed using IBM SPSS 27.0. Line charts, dose–response curve fitting and multiple linear regression were conducted using Origin 2021.3 Results and discussion
3.1 Effects of PHE on plant growth
Plant total fresh weight, root length inhibition rate, stem length and germination index were determined after culturing in PHE-polluted soil (Fig. S1–S16). Briefly, plant fresh weights were inversely proportional to the PHE concentrations in the soil (Fig. S1–S4). Furthermore, the stress of PHE on certain plant was influenced by soil type. In paddy soils, the fresh weight of rice exhibited a low-dose stimulation and high-dose inhibition pattern with the increase in PHE concentrations, whereas in upland soils, it showed an inverse relationship with PHE concentrations. The effect of PHE on root length varied among plant species (Fig. S5–S8). In paddy soils, the root length inhibition rate of most plants first decreased and then increased with higher PHE concentrations. However, in upland soils, the root length inhibition rates of most plants were proportionate to the increased PHE concentration. The plant stem length was a typical index for observing the stress of PHE on plant growth (Fig. S9–S12). Almost all the stem lengths of plants showed a negative correlation with the PHE concentration. It was obviously that the germination index and root length inhibition rate had a negative correlation (Fig. S13–S16). In general, the germination rate of most plants decreased with the increase in PHE concentrations. However, the changes in the germination rate were also affected by the plant species and soil type. In summary, fresh weight and stem length better reflected the characteristics of plants under PHE stress in the soil.The plant fresh weight and stem length were the important indexes for assessing the plant growth in the pollution site. Some studies indicated that PAHs can decrease plant (such as barley, broad bean, lettuce, and Chinese cabbage) growth because the PAHs may cause damages to the cell membrane and organelles.22–25 In our study, most of the plant fresh weight and stem length were inhibited by PHE and the inhibition was positively correlated with the PHE concentration, indicating that fresh weight and stem length should be visualized indexes for observing the PHE toxicity to the plants. Roots are in direct contact with contaminants, making themselves more susceptible to damage. The germination index was correlated with the root length. In our study, root-correlated indexes were not uniform as some plant roots had low sensitivity to PHE. Previous studies have also found that PAHs can inhibit the root growth of wheat seedling/lawn grasses.23,26 However, some plant roots extended into uncontaminated areas to cope with PHE stress (such as lettuce). One possible reason was that when roots first come into contact with PHE, it may stimulate root growth, especially the new one.27 Another potential reason may be the short growth period and minimal individual variation of plants during the early developmental stages, as well as differences among plant species.28
3.2 Toxicity of PHE on plant growth factor models
Since the changes in fresh weight and stem length of plants during the experiment were more stable and the toxic effects were more pronounced under PHE contamination, this study established EC10 toxicity effect curves based on fresh weight and stem length to determine plant toxicity thresholds. The logistic model was used to fit the dose–response curves of the results. The dose–response relationship fitting curves based on the fresh weight/stem length for the four types of soils (Table 1) were investigated in this study (Fig. 1, 2 and S17–S22). Generally, compared to the fresh weight, the stem length showed better fit quality in the curve fitting process. When using stem length as the indicator for curve fitting, the logistic model provided better results for most plants except for onion in Yingtan upland soil. | ||
| Fig. 1 Dose–response relationship curves between phenanthrene concentration and plant fresh weight in Ningbo paddy soil. FW: fresh weight; PHE: phenanthrene. | ||
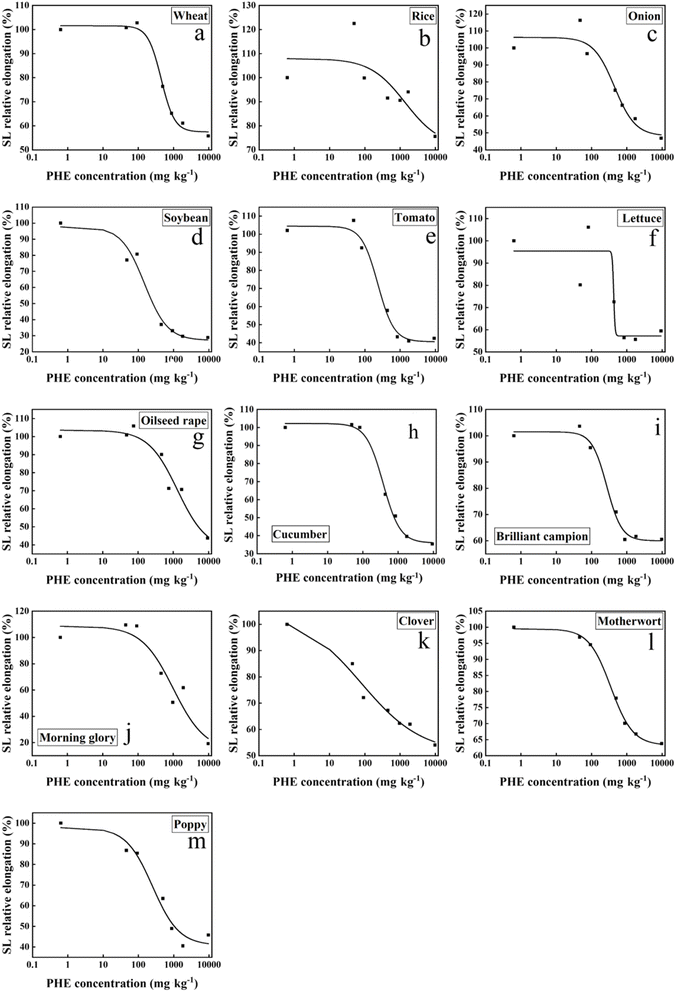 | ||
| Fig. 2 Dose–response relationship curves between phenanthrene concentration and plant stem length in Ningbo paddy soil. SL, stem length; PHE, phenanthrene. | ||
| Physiochemical properties | Ningbo paddy soil | Ningbo upland soil | Yingtan paddy soil | Yingtan upland soil |
|---|---|---|---|---|
| a TIC, total inorganic carbon; TN, total, nitrogen; SOM, soil organic matter; EC, electrical conductivity; CEC, cation exchange capacity. | ||||
| pH | 5.40 | 4.46 | 4.74 | 4.55 |
| CEC (cmol kg−1) | 19.12 | 10.95 | 12.81 | 11.73 |
| SOM (g kg−1) | 79.40 | 31.09 | 19.59 | 15.48 |
| EC (µS cm−1) | 155.50 | 40.41 | 77.37 | 43.72 |
| TN (g kg−1) | 5.47 | 1.51 | 1.13 | 1.02 |
| TIC (g kg−1) | 0.71 | 0.80 | 0.65 | 0.53 |
| Sand (%) | 37.2 | 43.1 | 35.6 | 25.2 |
| Silt (%) | 43.5 | 26.3 | 27.3 | 25.2 |
| Clay (%) | 19.3 | 30.6 | 37.1 | 49.6 |
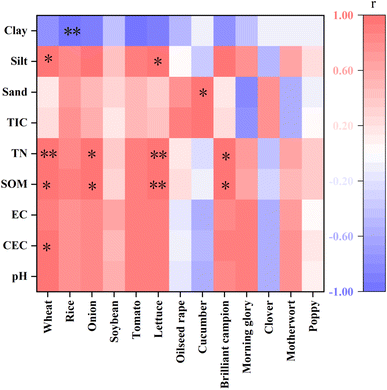
A comparison of EC10 values revealed that EC10 for stem length was lower than that for fresh weight, indicating that the stem length was more sensitive to PHE contamination in soil (Table S1). There were significant differences in EC10 values for the same plant across different soils, for example, when using fresh weight as an indicator to fit the dose–response curve for rice, the EC10 values ranged from 49.00 mg kg−1 to 440.57 mg kg−1, with the maximum being 8.99 times higher than the minimum (Table 2). In contrast, when the stem length was used as the indicator to fit the dose–response curve, the EC10 values ranged from 5.04 mg kg−1 to 111.87 mg kg−1, showing a 22-fold difference. This phenomenon suggested that the impact of exogenous PHE on plant growth was closely related to the physicochemical properties of the soil. Hence, it is necessary to analyze the relationship between soil physicochemical properties and the ecotoxicity data of organic pollutants. Using IBM SPSS 27.0 for Pearson's correlation analysis, a significant relationship was identified between soil properties and the toxicity threshold EC10. A higher absolute value of the correlation coefficient between soil properties and plant EC10 indicates a stronger correlation. Among the 13 plants studied, wheat showed the highest correlation coefficient between EC10 and soil pH, reaching 0.943 (Fig. 3). The toxicity thresholds of PHE for wheat, onion, lettuce, and catchfly were positively correlated with the soil organic matter content. For most plants, except cucumber and clover, the EC10 values of PHE toxicity also exhibited positive correlations with the soil pH, CEC, and EC values. The Ningbo paddy soil had the highest pH (5.40), CEC (19.12 cmol kg−1), soil organic matter content (79.40 g kg−1), and EC (155.50 µS cm−1) among the four soil types; correspondingly, the EC10 value for plants was also higher than that of the other three soils.
| Species | Fresh weight (mg kg−1) | Stem length (cm) |
|---|---|---|
| EC10 | EC10 | |
| a ND indicates that EC10 cannot be derived by logistic curve fitting. | ||
| Wheat | 213.78–859.78 | 6.22–170.72 |
| Rice | 49.00–440.57 | 5.04–111.87 |
| Onion | 0.94–194.28 | 7.70–98.39 |
| Soybean | 45.29–316.75 | 8.81–36.29 |
| Tomato | 11.73–31.66 | 0.64–72.99 |
| Lettuce | 9.24–578.14 | 0.62–383.48 |
| Oilseed rape | 5.07–255.35 | 5.31–406.47 |
| Cucumber | 49.90–171.15 | 39.30–330.97 |
| Brilliant campion | 0.018–444.73 | 1.20–92.38 |
| Morning glory | 5.91–289.44 | 19.75–101.55 |
| Clover | 11.13-ND | 0.97–23.77 |
| Motherwort | 2.65–49.19 | 3.44–72.03 |
| Poppy | 4.52–16.88 | 10.00–39.05 |
The physicochemical properties of soil can influence the bioavailability of PAHs, thereby affecting their toxicity to plants, with soil organic matter being an important influencing factor.29 PAH adsorption in soil can be enhanced by soil organic matters, leading to a potential risk to the plants.30 For example, sand with a lower soil organic matter concentration had a higher PAH degradation rate, but a lower adsorption capacity for PAHs than soil.31 Similarly, previous study found that the EC50 value of PAHs in black soil was three times that in red soil, which is attributed to the higher soil organic matter content in black soil.32 The finding is consistent with our results. The pH is a key factor in microbial degradation of organic pollutants, which extremely affects the soil fertility, plant growth, and microbial activity, leading to nutrient deficiencies. Studies have shown that in PAH toxicity tests on lettuce, a lower soil pH value leads to slower PAH degradation, resulting in higher accumulation of PAHs in organisms and exerting pressure on terrestrial organism growth.32–34 In addition, other physicochemical properties of soil, such as CEC and EC, can significantly influence the ecological toxicity of PAHs.35,36 The CEC value mainly depends on the content of clay and soil organic matter in the soil; higher CEC values indicate greater soil fertility.
3.3 Ecological safety threshold for PHE
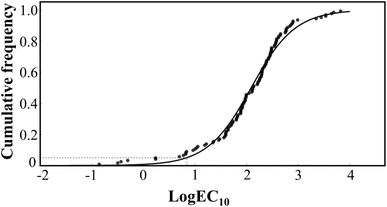
To improve the accuracy of the fitting model, it is necessary to increase the toxicity data of other species in the database. The toxicity data screening principle was shown in SI (Text S1). After reliability evaluation, a total of 17 toxicity data points were deemed suitable for fitting. In combination with the experiment results, the toxicity data used for fitting covered a total of 26 species (Table S2), including 16 terrestrial plant species, 7 terrestrial invertebrate species, and 3 soil microbial species or microbial-driven soil ecological processes. The details are shown in Tables S3–S5 in the SI. Three equations were derived by the toxicological data available from the lab experiments and literature (Table S6). By comparing the goodness of fit, the logistic equation was found to provide the best fitting performance (Table S7). Typically, prior to using the SSD method to model the distribution curves of toxicity effect parameters for various ecological receptors or processes, it is necessary to normalize the ecotoxicity effect parameters of the same species across different soil conditions to a uniform soil property baseline.32 Therefore, the EC10 values for species were adjusted to correspond to a standard soil condition (pH = 6.5, SOM = 20 g kg−1, or CEC = 20 cmol kg−1), and the hazardous concentrations (HCx) of PHE in the standard soil were subsequently calculated (Fig. 4). Based on the SSD curve, the estimated HC5, HC20, HC40 and HC50 values for standard soil were 7, 35, 95 and 122 mg kg−1, respectively (Table 3).| Land types | Hazardous concentration | Ecological safety threshold (mg kg−1) |
|---|---|---|
| a HC5: hazardous concentrations for 5% of the species affected; HC20: hazardous concentrations for 20% of the species affected; HC40: hazardous concentrations for 40% of the species affected; HC50: hazardous concentrations for 50% of the species affected. | ||
| Agricultural and forestry land | HC5 | 7 |
| Green space and square land | HC20 | 35 |
| Residential land | HC40 | 95 |
| Commercial and industrial land | HC50 | 122 |
Through the data, we can see that different species tested in various soils showed varying sensitivities to PHE toxicity, and the EC10 values obtained from different text indexes for the same species varied considerably (Tables S3–S5). Therefore, the toxicity data obtained in this study differed in the selection of test indexes for each species, as certain toxicity indicators are known to more effectively represent the ecotoxicity of PHE when determining ecological safety thresholds. The SSD method has proven to be a reliable approach for establishing ecological safety thresholds.37 In SSD interpretation, HCx is typically defined to provide a protective effect for the x% of the group.32 It is crucial to understand that the HCx values calculated from the SSD curve do not indicate that x% species are “sacrificed,” with the remaining (1−x)% being safeguarded.38 SSD curves are typically fitted using distributions such as Burr type III, logistic, normal, gamma, and Gompertz, with the model selection based on the data fitting accuracy,19,39 and the logistic model was used to fit the SSD curves in our study. Land use planning requires balancing ecological protection with land utilization efficiency, and the hierarchical HC value setting aligns with land management policies, serving as a basis for establishing soil quality standards.15,32,40 In China, the Code for Classification of Urban Land Use and Planning Standards of Development Land (GB 50137-2011) partitions land into residential/public green/open space versus commercial/industrial (among others), which provides the policy basis for differentiated environmental targets; a single phenanthrene threshold is therefore inappropriate. Accordingly, we provide a suite of SSD-based thresholds and map them to land use: HC5 (95% protection) for agricultural and forestry land, HC20 (80% protection) for green spaces and squares, HC40 (60% protection) for residential areas, and HC50 (50% protection) for commercial and industrial zones. Existing literature reveals that few studies have employed the SSD method to establish the environmental threshold value of PHE in soil, and there is a lack of comprehensive toxicity data for PHE. Consequently, this paper represents the first use of the SSD approach to define the soil environmental criteria for PHE.
Conclusions
Based on 26 species/soil ecological processes (including 16 plants, 7 invertebrate and 3 soil ecological processes), new ecological safety thresholds for soil PHE were proposed. Through the experiment results, we found that the EC10 values of most plants were positively correlated with pH, soil organic matter content, CEC, and EC values. The EC10 value of PHE that we used to fit the SSD curve ranged from 0.018 to 578.14 mg kg−1, including the experimental results and the toxicity data searched from the literature. Last, the HC5 (agricultural and forestry land, 7 mg kg−1), HC20 (green spaces and squares, 35 mg kg−1), HC40 (residential land, 95 mg kg−1) and HC50 (commercial and industrial land, 122 mg kg−1) values were obtained by the SSD method. In practice, users select the HCx value according to land-use protection goals and then adjust the threshold to local soils via pH, SOM, and CEC, so that decisions reflect bioavailability rather than total concentration. Our study provides a typical example of using the SSD method to determine the toxicity threshold of PHE, offering valuable reference information for further consideration and research by regulators and scientists. Despite integrating pot tests and extensive literature, reliance on four eastern Chinese soils limits representativeness to some extent.Author contributions
Dr Jiahui Zhu, Ms Qian Yang and Prof. Xinhua Zhan: data analysis, investigation, methodology and formal analysis; Ms. Qian Yang, Ms. Xuke Wang and Dr Jiawei Wang: experimental part, investigation and methodology; Ms Xuke Wang and Ms. Shuilin Zhu: methodology and data curation; Dr Jiahui Zhu and Ms. Qian Yang: writing the original draft, visualization; Dr Jiahui Zhu and Prof. Xinhua Zhan: conceptualization, writing—review & editing, validation, supervision; Prof. Xinhua Zhan: funding acquisition.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: toxicity data screening principle; toxicity data retrieval requirements; reliability data relating to species distribution, toxicity data of phenanthrene to terrestrial plants; toxicity data of phenanthrene to terrestrial invertebrates; toxicity data of phenanthrene on soil microorganisms and microbial-driven soil ecological processes; equations; the goodness of fit of three equations; effects of different phenanthrene concentrations on plant fresh weight, root length inhibition rate, stem length, and germination index in different soils; dose-response relationship curves between phenanthrene concentration and plant fresh weight/stem length in different soils; and the experimental setup. See DOI: https://doi.org/10.1039/d5em00664c.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2021YFC1809103), the National Natural Science Foundation of China (42477021, 42407328, 31770546), the Jiangsu Funding Program for Excellent Postdoctoral Talent (2023ZB159) and the Natural Science Foundation of Jiangsu Province (BK20241583).References
- Y. Shen, Y. Sheng, J. Li, J. Zhu, S. Shi and X. Zhan, The role of temperature in phenanthrene transfer and accumulation in crop leaves, Environ. Pollut., 2020, 258, 113827 CrossRef CAS PubMed.
- J. Zhu, R. Chen, Q. Feng, C. Huang, F. Huang, J. Du, J. Wang and X. Zhan, Mechanistic insights into auxin-enhancing polycyclic aromatic hydrocarbon uptake by wheat roots: Evidence from in situ intracellular pH and root-surface H(+) flux, J. Hazard. Mater., 2024, 465, 133077 CrossRef CAS PubMed.
- X. Zhan, X. Zhang, X. Yin, H. Ma, J. Liang, L. Zhou, T. Jiang and G. Xu, H+/phenanthrene symporter and aquaglyceroporin are implicated in phenanthrene uptake by wheat (Triticum aestivum L.) roots, J. Environ. Qual., 2012, 41, 188–196 CrossRef CAS PubMed.
- Y. Y. Qiu, Y. X. Gong and H. G. Ni, Contribution of soil erosion to PAHs in surface water in China, Sci. Total Environ., 2019, 686, 497–504 CrossRef CAS PubMed.
- C. Wang, X. Wang, P. Gong and T. Yao, Polycyclic aromatic hydrocarbons in surface soil across the Tibetan Plateau: spatial distribution, source and air-soil exchange, Environ. Pollut., 2014, 184, 138–144 Search PubMed.
- T. Wang, D. Su, X. Li, X. Wang and Z. He, Adsorption behavior of phenanthrene in soil amended with modified loofah sponge, J. Clean. Prod., 2021, 298, 126845 Search PubMed.
- A. Muratova, N. Pozdnyakova, O. Makarov, M. Baboshin, B. Baskunov, N. Myasoedova, L. Golovleva and O. Turkovskaya, Degradation of phenanthrene by the rhizobacterium Ensifer meliloti, Biodegradation, 2014, 25, 787–795 CrossRef CAS PubMed.
- X. Zhan, X. Yi, L. Yue, X. Fan, G. Xu and B. Xing, Cytoplasmic pH-Stat during Phenanthrene Uptake by Wheat Roots: A Mechanistic Consideration, Environ. Sci. Technol., 2015, 49, 6037–6044 CrossRef CAS PubMed.
- X. H. Zhan, H. L. Ma, L. X. Zhou, J. R. Liang, T. H. Jiang and G. H. Xu, Accumulation of phenanthrene by roots of intact wheat (Triticum acstivnm L.) seedlings passive or active uptake, BMC Plant Biol., 2010, 10, 1–8 CrossRef PubMed.
- V. S. Richardi, M. Vicentini, G. S. Morais, D. Rebechi, T. A. da Silva, L. F. Favaro and M. A. Navarro-Silva, Effects of phenanthrene on different levels of biological organization in larvae of the sediment-dwelling invertebrate Chironomus sancticaroli (Diptera: Chironomidae), Environ. Pollut., 2018, 242, 277–287 CrossRef CAS PubMed.
- A. A. Jennings, Worldwide regulatory guidance values for surface soil exposure to noncarcinogenic polycyclic aromatic hydrocarbons, J. Environ. Manage., 2012, 101, 173–190 CrossRef CAS PubMed.
- D. F. Kalf, T. Crommentuijn and E. J. v. d. Plassche, Environmental Quality Objectives for 10 Polycyclic Aromatic Hydrocarbons (PAHs), Ecotoxicol. Environ. Saf., 1997, 36, 89–97 Search PubMed.
- CCME, Canadian soil quality guidelines for the protection of environmental and human health, National Guidelines and Standards Office, Quebec, Canada, 2007 Search PubMed.
- Q. You, K. Yan, Z. Yuan, D. Feng, H. Wang, L. Wu and J. Xu, Polycyclic aromatic hydrocarbons (PAHs) pollution and risk assessment of soils at contaminated sites in China over the past two decades, J. Clean. Prod., 2024, 450 Search PubMed.
- Y. Huang, N. Zhang, Z. Ge, C. Lv, L. Zhu, C. Ding, C. Liu, P. Peng, T. Wu and Y. Wang, Determining soil conservation strategies: Ecological risk thresholds of arsenic and the influence of soil properties, Eco-Environ. Health, 2024, 3, 238–246 CrossRef PubMed.
- C. Ding, Y. Ma, X. Li, T. Zhang and X. Wang, Derivation of soil thresholds for lead applying species sensitivity distribution: A case study for root vegetables, J. Hazard. Mater., 2016, 303, 21–27 CrossRef CAS PubMed.
- G. Han, Y. Huang, M. Yang, D. Meng, H. Xia, H. Cui and D. Du, Ecological risk-based soil thresholds for polycyclic aromatic hydrocarbons (PAHs) addressing direct contact and food chain exposure in China, Sci. Total Environ., 2025, 992, 179978 CrossRef CAS PubMed.
- Z. Wang, C. Qu, J. Zhang, L. Zhi, T. Tang, H. Yao, W. Li, C. Shi and S. Qi, Constructing model-averaging species sensitivity distributions of Phenanthrene based on reproductive fitness: Implications for assessing ecological risk in urban watershed, J. Hazard. Mater., 2023, 443, 130296 CrossRef CAS PubMed.
- W. He, N. Qin, X. Kong, W. Liu, W. Wu, Q. He, C. Yang, Y. Jiang, Q. Wang, B. Yang and F. Xu, Ecological risk assessment and priority setting for typical toxic pollutants in the water from Beijing-Tianjin-Bohai area using Bayesian matbugs calculator (BMC), Ecol. Indic., 2014, 45, 209–218 Search PubMed.
- G. S. Siemering and R. Thiboldeaux, Background concentration, risk assessment and regulatory threshold development: Polycyclic aromatic hydrocarbons (PAH) in Milwaukee, Wisconsin surface soils, Environ. Pollut., 2021, 268, 115772 Search PubMed.
- Z. Pan, Y. Dong, G. He, T. Guo and N. Zhu, Spatial heterogeneity of soil properties in planted mixed forests in the rocky desertification areas of the Wuling Mountain, PeerJ, 2024, 12, e18724 Search PubMed.
- A. M. M. Mawad, E. S. E. Aldaby, M. M. Y. Madany and M. F. A. Dawood, The application of PAHs-Degrading Pseudomonas aeruginosa to mitigate the phytotoxic impact of pyrene on barley (Hordeum vulgare L.) and broad bean (Vicia faba L.) plants, Plant Physiol. Biochem., 2024, 215, 108959 CrossRef CAS PubMed.
- J. Zhu, Z. Zou, Y. Shen, J. Li, S. Shi, S. Han and X. Zhan, Increased ZnO nanoparticle toxicity to wheat upon co-exposure to phenanthrene, Environ. Pollut., 2019, 247, 108–117 CrossRef CAS PubMed.
- I. C. Eom, C. Rast, A. M. Veber and P. Vasseur, Ecotoxicity of a polycyclic aromatic hydrocarbon (PAH)-contaminated soil, Ecotoxicol. Environ. Saf., 2007, 67, 190–205 Search PubMed.
- S. Deng, T. Ke, Y. Wu, C. Zhang, Z. Hu, H. Yin, L. Guo, L. Chen and D. Zhang, Heavy Metal Exposure Alters the Uptake Behavior of 16 Priority Polycyclic Aromatic Hydrocarbons (PAHs) by Pak Choi (Brassica chinensis L.), Environ. Sci. Technol., 2018, 52, 13457–13468 CrossRef CAS PubMed.
- A. Gawryluk, A. Stepniowska and H. Lipinska, Effect of soil contamination with polycyclic aromatic hydrocarbons from drilling waste on germination and growth of lawn grasses, Ecotoxicol. Environ. Saf., 2022, 236, 113492 CrossRef CAS PubMed.
- J. Li, H. Zhang, J. Zhu, Y. Shen, N. Zeng, S. Liu, H. Wang, J. Wang and X. Zhan, Role of miR164 in the growth of wheat new adventitious roots exposed to phenanthrene, Environ. Pollut., 2021, 284, 117204 CrossRef CAS PubMed.
- S. H. Lee, W. S. Lee, C. H. Lee and J. G. Kim, Degradation of phenanthrene and pyrene in rhizosphere of grasses and legumes, J. Hazard. Mater., 2008, 153, 892–898 CrossRef CAS PubMed.
- W. Wilcke, Synopsis Polycyclic Aromatic Hydrocarbons (PAHs) in Soil — a Review, J. Plant Nutr. Soil Sci., 2000, 163, 229–248 Search PubMed.
- D. Werner, C. P. Higgins and R. G. Luthy, The sequestration of PCBs in Lake Hartwell sediment with activated carbon, Water Res., 2005, 39, 2105–2113 Search PubMed.
- W. D. Weissenfels, H. J. Klewer and J. Langhoff, Adsorption of polycyclic aromatic hydrocarbons (PAHs) by soil particles influence on biodegradability and biotoxicity, Appl. Microbiol. Biotechnol., 1992, 36, 689–696 CrossRef CAS PubMed.
- J. Deng, H. Wang, Y. Su, H. Zhang, C. Qin, A. Mosa and W. Ling, Determination of soil environment criteria for ecological safety of benzo[a]pyrene in soil based on the species sensitivity distribution approach, Appl. Soil Ecol., 2024, 195, 105213 CrossRef.
- X. Gong, S. Wang, Z. Wang, Y. Jiang, Z. Hu, Y. Zheng and S. Scheu, Earthworms modify soil bacterial and fungal communities through enhancing aggregation and buffering pH, Geoderma, 2019, 347, 59–69 CrossRef CAS.
- K. Wielgusz, M. Praczyk, L. Irzykowska and D. Świerk, Fertilization and soil pH affect seed and biomass yield, plant morphology, and cadmium uptake in hemp (Cannabis sativa L.), Ind. Crop. Prod., 2022, 175, 114245 Search PubMed.
- T. Wang, D. Su, X. Wang and Z. He, Adsorption-Degradation of Polycyclic Aromatic Hydrocarbons in Soil by Immobilized Mixed Bacteria and Its Effect on Microbial Communities, J. Agric. Food Chem., 2020, 68, 14907–14916 CrossRef CAS PubMed.
- L. Yang, D. Han, D. Jin, J. Zhang, Y. Shan, M. Wan, Y. Hu and W. Jiao, Soil physiochemical properties and bacterial community changes under long-term polycyclic aromatic hydrocarbon stress insitu steel plant soils, Chemosphere, 2023, 334, 138926 Search PubMed.
- A. Del Signore, A. J. Hendriks, H. J. Lenders, R. S. Leuven and A. M. Breure, Development and application of the SSD approach in scientific case studies for ecological risk assessment, Environ. Toxicol. Chem., 2016, 35, 2149–2161 CrossRef CAS PubMed.
- L. Qin, X. Sun, L. Yu, J. Wang, S. Modabberi, M. Wang and S. Chen, Ecological risk threshold for Pb in Chinese soils, J. Hazard. Mater., 2023, 444, 130418 CrossRef CAS PubMed.
- S. Belanger, M. Barron, P. Craig, S. Dyer, M. Galay-Burgos, M. Hamer, S. Marshall, L. Posthuma, S. Raimondo and P. Whitehouse, Future needs and recommendations in the development of species sensitivity distributions: Estimating toxicity thresholds for aquatic ecological communities and assessing impacts of chemical exposures, Integr. Environ. Assess. Manag., 2017, 13, 664–674 CrossRef PubMed.
- R. Checkai, E. Van Genderen, J. P. Sousa, G. Stephenson and E. Smolders, Deriving site-specific clean-up criteria to protect ecological receptors (plants and soil invertebrates) exposed to metal or metalloid soil contaminants via the direct contact exposure pathway, Integr. Environ. Assess. Manag., 2014, 10, 346–357 CrossRef CAS PubMed.
Footnote |
| † These two authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |