Open Access Article
Open Access ArticleAnion-decoupled electrolytes enable stable cycling and fast interfacial kinetics for calcium metal anodes
Qi
Meng
a,
Zhen
Zhan
b,
Yiyuan
Ma
a,
Qi
Qi
a,
Yingkai
Hua
a,
Yuyang
Yi
a,
Jingya
Yu
a,
Mengcheng
Wu
a,
Jingjing
Tang
 e,
Songhua
Cai
e,
Songhua
Cai
 b,
Kang Cheung
Chan
b,
Kang Cheung
Chan
 ac and
Zheng-Long
Xu
ac and
Zheng-Long
Xu
 *acd
*acd
aDepartment of Industrial and Systems Engineering, The Hong Kong Polytechnic University, Hung Hom, Hong Kong SAR, China. E-mail: zhenglong.xu@polyu.edu.hk
bDepartment of Applied Physics, The Hong Kong Polytechnic University, Hung Hom, Hong Kong, China
cResearch Institute for Advanced Manufacturing, The Hong Kong Polytechnic University, Hung Hom, Hong Kong SAR, China
dShenzhen Research Institute, The Hong Kong Polytechnic University, Shenzhen, China
eSchool of Metallurgy and Environment, Central South University, Changsha, China
First published on 2nd April 2026
Abstract
Calcium (Ca) metal batteries are a promising post-lithium-ion technology due to the high energy density of Ca and its crustal abundance. However, the strong cation–anion interactions of divalent Ca2+ in conventional electrolytes promote the formation of a Ca2+ ion-blocking solid electrolyte interphase (SEI) that hinders Ca metal electrodeposition. Here, we introduce an anion-decoupling strategy that combines a strongly coordinating solvent, dimethylacetamide (DMAc), with an anion-pulling 1,2-dibromobenzene (1,2-DBB) additive to regulate the widely accessible Ca(TFSI)2-based electrolytes for reversible Ca metal anodes. Spectroscopic and theoretical analyses reveal that DMAc induces a solvent-rich coordination structure and suppresses Ca2+-TFSI− ion pairs, while 1,2-DBB further pulls TFSI− out of the primary solvation shell, hindering its decomposition into unfavorable CaF2 in the SEI. The resulting inorganic-poor SEI enables fast Ca2+ transport and highly reversible Ca metal plating/stripping at low overpotentials of <0.19 V, compared to those of >5 V in conventional Ca(TFSI)2 electrolytes. The optimized electrolyte supports stable cycling of Ca//Ca symmetric cells for over 340 hours and delivers >90% capacity retention over 200 cycles in Ca//graphite and Ca//9,10-phenanthrenequinone full cells. This work establishes anion-decoupling electrolyte chemistry, distinct from the well-established anion-coupling electrolyte chemistry for monovalent metal anodes (Li and Na), enabling fast interfacial kinetics and stable cycling of divalent metal anodes.
Broader contextWith performance requirements constantly increasing for energy storage devices, there is much demand for highly affordable and high-energy density batteries that extend the capabilities of conventional lithium-ion systems. For battery anodes, calcium (Ca) metal is considered to be one of the most attractive choices because of its negative electrochemical potential, exceptional nature abundance, and high theoretical capacities. However, reversible cycling of Ca metal in aprotic organic electrolytes has been extremely difficult due to the anion-derived Ca ion-blocking passivation layers. Unlike monovalent Li electrolytes, divalent Ca electrolytes exhibit stronger cation–anion coordination that promotes anion decomposition and the aggressive formation of inorganic-rich solid–electrolyte interphases (SEIs). To address these fundamental challenges, this work introduces a two-pronged approach that decouples Ca ion–anion pairs in the bulk electrolyte and stabilizes the anion species to promote the formation of organic-rich SEI layers. The tailored interface supports fast Ca ion transport and enables Ca metal stripping/plating with low overpotentials. The anion-decoupling electrolyte design diverges conceptually from the widely explored anion-coupling paradigm for Li metal anodes, which offers a new mechanistic framework for advancing multivalent metal battery chemistries. |
1. Introduction
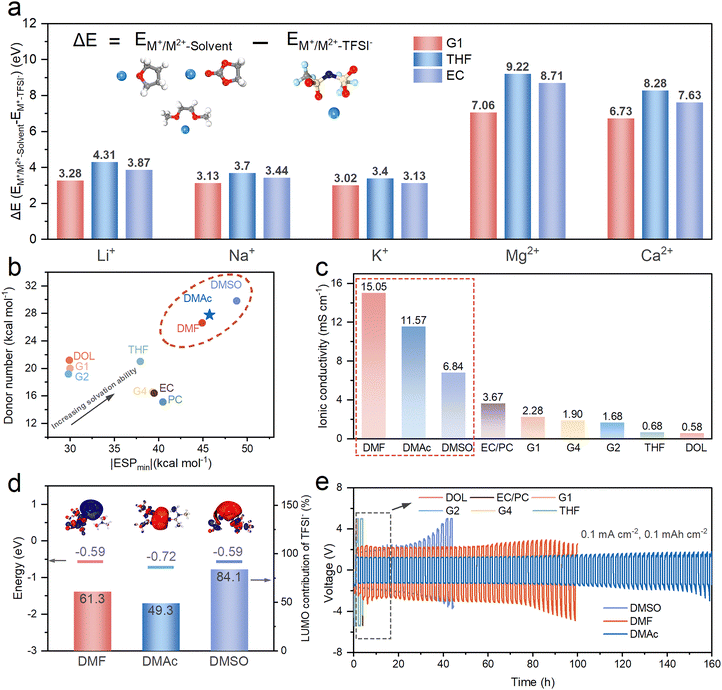
Driven by the rapid expansion of the electrochemical energy storage market, post-lithium metal batteries have emerged as promising alternatives to current Li-ion batteries, owing to their potential for higher energy densities from metal anodes and better affordability from abundant elements.1–3 Among the post-Li battery systems, calcium (Ca) metal batteries are particularly promising due to their low electrochemical potential (−2.87 V vs. the standard hydrogen electrode), high theoretical capacities (1337 mAh g−1 and 2073 mAh cm−3), excellent thermal stability (melting point: ∼842 °C), and crustal abundance (ranking as the 5th richest element in the Earth's crust).4–7 Nevertheless, reversible Ca metal anodes with compelling stripping/plating reaction kinetics remain inaccessible in most organic electrolytes.The problem roots in two intrinsic constraints. First, compared to monovalent cations like Li+ and Na+, Ca2+ with stronger electrostatic interactions hinders the ion-pair dissociation, leading to sluggish ion transport in the bulk electrolyte and high energy barriers for ion desolvation at the anode/electrolyte interface.8–10 Second, the aggressive decomposition of anions in organic electrolytes readily forms thick and ionically insulating inorganic phases on the surface of the Ca metal, thus disabling reversible Ca plating/stripping reactions (Fig. 1a).11–14 Given the critical role of the electrolyte in determining the solid electrolyte interphase (SEI) chemistry and interfacial kinetics,15–17 rational electrolyte design is under the spotlight for advancing Ca metal batteries. The first reversible Ca metal anode was achieved using a Ca(BF4)2–ethylene carbonate (EC)/propylene carbonate (PC) electrolyte.18 The strong Ca2+–BF4− ion pair coupling and the CaF2-rich SEI layers demanded elevated temperatures of above 75 °C to activate effective Ca ion migration.10 Room-temperature operation of Ca metal anodes was realized in a Ca(BH4)2/tetrahydrofuran (THF) electrolyte.19 The relatively conductive CaH2 component in the SEI from BH4− decomposition was found to enable Ca metal cycling at moderate overpotentials (<0.5 V). However, the narrow electrochemical stability window (<2.5 V) and the high cost of borohydride salts limit their practicality for Ca metal batteries using high-voltage cathodes. To weaken Ca2+–anion coordination, large and weakly coordinating anions such as [B(hfip)4]−,20,21 (CB11H12)−,22 and [Al(hfip)4]− (ref. 23) have been developed for reversible Ca metal anodes, but their practicality is limited by the expensive precursors and complex syntheses.
By contrast, Ca(TFSI)2 is an affordable and widely available salt, but the strong Ca2+–TFSI− interactions in organic electrolytes have demonstrated irreversibility for Ca metal anodes. This behavior contrasts with monovalent-ion (i.e., Li+ and Na+) electrolyte systems, which prefer weakly solvating solvents (e.g., fluorinated ethers) and enhanced contact-ion-pair (CIP) or aggregate (AGG) solvation structures to yield inorganic-rich SEI layers (i.e., LiF and NaF),24–26 thus stabilizing metal surfaces and promoting the uniform deposition of Li and Na metal anodes.27,28 In contrast, in the Ca(TFSI)2–DME or EC/PC electrolytes, the tightly bound Ca2+–TFSI− complexes lead to the formation of passivation layers mainly containing CaF2, CaO and CaCO3,29,30 which impose pronounced Ca2+ migration energy barriers (>1 eV).1,10,31 To alleviate the formation of inorganic-rich SEIs, strongly coordinating co-solvents (i.e., 4-methoxybutan-2-amine added in Ca[B(hfip)4]2–DME (G1) electrolyte)32 and ionic liquids (i.e., EmimBF4 added in Ca(TFSI)2–dimethyl sulfoxide (DMSO) electrolyte)33 have been introduced to decrease the coordination between anions and Ca2+. Although the cycling lifespan of Ca metal anodes was extended, the overpotential remains above 0.5 V. Therefore, developing new electrolytes or strategies that can unlock Ca(TFSI)2-based electrolytes enabling reversible Ca plating/stripping with low overpotentials remains challenging.
In this work, we introduce a two-pronged approach by systematically screening strongly coordinating solvents to reinforce Ca2+–solvent coordination (Fig. 1b) and an anion-pulling additive, 1,2-dibromobenzene (1,2-DBB), to further extract TFSI− out of the primary solvation shell in Ca(TFSI)2-based electrolytes (Fig. 1c). Theoretical calculations of solvation strengths across a series of solvents (i.e., ethers, carbonates, amides, and sulfoxides) in Ca(TFSI)2-based electrolyte solutions, correlated with the electrochemical responses of Ca metal anodes, reveal a direct relationship between Ca reversibility and the ion-pair decoupling capability, thus identifying dimethylacetamide (DMAc) as the optimal solvent in this system. Although DMAc solvent has been explored for Ca anodes, strong cation–anion coupling persists and interfacial kinetics remain sluggish,34 necessitating the introduction of “anion-pulling” chemistry to overcome this performance hurdle. By comparing the binding energies of halogen–benzene molecules with TFSI−, 1,2-dibromobenzene (1,2-DBB) is selected as an effective additive for weakening Ca2+–TFSI− coordination. Spectroscopic and theoretical analyses demonstrate that the 1,2-DBB additive modulates the solvation structure and increases the chemical bond orders of TFSI− anions, thereby suppressing the decomposition of TFSI− into unfavorable inorganic compounds in the SEI as demonstrated by high-resolution transmission electron microscopy (HRTEM) observations. The resulting Ca(TFSI)2–DMAc/1,2-DBB electrolyte exhibits high ionic conductivity (σ = 12.75 mS cm−1), low desolvation energy barriers (ΔE = 27.02 kJ mol−1), and accelerated Ca2+ transport kinetics through the electrode/electrolyte interface. Consequently, Ca//Ca symmetric cells operate with low overpotentials (<0.2 V vs. > 5 V in conventional Ca(TFSI)2-G1, EC/PC, or THF electrolytes) and extended cycle life (over 340 hours). The superiority of our anion-decoupled electrolytes is also demonstrated by the excellent cyclability of Ca//graphite (over 200 cycles with >90% capacity retention) and Ca//9,10-phenanthrenequinone (PQ) full cells (290 cycles at 50 mA g−1). Overall, this work unlocks the feasibility of Ca(TFSI)2-based electrolytes that were previously unsuitable for Ca metal anodes through anion-decoupling chemistry. Our findings emphasize fundamentally different electrolyte design principles from those established in monovalent metal anode systems, offering new insights towards developing feasible electrolytes for multivalent metal batteries.
2. Results and discussion
2.1 Strongly coordinating solvents for decoupling Ca2+–TFSI− pairs
Density functional theory (DFT) calculations of the binding energy differences (ΔE) between the representative alkali and alkaline earth metal ions (e.g., Li+, Na+, K+, Ca2+, and Mg2+) with typical solvents (e.g., G1, THF, and EC) and TFSI− anions reveal that ΔE for divalent ions (Ca2+ and Mg2+) are two times higher than those of monovalent ions (Li+, Na+, and K+) across all the representative solvents (Fig. 2a). This result illustrates the stronger cation–anion coupling strength in Ca-based electrolyte systems. Notably, Ca2+ presents stronger binding energies with TFSI− than FSI−, OTF−, and BF4− (Fig. S1), highlighting the difficulty in decoupling Ca2+–TFSI− ion pairs in the electrolyte. Furthermore, the lowest unoccupied molecular orbital (LUMO) energy levels of TFSI− shift markedly from 0.12 eV (free anion) to −0.68 eV upon coupling with Ca2+ (Fig. S2a). The strong Ca2+–TFSI− interaction reduces the Mayer bond orders across the internal bonds (C–F, S–N, and S–O) in TFSI−, thus enhancing their cleavage tendency to form the CaF2-rich SEI (Fig. S2b).35 It was reported that CaF2 exhibits the largest Ca2+ migration energy barriers (∼2046 meV) among the potential inorganic SEI components (i.e., ∼997 meV for CaO, ∼540 meV for CaH2, and ∼1436 meV for CaCO3).10 Therefore, it is essential to exclude TFSI− from the primary solvation shell and suppress its decomposition for feasible Ca metal cycling in the Ca(TFSI)2-based electrolytes.To approach a solvent-rich and anion-poor solvation configuration, we examined the donor number (DN) and the minimum electrostatic potential (ESPmin) of ten representative solvent molecules, including ethers, carbonates, amides, and sulfoxide. The DN reflects the tendency of electron donating ability of a solvent towards cations,36 while ESPmin represents the lowest electrostatic potential on the solvent surface, indicating the strength of cation–solvent interactions.37 Accordingly, solvents with high DN and large |ESPmin| are expected to deliver strong coordination with Ca2+. Fig. S3 and Fig. 2b summarize the |ESPmin| and DN distributions of nine representative solvents, which shows that amides (dimethylformamide (DMF) and DMAc) and the sulfoxide (DMSO) lie in the upper-right region with a DN of >26 kcal mol−1 and an |ESPmin| of >44 kcal mol−1, identifying them as highly coordinating solvent candidates. Ethers (i.e., dioxolane DOL, G1, diglyme G2, and tetraglyme G4) and carbonates (i.e., EC and PC) present relatively low DN (<15 kcal mol−1) or |ESPmin| (<30 kcal mol−1).
To validate this prediction, Raman measurements and molecular dynamics (MD) simulations were performed for 0.3 M Ca(TFSI)2 G1 (a weakly coordinating solvent) and 0.3 M Ca(TFSI)2 DMAc (a strongly coordinating solvent) electrolyte solutions. Raman spectra related to the S–N–S vibration of TFSI− reveal that the coordinated TFSI− species (at 749.5 cm−1) predominate in the G1-based electrolyte, whereas the free TFSI− peak (at 738.9 cm−1) is more pronounced in the DMAc-based electrolyte (Fig. S4). MD simulations further reveal the distinct solvation features: the Ca(TFSI)2-G1 electrolyte exhibits pronounced aggregations of Ca2+–TFSI− ion pairs (Fig. S5a), whereas Ca(TFSI)2–DMAc displays more dispersed solvated ion pair configurations (Fig. S5b). Radial distribution functions (RDFs) show a higher coordination number of 3.5 for Ca–OTFSI− (oxygen in TFSI−) in the Ca(TFSI)2–G1 electrolyte than that of 2.1 for the Ca(TFSI)2–DMAc electrolyte (Fig. S5c), indicating more Ca2+–TFSI− ion pairs in the former electrolyte. On the other hand, the coordination number for the solvent (Ca–Osolvent) increases from 4.6 in the G1-based electrolyte to 5.7 in the DMAc-based electrolyte (Fig. S5d). These comparisons corroborate the effectiveness of DMAc solvent with high DN and |ESPmin| values in decoupling TFSI− from Ca2+.
The improved ion decoupling translates directly to enhanced ion transport in the bulk electrolyte. Strongly coordinating electrolytes (0.3 M Ca(TFSI)2 in DMF, DMAc, or DMSO) exhibit higher ionic conductivities (σ = 6.84 to 15.05 mS cm−1) than the moderate or weakly solvating systems (σ = 0.58 to 3.67 mS cm−1) under the identical 0.3 M salt concentration (Fig. 2c and Fig. S6). Interestingly, galvanostatic cycling of Ca//Ca symmetric cells in these electrolytes at 0.1 mA cm−2 and 0.1 mAh cm−2 (Fig. 2e) displays reversibility only in the DMF, DMAc, and DMSO-based electrolytes, whereas the cells employing the G1, G2, G4, THF, DOL and EC/PC-based electrolytes rapidly fail by exceeding the 5 V cutoff voltage.
Among the three workable electrolytes, the cyclability of Ca//Ca cells follows the order of DMAc (160 h) > DMF (100 h) > DMSO (40 h) at 0.1 mA cm−2, despite the calculated binding energies between solvents and Ca2+ following a different trend (DMSO > DMAc > DMF, Fig. S7). This discrepancy suggests that, beyond solvation strength, the electrochemical stability of Ca2+–solvent–TFSI− complexes and the resulting SEI composition play an unignorable role in determining the reversibility of Ca metal anodes. Accordingly, we calculated the LUMO levels of Ca2+–solvent–TFSI− (solvent = DMF, DMAc, and DMSO) complexes and analyzed the LUMO contributions from TFSI− (Fig. 2d). Although their overall LUMO levels are comparable (−0.59 to −0.72 eV), corresponding to similar reductive stability, the TFSI− contributions to the LUMO are largely different, namely, 49.3% for Ca2+–DMAc–TFSI−, 61.3% for Ca2+–DMF–TFSI−, and 84.1% for Ca2+–DMSO–TFSI−. Interestingly, the observed cyclability (DMAc > DMF > DMSO) of Ca electrodes correlated inversely with the TFSI− contribution to the LUMO. It means that higher TFSI− stability in the Ca2+–solvent–TFSI− complex can suppress TFSI− decomposition into unfavorable inorganic SEI species, thus stabilizing the electrochemical reduction of Ca metal anodes. Overall, these results establish that strong Ca2+ solvating capability and the high stabilization degree of TFSI− can be considered as the primary criteria for selecting optimal solvents in Ca(TFSI)2-based electrolytes. Having established DMAc as the ideal solvent, we further evaluated the concentration-dependent anion-coupling behavior. The physicochemical properties of electrolytes and resulting electrochemical performance of Ca electrodes recommend 0.3 M Ca(TFSI)2–DMAc as the optimal choice (Fig. S8).
2.2 Anion-pulling halogen benzene additives
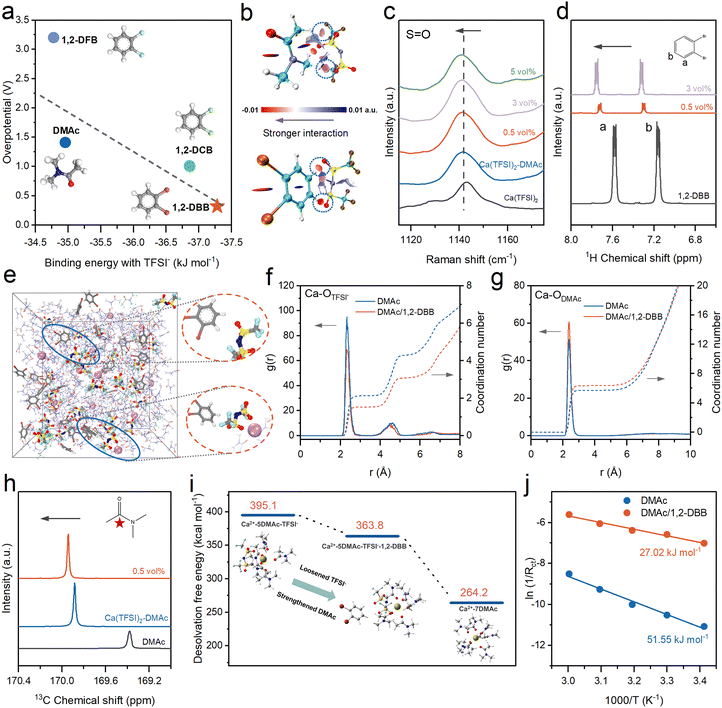
Despite the reversibility achieved in Ca(TFSI)2–DMAc electrolytes, the high overpotentials of >1 V for Ca metal plating/stripping reactions remain unsatisfactory. To further ameliorate the reaction kinetics, a series of halogenated benzenes were screened as anion-pulling additives to further decouple the Ca2+–TFSI− ion pairs. The polarizable aromatic framework and electron-withdrawing halogens confer strong electropositivity for the aromatic C–H bonds (Fig. S9), which favors the formation of C–H⋯O hydrogen bonds with TFSI−. Fig. S10a presents the C–H⋯O binding energies of various halogenated benzenes and DMAc with TFSI−, namely, −36.86 kJ mol−1 for 1,2-dichlorobenzene (1,2-DCB), −37.28 kJ mol−1 for 1,2-DBB, −34.78 kJ mol−1 for 1,2-difluorobenzene (1,2-DFB), and −34.96 kJ mol−1 for DMAc, respectively. Remarkably, the Ca deposition/stripping overpotential decreases nearly linearly with increasing C–H⋯O binding energies, reaching a minimum of 0.32 V at 0.2 mA cm−2 in the 0.3 M Ca(TFSI)2–DMAc/1,2-DBB electrolyte (Fig. 3a and Fig. S10b). The 1.1 V reduction from Ca(TFSI)2–DMAc to Ca(TFSI)2–DMAc/1,2-DBB suggests the effectiveness of the anion-pulling strategy. It is noted that although 1,2-DFB and DMAc present comparable binding energies with TFSI−, the 1,2-DFB additive significantly increases the overpotential to 3.2 V (from 1.4 V for the baseline DMAc electrolyte), which is likely attributable to the 1,2-DFB-derived fluorinated inorganic species in the SEI layer.For the interaction between 1,2-DBB additives and Ca2+, DFT calculations exhibit markedly lower |ESPmin| values (i.e., 14.79 kcal mol−1 for 1,2-DBB vs. 45.78 kcal mol−1 for DMAc) and binding energies (i.e., −83.83 kcal mol−1 for Ca2+-1,2-DBB vs. −115.19 kcal mol−1 for Ca2+–DMAc) (Fig. S11). It is inferred that the 1,2-DBB additive does not enter the first solvation shell but instead exerts its pulling effect externally on TFSI−. Other bromine benzene additives with different numbers of bromine substituents, like 1-bromobenzene (1-BB) and 1,3,5-tribromobenzene (1,3,5-TBB), were also examined that present lower binding energies with TFSI− and inferior electrochemical performance for Ca metal anodes (Fig. S12). By cycling Ca//Ca symmetric cells in DMAc/1,2-DBB electrolytes with different 1,2-DBB ratios (0.1 vol%, 0.2 vol%, 0.5 vol%, and 1.0 vol%, Fig. S13), the 0.5 vol% 1,2-DBB was determined as the optimal choice with the best cyclability of Ca metal for the subsequent studies.
Raman and nuclear magnetic resonance (NMR) measurements were carried out to evaluate the solvation perturbation induced by 1,2-DBB. Upon dissolving Ca(TFSI)2 in DMAc, the characteristic Raman peak at 1143.8 cm−1 referring to the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O vibration of TFSI− downshifts to 1141.5 cm−1, and further to 1140.4 cm−1 with 1,2-DBB addition (Fig. 3c), signifying noticeable decoupling of TFSI− anions. The strong interaction between TFSI− and 1,2-DBB is further evidenced by the concurrent redshifts in the C–H in-plane and C–C–C out-of-plane bending vibrations of 1,2-DBB Raman spectra (Fig. S14). These spectral changes can be attributed to the redistribution of electron density at the C–H donor site and distortion of the aromatic ring in 1,2-DBB upon binding with TFSI−.38 Moreover, the downfield shift of 1H NMR peaks for 1,2-DBB (at both 0.5 vol% and 3 vol% concentrations) further illustrates the electron-withdrawing effect of the C–H sites in 1,2-DBB on TFSI− (Fig. 3d). Similarly, the downfield shift in the 19F NMR chemical shifts of TFSI− anions upon adding 1,2-DBB (Fig. S15) also supports the anion-pulling mechanism.39
O vibration of TFSI− downshifts to 1141.5 cm−1, and further to 1140.4 cm−1 with 1,2-DBB addition (Fig. 3c), signifying noticeable decoupling of TFSI− anions. The strong interaction between TFSI− and 1,2-DBB is further evidenced by the concurrent redshifts in the C–H in-plane and C–C–C out-of-plane bending vibrations of 1,2-DBB Raman spectra (Fig. S14). These spectral changes can be attributed to the redistribution of electron density at the C–H donor site and distortion of the aromatic ring in 1,2-DBB upon binding with TFSI−.38 Moreover, the downfield shift of 1H NMR peaks for 1,2-DBB (at both 0.5 vol% and 3 vol% concentrations) further illustrates the electron-withdrawing effect of the C–H sites in 1,2-DBB on TFSI− (Fig. 3d). Similarly, the downfield shift in the 19F NMR chemical shifts of TFSI− anions upon adding 1,2-DBB (Fig. S15) also supports the anion-pulling mechanism.39
The binding configurations between TFSI− and DMAc or 1,2-DBB were calculated using DFT. It shows a shorter C–H⋯O length of 2.40 Å for 1,2-DBB–TFSI− than that of 2.66 Å for DMAc–TFSI− (Fig. S16a). Meanwhile, enhanced C–F, S–N and S–O bonding orders in TFSI− upon bonding with 1,2-DBB suggests greater thermodynamic stability of TFSI− (Fig. S16b). The strong noncovalent interactions were further visualized via interaction region indicator (IRI) isosurfaces (Fig. 3b).40 MD simulations reveal that 1,2-DBB forms external complexes with TFSI− without entering the primary Ca2+ solvation shell (Fig. 3e). RDFs (Fig. 3f and g) show that the coordination numbers of Ca–OTFSI− decrease from 2.1 to 1.5, while the Ca–ODMAc increases from 5.7 to 6.3 to preserve solvation stability. Critically, 1,2-DBB was not observed in the primary solvation shell, consistent with their weaker binding energy to Ca2+ (Fig. S17). The rearrangement of DMAc coordination was further validated by spectroscopic evidence: the downshifts of the 13C NMR chemical shift (Fig. 3h and Fig. S18) in the DMAc solvent following the 1,2-DBB addition indicated enhanced Ca2+–DMAc interaction. This enhancement is corroborated by Raman spectra, which reveal an increased percentage of coordinated DMAc molecules from 42.7% to 49.2% (Fig. S19). The effective anion-decoupling resulted in an increased ionic conductivity from 11.57 to 12.75 mS cm−2 (Fig. S20), confirming the accelerated Ca2+ transport in the DMAc/1,2-DBB electrolyte.
Building on the MD-derived coordination number (7.8), a representative Ca2+ solvation structure comprising five DMAc and one TFSI− anion was modeled. In the presence of 1,2-DBB, DFT results show elongated and weakened Ca2+–TFSI− bonds arising from the extraction of TFSI− from the solvation shell by 1,2-DBB. Conversely, the Ca2+–DMAc coordination presents shorter bond lengths and higher bond orders to compensate for the Ca2+ polarization left by TFSI− deficiency (Fig. S21). Moreover, the presence of 1,2-DBB reduces the desolvation free energy of Ca2+, reaching a minimum upon complete TFSI− removal, indicating that anion decoupling accelerates desolvation kinetics (Fig. 2i). Temperature-dependent electrochemical impedance spectroscopy (EIS) analyses (Fig. 2j and Fig. S22) also reveal lower Ca2+ desolvation energies in the DMAc/1,2-DBB electrolyte (ΔE = 27.02 kJ mol−1) compared with pristine DMAc (ΔE = 51.55 kJ mol−1), consistent with the faster interfacial kinetics. In addition, the TFSI− contribution to the LUMO decreases from 11.1% to 3.4% upon 1,2-DBB addition (Fig. S23), suggesting improved reductive stability of TFSI− and regulated SEI chemistry to be discussed below.
2.3 Solid electrolyte interphases from anion-decoupled electrolytes
The anion-decoupling strategy not only facilitates ion transport in the bulk electrolyte and the Ca2+ desolvation kinetics at the electrolyte/electrode interface but also importantly influences the SEI composition and Ca stripping/plating behaviors. After 20 cycles in Ca//Ca symmetric cells in the DMAc electrolyte, the cycled Ca anodes were covered by a fluorine (F)-rich passivation layer (Fig. 4a) in comparison to pristine Ca foil (Fig. S24a and b). XRD analysis identifies crystalline Ca(OH)2, CaO, and CaF2 as the dominant inorganic species on the Ca metal surface (Fig. 4c). In contrast, Ca metal electrodes cycled in the DMAc/1,2-DBB electrolyte present a uniform surface with significantly reduced F content (Fig. 4b and Fig. S24c), consistent with the negligible CaF2 peaks in the XRD result. HRTEM was employed to examine the microstructure and composition of the SEI. Ca metal anodes cycled in the DMAc electrolyte present a thick SEI layer (∼23.5 nm) containing CaF2 nanocrystals with a lattice fringe of 0.273 nm (Fig. 4d). The SEI formed in the DMAc/1,2-DBB electrolyte is thinner (∼11.5 nm) and shows an amorphous organic layer on the surface of metallic Ca deposits (Fig. 4e). Furthermore, characterization of SEI layers formed in the DMAc/1,2-DCB and DMAc/1,2-DFB electrolytes reveals a clear correlation between the anion-decoupling ability of the additives and the resulting SEI properties (Fig. S25).The chemical composition and depth distribution of SEI layers were further elucidated by X-ray photoelectron spectroscopy (XPS) depth profiling and time-of-flight secondary ion mass spectrometry (TOF-SIMS). After cycling in the DMAc electrolyte, the F 1s spectrum shows a high content of Ca–F species (Fig. S26a), which become more prominent with increasing Ar+ etching time. This result indicates abundant CaF2 throughout the SEI layer on the Ca metal surface, a consequence of aggressive TFSI− decomposition. Conversely, in the DMAc/1,2-DBB electrolyte, the normalized F 1s peak intensities for inorganic species are much lower (Fig. S26b) along with increased C–Fx peaks (Fig. S27). Such a difference confirms the suppressed TFSI− decomposition and stabilization of C–F bonds by the 1,2-DBB additive. Similar trends are also observed in the S 2p and N 1s spectra (Fig. S28 and S29).
XPS depth profiling revealed a hierarchical SEI structure in the DMAc electrolyte, where the inorganic components (i.e., CaF2 and CaSx) from TFSI− decomposition become increasingly prominent upon etching, while organic components diminished rapidly with depth (Fig. S27). Conversely, the SEI from the DMAc/1,2-DBB electrolyte exhibited a reduced inorganic content and abundant organic components upon etching, indicating the formation of a solvent-derived, organic-rich SEI promoted by the anion-pulling effect. These SEI characteristics were further corroborated by TOF-SIMS. The SEI formed in the DMAc/1,2-DBB electrolyte was dominated by solvent-derived organic species (CN− and C2−), alongside minor inorganic fluorides (Fig. 4f–h and Fig. S30). In contrast, the SEI derived from the DMAc electrolyte shows a high prevalence of CaF+, Ca2OF+, and Ca3O2F+ throughout the depth, while organic components are rich only on the top surface (Fig. 4g and Fig. S31a–c). Sulfur-containing species (S−, SO−, and SO2−), which are solely derived from TFSI− anions, are also greatly reduced upon 1,2-DBB addition (Fig. S31d). Quantitative analysis of the organic-to-inorganic component (O/I) ratios from TOF-SIMS depth profiles (Fig. 4h) highlights this contrast: the internal SEI formed in the DMAc/1,2-DBB electrolyte reached an O/I value of 2.1, compared to 0.8 for the SEI in the DMAc electrolyte demonstrates the suppression of TFSI− decomposition. To confirm the superiority of the organic-rich SEI, an electrolyte replacement protocol was implemented (Fig. 4i and j). When cycled directly in the DMAc electrolyte, the TFSI−-derived SEI with sluggish Ca2+ diffusion feature results in a high overpotential of 1.27 V. In contrast, Ca metal electrodes pre-cycled in the DMAc/1,2-DBB electrolyte and subsequently transferred to the DMAc electrolyte exhibited a much lower overpotential of 0.55 V, indicating the persistent benefit of the pre-formed organic-rich SEI.
To evaluate the possible reduction of the 1,2-DBB additive on Ca metal, Ca electrodes were immersed in pure 1,2-DBB for 24 h before assembling symmetric cells with DMAc electrolyte. The resulting voltage profiles remain unchanged relative to the untreated Ca//Ca cells (Fig. S32). Furthermore, in situ Raman spectroscopy monitored the electrolyte evolution during cycling (Fig. S33). The 1,2-DBB signal at 399.1 cm−1 remained constant in both the intensity and the peak position, whereas the characteristic DMAc peak at 589.7 cm−1 presents apparent attenuation during the initial cycles before approaching stabilization. Consistently, no bromine-containing species were detected in the TOF-SIMS spectrum for the SEI (Fig. S34). These results collectively demonstrate that 1,2-DBB acts solely as an anion-pulling modulator without direct participation in interfacial reactions, which differs from the reported role of 1,2-DBB decomposition in forming a NaBr-rich SEI in Na metal batteries.41
2.4 Electrochemical performance of Ca metal anodes
To validate the efficacy of the anion-decoupled electrolyte, electrochemical performance of Ca metal anodes was systematically evaluated. As shown in Fig. 5a, the Ca//Ca symmetric cell in the DMAc/1,2-DBB electrolyte achieves an extended cycling lifetime of 340 h at 0.05 mA cm−2 and 0.025 mAh cm−2. In comparison with a high overpotential of 1.68 V in the DMAc electrolyte, the overpotential reduced by nearly nine-fold to 0.19 V. Even under a ten-fold higher current density of 0.5 mA cm−2, the cells with DMAc/1,2-DBB electrolyte persisted stability over 65 cycles (Fig. S35). Furthermore, the DMAc/1,2-DBB electrolyte enabled superior rate capability (Fig. 5b), reducing the overpotentials by two to five times compared to the DMAc electrolyte across current densities ranging from 0.1 to 2 mA cm−2 (Fig. 5c). Consistently, the cyclic voltammetry (CV) profiles exhibited pronounced stripping and plating peak currents (Fig. S36), indicative of enhanced reaction kinetics. Benchmarking against state-of-the-art Ca anode performance in various electrolytes highlights that the present anion-decoupled formulation achieves one of the best combinations of low overpotentials, high current responses, and extended cycling stability (Fig. 5d and Table S1).34,42–45 | ||
| Fig. 5 (a) Cycling performance of Ca//Ca symmetric cells in DMAc and DMAc/1,2-DBB electrolytes at 0.05 mA cm−2 and 0.025 mAh cm−2. (b) Rate performance of Ca//Ca symmetric cells in DMAc and DMAc/1,2-DBB electrolytes at a constant capacity of 0.1 mAh cm−2. (c) Overpotentials of Ca//Ca cells at increasing current rates derived from (b). (d) Comparison of the electrochemical performance of Ca//Ca symmetric cells in recently reported electrolytes.40–44 (e) Cycling performance, (f) rate performance and (g) charge–discharge curves of the Ca//Gra cell in the DMAc/1,2-DBB electrolyte. (h) Cycling performance of the Ca//PQ full cell in the DMAc/1,2-DBB electrolyte. | ||
Electrochemical impedance spectroscopy (EIS) was employed to probe the interfacial kinetics and their evolution during cycling (Fig. S37). For fresh cells, the charge transfer resistance (RCT) of Ca//Ca symmetric cells in the DMAc/1,2-DBB electrolyte is more than twenty times lower than that in the DMAc electrolyte (Fig. S37a and b). Although impedances increased upon cycling for both systems, the cell with the DMAc/1,2-DBB electrolyte maintained exceptional interfacial stability, with both RCT and SEI resistance (RSEI) reduced by over seven times compared to those in the DMAc electrolyte after 20 cycles (Fig. S37c and d), consistent with the low overpotential and superior rate performance.
CV curves of Ca//carbon paper (CC) asymmetric cells (Fig. S38) exhibited reduced voltage hysteresis and larger current response in the DMAc/1,2-DBB electrolyte than those in the DMAc electrolyte. SEM images and corresponding EDS mapping demonstrated uniform Ca deposits on the CC current collector (Fig. S39). To further verify the kinetic advantage in the DMAc/1,2-DBB electrolyte, we then paired Ca metal with graphite (Gra) electrodes undergoing a rapid co-intercalation reaction in the DMAc-based electrolyte.46,47 In the DMAc system, Ca//Gra cells suffered severe polarization and negligible capacities due to the high overpotential and poor reversibility of Ca metal anodes (Fig. S40). In contrast, the DMAc/1,2-DBB electrolyte enabled highly reversible discharge/charge over 200 cycles at 100 mA g−1 with a stable capacity of 78 mAh g−1 (Fig. 5e). When the current density increased by 20-fold from 50 mA g−1 to 1000 mA g−1, 79% of the initial capacity was preserved (Fig. 5f) with a distinct discharge plateau at 0.37 V throughout the high-rate cycling (Fig. 5g). The remarkable high-rate capability can be attributed to the appealing reaction kinetics of the Ca metal electrode. The Ca//9,10-phenanthrenequinone (PQ) full cell was also built to deliver decent cyclic capacities exceeding 90 mAh g−1 and stable cycling for over 290 cycles (Fig. 5h and Fig. S41), supporting the practical applicability of the tailored electrolyte in Ca-based energy storage, whereas it fails to deliver appreciable capacity in the DMAc electrolyte due to the severe kinetic limitation of the Ca anode (Fig. S42). When extended to other fluorinated Ca salts (i.e., Ca(BF4)2 and Ca(OTf)2), the addition of 1,2-DBB can also significantly reduce the Ca plating/stripping overpotentials, which suggests the broad applicability of our anion-decoupling strategy across diverse Ca electrolytes (Fig. S43). Finally, we also prove the effectiveness of our anion-decoupling strategy for Mg metal anodes by cycling Mg//Mg cells in Mg(TFSI)2-G1, DMAc, and DMAc/1,2-DBB electrolytes, where similar reductions in the plating/stripping overpotentials are observed (Fig. S44), implying the general applicability of this electrolyte design principle for multivalent metal anodes.
3. Conclusions
In summary, we revealed the critical role of anion-decoupling chemistry in enhancing the reaction kinetics and cycling stability of Ca metal anodes in Ca(TFSI)2-based electrolytes. By integrating a strongly coordinating DMAc solvent with the anion-pulling 1,2-DBB additive, the Ca2+–TFSI− coupling is significantly weakened, rendering the strength of Ca2+–solvent coordination, high ionic conductivity (12.75 mS cm−1), and reduced desolvation energy barriers at the electrode/electrolyte interface. More importantly, this decoupling strategy improves the stability of TFSI− and suppresses its decomposition into undesirable inorganic compounds. The organic-rich SEI from a solvent-rich electrolyte configuration facilitates Ca2+ transport and electrodeposition with low overpotentials. Effectiveness of the anion-decoupled electrolyte is demonstrated by the stable cycling of Ca//Ca symmetric cells in the Ca(TFSI)2–DMAc/1,2-DBB electrolyte, in comparison to the high overpotentials of >5 V and short cycle life of Ca metal electrodes in conventional electrolytes. The anion-decoupled DMAc/1,2-DBB electrolyte also sustains Ca//Gra and Ca//PQ full cells over 200 cycles with >90% capacity retention. These findings highlight a unique electrolyte design paradigm, fundamentally distinct from monovalent metal anodes which prefer strong anion coordination in the electrolyte systems, offering new insights for advancing sustainable and safe multivalent metal anodes.Author contributions
Conceptualization, methodology and formal analysis: Q. Meng and Z.-L. Xu; data curation, investigation, project administration, visualization and writing – original draft: Q. Meng; resources and validation: Z. Zhan; funding acquisition and resources: Z.-L. Xu, Y. Y. Ma, Q. Qi, Y. K. Hua, Y. Y. Yi, J. Y. Yu, and M. C. Wu; supervision: Z.-L. Xu; writing – reviewing and editing: J. J. Tang, S. H. Cai, K. C. Chan and Z.-L. Xu.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ee07317k.Acknowledgements
This work described in this paper was fully supported by grants from the Research Grants Council of the Hong Kong Special Administrative Region, China (Project No. PolyU15305022 and PolyU15304723), the Research Institute for Advanced Manufacturing (Project No. 1-CDLR), the Research Committee of the Hong Kong Polytechnic University (Project No. G-UARH, 1-BBR0, and 4-W43C), the General Program Sponsored by Basic Research Fund in Shenzhen Natural Science Foundation (Project No. JCYJ20250604184257074), and the NSFC/RGC Joint Research Scheme (Project No. N_PolyU5117/25).References
- J. Kim, M. Kim, J. Lee, J. An, S. Yang, H. C. Ahn, D.-J. Yoo and J. W. Choi, Chem. Soc. Rev., 2024, 53, 8878–8902 RSC.
- Y. Liang, H. Dong, D. Aurbach and Y. Yao, Nat. Energy, 2020, 5, 646–656 CrossRef CAS.
- H. Lin, J. Yu, F. Chen, R. Li, B. Y. Xia and Z. L. Xu, Small Methods, 2023, 7, 2300561 CrossRef CAS PubMed.
- I. D. Hosein, ACS Energy Lett., 2021, 6, 1560–1565 CrossRef CAS.
- Y. Hua, Y. Ma, Q. Qi and Z.-L. Xu, Nanoscale, 2024, 16, 17683–17698 RSC.
- R. Li, Y. Lee, Z. Song, S. Ma, Y. Yi, H. Lin, Y. Hua, P. Jiang, F. Chen, J. Yu, X. Pu, Z. Chen, K. C. Chan, K.-Y. Park and Z.-L. Xu, Adv. Mater., 2025, 37, e06603 CrossRef CAS PubMed.
- H. Lin, Q. Meng, J. Long, C. Cao and Z. L. Xu, Adv. Funct. Mater., 2026, e00035 CrossRef.
- S. Li, J. Zhang, S. Zhang, Q. Liu, H. Cheng, L. Fan, W. Zhang, X. Wang, Q. Wu and Y. Lu, Nat. Energy, 2024, 9, 285–297 CrossRef CAS.
- M. E. Arroyo-de Dompablo, A. Ponrouch, P. Johansson and M. R. Palacín, Chem. Rev., 2019, 120, 6331–6357 CrossRef PubMed.
- J. Forero-Saboya, C. Davoisne, R. Dedryvère, I. Yousef, P. Canepa and A. Ponrouch, Energy Environ. Sci., 2020, 13, 3423–3431 RSC.
- D. Aurbach, R. Skaletsky and Y. Gofer, J. Electrochem. Soc., 1991, 138, 3536 CrossRef CAS.
- S. Yang, X. Wang, R. Li, Y. Zhou, H. Huang, M. Zhou, Y. Gao, W. Zhao, Y. Gao and Z. Pan, Energy Environ. Sci., 2025, 18, 1941–1951 RSC.
- A. M. Melemed and B. M. Gallant, J. Electrochem. Soc., 2020, 167, 140543 CrossRef CAS PubMed.
- B. Ji, H. He, W. Yao and Y. Tang, Adv. Mater., 2021, 33, 2005501 CrossRef CAS PubMed.
- H. Wan, J. Xu and C. Wang, Nat. Rev. Chem., 2024, 8, 30–44 CrossRef CAS PubMed.
- X. Liu, X. Dong, H. Adenusi, Y. Wu and S. Passerini, Nat. Rev. Chem., 2025, 1–12 Search PubMed.
- H. Lin, Z. Zhan, H. Zeng, R. Li, Y. Yi, F. Chen, S. Cai, Y. Zhu, C. F. Cheung and Z.-L. Xu, Adv. Mater., 2025, e10711 CrossRef CAS PubMed.
- A. Ponrouch, C. Frontera, F. Barde and M. R. Palacin, Nat. Mater., 2016, 15, 169–172 CrossRef CAS PubMed.
- D. Wang, X. Gao, Y. Chen, L. Jin, C. Kuss and P. G. Bruce, Nat. Mater., 2018, 17, 16–20 CrossRef CAS PubMed.
- Z. Li, O. Fuhr, M. Fichtner and Z. Zhao-Karger, Energy Environ. Sci., 2019, 12, 3496–3501 RSC.
- A. Shyamsunder, L. E. Blanc, A. Assoud and L. F. Nazar, ACS Energy Lett., 2019, 4, 2271–2276 CrossRef CAS.
- K. Kisu, S. Kim, T. Shinohara, K. Zhao, A. Zuttel and S. I. Orimo, Sci. Rep., 2021, 11, 7563 CrossRef CAS PubMed.
- T. Pavcnik, J. D. Forero-Saboya, A. Ponrouch, A. Robba, R. Dominko and J. Bitenc, J. Mater. Chem. A, 2023, 11, 14738–14747 RSC.
- Y. Zhao, T. Zhou, M. Mensi, J. W. Choi and A. Coskun, Nat. Commun., 2023, 14, 299 CrossRef CAS PubMed.
- S. Wang, X.-G. Zhang, Y. Gu, S. Tang and Y. Fu, J. Am. Chem. Soc., 2024, 146, 3854–3860 CrossRef CAS PubMed.
- S. Ilic, S. N. Lavan and J. G. Connell, Chem, 2024, 10, 2987–3007 CAS.
- J. Tan, J. Matz, P. Dong, J. Shen and M. Ye, Adv. Energy Mater., 2021, 11, 2100046 CrossRef CAS.
- H. Yildirim, A. Kinaci, M. K. Y. Chan and J. P. Greeley, ACS Appl. Mater. Interfaces, 2015, 7, 18985–18996 CrossRef CAS PubMed.
- J. D. Forero-Saboya, E. Marchante, R. B. Araujo, D. Monti, P. Johansson and A. Ponrouch, J. Phys. Chem. C, 2019, 123, 29524–29532 CrossRef CAS PubMed.
- R. B. Araujo, V. Thangavel and P. Johansson, Energy Storage Mater., 2021, 39, 89–95 CrossRef.
- Z. Hou, R. Zhou, Z. Min, Z. Lu and B. Zhang, ACS Energy Lett., 2023, 8, 274–279 CrossRef CAS.
- S. Hou, X. Ji, K. Gaskell, P.-F. Wang, L. Wang, J. Xu, R. Sun, O. Borodin and C. Wang, Science, 2021, 374, 172–178 CrossRef CAS PubMed.
- L. Ye, M. Liao, K. Zhang, M. Zheng, Y. Jiang, X. Cheng, C. Wang, Q. Xu, C. Tang, P. Li, Y. Wen, Y. Xu, X. Sun, P. Chen, H. Sun, Y. Gao, Y. Zhang, B. Wang, J. Lu, H. Zhou, Y. Wang, Y. Xia, X. Xu and H. Peng, Nature, 2024, 626, 313–318 CrossRef CAS PubMed.
- Z. Hou, R. Zhou, Y. Yao, Z. Min, Z. Lu, Y. Zhu, J. M. Tarascon and B. Zhang, Angew. Chem., Int. Ed., 2022, 61, e202214796 CrossRef CAS PubMed.
- I. Mayer, J. Comput. Chem., 2007, 28, 204–221 CrossRef CAS PubMed.
- P. Zhou, Y. Xiang and K. Liu, Energy Environ. Sci., 2024, 17, 8057–8077 RSC.
- Y. Wu, Q. Hu, H. Liang, A. Wang, H. Xu, L. Wang and X. He, Adv. Energy Mater., 2023, 13, 2300259 CrossRef CAS.
- G. Shakila, S. Periandy and S. Ramalingam, Spectrochim. Acta, Part A, 2012, 86, 449–455 CAS.
- Z. Zhao, B. Nian, Y. Lei, L. Zhao, M. N. Hedhili, D. Guo, Z. Shi, W. Zhao, J. K. El-Demellawi and Y. Wang, Adv. Mater., 2024, 36, 2402626 CrossRef CAS PubMed.
- T. Lu and Q. Chen, Chem.: Methods, 2021, 1, 231–239 CAS.
- L. Wang, N. Ren, Y. Yao, H. Yang, W. Jiang, Z. He, Y. Jiang, S. Jiao, L. Song, X. Wu, Z.-S. Wu and Y. Yu, Angew. Chem., Int. Ed., 2023, 135, e202214372 CrossRef.
- Z. Hou, R. Zhou, K. Liu, J. Zhu and B. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202413416 CrossRef CAS PubMed.
- H. Lin, J. Meng, W. Guo, R. Li, Y. Yi, Y. Ma, C. F. Cheung, D. Aurbach and Z.-L. Xu, Energy Environ. Sci., 2024, 17, 6548–6558 RSC.
- S. Yang, X. Wang, R. Li, Y. Zhou, H. Huang, M. Zhou, Y. Gao, W. Zhao, Y. Gao, Z. Pan and X. Yang, Energy Environ. Sci., 2025, 18, 1941–1951 RSC.
- Z. Slim, C. Cruz-Cardona, C. Pechberty, T. Hosaka, Z. Mandić, V. Panic and P. Johansson, ACS Mater. Lett., 2025, 7, 3235–3242 CrossRef.
- Y. Yi, Y. Xing, H. Wang, Z. Zeng, Z. Sun, R. Li, H. Lin, Y. Ma, X. Pu, M. M. Li, K. Y. Park and Z. L. Xu, Angew. Chem., Int. Ed., 2024, 63, e202317177 CrossRef CAS PubMed.
- J. Park, Z.-L. Xu, G. Yoon, S. K. Park, J. Wang, H. Hyun, H. Park, J. Lim, Y.-J. Ko, Y. S. Yun and K. Kang, Adv. Mater., 2020, 32, 1904411 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |