Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Novel fast Li-ion conductors for solid-state electrolytes from first-principles
Tushar Singh
Thakur
 *a,
Loris
Ercole
*a,
Loris
Ercole
 a and
Nicola
Marzari
a and
Nicola
Marzari
 abc
abc
aTheory and Simulation of Materials (THEOS), and National Centre for Computational Design and Discovery of Novel Materials (MARVEL), École Polytechnique Fédérale de Lausanne, 1015 Lausanne, Switzerland. E-mail: tushar.thakur@epfl.ch
bPSI Center for Scientific Computing, Theory and Data, Paul Scherrer Institute, 5232 Villigen PSI, Switzerland
cTheory of Condensed Matter, Cavendish Laboratory, University of Cambridge, Cambridge CB3 0US, UK
First published on 22nd April 2026
Abstract
We present a high-throughput computational screening for fast lithium-ion conductors to identify promising materials for application in all solid-state electrolytes. Starting from more than 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Li-containing experimental structures sourced from the Crystallography Open Database, Inorganic Crystal Structure Database and Materials Platform for Data Science, we perform highly automated calculations to identify electronic insulators. On these ∼1000 structures, we use molecular dynamics simulations to estimate Li-ion diffusivities using the pinball model, which describes the potential energy landscape of diffusing lithium with accuracy similar to density functional theory while being 200–500 times faster. Then we study the ∼60 most promising and previously unknown fast conductors using full first-principles molecular dynamics simulations at several temperatures to estimate their activation barriers. The results are discussed in detail for the 9 fastest conductors, including Li7NbO6, which shows a remarkable ionic conductivity of ∼5 mS cm−1 at room temperature. We further present the entire screening protocol, including the workflows where the accuracy of the pinball model is improved self-consistently, necessary for automatically running the required calculations and analysing their results.
000 Li-containing experimental structures sourced from the Crystallography Open Database, Inorganic Crystal Structure Database and Materials Platform for Data Science, we perform highly automated calculations to identify electronic insulators. On these ∼1000 structures, we use molecular dynamics simulations to estimate Li-ion diffusivities using the pinball model, which describes the potential energy landscape of diffusing lithium with accuracy similar to density functional theory while being 200–500 times faster. Then we study the ∼60 most promising and previously unknown fast conductors using full first-principles molecular dynamics simulations at several temperatures to estimate their activation barriers. The results are discussed in detail for the 9 fastest conductors, including Li7NbO6, which shows a remarkable ionic conductivity of ∼5 mS cm−1 at room temperature. We further present the entire screening protocol, including the workflows where the accuracy of the pinball model is improved self-consistently, necessary for automatically running the required calculations and analysing their results.
Broader contextSolid-state electrolytes have emerged as key components in the development of the next generation of energy storage devices. Their inherent safety and superior performance compared to conventional liquid electrolytes have attracted increased attention in the field of sustainable energy. Despite the tremendous attention, the design and discovery of a novel solid-state electrolyte with high Li-ion conductivity remain a significant challenge. While many structural families have been identified over the years, the progress has been slow and discovering new fast Li-ion conductors for solid-state electrolytes would have a major impact. Unlike all experimental procedures that can be human intensive, computational methods for automated discovery are readily parallelisable and require much fewer resources. Nevertheless, a computational strategy relying on full first-principles methods can be exceptionally expensive, and hence there is a need for methods that are sufficiently inexpensive to be able to run thousands of appropriate calculations while being accurate enough to yield meaningfully predictive results. This screening identifies fast Li-ion conductors by estimating Li-ion diffusivity using molecular dynamics simulations with the pinball model. This approach is typically two orders of magnitude faster than first-principles molecular dynamics simulations while retaining a similar level of accuracy. We emphasise that we exclusively study experimentally known materials, ensuring that the fast ionic conductors we suggest are actually synthesisable and ready for in-depth experimental investigation. |
1 Introduction
All-solid-state Li-ion batteries (ASSLBs) have been intensively studied1–3 particularly for applications in electric vehicles4,5 and mobile devices.6 This growing interest is primarily attributed to ASSLBs' higher energy densities and enhanced safety profiles compared to their conventional liquid counterparts.2,5,7 Besides this, ASSLBs' lightweight nature facilitates improved battery miniaturisation and easier assembly process,8 and they exhibit superior mechanical, thermal and electrochemical stability.9,10 Despite the significant attention ASSLBs have received, no known solid-state material satisfies all of the desirable requirements needed for their application, including high ionic conductivity.9,11 While many structural families have been identified, progress remains slow, underscoring the importance of searching new materials for ASSLBs.12,13In the past, materials discovery has relied on experimental approaches guided by chemical intuition.14 As a first example, phosphate based Li-containing materials were derived from NASICONs (Na Super Ionic CONductors)15,16 with the structural formula LiM2(PO4)3 (M = Ti, Zr).17,18 These so-called Li-NASICONs exhibit high Li-ion conductivity19 and continue to be the subject of ongoing research.20,21 Further examples include the gradual and systematic exploration of various inorganic families such as nitrides,22,23 halides,24,25 hydrides,26,27 perovskites with the general formula La3xLa2/3−xTiO328,29 and Li-argyrodites with the formula Li6PS5X (X = Cl, Br, I).30 A final example is the development of Li-containing garnet structures, with chemical composition Li5La3M2O12 (M = Ta, Nb), which were identified to be promising conductors, albeit with limited ionic conductivity.31 However, the chemical substitution with aliovalent ions led to the discovery of Li7La3Zr2O12, commonly known as LLZO, that demonstrates significantly higher ionic conductivity.32
The development of LLZO also serves as an example of chemical substitution in well-known ionic conductor families to explore the vast chemical space and identify new ionic conductors. Another example is the extensively studied family of LISICONs (Li-superionic conductors) with the formula Li14Zn(GeO4)4.33 Over time, numerous new LISICON-type materials were discovered,34–37 which can be represented by a more general formula of Li4XO4−Li3YO4 (X = Si, Ge, Ti; Y = P, As, V).14 LISICONs also serve as the precursors to the thio-LISICON family,38 which consists of a more polarisable sulphide anionic framework rather than an oxide sublattice, thereby enhancing their ionic conductivity.39 Further substitution of the cations led to the discovery of tetragonal-Li10GeP2S12 (LGPS),40 which is widely regarded as one of the best solid-state ionic conductors11 and has motivated the development of numerous promising derivative structures.41 To summarise, significant breakthroughs have primarily resulted from chemical intuition or by systematic substitution in known materials, motivated by the keen understanding of the underlying chemistry. Besides this, combinatorial methods42 and straightforward high-throughput experimental approaches43,44 have also contributed to the discovery of new superionic conductors, albeit with mixed success.
However, these experimental approaches do not scale as effectively as computational methods, which can be highly efficient in materials discovery by allowing for the exploration of a vast number of structural families within a short time frame (Fig. 1).45–47 Furthermore, computer simulations have primarily been limited to understanding the underlying diffusion mechanism, which in turns contributes to developing deeper chemical intuition. As a result, many computational screenings are typically motivated by established chemical knowledge, focusing on specific ion-conduction mechanisms or space-groups to propose new materials.48 For instance, Xiao et al.49 performed a computational screening motivated by the diffusion network in garnets and NASICON type conductors; Muy et al.50 explored all the possible doping strategies within the argyrodite family. In contrast, a screening approach that is agnostic to the underlying chemistry of structures can probe a much more expansive chemical space and potentially identify novel materials that have no apparent connection to the existing materials.
Consequently, it is essential to establish robust screening criteria motivated by physical properties to effectively identify the most suitable candidates for solid-state electrolytes. To prevent self-discharge in a battery, an SSE ought to exhibit low electron mobility, which is determined by the material's electronic band gap. The most accessible first-principles method for estimating band gaps is Kohn–Sham density functional theory (DFT).51,52 Although more advanced approaches, such as GW,53 Koopmans-compliant functionals,54 hybrid functionals,55 Hubbard-corrected DFT56 and many others,57 can yield band gap values that are predictive, these methods are significantly more computationally demanding compared to single point DFT calculation. Thus, for screening purposes, DFT offers a satisfactory balance between computational efficiency and accuracy for band gap estimates, despite its tendency to underestimate band gaps.58 This was also utilised by the screening studies of Muy et al.59 and Sendek et al.,60 who calculated band gaps at the level of DFT-PBE61 and applied a filtering criterion treating any material with a band gap greater than 1 eV as an insulator.
Electrochemical stability can be estimated using first-principles calculations as well,62 and it can be estimated in a high-throughput mode.63 While a broad electrochemical stability window is desirable for SSEs, many currently utilised electrolytes exhibit narrow stability windows.14 A notable example is LGPS which is stabilised with interphases and protective coatings.64,65 In the same vein, although low interfacial resistance and high interfacial compatibility between electrolytes and electrodes are important for optimum performance, higher resistance (and lower compatibility) can be mitigated by incorporating appropriate interfacial materials.65,66 Therefore, we emphasise that while electrochemical stability and interfacial compatibility are important considerations, they are not essential for a screening process, and thus, we have opted not to calculate these in this study.
Mechanical properties such as bulk and shear moduli can be readily obtained from simulations.67,68 However, the relevance of this information remains somewhat ambiguous. For instance, preventing or retarding the unwanted growth of Li-dendrites is achieved not merely through the use of a high-modulus material, but rather through defect engineering.69,70 Consequently, while bulk properties can be calculated easily, their utility as screening criteria is not well understood and as such we have chosen not to incorporate them in our study.
In summary, many challenges persist that limit the selection of materials for use as SSEs;71,72 still, achieving high ionic conductivity remains the most critical criterion.73,74 Ionic diffusion can be estimated from atomistic simulations directly through MD,75,76 with the accuracy dependent on the underlying potential energy surface (PES), which can be computed using empirical or machine-learned force-fields or using first-principles methods.77–79 While empirical force-fields may be sufficiently accurate to model Li-diffusion,37,80 they require precise fitting of the parameters to the specific system under consideration, which limits their applicability in exploring a vast chemical space. DFT can provide highly accurate and general PES applicable to a wide variety of chemical compositions. However, first-principles molecular dynamics (FPMD) in the Born–Oppenheimer81 approximation relies on performing single point DFT calculations at every MD step, rendering it prohibitively expensive.82 Another variant of FPMD, Car–Parrinello molecular dynamics,83 is computationally more efficient, but requires careful tuning to the system being studied. While this method can be highly useful for investigating diffusion mechanisms within a single system,84 it is non-trivial to calibrate its parameters across a multitude of systems. In addition to MD, ionic conductivity can be estimated in the simplest Arrhenius picture by calculating migration barriers for Li-diffusion, which can be obtained from inexpensive static calculations.85,86 However, identifying barriers is a highly complex task that often requires human intervention and is thus challenging to automate.87–89 Other methods attempt to link diffusion to more easily accessible properties: for example, the bond-valence method90 has been used to inexpensively calculate Li-ion conductivity in several independent screenings,91–93 though with limited accuracy due to the limitations of the method.94,95 Another approach involved deriving diffusion coefficients using specific phonon frequencies.96,97 In all cases, the aim to reduce computational costs goes directly against the requirement of reliable predictions across a broad range of materials.
In the past few years, universal machine learning interatomic potentials (MLIPs) have also emerged as an one-stop solution for running cheap and accurate MD simulations, including MACE-MP0,98,99 M3Gnet,100 CHGnet101 and the proprietary GNOME.102 These universal MLIPs are intended to be systems agnostic, can supposedly model most elements in the periodic table, and most importantly work out-of-the-box. Before deployment, their suitability needs to be thoroughly tested. Besides the initial applications, a few independent performance assessments of the universality have been performed.103–106 Both Yu et al.104 and Focassio et al.105 concluded that universal MLIPs are not yet accurate enough to reproduce first-principles results and show significant error in the estimation of properties under consideration. Both suggested that the current best use case is as a foundation onto which a more appropriate model can be trained. These shortcomings are also noted by the original authors.99 Nevertheless, these universal MLIPs promise a most promising way forward and are starting to be employed in high-throughput screenings.107,108
Besides universal MLIPs, several other powerful predictive models exist.109 The most common approach is to use descriptors to directly predict properties, like ionic conductivity, from the structures and or chemical phase space,59,110,111 by unsupervised or semi-supervised learning due to the lack of labelled data,112 or atypically by training directly on experimental data.113 Another approach that has garnered significant attention in the past year is inverse modelling, facilitated by artificial intelligence for materials discovery.102,114 These methods involve proposing hypothetical materials that may not necessarily be experimentally synthesisable.100,101 Nonetheless, predicting materials that are not merely synthesisable but also technologically relevant is highly non-trivial,115 which suggests that the underlying premise may require further examination.116 This stands in direct contrast to the present work, where we screen experimentally known materials whose synthesis recipes are known. It is important to note that several well-regarded screenings in the past few years59,110,117,118 also utilised structures from the same repositories as ours. However, our workflow was able to identify promising conductors that were not highlighted in those earlier efforts, underscoring the effectiveness of our approach.
We conclude this brief review of computational methods for modelling ionic diffusion by noting that screening fast Li-ion conductors remains a challenging undertaking. This difficulty arises either from the limited transferability and/or accuracy of descriptors, force-fields and universal MLIPs or due to the cost of first-principles approaches. Thus, accurately modelling the diffusion of Li-ions in a large-scale screening with MD simulations necessitates a computational approach that combines the low computational cost of force-fields with the precision and generality of DFT. In this study, this is achieved using the pinball model, which describes the potential energy surface of lithium diffusing in an SSE and is on average about 200–500 times faster than DFT, while offering often comparable accuracy.119 It is based on two key assumptions: (1) all Li atoms are completely ionised and are referred to as pinballs and (2) the host lattice (all non-Li atoms along with the valence electrons of Li atoms) is fixed at the equilibrium positions, and the charge density is frozen. The pinball model forms the backbone of our screening, as detailed in Section 2.3.1, enabling the identification of promising Li-ion conductors for further investigation using full first-principles simulations.
As a final note, we highlight a previous screening117 conducted using a similar framework based on the pinball model. The critical distinctions are as follows: (1) the present study utilises a more expansive database, encompassing over twice the number of structures; (2) we include non-local interactions within the pinball model; (3) we have implemented a self-consistent workflow that iteratively enhances the accuracy of the pinball model; (4) we apply more stringent criteria across all filtering parameters, for instance by tightening the tolerances used to compare crystal structures, we classify nearly 30% more structures as duplicates in this screening; (5) we use different functionals and pseudopotentials along with different sets of input parameters for all electronic structure calculations. These differences and the advantages they offer are described in more detail along with methods in Section 2, followed by a discussion of results in Section 3. Last, we summarise this screening and present our conclusions, followed by an outlook on the development of a universal machine learning potential to model Li-ion diffusion in Section 4.
2 Methods
Any computational screening of this magnitude requires a robust framework to automatically launch and monitor calculations, handle errors on-the-fly, and link data generated during calculations.120,121 Furthermore, it is necessary that this infrastructure explicitly preserves the provenance for easy reproducibility, queryability, and shareability of the results.122,123 To achieve this twofold goal of automating and managing complex workflows and storing full provenance of all related data, we used the Automated Interactive Infrastructure and Database for Computational Science (AiiDA), which is a Python-based infrastructure and workflow manager.46,124,125 The key advantage of AiiDA over other workflow managers lies in its ability to preserve the provenance of a calculation in its entirety. This includes storing the complete history of a calculation along with an exhaustive list of all inputs that led to the creation of that piece of data, as a directed acyclic graph within a relational database. This feature allows one to query any data point as a graph node in an easy to navigate fashion and assess the causal relationship between nodes. Fig. 2 illustrates this capability in an acyclic graph, taken from this work, that illustrates the entire screening path for one structure. This approach not only supports Open Science but goes beyond the well-known FAIR principle.126 Additionally, AiiDA facilitates a high degree of automation and parallelisation to easily run calculations on high-performance computing platforms, and every calculation in this screening is run using AiiDA.2.1 Preliminary filters
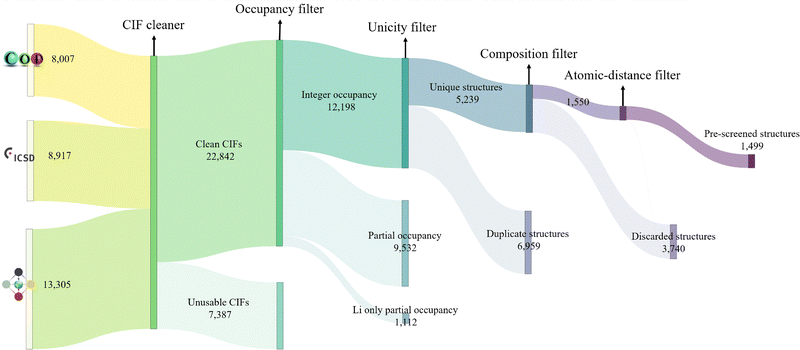
Starting from experimental structures sourced from the Crystallography Open Database (COD),127 Inorganic Crystal Structure Database (ICSD)128 and Materials Platform for Data Science (MPDS)129 repositories, we identify more than 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 lithium containing structures, which are imported as CIF files using AiiDA. These files sometimes contain syntax errors or extraneous information that require correction before they can be used. The issues and their corresponding solutions are comprehensively described in the work by Mounet et al.47 We follow that protocol to clean, parse and standardise CIF files using COD-tools.130–132 Finally, on the cleaned CIF files, we apply a sequence of filters to systematically narrow down the list of promising structures, as illustrated in Fig. 3.
000 lithium containing structures, which are imported as CIF files using AiiDA. These files sometimes contain syntax errors or extraneous information that require correction before they can be used. The issues and their corresponding solutions are comprehensively described in the work by Mounet et al.47 We follow that protocol to clean, parse and standardise CIF files using COD-tools.130–132 Finally, on the cleaned CIF files, we apply a sequence of filters to systematically narrow down the list of promising structures, as illustrated in Fig. 3.
We note that thus far we have conducted data analysis. The subsequent sections describe the final two filters wherein we perform electronic-structure calculations.
2.2 Electronic filters
To classify the filtered structures as electronic insulators, we calculate the band gap at the level of DFT. As a rule of thumb, we categorise structures with a band gap greater than 1 eV as electronically insulating. Generally, DFT underestimates the band gap for most materials.58 All DFT calculations are performed using the pw.x code from the Quantum ESPRESSO distribution,138,139 using experimental geometry, and with the PBEsol61,140 exchange–correlation functional. Pseudopotentials and their corresponding cut-offs are sourced from the Standard Solid-State Pseudopotential (SSSP) Efficiency 1.2.1 library,141 which provides comprehensive validation of pseudopotentials across various libraries and methods.142–147 For each SCF calculation, we use Marzari-Vanderbilt cold smearing148 and increase the number of bands by 20%, while the Brillouin zone is sampled with a Monkhorst–Pack grid of density 0.15 Å−1.Besides this, we perform variable-cell relaxation on about 20% of the structures to investigate the effects of geometry optimisation on band gap estimation.
2.3 Diffusivity filter
To run MD simulations, we generate supercells based on experimental geometries, ensuring a minimum separation of 8 Å between opposite faces, using the supercellor package.150 We run MD simulations with Born–Oppenheimer approximation81 in the canonical ensemble. Temperature is controlled with the stochastic velocity rescaling thermostat.151From the Einstein relation78 we can write the tracer diffusion coefficient Dtr as:
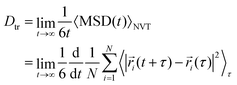 | (1) |
The tracer diffusion coefficient is related to the charge diffusion coefficient with Haven's ratio as H = Dtr/Dσ, which is a measure of correlated motion of the particles.152 In the dilute limit, we assume it to be 1, though in practice it is often less than 1, implying that correlated motion can enhance conductivity.87 Consequently, we do not overestimate conductivities. And from the Nernst–Einstein equation,153 we can calculate the ionic conductivity σ as:
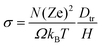 | (2) |
All analysis of trajectories including the calculation of MSD was done using the open-source tool Suite for analysis of molecular simulations (SAMOS).154
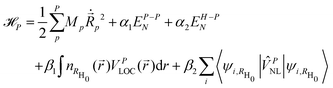 | (3) |
![[R with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0052_20d1.gif) and
and 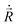 are respectively the positions and velocities of the pinballs i.e. the Li-ions, EA−BN is the electrostatic interaction between the frozen core electrons of species A and B, VPLOC/NL are the local and non-local external pseudopotential components of pinballs, which act on the charge density
are respectively the positions and velocities of the pinballs i.e. the Li-ions, EA−BN is the electrostatic interaction between the frozen core electrons of species A and B, VPLOC/NL are the local and non-local external pseudopotential components of pinballs, which act on the charge density 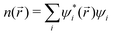 , which is frozen for the host lattice H0. The final term is responsible for non-local interactions, which further improves the accuracy of the model with additional computational cost. α1, α2, β1 and β2 are phenomenological coefficients (referred to as pinball coefficients) introduced to further improve the accuracy that can be computed by fitting the pinball forces with DFT forces.
, which is frozen for the host lattice H0. The final term is responsible for non-local interactions, which further improves the accuracy of the model with additional computational cost. α1, α2, β1 and β2 are phenomenological coefficients (referred to as pinball coefficients) introduced to further improve the accuracy that can be computed by fitting the pinball forces with DFT forces.
For this screening, we designed and implemented a highly automated and powerful workflow in AiiDA as a plugin called aiida-flipper.149 All supercells are passed to the diffusion workflow, which iteratively runs MD simulations with the pinball Hamiltonian and self-consistently refines the pinball coefficients, thereby progressively enhancing the accuracy in determining Li-ion conductivity. Fig. 4 illustrates the details of the workflow. This workflow represents a significant improvement with respect to the previous pinball screening,117 as it ensures the convergence of the pinball coefficients and thus improves the quality of the forces.
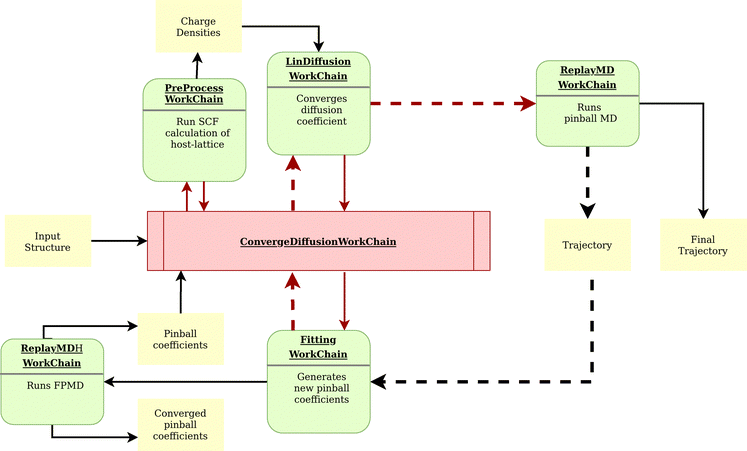 | ||
Fig. 4 A schematic representation of the self-consistent workflow of AiiDA-flipper,149 the python package employed to run MD simulations using the pinball model119 and compute ionic conductivity of lithium. The nomenclature depicted corresponds exactly to the Python classes within the plugin. The ConvergeDiffusion workchain initiates the process by launching the PreProcess workchain, which runs a single point calculation and stores the charge densities of the host lattice to be used in all subsequent pinball MD simulations. Next, the Fitting workchain is launched, generating sufficient snapshots with random displacement of Li-ions in the supercell to fit 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 force components through calculations at both the pinball and DFT levels. This initial estimate of pinball coefficients is then used to initiate the LinDiffusion workchain, which runs a long MD simulation at the pinball level to converge the diffusion coefficient to a predetermined threshold. From the trajectory of this MD run, uncorrelated configurations are extracted, and a new set of pinball coefficients are derived through linear regression of the DFT and pinball forces. This iterative cycle continues self-consistently until the pinball coefficients converge. Once convergence is achieved, a final MD simulation is performed using the converged pinball coefficients, and the final MD trajectory is used to compute the diffusion coefficient. This workflow ensures accurate and reliable computation of ionic conductivity, leveraging the self-consistent refinement of pinball coefficients through iterative MD simulations and force component fitting. 000 force components through calculations at both the pinball and DFT levels. This initial estimate of pinball coefficients is then used to initiate the LinDiffusion workchain, which runs a long MD simulation at the pinball level to converge the diffusion coefficient to a predetermined threshold. From the trajectory of this MD run, uncorrelated configurations are extracted, and a new set of pinball coefficients are derived through linear regression of the DFT and pinball forces. This iterative cycle continues self-consistently until the pinball coefficients converge. Once convergence is achieved, a final MD simulation is performed using the converged pinball coefficients, and the final MD trajectory is used to compute the diffusion coefficient. This workflow ensures accurate and reliable computation of ionic conductivity, leveraging the self-consistent refinement of pinball coefficients through iterative MD simulations and force component fitting. | ||
We perform both pinball MD and FPMD on the experimentally reported crystal structures available in crystallographic databases, i.e., idealised ordered structures derived from CIF files. However, real materials often deviate from these ideal structures through point defects, non-stoichiometry, partial occupancies, disorder, grain boundaries, or local structural distortions, all of which can affect ion conduction.157–160 In many cases, such deviations may enhance Li mobility, for example, the introduction of Li vacancies or aliovalent substitutions can create additional diffusion pathways or lower migration barriers, potentially transforming a poor conductor in its ideal stoichiometric form into a significantly better conductor in practice.161 However, a systematic treatment of such effects in a screening study of this scale is highly non-trivial,50 and as such we consider this a promising avenue for future screenings, but ultimately outside the scope of present screening.
3 Results and discussion
The pre-screening phase, which does not involve any electronic structure calculations, is illustrated in Fig. 3. Starting with approximately 8000, 9000, and 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 experimental structures sourced from COD,127 ICSD,128 and MPDS129 respectively, we extract nearly 23
000 experimental structures sourced from COD,127 ICSD,128 and MPDS129 respectively, we extract nearly 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 clean CIF files, discarding the unsalvageable ones. All subsequent filters are applied to structures derived from these clean CIF files. We eliminate approximately 10
000 clean CIF files, discarding the unsalvageable ones. All subsequent filters are applied to structures derived from these clean CIF files. We eliminate approximately 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 structures with partial occupancies, and from the remaining 12
600 structures with partial occupancies, and from the remaining 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 structures with integer atomic occupancies, 5200 are identified as unique using the structure matcher algorithm of pymatgen.137 Further filtering removes structures containing unwanted elements and those with unwanted bond lengths, resulting in 1499 structures that advance to the next phase of the screening.
000 structures with integer atomic occupancies, 5200 are identified as unique using the structure matcher algorithm of pymatgen.137 Further filtering removes structures containing unwanted elements and those with unwanted bond lengths, resulting in 1499 structures that advance to the next phase of the screening.
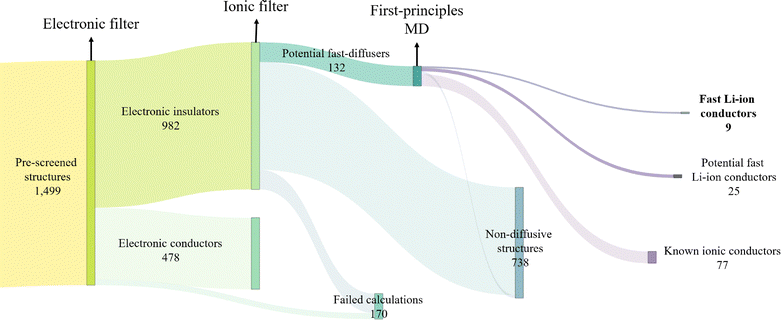
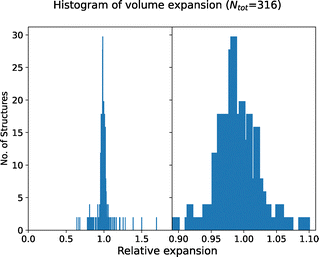
We perform single-point calculations on these structures at the level of DFT-PBEsol.140 Out of these, 251 calculations fail to converge due to issues in the self-consistent electronic cycle. These are subsequently rerun using the non-linear conjugate gradient method within SIRIUS162 enabled Quantum ESPRESSO. Following this, we calculate the band gap for all structures and classify a structure as an electronic insulator if its band gap exceeds 1 eV. Out of the 1499 unique structures, 982 are identified as electronic insulators, and 39 calculations failed, representing the first filter illustrated in Fig. 5. To assess the impact of geometry optimisation on our filtering criterion, we performed additional variable-cell relaxation on 25% of these 1499 structures, of which 316 finished successfully. Fig. 6 compares the band gaps between relaxed and experimental geometries. Our findings indicate that only 5 out of the 316 structures, or less than 2%, are identified as insulators when calculated using experimental geometry instead of performing full variable-cell relaxation, representing false positive results. The reverse scenario, where metallic structures turn into insulators upon relaxation (false negatives), is slightly more common with 10 out of 316 structures. Given that all MD simulations are performed at experimental geometries, we opt not to relax any other structures, considering a less than 2% false positive rate acceptable given the significantly higher computational cost of variable-cell relaxation and the additional failure due to issues in the ionic convergence cycle.
 | ||
| Fig. 6 Comparison of band gaps at optimised geometry (Vrel) and experimental geometry (Vexp). For the majority of the structures, the classification as insulators remains unchanged upon relaxation. | ||
Fig. 7 presents a histogram of the relative volume change upon geometry optimisation, defined as the optimised volume divided by the experimental volume. Utilising the PBEsol functional, we achieve a narrow and uniform distribution of volume changes, maintaining lattice parameters that more closely match experimental values. This contrasts with the standard PBE functional,61 where structures are more likely to exhibit expansion,163 as observed by Kahle et al.117
3.1 Pinball MD
All the MD simulations are performed on the supercells generated from the 982 insulators identified in the previous step. To evaluate the significance of including non-local interactions within the pinball model, we conducted tests on a few systems both with and without non-local interactions. The MSD plots of this comparison, shown in Fig. 8, reveal that using only local projectors typically leads to an underestimation of Li-ion diffusion. Consequently, we opt to include it in our screening, despite the higher computational cost. | ||
| Fig. 8 MSD plot of two materials comparing Li-diffusion with and without non-local interactions within the pinball model at 1000 K. Using only local projectors typically leads to an underestimation of Li-ion diffusion. The r2 coefficient of determination for the pinball forces improves from 0.83 to 0.99 for Li3Y(PS4)2 and from 0.91 to 0.99 for Li2CsI3 when non-local interactions are included. Based on first-principles simulations Li3Y(PS4)2 and Li2CsI3 show ionic conductivities of 2.16 mS cm−1 at 300 K164 and 0.22 mS cm−1 at 500 K117 respectively. | ||
Next, we derive an initial estimate of the pinball coefficients through the linear regression of forces calculated at both DFT and pinball levels for all supercells. The quality of these coefficients is evaluated using the r2 correlation between DFT and pinball forces. We ensure that the r2 correlation for the converged pinball coefficients exceeds 0.95, with the majority of cases exceeding 0.99. If this criterion is not met, additional self-consistent pinball MD iterations are performed, allowing for the extraction of further uncorrelated configurations from these extended MD simulations. These serve as additional data points for improving the fit until full convergence is achieved, as indicated by stable pinball coefficients and an r2 value approaching 1. Out of 982 structures, we achieve convergence for 914, with failures occurring due to issues in the self-consistent electronic cycle when calculating DFT forces. An additional 63 structures failed the pinball MD simulations due to drift in the constant of motion, leading to 851 structures with a final iteration of the pinball MD run with converged coefficients and a total simulation time of 22.1 µs. As illustrated in Fig. 9, the pinball coefficients readily converge for most structures. Based on the slope of the MSD plot from the final MD iteration and eqn (2), we estimate Li-ion conductivity. An ionic conductivity of 1 mS cm−1 at 1000 K is chosen as the threshold to categorise potential fast ionic conductors at the pinball level.
 | ||
| Fig. 9 Self-consistent iterations for the MSD plots of Li, along with the convergence of pinball coefficients for several fast Li-ion conductors. The zeroth pinball coefficients are derived by fitting DFT and pinball forces on randomly rattled structures, which are then used to perform the first MD iteration. Force fitting is subsequently performed on configurations obtained from the first MD iteration to obtain the first pinball coefficients, which are then used in the second MD iteration. For most structures pinball coefficients converge after two of these self-consistent iterations, with the estimate of Li-diffusion remaining largely unchanged, illustrating the need for a self-consistent cycle. For a select few structures, additional iterations are performed after the convergence of the pinball parameters to verify that no divergence occurred in subsequent steps, ensuring the robustness of the workflow. We attribute the slight change in dynamics in some of the MD simulations to the inherent stochasticity of the thermostat that is used.151 | ||
At the end of this process, 132 structures are identified for further studies using first-principles calculations. Of these 132 structures, 49 originate from the newly considered MPDS repository, while the remaining structures were present in the overlapping datasets in the previous screening.117 We attribute the inclusion of non-local interactions within the pinball model along with the self-consistent fitting of the pinball coefficients as the main reasons for the increased predictive power of current workflow over the previous screening.
3.2 First-principles MD
We classify the 132 structures obtained from the self-consistent pinball workflow into four categories: (1) structures already identified in the literature as Li-ion conductors, (2) structures that do not exhibit diffusion within FPMD, (3) structures that show negligible diffusion at lower temperatures but may still be of interest, and (4) fast Li-ion conductors.In the following short review, we report these 77 structures and their current use case if applicable. Li2Ti6O13 is a known ionic conductor165 and was recently proposed as a cathode material,166 while sodium substituted Li2Ti6O13 is used as an intercalation anode.167 Li7P3S11 is a well-known superionic conductor.168,169 Li2TeO4 is a known superionic conductor170 and was proposed as an electrode material.171 Li6NBr3 was experimentally shown to be a fast ionic conductor,172 but worse than the well known Li3N,173,174 which we also identified. LiI is a well known ionic conductor,175 while LiBr and LiCl show negligible Li-ion conduction without doping.176 Li2Se is used as a cathode material177 and also as an interface material.178 Li3BN2 is a well known fast ionic conductor.179 Li3BS3 was reported in the computational screening by Laskowski et al.,180 despite an earlier computational study181 that proposed it as a potential ionic conductor. Li4SnSe4 is a known ionic conductor.182 Li2SiN2 is used as an anode material183 and Li anode coatings are used to increase electrochemical stability.184 Li2SiP2 is a known ionic conductor185 and was proposed as a potential solid-state electrolyte material.186 Li2SiS3 is a known ionic conductor.187,188 LiBF4 has been used as non-aqueous electrolyte for two decades.189 Li3BrO is a known superionic conductor.190,191 Li3Y(PS4)2 was proposed in a computational study with very high ionic conductivity.164 Li3PS4 is a known ionic conductor192 and has been engineered with much better properties in the past decade.193 Li4PN3 was recently discovered using first principles simulations.194,195 Li5AlS4 was experimentally reported to have low ionic conductivity at room temperature,196 but is otherwise known in the argyrodite family.197 Li5NCl2 has been known to be an ionic conductor for a long time,198 but was recently studied in greater detail by Landgraf et al.199 Li7BiO6 has been known for a long time to be an ionic conductor.200 LiGa(SeO3)2 was proposed recently by Jun et al.118 LiHf2(PO4)3 is a known ionic conductor,201,202 but Al substituted Li1+xAlxHf2−x(PO4)3 showed more promise.203 LiInS2 is a known ionic conductor204 and was recently studied within the LiXS2 family as a cathode material.205 LiS206 is a part of the Li–S battery system, while Li2S is used as a cathode material.207 Li3InO3 is a known ionic conductor.208 LiZnPS4 is a poor ionic conductor, but with defect engineering shows more promise.209,210 LiTi2(PO4)3 is used as a cathode material in aqueous batteries especially when doped as LiMnxTi2−x(PO4)3;211,212 further doping has yielded promising results as an SSE.213 LiNbO3 is used as a coating on cathode materials214 and also as an anode material in Li-ion capacitors.215 LiAlCl4 is not well studied, despite being a known ionic conductor for a long time.216 Li2O is a well known ionic conductor.73,217 Li2Mo4O13 was recently proposed as an anode material.218 LiSn2(PO4)3 is a well known anode material with various different synthesis methods.219–221 Li4SnS4 is a known ionic conductor.222 Li9S3N is a known ionic conductor223 and was proposed as a barrier coating between electrolyte and the Li metal anode.224 Li4GeS4 is a well known ionic conductor.225 LiCF3SO3 is a known ionic conductor along with sodium, caesium and rubidium substitutes.226,227 Li5NBr2 and Li10N3Br were investigated recently in the halogen-nitride system Li3a+bNaXb, with Li10N3Br found to be an excellent ionic conductor.228 Li3In2(PO4)3 is a known superionic conductor.229 Li2B6O9F2 is a known ionic conductor.230 Li2SrTa2O7 is a known ionic conductor but other substitution compounds are more promising.231 Li7SbO6 is a known ionic conductor.232 Besides this, Kahle et al.117 proposed the following as fast Li-ion conductors: Li5Cl3O, Li7TaO6, LiGaI4, LiGaBr3, and Li3CsCl4 and Li2CsI3, which are theoretical structures,233 Li2WO4, which is used to improve conductivity of other materials either as a solid mixture234 or in solid solution,235 and LiAlSiO4, whose suitability was systematically studied with Al doping by Ryu et al.236 Last, FPMD simulations performed by Kahle et al.117 showed insignificant diffusion in the following structures at lower temperatures: LiAlSe2, Li4Re6S11, LiPO3, Li3Sc2(PO4)3, Li4P2O7, (LiI)2Li3SbS3, Li6PS5I, Li5P(S2Cl)2, Li3P7, Li3SbS3, Li2B3O4F3, Li2Mg2(SO4)3, Li3AsS3, Li2Si2O5, Li2NaB(PO4)2, Li6Y(BO3)3, and LiAuF4.
| Structure | Database | Database-id |
|---|---|---|
| Li2Te2O5 | ICSD | 26451, 26452237 |
| Li2CsCl3 | MPDS | S1022277238 |
| LiKSe | ICSD | 67277239 |
| LiYS2 | MPDS | S537670240 |
| LiInSe2 | MPDS | S1214509241 |
| LiAlS2 | ICSD | 608360242 |
| LiLuS2 | MPDS | S307222243 |
| Li7Te3O9F | MPDS | S1533619244 |
| Li5SiP3 | MPDS | S1145472245 |
| Li6RbBiO6 | MPDS | S1408313246 |
| LiAuF6 | MPDS | S1904723247 |
| Li3Na3Ga2F12 | MPDS | S1836948248 |
| LiZrS2 | MPDS | S301115249 |
| Li2CdSnSe4 | MPDS | S1952801250 |
| LiBa4Ga5Se12 | MPDS | S1021504251 |
| Li3Na3Rh2F12 | MPDS | S307582252 |
| Li2HgO2 | MPDS | S1702887253 |
| Li2Ca2Ta3O10 | ICSD | 88497254 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times greater than that of the tetragonal phase,255 corresponding to an MSD of 0.01 Å2 ps−1. Therefore, simulations with durations of 100–200 ps are insufficient to accurately resolve diffusion in such systems. Consequently, the activation barriers that we report may not be entirely accurate due to lack of sufficient statistics at lower temperatures. Similar to the previous section, we observe materials that include oxides, halides, phosphides and additional nitrides.
000 times greater than that of the tetragonal phase,255 corresponding to an MSD of 0.01 Å2 ps−1. Therefore, simulations with durations of 100–200 ps are insufficient to accurately resolve diffusion in such systems. Consequently, the activation barriers that we report may not be entirely accurate due to lack of sufficient statistics at lower temperatures. Similar to the previous section, we observe materials that include oxides, halides, phosphides and additional nitrides.
| Structure | Database | Database-id | Bandgap | Ionic conductivity | Ionic conductivity |
|---|---|---|---|---|---|
| DFT-PBEsol (eV) | Pinball (mS cm−1) | FPMD (mS cm−1) | |||
| Li2BeF4 | MPDS | S1935520256,257 | 7.49 | 10 | 1822 |
| Li2Ti4O9 | MPDS | S559372258 | 3.2 | 1042 | 1348 |
| LiY2Ti2S2O5 | COD | 4124533259 | 1.25 | 74 | 1251 |
| Li10BrN3 | MPDS | S1614518260 | 1.79 | 4247 | 886 |
| Li2Cs3Br5 | ICSD | 245978233 | 3.79 | 106 | 594 |
| Li8SeN2 | MPDS | S1931016261 | 1.88 | 467 | 588 |
| Li8TeN2 | MPDS | S1931019261 | 2.28 | 214 | 446 |
| LiCF3SO3 | ICSD | 110018262 | 6.78 | 40 | 384 |
| Li2ZnBr4 | COD | 1517836263 | 3.75 | 8 | 357 |
| LiBeP | ICSD | 670551,264 42037265 | 2.75 | 18 | 356 |
| Li5Br2N | ICSD | 78836260 | 2.29 | 1351 | 344 |
| Li10Si2PbO10 | ICSD | 78326266 | 2.86 | 29 | 342 |
| Li2ZnGeSe4 | COD | 7031897267 | 1.89 | 63 | 291 |
| LiCs2I3 | ICSD | 245984233 | 3.41 | 1410 | 280 |
| LiSr2Br5 | MPDS | S1941469268 | 3.53 | 176 | 263 |
| LiGaSe2 | COD | 1531591269 | 2.23 | 5 | 173 |
| LiP7 | ICSD | 23621270 | 1.56 | 11 | 133 |
| LiMoPO6 | COD | 7701361271 | 2.52 | 8 | 132 |
| LiY(MoO4)2 | COD | 1008103272,273 | 3.22 | 660 | 68 |
| Li10B14Cl2O25 | MPDS | S1803375274 | 6.30 | 13 | 65 |
| Li2P2PdO7 | COD | 1000333275 | 1.39 | 513 | 485 |
| Li2B3PO8 | MPDS | S1614518276 | 5.49 | 45 | 41 |
| Li2B2Se5 | COD | 1510746277 | 1.84 | 84 | 20 |
| Li8Bi2(MoO4)7 | ICSD | 54021278 | 2.95 | 7 | 12 |
| Li3AuS2 | COD | 4319430274 | 1.86 | 895 | 8 |
3.2.3.1 Li10Si2PbO10. Originally synthesised in 1994 by Brandes et al.,266 this material has received limited attention, particularly in the context of fast Li-ion conduction. Lead-silicate glasses, including this compound, are known for diverse optical properties, such as transparency, refractive index, colouration, electrical conductivity, and chemical durability279 and have found application in areas such as the monitoring of radioactive materials.280 However, their potential as solid-state electrolytes remains largely unexplored. In our simulations, Li10Si2PbO10 displays Li-ion diffusion at lower temperatures, as shown in Fig. 10; yet, its relatively high activation energy of 0.35 eV results in an estimated room-temperature ionic conductivity of less than 0.1 mS cm−1, limiting its viability as a potential electrolyte material.
 | ||
| Fig. 10 MSD plot of Li along with host-lattice species of Li10Si2PbO10 and Li2B3PO8 at 500 K from FPMD. | ||
3.2.3.2 Li2B3PO8. Synthesised relatively recently in 2014 by Hasegawa et al.,276 this material has yet to receive significant attention, particularly in the context of Li-ion batteries. Borophosphates of this kind have primarily been investigated in the semiconductor industry for their magnetic coupling mechanisms, optical characteristics, and catalytic behaviour.281,282 Similar to Li10Si2PbO10, our simulations are able to resolve Li-ion diffusion at lower temperatures for Li2B3PO8, as shown in Fig. 10; the relatively high activation energy of 0.28 eV renders it less suitable for room-temperature applications, where the estimated ionic conductivity falls well below 0.1 mS cm−1.
3.2.3.3 Li2BeF4. This material was first synthesised in 1952 by Novoselova et al.256 and exhibits one of the highest Li-diffusion at elevated temperatures. However, due to the inability to resolve diffusion at lower temperatures, accurately quantifying its activation barrier remains challenging. We anticipate that with sufficiently long simulations, on the order of several nanoseconds, it would be possible to quantify diffusion at lower temperatures as well, making this material an excellent candidate for further investigation using machine learning techniques. Furthermore, it has notably been used as a coolant in nuclear reactors,283 highlighting the established interest in its synthesisability within experimental settings. Given these factors and the toxic nature of beryllium, it remains an interesting case study.
3.2.3.4 Li8SeN2 and Li8TeN2. Both selenium and tellurium nitrides, which demonstrate excellent Li-ion diffusion at higher temperatures, were first synthesised in 2010 by Bräuling et al.261 They are three-dimensional diffusers, but at lower temperatures they do not show high diffusion. Furthermore, nitrides are generally among the most challenging materials to process due to relatively high-temperature synthesis routes;284 we refrain from classifying them as the most promising candidates within this screening.
 | ||
| Fig. 11 Diffusion coefficients derived from our FPMD simulations for the most promising oxides and nitrides. The dashed line represents the best-fit line, with the slope corresponding to the activation barriers, indicated in brackets (eV). We additionally show LGPS for comparison, with data taken from the work of Kahle et al.117 | ||
 | ||
| Fig. 12 Diffusion coefficients derived from our FPMD simulations for the most promising halides. The dashed line represents the best-fit line, with the slope corresponding to the activation barriers, indicated in brackets (eV). We additionally show LGPS for comparison, with data taken from the work of Kahle et al.117 | ||
| Structure | Database | Database-id | Bandgap | Ionic conductivity | Ionic conductivity | Ionic conductivity | Activation energy |
|---|---|---|---|---|---|---|---|
| DFT-PBEsol (eV) | at 500 K (mS cm−1) | at 750 K (mS cm−1) | at 1000 K (mS cm−1) | (eV) | |||
| Li4CO4 | ICSD | 245389286 | 5.26 | 235 | 551 | 726 | 0.15 |
| LiCsI2 | ICSD | 245986287,288 | 3.32 | 203 | 340 | 698 | 0.16 |
| Li3CsBr4 | ICSD | 245982233 | 3.73 | 456 | 1035 | 1616 | 0.17 |
| Li3Cs2Br5 | ICSD | 245980233 | 3.90 | 181 | 358 | 844 | 0.18 |
| Li7NbO6 | MPDS | S1818764289 | 3.58 | 77 | 288 | 418 | 0.21 |
| Li3Cs2I5 | ICSD | 245987290,291 | 3.43 | 161 | 554 | 1112 | 0.23 |
| LiCs3Cl4 | ICSD | 245969233,292 | 4.38 | 35 | 112 | 283 | 0.23 |
| Li4Mo3O8 | MPDS | S1614518293 | 1.17 | 6 | 32 | 54 | 0.25 |
| Li5NaN2 | ICSD | 92313294 | 1.49 | 279 | 1268 | 3609 | 0.28 |
3.2.4.1 Li7NbO6. First synthesised in 1969,289 this material has been the subject of multiple experimental studies;232,295 however it has never been investigated as a Li-ion conductor until He et al. proposed Li7NbO6 as a potential ionic conductor.296 Subsequent computational work by Feng et al. reported a low ionic conductivity of 0.008 mS cm−1 along with a significantly higher activation barrier and lower diffusion than our predictions,297 as illustrated in Fig. 13. We note, however, that our study and that of Feng et al. address two distinct structural phases of the same composition. Our calculations indicate that the phase considered in the present work is higher in total energy by approximately 1 eV per formula unit relative to the phase reported by Feng et al., suggesting that it is metastable within our DFT framework, despite having been reported experimentally. This makes the Li7NbO6 system particularly interesting from a materials-design perspective, since it raises the possibility that the high-conductivity phase identified here could be stabilised under suitable synthesis conditions or through chemical substitution. In this context, Feng et al. also investigated tungsten doping and found an enhancement of the room temperature conductivity to 0.28 mS cm−1, which remains an order of magnitude lower than our estimated value of 5 mS cm−1. Nevertheless, these findings support doping as an effective strategy to further improve the ionic conductivity. Given these promising properties and the substantial experimental background already established, we propose that Li7NbO6 holds significant potential as an excellent electrolyte for future applications.
 | ||
| Fig. 13 MSD plot of Li along with host-lattice species of the oxides Li7NbO6, Li4Mo3O8, and Li4CO4, and the nitride Li5NaN2 at 600 K from FPMD. | ||
3.2.4.2 Li4Mo3O8. This molybdenum oxide exhibits high Li-ion conductivity, as illustrated in Fig. 13. The yttrium-doped variant was first synthesised in 1980, while the undoped form was synthesised in 1999. Our FPMD simulations indicate that both materials possess low activation barriers, with the yttrium-doped version performing slightly better. Based on the activation energies, we estimate the ionic conductivities at room temperature to be 0.2 mS cm−1. We strongly recommend further experimental studies to validate these findings and confirm the potential as solid-state electrolytes.
3.2.4.3 Li5NaN2. The well known Li3N was first proposed in 1935298 and has since spawned a broad class of Li-ion conductors that continue to attract attention today.299 While studying Li3N in 2000, Schön et al. proposed the metastable Li5NaN2, as a derivative of Li3N, with relatively low formation energy.294 Our calculations are able to resolve Li-ion diffusion at lower temperatures as shown in Fig. 13 and indicate a relatively higher activation barrier of 0.28 eV, which corresponds to an estimated room-temperature ionic conductivity of 4 mS cm−1. Given the success of doping in enhancing the conductivity of Li3N,300,301 we are optimistic that similar strategies could improve the performance of Li5NaN2, making it a promising candidate. However, a notable challenge is the concurrent diffusion of Na-ions, highlighting the need for targeted compositional or structural engineering to restrict Na-ion mobility.
3.2.4.4 Li4CO4. We examined this material in four distinct crystal structures with the same stoichiometry, all of which exhibited excellent Li-ion diffusion. However, this material remains a theoretical structure that exists at high pressure and appears to simply be a variant of Li-doped carbonates, which may decompose at ambient temperature and pressure.286 Given these uncertainties, we are cautious about its potential as an electrolyte. Despite its low activation barrier, we have opted not to list it as a promising candidate until further validation can be conducted, and it is established that these materials can exist at normal temperature and pressure without decomposing into simple carbonates. Based on the activation energy, we estimate the ionic conductivity at room temperature to be 37 mS cm−1.
3.2.4.5 Cs-containing halides. Amongst the most promising materials we identify are Li3CsBr4, Li3Cs2Br5, LiCs3Cl4, LiCsI2 and Li3Cs2I5, some of which were first proposed by Pentin et al. using ab initio methods.233 Each of these materials demonstrates high ionic conductivity as shown in Fig. 14–16; and low activation barrier ranging from 0.15 to 0.25 eV as illustrated in Fig. 12. Although experimental validation for these materials is still pending, their synthesis appears to be feasible. Most notably, LiCsI2,288 Li3Cs2I5291 and Li2CsI3290 (which was also proposed by Kahle et al.117) have all been successfully synthesised, and Li3Cs2Br5 may also be synthesised following a similar approach to Li3Cs2I5. While these compounds have not yet been explored as ionic conductors, our screening suggests significant potential for future experimental validation. Though the synthesisability of the other Cs–Li–halides remains uncertain, they may depend on methodologies similar to those used for this ternary system.290 Given these considerations, we hesitate from designating these materials as the top candidates within this screening, pending further experimental investigations. However, it is important to emphasise that despite these uncertainties, this system represents a promising avenue for further exploration and warrants both experimental and theoretical pursuits.
 | ||
| Fig. 14 MSD plot of Li along with host-lattice species in Cs-containing chloride at 600 K from FPMD. | ||
 | ||
| Fig. 15 MSD plot of Li along with host-lattice species in Cs-containing bromides at 600 K from FPMD. | ||
As a final validation of the pinball model, we compare the diffusion coefficients obtained from FPMD and pinball MD for the structures discussed in Sections 3.2.2–3.2.4, as shown in Fig. 17. At first glance, first-principles diffusion is not quantitatively reproduced by the pinball model, which generally tends to overestimate the diffusion coefficient. This systematic bias is consistent with the underlying approximations of the method, in particular the frozen host-lattice treatment, which neglects lattice dynamics and the feedback of the mobile Li ions on the host sublattice.119 While the empirically observed false-positive rate among the top candidates (that were subsequently validated by FPMD) is low, the same cannot be said about the false-negatives, since there are examples of materials that are classified as non-conducting at the level of pinball MD that might be conducting,302 which underscores the difficulty in quantifying the true predictive power of the pinball model. A more thorough discussion of false positives and negatives can be found in ref. 117. Considering that the pinball workflow successfully rediscovered 77 known ionic conductors, the number of false positives appears to be low, setting an upper bound of ∼85% on the overall predictive rate of our workflow.
4 Conclusions and outlook
We conducted a high-throughput computational screening of over 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 lithium containing experimental structures sourced from the MPDS, ICSD, and COD repositories. Through the application of several structural filters, we identified approximately 1500 unique crystal structures suitable for electronic structure calculations. We determined the band gaps for these structures at the level of DFT with the PBEsol functional and identified nearly 1000 as electronic insulators. To investigate Li-ion diffusion, we implemented a self-consistent MD workflow in AiiDA, utilising the computationally efficient and highly accurate pinball model. From these simulations, we identified 132 fast Li-ion diffusers, 77 of which were previously recognised in the literature as Li-ion conductors. The remaining 55 materials were further examined using full first-principles MD simulations, leading to the discovery of seven promising materials, including the oxides LiY(MoO4)2, Li4Mo3O8 and Li7NbO6, the nitrides Li8SeN2 and Li8TeN2, and Cs-containing iodides LiCsI2 and Li3Cs2I5. These materials demonstrated excellent activation barriers and Li-ion diffusion near room temperature comparable to or exceeding that of LGPS, a well-known Li-ion superconductor. However, it is important to note that this estimation is based on the extrapolation of the Arrhenius plot, where a change of slope is possible. Additionally, we identified five other materials with similar levels of ionic conductivity, although their synthesisability remains uncertain. Furthermore, we identified 25 potential fast Li-ion conductors, including Li2BeF4 and Li8SeN2, that exhibit high Li-ion diffusion at elevated temperatures. However, due to the limited timescales accessible to first-principles MD simulations, we were unable to resolve their diffusion behaviour at lower temperatures. These materials may be promising candidates for further study using machine learning techniques, which could enable more extended simulations at lower temperatures.
000 lithium containing experimental structures sourced from the MPDS, ICSD, and COD repositories. Through the application of several structural filters, we identified approximately 1500 unique crystal structures suitable for electronic structure calculations. We determined the band gaps for these structures at the level of DFT with the PBEsol functional and identified nearly 1000 as electronic insulators. To investigate Li-ion diffusion, we implemented a self-consistent MD workflow in AiiDA, utilising the computationally efficient and highly accurate pinball model. From these simulations, we identified 132 fast Li-ion diffusers, 77 of which were previously recognised in the literature as Li-ion conductors. The remaining 55 materials were further examined using full first-principles MD simulations, leading to the discovery of seven promising materials, including the oxides LiY(MoO4)2, Li4Mo3O8 and Li7NbO6, the nitrides Li8SeN2 and Li8TeN2, and Cs-containing iodides LiCsI2 and Li3Cs2I5. These materials demonstrated excellent activation barriers and Li-ion diffusion near room temperature comparable to or exceeding that of LGPS, a well-known Li-ion superconductor. However, it is important to note that this estimation is based on the extrapolation of the Arrhenius plot, where a change of slope is possible. Additionally, we identified five other materials with similar levels of ionic conductivity, although their synthesisability remains uncertain. Furthermore, we identified 25 potential fast Li-ion conductors, including Li2BeF4 and Li8SeN2, that exhibit high Li-ion diffusion at elevated temperatures. However, due to the limited timescales accessible to first-principles MD simulations, we were unable to resolve their diffusion behaviour at lower temperatures. These materials may be promising candidates for further study using machine learning techniques, which could enable more extended simulations at lower temperatures.
Finally, we expect that the extensive first-principles data generated through this study will play a crucial role in training the next generation of machine learning interatomic potentials (MLIPs). To facilitate this, we have made all our first-principles data, along with comprehensive provenance, publicly available on the open-source Materials Cloud archive platform.303 This dataset could be particularly instrumental in developing a “universal-Li” MLIP, which has the potential to unlock new and intriguing systems in the future and serve as a foundational tool for the study of next-generation solid-state Li-ion batteries.
Author contributions
T. S. T.: conceptualisation, data curation, formal analysis, methodology, software, validation, visualisation, writing – original draft; L. E.: methodology, software; N. M.: funding acquisition, project administration, supervision; all authors: writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article, including first-principles molecular dynamics trajectories and input crystal structures are available at Materials Cloud Archive at: https://doi.org/10.24435/materialscloud:xm-46.The code for running all the calculations can be found at: https://github.com/epfl-theos/aiida-flipper. The version of the code employed for this study is version 1.4.
Supplementary information (SI) provides MSD plots for all materials studied using FPMD simulations, and is available. See DOI: https://doi.org/10.1039/d5ee07336g.
Acknowledgements
This project has received funding from the European Union Horizon 2020 research and innovation programme under grant agreement no. 957189. The project is part of BATTERY 2030+, the large-scale European research initiative for inventing the sustainable batteries of the future. We acknowledge support from the NCCR MARVEL, a National Centre of Competence in Research, funded by the Swiss National Science Foundation (grant number 205602). This work was supported by a grant from the Swiss National Supercomputing Centre (CSCS) under project ID 465000416 (LUMI-G) and project ID mr33 (EIGER).Notes and references
- M. Armand and J.-M. Tarascon, Nature, 2008, 451, 652–657 CrossRef CAS PubMed.
- J. Janek and W. G. Zeier, Nat. Energy, 2016, 1, 1–4 Search PubMed.
- X. Yao, B. Huang, J. Yin, G. Peng, Z. Huang, C. Gao, D. Liu and X. Xu, Chin. Phys. B, 2015, 25, 18802 CrossRef.
- R. Schmuch, R. Wagner, G. Hörpel, T. Placke and M. Winter, Nat. Energy, 2018, 3, 267–278 CrossRef CAS.
- B. Scrosati and J. Garche, J. Power Sources, 2010, 195, 2419–2430 CrossRef CAS.
- J. Bates, N. Dudney, B. Neudecker, A. Ueda and C. Evans, Solid State Ionics, 2000, 135, 33–45 CrossRef CAS.
- Y. Nishi, J. Power Sources, 2001, 100, 101–106 CrossRef CAS.
- H. Liu, G. Zhang, X. Zheng, F. Chen and H. Duan, Int. J. Extreme Manuf., 2020, 2, 042001 CrossRef CAS.
- J. C. Bachman, S. Muy, A. Grimaud, H.-H. Chang, N. Pour, S. F. Lux, O. Paschos, F. Maglia, S. Lupart and P. Lamp, et al. , Chem. Rev., 2016, 116, 140–162 CrossRef CAS PubMed.
- H.-D. Lim, J.-H. Park, H.-J. Shin, J. Jeong, J. T. Kim, K.-W. Nam, H.-G. Jung and K. Y. Chung, Energy Storage Mater., 2020, 25, 224–250 CrossRef.
- S. Ohno, A. Banik, G. F. Dewald, M. A. Kraft, T. Krauskopf, N. Minafra, P. Till, M. Weiss and W. G. Zeier, Prog. Energy, 2020, 2, 022001 CrossRef CAS.
- F. Zheng, M. Kotobuki, S. Song, M. O. Lai and L. Lu, J. Power Sources, 2018, 389, 198–213 CrossRef CAS.
- Y. Meesala, A. Jena, H. Chang and R.-S. Liu, ACS Energy Lett., 2017, 2, 2734–2751 CrossRef CAS.
- F. Zheng, M. Kotobuki, S. Song, M. O. Lai and L. Lu, J. Power Sources, 2018, 389, 198–213 CrossRef CAS.
- L.-O. Hagman, P. Kierkegaard, P. Karvonen, A. I. Virtanen and J. Paasivirta, Acta Chem. Scand., 1968, 22, 1822–1832 CrossRef CAS.
- M. Guin and F. Tietz, J. Power Sources, 2015, 273, 1056–1064 CrossRef CAS.
- F. Sudreau, D. Petit and J. Boilot, J. Solid State Chem., 1989, 83, 78–90 CrossRef CAS.
- Z. Jian, Y.-S. Hu, X. Ji and W. Chen, Adv. Mater., 2017, 29, 1601925 CrossRef PubMed.
- H. Aono, E. Sugimoto, Y. Sadaoka, N. Imanaka and G.-Y. Adachi, J. Electrochem. Soc., 1990, 137, 1023 CrossRef CAS.
- A. Loutati, O. Guillon, F. Tietz and D. Fattakhova-Rohlfing, Open Ceram., 2022, 12, 100313 CrossRef CAS.
- G. Kaur, S. C. Sivasubramanian and A. Dalvi, Electrochim. Acta, 2022, 434, 141311 CrossRef CAS.
- U. v Alpen, A. Rabenau and G. Talat, Appl. Phys. Lett., 1977, 30, 621–623 CrossRef.
- R. Adalati, M. Sharma, S. Sharma, A. Kumar, G. Malik, R. Boukherroub and R. Chandra, J. Energy Storage, 2022, 56, 105912 CrossRef.
- X. Li, J. Liang, N. Chen, J. Luo, K. R. Adair, C. Wang, M. N. Banis, T.-K. Sham, L. Zhang and S. Zhao, et al. , Angew. Chem., 2019, 131, 16579–16584 CrossRef.
- X. Nie, J. Hu and C. Li, Interdiscip. Mater., 2023, 2, 365–389 Search PubMed.
- M. Matsuo and S.-i Orimo, Adv. Energy Mater., 2011, 1, 161–172 CrossRef CAS.
- R. Mohtadi and S.-i Orimo, Nat. Rev. Mater., 2016, 2, 1–15 Search PubMed.
- Y. Inaguma, C. Liquan, M. Itoh, T. Nakamura, T. Uchida, H. Ikuta and M. Wakihara, Solid State Commun., 1993, 86, 689–693 CrossRef CAS.
- S. Stramare, V. Thangadurai and W. Weppner, Chem. Mater., 2003, 15, 3974–3990 CrossRef CAS.
- H.-J. Deiseroth, S.-T. Kong, H. Eckert, J. Vannahme, C. Reiner, T. Zaiß and M. Schlosser, Angew. Chem., 2008, 120, 767–770 CrossRef.
- V. Thangadurai, H. Kaack and W. J. Weppner, J. Am. Ceram. Soc., 2003, 86, 437–440 CrossRef CAS.
- R. Murugan, V. Thangadurai and W. Weppner, et al. , Angew. Chem., Int. Ed., 2007, 46, 7778 CrossRef CAS PubMed.
- H.-P. Hong, Mater. Res. Bull., 1978, 13, 117–124 CrossRef CAS.
- Y.-W. Hu, I. Raistrick and R. A. Huggins, J. Electrochem. Soc., 1977, 124, 1240 CrossRef CAS.
- A. Khorassani and A. West, Solid State Ionics, 1982, 7, 1–8 CrossRef CAS.
- A. Rodger, J. Kuwano and A. West, Solid State Ionics, 1985, 15, 185–198 CrossRef CAS.
- Y. Deng, C. Eames, J.-N. Chotard, F. Lalère, V. Seznec, S. Emge, O. Pecher, C. P. Grey, C. Masquelier and M. S. Islam, J. Am. Chem. Soc., 2015, 137, 9136–9145 CrossRef CAS PubMed.
- R. Kanno and M. Murayama, J. Electrochem. Soc., 2001, 148, A742 CrossRef CAS.
- B. Tao, C. Ren, H. Li, B. Liu, X. Jia, X. Dong, S. Zhang and H. Chang, Adv. Funct. Mater., 2022, 32, 2203551 CrossRef CAS.
- N. Kamaya, K. Homma, Y. Yamakawa, M. Hirayama, R. Kanno, M. Yonemura, T. Kamiyama, Y. Kato, S. Hama and K. Kawamoto, et al. , Nat. Mater., 2011, 10, 682–686 CrossRef CAS PubMed.
- F. Liang, Y. Sun, Y. Yuan, J. Huang, M. Hou and J. Lu, Mater. Today, 2021, 50, 418–441 CrossRef CAS.
- H. Koinuma and I. Takeuchi, Nat. Mater., 2004, 3, 429–438 CrossRef CAS PubMed.
- X.-D. Xiang, X. Sun, G. Briceno, Y. Lou, K.-A. Wang, H. Chang, W. G. Wallace-Freedman, S.-W. Chen and P. G. Schultz, Science, 1995, 268, 1738–1740 CrossRef CAS PubMed.
- I. Takeuchi, O. Famodu, J. Read, M. Aronova, K.-S. Chang, C. Craciunescu, S. Lofland, M. Wuttig, F. Wellstood and L. Knauss, et al. , Nat. Mater., 2003, 2, 180–184 CrossRef CAS PubMed.
- I. E. Castelli, T. Olsen, S. Datta, D. D. Landis, S. Dahl, K. S. Thygesen and K. W. Jacobsen, Energy Environ. Sci., 2012, 5, 5814–5819 RSC.
- G. Pizzi, A. Cepellotti, R. Sabatini, N. Marzari and B. Kozinsky, Comput. Mater. Sci., 2016, 111, 218–230 CrossRef.
- N. Mounet, M. Gibertini, P. Schwaller, D. Campi, A. Merkys, A. Marrazzo, T. Sohier, I. E. Castelli, A. Cepellotti and G. Pizzi, et al. , Nat. Nanotechnol., 2018, 13, 246–252 CrossRef CAS PubMed.
- M. K. Tufail, P. Zhai, M. Jia, N. Zhao and X. Guo, Energy Mater. Adv., 2023, 4, 15 CrossRef CAS.
- Y. Xiao, K. Jun, Y. Wang, L. J. Miara, Q. Tu and G. Ceder, Adv. Energy Mater., 2021, 11, 2101437 CrossRef CAS.
- S. Muy, T. Le Mercier, M. Dufour, M.-D. Braida, A. A. Emery and N. Marzari, Chem. Mater., 2025, 37, 2395–2403 CrossRef CAS.
- P. Hohenberg and W. Kohn, Phys. Rev., 1964, 136, B864 CrossRef.
- W. Kohn and L. J. Sham, Phys. Rev., 1965, 140, A1133 CrossRef.
- M. Shishkin, M. Marsman and G. Kresse, Phys. Rev. Lett., 2007, 99, 246403 CrossRef CAS PubMed.
- R. De Gennaro, N. Colonna, E. Linscott and N. Marzari, Phys. Rev. B, 2022, 106, 035106 CrossRef CAS.
- G. Miceli, W. Chen, I. Reshetnyak and A. Pasquarello, Phys. Rev. B, 2018, 97, 121112 CrossRef CAS.
- B. Himmetoglu, A. Floris, S. De Gironcoli and M. Cococcioni, Int. J. Quantum Chem., 2014, 114, 14–49 CrossRef CAS.
- N. Marzari, A. Ferretti and C. Wolverton, Nat. Mater., 2021, 20, 736–749 CrossRef CAS PubMed.
- A. J. Cohen, P. Mori-Sánchez and W. Yang, Chem. Rev., 2012, 112, 289–320 CrossRef CAS PubMed.
- S. Muy, J. Voss, R. Schlem, R. Koerver, S. J. Sedlmaier, F. Maglia, P. Lamp, W. G. Zeier and Y. Shao-Horn, iScience, 2019, 16, 270–282 CrossRef CAS PubMed.
- A. D. Sendek, Q. Yang, E. D. Cubuk, K.-A. N. Duerloo, Y. Cui and E. J. Reed, Energy Environ. Sci., 2017, 10, 306–320 RSC.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- W. D. Richards, L. J. Miara, Y. Wang, J. C. Kim and G. Ceder, Chem. Mater., 2016, 28, 266–273 CrossRef CAS.
- O. Borodin, M. Olguin, C. E. Spear, K. W. Leiter and J. Knap, Nanotechnology, 2015, 26, 354003 CrossRef PubMed.
- Y. Sun, W. Yan, L. An, B. Wu, K. Zhong and R. Yang, Solid State Ionics, 2017, 301, 59–63 CrossRef CAS.
- S. Jian, H. Li, X. Jia, D. Zhong, B. Tao, X. He, G. Wang and H. Chang, FlatChem, 2024, 100693 CrossRef CAS.
- K. H. Park, Q. Bai, D. H. Kim, D. Y. Oh, Y. Zhu, Y. Mo and Y. S. Jung, Adv. Energy Mater., 2018, 8, 1800035 CrossRef.
- J. Pokluda, M. Černy, M. Šob and Y. Umeno, Prog. Mater. Sci., 2015, 73, 127–158 CrossRef CAS.
- G. Materzanini, T. Chiarotti and N. Marzari, npj Comput. Mater., 2023, 9, 10 CrossRef CAS PubMed.
- Y. Ren, Y. Shen, Y. Lin and C.-W. Nan, Electrochem. Commun., 2015, 57, 27–30 CrossRef CAS.
- L. Porz, T. Swamy, B. W. Sheldon, D. Rettenwander, T. Frömling, H. L. Thaman, S. Berendts, R. Uecker, W. C. Carter and Y.-M. Chiang, Adv. Energy Mater., 2017, 7, 1701003 CrossRef.
- K. Kerman, A. Luntz, V. Viswanathan, Y.-M. Chiang and Z. Chen, J. Electrochem. Soc., 2017, 164, A1731 CrossRef CAS.
- Z. Gao, H. Sun, L. Fu, F. Ye, Y. Zhang, W. Luo and Y. Huang, Adv. Mater., 2018, 30, 1705702 CrossRef PubMed.
- S. Hull, Rep. Prog. Phys., 2004, 67, 1233 CrossRef CAS.
- P. Knauth and H. L. Tuller, J. Am. Ceram. Soc., 2002, 85, 1654–1680 CrossRef CAS.
- B. J. Alder and T. E. Wainwright, J. Chem. Phys., 1959, 31, 459–466 CrossRef CAS.
- A. Rahman, Phys. Rev., 1964, 136, A405 CrossRef.
- D. Frenkel and B. Smit, Understanding molecular simulation: from algorithms to applications, Elsevier, 2023 Search PubMed.
- M. Tuckerman, Statistical Mechanics: Theory and Molecular Simulation, OUP, Oxford, 2010 Search PubMed.
- M. P. Allen and D. J. Tildesley, Computer simulation of liquids, Oxford University Press, 2017 Search PubMed.
- S. Adams and R. P. Rao, J. Mater. Chem., 2012, 22, 7687–7691 RSC.
- M. Born and W. Heisenberg, Original Scientific Papers Wissenschaftliche Originalarbeiten, 1985, pp. 216–246 Search PubMed.
- M. E. Tuckerman, J. Phys.: Condens. Matter, 2002, 14, R1297 CrossRef CAS.
- R. Car and M. Parrinello, Phys. Rev. Lett., 1985, 55, 2471 CrossRef CAS PubMed.
- G. Materzanini, L. Kahle, A. Marcolongo and N. Marzari, Phys. Rev. Mater., 2021, 5, 035408 CrossRef CAS.
- Y. A. Du and N. Holzwarth, J. Electrochem. Soc., 2007, 154, A999 CrossRef CAS.
- N. Lepley, N. Holzwarth and Y. A. Du, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 88, 104103 CrossRef.
- A. Marcolongo and N. Marzari, Phys. Rev. Mater., 2017, 1, 025402 CrossRef.
- X. He, Y. Zhu and Y. Mo, Nat. Commun., 2017, 8, 15893 CrossRef CAS PubMed.
- B. J. Morgan, R. Soc. Open Sci., 2017, 4, 170824 CrossRef PubMed.
- I. Brown, Acta Crystallogr., Sect. B: Struct. Sci., 1992, 48, 553–572 CrossRef.
- N. Anurova, V. Blatov, G. Ilyushin, O. Blatova, A. Ivanov-Schitz and L. Dem'Yanets, Solid State Ionics, 2008, 179, 2248–2254 CrossRef CAS.
- M. Avdeev, M. Sale, S. Adams and R. P. Rao, Solid State Ionics, 2012, 225, 43–46 CrossRef CAS.
- R. Xiao, H. Li and L. Chen, J. Materiomics, 2015, 1, 325–332 CrossRef.
- J. Swenson, et al. , Phys. Rev. Lett., 2000, 84, 4144 CrossRef PubMed.
- C. Müller, E. Zienicke, S. Adams, J. Habasaki and P. Maass, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 75, 014203 CrossRef.
- M. Aniya and K. Wakamura, Phys. B, 1996, 219, 463–465 CrossRef.
- K. Wakamura, Phys. Rev. B: Condens. Matter Mater. Phys., 1997, 56, 11593 CrossRef CAS.
- I. Batatia, D. P. Kovacs, G. Simm, C. Ortner and G. Csányi, Adv. Neural Inf. Process. Syst., 2022, 35, 11423–11436 Search PubMed.
- I. Batatia, P. Benner, Y. Chiang, A. M. Elena, D. P. Kovács, J. Riebesell, X. R. Advincula, M. Asta, M. Avaylon, W. J. Baldwin, F. Berger, N. Bernstein, A. Bhowmik, F. Bigi, S. M. Blau, V. Carare, M. Ceriotti, S. Chong, J. P. Darby, S. De, F. D. Pia, V. L. Deringer, R. ElijoÅ¡ius, Z. El-Machachi, E. Fako, F. Falcioni, A. C. Ferrari, J. L. A. Gardner, M. J. Gawkowski, A. Genreith-Schriever, J. George, R. E. A. Goodall, J. Grandel, C. P. Grey, P. Grigorev, S. Han, W. Handley, H. H. Heenen, K. Hermansson, C. H. Ho, S. Hofmann, C. Holm, J. Jaafar, K. S. Jakob, H. Jung, V. Kapil, A. D. Kaplan, N. Karimitari, J. R. Kermode, P. Kourtis, N. Kroupa, J. Kullgren, M. C. Kuner, D. Kuryla, G. Liepuoniute, C. Lin, J. T. Margraf, I.-B. Magdau, A. Michaelides, J. H. Moore, A. A. Naik, S. P. Niblett, S. W. Norwood, N. O’Neill, C. Ortner, K. A. Persson, K. Reuter, A. S. Rosen, L. A. M. Rosset, L. L. Schaaf, C. Schran, B. X. Shi, E. Sivonxay, T. K. Stenczel, C. Sutton, V. Svahn, T. D. Swinburne, J. Tilly, C. van der Oord, S. Vargas, E. Varga-Umbrich, T. Vegge, M. Vondrák, Y. Wang, W. C. Witt, T. Wolf, F. Zills and G. Csányi, J. Chem. Phys., 2025, 163, 0021–9606 CrossRef PubMed.
- C. Chen and S. P. Ong, Nat. Comput. Sci., 2022, 2, 718–728 CrossRef PubMed.
- B. Deng, P. Zhong, K. Jun, J. Riebesell, K. Han, C. J. Bartel and G. Ceder, Nat. Mach. Intell., 2023, 5, 1031–1041 CrossRef.
- A. Merchant, S. Batzner, S. S. Schoenholz, M. Aykol, G. Cheon and E. D. Cubuk, Nature, 2023, 624, 80–85 CrossRef CAS PubMed.
- J. Riebesell, R. E. Goodall, A. Jain, P. Benner, K. A. Persson and A. A. Lee, arXiv, 2023, preprint, arXiv:2308.14920 DOI:10.48550/arXiv.2308.14920, https://arxiv.org/abs/2308.14920.
- H. Yu, M. Giantomassi, G. Materzanini and G.-M. Rignanese, arXiv, 2024, preprint, arXiv:2403.05729 DOI:10.48550/arXiv.2403.05729, https://arxiv.org/abs/2403.05729.
- B. Focassio, L. P. M. Freitas and G. R. Schleder, arXiv, 2024, preprint, arXiv:2403.04217 DOI:10.48550/arXiv.2403.04217, https://arxiv.org/abs/2403.04217.
- D. Wines and K. Choudhary, arXiv, 2024, preprint, arXiv:2412.10516 DOI:10.48550/arXiv.2412.10516, https://arxiv.org/abs/2412.10516.
- Z. Li, K. Jun, B. Deng and G. Ceder, arXiv, 2025, preprint, arXiv:2511.20964 DOI:10.48550/arXiv.2511.20964, https://arxiv.org/abs/2511.20964.
- J. Lian, X. Fu, X. Gong, R. Xiao and H. Li, arXiv, 2025, preprint, arXiv:2507.02334 DOI:10.48550/arXiv.2507.02334, https://arxiv.org/abs/2507.02334.
- A. Agrawal and A. Choudhary, APL Mater., 2016, 4, 53208 CrossRef.
- F. A. L. Laskowski, D. B. McHaffie and K. A. See, Energy Environ. Sci., 2023, 16, 1264–1276 RSC.
- A. D. Sendek, Q. Yang, E. D. Cubuk, K.-A. N. Duerloo, Y. Cui and E. J. Reed, Energy Environ. Sci., 2017, 10, 306–320 RSC.
- Y. Zhang, X. He, Z. Chen, Q. Bai, A. M. Nolan, C. A. Roberts, D. Banerjee, T. Matsunaga, Y. Mo and C. Ling, Nat. Commun., 2019, 10, 5260 CrossRef PubMed.
- C. J. Hargreaves, M. W. Gaultois, L. M. Daniels, E. J. Watts, V. A. Kurlin, M. Moran, Y. Dang, R. Morris, A. Morscher and K. Thompson, npj Comput. Mater., 2023, 9, 9 CrossRef CAS.
- C. Chen, D. T. Nguyen, S. J. Lee, N. A. Baker, A. S. Karakoti, L. Lauw, C. Owen, K. T. Mueller, B. A. Bilodeau and V. Murugesanet al., arXiv, 2024, preprint, arXiv:2401.04070 DOI:10.48550/arXiv.2401.04070, https://arxiv.org/abs/2401.04070.
- A. Zunger, Nat. Rev. Chem., 2018, 2, 0121 CrossRef CAS.
- A. K. Cheetham and R. Seshadri, Chem. Mater., 2024, 36, 3490–3495 CrossRef CAS PubMed.
- L. Kahle, A. Marcolongo and N. Marzari, Energy Environ. Sci., 2020, 13, 928–948 RSC.
- K. Jun, Y. Sun, Y. Xiao, Y. Zeng, R. Kim, H. Kim, L. J. Miara, D. Im, Y. Wang and G. Ceder, Nat. Mater., 2022, 21, 924–931 CrossRef CAS PubMed.
- L. Kahle, A. Marcolongo and N. Marzari, Phys. Rev. Mater., 2018, 2, 065405 CrossRef.
- S. Curtarolo, G. L. Hart, M. B. Nardelli, N. Mingo, S. Sanvito and O. Levy, Nat. Mater., 2013, 12, 191–201 CrossRef CAS PubMed.
- K. Alberi, M. B. Nardelli, A. Zakutayev, L. Mitas, S. Curtarolo, A. Jain, M. Fornari, N. Marzari, I. Takeuchi and M. L. Green, et al. , J. Phys. D: Appl. Phys., 2018, 52, 013001 CrossRef PubMed.
- P. Buneman, S. Khanna and T. Wang-Chiew, Database Theory—ICDT 2001: 8th International Conference London, UK, January 4-6, 2001 Proceedings 8, 2001, pp. 316-330.
- L. Moreau and P. Groth, Provenance: an introduction to PROV, Springer Nature, 2022 Search PubMed.
- S. P. Huber, S. Zoupanos, M. Uhrin, L. Talirz, L. Kahle, R. Häuselmann, D. Gresch, T. Müller, A. V. Yakutovich and C. W. Andersen, et al. , Sci. Data, 2020, 7, 300 CrossRef PubMed.
- M. Uhrin, S. P. Huber, J. Yu, N. Marzari and G. Pizzi, Comput. Mater. Sci., 2021, 187, 110086 CrossRef.
- M. D. Wilkinson, M. Dumontier, I. J. Aalbersberg, G. Appleton, M. Axton, A. Baak, N. Blomberg, J.-W. Boiten, L. B. da Silva Santos and P. E. Bourne, et al. , Sci. Data, 2016, 3, 1–9 Search PubMed.
- S. Gražulis, A. Daškevič, A. Merkys, D. Chateigner, L. Lutterotti, M. Quirós, N. R. Serebryanaya, P. Moeck, R. T. Downs and A. Le Bail, Nucleic Acids Res., 2011, 40, D420–D427 CrossRef PubMed.
- A. Belsky, M. Hellenbrandt, V. L. Karen and P. Luksch, Acta Crystallogr., Sect. B: Struct. Sci., 2002, 58, 364–369 CrossRef PubMed.
- E. Blokhin and P. Villars, The PAULING FILE Project and Materials Platform for Data Science: From Big Data Toward Materials Genome, Springer International Publishing, Cham, 2018, pp. 1–26 Search PubMed.
- A. Vaitkus, A. Merkys and S. Gražulis, J. Appl. Crystallogr., 2021, 54, 661–672 CrossRef CAS PubMed.
- A. Merkys, A. Vaitkus, J. Butkus, M. Okulič-Kazarinas, V. Kairys and S. Gražulis, J. Appl. Crystallogr., 2016, 49, 292–301 CrossRef CAS PubMed.
- S. Gražulis, A. Merkys, A. Vaitkus and M. Okulič-Kazarinas, J. Appl. Crystallogr., 2015, 48, 85–91 CrossRef PubMed.
- G. L. Hart and R. W. Forcade, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 77, 224115 CrossRef.
- R. Grau-Crespo, S. Hamad, C. R. A. Catlow and N. De Leeuw, J. Phys.: Condens. Matter, 2007, 19, 256201 CrossRef.
- S. Mustapha, P. D’Arco, M. De La Pierre, Y. Noël, M. Ferrabone and R. Dovesi, J. Phys.: Condens. Matter, 2013, 25, 105401 CrossRef CAS PubMed.
- R. Hundt, J. Schön and M. Jansen, J. Appl. Crystallogr., 2006, 39, 6–16 CrossRef CAS.
- S. P. Ong, W. D. Richards, A. Jain, G. Hautier, M. Kocher, S. Cholia, D. Gunter, V. L. Chevrier, K. A. Persson and G. Ceder, Comput. Mater. Sci., 2013, 68, 314–319 CrossRef CAS.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 (19pp) CrossRef PubMed.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. B. Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, M. Cococcioni, N. Colonna, I. Carnimeo, A. D. Corso, S. de Gironcoli, P. Delugas, R. A. D. Jr, A. Ferretti, A. Floris, G. Fratesi, G. Fugallo, R. Gebauer, U. Gerstmann, F. Giustino, T. Gorni, J. Jia, M. Kawamura, H.-Y. Ko, A. Kokalj, E. Küçükbenli, M. Lazzeri, M. Marsili, N. Marzari, F. Mauri, N. L. Nguyen, H.-V. Nguyen, A. O. de-la Roza, L. Paulatto, S. Poncé, D. Rocca, R. Sabatini, B. Santra, M. Schlipf, A. P. Seitsonen, A. Smogunov, I. Timrov, T. Thonhauser, P. Umari, N. Vast, X. Wu and S. Baroni, J. Phys.: Condens. Matter, 2017, 29, 465901 CrossRef CAS PubMed.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 CrossRef PubMed.
- G. Prandini, A. Marrazzo, I. E. Castelli, N. Mounet and N. Marzari, npj Comput. Mater., 2018, 4, 72 CrossRef.
- A. Willand, Y. O. Kvashnin, L. Genovese, Á. Vázquez-Mayagoitia, A. K. Deb, A. Sadeghi, T. Deutsch and S. Goedecker, J. Chem. Phys., 2013, 138, 104–109 CrossRef PubMed.
- A. Dal Corso, Comput. Mater. Sci., 2014, 95, 337–350 CrossRef CAS.
- K. F. Garrity, J. W. Bennett, K. M. Rabe and D. Vanderbilt, Comput. Mater. Sci., 2014, 81, 446–452 CrossRef CAS.
- M. Topsakal and R. Wentzcovitch, Comput. Mater. Sci., 2014, 95, 263–270 CrossRef CAS.
- M. Schlipf and F. Gygi, Comput. Phys. Commun., 2015, 196, 36–44 CrossRef CAS.
- M. J. Van Setten, M. Giantomassi, E. Bousquet, M. J. Verstraete, D. R. Hamann, X. Gonze and G.-M. Rignanese, Comput. Phys. Commun., 2018, 226, 39–54 CrossRef CAS.
- N. Marzari, D. Vanderbilt, A. De Vita and M. Payne, Phys. Rev. Lett., 1999, 82, 3296 CrossRef CAS.
- T. Thakur, AiiDA-flipper, 2024, https://github.com/epfl-theos/aiida-flipper.
- L. Kahle, The supercell creator, 2019, https://github.com/lekah/supercellor.
- G. Bussi, D. Donadio and M. Parrinello, J. Chem. Phys., 2007, 126, 014–101 CrossRef PubMed.
- Y. Haven, Phys. Chem. Glasses, 1965, 6, 38 CAS.
- A. Einstein, Ann. Phys., 1905, 4, 1–10 Search PubMed.
- L. Kahle, Suite for Analysis of Molecular Simulations (SAMOS), 2024, https://github.com/lekah/samos Search PubMed.
- X. He, Y. Zhu, A. Epstein and Y. Mo, npj Comput. Mater., 2018, 4, 18 CrossRef.
- D. Sivia and J. Skilling, Data analysis: a Bayesian tutorial, OUP, Oxford, 2006 Search PubMed.
- Y. Wang, W. D. Richards, S. P. Ong, L. J. Miara, J. C. Kim, Y. Mo and G. Ceder, Nat. Mater., 2015, 14, 1026–1031 CrossRef CAS PubMed.
- G. K. P. Dathar, J. Balachandran, P. R. Kent, A. J. Rondinone and P. Ganesh, J. Mater. Chem. A, 2017, 5, 1153–1159 RSC.
- L. Zhou, N. Minafra, W. G. Zeier and L. F. Nazar, Acc. Chem. Res., 2021, 54, 2717–2728 CrossRef CAS PubMed.
- H. Yang and N. Wu, Energy Sci. Eng., 2022, 10, 1643–1671 CrossRef CAS.
- D. Wang, H. Shi, S. Wang, X. Wu, W. Jiang, S. Liang and Z. Xu, Coord. Chem. Rev., 2024, 508, 215776 CrossRef CAS.
- A. Kozhevnikov and S. Pintarelli, SIRIUS, 2023, https://github.com/electronic-structure/SIRIUS.
- M. De La Pierre, R. Orlando, L. Maschio, K. Doll, P. Ugliengo and R. Dovesi, J. Comput. Chem., 2011, 32, 1775–1784 CrossRef CAS PubMed.
- Z. Zhu, I.-H. Chu and S. P. Ong, Chem. Mater., 2017, 29, 2474–2484 CrossRef CAS.
- K. Kataoka, J. Awaka, N. Kijima, H. Hayakawa, K. Ohshima and J. Akimoto, Chem. Mater., 2011, 23, 2344–2352 CrossRef CAS.
- J. Fernández-Gamboa, F. Tielens and Y. Zulueta, Mater. Sci. Semicond. Process., 2022, 152, 107074 CrossRef.
- A. Kuhn, J. Mater. Chem. A, 2018, 6, 443–455 RSC.
- I.-H. Chu, H. Nguyen, S. Hy, Y.-C. Lin, Z. Wang, Z. Xu, Z. Deng, Y. S. Meng and S. P. Ong, ACS Appl. Mater. Interfaces, 2016, 8, 7843–7853 CrossRef CAS PubMed.
- H. Yamane, M. Shibata, Y. Shimane, T. Junke, Y. Seino, S. Adams, K. Minami, A. Hayashi and M. Tatsumisago, Solid State Ionics, 2007, 178, 1163–1167 CrossRef CAS.
- Y. Xiao, K. Jun, Y. Wang, L. Miara, Q. Tu and G. Ceder, Adv. Energy Mater., 2021, 11, 2101437 CrossRef CAS.
- S. Kirklin, M. Chan, L. Trahey, M. Thackeray and C. Wolverton, Phys. Chem. Chem. Phys., 2014, 16, 22073–22082 RSC.
- M. Howard, O. Clemens, P. Slater and P. Anderson, J. Alloys Compd., 2015, 645, S174–S177 CrossRef CAS.
- B. Boukamp and R. Huggins, Phys. Lett. A, 1976, 58, 231–233 CrossRef.
- B. Boukamp and R. Huggins, Mater. Res. Bull., 1978, 13, 23–32 CrossRef CAS.
- R. Shannon, B. Taylor, A. English and T. Berzins, International Symposium on Solid Ionic and Ionic-Electronic Conductors, 1977, pp. 783–796 Search PubMed.
- N. Adelstein and B. Wood, Chem. Mater., 2016, 28, 7218–7231 CrossRef CAS.
- F. Wu, J. Lee, Y. Xiao and G. Yushin, Nano Energy, 2016, 27, 238–246 CrossRef CAS.
- W. Yang, ACS Appl. Mater. Interfaces, 2022, 14, 50710–50717 CrossRef CAS PubMed.
- H. Yamane, S. Kikkawa and M. Koizumi, J. Solid State Chem., 1987, 71, 1–11 CrossRef CAS.
- F. Laskowski, D. McHaffie and K. See, Energy Environ. Sci., 2023, 16, 1264–1276 RSC.
- F. Bianchini, H. Fjellvåg and P. Vajeeston, Mater. Lett., 2018, 219, 186–189 CrossRef CAS.
- T. Kaib, P. Bron, S. Haddadpour, L. Mayrhofer, L. Pastewka, T. T. Järvi, M. Moseler, B. Roling and S. Dehnen, Chem. Mater., 2013, 25, 2961–2969 CrossRef CAS.
- A. Ulvestad, J. Mæhlen and M. Kirkengen, J. Power Sources, 2018, 399, 414–421 CrossRef CAS.
- D. Snydacker, V. Hegde and C. Wolverton, J. Electrochem. Soc., 2017, 164, 3582 CrossRef.
- L. Toffoletti, Chem. – Eur. J., 2016, 22, 17635–17645 CrossRef CAS PubMed.
- S. Yeandel, D. Scanlon and P. Goddard, J. Mater. Chem. A, 2019, 7, 3953–3961 RSC.
- B. Hang, T. Ohnishi, M. Osada, X. Xu, K. Takada and T. Sasaki, J. Power Sources, 2010, 195, 3323–3327 CrossRef CAS.
- W. Huang, J. Am. Chem. Soc., 2022, 144, 4989–4994 CrossRef CAS PubMed.
- K. Xu, Chem. Rev., 2004, 104, 4303–4418 CrossRef CAS PubMed.
- A. Emly, E. Kioupakis and A. Ven, Chem. Mater., 2013, 25, 4663–4670 CrossRef CAS.
- Y. Zhao and L. Daemen, J. Am. Chem. Soc., 2012, 134, 15042–15047 CrossRef CAS PubMed.
- M. Tachez, J.-P. Malugani, R. Mercier and G. Robert, Solid State Ionics, 1984, 14, 181–185 CrossRef CAS.
- Z. Liu, J. Am. Chem. Soc., 2013, 135, 975–978 CrossRef CAS PubMed.
- A. Al-Qawasmeh, A. Obeidat, A. Shukri and N. Holzwarth, J. Electrochem. Soc., 2020, 167, 60505 CrossRef CAS.
- A. Obeidat, A. Al-Qawasmeh and A. Shukri, J. Electrochem. Soc., 2022, 169, 50521 CrossRef CAS.
- H. Lim, S.-C. Kim, J. Kim, Y.-I. Kim and S.-J. Kim, J. Solid State Chem., 2018, 257, 19–25 CrossRef CAS.
- W. Huang, J. Solid State Chem., 2019, 270, 487–492 CrossRef CAS.
- W. Weppner, P. Hartwig and A. Rabenau, J. Power Sources, 1981, 6, 251–259 CrossRef CAS.
- V. Landgraf, T. Famprikis, J. Leeuw, L. Bannenberg, S. Ganapathy and M. Wagemaker, ACS Appl. Energy Mater., 2023, 6, 1661–1672 CrossRef CAS PubMed.
- E. Nomura and M. Greenblatt, J. Solid State Chem., 1984, 52, 91–93 CrossRef CAS.
- H. Aono, E. Sugimoto, Y. Sadaoka, N. Imanaka and G.-Y. Adachi, Solid State Ionics, 1993, 62, 309–316 CrossRef CAS.
- A. Martínez-Juárez, J. Iglesias and J. Rojo, Solid State Ionics, 1996, 91, 295–301 CrossRef.
- C.-M. Chang, S.-H. Hong and H.-M. Park, Solid State Ionics, 2005, 176, 2583–2587 CrossRef CAS.
- B. Tell, S. Wagner and H. Kasper, J. Electrochem. Soc., 1977, 124, 536 CrossRef CAS.
- F.-Y. Rao, Chin. Phys. B, 2016, 25, 28202 CrossRef.
- A. Rosenman, E. Markevich, G. Salitra, D. Aurbach, A. Garsuch and F. Chesneau, Adv. Energy Mater., 2015, 5, 1500212 CrossRef.
- D. Su, D. Zhou, C. Wang and G. Wang, Adv. Funct. Mater., 2018, 28, 1800154 CrossRef.
- G. Eichinger, Solid State Ionics, 1981, 2, 289–295 CrossRef CAS.
- W. D. Richards, Y. Wang, L. J. Miara, J. C. Kim and G. Ceder, Energy Environ. Sci., 2016, 9, 3272–3278 RSC.
- K. Kaup, F. Lalère, A. Huq, A. Shyamsunder, T. Adermann, P. Hartmann and L. F. Nazar, Chem. Mater., 2018, 30, 592–596 CrossRef CAS.
- X. Jiang, H. Xu, H. Mao, J. Yang and Y. Qian, J. Power Sources, 2016, 320, 94–103 CrossRef CAS.
- Y. Wang, X. Xiao, Q. Li and H. Pang, Small, 2018, 14, 1802193 CrossRef PubMed.
- Y. Huang, Y. Jiang, Y. Zhou, Z. Hu and X. Zhu, ChemElectroChem, 2019, 6, 6016–6026 CrossRef CAS.
- F. Walther, F. Strauss, X. Wu, B. Mogwitz, J. Hertle, J. Sann, M. Rohnke, T. Brezesinski and J. Janek, Chem. Mater., 2021, 33, 2110–2125 CrossRef CAS.
- H. Jiang, S. Wang, B. Zhang, Y. Shao, Y. Wu, H. Zhao, Y. Lei and X. Hao, Chem. Eng. J., 2020, 396, 125207 CrossRef CAS.
- M. Spiesser, P. Palvadeau, C. Guillot and J. Cerisier, Solid State Ionics, 1983, 9, 103–106 CrossRef.
- A. V. Chadwick, Defect and Diffusion Forum, 1993, pp. 1015–1040 Search PubMed.
- R. Verma, C.-J. Park, R. Kothandaraman and U. Varadaraju, Electrochim. Acta, 2017, 258, 1445–1452 CrossRef CAS.
- C. M. Burba and R. Frech, J. Electrochem. Soc., 2005, 152, A1233 CrossRef CAS.
- W.-J. Cui, J. Yi, L. Chen, C.-X. Wang and Y.-Y. Xia, J. Power Sources, 2012, 217, 77–84 CrossRef CAS.
- J. Tian, D. Wang and Z. Shan, et al. , J. Power Sources, 2017, 361, 96–104 CrossRef.
- T. Kaib, S. Haddadpour, M. Kapitein, P. Bron, C. Schröder, H. Eckert, B. Roling and S. Dehnen, Chem. Mater., 2012, 24, 2211–2219 CrossRef CAS.
- R. Marx, F. Lissner and T. Schleid, Z. Anorg. Allg. Chem., 2006, 632, 2151 CrossRef.
- L. J. Miara, N. Suzuki, W. D. Richards, Y. Wang, J. C. Kim and G. Ceder, J. Mater. Chem. A, 2015, 3, 20338–20344 RSC.
- M. Murayama, R. Kanno, Y. Kawamoto and T. Kamiyama, Solid State Ionics, 2002, 154, 789–794 CrossRef.
- M. Pompetzki, L. van Wüllen and M. Jansen, Z. Anorg. Allg. Chem., 2004, 630, 484–490 CrossRef CAS.
- L. van Wüllen, L. Hildebrandt and M. Jansen, Solid State Ionics, 2005, 176, 1449–1456 CrossRef.
- J. Sang, Y. Yu, Z. Wang and G. Shao, Phys. Chem. Chem. Phys., 2020, 22, 12918–12928 RSC.
- V. Kravchenko and S. Sigaryov, J. Mater. Sci., 1994, 29, 6004–6010 CrossRef CAS.
- T. Pilz and M. Jansen, Z. Anorg. Allg. Chem., 2011, 637, 2148–2152 CrossRef CAS.
- S. J. Fanah and F. Ramezanipour, Solid State Sci., 2019, 97, 106014 CrossRef CAS.
- C. Muehle, R. E. Dinnebier, L. van Wüllen, G. Schwering and M. Jansen, Inorg. Chem., 2004, 43, 874–881 CrossRef CAS PubMed.
- I. Pentin, J. Schön and M. Jansen, Solid State Sci., 2008, 10, 804–813 CrossRef CAS.
- A. Ahmad and A. Arof, Ionics, 2002, 8, 433–438 CrossRef CAS.
- M. Dissanayake, M. Careem, P. Bandaranayake and C. Wijayasekera, Solid State Ionics, 1991, 48, 277–281 CrossRef CAS.
- J.-G. Ryu, R. Balasubramaniam, V. Aravindan, S. Park, S. J. Cho and Y.-S. Lee, ACS Appl. Mater. Interfaces, 2023, 16, 761–771 CrossRef PubMed.
- D. Cachau-Herreillat, A. Norbert, M. Maurin and E. Philippot, J. Solid State Chem., 1981, 37, 352–361 CrossRef CAS.
- I. V. Pentin, V. Saltykov, J. Nuss, J. C. Schön and M. Jansen, Chem. – Eur. J., 2012, 18, 3559–3565 CrossRef CAS PubMed.
- H. Sabrowsky, Z. Naturforsch., 1989, 44, 1607–1609 CrossRef.
- T. Ohtani, H. Honjo and H. Wada, Mater. Res. Bull., 1987, 22, 829–840 CrossRef CAS.
- B. Greuling, Cryst. Res. Technol., 1987, 22, K27–K29 CrossRef CAS.
- E. Hellstrom and R. Huggins, Mater. Res. Bull., 1979, 14, 881–889 CrossRef CAS.
- M. Tromme, CR Seances Acad. Sci., Ser. C, 1971, 273, 849–851 CAS.
- J.-H. Feng, C.-L. Hu, H.-P. Xia, F. Kong and J.-G. Mao, Inorg. Chem., 2017, 56, 14697–14705 CrossRef CAS PubMed.
- H. Eickhoff, L. Toffoletti, W. Klein, G. Raudaschl-Sieber and T. F. Fässler, Inorg. Chem., 2017, 56, 6688–6694 CrossRef CAS PubMed.
- V. A. Carlson and A. M. Stacy, J. Solid State Chem., 1992, 96, 332–343 CrossRef CAS.
- O. Graudejus, A. P. Wilkinson, L. C. Chacón and N. Bartlett, Inorg. Chem., 2000, 39, 2794–2800 CrossRef CAS PubMed.
- J. Chassaing, PhD thesis, Gauthier-Villars, 1968 Search PubMed.
- M. S. Whittingham and F. R. Gamble Jr, Mater. Res. Bull., 1975, 10, 363–371 CrossRef CAS.
- J.-H. Zhang, D. J. Clark, A. Weiland, S. S. Stoyko, Y. S. Kim, J. I. Jang and J. A. Aitken, Inorg. Chem. Front., 2017, 4, 1472–1484 RSC.
- W. Yin, K. Feng, D. Mei, J. Yao, P. Fu and Y. Wu, Dalton Trans., 2012, 41, 2272–2276 RSC.
- R. De Pape, J. Portier, J. Grannec, G. Gauthier and P. Hagenmuller, CR Acad. Sc. Paris, Sér. C, 1969, 269, 1120–1121 CAS.
- R. Hoppe and H.-J. Röhrborn, Z. Anorg. Allg. Chem., 1964, 329, 110–122 CrossRef CAS.
- K. Toda, M. Takahashi, T. Teranishi, Z.-G. Ye, M. Sato and Y. Hinatsu, J. Mater. Chem., 1999, 9, 799–803 RSC.
- J. Wolfenstine, E. Rangasamy, J. L. Allen and J. Sakamoto, J. Power Sources, 2012, 208, 193–196 CrossRef CAS.
- A. Novoselova and Y. P. Simanov, Thermal and X-ray analysis of the system LiF-BeF2, MAIK Nauka/Interperiodica Publishers, 1952 Search PubMed.
- D. Roy, R. Roy and E. Osborn, J. Am. Ceram. Soc., 1954, 37, 300–305 CrossRef CAS.
- M. Dion, Y. Piffard and M. Tournoux, J. Inorg. Nucl. Chem., 1978, 40, 917–918 CrossRef CAS.
- G. Hyett, O. J. Rutt, Z. A. Gál, S. G. Denis, M. A. Hayward and S. J. Clarke, J. Am. Chem. Soc., 2004, 126, 1980–1991 CrossRef CAS PubMed.
- R. Marx, Z. Naturforsch. B, 1995, 50, 1061–1066 CrossRef CAS.
- D. Bräunling, O. Pecher, D. M. Trots, A. Senyshyn, D. A. Zherebtsov, F. Haarmann and R. Niewa, Synthesis, crystal structure and lithium motion of Li8SeN2 and Li8TeN2, 2010 Search PubMed.
- M. Bolte and H.-W. Lerner, Acta Crystallogr., Sect. E: Crystallogr. Commun., 2001, 57, m231–m232 CrossRef CAS.
- A. Pfitzner, J. Cockcroft, I. Solinas and H. Litz, Z. Anorg. Allg. Chem., 1993, 619, 993–998 CrossRef CAS.
- M. K. Yadav and B. Sanyal, J. Alloys Compd., 2015, 622, 388–393 CrossRef CAS.
- A. El Maslout, J.-P. Motte, A. Courtois and C. Gleitzer, J. Solid State Chem., 1975, 15, 213–217 CrossRef CAS.
- R. Brandes and R. Hoppe, Z. Anorg. Allg. Chem., 1994, 620, 2026–2032 CrossRef CAS.
- J.-H. Zhang, D. J. Clark, J. A. Brant, C. W. Sinagra, Y. S. Kim, J. I. Jang and J. A. Aitken, Dalton Trans., 2015, 44, 11212–11222 RSC.
- K. Mahendran, K. Sujatha, R. Sridharan and T. Gnanasekaran, J. Alloys Compd., 2003, 358, 42–47 CrossRef CAS.
- L. Isaenko, A. Yelisseyev, S. Lobanov, A. Titov, V. Petrov, J.-J. Zondy, P. Krinitsin, A. Merkulov, V. Vedenyapin and J. Smirnova, Cryst. Res. Technol., 2003, 38, 379–387 CrossRef CAS.
- H. v Schnering and W. Wichelhaus, Naturwissenschaften, 1972, 59, 78–79 CrossRef.
- H. Liu, H. Wu, H. Yu, Z. Hu and Y. Wu, Dalton Trans., 2019, 48, 16626–16632 RSC.
- Y. Le Page and P. Strobel, Acta Crystallogr., Sect. B: Struct. Sci., 1980, 36, 1919–1920 CrossRef.
- N. Peng, X. Cheng, H. Yu, H. Zhu, T. Liu, R. Zheng, M. Shui, Y. Xie and J. Shu, Energy Storage Mater., 2019, 21, 297–307 CrossRef.
- F. Q. Huang, Y. Yang, C. Flaschenriem and J. A. Ibers, Inorg. Chem., 2001, 40, 1397–1398 CrossRef CAS PubMed.
- Y. Laligant, Eur. J. Solid State Inorg. Chem., 1992, 29, 239–247 CAS.
- T. Hasegawa and H. Yamane, Dalton Trans., 2014, 43, 14525–14528 RSC.
- A. Lindemann, J. Kuchinke, C. Köster, A. Hammerschmidt, M. Döch, T. Pruss and B. Krebs, Phosphorus, Sulfur Silicon Relat. Elem., 2001, 169, 169–172 CrossRef.
- R. Klevtsova, S. Solodovnikov, L. Glinskaya, V. Alekseev, K. Khalbaeva and E. Khaikina, J. Struct. Chem., 1997, 38, 89–95 CrossRef CAS.
- D. Caurant, G. Wallez, O. Majérus, G. Roisine and T. Charpentier, Lead in Glassy Materials in Cultural Heritage, 2024, pp. 37–92 Search PubMed.
- I. B. Kacem, L. Gautron, D. Coillot and D. R. Neuville, Chem. Geol., 2017, 461, 104–114 CrossRef.
- V. Tomashyk, Quaternary alloys based on II-VI semiconductors, CRC Press, 2014 Search PubMed.
- M. Li and A. Verena-Mudring, Cryst. Growth Des., 2016, 16, 2441–2458 CrossRef CAS.
- B. Kelleher, K. Dolan, M. Anderson and K. Sridharan, Nucl. Technol., 2016, 195, 239–252 CrossRef.
- J. Xie and Y. Xie, Chem. – Eur. J., 2016, 22, 3588–3598 CrossRef CAS PubMed.
- R. Jalem, Y. Yamamoto, H. Shiiba, M. Nakayama, H. Munakata, T. Kasuga and K. Kanamura, Chem. Mater., 2013, 25, 425–430 CrossRef CAS.
- Ž. P. Čančarević, J. C. Schön and M. Jansen, Chem. – Eur. J., 2007, 13, 7330–7348 CrossRef PubMed.
- E. Dvoryanova, I. Kondratyuk and I. Garkushin, Russ. J. Inorg. Chem., 2010, 55, 1136–1141 CrossRef CAS.
- A. Manyakova, E. Egorova and I. Garkushin, Russ. J. Inorg. Chem., 2018, 63, 1232–1235 CrossRef CAS.
- J. Hauck, Z. Naturforsch. B, 1969, 24, 1067–1068 CrossRef CAS.
- G. Meyer and H.-C. Gaebell, Mater. Res. Bull., 1983, 18, 1353–1360 CrossRef CAS.
- V. Nagarkar, E. Ovechkina, H. Bhandari, L. Soundara-Pandian, M. More, R. Riedel and S. Miller, Phys. Proc., 2015, 69, 161–168 CrossRef CAS.
- T. S. Ortner, J. P. Scheifers, J. Flores, Y. Zhang, A. K. Iyer, T. M. Gesing and B. P. Fokwa, Eur. J. Inorg. Chem., 2019, 3526–3535 CrossRef CAS PubMed.
- S. Hibble, I. Fawcett and A. Hannon, Acta Crystallogr., Sect. B: Struct. Sci., 1997, 53, 604–612 CrossRef.
- J. Schön, M. Wevers and M. Jansen, Solid State Sci., 2000, 2, 449–456 CrossRef.
- M. Sorescu, E. Knobbe, J. Martin, J. Barrie and D. Barb, J. Mater. Sci., 1995, 30, 5944–5952 CrossRef CAS.
- X. He, Q. Bai, Y. Liu, A. M. Nolan, C. Ling and Y. Mo, Adv. Energy Mater., 2019, 9, 1902078 CrossRef CAS.
- S. Feng, Z. Wang, G. Zhang, P. Yue, W. Pan, Q. Lu, H. Guo, X. Li, G. Yan and J. Wang, Phys. Chem. Chem. Phys., 2024, 26, 25881–25889 RSC.
- E. Zintl and G. Brauer, Z. Elektrochem. Angew. Phys. Chem., 1935, 41, 102–107 CrossRef CAS.
- W. Li, M. Li, H. Ren, J. T. Kim, R. Li, T.-K. Sham and X. Sun, Energy Environ. Sci., 2025, 18, 4521–4554 RSC.
- T. Lapp, S. Skaarup and A. Hooper, Solid State Ionics, 1983, 11, 97–103 CrossRef CAS.
- N. Tapia-Ruiz, M. Segalés and D. H. Gregory, Coord. Chem. Rev., 2013, 257, 1978–2014 CrossRef CAS.
- S. R. Xie, S. J. Honrao and J. W. Lawson, Chem. Mater., 2024, 36, 9320–9329 CrossRef CAS.
- T. S. Thakur, L. Ercole and N. Marzari, Energy Environ. Sci., 2026 10.1039/D5EE07336G.
| This journal is © The Royal Society of Chemistry 2026 |