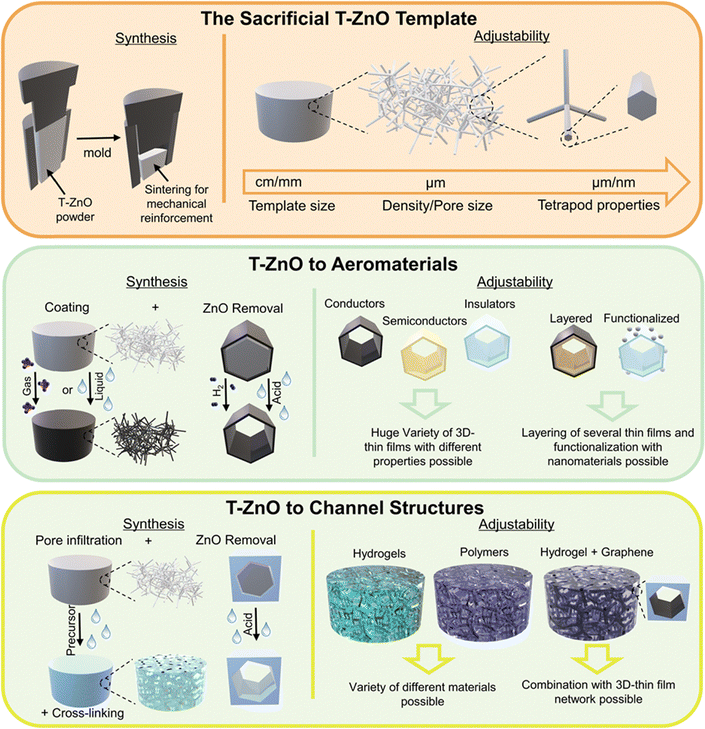
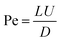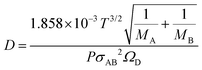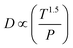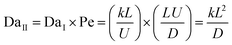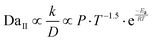Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Functional tetrapodal zinc oxide: from synthesis and multiphysics to advanced applications
Mozaffar
Abdollahifar†
 *ab,
Erik
Greve†
*ab,
Erik
Greve†
 a,
Jonas
Lumma†
a and
Rainer
Adelung
a,
Jonas
Lumma†
a and
Rainer
Adelung
 *ab
*ab
aChair for Functional Nanomaterials, Department of Materials Science, Faculty of Engineering, Kiel University, Kaiserstr. 2, 24143 Kiel, Germany. E-mail: moza@tf.uni-kiel.de; ra@tf.uni-kiel.de
bKiel Nano, Surface and Interface Science (KiNSIS), Kiel University, Kaiserstr. 2, 24143 Kiel, Germany
First published on 19th March 2026
Abstract
Tetrapodal zinc oxide (T-ZnO) represents a distinctive ZnO architecture, characterized by a central core and four tetrahedrally arranged monocrystalline arms. Beyond its role as a conventional particle, T-ZnO is increasingly recognized as a microscopic building block whose three-dimensional geometry governs tightly coupled multiphysics properties. This review critically examines the structural lifecycle of T-ZnO across multiple length scales. We evaluate scalable synthesis methodologies and competing thermodynamic models governing early-stage nucleation, followed by a systematic analysis of structure–property relationships. Structure–property correlations are addressed from the atomic scale of the crystal lattice, through the microscopic scale of individual tetrapodal particles, to the macroscopic aggregation of tetrapods into functional networks. Across these scales, the tetrapodal morphology dictates properties like mechanics, piezoelectricity, and optoelectronics via diverse and often competing effects. These properties are typically intertwined, producing physical coupling that generates both performance synergies and fundamental design trade-offs. The utility of this architecture is demonstrated in two principal domains: first, as an active functional particle network leveraging geometric stress concentration and percolation effects for applications such as advanced sensing; and second, as a sacrificial structural template for the fabrication of ultralight, highly porous aeromaterials that decouple the architectural advantages of the network from the intrinsic chemical limitations of ZnO. Ultimately, advancing T-ZnO from empirical optimization to rational technological deployment requires predictive multiphysics computational frameworks capable of guiding the design of its complex functionality.
1. Introduction
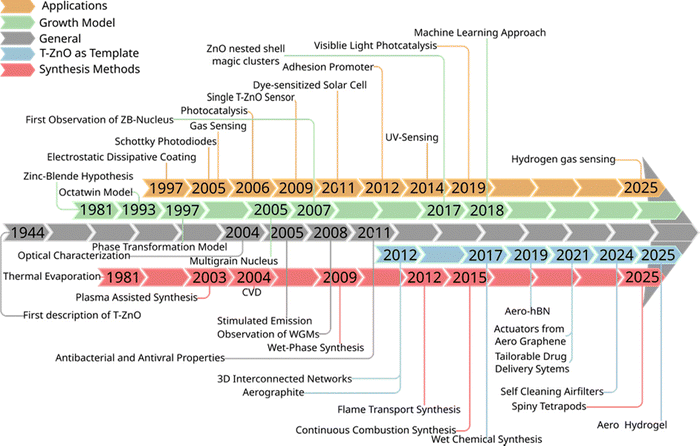
Predictive control over functional materials remains one of the foremost challenges in materials science. This problem is deeply rooted in the synthesis-structure–property relationship that governs material performance. Tetrapodal zinc oxide (T-ZnO) exemplifies this challenge. Its functional properties are governed not only by its intrinsic chemistry but also by its distinct morphology (a particle with a central core and four arms extending in different directions at roughly 109.5° angles) and are highly sensitive to its synthesis conditions. The scientific journey of T-ZnO, chronicled over eight decades in Fig. 1, began with the foundational crystallographic description of T-ZnO twinning in 1944.1 It evolved through the postulation of seminal yet contested growth models, ranging from the simple metastable zinc blende (ZB) nucleus hypothesis (1981)2 to the complex octatwin (1993)3 and multi-grain wurtzite (WZ) (2005)4 theories. Furthermore, the timeline in Fig. 1 marks the transition from fundamental understanding to a dual yet interconnected landscape of technological applications. Early breakthroughs demonstrated its utility as an active material in photocatalysis (2007),5 single-tetrapod sensors (2009),6 and as a light-scattering layer in dye-sensitized solar cells (DSSC) (2011).7 Concurrently, a transformative application emerged with the synthesis of aerographite (2012),8 as illustrated by the emergence of the blue “template” stream in Fig. 1. This established T-ZnO not just as a functional component, but as a revolutionary sacrificial template for creating entirely new classes of microstructured materials, such as ultralightweight three-dimensionally (3D) interconnected thin films, the so called “aeromaterials”.8,9Despite extensive studies on T-ZnO synthesis, the fundamental atomistic mechanisms driving the formation of its characteristic four-armed morphology are still not fully understood.2,3,10,11 This fundamental uncertainty creates a critical knowledge gap between macroscopic synthesis parameters and atomistic control, limiting the rational and predictive design of T-ZnO. In addition to the knowledge gap regarding its formation, T-ZnO is characterized by structural features such as a defect-rich core, a non-centrosymmetric WZ-lattice in its arms, and high-energy polar facets. These features are often dependent on synthesis conditions, closely related, and can influence each other. For instance, intrinsic defects enable n-type conductivity,12,13 and the concentration of these defects, directly affects the material's conductivity. These intrinsic defects also influence the piezoelectric properties of the WZ arms through free charge carriers14,15 and reduce optical efficiency via non-radiative recombination centers.16 Furthermore, structural features such as the core junction, also play an important role.14 In this junction, the degree of lattice mismatch and the presence of defects further influence the behavior of T-ZnO in various regards. This complex interdependency of the structural features and their effects to the properties needs to be tuned for desired application but also enables multi-terminal functionalities.17
The systematic analysis of these interconnected phenomena establishes a foundation for the practical use of T-ZnO in materials science. Crucially, our analysis reveals that T-ZnO acts as a material nexus, uniting functional and structural domains. T-ZnO can be rationally applied in two synergistic modes. First, it can serve as an active material, where its intrinsic properties such as n-type conductivity, piezoelectricity, or photoluminescence (PL) are directly exploited and often enhanced by its tetrapodal structure. Second, T-ZnO can function as a load-bearing structural element, where its morphology enables new properties, such as the formation of three-dimensionally percolating networks with low percolation thresholds. Moreover, the architecture of these networks can be transferred to other materials. In such a templating approach, T-ZnO can imprint its structural properties onto materials like graphene,18 SiO219 or ceramics,20 which are otherwise difficult to produce in such a morphology. This strategy effectively decouples morphology from chemistry, allowing T-ZnO to export its structural benefits to other functional chemistries.
This review provides a critical, multi-scale framework to clarify the current challenges regarding T-ZnO. We begin with a critical assessment of the synthesis and unresolved growth mechanisms of T-ZnO (Section 2). We then trace how these conditions shape the structure–property relationships (Section 3), which govern the material's vast application potential (Section 4). Following the detailed discussion of synthesis, properties and applications of T-ZnO, the review culminates in a forward-looking perspective (Section 5). This section outlines key experimental and theoretical frontiers. Progress in these areas may move T-ZnO from a model system of study toward a blueprint for a new paradigm: the predictive, on-demand design of three-dimensionally microstructured materials.
2. Synthesis and growth mechanisms of T-ZnO
Despite decades of research and a large variety of synthesis routes, the predictable design of T-ZnO with tailored arm morphologies and core structures remains difficult. This challenge stems from the material's distinctive architecture. Unlike zero-dimensional (0D), one-dimensional (1D), or two-dimensional (2D) nanostructures, T-ZnO exhibits a spatially open three-dimensional (3D) tetrapodal morphology. This unique morphology comprises a central core from which four high-aspect-ratio arms extend. This geometric configuration imparts properties that differentiate it from other ZnO forms. Consequently, deviations in morphology directly alter material properties. This structure–property relationship is central to governing T-ZnO's performance across all scales, from individual arms to assembled networks. Consequently, it emphasizes the importance of a combined theoretical and empirical understanding of nucleation and growth.This section critically assesses the synthesis-mechanism, arguing that a unified growth theory, bridging atomistic nucleation with macroscopic process parameters, is the critical missing link for realizing the full potential of these unique 3D nano-/micromaterials. To address this gap, the section first examines the fundamental growth mechanisms of the nucleus (Section 2.1). Building on this foundation, Section 2.2 develops the underlying thermodynamic principles and general design rules, which are then applied to the primary fabrication methods for T-ZnO in Section 2.3. This creates a rational framework that enables deliberate control over synthesis parameters, shifting the field from empirical recipes to first-principles design.
2.1 Fundamental growth mechanism
This section traces the evolution of scientific thought on the nature of the T-ZnO nucleus. Early models postulated that a core of a metastable crystal phase, specifically cubic ZB, provides the necessary tetrahedral symmetry.11 Conversely, later theories argued that the structure could arise entirely from the thermodynamically stable WZ phase through complex twinning and strain accommodation mechanisms. This progression reveals how increasingly sophisticated characterization methods have deepened our understanding of symmetry breaking and crystal growth at the nanoscale.
2.1.1.1 Model A: the epitaxial growth model (1981). The first and most intuitive explanation for the tetrapod morphology, proposed by Shiojiri and Kaito in 1981, is the epitaxial growth model.11,15 This framework suggests that the tetrapod does not nucleate from the stable WZ phase, but rather from a core composed of the metastable cubic ZB polymorph. The formation of such a metastable phase is theorized to be plausible under the high-supersaturation, non-equilibrium conditions characteristic of vapor-phase synthesis.
In this model, the ZB nucleus forms an octahedron, exposing four energetically equivalent Zn-terminated facets. These surfaces act as a template for the epitaxial growth of WZ arms. As deposition proceeds along the crystallographically equivalent directions, the arms extend to form a structure with ideal tetrahedral symmetry, producing the characteristic tetrapod.26,27 This epitaxial relationship explains the fourfold symmetry and predicts a tetrahedral arm angle of 109.5°, the ideal tetrahedral angle.24,28 This model predicts a perfect tetrahedral alignment of WZ arms extending from the ZB octahedral nucleus (Fig. 2a). Direct high-resolution transmission electron microscopy (HRTEM) analysis confirms this ZB core in specific samples, validating the epitaxial relationship between the ZB {111} and WZ 〈0001〉 growth directions (Fig. 2b).
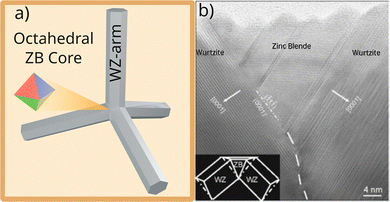 | ||
| Fig. 2 Epitaxial growth model for tetrapod ZnO. (a) Schematic illustration of a ZB octahedral nucleus (left) and the resulting WZ tetrapod with arms aligned in perfect tetrahedral geometry (right). (b) High-resolution TEM image showing a ZB core enclosed by WZ domains, with the corresponding structural model inset, providing direct evidence for the epitaxial relationship between ZB {111} and WZ {0001} growth directions. (b) Reproduced from ref.10 with permision from AIP Publishing,10 Copyright 2007. | ||
While this simplicity makes model A conceptually attractive, it also defines its principal limitations. Several factors limit the applicability of this model: systematic experimental deviations from ideal tetrahedral geometry, the energetic cost of maintaining a coherent ZB–WZ interface, and the rarity of direct ZB-core observations. Collectively, these suggest that this mechanism, if operative, is restricted to a narrow range of synthesis conditions. These shortcomings motivated the development of alternative models that relax the requirement of a perfect crystallographic template and instead incorporate strain, defects, or phase transformation as central elements of tetrapod nucleation.
2.1.1.2 Model B: the octa-twin nucleus model (1993). To address the limitations of the epitaxial model, Iwanaga et al. developed the octa-twin model, which rationalizes tetrapod formation entirely within the stable WZ phase.3,24,25 A crucial advantage of this model is its ability to account for the persistent ∼105° arm angle frequently observed in experiments.21,29
This model postulates a nucleus composed of eight trigonal pyramidal WZ sub-crystals. Each sub-crystal exposes three outward-facing {1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 22} facets and one basal plane. All sub-crystals are twinned along the outward facing facets, which are the most energetically favorable twinning planes in the WZ structure. These twinned domains arrange into an octahedron, introducing significant internal strain due to angular mismatches of the twin boundaries.30 This strain is partially relieved by microcracking or dislocation formation, resulting in a distorted nucleus. Growth then proceeds preferentially from four sub-crystals whose basal planes are oriented along the +c direction, producing the characteristic four-armed tetrapod morphology.25 As the nucleus evolves, it transitions into a hexa-twin configuration (Fig. 3), where mutual accommodation of twin interfaces minimizes interfacial stress.
22} facets and one basal plane. All sub-crystals are twinned along the outward facing facets, which are the most energetically favorable twinning planes in the WZ structure. These twinned domains arrange into an octahedron, introducing significant internal strain due to angular mismatches of the twin boundaries.30 This strain is partially relieved by microcracking or dislocation formation, resulting in a distorted nucleus. Growth then proceeds preferentially from four sub-crystals whose basal planes are oriented along the +c direction, producing the characteristic four-armed tetrapod morphology.25 As the nucleus evolves, it transitions into a hexa-twin configuration (Fig. 3), where mutual accommodation of twin interfaces minimizes interfacial stress.
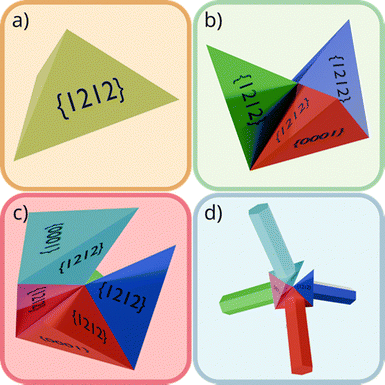 | ||
| Fig. 3 Twin-mediated nuclei with increasing multiplicity. (a) Single pyramidal twin with an equilateral triangle as base. (b) Three additional twins grown on the {1212} facets of the first twin. (c) Hexa-twin configuration (after Iwanaga et al.25), in which all twinned interfaces are mutually accommodated, releasing interfacial stress and yielding a distorted tetrahedral arrangement of the c+ axes; the {0001} facet is indicated. (d) Resulting distorted tetrapod morphology. | ||
In direct contrast to the epitaxial growth model, the octa-twin nucleus model abandons the concept of a predefined symmetry template and instead explains tetrapod formation entirely within the stable WZ phase. Here, the morphology results not from the crystallographic properties but emerges from strain-driven twinning during nucleation. This distinguishes model B from model A and marks the first explicit incorporation of defect-mediated growth.
The octa-twin model offers an explanation for the consistent ∼105° arm angles seen in experiments, attributing the deviation from 109.5° to the relaxation of the twinned nucleus structure.25 This interpretation is supported by angular measurements on multiple tetrapods and by indirect TEM evidence suggesting the presence of twinned subdomains, even though direct imaging of a complete eight-domain nucleus remains elusive.3,24,25 Lastly, the complexity of the proposed nucleus structure and variability in observed tetrapod morphologies suggest this may be one of several possible growth pathways, rather than a universal mechanism for all T-ZnO structures.
2.1.1.3 Model C: the nucleus transformation model (1997). A third growth mechanism proposed by Nishio et al.2 presents a hybrid model that links cubic and hexagonal phases in ZnO tetrapod formation. Growth initiates with a transient ZB nucleus formed under high supersaturation and elevated temperatures typical of vapor-phase synthesis.2 The metastable ZB phase subsequently undergoes a solid-state transformation into the WZ structure via crystallographic slip, accompanied by the formation of stacking faults and twin boundaries. This transformation preserves the octahedral geometry of the original ZB nucleus but produces a multiply twinned WZ core. Depending on the slip system, the core typically consists of either four or six coherently twinned WZ domains. In the six-domain configuration, specific twins form the internal nucleus boundaries, while the remaining domains elongate outward to form the arms. Such a sixfold twinned core cannot be accounted for by the previously proposed octa-twin model. From this multiply twinned nucleus, four WZ domains aligned along the c+ direction extends outward to form the characteristic tetrapod arms. Diffraction data confirms the existence of multiply twinned {1
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 24} domains (Fig. 4), supporting the transformation-based mechanism by validating the specific orientation of the WZ elements.
24} domains (Fig. 4), supporting the transformation-based mechanism by validating the specific orientation of the WZ elements.
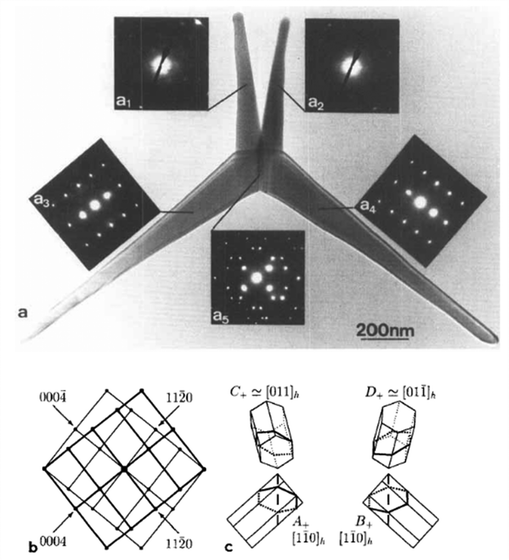 | ||
Fig. 4 TEM analysis of a T-ZnO particle with a multiply twinned {1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 24} core. (a) TEM image of a tetrapod with four arms, accompanied by selected-area electron diffraction (ED) patterns for each domain (a1–a5). (b) Schematic of the ED pattern highlighting the characteristic reflections of the twinned domains. (c) Crystal orientation models of the four WZ elements (A+, B+, C+, D+), showing the hexagonal prism geometry and twinning relationships. (a)–(c) Reproduced from ref. 2 with permision from Taylor & Francis,2 Copyright 1997. 24} core. (a) TEM image of a tetrapod with four arms, accompanied by selected-area electron diffraction (ED) patterns for each domain (a1–a5). (b) Schematic of the ED pattern highlighting the characteristic reflections of the twinned domains. (c) Crystal orientation models of the four WZ elements (A+, B+, C+, D+), showing the hexagonal prism geometry and twinning relationships. (a)–(c) Reproduced from ref. 2 with permision from Taylor & Francis,2 Copyright 1997. | ||
This model is primarily supported by Nishio et al.'s2 observation of a six-grained tetrapod, which has not been independently confirmed. It does, however, account for the experimentally observed inter-arm angles.
2.1.1.4 Model D: the multi-grain wurtzite model (2005). Ronning et al.4 proposed an alternative model based on direct HRTEM evidence and supported by density functional theory (DFT) calculations. In this model, the tetrapod core consists of four single-crystalline WZ grains arranged in a distorted tetrahedral geometry, with misfit strain accommodated by a high density of structural defects. They suggest a self-assembly nucleation mechanism resembling a phase transition, similar to that described by Nishio et al.2 Zn vapor initially forms small Zn clusters that oxidize into ZnO spheroids. Upon further growth, these spheroids collapse into a more stable tetrahedral structure. This final structure is composed of four triangular dipyramidal ZnO grains with their c-axes pointing toward the apexes (Fig. 5a). High-angle grain boundaries accommodate the internal misfit strain, effectively stabilizing the distorted tetrahedral arrangement (Fig. 5b–e).
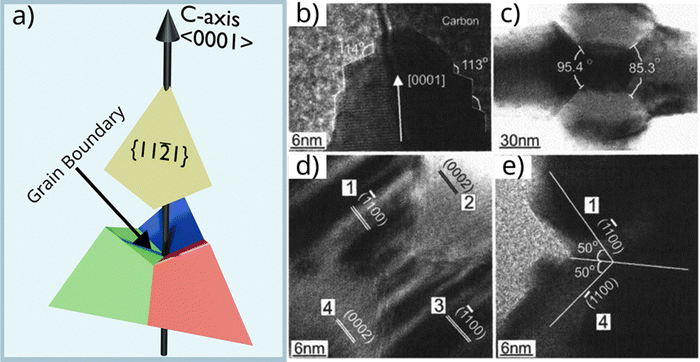 | ||
Fig. 5 Structural analysis of the multi-grain wurtzite (WZ) nucleus. (a) Schematic representation of the self-assembled tetrahedral core, comprising four WZ grains bounded by {1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 21} facets with outward-facing c-axes. (b) HRTEM image of a leg tip exhibiting 113°–114° growth steps. (c) Low- and (d) high-magnification TEM images of the central core, revealing four distinct hexagonal grains separated by high-angle grain boundaries. (e) HRTEM image of the boundary region detailing the twin relationship between adjacent arms.4 (b)–(e) Reproduced from ref. 4 with permision from AIP Publishing,4 Copyright 2005. 21} facets with outward-facing c-axes. (b) HRTEM image of a leg tip exhibiting 113°–114° growth steps. (c) Low- and (d) high-magnification TEM images of the central core, revealing four distinct hexagonal grains separated by high-angle grain boundaries. (e) HRTEM image of the boundary region detailing the twin relationship between adjacent arms.4 (b)–(e) Reproduced from ref. 4 with permision from AIP Publishing,4 Copyright 2005. | ||
The key contribution of this model is its emphasis on nanoscale thermodynamics. Rather than invoking a predefined crystallographic template such as a ZB phase or octa-twin, it describes a dynamic process in which the core structure emerges from energy minimization through particle aggregation and rearrangement. However, it remains unclear whether the initial nucleus originates from Zn condensation, as supersaturation of Zn vapor with respect to metallic Zn is not expected under the relevant conditions,31 making homogeneous Zn nucleation unlikely.
2.1.2.1 Condition-dependent pathways (thermodynamic vs. kinetic control). The diversity of synthesis methods implies that a universal nucleation pathway likely does not exist. Instead, the dominant mechanism appears to be dictated by the thermodynamic landscape of the specific synthesis method. Models A and C, which invoke metastable ZB phases, are predominantly supported by data from high-temperature vapor-phase synthesis. Under these far-from-equilibrium conditions, rapid kinetics can stabilize metastable phases before they relax into the equilibrium structure. In contrast, model D (multi-grain WZ) often aligns with wet-chemical or slower oxidation data. Here, slower kinetics allow the system to relax into the thermodynamically stable WZ phase, resolving the tetrapod geometry via defect accommodation rather than phase changes. It is physically plausible that distinct nucleation mechanisms operate in parallel, with the dominant pathway determined by local parameters such as temperature, supersaturation, and precursor chemistry.
2.1.2.2 Limits of ex situ characterization. A major obstacle is the temporal disparity between the nucleation event and structural characterization. Nucleation is a transient, non-equilibrium process occurring on rapid timescales at high temperatures. However, current evidence, such as the HRTEM imaging supporting models A and D, is predominantly obtained ex situ on mature nanostructures.
Researchers are therefore analyzing the thermodynamically relaxed state of the nucleus rather than the active nucleation complex. Distinguishing between a structure that initiated growth (e.g., a metastable ZB core) and one that formed via stress relaxation during cooling (e.g., a multi-grain WZ core) remains a significant analytical challenge. While model A relies on geometric elegance, it lacks broad statistical backing; conversely, model D provides direct imaging evidence but struggles to definitively explain the initial symmetry-breaking event without in situ verification.
2.1.2.3 Strain-induced structural reconfiguration. Finally, T-ZnO growth involves a complex coupling of atomistic and mesoscopic scales. The macroscopic morphology is governed by the core's crystallographic defects, such as twin boundaries and inversion domains. However, as the arms extend, they exert retroactive strain on the central core. This feedback loop can induce plastic deformation or defect migration that obscures the original crystallographic signature of the nucleation event. Consequently, the final core structure observed in microscopy may differ significantly from the initial nucleus, complicating efforts to reverse-engineer the mechanism solely from the final morphology.
Summary: evolution of nucleation concepts. The historical progression of T-ZnO nucleation models represents a conceptual evolution from idealized crystallography toward defect-mediated, thermodynamically driven theories. Early hypotheses, such as the epitaxial model (A), were notable for their geometric simplicity, relying on a perfect ZB core to explain the tetrapod's symmetry. However, the persistent experimental observation of non-ideal arm angles (∼105°) necessitated more physically robust descriptions. This drove the development of the octa-twin (B) and multi-grain (D) models, which do not require a perfect template. Instead, these modern models propose that the tetrapod morphology arises from the WZ phase itself, where the core structure is determined by the energetic accommodation of strain via defects. Current understanding thus positions T-ZnO nucleation not as a single fixed event, but as a dynamic competition between kinetic trapping and thermodynamic relaxation, as summarized in Table 1.
| Model (first proposed) | Nature of the nucleus | Primary driving force | Key supporting evidence | Key challenges/limitations |
|---|---|---|---|---|
| A: epitaxial growth (1981) | Metastable ZB crystal core. | Epitaxial growth of WZ arms on the four {111} faces of the ZB seed. | HRTEM/ED showing a distinct ZB core in some rare samples.10 | • Persistent angle discrepancy (∼105° vs. 109.5°) |
| • High energetic cost of ZB/WZ interface | ||||
| • Core is very rarely observed | ||||
| B: octa-twin nucleus (1993) | A complex, strained WZ octa-twin embryo composed of eight sub-crystals. | Accommodation and release of internal strain to explain the non-ideal (∼105°) arm angles. | Explains the experimentally observed non-ideal (∼105°) arm angles.25 | • Proposed nucleus is extremely complex |
| • Has not been directly and unambiguously observed | ||||
| C: nucleus transformation (1997) | An initial, transient ZB crystal that transforms into a twinned WZ structure. | Solid-state phase transformation from metastable ZB to stable, twinned WZ via crystallographic slips. | Reconciles evidence for a ZB origin with the final, defect-rich WZ structure, supported by the rare observation of a six-grained core.2 | • The proposed rapid, solid-state transformation is highly complex |
| • Key observation of a six-grained core has not been independently reproduced | ||||
| D: multi-grain wurtzite (2005) | Four distinct WZ grains that self-assemble in a twin-like relationship. | Thermodynamic energy minimization by reducing the high surface area of precursor clusters. | Direct HRTEM imaging of a four-grain core; strongly supported by DFT calculations.4 | Evidence for the final core is strong, but the dynamic self-assembly pathway remains a theoretical assumption |
2.2 Unifying principles: kinetic vs. thermodynamic control
The chemical state of the reactor, specifically the degree of supersaturation, acts as the unifying principle that governs the formation of T-ZnO. Synthesis parameters dictate the transport rates, determining whether the system has sufficient energy to overcome activation barriers or if it operates under strict kinetic control. These transport conditions directly dictate the atomic arrangement, determining whether atoms relax into stable lattices or become trapped in metastable states. As illustrated in Fig. 6, these structural differences dictate the final macroscopic shape, separating kinetically trapped products (such as hexagonal prisms) from thermodynamically stable morphologies (such as bipyramids).32 Therefore, understanding how macroscopic parameters shift the regime between thermodynamic and kinetic control is essential for predictive synthesis.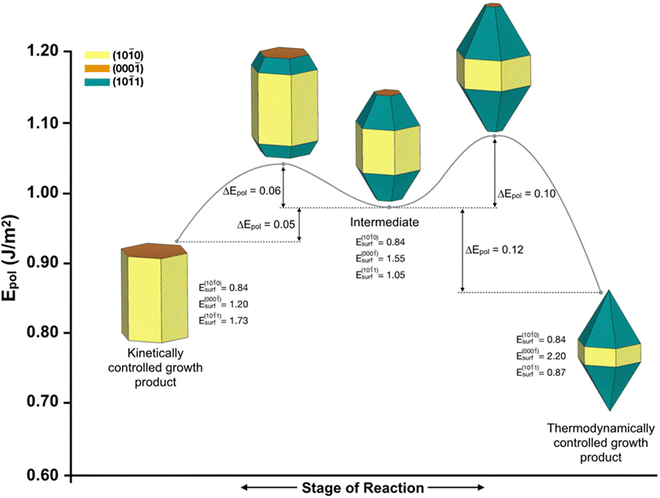 | ||
| Fig. 6 Quantitative energy landscape governing ZnO morphology. The reaction pathway illustrates the structural transition from a high-energy kinetically controlled product (left) to a surface-energy-minimized thermodynamically controlled product (right). The denoted energy barriers (ΔEpol) represent the activation energy required for these atomic rearrangements. Macroscopic synthesis parameters dictate whether the system overcomes these barriers or remains locked in a metastable morphology. Reproduced from ref. 32 with permision from MDPI,32 Copyright 2023. | ||
When a reactor operates at high temperatures but low supersaturation, the system falls under thermodynamic control where the surface diffusion of species exceeds their arrival rate.32,33 This high atomic mobility allows the atoms to bypass the formation of internal defects and relax into their lowest-energy lattice sites. Consequently, the crystal minimizes its overall surface energy to form compact, low-aspect-ratio prisms or quasi-spherical aggregates.27,32,34 A clear illustration of this transport behavior is Ostwald ripening, frequently observed in the wet-chemical synthesis of ZnO nanoparticles.35 As demonstrated by Wong et al.35 smaller, highly curved particles dissolve, releasing species that diffuse and deposit onto larger, more stable particles. This process confirms that thermodynamic control inherently erases high-energy anisotropic features in favor of compact, energetically stable structures.35
Conditions of high precursor flux and extreme supersaturation, typical of vapor-phase synthesis, drive the system into strict kinetic control.27,34 Elevated synthesis temperatures (typically >900 °C) increase the zinc vapor pressure, accelerating the arrival rate of growth species so that it overwhelmingly exceeds the surface diffusion rate.4,21,36–38 This kinetic arrest forces the system to initially form a nucleus via homogeneous nucleation in the gas phase, locking atoms into specific facets before they can relax into equilibrium sites.39 Subsequently, the rapid accumulation of material restricts growth to the most exposed facets, driving the anisotropic extension of the four arms along the 〈0001〉 directions.
Within this highly supersaturated gas phase, the effective nucleation density remains low because most subcritical clusters fail to overcome the critical nucleation barrier.31 Similar to Ostwald ripening during ZnO annealing,40 these structurally unstable atomic clusters re-evaporate rather than forming stable defect centers. The resulting zinc and oxygen species are then transported and recycled to feed the rapid elongation of the already established tetrapodal arms. If these intermediate nuclei were stabilized instead, the chemical supersaturation of the reactor would be rapidly depleted. This depletion would remove the driving force for kinetic control, inherently suppressing anisotropic extension and forcing a return to isotropic morphologies.
The temporal dynamics of this process were elucidated by Zhou et al.,41 who demonstrated how the chemical environment drives a two-step growth mechanism. Initially, a slower nucleation step locks the atomic arrangement of the core, after which extreme kinetic control forces arriving species onto specific outward-facing facets.31,41 This rapid transport sequence ultimately yields the high-aspect-ratio elongation of the tetrapodal arms. However, as demonstrated by the pivotal work of Ali and Winterer42 and supported by thermodynamic calculations,31 simply increasing the reactor temperature can raise the equilibrium vapor pressure and inherently reduce the degree of supersaturation. This loss of chemical driving force favors the gas phase, potentially suppressing the initial atomic nucleation event and preventing the required defect center from forming altogether.31
These findings provide critical experimental validation: reactor conditions must be deliberately engineered to maintain continuous, high supersaturation in order to sustain strict kinetic control.31,42 If this transport-driving force is lost, atoms will relax into equilibrium lattices rather than forming structural defects, and the highly anisotropic tetrapodal arms cannot be achieved. To rigorously resolve this causal interplay between the chemical environment and physical transport, it is necessary to quantify the system using the two fundamental dimensionless numbers that govern reactive flows.
To describe the physical transport of species within the highly supersaturated reactor environment, we first employ the Péclet number (Pe). This dimensionless number quantifies the regime of kinetic control by representing the ratio of the advective transport rate (bulk fluid flow) to the diffusive transport rate. Mathematically, it is expressed as:
In vapor-phase systems typical for T-ZnO synthesis, the mass diffusion coefficient (D) is dynamically governed by the local supersaturation and the surrounding thermal environment. The gas-kinetic dependence of this diffusivity dictates the threshold for kinetic control, which is rigorously described by the Chapman–Enskog equation:
By applying this specifically to gas-phase transport, we derive the critical scaling law that governs the Péclet regime in chemical vapor transport (CVT):
| Pegas ∝ P |
Conversely, fluids in hydrothermal or liquid-phase synthesis are effectively incompressible, meaning pressure cannot be used to tune the chemical supersaturation or D. Therefore, strict kinetic control via the Pe in these systems is achieved entirely by manipulating the advective precursor U. Martin et al.45 experimentally demonstrated this transport universality by tuning the flow rate to balance advection and diffusion (Pe ∼ 1.1). This specific transport balance allows arriving atoms to arrange into oriented, stable lattices rather than chaotic structural defects, ultimately dictating the uniform morphology of ZnO nanowires.45 However, as flow velocity increases further (Pe > 2), the extreme kinetic control forces atoms to precipitate too rapidly, preventing proper lattice arrangement and yielding severe macroscopic agglomeration.45
Second, to capture the chemical kinetics within a highly supersaturated environment, we introduce the first Damköhler number (DaI). This dimensionless ratio quantifies kinetic control by comparing the chemical reaction rate to the advective fluid transport rate:
In complex, high-temperature environments like FTS, a singular metric cannot fully capture the dynamic supersaturation. The reliable formation of T-ZnO requires a strict sequence of kinetic control: first initiating rapid homogeneous gas-phase nucleation, followed by sustained advection to feed the growing facets. To predict this sequential atomic arrangement, first forming a twinned defect core and then extending into a crystalline lattice, we couple the transport (Pe) and kinetic (DaI) descriptors into a unified master scaling law. By multiplying them, we eliminate the flow velocity (U) and derive the second Damköhler number (DaII), which serves as the ultimate predictor of the final tetrapodal arm morphology:
This unified framework mathematically explains the extreme sensitivity of the synthesis environment to temperature. By substituting the Arrhenius dependence for the chemical reaction rate (k) and the Chapman–Enskog dependence for gas-phase diffusion (D) into the second Damköhler equation, we derive the constitutive scaling law that governs strict kinetic control in vapor transport:
By formally linking these dimensionless transport parameters to their resulting crystallographic structures, this theoretical framework provides a powerful predictive tool for rational synthesis (summarized in Table 2). Crucially, this analysis resolves the apparent paradox of temperature in T-ZnO synthesis. It clarifies that while a high-temperature environment is initially essential to generate the extreme supersaturation required to trigger kinetic control (DaII ≫ 1) and form the foundational gas-phase defect core, this exact same thermal energy becomes a double-edged sword during the subsequent growth phase.
| Experimental parameter | Mechanistic impact & scaling law (kinetics) | Primary outcome: nucleation (DaII regime) | Secondary trade-off: growth (Pe regime) |
|---|---|---|---|
| Temperature (T) (universal) | Reaction dominates (k↑↑): rate constant k rises exponentially (Arrhenius), overwhelmingly outpacing gas-phase diffusion D. | Establishing the defect core: the extreme initial supersaturation drives DaII ≫ 1, forcing gas-phase atoms to instantly lock into twinned defects rather than relaxing into surface films.22,31 | Loss of supersaturation: sustained high T eventually undersaturates the environment. If kinetic control is not maintained via rapid quenching, atoms re-evaporate and the emerging arms dissolve.31 |
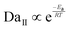
|
|||
| Reactor pressure (P) (vapor-phase specific) | Diffusion suppression (D↓): in highly supersaturated compressible gases, diffusivity is strictly suppressed inversely with total pressure (D ∝ 1/P) (note: in liquid/hydrothermal, P has negligible effect on D). | Transition to gas-phase defect centers increasing P drives DaII ≫ 1 by trapping reactants, ensuring the rapid formation of structural defects rather than equilibrium substrate lattices.43 | Anisotropic shaping: high P sustains strict kinetic control (high Pe), preventing lateral lattice relaxation and forcing atoms to extend into sharp, high-aspect-ratio tetrapodal arms.32 |
| Flow velocity (U) (universal) | Advection dominates (Pe↑): directly establishes kinetic control by overwhelmingly increasing the advective transport rate over lateral surface diffusion. | Yielding free-standing cores: rapid advection sweeps precursor species into the fluid stream, preventing them from structurally relaxing onto the reactor walls. | Morphology tuning: extreme kinetic control (high Pe) forces atoms into tapered tetrapodal arms. Conversely, a loss of advection (low Pe) allows atoms to relax into isotropic agglomerations.45 |
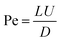
|
|||
If the newly formed nuclei are not rapidly transported to a cooler zone, the local environment loses its effective supersaturation. As shown by Saunders et al.,31 the zinc vapor actually becomes undersaturated at these sustained high temperatures. Without rapid quenching to maintain kinetic control (high Pe), the thermodynamic drive to re-evaporate completely overwhelms the advective growth rate.31 This thermal destruction erases the internal defects and dissolves the emerging tetrapodal arms. Consequently, the rational design of high-quality T-ZnO requires spatially decoupled reactors: an initial high-temperature zone is required to trigger extreme supersaturation and core nucleation (high DaII), followed immediately by a high-velocity, lower-temperature zone to sustain the kinetic control required to rapidly extend the arms (high Pe).15,41
The specific compositional gradient in the Zn/O ratio, measured from the core to the arm tip, physically records the fluctuating supersaturation and transport rates experienced by the particle as it traverses the reactor.45,47 This macroscopic behavior is directly supported by atomistic theory. Specifically, DFT and machine learning (ML) predictions demonstrate how these shifting chemical environments alter the localized energetics of individual atomic fragments. By dynamically tuning the relative stability of specific structural facets, these fundamental changes in kinetic control directly drive the structural transitions in the macroscopic morphology at the nanoscale.32
Therefore, the scaling laws summarized in Table 2 offer a predictive pathway for rational reactor engineering. For example, to force the elongation of blunt structures into sharp tetrapodal arms, the system must be pushed deeper into strict kinetic control (Pe ≫ 1). In gas-phase reactors, increasing pressure inherently suppresses lateral diffusion; in continuous hydrothermal systems, increasing precursor flow enhances advection. Both actions prevent atoms from relaxing into equilibrium lattices, successfully locking them into the extending tips (note: in static batch reactors where flow is negligible, the lack of advection naturally reverts the system to thermodynamic control, allowing atoms to form stable lattices that yield low-aspect-ratio isotropic prisms). Conversely, to increase the yield of free-standing tetrapods rather than substrate films, the priority shifts to the DaII. A sharp increase in temperature elevates the supersaturation, driving the reaction locus entirely into the gas phase (DaII ≫ 1). This rapid kinetic control ensures that precursors instantly bond to form twinned defect seeds before they can physically diffuse to the reactor walls. Ultimately, this decoupled transport framework moves the field beyond empirical trial-and-error, enabling the deliberate design of both initial yield and precise final morphology.21,37,43,45,48 This theoretical foundation provides a direct transition to the applied synthesis methods discussed in the following section.
2.3 Synthesis methods of T-ZnO
Tetrapodal ZnO can be synthesized via a variety of routes, which are broadly divided into liquid-phase and vapor-phase methods, which are discussed below. While liquid-phase routes offer potential advantages in low-temperature processing, vapor-phase approaches are the most established, widely utilized, and best understood for producing high-quality crystalline T-ZnO. As such, they will be discussed first.As established in Section 2.2, these methods rely on generating a high zinc flux to drive the system into the kinetic controlled regime, ensuring the formation of tetrpodal structures rather than compact crystals. Different vapor-phase methods, such as thermal evaporation, chemical vapor deposition (CVD), FTS, plasma-assisted growth, and combustion-based approaches can be distinguished by the zinc precursor, the heat source, and the strategy used to control the vapor pressures. Each method provides a unique way of creating the required non-equilibrium conditions for the self-organized emergence of tetrapodal morphology, each with its own profile regarding cost, scalability, energy consumption, and the precision of morphological control. Each vapor-based method is discussed in detail in the following sections.
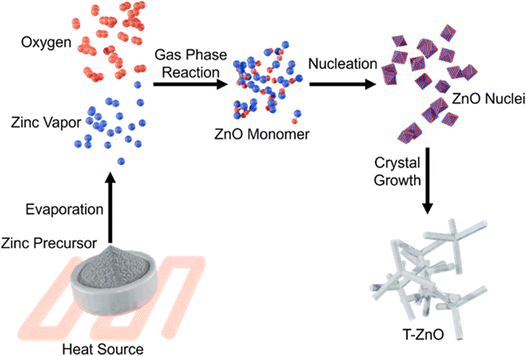
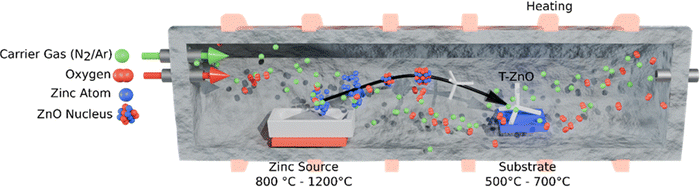
2.3.1.1 Thermal evaporation. Thermal evaporation represents the most classical route to synthesize T-ZnO. Thermal evaporation allows for high-throughput synthesis, but only offers limited control over the final morphology. The process relies on the direct thermal oxidation of a metallic zinc source (powder, foil, wires) in an oxygen-containing environment.4,49 Its key advantage is the simplicity of the setup (often just a tube furnace) which translates to low capital cost. Oxygen can be supplied either by ambient air4,49,50 or by a controlled atmosphere with a carrier gas such as Ar/N2.4,51 However, this simplicity is offset by high operational costs, as the process is energy-intensive, using external heat sources, with typical synthesis temperatures ranging from 650 °C to 1100 °C.4,21,22,33,52–55Fig. 7 schematically illustrates the thermal evaporation process. Zinc atoms evaporate and react with oxygen to form ZnO molecules, which nucleate and aggregate into ZnO nuclei. These clusters evolve into crystalline domains, ultimately growing into T-ZnO.
Depending on the setup, the oxidizing atmosphere can be static (sealed batch systems) or utilize a continuous input of oxidizer and/or zinc precursor. The process is most often conducted as a batch-type reaction, where Zn powder, foils or pellets are evaporated inside a tube furnace.49,51,53,55
The final dimensions are highly sensitive to the temperature gradient; higher deposition temperatures favor the growth of larger tetrapods with longer arms,4,27,53 while lower temperatures often yield shorter or irregular arms, as described in Section 2.1. Reaction times vary and exert a significant influence on final dimensions.4,53 The resulting tetrapods typically exhibit arm lengths between 0.3 and 10 µm and with diameters in the range of 30–650 nm, depending on exact synthesis conditions.4,49,50,56
Thermal evaporation enables the rapid and direct synthesis of T-ZnO in comparatively simple setups.2,52,54,57 This makes it attractive for applications requiring large amounts of material. The primary drawback of this method, however, is the inherent difficulty in precisely controlling these thermal and concentration gradients, leading to inhomogeneous nucleation and growth conditions across the deposition zone, resulting in a product with broad size and morphology distributions.23,34,56 This lack of monodispersity is a significant limitation for advanced electronic or optical applications where uniformity is paramount. Consequently, post-synthesis processing or selection is often necessary if narrow dimensional control is desired.52 Overall, thermal evaporation lacks the fine tunability of more advanced techniques like CVD or plasma-assisted processes. However, its low setup cost and proven ability to produce gram-scale quantities27,52 ensure its continued relevance. It remains particularly useful for mechanistic studies and applications requiring large volumes of material, such as fillers for polymer composites, where precise morphological uniformity is a secondary concern.
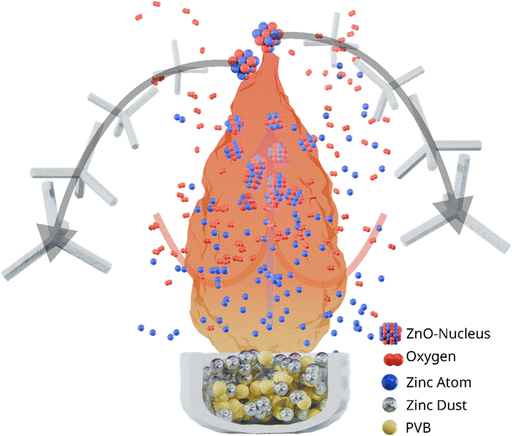
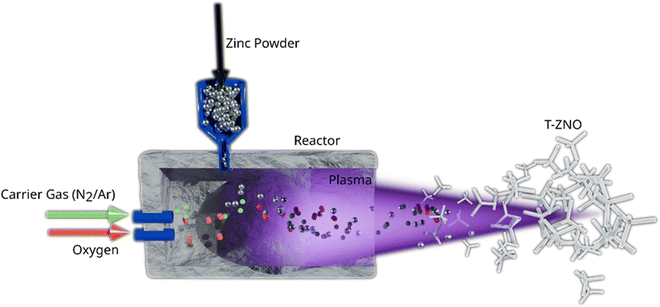
2.3.1.2 Combustion-driven synthesis: flame transport synthesis (FTS) and related methods. Combustion-driven approaches represent a scalable alternative to furnace-based heating, aiming to improve energy efficiency by utilizing the chemical energy of fuels. Among these, FTS is one of the most effective and widely adopted techniques for producing T-ZnO.12,21,58Fig. 8 illustrates this process, where a mixture of zinc microparticles and a sacrificial organic component undergoes combustion, generating a highly reactive flame atmosphere that vaporizes zinc and drives the nucleation and growth of T-ZnO. This method occupies a unique space in the synthesis landscape, offering very high throughput and reduced reliance on external energy, but at the cost of precise, steady-state process control.
A mixture of Zn microparticles and a sacrificial organic fuel undergoes combustion, generating a localized high-temperature, reactive atmosphere. The exothermic flame vaporizes zinc and creates transient Zn/O ratios that drive rapid nucleation and anisotropic growth of ZnO tetrapods.28,52 In a typical approach, a Zn microparticles: polyvinyl butyral (PVB) mixture is heated in a muffle furnace at ∼900–950 °C. The process combines zinc microparticles with a sacrificial organic component, such as PVB or ethanol. Upon combustion, this mixture generates a highly reactive atmosphere.21,37 Instead of relying entirely on external heating to vaporize the zinc, an external heat source is used to ignite the organic additive. The subsequent exothermic combustion generates a rapid, intense, and localized high-temperature environment that vaporizes zinc and establishes the supersaturation conditions required for tetrapod nucleation and growth.21,59 The PVB combustion releases Zn vapor along with CO2, H2O, and other volatiles.21
The advantage of FTS results from its dynamic, self-regulating chemical environment, which directly dictates the final morphology. Initially, the rapid combustion of the polymer consumes local oxygen, creating a transient, Zn-rich atmosphere. Initially, Zn and O atoms combine to form a primary cluster. Additional Zn and O atoms then attach to this cluster at different facets, leading to the formation of a stable nucleus. Once this nucleus is established, subsequent Zn and O atoms drive conventional one-dimensional spike growth along the c-axis (0001). Nucleation represents a critical step, while homogeneous nucleation predominantly yields tetrapod-type structures, heterogeneous nucleation can give rise to diverse morphologies, including multipods, flowers, and tree-like architectures.21,58,59 This intrinsic time-dependence is also seen in variants like ethanol-assisted flame transport synthesis (E-FTS), which yield morphologically distinct tetrapods.37,60 From a critical perspective, this dynamic nature is both a feature and a flaw. While it allows for the single-step synthesis of complex morphologies and facilitates the incorporation of dopant metals such as Cu, Al, and Sn,58,61 the lack of steady-state control makes achieving high batch-to-batch reproducibility and morphological uniformity a significant challenge. Furthermore, the use of a sacrificial fuel results in poor atom economy, a key consideration from a green chemistry standpoint.
Other combustion-based strategies have been explored to gain better control over the reaction environment. Xu et al.62 demonstrated counterflow diffusion flame (CDF) synthesis, where zinc substrates are exposed to a methane-air flame. The controlled gradients of temperature and oxygen yield diverse morphologies, including rods, ribbons, flowers, and tetrapods, with morphology being strongly dependent on flame conditions. Chen et al.63 reported a melting-combustion method (MCM), directly igniting molten zinc with an acetylene-oxygen flame to produce high-purity tetrapods without catalysts or volatile precursors. Growth follows a vapor-solid mechanism and occasionally shows bottleneck features along the arm.63 Offering perhaps the highest energy efficiency, Hwang et al.64 introduced a self-propagating high-temperature synthesis (SHS) approach. In this method, compacted Zn powder with an oxidizer (NH4NO4) is ignited by a heating coil in a closed chamber, the exothermic reaction sustains itself, generating mainly tetrapodal ZnO with aspect ratios up to 50 and yields of ∼95% under optimized conditions.
Collectively, these methods highlight the versatility of combustion as a scalable energy source for T-ZnO synthesis, though precise morphological control remains a common challenge across all variants. The methods also demonstrate that tetrapod growth is driven by strong advection. The formation of high-quality crystals in this chaotic environment confirms that T-ZnO growth thrives under conditions of strong advection (), where reaction rates dominate over diffusion.
2.3.1.3 Chemical vapor deposition (CVD). Representing the high-precision, moderate-throughput approach to T-ZnO synthesis, CVD and related vapor-transport methods offer a significant leap in morphological control compared to direct evaporation or combustion. Fig. 9 illustrates the principle of this process in a two-zone furnace setup, where Zn-based precursors are converted into a supersaturated Zn vapor that is transported and subsequently nucleates into T-ZnO crystals under controlled conditions. The key advantage of CVD is the spatial decoupling of the precursor source from the growth substrate, typically achieved within a two-zone furnace.15,53,65–68 This arrangement allows for independent and precise control over vapor generation and crystal nucleation. Consequently, it enables a level of tunability that is difficult to achieve with other methods. In contrast to FTS, which operates in highly turbulent conditions, CVD allows for the independent tuning of reaction temperature and precursor flux and ratio.
In a typical approach, carbothermal reduction of Zn-based precursors such as Zn, ZnO, or ZnCO3 with graphite generates a supersaturated Zn vapor that oxidizes under controlled atmospheres in a two-zone furnace.15,53,65–69 Using this strategy, Yu et al.70 produced high-quality, uniform T-ZnO nanocrystals with arm diameters of 80–100 nm and lengths of 600–1000 nm, guided by NiO nanoparticles that acted as catalytic nucleation sites on substrates held at ∼600 °C. Numerous catalyst-free variations have also been reported.53 For example, Newton et al.15 synthesized tetrapods with arm lengths from 200 nm to 10 µm and diameters between 50 and 500 nm simply by varying the oxygen content (0.5–5%) in the carrier gas, demonstrating the strong influence of Zn/O ratios on growth.15 This highlights the method's capacity to systematically explore the growth parameter space, where subtle shifts in partial pressure and substrate temperature determine the final morphology.65
Alternative variants further expand the accessible parameter space. Self-catalytic growth using zinc mixed with zinc acetate has been shown to promote anisotropic growth through a vapor-liquid–solid mechanism at temperatures as low as 650 °C.71 Catalyst-free CVD oxidation of pure Zn powders at 940–990 °C in Ar/O2 atmospheres yields tetrapod arms of 20–100 nm diameter and 1–15 µm length, with O2 concentration governing not only morphology67 but also optical properties. Higher oxygen flows enhance visible luminescence (590–610 nm) linked to oxygen vacancies, whereas moderate O2 favours stronger UV emission (∼3.2 eV excitons), directly connecting synthesis conditions to functional behaviour.67
Overall, CVD-based routes provide significant tunability, combining the simplicity of thermal evaporation methods with a good morphology control. Despite their advantages, CVD-based methods face several key limitations. They require advanced equipment, including high-temperature furnaces, gas flow controllers, and vacuum systems, which significantly increase both the cost and technical complexity. Additionally, the use of catalysts such as NiO or Zinc acetate, introduces health and environmental hazards.70,72 Scalability also remains limited by furnace volume and substrate area, making CVD not suitable for mass production of T-ZnO. Finally, the high operating temperatures (600–900 °C) contribute to energy consumption and carbon emissions, limiting the sustainability.
2.3.1.4 Plasma-assisted synthesis of T-ZnO. Plasma-assisted methods exploit highly reactive, non-equilibrium environments to enable rapid ZnO formation while simultaneously offering a pathway to sophisticated defect engineering. The fundamental advantage of plasma lies in its unique chemistry. Fig. 10 illustrates this process, where precursor gases are ionized into a plasma containing energetic ions, electrons, and radicals that drive ultrafast nucleation and growth of T-ZnO. By applying strong electromagnetic fields (microwave, DC), the precursor gases are ionized into a highly energetic state containing ions (Zn2+, O2−), electrons, and reactive radicals (O˙). These species possess much higher chemical potential than their neutral counterparts in thermal methods, allowing for extremely rapid nucleation and growth at lower overall substrate temperatures. This can also suppress thermal agglomeration, enabling the formation of much finer nanostructures.73
Early work by Zhang et al.73 used a microwave plasma furnace (400 W, 4 Torr, Ar/O2) to produce crystalline tetrapods with fine arms (10–25 nm), showing that plasma conditions can inhibit agglomeration and stabilize anisotropic growth. Lin et al.74 expanded this with a DC plasma reactor, where injected zinc powder was vaporized and oxidized, yielding tetrapods along with rods and nanoplatelets. The tetrapod yield (15–65%) depended on plasma power and particle residence time, and the process scaled up to ∼1 kg h−1, though post-processing was required. Lee et al.75 applied a 2.45 GHz microwave plasma (∼6500 K) with an N2/O2 swirl gas, producing tetrapods (∼30 nm diameter, ∼250 nm length). Yang et al.46 synthesized morphology-controlled ZnO tetrapods using a direct arc plasma discharge, showing that changing oxygen partial pressure-controlled shape and structure.
Recent atmospheric-pressure microwave plasma (APMP) systems allowed for more control. Yang and Jeong76 demonstrated that adjusting power and collection distance tuned tetrapod size, with larger structures forming at greater distances. Padmanaban et al.29 further showed that RF-powered microplasmas can produce defect-free nanoscale tetrapods (∼65 nm arms, ∼11 nm diameters).
High plasma powers or reduced oxygen concentrations strongly promote the formation of anisotropic ZnO microstructures, including well-defined tetrapods.77 While the influence of plasma pressure has not yet been systematically investigated, it is expected that lower pressures would favour the development of more faceted tetrapods. Plasma-assisted methods, however, inherently increase both the complexity and energy demands of synthesis, resulting in higher costs compared even conventional CVD approaches. Despite these drawbacks, certain plasma configurations have demonstrated the ability to produce high-quality T-ZnO with controlled morphology. One important advantage of plasma-based techniques is that they do not require specialized or expensive precursors, instead relying on elemental zinc. The costs increase even further considering the high energy demands for operating the plasma and possible external heat sources. Plasma-based methods demonstrate that extreme non-equilibrium conditions can be achieved by means other than temperature. This allows for the study of precursor ratios independently of thermal effects, offering a unique perspective on the growth mechanism. Table 3 summarizes the geometries and morphologies reported for each vapor-phase synthesis method. It highlights typical ranges in arm length/diameter and key features influencing morphology.
| Method | Typical conditions | Tetrapod arm size/morphology | Key advantages | Key limitations |
|---|---|---|---|---|
| Thermal evaporation | 650–1100 °C in a tube furnace; ambient or controlled O2 | Arms 0.3–10 µm; diameters 30–650 nm; sensitive to thermal gradients | Low capital cost, simple setup, high material throughput. | Poor morphological uniformity (broad size distribution), high energy consumption. |
| Combustion-driven (FTS, SHS, etc.) | Ignition of Zn ion fuel/oxidizer mixture (e.g., PVB, NH4NO3); ∼900–950 °C | Arms from nm to tens of µm; often features tapered tips due to dynamic gas environment. | High throughput, potentially energy-efficient (exothermic), single-step process. | Poor uniformity and reproducibility due to lack of steady-state control; poor atom economy. |
| Chemical vapor deposition (CVD) | Carbothermal reduction of ZnO in a 2-zone furnace; controlled gas flow. | Arms 200 nm-10 µm; diameters 20–500 nm; capable of highly uniform, high-quality crystals. | Excellent morphological and dimensional control, allows for in situ defect engineering. | Higher capital cost, complex setup, scalability limited by reactor size and deposition rate. |
| Plasma-assisted synthesis | Microwave/DC/RF plasma; high power density; low- or atmospheric-pressure. | Very fine arms 10–65 nm; can produce defect-free or intentionally engineered crystals. | Extremely rapid growth rates, simultaneous control over morphology and defects. | Very high capital and operational (energy) costs, complex process control. |
Ultimately, the selection of a vapor-phase synthesis route is a strategic decision dictated by the classic engineering trade-off between production scale, material quality, and cost. The methods discussed represent a spectrum of choices along this continuum.
At one end, thermal evaporation and combustion-based approaches offer pathways to high-throughput, gram-scale production, making them suitable for bulk applications like composites and fillers; however, this scalability is fundamentally coupled with poor morphological homogeneity and, in some cases, high energy consumption or low atom economy. In contrast, CVD provides unparalleled precision, enabling the fabrication of uniform, defect-engineered T-ZnO crystals essential for high-performance devices, albeit with significant penalties in setup complexity and scalability.
Plasma-assisted synthesis emerges as a compelling outlier, a potential paradigm-shifter that promises both extreme speed and fine nanostructure control. However, its very high energy and capital costs currently remain a significant barrier to widespread adoption. The persistent challenge in the field, therefore, is to develop hybrid or novel synthetic routes that unite the scalability of combustion with the precision of CVD in an economically and environmentally sustainable manner.
2.3.2.1 Aqueous solution growth. The simplest wet-chemical approach involves direct precipitation from an aqueous solution. Using Zinc sulfate and ammonia, branched rods, crosses, and tetrapods can form within 15 minutes at ∼90 °C.6,78 The chemistry of this process is far more nuanced than the simple addition of Zn2+ and OH− ions. In reality, the zinc ions exist as stable coordination complexes (e.g., tetraamminezinc, [Zn(NH3)4]2+). The growth of ZnO occurs via the slow, controlled decomposition of these complexes, which provides a steady supply of the fundamental growth units to the crystal surfaces. The process also relies on the nucleation of a ZnO nucleus, from which arms extend by the addition of Zn2+ and OH− ions in the complex solution.6 The resulting tetrapods typically display arm lengths of about 5 µm and diameters below 1 µm, with rather good homogeneity.6 While straightforward, this method's outcome is extremely sensitive to subtle fluctuations in pH, temperature, and concentration, which alter the delicate equilibrium of the zinc complexes, making precise morphological control and reproducibility a significant challenge.
2.3.2.2 Mechanisms of aqueous growth and morphological control. The wet phase synthesis employ Zn2+ and OH, for example zinc sulfate, Zinc acetate and ammonia or sodium hydroxide. For the case of ammonium containing solutions, ZnO formation is governed by the equilibria between [Zn(OH)3]2− and [Zn(NH3)4]2+, which therefore determines the activity of Zn2+. Thus, they control axial and lateral growth at pH's of 10–11 and temperatures above 75 °C as they decompose to feed anisotropic extension along 〈0001〉 directions.79 Fast nucleation under these conditions yields a ZB nucleus (as described in the Epitaxial growth model, Section 2.2.2) from which four WZ arms grow, leading to the tetrapodal morphology.6 The exact morphology is further tuned by capping molecules that selectively adsorb on polar or non-polar facets. Ammonia favors axial growth, whereas citrate or PVA can suppress 〈0001〉 extension or induce twinning, leading to the growth toward disks or dumbbells via facet passivation or bridging at (0001).80 Accordingly, these outcomes reflect shifts in the surface-energy landscape which can be explained on the basis of Wulff-based morphology maps and how ligands, solvent, and temperature alter relative facet energies to drive branching and arm growth.32 Integrating supersaturation kinetics with complexation chemistry and targeted facet capping thus enables reproducible wet-chemical synthesis of T-ZnO.
2.3.2.3 Microemulsion-mediated and solvothermal methods. To exert greater control over nucleation and growth, more sophisticated methods have been developed. Microemulsion-mediated hydrothermal methods attempt to address morphology control by confining nucleation and growth within surfactant-stabilized droplets. These droplets act as individual nanoreactors, limiting nucleus size and promoting uniformity. Jiang et al.81 used this approach to produce highly crystalline ZnO nanotetrapods with uniform leg lengths (200–300 nm) at 120 °C for 12 h. The confinement effect of the reverse micelles, together with surfactant interactions with ZnO surfaces, is believed to bias growth along the [0001] axis. This mechanism, however, is still not well understood, and the method depends on a balance of surfactant, co-surfactant, and precursor concentrations.81
Alcohol-based wet chemical synthesis has also been described in the literature,82 where zinc alkoxides or acetates are decomposed in alcohol solutions, sometimes with stabilizers or surfactants. This approach can generate tetrapods with arm diameters below 100 nm and relatively uniform morphologies. The use of organic solvents and stabilizers can provide different coordination environments for the zinc precursor compared to water, offering another lever for controlling growth kinetics. Variants that incorporate dopants such as Mg have been shown to increase yield and improve structural homogeneity.82 Nevertheless, these alcoholysis routes face similar challenges to hydrothermal methods: they are highly sensitive to precursor chemistry, reaction conditions, and additives, which makes reproducibility and scale-up difficult.
To sum up, wet-chemical synthesis provides a low-cost, low-temperature alternative to vapor-phase methods for T-ZnO, enabling direct formation of nanoscale tetrapods without specialized hardware and offering compatibility with flexible substrates for applications in catalysis and sensing. Compared to vapor-phase approaches, however, these methods often suffer from lower morphological uniformity, high sensitivity to precursor chemistry, and limited reproducibility, which complicates scale-up. Their main practical advantage lies in accessibility and affordability, but they also introduce environmental problems, the solvents, surfactants, and additives contaminate water, requiring removal or disposal. The resulting tetrapods must be thoroughly washed to eliminate residual chemicals. Future progress will depend on greener chemistries and simplified purification strategies that preserve the low-threshold nature of wet synthesis while improving uniformity, scalability, and sustainability.
2.4 Computational and ML-driven insights
The competing experimental growth models highlight the current limitations in capturing nanoscale nucleation events. To overcome these limitations, the field is increasingly turning to advanced computational methods, particularly the integration of first-principles calculations with ML. This approach marks a crucial shift from describing a few plausible-but-unproven models to building a truly predictive framework for nanoparticle formation. This trend is underscored by the 2024 Nobel Prize in Chemistry for breakthroughs in computational protein design.A prime example of this paradigm is the work by Chen and Dixon,83 who developed a fragment-based energy decomposition method enhanced with ML to rapidly and accurately predict the stability of ZnO nanoparticles across different sizes and polymorphs. Instead of relying solely on computationally expensive (DFT calculations for each potential structure, they trained a ML model on the energies of small atomic fragments). This allowed them to predict the stability of large, complex nanoclusters with DFT-level accuracy at a fraction of the cost. The insights from this predictive approach are profound because they begin to unify the preceding models:
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0} and {11
0} and {11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0} are significantly more stable than the polar {0001} faces. This provides a clear energetic explanation for the rapid, anisotropic c-axis growth observed in all tetrapod synthesis. These calculations confirm that the arm elongation observed in experiments is the direct result of the kinetic suppression of diffusion to the stable side facets, validating the transport model proposed in Section 2.2.
0} are significantly more stable than the polar {0001} faces. This provides a clear energetic explanation for the rapid, anisotropic c-axis growth observed in all tetrapod synthesis. These calculations confirm that the arm elongation observed in experiments is the direct result of the kinetic suppression of diffusion to the stable side facets, validating the transport model proposed in Section 2.2.
This predictive link between atomistic surface energetics and macroscopic shape is further strengthened by the work of Gouveia and co-workers.32 As illustrated in Fig. 11, they combined DFT surface energy calculations with the Wulff construction to generate comprehensive morphology maps. These diagrams successfully reproduce experimentally observed ZnO shapes (from prisms to plates) by simulating how growth conditions (governed by the local Pe) alter the relative stability of the polar and non-polar surfaces.32
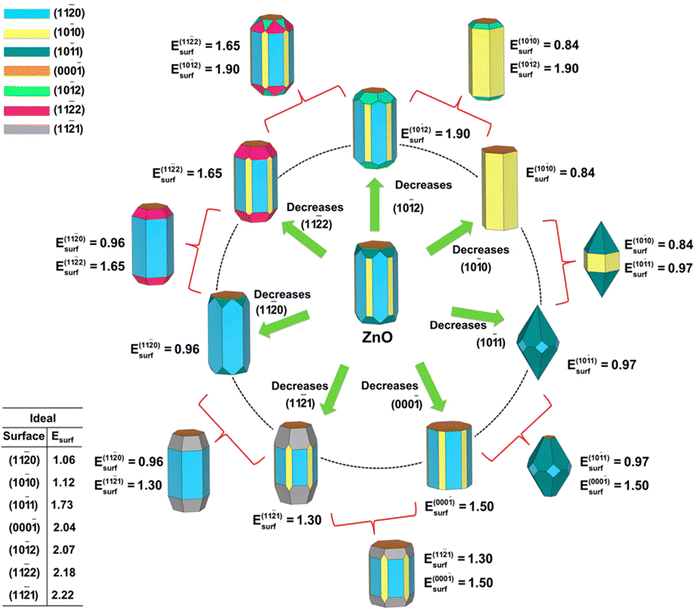 | ||
Fig. 11 Computational morphology map of wurtzite (WZ) ZnO generated via Wulff constructions. The central structure represents the equilibrium morphology based on standard surface energies. The surrounding pathways illustrate how the selective reduction of specific surface energies (e.g., stabilizing the non-polar {10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0} yellow facets vs. the polar {0001} orange facets) drives the morphological transition from compact crystals to elongated prisms or flat plates. Reproduced from ref. 32 with permision from MDPI,32 Copyright 2023. 0} yellow facets vs. the polar {0001} orange facets) drives the morphological transition from compact crystals to elongated prisms or flat plates. Reproduced from ref. 32 with permision from MDPI,32 Copyright 2023. | ||
Ultimately, this computational framework offers a potential pathway to settle the long-standing debate. By simulating the energy landscapes under varied chemical potentials, ML-enhanced models could predict which nucleation pathway. Specifically, they could determine if a ZB core, a twinned WZ embryo, or a multi-grain assembly dominates under specific synthesis conditions. This approach bridges the atomistic world of precursor chemistry with the nanoscale world of crystallography, representing the most promising route toward a unified, multi-scale understanding that could finally enable the rational, predictive design of T-ZnO morphologies.
2.5 Future frontiers: integrating experiment and theory
The synthesis of T-ZnO is a mature field, yet it remains governed by a fundamental paradox: the remarkable reproducibility of a complex 3D morphology that emerges from a poorly understood, atomistically-contested nucleation event. This review has traced the evolution of this challenge, from the debate over discrete crystallographic models to the practical trade-offs between various synthesis routes. The path forward, however, lies not in finding a single “winner” among the historical growth models. Instead, it requires adopting a unified, multi-scale framework. This framework must directly link controllable macroscopic process parameters to the underlying nanoscale physics of nucleation and growth.The foundation for this framework is now in place. The competition between kinetic and thermodynamic forces, quantified by the Pe and DaII, provides the essential physical language to connect reactor conditions to morphological outcomes. This transport-reaction triad clarifies that complex methods like FTS are not chaotic. Rather, they are governed by definable regimes where the second Damköhler number (DaII) dictates the nucleation locus and the Pe controls the arm extension. Concurrently, the rise of ML-enhanced computational methods provides the predictive engine capable of navigating these vast energy landscapes.
The critical challenge is no longer a lack of plausible theories, but rather the lack of a direct, data-rich feedback loop between synthesis, in situ characterization, and predictive simulation. Achieving true synthesis-by-design therefore requires a concerted, three-pronged research initiative. This strategy aims to bridge the gap between macroscopic processing and atomistic control through the following interconnected approaches:
2.5.2.1 Environmental TEM (ETEM) on model systems. We cannot put a turbulent flame inside a TEM, but we can replicate the chemical potential. By using an ETEM equipped with a specialized micro-electro-mechanical systems (MEMS) heating holder, researchers can replicate the chemical potential of the reactor. This setup allows them to generate the exact same zinc vapor and oxygen supersaturation conditions in a controlled, low-pressure environment. This allows for real-time observation of the crystal's birth, determining if the seed starts as a metastable zinc blende cube, a magic cluster, or a multi-grain assembly, effectively validating the crystallographic pathway without macroscopic turbulence.
2.5.2.2 In situ synchrotron X-ray scattering. To understand the dense, turbulent environments of real-world reactors where electrons cannot penetrate, high-energy synchrotron X-rays (30–100 keV) are required. These hard X-rays can pass through thick reactor walls and flames as if they were transparent. By placing portable flame reactors directly in the beamline, small-angle X-ray scattering (SAXS) can reveal real-time growth rates. Simultaneously, wide-angle X-ray scattering (WAXS) captures the exact millisecond of phase transition from molten Zn to crystalline WZ. This provides kinetic data that ETEM cannot.
Ultimately, solving the T-ZnO growth puzzle is a model problem for a grander challenge in materials science: the rational design of complex, non-equilibrium 3D nanostructures. The strategies outlined here unite dimensionless physical descriptors with ML-driven simulation and targeted in situ validation. They represent a transferable blueprint for moving beyond empirical trial-and-error, guiding the field toward the predictive synthesis of functional materials with unparalleled architectural control.
3 Fundamental properties and structure–property relationships
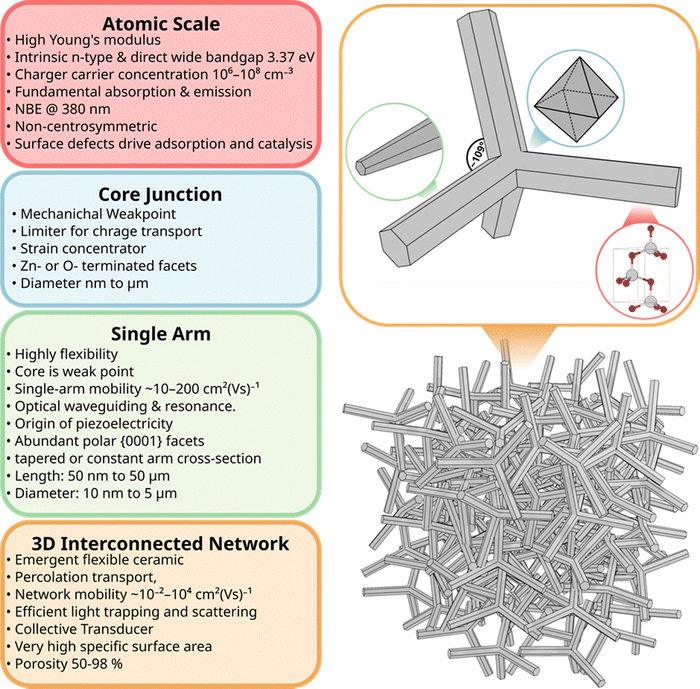
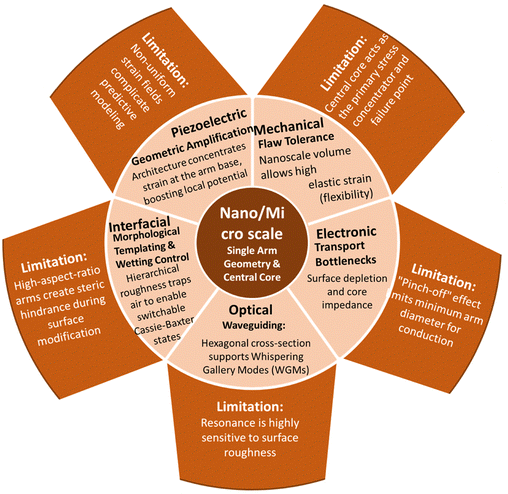
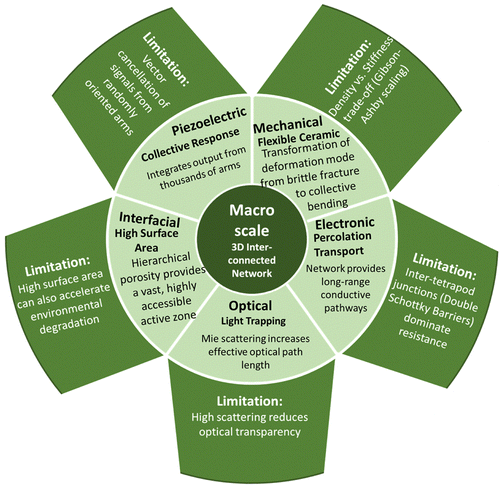
The scientific relevance of T-ZnO extends beyond its synthesis and arises from the way its distinctive three-dimensional morphology governs and couples a wide range of material properties. Rather than acting as a passive scaffold, the tetrapodal architecture strongly influences how optical, electronic, mechanical, and chemical properties are expressed and interact. Many of the functional characteristics of T-ZnO therefore emerge not from its chemistry alone, but from the combination of intrinsic ZnO properties with geometric confinement, anisotropy, and connectivity across multiple length scales. This section adopts a general, property-centered perspective and aims to clarify how the characteristic architecture of T-ZnO gives rise to coupled physical responses. These couplings are central to understanding why T-ZnO often exhibits behavior that differs markedly from that of simpler ZnO morphologies, such as particles, rods, or films. Understanding these built-in multi-physical interactions is essential for developing rational synthesis strategies. Furthermore, it facilitates the translation of T-ZnO from an empirical material system toward targeted, application-driven design.To provide a structured framework for these interdependencies, the properties of T-ZnO are discussed hierarchically across three length scales. At the atomic scale, the WZ crystal structure and the defect landscape define the intrinsic electronic, optical, and piezoelectric properties and their fundamental limits. At the microscale, the single tetrapod geometry introduces additional functionalities, including anisotropic mechanical response, stress concentration, and directional charge transport. At the macroscale, the assembly of tetrapods into interconnected networks leads to emergent properties governed by percolation, porosity, and collective deformation.
Following this hierarchy, the section begins with the crystal structure and defect chemistry of T-ZnO (Section 3.1) and proceeds to discuss its electronic (Section 3.2), optical (Section 3.3), mechanical (Section 3.4), piezoelectric (Section 3.5), and surface chemical properties (Section 3.6). These individual aspects are then brought together in Section 3.7 to highlight overarching structure–property relationships and inherent trade-offs. This integrated view provides the conceptual foundation for the application-oriented discussions in Section 4. To support this multi-scale perspective, Fig. 12 summarizes the key structural and functional domains of T-ZnO and illustrates how atomic-scale characteristics translate into emergent behavior at the network level.
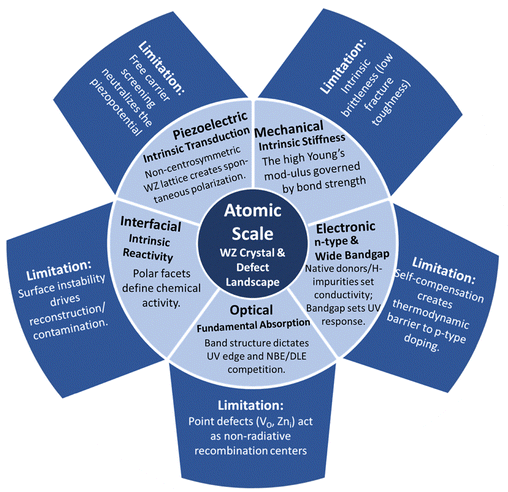
3.1 Crystal structure and defect engineering
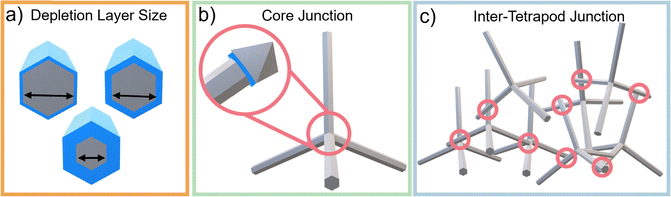
The functionality of T-ZnO emerges from the interplay between its macroscopic 3D architecture and atomic-scale imperfections. This section deconstructs the anisotropic WZ arm structure (Section 3.1.1), the structural ambiguity of the central junction (Section 3.1.2), and the resulting intrinsic defect landscape (Section 3.1.3). We then frame defect manipulation as an exercise in applied thermodynamics (Section 3.1.4) and assess the characterization suite required to resolve these features (Section 3.1.5).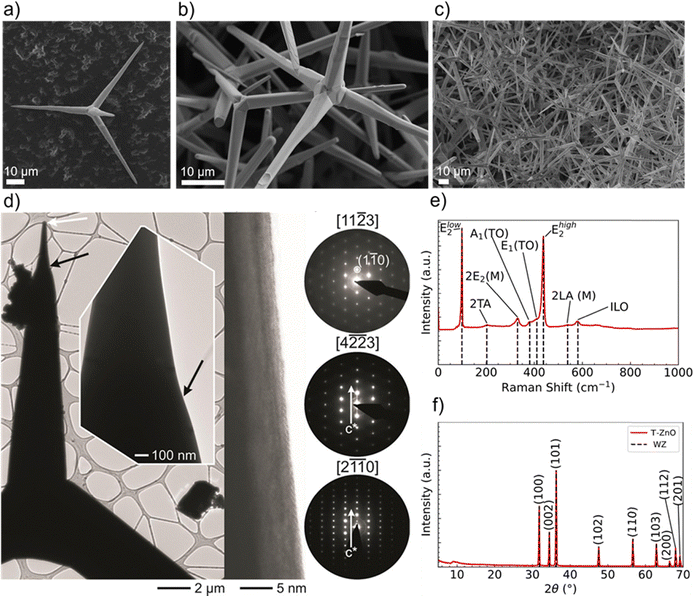 | ||
| Fig. 13 SEM images of T-ZnO showing its tetrapodal morphology, with (a) a single tetrapod, (b) and (c) in a network at different magnifications. (d) HRTEM and SAED analysis confirming the single-crystalline nature of a T-ZnO arm. A bright-field TEM image, a high-resolution micrograph taken from the arm, revealing the atomic lattice. and a SAED tilting series, which confirms the arm is a single crystal that grew along the c-axis. (e) Raman spectrum of T-ZnO and (f) XRD spectrum of T-ZnO synthesized via the FTS method. (d) Reproduced from ref. 21 with permision from American Chemical Society,21 Copyright 2015. | ||
The WZ is inherently non-centrosymmetric due to the tetrahedral coordination of Zn2+ and O2− ions.93,94 This lack of inversion symmetry leads to two primary functional consequences: a net spontaneous polarization along the c-direction,88,95,96 and critically, strong piezoelectric properties.94,95 This intrinsic electromechanical coupling provides the physical foundation for T-ZnO applications in strain sensing and energy harvesting (Section 3.4). While the arms typically maintain high crystalline perfection,12,13,29,37,38,86,88,89 they may host planar defects like stacking faults and are often terminated by reactive polar surfaces that act as sinks or sources for point defects.21,86,88 This relative structural integrity of the arms contrasts sharply with the complex, defect-rich central junction discussed in the following section.
Defining the T-ZnO core structure is critical, as divergent atomic arrangements produce distinct functional signatures. A persistent ZB core, for instance, would create ZB/WZ polymorphic homojunctions at the arm interfaces. The resulting band offsets likely introduce significant potential energy barriers that act as scattering centers, severely hindering inter-arm charge transport.12,26,95,97 Conversely, a highly strained, twinned WZ core would serve as a preferential sink for defect accumulation.21,47,85,97 Local strain fields around twin boundaries can lower the defect formation energy (Ef), driving defects toward the core region.100 This defect-rich environment influences optical and electronic performance by promoting non-radiative recombination and introducing localized electronic states at internal interfaces97 (Fig. 14). Furthermore, these defect-laden boundaries function as both mechanical stress concentrators for crack initiation and electronic resistive bottlenecks, ultimately defining the material's failure points and conductivity limits under load.101
 | ||
| Fig. 14 Correlative analysis of the structural and optical properties of a single T-ZnO particle. (a) High-resolution scanning transmission electron microscopy (STEM) image. (b) Corresponding panchromatic cathodoluminescence (CL) map, showing highly luminescent arms contrasting with a non-luminescent (dark) central junction. (c) Overall CL spectrum from the tetrapod, displaying the dominant UV near-band-edge (NBE) peak (∼3.20 eV).97 (a)–(c) Reproduced from ref. 97 with permision from IOP Science,97 Copyright 2021. | ||
Beyond electronic and optical effects, the core structure also plays a key role in the mechanical response of T-ZnO. Defect-decorated twin boundaries can serve as stress concentrators and preferential sites for crack initiation under load. Simultaneously, they serve as a resistive bottleneck for charge transport. This dual role inextricably links mechanical deformation to electrical performance. Together, these considerations highlight that the unresolved core structure is a central element in understanding the coupled mechanical, electronic, and optical properties of T-ZnO.12,21,29,38,85
3.1.3.1 Primary intrinsic point defects. At the heart of T-ZnO's defect landscape are the primary point defects: vacancies, interstitials, and antisites. These defects and their roles will be discussed in the following sections and Table 4 provides a summary of the primary intrinsic point defects of T-ZnO.
| Defect | Primary functional role | Electronic nature | Favored conditions | Key experimental signature(s) |
|---|---|---|---|---|
| Oxygen vacancy (VO) | Charge compensation center; radiative & non-radiative recombination center. | Deep donor105 | O-deficient (reducing) | Photoluminescence (PL) (green emission, debated); electron paramagnetic resonance (EPR) (for paramagnetic V+O); positron annihilation spectroscopy (PAS) |
| Zinc vacancy (VZn) | Primary compensating acceptor in n-type ZnO. | Deep acceptor88 | O-rich (oxidizing) | PAS |
| Zinc interstitial (Zni) | Potential source of temporary n-type conductivity; charge compensation center. | Shallow donor (high energy needed to form, limiting its amount) | Zn-rich | PL (violet/blue emission); difficult to detect directly |
| Oxygen interstitial (Oi) | Compensating deep acceptor. | Deep acceptor88 | O-rich | PL (yellow/orange/red emission); Difficult to detect directly |
| Oxygen antisite (OZn) | Likely insignificant due to very high energy needed to form. | Deep acceptor | Extreme lack of zinc or oxygen | No clear signature |
| Zinc antisite (ZnO) | Donor |
Oxygen vacancy (VO). An oxygen atom missing from its lattice site, is one of the most studied defects in ZnO.36,92,100,102 While historically and empirically linked to n-type conductivity,12,13 foundational DFT studies suggest it is a deep donor, making it an unlikely source for the high background carrier concentrations (Fig. 15).105,106 Instead, it likely functions as a charge compensation center and a primary contributor to visible PL, specifically the green emission “g” band (Fig. 15b), though surface states remain a debated alternative origin.87,97,107 VO exists in multiple charge states (V0O, V+O, V2+O) contingent on the Fermi level.29 Its paramagnetic state (V+O) is detectable via electron paramagnetic resonance (EPR) but is thermodynamically unstable (Fig. 15c).47,85,105
 | ||
| Fig. 15 First-principles calculations of intrinsic defect formation energies (ΔEF) as a function of the Fermi level (EF), with the valence band maximum set to 0.0 eV. (a) Comparative plot of the primary donor defects, the oxygen vacancy (VO) and the zinc interstitial (Zni). (b) Representative PL spectrum of ZnO highlighting visible emission bands (green “g” and yellow “y”) linked to intrinsic defects. (c) Detailed formation energies of the VO charge states, illustrating its “negative-U” property where the paramagnetic V+O state is thermodynamically unstable. (d) Schematic energy band diagram of the ZnO intrinsic defect landscape, mapping the deep and shallow energy levels and their proposed radiative transitions for UV and visible emissions. (a) Reproduced from ref. 106 with permision from American Chemical Society,106 Copyright 2014, (b) and (c) reproduced from ref. 105 with permision from Elsevier,105 Copyright 2001, (d) reproduced from ref. 29 with permision from Royal Chemical Society,29 Copyright 2024. | ||
Zinc vacancy (VZn). A zinc atom absent from its lattice site,36,85,87 the VZn is an acceptor-type defect,88,103 which is more likely to form under oxygen-rich conditions. It acts to compensate n-type conductivity and is also linked to visible PL. While often associated with the green luminescence band, sometimes as part of a defect complex, theoretical and experimental studies have also linked it to emissions in the yellow or orange regions.88
Zinc interstitial (Zni). This occurs when a zinc atom occupies on an interstitial site in the crystal lattice. Zni is considered a mobile shallow donor (can donate an electron).92,106 However, its high Ef limits its concentration under equilibrium, making it an improbable source for stable n-type conductivity.105 Since neither VO nor Zni are thermodynamically favorable as stable shallow donors, foundational DFT suggests that persistent n-type conductivity originates from extrinsic sources, most notably unintentional hydrogen (H) impurities.105 Experimentally, Zni is often associated with violet/blue PL emissions (e.g., ∼420–450 nm).36
Oxygen interstitial (Oi). This is when an oxygen atom is in a space where it doesn't belong, often sharing a spot with another oxygen atom (split-interstitial configuration),103 the Oi is a deep acceptor favored under O-rich conditions.88,103 Although having a high Ef, it has been identified as a potentially dominant defect in certain electrochemical environments103 and is linked to yellow, orange, or red PL bands (e.g., a weak red emission around 705 nm).87,108
Antisite defects (OZn, ZnO). These are defects where one type of atom is on the site of the other (e.g., an oxygen atom on a zinc site). They are generally considered to have very high formation energies, Ef,26 consequently, they are not expected to be present in significant concentrations under most conditions, and their direct contribution to properties is considered minor.96
A schematic of the energy levels for these primary defects and their corresponding radiative transitions is provided in Fig. 15d.
3.1.3.2 Defect complexes: the importance of interactions. Treating defects as isolated entities is an oversimplification; attractive electrostatic and strain-field interactions in the lattice often lead to the formation of defect complexes with electronic and optical signatures distinct from their constituents. Examples include donor–acceptor pairs (Zni and VZn) or vacancy pairs (VZn and VO). Although potentially electrically neutral, these complexes function as significant scattering centers that impede carrier mobility and provide unique radiative or non-radiative pathways, such as the ∼400 nm PL band, often attributed to Zni/VZn pairs.29
3.1.3.3 Spatial heterogeneity: a non-uniform defect landscape. Evidence indicates a spatially heterogeneous defect landscape where defects preferentially accumulate in specific regions.37,47,102,109 The structurally disordered central core acts as a sink for point defects to relieve lattice strain, creating a high-density defect zone that hinders charge transport and initiates mechanical failure. Additionally, defects segregate toward the surfaces and arm tips. Energy dispersive X-ray (EDX) analysis confirms this non-uniformity, showing VO enrichment at the arm tips and zinc deficiency near the bases (Fig. 16).47 This thermodynamically driven segregation dictates that the most catalytically and sensorially active sites reside at the arm extremities, a critical factor for device design.
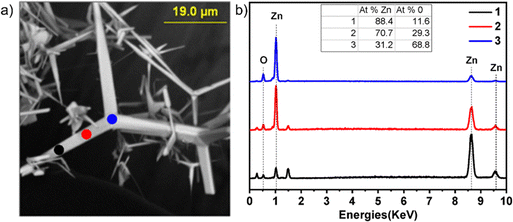 | ||
| Fig. 16 Spatial defect heterogeneity in T-ZnO. (a) SEM image of a tetrapod indicating the points of EDX analysis: the arm tip (1, black), arm middle (2, red), and base/core (3, blue). (b) Corresponding EDX spectra and inset data table. The elemental quantification highlights an oxygen deficiency at the arm tip (point 1, 11.6% O) and a zinc deficiency near the base (point 3, 31.2% Zn). (a) and (b) Reproduced from ref. 47 with permision from MDPI,47 Copyright 2023. | ||
This more detailed picture of an interacting and unevenly distributed “defect landscape” (rather than a simple list of defects) is essential for moving toward a predictive understanding of T-ZnO's properties.
3.1.4.1 Foundational principles: defect formation energy. Defect concentration in a crystal scales exponentially with the Gibbs free energy of formation, which is dominated at a given temperature by the formation enthalpy, or formation energy (Ef).106 For a defect D in charge state q, its Ef is not a fixed constant but a variable dependent on the atomic and electronic reservoirs in equilibrium with the host lattice:103,105
 | (1) |
| μZn + μO = ΔHf(ZnO) | (2) |
3.1.4.2 Synthesis-level control of defect chemistry. The most fundamental strategy for defect engineering involves governing native defect chemistry during initial formation by modulating the thermodynamic parameters of the synthesis environment (Section 2).
Vapor-phase methods. In high-temperature routes such as CVD and thermal evaporation, gas-phase partial pressures directly establish the reactor's chemical potentials (μZn and μO). Zn-rich conditions, achieved with a high Zn/O precursor ratio, establish a high μZn and correspondingly low μO, thereby lowering Ef(VO) and Ef(Zni) and promoting the formation of n-type conductivity.96 Conversely, O-rich conditions lower μZn and increase the Ef of these donor defects, while making acceptor-like defects such as the VZn more stable.103 The FTS presents a more complex scenario: rapid, localized combustion generates dynamic, non-equilibrium temperature and chemical potential gradients that uniquely shape the defect landscape.21,37 Such synthesis-level control can yield counter-intuitive results; for example, adding an oxidizing agent (H2O2) during vapor-phase synthesis has been shown to paradoxically increase the final oxygen vacancy concentration.38
Solution-phase (wet) methods. In low-temperature solution-based synthesis, the chemical potentials are not set by gas pressures but by the concentrations of dissolved precursors, pH, and the stability of complex ions (e.g., [Zn(NH3)4]2+).78 The controlled decomposition of these complexes ensures a steady-state flux of growth species, creating a chemical potential regime distinct from vapor-phase equilibrium.
It is crucial to note that while basic DFT studies have established these principle,105 more recent work highlights the critical role of temperature.37 High-temperature phonon contributions can significantly alter the Gibbs free energy of formation, particularly for VO, thereby shifting the thermodynamic equilibrium and relative defect concentrations predicted by calculations at absolute zero (0 K) alone.106
3.1.4.3 Post-synthesis treatments: thermodynamic re-equilibration and surface modification. Post-synthesis treatments allow for the precise modification of T-ZnO defect populations through bulk thermodynamic re-equilibration or direct surface engineering.47,110–113
Atmosphere-controlled annealing. Thermal annealing is a primary method for re-equilibrating bulk defect populations under controlled thermodynamic conditions.106 By exposing the material to specific atmospheres and temperatures, the external chemical potentials drive point defect diffusion and reaction toward a new equilibrium.103 For example, annealing in O2 or air raises the μO, which in turn increases the Ef of VO and can facilitate their annihilation.103,105 This is experimentally observed as a suppression of the defect-related green PL.88 Li and Seebauer104 validated this principle by demonstrating that the surface potential, a direct proxy for the near-surface defect population, could be precisely and reversibly tuned simply by controlling temperature and the external oxygen partial pressure. Their work confirms that the macroscopic environment dictates the microscopic defect thermodynamics.103,106
However, this equilibrium is governed by the competition between chemical enthalpy and thermal entropy. While moderate temperatures favor oxidation, annealing at extremes (e.g., >1000C) can paradoxically increase oxygen vacancy concentrations, even in oxidizing atmospheres.96,114 At these temperatures, the entropic driving force (ΔS) for lattice disorder overrides the chemical potential of the atmosphere; upon rapid cooling, these high-temperature vacancy populations are “frozen in,” resulting in the oxygen-deficient surfaces characteristic of high-temperature processing.96,114
Energetic surface modification. Beyond thermal routes, post-synthesis UV irradiation provides a non-contact method to engineer surface defects. UV exposure can trigger a reversible switch from hydrophobic to hydrophilic wettability, which ESR and XPS correlate directly with an increased surface concentration of oxygen vacancies.47
Surface passivation and heterostructures. Surface engineering focuses on passivating active sites or constructing heterostructures to define functional properties. Surface passivation neutralizes electronic states that act as non-radiative recombination centers, often via conformal organic or inorganic coatings.107 For instance, (3-aminopropyl)triethoxysilane (APTES) has been utilized to passivate surface traps, confirmed by shifts in defect-related PL (Fig. 17) and carrier dynamics evaluated through time-resolved PL (TRPL).89,101,107
 | ||
| Fig. 17 Optical signatures of surface defect passivation. (a) PL spectra of bare ZnO (dark green) and APTES-passivated ZnO (light green), highlighting the relative intensities of the near-band-edge (NBE, ∼378 nm) and defect-related deep-level emission (DLE, ∼510 nm) bands. (b) Photoluminescence excitation (PLE) spectra showing a tail of states (arrow) associated with the surface defect landscape before and after functionalization. (a) and (b) Reproduced from ref. 107 with permision from American Chemical Society,107 Copyright 2022. | ||
Alternatively, the T-ZnO network can serve as a high-surface-area scaffold for core–shell heterostructure growth.85,87,88,97,107,115 Sequential dipping procedures have been used to grow complex shells, such as ZnSe/CdSe/ZnSe, onto T-ZnO templates.89 This enables the engineering of tailored band alignments to optimize charge separation for photovoltaics or to achieve wavelength-specific optical emissions by creating entirely new electronic interfaces.
3.1.4.4 Extrinsic doping. Extrinsic doping overrides native defect chemistry to precisely manipulate the thermodynamic landscape of T-ZnO by shifting the Fermi level (EF) and modifying the local atomic environment.91,100,109
Fermi level and carrier control. Strategic shifting of EF allows for precise conductivity control, though outcomes differ significantly between n-type and p-type regimes.91
• N-type doping: reliable n-type conductivity is achieved using group III elements (Ga, Al, In) as substitutional donors for Zn. These elements substitute for Zn and act as shallow donors, increasing the electron concentration and making the material suitable for applications like transparent conductive oxides (TCOs).87,88
• P-type doping: in contrast, achieving stable and reproducible p-type ZnO remains a major challenge.88,116 Nitrogen is the most explored p-type dopant, aiming to substitute for oxygen and act as a shallow acceptor, pushing EF towards the valence band.13,90,105 However, as the EF is reduced, the Ef of native donor defects like the VO, decreases dramatically. This triggers the spontaneous formation of donors that counteract the intended acceptor levels, pinning EF and hindering p-type stability.105 Efforts to overcome this hurdle include nitrogen doping via urea solid-state reactions109 or ammonia-flow nitridation,90 alongside co-doping strategies with Fe, Cu, or Al to mitigate compensation effects.87
Modification of host energetics and reactivity. Beyond electronic control, dopants alter the host lattice's chemical reactivity by modifying local strain and bonding environments. Pala and Metiu100 systematically demonstrated that substituting surface Zn with lower-valency cations (e.g., Li+, Ag+) induces an “electron deficit,” significantly reducing the Oxygen Vacancy Formation Energy (OVFE). This reduction increases lattice oxygen reactivity, enhancing oxidative catalytic power. Conversely, dopants that form stronger oxygen bonds (e.g., Al3+, Ti4+) elevate the OVFE, rendering the oxide more stable and less reactive. This provides a predictive link between dopant chemistry and the thermodynamic stability of native surface defects.
These multifaceted strategies are consolidated in Table 5, which serves as a “control panel” linking functional goals to thermodynamic principles and experimental levers.
| Desired functional goal | Thermodynamic lever | Experimental action | Verification method |
|---|---|---|---|
| Increase n-type conductivity | Decrease formation energy (Ef) of native donors (VO, Zni) | Synthesis: use Zn-rich precursors. | Electrical: four-point probe. |
| Post-synthesis: anneal in reducing atmosphere (e.g., H2/Ar). | Spectroscopic: PL shows an increased NBE/DLE ratio. | ||
| Suppress defect-related PL (green luminescence) | Increase formation energy (Ef) of VO | Post-synthesis: anneal in oxidizing atmosphere (e.g., O2/air) at moderate temperatures (<800 °C). | Optical: PL spectroscopy shows a suppressed green emission peak. |
| Achieve p-type conductivity | Decrease Ef of acceptors (e.g., NO (N on O site)) while managing self-compensation | Doping: introduce nitrogen dopants (e.g., via NH3 anneal) potentially with co-dopants. | Electrical: hall effect measurement showing positive Hall coefficient. |
| Enhance oxidative catalytic activity | Decrease oxygen vacancy formation energy (OVFE) at the surface | Doping: introduce lower-valency dopants (e.g., Li+, Ag+) to create an “electron deficit.” | Chemical: catalytic performance testing (e.g., CO oxidation). |
| Surface: XPS analysis. | |||
| Passivate surface recombination centers | Neutralize electronically active surface states | Post-synthesis: apply a conformal surface coating (e.g., APTES, Al2O3). | Optical: time-resolved PL shows an increased carrier lifetime. |
| Technique | Scale of information | Primary application | Key limitations & things to note |
|---|---|---|---|
| Scanning electron microscopy (SEM) | Micro- to macro-scale | Visualizing overall morphology, network connectivity, and porosity. | Provides no information on crystal structure or atomic-scale defects. |
| Transmission electron microscopy (TEM)/high-resolution TEM (HRTEM)/selected area electron diffraction (SAED) | Nano- to atomic-scale | Confirming that arms are single crystals (SAED); Seeing the atomic lattice and planar defects (HRTEM). | Local probe: extremely small sampling volume; requires destructive preparation; difficult to obtain statistically relevant data. |
| X-ray diffraction (XRD) | Bulk/large area | Confirming the primary crystal phase (wurtzite) and phase purity; measuring average lattice strain. | Averaging: provides no information on local heterogeneity (e.g., core vs. arm) or point defects. |
| Raman spectroscopy | Bulk/large area | Evaluating overall crystal quality and lattice vibrational modes; detecting local strain and disorder. | Interpretation of defect-induced modes can be complex; often requires low temperature for clear resolution. |
| Photoluminescence (PL) spectroscopy | Bulk/large area | Evaluating overall optical quality (near-band-edge/deep-level emission (NBE/DLE) ratio); investigating the presence of deep-level electronic states. | Highly debated interpretation: the origin of the visible deep-level emission (e.g., green luminescence) is controversial. It is often attributed to VO, but contributions from VZn and surface states are widely argued in literature. Cannot identify specific defects in isolation. |
| Electron paramagnetic resonance (EPR) spectroscopy | Bulk/large area | Clearly identifying specific paramagnetic defects, such as the singly-ionized oxygen vacancy (V+O). | Selective: only sensitive to defects with unpaired spins; “silent” to neutral or fully ionized defects V2+O |
| X-ray photoelectron spectroscopy (XPS) | Surface (∼2–10 nm) | Quantifying surface elemental composition and chemical states (e.g., distinguishing lattice O from hydroxyls). | Surface only: provides no information about the bulk crystal; requires ultra-high vacuum (UHV). |
| Conductive atomic force microscopy (c-AFM) | Nanoscale (surface) | Mapping local conductivity across surfaces and junctions; quantifying the electronic barrier height of individual T-ZnO-to-T-ZnO contacts. | Mainly a surface probe; does not provide simultaneous atomic-level structural information of the underlying junction. |
| In situ TEM with nano-probes | Nanoscale (single junction) | Directly correlating the atomic structure of a specific junction (e.g., degree of sintering) with its measured current–voltage (I–V) characteristics. | Technically demanding: high risk of beam damage or contact resistance artifacts; represents the most advanced single-junction characterization. |
The contentious interpretation of PL spectroscopy illustrates this challenge. Although PL is a standard defect probe, attributing specific signals, particularly the ubiquitous green luminescence, exclusively to VO remains highly debated.87,88,107 Conflicting data exist, with some models linking VO to red emission bands,26 while others implicate surface states or adsorbed species.36,104,107 Furthermore, photoluminescence excitation (PLE) spectroscopy reveals a complex near the band edge, providing electronic signatures of surface functionalization that PL alone cannot resolve.107 Consequently, bulk PL measurements remain insufficient for definitive defect identification.47,87,88 To achieve predictive control, the field must advance through two primary strategies:
(1) Correlative microscopy: future defect identification relies on multi-modal analysis performed on individual nanostructures.21,38,47,85,86,97,107 Applying HRTEM for atomic structure,38,86 Electron energy loss spectroscopy (EELS) for local chemistry, and CL for emission mapping,47,97 to the exact same tetrapod eliminates the ambiguity of ensemble averaging. This suite must incorporate spatially resolved spectroscopy; for example, EPR can distinguish V+O populations at the T-ZnO core (g = 1.962) v from those at the surface (g = 1.998).85 Additionally, ultraviolet photoemission spectroscopy (UPS) is essential for measuring Fermi levels and constructing precise band alignments in core–shell heterostructures.89 This approach, combining high-resolution imaging like HRTEM and CL38,47,86,97 with advanced electronic and chemical-state mapping (EELS, UPS, EPR), would create a clear connection between a directly observed atomic-scale feature (e.g., a specific twin boundary or defect cluster) and its measured optical and electronic properties, eliminating the uncertainty found in measurements that average over a large sample.
(2) In situ and operando characterization: moving beyond static snapshots to real-time observation is critical for understanding T-ZnO dynamics. ETEM offers the potential to visualize nucleation events during synthesis and track defect migration under controlled thermal or atmospheric loads.12,104 Furthermore, operando measurements allow for monitoring structural changes within operating devices. These techniques are essential to resolve long-standing structural questions, such as the precise atomic arrangement of the central core, and to provide the experimental evidence required for the rational design of T-ZnO properties.
3.2 Electronic and transport properties
The intrinsic semiconducting nature of the WZ unit cell forms the foundation of T-ZnO's electronic behavior, yet the overall conductivity is shaped by a hierarchy of multi-scale effects. At the atomic level, point defects such as vacancies and interstitials introduce donor and trap states that modulate carrier concentration and mobility. At the nanoscale, surface defects and arm-tip states influence local charge distribution and depletion layers, affecting transport along individual arms. The central core, typically rich in defects, can act as a resistive bottleneck, limiting inter-arm conduction. Finally, at the macroscale, the connectivity and percolation of tetrapods within the network govern the global transport properties, as even well-conducting arms are constrained by junctions and network topology. Understanding this multi-level interplay is key to interpreting T-ZnO's electronic behavior and guiding the rational design of functional devices.1. The wide bandgap: at room temperature, ZnO's bandgap of approximately 3.37 eV52 (which increases to ∼3.44 eV at low temperatures96). This wide gap ensures high optical transparency and renders the material ideal for UV-regime applications, including photodetectors and light-emitting diodes (LEDs).29,48,86,88,90,92,111,117–119 However, the wide bandgap is also the primary barrier to stable p-type doping; it favors the formation of deep rather than shallow acceptor levels and creates a thermodynamic drive for the spontaneous formation of compensating native donor defects (Section 3.2.2).
2. The exciton binding energy trade-off: ZnO possesses an exceptionally large exciton binding energy of 60 meV,13,29,88,92,111,118,120–122 significantly exceeding the room-temperature thermal energy (∼26 meV). While this stability facilitates efficient radiative recombination for light-emitting devices (Section 3.3.2), it complicates light-harvesting and detection mechanisms. Photogenerated electron–hole pairs tend to remain as bound, neutral excitons rather than free carriers. To generate a measurable photocurrent (Fig. 18), these excitons must be dissociated, a process requiring high internal or external electric fields to overcome the associated energy barrier.37 Consequently, this requirement can limit the conversion efficiency of ZnO-based photodetectors and photovoltaic cells.
 | ||
| Fig. 18 Dynamic photocurrent response to UV light for T-ZnO networks synthesized at (a) 920 °C and (b) 1000 °C. The 920 °C sample exhibits a slow response with persistent photocurrent, whereas the 1000 °C sample demonstrates a fast, reversible on/off response. (a) and (b) Reproduced from ref. 37 with permision from American Chemical Society,37 Copyright 2025. | ||
The modern consensus, strongly supported by this computational work. It points to the unintentional incorporation of hydrogen impurities as the primary source of ZnO's intrinsic n-type behavior.103,105 Ubiquitous in growth environments, hydrogen acts as a robust shallow donor in ZnO, occupying interstitial or substitutional sites (Fig. 19). Despite this shift, native defects remain critical as charge-compensating entities.85 The thermodynamic framework established in Section 3.1.4 shows that their formation energies are strongly dependent on the EF. Any attempt to make the material p-type (e.g., by nitrogen doping) lowers the EF, which in turn dramatically lowers the Ef of these native donors.103 This triggers the spontaneous formation of donors that neutralize introduced acceptors, a self-compensation mechanism that remains the fundamental barrier to stable p-type ZnO.88,96,116
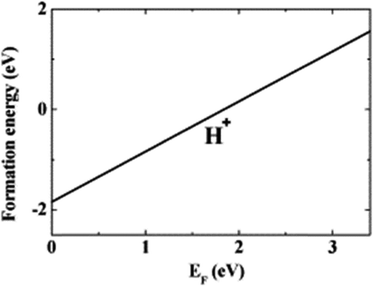 | ||
| Fig. 19 Calculated formation energy (Ef) of hydrogen (H) in ZnO as a function of the Fermi level (EF), with the valence band maximum (EV) set to 0.0 eV and the conduction band minimum (EC) at ∼3.37 eV. The plot demonstrates that hydrogen exists exclusively in the positive charge state (H+), as the neutral (H0) and negative (H−) charge states remain thermodynamically unstable across the entire bandgap. Reproduced from ref. 105 with permision from Elsevier,105 Copyright 2001. | ||
Ultimately, the carrier concentration in T-ZnO is a multifaceted function of its synthesis and processing history. Concentrations vary with intrinsic defect density, notably low in FTS T- products,102 intentional doping, and post-treatment conditions.87 Furthermore, secondary factors such as alloying,87 which can widen the bandgap,91 or the presence of extended defects like dislocations that can act as electron traps and scattering centers, thereby reducing carrier mobility, can also influence the overall electronic behavior.47,125
3.2.3.1 Bottlenecks of the electronic pathway.
The ideal single-crystalline arm. At the most fundamental level, each arm of a T-ZnO tetrapod is a single-crystalline WZ structure. In an ideal, bulk crystal of this quality, electron mobility would be high, naturally limited only by phonon scattering at room temperature.125,127,128 This highly crystalline arm represents the intrinsic limit of conductivity, a low-resistance pathway relative to the network architecture. In practice, however, this ideal pathway is immediately compromised by a series of bottlenecks, which effectively degrade this performance long before the macroscopic scale is reached (Fig. 20).
Bottleneck I (nanoscale): surface scattering and pinch-off. The first major degradation to this ideal transport occurs at the nanoscale. In arms with small diameters (<100 nm), the high surface-to-volume ratio means that surface scattering from charged defect states88,107 and physical surface roughness becomes a dominant limiting factor, significantly reducing the effective mobility compared to bulk ZnO.104,107 This effect is physically governed by the Debye length (LD), which defines the width of the surface depletion region caused by trapped charge at surface states.52,86,104 This principle leads to a critical design constraint known as the “pinch-off” effect (Fig. 20a): as the arm diameter is reduced to twice the LD (d ∼2LD), these depletion regions can merge and deplete the entire arm of free carriers, making it highly resistive.37
Bottleneck II (single tetrapod): the central core junction. The second bottleneck impedes transport within a single tetrapod. As detailed in Section 3.1.2, the structurally complex and often defect-rich central core, which contains a high density of defects and misfit stresses from its complex growth, acts as a potent scattering center (Fig. 20b). Direct impedance spectroscopy by Huh et al.129 further demonstrate that this junction region contributes disproportionately to the overall electrical resistance of a ZnO tetrapod. Although its volume fraction is small, the junction exhibits a higher local resistance than the WZ arms, thereby dominating the total resistance pathway, shown by Fig. 21. This junction introduces significant potential energy barriers52 that hinder the efficient flow of charge from one arm to another, directly impacting the measured I-V characteristics of individual tetrapods and manifesting in techniques like intensity modulated photocurrent spectroscopy (IMPS).89
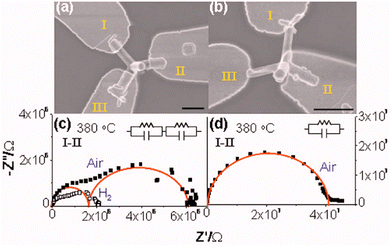 | ||
| Fig. 21 The SEM images of (a) two T-ZnO arms measured in series with the T-ZnO core and (b) only one contacted T-ZnO arm. The impedance spectra measured from (c) of the setup shown in (a) and (d) of setup shown in (b), respectively, at 380 °C under air (solid squares). Also included in (c) is the impedance spectrum measured under hydrogen at 380 °C (open squares). The scale bars shown in (a) and (b) indicate 1 micrometer. (a)–(d) Reproduced from ref. 129 with permision from AIP Publishing,129 Copyright 2008. | ||
Bottleneck III (the network): the dominant inter-tetrapod junction. While nanoscale effects and the core structure influence transport, experimental evidence indicates that the dominant bottleneck in any macroscopic T-ZnO assembly arises at the T-ZnO-to-T-ZnO junctions (Fig. 20c).87,121 Conduction across the 3D network relies on these inter-tetrapod contacts, which provide multiple interconnected pathways.13,88,95,121,124 This has shown to improve carrier collection in devices such as solar cells and sensors when compared to nanoparticle films.13 However, for high-mobility electronics, transport is ultimately limited by the high resistance at these junctions.87,121
This bottleneck can be understood by modeling the junction as a nanoscale grain boundary.96 irst, the cross-sectional area of the junction is typically much smaller than that of a single tetrapod arm, limiting the available conduction pathway. Second, electronic defect states at the interface trap charges, creating a plane of fixed negative charge that further hinders electron flow. This charge repels mobile electrons in the adjacent arms, resulting in two opposing depletion zones at the junction. The resulting potential profile is commonly described as a double Schottky barrier (DSB).130 This explains the experimentally observed low effective mobility of T-ZnO networks, which remains far below the mobility of a single, defect-free arm.
3.2.3.2 Junction engineering: targeting the dominant bottleneck. The hierarchical analysis of transport dynamics dictates that while all bottlenecks influence performance, enhancing macroscopic conductivity necessitates targeting the dominant inter-tetrapod junction. This “junction engineering” via post-synthesis modification is established as a critical strategy for optimizing network-level transport properties.87 Mitigation of the junction barrier is primarily pursued through two complementary paradigms:
1. Geometric modification via sintering: thermal annealing at high temperatures promotes stronger interconnections between tetrapods. This geometric solution increases the physical cross-sectional area for current flow, thereby directly reducing the junction resistance.
2. Electronic modification via passivation: surface passivation offers a chemical/electronic solution. By coating the T-ZnO network with a suitable passivating layer, it is possible to chemically neutralize the electronic trap states at the grain boundary facilitating more efficient charge transport.
The fundamental divergence in transport physics between an isolated, single-crystalline arm and the macroscopic assembly is central to T-ZnO electronics. These scale-dependent distinctions, which define the hierarchy of transport bottlenecks, are summarized in Fig. 22. Resolving the junction-limited nature of these networks remains the prerequisite for the full realization of T-ZnO-based electronic and optoelectronic architectures.
3.2.4.1 Alloying: tuning the bandgap at a thermodynamic cost. Alloying ZnO with isovalent elements provides a direct method for bandgap tuning. Introducing magnesium (forming MgxZn1−xO) widens the bandgap towards the deep-UV, while alloying with cadmium (forming CdxZn1−xO) narrows it towards the visible region.96 This allows for precise tailoring of the material's spectral response for applications like wavelength-selective photodetectors or solar cells. However, this engineering comes at a thermodynamic cost. Alloying alters the host lattice parameters and chemical bond energies, which in turn modifies the Ef of native point defects. For instance, theoretical studies have shown that in MgZnO alloys, the Ef of key compensating donor defects, like the VO, can be even lower than in pure ZnO under certain conditions.103 This exacerbates the material's intrinsic tendency to self-compensate, making the already difficult challenge of achieving p-type conductivity even more challenging in these wide-bandgap alloys.
3.2.4.2 Doping: a thermodynamic competition. Doping with aliovalent elements is primarily used to control carrier concentration by shifting the Ef,105 which leads to the following two scenarios:
• N-type doping: this is a thermodynamically straightforward process. Doping with group III elements like gallium (Ga) or aluminum (Al) introduces shallow donor states that readily ionize, reliably increasing the n-type conductivity for applications like TCOs.88
• P-type doping: in contrast, p-type doping is a thermodynamic competition. The goal of introducing acceptors, such as nitrogen substituting for oxygen, is to lower the EF towards the valence band.96 However, as EF moves lower, the Ef of native donor defects (like VO) decreases dramatically, as determined by the qEF term in the formation energy equation (eqn (1)). The material responds to the introduction of acceptors by spontaneously forming these low-energy donor defects to counteract them. This powerful self-compensation mechanism, predicted by first-principles thermodynamics,105 is the fundamental reason why achieving stable p-type ZnO remains a major challenge (Fig. 23).88,90,96,131 This illustrates that doping is not simply a matter of adding impurities but a battle against the intrinsic thermodynamic tendencies of the host crystal.
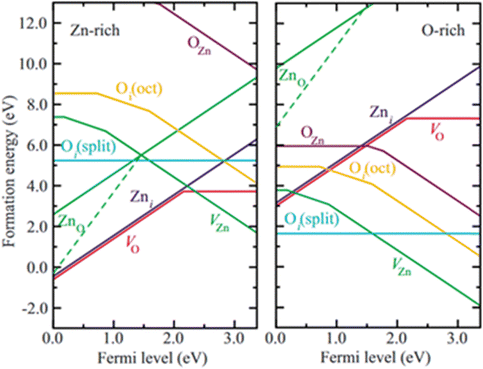 | ||
| Fig. 23 Calculated formation energies (Ef) of native point defects in ZnO as a function of the Fermi level (EF) under (left) Zn-rich and (right) O-rich conditions. The Fermi level is referenced to the valence band maximum, 0.0 eV. Solid lines represent the most thermodynamically stable charge state for a given defect, where the slope indicates the specific charge state and the kinks denote thermodynamic transition levels. Reproduced from ref. 131 with permision from American Physical Society,131 Copyright 2007. | ||
Table 7 consolidates this multi-scale perspective, contrasting historical paradigms with the modern consensus to identify critical engineering targets. This synthesis illustrates the interplay between intrinsic semiconducting behavior, the hierarchy of transport bottlenecks, and the thermodynamic boundaries of band engineering.
| Core electronic challenge | Conventional view (past) | Modern consensus (present) | Engineering imperative (future) | T-ZnO specific implication |
|---|---|---|---|---|
| Undoped n-type conductivity | Native defects (VO, Zni) act as primary donors. | Hydrogen impurities: unintentional H-incorporation is the dominant shallow donor; native defects act as compensators. | Impurity control: mitigate H-incorporation for high-purity applications, or exploit it for robust n-type transparent conductors. | Doping baseline: defines the carrier concentration floor for T-ZnO networks; shifts focus from vacancy control to hydrogen management. |
| P-type doping difficulty | High formation energy of acceptors; low solubility. | Self-compensation: lowering the Fermi level (EF) spontaneously lowers the formation energy of native donors, which neutralize acceptors. | Fermi level engineering: develop co-doping or non-equilibrium strategies to kinetically suppress compensating donor formation. | Device limitation: hinders T-ZnO p–n homojunctions; limits applications to unipolar (n-type) or heterojunction devices. |
| Wide bandgap/exciton energy | Stable UV emission; optical transparency. | Recombination trade-off: high exciton binding energy (∼60 meV) favors emission but reduces free carrier generation for photocurrents. | Field management: integrate strong internal fields to dissociate excitons (for PV/detectors) or enhance confinement (for LEDs). | Surface dominance: high surface-to-volume ratio of T-ZnO arms exacerbates surface recombination, competing with intrinsic bulk emission. |
| Macroscopic charge transport | Modeled as a simple resistor network. | Junction dominated: transport is limited by a hierarchy of bottlenecks: inter-tetrapod DSBs > core impedance > surface scattering. | Junction engineering: direct modification of the barrier (sintering, chemical passivation) and characterization via c-AFM/in situ TEM. | The critical bottleneck: network mobility is decoupled from crystal quality; high-performance requires solving the junction bottleneck. |
3.3 Optical properties
Building upon the understanding of T-ZnO's electronic structure, this section delves into its equally crucial optical properties. The unique functionality of T-ZnO arises from a complex, multi-scale interaction with light. This section deconstructs this hierarchy, beginning with the intrinsic absorption and emission processes dictated by the atomic-scale electronic structure. It then explores the primary strategies used to engineer these core properties, such as bandgap tuning and hybridization. From there, the discussion moves to the resonant cavity effects governed by the nano- to microscale arm geometry and finally explores the network-level light scattering that emerges at the macroscale. This framework provides a cohesive story of how T-ZnO's structure engineers its interaction with light from the quantum to the macroscopic level.3.3.1.1 Absorption. The primary optical absorption feature of T-ZnO is a sharp UV absorption edge below 400 nm.29,48,52,89,90,120,132–134 This is defined by a strong excitonic absorption peak often observed between 370–390 nm (Fig. 15b).26,29,52,86,89,90,119,120,132 With absorption coefficients exceeding105 cm−1,89 this peak corresponds to the fundamental, direct bandgap transition from the O2p-derived valence band to the Zn4s-derived conduction band.88,93 This intrinsic UV sensitivity is critical for applications like photocatalysis, where absorbed photons generate the electron–hole pairs necessary for redox reactions,86,90,110–113,133,135,136 and for UV photodetectors.48,52,86,97,120,121
While pristine ZnO primarily absorbs in the UV, defects (Section 3.1.3) introduce mid-gap electronic states that facilitate sub-bandgap absorption.90,109 This allows limited, but functionally important photocatalytic activity under visible light,13,47,87,90,109,135,137 although this is often insufficient for high-efficiency applications without further engineering.88,90
3.3.1.2 Photoluminescence (PL). PL serves as a critical diagnostic for evaluating T-ZnO quality and its defect landscape. T-ZnO typically exhibits two competing emission channels (representative spectrum in Fig. 24).
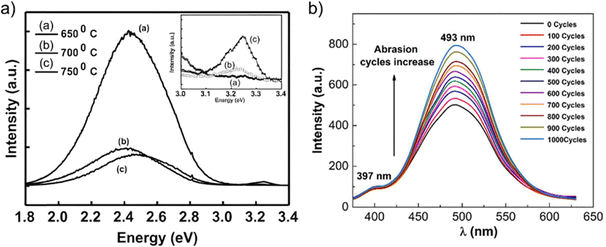 | ||
| Fig. 24 PL spectra of T-ZnO illustrating the competition between the UV near-band-edge (NBE) and green deep-level emission (DLE) bands. (a) Spectra as a function of growth temperature, demonstrating the relative suppression of the green DLE and dominance of the NBE emission at higher temperatures. (b) Spectra as a function of mechanical abrasion, showing the proportional increase of the green DLE peak (∼493 nm) alongside a stable UV NBE peak (∼397 nm). (a) Reproduced from ref. 92 with permision from Elsevier,92 Copyright 2015, (b) reproduced from ref. 135 with permision from Elsevier,135 Copyright 2022. | ||
(I) UV near-band-edge (NBE) emission: this is a sharp, intense emission band observed near 380–390 nm at room temperature.36,107,132,135 Driven by ZnO's high exciton binding energy (Section 3.2.1), it originates from the radiative recombination of excitons. The NBE is a composite of transitions involving free excitons and excitons bound to neutral donors (D0X) or acceptors (A0X).29,87,88,105,137–139 A dominant NBE signal is the hallmark of high crystalline perfection and low defect density.29,88,97,107,132,134,138–140 Under high excitation densities, this excitonic recombination can enable optically pumped UV lasing.122
(II) Deep-level emission (DLE): a broad emission band typically spanning the green-yellow-orange region (∼500–650 nm) and frequently centered at ∼510 nm.107,132,135,137,141 This band represents an inefficient “energy leak,” where energy is lost through light-producing transitions involving deep-level electronic states.29,88,105,107,132,135,137,142 These include the point defects (VO and Zni), and complexes described in Section 3.1.3, as well as extrinsic impurities (Cu, Li, N).87,88,96,106,107,134,135,139,142
The green luminescence debate: the origin of the prominent green luminescence (GL) band (∼500–550 nm) is particularly widely debated.87,88,134,137,143 The debate is polarized between two primary viewpoints: the intrinsic bulk defect model and the surface state model. A critical comparison of the evidence reveals why consensus remains elusive, and there are two viewpoints:
Viewpoint 1: the oxygen vacancy (VO) model. This classic model attributes GL to the recombination of a photogenerated hole with an electron trapped at a singly ionized VO (V+O) deep within the bulk lattice.144 Experimentally, GL intensity often increases in samples synthesized under reducing (oxygen-poor) conditions, which thermodynamically favors VO formation. EPR studies have frequently shown a correlation between GL intensity and the g ∼ 1.96 signal attributed to paramagnetic defects.145,146 Foundational DFT calculations indicate that the formation energy of VO is high in n-type ZnO147 and that its thermodynamic transition levels do not perfectly align with the green spectral emission, casting doubt on it being the sole origin.
Viewpoint 2: the surface state model. This model suggests that GL originates from electron transitions involving surface-adsorbed species (e.g., –OH groups) or surface vacancies/dangling bonds, rather than bulk defects. The high surface-to-volume ratio of T-ZnO makes surface effects dominant.148 Strong evidence comes from size-dependent studies where GL intensity scales with surface area.148 Crucially, surface passivation experiments (e.g., coating with polymers)149 often quench the GL band without altering the bulk crystal, directly implicating the surface as the emission source. This model struggles to explain why GL is observed in large, bulk-like single crystals where surface contributions should be negligible.150
While frequently associated with defects such as the VO or surface imperfection,92,97,107,132,135,140 compelling evidence from time-resolved PL and CL studies suggests that surface-adsorbed species or complex surface oxygen dynamics may also be responsible.87,88,96,104 This ongoing uncertainty highlights the necessity of the advanced correlative microscopy techniques proposed in Section 3.1.5 to create a definite link between atomic structure and optical signature.
3.3.1.3 Photoluminescence as a diagnostic tool. The relative intensities, peak positions, and lifetimes of the NBE and DLE bands are highly sensitive to synthesis conditions (Fig. 24a), defect concentrations (Fig. 24b), and even spatial location on the tetrapod structure.26,29,87,97,135,137–139,143,151 This makes PL a valuable diagnostic tool, allowing researchers to assess the properties of T-ZnO and its performance in several key ways:
Assessing quality: the ratio of the NBE to DLE intensity (NBE/DLE) serves as a key qualitative metric for material quality, where a higher ratio generally indicates a lower density of non-radiative defect pathways and often correlates with higher photocatalytic or photoelectrochemical efficiency.36,92,152
Probing charge transfer: TRPL is a powerful technique for studying charge transfer dynamics. In hybrid systems, the quenching of PL intensity is a direct indicator of efficient charge transfer from the ZnO to an attached molecule or nanoparticle (Fig. 25), signifying reduced recombination and improved device performance.89,97,107,132,134
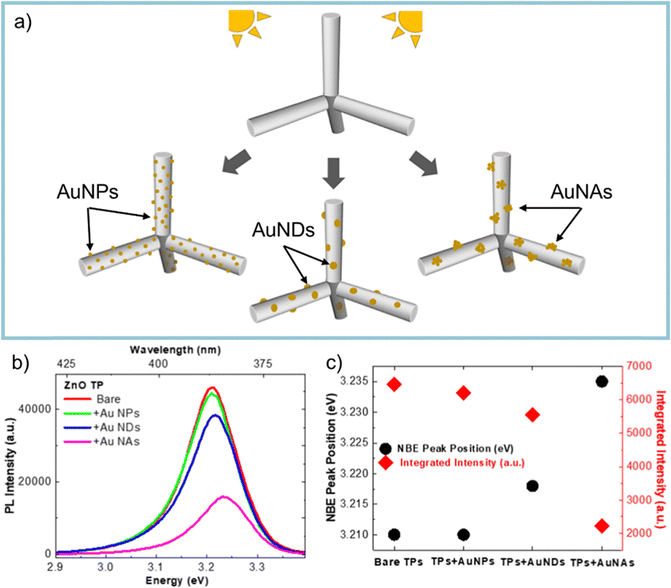 | ||
| Fig. 25 PL quenching in T-ZnO/Au hybrid systems. (a) Schematic illustrating the in situ photochemical growth of various gold nanostructures, nanoparticles (AuNPs), nano-domes (AuNDs), and nanoparticle aggregates (AuNAs), onto the T-ZnO scaffold. (b) Comparative PL spectra demonstrating the progressive quenching of the near-band-edge (NBE) emission of bare T-ZnO upon hybridization. (c) Quantification of the integrated NBE intensity drop as a function of the Au nanostructure morphology. (a-c) Reproduced from ref. 97 with permision from IOP Science,97 Copyright 2021. | ||
Overall, the PL spectrum provides a rich fingerprint of T-ZnO's internal workings, revealing both its ideal performance (NBE) and its efficiency losses (DLE). While its use for identifying specific defects is filled with uncertainty, its role as a comprehensive diagnostic for material quality and charge transfer dynamics is essential. To provide a detailed reference for these various emission bands and their debated origins, the key PL signatures are summarized and critically assessed in Table 8.
| Radiative channel | Emission | Approx. wavelength (nm) | Proposed origin | Diagnostic significance & impact on efficiency |
|---|---|---|---|---|
| Primary excitonic channel (efficient pathway) | NBE (UV) | ∼375–385 | Excitonic recombination (e.g., FX, D0X);88,92,107,132,139 exciton–exciton collision26,36,137 | A key metric of crystal quality; a high NBE/DLE ratio indicates low defect density.92,151,152 Its intensity also measures energy transfer at interfaces in hybrid system.140 |
| Secondary defect channels (loss pathways) | Violet/blue | ∼400–450 | Zni-related transitions; possibly Zni–VZn pairs29,36 | Indicates the presence of interstitial-type donor defects. |
| Green | ∼490–550 | Highly debated: VO is the common proposal, but surface states and other defects are also candidate.85,87,88,92,107,132,135,137,153 | Represents a primary “energy leak.” While its origin is ambiguous, its presence signifies a high density of deep-level traps that reduce overall quantum efficiency.105,107,135,137,151,153 | |
| Yellow/orange | ∼550–650 | Oi; VZn-related complexes,88 VO29 | Indicates the presence of deep acceptor-type defects or complexes, another energy-loss pathway. | |
| Red | ∼640–710 | VO or Oi states; N-doping.26,88,137 | Can be a signature of specific processing conditions (e.g., excess oxygen) or intentional doping. | |
3.3.2.1 Bandgap and absorption tuning. The precise optical bandgap and absorption characteristics of T-ZnO can be intentionally modified through several routes. At the nanoscale, quantum confinement in smaller nanostructures can induce a blue-shift in the absorption edge, effectively widening the bandgap.86,89,96 Variations in lattice strain (Section 3.1.1), controlled during synthesis, can also subtly tune the bandgap.29 More powerfully, doping with elements like nitrogen can introduce mid-gap states that narrow the effective bandgap and extend the material's absorption response into the visible region (Fig. 26a),88,90,109,154 a chemical and structural modification confirmed by XPS and Raman spectroscopy (Fig. 26b and c).90 This is a key strategy for developing visible-light-driven photocatalysts and photodetectors. Conversely, surface engineering can be used to passivate the ZnO surface and intentionally suppress its intrinsic photo-activity. Applying conformal coatings like polydimethylsiloxane (PDMS) or diamond-like carbon has been shown to hinder ZnO's photocatalytic response or prevent UV-induced wettability changes, which is crucial for applications where long-term stability is required.119,155
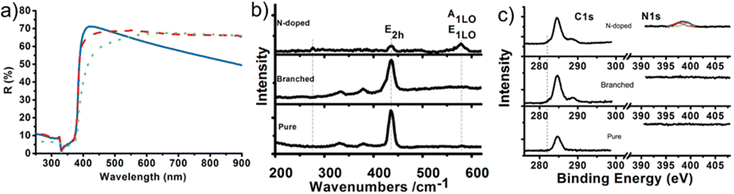 | ||
| Fig. 26 Spectroscopic evidence for nitrogen doping in T-ZnO and its effect on optical absorption. (a) Diffuse reflectance spectra comparing pure T-ZnO (blue) and N-doped T-ZnO (green), highlighting a sub-bandgap absorption tail in the visible region (∼400–650 nm) for the doped sample. (b) Raman spectra showing the emergence of a new defect-related peak (∼275 cm−1) and the suppression of the primary ZnO (E2h) mode in the N-doped sample. (c) XPS spectra displaying the N 1s peak (inset) in the N-doped sample, which is absent in pure T-ZnO. (a)–(c) Reproduced from ref. 90 with permision from American Chemical Society,90 Copyright 2012. | ||
3.3.2.2 Hybridization for visible light harvesting. Hybridization leverages the high surface-to-volume ratio and ultra-porous topology of the T-ZnO network (Section 3.6.1) to create intimate interfaces with sensitizing materials.88,93,109 These heterostructures bypass intrinsic absorption limits through three main strategies:
• Quantum dots (QDs): to create type-II heterojunctions that facilitate efficient charge separation after light absorption in the QD.89
• Plasmonic nanoparticles: noble metals like Ag or Au97,140 are used to leverage localized surface plasmon resonance (LSPR). The intense, localized electromagnetic fields generated by the LSPR dramatically enhance light absorption in the adjacent ZnO. The processes efficiency is highly sensitive to the size and morphology of the nanoparticles.97,140
• Carbon materials, dyes, and polymers: heterostructures with carbon-based materials like graphene oxid,119,132,134,143 polymers135 are formed to create junctions that promote charge separation and extend spectral absorption.
Hybrid systems, which increase the visible light harvesting of T-ZnO, utilize a diverse toolkit of physical mechanisms to overcome its intrinsic limitation.47 These include LSPR-induced near-field enhancement97,140 and better charge separation across Schottky barriers (for metal-ZnO hybrids97) or semiconductor heterojunctions.87–89,97,133,134 The effectiveness of the charge separation processes can be seen for example via the quenching of CL (Fig. 27), which provides direct proof of energy transfer from the T-ZnO to the plasmonic sensitize.140 This can also be studied in detail using TRPL.89,137
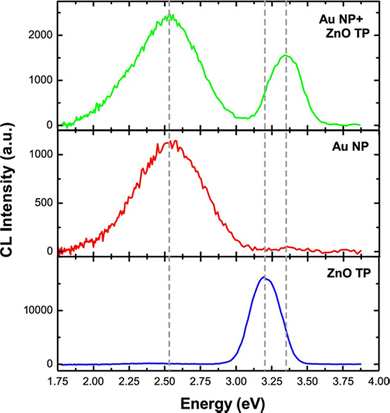 | ||
| Fig. 27 Cathodoluminescence (CL) spectra of a T-ZnO/Au plasmonic hybrid. The spectrum of the bare T-ZnO (blue) shows a dominant UV near-band-edge (NBE) emission (∼3.3 eV). In the Au NP/ZnO hybrid (green), this UV emission is quenched (∼10×), while the Au localized surface plasmon resonance (LSPR) emission peak (∼2.4 eV) is simultaneously enhanced. Reproduced from ref. 140 with permision from Springer Nature,140 Copyright 2016. | ||
Such engineered absorption properties are pivotal for advancing applications in photocatalysis,13,21,88,132–135,137 Surface-enhanced Raman spectroscopy (SERS),133 and visible-light photodetectors.86
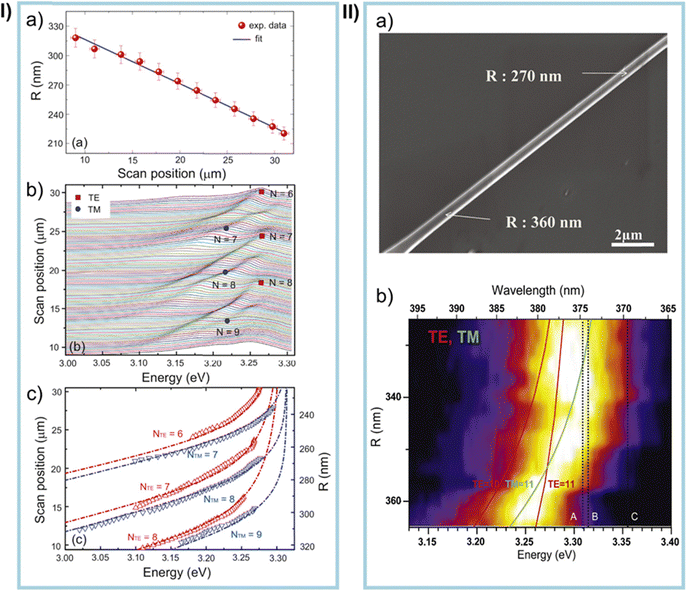 | ||
| Fig. 28 Whispering gallery mode (WGM) formation and exciton-polariton coupling in T-ZnO arms. (I) WGM peak observation: (a) T-ZnO arm radius (R) profile along the scan position. (b) Waterfall plot of spatially resolved PL spectra showing broad UV near-band-edge (NBE) emission modulated by periodic WGM peaks. (c) WGM energy shift versus arm radius fitted with a WGM-polariton model (dashed lines). (II) WGM-polariton dispersion: (a) SEM image of a tapered arm. (b) Spatially resolved cathodoluminescence (CL) map showing the NBE emission splitting into discrete WGM modes, with solid lines representing theoretical fits for the characteristic anti-crossing dispersion curve. (I (a)–(c)) Reproduced from ref. 138 with permision from Elsevier,138 Copyright 2018, (II (a) and (b)) reproduced from ref. 139 with permision from Elsevier,139 Copyright 2013. | ||
• Surface roughness: any significant roughness on the crystal facets will scatter light out of the resonant cavity, preventing mode formation or dramatically lowering the quality factor (Q-factor).139,142,151
• Crystalline defects: internal defects can act as non-radiative recombination or absorption centers, increasing optical losses and destroying the resonance.142
Consequently, the experimental observation of high-finesse WGM peaks validates a synthesis process's ability to yield near-perfect, defect-minimized crystal facets, linking the advanced optics of T-ZnO directly to the precision growth methods detailed in Section 2.142,151 In exceptionally high-purity arms, the strong coupling regime between confined WGM photons and intrinsic excitons facilitates the formation of hybrid light-matter quasiparticles known as exciton-polaritons.138,139 This state is characterized by a distinctive anti-crossing in the polariton dispersion curve (Fig. 28II), signaling a structurally and electronically pristine system capable of supporting stable excitons. Beyond linear interactions, the non-centrosymmetric wurtzite lattice enables nonlinear phenomena, such as second-harmonic WGMs within the T-ZnO legs.156 These advanced optical properties position T-ZnO-based resonators as promising candidates for polariton-based lasers, UV emitters with tunable decay dynamics,138,139 and ultrasensitive optical biosensors,142,151 all of which require a high degree of structural perfection.
3.3.4.1 The mechanism of light trapping. The physical origin of this behavior lies in the material's hierarchical architecture, where micron-scale tetrapods and high porosity create a spatially varying refractive index landscape. This network functions as a three-dimensional array of scattering centers where Mie scattering dominates photon dynamics.119 This process randomizes photon trajectories, inducing a “light trapping” effect wherein incident photons are constrained to tortuous paths.7,143 Consequently, the effective optical path length is dramatically increased, significantly boosting the probability of photon-material interaction and absorption.7,13,143 This trapping effect significantly boosts the probability that the photon will be absorbed.48 The functional implications include:
• Enhanced light harvesting: in devices like DSSCs, this light trapping leads to an increase in light harvesting efficiency.90,154
• Improved photocatalysis: for photocatalysis, the increased photon absorption probability directly leads to a higher rate of electron–hole pair generation, improving the overall reaction efficiency.137,143
3.3.4.2 Architectural synergy and signal amplification. The magnitude of this scattering can be so intense that the network's effective scattering cross-section exceeds the material's intrinsic absorption coefficient by an order of magnitude across broad spectral ranges.47,143 Crucially, a synergy exists between macroscale scattering and the atomic-scale defect landscape.90 While the porous architecture traps light to increase interaction frequency,7 surface defects, specifically oxygen vacancies facilitate the absorption of lower-energy visible photons.47 In this configuration, the scattering network acts as a geometric amplifier, enhancing the efficacy of defect-mediated absorption and substantially boosting the overall photoresponse of the system.
The preceding sections have described the multi-scale nature of T-ZnO's interaction with light, revealing a sophisticated hierarchy of optical phenomena. To consolidate these distinct levels, Table 9 provides a summary in the form of an optical engineering toolkit. This framework highlights how each structural scale (from the atomic to the macroscopic) offers a unique physical phenomenon that can be leveraged as a distinct tool for manipulating light, along with the key experimental strategies for tuning its performance.
| Scale of interaction | Dominant physical phenomenon | Primary engineering “Tool” | Key paramter (strategy) | Primary diagnostic method |
|---|---|---|---|---|
| Atomic scale (Section 3.3.1) | Bandgap absorption & PL | Control spectral sensitivity and diagnose material quality. | Bandgap tuning: doping, alloying. Hybridization: sensitizing with QDs, plasmonic nanoparticles, or dyes. | UV-Vis spectroscopy, PL, time-resolved PL. |
| Nano- to microscale (Section 3.3.3) | Waveguiding & WGMs | Create resonant microcavities for high-Q sensing, micro-lasers, and advanced emitters. | Synthesis perfection: growing arms with optically smooth facets and minimal crystalline defects. | High-resolution PL spectroscopy to resolve sharp, periodic WGM peaks. |
| Macroscale (Section 3.3.4) | Mie scattering & light trapping | Enhance overall light harvesting efficiency by increasing the effective optical path length. | Architectural control: tuning network porosity, tetrapod size, and density during synthesis. | Diffuse reflectance spectroscopy; measuring quantum efficiency in devices (e.g., solar cells, photocatalysts). |
However, the field's understanding of the energy relaxation pathways following photon absorption remains fragmented. While individual radiative and non-radiative channels are qualitatively identified, a quantitative map of the competing energy decay pathways within the complex network topology is still missing. Consequently, the grand challenge for T-ZnO photonics is to transcend static characterization and elucidate the complete energy cascade. This requires resolving a fundamental question: How is excitation energy partitioned among competing channels?
Resolving this partition requires determining specific quantum yields for each pathway:
• Non-radiative losses: quantifying energy dissipation at spatially localized defect sinks (the disordered central core) versus dispersed surface states.
• Radiative branching: distinguishing the ratio of excitonic NBE UV emission to defect-mediated DLE.
• Charge separation: determining the efficiency of carrier extraction for photocatalytic redox reactions or photocurrent generation.
Overcoming this challenge necessitates a shift from steady-state, ensemble-averaged measurements to advanced, time-resolved, and spatially-resolved spectroscopies. Future research must utilize femtosecond transient absorption spectroscopy to track ultrafast carrier cooling and spatially-resolved time-correlated single photon counting (TCSPC) to map recombination lifetimes across the specific arm-core-arm architecture. Mapping these energetic processes is the prerequisite for transitioning from empirical optimization to predictive, efficiency-focused optical engineering.
Table 10 deconstructs this energy cascade, identifying the dominant structural features, loss mechanisms, and specific engineering imperatives for each stage of the light-material interaction.
| Stage of interaction | Dominant structural feature | Physical mechanism | Primary loss channel | Engineering target |
|---|---|---|---|---|
| 1. Light harvesting (photon entry) | Macroscopic network: porosity and random arm orientation. | Multiple scattering (Mie): the 3D architecture extends the optical path length, increasing interaction probability. | Reflection/transmission: light escaping the network before absorption occurs. | “Black” ZnO: optimize packing density to maximize scattering events (light trapping). |
| 2. Excitation (absorption) | Crystal lattice: wurtzite band structure (Eg ∼ 3.37 eV). | Exciton generation: formation of stable electron–hole pairs with high binding energy (60 meV). | Thermalization: hot carrier cooling to the band edge (phonon emission). | Bandgap tuning: doping (Mg/Cd) to shift the absorption edge for solar spectrum overlap. |
| 3. Recombination (carrier dynamics) | Surface & core: high surface-area-to-volume ratio; disordered core. | Radiative vs. non-radiative: competition between UV emission and trap-assisted decay. | Surface traps: non-radiative recombination at surface states or core defects. | Passivation: core–shell structures to eliminate surface traps and boost quantum yield. |
| 4. Extraction (photon exit) | Microscopic geometry: hexagonal arm cross-section. | Resonance: arms act as Fabry–Pérot or WGM cavities. | Reabsorption: emitted photons being re-absorbed by the network before escaping. | Lasing cavities: exploit arm geometry to achieve coherent random lasing or WGM lasing. |
3.4 Mechanical properties
The mechanical performance of T-ZnO is a critical enabler for its application in flexible electronics, structural composites, and functional supports. Unlike typically brittle bulk ceramics, T-ZnO exhibits counter-intuitive resilience and flexibility, emergent properties derived not from the material itself, but from its hierarchical architecture. This analysis deconstructs these properties from the single-arm scale to the interconnected network level.However, this ideal behavior is weakened by the arm's real-world structure. The practical mechanical response is significantly influenced by the landscape of defects,161 particularly in the structurally complex and strained central core (Section 3.1.2), which can be considered the most susceptible region to stress concentration of the entire tetrapod. Extended defects like twin boundaries and stacking faults are concentrated in this region. These defects can act as stress concentrators and crack initiation sites. Detailed nanomechanical studies on individual carbon-based tetrapods derived from ZnO templates confirm this behavior. Specifically, they show that the structure's deformation is governed by a reversible localized buckling mechanism at the central joint (Fig. 29).163 Although this model was developed for hollow carbon-based microtubes, Veys et al.164 suggest that a similar buckling mechanism also occurs in solid ZnO tetrapods. The quantitative relationship between specific defect populations and key metrics like fracture toughness or fatigue resistance under cyclic loading remains a critical but under-investigated area. This lack of a predictive model means that the design of durable T-ZnO-based devices currently relies on empirical observation rather than rational engineering, highlighting a major challenge for the field.
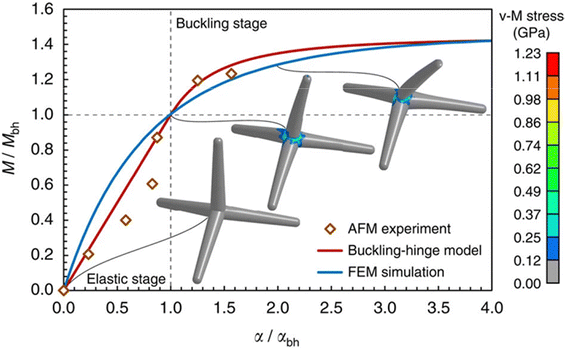 | ||
| Fig. 29 Simulation and modeling of localized buckling in a single T-ZnO tetrapod. The main plot compares experimental bending data (dots) with an analytical model (red line) and a finite element method (FEM) simulation (blue line). The inset FEM contour plots visualize the concentration of von Mises stress at the central joint. Reproduced from ref. 163 with permision from Springer Nature,163 Copyright 2017. | ||
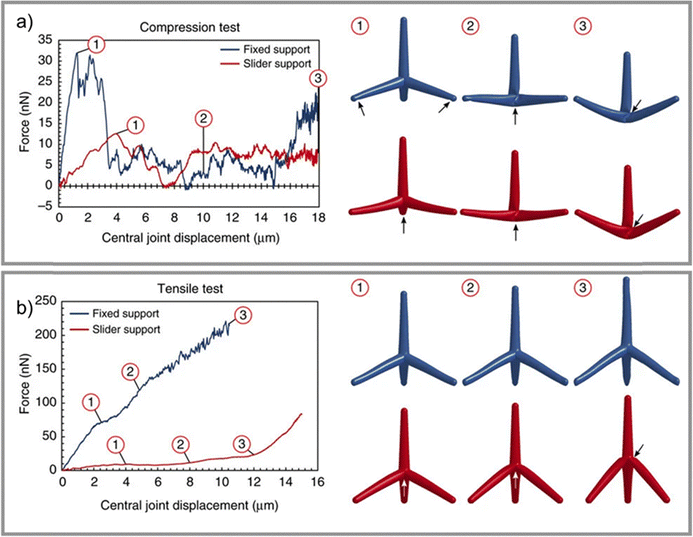 | ||
| Fig. 30 Finite element method (FEM) simulations of a single T-ZnO tetrapod's mechanical response. (a) Compression test showing a localized buckling mechanism, with arrows indicating the formation of buckling hinges that lead to a non-linear snap-through instability. (b) Tensile test showing the structural response under tension, plotted on a 10 times higher force scale compared to compression. (a) and (b) Reproduced from ref. 163 with permision from Springer Nature,163 Copyright 2017. | ||
This behavior creates a unique set of macroscopic properties. The networks are characterized by low density and high porosity (Fig. 31),8,85,120,165–168 which can be tuned over a wide range. Specific porosity values of derived materials/composites are often reported to exceed 90%,120,165–168 with the ability to be tuned between 50–98% via synthesis and post-processing.120,165,168,169 This lightness and mechanical adaptability is clearly exemplified in architectures derived from T-ZnO networks, such as aero-boron nitride (Aero-BN),167 three-dimensional carbon nanotube assemblies (t-CNTs),166,168 stretchable aerographite hybrids,8 hybrid T-ZnO foams,87 or necklace-like N-doped tubular carbon structures.85
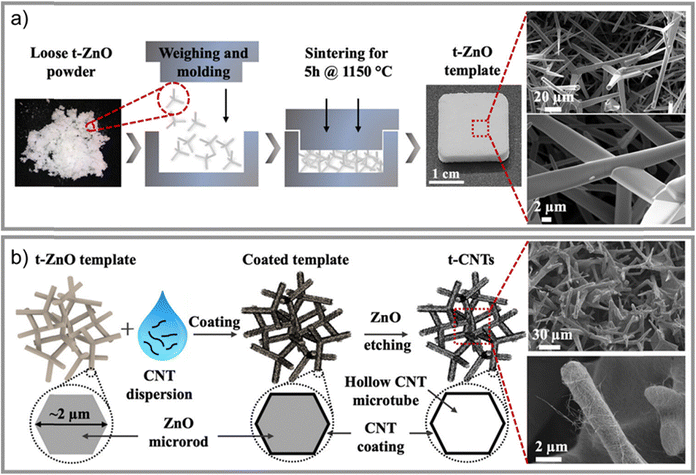 | ||
| Fig. 31 Synthesis and hierarchical architecture of T-ZnO networks and derived three-dimensional carbon nanotube (t-CNT) assemblies. (a) Fabrication process of the T-ZnO template from loose powder to a sintered, interconnected network, alongside SEM images of the resulting porous 3D architecture. (b) Sacrificial templating process illustrating the coating of a CNT dispersion onto the T-ZnO scaffold followed by ZnO etching, yielding a freestanding, hollow t-CNT assembly. (a) and (b) Reproduced from ref. 166 with permision from American Chemical Society,166 Copyright 2024. | ||
Critically, the effective Young's modulus (Eeff) of the network does not behave like a simple solid but follows a power-law relationship with network density (ρ) (Fig. 32): Eeff ∝ ρn, where the exponent n (typically 2–3) depends on the network architecture and the nature of the junctions.8 This relationship identifies density as the primary lever for tuning structural stiffness. However, global mechanical integrity remains contingent on the nature of inter-tetrapod contacts, which range from weak physical entanglement to robust, sintered necks87 that provide the necessary support for structural loads.8,121,167
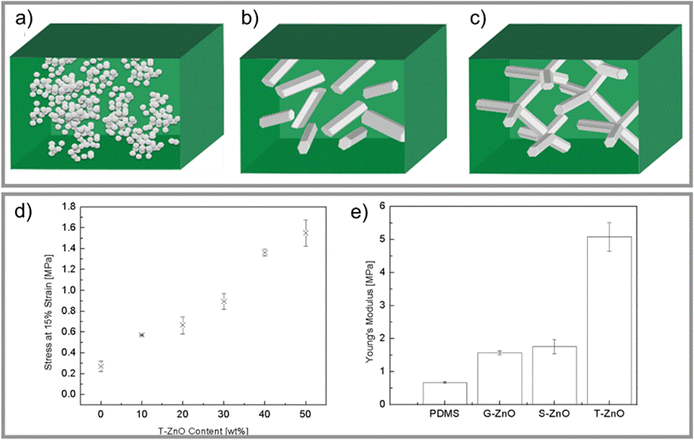 | ||
| Fig. 33 Schematic illustrating different filler geometries in polymer composites and their mechanical performance. (a) 0D nanosized spherical particles (S-ZnO). (b) 1D microsized ground short fibers (G-ZnO). (c) 3D microsized tetrapodal particles (T-ZnO). (d) Stress at 15% strain versus T-ZnO filler fraction in a PDMS composite. (e) Comparison of the Young's modulus of pure PDMS and composites containing 50 wt% of S-ZnO, G-ZnO, and T-ZnO fillers. (a)–(e) Reproduced from ref. 171 with permision from PloS One,171 Copyright 2014. | ||
Under external loading, the percolated T-ZnO network provides a high-efficiency pathway for stress transfer. This mechanism is the mechanical analogue to the electrical percolation discussed in Section 3.2.3: whereas the network in Section 3.2.3 provides a continuous path for carriers, here it provides a continuous path for mechanical flux. Applied stress is effectively shunted from the compliant polymer matrix into the rigid, interconnected inorganic framework. This transition from matrix-dominated to filler-dominated load-bearing results in dramatic enhancements to composite stiffness, tensile strength, and fatigue resistance (Fig. 34).36,94,115,135,170,171
| Scale/system | Key mechanical behavior | Basic physical principle | Primary engineering application | Critical limitation/design trade-off |
|---|---|---|---|---|
| Tetrapod arm | High stiffness & flexibility157,158,161 | Intrinsic WZ crystal stiffness157 combined with nanoscale flaw tolerance (low probability of critical-sized defects).158,159,161 | The basic, stiff building block for the macroscopic network. | The defect-rich central core acts as a stress concentrator158,163 and the final limit on the arm's ideal strength. |
| Macroscopic network | “Flexible ceramic” behavior (high compressibility & resilience)8,120,166 | 1. Transformation of deformation mode (fracture to bending).163,166 | Flexible sensor,120 compressible catalyst supports, lightweight structural materials.8,154,165,167 | A trade-off between stiffness (from strongly fused junctions) and flexibility/ability to spring back (from weak, physical junctions).87,121,154 |
| 2. Stiffness scales with network density (power law: E ∝ ρn). | ||||
| Composite reinforcement | Efficient mechanical reinforcement94,115,135,168,171 | The 3D shape forms a continuous, stress-bearing network at an extremely low volume fraction, enabling efficient stress transfer.48,115,135,168,171 | High-performance polymer composites with enhanced strength and durability.36,94,115,171 | Reinforcement is limited by the quality of the bonding at the filler-matrix interface. |
| Poor bonding leads to early failure. | ||||
As Table 11 elucidates, the mechanical response is not governed by a single variable, but by the interplay between network density and junction connectivity. Stiffness is primarily governed by the relative density (porosity). T-ZnO networks behave as open-cell cellular solids where the effective Young's modulus (Eeff) follows a classic power-law relationship with the relative density (ρ):
Second, the inter-tetrapod junction acts as the specific determinant of the deformation mode. While density sets the magnitude of the stiffness, the junction determines the material's resilience and operational range. Creating strong covalent necks via high-temperature sintering maximizes connectivity, yielding a rigid, stiff foam ideal for static structural support, though this comes at the cost of reduced elastic recovery. In contrast, relying on weak physical contacts results in a highly compliant network capable of large-scale reversible deformation (spring-back behavior). This configuration is perfectly suited for flexible sensors, albeit with a lower overall stiffness.121 Therefore, rational mechanical design requires simultaneously tuning the macroscopic density to set the modulus and the microscopic junction chemistry to define the failure mode.
This complexity points directly to the ultimate goal of the field: the development of a “digital twin” for T-ZnO. The grand challenge is to create a predictive, multi-scale computational model that can accurately forecast the macroscopic mechanical response based on the microscopic parameters of the network. Initial steps, such as combining finite element simulations with analytical models to capture the non-linear buckling mechanics of a single tetrapod, have been successfully demonstrated and serve as key validation for this approach (Fig. 29). However, extending this to a full network requires solving three difficult problems: capturing the non-linear mechanics of arm buckling,163 modeling the complex friction/adhesion physics of the junctions, and representing the random topology of the network. Success necessitates hybrid approaches integrating physics-based finite element analysis (FEA) with ML to handle topological complexity. Solving this challenge will finalize the evolution of T-ZnO from a laboratory discovery to a predictably engineered ceramic metamaterial.
3.5 Piezoelectric properties
Beyond its role as a passive electronic and optical medium, the T-ZnO architecture functions as an active electromechanical transducer. It converts mechanical energy into a suite of functional outputs, serving as a platform where single-strain inputs are translated into tailored electronic, optical, or chemical work.36,51,94 The operative mechanism is the intrinsic piezoelectricity of the WZ lattice (Section 3.1.1), which generates an internal electric field or piezopotential, under mechanical stress.94,172,173 The 3D tetrapodal geometry amplifies this effect by routing and concentrating strain at critical junctions, creating complex potential landscapes.95 This transducer functionality is deconstructed across three coupled domains:• Piezotronics: how the piezopotential is used to directly control the flow of charge carriers, creating an electronic signal from a mechanical force.
• Piezo-phototronics: how the piezopotential is used to dynamically alter the band structure at interfaces, controlling the generation, separation, or recombination of photogenerated carriers and creating a strain-tunable optical response.
• Piezocatalysis: how the strain-induced electric fields are used to enhance the efficiency of chemical reactions at the material's surface.
By establishing the physical origins and inherent limitations of the piezopotential, this section demonstrates how T-ZnO's architecture transforms simple mechanical deformation into a versatile energy-conversion engine.172
3.5.1.1 The piezopotential mechanism. Mechanical stress induces a relative displacement between the Zn2+ cations and O2− anions, creating an internal ionic dipole.51,94,172,174 The resulting macroscopic electric field, the piezopotential, locally modulates the electronic band structure by shifting the conduction and valence band edges.88,95 This piezopotential serves as a dynamic “gate” to modulate carrier transport (piezotronic effect).93,95,175 This coupling of ZnO's dual semiconducting (Section 3.2) and piezoelectric properties36,88,93,172,175,176 also allows mechanical stress to profoundly influence optoelectronic processes. The piezopotential can modulate the generation, separation, transport, or recombination of photogenerated carrier,174,176 thereby tuning light emission or detection efficiency in what is termed the piezo-phototronic effect.172,176 Further discussion on the origin of piezoelectricity in WZ ZnO can be found in.172
3.5.1.2 The critical limitation. The primary bottleneck for T-ZnO piezoelectricity is free carrier screening.93,172 Because ZnO is an n-type semiconductor88 with a high background electron concentration (Section 3.2.2), mobile electrons93 migrate in response to the strain-induced field. These electrons are attracted to positively polarized zones and repelled from negative ones, effectively neutralizing the ionic polarization and weakening the net electric field.95,172 Consequently, the observable piezoelectric response is governed by a dynamic competition between the rate of piezopotential generation (strain-driven) and the rate of carrier screening (conductivity-driven).95 Efficient device design requires not only a high piezoelectric coefficient but also precise engineering of the material's electronic properties to mitigate this screening effect (Fig. 35).172
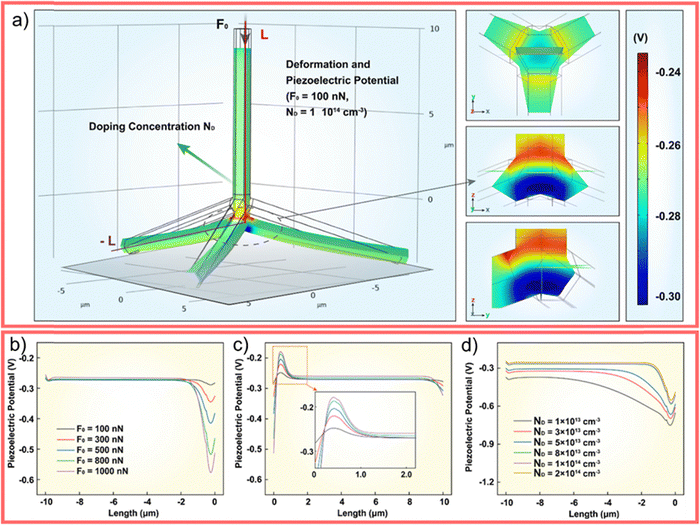 | ||
| Fig. 35 Finite element method (FEM) simulations of the T-ZnO piezopotential and its governing factors. (a) 3D model of the T-ZnO structure displaying the piezopotential distribution generated by an applied force (F0). Insets show the strain and potential concentrations at the core junctions. (b) and (c) Piezopotential profiles along the T-ZnO arms as a function of the applied force magnitude (F0). (d) Calculated piezopotential under a constant force versus background donor concentration (ND), illustrating the carrier screening effect. (a)–(d) Reproduced from ref. 95 with permision from Elsevier,95 Copyright 2024. | ||
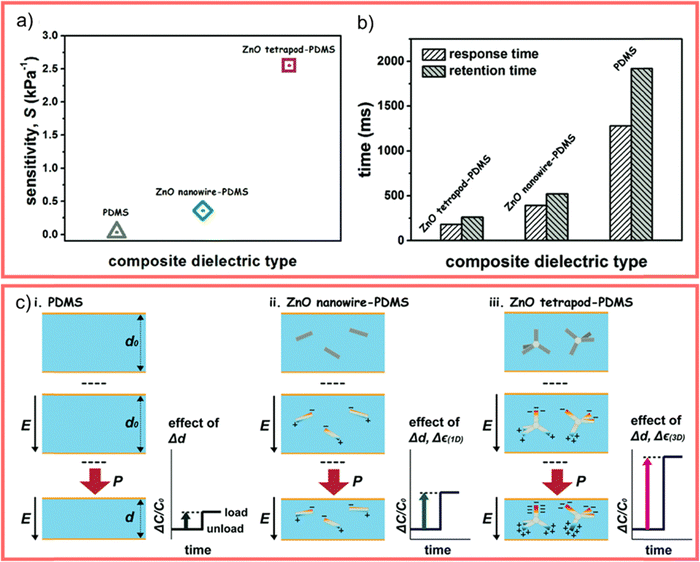 | ||
| Fig. 36 Comparison of piezocapacitive sensor performance using 3D T-ZnO tetrapods, 1D ZnO nanowires (NWs), and pristine PDMS. (a) Pressure sensitivity curves for the T-ZnO composite (2.55 kPa−1), 1D ZnO NW sensor (0.36 kPa−1), and pristine PDMS device (0.034 kPa−1). (b) Response and retention time profiles for the T-ZnO device. (c) Schematic illustrating the changes in polarization and permittivity (Δε(3D)) under applied pressure for 3D T-ZnO geometries compared to 1D nanowires under pressure. (a)–(c) Reproduced from ref. 51 with permision from Royal Chemical Society,51 Copyright 2021. | ||
3.5.2.1 The mechanism of strain concentration. The primary physical driver of this amplification is architected strain concentration at the arm roots.95 Unlike 1D structures where stress is more uniformly distributed, the T-ZnO geometry “funnels” mechanical energy toward the base of the arms and the structurally complex central core (Section 3.1.2).163 Since the piezopotential is directly proportional to local strain, this concentration locally amplifies the piezoelectric output (Fig. 36b–d), rendering the tetrapod a significantly more sensitive transducer than linear nanostructures. This geometric configuration offers two decisive operational advantages:
• Independent cantilever action: each arm can bend independently like a cantilever.163,175 This enables a single tetrapod to resolve the magnitude and 3D direction of complex stress vectors simultaneously, a multi-axial sensing capability inherently absent in 1D rods.95,175
• Complex potential landscapes: the 3D architecture supports complex deformation modes, including concurrent bending and torsion across multiple arms. This generates a spatially varying piezopotential distribution, particularly near the core junction (Fig. 36a).36,95 Such complex landscapes enable advanced functional devices,95 such as three-terminal logic switches, where localized mechanical force on a single “gate” leg modulates carrier transport between the remaining arms.175
This geometrically amplified transduction allows for the precise mechanical tuning of conductance175 and enhance the response of piezocapacitive sensors51 establishing T-ZnO as a superior platform for active electromechanical devices.
3.5.3.1 The mechanical gate mechanism. When a T-ZnO arm is bent, the resulting piezopotential locally raises or lowers the conduction and valence band edges. This band modulation can alter the height of a Schottky barrier at a metal contact or change the local carrier concentration within the semiconductor channel,172 thereby controlling the flow of current.95,175 This process is functionally analogous to a field-effect transistor (FET), but where the gating input is a mechanical force rather than an electrical voltage.172
3.5.3.2 Spatial localization and screening dynamics. The sensitivity of this gating effect is maximized at regions of peak strain, specifically the arm roots and the central core junction (Section 3.5.2).163 These localized high-strain zones enable high-precision control over charge transport. However, as noted in Section 3.5.1, the operational efficiency remains governed by the carrier screening effect. The net modulation of current depends on the kinetic equilibrium between the strain-induced ionic field and its electrostatic neutralization by mobile background electrons.
3.5.3.3 The single-tetrapod logic switch. The pinnacle of this application is the single-tetrapod electromechanical switch (Fig. 37).175 By applying a localized force to a single “gate” arm, a piezopotential is generated at the central junction. This potential acts to either enhance or deplete the conductive pathway between the remaining “source” and “drain” arms, facilitating discrete “ON” and “OFF” logic states. The output is hypersensitive to both force direction and source–drain bias, providing a level of control175 that underpins the development of advanced flexible logic, multi-axial strain sensors, and integrated electromechanical circuits.36,95
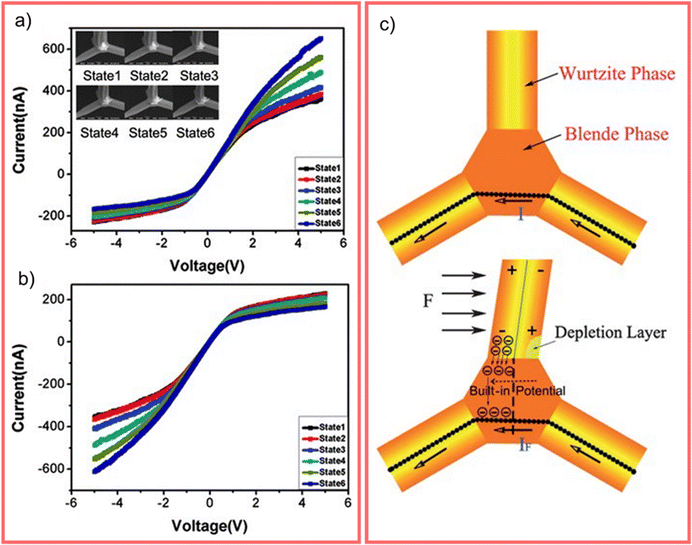 | ||
| Fig. 37 Experimental data and theoretical modeling of a single-tetrapod T-ZnO piezotronic logic device. (a) and (b) Current–voltage (I–V) curves measured while applying a mechanical force to bend the “gate” leg towards the (a) source electrode and (b) drain electrode. (c) Theoretical model illustrating the generation of a localized piezopotential and depletion layer at the core junction under an applied force (F). (a)–(c) Reproduced from ref. 175 with permision from Royal Chemical Society,175 Copyright 2011. | ||
When a T-ZnO structure is strained, the resulting piezopotential can change the Schottky barrier height,176 tilt the energy profile of a junction, or modify the width of a depletion region. This directly influences the key processes governing optoelectronic device performance: the generation, separation, transport, and/or recombination of photogenerated charge carriers (linking to concepts in Sections 3.2 and 3.3). For example, a strategically applied strain can create an electric field that assists in separating electron–hole pairs at a junction, reducing recombination and thereby enhancing the efficiency of a photodetector or photovoltaic cell.172,176 As with piezotronics, the effectiveness of this modulation is subject to the fundamental limitation of carrier screening.172,176 The ability of the piezopotential to sustainably alter the band structure is in constant competition with its neutralization by the material's mobile charge carriers.
A definitive demonstration of this principle is the strain-tunable UV sensor (Fig. 38).176 Under UV illumination, the sensitivity of a T-ZnO-based sensor to mechanical force is dramatically amplified. This is driven by a synergy at the Schottky contact: the mechanically induced piezopotential and the accumulation of photogenerated holes work in tandem to lower the SBH. This combined stimulus produces a current response far exceeding the sum of individual force or light inputs, enabling high-performance, multi-modal sensing.176
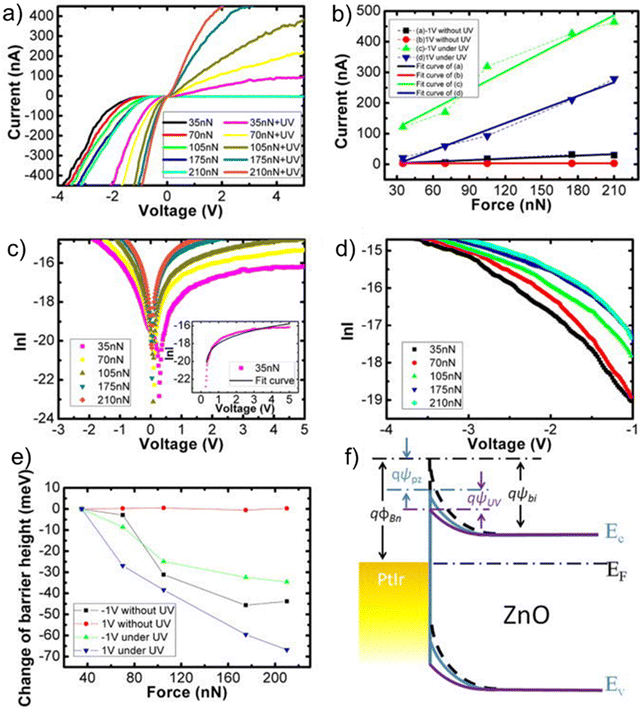 | ||
| Fig. 38 The piezo-phototronic effect: experimental data and theoretical model for a strain-tunable force sensor, demonstrating the synergistic coupling of mechanical strain and UV illumination. (a) and (b) Experimental I–V curves and the resulting current–force (sensitivity) plot. The sensitivity (the slope of the line in b) is dramatically enhanced under UV illumination, increasing from 0.17 A N−1 (dark, red line) to 2.05 A N−1 (UV, green line). (e) Quantification of the mechanism, showing the change in Schottky barrier height as a function of force. The barrier is lowered significantly more under the combined effect of strain and UV light. (f) The theoretical energy band diagram explaining the synergy. Both the strain-induced piezopotential (qΨbi) and the accumulation of photogenerated holes (qΨUV) work together to lower the SBH, boosting the device's current and sensitivity. (a)–(f) Reproduced from ref. 176 with permision from AIP Publishing,176 Copyright 2013. | ||
The tetrapodal architecture expands these possibilities through independent arm control. The ability to strain one arm to modulate the photoresponse in another suggests a path toward integrated opto-electromechanical logic, a degree of spatial and functional complexity unattainable in simple 1D nanowire systems.175,176
3.5.5.1 The mechanism of piezochemical transduction. The central role of the strain-induced piezopotential is to mitigate the primary bottleneck in catalysis: the rapid recombination of charge carriers.172–174 When T-ZnO is subjected to mechanical stress (e.g., from ultrasound or vibration), the resulting piezopotential creates a strong internal electric field. This field tilts the band structure and drives the spatial separation of charge carriers,172–174 in piezo-photocatalysis, these are photogenerated electron–hole pairs; in piezocatalysis, they are mobile surface charges or thermally generated carriers.174 This improved charge separation is the key to enhanced catalytic activity. By extending carrier lifetimes, the piezopotential increases the availability of charges for critical redox reactions, facilitating the generation of reactive oxygen species (ROS).173 These highly reactive radicals serve as the primary agents for the degradation of organic pollutants and the inactivation of bacteria.
The T-ZnO network is uniquely optimized for this “chemical work” compared to 1D structures.88,120 The independent deformation of multiple arms across the 3D topology maximizes the generation of diverse piezopotential gradients throughout the network, significantly enhancing global catalytic efficiency relative to simple nanorods.137 This architectural advantage has been validated in environmental remediation and disinfection, providing a robust pathway for converting ambient vibrations into high-value chemical work.173,174
| Device channel | Controlling principle | Basic physical mechanism | Key engineering application | Critical limiting factor |
|---|---|---|---|---|
| Electronic domain | Piezotronics | Piezopotential acts as a gate to control the flow of charge by changing energy barriers. | Strain/force sensors, electromechanical logic switches. | Free carrier screening: the neutralization of the piezopotential by background free electrons, reducing the gating effect |
| Optoelectronic domain | Piezo-phototronics | Strain-induced fields modify band alignment at interfaces, controlling the separation or recombination of electron–hole pairs. | Strain-tunable photodetectors, enhanced-efficiency solar cells. | Screening-induced quenching: high carrier concentration weakens the internal field necessary for assisting charge separation. |
| Chemical domain | Piezocatalysis | The surface piezopotential drives the spatial separation of charge carriers, prolonging their lifetime for redox reactions (e.g., ROS formation) | Enhanced pollutant degradation; Vibration-assisted disinfection | Surface potential reduction: screening reduces the magnitude of the surface potential available to drive redox chemistry |
As Table 12 elucidates, while the functional outputs are diverse, they are unified by a common physical bottleneck: the electrostatic screening of the piezopotential. The ultimate objective for the field is the evolution of T-ZnO into a “programmable piezotronic metamaterial.” This paradigm envisions a macroscopic network that can be dynamically reconfigured to perform disparate functions on demand through the application of precisely tuned, anisotropic strain fields. The central scientific question is whether T-ZnO can be “programmed” via its stress state: could an X-axis strain maximize piezotronic sensitivity for force sensing, while a transition to Z-axis strain reconfigures surface band-bending to optimize the network for piezocatalytic disinfection?
Realizing this level of control is a formidable “grand challenge” requiring:
1. Multi-physics modeling capable of mapping complex strain vectors onto the 3D piezopotential landscape of an entire stochastic network.
2. Kinetic accounting for the competitive screening rates of mobile carriers in real-time.
Solving these problems would mark the transition of T-ZnO from a material with isolated properties to a fully integrated, programmable electromechanical device platform.
3.6 The tunable functional interface
Beyond its intrinsic electronic and mechanical properties, the functional efficacy of T-ZnO is determined by its interaction with the external environment. This interaction is mediated not by a static boundary, but by a programmable “tunable functional interface.” This section frames the T-ZnO surface as the primary active zone, the locus for all heterogeneous processes including catalysis,133 sensing,102 and bio-integration occur.88,166 The utility of this interface is derived from four distinct engineering vectors that allow for the precise programming of interfacial behavior:• Hierarchical porosity and scale: an architectural feature that defines the magnitude and accessibility of the active surface area.48
• Atomic-scale landscape: the intrinsic chemical reactivity governed by specific crystal facets and point defect distributions.
• Surface functionalization: the capacity for covalent or non-covalent modification to introduce exogenous chemical functionalities.
• Dynamic wettability: a switchable gating mechanism that regulates molecular access to the interface through stimulus-responsive surface energy changes.47
Synergistically optimizing these parameters transforms T-ZnO from a passive scaffold into a sophisticated, multi-functional platform for advanced interfacial engineering.
• Large super-macropores between clusters of tetrapods act as highways for rapid, convective mass transport.
• Smaller macropores within the interlocked arms allow for diffusive access to the deeper regions of the network.
This system is paramount for any heterogeneous application, as it ensures that reactants can efficiently reach the high density of active sites on the internal surfaces.13,179 Total network porosity is highly tunable, frequently exceeding 90%.13,37,120,165
This unique combination of high, accessible surface area and complex 3D shape also provides distinct advantages in composite materials.48,135 The tetrapod geometry promotes homogeneous self-dispersion within a matrix and ensures that its nanoscopic features remain accessible, unlike simple nanoparticle agglomerates.38,48 The branching structure can also increase the effective roughness factor of a surface,90 a key parameter for tuning wettability. Furthermore, t his entire highly accessible architecture is a transferable property, when T-ZnO networks are used as sacrificial templates, the resulting aeromaterials inherit this high porosity, demonstrating the power of T-ZnO as an architectural design.120,143,165
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) facet.180 The non-polar facets are charge-neutral and thermodynamically stable. In contrast, the polar (0001) and (000
) facet.180 The non-polar facets are charge-neutral and thermodynamically stable. In contrast, the polar (0001) and (000![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) facets are terminated exclusively by a single ion type (either Zn2+ or O2−), creating a net surface charge and a diverging electrostatic potential.93 This configuration is thermodynamically unstable, an effect known as the “polar catastrophe”.180
) facets are terminated exclusively by a single ion type (either Zn2+ or O2−), creating a net surface charge and a diverging electrostatic potential.93 This configuration is thermodynamically unstable, an effect known as the “polar catastrophe”.180
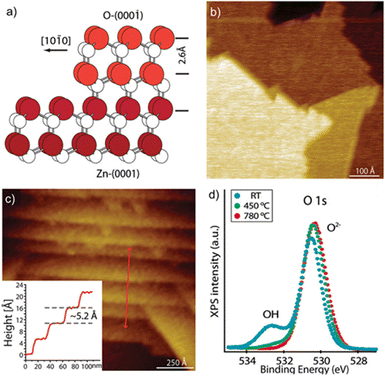 | ||
Fig. 39 Structural and chemical characterization of the polar O-terminated (000![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) facet. (a) Ball model of the wurtzite crystal structure illustrating the polar stacking of Zn and O atoms. (b) and (c) Scanning tunneling microscopy (STM) and AFM images displaying the step-terrace surface morphology, with the inset in (c) showing a height profile across the steps. (d) O 1s XPS spectra showing peaks corresponding to bulk lattice oxygen (O2−) and surface hydroxyl (–OH) groups as a function of temperature. (a)–(d) Reproduced from ref. 180 with permision from American Chemical Society,180 Copyright 2011. ) facet. (a) Ball model of the wurtzite crystal structure illustrating the polar stacking of Zn and O atoms. (b) and (c) Scanning tunneling microscopy (STM) and AFM images displaying the step-terrace surface morphology, with the inset in (c) showing a height profile across the steps. (d) O 1s XPS spectra showing peaks corresponding to bulk lattice oxygen (O2−) and surface hydroxyl (–OH) groups as a function of temperature. (a)–(d) Reproduced from ref. 180 with permision from American Chemical Society,180 Copyright 2011. | ||
To stabilize themselves, these polar surfaces must undergo significant atomic or electronic reconstructions. This can involve the formation of triangular surface pits, the adsorption of counter-ions (like hydroxyl groups), or the creation of a high density of surface vacancies.38,180 This mandatory reconstruction is the physical origin of the polar facets' superior reactivity compared to non-polar planes.87,173 These reconstructed surfaces present a high density of unique, low-coordination (atoms with fewer bonds than usual), and electronically active sites that are ideal for molecular adsorption and catalysis.87,93,179
3.6.2.1 The role of surface defects and adsorbates. The atomic landscape is further populated by intrinsic defects and ambient adsorbates that act as the primary engines for heterogeneous redox reactions.87,105 The two main types are:
• Surface defects: intrinsic point defects, particularly VO at or near the surface, act as critical sites for dissociative molecular adsorption and electron transfer.38,47,87,107 Their concentration and chemical state, which can be measured by surface-sensitive techniques like XPS,38,88,115,179 directly correlate with photocatalytic activity.137,181
• Adsorbates: under ambient conditions, the T-ZnO surface is not pristine but is typically covered with a layer of adsorbed species, primarily hydroxyl groups (OH)180 and molecular oxygen (O2).86,87,120,179 These adsorbates are not just contaminants; they are essential parts of the active interface.103,179 Surface hydroxyls can act as proton donors/acceptors or as sites for generating highly reactive hydroxyl radicals (˙OH) during photocatalysis,47,109 while adsorbed oxygen is the primary electron acceptor for generating superoxide radicals (O2˙−). The presence and nature of these species can be readily identified using techniques like Fourier-transform infrared spectroscopy (FTIR).86,135,179
Mastering this atomic landscape, specifically the interplay between polar stabilization and defect distribution, is the prerequisite for engineering high-selectivity sensors and high-efficiency catalysts.100
3.6.3.1 Covalent attachment. The primary method for creating robust, permanent modifications is through covalent attachment, most commonly achieved via silanization. The high density of surface hydroxyls provides ideal reaction sites for silane coupling agents. For example, octadecyltrichlorosilane (OTS) can be used to create a low-surface-energy layer.113 Alternatively, aminosilanes like APTES provide anchor points for further functionalization.107,182 This creates a stable baselayer that can then be used to chemically bond a wide variety of functional molecules, including the following:
• Biomolecules: for creating highly specific bio-interfaces, such as immobilizing Deoxyribonucleic Acid (DNA) for gene delivery182 or antibodies for biosensing applications.88,107Fig. 40 provides a complete model for this process, showing both the step-by-step chemical functionalization (Fig. 40a) and the resulting changes in the electronic band structure (Fig. 40b–e) that create the sensing mechanism.107
 | ||
| Fig. 40 Schematic model of T-ZnO biosensor functionalization and interfacial band bending. (a) Chemical procedure for covalent attachment (silanization), detailing the functionalization of the native ZnO surface with APTES to provide amine (–NH2) anchors for antibody binding. (b)–(e) Energy band diagrams illustrating the progressive modification of surface band bending following each functionalization step. (a)–(e) Reproduced from ref. 107 with permision from American Chemical Society,107 Copyright 2022. | ||
• Polymers or nanoparticles: for improving filler-matrix adhesion in composites or creating hybrid materials.48,135,140,171,179
3.6.3.2 Conformal coating and shell accretion. The open, 3D topology of the T-ZnO network serves as a superior template for the accretion of uniform, continuous coatings or shells,102,133 effectively generating a new functional interface atop the ZnO core. A broad spectrum of materials can be deposited to form core–shell heterostructures with synergistic properties:
• Plasmonic nanoparticles: decorating the surface with Ag12,13,88 or Au97,140 nanoparticles is a primary strategy for inducing LSPR. This effect enhances photocatalytic efficiency and enables SERS (Fig. 41).140
 | ||
| Fig. 41 Surface functionalization of T-ZnO with plasmonic gold (Au) nanoparticles. (a) Schematic of the photochemical synthesis for decorating a T-ZnO scaffold with Au nanoparticles. (b) Photographs of the T-ZnO dispersion displaying color changes during progressive Au nanoparticle formation. (c) HAADF-STEM image of a T-ZnO arm uniformly decorated with Au nanoparticles. (e) High-resolution TEM image showing the interface between the crystalline Au nanoparticles and the T-ZnO surface. (a)–(e) Reproduced from ref. 140 with permision from Springer Nature,140 Copyright 2016. | ||
• Other semiconductors: depositing shells of materials like TiO2,12,13,133 or ZnSe/CdSe (via dipping procedures89) creates heterojunctions with staggered band alignments. These configurations are engineered to maximize charge separation and minimize recombination losses.87,88,97
• Protective layers: ultrathin protective layers of materials such as Al2O3,88 TiO2,90 amorphous silica derived from TEOS,178 or diamond-like carbon nanocomposites119 are utilized to passivate electronic surface states. These coatings serve as a critical defense against photocorrosion, ensuring long-term device stability in harsh environments.
• Polymers and metal–organic framework (MOFs) and 2D-nanomaterials: the network can be coated with polymers like PDMS,155 polymethacrylates135,136 or poly(N-isopropylacrylamide) (PNIPAM) hydrogels,165 or with MOFs like zeolitic imidazolate framework, ZIF-8,12,102 or 2D-nanomaterials like two-dimensional transition metal carbides/nitrides (MXenes),183 to create hybrid materials for specialized applications.
3.6.3.2 Molecular immobilization. Finally, the extensive and accessible surface, combined with its chemically active sites (Section 3.6.2), allows for the efficient adsorption and immobilization of specific molecules. Fig. 42 presents a definitive case study of this mechanism, detailing both the synthesis of a T-ZnO hybrid (Fig. 42a) and the precise electrostatic mechanism (Fig. 42b) by which surface sites adsorb target analytes.179 This is a key strategy for creating interfaces with highly targeted functions, for instance:
 | ||
| Fig. 42 A case study of T-ZnO functionalization for molecular adsorption. (a) A schematic showing the synthesis of a T-ZnO/Activated Carbon hybrid material. (b) The detailed mechanism for molecular immobilization, demonstrating how surface hydroxyls (−OH) act as essential active sites that are protonated to electrostatically attract and adsorb a target chemical analyte (Cr(VI)). (a) and (b) Reproduced from ref. 179 with permision from Elsevier,179 Copyright 2019. | ||
• Dye sensitization: adsorbing dye molecules to enhance visible light absorption for photocatalysis.12,135
• Selective sensing: creating surfaces tailored for the specific adsorption of gas molecules12,13,37,102 or other chemical analytes88,179 for high-sensitivity sensor devices.86,120
• biomolecular immobilization: attaching specific biomolecules for biosensing12,88,107,120 or advanced biomedical applications. A notable example involves amino-modified tetrapods used to electrostatically bind plasmid DNA. The unique tetrapodal geometry allows the structures to ‘stand’ on cell membranes, facilitating gene delivery while minimizing cytotoxicity by preventing full cellular uptake.182
Through these diverse strategies, the T-ZnO surface can be transformed from its native state into a highly programmed, multifunctional interface, precisely tailored for a wide variety of advanced applications.
Crucially, this wettability is not static and can be reversibly switched using various external stimuli, including UV irradiation, thermal annealing, and applied electric fields.110–113,184 The most widely studied method is photoswitching. Under UV irradiation with photon energy greater than ZnO's bandgap, electron–hole pairs are generated. The holes migrate to the surface and react with lattice oxygen ions (O2−) to create surface oxygen vacancies. These defect sites are kinetically favorable for the dissociative adsorption of water molecules from the ambient air. This leads to a high density of surface hydroxyl groups and a transition to a hydrophilic or superhydrophilic state.110,112,113 Conversely, this process can be reversed and the hydrophobic state restored through thermal annealing135 or by extended storage in the dark, which removes the adsorbed hydroxyl groups.110,112 While this intrinsic photoswitching is a property of the ZnO material itself, hybrid systems can also be designed where the switching is governed by organic photochromic molecules (like azobenzene derivatives) integrated into the T-ZnO composite network.155
This switchable behavior has profound functional consequences, allowing the surface to be actively “gated” on or off for specific applications. A hydrophilic surface promotes intimate contact between the T-ZnO lattice and the surrounding aqueous medium. This maximizes the interaction between reactants and active sites, which can accelerate interfacial processes such as photocatalysis by over 2.5 times compared to the hydrophobic state (Fig. 43).47 Conversely, a superhydrophobic state, by trapping a layer of air, can effectively shut down the same reaction by preventing aqueous reactants from reaching the catalytic surface; experiments on ZnO nanorods confirm that achieving a superhydrophobic state leads to minimal photocatalytic activity.111
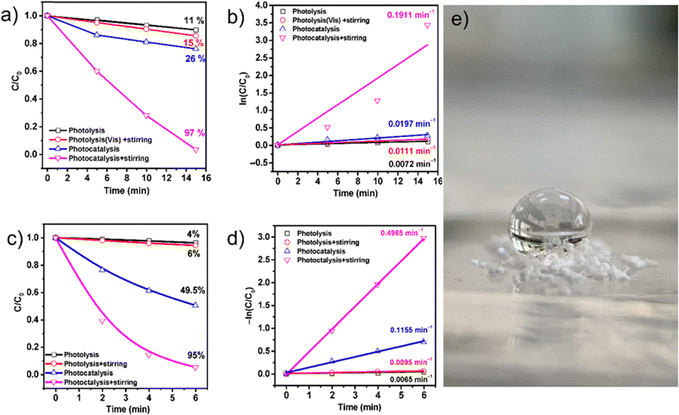 | ||
| Fig. 43 (a) and (b) Changes in the concentration of MB and kinetic curves for hydrophobic ZnO-Ts. (c) and (d) Changes in the concentration of MB and kinetic curves for hydrophilic ZnO-Ts. (e) A photograph of a water droplet on the surface of the hydrophobic ZnO-T powder. (a)–(e) Reproduced from ref. 47 with permision from MDPI,47 Copyright 2023. | ||
This inherent photosensitivity, however, is not always desirable, as uncontrolled changes in wettability can compromise the long-term stability of a device. This effect can be intentionally suppressed through surface engineering. For instance, applying a thin, conformal passivating layer, such as a diamond-like carbon nanocomposite film, can physically block the UV-induced mechanism and stabilize the surface wettability over time, even under UV exposure.119 Thus, the wettability of a T-ZnO interface is a highly tunable property that can be used as a functional gate or protected for stability, depending on the specific application requirements.
| Primary engineering goal | Functionalization strategy & physical mechanism | Resulting interfacial property | Target application |
|---|---|---|---|
| Enhance visible light absorption | Hybridization with sensitizers: LSPR from plasmonic NPs (Ag, Au)97,140 | Increased optical cross-section in the visible range; improved charge separation. | Visible-light photocatalysis, solar cells, photodetectors. |
| Heterojunctions with QDs or dyes89 | |||
| Engineer surface reactivity | Catalytic decoration: attachment of noble metal NPs (e.g., Au, Pt); creation of heterojunctions with co-catalysts (e.g., TiO2)133 | Creation of new active sites; modified adsorption energies; enhanced charge separation at interfaces. | Heterogeneous catalysis, surface-enhanced Raman spectroscopy.140 |
| Create bio-specific interfaces | Biomolecular attachment: covalent anchoring of enzymes, antibodies, or DNA via silanization13,107,182 | Specific molecular recognition capability; tailored biocompatibility. | Biosensing, targeted drug/gene delivery, bio-scaffolds. |
| Tune surface wetting & adhesion | Surface energy engineering: modification with low-surface-energy molecules (e.g., PFO,178 OTS113) or uniform coating with polymers (e.g., PDMS155) to engineer hierarchical roughness and control surface energy. | Tailored surface energy, leading to superhydrophobicity or superhydrophilicity | Self-cleaning surfaces, anti-fouling coatings, water-oil separation membranes |
| Improve stability & passivation | Dielectric coating: conformal deposition of chemically stable, wide-bandgap oxides (e.g., Al2O3, TiO2)87,88,90 | Passivation of electronic surface states; creation of a protective barrier against photocorrosion. | Stable photoanodes for water splitting, durable optoelectronic devices. |
As Table 13 illustrates, mastering these individual parameters to create single-function interfaces represents the current state-of-the-art. The next frontier lies in multi-functional integration. Therefore, the grand challenge for the field is the development of a “spatially-addressable, multi-functional bio-interface.”
This vision entails using the T-ZnO network not just as a uniform scaffold, but as an integrated device with distinct, spatially defined functional zones. The central scientific objective is to achieve a level of regional control that allows different functions to be programmed onto specific segments of the same tetrapod network, the “lab-on-a-tetrapod” concept. Future research must establish the feasibility of compartmentalizing the 3D architecture such that:
• Zone A is functionalized with enzymes for specific bio-detection.
• Zone B is decorated with plasmonic nanoparticles to serve as a SERS hotspot.
• Zone C features dynamically switchable wettability to function as a microfluidic gate or valve.
Achieving this requires the development of spatially selective functionalization techniques and a predictive understanding of how these distinct engineered zones interact within a continuous 3D architecture. Solving this challenge will transform T-ZnO from a passive scaffold into a truly programmable platform for the most complex, integrated interfacial systems in modern materials science.
3.7 Integrated structure–property nexus: from coupled physics to core design principles
The true structure–property nexus in T-ZnO demonstrates how a shared, hierarchical morphology forces distinct physical domains into a simultaneous, tightly coupled state. Rather than acting as a passive scaffold, the tetrapodal architecture dictates an instantaneous transduction of energy across multiple physical regimes. For instance, the application of a macroscopic mechanical force to the network does not yield a simple, isolated structural deformation. Instead, the force is geometrically concentrated as localized strain at the arm roots, which instantaneously generates a highly localized piezopotential. This strain-induced field simultaneously redefines the electronic landscape by shifting charge carrier transport pathways, alters the optical recombination rates of excitons, and induces surface band-bending that immediately modifies the rates of solid–liquid interfacial catalysis. Consequently, engineering T-ZnO requires acknowledging that any single input will invariably trigger a tightly coupled, multi-physics response.3.7.1.1 Atomic scale (Fig. 44). The fundamental characteristics of T-ZnO are dictated by WZ thermodynamics. At this level, the crystal lattice provides the intrinsic high mechanical stiffness, the wide bandgap necessary for UV optics, and the non-centrosymmetry required for piezoelectricity. Furthermore, the defect landscape, primarily governed by unintentional hydrogen impurities and intrinsic thermodynamic limits, serves as the baseline tuning layer for conductivity and visible-range emission. However, this scale also introduces absolute thermodynamic constraints, most notably the spontaneous formation of native donor defects (self-compensation) that severely limits the feasibility of stable p-type doping.
3.7.1.2 Nano/micro scale (Fig. 45). The geometry of the individual tetrapod arms translates these atomic properties into localized functionalities. The high aspect ratio and single-crystalline nature of the arms enable nanoscale flaw tolerance and permit them to function as optical microcavities supporting WGMs. Simultaneously, this scale introduces the system's primary structural bottlenecks: nanoscale surface depletion layers that can pinch off charge transport, and the highly defective central core, which acts as a mechanical stress concentrator and a primary scattering center for electrons.
3.7.1.3 Macroscale (Fig. 46). The collective behavior of the interconnected 3D network yields macroscopic properties that differ fundamentally from the bulk material. The tetrapodal geometry transforms the intrinsically brittle ceramic into a flexible, compressible macroscopic foam and converts a pristine UV absorber into a highly efficient, broad-spectrum light scatterer via Mie scattering. The ultimate performance limitation at this scale is defined by the inter-tetrapod junctions, where the formation of double Schottky barriers dictates the macroscopic electrical and thermal transport efficiency of the entire network.
3.7.2.1 Axiom 1: morphological translation of intrinsic properties. The 3D architecture dictates how bulk material properties manifest at the macroscale. The tetrapodal structure acts as a geometric translator, enabling the emergence of properties, such as macroscopic mechanical flexibility and extreme porosity, that the intrinsic ceramic constituents cannot achieve in thin-film or bulk forms.
3.7.2.2 Axiom 2: the inevitability of multi-physics trade-offs. Because the physical domains in T-ZnO are inherently coupled, optimizing one parameter frequently degrades another. For example, heavily doping the network to maximize electrical conductivity inherently increases the concentration of free charge carriers, which subsequently screen and dampen the strain-induced piezoelectric response. Similarly, enhancing the mechanical strength of the macroscopic network via high-temperature sintering creates rigid covalent bonds that destroy its structural flexibility. Engineering T-ZnO, therefore, requires managing these compromises rather than attempting to eliminate them.
3.7.2.3 Axiom 3: dynamic evolution of the functional interface. The structure–property relationships within T-ZnO are not static. Under operational conditions, the material undergoes continuous evolution: the surface defect landscape interacts with environmental adsorbates, the central cores accumulate mechanical fatigue, and the functional interface degrades. This dynamic behavior highlights a critical “synthesis-stability nexus”, wherein the initial synthesis parameters dictate not only the as-grown properties but also the evolutionary degradation and ultimate operational lifetime of the material.
4 T-ZnO applications
The true promise of T-ZnO lies not only in enhancing existing technologies but also in enabling entirely new approaches, ranging from innovative microstructured actuators to wearable sensors that monitor health in real-time and ultralightweight materials with the potential to revolutionize aerospace, medicine and soft robotics. This section highlights the applications that are beginning to translate T-ZnO's potential into reality. The unique combination of properties detailed in Section 3, from its semiconducting (Section 3.2) and piezoelectric nature (Section 3.5) to its remarkable mechanical resilience (Section 3.4) and high specific surface area (Section 3.6.1), establishes T-ZnO as a uniquely versatile platform for a wide range of advanced applications. The utility of T-ZnO is not just a sum of these individual characteristics. Instead, it is a result of their synergistic interplay, as summarized in the integrated structure–property nexus (Section 3.7). The applications that stem from this versatility can be broadly categorized based on the primary role the material plays. On one hand, many applications exploit the intrinsic properties of ZnO, where the material itself is the active component in sensing, catalysis, or optoelectronics. On the other hand, a ground-breaking class of applications leverages its unique 3D morphology. Here, the tetrapod network acts purely as a structural backbone for mechanical reinforcement or, more innovatively, as a sacrificial template. This templating capability allows researchers to architect entirely new materials with unprecedented properties, literally sculpting other materials from the micro to the macro scale using the T-ZnO network as a removable template. A visual overview of these distinct application pathways, from active sensors to templated aeromaterials, is presented in Fig. 47. This section will explore these diverse applications, highlighting how T-ZnO's hierarchical structure is leveraged to achieve novel functionalities.4.1 T-ZnO as an active functional material
Applications in this category exploit the intrinsic electronic (Section 3.2), optical (Section 3.3), piezoelectric (Section 3.5), or chemical properties (Section 3.6) of ZnO itself. Here, the material's high surface area and unique morphology serve to enhance its inherent performance. In its role as an active functional material, T-ZnO directly employs its fundamental material characteristics to interact with and respond to its environment. This includes leveraging its intrinsic semiconducting nature (Section 3.2), where electrical conductivity can be precisely modulated by external stimuli like UV light or gas adsorption. As well as its distinct optical properties, governed by a wide bandgap (Section 3.3.1) and characteristic PL (Section 3.3.2) and its piezoelectric effect, which originates from its non-centrosymmetric crystal structure (Section 3.5.1) and allows for the conversion of mechanical stress into electrical energy.Crucially, performance in these applications is not merely a function of bulk ZnO properties but is dramatically amplified by the tetrapod architecture. The high surface-to-volume ratio of the nanoscale arms (Section 3.6.1) creates a vast interface for interaction, making the material exceptionally sensitive. Furthermore, the interconnected 3D network ensures this surface is highly accessible and provides robust pathways for efficient charge transport (Section 3.2.3). This prevents the performance losses often seen in simple nanoparticle or nanowire agglomerates. In essence, the unique morphology acts as a structural amplifier. This makes T-ZnO a superior platform for devices like highly sensitive sensors and materials for advanced surface engineering and environmental control.
4.1.1.1 Chemiresistive sensors (gas). The high specific surface area (Section 3.6.1) and semiconducting nature (Section 3.2) of T-ZnO make it well suited for chemiresistive sensing, where changes in electrical resistance are induced by interactions with the chemical environment or radiation. In gas and UV sensors, the tetrapodal morphology offers a clear advantage over simpler particle shapes. It suppresses dense agglomeration and preserves a high accessible surface area.
Gas sensing with T-ZnO has been extensively studied. The sensing mechanism is based on resistance changes caused by the interaction of target gas molecules with chemisorbed oxygen ions on the ZnO surface. These interactions modulate the electron depletion layer and alter conductive pathways (Fig. 48a). This field was comprehensively reviewed in 201812 and significant progress has since been made, particularly in improving sensitivity and selectivity (Section 3.6.4). For example, Poschmann et al.102 coated T-ZnO with a thin ZIF-8 layer. This formed a molecular sieve that enabled highly selective hydrogen sensing even in the presence of methane. Their device achieved fast response times (1–2 s), low operating temperature (100 °C), and a reported 60-fold sensitivity enhancement. While promising, such approaches raise concerns regarding long-term stability in humid environments and the scalability of multi-step fabrication processes.
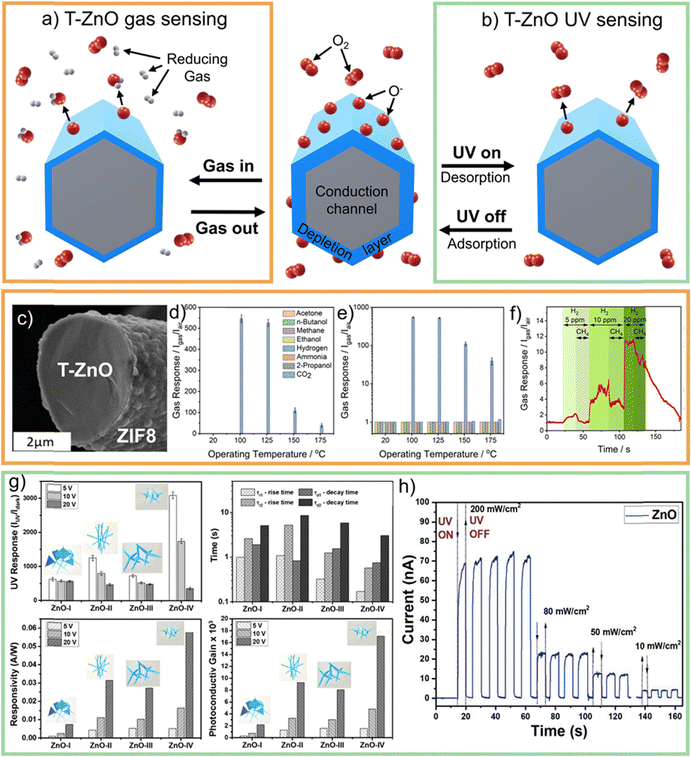 | ||
| Fig. 48 Chemoresistive sensing mechanism in T-ZnO. A cross-section of a T-ZnO arm illustrates surface depletion layer modulation. In the dark or with oxidizing gases, adsorbed oxygen ions (O2−) capture electrons, narrowing the conductive channel and increasing resistance. Conversely, (a) reducing gases or (b) UV irradiation release trapped electrons, widening the channel and lowering resistance.37,86 (c)–(f) Surface modification of T-ZnO with ZIF-8, detailing gas sensing responses to various gases across different temperatures on (d) linear and (e) logarithmic scales, alongside response curves for H2/CH4 mixtures, collectively highlighting enhanced H2 sensitivity.102 (g) UV sensitivity of gas sensor measurements for different T-ZnO morphologies and (h) dynamic response under varying UV light intensities.202 (c)–(f) Reproduced from ref. 102 with permision from American Chemical Society,102 Copyright 2023, (g)–(h) reproduced from ref. 202 with permision from Elsevier,202 Copyright 2019. | ||
In a similar vein, the combination of T-ZnO with graphene oxide185 and CuO186,187 have been reported as materials for selective hydrogen gas sensors. A further effective method involves surface modification of T-ZnO with ZnFe2O4. This has also demonstrated promising results in enhancing selectivity in gas sensing applications. For example, Mei et al.188 coated the surface of T-ZnO via a hydrothermal approach and tested the material as a gas sensor for different volatile organic compounds (VOCs) and reported a high selectivity for ethanol. Li et al.189 coated the surface of T-ZnO with a ZnFe2O4 as well, using a wet-chemical approach followed by a calcination step. Their material shows satisfactory results as a H2S sensor with a detection limit of 2 ppm at 250 °C. Next to the detection of hydrogen,102,185,186,190,191 ethanol188,192 and H2S,189 T-ZnO based gas sensors have been discussed for the detection of O2,193 NO,194 methane195,196 and different VOCs.197,198
Interestingly, these results are obtained even though the most chemically active sites, the polar {0001} facets at the tetrapod tips (Section 3.6.2), contribute only weakly to the electrical percolation pathways. These pathways are dominated by less reactive non-polar side-facet junctions. Moreover, most studies employ tetrapods with micrometer-scale arm diameters. However, Gupta et al.196 showed that significantly higher sensitivities are achieved when the arm diameter approaches the nanometer-scale depletion layer thickness, where charge transport becomes strongly surface-controlled.
Despite promising laboratory-scale results, several challenges remain critical for real-world implementation. Humidity cross-sensitivity, the frequent need for elevated operating temperatures (100–250 °C),199,200 and insufficient long-term stability testing remain recurring issues. From an outlook perspective, future research should focus less on demonstrating new analytes. Instead, it should prioritize systematic optimization of morphology, defect concentration, and network architecture, as well as strategies enabling room-temperature operation and robust signal compensation.
4.1.1.2 Chemiresistive sensors (UV). Complementing its utility in gas detection, T-ZnO is also highly effective for UV sensing. UV light below ∼375 nm excites electrons from the valence to the conduction band (Section 3.3.1), generating charge carriers that decrease resistance. Simultaneously, holes migrate to the ZnO surface, react with adsorbed oxygen, and release electrons back into the conduction band (Fig. 48b). This combines photogeneration with surface chemistry (Section 3.6.2). The high exciton binding energy of ZnO (60 meV, Section 3.2.1) enables room-temperature UV detection. Compared to spherical ZnO particles, T-ZnO exhibits superior sensitivity, response, and recovery times. This is primarily due to its highly porous, three-dimensional network, which increases accessible surface area and enhances interactions with oxygen and UV radiation.52,86,120,197,201–203 Although T-ZnO generally exhibits superior UV-sensing properties compared to spherical ZnO particles, further optimization of its morphology and size holds the potential to enhance sensor device performance. Ilickas et al.86 compared different T-ZnO sizes regarding UV sensitivity. T-ZnO arm diameters of approximately twice the LD showed the best sensing performance. This is a key parameter whose influence on charge transport was introduced in Section 3.2.3. These results agree with the findings by Postica et al.,202 who discussed the influence of different T-ZnO morphologies on UV sensitivity. They demonstrated that T-ZnO with thin and long tips and diameters close to the LD shows the best UV sensitivity (Fig. 48g and h). Via a functionalization of T-ZnO with CNTs further improvement of the UV sensing performances is possible due to an improved carrier separation at the T-ZnO/CNT interface. The superior UV-detection capabilities of T-ZnO are increasingly being validated in real-world applications (e.g., monitoring UV exposure in ruminant animals201). This demonstrates that the reliability and robustness of T-ZnO-based UV sensors have already reached a high level.
Collectively, these findings for both gas and UV sensing highlight a key design principle. The unique, high-surface-area network of T-ZnO amplifies its intrinsic semiconducting properties, making it an excellent platform for next-generation chemiresistive sensors. An additional critical design consideration, often overlooked, is the tetrapod arm diameter. Ideally, this should match the depletion layer thickness to maximize sensitivity.86,196,202
4.1.1.3 Electromechanical sensors (pressure & strain). The piezoelectric properties (Section 3.5) and mechanical robustness (Section 3.4) of T-ZnO enable its use in pressure sensors. These devices either generate a voltage proportional to applied force via the piezoelectric effect or show a change in capacitance via the capacitive effect (Fig. 49a). Although T-ZnO is intrinsically piezoelectric, the random orientation of tetrapods in a composite matrix causes the piezoelectric potentials of individual arms to largely cancel, limiting the macroscopic response. Consequently, capacitive sensors are often more effective. Here, T-ZnO is embedded in a polymer matrix (e.g., PDMS or polyvinylidene fluoride, PVDF) to form dielectric structures whose capacitance changes under mechanical force. The interlocking tetrapod network provides high compressibility and resilience for enhanced sensitivity (Fig. 49b). These T-ZnO/polymer composite sensors204 yield mechanically robust devices while maintaining high sensitivity, with detection limits as low as 1 Pa.36,51,94,205–208 These sensors have been tested for advanced technologies especially in biomedical applications. For example, Wang et al.94 presented a pressure sensor based on a T-ZnO/PDMS composite with a sensitivity of 182.5 mV N−1. This demonstrated distinct responses to objects of varying stiffness and highlighted its potential use as an end-effector component in surgical robots (Fig. 49c–f). This technology is further developed in several studies enabling detection of body movements by athletes206 or the flexion and detection of fingers, which is usable to directly translate sign language.205
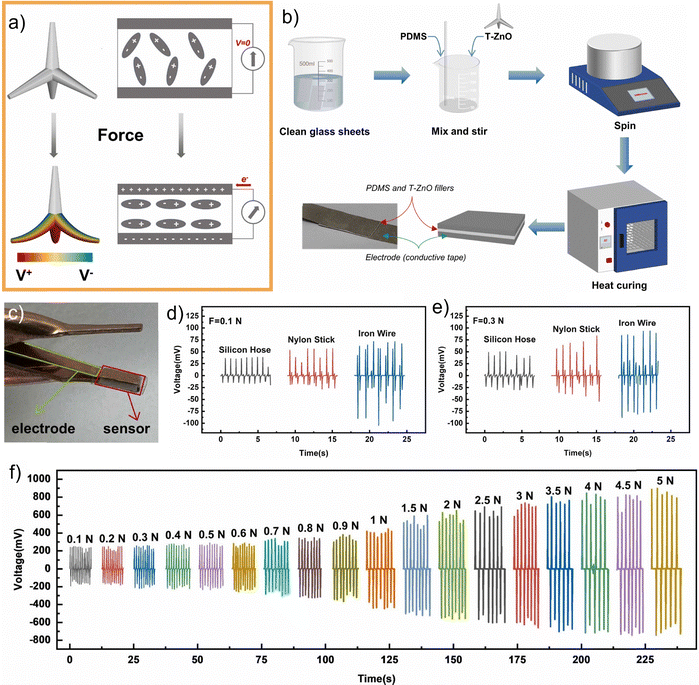 | ||
| Fig. 49 Piezocapacitive sensing mechanism in T-ZnO. (a) Schematic illustration of the piezocapacitive effect. (b) Fabrication process of a T-ZnO/PDMS thin film sensor. (c) Photograph of the T-ZnO/PDMS-based piezocapacitive device. (d) and (e) Output voltage signals when gripping different materials under applied forces of 0.1 N and 0.3 N, respectively. (f) Voltage response of the sensor to varying gripping forces from 0.1 N to 5 N. (a)–(f) Reproduced from ref. 94 with permision from Elsevier,94 Copyright 2024. | ||
The practical application of these sensors, however, requires addressing several challenges. One key issue is mechanical hysteresis in the polymer matrix, where the material does not immediately return to its initial state, potentially causing signal drift. The long-term durability and fatigue resistance under thousands of compression cycles are also critical for wearable or robotic applications. These factors are highly dependent on the interfacial adhesion between the T-ZnO filler and the polymer matrix. Moreover, as noted by Liu et al.206 these composites can exhibit cross-sensitivity to temperature and humidity, which can interfere with the pressure signal. Future work must focus on developing materials with reduced hysteresis and implementing compensation algorithms to deconvolve pressure signals from these environmental variables.
Such signal disentanglement can even enable multifunctional sensing, as demonstrated by Wang et al.,209 who reported a sensor capable of independently detecting multiple stimuli, including strain, temperature, and the contacted material. This was achieved by minimizing signal interference via machine-learning-based separation of the underlying physical effects. Furthermore, functionalization of T-ZnO (e.g., with laccase-like oxidase) enables additional sensing modalities, such as lactate concentration.207 This establishes T-ZnO as a versatile platform for pressure sensors with advanced signal-processing capabilities.
To proceed in the field of T-ZnO based pressure sensors, systematic investigations of tetrapod size, network concentration, and defect density are needed. Understanding their effects on sensor sensitivity would be highly valuable for identifying optimal T-ZnO design parameters. At present, existing studies vary widely in these and other parameters. Tetrapod sizes, for example, range from a few micrometers205,209 to several hundred micrometers.207,208 This variability makes it difficult to draw clear conclusions about their influence on performance.
4.1.1.4 Optical & electrochemical biosensors. Beyond chemiresistive and mechanical sensing, T-ZnO's unique combination of high surface area, strong optical response, and good electrical conductivity makes it an ideal scaffold for advanced biosensors. Its 3D architecture provides a stable and accessible platform for immobilizing biological recognition elements (like enzymes or antibodies), whose activity can then be read out using various signal transduction methods, primarily optical and electrochemical (Fig. 50a).
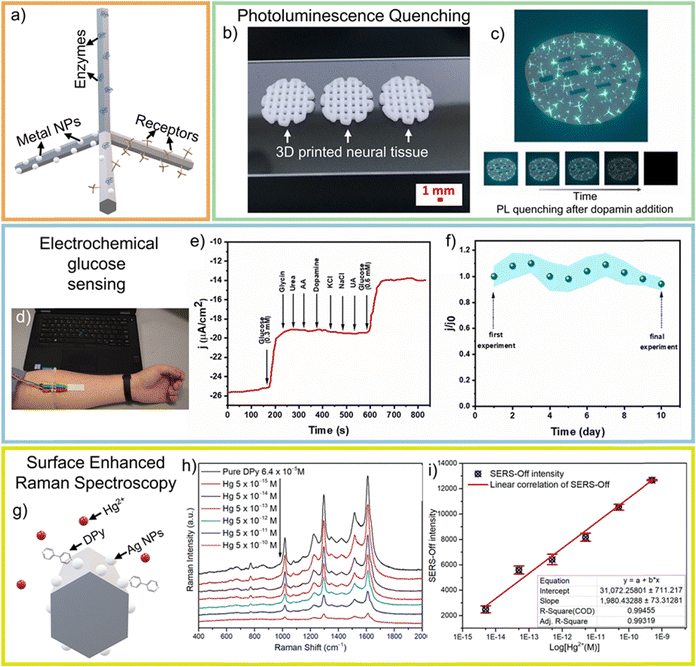 | ||
| Fig. 50 Optical & electrochemical biosensors. (a) For biosensing, T-ZnO can be surface-functionalized with various materials such as metal nanoparticles, enzymes, or other bioactive molecules. (b) Photograph of a T-ZnO-containing bioprinted scaffold. (c) PL quenching observed after dopamine addition to the scaffold.210 (d) Photograph of an electrochemical glucose sensor incorporating T-ZnO. (e) High selectivity of the sensor toward glucose. (f) Stable performance for sweat-based glucose detection over several days.213 (g) Schematic of a T-ZnO arm functionalized with Ag nanoparticles and DPy in a mercury ion environment. (h) Detection of Hg2+ ions using this composite via SERS (i) Sensitivity reaching the femtomolar range.219 (a)–(c) Reproduced from ref. 210 with permision from Elsevier,210 Copyright 2023, (d)–(f) reproduced from ref. 213 with permision from Elsevier,213 Copyright 2022, (h)–(i) reproduced from ref. 219 with permision from MDPI,219 Copyright 2021. | ||
The optical properties of T-ZnO (Section 3.3) expand the materials sensing capabilities to further sensing applications under wet conditions. In such sensing devices, detection often relies on the quenching effect (decreasing effect on the PL intensity) of target molecules on the PL of T-ZnO under UV irradiation. The high crystal quality of T-ZnO arms provides a strong and stable baseline PL signal, while their high surface area allows for a dense functionalization with bioreceptors, amplifying the quenching effect and boosting sensitivity. Fundamentally, the efficiency of this quenching process depends on the precise energy level alignment between the T-ZnO band edges and the analyte's molecular orbitals. This factor can be significantly influenced by surface defects (Section 3.1.3), which act as competing recombination pathways. For example, Li et al.210 used this mechanism to detect dopamine in a 3D-printed hydrogel containing neurons, opening avenues for creating self-monitoring, 3D-printed tissues as in-vitro models for neurological diseases. The self-monitoring 3D tissue might also become an alternative for animal testing regarding its higher degree of conformity with real human tissue compared to 2D cultures (Fig. 50b and c). The PL modifiying sensing mechanism has also shown its usability for tetracycline,107 streptavidine211 and SARS-CoV-2212 sensors. However, regarding real-world applicability, several challenges remain. In particular, small variations in synthesis conditions, defect concentration, and the degree of broken tetrapods can significantly affect luminescence properties (e.g., intensity; Section 3.3), posing challenges for the reproducible production of sensors with consistent performance.
Moreover, T-ZnO has been explored as a sensing material for glucose detection, where its interconnected network provides efficient pathways for electrochemical signal transduction.213–215 For example, Myndrul et al.213 coated a flexible electrode with MXene coated T-ZnO, which was modified with glucose oxidase and coated with a Nafion layer to fix the enzyme. The resulting sensor is flexible and can be attached to the skin (Fig. 50d–f). There it can selectively determine the glucose concentration of the sweat, showing good correlation to the glucose concentration in the blood and with a detection limit of 17 µM. This non-invasive approach could be a relief for diabetes patients. While promising, it is important to note that the physiological correlation between sweat and blood glucose is complex and remains a subject of clinical debate, representing a significant challenge for the real-world application of any sweat-based glucose sensor.216 Such electrochemical sensing mechanisms for biomarker molecules are not limited to sugars but can also be extended to other analytes such as uric acid217 or epidermal growth factor receptor 2 (HER-2),218 a key biomarker for breast cancer. Despite these initial promising results on electrochemical sensing with T-ZnO-based sensors, further studies are needed to evaluate long-term stability. Early reports indicate changes in sensor performance218 and many studies do not assess long-term effects.214,217 With improved understanding in this direction, electrochemical T-ZnO-based sensors could become a cost-effective and minimally invasive option for clinical biomarker monitoring.
Furthermore, The unique geometry of T-ZnO can be leveraged to create plasmonic “hot spots” for SERS biosensor,219–221 enabling ultra-low detection limits. Tangsuwanjinda et al.219 demonstrated this by decorating T-ZnO with Ag nanoparticles and 4,4′-dipyridyl (DPy), which allowed the system to detect Hg2+ ions in the femtomolar range (Fig. 50g–i). However, the use of this sensor is tied to the possession of a typically expensive Raman spectrometer, which may be a barrier to the widespread adoption of this technique.
Overall, T-ZnO-based biosensors represent a promising branch of T-ZnO-based sensing technologies, but they typically face challenges common to the field.222 A primary challenge is non-specific binding, where unwanted molecules from complex biological fluids (like blood) adhere to the surface, causing false signals.223 The long-term stability of the immobilized bioreceptors and the potential for Zn2+ ion leaching, which could affect both the sensor's function and local cell viability, also require careful consideration.224 Finally, developing robust methods for calibrating these sensors, especially for wearable and in-vivo applications, remains a critical step for their translation from the lab to clinical use.
T-ZnO has been explored as an active component in various optical and electronic devices.225–228 For instance, Giubileo et al.225 investigated the application of T-ZnO tips as electron-emitting elements. Their study demonstrated that T-ZnO exhibits stable electron emission with a low turn-on voltage of just 7 V. This combination of stability and low operating voltage suggests its potential utility in applications where energy efficiency and compact device design are critical. However, for practical field emission devices, the crucial next step is to evaluate the long-term operational lifetime under continuous operation, as high-field emission can lead to tip degradation.
Karthick et al.226 demonstrated that the integration of T-ZnO with a p-type silicon wafer results in a robust photodetector, where the 3D network can improve performance through enhanced light scattering and efficient charge collection pathways. This benefit, however, comes with a critical trade-off: the numerous inter-tetrapod junctions that form the network can also act as potential barriers or trap states, which may limit the device's response speed. Furthermore, PL,88,97,229,230 WGMs231(which make individual arms ideal micro-resonators for lasing), and second harmonic oscillation232 of T-ZnO are discussed in the literature,88,97,229–232 regarding their use in different optoelectronic devices.
Realizing high-quality WGM lasing, however, is critically dependent on synthesizing T-ZnO with exceptionally smooth crystallographic facets, as surface roughness can dramatically suppress the resonant modes (Section 3.3.3). While these phenomena are promising for different optoelectronic devices, the long-standing difficulty in achieving stable p-type ZnO remains a significant hurdle for developing efficient T-ZnO-based LEDs. Ultimately, a key challenge for the field is bridging the gap from demonstrating these powerful optical phenomena in single, isolated tetrapods to developing scalable fabrication methods for creating reliable, large-area devices.
4.1.3.1 Advanced wettability and coatings. The development of superhydrophobic and superhydrophilic surfaces plays a major role in materials science due to their broad applicability, including water harvesting and antifouling. T-ZnO is a versatile material for tuning surface wettability, as it can exhibit both hydrophilic and hydrophobic behavior. This switchable wettability arises from changes in the surface concentration of oxygen vacancies, which can be induced, for example, by UV irridiation or thermal treatment (Fig. 51a). T-ZnO can also increase surface roughness, reducing solid–liquid contact and thereby increasing the contact angle, which enables superhydrophobic surfaces, especially when combined with hydrophobic polymers (Section 3.6.3). The combination of tunable surface chemistry and inherent 3D microtexturing makes T-ZnO a highly effective component for advanced functional coatings.
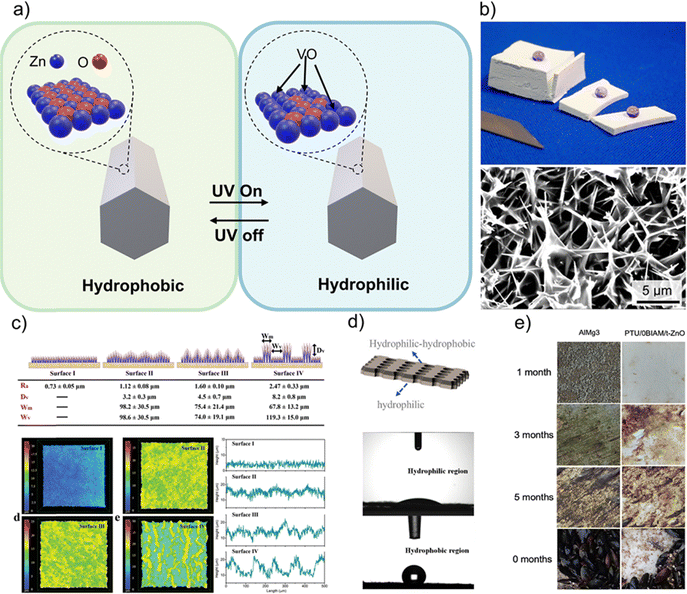 | ||
| Fig. 51 T-ZnO wettability and coatings. (a) UV irradiation enhances T-ZnO water wettability by increasing the concentration of surface oxygen vacancies (VO). (b) Superhydrophobic T-ZnO/PDMS composite and its corresponding SEM microstructure.233 (c) Tailoring of the T-ZnO/PDMS microstructure enables precise control over hydrophilicity.234 (d) Schematic of T-ZnO-incorporated Janus membranes exhibiting distinct dual-sided wettability for fog-harvesting applications.249 (e) Antifouling effects of T-ZnO-based coatings against marine biofouling.253 (b) Reproduced from ref. 233 with permision from American Chemical Society,233 Copyright 2019, (c) Reproduced from ref. 234 with permision from Wiley,234 Copyright 2022, (d) Reproduced from ref. 249 with permision from American Chemical Society,249 Copyright 2023, (e) Reproduced from ref. 253 with permision from Elsevier,253 Copyright 2023. | ||
Superhydrophobicity has been achieved in several studies utilizing T-ZnO. Typically, a polymer precursor is mixed with T-ZnO and applied to a surface, for example by spray coating. Yamauchi et al.233 presented such an approach, combining T-ZnO with PDMS to either cast a monolith or spray-coat a thin film onto a target substrate. The authors describe the resulting composite structure as analogous to a porcupine fish, where the flexible PDMS matrix mimics the skin and the rigid T-ZnO tetrapods resemble scales that create surface roughness. The resulting composite exhibits superhydrophobic behavior, with a water contact angle of 150°, which remains stable even after mechanical abrasion and bending (Fig. 51b).
Through adapting the microstructure in to a gecko-, petal- or lotus-like surface structure of a PDMS/T-ZnO composite via different coating approaches, the water contact angle is modifiable as shown by Weng et al.234 (Fig. 51c). Furthermore, the quality of T-ZnO-based superhydrophobic coatings can be monitored via their PL response after abrasion. Increased abrasion cycles cause more cracking of the T-ZnO particles, generating new surfaces and defects, which in turn increase the overall PL. This effect provides a non-destructive method for quality control, as demonstrated via T-ZnO-methacrylate copolymer composites by Liu et al.135 Additional composites exhibiting similarly pronounced superhydrophobic properties have been reported, further demonstrating the versatility and applicability of T-ZnO-based materials for the fabrication of superhydrophobic surfaces.119,235–242
Wettability is not only relevant in water-based systems. Bajpayee et al.243 developed a superhydrophobic composite coating comprising T-ZnO, amorphous silica, and fluorinated polyhedral oligomeric silsesquioxane (F-POSS). The combination of multiscale surface texturing, enabled by the T-ZnO and chemical surface functionalization with F-POSS not only renders the surface non-wettable by low surface tension liquids such as alcohols and n-hexadecane. The resulting minimization of the contact area between the liquid and the surface further effectively suppresses ice nucleation by inhibiting heterogeneous nucleation at the solid–liquid interface. Furthermore, anti-icing surfaces based on T-ZnO/PDMS composites have been reported by Weng et al.244,245
Although most studies on T-ZnO composite coatings focus on superhydrophobicity, T-ZnO can also be employed to create superhydrophilic surfaces. This has been demonstrated for a T-ZnO coating functionalized with perfluorooctanesulfonate, achieving a water contact angle of 0° and a contact angle of 160° for viscous oil. When applied to a stainless-steel mesh with micrometer-scale pores, the coating functions as an effective separation membrane, enabling the removal of residual oil from water-oil mixtures, with purified water containing less than 300 ppm of oil.178
In conclusion, T-ZnO is a versatile material for creating both superhydrophobic and superhydrophilic surfaces. Tetrapod sizes in the micrometer range from a few micrometers233,237,240 up to hundred micrometers235,236 appear sufficient for these applications. Surface homogeneity does not seem critical, as both uniform233 and non-uniform tetrapods236,238 can achieve superhydrophobicity. However, tetrapod density appears to influence hydrophilicity.236 To confirm these trends, further studies with more systematically comparable parameters are desirable. Furthermore, several challenges remain to be addressed. Although abrasion stability has been tested with promising results,233 the long-term stability of T-ZnO-based coatings is still uncertain, since the switchable hydrophilicity may become problematic if oxygen vacancy concentrations change over time, requiring repeated treatments such as UV irradiation. Yet, UV exposure could also activate the photocatalytic properties of T-ZnO, potentially degrading both the coating itself and commonly used polymer matrices. In particular, polymers such as PDMS are prone to UV-induced decomposition.246 Furthermore, many reported composites combine T-ZnO with fluorinated polymer matrices.178,235,243 While such systems demonstrate excellent performance, fluorinated polymers pose significant health and environmental risks and are subject to increasing regulatory restrictions.247 Their use should therefore be avoided. T-ZnO, however, may enable the development of coatings with comparable performance without relying on fluorinated polymers.233,234
In addition to superhydrophobic and superhydrophilic surfaces, advanced T-ZnO-based composites with switchable, heterogeneous, or patterned wettability have been investigated in several studies, enabling their use in applications such as oil–water separation and fog harvesting.
Switchable wettability can be achieved, for example, by functionalizing a T-ZnO/PDMS composite with azobenzene molecules. Upon UV irradiation, the azobenzene moieties undergo a trans-to-cis isomerization, rendering the surface hydrophilic. Subsequent exposure to visible light reverses this process, restoring the trans configuration and thus hydrophobic surface properties. The switching occurs within several minutes, with the water contact angle shifting from approximately 140° (hydrophobic) to 40° (hydrophilic) and back. This dynamic behavior enables the material to be used for selective separation processes, such as oil removal from water-oil mixtures or water removal from oil–water mixtures, depending on the desired wettability state.155,248
Furthermore, the design of surfaces with spatially heterogeneous wettability has attracted considerable interest for enabling multifunctional membranes with directionally controlled fluid transport. In this context, Janus membranes, characterized by distinctly different surface properties on each side, offer unique opportunities for applications such as fog harvesting (Fig. 51d). Liu et al.135 proposed an approach for fog harvesting using T-ZnO coatings, in which a cotton fabric was coated with a T-ZnO/poly(methyl methacrylate) (PMMA) composite. Patterned wettability was achieved through selective UV illumination. During fog harvesting experiments, water droplets nucleated and accumulated on the hydrophilic regions, while droplets on the hydrophobic areas rolled off, enabling efficient water transport. These patterned wettability fog harvesters demonstrated a water collection rate of 3.4 mg min−1 cm−2. Following a similar concept, Chenxi et al.249 synthesized Janus membranes for fog harvesting, showing high stability against UV exposure and abrasion by sand, conditions typical of dry environments such as deserts. Their membranes exhibited fog harvesting efficiencies of 28.3 mg min−1 cm−2. Li et al.250 developed a Janus membrane by spray-coating one side of a copper mesh with a T-ZnO/PDMS composite while growing Cu(OH)2 nanoneedles on both sides. The T-ZnO/PDMS-coated side exhibited hydrophobicity with a water contact angle of 153°, whereas the opposite side, composed solely of Cu(OH)2 nanoneedle arrays, was hydrophilic. This asymmetric wettability facilitated condensation on the hydrophobic side, and the resulting surface energy gradient induced a Laplace pressure-driven transport of water to the hydrophilic side, where it could be efficiently harvested at rates up to 30.8 mg min−1 cm−2. These fog-harvesting efficiencies are comparable to those of the latest high-performance fog-harvesting devices.251
Overall, T-ZnO demonstrates significant potential for applications requiring switchable, heterogeneous, or patterned wettability. While preliminary studies have shown promising performance in abrasion tests155,248 and repeated cycle,250 the long-term stability and effectiveness of the necessary complex composites remain untested. With further investigation into their durability, both intrinsic properties and microscale structural features of T-ZnO-based materials could be leveraged to address challenging wettability application requirements. These technologies may find broader applications in areas such as fog harvesting.135,249,250
Applied research on T-ZnO composite coatings is also highly relevant in the field of antifouling. Fouling presents substantial challenges, particularly in the shipping industry, where surface deterioration and increased roughness lead to higher fuel consumption and thus to elevated CO2 emissions. The surface structure and wettability of T-ZnO-based antifouling coatings reduce the adhesion of fouling organisms, resulting in decreased growth of marine organisms at the surfaces and surfaces that are easier to clean (Fig. 51e).252–254 Additionally, some antifouling coatings have demonstrated a reduction in CaCO3 crystal deposition, which commonly occurs in water pipes and can cause serious issues such as blockages and corrosion.255 These findings demonstrate that T-ZnO enables coatings capable of significantly extending duty cycles for surfaces exposed to aqueous media, from marine structures such as ship hulls252,253 and fishing nets254 to water distribution system,255 by controlling surface structure and wettability. Moreover, T-ZnO-based coatings have been shown to be biocompatible and could therefore represent an eco-friendly alternative to the copper-based biocidal coatings commonly employed in marine environments. Nonetheless, the material quantities required for large-scale applications, such as ship hulls, are considerable, and industrial synthesis capacity is only beginning to reach the necessary scale.
Aside from T-ZnO-based coatings designed to engineer surface wettability, leveraging the intrinsic wettability of T-ZnO and its unique structural features, several studies have introduced advanced coatings and composites that further exploit further properties of T-ZnO, such as its chemical reactivity and interlocking structures.256–258
For instance, Liu et al.256 developed an aluminum phosphate coating for Ti-6Al-4V alloys incorporating Al2O3-coated T-ZnO particles. The T-ZnO releases Zn2+ ions, which facilitate crosslinking within the aluminum phosphate matrix. The Al2O3 coating on T-ZnO controls the ion release rate, enabling smooth and homogeneous crosslinking. This coating process occurs at a low temperature of 70 °C, resulting in good adhesion, increased hardness, and crack-free coatings. The low processing temperature is energy-efficient and prevents phase transformations in the alloy that could lead to increased brittleness. Another strategy, presented by Brindha et al.258 used the unique tetrapodal structure of T-ZnO to develop polymer composite coatings with enhanced adhesion to Mg alloys. These coatings form effective barriers against aqueous electrolytes such as KOH, making them promising candidates for anode protection in primary Mg batteries. These findings demonstrate that coatings containing T-ZnO can exploit not only the interplay between wettability and microstructure but also other intrinsic properties, enabling the design of advanced high-performance coatings.
4.1.3.2 Particle-based systems for decontaminant adsorption. As discussed in the theoretical section (Section 3.6), T-ZnO exhibits noteworthy surface properties. In particular, individual tetrapods and T-ZnO networks possess a high and readily accessible specific surface area, which can be easily modified. These characteristics make T-ZnO highly suitable for decontamination applications, where contaminants are removed from a medium via adsorption. To enhance selectivity or maximize adsorption capacity, T-ZnO is frequently incorporated into composite materials.
In the literature, the main focus is on the development of T-ZnO-based adsorbents for the removal of heavy metal ions179,259,260 or oil.261 For example Sharma et al.179 synthesized a composite containing T-ZnO and activated carbon, which demonstrated removal rates of Cr(VI) ions of up to 97%. In another approach, T-ZnO was coated with Fe2O3 nanorods and PDMS to form a nanocomposite capable of efficiently removing oil from wastewater. For instance, diesel removal rate is up to 96% and adsorption capacity 1135 mg g−1. Through the magnetic Fe2O3 nanorods the composite can easily be removed from the cleaned water after adsorption of the oil.261
Although T-ZnO shows promising potential for wastewater decontamination, primarily due to its large and tunable surface area that enables efficient adsorption of contaminants, several drawbacks must be considered. ZnO itself is toxic to aquatic organisms, and if T-ZnO particles are not fully recovered after treatment, they may increase rather than reduce water toxicity. In acidic media, zinc ions can leach into the water, further contributing to ecotoxicity.262,263 In addition, compared to established adsorbents such as activated carbon or zeolites,264 T-ZnO remains relatively costly, which currently limits its broader applicability.
In contrast, T-ZnO microparticles hold significant promise for biomedical applications. Due to their size, being larger than typical cells, they are typically not internalized via endocytosis, yet they still retain key nanoscale properties for medical applications. Moreover, compared to conventional ZnO nanoparticles with diameters of just a few nanometers, T-ZnO microparticles have a substantial, yet much lower, surface-to-volume ratio, which may reduce the release of Zn2+ ions. This characteristic may contribute to a lower cytotoxicity, making them a safer alternative for biomedical use.58,182,268,269 Moreover, T-ZnO structures circumvent common drawbacks associated with conventional nanomaterial morphologies, such as nanoparticles or nanorods, including the tendency to agglomerate.12
As a result, research into the biomedical applications of T-ZnO has expanded significantly, culminating in the establishment of Phi-Stone AG, a company that produces good manufacturing practice (GMP)-certified, pharmaceutical-grade T-ZnO microparticles for medical applications. A notable commercial product is Afinovir, a wound protection gel that incorporates T-ZnO.270
4.1.4.1 Antibacterial and antiviral applicationsv. Several studies have reported the antibacterial and antiviral properties of T-ZnO. Among these, its antibacterial activity is the most extensively documented. Nevertheless, the underlying mechanism remains a subject of ongoing investigation. It is commonly proposed that the antibacterial effect arises either from the release of Zn2+ ions and/or the generation of ROS.268,271 T-ZnO has been evaluated against a variety of bacterial strains, consistently demonstrating superior antibacterial performance (Fig. 52a). For instance, Collares et al.272 incorporated T-ZnO as a filler in dental resin sealers and observed effective antibacterial activity against Enterococcus faecalis. Furthermore, the combination of T-ZnO with thin film based surface acoustic waves showed increased antibacterial properties.273 Additional studies have further substantiated the antibacterial potential of T-ZnO.271,274–281
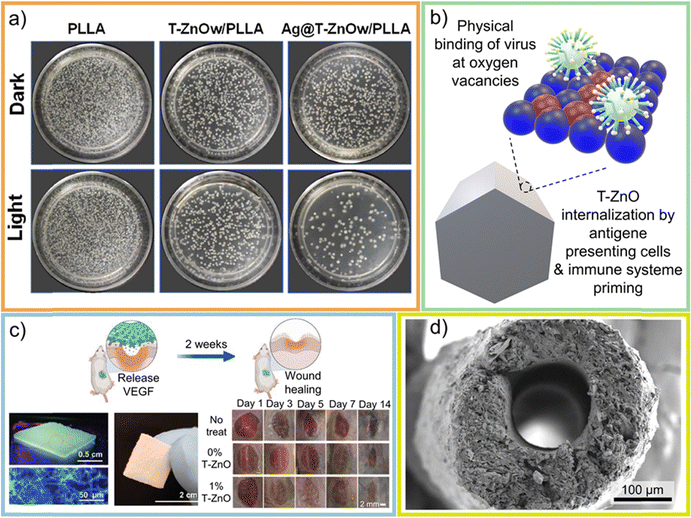 | ||
| Fig. 52 (a) Antibacterial activity of T-ZnO/Ag NP/poly-L-lactic acid composites against E. coli on agar plates.276 (b) Schematic of the proposed virus-trapping mechanism at oxygen vacancies on the T-ZnO surface, followed by internalization by antigen-presenting cells and subsequent immune system priming.284 (c) Smart wound scaffold composed of T-ZnO, VEGF, and a hydrogel matrix, with in vivo testing on mouse wounds.268 (d) SEM image of a glaucoma stent containing T-ZnO.170 (a) Reproduced from ref. 276 with permision from Elsevier,276 Copyright 2022, (c) Reproduced from ref. 268 with permision from Wiley,268 Copyright 2021, (d) Reproduced from ref. 170 with permision from American Chemical Society,170 Copyright 2023. | ||
In addition to its antibacterial properties, T-ZnO also exhibits promising antiviral activity. Beyond the commonly discussed mechanisms, such as the release of Zn2+ ions and the generation of ROS, further antiviral mechanisms have been proposed. For instance, studies have shown that infections caused by Hepatitis E and Hepatitis C viruses can be inhibited by ZnO nanoparticles and T-ZnO structures.282 This occurs through a dual mechanism. First, physical binding and entrapment of the viruses at the material surface prevents viral entry into host cells. Second, the material inhibits viral replication. However, compared to conventional ZnO nanoparticles, T-ZnO has demonstrated superior antiviral efficacy, which may be attributed to its unique morphology and the distinct crystal structures and surface facets.282 The virus-trapping capability is believed to be driven by oxygen vacancies at the surface of T-ZnO.283 Additionally, viruses bound to T-ZnO can be internalized by antigen-presenting cells, thereby priming the immune system for a more effective response (Fig. 52b).284 This antiviral activity makes T-ZnO a promising candidate not only for direct therapeutic applications but also for the development of antiviral surface coatings. Such coatings have been successfully demonstrated, for example, to reduce the infectivity of SARS-CoV-2.285
Antibacterial and antiviral applications represent one of the most advanced research areas for T-ZnO. Particularly noteworthy is its ability to entrap viruses at the T-ZnO particle surface283 and promote their exposure to antigen-presenting cells, thereby priming the immune system.284 These properties make T-ZnO highly attractive for tackling of viral diseases like Hepatitis E and C.282 Intensive research in this field has already led to the development of first market-ready pharmaceutical products. Further long-term studies evaluating T-ZnO's effectiveness against additional diseases could further enhance the medical relevance of T-ZnO.
4.1.4.2 T-ZnO containing composites for biomedical applications. Not only the direct antibacterial, antiviral, or antifungal effects of T-ZnO are relevant for biomedical applications. Other properties, such as its mechanical stability and PL, are also of great interest. In many cases, it is the combination of multiple T-ZnO properties within composites that leads to highly effective biomedical products.
For example, Siebert et al.268 developed a smart wound dressing platform by incorporating H2O2-modified and vascular endothelial growth factor (VEGF)-decorated T-ZnO into 3D-printed gelatin methacryloyl (GelMA) hydrogel wound patches. In this multifunctional system, T-ZnO plays several crucial roles. First, its intrinsic antibacterial properties reduce bacterial load at the wound site. Second, due to its large and accessible surface area, T-ZnO offers binding sites for bioactive molecules such as VEGF, which, in this design, can be released in a light-triggered manner within the optical spectrum (Fig. 52c). Additionally, the incorporation of T-ZnO as a filler material enhances the mechanical integrity of the hydrogel scaffold, contributing to the structural robustness of the wound patch. T-ZnO has also demonstrated its potential in other advanced wound dressing platforms, e.g. in a composite with alginate.274 Furthermore, T-ZnO has been integrated with PDMS for the manufacturing of stents used in minimally invasive glaucoma surgery to effectively reduce intraocular pressure. Compared to stents composed solely of PDMS, the composite stents exhibit an increased elastic modulus while maintaining sufficient flexibility to conform to the curvature of the eye (Fig. 52d). Moreover, the incorporation of T-ZnO reduces fibroblast viability, leading to antifibrotic properties upon implantation, likely due to the combined effects of surface roughness and the release of Zn2+ ions.170,286,287
Altogether, the combination of T-ZnO with other materials for biomedical applications has been explored through a variety of approaches. In these studies, multiple intrinsic properties of T-ZnO are often leveraged simultaneously. Most commonly, its inherent antibacterial activity is coupled with either its surface modifiability or its favorable mechanical properties.170,268,274,288 Given the commercial availability of pharmaceutical-grade T-ZnO, the reported findings hold significant promise and are of considerable interest for further research and development.
4.1.5.1 Thermal energy storage and batteries. Energy storage is one of the central challenges in the global transition from a fossil-based economy to one relying on renewable energy sources such as wind, solar, and hydropower. Unlike conventional fossil-fuel power plants that provide a stable and continuous energy output, renewable energy systems are inherently intermittent, with their output fluctuating according to diurnal cycles, weather conditions, and seasonal variations. As a result, robust and scalable energy storage solutions are essential to bridge the gap between energy supply and demand, both in the short and long term. These solutions range from widely adopted technologies like batteries to less conventional approaches such as thermal energy storage. T-ZnO has been investigated in several of these systems and has demonstrated the potential to enhance their performance at the laboratory scale.
One promising strategy to enhance the performance of energy storage systems is the integration of T-ZnO networks into phase change material (PCM) matrices.48,289–291 For example, Balasubramanian et al.48 utilized paraffin wax as an organic PCM. This material is capable of storing substantial amounts of thermal energy during its phase transition from solid to liquid and releasing it upon solidification. However, the practical application of paraffin wax is limited by its inherently low thermal conductivity. By incorporating T-ZnO structures into the paraffin matrix, the thermal conductivity was improved by up to 94% without compromising the material's latent heat capacity. This significant enhancement enables more efficient solar thermal energy harvesting and improved thermal management.
Moreover, T-ZnO structures have shown utility beyond organic PCMs. Zhu et al.289 demonstrated that also in inorganic systems, such as CaCO3-based PCMs, the addition of T-ZnO (at a mole ratio of just 5%) can increase thermal conductivity by 17.8%. This improvement is particularly beneficial at high heating rates, where enhanced thermal transport directly accelerates reaction kinetics. These findings highlight the potential of T-ZnO templated networks to improve both organic and inorganic PCM systems, broadening their applicability in thermal energy storage technologies.
In addition to its role in thermal energy storage, T-ZnO has also demonstrated potential in the field of electrochemical energy storage. Chang et al.292 investigated the use of T-ZnO as a coating for graphite anodes in lithium-ion batteries. Safety remains a critical issue in lithium-ion battery technology, with internal short circuits posing a major risk. These can initiate localized heating, triggering a chain of exothermic reactions that lead to a phenomenon known as thermal runaway. By applying a T-ZnO coating to the graphite electrode, direct contact between the anode and cathode can be mitigated, thereby reducing the likelihood of internal short circuits. The unique open-porous architecture of the T-ZnO network provides thermal stability while maintaining efficient ion diffusion pathways. Unlike dense nanoscale ZnO coatings such as short rods, the tetrapodal structure minimizes obstruction to lithium-ion transport. This approach substantially improves the safety of lithium-ion batteries, without compromising ion transport due to the open porous architecture of the T-ZnO network.
To conclude, in energy storage, thermal systems as well as batteries, T-ZnO is not typically employed as a direct storage material. Instead, its incorporation into existing systems can significantly enhance performance or safety. This improvement arises mainly from the combination of its high thermal conductivity and its ability to form highly porous networks, which facilitate or enable efficient transfer processes such as heat and ion transport.48,292 However, for thermal energy storage, the transition of T-ZnO into real-life applications would require large amounts of low-cost material, with industrial production just starting to scale up. In batteries, by contrast, the potential benefits must be carefully balanced with the complex electrochemistry of these systems, where ZnO may introduce additional effects that still need systematic investigation. However, another major potential in the field of electrochemical energy storage lies in the use of T-ZnO as a sacrificial template to fabricate hierarchically structured electrodes. Such architectures can significantly improve electrode performance by combining large accessible surface areas with efficient transport pathways, as discussed in Section 4.3.2.4.
4.1.5.2 (Photo-)catalysis. ZnO absorbs light in the UV region due to its band gap of approximately 3.2 eV (see Section 3.3.1). It also exhibits a large exciton binding energy of 60 meV (see Section 3.2.1) and is known for its photocatalytic activity. Combined with its chemical stability, radiation resistance, relative abundance, and low toxicity, ZnO has become one of the most extensively studied photocatalysts, with a wide range of nano- and microstructured morphologies explored. Among these, the tetrapod morphology has demonstrated superior photocatalytic performance in various studies.13,293–296 This is attributed to a lower concentration of nonradiative defects, minimizing energy losses, a high accessible specific surface area and reduced electron–hole recombination facilitated by abundant surface states. As the photocatalytic properties of T-ZnO have been comprehensively reviewed in a recent publication by Pujara et al.,13 they will not be discussed in further detail here.
An aspect of T-ZnO not addressed in the review by Pujara et al.13 is its unique capability to form interconnected networks that serve as sacrificial templates for the synthesis of novel materials. This templating strategy enables the fabrication of photocatalytically active materials with complex, hierarchically porous architectures. Such structures can enhance photocatalytic efficiency by providing a high surface area, improved mass transport, and better light-harvesting capabilities, features particularly advantageous for gas-phase and high-throughput catalytic applications (see Section 4.3.2.4).
Beyond photocatalysis, T-ZnO has also been investigated in several direct catalytic applications. In these contexts, T-ZnO typically serves as a support material, for instance for metallic nanoparticles. Compared to other ZnO-based substrates, T-ZnO often demonstrates enhanced catalytic performance, likely due to specific interactions between the catalyst and the unique surface properties of the T-ZnO structure.297–299
4.2 T-ZnO as active structural material
Beyond applications that leverage its intrinsic properties, a ground-breaking class of technologies utilizes T-ZnO primarily for its unique 3D shape. The tetrapods ability to form robust, mechanically interlocked networks (achieving three-dimensional percolation with a low filling factor) makes the architecture itself the key functional feature (Section 3.4.2). This structure is exploited in two main ways. First, it can serve as a permanent structural backbone within a composite material. Second, it acts as a removable, sacrificial template to architect entirely new classes of materials from the micro to the macro scale. The latter will be discussed in a Section 4.3.To summarize T-ZnO acts as more than a simple filler: its tetrapodal geometry enables strong mechanical interlocking, improves adhesion and enhances matrix reinforcement. In addition, it supports the dispersion of other nanofillers like CNTs, fostering well-connected networks and thus enabling multifunctional, high-performance composites.300–305 Nevertheless, the limited lab- and small-scale industrial production of T-ZnO composites still hinders real-world applications. This is mainly due to the large T-ZnO loadings required in composites and the currently high, non-competitive cost of T-ZnO for fillers.
To overcome this obstacle several approaches using T-ZnO in composite shielding materials are proposed.115,310–314 These methods leverage the high surface area (Section 3.6.1) and amenability to surface functionalization (Section 3.6.4) of the T-ZnO network. For example, Xu et al.115 mixed electroless silver plated T-ZnO with Fe3O4 coated rGO in a water based polyurethane emulsion, which forms a gradient thin film during evaporation of the water, because the Ag coated T-ZnO is denser then the Fe3O4 coated reduced Graphene Oxide. This gradient is responsible for the material's excellent microwave shielding performance, which works as follows: the microwaves enter the material through the Fe3O4@rGO-rich side, which has low reflectivity. There, the microwaves are attenuated through magnetic and dielectric losses. The conductive Ag@T-ZnO-rich side has high reflectivity and reflects the microwaves back into the Fe3O4@rGO-rich side, where they are attenuated again. With this approach the authors achieve a shielding effectiveness of up to 87 dB in the X-band with the shielding effectiveness contributing mainly from absorptive mechanisms and just 2.4 dB contributing from reflection.115 Further developments involve composites of T-ZnO with carbon cloth as a broadband absorbers,310 or the use of T-ZnO as a sacrificial template to synthesize a lightweight and absorbing aeromaterial based on a carbon backbone decorated with MnO2 and poly aniline311 or a combination of PNIPAM hydrogel with MXenes and silver nanowires, which allows switchable shielding performance due to stimuli responsive behaviour of the hydrogel.312
In conclusion, T-ZnO serves as a versatile structural backbone in EMI shielding composites, enabling high absorption and efficient reflection management through its 3D network architecture, large surface area, and functionalizability. The published composites surpass shielding effectiveness above 40 dB, which is suitable for most applications.315 Although recently reported porous materials with similarly complex synthesis achieve shielding effectiveness up to 109 dB,316 T-ZnO-based composites exhibit lower absolute shielding. However, their tunable surface properties open opportunities for designing complex, lightweight shielding materials with enhanced or even switchable capabilities, representing promising directions for future research.
4.3 T-ZnO as sacrificial template for novel architectures
The most innovative application of T-ZnO's structure, is its use as a sacrificial template. This approach does not leverage the intrinsic properties of ZnO, but its unique morphology to architect entirely new materials. A template is a material that prescribes the shape of (nano-)material growth or agglomeration. A template-based synthesis approach consists of three steps: (i) template preparation, (ii) template-guided synthesis/agglomeration of the target molecule/material in the desired shape and (iii) removal of the template structure. There is a huge variety of different template synthesis approaches.317 T-ZnO is particularly well-suited as a template to prescribe the structure for a multitude of different advanced materials, because of its unusual shape, its ability to form highly porous and accessible networks as described in Section 3.6.1, its insolubility in many environments (except acidic environments with pH < 6), which makes it easily removable via acids, its rather low production costs and its low toxicity.12,318The seminal example of aeromaterials is “aerographite”, the first structure described in the literature, which is synthesized via a T-ZnO templating approach. Aerographite is synthesized following a dry-chemical CVD approach, in which a thin layer of graphite is deposited at the surface of a T-ZnO network. The hydrogen present in the reaction atmosphere is capable of reducing the ZnO to Zn, which evaporates at elevated temperatures and allows a simultaneous template removal. At its publication date in 2012 aerographite was the most lightweight material on earth with a density of 0.2 mg cm−3, illustrating that extreme material properties are possible by using an inherently low-density, high-porosity T-ZnO network (Section 3.6.1) as a template structure.8 Since the discovery of aerographite, numerous CVD-based approaches for synthesizing diverse aeromaterials have been reported. Building on this, a related technique known as hybrid vapor phase epitaxy (HVPE)319 has been employed to grow a wide range of thin films directly on T-ZnO templates. This method also enables subsequent aerofication, expanding the versatility and material diversity of aeromaterial fabrication Table 14. A review article focusing on the gas-phase synthesis of aeromaterials, particularly semiconducting aeromaterials, was published in 2025.320
| Material | type | Synthesis approach | Application (scenarios) | Author (pulication date) |
|---|---|---|---|---|
| Graphite | Aeromaterial | CVD | Supercondensator | Mecklenburg et al. (2012)8 |
| Graphite + GaN | Aeromaterial | CVD + HVPE | Sensors | Schuchardt et al. (2015)319 |
| Graphite + epoxy | Channel | CVD + infiltration | Conductive composite | Chandrasekaran et al. (2016)331 |
| Graphite + peptides + PEG | Aeromaterials | CVD + infiltration | Cell growth | Lamprecht et al. (2016)367 |
| Graphite | Aeromaterial | CVD | Gas sensor | Lupan et al. (2016)362 |
| Silicon | Aeromaterial | Plasma-enhanced CVD | Gas sensor | Hölken et al. (2016)365 |
| Graphite + nano T-ZnO | Aeromaterial | CVD + sputtering | Optoelectronics | Tiginyanu et al. (2016)143 |
| Graphite + epoxy | Channel | CVD + infiltration | Pressure sensor | Garlof et al. (2016)332 |
| Graphite + epoxy | Aeromaterial + Channel | CVD + infiltration | Electronics | Garlof et al. (2017)333 |
| Graphite | Aeromaterial | CVD | Supercapacitor | Parlak et al. (2017)368 |
| Graphite | Aeromaterial | CVD | Theoretical | Meija et al. (2017)163 |
| Carbon + diamond + SnO2 | Aeromaterial | Hot-filament chemical vapor deposition (HFCVD) | Electro wettability | Silva et al. (2017)369 |
| Graphite | Aeromaterial | CVD | Gas sensing | Lupan et al. (2017)364 |
| MWCNT | Aeromaterial + Channel | Infiltration | Electronics | Schütt et al. (2017)321 |
| Graphite + Si/SiOx NP | Aeromaterial | CVD + plasma jet | Biomedical | Daria et al. (2018)370 |
| Graphite + InP | Aeromaterial | CVD + HVPE | Strain sensors | Plesco et al. (2018)363 |
| MWCNT + hydroxyapatite + SiO2 | Aeromaterial | Infiltration | Cell growth | Taale et al. (2018)346 |
| GaN | Aeromaterials | HVPE | Electronics | Tiginyanu et al. (2019)371 |
| Graphite + CdTe | Aeromaterial | CVD | Gas sensing | Strobel et al. (2019)372 |
| MWCNT, graphite, graphene | Aeromaterial | Infiltration, CVD | Cell growth | Taale et al. (2019)347 |
| Carbon | Aeromaterial | CVD | Lithium-sulfur batteries | Yang et al. (2019)354 |
| GaN | Aeromaterial | HVPE | Pressure sensor | Dragoman et al. (2019)360 |
| Graphite | Aeromaterial | CVD | Theoretical | Marx et al. (2019)373 |
| MWCNT | Aeromaterial | Infiltration | Electronics | Pöhls et al. (2019)168 |
| Graphite + hydroxyapatite | Aeromaterial | CVD + electrophoresis | Cell growth | Taale et al. (2019)345 |
| Graphene, graphene oxide, reduced graphene oxide | Aeromaterial | Infiltration | Electronics | Rasch et al. (2019)18 |
| Carbon + ZIF-8 | Aeromaterial | Wet-chemical growth + pyrolysis | Supercapacitors | Yuksel et al. (2020)85 |
| Carbon + polyaniline | Aeromaterial | Carbonization + polymerization | EMI | Li et al. (2020)311 |
| Hexagonal BN | Aeromaterial | CVD | Laser light scattering | Schütt et al. (2020)167 |
| PDMS | Channel | Infiltration | Drug delivery | Rasch et al. (2020)327 |
| Ga2O3 | Aeromaterial | HVPE | EM transparency | Braniste et al. (2020)374 |
| ZnS | Aeromaterial | HVPE | Switchable hydrophilicity | Plesco et al. (2020)343 |
| MWCNT + sulfur | Aeromaterial | Infiltration + vapor infiltration | Lithium sulfur batteries | Cavers et al. (2020)355 |
| Graphene | Aeromaterial | Infiltration | Theoretical | Rezvani et al. (2021)375 |
| Ga2O3 + Pt/Au | Aeromaterial | HVPE | Photocatalysis | Plesco et al. (2021)376 |
| Hydrogel + graphene | Channel | Infiltration + crosslinking | Conductive hydrogel | Arndt et al. (2021)334 |
| GaN | Aeromaterial | HVPE | Actuation | Braniste (2021)324 |
| Graphene, reduced graphene oxide | Aeromaterial | Infiltration | Electrically powered repeatable air explosions (EPRAE) (actuation) | Schütt et al. (2021)336 |
| Hydrogel | Channel | Infiltration + crosslinking | Cell migration | Siemsen et al. (2021)328 |
| Hydrogel (PNIPAM) | Channel | Infiltration + crosslinking | Actuation | Spratte et al. (2022)165 |
| Graphene oxide | Aeromaterial | Infiltration | Cell growth | Wang et al. (2022)349 |
| PDMS | Channel | Infiltration | Drug delivery | Hauck et al. (2022)330 |
| MoS2 | Aeromaterial | Hydrothermal | Potassium batteries | Ma et al. (2022)377 |
| TiO2 | Aeromaterial | HVPE | Photocatalysis | Ciobanu et al. (2022)358 |
| Partially reduced graphene oxide | Channel (Membrane) | Vacuum filtration | Pressure sensor | Ren et al. (2023)361 |
| Ga2O3/ZnGa2O4 | Aeromaterial | HVPE | Lithium batteries | Wolff et al. (2023)357 |
| (Poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) + polytetrafluoroethylene (PTFE)) | Aeromaterial | Infiltration + initiated CVD | Strain invariant conductor | Barg et al. (2023)344 |
| Hydrogel (PNIPAM) + graphene | Channel | Infiltration + crosslinking | Soft actuation | Hauck et al. (2023)335 |
| Graphene | Aeromaterial | Infiltration | Air filtration | Reimers et al. (2023)323 |
| SiO2 + reduced graphene oxide | Aeromaterial | Wet chemical growth + infiltration | Light driven actuators | Saure et al. (2023)19 |
| MXene | Aeromaterial | Infiltration | Supercapacitors | Spurling et al. (2024)353 |
| CNT | Aeromaterial | Infiltration | Underwater thermoacoustic | Liu et al. (2024)166 |
| Ce1−xZnxO2−δ | Aeromaterial | Wet-chemical growth | Photocatalysis | Lumma et al. (2024)20 |
| SiO2 + CsPbBr3 | Aeromaterial | Wet-chemical growth | Laser light scattering + light conversion | Saure et al. (2024)322 |
| Alginate | Channel | Infiltration + crosslinking | Tissue engineering | Li et al. (2024)329 |
| ZnS | Aeromaterial | HVPE | Physical vapor transport | Ursaki et al. (2024)378 |
| GaN | Aeromaterial | HVPE | Photocatalysis | Ciobanu et al. (2024)379 |
| TiO2 | Aeromaterial | HVPE | Photocatalysis | Ciobanu et al. (2024)325 |
| Graphene | Aeromaterial | Infiltration | Actuation | Reimers et al. (2025)326 |
| TiO2 | Aeromaterial | ALD | UV Photodetector | Nicolaescu et al. (2025)366 |
The synthesis of ultralightweight aeromaterials is not limited to gas phase processes, which can generate different homogeneous thin films at the T-ZnO template, but are technically rather complex setups. It is also possible to wet-chemically coat T-ZnO with a thin film of a target material. This can be done either by infiltrating nanomaterial dispersions into a T-ZnO network or by growing a thin film from solution at the surface of the T-ZnO. The infiltration approach was first published in 2017 by Schütt et al.321 A comprehensive explanation of the infiltration method is provided by Rasch et al.18 Usually a 1D or 2D nanomaterial dispersion containing e.g. exfoliated graphene, graphene oxide, MXenes, MoS2 or CNTs is infiltrated into the highly accessible pores of a T-ZnO network (Section 3.6.1). Through evaporating the solvent, a thin layer of nanomaterial deposits at the surface of the T-ZnO. This simple infiltration procedure can be repeated to increase the layer thickness of the nanomaterial. The direct wet-chemical growth of a material at the surface of T-ZnO was first published in 2023.19 Especially the growth of SiOx films at the surface of T-ZnO has been investigated in several publications, because it is simple to achieve compared to other transparent and light scattering aeromaterials e.g. Aero-hBN.19,322 The wet-chemical approach (infiltration; direct growth) allows to synthesize samples with several centimetre side length, which enables use cases with a demand for large volumes, e.g. air filters.323
A key advantage of the T-ZnO template approach, compared to the synthesis of other ultralightweight structures like aerogels, is the ease with which composite aeromaterials can be created. This is due to the fact that different nanomaterials can be introduced or synthesized step by step on the mechanically robust (Section 3.4) and chemically amenable surface of the T-ZnO network.19,20 In conventional aerogel synthesis, all nanomaterials are typically introduced simultaneously, which frequently complicates or even precludes the process, as their incorporation may require incompatible reaction conditions.
To summarize the synthesis of aeromaterials is methodologically diverse. The thin-film formation on the T-ZnO scaffold has been realized through three primary routes: gas-phase deposition techniques like CVD or HVPE, the infiltration of pre-synthesized nanomaterial dispersions (e.g., graphene oxide, MXenes, CNTs, etc.), or direct wet-chemical growth from precursor solutions. Gas-phase methods were the first to be reported and remain a powerful tool for discovering novel aeromaterials with high-quality, homogeneous coatings.8,319,324,325 However, their high synthetic complexity (often requiring vacuum systems and high temperatures) limits scalability in terms of both sample size and production quantity. In contrast, wet-chemical approaches are generally less demanding and more amenable to upscaling, making them promising for applications requiring larger material volumes.323,326
In addition to the template-guided synthesis of aeromaterials based on T-ZnO network structures, another important class of materials can be structurally defined using these templates, so-called channel structures. In this approach, the interconnected pores of the T-ZnO network are fully infiltrated with a matrix material, such as a polymer (e.g., PDMS)327 or a hydrogel (e.g., PNIPAM).165 After complete infiltration, the T-ZnO template is selectively removed, leaving behind a continuous matrix embedded with a network of interconnected microchannels that mirror the original T-ZnO architecture.165,327–330
Moreover, it is also possible to synthesize an aeromaterial and subsequently infiltrate its entire pore structure with a matrix material, such as epoxy. This process results in a continuous matrix embedded with a three-dimensional thin-film network replicating the geometry of the aeromaterial. In this way, percolating networks with ultra-low filler loadings can be achieved. This combines mechanical integrity with functional properties such as electrical conductivity or thermal management.331–333
An additional approach involves coating the T-ZnO network with a thin film of another material prior to infiltrating its pores with a matrix material. After infiltration, the T-ZnO template can be removed, resulting in a three-dimensional channel system with functionalized or coated channel walls. This method enables the fabrication of complex hierarchical structures, where the coating imparts additional chemical, electrical, or mechanical functionality to the internal surfaces of the channel network.321,334,335
4.3.2.1 Structural porosity and transport. First, the structure of aeromaterials is highly open-porous, characterized by a network of macropores that is typically interconnected throughout the entire structure. This unique open porosity allows for the rapid transport of gases or liquids between the internal pore volume of the aeromaterial and the surrounding environment. Compared to other low-density structures, such as aerogels which often contain closed or rather closed pore architectures, this degree of openness is untypical. This structural openness is a key element for many specific applications, such as actuation and tissue growth scaffolds, where mass transport is critical.336
Only a few materials exhibit structures comparable to T-ZnO-derived aeromaterials. The closest analogues are materials derived from open-porous melamine foams (e.g., Basotect). These melamine foams are inexpensive and industrially mass-produced. However, their use as templates for lightweight structures has several significant drawbacks. In particular, the removal of the melamine backbone is challenging; unlike T-ZnO, it is not soluble under mild conditions and tends to melt before decomposing, which typically destroys the fragile three-dimensional thin films deposited on the structure. A commonly employed alternative is to first carbonize the melamine foam, deposit the thin film, and subsequently remove the carbonized backbone by oxidation.337 The harsh conditions required for this backbone removal limit the use of melamine foams as templates to only a few aeromaterial classes, such as SiO2-based systems, and hinder their applicability to others, including carbon-based aeromaterials which would burn away alongside the template.
4.3.2.2 Mechanical properties and stability. Beyond their structural similarities, aeromaterials typically exhibit low absolute mechanical strength. However, when normalized to their extremely low density, their mechanical performance is often remarkable and has been rigorously tested in several studies.8,18,321 This behavior can likely be attributed to a specific buckling-hinge mechanism observed in the nodes of the hollow tetrapods.163 Mechanical stress remains a serious threat to the structural integrity of these materials, as most aeromaterials can be destroyed under direct compression. Nevertheless, they are sufficiently stable to be carefully integrated into devices and can withstand notable pneumatic pressures, such as those generated by gas streams. This robustness makes them attractive for applications including air filtration,323 gas-phase photocatalysis,20 and actuators.326,336 Furthermore, the shelf life of typical aeromaterials (whether carbon-based, semiconducting, or SiO2-based) under standard conditions is long, extending over years to decades. In addition, exceptional application-related cycle stability against temperature and heat has been demonstrated over thousands of cycles.336
4.3.2.3 Environmental impact and safety. The environmental impact of degraded or destroyed aeromaterials has not yet been investigated in detail. However, this impact is expected to depend strongly on both the constituent materials and the distinct microstructure of the aeromaterials. With respect to material composition: (i) aeromaterials are extremely lightweight, resulting in a very low total material mass released into the environment, which reduces potential impact particularly when toxic components are present; and (ii) a large proportion of investigated aeromaterials are carbon-based and, in principle, biodegradable. Regarding the microstructure, aeromaterials are typically readily destroyed by (i) mechanical stress or (ii) heat. Nevertheless, the environmental and biological impact of aeromaterial fragments, which retain microrod-like structures, has not yet been assessed. We therefore strongly recommend further scientific investigation, particularly concerning the potential risks and consequences of the inhalation of aeromaterial fragments.
4.3.2.4 Scalability and commercialization. To date, most aeromaterials have been synthesized only in laboratory-scale quantities. Nevertheless, initial studies have demonstrated the fabrication of large area aeromaterial plates, indicating the feasibility of upscaling. This upscalability is closely linked to increasing supplies from T-ZnO manufacturers, such as the Phi-Stone AG company.270 An important advantage is that the most extensively studied carbon-based aeromaterials do not depend on critical or costly resources such as rare earth elements or noble metals. Despite this, the overall production process, including T-ZnO synthesis, template construction, template coating, and template removal, remains highly specialized and complex. Consequently, aeromaterials are likely to remain relatively expensive. This makes them most suitable for applications where: (i) only small amounts are required, (ii) long-term use is feasible, and (iii) the aeromaterial enables significantly superior performance compared with conventional approaches. Despite these challenges, a company named Aero Materials GmbH initiated the commercialization of aeromaterials this year.338
4.3.2.5 Channel networks. Compared to aeromaterials, channel networks derived from T-ZnO exhibit a relatively dense, matrix-dominated structure. They consist of a matrix material permeated by interconnected, tube-shaped pores, with the pore volume typically accounting for only 5–10% of the total volume. Nevertheless, even this volumetrically small pore fraction can significantly alter material properties, for example by strongly affecting flow behavior in hydrogels.335 Generating such a well-percolating pore network within a dense matrix while maintaining such a high solid volume fraction is challenging, and only a few comparable manufacturing approaches exist to achieve this topology. A widely known technique to fabricate microchannels is using standard lithographic methods. However, these methods limit channel density and depth, making it difficult to realize true three-dimensional microchannel architectures. To date, only a few methods exist that successfully enable true 3D microfluidics.339 The serial nature of many 3D printing processes creates practical difficulties in realizing dense structures over large areas, and inherent resolution limits often prevent the routine construction of channels with inner diameters smaller than 50 µm.340 Unlike tetrapodal T-ZnO templates, a 3D-printed interconnected backbone is not self-assembled and requires precise, active fabrication. However, to advance the application of T-ZnO-derived microchannel networks to microfludic applications, the microchannels must be maniuplated individually. Furthermore, other templates than T-ZnO can be used to achive channel structures (e.g., sugar crystals). While these are typically more cost-effective than T-ZnO, they lack the highly anisotropic tetrapodal morphology necessary to achieve sufficient percolation at low filler concentrations.
Because channel structures predominantly consist of the matrix material (e.g., polymers such as PDMS), their mechanical properties are typically close to those of the matrix itself. These properties can be modified by introducing fillers into the pore network. Owing to their similarity to bulk materials, the environmental impact and durability of T-ZnO-derived channel networks are likewise largely determined by the matrix material used to form the pore network. To date, channel networks have only been produced at the laboratory scale. In principle, upscaling of this class of materials is feasible, but as with aeromaterials, it strongly depends on the availability of scalable T-ZnO production. However, compared to aeromaterials, far fewer applications currently justify the production costs of channel structures. Consequently, no industrial approaches toward their commercialization are known so far.
 | ||
| Fig. 54 Applications of aeromaterials and channel structures. Materials derived from T-ZnO templates enable a wide range of applications. | ||
4.3.3.1 Actuation and smart materials. The unique structure of T-ZnO is particularly advantageous for the development of actuators and smart materials. These are materials that can significantly change their properties in a controlled manner in response to external stimuli such as temperature or pressure. T-ZnO enables precise three-dimensional microstructuring within macroscopic samples, unlocking novel functionalities or significantly enhancing performance.
An innovative actuation concept introduced by Schütt et al.336 is based on low-density aeromaterials composed of conductive, two-dimensional carbon materials arranged in a three-dimensional architecture defined by a T-ZnO template. Upon Joule heating, these structures can be rapidly heated by several hundred Kelvin within just a few milliseconds. The high specific surface area of the aeromaterial facilitates efficient heat transfer to the surrounding gas molecules. This results in a rapid and significant expansion of gas volume. Due to the aeromaterial's low heat capacity, it quickly returns to its initial temperature. This process, referred to as electrically powered repeatable air explosion (EPRAE), can be repeated up to six times per second. It shows great promise for a large variety of applications, including lightweight electrically driven pneumatic systems, catalysis, sensing, sorption, propulsion, thermoacoustics, and microfluidics (Fig. 55I). This actuation method has been extensively investigated in the context of onboard pressure supply systems for soft robotics.326 Furthermore, Saure et al.19 developed an alternative aeromaterial based on SiO2, which is both transparent and highly scattering in the visible spectrum. By incorporating small amounts of homogeneously distributed rGO flakes, a three-dimensional light-absorbing structure was synthesized. Due to the material's low heat capacity, exposure to high-intensity light, such as from a powerful lamp, can rapidly increase its temperature by several hundred degrees Celsius throughout the entire volume. This results in a gas expansion effect comparable to that observed in the EPRAE process. Further publications have demonstrated the rapid heating capabilities of ultralight aeromaterials for applications such as underwater acoustic wave generation166 and self-cleaning air filtration system.323
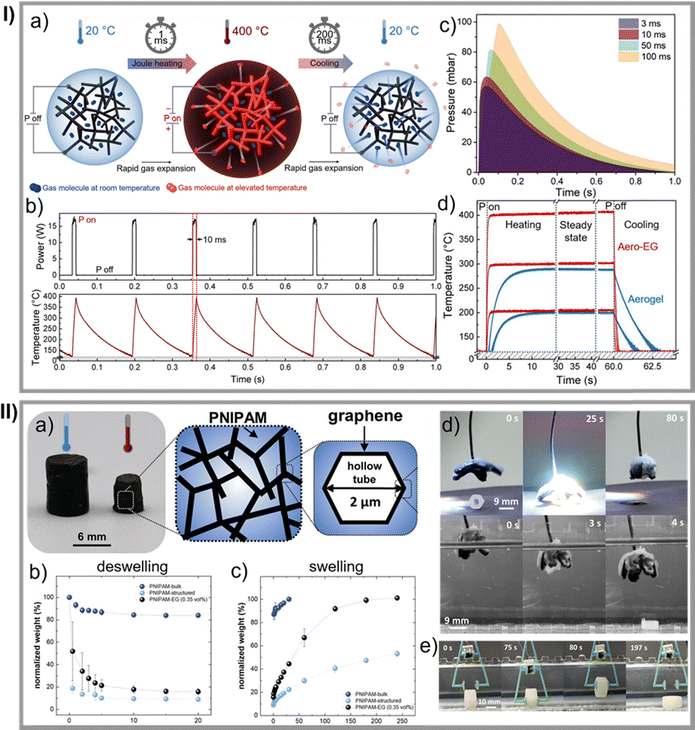 | ||
| Fig. 55 Actuation concepts based on T-ZnO-templated materials. (I) EPRAE concept:336 (a) Schematic of EPRAE, where Joule heating of a conductive aeromaterial causes instantaneous heat transfer and rapid air expansion. (b) Typical temperature-time profile. (c) Pressure generated across various power-pulse widths. (d) EPRAE effect comparison between conductive aeromaterials and aerogels. (II) Microstructured hydrogels:335 (a) image and schematic of T-ZnO-templated PNIPAM hydrogels with graphene channel walls. (b) Temperature-induced deswelling. (c) Swelling behavior comparison of unstructured bulk, structured, and graphene-walled structured PNIPAM. (d) and (e) Gripper concepts utilizing the swelling/deswelling mechanics of these hydrogels as artificial muscles. (I (a)–(d)) Reproduced from ref. 336 with permision from Elsevier,336 Copyright 2021, (I (a)–(e)) reproduced from ref. 335 with permision from Wiley,335 Copyright 2023. | ||
Actuators based on the EPRAE mechanism, represent an aeromaterial technology with the potential to significantly impact industrial production. In such environments, compressed air systems are among the largest electricity consumers, accounting for up to 10% of industrial energy use.341 However, these systems are typically inefficient, with leakage and poor maintenance leading to energy losses of 30% or more.342 The EPRAE mechanism enables localized, on-demand pressure generation, thereby eliminating the need for complex and leak-prone compressed-air distribution networks. Moreover, the EPRAE process generates pressure without relying on moving parts, in contrast to conventional mechanical compressors. As a result, EPRAE technology has the potential to substantially reduce both maintenance requirements and the overall energy demand associated with compressed-air systems. Recent studies have demonstrated that carbon-based conductive aeromaterials show promising cycling stability,336 scalability,323 and effective pressure generation326,336via EPRAE. Future research should aim at improving power-to-pressure conversion efficiency, increasing maximum pressure output, and identifying specific industrial applications where localized pressure supply would provide the greatest benefit. Even at its present stage of development, the EPRAE process may already provide valuable solutions for specific industrial applications.
Other aeromaterials, such as Aero-ZnS and Aero-GaN, exhibit distinct properties that enable self-propelling behavior at water surfaces, making them promising candidates for actuation-driven applications.324,343 Moreover, Barg et al.344 developed a multilayered aeromaterial composed of a conductive poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) core coated with an insulating polytetrafluoroethylene (PTFE) layer, resulting in a strain-invariant conductor. This material shows promise for a range of applications, including soft electronics and energy storage systems. However, due to the complexity of its synthesis and the limited scalability of the approaches discussed, these concepts are currently more likely to remain of primarily scientific interest rather than being readily transferable to large-scale applications.
Another approach to designing advanced actuators involves channel structures templated by a porous T-ZnO network. Spratte et al.165 filled the pores of such networks with a stimuli-responsive hydrogel, namely PNIPAM, which exhibits a volume change at a specific temperature. However, the swelling and deswelling capacity of bulk hydrogels is typically limited; the authors measured only a 12% volume reduction for unstructured hydrogels. By introducing microchannel structuring within the hydrogel network, a volume reduction of up to 90% was achieved, while the stiffness of the swollen hydrogel remained unchanged. Additionally, the response rate was significantly enhanced. This microchannel-structured hydrogel was successfully demonstrated as an artificial muscle to power a gripper (Fig. 55II).165 By coating the T-ZnO network with a thin layer of conductive material, such as exfoliated graphene flakes, conductive hydrogels with exceptional conductivity and extremely low filler loading can be synthesized334 Hauck et al.335 further investigated composites combining the high conductivity of microchannel-structured hydrogels with the thermoresponsive behavior of PNIPAM. This composite material exhibited a 400% enhancement in actuation dynamics and a 4000% increase in actuation stress by effectively overcoming water transport limitations inherent to hydrogels. The volume change of the hydrogel can be triggered electrically or optically, enabling advanced actuation concepts for soft actuators. Channel-network-based actuators have demonstrated impressive performance. However, their synthesis and handling remain challenging. Consequently, alternative actuation technologies based on T-ZnO-templated materials, such as the EPRAE process, appear more promising for industrial applications. With further progress toward simplified synthesis routes and improved usability, this approach could become highly attractive for soft robotics.
4.3.3.2 Photonics and laser light scattering. T-ZnO networks exhibit unique optical properties, as discussed in Section 3.3. Many of these properties are directly transferred to aeromaterials structurally derived from T-ZnO and are further enhanced by the characteristics of the materials composing the new aeromaterial. Such materials are promising for a range of optical applications. For instance, an aeromaterial consisting of interconnected hollow h-BN microtubes and investigated by Schütt et al.167 might be useful as a laser light diffuser. Due to the wide band gap of hBN (∼6.5 eV), these aeromaterials exhibit extremely low absorption in the optical range. The interconnected h-BN microtubes act as individual Rayleigh scattering centers for incident laser light, diffusely scattering it in all directions with low speckle contrast. By simultaneously scattering blue, green, and red laser beams within the Aero-hBN, white light generation is achieved, which is conceptualized in Fig. 56a. This approach holds promise for high-brightness lighting applications with light outputs surpassing those of conventional LEDs, which are typically limited to about 5 kW cm−2. However, the efficiency of laser diodes, especially in the green spectral region, remains a bottleneck. Saure et al.322 have proposed an approach to address this challenge. Their work combines an aeromaterial based on SiO2 (Fig. 56b), which exhibits optical properties in the visible spectrum comparable to boron nitride, with halide perovskite QDs (Fig. 56c), known for their high quantum yield. The CsPbBr3 QDs used efficiently convert UV light into green emission at 505 nm, as demonstrated in Fig. 56d. While halide perovskite QDs are typically prone to degradation under environmental stressors, the authors present initial strategies to improve stability, including encapsulation with poly(ethylene glycol)dimethacrylate (pEGDMA).
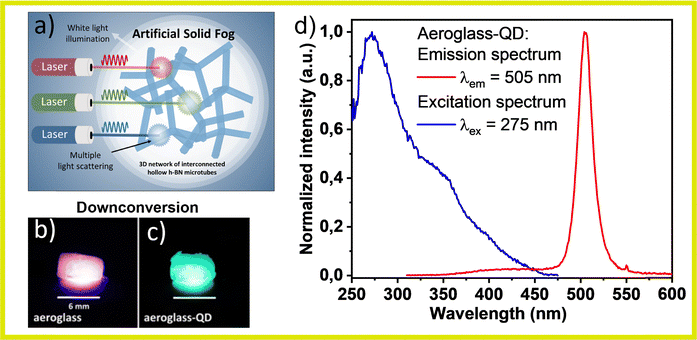 | ||
| Fig. 56 (a) White-light generation via “artificial solid fog” using a 3D network of interconnected hollow h-BN microtubes (>99.99% porosity). Multiple light scattering within the foam converts directional RGB laser inputs into an isotropic, high-brightness white-light source.167 (b) and (c) Photographs of pristine aeroglass and aeroglass-QD, respectively, under 275 nm excitation.322 (d) Emission (λex = 275 nm) and excitation (λem = 505 nm) spectra of the aeroglass-QD sample, demonstrating the down-conversion behavior of embedded QDs.322 (a) Reproduced from ref. 167 with permision from Springer Nature,167 Copyright 2020, (b)–(d) reproduced from ref. 322 with permision from Springer Nature,322 Copyright 2024. | ||
These findings highlight the potential to incorporate QDs with different emission wavelengths into a laser-scattering aeromaterial. This enables a single, efficient laser diode to generate light that is both scattered and spectrally converted into the desired color output. For the transfer of this technology into real-world applications, emphasis should be placed on avoiding lead-based materials such as the QDs reported by Saure et al.,322 as well as other highly toxic components. Ensuring the long-term stability of the laser light transformer and scatterer is also paramount. Furthermore, lasers can pose a serious health risk, particularly to the eyes, if the beam escapes a lighting device before being scattered by an aeromaterial. Therefore, developing safety mechanisms to ensure proper scattering and spatial distribution of the laser light is essential before this technology can be considered for real-world applications.
4.3.3.3 Biomedical applications. The hierarchical design capabilities enabled by the T-ZnO templating method also hold great potential for biomedical applications such as cell culture and drug delivery. In particular, aeromaterials are highly attractive as scaffolds for tissue engineering. This is due to their combination of a large, adaptable surface area for cell colonization and an open porous structure that facilitates effective nutrient transport (Fig. 57a).329,345–349 This concept was demonstrated for instance by Taale et al.346 who developed a multi-walled carbon nanotube (MWCNT)-based aeromaterial as a scaffold for osteoblast and fibroblast growth. The MWCNT scaffold was readily functionalized with biomineralizing agents, such as hydroxyapatite and bioactive glass nanoparticles, resulting in high cell viability and promising biocompatibility. When assessing aeromaterials for cell scaffolds, a particularly advantageous aspect is their hierarchical architecture, which closely mimics the multiscale structural complexity of natural biological systems. The open porosity of aeromaterial networks provides space for tissue growth while their interconnected pores facilitate nutrient transport.329,345–349 Both of which are key challenges in designing functional tissue scaffolds.350 Future research should focus on further enhancing this multiscale porosity to more closely replicate the extreme hierarchical architecture of biological systems. Increasing template complexity through methods such as laser cutting or additive manufacturing may enable the development of biomimetic scaffolds. These could support the simultaneous growth of multiple biological structures, including cells and blood vessels, marking an important step toward the artificial fabrication of functional organs.351
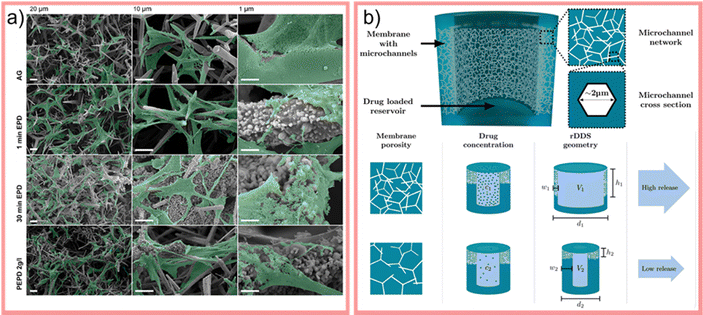 | ||
| Fig. 57 Biomedical applications based on T-ZnO-templated materials. (a) SEM images of aerographite scaffolds for cell growth. The aerographite was decorated with hydroxyapatite nanoparticles via periodic electrophoretic deposition and cultivated with pre-osteoblast cells (green).345 (b) Drug-reservoir concept using a T-ZnO-templated PDMS microchannel network. Release occurs via diffusion, with rates tunable by adjusting channel density (membrane porosity) and membrane thickness.330 (a) Reproduced from ref. 345 with permision from American Chemical Society,345 Copyright 2019, (b) reproduced from ref. 330 with permision from MDPI.,330 Copyright 2022. | ||
In addition to aeromaterials, channel structures templated via T-ZnO networks offer significant potential for biomedical applications, particularly in the field of controlled drug delivery. One notable example is the work by Rasch et al.327 who developed a PDMS-based microchannel network that functions as a drug reservoir and enables diffusion-controlled release. By adjusting the density and distribution of the drug-releasing channels, they achieved a highly tunable drug release profile (Fig. 57b). This simple yet effective system shows promise for applications such as glioblastoma therapy. Building on this concept, Hauck et al.330 expanded the approach through detailed computational modeling of T-ZnO-templated channel networks, enhancing understanding and design optimization of such drug-delivery systems. Using T-ZnO-templated channel networks as drug delivery systems is a highly promising approach, as demonstrated in the literature. The commonly used PDMS matrix is easy to incorporate, and with further clinical evaluation, this straightforward technology could become a viable real-world application.
Hydrogel-based channel structures created via a T-ZnO templating approach can further serve as biomimetic models for human tissue. Siemsen et al.328 investigated the migration behavior of cancer cells within these structures. They demonstrated that large-scale scaffolds can be infiltrated by cancer cells within just a few days. Their study also revealed that both the stiffness and the microarchitecture of the hydrogel significantly influence cellular invasion and nuclear dynamics. This underscores the importance of mechanical and structural cues in tumor progression. This study further highlights the potential of T-ZnO-templated materials for applications where diverse and complex biological architectures need to be mimicked.
4.3.3.4 Energy storage and conversion. The ability to fabricate hierarchically structured materials with large surface areas, tunable surfaces, and interconnected open porosity is highly attractive for applications in energy storage and conversion.
In the context of energy storage, T-ZnO-templated structures are particularly promising for the development of advanced supercapacitor electrodes.352 High-performance supercapacitors require thick electrodes that combine high areal mass loading with efficient ion and electron transport, which can be achieved through a carefully engineered pore architecture. The T-ZnO templating approach has demonstrated impressive capabilities in this regard, as shown by Spurling et al.353 who developed an MXene-based aeromaterial for supercapacitor applications (Fig. 58). Their design achieved a high areal loading of 7.2 mg cm−2, a material density of 1.44 g cm−3, and an excellent specific capacitance of 240 F g−1 (140 F cm−3). Remarkably, even at high scan rates of 200 mV s−1, the electrodes maintained an areal capacitance of 1.4 F cm−2, outperforming state-of-the-art MXene-based supercapacitor electrodes. In a different approach, Yuksel et al.85 developed an aerocomposite composed of nitrogen-doped tubular carbon and ZIF-8 particles, which was evaluated as an electrode material for zinc-ion supercapacitors. The resulting device exhibited an ultrahigh rate capability of up to 2000 mV s−1 and achieved a specific capacitance of 341.2 F g−1 at a current density of 0.1 A g−1.
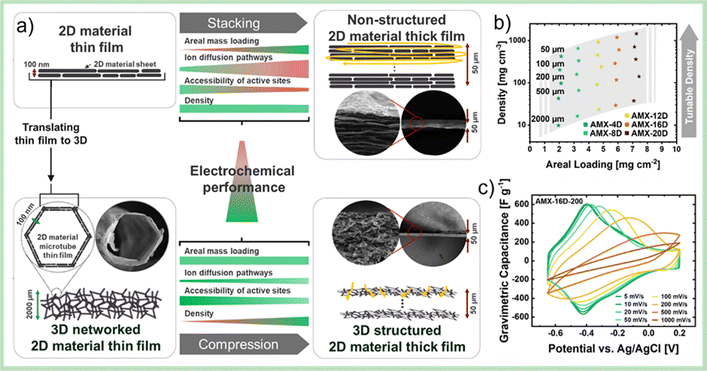 | ||
| Fig. 58 Supercapacitors based on aero-MXenes. (a) Comparison between a conventional 2D nanomaterial thick film and a thick film produced by compressing an MXene-based aeromaterial, resulting in a 3D-structured thick film with a pronounced influence on electrochemical performance. (b) Density and areal loading of 3D-structured MXene (the grey area indicates the feasible electrode region; stars indicate the prepared and tested samples). (c) Cyclic voltammogram of a 3D-structured MXene film with a mass loading of 5.8 mg cm−2 in 0.5 M H2SO4 at scan rates from 5 mV s−1 to 1000 mV s−1. (a) Reproduced from ref. 353 with permision from Elsevier,353 Copyright 2024. | ||
In addition to supercapacitors, hierarchically porous materials derived from T-ZnO have also been investigated for battery applications, particularly in lithium–sulfur (Li–S) batteries. In this context, the primary function of the T-ZnO-derived frameworks is to immobilize polysulfides. This mitigates the shuttle effect, thereby significantly enhancing the cycling stability and overall lifespan of the battery.354–356 Furthermore, an atypical material for battery applications, composed of β-Ga2O3/ZnGa2O4, has been investigated in this context. It demonstrated promising initial capacities of 714 mAh g−1 and, beyond its electrochemical applicability, exhibited narrow-band PL emission at 504 nm under UV irradiation, thereby highlighting the multifunctional versatility of aeromaterials derived from T-ZnO templates.357
Despite promising lab-scale results, particularly for T-ZnO-templated supercapacitor electrodes, this technology remains far from real-world application. The high costs associated with T-ZnO, nanomaterials and aerofication make energy storage devices economically unfeasible. This is especially true when compared to other T-ZnO-templated technologies, such as EPRAE-based actuation, which offer unique functionalities and thus greater commercial potential despite high production costs.
Regarding energy conversion, materials synthesized via a T-ZnO templating approach have not yet been extensively studied. However, they hold significant potential, particularly in (photo)catalysis. This is due to their open porous architecture, which facilitates high flow conditions in gas-phase reactions while maintaining a large surface area and, consequently, a high density of catalytically active sites. This has been explored in several publications, with typically photocatalytic materials such as zinc-doped cerium oxide20 (Fig. 59a) and titanium dioxide325,358 (Fig. 59b). The structural benefits of these aeromaterials extend beyond morphology, as their mechanical robustness under gas flow20 (Fig. 59c) enables stable operation in continuous-flow reactions. Their photocatalytic activity has also been demonstrated through the degradation of tetracycline using Aero-TiO2 under visible and UV irradiation, including continuous-flow UV conditions (Fig. 59d).325 Furthermore, among the many strategies aimed at enhancing photocatalytic performance, most of which focus on modifications at the nano- and microscale, the use of sacrificial T-ZnO templates presents a unique opportunity. It allows for structural control across multiple length scales, from the nanoscale to the macroscale. Notably, the macroscale is often overlooked in photocatalyst design. Yet, the ability to tailor features across all relevant scales, including the macroscale, may significantly improve photocatalytic efficiency.20,359
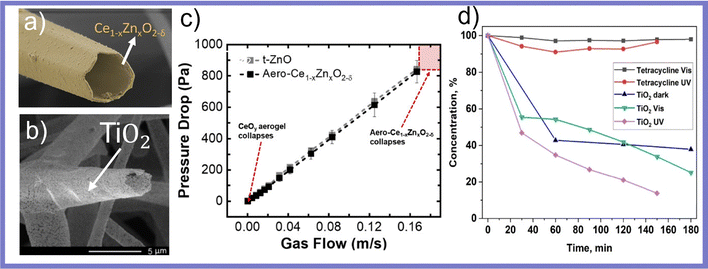 | ||
| Fig. 59 Applications of aeromaterials as photocatalysts: (a) SEM image of Aero-Ce1−xZnxO2−δ showing the hollow, architecture. (b) SEM image of Aero-TiO2 highlighting the open porous framework. (c) Pressure-drop curves of Aero-Ce1−xZnxO2−δ compared with pristine T-ZnO, illustrating the mechanical stability and gas-flow characteristics of the aeromaterial structure. (d) Photocatalytic degradation of tetracycline using Aero-TiO2 under visible and UV irradiation. (a) and (c) Reproduced from ref. 20 with permision from American Chemical Society,20 Copyright 2024, (b) reproduced from ref. 358 with permision from MDPI,358 Copyright 2022, (d) reproduced from ref. 325 with permision from Springer Nature,325 Copyright 2024. | ||
4.3.3.5 Sensing. T-ZnO itself is a versatile and promising material for various sensing applications (see Section 4.1.1). Its primary advantages in this field stem from its unique three- dimensional architecture. This provides, for example, a high surface area, excellent accessibility of active sites, and enhanced interaction with analytes. By employing T-ZnO as a structural template for the fabrication of three-dimensionally structured materials, these benefits can be transferred to other functional materials. This significantly improves their sensing performance. For example, Dragoman et al.360 employed T-ZnO networks as sacrificial templates to synthesize an aeromaterial composed of GaN, which exhibits a high piezoelectric coefficient and demonstrates excellent sensitivity to pressures ranging from 5 to 40 atm. Due to its low density and mechanical robustness, this material holds significant promise for aerospace applications. In another innovative approach, Ren et al.361 utilized the unique geometry of T-ZnO to fabricate a bio-inspired pressure sensor. By combining T-ZnO and graphene oxide via vacuum filtration, followed by T-ZnO etching and partial reduction, they created a selective membrane for cations. When placed between NaCl solution reservoirs, pressure applied to one reservoir induces the migration of cations through the membrane, generating a measurable signal as a result of net charge flow. This sensor demonstrated a sensitivity of 0.282 nA Pa−1 and response and recovery times of 90 ms and 110 ms, respectively. Further studies have highlighted the gas-sensing capabilities.362–365 and UV-Photodetection capabilities366 of T-ZnO-templated materials. (Fig. 60a) shows a schematic gas sensor based on aerographite by Lupan et al.362 (Fig. 60b) shows the response of the sensor after exposition to various gases and concentrations as a function of the applied bias voltage.362 These examples demonstrate how the distinctive architecture of T-ZnO can be exploited as a microstructural template for designing advanced and highly responsive sensing devices at the lab scale. However, mass production of aeromaterial-based sensors remains challenging due to the need for highly reproducible control of film thickness, conduction pathways, and contacts. Such precision is less critical in other applications (e.g., cell scaffolds or EPRAE-based systems), suggesting that the commercialization of T-ZnO-templated sensors will likely lag behind other T-ZnO-templated material technologies.
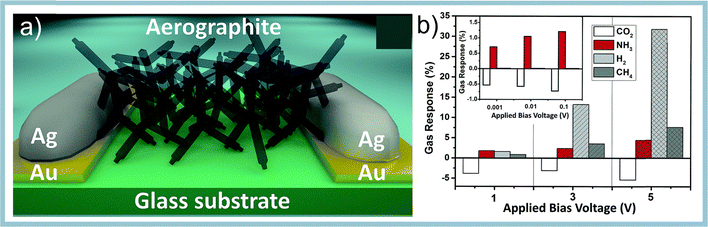 | ||
Fig. 60 (a) Schematic representation of the aerographite-based gas sensor structure, showing the interconnected aerographite network bridging Ag/Au contacts on a glass substrate. (b) Gas response of the aerographite sensor at room temperature as a function of applied bias voltage for CO2 (500 ppm), NH3 (100 ppm), H2 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm), and CH4 (10 000 ppm), and CH4 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm). (a) and (b) Reproduced from ref. 362 with permision from Royal Chemical Society,362 Copyright 2016. 000 ppm). (a) and (b) Reproduced from ref. 362 with permision from Royal Chemical Society,362 Copyright 2016. | ||
The extensive list of materials and applications catalogued in Table 14 illustrates a dynamic and rapidly expanding field of research. To better synthesize these developments and identify future opportunities, we have distilled this information into a strategic “opportunity matrix” (Fig. 61). This matrix maps the primary material classes against their application domains, with the research maturity of each area color-coded according to its chronological emergence. The matrix reveals a clear three-phase evolutionary trajectory. The field was founded on established (green) research into carbon and semiconductor materials mainly for sensing applications, which served as the initial proof-of-concept. A second wave of growing (light green) research, beginning around 2017, saw a diversification into new applications like actuation, largely driven by the adoption of more scalable synthesis methods. From 2022 until 2025 (light red) the field of T-ZnO templated materials made further progress in refining existing materials and synthesis approaches to tackle larger scale applications like air filters. Furthermore, more complex materials were synthesized like rGO flake loaded SiO2 aeromaterials, which can be used for actuation via a volumetric photothermal response. In addition, the opportunity matrix shows material class application combinations, which have not been explored yet (red), but may represent significant opportunities for future investigation.
 | ||
| Fig. 61 The T-ZnO templating landscape, an opportunity matrix. This matrix provides a strategic overview of the research field, mapping material classes against key application domains. The color-coding indicates the research maturity for each intersection, based on the chronology of publications in Table 14: established (green, foundational work, since 2012), growing (light green, active diversification, since 2017), emerging (light red, recent breakthroughs, since 2022), and unexplored (red, potential future research avenues). The matrix highlights the field's evolution from foundational carbon-based materials towards a diverse and functionally driven frontier. | ||
4.4 Key learnings from T-ZnO applications
The application landscape of T-ZnO is defined by a powerful duality. This is a direct consequence of the unique structure–property nexus described in Section 3. The material can be leveraged either as a functionally active component, where its intrinsic properties are paramount, or as a structurally transformative sacrificial template. To provide a critical overview, the diverse application areas discussed in this section are systematically summarized and evaluated in Table 15. This table offers a comparative analysis of each domain's technological maturity and the key challenges hindering its translation from the laboratory to industrial scale.| Application area | Primary T-ZnO property leveraged | Current TRL (estimated) | Primary bottleneck for scale-up |
|---|---|---|---|
| Sensing applications (gas, UV, pressure, bio) | Chemiresistive effect, piezoelectricity, high surface area, and functionalizable surface | 3–6 (lab-scale to pilot-scale; prototypes in real-world scenarios) | Long-term stability & drift, cross-sensitivity (to humidity/temp), and reproducibility in complex biological media. |
| Surface engineering & environmental (coatings, decontamination, catalysis) | Switchable wettability, hierarchical porosity/high surface area, and photocatalytic activity | 2–6 (proof-of-concept to early industrial trials) | Long-term durability (especially under UV), cost-competitiveness with existing solutions (e.g., activated carbon), and environmental concerns of some components (e.g., fluorinated polymers). |
| Biomedical & healthcare (antibacterial, wound dressings, stents) | Biocompatibility, antibacterial/antiviral activity, and mechanical reinforcement in composites. | 5–10 (advanced development to early commercialization) | Ensuring long-term biocompatibility, navigating complex regulatory approval pathways, scaling-up pharma grade T-ZnO production |
| Energy storage & conversion (batteries, thermal storage) | Thermal conductivity, hierarchical porosity (for ion transport), and photocatalytic properties. | 2–4 (lab-scale/proof-of-concept) | Scalable, low-cost synthesis for bulk applications; improving overall energy conversion/storage efficiency; ensuring long-term cycling stability. |
| Composite reinforcement & EMI shielding (structural fillers, shielding materials) | Mechanical percolation and 3D interlocking network to create a functional backbone. | 2–4 (advanced research with promising laboratory results) | Optimizing filler-matrix interfacial adhesion for mechanical strength; balancing absorption vs. reflection for EMI shielding; cost. |
| Sacrificial templating (aeromaterials & channel structures) | Unique 3D morphology, hierarchical porosity, and chemical removability (via etching). | 2–s6 (advanced research/prototypes in real-world scenarios/emerging field) | Complexity of multi-step processing; scalability of high-quality coating methods (gas-phase vs. wet-chemical); cost of template production coating and template removal at industrial scale. |
5. A practical outlook for T-ZnO architected materials
This review has outlined the key structure–property relationships of T-ZnO. Its utility does not arise from ideal crystallinity, but from a complex, defect-rich architecture that is arrested far from its thermodynamic ground state. The same atomic defects that enable n-type conductivity can also quench luminescence or initiate mechanical fatigue. This trade-off is intrinsic and defines the performance envelope of T-ZnO-based systems. Rather than viewing defect-dependency as a liability, we propose treating it as a programmable feature. T-ZnO is a material in which defects, junctions, and network topology are not accidents, but control knobs. These effects are supported by experimental observations reported in the literature. Charge trapping at defect sites can record prior exposure to stress, light, or electrical bias. Meanwhile, junctions govern how electrical or mechanical signals propagate through the network.5.1 Templating as a manufacturing approach
T-ZnO-based materials are best understood through the concept of architectural templating, which occupies a distinct position within advanced manufacturing. Additive manufacturing enables precise macroscopic shape control but provides limited access to complex nano- and microscale networks. Conversely, self-ordered assembly produces nanoscale features but offers little control over macroscopic form. T-ZnO templating bridges this critical gap. T-ZnO solves the “scalability gap” in nanotechnology by using self-assembly to create macroscopic, manipulatable parts. It allows for the transfer of a pre-formed, self-assembled nano-micro architecture into bulk materials with high fidelity. This effectively scales the benefits of the nanoscale to the human hand. T-ZnO performs best when function arises from the tetrapodal network itself. Examples include high porosity, rapid mass transport, low density, and large accessible surface area. Comparable benefits can also be achieved when these network properties are transferred into new material classes. In this way, T-ZnO enables functional systems that remain difficult to achieve using conventional fillers or fabrication strategies.5.2 From laboratory studies to practical systems
Looking ahead, future progress in T-ZnO design depends on controlling morphology together with the type, density, and spatial distribution of imperfections. This is necessary to achieve defined and testable functions. This requires correlating synthesis parameters with measurable outputs such as junction density, percolation threshold, carrier mobility, and mechanical compliance. Two complementary strategies can be distinguished. First, T-ZnO can act as an active framework in which sensing, memory-related effects, and signal routing arise directly from the network architecture. Second, it can serve as a sacrificial template to transfer this architecture into polymers, carbons, or oxides. This separates structural function from ZnO chemistry. Achieving either strategy depends on predictive synthesis and scalable processing. From a development perspective, bringing T-ZnO from laboratory studies to practical use requires a clear and structured development path. Recent advances in synthesis, structural control, and integration indicate that T-ZnO can act as more than a passive functional filler. It can function as a connected network whose macroscopic response is governed by interactions between many junctions. This, in turn, requires control over individual tetrapods, their connections, and the integration of resulting networks into stable and scalable material systems.Together, the technical directions described above form a single, connected development path. Predictive synthesis, network integration, and scalable manufacturing inform and constrain each other through continuous feedback.
5.3 Development roadmap and application perspectives
Table 16 summarizes a development roadmap for T-ZnO network materials, highlighting current practice, key limitations, and concrete steps required for progress.| Area | Current practice | Main limitation | Required advance | TRL | Outcome | Feedback |
|---|---|---|---|---|---|---|
| Predictive synthesis | Trial-and-error synthesis | Weak process-structure link | Link reactor conditions to tetrapod geometry and junction density | 2 → 4 | Controlled network topology | Enables integration and scale-up |
| Network integration | Single-function composites | Poor interface control | Control contact resistance, adhesion, and strain transfer | 1 → 3 | Predictable electrical and mechanical response | Defines failure modes that dictate synthesis targets. |
| Manufacturing scale-up | Lab-scale batch synthesis | Low throughput, high energy cost | Develop continuous, energy-efficient reactors | 2 → 5 | Scalable production of T-ZnO networks | Sets hard cost limits that eliminate non-viable precursors. |
| Interdisciplinary expertise | Disciplinary separation | Fragmented workflows | Integrate synthesis, characterization, and system design | N/A | Faster iteration and reproducibility | Closes the loop between atomistic simulation and reactor engineering. |
If these challenges can be addressed, the T-ZnO platform could enable several application directions that rely on controlled network architecture rather than material composition alone. In catalysis and energy-related systems, the well-defined geometry of tetrapod junctions can be used to create microstructured catalysts and battery or supercapacitor electrodes. These would feature high accessible surface area, efficient mass transport, and improved mechanical stability. T-ZnO-derived networks can also be used as physical systems in which charge transport is distributed across many junctions. Their electrical response is governed by network topology and junction properties. This leads to history-dependent behavior without relying on conventional circuit design. This nonlinear, memory-driven response suggests that T-ZnO networks could serve as physical substrates for reservoir computing and neuromorphic hardware. For example, an elastomeric patch embedded with a T-ZnO network could be utilized as an adaptive tactile sensor (a smart skin), where the network inherently processes and filters complex mechanical stimuli, such as human touch or robotic grasping forces, without the need for external analog-to-digital microprocessors. The same open, multiscale architectures make T-ZnO templates suitable as scaffolds for biological systems or hybrid bio-inorganic materials. Here, surface chemistry can be tailored to control interactions with organic matter. Beyond static structures, these networks further enable materials whose properties evolve over time. Examples include actuators with tunable porosity or conductive pathways that change under electrical or mechanical loading. Together, these examples illustrate how control over network architecture can translate into additional, application-specific functionality.
Ultimately, T-ZnO represents a paradigm shift from Material-Centric to Architecture-Centric design. It is a versatile building block for architected materials in which function is encoded in network structure rather than added through composition alone. Continued progress will depend on closing feedback loops between synthesis, structure, and performance. This must be supported by coordinated experimental, analytical, and processing expertise. With these elements in place, T-ZnO-based systems provide a well-defined platform for exploring structure-driven functionality across a range of material applications.
Conflicts of interest
The authors declare that they have no conflicts of interest.Glossary of abbreviations
| 0D/1D/2D/3D | Zero-/one-/two-/three-dimensional |
| Aero-BN | Aero-boron nitride |
| APMP | Atmospheric-pressure microwave plasma |
| APTES | (3-Aminopropyl)triethoxysilane |
| c-AFM | Conductive atomic force microscopy |
| CDF | Counterflow diffusion flame |
| CFD | Computational fluid dynamics |
| CL | Cathodoluminescence |
| CNT | Carbon nanotube |
| CVD | Chemical vapor deposition |
| Da | Damköhler number (DaI, DaII) |
| DFT | Density functional theory |
| DLE | Deep-level emission |
| DNA | Deoxyribonucleic acid |
| DPy | 4,4′-Dipyridyl |
| DSSC | Dye-sensitized solar cell |
| E-FTS | Ethanol-assisted flame transport synthesis |
| ED | Electron diffraction |
| EDX | Energy dispersive X-ray (analysis) |
| EELS | Electron energy loss spectroscopy |
| EMI | Electromagnetic interference |
| EPRAE | Electrically powered repeatable air explosion |
| EPR | Electron paramagnetic resonance |
| ETEM | Environmental transmission electron microscopy |
| F-POSS | Fluorinated polyhedral oligomeric silsesquioxane |
| FEA | Finite element analysis |
| FEM | Finite element method |
| FET | Field-effect transistor |
| FTIR | Fourier-transform infrared spectroscopy |
| FTS | Flame transport synthesis |
| GelMA | Gelatin methacryloyl |
| GMP | Good manufacturing practice |
| HER-2 | Epidermal growth factor receptor 2 |
| HFCVD | Hot-filament chemical vapor deposition |
| HRTEM | High-resolution transmission electron microscopy |
| HVPE | Hybrid vapor phase epitaxy |
| IMPS | Intensity modulated photocurrent spectroscopy |
| L D | Debye length |
| LED | Light-emitting diode |
| LSPR | Localized surface plasmon resonance |
| MCM | Melting-combustion method |
| MEMS | Micro-electro-mechanical systems |
| ML | Machine learning |
| MOF | Metal–organic framework |
| MWCNT | Multi-walled carbon nanotube |
| NBE | Near-band-edge (Emission) |
| OTS | Octadecyltrichlorosilane |
| OVFE | Oxygen vacancy formation energy |
| PCM | Phase change material |
| PDMS | Polydimethylsiloxane |
| Pe | Péclet number |
| PEDOT:PSS | Poly(3,4-ethylenedioxythiophene)polystyrene sulfonate |
| pEGDMA | Poly(ethylene glycol)dimethacrylate |
| PL | Photoluminescence |
| PLE | Photoluminescence excitation |
| PMMA | Poly(methyl methacrylate) |
| PNIPAM | Poly(N-isopropylacrylamide) |
| PTFE | Polytetrafluoroethylene |
| PVB | Polyvinyl butyral |
| PVDF | Polyvinylidene fluoride |
| QD | Quantum dot |
| ROS | Reactive oxygen species |
| SAED | Selected area electron diffraction |
| SAXS | Small-angle X-ray scattering |
| SEM | Scanning electron microscopy |
| SERS | Surface-enhanced Raman spectroscopy |
| SHS | Self-propagating high-temperature synthesis |
| STEM | Scanning transmission electron microscopy |
| STM | Scanning tunneling microscopy |
| TCO | Transparent conductive oxide |
| TEM | Transmission electron microscopy |
| TRPL | Time-resolved photoluminescence |
| T-ZnO | Tetrapodal zinc oxide |
| UPS | Ultraviolet photoemission spectroscopy |
| VEGF | Vascular endothelial growth factor |
| VOCs | Volatile organic compounds |
| WAXS | Wide-angle X-ray scattering |
| WGM | Whispering gallery mode |
| WZ | Wurtzite (crystal structure) |
| XPS | X-ray photoelectron spectroscopy |
| XRD | X-ray diffraction |
| ZB | Zinc-blende (crystal structure) |
| ZIF | Zeolitic imidazolate framework |
Master glossary
| Term | Definition |
|---|---|
| Active functional material | A design paradigm where the T-ZnO tetrapod itself performs the primary task (e.g., sensing, catalysis) by leveraging its intrinsic electronic, optical, or piezoelectric properties. |
| Active structural material | A design paradigm where the T-ZnO network serves as a physical backbone. Its function relies on the 3D geometry and mechanical connectivity (e.g., for reinforcement or EMI shielding) rather than chemical activity. |
| Aeromaterial | An ultra-lightweight, highly porous material synthesized by coating a sacrificial T-ZnO template with a target material (e.g., Carbon, GaN) and subsequently etching away the ZnO, leaving a hollow, interconnected tubular network. |
| Antifouling | The capability of a surface coating to inhibit the attachment and growth of bio-organisms (e.g., marine biofouling). In T-ZnO coatings, this is achieved through tunable wettability and surface micro-texture. |
| Architectural templating | The strategic manufacturing niche of T-ZnO. It involves using the self-assembled tetrapod network to transfer complex, high-fidelity micro-architectures to macroscopic materials, effectively bridging the scale gap between bottom-up synthesis and top-down fabrication. |
| Atom economy | A green chemistry metric that evaluates the efficiency of a chemical reaction by calculating how many atoms from the reactants are incorporated into the final product versus how many are lost as waste. |
| Band structure engineering | The modification of a material's electronic structure (specifically the bandgap and band edges) through alloying (e.g., MgZnO) or doping to tailor its optical absorption or electrical properties. |
| Carbothermal reduction | A high-temperature synthesis mechanism used in CVD where carbon acts as a reducing agent to strip oxygen from a metal oxide (e.g., ZnO) to generate metal vapor. |
| Cassie–Baxter state | A wetting regime where a liquid droplet sits on top of surface microstructures with trapped air pockets underneath, resulting in superhydrophobicity and high contact angles. |
| Channel structure | An inverse-opal-like material created by infiltrating the void space of a T-ZnO network with a matrix (e.g., polymer, hydrogel) and removing the template, resulting in a solid block permeated by interconnected micro-channels. |
| Chemiresistive sensor | A sensor device that detects analytes (gases) or radiation (UV) by measuring changes in electrical resistance caused by the modulation of the surface depletion layer width. |
| Correlative microscopy | An analytical approach that applies multiple characterization techniques (e.g., CL, SEM, EELS) to the exact same single nanostructure to directly link atomic-scale features to functional performance. |
| Damköhler number, second (DaII) | A dimensionless number representing the ratio of the chemical reaction rate to the diffusive mass transfer rate. In T-ZnO synthesis, a high DaII indicates the system is reaction-dominated, favoring gas-phase nucleation of tetrapods. |
| Depletion layer | A space-charge region at the semiconductor surface depleted of mobile carriers. Its modulation by adsorbed oxygen or target gases is the fundamental transduction mechanism for T-ZnO gas sensors. |
| Digital twin | A comprehensive, multi-physics computational model capable of simulating the material's entire lifecycle. It connects macroscopic reactor parameters (T,P,Flow) directly to the resulting atomic structure and final functional performance. |
| Double Schottky barrier (DSB) | The potential energy barrier formed at the grain boundary between two contacting tetrapod arms. It creates back-to-back depletion regions, acting as the dominant resistive bottleneck for charge transport in the network. |
| Endocytosis | The cellular uptake mechanism for small particles. T-ZnO microparticles are typically larger than the threshold for endocytosis, which prevents cellular internalization and significantly reduces cytotoxicity compared to nanoparticles. |
| Exciton | A quasiparticle consisting of an electron and a hole bound together by electrostatic attraction. ZnO has a high exciton binding energy (60 meV), enabling stable room-temperature light emission. |
| Exciton-polariton | A hybrid light-matter quasiparticle resulting from the strong coupling of photons and excitons, typically observed in high-quality T-ZnO optical cavities. |
| Flaw tolerance | A nanomechanical principle based on Griffith's theory: as material volume decreases (to the nanoscale), the probability of containing a critical failure-inducing flaw drops, allowing ceramic T-ZnO arms to bend elastically without fracturing. |
| Fog harvesting | The collection of water from the atmosphere using functionalized surfaces. T-ZnO composites, particularly those with patterned wettability (Janus membranes), optimize the nucleation and transport of water droplets. |
| Formation energy (Ef) | The thermodynamic energy cost required to create a specific defect. It is not fixed but varies with the Fermi level and chemical potential (e.g., Zn-rich vs. O-rich conditions). |
| Free carrier screening | The phenomenon where mobile charge carriers (electrons) redistribute to neutralize the strain-induced piezopotential, fundamentally limiting the efficiency of piezoelectric sensing in semiconducting ZnO. |
| Geometric amplification | The structural mechanism where the tetrapod geometry concentrates applied macroscopic force at the arm roots (junctions), generating a local piezopotential significantly higher than in simple nanowires. |
| Janus membrane | A membrane engineered with asymmetric wettability (e.g., one side hydrophobic, the other hydrophilic) to drive directional water transport, utilized in fog harvesting and separation applications. |
| Kinetic control | A growth regime dominated by high energy and rapid flux. Atoms adhere to the crystal surface faster than they can diffuse to equilibrium positions, leading to anisotropic, high-energy shapes like tetrapods. |
| Localized buckling | The specific deformation mode of T-ZnO under compression, where the central core acts as a compliant hinge, allowing the network to deform reversibly rather than fracturing. |
| Materials-by-design | A paradigm shifting from empirical trial-and-error to the predictive, rational engineering of materials with specific target properties using computational modeling and fundamental physical laws. |
| Metastable | A state that is energetically higher than the ground state but kinetically stable over long periods. The cubic zinc blende (ZB) phase is hypothesized to be the metastable nucleus for T-ZnO. |
| Mie scattering | The elastic scattering of light by particles comparable in size to the light's wavelength. This is the dominant mechanism driving the strong “light trapping” and path-length enhancement in T-ZnO networks. |
| Molecular sieve | A porous coating (e.g., ZIF-8) applied to T-ZnO sensors that filters gas molecules based on kinetic diameter, thereby enhancing selectivity (e.g., allowing H2 to pass while blocking larger molecules). |
| Monodispersity | The state of a collection of particles having uniform size and shape. |
| Near-band-edge (NBE) emission | Sharp, UV photoluminescence resulting from the direct recombination of excitons. A high NBE/DLE ratio is the primary metric for high crystal quality. |
| Non-centrosymmetric | A crystal lattice lacking a center of inversion (symmetry). This structural asymmetry in wurtzite ZnO leads to charge polarization under strain, the origin of the piezoelectric effect. |
| Nucleation | The initial symmetry-breaking event in crystal growth where a small cluster of atoms rearranges into a stable thermodynamic phase, providing the template for subsequent growth. |
| Passivation | The application of a coating or chemical treatment to neutralize surface defect states, thereby reducing non-radiative recombination and improving carrier mobility. |
| Piezo-phototronics | A field of coupled physics using the strain-induced piezopotential to modulate the band structure at an interface, thereby controlling the generation, separation, and transport of photo-excited carriers. |
| Piezotronics | A field using the piezoelectric potential as a “gate” voltage to tune the charge carrier transport in a semiconductor device (effectively a mechanical field-effect transistor). |
| Pinch-off effect | A transport condition in nanowires where the surface depletion regions overlap, causing the entire cross-section to become insulating. |
| Polar catastrophe | The thermodynamic instability of crystal facets terminated by a single ionic species (e.g., the polar {0001} face). The surface reconstructs or adsorbs species (like −OH) to neutralize this diverging potential, driving high chemical reactivity. |
| Predictive synthesis | The transition from empirical material discovery to data-driven manufacturing. It relies on establishing quantitative scaling laws (like DaII and Pe) to accurately forecast morphological outcomes before physical synthesis begins. |
| Rational design | An engineering approach based on the predictive understanding of fundamental chemical and physical principles to create materials with specific, pre-determined functionalities. |
| Sacrificial templating | A manufacturing strategy where the T-ZnO network defines the architecture of a new material. The “transcription” of the tetrapodal geometry allows the fabrication of complex 3D structures from materials that cannot self-assemble into such shapes. |
| Self-compensation | A thermodynamic feedback loop where shifting the Fermi level (e.g., to achieve p-type doping) lowers the formation energy of opposing native defects (e.g., donors), which spontaneously form to neutralize the intended doping. |
| Supersaturation | A non-equilibrium state where a vapor or solution contains a concentration of solute exceeding the thermodynamic solubility limit. It is the primary thermodynamic driving force for nucleation. |
| Switchable wettability | The ability of T-ZnO surfaces to reversibly transition between hydrophobic and hydrophilic states under external stimuli (UV light, heat), acting as a gate for molecular access in sensors and coatings. |
| Synthesis-stability nexus | The principle that a material's ultimate lifecycle and failure modes are inextricably pre-determined by the thermodynamic path taken during its initial synthesis. |
| Synthesis-structure–property nexus | The foundational concept of materials science, describing the causal link where processing conditions determine atomic/micro-structure, which in turn dictates macroscopic material performance. |
| Thermal runaway | An uncontrolled exothermic chain reaction in batteries. T-ZnO coatings on separators or electrodes help prevent this by providing thermal stability and preventing internal short circuits. |
| Thermodynamic control | A growth regime dominated by stability and slow rates. Atoms have sufficient time to diffuse to low-energy sites, resulting in compact, faceted shapes like spheres or prisms to minimize surface energy. |
| Whispering gallery mode (WGM) | An optical resonance effect where light propagates around the hexagonal circumference of a T-ZnO arm via total internal reflection, acting as a high-quality optical cavity. |
| Wurtzite (WZ) | The thermodynamically stable, hexagonal crystal polymorph of ZnO. It is non-centrosymmetric and forms the arms of the tetrapod. |
| Zinc-blende (ZB) | The metastable, cubic crystal polymorph of ZnO. Several growth models propose that a ZB seed is required to initiate the tetrahedral symmetry of the tetrapod. |
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors acknowledge the German Aerospace Centre (DLR – FKZ: 50RP2300A) and the German Research Foundation (DFG – 501386727) for the financial support of this work. Furthermore, this project received support from the German Research Foundation (DFG) through the German-Taiwan Collaborative Project (AD 183/35-1).References
- M. L. Fuller, Twinning in Zinc Oxide, J. Appl. Phys., 1944, 15, 164–170 CrossRef CAS.
- K. Nishio, T. Isshiki, M. Kitano and M. Shiojiri, Structure and growth mechanism of tetrapod-like ZnO particles, Philos. Mag. A, 1997, 76, 889–904 Search PubMed.
- H. Iwanaga, M. Fujii and S. Takeuchi, Growth model of tetrapod zinc oxide particles, J. Cryst. Grow., 1993, 134, 275–280 CrossRef CAS.
- C. Ronning, N. G. Shang, I. Gerhards, H. Hofsäss and M. Seibt, Nucleation mechanism of the seed of tetrapod ZnO nanostructures, J. Appl. Phys., 2005, 98, 34307 CrossRef.
- Q. Zhang, W. Fan and L. Gao, Anatase TiO2 nanoparticles immobilized on ZnO tetrapods as a highly efficient and easily recyclable photocatalyst, Appl. Catal., B, 2007, 76, 168–173 Search PubMed.
- O. Lupan, L. Chow and G. Chai, A single ZnO tetrapod-based sensor, Sens. Actuators, B, 2009, 141, 511–517 Search PubMed.
- W. Chen and S. Yang, Dye-sensitized solar cells based on ZnO nanotetrapods, Front. Optoelectron., 2011, 4, 24–44 Search PubMed.
- M. Mecklenburg, A. Schuchardt, Y. K. Mishra, S. Kaps, R. Adelung, A. Lotnyk, L. Kienle and K. Schulte, Aerographite: ultra lightweight, flexible nanowall, carbon microtube material with outstanding mechanical performance, Adv. Mater., 2012, 24, 3486–3490 CrossRef CAS PubMed.
- F. Rasch, F. Schütt, L. M. Saure, S. Kaps, J. Strobel, O. Polonskyi, A. S. Nia, M. R. Lohe, Y. K. Mishra, F. Faupel, L. Kienle, X. Feng and R. Adelung, Wet-Chemical Assembly of 2D Nanomaterials into Lightweight, Microtube-Shaped, and Macroscopic 3D Networks, ACS Appl. Mater. Interfaces, 2019, 11, 44652–44663 CrossRef CAS PubMed.
- Y. Ding, Z. L. Wang, T. Sun and J. Qiu, Zinc-blende ZnO and its role in nucleating wurtzite tetrapods and twinned nanowires, Appl. Phys. Lett., 2007, 90, 153510 CrossRef.
- M. Shiojiri and C. Kaito, Structure and growth of ZnO smoke particles prepared by gas evaporation technique, J. Cryst. Grow., 1981, 52, 173–177 CrossRef CAS.
- Y. K. Mishra and R. Adelung, ZnO tetrapod materials for functional applications, Mater. Today, 2018, 21, 631–651 CrossRef CAS.
- A. Pujara, R. Sharma, S. Sharma, M. Bechelany, Y. K. Mishra and J. Prakash, Novel zinc oxide 3D tetrapod nano-microstructures: recent progress in synthesis, modification and tailoring of optical properties for photocatalytic applications, Mater. Adv., 2025, 6(7), 2123–2153 RSC.
- J. S. Tawale, K. K. Dey, R. Pasricha, K. N. Sood and A. K. Srivastava, Synthesis and characterization of ZnO tetrapods for optical and antibacterial applications, Thin Solid Films, 2010, 519, 1244–1247 Search PubMed.
- M. C. Newton and P. A. Warburton, ZnO tetrapod nanocrystals, Mater. Today, 2007, 10, 50–54 Search PubMed.
- M. Y. Guo, A. M. C. Ng, F. Liu, A. B. Djurisic, W. K. Chan, H. Su and K. S. Wong, Effect of native defects on photocatalytic properties of ZnO, J. Phys. Chem. C, 2011, 115, 11095–11101 Search PubMed.
- B. Yin, Y. Qiu, H. Zhang, J. Lei, Y. Chang, J. Ji, Y. Luo, Y. Zhao and L. Hu, Piezoelectric effect of 3-D ZnO nanotetrapods, RSC Adv., 2015, 5, 11469–11474 Search PubMed.
- F. Rasch, F. Schütt, L. M. Saure, S. Kaps, J. Strobel, O. Polonskyi, A. S. Nia, M. R. Lohe, Y. K. Mishra, F. Faupel, L. Kienle, X. Feng and R. Adelung, Wet-Chemical Assembly of 2D Nanomaterials into Lightweight, Microtube-Shaped, and Macroscopic 3D Networks, ACS Appl. Mater. Interfaces, 2019, 11, 44652–44663 Search PubMed.
- L. M. Saure, N. Kohlmann, H. Qiu, S. Shetty, A. Shaygan Nia, N. Ravishankar, X. Feng, A. Szameit, L. Kienle, R. Adelung and F. Schütt, Hybrid Aeromaterials for Enhanced and Rapid Volumetric Photothermal Response, ACS Nano, 2023, 17, 22444–22455 Search PubMed.
- J. Lumma, T. Tjardts, E. Greve, L. M. Saure, S. Veziroglu, R. Adelung, L. Kienle, N. Wolff and F. Schütt, Synthesis of Highly Porous 3D Cerium Oxide Networks Designed for Catalytic Applications, Cryst. Growth Des., 2024, 24, 4914–4923 Search PubMed.
- Y. K. Mishra, G. Modi, V. Cretu, V. Postica, O. Lupan, T. Reimer, I. Paulowicz, V. Hrkac, W. Benecke, L. Kienle and R. Adelung, Direct Growth of Freestanding ZnO Tetrapod Networks for Multifunctional Applications in Photocatalysis, UV Photodetection, and Gas Sensing, ACS Appl. Mater. Interfaces, 2015, 7, 14303–14316 Search PubMed.
- N. Hongsith, T. Chairuangsri, T. Phaechamud and S. Choopun, Growth kinetic and characterization of tetrapod ZnO nanostructures, Solid State Commun., 2009, 149, 1184–1187 Search PubMed.
- G. Modi, Zinc oxide tetrapod: a morphology with multifunctional applications, Adv. Nat. Sci.: Nanosci. Nanotechnol., 2015, 6, 33002 Search PubMed.
- H. Iwanaga, M. Fujii and S. Takeuchi, Inter-leg angles in tetrapod ZnO particles, J. Cryst. Grow., 1998, 183, 190–195 Search PubMed.
- H. Iwanaga, M. Fujii, M. Ichihara and S. Takeuchi, Some evidence for the octa-twin model of tetrapod ZnO particles, J. Cryst. Grow., 1994, 141, 234–238 CrossRef CAS.
- M. S. Riahimadvar and M. Tajaldini, Fast and one-step synthesis of small ZnO nano-tetrapods Using CO 2 laser in ambient air: physical properties, Phys. Scr., 2022, 97, 105811 Search PubMed.
- S. Rackauskas, K. Mustonen, T. Järvinen, M. Mattila, O. Klimova, H. Jiang, O. Tolochko, H. Lipsanen, E. I. Kauppinen and A. G. Nasibulin, Synthesis of ZnO tetrapods for flexible and transparent UV sensors, Nanotechnology, 2012, 23, 95502 Search PubMed.
- B.-B. Wang, J.-J. Xie, Q. Yuan and Y.-P. Zhao, Growth mechanism and joint structure of ZnO tetrapods, J. Phys. D: Appl. Phys., 2008, 41, 102005 Search PubMed.
- D. B. Padmanaban, P. Maguire and D. Mariotti, Non-equilibrium defect chemistry in oxygen-rich zinc oxide nano-tetrapods synthesized using atmospheric pressure microplasma, J. Mater. Chem. A, 2024, 12, 9212–9231 RSC.
- Y. Qiu and S. Yang, ZnO Nanotetrapods: Controlled Vapor-Phase Synthesis and Application for Humidity Sensing, Adv. Funct. Mater., 2007, 17, 1345–1352 CrossRef CAS.
- R. B. Saunders, E. McGlynn and M. O. Henry, Theoretical Analysis of Nucleation and Growth of ZnO Nanostructures in Vapor Phase Transport Growth, Cryst. Growth Des., 2011, 11, 4581–4587 CrossRef CAS.
- A. F. Gouveia, S. C. S. Lemos, E. R. Leite, E. Longo and J. Andrés, Back to the Basics: Probing the Role of Surfaces in the Experimentally Observed Morphological Evolution of ZnO, Nanomaterials, 2023, 13(6), 978 CrossRef CAS PubMed.
- K. Zheng, C. X. Xu, G. P. Zhu, X. Li, J. P. Liu, Y. Yang and X. W. Sun, Formation of tetrapod and multipod ZnO whiskers, Phys. E, 2008, 40, 2677–2681 CrossRef CAS.
- S. Rackauskas, O. Klimova, H. Jiang, A. Nikitenko, K. A. Chernenko, S. D. Shandakov, E. I. Kauppinen, O. V. Tolochko and A. G. Nasibulin, A Novel Method for Continuous Synthesis of ZnO Tetrapods, J. Phys. Chem. C, 2015, 119, 16366–16373 CrossRef CAS.
- E. M. Wong, J. E. Bonevich and P. C. Searson, Growth kinetics of nanocrystalline ZnO particles from colloidal suspensions, J. Phys. Chem. B, 1998, 102, 7770–7775 CrossRef CAS.
- Y. Liu, G. Lu, L. Guo, Y. Zhang and M. Chen, Flexible Pressure Sensor Based on Tetrapod-Shaped ZnO-PDMS Piezoelectric Nanocomposites, IEEE Sens. J., 2023, 23, 3532–3540 CAS.
- S. Shree, V. Postica, L. Voß, C. Lupan, Y. K. Mishra, L. Kienle, R. Adelung and O. Lupan, Optimization of T-ZnO Process for Gas and UV Sensors, ACS Appl. Electron. Mater., 2025, 7, 3848–3863 Search PubMed.
- D. Yang, R. A. Gopal, T. Lkhagvaa and D. Choi, Oxidizing agent impacting on growth of ZnO tetrapod nanostructures and its characterization, Environ. Res., 2021, 197, 111032 CrossRef CAS PubMed.
- H. Yamamoto, Y. Otani, T. Seto, P. Nartpochananon and T. Charinpanitkul, Generation of uniform tetrapod-shaped zincoxide nanoparticles by gas-phase reaction with using flow restrictor, Adv. Powder Technol., 2012, 23, 71–79 CrossRef CAS.
- A. Krost, J. Christen, N. Oleynik, A. Dadgar, S. Deiter, J. Bläsing, A. Krtschil, D. Forster, F. Bertram and A. Diez, Ostwald ripening and flattening of epitaxial ZnO layers during in situ annealing in metalorganic vapor phase epitaxy, Appl. Phys. Lett., 2004, 85, 1496–1498 CrossRef CAS.
- Z. Zhou, J. Liu and S. Hu, Studies on the kinetics process of tetra-needle-like ZnO whisker growth, J. Cryst. Grow., 2005, 276, 317–320 CrossRef CAS.
- M. Ali and M. Winterer, Influence of nucleation rate on the yield of ZnO nanocrystals prepared by chemical vapor synthesis, J. Phys. Chem. C, 2010, 114, 5721–5726 CrossRef CAS.
- E. S. Babu, S. Kim, J.-H. Song and S.-K. Hong, Effects of growth pressure on morphology of ZnO nanostructures by chemical vapor transport, Chem. Phys. Lett., 2016, 658, 182–187 Search PubMed.
- R. P. Sugavaneshwar and K. K. Nanda, Uninterrupted and reusable source for the controlled growth of nanowires, Sci. Rep., 2013, 3, 1172 CrossRef CAS PubMed.
- N. Martin, V. Lacour, C. M.-T. Perrault, E. Roy and Y. Leprince-Wang, High flow rate microreactors integrating in situ grown ZnO nanowires for photocatalytic degradation, React. Chem. Eng., 2022, 7, 750–757 RSC.
- S. L. Yang, R. S. Gao, B. Yang, P. L. Niu and R. H. Yu, Morphology-controlled growth of tetrapod ZnO nanostructures by direct arc discharge, Appl. Phys. A: Mater. Sci. Process., 2010, 99, 9–13 CrossRef CAS.
- F. Orudzhev, A. Muslimov, D. Selimov, R. R. Gulakhmedov, A. Lavrikov, V. Kanevsky, R. Gasimov, V. Krasnova and D. Sobola, Oxygen vacancies and surface wettability: key factors in activating and enhancing the solar photocatalytic activity of ZnO tetrapods, Int. J. Mol. Sci., 2023, 24, 16338 Search PubMed.
- K. Balasubramanian, A. K. Pandey, R. Abolhassani, H.-G. Rubahn, S. Rahman and Y. K. Mishra, Tetrapods based engineering of organic phase change material for thermal energy storage, Chem. Eng. J., 2023, 462, 141984 CrossRef CAS.
- Q. Wan, K. Yu, T. H. Wang and C. L. Lin, Low-field electron emission from tetrapod-like ZnO nanostructures synthesized by rapid evaporation, Appl. Phys. Lett., 2003, 83, 2253–2255 CrossRef CAS.
- N. Babayevska, Ł. Przysiecka, I. Iatsunskyi, G. Nowaczyk, M. Jarek, E. Janiszewska and S. Jurga, ZnO size and shape effect on antibacterial activity and cytotoxicity profile, Sci. Rep., 2022, 12, 8148 CrossRef CAS PubMed.
- G.-W. Hsieh, S.-R. Ling, F.-T. Hung, P.-H. Kao and J.-B. Liu, Enhanced piezocapacitive response in zinc oxide tetrapod-poly(dimethylsiloxane) composite dielectric layer for flexible and ultrasensitive pressure sensor, Nanoscale, 2021, 13, 6076–6086 RSC.
- M. Ilickas, M. Marčinskas, D. Peckus, R. Mardosaitė, B. Abakevičienė, T. Tamulevičius and S. Račkauskas, ZnO UV sensor photoresponse enhancement by coating method optimization, J. Photochem. Photobiol., 2023, 14, 100171 CrossRef CAS.
- L. Lazzarini, G. Salviati, F. Fabbri, M. Zha, D. Calestani, A. Zappettini, T. Sekiguchi and B. Dierre, Unpredicted nucleation of extended zinc blende phases in wurtzite ZnO nanotetrapod arms, ACS Nano, 2009, 3, 3158–3164 CrossRef CAS PubMed.
- F. Z. Wang, Z. Z. Ye, D. W. Ma, L. P. Zhu and F. Zhuge, Novel morphologies of ZnO nanotetrapods, Mater. Lett., 2005, 59, 560–563 CrossRef CAS.
- G.-H. Lee, Effect of annealing temperature on the morphology and optical properties of ZnO tetrapods grown by a thermal evaporation technique, Ceram. Int., 2016, 42, 14452–14455 CrossRef CAS.
- F. Liu, P. J. Cao, H. R. Zhang, J. Q. Li and H. J. Gao, Controlled self-assembled nanoaeroplanes, nanocombs, and tetrapod-like networks of zinc oxide, Nanotechnology, 2004, 15, 949–952 Search PubMed.
- G. Zhao, L. Xia, S. Wu, L. Song, A. Wei and G. Wen, Ultrafast and mass production of ZnO nanotetrapods by induction-heating under air ambient, Mater. Lett., 2014, 118, 126–129 Search PubMed.
- H. Papavlassopoulos, Y. K. Mishra, S. Kaps, I. Paulowicz, R. Abdelaziz, M. Elbahri, E. Maser, R. Adelung and C. Röhl, Toxicity of functional nano-micro zinc oxide tetrapods: impact of cell culture conditions, cellular age and material properties, PLoS One, 2014, 9, e84983 CrossRef PubMed.
- Y. K. Mishra, S. Kaps, A. Schuchardt, I. Paulowicz, X. Jin, D. Gedamu, S. Freitag, M. Claus, S. Wille, A. Kovalev, S. N. Gorb and R. Adelung, Fabrication of Macroscopically Flexible and Highly Porous 3D Semiconductor Networks from Interpenetrating Nanostructures by a Simple Flame Transport Approach, Part. Part. Syst. Charact., 2013, 30, 775–783 CrossRef CAS.
- L. I. Istomina, K. A. Sakharov, E. I. Anosova, S. G. Protasova, A. M. Ionov, A. S. Starikova, A. V. Ilyakova, Y. A. Zakharova and S. V. Andreev, New method of synthesizing ZnO/ZnO2 particles of different morphologies in hydrogen peroxide vapor, Mater. Chem. Phys., 2025, 131810 Search PubMed.
- J. Gröttrup, I. Paulowicz, A. Schuchardt, V. Kaidas, S. Kaps, O. Lupan, R. Adelung and Y. K. Mishra, Three-dimensional flexible ceramics based on interconnected network of highly porous pure and metal alloyed ZnO tetrapods, Ceram. Int., 2016, 42, 8664–8676 CrossRef.
- F. Xu and S. Tse, Flame Synthesis of Zinc Oxide Nanostructures, 2012 DOI:10.2514/6.2008-1454.
- L. Chen, W. Song, C. Xie, L. Lin and J. Wang, A novel combustion method to prepare tetrapod nano-ZnO, Mater. Lett., 2007, 61, 4603–4605 CrossRef CAS.
- C.-C. Hwang, C.-S. Lin, G.-P. Wang, C.-H. Peng and S.-L. Chung, A self-propagating high-temperature synthesis method for synthesis of zinc oxide powder, J. Alloys Compd., 2009, 467, 514–523 CrossRef CAS.
- T.-L. Phan, V. Le Cuong, V. D. Lam and N. T. Dang, Various CVD-grown ZnO nanostructures for nanodevices and interdisciplinary applications, Beilstein J. Nanotechnol., 2024, 15, 1390–1399 CrossRef CAS PubMed.
- F. H. Alsultany, Z. Hassan and N. M. Ahmed, Large-scale uniform ZnO tetrapods on catalyst free glass substrate by thermal evaporation method, Mater. Res. Bull., 2016, 79, 63–68 CrossRef CAS.
- S. A. Al Rifai and E. P. Domashevskaya, The synthesis and optical properties of different zinc oxide nanostructures, Russ. J. Phys. Chem., 2013, 87, 2246–2252 CrossRef CAS.
- Y. Qiu, L. Hu, H. Zhang, L. Wang, J. Ma and B. Wang, Changing the Shape of ZnO Nanostructures by Controlling Oxygen Concentration: From Tetrapod-Like to Dandelion-Like Nanostructures, Integr. Ferroelectr., 2011, 127, 134–140 CrossRef CAS.
- K.-S. Yun, K.-I. Ri, Y.-G. Choe, K.-U. Kim and C.-H. Jang, Novel hierarchical structure of ZnO: Growth of spiny tetrapod via vapor phase synthesis, Funct. Mater. Lett., 2025, 18(7), 2551067 CrossRef CAS.
- W. Yu, X. Li and X. Gao, Catalytic Synthesis and Structural Characteristics of High-Quality Tetrapod-Like ZnO Nanocrystals by a Modified Vapor Transport Process, Cryst. Growth Des., 2005, 5, 151–155 CrossRef CAS.
- H. Li, Y. Huang, Y. Zhang, J. Qi, X. Yan, Q. Zhang and J. Wang, Self-catalytic Synthesis, Structures, and Properties of High-Quality Tetrapod-Shaped ZnO Nanostructures, Cryst. Growth Des., 2009, 9, 1863–1868 CrossRef CAS.
- S. L. More, M. Kovochich, T. Lyons-Darden, M. Taylor, A. M. Schulte and A. K. Madl, Review and Evaluation of the Potential Health Effects of Oxidic Nickel Nanoparticles, Nanomaterials, 2021, 11(3), 642 CrossRef CAS PubMed.
- X.-H. Zhang, S.-Y. Xie, Z.-Y. Jiang, Z.-X. Xie, R.-B. Huang, L.-S. Zheng, J.-Y. Kang and T. Sekiguchi, Microwave plasma growth and high spatial resolution cathodoluminescent spectrum of tetrapod ZnO nanostructures, J. Solid State Chem., 2003, 173, 109–113 CrossRef CAS.
- H.-F. Lin, S.-C. Liao and C.-T. Hu, A new approach to synthesize ZnO tetrapod-like nanoparticles with DC thermal plasma technique, J. Cryst. Grow., 2009, 311, 1378–1384 Search PubMed.
- B.-J. Lee, S.-I. Jo and G.-H. Jeong, Synthesis of ZnO Nanomaterials Using Low-Cost Compressed Air as Microwave Plasma Gas at Atmospheric Pressure, Nanomaterials, 2019, 9(7), 942 CrossRef CAS PubMed.
- J.-Y. Yang and G.-H. Jeong, Synthesis of ZnO Tetrapods Using Atmospheric Plasma Jet and Their Size-Dependent Photosensing Properties, Appl. Sci. Converg. Technol., 2022, 31, 149–152 CrossRef.
- A. M. Schwan, S. Chwatal, C. Hendler, D. Kopp, J. M. Lackner, R. Kaindl, M. Tscherner, M. Zirkl, P. Angerer, B. Friessnegger, S. Augl, D. Heim, A. Hinterer, M. Stummer and W. Waldhauser, Morphology-controlled atmospheric pressure plasma synthesis of zinc oxide nanoparticles for piezoelectric sensors, Appl. Nanosci., 2023, 13, 6421–6432 CrossRef CAS.
- O. Lupan, L. Chow, G. Chai, B. Roldan, A. Naitabdi, A. Schulte and H. Heinrich, Nanofabrication and characterization of ZnO nanorod arrays and branched microrods by aqueous solution route and rapid thermal processing, Mater. Sci. Eng., B, 2007, 145, 57–66 CrossRef CAS.
- O. Lupan, G. Chai and L. Chow, Novel hydrogen gas sensor based on single ZnO nanorod, Microelectron. Eng., 2008, 85, 2220–2225 CrossRef CAS.
- H. Zhang, D. Yang, D. Li, X. Ma, S. Li and D. Que, Controllable Growth of ZnO Microcrystals by a Capping-Molecule-Assisted Hydrothermal Process, Cryst. Growth Des., 2005, 5, 547–550 CrossRef CAS.
- J. Jiang, Y. Li, S. Tan and Z. Huang, Synthesis of zinc oxide nanotetrapods by a novel fast microemulsion-based hydrothermal method, Mater. Lett., 2010, 64, 2191–2193 CrossRef CAS.
- Y. Yang, Y. Jin, H. He, Q. Wang, Y. Tu, H. Lu and Z. Ye, Dopant-induced shape evolution of colloidal nanocrystals: the case of zinc oxide, J. Am. Chem. Soc., 2010, 132, 13381–13394 CrossRef CAS PubMed.
- M. Chen and D. A. Dixon, Machine-Learning Approach for the Development of Structure–Energy Relationships of ZnO Nanoparticles, J. Phys. Chem. C, 2018, 122, 18621–18639 CrossRef CAS.
- A. Dmytruk, I. Dmitruk, Y. Shynkarenko, R. Belosludov and A. Kasuya, ZnO nested shell magic clusters as tetrapod nuclei, RSC Adv., 2017, 7, 21933–21942 RSC.
- R. Yuksel, O. Buyukcakir, P. K. Panda, S. H. Lee, Y. Jiang, D. Singh, S. Hansen, R. Adelung, Y. K. Mishra, R. Ahuja and R. S. Ruoff, Necklace-like Nitrogen-Doped Tubular Carbon 3D Frameworks for Electrochemical Energy Storage, Adv. Funct. Mater., 2020, 30, 1909725 CrossRef CAS.
- M. Ilickas, R. Mardosaite, F. Cesano, S. Cravanzola, C. Barolo, D. Scarano, G. Viscardi and S. Rackauskas, ZnO tetrapod morphology influence on UV sensing properties, Nanotechnology, 2023, 35, 15502 CrossRef PubMed.
- O. Lupan, V. Postica, J. Gröttrup, A. K. Mishra, N. H. de Leeuw, J. F. C. Carreira, J. Rodrigues, N. Ben Sedrine, M. R. Correia and T. Monteiro, Hybridization of zinc oxide tetrapods for selective gas sensing applications, ACS Appl. Mater. Interfaces, 2017, 9, 4084–4099 CrossRef CAS PubMed.
- J. Rodrigues, N. Ben Sedrine, M. R. Correia and T. Monteiro, Photoluminescence investigations of ZnO micro/nanostructures, Mater. Today Chem., 2020, 16, 100243 CrossRef CAS.
- K. Yan, L. Zhang, J. Qiu, Y. Qiu, Z. Zhu, J. Wang and S. Yang, A quasi-quantum well sensitized solar cell with accelerated charge separation and collection, J. Am. Chem. Soc., 2013, 135, 9531–9539 CrossRef CAS PubMed.
- Y. Qiu, K. Yan, H. Deng and S. Yang, Secondary branching and nitrogen doping of ZnO nanotetrapods: building a highly active network for photoelectrochemical water splitting, Nano Lett., 2012, 12, 407–413 CrossRef CAS PubMed.
- T. Santhaveesuk, D. Wongratanaphisan and S. Choopun, Enhancement of sensor response by TiO2 mixing and Au coating on ZnO tetrapod sensor, Sens. Actuators, B, 2010, 147, 502–507 CrossRef CAS.
- N. Roy and A. Roy, Growth and temperature dependent photoluminescence characteristics of ZnO tetrapods, Ceram. Int., 2015, 41, 4154–4160 CrossRef CAS.
- D. Debnath, D. Sen, T. T. Neog, B. Saha and S. K. Ghosh, Growth of ZnO Polytypes: Multiple Facets of Diverse Applications, Cryst. Growth Des., 2024, 24, 871–885 CrossRef CAS.
- K. Wang, J.-N. Ma, C.-Y. Zhang, Z. Pei, W.-T. Tang and Q. Zhang, Self-powered high-sensitivity piezoelectric sensors for end-fixture force sensing in surgical robots based on T-ZnO, Colloids Surf., A, 2024, 697, 134424 CrossRef CAS.
- Z. Zhang, M. Willatzen, Y. K. Mishra and Z. L. Wang, Transport studies in piezo-semiconductive ZnO nanotetrapod based electronic devices, Mater. Today Electron., 2024, 8, 100102 CrossRef.
- A. Janotti and C. G. van de Walle, Fundamentals of zinc oxide as a semiconductor, Rep. Prog. Phys., 2009, 72, 126501 CrossRef.
- M. Villani, F. Rossi, D. Calestani, G. Salviati and F. Fabbri, Evaluating the plasmon-exciton interaction in ZnO tetrapods coupled with gold nanostructures by nanoscale cathodoluminescence, Nano Express, 2021, 2, 14004 CrossRef CAS.
- W. D. Yu, X. M. Li and X. D. Gao, Homogeneous-catalytic synthesis of tetrapodlike ZnO nanocrystals and their photoluminescence properties, Chem. Phys. Lett., 2004, 390, 296–300 CrossRef CAS.
- L. Manna, D. J. Milliron, A. Meisel, E. C. Scher and A. P. Alivisatos, Controlled growth of tetrapod-branched inorganic nanocrystals, Nat. Mater., 2003, 2, 382–385 CrossRef CAS PubMed.
- R. G. S. Pala and H. Metiu, Modification of the Oxidative Power of ZnO(10
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) Surface by Substituting Some Surface Zn Atoms with Other Metals, J. Phys. Chem. C, 2007, 111, 8617–8622 CrossRef CAS.
0) Surface by Substituting Some Surface Zn Atoms with Other Metals, J. Phys. Chem. C, 2007, 111, 8617–8622 CrossRef CAS. - C. Klingshirn, ZnO: From basics towards applications, Phys. Status Solidi B, 2007, 244, 3027–3073 CrossRef CAS.
- M. P. M. Poschmann, L. Siebert, C. Lupan, O. Lupan, F. Schütt, R. Adelung and N. Stock, Surface Conversion of ZnO Tetrapods Produces Pinhole-Free ZIF-8 Layers for Selective and Sensitive H2 Sensing Even in Pure Methane, ACS Appl. Mater. Interfaces, 2023, 15, 38674–38681 CrossRef CAS PubMed.
- M. Todorova and J. Neugebauer, Identification of bulk oxide defects in an electrochemical environment, Faraday Discuss., 2015, 180, 97–112 RSC.
- M. Li and E. G. Seebauer, Defect engineering in semiconducting oxides: Control of ZnO surface potential via temperature and oxygen pressure, AIChE J., 2016, 62, 500–507 CrossRef CAS.
- C. G. van de Walle, Defect analysis and engineering in ZnO, Phys. B, 2001, 308–310, 899–903 CrossRef CAS.
- T. S. Bjørheim and E. Kotomin, Ab Initio Thermodynamics of Oxygen Vacancies and Zinc Interstitials in ZnO, J. Phys. Chem. Lett., 2014, 5, 4238–4242 CrossRef PubMed.
- M. Brás, J. Zanoni, B. P. Falcão, J. P. Leitão, F. M. Costa, T. Monteiro, S. O. Pereira and J. Rodrigues, Label-Free Nanoscale ZnO Tetrapod-Based Transducers for Tetracycline Detection, ACS Appl. Nano Mater., 2022, 5, 1232–1243 CrossRef.
- M. S. Riahimadvar and M. Tajaldini, Fast and one-step synthesis of small ZnO nano-tetrapods Using CO2 laser in ambient air: physical properties, Phys. Scr., 2022, 97, 105811 CrossRef CAS.
- Y. Qiu, M. Yang, H. Fan, Y. Xu, Y. Shao, X. Yang and S. Yang, Synthesis and characterization of nitrogen doped ZnO tetrapods and application in photocatalytic degradation of organic pollutants under visible light, Mater. Lett., 2013, 99, 105–107 CrossRef CAS.
- R. Mardosaitė, A. Jurkevičiūtė and S. Račkauskas, Superhydrophobic ZnO Nanowires: Wettability Mechanisms and Functional Applications, Cryst. Growth Des., 2021, 21, 4765–4779 CrossRef.
- H. Ennaceri, L. Wang, D. Erfurt, W. Riedel, G. Mangalgiri, A. Khaldoun, A. El Kenz, A. Benyoussef and A. Ennaoui, Water-resistant surfaces using zinc oxide structured nanorod arrays with switchable wetting property, Surf. Coat. Technol., 2016, 299, 169–176 CrossRef CAS.
- M. T. Z. Myint, N. S. Kumar, G. L. Hornyak and J. Dutta, Hydrophobic/hydrophilic switching on zinc oxide micro-textured surface, Appl. Surf. Sci., 2013, 264, 344–348 CrossRef CAS.
- J. Li, Q. Sun, S. Han, J. Wang, Z. Wang and C. Jin, Reversibly light-switchable wettability between superhydrophobicity and superhydrophilicity of hybrid ZnO/bamboo surfaces via alternation of UV irradiation and dark storage, Prog. Org. Coat., 2015, 87, 155–160 CrossRef CAS.
- F. Oba, S. R. Nishitani, S. Isotani, H. Adachi and I. Tanaka, Energetics of native defects in ZnO, J. Appl. Phys., 2001, 90, 824–828 CrossRef CAS.
- Y. Xu, Y. Yang, D.-X. Yan, H. Duan, G. Zhao and Y. Liu, Flexible and conductive polyurethane composites for electromagnetic shielding and printable circuit, Chem. Eng. J., 2019, 360, 1427–1436 CrossRef CAS.
- C. Klingshirn, ZnO: material, physics and applications, Chem. Phys. Chem., 2007, 8, 782–803 CrossRef CAS PubMed.
- S. Xu, Y. Qin, C. Xu, Y. Wei, R. Yang and Z. L. Wang, Self-powered nanowire devices, Nat. Nanotechnol., 2010, 5, 366–373 CrossRef CAS PubMed.
- R. K. Mishra, V. Kumar, G. Le Trung, G. J. Choi, J. W. Ryu, R. Bhardwaj, P. Kumar, J. Singh, S. H. Lee and J. S. Gwag, Recent advances in ZnO nanostructure as a gas-sensing element for an acetone sensor: a short review, Luminescence, 2023, 38, 1087–1101 CrossRef CAS PubMed.
- A. Tamulevičienė, R. Mardosaitė, M. Ilickas, B. Abakevičienė, T. Tamulevičius, Š. Meškinis and S. Račkauskas, Highly-hydrophobic, transparent, and durable coatings based on ZnO tetrapods with diamond-like carbon nanocomposite, Surf. Coat. Technol., 2023, 470, 129863 CrossRef.
- X. Tao, H. Jin, M. Mintken, N. Wolff, Y. Wang, R. Tao, Y. Li, H. Torun, J. Xie, J. Luo, J. Zhou, Q. Wu, S. Dong, J. Luo, L. Kienle, R. Adelung, Y. K. Mishra and Y. Q. Fu, Three-Dimensional Tetrapodal ZnO Microstructured Network Based Flexible Surface Acoustic Wave Device for Ultraviolet and Respiration Monitoring Applications, ACS Appl. Nano Mater., 2020, 3, 1468–1478 CrossRef CAS.
- D. Gedamu, I. Paulowicz, S. Kaps, O. Lupan, S. Wille, G. Haidarschin, Y. K. Mishra and R. Adelung, Rapid fabrication technique for interpenetrated ZnO nanotetrapod networks for fast UV sensors, Adv. Mater., 2014, 26, 1541–1550 CrossRef CAS PubMed.
- R. Chen, B. Ling, X. W. Sun and H. D. Sun, Room temperature excitonic whispering gallery mode lasing from high-quality hexagonal ZnO microdisks, Adv. Mater., 2011, 23, 2199–2204 CrossRef CAS PubMed.
- R. Rathnasekara, G. Mayberry and P. Hari, Thermoelectric, electrochemical, & dielectric properties of four ZnO nanostructures, Materials, 2022, 15, 8816 CrossRef CAS PubMed.
- H. Zhao, B. Ouyang, L. Han, Y. K. Mishra, Z. Zhang and Y. Yang, Conjuncted photo-thermoelectric effect in ZnO–graphene nanocomposite foam for self-powered simultaneous temperature and light sensing, Sci. Rep., 2020, 10, 11864 CrossRef CAS PubMed.
- X. Liang and C. Wang, Electron and phonon transport anisotropy of ZnO at and above room temperature, Appl. Phys. Lett., 2020, 116(4), 043903 CrossRef CAS.
- M. Ahmad, C. Pan, J. Zhao, J. Iqbal and J. Zhu, Electron irradiation effect and photoluminescence properties of ZnO-tetrapod nanostructures, Mater. Chem. Phys., 2010, 120, 319–322 CrossRef CAS.
- A. R. Hutson, Electronic properties of ZnO, J. Phys. Chem. Solids, 1959, 8, 467–472 CrossRef CAS.
- T. Makino, Y. Segawa, A. Tsukazaki, A. Ohtomo and M. Kawasaki, Electron transport in ZnO thin films, Appl. Phys. Lett., 2005, 87(2), 022101 Search PubMed.
- J. Huh, G.-T. Kim, J. S. Lee and S. Kim, A direct measurement of the local resistances in a ZnO tetrapod by means of impedance spectroscopy: The role of the junction in the overall resistance, Appl. Phys. Lett., 2008, 93(4), 042111 Search PubMed.
- G. Blatter and F. Greuter, Carrier transport through grain boundaries in semiconductors, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 3952 Search PubMed.
- A. Janotti and C. G. van de Walle, Native point defects in ZnO, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 165202 Search PubMed.
- R. A. Fernandes, M. J. Sampaio, E. S. Da Silva, P. Serp, J. L. Faria and C. G. Silva, Synthesis of selected aromatic aldehydes under UV-LED irradiation over a hybrid photocatalyst of carbon nanofibers and zinc oxide, Catal. Today, 2019, 328, 286–292 Search PubMed.
- Q. Zhang, W. Fan and L. Gao, Anatase TiO2 nanoparticles immobilized on ZnO tetrapods as a highly efficient and easily recyclable photocatalyst, Appl. Catal., B, 2007, 76, 168–173 Search PubMed.
- J. Chebwogen, F. W. Nyongesa, J. M. Mwabora, G. M. Ntshani and P. G. Ndungu, A Comparative Study of Sol–Gel Synthesized ZnO/GO and ZnO/g-C3N4 Nanocomposites in the Photocatalytic Degradation of Acetaminophen, Int. J. Photoenergy, 2025, 2025, 9216858 Search PubMed.
- J. Liu, M. Li, C. Luo, S. Zhou and W. Chen, Eco-friendly synthesis of self-reporting robust superhydrophobic coatings with damage sensitive photoluminescence, Chem. Eng. J., 2022, 431, 134162 Search PubMed.
- C. Li, B. Lee, C. Wang, A. Bajpayee, L. D. Douglas, B. K. Phillips, G. Yu, N. Rivera-Gonzalez, B.-J. Peng, Z. Jiang, H.-J. Sue, S. Banerjee and L. Fang, Photopolymerized superhydrophobic hybrid coating enabled by dual-purpose tetrapodal ZnO for liquid/liquid separation, Mater. Horiz., 2022, 9, 452–461 Search PubMed.
- T. Reimer, I. Paulowicz, R. Röder, S. Kaps, O. Lupan, S. Chemnitz, W. Benecke, C. Ronning, R. Adelung and Y. K. Mishra, Single step integration of ZnO nano- and microneedles in Si trenches by novel flame transport approach: whispering gallery modes and photocatalytic properties, ACS Appl. Mater. Interfaces, 2014, 6, 7806–7815 CrossRef CAS PubMed.
- F. Sun, L. Sun, B. Zhang and H. Wang, Spatially resolved surface-related exciton polariton dynamics in a single ZnO tetrapod, Solid State Commun., 2018, 270, 107–110 Search PubMed.
- S.-K. Lee, W. M. Chen, D. Hongxing, Z. Chen and I. A. Buyanova, Cathodoluminescence characterization of ZnO tetrapod structures, Thin Solid Films, 2013, 543, 114–117 CrossRef CAS.
- G. Bertoni, F. Fabbri, M. Villani, L. Lazzarini, S. Turner, G. van Tendeloo, D. Calestani, S. Gradečak, A. Zappettini and G. Salviati, Nanoscale mapping of plasmon and exciton in ZnO tetrapods coupled with Au nanoparticles, Sci. Rep., 2016, 6, 19168 CrossRef CAS PubMed.
- A. E. Muslimov, V. V. Krasnova, L. Zadorozhnaya and V. M. Kanevsky, Influence of Morphological and Dimensional Parameters on Cathodoluminescent Properties of ZNO Microstructure Ensemble, J. Surf. Investig., 2024, 18, 1640–1644 CrossRef CAS.
- L. A. Zadorozhnaya, A. P. Tarasov and V. M. Kanevsky, The Growth Mechanism, Luminescence, and Lasing of Polyhedral ZnO Microcrystals with Whispering-Gallery Modes, Photonics, 2023, 10(12), 1328 CrossRef CAS.
- I. Tiginyanu, L. Ghimpu, J. Gröttrup, V. Postolache, M. Mecklenburg, M. A. Stevens-Kalceff, V. Ursaki, N. Payami, R. Feidenhansl, K. Schulte, R. Adelung and Y. K. Mishra, Strong light scattering and broadband (UV to IR) photoabsorption in stretchable 3D hybrid architectures based on Aerographite decorated by ZnO nanocrystallites, Sci. Rep., 2016, 6, 32913 CrossRef CAS PubMed.
- K. Vanheusden, C. H. Seager, W. T. Warren and J. A. Voigt, Correlation between photoluminescence and oxygen vacancies in ZnO phosphors, Appl. Phys. Lett., 1996, 68, 403–405 CrossRef CAS.
- K. Vanheusden, W. L. Warren, C. H. Seager, J. A. Voigt and B. E. Gnade, Mechanisms behind green photoluminescence in ZnO phosphor powders, J. Appl. Phys., 1996, 79, 7983–7990 CrossRef CAS.
- F. A. La Porta, J. Andres, M. V. Vismara, C. Frederico de Oliveira, J. R. Sambrano, M. S. Li, J. A. Varela and E. Longo, Correlation between structural and electronic order–disorder effects and optical properties in ZnO nanocrystals, J. Mater. Chem. C, 2014, 2, 10164–10174 RSC.
- Z. G. Yu, P. Wu and H. Gong, Theoretical and experimental studies on oxygen vacancy in p-type ZnO, Phys. B, 2007, 401, 417–420 CrossRef.
- I. Shalish, H. Temkin and V. Narayanamurti, Size-dependent surface luminescence in ZnO nanowires, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 69, 245401 CrossRef.
- J.-P. Richters, T. Voss, L. Wischmeier, I. Rückmann and J. Gutowski, Influence of polymer coating on the low-temperature photoluminescence properties of ZnO nanowires, Appl. Phys. Lett., 2008, 92(1), 011103 CrossRef.
- F. Leiter, H. Zhou, F. Henecker, A. Hofstaetter, D. M. Hofmann and B. K. Meyer, Magnetic resonance experiments on the green emission in undoped ZnO crystals, Phys. B, 2001, 308, 908–911 CrossRef.
- M. R. C. Sytu and J.-I. Hahm, Principles and Applications of ZnO Nanomaterials in Optical Biosensors and ZnO Nanomaterial-Enhanced Biodetection, Biosensors, 2024, 14(10), 480 CrossRef CAS PubMed.
- N. K. Hassan, M. R. Hashim and N. K. Allam, ZnO nano-tetrapod photoanodes for enhanced solar-driven water splitting, Chem. Phys. Lett., 2012, 549, 62–66 CrossRef CAS.
- M. Villani, F. Rossi, D. Calestani, G. Salviati and F. Fabbri, Evaluating the plasmon-exciton interaction in ZnO tetrapods coupled with gold nanostructures by nanoscale cathodoluminescence, Nano Ex., 2021, 2, 14004 CrossRef CAS.
- W. Chen, Y. Qiu and S. Yang, Branched ZnO nanostructures as building blocks of photoelectrodes for efficient solar energy conversion, Phys. Chem. Chem. Phys., 2012, 14, 10872–10881 RSC.
- L. Juan, Y. Chenxi, N. Ziru, Y. Shenglan and H. Dongwen, Fabrication of flexible photo-responsive mesh with reversible wettability and enhanced mechanical damage resistance for efficient oil/water separation, J. Environ. Chem. Eng., 2024, 12, 114857 CrossRef.
- Y. Zhang, H. Zhou, S. W. Liu, Z. R. Tian and M. Xiao, Second-harmonic whispering-gallery modes in ZnO nanotetrapod, Nano Lett., 2009, 9, 2109–2112 CrossRef CAS PubMed.
- D. Jiang, C. Tian, Q. Liu, M. Zhao, J. Qin, J. Hou, S. Gao, Q. Liang and J. Zhao, Young's modulus of individual ZnO nanowires, Mater. Sci. Eng., A, 2014, 610, 1–4 Search PubMed.
- F. Muktepavela, L. Grigorjeva, K. Kundzins, E. Gorokhova and P. Rodnyi, Structure, nanohardness and photoluminescence of ZnO ceramics based on nanopowders, Phys. Scr., 2015, 90, 94018 Search PubMed.
- N. Zhang and M. Asle Zaeem, Nanoscale flaw tolerance behaviour of polycrystalline tetragonal zirconia nanopillars, Int. J. Mech. Sci., 2020, 173, 105405 CrossRef.
- Z. Qin and M. J. Buehler, Flaw Tolerance of Nuclear Intermediate Filament Lamina under Extreme Mechanical Deformation, ACS Nano, 2011, 5, 3034–3042 Search PubMed.
- M.-R. He, P. Xiao, J. Zhao, S. Dai, F. Ke and J. Zhu, Quantifying the defect-dominated size effect of fracture strain in single crystalline ZnO nanowires, J. Appl. Phys., 2011, 109(12), 123504 Search PubMed.
- C. Q. Chen and J. Zhu, Bending strength and flexibility of ZnO nanowires, Appl. Phys. Lett., 2007, 90(4), 043105 CrossRef.
- R. Meija, S. Signetti, A. Schuchardt, K. Meurisch, D. Smazna, M. Mecklenburg, K. Schulte, D. Erts, O. Lupan, B. Fiedler, Y. K. Mishra, R. Adelung and N. M. Pugno, Nanomechanics of individual aerographite tetrapods, Nat. Commun., 2017, 8, 14982 Search PubMed.
- E. Veys, L. Makower, M. Williamson, L. M. Saure, R. Adelung, F. Schütt, N. M. Pugno and T. J. Marrow, In situ observation of compressive deformation of an interconnected network of zinc oxide tetrapods, Scr. Mater., 2023, 224, 115153 CrossRef CAS.
- T. Spratte, C. Arndt, I. Wacker, M. Hauck, R. Adelung, R. R. Schröder, F. Schütt and C. Selhuber-Unkel, Thermoresponsive Hydrogels with Improved Actuation Function by Interconnected Microchannels, Adv. Intell. Syst., 2022, 4(3), 2100081 Search PubMed.
- N. Liu, L. M. Saure, R. Sriramdas, F. Schütt, K. Wang, A. Nozariasbmarz, Y. Zhang, R. Adelung, R. H. Baughman, S. Priya, W. Li and B. Poudel, Underwater Thermoacoustic Generation by a Hierarchical Tetrapodal Carbon Nanotube Network, ACS Nano, 2024, 18, 8988–8995 CrossRef CAS PubMed.
- F. Schütt, M. Zapf, S. Signetti, J. Strobel, H. Krüger, R. Röder, J. Carstensen, N. Wolff, J. Marx, T. Carey, M. Schweichel, M.-I. Terasa, L. Siebert, H.-K. Hong, S. Kaps, B. Fiedler, Y. K. Mishra, Z. Lee, N. M. Pugno, L. Kienle, A. C. Ferrari, F. Torrisi, C. Ronning and R. Adelung, Conversionless efficient and broadband laser light diffusers for high brightness illumination applications, Nat. Commun., 2020, 11, 1437 CrossRef PubMed.
- J.-H. Pöhls, F. Schütt, C. O'Neill, S. Shree, M. B. Johnson, Y. K. Mishra, R. Adelung and M. A. White, Thermal and electrical transport properties in multi-walled carbon nanotube-coated ZnO tetrapods and self-entangled multi-walled carbon nanotube tubes, Carbon, 2019, 144, 423–432 CrossRef.
- E. Veys, L. Makower, M. Williamson, L. M. Saure, R. Adelung, F. Schütt, N. M. Pugno and T. J. Marrow, In situ observation of compressive deformation of an interconnected network of zinc oxide tetrapods, Scr. Mater., 2023, 224, 115153 CrossRef CAS.
- A. Gapeeva, H. Qiu, A. Cojocaru, C. Arndt, T. Riaz, F. Schütt, C. Selhuber-Unkel, Y. K. Mishra, A. Tura, S. Sonntag, S. Gniesmer, S. Grisanti, S. Kaps and R. Adelung, Tetrapodal ZnO-Based Composite Stents for Minimally Invasive Glaucoma Surgery, ACS Biomater. Sci. Eng., 2023, 9, 1352–1361 Search PubMed.
- X. Jin, M. Deng, S. Kaps, X. Zhu, I. Hölken, K. Mess, R. Adelung and Y. K. Mishra, Study of tetrapodal ZnO-PDMS composites: A comparison of fillers shapes in stiffness and hydrophobicity improvements, PLoS One, 2014, 9, e106991 Search PubMed.
- R. K. Pandey, J. Dutta, S. Brahma, B. Rao and C.-P. Liu, Review on ZnO-based piezotronics and piezoelectric nanogenerators: aspects of piezopotential and screening effect, J. Phys. Mater., 2021, 4, 44011 CrossRef CAS.
- J. He, C. Dong, X. Chen, H. Cai, X. Chen, X. Jiang, Y. Zhang, A. Peng and M. A. H. Badsha, Review of piezocatalysis and piezo-assisted photocatalysis in environmental engineering, Crystals, 2023, 13, 1382 CrossRef CAS.
- P. Jia, J. Li and H. Huang, Piezocatalysts and Piezo-Photocatalysts: From Material Design to Diverse Applications, Adv. Funct. Mater., 2024, 34, 2407309 CrossRef CAS.
- K. Sun, J. Qi, Q. Zhang, Y. Yang and Y. Zhang, A novel logic switch based on individual ZnO nanotetrapods, Nanoscale, 2011, 3, 2166–2168 RSC.
- Z. Wang, J. Qi, S. Lu, P. Li, X. Li and Y. Zhang, Enhancing sensitivity of force sensor based on a ZnO tetrapod by piezo-phototronic effect, Appl. Phys. Lett., 2013, 103(14), 143125 CrossRef.
- G. S. Wood, K. Jeronimo, C. Mahzan, M. Ammar Bin, R. Cheung and E. Mastropaolo, Zinc oxide nanowires-based flexible pressure sensor, Micro Nano Lett., 2021, 16, 432–435 CrossRef CAS.
- A. Bajpayee, T. E. G. Alivio, P. McKay and S. Banerjee, Functionalized Tetrapodal ZnO Membranes Exhibiting Superoleophobic and Superhydrophilic Character for Water/Oil Separation Based on Differential Wettability, Energy Fuels, 2019, 33, 5024–5034 CrossRef CAS.
- M. Sharma, M. Joshi, S. Nigam, S. Shree, D. K. Avasthi, R. Adelung, S. K. Srivastava and Y. Kumar Mishra, ZnO tetrapods and activated carbon based hybrid composite: Adsorbents for enhanced decontamination of hexavalent chromium from aqueous solution, Chem. Eng. J., 2019, 358, 540–551 CrossRef CAS.
- J. V. Lauritsen, S. Porsgaard, M. K. Rasmussen, M. C. R. Jensen, R. Bechstein, K. Meinander, B. S. Clausen, S. Helveg, R. Wahl, G. Kresse and F. Besenbacher, Stabilization Principles for Polar Surfaces of ZnO, ACS Nano, 2011, 5, 5987–5994 CrossRef CAS PubMed.
- S.-G. Heo, S.-I. Jo and G.-H. Jeong, Revealing the enhanced photocatalytic properties of ZnO tetrapods produced by atmospheric-pressure microwave plasma jet system, Curr. Appl. Phys., 2023, 46, 46–54 CrossRef.
- L. Nie, L. Gao, P. Feng, J. Zhang, X. Fu, Y. Liu, X. Yan and T. Wang, Three-dimensional functionalized tetrapod-like ZnO nanostructures for plasmid DNA delivery, Small, 2006, 2, 621–625 CrossRef CAS PubMed.
- S. Jangra, A. Raza, B. Kumar, J. Sharma, S. Das, K. Pandey, Y. K. Mishra and M. S. Goyat, MXene decorated ZnO-tetrapod for efficient degradation of Methyl Orange, Methylene Blue, and Rhodamine B dyes, Mater. Sci. Eng., B, 2025, 311, 117832 CrossRef CAS.
- Q. Gao, J. Zhao, J. Hu and M. Wang, Applying a switchable superhydrophobic and hydrophilic ZnO nanorod array-coated stainless-steel mesh to electrically-induced oil/water separation, Colloids Surf., A, 2021, 628, 127231 CrossRef CAS.
- F. Rasch, V. Postica, F. Schütt, Y. K. Mishra, A. S. Nia, M. R. Lohe, X. Feng, R. Adelung and O. Lupan, Highly selective and ultra-low power consumption metal oxide based hydrogen gas sensor employing graphene oxide as molecular sieve, Sens. Actuators, B, 2020, 320, 128363 CrossRef CAS.
- B. Chakraborty, D. Litra, A. K. Mishra, C. Lupan, R. Nagpal, S. Mishra, H. Qiu, S. Railean, O. Lupan, N. H. de Leeuw, R. Adelung and L. Siebert, Ultra-selective hydrogen sensors based on CuO - ZnO hetero-structures grown by surface conversion, J. Alloys Compd., 2024, 1002, 175385 CrossRef CAS.
- B. Chakraborty, D. Litra, A. K. Mishra, C. Lupan, R. Nagpal, S. Mishra, H. Qiu, S. Railean, O. Lupan and N. H. de Leeuw, Ultra-selective hydrogen sensors based on CuO-ZnO hetero-structures grown by surface conversion, J. Alloys Compd., 2024, 1002, 175385 CrossRef CAS.
- H. Mei, S. Zhou, M. Lu and L. Cheng, Tetrapod-like ZnO/ZnFe2O4 based heterostructure for enhanced ethanol detection, J. Alloys Compd., 2020, 840, 155583 CrossRef CAS.
- D. Li, J. Ma and K. Chen, 2-D zinc ferrite “moss” furred on 3-D zinc oxide tetrapods to boost detection sensitivity of hydrogen sulfide, J. Phys. Chem. Solids, 2021, 148, 109656 CrossRef CAS.
- C. Lupan, In-Depth Properties Analysis Of Znal2o4/Zno Micro-Nanostructures, J. Eng. Sci., 2025, 32, 35–45 CrossRef PubMed.
- F. Rezaei, P. Kameli, S. Hakimpour, S. Salari, M. Ranjbar, T. Sarkar and B. Aslibeiki, Hydrogen Gas Sensing Properties of Pd-decorated ZnO Tetrapod Nanostructures, Ceram. Int., 2025, 51(27), 54582–54590 Search PubMed.
- J. Hu, H. Ma, Y. Zhou, L. Ma, S. Zhao, S. Shi, J. Li and Y. Chang, Gas-Sensing Properties and Mechanisms of 3D Networks Composed of ZnO Tetrapod Micro-Nano Structures at Room Temperature, Materials, 2024, 17(1), 203 CrossRef CAS PubMed.
- Y. Lin, Z. Long, S. Liang, T. Zhong and L. Xing, A wearable exhaling-oxygen-sensing mask based on piezoelectric/gas-sensing coupling effect for real-time monitoring and uploading lung disease information, J. Phys. D: Appl. Phys., 2022, 55, 224001 CrossRef CAS.
- K. Lee, M. Sahu, S. Hajra, R. Abolhassani, K. Mistewicz, B. Toroń, H.-G. Rubahn, Y. K. Mishra and H. J. Kim, Zinc oxide tetrapod sponges for environmental pollutant monitoring and degradation, J. Mater. Res. Technol., 2023, 22, 811–824 CrossRef CAS.
- A. Knoepfel, B. Poudel and S. Gupta, Surface-Catalyzed Zinc Oxide Nanorods and Interconnected Tetrapods as Efficient Methane Gas Sensing Platforms, Chemosensors, 2023, 11, 506 CrossRef CAS.
- S. Gupta and H. Zou, Implementing an Analytical Model to Elucidate the Impacts of Nanostructure Size and Topology of Morphologically Diverse Zinc Oxide on Gas Sensing, Chemosensors, 2025, 13, 38 CrossRef CAS.
- R. Nagpal, C. Lupan, A. Bîrnaz, A. Sereacov, E. Greve, M. Gronenberg, L. Siebert, R. Adelung and O. Lupan, Multifunctional three-in-one sensor on t-ZnO for ultraviolet and VOC sensing for bioengineering applications, Biosensors, 2024, 14, 293 CrossRef CAS PubMed.
- F. Xu, W. Li, S. Sun, A. Zhong, X. Cheng, J. Shi, Z. Li, J. Li, W. Zhang and X. Wang, Expanding selectivity functionality of a ZnO nanotetrapod-based volatile organic compound sensor using Au nanoparticle decoration, ACS Appl. Nano Mater., 2023, 6, 8335–8345 CrossRef CAS.
- M. Waqas Alam, A. Sharma, A. Sharma, S. Kumar, P. Mohammad Junaid and M. Awad, VOC Detection with Zinc Oxide Gas Sensors: A Review of Fabrication, Performance, and Emerging Applications, Electroanalysis, 2025, 37(1), e202400246 CrossRef CAS.
- H. Chen, L. Lv, K. Xue, P. Zhang, L. Du and G. Cui, Oral Exhalation H2S Sensor Based on Cu2O/ZnO Heterostructures, ACS Sens., 2025, 10, 2579–2588 CrossRef CAS PubMed.
- A. Knoepfel, N. Liu, Y. Hou, S. Sujani, B. R. Dos Reis, R. White, K. Wang, B. Poudel, S. Gupta and S. Priya, Development of Tetrapod Zinc Oxide-Based UV Sensor for Precision Livestock Farming and Productivity, Biosensors, 2022, 12(10), 837 CrossRef CAS PubMed.
- V. Postica, I. Paulowicz, O. Lupan, F. Schütt, N. Wolff, A. Cojocaru, Y. K. Mishra, L. Kienle and R. Adelung, The effect of morphology and functionalization on UV detection properties of ZnO networked tetrapods and single nanowires, Vacuum, 2019, 166, 393–398 CrossRef CAS.
- P. Schadte, R. Madurawala, M.-I. Terasa, M. Tienken, L. Joswig, J. Bahr, S. Kaps, L. Siebert and R. Adelung, Flexible, fast and freestanding UV sensors by printing of networked materials, Appl. Mater. Today, 2025, 45, 102846 CrossRef.
- Y. Liu, X. Zeng, L. Zhu, C. Wang, K. Geng and R. Yao, Effect of nanostructure morphology and concentration on the piezoelectric performance of flexible pressure sensor based on PVDFTRFE/nano-ZNO composite thin film, Curr. Nanosci., 2024, 20, 543–553 CrossRef CAS.
- C. Gao, Z. Long, T. Zhong, S. Liang and L. Xing, A self-powered intelligent glove for real-time human-machine gesture interaction based on piezoelectric effect of T-ZnO/PVDF film, J. Phys. D: Appl. Phys., 2022, 55, 194004 CrossRef CAS.
- B. Liu, M. Shen, L. Mao, Y. Mao and H. Ma, Self-powered Biosensor Big Data Intelligent Information Processing System for Real-time Motion Monitoring, Z. Anorg. Allg. Chem., 2020, 646, 500–506 CrossRef CAS.
- Y. Mao, M. Shen, B. Liu, L. Xing, S. Chen and X. Xue, Self-Powered Piezoelectric-Biosensing Textiles for the Physiological Monitoring and Time-Motion Analysis of Individual Sports, Sensors, 2019, 19(15), 3310 CrossRef CAS PubMed.
- P. Pandit, M. Y. Chougale, D. Dubal, Y. K. Mishra, G. Kerr and A. K. Pandey, Amplifying Touch Using 3D ZnO Tetrapods for Tactile and Haptic Intelligence, Small, 2025, 21, e2408414 CrossRef PubMed.
- L. Wang, X. Qi, C. Li and Y. Wang, Multifunctional tactile sensors for object recognition, Adv. Funct. Mater., 2024, 34, 2409358 CrossRef CAS.
- J. Li, A. Reimers, K. M. Dang, M. G. K. Brunk, J. Drewes, U. M. Hirsch, C. Willems, C. E. H. Schmelzer, T. Groth, A. S. Nia, X. Feng, R. Adelung, W. D. Sacher, F. Schütt and J. K. S. Poon, 3D printed neural tissues with in situ optical dopamine sensors, Biosens. Bioelectron., 2023, 222, 114942 CrossRef CAS PubMed.
- M. Terracciano, S. Račkauskas, A. P. Falanga, S. Martino, G. Chianese, F. Greco, G. Piccialli, G. Viscardi, L. de Stefano, G. Oliviero, N. Borbone and I. Rea, ZnO Tetrapods for Label-Free Optical Biosensing: Physicochemical Characterization and Functionalization Strategies, Int. J. Mol. Sci., 2023, 24(5), 4449 CrossRef CAS PubMed.
- R. Viter, I. Tepliakova, M. Drobysh, V. Zbolotnii, S. Rackauskas, S. Ramanavicius, K. Grundsteins, V. Liustrovaite, A. Ramanaviciene and V. Ratautaite, Photoluminescence-based biosensor for the detection of antibodies against SARS-CoV-2 virus proteins by ZnO tetrapod structure integrated within microfluidic system, Sci. Total Environ., 2024, 939, 173333 CrossRef CAS PubMed.
- V. Myndrul, E. Coy, N. Babayevska, V. Zahorodna, V. Balitskyi, I. Baginskiy, O. Gogotsi, M. Bechelany, M. T. Giardi and I. Iatsunskyi, MXene nanoflakes decorating ZnO tetrapods for enhanced performance of skin-attachable stretchable enzymatic electrochemical glucose sensor, Biosens. Bioelectron., 2022, 207, 114141 CrossRef CAS PubMed.
- V. Myndrul, I. Iatsunskyi, N. Babayevska, M. Jarek and T. Jesionowski, Effect of Electrode Modification with Chitosan and Nafion® on the Efficiency of Real-Time Enzyme Glucose Biosensors Based on ZnO Tetrapods, Materials, 2022, 15(13), 4672 CrossRef CAS PubMed.
- C.-E. Cheng, S. Tangsuwanjinda, H.-M. Cheng and P.-H. Lee, Copper Oxide Decorated Zinc Oxide Nanostructures for the Production of a Non-Enzymatic Glucose Sensor, Coatings, 2021, 11, 936 CrossRef CAS.
- X. Li, H. Deng and K. Wu, Sweat-Based Glucose Sensor Based on a Graphene/Au Composite Electrode, Sci. Adv. Mater., 2022, 14, 55–61 CrossRef CAS.
- P. Pal, Y. Pratap and S. Kabra, T-ZnO/AlGaN/GaN HEMT uric acid sensor-sensitivity analysis and effect of surface wettability for improved performance, IEEE Sens. J., 2022, 22, 11819–11826 Search PubMed.
- R. Rawat, S. Roy, T. Goswami, F. S. Mirsafi, M. Ismael, T. Leissner, Y. K. Mishra, J. McLaughlin, K. Kant and A. Mathur, Aptamer-enhanced ultrasensitive electrochemical detection of HER-2 in breast cancer diagnosis using ZnO tetrapod-K4PTC nanohybrids, Sci. Rep., 2025, 15, 17173 CrossRef CAS PubMed.
- S. Tangsuwanjinda, Y.-Y. Chen, C.-H. Lai, G.-T. Jhou, Y.-W. Chiang and H.-M. Cheng, Microporous Oxide-Based Surface-Enhanced Raman Scattering Film for Quadrillionth Detection of Mercury Ion (II), Processes, 2021, 9, 794 CrossRef CAS.
- E. Vanags, I. Bite, L. Ignatane, R. Ignatans, A. Trausa, C. F. Tipaldi, K. Vilks and K. Smits, Zinc Oxide Tetrapods Doped with Silver Nanoparticles as a Promising Substrate for the Detection of Biomolecules via Surface-Enhanced Raman Spectroscopy, ChemEngineering, 2024, 8, 19 CrossRef CAS.
- P.-T. Chen, Y.-C. Lu, S. Tangsuwanjinda, R.-J. Chung, R. Sakthivel and H.-M. Cheng, Irradiation-induced synthesis of Ag/ZnO nanostructures as surface-enhanced Raman scattering sensors for sensitive detection of the pesticide acetamiprid, Sensors, 2022, 22, 6406 CrossRef CAS PubMed.
- Z. Zhao, W. Lei, X. Zhang, B. Wang and H. Jiang, ZnO-based amperometric enzyme biosensors, Sensors, 2010, 10, 1216–1231 CrossRef CAS PubMed.
- A. Sharma, A. Agrawal, G. Pandey, S. Kumar, K. Awasthi and A. Awasthi, Carbon Nano-Onion-Decorated ZnO Composite-Based Enzyme-Less Electrochemical Biosensing Approach for Glucose, ACS Omega, 2022, 7, 37748–37756 CrossRef CAS PubMed.
- S. Anbu, R. Ravishankaran, M. F. C. Da Guedes Silva, A. A. Karande and A. J. L. Pombeiro, Differentially selective chemosensor with fluorescence off-on responses on Cu(2 +) and Zn(2 +) ions in aqueous media and applications in pyrophosphate sensing, live cell imaging, and cytotoxicity, Inorg. Chem., 2014, 53, 6655–6664 CrossRef CAS PubMed.
- F. Giubileo, E. Faella, A. Kumar, S. de Stefano, L. Viscardi, K. Intonti, O. Durante, A. Pelella, A. Mazzotti, N. Martucciello, E. Beliayev, Y. K. Mishra and M. Passacantando, and A. Di Bartolomeo, Zinc oxide tetrapods as novel field emitters with low turn-on voltage, Nano Express, 2024, 5, 45017 CrossRef CAS.
- K. Karthick, P. Kathirvel, R. Marnadu, S. Chakravarty and M. Shkir, Ultrafast one step direct injection flame synthesis of zinc oxide nanoparticles and fabrication of p-Si/n-ZnO photodiode and characterization, Phys. B, 2021, 612, 412971 CrossRef CAS.
- M. Oproescu, A.-G. Schiopu, V.-M. Calinescu and J. D. Fidelus, Enhanced Efficiency of Polycrystalline Silicon Solar Cells Using ZnO-Based Nanostructured Layers, Crystals, 2025, 15, 398 CrossRef CAS.
- W. Z. Lai, X. Huang, X. F. Wang, Z. H. Wei, Q. T. Wang, L. Sun, L. Zhang, X. Y. Ye and L. A. Ma, Field emission properties of tetrapod-shaped ZnO/TiN point light source with semicircle reflecting anode, Vacuum, 2022, 205, 111408 CrossRef CAS.
- B. Aziz, A. Majid, L. Ghani and I. Aziz, Photoluminescence effect on phosphorous irradiated zinc oxide (ZnO) nanotetrapods synthesized by simple thermal oxidation method, J. Phys. Appl., 2020, 3, 107–112 Search PubMed.
- M. Zhang, F. Averseng, J.-M. Krafft, P. Borghetti, G. Costentin and S. Stankic, Controlled Formation of Native Defects in Ultrapure ZnO for the Assignment of Green Emissions to Oxygen Vacancies, J. Phys. Chem. C, 2020, 124, 12696–12704 CrossRef CAS.
- A. P. Tarasov, C. M. Briskina, V. M. Markushev, L. A. Zadorozhnaya, A. S. Lavrikov and V. M. Kanevskii, Analysis of Laser Action in ZnO Tetrapods Obtained by Carbothermal Synthesis, JETP Lett., 2019, 110, 739–743 CrossRef CAS.
- J. Zhao, J. Fan, W. Liu, H. Shi, N. Xiao and M. Hu, Ultra-Broadband Second-Harmonic Generation in ZnO Nano-Tetrapod With Over-One-Octave Bandwidth, IEEE Photonics Technol. Lett., 2019, 31, 250–252 CAS.
- Y. Yamauchi, M. Tenjimbayashi, S. Samitsu and M. Naito, Durable and Flexible Superhydrophobic Materials: Abrasion/Scratching/Slicing/Droplet Impacting/Bending/Twisting-Tolerant Composite with Porcupinefish-Like Structure, ACS Appl. Mater. Interfaces, 2019, 11, 32381–32389 CrossRef CAS PubMed.
- W. Weng, M. Tenjimbayashi, W. H. Hu and M. Naito, Evolution of and Disparity among Biomimetic Superhydrophobic Surfaces with Gecko, Petal, and Lotus Effect, Small, 2022, 18, e2200349 CrossRef PubMed.
- L.-H. Kao and Q.-H. Chen, An integrated approach for developing a durable and superhydrophobic anti-corrosion coating inspired by biomimetic starfish surface structures, Prog. Org. Coat., 2024, 197, 108825 CrossRef CAS.
- Z. Zhang, Preparation of hydrophobic silicone rubber composite insulators and the research of anti-aging performance, e-Polymers, 2024, 24(1) DOI:10.1515/epoly-2024-0079.
- R. D. Davidson, T. E. O’Loughlin, T. E. G. Alivio, S.-M. Lim and S. Banerjee, Thermodynamics of Wettability: A Physical Chemistry Laboratory Experiment, J. Chem. Educ., 2022, 99, 2689–2696 CrossRef CAS.
- Y. Cao, C. Wang, F. Tie and W. Dong, Superhydrophobic alkoxysilane/T-ZnO/SiO2 nanocomposite coatings enhance mechanical properties of porous building substrates: An experimental and multi-physics simulation study, Constr. Build. Mater., 2025, 467, 140412 CrossRef CAS.
- W. Weng, Y. Yu and M. Naito, Antifreeze-Infused Superhydrophobic Surface with Superb Anti-icing and Antifrosting Performance, Langmuir, 2025, 41, 18741–18753 CrossRef CAS PubMed.
- J. Bai, Q. Wang, Y. Zhou, Q. Peng, Q. Fu, Z. Peng, Z. Zhang, J. Long, S. Zhao, G. Li and H. Lei, Preparation and properties of wear resistant thermochromic superhydrophobic coatings, Colloids Surf., A, 2025, 707, 135882 CrossRef CAS.
- L. Vorotinskienė, A. Antanavičė, R. Skvorčinskienė, S. Račkauskas, M. Urbonavičius, R. Kriūkienė and M. Maziukienė, Effect of hydrophobicity of ZnO tetrapods coating on vapour film formation and friction reduction: A study using complex approach, Int. J. Therm. Sci., 2026, 220, 110372 CrossRef.
- H. Zhu, B. Wang, D. Liu, Y. Lei, J. Wen, S. Yang and P. Yu, Preparation of superhydrophobic polyurethane sponge with photodegradation function and study on its oil-water separation performance, Mater. Today Commun., 2024, 41, 110831 CrossRef CAS.
- A. Bajpayee, N. Rivera-Gonzalez, E. J. Braham, T. E. G. Alivio, A. Fnu, S. Alvi, C. Li, N. Cool, M. Al-Hashimi, L. Fang and S. Banerjee, Multiscale Textured Mesh Substrates that Glide Alcohol Droplets and Impede Ice Nucleation, Adv. Eng. Mater., 2022, 24(8), 2101524 CrossRef CAS.
- W. Weng, M. Tenjimbayashi and M. Naito, Role variability of surface chemistry and surface topography in anti-icing performance, iScience, 2024, 27(11), 111039 CrossRef CAS PubMed.
- W. Weng, X. Zheng, M. Tenjimbayashi, I. Watanabe and M. Naito, De-icing performance evolution with increasing hydrophobicity by regulating surface topography, Sci. Technol. Adv. Mater., 2024, 25, 2334199 CrossRef PubMed.
- Z. Wang, L. Qian, X. Peng, Z. Huang, Y. Yang, C. He and P. Fang, New Aspects of Degradation in Silicone Rubber under UVA and UVB Irradiation: A Gas Chromatography-Mass Spectrometry Study, Polymers, 2021, 13(13), 2215 CrossRef CAS PubMed.
- R. Lohmann, I. T. Cousins, J. C. DeWitt, J. Glüge, G. Goldenman, D. Herzke, A. B. Lindstrom, M. F. Miller, C. A. Ng, S. Patton, M. Scheringer, X. Trier and Z. Wang, Are Fluoropolymers Really of Low Concern for Human and Environmental Health and Separate from Other PFAS?, Environ. Sci. Technol., 2020, 54, 12820–12828 CrossRef CAS PubMed.
- C. Yang, J. Wang, J. Li, H. Zhang, C. Shi, Z. Guo and B. Bai, Facile fabrication of durable mesh with reversible photo-responsive wettability for smart oil/water separation, Prog. Org. Coat., 2021, 160, 106520 CrossRef CAS.
- Y. Chenxi, W. Jian, L. Juan, Z. Haiou, C. Tianqing, G. Zhen, W. Yingguo and B. Bo, Multibioinspired Design of a Durable Janus Copper Foam with Asymmetric and Cooperative Alternating Wettability for Efficient Fog Harvesting, ACS Sustainable Chem. Eng., 2023, 11, 3147–3159 CrossRef.
- D. Li, Y. Fan, G. Han and Z. Guo, Multibioinspired Janus membranes with superwettable performance for unidirectional transportation and fog collection, Chem. Eng. J., 2021, 404, 126515 CrossRef CAS.
- Z. Yu, T. Zhu, J. Zhang, M. Ge, S. Fu and Y. Lai, Fog Harvesting Devices Inspired from Single to Multiple Creatures: Current Progress and Future Perspective, Adv. Funct. Mater., 2022, 32(26), 2200359 CrossRef CAS.
- H. Qiu, A. Gapeeva, I. Hölken, S. Kaps, R. Adelung and M. J. Baum, Preventing algae adhesion using lubricant-modified polydimethylsiloxane/polythiourethane nanocomposite, Mater. Des., 2022, 214, 110389 CrossRef CAS.
- K. Feng, H. Qiu, A. Gapeeva, X. Li, Y. Li, S. Kaps, Y. K. Mishra, R. Adelung, M. Baum and L. Yu, Indole-functionalized polythiourethane/tetrapodal shaped ZnO nanocomposites for eco-friendly marine biofouling control, Prog. Org. Coat., 2023, 185, 107939 CrossRef CAS.
- H. Qiu, A. Gapeeva, S. Kaps, R. Adelung and M. J. Baum, Modification of Nylon Nets with Poly(dimethylsiloxane)/Tetrapodal-Shaped ZnO Composite for Aquaculture Biofouling Control, ACS Appl. Polym. Mater., 2021, 3, 6598–6607 CrossRef CAS.
- N. Yamamoto, A. Kioka and Y. Yamada, On the deposition and polymorphism of CaCO3 crystals in the presence of the tetrapod-shaped ZnO nanomaterials and polydimethylsiloxane composite, Results Surf. Interfaces, 2023, 12, 100138 CrossRef.
- F. Liu, M. Yang, B. Han and J. Long, Development of T-ZnOw@Al2O3-incorporated low-temperature curing aluminium phosphate coating on Ti–6Al–4V alloy, Ceram. Int., 2019, 45, 18406–18412 CrossRef CAS.
- Z. Zhao, R. Balu, N. R. Choudhury and N. K. Dutta, The effect of tetra-needle-like zinc oxide whisker as additive on the crystallization kinetics of polybutylene adipate terephthalate/polylactic acid blend, Polym. Eng. Sci., 2024, 64, 4079–4098 CrossRef CAS.
- R. Brindha, S. S. R. Ajith, M. Nandhini, M. Selvam, K. Subannajui, K. Khotmungkhun and K. Sakthipandi, Evaluation of anticorrosive behaviour of ZnO nanotetra-pods on a AZ91-grade Mg alloy, Bull. Mater. Sci., 2019, 42, 221 CrossRef.
- L. Liu, S. Xu, Z. Wang, X. Chen, M. Cao, S. Zhang, Y. Liu and J. Cui, Building of soft-hard compound brush in porous PVA/NH2@TAtZnO plural gel and the high-efficiency anti-interference removal on Pb(II), Chemosphere, 2023, 319, 137990 CrossRef CAS PubMed.
- S. Xu, X. Jiang, L. Liu, Z. Wang, X. Zhang, Y. Peng and M. Cao, Preparation of PVA/tetra-ZnO composite with framework-supported pore-channel structure and the removal research of lead ions, Environ. Sci. Pollut. Res. Int., 2019, 26, 24062–24074 CrossRef CAS PubMed.
- M. Sharma, M. Joshi, S. Nigam, D. K. Avasthi, R. Adelung, S. K. Srivastava and Y. K. Mishra, Efficient oil removal from wastewater based on polymer coated superhydrophobic tetrapodal magnetic nanocomposite adsorbent, Appl. Mater. Today, 2019, 17, 130–141 CrossRef.
- F. Wu, B. J. Harper and S. L. Harper, Comparative dissolution, uptake, and toxicity of zinc oxide particles in individual aquatic species and mixed populations, Environ. Toxicol. Chem., 2019, 38, 591–602 CrossRef CAS PubMed.
- S. W. Y. Wong, P. T. Y. Leung, A. B. Djurisić and K. M. Y. Leung, Toxicities of nano zinc oxide to five marine organisms: influences of aggregate size and ion solubility, Anal. Bioanal. Chem., 2010, 396, 609–618 CrossRef CAS PubMed.
- E. Iakovleva and M. Sillanpää, The use of low-cost adsorbents for wastewater purification in mining industries, Environ. Sci. Pollut. Res. Int., 2013, 20, 7878–7899 CrossRef CAS PubMed.
- 2000 years of zinc and brass, ed. P. T. Craddock, British Museum Press, London, 1998, vol. 50 Search PubMed.
- B. de Berardis, G. Civitelli, M. Condello, P. Lista, R. Pozzi, G. Arancia and S. Meschini, Exposure to ZnO nanoparticles induces oxidative stress and cytotoxicity in human colon carcinoma cells, Toxicol. Appl. Pharmacol., 2010, 246, 116–127 CrossRef CAS PubMed.
- D. Xiong, T. Fang, L. Yu, X. Sima and W. Zhu, Effects of nano-scale TiO2, ZnO and their bulk counterparts on zebrafish: acute toxicity, oxidative stress and oxidative damage, Sci. Total Environ., 2011, 409, 1444–1452 CrossRef CAS PubMed.
- L. Siebert, E. Luna-Cerón, L. E. García-Rivera, J. Oh, J. Jang, D. A. Rosas-Gómez, M. D. Pérez-Gómez, G. Maschkowitz, H. Fickenscher, D. Oceguera-Cuevas, C. G. Holguín-León, B. Byambaa, M. A. Hussain, E. Enciso-Martinez, M. Cho, Y. Lee, N. Sobahi, A. Hasan, D. P. Orgill, Y. K. Mishra, R. Adelung, E. Lee and S. R. Shin, Light-controlled growth factors release on tetrapodal ZnO-incorporated 3D-printed hydrogels for developing smart wound scaffold, Adv. Funct. Mater., 2021, 31(22), 2007555 CrossRef CAS PubMed.
- M.-Z. Pan, S.-B. Hua, J.-M. Wu, X. Yuan, Z.-L. Deng, J. Xiao and Y.-S. Shi, Preparation and properties of T-ZnOw enhanced BCP scaffolds with double-layer structure by digital light processing, J. Adv. Ceram., 2022, 11, 570–581 CrossRef CAS.
- A. G. Phi-Stone, https://www.phi-stone.de/en/.
- A. Büter, G. Maschkowitz, M. Baum, Y. K. Mishra, L. Siebert, R. Adelung and H. Fickenscher, Antibacterial Activity of Nanostructured Zinc Oxide Tetrapods, Int. J. Mol. Sci., 2023, 24(4), 3444 CrossRef PubMed.
- F. M. Collares, I. M. Garcia, M. Klein, C. F. Parolo, F. A. L. Sánchez, A. Takimi, C. P. Bergmann, S. M. W. Samuel, M. A. Melo and V. C. Leitune, Exploring Needle-Like Zinc Oxide Nanostructures for Improving Dental Resin Sealers: Design and Evaluation of Antibacterial, Physical and Chemical Properties, Polymers, 2020, 12(4), 789 CrossRef CAS PubMed.
- H. L. Ong, B. M. Dell'Agnese, Y. Jiang, Y. Guo, J. Zhou, J. Zhang, J. Luo, R. Tao, M. Zhang and L. G. Dover, Controlling bacterial growth and inactivation using thin film-based surface acoustic waves, Lab Chip, 2024, 24, 4344–4356 RSC.
- P. Schadte, F. Rademacher, G. Andresen, M. Hellfritzsch, H. Qiu, G. Maschkowitz, R. Gläser, N. Heinemann, D. Drücke, H. Fickenscher, R. Scherließ, J. Harder, R. Adelung and L. Siebert, 3D-printed wound dressing platform for protein administration based on alginate and zinc oxide tetrapods, Nano Convergence, 2023, 10, 53 CrossRef CAS PubMed.
- Y. Du, D. Ma, J. Mao, J. Yao, Q. Ma, J. Lu, F. Luo, C. Luo and L. Li, Improving the antistatic and antibacterial properties of polypropylene via tetrapod-shaped ZnO@Ag particles, Polym. Test., 2021, 101, 107301 CrossRef CAS.
- F. Qi, X. Gao, C. Wang, Y. Shuai, L. Yang, R. Liao, J. Xin, S. Peng and C. Shuai, In situ grown silver nanoparticles on tetrapod-like zinc oxide whisker for photocatalytic antibacterial in scaffolds, Mater. Today Sustainability, 2022, 19, 100210 CrossRef.
- J. Vachon, J. Alfurhood, M. Al-Subaie, L. Yang and S. Pierrat, Antibacterial, Antiviral and Antifungal Activity of Zinc Oxide Tetrapod in Polypropylene Compared to Other Zinc Based Additives, J. Appl. Polym. Sci., 2025, 142(25), e57051 CrossRef CAS.
- Q. Li, R. Lin, Z. Tang, S. Liang, X. Xue and L. Xing, A flexible self-cleaning/antibacterial PVDF/T-ZnO fabric based on piezo-photocatalytic coupling effect for smart mask, J. Phys. D: Appl. Phys., 2024, 57, 305106 CrossRef CAS.
- Z. Zhao, R. Balu, S. Gangadoo, N. K. Duta and N. R. Choudhury, Poly(butylene adipate-co-terephthalate)/Polylactic Acid/Tetrapod-Zinc Oxide Whisker Composite Films with Antibacterial Properties, Polymers, 2024, 16, 1039 CrossRef CAS PubMed.
- H. Lee, Y.-J. Kim, Y.-J. Yang, J.-H. Lee and H.-H. Lee, Development of antibacterial dual-cure dental resin composites via tetrapod-shaped zinc oxide incorporation, Dent. Mater., 2024, 40, 1762–1772 CrossRef CAS PubMed.
- J. Liu, J. Xiong, Q. Huang, T. Lu, W. Chen and M. Li, Eco-friendly synthesis of robust bioinspired cotton fabric with hybrid wettability for integrated water harvesting and water purification, J. Cleaner Prod., 2022, 350, 131524 CrossRef CAS.
- J. Gupta, M. Irfan, N. Ramgir, K. P. Muthe, A. K. Debnath, S. Ansari, J. Gandhi, C. T. Ranjith-Kumar and M. Surjit, Antiviral Activity of Zinc Oxide Nanoparticles and Tetrapods Against the Hepatitis E and Hepatitis C Viruses, Front. Microbiol., 2022, 13, 881595 CrossRef PubMed.
- Y. K. Mishra, R. Adelung, C. Röhl, D. Shukla, F. Spors and V. Tiwari, Virostatic potential of micro-nano filopodia-like ZnO structures against herpes simplex virus-1, Antiviral Res., 2011, 92, 305–312 CrossRef CAS PubMed.
- T. E. Antoine, S. R. Hadigal, A. M. Yakoub, Y. K. Mishra, P. Bhattacharya, C. Haddad, T. Valyi-Nagy, R. Adelung, B. S. Prabhakar and D. Shukla, Intravaginal Zinc Oxide Tetrapod Nanoparticles as Novel Immunoprotective Agents against Genital Herpes, J. Immunol., 2016, 196, 4566–4575 CrossRef CAS.
- M. Hosseini, S. Behzadinasab, A. W. H. Chin, L. L. M. Poon and W. A. Ducker, Reduction of Infectivity of SARS-CoV-2 by Zinc Oxide Coatings, ACS Biomater. Sci. Eng., 2021, 7, 5022–5027 CrossRef CAS PubMed.
- S. R. Sonntag, S. Gniesmer, A. Gapeeva, R. Adelung, A. Cojocaru, Y. K. Mishra, S. Kaps, A. Tura, S. Grisanti and S. Grisanti, Zinc oxide tetrapods modulate wound healing and cytokine release in vitro—a new antiproliferative substance in glaucoma filtering surgery, Life, 2022, 12, 1691 CrossRef CAS PubMed.
- S. R. Sonntag, S. Gniesmer, A. Gapeeva, K. J. Offermann, R. Adelung, Y. K. Mishra, A. Cojocaru, S. Kaps, S. Grisanti and S. Grisanti, In vitro evaluation of zinc oxide tetrapods as a new material component for glaucoma implants, Life, 2022, 12, 1805 CrossRef CAS PubMed.
- Y. Xu, P. Chen, X. Guo, C. Li, Z. Li, Z. Zhou, M. Li, N. Long and D. Li, Tetraneedlelike ZnO Whiskers/Tetracalcium Phosphate Synergistically Improved the Mechanical, Degradation and Biological Properties of Polycaprolactone Scaffolds, J. Inorg. Organomet. Polym., 2025, 1–13 Search PubMed.
- Q. Zhu, Y. Xuan and X. Liu, Enhancing direct solar thermochemical performance of modified CaCO3 with thermal transport networks composed of tetrapod-shaped ZnO whiskers, Sol. Energy Mater. Sol. Cells, 2022, 248, 111981 CrossRef CAS.
- B. Kalidasan, A. K. Pandey, F. S. Mirsafi, T. Leissner, H.-G. Rubahn, S. Rahman and Y. K. Mishra, Tetrapod-engineered eutectic salt hydrate composites for advanced thermal energy storage, J. Energy Storage, 2025, 124, 116680 CrossRef CAS.
- Y. Li, G. Hu, Q. Wang, F. Dong and Y. Xiong, Design of PVA/multilayer hybrid particle composite aerogel skeleton supported form-stable phase change materials with high thermal conductivity and solar-to-thermal conversion efficiency, J. Energy Storage, 2024, 77, 109966 CrossRef CAS.
- C.-M. Chang, H.-F. Lin, S.-C. Liao, H.-T. Chiu, C.-E. Liu and H.-L. Guo, Effects of Negative Electrodes Coated by ZnO with Different Morphologies on Electrochemical Performances and Safety of Lithium Ion Batteries, Int. J. Electrochem. Sci., 2019, 14, 1197–1207 CrossRef CAS.
- M. Jia, S. Yu, D. Li, C. Liu, Y. Bao, X. Yu and K. Chen, Preparation and Photocatalytic Performance of MoS2/ZnS/ZnO-T Heterojunction Photocatalyst for Dye Degradation in Water, Part. Part. Syst. Charact., 2022, 39, 2200034 CrossRef CAS.
- J. Tang, Y. Xue, C. Ma, S. Zhang and Q. Li, Facile preparation of BiOI/T-ZnOw p–n heterojunction photocatalysts with enhanced removal efficiency for rhodamine B and oxytetracycline, New J. Chem., 2022, 46, 13010–13020 RSC.
- Y. He, A. U. Rehman, M. Xu, C. A. Not, A. M. C. Ng and A. B. Djurišić, Photocatalytic degradation of different types of microplastics by TiOx/ZnO tetrapod photocatalysts, Heliyon, 2023, 9, e22562 CrossRef CAS PubMed.
- A. K. Mourya, R. P. Singh, T. Kumar, A. S. Talmale, G. S. Gaikwad and A. V. Wankhade, Tuning the morphologies of ZnO for enhanced photocatalytic activity, Inorg. Chem. Commun., 2023, 154, 110850 CrossRef CAS.
- S.-H. Hung and K. McKenna, First-Principles Investigation of the Structure and Properties of Au Nanoparticles Supported on ZnO, J. Phys. Chem. C, 2019, 123, 21185–21194 CrossRef CAS PubMed.
- A. G. Ramu, D. J. Yang, E. M. Al Olayan, O. D. AlAmri, A. S. Aloufi, J. O. Almushawwah and D. Choi, Synthesis of hierarchically structured T-ZnO-rGO-PEI composite and their catalytic removal of colour and colourless phenolic compounds, Chemosphere, 2021, 267, 129245 CrossRef CAS PubMed.
- B. Bachiller-Baeza, A. Iglesias-Juez, G. Agostini and E. Castillejos-López, Pd–Au bimetallic catalysts supported on ZnO for selective 1,3-butadiene hydrogenation, Catal. Sci. Technol., 2020, 10, 2503–2512 RSC.
- X. Huang, L. Zeng, R. Li, Z. Xi and Y. Li, Manipulating conductive network formation via 3D T-ZnO: A facile approach for a CNT-reinforced nanocomposite, Nanotechnol. Rev., 2020, 9, 534–542 CrossRef CAS.
- R. Yan, W. Qin, G. Gong, W. Chen, H. Peng, R. Zhong, C. Mou, C. Huang and B. Zhou, Composite aramid aerogels with anisotropic layered structure reinforced by tetra-needle-like ZnO whiskers for acoustic and thermal insulations, J. Mater. Sci., 2025, 60, 2344–2362 CrossRef CAS.
- F. Scherer, S. Wille, L. Saure, F. Schütt, B. Wellhäußer, R. Adelung and M. Kern, Investigation of Mechanical Properties of Polymer-Infiltrated Tetrapodal Zinc Oxide in Different Variants, Materials, 2024, 17(9), 2112 CrossRef CAS PubMed.
- D. Meinderink, K. J. Nolkemper, J. Bürger, A. G. Orive, J. K. Lindner and G. Grundmeier, Spray coating of poly(acrylic acid)/ZnO tetrapod adhesion promoting nanocomposite films for polymer laminates, Surf. Coat. Technol., 2019, 375, 112–122 CrossRef CAS.
- Y. Su, Y. Chen, H. Zhang, S. Liu and P. Guo, Synergistic enhancement effect of multi-dimensional nanomaterials on high-damping polyurethane, J. Rubber Res., 2025, 28, 39–50 CrossRef CAS.
- X. Jin, J. Strueben, L. Heepe, A. Kovalev, Y. K. Mishra, R. Adelung, S. N. Gorb and A. Staubitz, Joining the un-joinable: adhesion between low surface energy polymers using tetrapodal ZnO linkers, Adv. Mater., 2012, 24, 5676–5680 CrossRef CAS PubMed.
- T. Peng, J. Wang, W. Chao, S. Zhang, J. Liu and J. Wang, Investigation of Space Charge Characteristics and Deep Charging Effects in T-ZnOw/Polyimide Composites, IEEE Trans. Dielectr. Electr. Insul., 2025, 99 Search PubMed.
- B. Wellhäußer, L. M. Saure, F. Schütt, F. Scherer, S. Wille and M. Kern, The Impact of an MDP-Containing Primer on the Properties of Zinc Oxide Networks Infiltrated with BisGMA-TEGDMA and UDMA-TEGDMA Polymers, Materials, 2024, 18, 137 CrossRef PubMed.
- H. Greijer, N. Mirotta, E. Treossi, F. Valorosi, F. Schütt, L. Siebert, Y. K. Mishra, R. Adelung, V. Palermo and H. Hillborg, Tuneable conductivity at extreme electric fields in ZnO tetrapod-silicone composites for high-voltage power cable insulation, Sci. Rep., 2022, 12, 6035 CrossRef CAS PubMed.
- X. Zeng, X. Cheng, R. Yu and G. D. Stucky, Electromagnetic microwave absorption theory and recent achievements in microwave absorbers, Carbon, 2020, 168, 606–623 CrossRef CAS.
- L. Wang, X. Li, Q. Li, X. Yu, Y. Zhao, J. Zhang, M. Wang and R. Che, Oriented Polarization Tuning Broadband Absorption from Flexible Hierarchical ZnO Arrays Vertically Supported on Carbon Cloth, Small, 2019, 15, e1900900 CrossRef PubMed.
- X. Li, L. Yu, W. Zhao, Y. Shi, L. Yu, Y. Dong, Y. Zhu, Y. Fu, X. Liu and F. Fu, Prism-shaped hollow carbon decorated with polyaniline for microwave absorption, Chem. Eng. J., 2020, 379, 122393 CrossRef CAS.
- J. Wang, Z. Xiang, Y. Yin, Z. Lu, Q. Ren, B. Yuan and W. Lu, Stomata-Inspired Intelligent High-Performance Hydrogel With on-Demand Gateable Electromagnetic-Interference Shielding, Small, 2025, e10156 CrossRef CAS.
- H. Cai, Y. Chen, C. Feng and Z. Lin, Solvent-controlled synthesis of bimetallic ZIF-coated tetra-needle ZnO whiskers toward hollow Co/C derivatives with enhanced microwave absorption, J. Mater. Sci., 2025, 60, 18824–18840 CrossRef CAS.
- X. Guo, X. Zhou, Y. Gao, Y. Qian, G. Wang and L. Lyu, Tetra-needle like ZnO whisker doped graphene composites for radar/infrared compatible stealth properties, Polym. Compos., 2025, 46, 9383–9399 CrossRef CAS.
- R. M. Simon, Emi Shielding Through Conductive Plastics, Polym.-Plast. Technol. Eng., 1981, 17, 1–10 CrossRef CAS.
- F. Peng, W. Zhu, Y. Fang, B. Fu, H. Chen, H. Ji, X. Ma, C. Hang and M. Li, Ultralight and Highly Conductive Silver Nanowire Aerogels for High-Performance Electromagnetic Interference Shielding, ACS Appl. Mater. Interfaces, 2023, 15, 4284–4293 CrossRef CAS PubMed.
- M. Pérez-Page, E. Yu, J. Li, M. Rahman, D. M. Dryden, R. Vidu and P. Stroeve, Template-based syntheses for shape controlled nanostructures, Adv. Colloid Interface Sci., 2016, 234, 51–79 CrossRef PubMed.
- C. Orha, M. Nicolaescu, M.-I. Morariu, T. Galatonova, S. Busuioc, C. Lazau and C. Bandas, A Comprehensive Review on Aero-Materials: Present and Future Perspectives, Coatings, 2025, 15, 754 CrossRef CAS.
- A. Schuchardt, T. Braniste, Y. K. Mishra, M. Deng, M. Mecklenburg, M. A. Stevens-Kalceff, S. Raevschi, K. Schulte, L. Kienle, R. Adelung and I. Tiginyanu, Three-dimensional Aerographite-GaN hybrid networks: single step fabrication of porous and mechanically flexible materials for multifunctional applications, Sci. Rep., 2015, 5, 8839 CrossRef CAS PubMed.
- C. Orha, M. Nicolaescu, M.-I. Morariu, T. Galatonova, S. Busuioc, C. Lazau and C. Bandas, A Comprehensive Review on Aero-Materials: Present and Future Perspectives, Coatings, 2025, 15, 754 CrossRef CAS.
- F. Schütt, S. Signetti, H. Krüger, S. Röder, D. Smazna, S. Kaps, S. N. Gorb, Y. K. Mishra, N. M. Pugno and R. Adelung, Hierarchical self-entangled carbon nanotube tube networks, Nat. Commun., 2017, 8, 1215 CrossRef PubMed.
- L. M. Saure, J. Lumma, N. Kohlmann, T. Hartig, E. E. S. Teotonio, S. Shetty, N. Ravishankar, L. Kienle, F. Faupel, S. Schröder, R. Adelung, H. Terraschke and F. Schütt, Functional light diffusers based on hybrid CsPbBr3/SiO2 aero-framework structures for laser light illumination and conversion, J. Mater. Sci., 2024, 59, 17382–17393 CrossRef CAS.
- A. Reimers, A. Bouhanguel, E. Greve, M. Möller, L. M. Saure, S. Kaps, L. Wegner, A. S. Nia, X. Feng, F. Schütt, Y. Andres and R. Adelung, Multifunctional, self-cleaning air filters based on graphene-enhanced ceramic networks, Device, 2023, 1, 100098 CrossRef.
- T. Braniste, V. Ciobanu, F. Schütt, H. Mimura, S. Raevschi, R. Adelung, N. M. Pugno and I. Tiginyanu, Self-Propelled Aero-GaN Based Liquid Marbles Exhibiting Pulsed Rotation on the Water Surface, Materials, 2021, 14(17), 5086 CrossRef CAS PubMed.
- V. Ciobanu, T. Galatonova, T. Braniste, P. Urbanek, S. Lehmann, B. Hanulikova, K. Nielsch, I. Kuritka, V. Sedlarik and I. Tiginyanu, Aero-TiO2 three-dimensional nanoarchitecture for photocatalytic degradation of tetracycline, Sci. Rep., 2024, 14, 31215 CrossRef CAS PubMed.
- A. Reimers, J. Rank, E. Greve, M. Möller, S. Kaps, J. Bahr, R. Adelung and F. Schütt, Graphene-Based Thermopneumatic Generator for On-Board Pressure Supply of Soft Robots, Soft Robot., 2025, 12, 124–134 CrossRef CAS PubMed.
- F. Rasch, C. Schmitt, L. M. Saure, R. Meyer, V. Adamski, D. Dengiz, R. Scherließ, R. Lucius, M. Synowitz, Y. K. Mishra, K. Hattermann, R. Adelung, J. Held-Feindt and F. Schütt, Macroscopic Silicone Microchannel Matrix for Tailored Drug Release and Localized Glioblastoma Therapy, ACS Biomater. Sci. Eng., 2020, 6, 3388–3397 CrossRef CAS PubMed.
- K. Siemsen, S. Rajput, F. Rasch, F. Taheri, R. Adelung, J. Lammerding and C. Selhuber-Unkel, Tunable 3D Hydrogel Microchannel Networks to Study Confined Mammalian Cell Migration, Advanced healthcare, Materials, 2021, 10, e2100625 Search PubMed.
- J. Li, B. Hietel, M. G. K. Brunk, A. Reimers, C. Willems, T. Groth, H. Cynis, R. Adelung, F. Schütt, W. D. Sacher and J. K. S. Poon, 3D-printed microstructured alginate scaffolds for neural tissue engineering, Trends Biotechnol., 2025, 43, 447–461 CrossRef CAS PubMed.
- M. Hauck, J. Dittmann, B. Zeller-Plumhoff, R. Madurawala, D. Hellmold, C. Kubelt, M. Synowitz, J. Held-Feindt, R. Adelung, S. Wulfinghoff and F. Schütt, Fabrication and Modelling of a Reservoir-Based Drug Delivery System for Customizable Release, Pharmaceutics, 2022, 14(4), 777 CrossRef CAS PubMed.
- S. Chandrasekaran, W. V. Liebig, M. Mecklenburg, B. Fiedler, D. Smazna, R. Adelung and K. Schulte, Fracture, failure and compression behaviour of a 3D interconnected carbon aerogel (Aerographite) epoxy composite, Compos. Sci. Technol., 2016, 122, 50–58 CrossRef CAS.
- S. Garlof, T. Fukuda, M. Mecklenburg, D. Smazna, Y. K. Mishra, R. Adelung, K. Schulte and B. Fiedler, Electro-mechanical piezoresistive properties of three dimensionally interconnected carbon aerogel (Aerographite)-epoxy composites, Compos. Sci. Technol., 2016, 134, 226–233 CrossRef CAS.
- S. Garlof, M. Mecklenburg, D. Smazna, Y. K. Mishra, R. Adelung, K. Schulte and B. Fiedler, 3D carbon networks and their polymer composites: Fabrication and electromechanical investigations of neat Aerographite and Aerographite-based PNCs under compressive load, Carbon, 2017, 111, 103–112 CrossRef CAS.
- C. Arndt, M. Hauck, I. Wacker, B. Zeller-Plumhoff, F. Rasch, M. Taale, A. S. Nia, X. Feng, R. Adelung, R. R. Schröder, F. Schütt and C. Selhuber-Unkel, Microengineered Hollow Graphene Tube Systems Generate Conductive Hydrogels with Extremely Low Filler Concentration, Nano Lett., 2021, 21, 3690–3697 CrossRef CAS PubMed.
- M. Hauck, L. M. Saure, B. Zeller-Plumhoff, S. Kaps, J. Hammel, C. Mohr, L. Rieck, A. S. Nia, X. Feng, N. M. Pugno, R. Adelung and F. Schütt, Overcoming Water Diffusion Limitations in Hydrogels via Microtubular Graphene Networks for Soft Actuators, Adv. Mater., 2023, 35, e2302816 CrossRef PubMed.
- F. Schütt, F. Rasch, N. Deka, A. Reimers, L. M. Saure, S. Kaps, J. Rank, J. Carstensen, Y. Kumar Mishra, D. Misseroni, A. Romani Vázquez, M. R. Lohe, A. Shaygan Nia, N. M. Pugno, X. Feng and R. Adelung, Electrically powered repeatable air explosions using microtubular graphene assemblies, Mater. Today, 2021, 48, 7–17 CrossRef.
- P. S. Pálvölgyi, D. Sebők, I. Szenti, E. Bozo, H. Ervasti, O. Pitkänen, J. Hannu, H. Jantunen, M. E. Leinonen, S. Myllymäki, A. Kukovecz and K. Kordas, Lightweight porous silica foams with extreme-low dielectric permittivity and loss for future 6G wireless communication technologies, Nano Res., 2021, 14, 1450–1456 CrossRef.
- AERO MATERIALS GmBH, https://aero-materials.com/.
- L. M. Bellan, M. Pearsall, D. M. Cropek and R. Langer, A 3D interconnected microchannel network formed in gelatin by sacrificial shellac microfibers, Adv. Mater., 2012, 24, 5187–5191 CrossRef CAS PubMed.
- H. Luan, Q. Zhang, T.-L. Liu, X. Wang, S. Zhao, H. Wang, S. Yao, Y. Xue, J. W. Kwak, W. Bai, Y. Xu, M. Han, K. Li, Z. Li, X. Ni, J. Ye, D. Choi, Q. Yang, J.-H. Kim, S. Li, S. Chen, C. Wu, D. Lu, J.-K. Chang, Z. Xie, Y. Huang and J. A. Rogers, Complex 3D microfluidic architectures formed by mechanically guided compressive buckling, Sci. Adv., 2021, 7, eabj3686 CrossRef CAS PubMed.
- R. Saidur, N. A. Rahim and M. Hasanuzzaman, A review on compressed-air energy use and energy savings, Renewable Sustainable Energy Rev., 2010, 14, 1135–1153 CrossRef.
- F. Doyle and J. Cosgrove, An approach to optimising compressed air systems in production operations, Int. J. Ambient Energy, 2018, 39, 194–201 CrossRef.
- I. Plesco, T. Braniste, N. Wolff, L. Gorceac, V. Duppel, B. Cinic, Y. K. Mishra, A. Sarua, R. Adelung, L. Kienle and I. Tiginyanu, Aero-ZnS architectures with dual hydrophilic–hydrophobic properties for microfluidic applications, APL Mater., 2020, 8(6), 061105 CrossRef CAS.
- I. Barg, N. Kohlmann, F. Rasch, T. Strunskus, R. Adelung, L. Kienle, F. Faupel, S. Schröder and F. Schütt, Strain-Invariant, Highly Water Stable All-Organic Soft Conductors Based on Ultralight Multi-Layered Foam-Like Framework Structures, Adv. Funct. Mater., 2023, 33(21), 2212688 CrossRef CAS.
- M. Taale, D. Krüger, E. Ossei-Wusu, F. Schütt, M. A. U. Rehman, Y. K. Mishra, J. Marx, N. Stock, B. Fiedler, A. R. Boccaccini, R. Willumeit-Römer, R. Adelung and C. Selhuber-Unkel, Systematically Designed Periodic Electrophoretic Deposition for Decorating 3D Carbon-Based Scaffolds with Bioactive Nanoparticles, ACS Biomater. Sci. Eng., 2019, 5, 4393–4404 CrossRef CAS PubMed.
- M. Taale, F. Schütt, K. Zheng, Y. K. Mishra, A. R. Boccaccini, R. Adelung and C. Selhuber-Unkel, Bioactive Carbon-Based Hybrid 3D Scaffolds for Osteoblast Growth, ACS Appl. Mater. Interfaces, 2018, 10, 43874–43886 CrossRef CAS PubMed.
- M. Taale, F. Schütt, T. Carey, J. Marx, Y. K. Mishra, N. Stock, B. Fiedler, F. Torrisi, R. Adelung and C. Selhuber-Unkel, Biomimetic Carbon Fiber Systems Engineering: A Modular Design Strategy To Generate Biofunctional Composites from Graphene and Carbon Nanofibers, ACS Appl. Mater. Interfaces, 2019, 11, 5325–5335 CrossRef CAS PubMed.
- C. Shuai, Z. Wang, F. Yang, H. Zhang, J. Liu and P. Feng, Laser additive manufacturing of shape memory biopolymer bone scaffold: 3D conductive network construction and electrically driven mechanism, J. Adv. Res., 2024, 65, 167–181 CrossRef CAS.
- F. Wang, L. M. Saure, F. Schütt, F. Lorich, F. Rasch, A. S. Nia, X. Feng, A. Seekamp, T. Klüter, H. Naujokat, R. Adelung and S. Fuchs, Graphene Oxide Framework Structures and Coatings: Impact on Cell Adhesion and Pre-Vascularization Processes for Bone Grafts, Int. J. Mol. Sci., 2022, 23(6), 3379 CrossRef CAS PubMed.
- T. S. Karande, J. L. Ong and C. M. Agrawal, Diffusion in musculoskeletal tissue engineering scaffolds: design issues related to porosity, permeability, architecture, and nutrient mixing, Ann. Biomed. Eng., 2004, 32, 1728–1743 CrossRef PubMed.
- C. D. Devillard and C. A. Marquette, Vascular Tissue Engineering: Challenges and Requirements for an Ideal Large Scale Blood Vessel, Front. Bioeng. Biotechnol., 2021, 9, 721843 CrossRef.
- D. A. Oliveira, R. A. Da Silva, M. O. Orlandi and J. R. Siqueira Jr, Exploring ZnO nanostructures with reduced graphene oxide in layer-by-layer films as supercapacitor electrodes for energy storage, J. Mater. Sci., 2022, 57, 7023–7034 CrossRef CAS.
- D. Spurling, H. Krüger, N. Kohlmann, F. Rasch, M. P. Kremer, L. Kienle, R. Adelung, V. Nicolosi and F. Schütt, 3D networked MXene thin films for high performance supercapacitors, Energy Storage Mater., 2024, 65, 103148 CrossRef.
- R. Yang, H. Du, Z. Lin, L. Yang, H. Zhu, H. Zhang, Z. Tang and X. Gui, ZnO nanoparticles filled tetrapod-shaped carbon shell for lithium-sulfur batteries, Carbon, 2019, 141, 258–265 CrossRef CAS.
- H. Cavers, H. Krüger, O. Polonskyi, F. Schütt, R. Adelung and S. Hansen, Temperature-Dependent Vapor Infiltration of Sulfur into Highly Porous Hierarchical Three-Dimensional Conductive Carbon Networks for Lithium Ion Battery Applications, ACS Omega, 2020, 5, 28196–28203 CrossRef CAS PubMed.
- H. Kruger, H. Cavers, O. Gronenberg, U. Schurmann, Y. K. Mishra, J. Jacobsen, J. Carstensen, N. Stock, L. Kienle, F. Schutt, R. Adelung and S. Hansen, in 2021 IEEE 11th International Conference Nanomaterials: Applications & Properties (NAP), IEEE, 2021, pp. 1–6 Search PubMed.
- N. Wolff, T. Braniste, H. Krüger, S. Mangelsen, M. R. Islam, U. Schürmann, L. M. Saure, F. Schütt, S. Hansen, H. Terraschke, R. Adelung, I. Tiginyanu and L. Kienle, Synthesis and Nanostructure Investigation of Hybrid β-Ga2 O3/ZnGa2 O4 Nanocomposite Networks with Narrow-Band Green Luminescence and High Initial Electrochemical Capacity, Small, 2023, 19, e2207492 CrossRef PubMed.
- V. Ciobanu, V. V. Ursaki, S. Lehmann, T. Braniste, S. Raevschi, V. V. Zalamai, E. V. Monaico, P. Colpo, K. Nielsch and I. M. Tiginyanu, Aero-TiO2 Prepared on the Basis of Networks of ZnO Tetrapods, Crystals, 2022, 12, 1753 CrossRef CAS.
- F. Matter and M. Niederberger, The Importance of the Macroscopic Geometry in Gas-Phase Photocatalysis, Adv. Sci., 2022, 9, e2105363 CrossRef PubMed.
- M. Dragoman, V. Ciobanu, S. Shree, D. Dragoman, T. Braniste, S. Raevschi, A. Dinescu, A. Sarua, Y. K. Mishra, N. Pugno, R. Adelung and I. Tiginyanu, Sensing up to 40 atm Using Pressure-Sensitive Aero-GaN, Phys. Status Solidi RRL, 2019, 13(6), 1900012 CrossRef.
- Z. Ren, H. Zhang, N. Liu, D. Lei, Q. Zhang, T. Su, L. Wang, J. Su and Y. Gao, Self-powered 2D nanofluidic graphene pressure sensor with Serosa-Mimetic structure, EcoMat, 2023, 5(3), e12299 CrossRef CAS.
- O. Lupan, V. Postica, M. Mecklenburg, K. Schulte, Y. K. Mishra, B. Fiedler and R. Adelung, Low powered, tunable and ultra-light aerographite sensor for climate relevant gas monitoring, J. Mater. Chem. A, 2016, 4, 16723–16730 RSC.
- I. Plesco, J. Strobel, F. Schütt, C. Himcinschi, N. Ben Sedrine, T. Monteiro, M. R. Correia, L. Gorceac, B. Cinic, V. Ursaki, J. Marx, B. Fiedler, Y. K. Mishra, L. Kienle, R. Adelung and I. Tiginyanu, Hierarchical Aerographite 3D flexible networks hybridized by InP micro/nanostructures for strain sensor applications, Sci. Rep., 2018, 8, 13880 CrossRef PubMed.
- O. Lupan, V. Postica, J. Marx, M. Mecklenburg, Y. K. Mishra, K. Schulte, B. Fiedler and R. Adelung, Individual hollow and mesoporous aero-graphitic microtube based devices for gas sensing applications, Appl. Phys. Lett., 2017, 110(26), 263109 CrossRef.
- I. Hölken, G. Neubüser, V. Postica, L. Bumke, O. Lupan, M. Baum, Y. K. Mishra, L. Kienle and R. Adelung, Sacrificial Template Synthesis and Properties of 3D Hollow-Silicon Nano- and Microstructures, ACS Appl. Mater. Interfaces, 2016, 8, 20491–20498 CrossRef PubMed.
- M. Nicolaescu, T. Braniste, C. Orha, M.-I. Morariu, S. Lehmann, K. Nielsch, I. M. Tiginyanu, R. Faur, V. Zalamai and C. Lazau, Room Temperature UV Photodetector Based on Aero-Titania, Int. J. Mol. Sci., 2025, 26, 11035 CrossRef CAS PubMed.
- C. Lamprecht, M. Taale, I. Paulowicz, H. Westerhaus, C. Grabosch, A. Schuchardt, M. Mecklenburg, M. Böttner, R. Lucius, K. Schulte, R. Adelung and C. Selhuber-Unkel, A Tunable Scaffold of Microtubular Graphite for 3D Cell Growth, ACS Appl. Mater. Interfaces, 2016, 8, 14980–14985 CrossRef CAS PubMed.
- O. Parlak, Y. Kumar Mishra, A. Grigoriev, M. Mecklenburg, W. Luo, S. Keene, A. Salleo, K. Schulte, R. Ahuja, R. Adelung, A. P. Turner and A. Tiwari, Hierarchical Aerographite nano-microtubular tetrapodal networks based electrodes as lightweight supercapacitor, Nano Energy, 2017, 34, 570–577 CrossRef CAS.
- E. L. Silva, Y. K. Mishra, A. J. S. Fernandes, R. F. Silva, J. Strobel, L. Kienle, R. Adelung, F. J. Oliveira and M. L. Zheludkevich, Direct Synthesis of Electrowettable Carbon Nanowall–Diamond Hybrid Materials from Sacrificial Ceramic Templates Using HFCVD, Adv. Mater. Interfaces, 2017, 4(10), 1700019 CrossRef.
- S. Daria, S. Sindu, H. Mathias, H. Luka, M. Janik, D. Jannes, K. Zaho, F. Bodo, K. Holger and A. Rainer, Surface modification of highly porous 3D networks via atmospheric plasma treatment, Contrib. Plasma Phys., 2018, 58, 384–393 CrossRef CAS.
- I. Tiginyanu, T. Braniste, D. Smazna, M. Deng, F. Schütt, A. Schuchardt, M. A. Stevens-Kalceff, S. Raevschi, U. Schürmann, L. Kienle, N. M. Pugno, Y. K. Mishra and R. Adelung, Self-organized and self-propelled aero-GaN with dual hydrophilic-hydrophobic behaviour, Nano Energy, 2019, 56, 759–769 CrossRef CAS.
- J. Strobel, L. Ghimpu, V. Postica, O. Lupan, M. Zapf, S. Schönherr, R. Röder, C. Ronning, F. Schütt, Y. K. Mishra, I. Tiginyanu, R. Adelung, J. Marx, B. Fiedler and L. Kienle, Improving gas sensing by CdTe decoration of individual Aerographite microtubes, Nanotechnology, 2019, 30, 65501 CrossRef CAS PubMed.
- J. Marx, M. Lewke, D. Smazna, Y. K. Mishra, R. Adelung, K. Schulte and B. Fiedler, Processing, growth mechanism and thermodynamic calculations of carbon foam with a hollow tetrapodal morphology – Aerographite, Appl. Surf. Sci., 2019, 470, 535–542 CrossRef CAS.
- T. Braniste, M. Dragoman, S. Zhukov, M. Aldrigo, V. Ciobanu, S. Iordanescu, L. Alyabyeva, F. Fumagalli, G. Ceccone, S. Raevschi, F. Schütt, R. Adelung, P. Colpo, B. Gorshunov and I. Tiginyanu, Aero-Ga2O3 Nanomaterial Electromagnetically Transparent from Microwaves to Terahertz for Internet of Things Applications, Nanomaterials, 2020, 10(6), 1047 CrossRef CAS PubMed.
- S. J. Rezvani, A. D'Elia, S. Macis, S. Nannarone, S. Lupi, F. Schütt, F. Rasch, R. Adelung, B. Lu, Z. Zhang, L. Qu, X. Feng, A. R. Vázquez and A. Marcelli, Structural anisotropy in three dimensional macroporous graphene: A polarized XANES investigation, Diamond Relat. Mater., 2021, 111, 108171 CrossRef CAS.
- I. Plesco, V. Ciobanu, T. Braniste, V. Ursaki, F. Rasch, A. Sarua, S. Raevschi, R. Adelung, J. Dutta and I. Tiginyanu, Highly Porous and Ultra-Lightweight Aero-Ga2O3: Enhancement of Photocatalytic Activity by Noble Metals, Materials, 2021, 14(8), 1985 CrossRef CAS PubMed.
- J. Ma, C. Liu and C. Song, Hollow MoS2 tetrapods for high-performance potassium-ion storage, J. Alloys Compd., 2022, 898, 162885 CrossRef CAS.
- V. Ursaki, T. Braniste, V. Zalamai, E. Rusu, V. Ciobanu, V. Morari, D. Podgornii, P. C. Ricci, R. Adelung and I. Tiginyanu, Aero-ZnS prepared by physical vapor transport on three-dimensional networks of sacrificial ZnO microtetrapods, Beilstein J. Nanotechnol., 2024, 15, 490–499 CrossRef CAS PubMed.
- V. Ciobanu, T. Galatonova, P. Urbanek, T. Braniste, F. Doroftei, M. Masar, P. Suly, V. Ursaki, B. Hanulikova, T. Sopik, V. Sedlarik, I. Kuritka and I. Tiginyanu, Enhanced solar light photocatalytic degradation of tetracycline by aero-GaN and ZnO microtetrapods functionalized with noble metal nanodots, Heliyon, 2024, 10, e40989 CrossRef CAS PubMed.
Footnote |
| † These authors have the same contribution to this work. |
| This journal is © The Royal Society of Chemistry 2026 |