The effect of heavy atoms on the thermally activated delayed fluorescence properties of naphthalimide–phenoselenazine electron donor–acceptor dyads: electron transfer and intersystem crossing
Peiwen
Jiang†
 ab,
Haotian
Bai†
c,
Yuying
Pei
a,
Antonio
Toffoletti
ab,
Haotian
Bai†
c,
Yuying
Pei
a,
Antonio
Toffoletti
 d,
Jianzhang
Zhao
d,
Jianzhang
Zhao
 *a,
Yanqin
Li
*a,
Yanqin
Li
 *b,
Yan
Wan
*b,
Yan
Wan
 *c and
Antonio
Barbon
*c and
Antonio
Barbon
 *d
*d
aState Key Laboratory of Fine Chemicals, Frontiers Science Center for Smart Materials, School of Chemical Engineering, Dalian University of Technology, Dalian, 116024, P. R. China. E-mail: zhaojzh@dlut.edu.cn
bSchool of Chemistry, Dalian University of Technology, Dalian, 116024, P. R. China. E-mail: liyanqin@dlut.edu.cn
cCollege of Chemistry, Beijing Normal University, Beijing, 100875, P. R. China. E-mail: wanyan@bnu.edu.cn
dDipartimento di Scienze Chimiche, Università degli Studi di Padova, Via Marzolo 1, 35131, Padova, Italy. E-mail: antonio.barbon@unipd.it
First published on 20th February 2026
Abstract
We prepared two electron donor–acceptor dyads based on naphthalimide (NI) as the electron acceptor and 10H-phenoxazine (PXZ) or 10H-phenoselenazine (PSeZ) as the electron donor. Both NI–PXZ and NI–PSeZ exhibit thermally activated delayed fluorescence (TADF). The purpose of this study is to unravel the effects of heavy atoms on the TADF properties, which are believed to enhance the reverse intersystem crossing (rISC); thus, more dark 3CS and 3LE states are transformed into the emissive 1CS state, so that the TADF properties are enhanced by the presence of heavy atoms such as Se in the molecular structure. Steady state UV-vis absorption and fluorescence studies show negligible interaction between the donor and acceptor at the ground state (S0), yet charge transfer (CT) emission was observed, indicating interaction between the radical anion and the radical cation of the CS state. We didn’t observe shortened delayed fluorescence lifetime for NI–PSeZ (τPF = 14.9 ns, τDF = 91.1 µs) as compared to the analogues of NI–PXZ (τPF = 24.3 ns, τDF = 57.8 µs) and the previously reported NI–PTZ (τPF = 11.9 ns, τDF = 82.1 µs); therefore the heavy atom effect for the rISC is not significant. Moreover, femtosecond transient absorption spectroscopy only captures the 1CS state as the final species and the charge separation and recombination become faster in polar solvetns. Nanosecond transient absorption spectra show the presence of a low-lying 3NI state in non-polar solvents (τT = 41.3 µs), an admixture of the 3NI state and the 3CS state (τT = 23.5 µs) in solvents with intermediate polarity, and only the 3CS state (τT = 0.8 µs) in polar solvents. These observations support the spin-orbit charge transfer ISC (SOCT-ISC) mechanism. The results further show that the rISC is not enhanced by the heavy atom effect, for NI–PSeZ, the krISC = 7.0 × 104 s−1, in comparison, krISC = 1.3 × 105 s−1 for NI–PXZ, and krISC = 1.4 × 107 s−1 for NI–PTZ. Time-resolved electron paramagnetic resonance (TREPR) spectral studies show that the localized triplet state (3NI) is the last triplet state for the dyads in a frozen solution at 80 K, and based on the selective population of the three sublevels of the T1 state, SOCT-ISC may contribute to the formation of the triplet states.
Introduction
Electron donor–acceptor compounds may undergo charge separation to form a charge-separated state upon photoexcitation,1–7 or charge recombination-induced intersystem crossing (ISC) to form a localized triplet state (3LE, LE: localized excited).8–11 These materials play a pivotal role in photovoltaics,12–14 photocatalysis,15,16 and organic light emitting diodes (OLEDs), such as the thermally activated delayed fluorescence (TADF) emitters,17–26 as well as in fundamental photochemistry studies.27,28 One of the conventional goals in studying these dyads is to achieve long-lived charge separation (CS) states, for mimicking the natural photosynthesis center.29–37 The long-lived CS states can function as the oxidant (the radical cation part) or the reductant (the radical anion part); thus the long-lived CS state can drive intermolecular electron transfer to perform photocatalytic reactions.3,15,16,38,39 In the conventional electron donor–acceptor compounds, several strategies were used to prolong the CS state lifetimes, such as long separation distance between the donor and acceptor to reduce the electronic coupling (VDA),40 or use of the Marcus inverted region effect (eqn (1)):27,29,41,42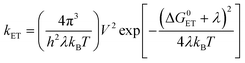 | (1) |
The potential of the traditional electron donor–acceptor compounds has been almost fully exploited after several decades of investigation.2,38,43,44 New strategies are desired for accessing the long-lived CS states in electron donor–acceptor compounds, for instance, in compact electron donor–acceptor compounds having a simple molecular structure, e.g. a short single bond as a linker between the donor and acceptor, instead of the long, saturated linker used in the synthetically demanding traditional electron donor–acceptor compounds. With this seemingly simple alteration of the molecular structure, the photophysics of the dyads will be substantially changed. First, the VDA increases substantially for these compact dyads, and thus both the CS and the charge recombination (CR) processes will be accelerated.8,9,43 Second, the molecular orbital overlap integral will increase significantly, leading to an increased electron exchange integral (J) of the two electrons in the overlap of singly occupied molecular orbitals (SOMO) of the radical anion and cations; thus, a larger energy gap between the 1CS and 3CS states (2J) is expected. This large 2J value is in sharp contrast to the negligible 2J value in conventional non-compact D–A compounds. In the conventional electron donor–acceptor compounds, the spin–spin interaction of the radical anion and cation of the CS state is very weak, and the CS state exists in spin correlated radical pair (SCRP), the 1CS and 3CS state share almost the same energy (2J can be down to a few MHz) and the interchange between the two species is fast.43 Because the CR from the 1CS state to the ground state (S0) is spin-allowed, the CS state should have a short lifetime. In contrast, in the compact electron donor–acceptor compounds, however, the 2J is large, thus the interconversion between the 1CS and 3CS state is inhibited, and a long-lived 3CS state can be formed because the CR from the 3CS state to the S0 state is electron spin-forbidden. Therefore, this large 2J value will affect the CS state profoundly.8,43 This so-called electron spin control method has been used to attain a long-lived 3CS state in compact electron donor–acceptor compounds.43 The formation of a 3CS state, instead of the SCRP, has been unambiguously confirmed via pulse laser excited time-resolved electron paramagnetic resonance (TREPR) spectroscopy.45–48
The photophysics of the compact electron donor–acceptor compounds is rich and interesting.49 For instance, if the 3LE state of the electron donor–acceptor molecule shares a similar energy with that of the CS state, then TADF properties can result. However, due to the complexity of the photophysical processes of these electron donor–acceptor compounds, the detailed mechanism of the electron donor–acceptor TADF emitters has not been fully unveiled. For instance, the experimental evidence for the three-state model (the spin–vibronic coupling mechanism) of the TADF emitters is rare,50–55 and controversial experimental evidence has been found for the recently proposed heavy atom effects on the TADF properties of the compounds.56–59 Notably, the heavy atom effect on TADF has been well investigated in traditional chromophores with heavy atom substitution (e.g., Br/I), where spin–orbit coupling (SOC) is effectively enhanced to accelerate the ISC.59 However, there is controversy regarding compact D–A TADF systems with short linkers between donor and acceptor.57 In such compact systems, the strong electronic orbital overlap and large 2J energy gap and spin–orbit charge transfer ISC (SOCT-ISC) mechanism may alter the role of the heavy atom effect, but this hypothesis lacks direct verification by model molecular systems.
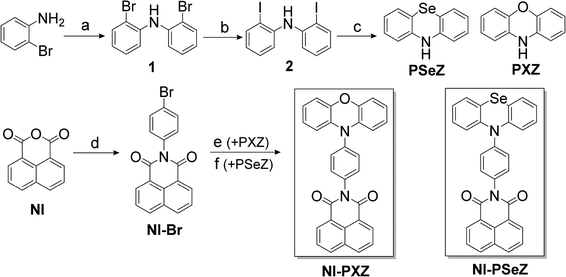
Herein we prepared two electron donor–acceptor compounds as model compounds to study the photophysics of the donor–acceptor TADF emitters, especially the heavy atom effect on the TADF photophysics. Steady state and femtosecond/nanosecond transient absorption and photoluminescence were used in the study. The dynamics of the transient paramagnetic species, such as the 3LE state, as well as the 3CS state, were studied via pulse laser excited TREPR spectroscopy.
Results and discussion
Molecular structure design and properties
Previously we prepared NI–PTZ dyads, with the N-phenyl PTZ attached at the imide position of the NI moiety and TADF properties were observed.60 It is interesting that a phenyl linker is used between the NI and the phenothiazine (PTZ) moiety, yet electronic coupling between the two units exists, and the 1CS state is emissive (without coupling between the donor and acceptor, the CS state would be a dark state). We also prepared NI–PXZ dyads in which the PXZ unit is connected at the 4-position of the NI moiety and TADF properties were observed.61 Herein in order to study the heavy atom effect on the photophysical properties of the dyads, we prepared a NI–PSeZ dyad, in which the heavy atom Se is embedded in the molecular skeleton, which is different from the molecular structures in which the bromo atoms are attached. Thus, NI–PSeZ was prepared (Scheme 1). As a reference compound, NI–PXZ was also prepared, the difference in the molecular structure as compared to that of NI–PSeZ and the previously reported NI–PTZ being only a one atom variation.60 This is a unique method to modify the molecular structure with minimal perturbation, leaving many interactions between the two units in the dyad intact, which is beneficial to unveil the effect of a specific factor on the photophysics of the dyads.62Single crystal X-ray diffraction analysis
The molecular structures of NI–PXZ and NI–PSeZ were determined via single crystal X-ray diffraction (Fig. 1). Single crystals of NI–PXZ and NI–PSeZ were obtained via slow diffusion of n-hexane (HEX) liquid into a chloroform (CHCl3) solution of the compound. The geometry of NI–PSeZ adopts a quasi-equatorial (eq) conformation, in contrast to the quasi-axial (ax) conformation previously observed in PSeZ-containing D–A TADF emitters.57 The torsion angles between the NI moiety and the phenyl unit are 86.7° (NI–PXZ) and 58.7° (NI–PSeZ) (Fig. 1a and b). The dihedral angle between the PXZ and phenyl unit is 78.3°, which is similar to that in NI–PSeZ (72.0°). The detailed parameters and the molecular structure of NI–PXZ and NI–PSeZ are presented in Table S1 (SI).UV-vis absorption and fluorescence emission spectra
The UV-vis absorption spectra of the compounds were studied (Fig. 2). For PXZ and PSeZ, broad, structureless absorption bands in the range of 300–350 nm were observed. For NI–Br, a stronger, structured absorption band in the slightly red-shifted range was observed. For NI–PXZ and NI–PSeZ, stronger absorption bands in a similar range were observed, and the absorption profile of the dyads is virtually the sum of the electron donor (PXZ or PSeZ) and the electron acceptor parts (NI unit); thus, the electronic interaction of the electron donor and acceptor is negligible at the ground state. This is reasonable since the electron donor and acceptor part are isolated by an orthogonal phenyl linker.60,63 In other words, the π-conjugation between the donor and the acceptor is excluded because the π-plane of phenyl linker adopts an orthogonal geometry against the NI part. This is supported by the lack of the S0 → 1CT absorption band, which was observed for an analogue NI–PTZ dyad, with the PTZ unit attached at the 4C position of the NI moiety (not the imide position as in NI–PXZ and NI–PSeZ).64,65The fluorescence properties of the compounds were studied (Fig. 3). First, the fluorescence emission of the dyads in air and in a N2 atmosphere were compared (Fig. 3a and b). For NI–PXZ, a broad, structureless emission band centered at 587 nm was observed under a N2 atmosphere (Fig. 3a). This is a typical CT state emission.64 However, the emission was quenched to a large extent in an aerated solution, and only a minor emission band at 535 nm was observed. Similar results were observed for NI–PSeZ (Fig. 3b). The sensitivity of the fluorescence emission intensity to O2 is one characteristic of TADF.66 Thus, we envision the TADF properties for these dyads. We propose that the minor emission band centered at 535 nm in an aerated solution originated from a CT state which has slightly higher energy than the major emissive CT state, giving an emission band centered at 587 nm. This is different from the previously reported NI–PTZ analogue.60
The fluorescence spectra of the two dyads in solvents with different polarities were studied (Fig. 3c and d). The results show that the fluorescence of the dyads is strongly quenched in polar solvents as compared to non-polar solvents. This result indicates that the emissive S1 state has significant charge transfer character.67 This is in agreement with the broad, structureless emission band of the compounds, as well as the TADF mechanisms, in which the 1CS state is the emissive state (3CS and 3LE states are dark states).60
Fluorescence lifetime studies
In order to confirm the TADF properties of the compounds, the fluorescence lifetimes of the dyads were studied (Fig. 4). Under a N2 atmosphere, NI–PXZ shows a biexponential decay for the fluorescence (Fig. 4a), the faster decaying component has a lifetime of 24.3 ns (86%), and the slower decaying component has a lifetime of 57.8 µs (14%). Note that this delayed fluorescence lifetime is much shorter than that of the previously reported analogue containing a PTZ electron donor (τDF = 82.1 µs).60 This result leads to the inference that a minor variation of the molecular structure by only one atom may alter the excited state properties of the compound significantly. Note the triplet state lifetimes have important implications in OLED devices because roll-off of the efficiency may result in a long triplet excited state lifetime.68,69 Triplet–triplet annihilation (TTA) may also result and it may affect the OLED device performance. On the other hand, for the dyad NI–PSeZ, the prompt and delayed fluorescence lifetimes are τPF = 14.9 ns (91%) and τDF = 91.1 µs (9%), respectively. These values are longer than those for the previously reported dyad of NI–PTZ (τPF = 11.9 ns/τDF = 82.1 µs).60 Under an air atmosphere, the luminescence lifetimes are shortened, especially the delayed fluorescence lifetimes.As compared to the luminescence lifetimes of NI–PTZ, NI–PSeZ shows longer luminescence lifetimes, i.e. there is no so-called heavy atom effect for the TADF process (accelerated rISC rate constant thus shortened triplet state lifetimes in the series analogues). The previously reported NI–N-PTZ dyad containing a C–N bond as the linker between the donor and acceptor also exhibits TADF properties, with a luminescence lifetime of 22.6 ns (96.6%)/2.6 µs (3.4%) in HEX.65 The currently reported delayed fluorescence lifetime is much longer, for instance, for NI–PXZ (τPF = 28.7 ns/τDF = 43.1 µs) and NI–PSeZ (τPF = 14.7 ns/τDF = 64.5 µs) in HEX. This difference may be attributed to the different energy level orders of the 3CS state and the 3LE state. For NI–PXZ and NI–PSeZ, the energy of the 3LE state is relatively lower than that of the 3CS state. Therefore, the energy storage effect of the 3LE state results in a longer delayed fluorescence lifetime. In contrast, the 3CS state of NI–N-PTZ exhibits a lower energy, consequently leading to a shorter delayed fluorescence lifetime. The photophysical parameters of the compounds are summarized in Table 1. It was found that the fluorescence quantum yields are usually low. On the other hand, as an approximation of ISC quantum yields, the singlet oxygen quantum yields (ΦΔ) of the compounds are high (ΦΔ = 41% or 49%), which are slightly higher that reported previously for the NI–PTZ analogues (ΦΔ = 38%)60 and NI–N-PTZ analogues (ΦΔ = 16%).65
| kISC = (1 − Φp)kp | (2) |
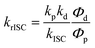 | (3) |
| kTnr = kd − ΦpkrISC | (4) |
| λ abs | ε | λ em | Φ Δ | τ F | Φ F | τ T | |
|---|---|---|---|---|---|---|---|
| a Maximal UV-vis absorption wavelength in CHX, c = 1.0 × 10−5 M, in nm. b Molar absorption coefficient at absorption maxima, in 104 M−1 cm−1. c Maximal fluorescence emission wavelength in CHX, λex = 340 nm, in nm. d Singlet oxygen quantum yields in CHX, Ru(bpy)3[PF6]2 was used as standard compound (ΦΔ = 57% in DCM), λex = 340 nm, in %. e Fluorescence lifetime in CHX under N2, λex = 340 nm. f Absolute fluorescence quantum yields under N2, determined using an optical integration sphere, λex = 340 nm, in %. g Triplet excited-state lifetimes in deaerated CHX, in µs. h Not observed. | |||||||
| PXZ | 313 | 0.8 | —h | —h | —h | —h | —h |
| PSeZ | 309 | 0.4 | —h | —h | —h | —h | —h |
| NI–Br | 332/348 | 1.5/1.3 | 377 | 12 | —h | —h | 44.2 |
| NI–PXZ | 329/345 | 2.7/2.1 | 610 | 49 | 24.3 ns/57.8 µs | 2.0 | 53.5 |
| NI–PSeZ | 327/342 | 2.3/1.9 | 616 | 41 | 14.9 ns/91.1 µs | 1.7 | 41.3 |
For compounds exhibiting TADF behavior, the rate constants for ISC (kISC), reverse intersystem crossing (krISC), and triplet non-radiative decay (kTnr) may be derived from equations 2–4 based on the luminescence lifetimes and luminescence quantum yields.70,71kp and kd represent the decay rate constants of prompt fluorescence and delayed fluorescence, respectively, where p and d denote the photoluminescence quantum efficiencies of prompt fluorescence and delayed fluorescence, respectively. For NI–PXZ, the rate constants kISC, krISC and kTnr were obtained as 4.1 × 107 s−1, 1.3 × 105 s−1 and 1.7 × 104 s−1, respectively. For NI–PSeZ, the rate constants were 6.7 × 107 s−1, 7.0 × 104 s−1 and 8.5 × 103 s−1, respectively. The key photophysical parameters for NI–PXZ, NI–PSeZ and the previously reported NI–PTZ clearly shows that the incorporation of the heavy atom Se in PSeZ does not enhance the rISC process: the krISC of NI–PSeZ (7.0 × 104 s−1) is lower than that of NI–PXZ (1.3 × 105 s−1) and far lower than that of NI–PTZ (1.4 × 107 s−1), accompanied by a longer delayed fluorescence lifetime (τDF = 91.1 µs) of NI–PSeZ compared with NI–PXZ (57.8 µs) and NI–PTZ (82.1 µs), which is in contrast to the expected acceleration of rISC by the heavy atom effect.59
We also measured the ΦΔ values of the compounds in solvents with different polarities (Table 2). The results show that for NI–Br, the ΦΔ values increase in polar solvents as compared to those in non-polar solvents. For the two dyads, however, the ΦΔ values became smaller in polar solvents, and no singlet oxygen photosensitizing ability was observed for the dyads in polar solvents, such as tetrahydrofuran (THF), DCM and ACN. These results show that in polar solvents, the CS state energy decreases, and becomes lower than in the 3NI state. Note the CS state energy is highly dependent on the solvent polarity, whereas the 3LE state energy is almost independent of the solvent polarity. Given that the 3CS state is the lowest state for the dyads upon photoexcitation, the singlet oxygen photosensitizing will be non-efficient because there is no triplet energy transfer to the ground state 3O2 to produce 1O2. These postulations were confirmed by the nanosecond transient absorption spectral studies (see a later section).
| Compounds | CHXb | HEXc | TOLd | THFe | DCMf | ACNg |
|---|---|---|---|---|---|---|
| a Singlet oxygen quantum yield (ΦΔ) of dyads with Ru(bpy)3[PF6]2 as the standard (ΦΔ = 0.57 in DCM) in different solvents, λex = 340 nm. b E T (30) = 30.9 kcal mol−1. c E T (30) = 31.0 kcal mol−1. d E T (30) = 33.9 kcal mol−1. e E T (30) = 37.4 kcal mol−1. f E T (30) = 40.7 kcal mol−1. g E T (30) = 45.6 kcal mol−1. h Not observed. | ||||||
| NI–Br | 12 | 6 | 25 | 19 | 44 | 61 |
| NI–PXZ | 49 | 43 | 12 | —h | —h | —h |
| NI–PSeZ | 41 | 30 | 27 | —h | —h | —h |
Electrochemical and spectroelectrochemical properties
In order to study the thermodynamics of the electron transfer in the dyads upon photoexcitation, the redox potentials of the compounds were studied (Fig. 5a). For both PXZ and PSeZ, similar reversible oxidation waves at +0.21 V (vs. Fc+/Fc) were observed (Fig. 5a and Table 3). For the dyads, similar oxidation potentials were observed (Fig. 5a and Table 3), indicating that the electronic coupling between the electron donor and acceptor is negligible. For NI–PXZ and NI–PSeZ, similar reversible reduction waves at −1.80 V (vs. Fc/Fc+) were observed. Compared to the similar structure NI–PTZ,60 the oxidation potential of NI–PSeZ was 0.06 V higher than that of NI–PTZ, but both have similar reduction potentials. The difference in the reduction and oxidation potentials of NI–PSeZ and NI–PXZ is small.| Compounds | E RED/Vb | E ox/Vb | ΔGCS/eV | E CS/eV | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HEX | CHX | TOL | DCM | ACN | HEX | CHX | TOL | DCM | ACN | |||
| a Cyclic voltammetry in N2-saturated DCM containing a 0.10 M Bu4NPF6 supporting electrolyte; Pt electrode was used as the counter electrode; the working electrode is the glassy carbon electrode; Ag/AgNO3 couple is the reference electrode. E00 = 3.52 eV. E00 is the energy level of the singlet excited state localized on the NI moiety (1NI*) approximated via the crossing point of UV-vis absorption and fluorescence emission spectra after normalization. b The value was obtained by setting the oxidation potential of Fc+/Fc as 0. c Not observed or not applicable. | ||||||||||||
| NI–PXZ | −1.79 | +0.33 | −0.86 | −0.96 | −1.05 | −1.57 | −1.71 | 2.66 | 2.56 | 2.47 | 1.95 | 1.80 |
| NI–PSeZ | −1.81 | +0.36 | −0.82 | −0.91 | −1.00 | −1.52 | −1.66 | 2.70 | 2.61 | 2.52 | 2.00 | 1.86 |
| NI–Br | −1.78 | —c | —c | —c | —c | —c | —c | —c | —c | —c | —c | —c |
Based on the Weller eqn (5)–(7), the Gibbs free energy changes of the intramolecular electron transfer (ΔGCS) were calculated (Table 3).27,72,73 The results show that electron transfer is thermodynamically allowed even in the non-polar solvent cyclohexane and it becomes more exothermic in polar solvents. Thus, electron transfer should be observed, especially in polar solvents. It should be noted that what was observed in transient absorption spectra may not necessarily follow this trend, because the electron transfer can be outcompeted kinetically by other photophysical processes, such as ISC to produce the 3LE state. Moreover, determination error may exist for the relative energy levels of the CS state and the 3LE state.
Moreover, it should be noted the CS state energies of NI–PXZ and NI–PSeZ are different, and the 3LE states localized on the NI moiety should be virtually the same. Thus, besides the possible heavy atom effect on the photophysics of NI–PSeZ, the effect of the variation of CS/3LE state energy gap should not be neglected.59 Previously we found similar variation of the CS state energy of brominated electron donor–acceptor TADF emitters.59 Thus, one should be careful in interpretation of the so-called heavy atom effect in the electron donor–acceptor TADF emitters. The 3NI state energy was determined as 2.25 eV,74 and thus in polar solvents such as DCM and ACN, the CS state should be the lowest state, which should be observed in the nanosecond transient absorption spectra (see a later section).
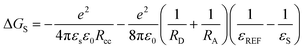 | (5) |
| ΔGCS = e[EOX − ERED] − E00 + ΔGs | (6) |
| ECS = e[EOX − ERED] + ΔGs | (7) |
In order to study the UV-vis absorption of the radical anions and radical cations, and thus to aid the analysis of the femtosecond transient absorption spectral data, spectroelectrochemical characterization of the dyads was performed (Fig. 5b and c). For NI–PSeZ, when a negative potential of −1.72 V (vs. Fc/Fc+) was applied, the original absorption band centered at 335 nm decreased, concomitantly, and two new absorption bands centered at 420 nm, 490 nm and 830 nm were observed, which were attributed to the NI−˙ radical anion.60 On the other hand, when a positive potential of +0.71 V (vs. Fc+/Fc) was applied, the original absorption band at 330 nm showed only a minor decrease, which is reasonable, because the NI unit should be intact, and only the PSeZ part should be oxidized. Indeed, new absorption bands centered at 525 nm and 835 nm were observed, which were attributed to the PSeZ+˙ radical cation.59 This information will be useful for assignment of the CS state in the femtosecond or nanosecond transient absorption spectra (see later sections). It should be noted that upon photoexcitation, CS states may be formed. In these CS states, both the radical anion and cation exist simultaneously, and thus there is coulombic interaction between the ions. Such interaction does not exist in the electrochemically generated radical anions or cations. However, this interaction may impose negligible effects on the UV-vis absorptions of the CS states.
Femtosecond transient absorption (fs-TA) spectra
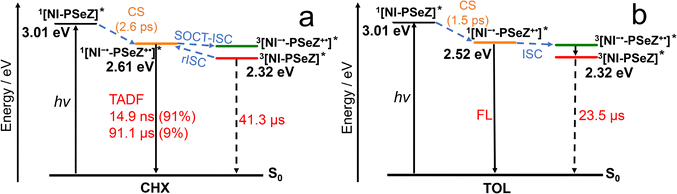
In order to study the excited kinetics of the dyads, the fs-TA spectra of the compounds were studied (Fig. 6). For NI–PXZ, positive absorption bands centered at 410 nm and 540 nm were observed, then red-shifted absorption bands developed at longer delay times, and the absorption band centered at ca. 410 nm became sharper. In order to analyse the transient species in detail, evolution associated difference spectra (EADS) were obtained via global fitting and target analysis, based on the sequential model (Fig. 6b). The first spectrum shows positive absorption bands centered at 410 nm and 490 nm, which are assigned to the 1NI state.60 The absorption profile is slightly different from the 1NI state with PXZ attached at the 4-position of the NI chromophore via a C–N bond.61 Then after 1.4 ps, the second spectrum developed, which shows drastically different absorption bands centered at 420 nm and a broad absorption band centered at 550 nm. These absorption bands are in agreement with the absorption of NI−˙ radical anions and the PXZ+˙ radical cations, respectively (Fig. S16). Thus, the second spectrum is attributed to the CS state, and the CS takes 1.4 ps.This CS process is slightly slower than for the previously reported NI–PTZ analogue, in which the CS takes 0.8 ps.60 After 12.5 ps, the third spectrum developed, which is very close to the second spectrum. Thus, the third spectrum is assigned to the solvation relaxed CS state. Then after ca. 542.9 ps, the system evolves to another state, the final state with a long lifetime is a CS state, and we propose that it is a 1CS state. The geometry relaxation may take 542.9 ps. The final spectrum is a CS state. This result indicates that the CR takes longer than 1.5 ns. This CS in cyclohexane is in agreement with the study of the thermodynamics of the CS (Table 3), which shows the CS from the 1LE state is thermodynamically allowed. Note the excited state dynamics is different from the NI–PXZ analogue with the PXZ unit attached at the 4-position of the NI moiety.61 Due to the simple molecular structure, we did not observe any upper intermediate states,75 or delocalized excited states.76
Similarly, we studied the excited state dynamics of the dyad NI–PSeZ (Fig. 6c and d). The CS takes 2.6 ps, which is two times longer than that of NI–PXZ. Then similar solvent and geometry relaxation were observed, and the final state was relaxed 1CS state. This assignment is supported by the long prompt fluorescence lifetime of 14.9 ns (Fig. 3b). These results show that the CR from the 1CS state to produce the 3NI state is slow.
The CS and CR of NI–PSeZ and NI–PXZ in TOL and ACN were also studied (Fig. S18 and S19, SI). With the increase in solvent polarity, for NI–PSeZ in toluene, 1NI was first observed to reach the 1CS state via rapid CS, and within 313.1 ps, the 1CS state relaxed to the lowest vibrational excited 1CS state through vibrational relaxation. A similar process was observed for NI–PXZ in toluene: it reached the 1CS state within 1.4 ps, followed by relaxation to the lowest vibrational excited 1CS state at 153.7 ps. Unlike the compound NI–PXZ reported in the literature,61 this study observed the long-lived 3CS state, likely due to differences arising from the varying positions at which the PXZ chromophore is attached to NI. In the highly polar ACN solvent, NI–PXZ and NI–PSeZ exhibit no TADF properties, and no triplet CS state was observed in fs-TA spectra (Fig. S18d and S19d). NI–PXZ and NI–PSeZ undergo CS at 770 fs and 448 fs, respectively, to generate the 1CS state, and subsequently undergo CR to the ground state at 59.8 ps and 265.6 ps, respectively. The signal decayed completely within the time window of the femtosecond measurement, indicating a negligible proportion of long-lived components. This is consistent with the photophysical process of NI–PXZ reported in the literature in which the PXZ unit is connected at the 4-position of the NI moiety.61 The charge recombination process in this compound occurs within 10.3 ps, which is faster than the rates observed in NI–PXZ and NI–PSeZ. Furthermore, due to the rapid CR of NI–PXZ and NI–PSeZ in ACN, triplet states cannot be generated via ISC. Consequently, no CS state signal was detected in ACN in longer-timescale ns-TA spectra.
Nanosecond transient absorption (ns-TA) spectra
In order to study the long-lived species formed in the dyads upon photoexcitation, the nanosecond transient absorption (ns-TA) spectra of the dyads were studied (Fig. 7). For NI–PSeZ in CHX, a prominent positive absorption band centered at 460 nm was observed (Fig. 7a), which is attributed to the triplet excited state absorption (ESA) of the NI T1 state (i.e. T1 → Tn transitions), this is supported by the ns-TA spectra of the reference compound NI–Br (Fig. S20). The triplet state lifetime of the 3NI state (3LE, i.e. localized excited state) was determined as 41.3 µs (Fig. 7d). In this case, the CS state has higher energy (2.61 eV, Table 3) than the 3NI state (ET1 = 2.25 eV).74In a more polar solvent TOL, the prominent absorption band centered at 470 nm is persistent, and moreover, two shoulder absorption bands centered at 410 nm and 540 nm were observed (Fig. 7b), these shoulder bands are attributed to NI−˙ and PSeZ+˙, respectively, based on the spectroelectrochemical studies (Fig. 7b and c). The transient decays had a lifetime of 23.5 µs, which is attributed to the intramolecular CS.
The decay kinetics monitored at three wavelengths (410 nm, 470 nm, and 540 nm) give the same decay rate constants, which suggests that the 3CS state and the 3LE state are in good equilibrium. This is in agreement with the spin–vibronic coupling mechanism of the TADF process. We also measured the ns-TA spectra of NI–PSeZ in a more polar solvent THF (Fig. 7c). The results show that the absorption bands centred at 410 nm and 500 nm are more prominent, and thus the CS state is dominant. The lifetime is shortened to 0.8 µs. These results show that even with small variation of the 3CS/3LE states energy gap, the photophysical properties of the electron donor–acceptor TADF emitter will change substantially.77 In the polar solvent ACN, no triplet CS state signal of NI–PSeZ was detected, which is consistent with the results from fs-TA. We found the CS state lifetime of NI–PSeZ in TOL (23.5 µs) is much longer than that of the reference dyad NI–PXZ (13.8 µs) and the previously reported analogue of NI–PTZ (ca. 0.03 µs).60 These results indicate that there is no simple heavy atom effect to enhance the rISC to shorten the triplet states lifetimes. Other factors, such as the energy gaps of the states may also play a role in determination of the photophysics of the electron donor–acceptor dyads. Moreover, the property of the current NI–PSeZ is also different from the NI–N-PTZ analogue with the PTZ moiety attached at the 4-position of the NI moiety,65 which shows a lifetime of 2.6 µs in hexane, and the 3CS state is dominant. In HEX, NI–PSeZ exhibits a 3LE state with the triplet-state lifetime of 43.7 µs, longer than that of NI–N-PTZ.
In order to study the ns-TA spectra of NI–PXZ, measurements were repeated in solvents with different polarities (Fig. S22). In the non-polar solvents CHX and HEX, identical triplet signals (3LE state) were detected, accompanied by relatively long triplet lifetimes of 53.5 µs and 25.7 µs (Fig. S22d and S21c), respectively. As the polarity of the solvent increased, the ns-TA signals were distinctly different. No TADF was observed in toluene, likely due to reduced CS energy levels caused by increased polarity. The ns-TA signals shifted to 410 nm and 540 nm, revealing a mixture of 3LE and 3CS states and a shortening of the triplet lifetime to 13.8 µs (Fig. S22e). In the more polar THF solvent, the triplet signal gradually evolved into a pure 3CS signal. The 3NI* signal at 470 nm completely disappeared, and the lifetime was shortened to 4.7 µs (Fig. S22f). Similar to NI–PSeZ, no signal for NI–PXZ was observed in ACN. Moreover, we found that NI–PXZ, which contains a phenyl linkage between the donor and acceptor moieties, exhibits longer triplet lifetimes than the previously reported NI–PXZ analogue when tested under identical solvent conditions.61,78 But the 3LE state lifetime in CHX is significantly shorter compared to NI–PTZ (ca. 274.7 µs).60
To confirm the formation of the CS state in polar solvents for NI–PSeZ and NI–PXZ, radical anion quenching experiments were performed. Electron acceptor 4-isopropyl-4′-methyldiphenyliodonium tetrakis(pentafluorophenyl)borate (DPI) was added to the solution and the change of ns-TA spectra was monitored (Fig. 8).39,79 In the presence of DPI, we observed the disappearance of the NI−˙ absorption band, whereas the PSeZ+˙ band remained unchanged.
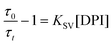 | (8) |
This is reasonable because the quencher, DPI, is an electron acceptor and only the radical anion NI−˙ can be quenched, and thus the radical cation (PSeZ+˙) cannot be quenched by DPI and remains intact (Fig. 8a). By incrementally adding the DPI to the solution, the lifetimes of the species become shorter (Fig. 8b). For the NI−˙ state, which absorbs at 410 nm, the lifetime became shorter upon the addition of DPI. For instance, the lifetime monitored by the trace at 410 nm changes from 0.85 µs (0 eq. DPI added), to 0.66 µs (1.67 eq. DPI) and 0.44 µs (2.58 eq. DPI). Accordingly, based on the Stern–Volmer eqn (8), KSV is calculated to be 2.8 × 104 M−1 (Fig. 8c). Similar behavior was observed for NI–PXZ, for which the KSV value is calculated to be 4.0 × 104 M−1.
Time-resolved electron paramagnetic resonance (TREPR) spectra
Both NI–PXZ and NI–PSeZ show TADF properties, thus, it is interesting to analyze the existence of the transient paramagnetic species for these TADF emitters, for instance, the localized triplet state (3LE) and the triplet CS state (3CS), as well as their relative energy level order. Electron spin dynamics, as well as electron spin polarization (ESP), offer information about the ISC mechanism, etc.53,80,81 TREPR spectroscopy is an ideal tool.82–84 First, we studied the triplet state TREPR spectra of the electron donors PXZ and PSeZ (Fig. 9). For PXZ (in frozen solution in TOL/2-MeTHF = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), a typical TREPR spectrum for the triplet state of a powder sample (randomly oriented in the magnetic field) was observed, with an ESP pattern of (e, e, e, a, a, a) (e: emission; a: enhanced absorption). Half-field transition at 155 mT was observed, demonstrating the signal is due to the triplet excited state. The population rates of the three sublevels of the T1 state were determined as Px
1, v/v), a typical TREPR spectrum for the triplet state of a powder sample (randomly oriented in the magnetic field) was observed, with an ESP pattern of (e, e, e, a, a, a) (e: emission; a: enhanced absorption). Half-field transition at 155 mT was observed, demonstrating the signal is due to the triplet excited state. The population rates of the three sublevels of the T1 state were determined as Px![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Py
Py![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pz = 0.88
Pz = 0.88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.58
0.58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06, based on spectral simulation. Spectral analysis gives the zero-field splitting (ZFS) |D| = 3738 MHz and |E| = 386 MHz (Table 4). These data obtained with the powder sample are similar to the previous report with single crystals for the triplet state of phenoxazine, |D| = 0.099 cm−1 (2970 MHz), |E| = 0.015 cm−1 (450 MHz).85 Similarly, we also recorded the triplet state TREPR spectrum of the PSeZ (Fig. 9). The ESP phase pattern of the spectrum is (e, e, e, a, a, a), half-field transition was also observed. Spectral simulation gives the population rates of the three sublevels as Px
0.06, based on spectral simulation. Spectral analysis gives the zero-field splitting (ZFS) |D| = 3738 MHz and |E| = 386 MHz (Table 4). These data obtained with the powder sample are similar to the previous report with single crystals for the triplet state of phenoxazine, |D| = 0.099 cm−1 (2970 MHz), |E| = 0.015 cm−1 (450 MHz).85 Similarly, we also recorded the triplet state TREPR spectrum of the PSeZ (Fig. 9). The ESP phase pattern of the spectrum is (e, e, e, a, a, a), half-field transition was also observed. Spectral simulation gives the population rates of the three sublevels as Px![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Py
Py![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pz = 0.45
Pz = 0.45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.58
0.58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06, and ZFS |D| = 3190 MHz and |E| = 494 MHz. These data are similar to the previous report for the triplet state of PTZ, measured with single crystal, ZFS |D| = 0.123 cm−1 (3690 MHz) and |E| = 0.014 cm−1 (420 MHz).85 These large D values are due to the small π-conjugation size of these chromophores.
0.06, and ZFS |D| = 3190 MHz and |E| = 494 MHz. These data are similar to the previous report for the triplet state of PTZ, measured with single crystal, ZFS |D| = 0.123 cm−1 (3690 MHz) and |E| = 0.014 cm−1 (420 MHz).85 These large D values are due to the small π-conjugation size of these chromophores.
| Compounds | |D| (MHz) | |E| (MHz) |
P
x
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Py Py![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pz Pz |
ΔPb |
|---|---|---|---|---|
| a Obtained from simulation of the triplet-state TREPR spectra of the indicated molecules in frozen solution (TOL/2-MeTHF, 3/1, v/v) at 80 K. b ΔP = |Px − Py|/|Pz − Py|. c The second species (relative intensity of 30%) exhibit the same polarization state, attributed to the formation of some molecular clusters due to the high concentration used because of the low intensity of PSeZ. | ||||
| NI–PXZ | 2478 | 126 | 0.45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.60 0.60 |
1.38 |
| NI–PSeZ | 2461 | 148 | 0.54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.43 0.43 |
0.81 |
| NI–Br | 2474 | 127 | 0.50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.46 0.46 |
0.93 |
| PXZ | 3738 | 386 | 0.88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.58 0.58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06 0.06 |
0.58 |
| PSeZ | 3190 | 494 | 0.45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.58 0.58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06 0.06 |
0.25 |
| 2461c | 148c | 0.45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.58 0.58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06 0.06 |
0.25 | |
Next, we studied the TREPR spectrum of the NI–Br. The bromophenyl group is not a typical electron donor, and so we presume that the ISC of this reference compound should be the SOC–ISC. The TREPR spectrum of this compound shows an ESP pattern of (e, e, a, e, a, a), which is the same as that observed with the NI derivative (with n-butylamine at the imide position).86 Spectral simulations show that the population rates of the three sublevels are Px![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Py
Py![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pz = 0.5
Pz = 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.46. The ZFS |D| was determined as 2473 MHz and |E| was determined as 127 MHz, which is similar to the previous report.86
0.46. The ZFS |D| was determined as 2473 MHz and |E| was determined as 127 MHz, which is similar to the previous report.86
Next, we studied the TREPR spectra of the electron donor–acceptor dyads. For NI–PXZ, a TREPR spectrum with an ESP phase pattern of (e, e, a, e, a, a) was observed, which is similar to that of NI–Br. Spectral simulations show the population rates of the three sublevels of the T1 state Px![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Py
Py![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pz = 0.45
Pz = 0.45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.6, and the ZFS to be |D| = 2478 MHz, |E| = 126 MHz. These values are similar to that of NI–Br. The slightly different population rates of the three sublevels of NI–PXZ and NI–Br may be attributed to the different ISC mechanisms. For NI–PXZ, the charge recombination induced ISC may contribute to the formation of the triplet state. In the fs-TA spectral study, we found CS and CR in CHX, although the process may be inhibited to some extent in a frozen solution at 80 K. No 3CS state was observed, which should show a much smaller |D| value than the 3LE state.87 This result shows that 3LE has a lower energy than the 3CS state for NI–PXZ, in non-polar solvents. This conclusion is supported by the ns-TA spectral studies (Fig. 9). The results of NI–PXZ are different from the previously studied analogue of the dyad with the PXZ unit attached at the 4-position of the NI chromophore, which has the 3LE state and the 3CS state showing similar energy, and a fast equilibrium between the two states, which leads to a much smaller |D| value of 1484 MHz.61 The elevated 3CS state energy for the current NI–PXZ is attributed to the larger distance between the NI and the PXZ units (a phenyl linker between the two units is used, for the previously reported analogue, a C–N single bond was used as the linker).61 For NI–PSeZ, similar results were observed as compared to those for NI–PXZ. For both dyads, only the 3NI state (the 3LE state) was observed. These results show that the 3CS state energy is elevated to be above that of the 3LE state. Previously with analogues with a dyad based on NI and PTZ units, a weak radical pair signal was observed,60 which is probably due to the slightly more polar solvent used in that case as well as the stronger electron donating ability of the PTZ unit.60 These results show that a TADF emitter can have a 3LE state as the lowest state,51,80 although it was shown that a good TADF emitter should show a more significant 3CS state in the TREPR spectrum.65,88 This information is useful for understanding the photophysics of the TADF emitters.
0.6, and the ZFS to be |D| = 2478 MHz, |E| = 126 MHz. These values are similar to that of NI–Br. The slightly different population rates of the three sublevels of NI–PXZ and NI–Br may be attributed to the different ISC mechanisms. For NI–PXZ, the charge recombination induced ISC may contribute to the formation of the triplet state. In the fs-TA spectral study, we found CS and CR in CHX, although the process may be inhibited to some extent in a frozen solution at 80 K. No 3CS state was observed, which should show a much smaller |D| value than the 3LE state.87 This result shows that 3LE has a lower energy than the 3CS state for NI–PXZ, in non-polar solvents. This conclusion is supported by the ns-TA spectral studies (Fig. 9). The results of NI–PXZ are different from the previously studied analogue of the dyad with the PXZ unit attached at the 4-position of the NI chromophore, which has the 3LE state and the 3CS state showing similar energy, and a fast equilibrium between the two states, which leads to a much smaller |D| value of 1484 MHz.61 The elevated 3CS state energy for the current NI–PXZ is attributed to the larger distance between the NI and the PXZ units (a phenyl linker between the two units is used, for the previously reported analogue, a C–N single bond was used as the linker).61 For NI–PSeZ, similar results were observed as compared to those for NI–PXZ. For both dyads, only the 3NI state (the 3LE state) was observed. These results show that the 3CS state energy is elevated to be above that of the 3LE state. Previously with analogues with a dyad based on NI and PTZ units, a weak radical pair signal was observed,60 which is probably due to the slightly more polar solvent used in that case as well as the stronger electron donating ability of the PTZ unit.60 These results show that a TADF emitter can have a 3LE state as the lowest state,51,80 although it was shown that a good TADF emitter should show a more significant 3CS state in the TREPR spectrum.65,88 This information is useful for understanding the photophysics of the TADF emitters.
Theoretical calculations
DFT computations were performed on the compounds to rationalize the photophysical properties. First, the ground state geometry of the dyads was optimized using the DFT method (Fig. 10a). For NI–PXZ, the dihedral angles between the PXZ and the NI moieties against the intervening phenyl linker are 87.9° and −69.5°, respectively. However, geometry fluctuation is possible in fluid solution at room temperature. Similar results were observed for NI–PSeZ (Fig. 10b).The frontier molecular orbitals of the dyads were examined. For NI–PXZ, the HOMO is confined on the PXZ part, and LUMO is confined on the NI moiety. Thus, the PXZ part is the electron donor and NI is the electron acceptor. Similar results were observed for NI–PSeZ (Fig. 10b). Moreover, a slight distribution of the electron density on the phenyl linker is observed, thus electronic coupling between the donor and acceptor exists, this is critical for the emissive character of the 1CS state, otherwise the 1CS state will be a dark state.43
The electron spin density surface of the T1 state of the dyads under different conditions was studied (Fig. 11) to unveil the electronic character of the T1 states. First, the T1 state spin density of the dyads in the gas phase was studied (this condition can be treated as an approximation of the situation in non-polar solvents). For NI–PXZ, the spin density is distributed on both the NI and the PXZ moieties. However, the density on the two parts is unequal, and higher spin density was observed for the NI part. Based on the spin density distribution of the triplet state of the NI moiety, and the PXZ+˙, we conclude that in the gas phase, the T1 state of NI–PXZ has significant 3LE character. Similar results were observed for NI–PSeZ (Fig. 11b). These results are in agreement with the ns-TA spectral results, in which a 3LE state was observed.
However, in the polar solvent THF, we found the electron spin density distributions on the two parts are more equal. This is in agreement with the character of a 3CS state. This postulation is supported by the spin density distribution pattern on NI, and is different from that in Fig. 11a. Moreover, the spin distribution pattern on the NI unit is similar to that of NI−˙ (Fig. S26). This is in agreement with the ns-TA spectral studies. Thus we conclude a 3CS state is formed for NI–PXZ. Similar results were observed for NI–PSeZ. Previously we found a similar scenario for the electron donor–acceptor compounds,59i.e. the PTZ unit adopts a bent geometry with the dyad populating the 3NI state, whereas the PTZ units change to a plane geometry in the CS state of the dyad (i.e. in its radical cation form). Interestingly, we found that the PSeZ unit adopts different geometries in the dyad in the gas phase and in THF solvent, i.e. the unit exhibits a twisted geometry in the gas phase and a planar geometry in THF. That means the neutral and the cationic forms of this chromophore adopt a different geometry, which is similar to the scenario of PTZ.89,90 In contrast, the configuration of PXZ is identical in the gas phase and in the THF solvent, both adopting a planar geometry.
We calculated the spin orbit coupling matrix elements (SOCMEs) of the possible ISC pathways of the compounds using the TDDFT method in the ORCA 6.0 program.91 First, we checked the reference compounds. For NI, a large SOCME of 14.9 cm−1 was obtained for the S1 → T3 ISC (the two states have energy of 3.67 eV and 3.57 eV, respectively), which is in good agreement with the previous study,92 and it should be responsible for the ISC of this chromophore. For PSeZ, large SOCMEs were also obtained, for example, 70.7 cm−1 for S1 → T2 and 20.9 cm−1 for S1 → T3, respectively, which are due to the heavy atom effect of Se atoms.93–95 For PXZ, however, much smaller SOCMEs were obtained (<0.05 cm−1). For the D–A dyads, much smaller SOCMEs were obtained. For NI–PXZ, the SOCMEs of the S1 → T1 and S1 → T2 are negligible. The optically transition is S0 → S7 (3.655 eV, oscillator strength is 0.325), the SOCMEs is 0.60 cm−1 for the ISC of S7 state to the triplet states sharing similar energy or lower energy. For NI–PSeZ, this value is 11.8 cm−1. However, the fs-TA study shows that upon photoexcitation, the 1LE state undergoes ultrafast CS to form the 1CS state with a time constant of 1.4 ps (NI–PXZ) and 2.6 ps (NI–PSeZ), and this CS process is kinetically far faster than the direct ISC from the LE state (the rate of direct ISC is much lower than that of CS based on the calculated SOCMEs). Thus, the direct ISC from the LE state is not the dominant photophysical process and makes a negligible contribution to the overall excited state dynamics of the dyads, so we do not discuss this process further.
The ZFS D tensor of the triplet state of the compounds was preliminarily studied using the ORCA program.91 The ZFS D of the NI triplet state was calculated as 0.036 cm−1, which is in reasonable agreement with the experimental results of 0.082 cm−1.86 The calculation also shows that the SOC contribute (0.057 cm−1) more significantly than the spin–spin coupling (SSC, −0.02 cm−1). For the compound PXZ, the calculated ZFS D is 0.036 cm−1, which is smaller than the experimental value of 0.12 cm−1 (with single crystals, the value was reported to be ±0.099 cm−1).96 In this case the SOC contribution (−0.035 cm−1) is smaller than the SSC (0.070 cm−1). For PSeZ, however, the ZFS D is largely overestimated (−9.85 cm−1), compared to the experimental value (0.11 cm−1). The SOC contributed substantially (−9.86 cm−1) more than the SSC (0.015 cm−1). For NI–PXZ, the ZFS D of the triplet state was calculated as −0.037 cm−1, which is close to the experimental value of 0.083 cm−1 (note that the sign of the ZFS D cannot be determined by experiments). However, for NI–PSeZ, the ZFS D (2.40 cm−1) is largely overestimated, compared to the experimental value (0.082 cm−1). This result could be due to the overestimated SOC part (2.39 cm−1).
The photophysical processes of NI–PSeZ in different solvents are summarized in Scheme 2. In the nonpolar solvent CHX, NI–PSeZ is excited to generate the 1LE state. This is followed by a rapid CS (2.6 ps) to generate the 1CS state. Driven by the SOCT-ISC mechanism, CR (386.2 ps) occurs, producing the 3LE localized on the NI moiety. It is known that the energy gap between 1CS and the 3CS is generally small. Under this condition, 1CS, 3LE, and 3CS share close energy levels. The spin–vibrational coupling between 3LE and 3CS facilitates rISC, which regenerates the 1CS state and emits delayed fluorescence. However, with increasing solvent polarity, the energy level of the CS state gradually decreases, the energy gap between 3LE and 3CS increases, inhibits the occurrence of spin–vibrational coupling and makes the rISC process difficult. These results indicate that the TADF of electron D–A dyads follows the three-state model (energy level equilibrium among 1CS, 3LE, and 3CS).
Conclusions
In summary, we prepared two electron donor–acceptor dyads as thermally activated delayed fluorescence (TADF) emitters in order to study the heavy-atom effect on the intersystem crossing (ISC) and reverse ISC (rISC) of these TADF emitters. The dyads are based on naphthalimide (NI) as the electron acceptor and 10H-phenoxazine (PXZ) or 10H-phenoselenazine (PSeZ) as the electron donor, with the N-phenyl PXZ or PSeZ attached at the imide position of the NI moiety (dyads of NI–PXZ and NI–PSeZ, respectively). The linker between the electron donor and acceptor is a phenyl moiety. Interestingly, the luminescence lifetime studies show that the delayed fluorescence lifetime of NI–PSeZ containing the heavy atom Se (τPF = 14.9 ns, τDF = 91.1 µs) is longer compared to the analogues of NI–PXZ (τPF = 24.3 ns, τDF = 57.8 µs) as well as the previously reported NI–PTZ (τPF = 11.9 ns, τDF = 82.1 µs). Thus, apparently the rISC is not accelerated by the presence of heavy atoms in the molecular structures of the currently investigated compounds. In femtosecond transient absorption spectroscopy, the 1CS state was observed in a non-polar solvent (CHX) with no evidence of a charge recombination (CR) process, consistent with the results of fluorescence lifetime analysis. As long-lived species are undetectable in the femtosecond time window, nanosecond transient absorption spectroscopy was used. Nanosecond transient absorption spectra show the low-lying 3NI state in a non-polar solvent (CHX, τ = 41.3 µs), an admixture of the 3NI state and 3CS state (τ = 23.5 µs) in a solvent with intermediate polarity (TOL), and only the 3CS state (τ = 0.8 µs) in polar solvents (THF). Combined with femtosecond and nanosecond results, these observations confirm the SOCT-ISC mechanism. These experimental observations are consistent with the Gibbs free energy changes of the electron transfer and the charge separation state energy. Furthermore, the CS state was observed in polar solvents, while no TADF phenomenon was detected, which also supports the spin-vibronic coupling theoretical model of TADF. Pulsed laser excited electron paramagnetic resonance (TREPR) spectral studies show that the localized triplet state (3NI) is the last triplet state for the dyads in frozen solution at 80 K, and based on the selective population of the three sublevels of the T1 state, spin orbit charge transfer ISC may contribute to the formation of the triplet states.Author contributions
J. Z.: acquired the funding, conceived the research study, directed the data analysis and the writing of the manuscript; P. J.: synthesized the compounds, collected and analyzed the data and modified the manuscript; Y. L. and Y. P.: revised parts of the manuscript; H. B. and Y. W.: recorded the femtosecond transient absorption spectra; A. T. and A. B.: recorded and simulated the TREPR spectra and analyzed the data.Conflicts of interest
There are no conflicts to declare.Data availability
All relevant data are presented in the main text and/or the supplementary information (SI). The supplementary information includes experimental details, molecular structure characterization (¹H/ ¹³C NMR, HRMS, single crystal X-ray diffraction with CCDC 2476452 and 2476453), photophysical measurements (UV−vis absorption/fluorescence spectra, fluorescence lifetimes), femtosecond/nanosecond transient absorption spectra, spectroelectrochemical data, and theoretical calculation results (DFT, SOCMEs, ZFS parameters). See DOI: https://doi.org/10.1039/d6cp00066e.CCDC 2476452 and 2476453 contain the supplementary crystallographic data for this paper.97a,b
Acknowledgements
J. Z. thanks the NSFC (22473021, U25A20619 and W2521100), the National Key Research and Development Program of China (the Ministry of Science and Technology, No. 2023YFE0197600), the Research and Innovation Team Project of Dalian University of Technology (DUT2022TB10), the Fundamental Research Funds for the Central Universities (DUT22LAB610) and the State Key Laboratory of Fine Chemicals for financial support. A. Barbon acknowledges the Italian Ministry of Foreign Affairs (Progetti Grande Rilevanza 2024, CN24GR01). Funding by the European Union – Next Generation UE (PRIN2022 PNRR PHOTOCORE) is gratefully acknowledged.Notes and references
- D. M. Guldi, Chem. Commun., 2000, 321–327 RSC.
- J. W. Verhoeven, H. J. van Ramesdonk, M. M. Groeneveld, A. C. Benniston and A. Harriman, ChemPhysChem, 2005, 6, 2251–2260 CrossRef CAS PubMed.
- S. Fukuzumi, Pure Appl. Chem, 2007, 79, 981–991 CrossRef CAS.
- S. Suzuki, M. Kozaki, K. Nozaki and K. Okada, J. Photochem. Photobiol. C: Photochem. Rev., 2011, 12, 269–292 CrossRef CAS.
- O. S. Wenger, Chem. Soc. Rev., 2011, 40, 3538–3550 RSC.
- E. Vauthey, ChemPhysChem, 2012, 13, 2001–2011 CrossRef CAS PubMed.
- H. Imahori, Bull. Chem. Soc. Jpn., 2023, 96, 339–352 CrossRef CAS.
- Y. Hou, X. Zhang, K. Chen, D. Liu, Z. Wang, Q. Liu, J. Zhao and A. Barbon, J. Mater. Chem. C, 2019, 7, 12048–12074 RSC.
- M. A. Filatov, Org. Biomol. Chem., 2020, 18, 10–27 RSC.
- D. J. Gibbons, A. Farawar, P. Mazzella, S. Leroy-Lhez and R. M. Williams, Photochem. Photobiol. Sci., 2020, 19, 136–158 CrossRef CAS PubMed.
- X. Zhang, Z. Wang, Y. Hou, Y. Yan, J. Zhao and B. Dick, J. Mater. Chem. C, 2021, 9, 11944–11973 RSC.
- S. Suzuki, Y. Matsumoto, M. Tsubamoto, R. Sugimura, M. Kozaki, K. Kimoto, M. Iwamura, K. Nozaki, N. Senju, C. Uragami, H. Hashimoto, Y. Muramatsu, A. Konno and K. Okada, Phys. Chem. Chem. Phys., 2013, 15, 8088–8094 RSC.
- A. Devižis, J. De Jonghe-Risse, R. Hany, F. Nüesch, S. Jenatsch, V. Gulbinas and J.-E. Moser, J. Am. Chem. Soc., 2015, 137, 8192–8198 CrossRef PubMed.
- E. Salvadori, N. Luke, J. Shaikh, A. Leventis, H. Bronstein, C. W. M. Kay and T. M. Clarke, J. Mater. Chem. A, 2017, 5, 24335–24343 RSC.
- M. Xiang, Q.-Y. Meng, X.-W. Gao, T. Lei, B. Chen, C.-H. Tung and L.-Z. Wu, Org. Chem. Front., 2016, 3, 486–490 RSC.
- S. M. Sartor, Y. M. Lattke, B. G. McCarthy, G. M. Miyake and N. H. Damrauer, J. Phys. Chem. A, 2019, 123, 4727–4736 CrossRef CAS PubMed.
- H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Nature, 2012, 492, 234–238 CrossRef CAS PubMed.
- Y. Tao, K. Yuan, T. Chen, P. Xu, H. Li, R. Chen, C. Zheng, L. Zhang and W. Huang, Adv. Mater., 2014, 26, 7931–7958 CrossRef CAS PubMed.
- Y. Im, S. Y. Byun, J. H. Kim, D. R. Lee, C. S. Oh, K. S. Yook and J. Y. Lee, Adv. Funct. Mater., 2017, 27, 1603007 Search PubMed.
- Z. Yang, Z. Mao, Z. Xie, Y. Zhang, S. Liu, J. Zhao, J. Xu, Z. Chi and M. P. Aldred, Chem. Soc. Rev., 2017, 46, 915–1016 RSC.
- X. Cai and S.-J. Su, Adv. Funct. Mater., 2018, 28, 1802558 CrossRef.
- T. Huang, W. Jiang and L. Duan, J. Mater. Chem. C, 2018, 6, 5577–5596 RSC.
- F. Ni, N. Li, L. Zhan and C. Yang, Adv. Opt. Mater., 2020, 8, 1902187 CrossRef CAS.
- Y. Xu, P. Xu, D. Hu and Y. Ma, Chem. Soc. Rev., 2021, 50, 1030–1069 RSC.
- L. Chen, W.-C. Chen, Z. Yang, J.-H. Tan, S. Ji, H.-L. Zhang, Y. Huo and C.-S. Lee, J. Mater. Chem. C, 2021, 9, 17233–17264 Search PubMed.
- Y.-Z. Shi, H. Wu, K. Wang, J. Yu, X.-M. Ou and X.-H. Zhang, Chem. Sci., 2022, 13, 3625–3651 Search PubMed.
- D. I. Schuster, P. Cheng, P. D. Jarowski, D. M. Guldi, C. Luo, L. Echegoyen, S. Pyo, A. R. Holzwarth, S. E. Braslavsky, R. M. Williams and G. Klihm, J. Am. Chem. Soc., 2004, 126, 7257–7270 CrossRef CAS PubMed.
- H. Imahori, Y. Kobori and H. Kaji, Acc. Mater. Res., 2021, 2, 501–514 Search PubMed.
- K. Ohkubo, H. Kotani, J. Shao, Z. Ou, K. M. Kadish, G. Li, R. K. Pandey, M. Fujitsuka, O. Ito, H. Imahori and S. Fukuzumi, Angew. Chem., Int. Ed., 2004, 43, 853–856 Search PubMed.
- S. Suzuki, R. Sugimura, M. Kozaki, K. Keyaki, K. Nozaki, N. Ikeda, K. Akiyama and K. Okada, J. Am. Chem. Soc., 2009, 131, 10374–10375 CrossRef CAS PubMed.
- J. Hankache and O. S. Wenger, Chem. Commun., 2011, 47, 10145–10147 RSC.
- N. L. Bill, M. Ishida, Y. Kawashima, K. Ohkubo, Y. M. Sung, V. M. Lynch, J. M. Lim, D. Kim, J. L. Sessler and S. Fukuzumi, Chem. Sci., 2014, 5, 3888–3896 RSC.
- V. M. Blas-Ferrando, J. Ortiz, K. Ohkubo, S. Fukuzumi, F. Fernández-Lázaro and Á. Sastre-Santos, Chem. Sci., 2014, 5, 4785–4793 RSC.
- S.-H. Lee, C. T.-L. Chan, K. M.-C. Wong, W. H. Lam, W.-M. Kwok and V. W.-W. Yam, J. Am. Chem. Soc., 2014, 136, 10041–10052 CrossRef CAS PubMed.
- T. Higashino, T. Yamada, M. Yamamoto, A. Furube, N. V. Tkachenko, T. Miura, Y. Kobori, R. Jono, K. Yamashita and H. Imahori, Angew. Chem., Int. Ed., 2015, 55, 629–633 CrossRef PubMed.
- N. Zarrabi, B. J. Bayard, S. Seetharaman, N. Holzer, P. Karr, S. Ciuti, A. Barbon, M. Di Valentin, A. van der Est, F. D’Souza and P. K. Poddutoori, Phys. Chem. Chem. Phys., 2021, 23, 960–970 Search PubMed.
- K. B. Daisymol and K. R. Gopidas, J. Phys. Chem. Lett., 2023, 14, 977–982 CrossRef CAS PubMed.
- K. Ohkubo and S. Fukuzumi, Bull. Chem. Soc. Jpn, 2009, 82, 303–315 CrossRef CAS.
- Y. Pei, X. Chen, Y. Hou, J. Zhao, Y. Li and S. Ji, Polym. Chem., 2025, 16, 2600–2610 Search PubMed.
- K.-Y. Chen, C.-C. Hsieh, Y.-M. Cheng, C.-H. Lai, P.-T. Chou and T. J. Chow, J. Phys. Chem. A, 2006, 110, 12136–12144 Search PubMed.
- E. H. Yonemoto, G. B. Saupe, R. H. Schmehl, S. M. Hubig, R. L. Riley, B. L. Iverson and T. E. Mallouk, J. Am. Chem. Soc., 1994, 116, 4786–4795 Search PubMed.
- X. Chen, A. A. Sukhanov, Y. Yan, D. Bese, C. Bese, J. Zhao, V. K. Voronkova, A. Barbon and H. G. Yaglioglu, Angew. Chem., Int. Ed., 2022, 61, e202203758 CrossRef CAS PubMed.
- J. W. Verhoeven, J. Photochem. Photobiol., C, 2006, 7, 40–60 CrossRef CAS.
- H. Kotani, K. Ohkubo and S. Fukuzumi, Faraday Discuss., 2012, 155, 89–102 RSC.
- Y. Kobori, M. Fuki and H. Murai, J. Phys. Chem. B, 2010, 114, 14621–14630 Search PubMed.
- R. Carmieli, A. L. Smeigh, S. M. M. Conron, A. K. Thazhathveetil, M. Fuki, Y. Kobori, F. D. Lewis and M. R. Wasielewski, J. Am. Chem. Soc., 2012, 134, 11251–11260 CrossRef CAS PubMed.
- H. Yonemura and M. D. E. Forbes, Photochem. Photobiol., 2015, 91, 672–677 CrossRef CAS PubMed.
- Y. Oka and K. Inoue, Phys. Chem. Chem. Phys., 2024, 26, 16444–16448 RSC.
- X. Chen, X. Zhang, X. Xiao, Z. Wang and J. Zhao, Angew. Chem., Int. Ed., 2023, 62, e202216010 CrossRef CAS PubMed.
- J. Gibson, A. P. Monkman and T. J. Penfold, ChemPhysChem, 2016, 17, 2956–2961 CrossRef CAS PubMed.
- P. K. Samanta, D. Kim, V. Coropceanu and J. L. Brédas, J. Am. Chem. Soc., 2017, 139, 4042–4051 CrossRef CAS PubMed.
- F. B. Dias, T. J. Penfold and A. P. Monkman, Methods Appl. Fluoresc., 2017, 5, 012001 CrossRef PubMed.
- E. W. Evans, Y. Olivier, Y. Puttisong, W. K. Myers, T. J. H. Hele, S. M. Menke, T. H. Thomas, D. Credgington, D. Beljonne, R. H. Friend and N. C. Greenham, J. Phys. Chem. Lett., 2018, 9, 4053–4058 CrossRef CAS PubMed.
- I. Kim, K. H. Cho, S. O. Jeon, W.-J. Son, D. Kim, Y. M. Rhee, I. Jang, H. Choi and D. S. Kim, JACS Au, 2021, 1, 987–997 CrossRef CAS PubMed.
- M. Hempe, N. A. Kukhta, A. Danos, M. A. Fox, A. S. Batsanov, A. P. Monkman and M. R. Bryce, Chem. Mater., 2021, 33, 3066–3080 CrossRef CAS PubMed.
- B. H. Drummond, G. C. Hoover, A. J. Gillett, N. Aizawa, W. K. Myers, B. T. McAllister, S. T. E. Jones, Y. J. Pu, D. Credgington and D. S. Seferos, J. Phys. Chem. C, 2020, 124, 6364–6370 CrossRef CAS.
- S. Goto, Y. Nitta, N. O. Decarli, L. E. de Sousa, P. Stachelek, N. Tohnai, S. Minakata, P. de Silva, P. Data and Y. Takeda, J. Mater. Chem. C, 2021, 9, 13942–13953 RSC.
- Y. X. Hu, J. Miao, T. Hua, Z. Huang, Y. Qi, Y. Zou, Y. Qiu, H. Xia, H. Liu, X. Cao and C. Yang, Nat. Photonics, 2022, 16, 803–810 CrossRef CAS.
- Y. Pei, A. A. Sukhanov, X. Chen, A. Iagatti, S. Doria, X. Dong, J. Zhao, Y. Li, W. Chi, V. K. Voronkova, M. Di Donato and B. Dick, Chem. Eur. J., 2024, 31, e202403542 CrossRef PubMed.
- X. Xiao, Y. Yan, A. A. Sukhanov, S. Doria, A. Iagatti, L. Bussotti, J. Zhao, M. Di Donato and V. K. Voronkova, J. Phys. Chem. B, 2023, 127, 6982–6998 CrossRef CAS PubMed.
- X. Zhang, X. Liu, M. Taddei, L. Bussotti, I. Kurganskii, M. Li, X. Jiang, L. Xing, S. Ji, Y. Huo, J. Zhao, M. Di Donato, Y. Wan, Z. Zhao and M. V. Fedin, Chem. Eur. J., 2022, 28, e202200510 CrossRef CAS PubMed.
- M. K. Etherington, J. Gibson, H. F. Higginbotham, T. J. Penfold and A. P. Monkman, Nat. Commun., 2016, 7, 13680 CrossRef CAS PubMed.
- H. Tanaka, K. Shizu, H. Nakanotani and C. Adachi, J. Phys. Chem. C, 2014, 118, 15985–15994 CrossRef CAS.
- S. Sasaki, K. Hattori, K. Igawa and G.-I. Konishi, J. Phys. Chem. A, 2015, 119, 4898–4906 CrossRef CAS PubMed.
- G. Tang, A. A. Sukhanov, J. Zhao, W. Yang, Z. Wang, Q. Liu, V. K. Voronkova, M. Di Donato, D. Escudero and D. Jacquemin, J. Phys. Chem. C, 2019, 123, 30171–30186 CrossRef CAS.
- H. Tanaka, K. Shizu, H. Miyazaki and C. Adachi, Chem. Commun., 2012, 48, 11392–11394 RSC.
- M. Baruah, W. Qin, C. Flors, J. Hofkens, R. A. L. Vallée, D. Beljonne, M. Van der Auweraer, W. M. De Borggraeve and N. Boens, J. Phys. Chem. A, 2006, 110, 5998–6009 CrossRef CAS PubMed.
- R. Wang, Z. Li, T. Hu, L. Tian, X. Hu, S. Liu, C. Cao, Z.-L. Zhu, J.-H. Tan, Y. Yi, P. Wang, C.-S. Lee and Y. Wang, ACS Appl. Mater. Interfaces, 2021, 13, 49066–49075 CrossRef CAS PubMed.
- Y. H. Lee, Y.-S. Shin, T. Lee, J. Jung, J.-H. Lee and M. H. Lee, Chem. Eng. J., 2021, 423, 130224 CrossRef CAS.
- K. Goushi, K. Yoshida, K. Sato and C. Adachi, Nat. Photonics, 2012, 6, 253–258 CrossRef CAS.
- R. J. Vázquez, J. H. Yun, A. K. Muthike, M. Howell, H. Kim, I. K. Madu, T. Kim, P. Zimmerman, J. Y. Lee and T. Goodson III, J. Am. Chem. Soc., 2020, 142, 8074–8079 CrossRef PubMed.
- R. Ziessel, B. D. Allen, D. B. Rewinska and A. Harriman, Chem. Eur. J., 2009, 15, 7382–7393 CrossRef CAS PubMed.
- A. Karimata, H. Kawauchi, S. Suzuki, M. Kozaki, N. Ikeda, K. Keyaki, K. Nozaki, K. Akiyama and K. Okada, Chem. Lett., 2013, 42, 794–796 CrossRef CAS.
- B. Ventura, A. Bertocco, D. Braga, L. Catalano, S. d'Agostino, F. Grepioni and P. Taddei, J. Phys. Chem. C, 2014, 118, 18646–18658 CrossRef CAS.
- H. Noda, X.-K. Chen, H. Nakanotani, T. Hosokai, M. Miyajima, N. Notsuka, Y. Kashima, J.-L. Brédas and C. Adachi, Nat. Mater., 2019, 18, 1084–1090 CrossRef CAS PubMed.
- T. Hosokai, H. Matsuzaki, H. Nakanotani, K. Tokumaru, T. Tsutsui, A. Furube, K. Nasu, H. Nomura, M. Yahiro and C. Adachi, Sci. Adv., 2017, 3, e1603282 CrossRef PubMed.
- P. L. dos Santos, M. K. Etherington and A. P. Monkman, J. Mater. Chem. C, 2018, 6, 4842–4853 RSC.
- J. Tang, X. Liu, X. Zhang, J. Zhao and Y. Wan, New J. Chem., 2023, 47, 22418–22429 RSC.
- Y. Toba, Y. Usui, M. M. Alam and O. Ito, Macromolecules, 1998, 31, 6022–6029 CrossRef CAS.
- T. Ogiwara, Y. Wakikawa and T. Ikoma, J. Phys. Chem. A, 2015, 119, 3415–3418 CrossRef CAS PubMed.
- S. Yumoto, J. Katsumata, F. Osawa, Y. Wada, K. Suzuki, H. Kaji and K. Marumoto, Sci. Rep., 2023, 13, 11109 CrossRef CAS PubMed.
- S. Weber, eMagRes, 2017, 6, 255–270 Search PubMed.
- T. Biskup, Front. Chem., 2019, 7, 10 CrossRef CAS PubMed.
- S. Richert, C. E. Tait and C. R. Timmel, J. Magn. Reson., 2017, 280, 103–116 CrossRef CAS PubMed.
- J. M. Lhoste, A. Haug and M. Ptak, J. Chem. Phys., 1966, 44, 648–654 CrossRef CAS.
- K. Chen, I. V. Kurganskii, X. Zhang, A. Elmali, J. Zhao, A. Karatay and M. V. Fedin, Chem. Eur. J., 2021, 27, 7572–7587 CrossRef CAS PubMed.
- L. Lv, K. Yuan, T. Zhao, H. Li and D. Wang, New J. Chem., 2023, 47, 18972–18982 Search PubMed.
- B. H. Drummond, N. Aizawa, Y. Zhang, W. K. Myers, Y. Xiong, M. W. Cooper, S. Barlow, Q. Gu, L. R. Weiss, A. J. Gillett, D. Credgington, Y.-J. Pu, S. R. Marder and E. W. Evans, Nat. Commun., 2021, 12, 4532 CrossRef CAS PubMed.
- N. Pearce, E. S. Davies, R. Horvath, C. R. Pfeiffer, X.-Z. Sun, W. Lewis, J. McMaster, M. W. George and N. R. Champness, Phys. Chem. Chem. Phys., 2018, 20, 752–764 RSC.
- T. Okamoto, M. Kuratsu, M. Kozaki, K. Hirotsu, A. Ichimura, T. Matsushita and K. Okada, Org. Lett., 2004, 6, 3493–3496 CrossRef CAS PubMed.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2025, 15, e70019 Search PubMed.
- P. K. Samanta and N. J. English, J. Phys. Chem. C, 2020, 124, 8178–8185 Search PubMed.
- V. V. Patil, H. J. Jang and J. Y. Lee, J. Mater. Chem. C, 2023, 11, 8084–8090 RSC.
- D. Zhong, S. Liu, L. Yue, Z. Feng, H. Wang, P. Yang, B. Su, X. Yang, Y. Sun and G. Zhou, Chem. Sci., 2024, 15, 9112–9119 Search PubMed.
- Z. Cheng, X. Wu, X. Zhang, S. Gao, H. Tong and L. Wang, Adv. Opt. Mater., 2025, 13, 2500517 CrossRef CAS.
- J.-M. Lhoste, M. Ptak and D. Lexa, J. Chim. Phys., 1968, 65, 1876–1888 CrossRef CAS.
- (a) CCDC 2476452: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2p3ykb; (b) CCDC 2476453: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2p3ylc.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © the Owner Societies 2026 |