Open Access Article
Open Access ArticleMicroscopic visualization of NiFeOx and NiOx cocatalyst effects on charge carrier dynamics of BiVO4 photoanodes
Yuki
Nakatsukasa
,
Haruka
Nobuoka
and
Kenji
Katayama
 *
*
Department of Applied Chemistry, Chuo University, Tokyo 112-8551, Japan. E-mail: kkata.33g@g.chuo-u.ac.jp; Tel: +81-3-3817-1913
First published on 19th February 2026
Abstract
Improving the interfacial charge carrier dynamics of BiVO4 photoanodes is essential for achieving higher efficiency in photoelectrochemical water oxidation. In this study, we investigated the effects of NiFeOx and NiOx cocatalysts on BiVO4 by combining patterned-illumination time-resolved phase microscopy (PI-PM) and photoelectrochemical measurements, including impedance spectroscopy. Both cocatalysts substantially enhance the photocurrent and charge-injection efficiency (ηinj), whereas the bulk charge-separation efficiency (ηsep) remains less changed (within ±6%), indicating that the improvements originate from the interfacial hole transfer process. PI-PM visualizes the local trapped-carrier dynamics and reveals the differences in trapped-carrier activity between the cocatalysts. NiFeOx introduces spatially localized slow-decay hole and electron components that correspond to reactive carrier populations, while NiOx generates a more uniform distribution of reactive domains with increased spatial coverage and prolonged carrier lifetimes. These microscopic differences correlate with macroscopic performance, with NiOx achieving the highest ηinj and photocurrent (1.98 mA cm−2). The combined results demonstrate that NiFeOx locally enhances the catalytic activity at selective surface regions, whereas NiOx promotes more homogeneous interfacial charge extraction across the BiVO4 surface.
Introduction
Bismuth vanadate (BiVO4) has been widely recognized as one of the most promising visible-light-responsive photocatalysts for oxygen evolution because of its suitable band gap (∼2.4 eV), relatively high chemical stability, and compatibility with various fabrication methods. Owing to these characteristics, BiVO4 has been extensively investigated for photocatalytic and photoelectrochemical water oxidation,1–8 where the overall performance is strongly governed by the generation, separation, and interfacial utilization of photogenerated charge carriers. Despite these advantages, the photocurrent and quantum efficiency of BiVO4-based systems remain significantly lower than the theoretical values due to carrier recombination, defect states and sluggish surface water-oxidation kinetics.Loading cocatalysts onto semiconductor surfaces is one of the most effective routes to improve photocatalytic and PEC performance. Cocatalysts generally accelerate interfacial charge transfer, suppress surface recombination, and modify local energetics to facilitate desired redox reactions.9 Among various candidates, NiFeOx and NiOx have attracted particular attention as oxygen-evolution cocatalysts (OECs) due to their earth-abundant composition and high intrinsic activity. NiFeOx has been widely used to improve PEC water oxidation, offering advantages such as enhanced hole accumulation and reduced surface recombination.10–14 In contrast, NiOx, although less explored, possesses distinct catalytic and electronic properties, such as p-type conductivity and favorable band alignment with BiVO4 that may lead to different pathways for charge accumulation, defect passivation, and carrier stabilization.15–18
Understanding the enhancement mechanisms provided by these cocatalysts requires direct observation of the underlying charge carrier dynamics. Such dynamics offer fundamental insight into how cocatalysts modify trapping, recombination, and interfacial transfer processes. From this perspective, in general, transient absorption (TA) spectroscopy19–22 and time-resolved photoluminescence (TRPL)23 have been employed to study carrier behavior in BiVO4. The cocatalyst-induced longer lifetime of charge carriers has been studied by monitoring appropriate probe wavelengths.24 These methods have been mainly used for monitoring the free charge carriers in the conduction and valence bands.
As a complementary approach to TA and TRPL, we have developed the patterned-illumination time-resolved phase microscopy (PI-PM) method,25 which visualizes local trapped-carrier (not free charge-carrier) dynamics through refractive index changes with microscopic spatial resolution, caused by charge accumulation at the interface. This technique has revealed key aspects of charge trapping, interfacial reactions, and cocatalyst functions on various photocatalysts. Our previous studies demonstrated the local charge carrier dynamics of BiVO4 prepared by different synthetic methods,26 as well as provided microscopic insight into cocatalyst-related processes such as CoOx and Rh on BiVO4.27,28 These reports highlight the utility of PI-PM in distinguishing electron and hole contributions, identifying reactive versus nonreactive trap states, and mapping heterogeneous surface activity.
In this study, we apply PI-PM to elucidate the effects of NiFeOx and NiOx cocatalysts on BiVO4 photoanodes. We found that NiOx fabrication leads to a remarkably high photocurrent of 1.98 mA cm−2, surpassing that of NiFeOx-modified BiVO4 under similar preparation conditions. By combining surface structural analysis, electrical characterization (PEIS), and microscopic charge-carrier imaging (PI-PM), we systematically investigate how these cocatalysts alter surface energetics and interfacial dynamics. Through this comprehensive approach, we clarify the origin of the superior performance of NiOx. Although PI-PM has been previously employed to investigate charge-carrier dynamics in photocatalytic systems, a direct microscopic comparison of multiple high-performance cocatalysts under identical conditions has not yet been reported. In this work, we comparatively examine NiFeOx and NiOx overlayers on BiVO4 to elucidate how differences in the spatial distribution and lifetime of interfacial carriers translate into distinct charge-injection efficiencies and PEC performance. This comparative approach enables a mechanistic understanding of cocatalyst functionality at the microscopic level. To clarify the overall conceptual framework, a schematic illustration of the PI-PM methodology and its relation to photoelectrochemical (PEC) performance is presented in Fig. 1.
Experimental
PI-PM method
The PI-PM technique is a time-resolved pump–probe microscopy method that visualizes the dynamics of photo-excited charge carriers through refractive index variations detected by phase-contrast imaging.25 In this setup, both the pump and probe lights are collimated to illuminate the sample surface over a region approximately 0.5 mm in diameter. A slight defocus adjustment converts the refractive index modulation into measurable phase contrast as an intensity contrast,29 allowing non-radiative processes such as charge trapping and interfacial transfer to be visualized as temporal image sequences. Both lights are nanosecond pulsed (3–4 ns), and time-resolved images are obtained by electronically controlling the delay between the two pulses.An arbitrary illumination pattern, generated using a digital micromirror device (DMD), is used as the pump light to selectively excite charge carriers within the sample. Following excitation, these carriers undergo processes such as trapping, recombination, and interfacial transfer, resulting in time-dependent refractive index changes that reflect carrier generation and decay. The spatial distribution of these processes is reconstructed as a microscopic image sequence. Because refractive index detection is sensitive to interfacial non-radiative carrier dynamics, PI-PM can probe surface-trapped carriers that are not accessible via TA or TRPL. The sign of the refractive index changes and the response to scavenger species at the solution interface enable distinction between different carrier types on the surface.
Images obtained under pulsed-laser illumination inherently contain intensity fluctuations due to shot noise and optical inhomogeneity. Therefore, the acquired image sequences are reconstructed using three-dimensional total-variation (TV) regularization across the spatial (x, y) and temporal (t) dimensions,30 which effectively suppresses noise while preserving the structural and kinetic information. Because this procedure requires image contrast between illuminated and non-illuminated regions, pattern illumination is essential for the analysis.
A schematic of the optical setup is shown in Fig. S1 of the SI. In this experiment, a horizontally striped light pattern was employed as the pump beam, and the central area of the field of view was analyzed (total image area ≈480.8 × 93.9 µm2, 1024 × 200 pixels, and pixel width = 469 nm). The pump and probe lights were generated from the third and second harmonics of Nd:YAG pulse lasers (GAIA, Rayture Systems) with pulse widths of ≈3 ns and wavelengths of 355 nm and 532 nm, respectively. The irradiated pump area had a diameter of 0.5 mm, and the pulse energies were 0.8 mJ (pump) and 0.02 mJ (probe). The optical resolution of the system was 2–3 µm. Although both refractive index and absorption changes can contribute to the detected signal, the absorption change of BiVO4 was negligible at the probe wavelength (532 nm), under our setup conditions (wide-field detection).27
Clustering analysis
In the PI-PM analysis, classification of the surface charge carrier types was performed using clustering analysis. A representative region (≈30 × 50 µm) within the pump-illuminated area was selected for evaluation. The time-resolved PI-PM dataset contains pixel-wise temporal signals, yielding more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 individual response curves, each composed of 100–200 data points spanning time scales from nanoseconds to milliseconds.
000 individual response curves, each composed of 100–200 data points spanning time scales from nanoseconds to milliseconds.
To categorize these responses, spectral clustering—a statistical classification method based on vector similarity—was employed.31 Each temporal response was treated as a numerical vector, and the similarity between all vectors was quantified using a normalized correlation matrix. The clustering algorithm then grouped the signals exhibiting similar temporal behaviors into distinct categories that reflect different charge carrier dynamics.
Clustering analysis was performed automatically for all pixel-wise temporal responses. Pixels exhibiting extremely low signal intensity, corresponding to noise-dominated or no-response regions, were categorized as outliers based on a predefined signal threshold rather than kinetic features. The fraction of these low-signal responses is summarized in the tables accompanying each PI-PM figure. Finally, the classified responses were spatially mapped onto the corresponding optical image, allowing visualization of the spatial distribution of different charge carrier types and identification of their localized behaviors within the photo-excited region.
Sign of refractive index change
When the probe wavelength (532 nm) lies above the material's bandgap absorption edge, the contribution from optical absorption is negligible. Under such conditions, the PI-PM method predominantly detects refractive index changes that arise from trapped charge carriers at interfaces rather than from electronic transitions.25 The accumulation of charges at an interface generates a local electric field, Elocal, which can be expressed as: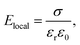 | (1) |
| Δε = εr2rElocal | (2) |
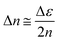 | (3) |
Sample preparation
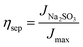 | (4) |
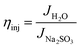 | (5) |
Results and discussion
Fig. 1 shows the SEM images of (a) bare BiVO4, (b) BiVO4/NiFeOx, and (c and d) BiVO4/NiOx photoanodes. The bare BiVO4 film exhibits a homogeneous surface morphology composed of densely packed, particle-like domains with an average diameter of approximately 0.1–0.2 µm. Such a granular surface is typical of BiVO4 films prepared by the MOD method32–35 and ensures sufficient surface area for subsequent co-catalyst deposition.Upon the introduction of NiFeOx, the overall surface morphology remained largely unchanged compared to that of the bare BiVO4. No distinctive aggregation or structural distortion was observed, suggesting that the NiFeOx layer was uniformly deposited on the BiVO4 surface. Energy-dispersive X-ray spectroscopy (EDS) confirmed the homogeneous distribution of Ni and Fe elements across the entire film (Fig. S2(A) in the SI), indicating uniform co-catalyst coverage without significant phase segregation.
In contrast, the BiVO4/NiOx sample exhibited a distinct thin overlayer covering the BiVO4 grains. Lower-magnification SEM images revealed the presence of microcracks on the NiOx layer (Fig. 1(c)), which may have originated during the calcination process due to differences in thermal expansion between the oxide layers but the nano-structure has not been modified much (Fig. 1(d)). EDS analysis confirmed the coexistence and even distribution of Ni and O signals across the surface (Fig. S2(B) in the SI), confirming the formation of a continuous NiOx layer. These surface cracks are considered to locally expose the BiVO4 substrate, potentially a partial contact between BiVO4 and an electrolyte solution. Although such cracks could influence local electrolyte access or interfacial electric fields, they are below the spatial resolution of the PI-PM technique and cannot be directly correlated with the observed carrier-mapping patterns. Therefore, their specific impact on the charge-carrier dynamics cannot be discussed within the present optical framework. In general, the morphology of Ni-based catalysts depends on the synthetic methods and preparation conditions,37,38 but NiFeOx and NiOx exhibit different surface morphologies under similar preparation conditions.
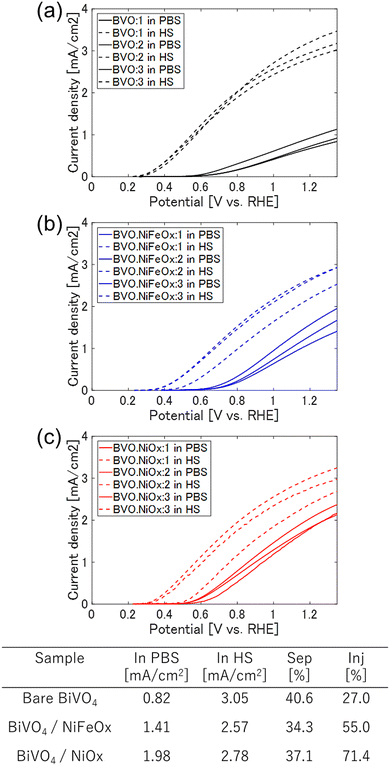
The photoelectrochemical (PEC) characteristics of the BiVO4-based photoanodes (3 samples for each type) were evaluated under AM 1.5G illumination, as shown in Fig. 2. Comparing the average values, the bare BiVO4 electrode exhibited an anodic photocurrent density of 0.82 mA cm−2 at 1.23 V vs. RHE, which was significantly enhanced to 1.41 and 1.98 mA cm−2 by the deposition of NiFeOx and NiOx co-catalysts, respectively. These enhancements correspond to 1.7- and 2.4-fold increases relative to the pristine BiVO4 sample, confirming that both co-catalysts effectively improve the water oxidation processes.
To clarify the origin of the enhanced PEC activity, the charge separation efficiency (ηsep) within the BiVO4 bulk and the charge injection efficiency (ηinj) at the photoanode–electrolyte interface were quantitatively evaluated (Fig. 3). These parameters were obtained from photocurrent measurements in the presence and absence of a hole scavenger and the theoretical maximum value. The bare BiVO4 exhibited limited efficiencies, with ηsep and ηinj of 40.6% and 27.0%, respectively, indicating that both bulk recombination and surface oxidation reactions restricted the overall photoresponse.
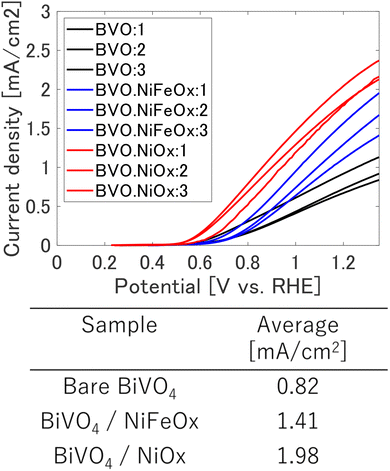 | ||
| Fig. 3 Linear sweep voltammograms (LSVs) of bare BiVO4, BiVO4/NiFeOx, and BiVO4/NiOx photoanodes measured under AM 1.5 G illumination (100 mW cm−2) in 0.1 M phosphate buffer (pH 7.2). | ||
After modification with NiFeOx and NiOx, the ηinj values increased markedly to 55.0% and 71.4%, while ηsep showed only slight variation (34.3% and 37.1%, respectively). Although the change in ηsep is smaller than that observed for ηinj, it exceeds experimental uncertainty and suggests that cocatalyst deposition may influence near-surface charge separation processes. Such effects could arise from the modifications of surface band bending, interfacial electric fields, or suppression of surface recombination. Nevertheless, the substantially larger enhancement in ηinj indicates that improved interfacial hole transfer remains the dominant factor governing the photocurrent increase. This interpretation is consistent with previous reports on metal–oxide cocatalyst modification of BiVO4, where acceleration of surface reaction kinetics plays a more significant role than changes in bulk charge transport.10–18
Among the two cocatalysts, NiOx produced the highest ηinj and the largest increase in photocurrent. This can be ascribed to its favorable band alignment with BiVO4 and high intrinsic hole conductivity, which together facilitate efficient hole extraction and water-oxidation catalysis. The improved ηinj correlates well with the onset-potential cathodic shift observed in the PEC curves (Fig. 2), confirming that NiOx provides superior interfacial energetics and charge-transfer pathways.
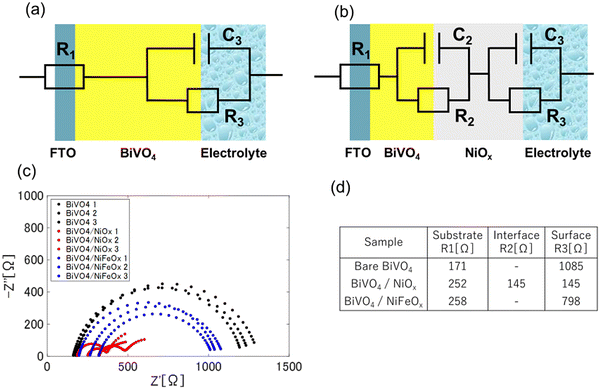
Under AM 1.5G illumination in 0.1 M PBS (pH 7.2), the Nyquist responses of bare BiVO4 and BiVO4/NiFeOx each display a single semicircle, whereas BiVO4/NiOx clearly shows two distinct semicircles, whose radius were smaller than the ones for the former two types. Consistent with this behavior, we employed two different equivalent circuit models (Fig. 4(a) and (b)): for bare BiVO4 and BiVO4/NiFeOx we assumed R1/(R3, C3), while for BiVO4/NiOx we assumed R1/(R2, C2)/(R3, C3). Here, R1 denotes the ohmic resistance due to the substrate/contacts, (R3, C3) represents the primary interfacial charge-transfer channel at the semiconductor/electrolyte boundary, where the charge transfer resistance in the bulk is typically negligible for the BiVO4 anode system. An additional (R2, C2) term in the NiOx case accounts for a second process that emerges after forming the NiOx overlayer due to transport/accumulation within the BiVO4–NiOx interface. The results for bare BiVO4 and BiVO4/NiFeOx are consistent with the previous result,35 while in the case of NiOx, a double-circuit model has not been observed for BiVO4.
The transition from a single to double semicircle upon NiOx loading indicates that, beyond lowering the primary interfacial resistance, NiOx introduces an additional kinetically distinguishable pathway. The reduced radius of the first arc together with the emergence of the second arc suggests (i) facilitated hole injection at the electrolyte interface and (ii) a concurrent NiOx-related storage/transfer process that becomes rate-limiting at lower frequencies.
Quantitative fitting of the PEIS spectra was carried out using equivalent circuit models. The extracted R3 values followed the order BiVO4 (1085 Ω) > BiVO4/NiFeOx (798 Ω) > BiVO4/NiOx (145 Ω), consistent with the PEC performance and ηinj trends obtained from photocurrent analysis. This reduction in R3 demonstrates that Ni-based cocatalysts substantially improve interfacial charge transfer, with NiOx showing the highest catalytic activity for surface oxidation.
Time-resolved image sequences of the refractive index changes upon patterned UV pump irradiation were obtained for (a) bare BiVO4, (b) BiVO4/NiFeOx, and (c) BiVO4/NiOx using the PI-PM method in acetonitrile (ACN), and shown in Fig. 5. The pump pattern is shown schematically at the bottom of each panel, and the analyzed regions are indicated by red rectangles. Upon excitation, a distinct contrast change corresponding to the refractive index modulation increased within 100 ns and decayed within 100 µs for all samples. The observed optical phase change arises from the locally generated photoinduced charge carriers at the semiconductor–electrolyte interface. There were no clear differences of the PI-PM image sequences observed for different types of the samples.
The local charge carrier dynamics of bare BiVO4 were analyzed by clustering the pixel-by-pixel temporal responses obtained from the PI-PM image sequence in ACN (Fig. 6 for region 1 and Fig. S3 in the SI for region 2). Two representative categories of dynamics were identified within the field of view corresponding to the regions marked in Fig. 5(a). One type exhibited a single rise-and-decay response (red) with a rise time of 12 ± 1 ns and a decay time of 0.8 ± 0.1 µs, whereas the other showed a single valley-and-recovery response (blue) with a fall time of 31 ± 2 ns and a recovery time of 1.0 ± 0.1 µs. Areas showing no measurable phase change were mapped in black.
Upon introducing the hole scavenger ethanol (EtOH) as a liquid phase, the red-colored response diminished, while the blue component remained, indicating that the positive (red) response corresponds to surface-trapped holes scavenged by EtOH. Conversely, the addition of nitrobenzene (NB) as an electron scavenger suppressed the blue signal, identifying the negative (blue) response as originating from electrons. The second region showed a similar tendency as the first region. These assignments are consistent with previous PI-PM investigations of TiO2 and BiVO4 particulate systems, where the sign of the refractive index changes due to trapped electron- and hole-related processes.26,28 The coexistence of both carrier types within the same microscopic field demonstrates the intrinsic spatial inhomogeneity of photo-excited carrier dynamics in BiVO4. Such inhomogeneity, linked to variations in the surface defect density and crystallographic orientation, contributes to the moderate charge-injection efficiency and recombination loss observed in the PEC measurements.
The local charge carrier dynamics of the BiVO4/NiFeOx sample were examined using PI-PM and the clustering analysis for two representative regions marked in Fig. 5(b). One example is shown in Fig. 7, while the other is provided in Fig. S4 in the SI. In ACN, compared with the bare BiVO4, the photo-induced responses were further separated into four distinct categories. Two types of rise-and-decay responses (red and cyan) were observed with rise and decay time constants of 16 ± 1 ns/0.4 ± 0.2 µs and 52 ± 3 ns/1.5 ± 0.1 µs, respectively, and two types of valley-and-recovery responses (blue and green) with fall and recovery time constants of 16 ± 1 ns/0.5 ± 0.1 µs and 23 ± 2 ns/1.9 ± 0.1 µs, respectively. The remaining regions showing no measurable change are indicated in black.
 | ||
| Fig. 7 The clustering analyses of the charge carrier responses of a bare BiVO4 in (A) ACN, (B) EtOH, and (C) NB/EtOH in region 1 in Fig. 5(a) correspond to a microscopic image, and the corresponding categorized map is shown in (b), and the scale bar corresponds to 10 µm. The averaged responses for the categorized responses are shown in (c). The area ratios of categories and the rise/decay times for the categories are shown in (d). | ||
Introduction of the hole scavenger (EtOH) selectively diminished only the slower positive (cyan) response, while the faster red response remained unchanged. This behavior suggests that the cyan component corresponds to reactive surface-trapped holes capable of oxidation, whereas the red component represents nonreactive or deeply trapped holes with limited oxidative activity. Conversely, upon addition of the electron scavenger (NB), only the slower negative (green) response decreased, confirming its assignment to reactive trapped electrons, while the faster blue response was unaffected. The second region shows an almost similar tendency to that of the first region. These findings indicate that the deposition of NiFeOx introduces energetically distinct cocatalyst states that selectively capture photogenerated holes, prolonging their lifetime and enhancing their oxidative potential. The emergence of the slow electron response (green) further implies that electron–hole recombination pathways are partially suppressed, extending electron lifetime through secondary trapping processes. The separation of both hole and electron responses into fast (nonreactive) and slow (reactive) components clearly demonstrates the role of NiFeOx in moderating charge recombination and stabilizing interfacial or near-surface accumulated reactive charge carriers participating in solution-phase reactions. In this work, “reactive” carriers are defined operationally as those whose PI-PM temporal responses are selectively altered by the introduction of electron or hole scavengers, indicating their participation in interfacial redox reactions. While PI-PM does not directly prove involvement in water oxidation, the correlation of these carrier populations with enhanced ηinj, reduced interfacial resistance, and improved PEC performance supports their functional relevance to the oxygen-evolution process.
It should be noted that PI-PM detects refractive index changes integrated along the optical path of the probe beam, and its depth resolution is limited by the diffraction-limited confocal parameter of the microscope (a few micron) rather than the optical penetration depth of the pump light. Therefore, nanometer-scale distinction among carriers located within BiVO4, within the cocatalyst overlayer, or exactly at the BiVO4–cocatalyst interface is not directly accessible.
However, the identification of the slow cyan and green components as reactive carriers is supported by their selective suppression in the presence of scavengers. Because scavenger reactions occur at the solid–liquid interface, the affected charge carriers must be located at, or able to reach, the solution interface. Thus, these components are attributed to interfacial or near-surface accumulated charge carriers participating in solution-phase reactions.
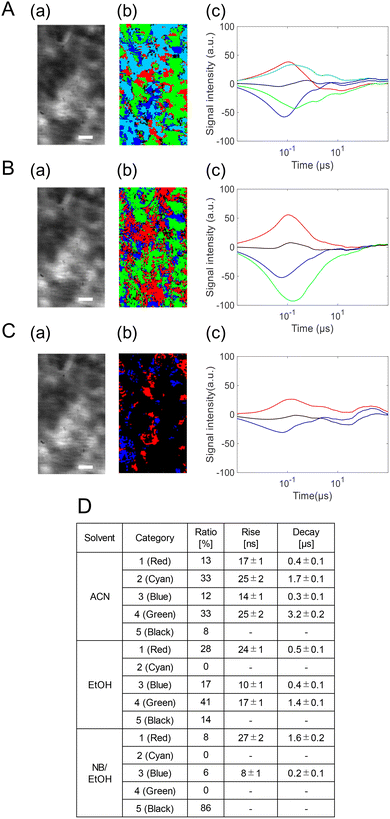
Finally, the PI-PM analysis for the BiVO4/NiOx photoanode revealed further evolution of the microscopic charge carrier behavior compared with BiVO4 and BiVO4/NiFeOx (Fig. 8). Two representative regions indicated in Fig. 5(c) were analyzed; one of them is shown here, and the other in Fig. S5 in the SI. Similarly to the NiFeOx case, both the hole- and electron-related responses were separated into two components, giving four distinct categories. Two rise-and-decay responses were identified with the rise and decay time constants of 17 ± 1 ns/0.4 ± 0.1 µs (red) and 25 ± 2 ns/1.7 ± 0.1 µs (cyan), and two valley-and-recovery responses with the fall and recovery constants of 14 ± 1 ns/0.3 ± 0.1 µs (blue) and 25 ± 2 ns/3.2 ± 0.2 µs (green). Regions without the measurable phase change were mapped in black.
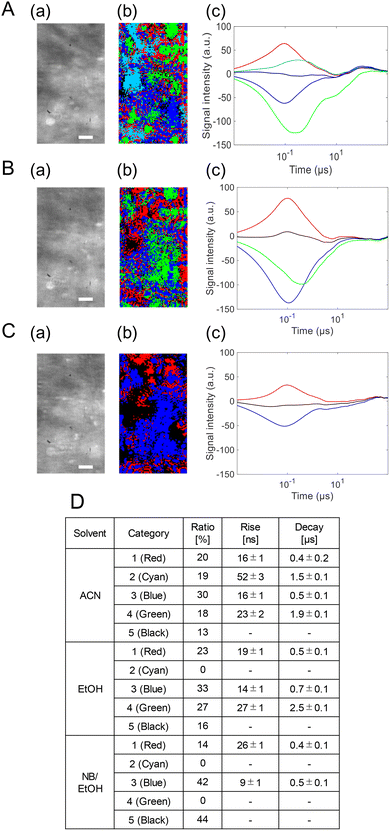 | ||
| Fig. 8 Clustering analyses of the charge carrier responses of the BiVO4/NiFeOx photoanode in (A) ACN, (B) EtOH, and (C) NB/EtOH in region 1 in Fig. 5(b) correspond to a microscopic image, and the corresponding categorized map is shown in (b), and the scale bar corresponds to 10 µm. The averaged responses for the categorized responses are shown in (c). The ratios of the categories and the rise/decay times for the categories are shown in (d). | ||
The scavenger effects were consistent with those observed for BiVO4/NiFeOx: the slower positive (cyan) and slower negative (green) components were selectively suppressed by EtOH (hole scavenger) and NB (electron scavenger), respectively. The second region also showed a similar tendency. This confirms their assignment as reactive trapped holes and reactive trapped electrons, while the faster (red and blue) components correspond to non-reactive or rapidly recombining carriers.
Compared with the NiFeOx case, the spatial fraction of the reactive carrier regions increased markedly: reactive holes from 19 → 33% and reactive electrons from 18 → 33%. Moreover, the lifetimes of the reactive carriers were extended: holes from 1.5 → 1.7 µs and electrons from 1.9 → 3.2 µs. These results indicate that the NiOx overlayer not only enhances charge separation but also prolongs carrier survival, thereby improving the overall reaction efficiency. The enhanced stabilization of both reactive holes and electrons implies that NiOx provides superior interfacial energetics and catalytic activity, consistent with the highest ηinj and PEC performance observed among the samples (Fig. 9).
 | ||
| Fig. 9 Clustering analyses of the charge carrier responses of the BiVO4/NiOx photoanode in (A) ACN, (B) EtOH, and (C) NB/EtOH in region 2 in Fig. 5(c) correspond to a microscopic image, and the corresponding categorized map is shown in (b), and the scale bar corresponds to 10 µm. The averaged responses for the categorized responses are shown in (c). The ratios of the categories and the rise/decay times for the categories are shown in (d). | ||
Discussion
To facilitate a unified comparison of the functional parameters extracted from PEC, PEIS, and PI-PM analyses, the key quantitative metrics are summarized in Table 1. This table highlights the systematic differences in the charge-injection efficiency, separation efficiency, carrier lifetime, and spatial fraction of reactive domains among the bare, NiFeOx-, and NiOx-modified BiVO4 photoanodes. The combined results from SEM, PEC, ηsep/ηinj analysis, and PI-PM measurements provide a consistent picture of how the Ni-based cocatalysts modulate charge separation and transfer in BiVO4 photoanodes. The SEM observations confirmed that both NiFeOx and NiOx form continuous overlayers on the BiVO4 surfaces while maintaining the underlying grain structure, suggesting that the differences in activity arise primarily from electronic and interfacial, rather than morphological variations. The PEC and efficiency analyses reveal that deposition of Ni-based oxides predominantly enhances the charge-injection efficiency (ηinj), while ηsep exhibits only moderate variation. Although cocatalyst loading may influence near-surface separation processes through the modifications of band bending or surface recombination, the substantially larger increase in ηinj indicates that acceleration of interfacial hole transfer is the primary factor governing the performance enhancement. This trend is consistent with previous studies of metal-oxide-modified BiVO4 systems.12 The improvement is most pronounced for NiOx, consistent with its higher photocurrent (PEC) and lower charge-transfer resistance in PEIS.| Sample | PEC [mA cm−2] | Scavenger analysis | PI-PM measurement | ||||
|---|---|---|---|---|---|---|---|
| Separation [%] | Injection [%] | Category | Ratio [%] | Rise [ns] | Decay [µs] | ||
| Bare BiVO4 | 0.82 | 40.6 | 27.0 | Holes | 60 | 12 ± 1 | 0.8 ± 0.1 |
| Electrons | 37 | 31 ± 2 | 1.0 ± 0.1 | ||||
| No response | 3 | — | — | ||||
| BiVO4/NiFeOx | 1.41 | 34.3 | 55.0 | Holes | 20 | 16 ± 1 | 0.4 ± 0.2 |
| Reactive holes | 19 | 52 ± 3 | 1.5 ± 0.1 | ||||
| Electrons | 30 | 16 ± 1 | 0.5 ± 0.1 | ||||
| Reactive electrons | 18 | 23 ± 2 | 1.9 ± 0.1 | ||||
| No response | 13 | — | — | ||||
| BiVO4/NiOx | 1.98 | 37.1 | 71.4 | Electrons | 13 | 17 ± 1 | 0.4 ± 0.1 |
| Reactive electrons | 33 | 25 ± 2 | 1.7 ± 0.1 | ||||
| Holes | 12 | 14 ± 1 | 0.3 ± 0.1 | ||||
| Reactive holes | 33 | 25 ± 2 | 3.2 ± 0.2 | ||||
| No response | 8 | — | — | ||||
The PI-PM results provide microscopic insight into these macroscopic behaviors. In BiVO4/NiFeOx, the cocatalyst introduces micron-scale active sites, generating spatially separated fast (nonreactive) and slow (reactive) hole/electron components. In this work, “reactive” carriers are defined operationally as those whose PI-PM temporal responses are selectively altered by the introduction of electron or hole scavengers, indicating their participation in interfacial redox reactions. While PI-PM does not directly prove involvement in water oxidation, the correlation of these carrier populations with enhanced ηinj, reduced interfacial resistance, and improved PEC performance supports their functional relevance to the oxygen-evolution process. The slower components (cyan and green) represent reactive trapped carriers that participate in surface redox reactions, whose distribution reflects the localized activity of the cocatalyst, even though they were homogeneously coated. In BiVO4/NiOx, a similar spatial distribution is observed, but the proportion of reactive regions increases (holes 19 → 33% and electrons 18 → 33%) and both the hole and electron lifetimes are further prolonged (holes 1.7 µs and electrons 3.2 µs). These changes signify more effective charge separation and stabilization at the NiOx interface. In the present work, the terms “localized” and “uniform” are defined operationally based on the spatial fraction and distribution of the reactive carrier pixels in the PI-PM categorization maps rather than on geometrical domain size analysis. Thus, “localized” refers to a smaller reactive area fraction with spatial discontinuity, whereas “uniform” indicates a larger and more spatially continuous reactive fraction.
The appearance of a second semicircle in the PEIS spectra for BiVO4/NiOx supports the presence of an additional slower charge transfer pathway, corresponding to the interfacial charge accumulation or transport within the BiVO4/NiOx junction, in agreement with the charge carrier dynamics observed in PI-PM. Overall, the synergistic interpretation of PEC, PEIS, and PI-PM results demonstrates that NiOx achieves the most efficient interfacial charge separation and transfer by stabilizing reactive holes and facilitating hole-driven oxidation while minimizing surface recombination losses.
The improvements observed for both NiFeOx- and NiOx-modified BiVO4 photoanodes in this study can be rationalized by considering previous work on their interfacial roles, catalytic characteristics, and electronic interactions with BiVO4. NiFeOx has long been recognized as one of the most active oxygen-evolution cocatalysts for BiVO4, owing to its Fe-mediated enhancement of NiOOH redox activity, its capacity for dynamic hole storage, and its ability to suppress surface recombination.12–14 The mechanistic picture in which NiFeOx accelerates the OER through rapid hole capture into Fe-tuned NiOOH motifs and stabilizes the semiconductor surface by mitigating recombination pathways.
NiOx, although chemically simpler, has been demonstrated to provide a different but complementary set of functionalities, including the p–n junction formation and an increase in surface band bending in BiVO4.17,18 Also, it is indicated that NiOx serves not only as a hole-extraction overlayer but also as a tunable electronic scaffold capable of passivating deep traps and facilitating efficient hole transfer to the electrolyte.15,16 Across these studies, NiOx is shown to improve BiVO4 performance by forming favorable interfacial band alignment, suppressing recombination through surface passivation, and enabling faster OER kinetics.
Although SEM–EDS analysis indicates homogeneous cocatalyst coverage, the PI-PM maps reveal spatially heterogeneous reactive domains, particularly in the NiFeOx case. Such functional inhomogeneity despite uniform structural coverage has been frequently observed in our previous cocatalyst mapping studies27,28,39 and reflects spatial variation in charge accumulation and interfacial transfer efficiency rather than discontinuity of the cocatalyst layer itself.
The localized, long-lived hole-accumulation domains observed for NiFeOx are consistent with previously reported hole-storage behavior associated with Ni–Fe–OOH species in similar systems, which explains why the photocurrent (1.41 mA cm−2) is enhanced yet remains below the highest reported values. However, the PI-PM and PEC measurements provide functional insight into charge accumulation and transfer dynamics rather than direct chemical-state identification. Definitive determination of the catalytic species would require complementary spectroscopic techniques such as XPS, XANES, or operando Raman spectroscopy. In contrast, NiOx forms a more uniform interfacial extraction layer, generating broader regions of slow-decay, hole-reactive signals in the PI-PM maps. Likewise, the broader spatial distribution and enhanced carrier stabilization observed for NiOx are consistent with literature reports suggesting favorable band alignment or p–n junction–like interfacial energetics.17,18 Further confirmation of the electronic-structure characterization is needed to support this interpretation.
Conclusion
In this study, we systematically investigated the effects of NiFeOx and NiOx cocatalysts on BiVO4 photoanodes using a combination of PEC characterization, impedance analysis, and patterned-illumination time-resolved phase microscopy (PI-PM). Both cocatalysts significantly enhanced the photocurrent and charge-injection efficiency (ηinj), confirming their essential roles in accelerating interfacial water-oxidation kinetics. However, the origin of these improvements differs between the two systems. NiFeOx increased ηinj to 55.0% and improved photocurrent through the formation of localized, catalytically active domains that stabilize reactive holes. In contrast, NiOx led to a higher ηinj of 71.4% and achieved the largest photocurrent (1.98 mA cm−2), consistent with its ability to form a uniform interfacial extraction layer with favorable band alignment and effective surface passivation.The PI-PM imaging revealed critical microscopic distinctions underlying these macroscopic enhancements. NiFeOx produced spatially discrete slow-decay carrier regions corresponding to reactive trapped holes and electrons, indicating that a fraction of the cocatalyst surface operates as active catalytic sites. NiOx, on the other hand, generated a broader distribution of reactive domains with prolonged carrier lifetimes, reflecting more efficient interfacial charge stabilization and a more continuous hole-extraction pathway. These PI-PM results provide the first direct visualization of how NiFeOx and NiOx modulate surface carrier populations differently. This insight not only explains the differing performance outcomes observed in this work but also provides design principles for future cocatalyst engineering—namely, optimizing both domain uniformity (as in NiOx) and catalytic site activity (as in NiFeOx) to approach the intrinsic limits of BiVO4-based photoanodes.
Beyond demonstrating the enhanced performance of Ni-based cocatalysts, this study establishes a quantitative link between spatially resolved carrier dynamics and macroscopic injection efficiency. By directly comparing NiFeOx and NiOx under identical conditions, we reveal that spatial continuity and stabilization of reactive carrier domains are key determinants of interfacial charge-transfer efficiency. This comparative framework highlights the utility of PI-PM as a functional diagnostic tool for rational cocatalyst design.
Author contributions
YN performed the experiments and analyses, HN supported the preparation of samples and KK guided the overall research. YN and KK wrote the manuscript and all the authors reviewed it.Conflicts of interest
The authors declare that they have no competing interests or other interests that might be perceived to influence the results and/or discussion reported in this article.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information: Fig. S1: PI-PM optical setup. Fig. S2: EDS results for BiVO4/NiFeOx and BiVO4/NiOx. Fig. S3: PI-PM results for BiVO4 (region 2). Fig. S4: PI-PM results for BiVO4/NiFeOx (region 2). Fig. S5: PI-PM results for BiVO4/NiOx (region 2). See DOI: https://doi.org/10.1039/d5cp05032d.Acknowledgements
This research was financially supported by the Institute of Science and Engineering, Chuo University, the Global - Learning & Academic Research Institution for Master's PhD students, and the Postdocs (LAMP) Program of the National Research Foundation of Korea (NRF) grant funded by the Ministry of Education (No. RS-2024-00445180).References
- A. Kudo, K. Omori and H. Kato, J. Am. Chem. Soc., 1999, 121, 11459–11467 Search PubMed.
- Y. Park, K. J. McDonald and K.-S. Choi, Chem. Soc. Rev., 2013, 42, 2321–2337 RSC.
- C. Martinez Suarez, S. Hernández and N. Russo, Appl. Catal., A, 2015, 504, 158–170 Search PubMed.
- M. Malathi, J. Madhavan, M. Ashokkumar and P. Arunachalam, Appl. Catal., A, 2018, 555, 47–74 CrossRef.
- Q. Wang and K. Domen, Chem. Rev., 2020, 120, 919–985 CrossRef CAS PubMed.
- T. Wang, K. Song, H. Liu, H. Li, Y. Zhang, W. Ren, R. Zhang, K. Li, F. He, Z. Qin and H. Hou, Chem. Eng. J., 2025, 509, 161333 CrossRef CAS.
- Z. Yuan, L. Jiang, H. Yang, Z. Liang, D. Zhang, B. Li, H. Hou, W. Yang and X. Zhan, Small, 2025, 21, e06133 CrossRef CAS PubMed.
- H. Hou, G. Shao, Y. Wang, W.-Y. Wong and W. Yang, Carbon Energy, 2024, 6, e373 CrossRef CAS.
- T. Liu, Z. Pan, K. Kato, J. J. M. Vequizo, R. Yanagi, X. Zheng, W. Yu, A. Yamakata, B. Chen, S. Hu, K. Katayama and C. Chu, Nat. Commun., 2022, 13, 7783 CrossRef CAS PubMed.
- X. Li, Z. Wang, A. Sasani, A. Baktash, K. Wang, H. Lu, J. You, P. Chen, P. Chen, Y. Bao, S. Zhang, G. Liu and L. Wang, Nat. Commun., 2024, 15, 9127 CrossRef CAS PubMed.
- J. Zhang, Y. Huang, X. Lu, J. Yang and Y. Tong, ACS Sustainable Chem. Eng., 2021, 9, 8306–8314 CrossRef CAS.
- B. Zhang, S. Yu, Y. Dai, X. Huang, L. Chou, G. Lu, G. Dong and Y. Bi, Nat. Commun., 2021, 12, 6969 CrossRef CAS PubMed.
- L. Fu, Z. Li, X. Shang and Y. Zheng, J. Mater. Chem. A, 2025, 13, 13951–13961 RSC.
- Y. Kuang, Q. Jia, H. Nishiyama, T. Yamada, A. Kudo and K. Domen, Adv. Energy Mater., 2016, 6, 1501645 CrossRef.
- S. Wang, Z. Shi, K. Du, Z. Ren, H. Feng, J. Wang, L. Wang, D. Cui, Y. Du and W. Hao, Small Methods, 2025, 9, 2401443 Search PubMed.
- D. A. Reddy, Y. Kim, K. A. J. Reddy, M. Gopannagari, A. P. Rangappa, D. Praveen Kumar, A. A. Sasikala Devi, D. Murali, H. S. Ahn and T. K. Kim, ACS Appl. Energy Mater., 2021, 4, 11353–11366 CrossRef CAS.
- M. Zhong, T. Hisatomi, Y. Kuang, J. Zhao, M. Liu, A. Iwase, Q. Jia, H. Nishiyama, T. Minegishi, M. Nakabayashi, N. Shibata, R. Niishiro, C. Katayama, H. Shibano, M. Katayama, A. Kudo, T. Yamada and K. Domen, J. Am. Chem. Soc., 2015, 137, 5053–5060 CrossRef CAS PubMed.
- M. Zhang, R. P. Antony, S. Y. Chiam, F. F. Abdi and L. H. Wong, ChemSusChem, 2019, 12, 2022–2028 Search PubMed.
- I. Grigioni, K. G. Stamplecoskie, E. Selli and P. V. Kamat, J. Phys. Chem. C, 2015, 119, 20792–20800 CrossRef CAS.
- J. Ravensbergen, F. F. Abdi, J. H. van Santen, R. N. Frese, B. Dam, R. van de Krol and J. T. M. Kennis, J. Phys. Chem. C, 2014, 118, 27793–27800 CrossRef CAS.
- S. Selim, L. Francàs, M. García-Tecedor, S. Corby, C. Blackman, S. Gimenez, J. R. Durrant and A. Kafizas, Chem. Sci., 2019, 10, 2643–2652 RSC.
- Y. Suzuki, D. H. K. Murthy, H. Matsuzaki, A. Furube, Q. Wang, T. Hisatomi, K. Domen and K. Seki, J. Phys. Chem. C, 2017, 121, 19044–19052 Search PubMed.
- H. L. Tan, X. Wen, R. Amal and Y. H. Ng, J. Phys. Chem. Lett., 2016, 7, 1400–1405 Search PubMed.
- A. Yamakata, C. S. K. Ranasinghe, N. Hayashi, K. Kato and J. J. M. Vequizo, ACS Appl. Energy Mater., 2020, 3, 1207–1214 Search PubMed.
- K. Katayama, Phys. Chem. Chem. Phys., 2024, 26, 9783–9815 RSC.
- Y. Nakatsukasa and K. Katayama, J. Phys. Chem. C, 2025, 129, 18935–18945 CrossRef CAS.
- T. Chugenji, Z. Pan and K. Katayama, J. Phys. Chem. C, 2022, 126, 19319–19326 CrossRef CAS.
- Y. Egawa, K. Kawaguchi, Z. Pan and K. Katayama, J. Chem. Phys., 2024, 160, 164705 CrossRef CAS PubMed.
- K. Katayama, T. Chugenji and K. Kawaguchi, AIP Adv., 2021, 11, 115215 CrossRef CAS.
- X. Huang, J. Fan, L. Li, H. Liu, R. Wu, Y. Wu, L. Wei, H. Mao, A. Lal, P. Xi, L. Tang, Y. Zhang, Y. Liu, S. Tan and L. Chen, Nat. Biotechnol., 2018, 36, 451–459 CrossRef CAS PubMed.
- U. von Luxburg, Stat. Comput., 2007, 17, 395–416 Search PubMed.
- K. Sayama, A. Nomura, T. Arai, T. Sugita, R. Abe, M. Yanagida, T. Oi, Y. Iwasaki, Y. Abe and H. Sugihara, J. Phys. Chem. B, 2006, 110, 11352–11360 CrossRef CAS PubMed.
- K. Sayama, A. Nomura, T. Arai, T. Sugita, R. Abe, M. Yanagida, T. Oi, Y. Iwasaki, Y. Abe and H. Sugihara, J. Phys. Chem. B, 2006, 110, 11352–11360 Search PubMed.
- S. Zhang, L. Shen, T. Ye, K. Kong, H. Ye, H. Ding, Y. Hu and J. Hua, Energy Fuels, 2020, 34, 5016–5023 Search PubMed.
- K. Kobayashi, Y. Nagai, Z. Pan and K. Katayama, J. Photochem. Photobiol., A, 2023, 440, 114651 CrossRef CAS.
- K. Katayama, Y. Takeda, K. Shimaoka, K. Yoshida, R. Shimizu, T. Ishiwata, A. Nakamura, S. Kuwahara, A. Mase, T. Sugita and M. Mori, Analyst, 2014, 139, 1953–1959 RSC.
- G. Shi, C. Arata, D. A. Tryk, T. Tano, M. Yamaguchi, A. Iiyama, M. Uchida, K. Iida, S. Watanabe and K. Kakinuma, ACS Omega, 2023, 8, 13068–13077 CrossRef CAS PubMed.
- M. K. Beisembekov, G. I. Omarbekova, S. K. Tazhibayev, A. K. Aimukhanov, A. S. Baltabekov, A. Z. Ziyat and A. K. Zeinidenov, Opt. Mater., 2024, 151, 115398 CrossRef CAS.
- T. Chugenji, Z. Pan, V. Nandal, K. Seki, K. Domen and K. Katayama, Phys. Chem. Chem. Phys., 2022, 24, 17485–17495 Search PubMed.
| This journal is © the Owner Societies 2026 |