 Open Access Article
Open Access ArticleNeat ionic liquids and deep eutectic solvents in photonics: status quo and future directions
Mila
Miroshnichenko
a and
Pankaj
Bharmoria
 *ab
*ab
aThe Institute of Materials Science of Barcelona, ICMAB-CSIC, 08193 Bellaterra, Barcelona, Spain
bDepartment of Chemical Engineering, Universitat Politècnica de Catalunya, EEBE, Eduard Maristany 10–14, 08019 Barcelona, Spain. E-mail: pankaj.bharmoria@upc.edu
First published on 14th January 2026
Abstract
In the past decades, ionic liquids (ILs) and deep eutectic solvents (DESs) have gained recognition as green solvents, mainly because of their low vapor pressure, potential for recycling, and customized synthesis. The customization leverages task specificity for a particular application. While the photoactive ILs and DESs have been used as adjuvants in aqueous or organic solution for a variety of applications, this perspective article discusses the relatively underexplored applications of neat ILs or DESs in the field of photonics. Specifically, customization of their chemical structure is discussed to facilitate a particular linear and non-linear optical response, serving as a solvent-free photonic component, including future directions.
Introduction
Ionic liquids (ILs), followed by deep eutectic solvents (DESs), have emerged as potential green solvents in the past two decades for multitude applications ranging from separation and purification,1,2 protein-stability,3–5 energy,6,7 pharmaceuticals,8,9 biotransformation,10,11 detergency,12 metal processing,13 and electrochemistry.14,15 This can be attributed to their properties including low vapour pressure, broad thermal ranges, recycling, biodegradability, specific solvation, and customized easy synthesis.16,17 Neat ILs are salts comprising discrete organic cations associated with discrete organic or inorganic anions via weak electrostatic, H-bonding or London dispersion interactions below 100 °C.18 Neat DESs consist of a eutectic mixture formed by Lewis or Brønsted acids and bases, which may encompass a diverse range of anion and/or cations. The liquidity of DESs at or near room temperature is supported by hydrogen bond interactions between the Lewis or Brønsted acid (donor), and Lewis base (acceptor).19 While both ILs and DESs share similar physical properties, differences in their chemical properties contribute to their applications in diverse fields.20 Photonics is one such field which requires specificity of the chemical nature of ILs or DESs. This is due to the fact that photonics involves the generation, detection, and manipulation of light; therefore, ions with a specific chemical structure can absorb, emit, and scatter the light. While both ILs and DESs offer advantage of the customization of the chemical structure, they have been underexplored in the field of photonics as neat-solvents. This perspective article discusses the limited work reported in relation to the specific chemical structure of IL or DES components, as well as their implications in future linear and non-linear optical applications. For better understanding of the cross-disciplinary readership ILs and DESs and linear and non-linear optics are briefly described before the main discussion.Ionic liquids and deep eutectic solvents
An ionic liquid can be simply described using the following formula.| M+·X− |
 | ||
| Fig. 1 Chemical equations and molecular structures of (a) ionic liquids and (b) deep eutectic solvents. | ||
A deep eutectic solvent is a eutectic mixture with a lower melting temperature than that of its constituent components. It can be described using the following formula.25
| M+·X−·zY |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio leads to the formation of DES; [Cho]+⋯Cl−⋯2HNCONH2 has a a lowered freezing temperature (Tf ≈ 12 °C) due to hydrogen bond interaction of urea with Cl− (see Fig. 1b).26 DESs can be of type I to IV depending on their structure, the details of which can be obtained from the following cited ref. 27.
2 molar ratio leads to the formation of DES; [Cho]+⋯Cl−⋯2HNCONH2 has a a lowered freezing temperature (Tf ≈ 12 °C) due to hydrogen bond interaction of urea with Cl− (see Fig. 1b).26 DESs can be of type I to IV depending on their structure, the details of which can be obtained from the following cited ref. 27.
Linear and non-linear optics
In linear optics, an incident light wave (generally weak) causes vibration of a molecule, followed by the emission of a light wave that has the same frequency as the incident light (Fig. 2a). Therefore, linear optics follows the superposition principle; A + B = X + Y,28 where A and B are input light waves hitting the molecule giving output light waves X and Y with the same frequency. Under the application of a weak electric field, the loosely bonded electrons in the materials undergo slight displacement from their equilibrium position to induce an electric dipole moment (![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) (t)). Since electrons near the nucleus are bound more strongly, their displacement potential is approximated as harmonic. Therefore, as per the harmonic approximation, the
(t)). Since electrons near the nucleus are bound more strongly, their displacement potential is approximated as harmonic. Therefore, as per the harmonic approximation, the ![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) scales linearly with the electric field (
scales linearly with the electric field (![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) ) amplitude of the incident light as per the following eqn (1).28,29
) amplitude of the incident light as per the following eqn (1).28,29![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) (t) = ε0χ (t) = ε0χ![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) (t) (t) | (1) |
![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) is the induced dipole moment per unit volume, ε0 is the permittivity of the free space, and χ is the polarization susceptibility. The common linear optical properties are reflection, refraction, scattering, and diffraction etc.
is the induced dipole moment per unit volume, ε0 is the permittivity of the free space, and χ is the polarization susceptibility. The common linear optical properties are reflection, refraction, scattering, and diffraction etc.
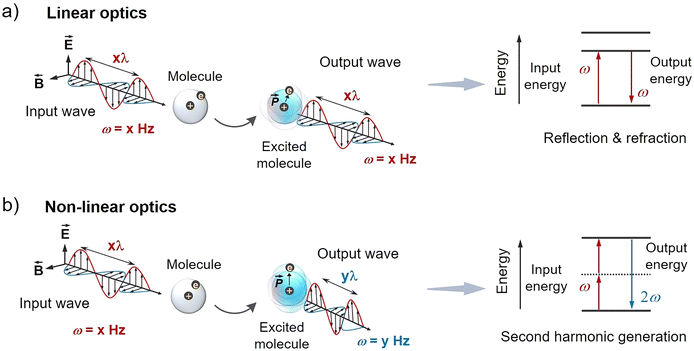 | ||
| Fig. 2 Illustration of (a) linear and (b) non-linear optical responses of a molecule upon excitation with a linearly polarized light wave. λ = wavelength; ω = frequency of the light wave. | ||
In non-linear optics, an intense light wave upon interaction with the molecule changes its optical properties such as frequency (Fig. 2b). Non-linear optics does not follow the superposition principle; A + B ≠ X + Y, hence the optical properties like frequency, polarization and phase of the incident light undergoes change as the polarization density of the medium reacts non-linearly to the electric field of the light. Non-linearity is displayed by the molecules when the electric field strength of the light is high compared to the intra-atomic electric field i.e. Ea ≈ 2 × 107 electrostatic unit.
Due to the strong electric field, the binding potential of electrons near the nucleus cannot be approximated as harmonic, resulting in large displacements from the equilibrium position. Therefore, the electron displacement does not scale linearly with the electric field as the non-linear electron motions become more significant. Hence, the induced polarization in non-linear optics is defined by eqn (2).30
![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) NL = χ(1) NL = χ(1)![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) + χ(2) + χ(2)![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) 2χ(3) 2χ(3)![[E with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0045_20d1.gif) 3 + ⋯ 3 + ⋯ | (2) |
Heterogeneity, polarity and viscosity of ionic liquids and deep eutectic solvents
During the light–matter interaction in solution, the solvent polarity and viscosity play a crucial role in altering the photonic properties by influencing the electronic transitions and molecular dynamics.31 The probability of electronic transition between different quantum states can be measured using the transition dipole moment integral eqn (3).32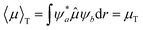 | (3) |
![[small mu, Greek, circumflex]](https://www.rsc.org/images/entities/i_char_e0b3.gif) ) between the quantum states
) between the quantum states 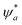 and ψb, integrated over all coordinates, dr. If μT ≠ 0, transition is allowed (e.g. singlet to singlet transition), and when μT = 0, transition is forbidden (e.g. singlet to triplet transition). The solvent polarity affects the transition dipole moment indirectly by altering the electronic distribution in the molecule in the ground and the excited state, which affects its interaction with light through induced polarization as per eqn (1)–(3) (Fig. 2). The nanoscale heterogeneity of ILs separates the ions into distinct polar and non-polar domains.33,34 Polar domains are formed by charged headgroups of cations and anions, stabilized by electrostatic and hydrogen-bonding interactions, whereas non-polar domains are formed by aggregation of alkyl chains due to van der Waals interactions (Fig. 3).35
and ψb, integrated over all coordinates, dr. If μT ≠ 0, transition is allowed (e.g. singlet to singlet transition), and when μT = 0, transition is forbidden (e.g. singlet to triplet transition). The solvent polarity affects the transition dipole moment indirectly by altering the electronic distribution in the molecule in the ground and the excited state, which affects its interaction with light through induced polarization as per eqn (1)–(3) (Fig. 2). The nanoscale heterogeneity of ILs separates the ions into distinct polar and non-polar domains.33,34 Polar domains are formed by charged headgroups of cations and anions, stabilized by electrostatic and hydrogen-bonding interactions, whereas non-polar domains are formed by aggregation of alkyl chains due to van der Waals interactions (Fig. 3).35
 | ||
| Fig. 3 Illustration of the segregated polar and non-polar domain in neat ILs. The domain length varies with an increase in the size of cation or anion headgroups and their alkyl chain length. | ||
More importantly the length and structure of diversity in the polar/non-polar domain can be altered by changing the molecular structure of the cation or anion and their alkyl chain length.36,37 For instance, the domain structure can vary from spatial heterogenous to liquid crystalline with an increase in the alkyl chain length.36 The distinct structure, local environment, diffusion, reorientation and association dynamics of IL ions in different domains play a key role in governing the optical properties of them or of the molecules dissolved in them.38–41 This is because the variations in polarity across domains alters the stability of the ground and excited state of the molecule due to the different interaction energies of the solvent dipoles with the dipoles of excited chromophores leading to inhomogeneous spectral broadening and bathochromic or hypochromic spectral shifts.42 Similar to NILs, DESs also exhibit differential polarity due to the heterogeneity in their structure which varies with the structure of their constituents.43–46 The differential polarity can also affect the molecular solubility of chromophores to influence the absorption coefficients and emission lifetimes between different states.47 The differential polarity of NILs and DESs can be investigated using Kamlet–Taft's solvent parameters (α, β and π*) using standard equations.48–50 These parameters denote the hydrogen bond donor capacity (α), hydrogen bond acceptor capacity (β), and polarizability (π*) of the liquid. Kamlet–Taft's parameters can be used to design ILs/DESs or their binary mixtures with specific polarity for a specific photonic application in the liquid-state. This is because the change in α, β and π* affects both the solvation and diffusion of the dissolved chromophores, which affects both excited-state dynamics (monomer vs excimer emission) and energy transfer.51–53 For example, aggregation induced emission enhancement (AIE), or quenching which are highly sensitive to solvent polarity.54–57 In the case of AIE the molecular motions (rotation and vibrations) cease due to the packing of molecules in a certain geometry, which blocks the non-radiative decay (knr) to boost the radiative decay (kr) to enhance the fluorescence quantum yield (Φf) according to the following eqn (4).
 | (4) |
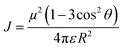 | (5) |
Viscosity is another key property which influences the photonic properties of molecules by controlling the polarization, diffusion of chromophores and oxygen. The viscosity (η) is related to the diffusion coefficient of a molecule (D) in the solution via Stokes–Einstein eqn (6).61
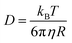 | (6) |
Neat ionic liquids as photonic components
The customized nature of ILs leverage the possibility of engineering the chemical structures of both cation and anions for responding to the light waves of a specific energy due to electric field polarization.78 However, the question, of what happens to NILs upon exposure to electromagnetic radiation of a specific wavelength is a key aspect of their photonic application,79 considering the change in structural heterogeneity,80 formation of possible transient photoexcited species,81,82 and changes in the properties of the input wave.83 The most common IL cation imidazolium [Cnmim]+ paired with an optically less active anion to leverage an inherent optical property in the broad UV-blue region.84 The neat [Cnmim]+ paired with the BF4− anion shows absorption maxima at ∼160 and 210 nm with the tail extending into the 400 nm range.84 However, their emission spectra display excitation wavelength dependent red shifts of the photoluminescence, known as the “red edge effect (REE) (Fig. 4a).”85,86 | ||
| Fig. 4 (a) Illustration of the modified Jablonski diagram showing electronic transitions, vibrational and dipolar relaxation during the red edge effect due to photoselection of certain chromophores experiencing different electric fields due to the interactions of dipoles of excited chromophores with the solvent dipole when excited at the edge of the absorption spectrum. (b) Illustration of the change in emission spectrum during the red-edge effect with a change in the excitation energy. (a) Was adapted from ref. 87 with permission from [Elsevier B.V.] [Demchenko, Biophys. Chem., 1982, 15, 101–10987], copyright 1982. | ||
In REE the emission spectra undergo a red-shift when excitation is performed at longer wavelengths (Fig. 4a and b). The REE is generally observed in condensed inhomogeneous medium with high polarity and restricted motion, like at low temperature or at high viscosity where dielectric relaxation slows down with a shift to low frequency and longer relaxation times (τR).85,86 In a typical case chromophores whose dipole moment and dipole orientation changes in the excited state (μe) with respect to the ground state (μg) show REE due to the interactions of medium dipole (μm) with the chromophore's dipoles (dipolar interactions). As a result, the chromophore experiences a different electric field with an added energy contribution from dipole–dipole interactions as per the following relationships.87
| E = E0 + Ω(Wdd) | (7) |
Wedd ∝ ![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) e·〈 e·〈![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) m〉 m〉 | (8) |
Wgdd ∝ ![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) g·〈 g·〈![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) m〉 m〉 | (9) |
Fluorescent imidazolium based NILs due to the segregation of polar/non-polar ensembles create ground-state heterogeneity, with a local energetically distinct species having low 0-0 transition energy, moreover enhanced viscosity with the increase in chain length slow down the dielectric relaxation of other components around photo selected imidazolium causing the red-edge effect (Fig. 4b).88,89 Moreover, these shifts become more prominent with an increase in the alkyl chain of the cation in neat IL ([C2mim][BF4] ∼ 280 nm; [C8mim][BF4] ∼ 390 nm).90 While the red-edge spectroscopy is exploited to study the dynamics of proteins in an aqueous solution of various additives by excitation at the red-edge of the tryptophan absorbance,91 considering the widespread use of neat imidazolium or pyridinium ILs in biocatalysis,92 the red-edge effect of ILs93 could be exploited as a probe to understand the structure–function relationship of enzymes upon cross-excitation at the red-edge of both tryptophan and neat ILs.
Besides inherent fluorescence, imidazolium cations have been chemically engineered with chiral emitters to display circularly polarized luminescence (CPL)94,95 for application as a CPL emitter (Fig. 4c) in electrochemical cells. The CPL is a phenomenon where the chiral chromophore with luminescence dissymmetry (glum) shows differential emission intensity of the right (IR) or left (IL) components of the circularly polarized light as per the following relationship (Fig. 5).96
| glum = 2(IL − IR)/IL − IR) | (10) |
Other than cations, anion customization has also been exploited to achieve suitably photoactive NILs. For instance, pairing of allyl-imidazolium cations of varying chain lengths with fluorescent [MnClxBry]n− anions resulted in phase change NILs with distinct thermochromic properties, which were exploited as sensors for temperature, relative humidity and alcohols by impregnating on the paper.98 Kovalenko and coworkers paired photo-emissive lead halide anions with phosphonium cation as scintillators for application in fast neutron imaging with recoil proton detection, with high resolution and low γ-ray sensitivity (Fig. 6).99
 | ||
| Fig. 6 (a) Illustration of the neutron scintillation by IL via generation of recoil proton after fast neutron bombardment, followed by recombination emission through the formation of electron–hole pairs. (b) Illustration of the neutron scintillation imaging using NIL as a scintillator. Fig. 6a and b adapted from ref. 99 with CC-BY-NC-ND 4.0 open access license from [American Chemical Society] [Kovalenko et al., ACS Photonics, 2021, 8, 3357–3364], copyright 2021. | ||
Neutron scintillation is an optical process where neutrons interact with the material to produce tiny flashes of light, detected further by the photomultiplier tube. These NILs exhibit specific properties such as proton rich cations, narrow excitation, low scattering, high photoluminescence quantum yield and large Stokes shift (1.7 eV) to avoid reabsorption desired for higher spatial resolution.99 The H-rich cations of IL generate recoil protons upon bombardment with fast neutrons, forming electron–hole pairs upon interaction with anions, which show high PL emission via charge recombination (Fig. 6a).
Hisamoto and coworkers demonstrated Förster resonance energy transfer (FRET) in NIL, containing phosphonium cations, paired with sulfonated pyrene anions as the donor (D), and lipophilic fluorescein anions as the acceptor (A).100
FRET is a non-radiative energy transfer process that occurs via dipole–dipole interactions between the donor and acceptor molecules separated by <100 Å. As a result, the emission of the acceptor is increased at the expense of the donor's emission due to FRET (Fig. 7). The energy transfer efficiency (E) in FRET can be estimated using the following relationship.28
| E = R06/R06 + r6 | (11) |
The FRET in the NIL mixture was utilized to form a solvent doped-PVC membrane with improved ion extraction-based sensing.100 The liquid nature of the IL played a key role here for supporting the molecular diffusion of anionic chromophores (donor and acceptor), in the trapped FRET liquid for efficient energy transfer.
Kimizuka and co-workers demonstrated Dexter energy transfer (DET) in NIL comprising tetra-alkyl phosphonium cations and photoactive anions such as sulfonated anthracene and 4-(2-phenyloxazol-5-yl) benzene for green to blue, and blue to UV photon upconversion via triplet–triplet annihilation photon upconversion (TTA-UC)101–103 (Fig. 8).
 | ||
| Fig. 8 (a) Illustration of the sequential energy transfer in TTA-UC. The sensitizer/donor upon absorption of low energy photons goes to the triplet-state via intersystem crossing (ISC), followed by the r triplet energy transfer to the annihilator (TET), which undergoes triplet–triplet annihilation (TTA), leading to the formation of an excited singlet-state of the annihilator, which show the anti-Stokes delayed emission. (b) Illustration of the change in emission spectra of photosensitizer and annihilator during TTA-UC. (c) Crystal structure of ionic crystals of [C14(C2)3P]+[DPASO3]− viewed along the b-axis, showing a lamellar structure consisting of ionic and non-ionic layers. Inset: The structure viewed along the c-axis to see a long tail alkyl chain sandwiched between two anthracene rings. (c) Has been reproduced from ref. 102 with permission from [The Royal Society of Chemistry, London] [Kimizuka et al., Phys. Chem. Chem. Phys., 2018, 20, 3233–3240], copyright 2018. | ||
TTA-UC is a non-linear optical process that occurs in the annihilator's ensembles doped with a triplet photosensitizer in the oxygen free conditions. The photosensitizer upon excitation at low energy undergoes intersystem to the triplet-state and while coming back to the ground-state sensitize the annihilator triplets via triplet–triplet energy transfer. The sensitized annihilator triplets then undergo annihilation via spin-exchange coupling, resulting in the formation of an emissive singlet-state of the annihilator which show upconversion emission (Fig. 8a). The probability of singlet formation (f) can be estimated using the following relationship.101
| f = 2 × ΦUC/Φisc·ΦTET·ΦTTA·ΦF | (12) |
The high viscosity of NILs plays a key role in TTA-UC as it decreases the oxygen diffusion which protects chromophore triplets oriented between non-polar domains of the IL, from quenching by the ground state triplet oxygen in aerated conditions (Fig. 8c).101–103 Moreover, the confinement of annihilators in contiguous ionic arrays allow effective triplet-energy transfer for an efficient energy upconversion.104
The customized nature of ILs has been exploited to generate laser pulses either in the bulk or confined liquid domain.105–108 In an interesting report Kupfer and coworkers utilized the vibrational energy in the chemical bonds of NIL, ethyl-3-methylimidazolium dicyanamide ([C2mim][dca]) to generate nanosecond mid infra-red orange radiation (603 nm) by down-conversion of 532-nm laser pulses via molecular stimulated Raman scattering (SRS) (Fig. 9).105
 | ||
| Fig. 9 Illustration of the spectral changes during photon downconversion of the 532 nm laser pulse due to stimulated Raman scattering in an IL; ([C2mim][dca] with Raman active dicyanamide anion. Adapted from ref. 105 with permission from [American Physical Society] [Palmer et al., Phys. Rev. Appl., 2023, 19, 014052], copyright 2023. | ||
SRS is a non-linear optical phenomenon that involves the inelastic scattering of photons from a coherently excited state of the Raman active material.
Consequently, the pump wavelength is transformed into Stokes and anti-Stokes wavelengths, which are shifted by the dominant bond vibrational frequency. The Raman frequency shift in SRS can be estimated using the following relationship.105
| ωR = ωL ± NRωv | (13) |
In the ethyl-3-methylimidazolium dicyanamide, the down converted emission corresponds to a Raman shift of 2200 cm−1 due to dominant bond vibration of the dicynamide anion at this frequency.105
Besides ethyl-3-methylimidazolium dicyanamide, molecular SRS was also observed with other NILs, comprising bis(trifluoromethylsulfonyl)amide anion paired with 1-butyl-1-methylpyrrolidinium and butylpyridinium cation for downconverting the 532 nm laser pulse to 578 nm, 599 and 632 nm via SRS.105 Further customization of the NIL structure with a suitable Raman active cation or anion can also lead to SRS based energy upconversion to produce high frequency pulses with lifetimes varying between nano to femtoseconds.105
Sun and coworkers used the NIL, 1-butyl-3-methyl imidazolium hexafluorophosphate, as a gain medium to develop a microfluidic ionic liquid dye laser using BODIPY as the dye operating via whispering gallery modes (WGMs) based lasing emission.106 WGM is an optical phenomenon generally observed in micro-sized circular resonators due to the circulation of light via total internal reflection along the curved boundary, resulting in the formation of resonance patterns.107,108 The microdroplets of 1-butyl-3-methyl imidazolium hexafluorophosphate doped with BODIPY when pumped with 485 nm pulsed laser above the lasing threshold of 40.1 µJ mm−2 showed WGM-based lasing emission at 527 nm different from the normal fluorescence emission of BODIPY.106 The refractive index and viscosity of the used IL played a key role to form droplets with optically smooth surfaces to avoid losses due to scattering during lasing.
NIL, Deng and coworkers reported electro-optical modulation by hydrophobic NILs, in the NIR region (λ = 1330 or 1530 nm) under the application of a strong electric field (E = ±4 V) due to N–H or C–H vibration and their overtone and combination transitions leading to radiationless deactivation or absorption in a microfluidic device.109 The electro-optical modulation amplitude increased due to the change in carrier concentration near the electrodes with an increase in the electrical conductivity of ILs.
Borra and coworkers exploited the higher reflectance in the infra-red region, low vapour pressure under vacuum, and low freezing temperature ∼ 175 K of silver coated NIL, 1-ethyl-3-methyl imidazolium ethylsulfate, to be used as a liquid mirror in an infra-red lunar telescope.110 The reflectance (R) is an optical property of the material indicating the fraction of light reflected in comparison to the transmitted light. The R depends on the angle of incidence (θ) and polarization of light (P), and refractive index (n) of the medium. The R of a liquid at θ = 0° can be calculated using the Fresnel equation.111
| R = (n1 − n2|n1 + n2)2 | (14) |
Besides reflectance, the liquidity and surface tension112 of NILs leverage the advantage of the shape change to create mirrors with high parabolic surfaces in microgravity. While it was considered a great breakthrough at that time, no further developments related to this were reported in the open literature until a recent internship report by Szobody et al.113 The team conducted the work at NASA Goddard Space Flight Center to develop self-healing silver nanoparticle coated NIL mirrors for the next generation of liquid space telescopes.113 This is an exciting, but highly underexplored area in NIL research with direct application in space exploration telescopes.114,115 A representative image showing the reflection of NIR light by the IL coated silver nanoparticles is shown as Fig. 10.
Due to their nano-heterogeneous structure and polarizability NILs can diffract the light of a specific wavelength corresponding to the dimension of nano-heterogeneity.116 This property makes the NILs a useful additive to increase the diffraction efficiency, resolution, and sensitivity of photopolymerizable hologram materials (Fig. 11).117,118 Moreover customization of their viscosity makes them suitable diffusing solvents during photopolymerization.117,118
 | ||
| Fig. 11 Figure showing the improved diffraction efficiency of the photopolymerizable material with the addition of ionic liquids. Has been adapted from ref. 117 with permission from [AIP Publishing} [Veith et al. Appl. Phys. Lett., 2008, 93, 141101], copyright 2008. | ||
Neat deep eutectic solvents as photonic components
Similar to ILs, the customized nature of DESs can be exploited to develop photo-responsive DESs by substitution of one of the components with a photoactive molecule. Neat DESs (NDESs) are considered next generation green solvents as replacements for ILs due to similarities in their physical properties.20,27 However, unlike NILs, NDESs have been less explored in photonics which gives an opportunity to develop this field. The refractive index (linear or no-linear) is among the most significant optical properties of DESs which leverage their potential applications as a medium for liquid-based optical communications.119–121 The non-linear refractive index (n2) varies with the light intensity (I) as per the following eqn (15).121| n − n0 = Δn = n2I | (15) |
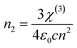 | (16) |
Gupta and coworkers developed a fluorescent NDESs (heptyltriphenylphosphine (M+), bromide (X−), and decanoic acid (zY)) which were used as sensors to detect D2O via an increase in fluorescence intensity.124 Hopkins and coworkers exploited the customized nature of DESs to synthesize chiral DESs comprising alkylammonium chloride salts as HBA and a chiral HBD (amino acids, organic acids and monosaccharide) to leverage the CPL property to a racemic mixture of luminescent dissymmetric lanthanide complexes.125,126 Further changing the HBA with alkylphosphonium chloride enhanced the enthalpy and entropy of chiral discrimination by 50%, demonstrating the advantage of the customized nature of DESs.127
Murakami and co-workers exploited the thermal stability of DESs as solvents to host organic chromophores (PtOEP, PdOEP and DPA) to achieve high efficiency upconversion of green photons to blue photons.128 Unlike the IL-based TTA-UC system where chromophores were part of the IL structure,101–104 DESs have been used just as native solvents. However, advancements can be made in this direction by introducing the HBD property to the TTA-UC chromophores to develop solvent free TTA-UC DESs.
The dynamic heterogeneity, hydrogen bond acidity or basicity of NDESs has also been exploited to study excited state proton-transfer (ESPT) operating at the nanosecond timescale using either pyranine or naphthol's as proton-transfer probes.129,130 ESPT is a photochemical process where a proton rapidly shifts within a molecule after absorbing light, creating a new, tautomeric form with different fluorescence or vibrational spectra.131 ESPT can be understood using the following equations.131
| R−OH (S0) + hν1 → R−OH* (S1) + A → R*O− (S1) + A–H+ | (17) |
| R*O− (S1) → RO− (S0) + hν2 | (18) |
| R*O− (S1) → RO− (S0) + heat | (19) |
Conclusions
The customized nature of NILs by large, and NDESs to some extent, has been exploited for chemical engineering of their structure for a specific linear or non-linear optical application. These processes include the red-edge effect, circularly polarized luminescence, neutron scintillation, photon conversion via FRET, DET, stimulated Raman scattering, electro-optical modulation, micro-fluidic dye lasers, reflector for liquid mirrors, directors in holograms, and refractors for liquid-based optical communication and medium for ESPT. However, these studies are limited to the proof-of-concept level, though with potential applications discussed in the respective sections. Other than the photonic part, properties like viscosity, polarity, and refractive index, broad thermal range, structural heterogeneity, conductivity, large electrochromic window and hydrogen bond acidity or basicity play a key role in their successful implementation in the reported photonic systems, as summarized in Table 1. Upon evaluation of the use of NDESs in comparison to NILs within photonic systems, it becomes evident that NDESs are still in the early stages of development, despite sharing numerous physicochemical characteristics with NILs. They are regarded as an advanced version of NILs primarily due to their environmental sustainability, which is attributed to their low toxicity, biodegradability, and low-cost synthesis.132 While these attributes may provide NDESs with certain advantages over NILs in certain applications, the requirements for many photonic applications necessitate photoactive materials that exhibit excellent photostability along with broad absorption and emission ranges. Consequently, one of the essential components must include either a polyaromatic hydrocarbon (which is typically considered toxic), metals,133 semiconductor nanoparticles,134,135 or perovskite compounds (may use toxic metals like Pb and Cd)136,137 and carbon nanoparticles.138,139 NIL also offers great structural versatility to design photoactive ionic liquids with a vast number of polyaromatic organic cations or anions because of its two-component nature. Whereas the structural complexity of DESs due to the multicomponent nature held together by hydrogen bonding limits its versatility beyond bio-derived components. Therefore, currently NILs are leading the photonic applications in the neat form compared to NDESs. However, the sustainable nature of DESs is of high value compared to NILs when considered only as solvents to disperse photoactive compounds to study various optical processes,128,130,131 followed by easy separation at the end-of-life. For consideration purely as a photonic component more NDESs with polyaromatic dyes as one of the components need to be synthesized for a valid comparison with NILs in photonic applications.| Neat ionic liquid | Photonic property | Properties of NIL exploited |
|---|---|---|
| [C2–8mim]+[BF4]− and [C4mim]+[PF6]− | Optical absorption and emission | Customization, and polarity84 |
| [Cnmim]+[X]− (n = 2, 4, 8; X = Cl, BF4, PF6) | Red edge effect | Heterogeneity, polarity and viscosity88–90,93 |
| [Cnmpy]+[X]− (n = 3, 4, 8; X = BF4, N(CN)2) | ||
| [CnRmim]+[I]− (R = diphenylacrylonitrile; n = 1, 7, 11) | Circularly polarized luminescence | Structural customization, chirality94,95,97 |
| [C2TCBNim]+[PF6]−, [(C4)4N]+[X]− (X = Pro, Val, Asp) | ||
| [C14(C6)3P]+[PbX4]−, [(C4)4P]+[PbX4]− | Neutron scintillation | Proton richness, viscosity, low scattering99 |
| [C14(C6)3P]+[X]− (X = HP-SO3, C12-FL) | Förster resonance energy transfer | Structure customization, viscosity, polarity100 |
| [C14(C6)3P]+[DPASO3]−, [C14(C2)3P]+[DPASO3]− | Dexter energy transfer in TTA-based photon upconversion | Structure customization, heterogeneity, viscosity, thermal stability, polarity101–104 |
| [C14(C6)3P]+[PPOSO3]− | ||
| [C2mim]+[N(CN)2]−, [X]+[NTf2]− (X = C4mPyr, C4Pyr) | Stimulated Raman scattering based photon up and down conversion | Structure customization, thermal stability105 |
| [C4mim]+[PF6]− | Gain medium in microfluidic dye laser operational via whispering gallery modes | Refractive index, and viscosity106 |
| [Cnmim]+[X]− (n = 1 or 4; X = NTf2, BF4, ClO4, PF6, N(CN)2) | Electro-optical modulation | Conductivity, viscosity, electrochromic window109 |
| [C2mim]+[C2OSO3]− | Reflectance | Liquidity, refractive index, surface tension, low vapour pressure, broad thermal range110,112 |
| [C4mim]+[PF6]− and [C4mim]+[BF4]− | Diffraction | Heterogeneity, polarizability, viscosity117,118 |
| Neat deep eutectic solvent | Photonic property | Properties of DES exploited |
|---|---|---|
| [Cho]+[Cl]−: lactic acid | Negative non-liner refraction due to the thermal lensing effect | Negative refractive index, heterogeneity, liquidity121 |
| Heptyltriphenylphosphine bromide: decanoic acid | Fluorescence | Structural customization, polarity124 |
| [C4)4N]+Cl−: zY (zY = D-(+)-α-glucose; D-(+)-β-glucose; L-(−)-α-glucose; D-(–)-β-fructose). [C4)4N]+Cl−: zY: zY or [Cho]Cl: zY (zY = L-lactic acid, L-leucic acid, L-ascorbic acid, R/S-acetoxypropionic acid, and methyl-(S)-lactate). [C4)4N]+Cl− or [C4)4P]+Cl−: zY (zY = L- or D-glutamic acid, L-proline, and L-arginine). | Circularly polarized luminescence | Structural customization, chirality125–127 |
| [Cn)4N]+Cl−: decanoic acid (n = 4 or 8) | Dexter energy transfer in TTA-based photon upconversion | Liquidity, polarity, thermal stability128 |
| [M]+[X]−: zY (M = [(C2)4N or Cho; X = Cl or Br; zY = ethylene glycol, or glycine or urea or tetraethylene glycol) | Excited-state proton transfer | Hydrogen bond acidity, liquidity, nano-heterogeneity and polarity130,131 |
What is next for NILs and DESs in photonics?
Thermal stability is among the key properties of NILs and DESs to be exploited in photonics, as it can address the heating effects during light–matter interactions.140–143 Heating can negatively alter the optical properties, such as resonance frequencies, and signal integrity due to the thermal cross-talk and change in the refractive index to affect the device performance.144 However, the higher heat capacities of NILs and DESs can serve as a heat sink in the case of thermo-optical effects. The studies on photonics in the liquid-state (chemical or metallic liquids) are slowly gaining momentum.145,146 NILs and DESs can contribute immensely as an organic liquid to this, with the customizable nature of their thermophysical and chemical properties. For example, liquid water, due to its absorption in the tetra-hertz range, generates a broadband terahertz wave.145,146 In case of liquid gallium, the high plasma frequency has been exploited to realize ultra-violet plasmonic structures.147 In case of liquid metal nanostructures, the absence of domain boundaries and the smoothness of surfaces results in an extended carrier relaxation time, consequently leading to reduced losses for surface plasmons in addition to broad thermal ranges.148 Along similar lines, NILs and DESs can be explored as liquid organic/inorganic semiconductors149,150 by synthesizing the variants comprising polyaromatic hydrocarbons and liquid metal salts. While a few proof-of-concept studies, especially on NILs, have already been reported in this direction,103,104 broadening the scope of detailed fundamental investigations related to applicable photonics can further attract the attention of the scientific community in this direction. For instance, the feasible advancement of liquid-junction solar cells151 or liquid solar cells152 as a sustainable clean energy alternative could significantly enhance the use of NIL or DES-based liquid organic semiconductors as in situ photon converters103,104,153,154 or solar concentrators155,156 to capture light either below or above the solar cell's band gap, thereby improving performance.155–158 While both NILs and DESs have been used as electrolyte or redox mediators in dye sensitized solar cells (DSSCs)159–161 due to their conductivity and non-volatility, using them as a dye, electrolyte and redox mediator in a single system would be an ideal advancement in this direction. For example, anthracene-based dyes have been used as dye and photon upconverters in DSSCs,157 hence suitable NDESs and NILs comprising redox active dyes if synthesized can serve both as sensitizer dyes, photon converters, and redox mediators in DSSCs. Both NILs and NDESs possess suitable properties for applications in liquid luminescent solar concentrators.162 For example, the high refractive index, heterogenous structure, differential polarity to solubilize luminophores and more importantly structural tunability to design liquids containing luminophores with high Stokes-shifts and thermal stability. Despite having suitable properties NILs163,164 or DESs have been rarely investigated for LSCs. With the emergence of liquid based LSCs, both NILs and NDESs could become promising future candidates for LSCs if suitable design principles considering the photostability of luminophores are envisioned. Owing to their thermochemical phase change characteristics, both NILs and DESs exhibit significant potential as materials for thermal energy storage.165–167 Molecular solar thermal energy storage systems (MOST) are emerging as clean energy alternatives where solar energy is stored as heat in molecular photoswitches.168–170 Besides fundamental research, recent years have seen a lot of reports on the development of MOST devices,167,171 in which photoswitches were dissolved in organic solvents (generally toxic), and were tested at different flow-rates, and residence-times to absorb maximum solar photons to be stored as heat by the photisomer.172 A typical example of MOST is the norbornadiene (NBD) ↔ quadricyclane (QC) system, where NBD, after absorbing UV photons, gets converted to the QC form, which stores heat as high as 93 kJ mol−1.169 Besides NBD, several other organic photoswitches like azobenzene, azopyrazole, dihydroazulene, and ortho-dianthrylbenzenes are emerging as suitable solar-thermal energy storage materials.169,173 As a green alternative to organic solvents, mixtures of water and non-ionic surfactants have been explored at high NBD concentrations, generally needed to store higher energy.174 However, water–surfactant mixtures are prone to phase separation above a certain concentration of the photoswitch or surfactant due to the solubility limitations. NILs or NDESs can overcome the solubility limitations as the photoswitches can be transformed directly into thermally stable NILs or DESs with required viscosities and vapor pressure through appropriate chemical engineering. This transformation can lead to improved heat storage capabilities, as it combines the heat storage resulting from the phase change in NILs or DESs with the heat absorbed from solar photons by the photoswitches.Author contributions
P. B. conceived the idea of this perspective article, wrote the first draft and revised the manuscript. M. M. assisted in editing the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information is available. See DOI: https://doi.org/10.1039/d5cp04142b. The Supplementary information Table S1, contains full names of the abbreviations used in this manuscript.All figures presented in this manuscript were prepared by the authors themselves and any information related to their drawing is available from the corresponding author upon reasonable request by email. Appropriate references have been cited where needed in relation to the presented figures.
Acknowledgements
P. B. acknowledges the Ramón y Cajal grant from the Spanish State Investigation Agency (grant no. J03416) and La-Caixa junior research leadership-post doctoral project PHOLCEB (ID: 100010434, fellowship code: LCF/BQ/P122/11910023) for financial support. M. M. and P. B. acknowledge the State Investigation Agency, through the Severo Ochoa Programme for Centres of Excellence in R&D (CEX2023-001263-S) and project PID2021-123873NB-I00 for financial support.Notes and references
- S. P. M. Ventura, F. A. Silva, M. V. Quental, D. Mondal, M. G. Freire and J. A. P. Coutinho, Sep. Sci., 2016, 39, 3505–3520 CrossRef PubMed.
- P. Bharmoria, A. A. Tietze, D. Mondal, T. S. Kang, A. Kumar and M. G. Freire, Chem. Rev., 2024, 124, 3037–3084 CrossRef CAS PubMed.
- N. Yadav and P. Venkatesu, Phys. Chem. Chem. Phys., 2022, 24, 13474–13509 RSC.
- A. Sanchez-Fernandez, J. H. Nicholson, S. M. M. Huaman, C. A. Romero, J.-F. Poon, S. Prevost and A. P. S. Brogan, Commun. Chem., 2025, 8, 173 CrossRef PubMed.
- T. Zhou, C. Gui, L. Sun, Y. Hu, H. Lyu, Z. Wang, Z. Song and G. Yu, Chem. Rev., 2023, 123, 12170–12253 CrossRef CAS PubMed.
- S. Chattopadhyay, S. Kataria, S. Prusty, M. S. ubban, A. Kumar and S. Sutradhar, ACS Symp. Ser., 2025, 1504, 135–153 CrossRef CAS.
- R. M. Moshikur, M. R. Chowdhury, M. Moniruzzaman and M. Goto, Green Chem., 2020, 22, 8116–8139 RSC.
- E. Chevé-Kools, Y. H. Choi, C. Roullier, G. Ruprich-Robert, R. Grougnet, F. Chapeland-Leclerc and F. Hollmann, Green Chem., 2025, 27, 8360–8385 RSC.
- Q. Hou, X. Qi, M. Zhen, H. Qian, Y. Nie, C. Bai, S. Zhang, X. Baia and M. Ju, Green Chem., 2021, 23, 119–231 RSC.
- Y. Gu and F. Jérôme, Chem. Soc. Rev., 2013, 42, 9550–9570 RSC.
- P. Bharmoria, M. J. Mehta, I. Pancha and A. Kumar, J. Phys. Chem. B, 2014, 118(33), 9890–9899 CrossRef CAS PubMed.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060–11082 CrossRef CAS PubMed.
- D. R. MacFarlane, M. Forsyth, P. C. Howlett, J. M. Pringle, J. Sun, G. Annat, W. Neil and E. I. Izgorodina, Acc. Chem. Res., 2007, 40, 1165–1173 CrossRef CAS PubMed.
- Z. Liu, F. Feng, W. Feng, G. Wang, B. Qi, M. Gong, F. Zhang and H. Pang, Energy Environ. Sci., 2025, 18, 3568–3613 RSC.
- S. Zhang, N. Sun, X. He, X. Lu and X. Zhang, J. Phys. Chem. Ref. Data, 2006, 35, 1475–1517 CrossRef CAS.
- D. Yu, Z. Xue and T. Mu, Chem. Soc. Rev., 2021, 50, 8596–8638 RSC.
- K. Fumino, S. Reimanna and R. Ludwig, Phys. Chem. Chem. Phys., 2014, 16, 21903–21929 RSC.
- L. J. B. M. Kollau, M. Vis, A. van den Bruinhorst, A. C. C. Esteves and R. Tuinier, Chem. Commun., 2018, 54, 13351–13354 RSC.
- H. Zhang, J. M. Vicent-Luna, S. Tao, S. Calero, R. J. J. Riobóo, M. L. Ferrer, F. del Monte and M. C. Gutiérrez, ACS Sustainable Chem. Eng., 2022, 10, 1232–1245 CrossRef CAS.
- R. Hayes, G. G. Warr and R. Atkin, Chem. Rev., 2015, 115, 6357–6426 CrossRef CAS PubMed.
- D. Lengvinaitė, S. Kvedaraviciute, S. Bielskutė, V. Klimavicius, V. Balevicius, F. Mocci, A. Laaksonen and K. Aidas, J. Phys. Chem. B, 2021, 125, 3255–13266 CrossRef PubMed.
- María J. Dávila, S. Aparicio, R. Alcalde, B. García and J. M. Leal, Green Chem., 2007, 9, 221–232 RSC.
- A. A. Bodour, N. Alomari, G. P. Duran, S. Rozas, G. I. Silva, S. Aparicio, A. G. Vega and M. Atilhan, Energy Fuels, 2025, 39, 12791–12829 CrossRef.
- Z. Lei, B. Chen, Y.-M. Koo and D. R. MacFarlane, Chem. Rev., 2017, 117, 6633–6635 CrossRef PubMed.
- B. B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang, J. M. Klein, A. Horton, L. Adhikari, T. Zelovich, B. W. Doherty, B. Gurkan, E. J. Maginn, A. Ragauskas, M. Dadmun, T. A. Zawodzinski, G. A. Baker, M. E. Tuckerman, R. F. Savinell and J. R. Sangoro, Chem. Rev., 2021, 121, 1232–1285 CrossRef CAS PubMed.
- J. A. Sirviö, M. Visanko and H. Liimatainen, Green Chem., 2015, 17, 3401–3406 RSC.
- V. Pandey, T. Pandey and R. S. Kaundal, Deep Eutectic Solvents ACS Symposium Series 2025, vol. 1504, ch. 3, pp. 77–95.
- P. Bharmoria and S. P. M. Ventura, Optical Applications of Nanomaterials, in Nanomaterials for healthcare, energy, and environment. Advanced structured materials, ed. A. Bhat, I. Khan, M. Jawaid, F. Suliman, H. Al-Lawati and S. Al-Kindy, vol. 118, Springer, Singapore, 2019 Search PubMed.
- https://www.chem.uci.edu/~potma/lecture3.pdf .
- G. Agrawal, Nonlinear fiber optics, Elsevier Inc., 5th edn, 2013, ch. 1, pp. 1–25 Search PubMed.
- S. L. Bondarev, V. N. Knyukshto and S. A. Tikhomirov, J. Appl. Spectrosc., 2002, 69, 230–237 CrossRef CAS.
- Transition moment in IUPAC Compendium of Chemical Terminology, International Union of Pure and Applied Chemistry, 5th edn, 2025.
- R. Hayes, G. G. Warr and R. Atkin, Chem. Rev., 2015, 115, 6357–6426 CrossRef CAS PubMed.
- M. Sha, Y. Liu, H. Dong, F. Luo, F. Jiang, Z. Tang, G. Zhu and G. Wu, Soft Matter, 2016, 12, 8942–8949 RSC.
- Y.-L. Wang, B. Li, S. Sarman, F. Mocci, Z.-Y. Lu, J. Yuan, A. Laaksonen and M. D. Fayer, Chem. Rev., 2020, 120, 5798–5877 CrossRef CAS PubMed.
- Y. Ji, R. Shi, Y. Wang and G. Saielli, J. Phys. Chem. B, 2013, 117, 1104–1109 Search PubMed.
- M. Torkzadeh and M. Moosavi, J. Phys. Chem. B, 2020, 124, 11446–11462 CrossRef CAS PubMed.
- Z. Hu and C. J. Margulis, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 831–836 CrossRef CAS PubMed.
- V. H. Paschoal, L. F. O. Faria and M. C. C. Ribeiro, Chem. Rev., 2017, 117, 7053–7112 Search PubMed.
- T. Frömbgen, K. Drysch, T. Tassaing, T. Buffeteau, O. Hollóczki and B. Kirchner, Angew. Chem., Int. Ed., 2025, 64, e202502885 CrossRef PubMed.
- P. Oulevey, S. Luber, B. Varnholt and T. Bürgi, Angew. Chem., Int. Ed., 2016, 55, 11787–11790 Search PubMed.
- A. Marini, A. M. Losa, A. Biancardi and B. Mennucci, J. Phys. Chem. B, 2010, 114, 17128–17135 CrossRef CAS PubMed.
- S. Spittle, D. Poe, B. Doherty, C. Kolodziej, L. Heroux, M. A. Haque, H. Squire, T. Cosby, Y. Zhang, C. Fraenza, S. Bhattacharyya, M. Tyagi, J. Peng, R. A. Elgammal, T. Zawodzinski, M. Tuckerman, S. Greenbaum, B. Gurkan, C. Burda, M. Dadmun and E. J. M. Joshua Sangoro, Nat. Commun., 2022, 13, 219 CrossRef CAS PubMed.
- M. N. Kamar, A. Mozhdehei, B. Dupont, R. Lefort, A. Moréac, J. Ollivier, M. Appel and D. Morineau, J. Chem. Phys., 2025, 163, 134506 CrossRef CAS PubMed.
- A. Malik and H. K. Kashyap, Phys. Chem. Chem. Phys., 2023, 5, 19693–19705 RSC.
- T. Rinesh, H. Srinivasan, V. K. Sharma, V. García Sakai and S. Mitra, J. Chem. Phys., 2025, 162, 244503 CrossRef PubMed.
- R. K. Venkatraman and A. J. Orr-Ewing, Acc. Chem. Res., 2021, 54, 4383–4394 CrossRef CAS PubMed.
- X. Wang, S. Zhang, J. Yao and H. Li, Ind. Eng. Chem. Res., 2019, 58, 7352–7361 CrossRef CAS.
- M. A. Ab Rani, A. Brant, L. Crowhurst, A. Dolan, M. Lui, N. H. Hassan, J. P. Hallett, P. A. Hunt, H. Niedermeyer, J. M. Perez-Arlandis, M. Schrems, T. Welton and R. Wilding, Phys. Chem. Chem. Phys., 2011, 13, 16831–16840 RSC.
- A. Pandey, R. Rai, M. Pala and S. Pandey, Phys. Chem. Chem. Phys., 2014, 16, 1559–1568 RSC.
- Y.-D. Song, Q. T. Wang and L. Chen, J. Phys. Org. Chem., 2023, 36, e4474 CrossRef CAS.
- R. K. Venkatraman and A. J. Orr-Ewing, Acc. Chem. Res., 2021, 54, 4383–4394 CrossRef CAS PubMed.
- Q. Zhou, M. Zhou, Y. Wei, X. Zhou, S. Liu, S. Zhang and B. Zhang, Phys. Chem. Chem. Phys., 2017, 19, 1516–1525 RSC.
- J. Mei, N. L. C. Leung, R. T. K. Kwok, J. W. Y. Lam and B. Z. Tang, Chem. Rev., 2015, 115, 11718–11940 CrossRef CAS PubMed.
- S. Suzuki, S. Sasaki, A. S. Sairi, R. Iwai, B. Z. Tang and G. Konishi, Angew. Chem., Int. Ed., 2020, 59, 9856–9867 CrossRef CAS PubMed.
- C. Gao, L. Fu, J. Wang, Y. Chu, L. Gao, H. Qiu and J. Chen, Chem. Biomed. Imaging, 2025, 3, 837–848 CrossRef CAS PubMed.
- A. Banerjee, K. De and U. Bhattacharjee, J. Phys. Chem. B, 2024, 128, 849–856 CrossRef CAS PubMed.
- C. R. Benson, L. Kacenauskaite, K. L. VanDenburgh, W. Zhao, B. Qiao, T. Sadhukhan, M. Pink, J. Chen, S. Borgi, C.-H. Chen, B. J. Davis, Y. C. Simon, K. Raghavachari, B. W. Laursen and A. H. Flood, Chem, 2020, 6, 1978–1997 CAS.
- X. Ma, R. Sun, J. Cheng, J. Liu, F. Gou, H. Xiang and X. Zhou, J. Chem. Educ., 2016, 93, 345–350 CrossRef CAS.
- Y. Huang, J. Xing, Q. Gong, L.-C. Chen, G. Liu, C. Yao, Z. Wang, H.-L. Zhang, Z. Chen and Q. Zhang, Nat. Commun., 2019, 10, 169 CrossRef PubMed.
- J. T. Edward, J. Chem. Educ., 1970, 47, 261 CrossRef CAS.
- C. Y. Victor Gray, J. Mårtensson and K. Börjesson, J. Am. Chem. Soc., 2019, 141, 9578–9584 CrossRef PubMed.
- A. A. Veldhorst and M. C. C. Ribeiro, J. Chem. Phys., 2018, 148, 193803 CrossRef PubMed.
- Y. L. Wang, B. Li, S. Sarman, F. Mocci, Z.-Y. Lu, J. Yuan, A. Laaksonen and M. D. Fayer, Chem. Rev., 2020, 120, 798–5877 Search PubMed.
- R. Clark, M. A. Nawawi, A. Dobre, D. Pugh, Q. Liu, A. P. Ivanov, A. J. P. White, J. B. Edel, M. K. Kuimova, A. J. S. McIntosh and T. Welton, Chem. Sci., 2020, 11, 6121–6133 RSC.
- S. Chatterjee, S. H. Deshmukh and S. Bagchi, J. Phys. Chem. B, 2022, 126, 8331–8337 CrossRef CAS PubMed.
- L. Naimovičius, M. Dapkevičius, E. Radiunas, M. Miroshnichenko, G. Kreiza, C. Alcaide, P. Baronas, Y. Sasaki, N. Yanai, N. Kimizuka, A. B. Pun, M. Solà, P. Bharmoria, K. Kazlauskas and K. Moth-Poulsen, Chem. Sci., 2025, 16, 20255–20264 RSC.
- H. Kouno, Y. Sasaki, N. Yanai and N. Kimizuka, Chem. – Eur. J., 2019, 25, 6042 CrossRef CAS.
- P. Bharmoria, S. Hisamitsu, H. Nagatomi, T. Ogawa, M.-A. Morikawa, N. Yanai and N. Kimizuka, J. Am. Chem. Soc., 2018, 34, 10848–10855 CrossRef PubMed.
- P. Bharmoria, S. Hisamitsu, Y. Sasaki, T. S. Kang, M.-A. Morikawa, B. Joarder, K. Moth-Poulsen, H. Bildirir, A. Mårtensson, N. Yanai and N. Kimizuka, J. Mater. Chem. C, 2021, 9, 11655–11661 RSC.
- F. J. Morgan and H. Dugan, Appl. Opt., 1979, 18, 4112–4115 CrossRef CAS PubMed.
- A. Muratsugu, J. Watanabe and S. Kinoshita, J. Chem. Phys., 2014, 140, 214508 CrossRef PubMed.
- A. Raj, P. Verma, A. Beliaev, P. Myllyperkiö and T. Kumpulainen, Chem. Sci., 2025, 16, 13935–13943 RSC.
- F. Serra and E. M. Terentjev, Macromolecules, 2008, 41, 981–986 CrossRef CAS.
- M. W. Dale, D. J. Cheney, C. Vallotto and C. J. Wedge, Phys. Chem. Chem. Phys., 2020, 22, 28173–28182 RSC.
- N. Gao, Y. Yang, Z. Wang, X. Guo, S. Jiang, J. Li, Y. Hu, Z. Liu and C. Xu, Chem. Rev., 2024, 124, 27–123 CrossRef CAS PubMed.
- L. V. T. D. de Alencar, S. B. Rodríguez-Reartes, F. W. Tavares and F. Llovell, ACS Sustainable Chem. Eng., 2024, 12, 7987–8000 CrossRef PubMed.
- T. S. Kang, M.-A. Morikawa, M. Singh and N. Kimizuka, J. Am. Chem. Soc., 2025, 147, 8809–8819 CrossRef CAS PubMed.
- M. Mahato, Y. Murakami and S. K. Das, Appl. Mater. Today, 2023, 32, 101808 CrossRef.
- Y. Wang, J. Phys. Chem. B, 2009, 113, 11058–11060 CrossRef CAS PubMed.
- J. Leier, N. C. Michenfelder and A.-N. Unterreiner, Chem. Open, 2021, 10, 72–82 CAS.
- K. P. Rola, A. Zając, A. Szpecht, D. Kowal, J. Cybińska, M. Śmiglak and K. Komorowska, Eur. Polym. J., 2021, 156, 110615 CrossRef CAS.
- T. S. Groves and S. Perkin, Faraday Discuss., 2024, 253, 193–211 RSC.
- I. Tanabe, Y. Kurawaki, Y. Morisawa and Y. Ozaki, Phys. Chem. Chem. Phys., 2016, 18, 22526–22530 RSC.
- A. P. Demchenko, Luminescence, 2002, 17, 19–42 CrossRef CAS PubMed.
- W. C. Galley and R. M. Purkey, Proc. Natl. Acad. Sci. U. S. A., 1970, 67, 1116–1121 CrossRef CAS PubMed.
- A. P. Demchenko, Biophys. Chem., 1982, 15, 101–109 CrossRef CAS PubMed.
- A. Paul, P. K. Mandal and A. Samanta, J. Phys. Chem. B, 2005, 109, 9148–9153 CrossRef CAS PubMed.
- Z. Hu and C. J. Margulis, Acc. Chem. Res., 2007, 40, 1097–1105 CrossRef CAS PubMed.
- T. Singh and A. Kumar, J. Phys. Chem. B, 2008, 112, 4079–4086 CrossRef CAS PubMed.
- Red-Edge Excitation Spectroscopy of Proteins with the FS5 Spectrofluorometer, Edinburgh Instruments Ltd., 2014. https://www.edinst.com/wp-content/uploads/2018/06/Red-Edge-Excitation-Proteins_App16.pdf Search PubMed.
- F. van Rantwijk and R. A. Sheldon, Chem. Rev., 2007, 107, 2757–2785 CrossRef CAS PubMed.
- I. Bandrés, M. Haro, B. Giner, H. Artigas and C. Lafuente, Phys. Chem. Liq., 2011, 49, 192–205 CrossRef.
- H. Guo, Q. Yu, Y. Xiong and F. Yang, J. Mol. Liq., 2021, 335, 116179 CrossRef CAS.
- C. Feng, K. Zhang, B. Zhang, L. Feng, L. He, C.-F. Chen and M. Li, Angew. Chem., Int. Ed., 2025, 64, e202425094 CrossRef CAS PubMed.
- G. Longhi, E. Castiglioni, J. Koshoubu and G. S. Abbate, Chirality, 2016, 28, 696–707 CrossRef CAS PubMed.
- B. Zercher and T. A. Hopkins, Inorg. Chem., 2016, 55, 10899–10906 CrossRef CAS PubMed.
- Z. Huang, M. Yi, Y. Xu, P. Qi, Y. Liu, A. Song and J. Hao, J. Mater. Chem. C, 2021, 9, 13276 RSC.
- V. Morad, K. M. McCall, K. Sakhatskyi, E. Lehmann, B. Walfort, A. S. Losko, P. Trtik, M. Strobl, S. Yakunin and M. V. Kovalenko, ACS Photonics, 2021, 8, 3357–3364 CrossRef CAS PubMed.
- T. Mizuta, K. Sueyoshi, T. Endo and H. Hisamoto, Anal. Chem., 2021, 93, 4143–4148 CrossRef CAS PubMed.
- S. Hisamitsu, N. Yanai and N. Kimizuka, Angew. Chem., Int. Ed., 2015, 54, 11550–11554 CrossRef CAS PubMed.
- S. Hisamitsu, N. Yanai, H. Kouno, E. Magome, M. Matsuki, T. Yamada, A. Monguzzi and N. Kimizuka, Phys. Chem. Chem. Phys., 2018, 20, 3233–3240 RSC.
- S. Hisamitsu, J. Miyano, K. Okumura, J. K.-H. Hui, N. Yanai and N. Kimizuka, Chem. Open, 2020, 9, 14–17 CAS.
- K. Shimizu, S. Hisamitsu, N. Yanai, N. Kimizuka and J. N. C. Lopes, J. Phys. Chem. B, 2020, 124, 3137–3144 CrossRef CAS PubMed.
- R. Kupfer, F. Wang, J. F. Wishart, M. Babzien, M. N. Polyanskiy, I. V. Pogorelsky, T. Rao, L. Cultrera, N. Vafaei-Najafabadi and M. A. Palmer, Phys. Rev. Appl., 2023, 19, 014052 CrossRef CAS.
- H. Zhang, C. Zhang, S. Vaziri, F. Kenarangi and Y. Sun, IEEE Photonics J., 2021, 13, 1500308 CAS.
- A. N. Oraevsky, Quantum Electron., 2002, 32, 377 CrossRef CAS.
- T. Reynolds, N. Riesen, A. Meldrum, X. Fan, J. M. M. Hall, T. M. Monro and A. François, Laser Photonics Rev., 2017, 11, 1600265 CrossRef.
- X. He, Q. Shao, P. Cao, W. Kong, J. Sun, X. Zhang and Y. Deng, Lab Chip, 2015, 15, 1311–1319 RSC.
- E. F. Borra, O. Seddiki, R. Angel, D. Eisenstein, P. Hickson, K. R. Seddon and S. P. Worden, Nature, 2007, 447, 979–981 CrossRef CAS PubMed.
- E. Hecht, Optics, Pearson Education, 4th edn, 2001 Search PubMed.
- M. Tariq, M. G. Freire, B. Saramago, J. A. P. Coutinho, J. N. C. Lopes and L. P. N. Rebelo, Chem. Soc. Rev., 2012, 41, 829–868 RSC.
- T. Szobody, Reflecting the future: Revolutionizing space exploration with liquid mirrors, NC Space Grant News, 2024 Search PubMed.
- N. Rowlands, Á. Romero-Calvo, D. Strafford, R. Kamire, A. Childers, S. F. Yates, E. Rahislic, J. Smoke, S.-H. Zheng, P. Cameron, G. Cano-Gómez, H. Chen, T. Hu, E. Comstock and M. Herrada, Proc. SPIE, 2024, 13100, Advances in Optical and Mechanical Technologies for Telescopes and Instrumentation VI, 131007H Search PubMed.
- X. Hou, Z. Wang, Z. Zheng, J. Guo, Z. Sun and F. Yan, ACS Appl. Mater. Interfaces, 2019, 11, 20417–20424 CrossRef CAS PubMed.
- I. Bandrés, M. Haro, B. Giner, H. Artigas and C. Lafuente, Phys. Chem. Liq., 2011, 49, 192–205 CrossRef.
- H. Lin, P. W. Oliveira and M. Veith, Appl. Phys. Lett., 2008, 93, 141101 CrossRef.
- H. Lin, P. W. de Oliveira and M. Veith, Opt. Mater., 2011, 33, 759–762 CrossRef CAS.
- Y. Chen, D. Zhao, Y. Bai, Y. Duan, C. Liu, J. Gu, X. Wang, X. Sun, Y. Li and L. Zhang, J. Mol. Liq., 2022, 348, 118031 CrossRef CAS.
- J. Luan, Y. Cheng, F. Xue, L. Cui and D. Wang, ACS Omega, 2023, 8, 25582–25591 CrossRef CAS PubMed.
- E. Ferreira, G. Ramos-Ortiz, A. Vazquez and M. Trejo-Durán, J. Mol. Liq., 2023, 384, 122253 CrossRef CAS.
- I. Betka, Nonlinear optical refractive index measurements of pure water via Z-scan technique at 800 nm, arXiv, 2025, preprint, arXiv:2502.02944 DOI:10.48550/arXiv.2502.02944.
- P. L. Rall, D. Förster, T. Graf and C. Pflaum, Opt. Express, 2022, 30, 38643–38662 CrossRef CAS PubMed.
- S. M. Patil, S. K. Gupta, D. Goswami and R. Gupta, ACS Omega, 2023, 8, 32444–32449 CrossRef CAS PubMed.
- L. V. Elzen and T. A. Hopkins, ACS Sustainable Chem. Eng., 2019, 7, 16690–16697 CrossRef.
- B. Nelson, L. V. Elzen, G. Whitacre and T. A. Hopkins, ChemPhotoChem, 2021, 5, 1071–1078 CrossRef CAS.
- C. R. Wright, L. V. Elzen and T. A. Hopkins, J. Phys. Chem. B, 2018, 122, 8730–8737 CrossRef CAS PubMed.
- Y. Murakami, S. K. Das, Y. Himuro and S. Maeda, Phys. Chem. Chem. Phys., 2017, 19, 30603–30615 RSC.
- V. Khokhar, Deepika and S. Pandey, J. Photochem. Photobiol., A, 2022, 427, 113798 CrossRef CAS.
- Sangeeta, A. Sil, V. Singh, R. Bhati and B. Guchhait, J. Phys. Chem. B, 2024, 128, 9805–9814 CrossRef CAS PubMed.
- P. Zhou and K. Han, Acc. Chem. Res., 2018, 51(7), 1681–1690 CrossRef CAS PubMed.
- Z. Usmani, M. Sharma, M. Tripathi, T. Lukk, Y. Karpichev, N. Gathergood, B. N. Singh, V. K. Thakur, M. Tabatabaei and V. K. Gupta, Sci. Total Environ., 2023, 88, 163002 CrossRef PubMed.
- K. L. Kelly, E. Coronado, L. L. Zhao and G. C. Schatz, J. Phys. Chem. B, 2003, 107, 668–677 CrossRef CAS.
- U. Banin, Y. Ben-Shahar and K. Vinokurov, Chem. Mater., 2014, 26, 97–110 CrossRef CAS.
- M. Bhar, N. Bhunia, G. H. Debnath, D. H. Waldeck and P. Mukherjee, Chem. Phys. Rev., 2024, 5, 011306 CrossRef CAS.
- J. Chang, H. Chen, G. Wang, B. Wang, X. Chena and H. Yuan, RSC Adv., 2019, 9, 7015–7024 RSC.
- T. Antrack, M. Kroll, M. Sudzius, C. Cho, P. Imbrasas, M. Albaladejo-Siguan, J. Benduhn, L. Merten, A. Hinderhofer, F. Schreiber, S. Reineke, Y. Vaynzof and K. Leo, Adv. Sci., 2022, 9, 2200379 CrossRef CAS PubMed.
- F. Migliorini, S. Belmuso, R. Dondè, S. D. Iuliis and I. Altman, Carbon Trend., 2022, 8, 100184 CrossRef CAS.
- N. Tarasenka, A. Stupak, N. Tarasenko, S. Chakrabarti and D. Mariotti, ChemPhysChem, 2017, 18, 1074–1083 CrossRef CAS PubMed.
- R. Jiang, Y. Guo and X. Guo, Sol. Energy Mater. Sol. Cells, 2026, 294, 113934 CrossRef CAS.
- Y. U. Paulechka, A. G. Kabo, A. V. Blokhin, G. J. Kabo and M. P. Shevelyova, J. Chem. Eng. Data, 2010, 55, 2719–2724 Search PubMed.
- B. D. Rabideau, K. N. West and J. H. Davis, Jr., Chem. Commun., 2018, 54, 5019–5031 Search PubMed.
- P. Dehury, J. Singh and T. Banerjee, ACS Omega, 2018, 3, 18016–18027 CrossRef CAS PubMed.
- E. Sperling, Mapping the Impact of heat on photonics, Semiconductor Engineering 2019, https://semiengineering.com/mapping-the-impact-of-heat-on-light/.
- Q. Jin, K. Williams, E. Yiwen, J. Dai and X. Zhang, Observation of broadband terahertz wave generation from liquid water, in Nonlinear Optics, OSA Technical Digest (online) (Optica Publishing Group, 2017), paper NW3A.1.
- Q. Jin, Y. W. E, J. M. Dai, L. L. Zhang, C. L. Zhang, A. Tcypkin, S. Kozlov and X. Zhang, Terahertz photonics in liquids, in Nonlinear Optics (NLO), OSA Technical Digest (Optica Publishing Group, 2019), paper NTu2B.3.
- P. Q. Liu, X. Miao and S. Datta, Opt. Mater. Express, 2023, 13, 699–727 Search PubMed.
- P. C. Wu, T.-H. Kim, A. S. Brown, M. Losurdo, G. Bruno and H. O. Everitt, Appl. Phys. Lett., 2007, 90, 103119 CrossRef.
- C. Kunkel, J. T. Margraf, K. Chen, H. Oberhofer and K. Reuter, Nat. Commun., 2021, 12, 2422 CrossRef CAS PubMed.
- C. Wang, H. Dong, L. Jiang and W. Hu, Chem. Soc. Rev., 2018, 47, 422–500 RSC.
- A. Chelsea, Are Liquid Solar Panels the Next Big Thing in Solar Power? Green Lancer, 2024, https://www.greenlancer.com/post/liquid-solar.
- P. V. Kamat, K. Tvrdy, D. R. Baker and E. J. Radich, Chem. Rev., 2010, 110, 6664–6688 Search PubMed.
- T. Ullrich, D. Munz and D. M. Guldi, Chem. Soc. Rev., 2021, 50, 3485–3518 RSC.
- P. Bharmoria, H. Bildirir and K. Moth-Poulsen, Chem. Soc. Rev., 2020, 49, 6529–6554 RSC.
- V. Sharma, R. Suthar, S. Karak and A. Sinha, ACS Appl. Opt. Mater., 2025, 3, 259–271 CrossRef CAS.
- W. G. J. H. M. van Sark, K. W. J. Barnham, L. H. Slooff, A. J. Chatten, A. Büchtemann, A. Meyer, S. J. McCormack, R. Koole, D. J. Farrell, R. Bose, E. E. Bende, A. R. Burgers, T. Budel, J. Quilitz, M. Kennedy, T. Meyer, C. D. M. Donegá, A. Meijerink and D. Vanmaekelbergh, Opt. Express, 2008, 16, 21773–21792 Search PubMed.
- L. Naimovičius, P. Bharmoria and K. Moth-Poulsen, Mater. Chem. Front., 2023, 7, 2297–2315 RSC.
- B. Daiber, K. van den Hoven, M. H. Futscher and B. Ehrler, ACS Energy Lett., 2021, 6, 2800–2808 CrossRef CAS PubMed.
- D. Nguyen, T. Van Huynh, V. S. Nguyen, P.-L. D. Cao, H. T. Nguyen, T.-C. Wei, P. H. Tran and P. T. Nguyen, RSC Adv., 2021, 11, 21560–21566 RSC.
- D. Nguyen, M. T. Nguyen, T. T. D. Nguyen, V. T. Huynh, B. P. N. Nguyen and P. T. Nguyen, Aust. Ceram. Soc., 2022, 58, 913–921 CrossRef CAS.
- M. Gorlova and L. Kloo, Dalton Trans., 2008, 2655–2666 Search PubMed.
- M. Hassan, T. A. Shifa, A. Vomiero, E. Moretti and K. B. Ibrahim, Small, 2025, 21, e09030 Search PubMed.
- S. Fernandes, J. C. G. E. da Silva and L. P. da Silva, Materials, 2022, 15, 3446 Search PubMed.
- C. Yang, H. A. Atwater, M. A. Baldo, D. Baran, C. J. Barile, M. C. Barr, M. Bates, M. G. Bawendi, M. R. Bergren, B. Borhan, C. J. Brabec, S. Brovelli, V. Bulovic, P. Ceroni, M. G. Debije, J.-M. Delgado-Sanchez, W.-J. Dong, P. M. Duxbury, R. C. Evans, S. R. Forrest, D. R. Gamelin, N. C. Giebink, X. Gong, G. Griffini, F. Guo, C. K. Herrera, A. W. Y. Ho-Baillie, R. J. Holmes, S.-K. Hong and T. Kirchartz, et al. , Joule, 2022, 6, 8 Search PubMed.
- M. Mokhtarpour, A. Rostami, H. Shekaari, A. Zarghami and S. Faraji, Sci. Rep., 2023, 13, 18936 CrossRef CAS PubMed.
- S. L. Piper, M. Kar, D. R. MacFarlane, K. Matuszek and J. M. Pringle, Green Chem., 2022, 24, 102–117 RSC.
- M. Królikowski, M. Więckowski, K. Żółtańska and M. Królikowska, J. Chem. Eng. Data, 2024, 69, 958–972 Search PubMed.
- Z. Wang, A. Roffey, R. Losantos, A. Lennartson, M. Jevric, A. U. Petersen, M. Quant, A. Dreos, X. Wen, D. Sampedro, K. Börjesson and K. Moth-Poulsen, Energy Environ. Sci., 2019, 12, 187–193 RSC.
- Z. Wang, H. Hölzel and K. Moth-Poulsen, Chem. Soc. Rev., 2022, 51, 7313–7326 RSC.
- X. Xu and G. Wang, Small, 2022, 18, e2107473 CrossRef PubMed.
- X. Xu, C. Li, W. Li, J. Feng and W.-Y. Li, Energy Environ. Sci., 2025, 18, 8990–9017 Search PubMed.
- N. Baggi, H. Hölzel, H. Schomaker, K. Moreno and K. Moth-Poulsen, ChemSusChem, 2023, 17, ge202301184 Search PubMed.
- N. Baggi, L. M. Muhammad, Z. Liasi, J. L. Elholm, P. Baronas, E. Molins, K. V. Mikkelsen and K. Moth-Poulsen, J. Mater. Chem. A, 2024, 12, 26457–26464 Search PubMed.
- L. Fernandez, H. Hölzel, P. Ferreira, N. Baggi, K. Moreno, Z. Wang and K. Moth-Poulsen, Surfactant-enabled strategy for molecular solar thermal energy storage systems in water, Green Chem., 2025, 27, 14119–14130 Search PubMed.
| This journal is © the Owner Societies 2026 |



