Open Access Article
Open Access ArticleCationic iridium-catalyzed enantioselective decarboxylative aryl addition of aromatic carboxylic acids to bicyclic alkenes
Ren
Asano†
a,
Reina
Nonami†
b,
Hina
Hamasaki
b,
Kazuya
Kanemoto
 c,
Harunori
Fujita
b,
Kaisei
Yamamoto
d,
Tomohiro
Seki
c,
Harunori
Fujita
b,
Kaisei
Yamamoto
d,
Tomohiro
Seki
 d,
Kazutaka
Shibatomi
d,
Kazutaka
Shibatomi
 e,
Shunsuke
Kuwahara
e,
Shunsuke
Kuwahara
 af and
Tomohiko
Shirai
af and
Tomohiko
Shirai
 *af
*af
aDepartment of Chemistry, Faculty of Science, Toho University, 2-2-1 Miyama, Funabashi, Chiba, Japan. E-mail: tomohiko.shirai@sci.toho-u.ac.jp
bDepartment of Social Design Engineering, National Institute of Technology, Kochi College, 200-1 Monobe Otsu, Nankoku, Kochi, Japan
cInstitute of Integrated Research, Institute of Science Tokyo, 2-3-10 Kanda-Surugadai, Chiyoda-ku, Tokyo, Japan
dDepartment of Chemistry, Faculty of Science, Shizuoka University, Shizuoka City, Shizuoka, Japan
eDepartment of Applied Chemistry and Life Science, Toyohashi University of Technology, 1-1 Hibarigaoka, Tempaku-cho, Toyohashi, Aichi, Japan
fResearch Center for Materials with Integrated Properties, Toho University, 2-2-1 Miyama, Funabashi, Chiba, Japan
First published on 14th February 2026
Abstract
Decarboxylative functionalization is an environmentally benign approach that enables the formation of new chemical bonds while emitting only CO2 as a byproduct. Herein, we report an asymmetric aryl addition to bicyclic alkenes involving the decarboxylation of aromatic carboxylic acids. The cationic iridium/S-Me-BIPAM catalyst system is effective in this reaction, enabling the enantioselective construction of chiral aryl-substituted bicyclo[2.2.1]heptane cores (up to 85% yield and up to 99% ee).
The chiral bicyclo[2.2.1] framework is an attractive structural motif and a prevalent substructure found in bioactive molecules such as SPK-601,1 AMG 221,2 setrobuvir, and sordarins (Fig. 1).3
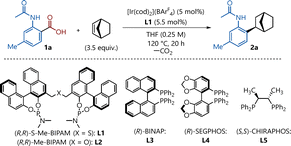
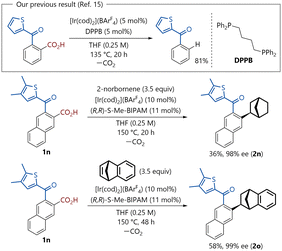
In addition, a chiral phosphine compound containing a bicyclo[2.2.1] moiety has also been employed as a chiral ligand in asymmetric synthesis.4 Consequently, substantial effort has been devoted to the development of various synthetic methodologies for the construction of these frameworks.5 In particular, in view of the pharmacological relevance of chiral aryl-substituted bicyclo[2.2.1]heptane derivatives,6 considerable attention has been directed toward the development of asymmetric arylation that provides direct access to these scaffolds via metal catalysis. Methods employing reactive reagents such as organometallic compounds7 or (pseudo)halides8 have been reported. Approaches based on the direct transformation of moieties that are ubiquitously present in organic molecules also provide an alternative entry to asymmetric arylation.9,10 Alongside catalytic asymmetric directed C(sp2)–H addition established by Hartwig11 and our group12 (Scheme 1a), the enantioselective ipso-transformation of ubiquitous carbonyl moieties represents an attractive strategy. In 2022, we reported that chiral cationic iridium catalysts could generate aryl–metal species via C–C bond activation through decarbonylation of aldehydes (Scheme 1b).13,14 These species enabled enantioselective decarbonylative aryl addition to bicyclic alkenes, constructing optically active bicyclo[2.2.1] skeletons. Our process represents the first highly enantioselective decarbonylative aryl addition to C–C double bonds; however, it has the potential drawback of catalyst deactivation through coordination of the generated CO. Thus, this reaction typically requires a high catalyst loading (at least 10 mol%), high temperatures, and long reaction times. To address these issues, we turned our attention to the decarboxylation of carboxylic acids and found that a cationic iridium complex with a chiral diphosphine ligand exhibited excellent catalytic activity for the decarboxylation of aromatic carboxylic acids.15 Aromatic carboxylic acids are abundant in nature and readily available as chemical feedstocks, and therefore have been widely utilized in conventional organic transformations as well as catalytic decarboxylative functionalization. Among the various methods, decarboxylative aryl addition reactions of aromatic carboxylic acids represent an environmentally benign and attractive approach that does not require organometallic reagents and does not generate strongly coordinating gases. However, asymmetric variants of decarboxylative aryl addition have not been reported to date.16–19 Herein, we report a cationic iridium-catalyzed asymmetric aryl addition to bicyclic alkenes via C–C bond activation through decarboxylation (Scheme 1c).
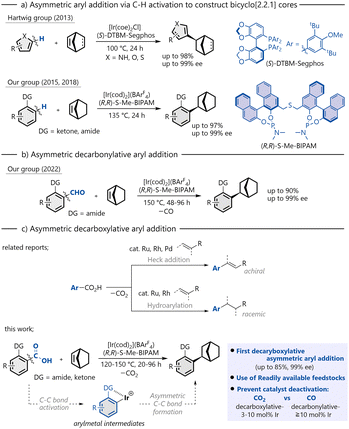 | ||
| Scheme 1 (a) Asymmetric aryl addition via C–H activation to construct bicyclo[2.2.1] cores. (b) Asymmetric decarbonylative aryl addition. (c) Asymmetric decarboxylative aryl addition. | ||
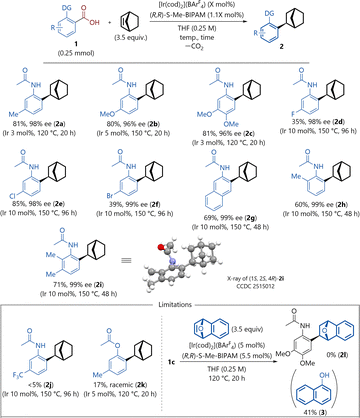
Our investigation started using carboxylic acid 1a, as the model substrate, with 2-norbornene over [Ir(cod)2](BArF4) (5 mol%, cod = 1,5-cyclooctadiene, BArF4 = tetrakis[3,5-bis(trifluoromethyl)phenyl]borate) and (R,R)-S-Me-BIPAM (5.5 mol%) in tetrahydrofuran (THF) at 120 °C in a sealed tube for 20 h under an N2 atmosphere, to give the desired adduct in 82% yield with 99% ee (Table 1, entry 1). The counterion strongly affects the decarboxylation process, presumably because enhanced Lewis acidity plays a crucial role in facilitating decarboxylation. Using [Ir(cod)2](OTf) or [Ir(cod)Cl]2 as precursors did not give the desired product (entries 2 and 3). When [Ir(cod)2](OTf) was employed, only the decarboxylation product was obtained in 13% yield, whereas [Ir(cod)Cl]2 did not promote the decarboxylation. Reactions with (R,R)-Me-BIPAM, (R)-BINAP, (R)-SEGPHOS, and (S,S)-CHIRAPHOS afforded the products in low yields and enantioselectivities (entries 4–7). The reaction was performed with 3 mol% catalyst loading, and the desired product 2a was obtained in 81% yield and 98% ee (entry 8). The iridium source and phosphine ligand are essential for reactivity (entries 9 and 10). Next, the scope of the reaction was explored by examining the functional groups on the aromatic ring of the carboxylic acids under the optimized conditions (Scheme 2). A substituent on aromatic carboxylic acid 1 significantly influenced the reactivity. Electron-donating substituents at the 4- or 5-position of the substrates were well tolerated (2a–2c). In contrast, when aromatic carboxylic acids bearing electron-withdrawing groups such as fluoro (1d) or bromo groups (1f) were used, the yields decreased, with the exception of the chloro group (1e), while enantioselectivities of over 90% ee were maintained. 3-Acetamido-2-naphthoic acid (1g) reacted by using 10 mol% of catalyst at 150 °C for 48 h, affording the product 2g in 69% yield with 99% ee. The presence of a methyl group at the 3-position of the aryl backbone (1h, 1i) had a negative effect on the reactivity, giving products 2h (60%, 99% ee) and 2i (71%, 99% ee), respectively, by using 10 mol% of catalyst at 150 °C for 48 h.
| Entry | Deviation from above | Yieldb (%) | eec |
|---|---|---|---|
| a Reactions were run on a 0.25 mmol scale. b Isolated yields. c Enantiomeric excess (ee) was determined by HPLC analysis with a chiral stationary phase. n.d. denotes not determined. | |||
| 1 | None | 82 | 99% |
| 2 | [Ir(cod)2](OTf) | 0 | — |
| 3 | [Ir(cod)Cl]2 (2.5 mol%) | 0 | — |
| 4 | (R,R)-Me-BIPAM (L2) | 20 | 92% |
| 5 | (R)-BINAP (L3) | 10 | n.d. |
| 6 | (R)-SEGPHOS (L4) | 57 | 28% |
| 7 | (S,S)-CHIRAPHOS (L5) | 72 | 0% |
| 8 | Ir/ligand (3 mol%) | 81 | 98% |
| 9 | No [Ir(cod)2](BArF4) | 0 | — |
| 10 | No ligand | Trace | n.d. |
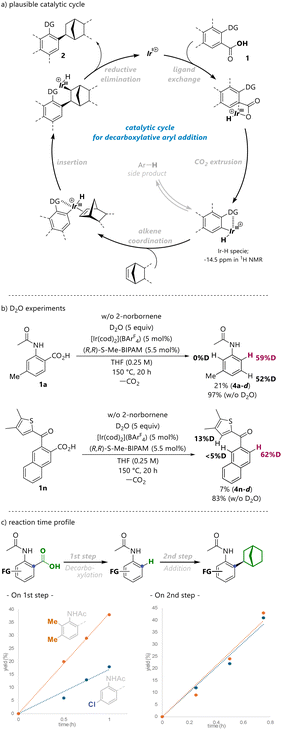
In addition, the absolute configuration of (1S, 2S, 4R)-2i was confirmed by X-ray crystallographic analysis. Substrate 1j, which bears a CF3 group at the 4-position, was unsuccessful. The reaction in the presence of an ester group at the 2-position gave the racemic product 2k in a diminished yield. The reaction of substrate 1c with oxa-benzonorbornadiene gave no desired addition product; instead, 1-naphthol 3 was obtained in 41% yield via the catalytic isomerization of bicycloalkene.20 Having identified the optimal reaction conditions, we investigated the reactions of aromatic carboxylic acids bearing ketone moieties (Scheme 3). Our previous studies on the decarboxylation of aromatic carboxylic acids showed that the 2-thienylcarbonyl group is a suitable directing group for the reaction, affording the desired arene product in high yield (81%).15 The first attempt was carried out using 3-(5-methylthiophene-2-carbonyl)-2-naphthoic acid (1m) and 2-norbornene, in the presence of 5 mol% Ir precursor and 5.5 mol% BIPAM ligand in THF at 150 °C. However, the desired adduct (2m) was obtained in only 9% yield after 20 h with the formation of undesired regioisomeric side products (see the SI for details). To suppress the formation of side products via C–H activation, a methyl group was introduced at the 4-position of the thiophene ring. Substrate bearing 4,5-dimethyl groups on the thiophene ring (1n) afforded the desired product (2n) in moderate yield with high enantioselectivity (36% yield, 98% ee). Next, the effects of bicyclic alkenes were explored. In the case of benzonorbornadiene, the reaction resulted in the formation of the desired product (2o) in improved yield with excellent enantioselectivity (58% yield, 99% ee). Based on the experimental observations and our previous studies, a plausible catalytic cycle for the reaction is proposed, as shown in Fig. 2a. Initially, the decarboxylation generates aryl–iridium hydride intermediates (δH −14.5, see the SI for details). Subsequently, the alkene inserts into the aryl–iridium bond to form an alkyl–iridium species. Finally, the cationic IrI active species are generated from the alkyl iridium complex via reductive elimination.
To gain further insight into the reaction mechanism, we performed D2O experiments to determine the site of activation by the catalyst (Fig. 2b). This catalytic system for decarboxylation was not effective in the presence of H2O or D2O, but predominant incorporation of a deuterium label at the ipso position was observed in the products (4a-d and 4n-d) (see the SI for details). In the amide product (4a-d), deuteration at the adjacent position was also observed.
In addition, the reaction time profiles were investigated (Fig. 2c). This reaction proceeds via decarboxylation followed by addition. Therefore, we compared the initial rates for each step using substrates with electron-donating and electron-withdrawing groups. As a result, the decarboxylation was accelerated in the presence of electron-donating groups, whereas no significant rate difference was observed for the addition step. This suggests that the CO2 extrusion process accompanied by carbon–carbon bond activation is facilitated by electron-donating groups and is likely involved in the rate-determining step. Next, 31P NMR was recorded before and after decarboxylation. For the active complex prepared from [Ir(cod)2](BArF4) and S-Me-BIPAM, a major peak was observed at 128.7 ppm (Fig. 3a), and no peak shift was observed even after decarboxylation (Fig. 3b). The peak position remained unchanged even in the presence of acetanilide, where reversible C–H activation proceeds (Fig. 3c). These results indicate that CO2 generated by the decarboxylation does not affect the catalyst structure, and hence its reactivity.
In conclusion, we have developed the first asymmetric decarboxylative aryl addition of aromatic carboxylic acids directed by anilide or ketone moieties, with excellent enantioselectivity (up to 99% ee). Although the reaction is limited to the use of carbon bridged bicyclic alkenes, our findings provide a new synthetic strategy for constructing chiral carbon–carbon bonds using metal catalysis. Furthermore, ongoing efforts are focused on elucidating the origins of enantioselectivity and extending this catalytic protocol to challenging molecular transformations.
All authors contributed to the writing of this manuscript and have approved the final submitted version.
Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, NMR spectra of the new compounds and X-ray crystallographic data. See DOI: https://doi.org/10.1039/d6cc00389c.CCDC 2515012 contains the supplementary crystallographic data for this paper.21
Acknowledgements
This work was financially supported by the Iwatani Naoji Foundation.References
- R. M. Adibhatla, J. F. Hatcher and A. Gusain, Neurochem. Res., 2012, 37, 671 CrossRef CAS PubMed.
- A. Li, C. C. Yuan, D. Chow, M. Chen, M. G. Emery, C. Hale, X. Zhang, R. Subramanian, D. J. Jean, R. Komorowski, M. Véniant, M. Wang and C. Fotsch, ACS Med. Chem. Lett., 2011, 2, 824 CrossRef CAS PubMed.
- H. Liang, Beilstein J. Org. Chem., 2008, 4, 31 Search PubMed.
- K. Vandyck, B. Matthys, M. Willen, K. Robeyns, L. Van Meervelt and J. Van der Eycken, Org. Lett., 2006, 8, 363 CrossRef CAS PubMed.
- (a) H. Sundén, R. Rios, Y. Xu, L. Eriksson and A. Córdova, Adv. Synth. Catal., 2007, 349, 2549 CrossRef; (b) X. Feng, Z. Zhou, R. Zhou, Q.-Q. Zhou, L. Dong and Y.-C. Chen, J. Am. Chem. Soc., 2012, 134, 19942 CrossRef CAS PubMed; (c) D. Liang, Y. Zou, Q. Wang and A. Goeke, J. Org. Chem., 2014, 79, 6726 Search PubMed; (d) R. Mose, M. E. Jensen, G. Preegel and K. A. Jørgensen, Angew. Chem., Int. Ed., 2015, 54, 13630 CrossRef CAS PubMed; (e) J.-G. Fu, Y.-F. Shan, W.-B. Sun, G.-Q. Lin and B.-F. Sun, Org. Biomol. Chem., 2016, 14, 5229 RSC.
- T. F. Spande, H. M. Garraffo, M. W. Edwards, H. J. C. Yeh, L. Pannell and J. W. Daly, J. Am. Chem. Soc., 1992, 114, 3475 CrossRef CAS.
- (a) F. Menard and M. Lautens, Angew. Chem., Int. Ed., 2008, 47, 2085 CrossRef CAS PubMed; (b) J. Panteleev, F. Menard and M. Lautens, Adv. Synth. Catal., 2008, 350, 2893 CrossRef CAS; (c) J. Bexrud and M. Lautens, Org. Lett., 2010, 12, 3160 Search PubMed.
- (a) S. Sakuraba, K. Awano and K. Achiwa, Synlett, 1994, 291 CrossRef CAS; (b) S. Sakuraba, T. Okada, T. Morimoto and K. Achiwa, Chem. Pharm. Bull., 1995, 43, 927 CrossRef CAS; (c) J. M. Brunel, M.-H. Hirlemann, A. Heumann and G. Buono, Chem. Commun., 2000, 1869 RSC; (d) X.-Y. Wu, H.-D. Xu, F.-Y. Tang and Q.-L. Zhou, Tetrahedron: Asymmetry, 2001, 12, 2565 CrossRef CAS; (e) D. Drago and P. S. Pregosin, Organometallics, 2002, 21, 1208 CrossRef CAS; (f) S. Liu and J. Zhou, Chem. Commun., 2013, 49, 11758 Search PubMed; (g) F. Zeng, C. Chen, L. Liu, Y. Li, B. Li, G. Lin, S. Huang, M. Zhang, Y. Yan, Y. Zhang, Y. Dang and J. Huang, ChemRxiv, 2021, preprint DOI:10.26434/chemrxiv-2021-tlgj2-v3.
- (a) R. Aufdenblatten, S. Diezi and A. Togni, Monatsh. Chem., 2000, 131, 1345 Search PubMed; (b) R. Dorta and A. Togni, Chem. Commun., 2003, 760 RSC.
- For representative examples of asymmetric intermolecular addition of arene C(sp2)–H bonds to alkenes using directing group strategies: (a) M. Hatano, Y. Ebe, T. Nishimura and H. Yorimitsu, J. Am. Chem. Soc., 2016, 138, 4010 CrossRef CAS PubMed; (b) D. Yamauchi, T. Nishimura and H. Yorimitsu, Chem. Commun., 2017, 53, 2760 Search PubMed; (c) Y. Ebe, M. Onoda, T. Nishimura and H. Yorimitsu, Angew. Chem., Int. Ed., 2017, 56, 5607 CrossRef CAS PubMed; (d) T. Shibata, M. Michino, H. Kurita, Y.-K. Tahara and K. S. Kanyiva, Chem. – Eur. J., 2017, 23, 88 Search PubMed; (e) S. Grélaud, P. Cooper, L. J. Feron and J. F. Bower, J. Am. Chem. Soc., 2018, 140, 9351 CrossRef PubMed; (f) K. Sakamoto and T. Nishimura, Adv. Synth. Catal., 2019, 361, 2124 CrossRef CAS; (g) K. Sakamoto and T. Nishimura, Org. Biomol. Chem., 2021, 19, 684 Search PubMed; (h) K. Murakami, K. Sakamoto and T. Nishimura, Synthesis, 2022, 4753 CAS; (i) K. Yamakawa, I. Nakamura, K. Sakamoto and T. Nishimura, J. Org. Chem., 2023, 88, 7858 Search PubMed; (j) T. P. Aldhous, R. Chung, A. Hassan, A. G. Dailing, P. Cooper, S. Grélaud, R. J. Mudd, L. J. Feron, P. D. Kemmitt and J. F. Bower, Angew. Chem., Int. Ed., 2025, 64, e202502569 Search PubMed.
- C. S. Sevov and J. F. Hartwig, J. Am. Chem. Soc., 2013, 135, 2116 CrossRef CAS PubMed.
- (a) T. Shirai and Y. Yamamoto, Angew. Chem., Int. Ed., 2015, 54, 9894 CrossRef CAS PubMed; (b) T. Shirai, T. Okamoto and Y. Yamamoto, Asian J. Org. Chem., 2018, 7, 1054 CrossRef CAS.
- T. Shirai, K. Sugimoto, M. Iwasaki, R. Sumida, H. Fujita and Y. Yamamoto, Synlett, 2019, 972 CAS.
- R. Nonami, Y. Morimoto, K. Kanemoto, Y. Yamamoto and T. Shirai, Chem. – Eur. J., 2022, 28, e202104347 CrossRef CAS PubMed.
- R. Nonami, Y. Kishino, T. Yamasaki, K. Kanemoto, K. Iwai, N. Nishiwaki, K. Shibatomi and T. Shirai, Chem. – Asian J., 2023, 18, e202300533 CrossRef CAS PubMed.
- For hydroarylation of alkynes using decarboxylation/C–H activation strategies; (a) J. Zhang, R. Shrestha, J. F. Hartwig and P. Zhao, Nat. Chem., 2016, 8, 1144 CrossRef CAS PubMed; (b) A. Biafora, B. A. Khan, J. Bahri, J. M. Hewer and L. J. Goossen, Org. Lett., 2017, 19, 1232 CrossRef CAS PubMed; (c) H. Chen, L. Ouyang, J. Liu, W.-J. Shi, G. Chen and L. Zheng, J. Org. Chem., 2019, 84, 12755 CrossRef CAS PubMed.
- For hydroarylation of alkenes using decarboxylation/C–H activation strategies: (a) A. Mandal, H. Sahoo, S. Dana and M. Baidya, Org. Lett., 2017, 19, 4138 CrossRef CAS PubMed; (b) K. R. Bettadapur, V. Lanke and K. R. Prabhu, Chem. Commun., 2017, 53, 6251 RSC; (c) X. Li, X. Wang and J. Zhang, Chem. Sci., 2023, 14, 5470 RSC.
- For Heck type addition to alkenes using decarboxylation/C–H activation strategies: (a) A. Maehara, H. Tsurugi, T. Satoh and M. Miura, Org. Lett., 2008, 10, 1159 CrossRef CAS PubMed; (b) S. Mochida, K. Hirano, T. Satoh and M. Miura, Org. Lett., 2010, 12, 5776 CrossRef CAS PubMed; (c) S. Mochida, K. Hirano, T. Satoh and M. Miura, J. Org. Chem., 2011, 76, 3024 CrossRef CAS PubMed; (d) N. Y. P. Kumar, A. Bechtoldt, K. Raghuvanshi and L. Ackermann, Angew. Chem., Int. Ed., 2016, 55, 6929 CrossRef CAS; (e) X.-Y. Jin, L.-J. Xie, H.-P. Cheng, A.-D. Liu, X.-D. Li, D. Wang, L. Cheng and L. Liu, J. Org. Chem., 2018, 83, 7514 CrossRef CAS PubMed; (f) M. S. Sherikar and K. R. Prabhu, Org. Lett., 2019, 21, 4525 CrossRef CAS PubMed; (g) S. Yugandar, T. Morita and H. Nakamura, Org. Biomol. Chem., 2020, 18, 8625 RSC; (h) J. Zhang, W. Xu, W. Zhuang, X. Chen, X. Zhang and Q. Huang, J. Org. Chem., 2023, 88, 15198 CrossRef CAS PubMed.
- For decarboxylative or decarbonylative aryl addition to alkenes using aromatic carboxylic acids: (a) A. G. Myers, D. Tanaka and M. R. Mannion, J. Am. Chem. Soc., 2002, 124, 11250 CrossRef CAS PubMed; (b) L. J. Gooβen, J. Paetzold and L. Winkel, Synlett, 2002, 1721 Search PubMed; (c) D. Tanaka and A. G. Myers, Org. Lett., 2004, 6, 433 CrossRef CAS PubMed; (d) D. Tanaka, S. P. Romeril and A. G. Myers, J. Am. Chem. Soc., 2005, 127, 10323 CrossRef CAS; (e) P. Hu, J. Kan, W. Su and M. Hong, Org. Lett., 2009, 11, 2341 CrossRef CAS PubMed; (f) Z.-M. Sun and P. Zhao, Angew. Chem., Int. Ed., 2009, 48, 6726 CrossRef CAS PubMed; (g) Z.-M. Sun, J. Zhang and P. Zhao, Org. Lett., 2010, 12, 992 CrossRef CAS PubMed; (h) S.-L. Zhang, Y. Fu, R. Shang, Q.-X. Guo and L. Liu, J. Am. Chem. Soc., 2010, 132, 638 CrossRef CAS PubMed; (i) L. J. Gooβen, B. Zimmermann and T. Knauber, Beilstein J. Org. Chem., 2010, 6, 43 Search PubMed; (j) Z. Fu, S. Huang, W. Su and M. Hong, Org. Lett., 2010, 12, 4992 CrossRef CAS PubMed; (k) F.-Q. Shi, Org. Lett., 2011, 13, 736 CrossRef CAS PubMed; (l) L. Huang, J. Qi, X. Wu, K. Huang and H. Jiang, Org. Lett., 2013, 15, 2330 CrossRef CAS PubMed; (m) A. Fardost, J. Lindh, P. J. R. Sjoberg and M. Larhed, Adv. Synth. Catal., 2014, 356, 870 Search PubMed; (n) S.-R. Ban, H.-N. Wang, V. Toader, D. S. Bohle and C.-J. Li, Org. Lett., 2014, 16, 6282 Search PubMed; (o) D. Yang, Y. Liu, P. Sun, N. Zhang, W. Wei, M. Sun, G. Chen, S. Bi and H. Wang, Org. Biomol. Chem., 2016, 14, 895 Search PubMed; (p) A. Hossian, S. K. Bhunia and R. Jana, J. Org. Chem., 2016, 81, 2521 Search PubMed; (q) X. Qin, C. Chen, L. Zhang, J. Xu, Y. Pan, H. Zhao, J. Han, H. Li and L. Xu, Tetrahedron, 2017, 73, 2242 CrossRef CAS; (r) J. Shen, B. Xu, M. Zhang and W. Su, Eur. J. Org. Chem., 2018, 2768 CrossRef CAS; (s) O. Bouazzaoui, K. Rousée, J. K. Mulengi, X. Pannecoucke, J.-P. Bouillon and S. Couve-Bonnaire, Eur. J. Org. Chem., 2018, 3705 CrossRef CAS; (t) W. Yu, L. Liu, T. Huang, X. Zhou and T. Chen, Org. Lett., 2020, 22, 7123 CrossRef CAS PubMed; (u) S. Jenthra, T. Mondal, G. Kemper, M. Lantzius-Beninga, M. Hölscher and W. Leitner, ACS Catal., 2023, 13, 10085 CrossRef CAS.
- For metal-catalyzed isomerization of oxabenzonorbornadienes to afford 1-naphthol: K. Villeneuve and W. Tam, J. Am. Chem. Soc., 2006, 128, 3514 CrossRef CAS PubMed.
- CCDC 2515012: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qf2fp.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |