Open Access Article
Open Access ArticleVisible-light-induced upcycling of perchloroethylene into trichloroacetamides using chlorine dioxide
Shohei
Ohno
 a,
Genta
Taniguchi
a,
Minami
Fukuhara
a,
Yuki
Itabashi
a,
Genta
Taniguchi
a,
Minami
Fukuhara
a,
Yuki
Itabashi
 b,
Tsuyoshi
Inoue
ab,
Kei
Ohkubo
b,
Tsuyoshi
Inoue
ab,
Kei
Ohkubo
 bc and
Haruyasu
Asahara
bc and
Haruyasu
Asahara
 *ab
*ab
aGraduate School of Pharmaceutical Sciences, The University of Osaka, 1-6 Yamadaoka, Suita, Osaka 565-0871, Japan. E-mail: asahara@phs.osaka-u.ac.jp
bInstitute for Open and Transdisciplinary Research Initiatives (OTRI), The University of Osaka, 1-6 Yamadaoka, Suita, Osaka 565-0871, Japan
cOrganization for Carbon Neutrality Collaboration (OCNC), The University of Osaka, 1-6 Yamadaoka, Suita, Osaka 565-0871, Japan
First published on 13th February 2026
Abstract
Herein, we report the visible-light-induced upcycling of environmentally problematic perchloroethylene into valuable trichloroacetamides using chlorine dioxide (ClO2˙). The reaction proceeds with stoichiometric amounts of reagents; under mild conditions, this enables broad applicability to the synthesis of aromatic trichloroacetamides, carbamates, urea, and heterocycles, offering a sustainable strategy for transforming an environmental pollutant into useful building blocks.
Perchloroethylene (PCE) is a chlorinated solvent widely used in industrial processes such as dry cleaning and metal degreasing. Owing to its high stability and resistance to degradation, PCE persists in the environment, raising concerns about its potential mutagenicity and carcinogenicity.1–3 In response, its production and use have been progressively restricted by environmental regulations.4 Nevertheless, PCE is still generated in various industrial processes.5 Accordingly, chemical upcycling technologies that convert PCE into valuable organic molecules represent a sustainable approach for reducing its environmental impact.6–8
Furthermore, PCE decomposes upon photo-oxidation to yield a complex mixture, including chlorine gas, carbon oxides (CO and CO2), phosgene, epoxides, and trichloroacetyl chloride.9–15 Recently, photochemical oxidation of PCE was reported, wherein irradiation with deep ultraviolet light (UV-C, λ < 280 nm) under an oxygen atmosphere produced trichloroacetyl chloride, which upon addition of alcohols or amines afforded trichlorocarbonates or trichloroacetamides.16,17 In 2024, a visible-light protocol using a small amount of chlorine gas as a radical initiator was reported.18 These methods have attracted considerable attention as chemical upcycling strategies. However, the method using deep ultraviolet light generates large amounts of highly toxic byproducts, such as phosgene, and requires a large amount of solvent for the substrate. The visible-light protocol overcomes some of these drawbacks but still requires chlorine gas, which presents safety and handling challenges. By contrast, chlorine dioxide (ClO2˙) can generate both chlorine radicals and singlet oxygen under visible light irradiation; hence, we have been working on developing oxidation reactions utilizing this property.19–22 ClO2˙ can be used as an oxidant under mild conditions, enabling improved reaction control and reducing safety risks.
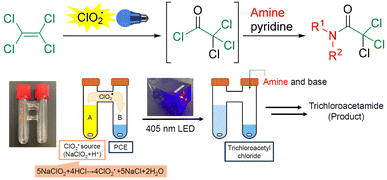
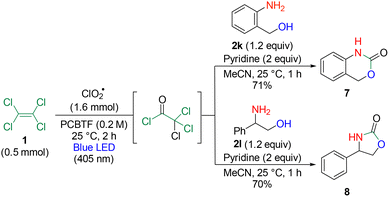
Herein, we report the visible light-induced photooxidation of PCE using ClO2˙ and establish a one-pot method for the synthesis of valuable trichloroacetamide23–27 derivatives (Scheme 1). The use of p-chlorobenzotrifluoride (PCBTF), which is inert to chlorine radicals and exhibits high solubility, as a solvent enables the reaction to be performed using stoichiometric amounts of PCE. The reaction proceeds under visible light at room temperature (25 °C), which not only minimizes the formation of highly toxic by-products such as phosgene but also expands the reaction scope to include a variety of aromatic trichloroacetamides.
An H-shaped reaction glass tube (COware) was employed as a two-chamber system.28–30 One side of the system (chamber A) contained an aqueous ClO2˙ solution prepared by mixing sodium chlorite (NaClO2) with HCl, and the other side (chamber B) was filled with a solution containing PCE. Upon irradiation of the entire vessel with a blue LED, ClO2˙ in chamber A was activated, resulting in gas formation. The gases passed through the glass-tube bridge connecting the two chambers and dissolved in the PCE-containing solution in chamber B. Subsequent treatment of chamber B with an amine and base afforded trichloroacetamide as the major product, which is regarded as a valuable intermediate for the synthesis of pharmaceuticals and agrochemicals.23–27
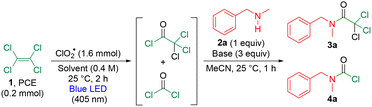
We conducted optimization studies of the visible-light-induced upcycling of PCE 1 to trichloroacetamide 3a (Table 1). PCE 1 was treated with ClO2˙ (8.0 equiv. relative to PCE),31 amine (1.0 equiv.), and a base (3.0 equiv.) under irradiation with a 405-nm blue LED. Under solvent-free conditions, using pyridine as a base, 3a and the phosgene-derived by-product 4a were obtained in a combined yield of 19% at an 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 1).
1 ratio (entry 1).
| Entry | Solvent | Base | Yielda (%) | ||
|---|---|---|---|---|---|
| 3a | 4a | ||||
| a NMR yield based on PCE. b Yield was calculated based on two molecules of phosgene per 1. c PCE (0.5 mmol), amine (1 equiv.), pyridine (2 equiv.). d PCE (0.5 mmol), amine (1.2 equiv.), pyridine (2 equiv.). e PCE (0.5 mmol), amine (1.2 equiv.), pyridine (2 equiv.), PCBTF (0.2 M). | |||||
| 1 | Neat | Pyridine | 18 | 1 |
|
| 2 | MeCN | Pyridine | 22 | 2 | |
| 3 | Toluene | Pyridine | 25 | 2 | |
| 4 | BTF | Pyridine | 63 | 4 | |
| 5 | PBTFB | Pyridine | 65 | 4 |
|
| 6 | PCBTF | Pyridine | 68 | 4 | |
| 7 | PCBTF | NEt3 | 36 | 2 | |
| 8 | PCBTF | DIPEA | 54 | 9 | |
| 9 | PCBTF | Cs2CO3 | 40 | 3 | |
| 10 | PCBTF | 2,6-Lutidine | 51 | 6 |
|
| 11c | PCBTF | Pyridine | 55 | 3 | |
| 12d | PCBTF | Pyridine | 76 | 4 | |
| 13e | PCBTF | Pyridine | 80 | 4 | |
Using acetonitrile as the solvent afforded a combined yield of 24% in a 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 2), and toluene afforded a combined yield of 27% in a 12.5
1 ratio (entry 2), and toluene afforded a combined yield of 27% in a 12.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 3). However, the reactions between the solvent and ClO2˙ also yielded side products. To suppress such the side reactions, benzotrifluoride (BTF)32–34 was employed as a solvent that is less reactive toward chlorine radicals; this improved the combined yield to 67% with a 15.8
1 ratio (entry 3). However, the reactions between the solvent and ClO2˙ also yielded side products. To suppress such the side reactions, benzotrifluoride (BTF)32–34 was employed as a solvent that is less reactive toward chlorine radicals; this improved the combined yield to 67% with a 15.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 4). Further improvement was achieved using p-bis(trifluoromethyl)benzene (PBTFB), affording a 69% combined yield in 16.3
1 ratio (entry 4). Further improvement was achieved using p-bis(trifluoromethyl)benzene (PBTFB), affording a 69% combined yield in 16.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 5). The best result was achieved with p-chlorobenzotrifluoride (PCBTF), which afforded a combined yield of 72% in a 17
1 ratio (entry 5). The best result was achieved with p-chlorobenzotrifluoride (PCBTF), which afforded a combined yield of 72% in a 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 6). A screening of green solvents and other electron-deficient aromatic solvents was conducted (Table S1). However, these alternative solvents resulted in significantly lower yields than PCBTF. Subsequently, we examined the effects of the base. When the base was changed to triethylamine, 3a and 4a were obtained in a combined yield of 38% in an 18
1 ratio (entry 6). A screening of green solvents and other electron-deficient aromatic solvents was conducted (Table S1). However, these alternative solvents resulted in significantly lower yields than PCBTF. Subsequently, we examined the effects of the base. When the base was changed to triethylamine, 3a and 4a were obtained in a combined yield of 38% in an 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 7). Using diisopropylethylamine as the base gave a combined yield of 63% in a 6
1 ratio (entry 7). Using diisopropylethylamine as the base gave a combined yield of 63% in a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 8). Changing the base to Cs2CO3 afforded a combined yield of 43% in a 13.3
1 ratio (entry 8). Changing the base to Cs2CO3 afforded a combined yield of 43% in a 13.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 9). The use of 2,6-lutidine afforded a combined yield of 57% at an 8.5
1 ratio (entry 9). The use of 2,6-lutidine afforded a combined yield of 57% at an 8.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 10). Subsequently, on using PCBTF as a solvent and pyridine as a base, the reaction of PCE (0.5 mmol) with amine (1.0 equiv.) and pyridine (2.0 equiv.) provided 3a and 4a in a combined yield of 58% in an 18.3
1 ratio (entry 10). Subsequently, on using PCBTF as a solvent and pyridine as a base, the reaction of PCE (0.5 mmol) with amine (1.0 equiv.) and pyridine (2.0 equiv.) provided 3a and 4a in a combined yield of 58% in an 18.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 11). When the amine loading was increased to 1.2 equiv. under identical conditions, the combined yield improved to 80% in a 19
1 ratio (entry 11). When the amine loading was increased to 1.2 equiv. under identical conditions, the combined yield improved to 80% in a 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 12). Further improvement was achieved when twice the amount of PCBTF was used, giving a combined yield of 84% and a 20
1 ratio (entry 12). Further improvement was achieved when twice the amount of PCBTF was used, giving a combined yield of 84% and a 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (entry 13). Therefore, the conditions in entry 13 were identified as optimal.
1 ratio (entry 13). Therefore, the conditions in entry 13 were identified as optimal.
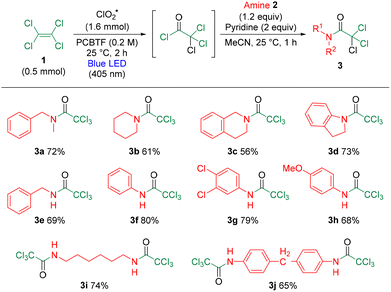
Next, the substrate scope with respect to the amines was investigated under the optimized conditions (Table 2). The reaction with methylbenzylamine afforded 3a in 72% yield. With an aliphatic heterocycle piperidine, 3b was obtained in 61% yield, whereas an aromatic heterocyclic tetrahydroisoquinoline gave 3c in 56% yield and dihydroindole provided 3d in 73% yield. In the case of primary amines, benzylamine furnished 3e in 69% yield, and aniline gave 3f in 80% yield. Additionally, the reaction with dichloroaniline afforded 3g, which is useful as an intermediate in the synthesis of pharmaceuticals and agrochemicals, in 79% yield. This reaction was also applicable to 4-methoxy aniline, affording 3h in 68% yield. Furthermore, long-chain aliphatic diamines underwent a smooth transformation to give 3i in 74% yield, and aromatic diamines afforded 3j in 65% yield. Notably, the reaction proceeded efficiently with a wide variety of aromatic amines under mild conditions, which was challenging in previous reports.
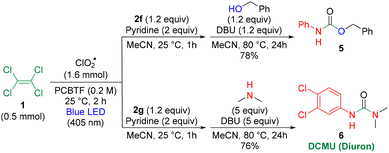
After the photooxidation of PCE 1 with ClO2˙, the addition of amine 2f and subsequent heating with benzyl alcohol in the presence of DBU afforded carbamate 5 without the isolation of trichloroacetamide (Scheme 2). Similarly, the reaction with amine 2g, followed by heating with dimethylamine and DBU, furnished herbicide DCMU 6.
Additionally, when an amino alcohol was used as the substrate, the formation of trichloroacetamide, followed by intramolecular cyclization accompanied by the elimination of the trichloro group, occurred sequentially, affording valuable heterocyclic scaffolds 7 and 8 for drug synthesis in 71% and 70% yields, respectively (Scheme 3).
Furthermore, trichloroethylene 9 was converted to dichloroacetamide 10 under the optimized conditions (Scheme 4). In this reaction, two types of intermediates were considered; however, the product derived from the other possible intermediate was not observed, which was likely due to the higher stability of the radical intermediate, leading to 10.35
We conducted the reaction using 1.1 g (6.5 mmol) of PCE 1 under the optimized conditions, affording the corresponding amide product 3a in 75% isolated yield (Scheme 5A). This result suggests that the reaction efficiency is maintained upon scale-up to the gram scale. Furthermore, when the reaction was performed with 0.5 equivalents of ClO2˙ relative to 1, 3a was obtained in 68% yield (Scheme 5B). This result indicates that the reaction proceeded via a radical-chain process.
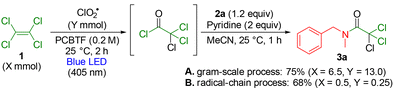 | ||
| Scheme 5 Gram-scale experiment and verification of the radical-chain mechanism in the visible-light-induced upcycling of PCE. | ||
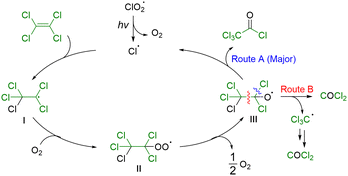
The proposed reaction mechanism is illustrated in Scheme 6. First, a chlorine radical generated by the photoirradiation of ClO2˙ adds to PCE to form radical intermediate I. Oxygen is added to this radical to afford peroxyl radical intermediate II, which is subsequently converted into alkoxyl radical intermediate III. From intermediate III, two pathways are possible: cleavage of the C–Cl bond affords the desired trichloroacetyl chloride (route A), whereas cleavage of the C–C bond yields the byproduct phosgene (route B).
A radical intermediate was detected by electron spin resonance (ESR) analysis performed at −120 °C. (Fig. S1).36 In dark, the ESR spectrum showed an anisotropic signal with four hyperfine lines owing to a chlorine nucleus (I = 3/2) corresponding to the ClO2˙. The average g value (gave) was consistent with the reported value of 2.014.37 After light irradiation and rapid freezing, a new signal was observed at gzz = 2.0337, with the disappearance of the signal owing to ClO2˙. A large gzz value was assigned to the corresponding peroxyl radical intermediate II.38
To elucidate the mechanistic origin of the observed selectivity, density functional theory (DFT) calculations were performed at the B3LYP/6-311+(2d) level, using the pentachloroethoxy radical as the reference state (0 kJ mol−1) (Fig. 1). Along route A, a transition state corresponding to chlorine radical dissociation was observed at a low activation free energy of 2.87 kJ mol−1. Structural analyses revealed substantial elongation of the cleaving C–Cl bond from 1.85 Å in the reactant to 2.07 Å at the transition state, consistent with a homolytic bond-cleavage process. The resulting product was significantly stable, lying 90.2 kJ mol−1 lower in energy than the reference state. By contrast, route B proceeded via C–C bond scission and exhibited a markedly higher activation barrier of 8.91 kJ mol−1. In the corresponding transition state, the C–C bond is elongated from 1.58 Å to 1.77 Å, accompanied by contraction of the adjacent C–O bond from 1.30 Å to 1.25 Å, indicating substantial bond reorganization and the development of carbonyl character. Although the product formed via route B is thermodynamically more stable (−163 kJ mol−1), the significantly lower activation barrier associated with route A clearly indicates that chlorine-radical dissociation is kinetically favored. This kinetic preference is fully consistent with the experimentally observed high selectivity toward the products derived from route A.
We developed a sustainable and environmentally friendly upcycling strategy that transforms PCE, a harmful pollutant, into valuable trichloroacetamides under visible-light irradiation with ClO2˙. Trichloroacetamides can be used directly in subsequent reactions without isolation, allowing for the efficient synthesis of various derivatives such as carbamate, urea, and heterocyclic compounds. When PCBTF was used as the solvent, the reaction proceeded with stoichiometric amounts of reagent. Under mild conditions, the formation of highly toxic byproducts such as phosgene was minimized. This protocol provides access to valuable trichloroacetamides, including challenging aromatic products and heterocycles. The reaction mechanism was clarified by DFT calculations and ESR measurements. These results demonstrate that our method is a practical strategy for the chemical upcycling of PCE and provides fundamental insights that can support the design of future transformations.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included in the supplementary information (SI). Supplementary information: experimental details, characterization data for new compounds, and copies of NMR spectra (PDF). See DOI: https://doi.org/10.1039/d6cc00273k.Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI grant (JP23K13709 to Y. I., JP24K21770 to K. O.), the New Energy and Industrial Technology Development Organization (NEDO) grants (17101509-0 & 24000999-0, H. A.), and the Masuyakinen basic research foundation (H. A.). In addition, we are deeply appreciative of the funding provided by the AGC Inc. We appreciate Dr Hidekazu Okamoto and Dr Hirokazu Takagi of AGC Inc. for various supports in this work.References
- P. G. Tratnyek, E. Edwards, L. Carpenter and S. Blossom, Environ. Sci.: Processes Impacts, 2020, 22, 465–471 RSC.
- M. Di Cicco, A. Tabilio Di Camillo, W. Di Marzio, M. E. Sáenz, D. M. P. Galassi, G. Pieraccini, A. Galante, D. Di Censo and T. Di Lorenzo, Environ. Toxicol. Chem., 2024, 43, 2515–2527 CrossRef CAS PubMed.
- J. H. Lee, A. K. Bryant, M. Alajlouni, B. E. Boor, A. Tasoglou and S. Liu, J. Breath Res., 2024, 18, 046008 CrossRef CAS PubMed.
- D. M. Ceballos, K. M. Fellows, A. E. Evans, P. A. Janulewicz, E. G. Lee and S. G. Whittaker, Front. Public Health, 2021, 9, 638082 Search PubMed.
- Perchloroethylene (PCE) can be generated as a side product in industrial chlorination and oxychlorination processes, for example during the production of trichloroethylene or other chlorinated hydrocarbons. Examples see: (a) S. Handa, M. Hiraoka, K. Yokoyama and T. Nakamura, Method for Producing Trichloroethylene and Tetrachloroethylene, JP Pat., JP4573147, 2010 Search PubMed; (b) I. W. Sutherland, N. G. Hamilton, C. C. Dudman, P. Jones, D. Lennon and J. M. Winfield, Appl. Catal., A, 2011, 399, 1–11 Search PubMed.
- P. A. Kots, B. C. Vance, C. M. Quinn, C. Wang and D. G. Vlachos, Nat. Sustainability, 2023, 6, 1258–1267 CrossRef.
- A. Jordan, Nat. Chem., 2024, 16, 676–677 CrossRef CAS PubMed.
- M. Liu, X. Wu and P. J. Dyson, Nat. Chem., 2024, 16, 700–708 CrossRef CAS PubMed.
- R. G. Dickinson and J. A. Leermakers, J. Am. Chem. Soc., 1932, 54, 3852–3862 CrossRef CAS.
- R. G. Dickinson and J. L. Carrico, J. Am. Chem. Soc., 1934, 56, 1473–1480 CrossRef CAS.
- D. M. Frankel, C. E. Johnson and H. M. Pitt, J. Org. Chem., 1957, 22, 1119–1120 CrossRef CAS.
- W. L. Dilling, C. J. Bredeweg and N. B. Tefertiller, Environ. Sci. Technol., 1976, 10, 351–356 CrossRef CAS.
- A. Yasuhara, Chemosphere, 1993, 26, 1507–1512 Search PubMed.
- S. Kutsuna, T. Ibusuki and K. Takeuchi, Environ. Sci. Technol., 2000, 34, 2484–2489 Search PubMed.
- P. B. Amama, K. Itoh and M. Murabayashi, J. Mol. Catal. A: Chem., 2004, 217, 109–115 CrossRef CAS.
- A. Zhang, Y. Kuwahara, Y. Hotta and A. Tsuda, Asian J. Org. Chem., 2013, 2, 572–578 CrossRef CAS.
- T. Akamatsu, M. Shele, A. Matsune, Y. Kashiki, F. Liang, T. Okazoe and A. Tsuda, ACS Omega, 2023, 8, 2669–2684 Search PubMed.
- I. Higashimura, M. Shele, T. Akamatsu, R. Ohmura, F. Liang, T. Okazoe and A. Tsuda, J. Org. Chem., 2024, 89, 1864–1872 CrossRef CAS PubMed.
- K. Ohkubo and K. Hirose, Angew. Chem., Int. Ed., 2018, 57, 2126–2129 CrossRef CAS PubMed.
- S. Ohno, H. Asahara, T. Inoue and K. Ohkubo, RSC Adv., 2022, 12, 31412–31414 RSC.
- H. Asahara, N. Takao, M. Moriguchi, T. Inoue and K. Ohkubo, Chem. Commun., 2022, 58, 6176–6179 Search PubMed.
- Y. Itabashi, H. Asahara and K. Ohkubo, Chem. Commun., 2023, 59, 7506–7517 RSC.
- A. Fantinati, V. Zanirato, P. Marchetti and C. Trapella, ChemistryOpen, 2020, 9, 100–170 CrossRef CAS PubMed.
- Q. Zhao, B. Li, X. Zhou, Z. Wang, F. L. Zhang, Y. Li, X. Zhou, Y. Fu and Y. F. Wang, J. Am. Chem. Soc., 2022, 144, 15275–15285 CrossRef CAS PubMed.
- F. Diaba and G. Trenchs, Org. Biomol. Chem., 2023, 22, 388–394 RSC.
- M. Bandyopadhyay, B. Bera, S. Pathak, D. Bhunia, S. Bhadra, S. Patra, M. Pal Chowdhury, J. Escorihuela and M. K. Bera, Adv. Synth. Catal., 2024, 366, 2696–2704 CrossRef CAS.
- T. T. Vo, M. Naher, C. M. Williams and P. V. Bernhardt, J. Org. Chem., 2025, 90, 12709–12716 CrossRef CAS PubMed.
- X. Jia, S. Kramer, T. Skrydstrup and Z. Lian, Angew. Chem., Int. Ed., 2021, 60, 7353–7359 CrossRef CAS PubMed.
- L. Chen, M. Zhou, L. Shen, X. He, X. Li and X. Zhang, Org. Lett., 2021, 23, 4991–4996 CrossRef CAS PubMed.
- K. Nozawa-Kumada, K. Noguchi, T. Akada, M. Shigeno and Y. Kondo, Org. Lett., 2021, 23, 6659–6663 Search PubMed.
- Based on the amount of sodium nitrite (0.8 equivalent of ClO2˙ was generated from ClO2− by dismutation reaction).
- A. Ogawa and D. P. Curran, J. Org. Chem., 1997, 62, 450–451 Search PubMed.
- M. Kirihara, Y. Sakamoto, S. Yamahara, A. Kitajima, N. Kugisaki and Y. Kimura, Synlett, 2022, 1670–1674 Search PubMed.
- M. Kirihara, T. Kawai, A. Kitajima, S. Yamahara, N. Mori and Y. Kimura, ChemistrySelect, 2024, 9, e202403692 Search PubMed.
- Y. Tian and Z. Q. Liu, RSC Adv., 2014, 4, 64855–64859 Search PubMed.
- A ClO2˙ aqueous solution (0.10–0.20 M) was prepared from sodium chlorite and hydrochloric acid, and the gaseous portion in the headspace was collected using a syringe. The gas was introduced directly into an ESR tube containing tetrachloroethylene. Introduction of ClO2˙ was continued until the solution turned yellow. The sample tube was then sealed with a rubber septum and subjected to ESR analysis.
- K. Ohkubo, K. Hirose, T. Shibata, K. Takamori and S. Fukuzumi, J. Phys. Org. Chem., 2017, 30, e3619 Search PubMed.
- K. Ohkubo, K. Suga, K. Morikawa and S. Fukuzumi, J. Am. Chem. Soc., 2003, 125, 12850–12859 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |