Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Make the ylides shine: synthesis and structure–property relationships of phospha-squaraines
Adisorn
Khantong
a,
Alexandre
Chemin
 a,
Olivier
Galangau
a,
Olivier
Galangau
 a,
Boris
Le Guennic
a,
Boris
Le Guennic
 a,
Denis
Jacquemin
a,
Denis
Jacquemin
 *bc and
Pierre-Antoine
Bouit
*bc and
Pierre-Antoine
Bouit
 *a
*a
aUniv Rennes, CNRS, ISCR – UMR 6226, Rennes F-35000, France. E-mail: pierre-antoine.bouit@univ-rennes.fr
bNantes Université, CNRS, CEISAM UMR 6230, F-44000, Nantes, France. E-mail: Denis.Jacquemin@univ-nantes.fr
cInstitut Universitaire de France (IUF), F-75005, Paris, France
First published on 2nd March 2026
Abstract
We report the synthesis and full characterization of a family of four phospha-squaraine dyes, obtained in a single step from squaric acid derivatives. Their structure–property relationships are investigated through a joint experimental/theoretical approach. All of these exotic ylides display strong absorbtion and emission in the red region of the spectrum, a rare feature for phosphorus ylides. Furthermore, they exhibit enhanced photostability compared to a standard squaraine reference.
Polymethine dyes have always been in the limelight in chemistry. They are derivatives in which an even number of electrons is delocalized between two heteroatoms across a π-bridge featuring an odd number of sp2 carbon atoms.1 This unique structure leads to a “cyanine” structure with hallmark properties, including an almost zero bond length alternation (BLA), that is all C–C bonds have lengths close to “one and a half” bonds. These dyes also exhibit sharp, exceptionally intense absorption bands that extend into the NIR region and can be tuned by varying the π-chain length, as well as luminescence with a very small Stokes shift and excellent brightness, including in the NIR.2 After their discovery, polymethines were first used as photography3 or textile dyes,4 and they are now widely applied as components in optoelectronic devices and as bioprobes.5 Owing to their absorption and emission in the biological transparency window, they are particularly suited for biological applications,5 but they are also valuable in material science, especially in nonlinear optics and photovoltaics.6 Squaraine dyes constitute a distinct class of zwitterionic polymethines. They feature an electron-deficient, four-membered central ring derived from squaric acid, substituted with electron-donating aza-heterocycles which structure is stabilized through resonance (Fig. 1a).7,8 Like polymethines, squaraines display intense absorption bands, high molar absorption coefficients, and good photoconductivity. As such, they are excellent chromophores, fluorophores, and semiconductors, with applications ranging from p-type semiconductors in bulk heterojunction solar cells (A, Fig. 1b) to fluorogenic imaging probes for tumor-selective optical imaging (2, Fig. 1b).9,10 In addition, their molecular engineering is rather straightforward, allowing effective tuning of their properties, including shifting their lowest-energy absorption into the NIR. Despite these advantages, further molecular engineering remains necessary to enhance their stability, performance, and tunability.11
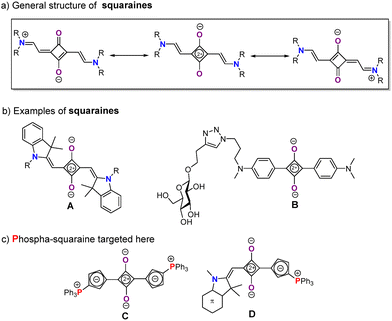 | ||
| Fig. 1 General structure (a), representative examples of squaraines dyes (b),9,10 and phospha-squaraines targeted here (c). | ||
In recent years, we and others have shown that substituting nitrogen with phosphorus in traditional chromophores/semiconductors effectively modulates their properties, leveraging the intrinsic properties of phosphorus in terms of electronegativity, valence, coordination number, aromaticity, and reactivity.12 For example, we recently applied this strategy to nanographenes13 or viologens.14 In 2023, we reported the first evidence of an “ideal polymethine state” (IPS) in phospha-cyanines.15 However, these exotic ylides displayed very weak luminescence. Building on these promising results, we now aim to extend this approach to phospha-squaraines, for which, to the best of our knowledge, no literature has been reported.16
In the present report, we describe the synthesis of a family of phospha-squaraines dyes and systematically investigate their optical properties through a combined experimental and theoretical approach. From this initial study, we conclude that phospha-squaraines are original fluorescent dyes featuring an ylidic fragment, with promising potential for applications in photobiology or material science.
Squaraines synthesis is generally based on Knoevenagel-type condensations with carbonyl derivatives. In this context, we selected cyclopentadienylidene triphenylphosphorane 1 (Scheme 1) as nucleophile to prepare the targeted phospha-squaraines (see Fig. 1c).17 Indeed, 1 is a “stable” ylide that has previously been used to design the first generation of phospha-cyanine dyes as well as other chromophores.18,19 By reacting 1 with squaric acid under standard conditions (nBuOH/toluene reflux), the symmetric phospha-squaraine 2 (Scheme 1) was obtained in low yield (22%) and isolated as a purple solid. The yield is low due to the degradation of ylide into triphenylphosphine/phosphine oxides inducing tedious purification steps. Variation of reaction time/temperature, as well as the use of other solvents such as THF or attempts to scavenge protons (with pyridine or NaOH) or water (with molecular sieves) did not allow to increase the yield. Using diethyl squarate instead of squaric acid as reactant led to monosubstituted derivative 9 featuring a single cyclopentadienylidene triphenylphosphorane (Scheme 1). However, such a compound cannot serve as intermediate toward 2 as deprotection of 9 with NaOH only led to ylide degradation. Finally, compound 2 was characterized by multinuclear NMR and mass spectrometry. In particular, the single resonance in 31P NMR (δ = +14.3 ppm) along with the overall symmetry of the NMR spectra, confirms a fully symmetric structure, consistent with a cyanine in its IPS.15 Based on these promising results, we expanded the molecular diversity by modifying either the central core or the lateral heterocycles. 9 was initially envisaged as key synthon for the divergent synthesis of dissymmetric squaraines 5–6, however its instability under the reaction conditions (vide supra) precluded this strategy. We thus prepared dissymmetric phospha-squaraines adapting Würthner's methodology.20 Condensation of 1 on mono-substituted squaric acids 3–4 afforded two new dyes 5–6 (Scheme 1), each featuring a P-heterocycle on one side and a N-heterocycle (indole) on the other. In this case, the yields were considerably improved (43–60%). Finally, we prepared the cyano-squaraine 8, in which one oxygen atom of the central core is substituted by a dicyanovinylene.20 Such compound also suffers from difficult separation from degradation product leading to very low isolated yield (4%).
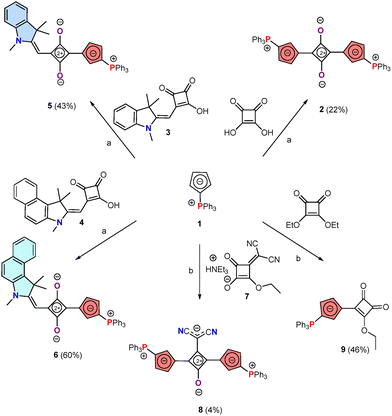 | ||
| Scheme 1 Synthesis of dyes 2, 5–6, 8–9. (a) nBuOH/Toluene, reflux, 2 h; (b) nBuOH/Toluene, reflux, 12 h). | ||
In short, by adapting established synthetic approaches, we synthesized four phospha-squaraines differing in their lateral substituents or central core. All compounds display good solubility in organic solvents and are air and moisture stable.
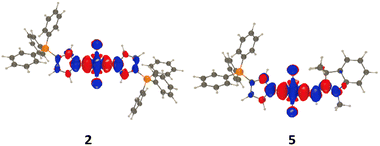
The structural parameters of the phospha-cyanines were investigated computationally at the M06-2X/6-311G(d,p) level (Fig. S37). As expected for such polymethine dyes, the conjugated backbone of all derivatives is fully planar. Regarding the bond distances, the classical features of squaraines dyes are observed. The C–C bonds within the 4-membered ring are rather long (d ∼ 1.46–1.47 Å), while the C–O bonds range from 1.22 to 1.23 Å. Along the polymethine chain, C–C bond distances fall within the “one and a half” bond range (1.38 Å < d < 1.43 Å). Finally, the C–P bonds (d ∼ 1.75 Å) are consistent with typical ylidic C–P bonds, as previously reported in phospha-cyanines.15 Based on these optimized geometries, the compounds exhibit the structural hallmarks of both squaraine cores and stabilized ylides.
The electrochemical properties of 2, 5–6, 8 were investigated by cyclic voltammetry (CV) in dichloromethane solution (Fig. S9–S15 and Table S2). The symmetric phospha-squaraine 2 displays irreversible oxidation and reduction at relatively low potential (Eox = +0.39 V vs. DmFc, Ered = −1.54 V vs. DmFc).21 The electrochemically determined energy gap (Eelec = 1.9 eV) is in good agreement with the optical one (Eopt = 2.2 eV). The irreversibility of these redox processes contrasts with classical squaraines A, which display fully reversible processes.9 Interestingly, structural modifications, whether on the lateral substituents or the central core, have a positive impact on the reversibility of the redox processes. Hence, the dissymmetric compounds 5 and 6 display quasi-reversible reductions at higher potentials, while their oxidations are only moderately affected, consistent with a slight reduction of the energy gap. In the case of 8, oxidation is slightly more difficult (Eox = +0.48 V vs. DmFc) while reduction is easier (Ered = −1.30 V vs. DmFc), resulting in a gap decrease, in agreement with the optical data (vide infra). Notably, both redox processes in 8 are quasi-reversible.
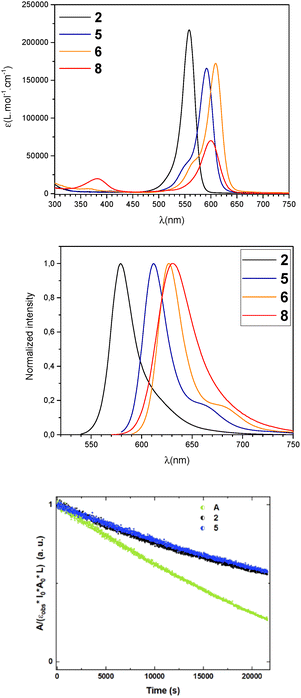
The optical properties of 2, 5–6, 8 were investigated in dilute CH2Cl2 solutions (c = 5 × 10−6 mol L−1, Fig. 2 and Table S2 and Fig. S2-S5).
All derivatives display similar absorption spectra, characterized by a sharp absorption band between 558 nm and 609 nm with high molar extinction coefficients (170![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L mol−1 cm−1 < ε < 220
000 L mol−1 cm−1 < ε < 220![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L mol−1 cm−1) and a vibronic shoulder. These spectra are nicely reproduced by TD-DFT calculations, vide infra. Only the dicyanovinylene-substituted squaraine 8 features a lower extinction coefficient (ε = 70
000 L mol−1 cm−1) and a vibronic shoulder. These spectra are nicely reproduced by TD-DFT calculations, vide infra. Only the dicyanovinylene-substituted squaraine 8 features a lower extinction coefficient (ε = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 L mol−1 cm−1). Overall, these measurements confirm that the new derivatives retain the typical optical features of squaraines dyes. Phospha-squaraine 2 displays a blue-shifted absorption compared to A (Δλ = 88 nm), reflecting the weaker electron-donating ability of the ylidic fragment 1 compared to the corresponding indole. Introducing indole lateral groups with increasing donor strength progressively redshifts the absorption, reaching λ = 609 nm for 6. Substituting one oxygen in the central core with a cyano group, as in 8, also induces a redshift to λ = 600 nm, consistent with trends observed in conventional squaraines. This shift is attributed to a charge-transfer contribution from the dicyanovinylene fragment to the polymethine bridge.9 From UV-Vis. measurements we can conclude that phospha-squaraines show no tendency to aggregate in dilute solution up to 10−4 mol L−1 (Fig. S6), probably due to the steric effect of the tetrahedral phosphorus, a behavior previously observed with phospha-cyanines,15 and contrasting with classical squaraines.22 Interestingly, the photostability of phospha-squaraines 2 and 5 is enhanced compared to reference squaraine A (Fig. 2 and Fig. S16–S21 and Table S3).
000 L mol−1 cm−1). Overall, these measurements confirm that the new derivatives retain the typical optical features of squaraines dyes. Phospha-squaraine 2 displays a blue-shifted absorption compared to A (Δλ = 88 nm), reflecting the weaker electron-donating ability of the ylidic fragment 1 compared to the corresponding indole. Introducing indole lateral groups with increasing donor strength progressively redshifts the absorption, reaching λ = 609 nm for 6. Substituting one oxygen in the central core with a cyano group, as in 8, also induces a redshift to λ = 600 nm, consistent with trends observed in conventional squaraines. This shift is attributed to a charge-transfer contribution from the dicyanovinylene fragment to the polymethine bridge.9 From UV-Vis. measurements we can conclude that phospha-squaraines show no tendency to aggregate in dilute solution up to 10−4 mol L−1 (Fig. S6), probably due to the steric effect of the tetrahedral phosphorus, a behavior previously observed with phospha-cyanines,15 and contrasting with classical squaraines.22 Interestingly, the photostability of phospha-squaraines 2 and 5 is enhanced compared to reference squaraine A (Fig. 2 and Fig. S16–S21 and Table S3).
All of these compounds also display luminescence in the red with low Stokes shifts (σ < 820 cm−1), as classically observed for squaraines.11 The trends observed in the absorption spectra are maintained in the corresponding emission spectra. All compounds display moderate fluorescence quantum yields (0.09 < ϕ < 0.35). While these values are below the best performing squaraines dyes,11 it is noteworthy that fluorescence is uncommon among ylidic derivatives, as only a few phosphorus ylides have been reported to show significant luminescence.23
All those experimental observations, including 0–0 energies, spectral shapes, and the effect of lateral heterocycles and central substituents, were satisfactorily reproduced computationally (Table S4 and Fig. S41). Notably, the calculated 0–0 energies are particularly accurate, with a mean absolute deviation of 0.03 eV only compared to experimental values. Analysis of the electron density difference (EDD, Fig. 3 and Fig. S38) confirmed π-delocalization across the conjugated backbone, displaying hallmark squaraine-like features: (i) alternation of electron density gain and loss at odd and even positions, and (ii) donating effects of the cyclopentadienyl and amino-heterocycles (in 5 and 6) as well as the squaric substituents (keto and/or dicyanovinylene) in all compounds.24 The P-atom is not significantly involved in the electronic absorption (Fig. 3 and Fig. S38), contrasting with our previous observations for phospha-cyanines.15 However, this is consistent with the strong ylidic character (with negligible ylene contribution) of the orbitals involved in the absorption process, confirmed by the aromatic character of the Cp ring as evidenced by NICS(0) calculations (Fig. S42) and NBO analysis (Table S6).25 All these calculations are consistent with the mesomeric structures depicted in Scheme 1. Although the P-atom does not directly participate in the transition, its presence is essential to ensure the chemical stability of the cyclopentadienyl based ylidic fragment, as evidenced by the hyperconjugation visible in the LUMOs of 2, 5–6, 8 (Fig. S39).26
We have reported the synthesis and full characterization of a family of four phospha-squaraine dyes (2, 5–6, 8), prepared in a single step from squaric acid or its derivatives. These ylides-containing dyes absorb and emit in the red region of the spectrum. Incorporation of phosphorus prevents aggregation and enhances photostability, making these dyes rare examples of luminescent compounds featuring a P-ylide. This study paves the way for the development of novel dyes featuring main-group elements, with potential applications in optoelectronic or photobiology.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, spectral data, and detailed DFT outputs. See DOI: https://doi.org/10.1039/d6cc00100a.CCDC 2471886 contains the supplementary crystallographic data for this paper.27
Acknowledgements
This work is supported by the CNRS, the University of Rennes, EUR LUMOMAT, the China-French associated international laboratory in “Functional Organophosphorus Materials”, the GDR Phosphore. A. C. thanks Univ Rennes for PhD grant. This research used resources of the GLiCID Computing Facility (Ligerien Group for Intensive Distributed Computing, https://doi.org/10.60487/glicid, Pays de la Loire, France).References
- (a) S. Dähne, Science, 1978, 199, 1163–1167 CrossRef PubMed; (b) J. Fabian, H. Nakazumi and M. Matsuoka, Chem. Rev., 1992, 92, 1197–1226 CrossRef CAS; (c) A. Mishra, R. Behera, P. K. Behera, B. K. Mishra and G. B. Behera, Chem. Rev., 2000, 100, 1973–2012 CrossRef CAS PubMed.
- S. Pascal, A. Haefele, C. Monnereau, A. Charaf-Eddin, D. Jacquemin, B. Le Guennic, C. Andraud and O. Maury, J. Phys. Chem. A, 2014, 118, 4038 CrossRef CAS PubMed.
- S. Fujita, Organic Chemistry of Photography 2013, Springer, Berlin (US) Search PubMed.
- S. Benkhaya, S. M’rabet and A. El Harfi, Inorg. Chem. Commun., 2020, 115, 107891 CrossRef CAS.
- W. Sun, S. Guo, C. Hu, J. Fan and X. Peng, Chem. Rev., 2016, 116, 7768–7817 CrossRef CAS PubMed.
- P.-A. Bouit, G. Wetzel, G. Berginc, B. Loiseaux, L. Toupet, P. Feneyrou, Y. Bretonnière, K. Kamada, O. Maury and C. Andraud, Chem. Mater., 2007, 19(22), 5325–5335 CrossRef CAS.
- J. J. Mc Ewen and K. J. Wallace, Chem. Commun., 2009, 6339–6351 RSC.
- A. Treibs and K. Jacob, Angew. Chem., Int. Ed. Engl., 1965, 8, 694 CrossRef.
- U. Mayerhöffer, K. Deing, K. Gruss, H. Braunschweig, K. Meerholz and F. Würthner, Angew. Chem., Int. Ed., 2009, 48, 8776–8779 CrossRef PubMed.
- M. Shimi, V. Sankar, M. K. Abdul Rahim, P. R. Nitha, S. Das, K. V. Radhakrishnan and K. G. Raghu, Chem. Commun., 2017, 53, 5433–5436 RSC.
- K. Ilina, W. M. MacCuaig, M. Laramie, J. N. Jeouty, L. R. McNally and M. Henary, Bioconjugate Chem., 2020, 31, 194–213 CrossRef CAS PubMed.
- Main Group Strategies toward Functional Hybrid Materials, ed. T. Baumgartner, F. Jaekle, 2018, John Wiley & Sons, UK Search PubMed.
- P.-A. Bouit, A. Escande, R. Szűcs, D. Szieberth, C. Lescop, L. Nyulászi, M. Hissler and R. Réau, J. Am. Chem. Soc., 2012, 134, 6524 CrossRef CAS PubMed.
- T. Delouche, A. Vacher, E. Caytan, T. Roisnel, B. Le Guennic, D. Jacquemin, M. Hissler and P.-A. Bouit, Chem. – Eur. J., 2020, 26, 8226–8229 CrossRef CAS PubMed.
- A. Chemin, I. Knysh, D. Ari, M. Cordier, T. Roisnel, B. L. Guennic, M. Hissler, D. Jacquemin and P.-A. Bouit, J. Phys. Chem. A, 2023, 127, 10457–10463 CrossRef CAS PubMed.
- There are some examples of squaraines with a pendant phosphoric acid anchoring group, but the P-atom is not involved in the squarainic π-system in these cases: F. M. Jradi, X. Kang, D. O’Neil, G. Pajares, Y. A. Getmanenko, P. Szymanski, T. C. Parker, M. A. El-Sayed and S. R. Marder, Chem. Mater., 2015, 27, 2480–2487 CrossRef CAS.
- F. Ramirez and S. Levy, J. Am. Chem. Soc., 1957, 79, 1957 Search PubMed.
- (a) H. Depoorter, J. Nys and A. van Dormael, Tetrahedron Lett., 1961, 6, 199–205 CrossRef; (b) L. Weimei, Z. Zhenghua, Y. Zhuguang, H. Mengzhen and W. Bingkui, Dyes Pigm., 1990, 14, 211–216 CrossRef.
- K. Zhang, X. Wang, Z. Zhou, J. Guo, H. Liu, Y. Zhai, Y. Yao, K. Yang and Z. Zeng, Angew. Chem., Int. Ed., 2025, 64, e202418520 CrossRef CAS PubMed.
- U. Mayerhöffer, M. Gsänger, M. Stolte, B. Fimmel and F. Würthner, Chem. – Eur. J., 2013, 19, 218–232 CrossRef PubMed.
- I. Noviandri, K. N. Brown, D. S. Fleming, P. T. Gulyas, P. A. Lay, A. F. Masters and L. Phillips, J. Phys. Chem. B, 1999, 103, 6713–6722 CrossRef CAS.
- (a) Y. Xu, Z. Li, A. Malkovskiy, S. Sun and Y. Pang, J. Phys. Chem. B, 2010, 114(25), 8574–8580 CrossRef CAS PubMed; (b) M. Schulz, J. Zablocki, O. S. Abdullaeva, S. Brück, F. Balzer, A. Lützen, O. Arteaga and M. Schiek, Nat. Commun., 2018, 9, 2413 CrossRef PubMed.
- (a) G. Pfeifer, F. Chahdoura, M. Papke, M. Weber, R. Szücs, B. Geffroy, D. Tondelier, L. Nyulászi, M. Hissler and C. Müller, Chem. – Eur. J., 2020, 26, 10534 CrossRef CAS PubMed; (b) T. Delouche, E. Caytan, M. Cordier, T. Roisnel, G. Taupier, Y. Molard, N. Vanthuyne, B. Le Guennic, M. Hissler, D. Jacquemin and P.-A. Bouit, Angew. Chem., Int. Ed., 2022, 61, e202205548 CrossRef CAS PubMed; (c) N. Hashimoto, R. Umano, Y. Ochi, K. Shimahara, J. Nakamura, S. Mori, H. Ohta, Y. Watanabe and M. Hayashi, J. Am. Chem. Soc., 2018, 140, 2046–2049 CrossRef CAS PubMed; (d) O. Fayafrou, E. Lognon, C. Duhayon, J.-B. Sortais, A. Molinari, O. Baslé and Y. Canac, Chem. Commun., 2024, 60, 13602–13605 RSC.
- F. Bassal, A. D. Laurent, B. Le Guennic and D. Jacquemin, Dyes Pigm., 2017, 138, 169–175 CrossRef CAS.
- L. Boubekeur, PhD thesis, École Polytechnique, Palaiseau, France, 2006.
- (a) H. L. Ammon, G. L. Wheeler and P. H. Watts Jr, J. Am. Chem. Soc., 1973, 95, 6158–6163 CrossRef CAS; (b) J. H. Brownie and M. C. Baird, Coord. Chem. Rev., 2008, 252, 1734–1754 CrossRef CAS.
- CCDC 2471886: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nz684.
| This journal is © The Royal Society of Chemistry 2026 |