Development and optimization of an LC-MS/MS method for the detection of Magnolia officinalis extracts in cosmetics: insights from DFT-assisted sample preparation
Yongqi
Yao
 *a,
Guangfeng
Zeng
a,
Zhiyuan
Wang
a,
Jianjun
Xie
a,
Cheng
Chen
*a,
Guangfeng
Zeng
a,
Zhiyuan
Wang
a,
Jianjun
Xie
a,
Cheng
Chen
 b,
Yingye
Hou
a,
Keyin
Xiao
a,
Cuiling
Cai
a,
Jie
Dong
a,
Qing
Liu
*a and
Longyan
Xu
*a
b,
Yingye
Hou
a,
Keyin
Xiao
a,
Cuiling
Cai
a,
Jie
Dong
a,
Qing
Liu
*a and
Longyan
Xu
*a
aFood and Cosmetics Testing Institute, Guangzhou Customs Technology Center, 510423 Guangzhou, People's Republic of China. E-mail: gziqtcyyq@163.com; gdciqlq@163.com; xlyciq@126.com
bSchool of Energy Science and Engineering, University of Science and Technology of China, 230026 Hefei, People's Republic of China
First published on 1st December 2025
Abstract
A sensitive LC-MS/MS method was developed for detecting Magnolia officinalis extracts, targeting magnolol, honokiol, magnoflorine and magnocurarine in complex cosmetic matrices, meeting the growing industry demand for reliable quality control solutions amidst the expanding applications of the extracts. Method optimization systematically addressed matrix effects and sample preparation, supported by density functional theory (DFT) calculations, which provided molecular-level insights into the analyte behavior. Validation confirmed a high extraction recovery of 90.7–106.2%, a low detection limit of 0.15 mg kg−1, excellent linearity with R2 ≥ 0.9994 over 0.5–10 mg kg−1, and precise and accurate intra- and inter-day results with an RSD ≤ 4.15% and accuracies of 90.6–106.3%. This method demonstrated reliability for quality control through its application in spiked creams, aqueous solutions, masks, and oils, concurrently verifying label claims for functional phenolics regarding content and composition and screening for prohibited/restricted alkaloid-substances, demonstrating its dual utility in quality assurance and regulatory compliance.
1 Introduction
Magnolia officinalis, commonly referred to as Houpu or Chuanpu, is a deciduous tree belonging to the Magnoliaceae family. This species is not only a critical component in pharmacology but has also attracted considerable interest due to its outstanding therapeutic potential in rapidly expanding medical aesthetics, food, and cosmetics. In the historical process of practice and exploration, Magnolia officinalis has been widely utilized for various medicinal purposes, particularly in addressing a range of health issues.1 Modern scientific studies have confirmed the presence of numerous bioactive compounds within Magnolia officinalis, such as phenols, alkaloids and flavonoids.2–5 Progressively, extensive research in biofunctional, toxicological, pharmacological, and biomedical areas has validated that key constituents—notably magnolol, honokiol, magnoflorine and magnocurarine—possess strong antioxidant, anti-inflammatory, anti-tumor, antibacterial, sedative, and anti-anxiety properties.6–9With the increasing interest in the use of Magnolia officinalis extracts, especially in the field of cosmetics, the determination and evaluation of the functional components have attracted attention owing to the medicinal value and unique biological activity of these components. Since the 1990s, research has progressively concentrated on refining the extraction methods and analytical techniques to more accurately quantify these bioactive compounds iteratively and incrementally, which not only enhances our understanding of their potential advantages but also facilitates the development of more effective natural products. The high performance liquid chromatography with diode array detection (HPLC-DAD) method, as described by Tsai and Chen in 1992, is recognized for its universality and the ability to verify peak purity using the full UV spectrum.10 This method utilizes lower wavelengths—209 nm for honokiol and 218 nm for magnolol—to improve quantification limits, which showed significant advantages in the early application of magnolia extracts. Additionally, the capillary zone electrophoresis with ultraviolet detection (CZE-UV) method, introduced by Chou's group, provides a viable alternative to high performance liquid chromatography (HPLC), offering comparable analysis times and requires only minimal sample volumes, which made it another effective option for analysis in 1996.11 With the development of detection technology, an optimized liquid chromatography-mass spectrometry (LC-MS) method was developed by Tsai in 2006 for the simultaneous determination of honokiol and magnolol in plant extracts or pharmaceutical preparations.12 The method showed good linearity, acceptable variability, and provided better specificity, sensitivity, and a faster analysis time compared with conventional methods. Hereupon, HPLC and LC-MS, which had become a prevalent analytical technique for the quantitative determination of bioactive compounds in extracts from Magnolia officinalis, were extensively applied across various formulations, including natural foods, functional health supplements, traditional Chinese medicine preparations, cosmetics and pharmaceutical products.13–19 To address the increased demand for incorporating Magnolia officinalis extracts into cosmetics, there is a corresponding rise in the requirement for advanced analytical methods that can accurately and reliably assess both the integrity and concentration of bioactive compounds in complex cosmetic formulations. However, some essential components in cosmetics have negative impacts on the detection and evaluation of functional ingredients in Magnolia officinalis, particularly during the manufacturing, packaging, and analysis processes of cosmetic products. Sunscreen reagents, such as titanium dioxide (TiO2), zinc oxide (ZnO), avobenzone and octinoxate, are efficient photocatalysts, generating electron–hole pairs under UV or visible light.20–22 The pairs facilitate the formation of superoxide radicals and the oxidation of phenols:23 photogenerated electrons reduce the molecular oxygen to form superoxide radicals (O2−), while photogenerated holes oxidize phenolic compounds, generating phenoxyl radicals (Ph-O˙), which further oxidize to form quinone compounds.24 Surface active agents can promote the evolution of phenols, including increasing the contact area of the phenolic molecule, optimizing the polarity of the extraction solvent, forming micelles, adjusting the reaction kinetics model, and enhancing the stability of the quinone products.25 Alkaloids and active enzymes, in conjunction with base additives, such as NaOH and KOH, effectively stabilize aryl oxygen anions, which increases the HOMO energy level of phenolic substances by the formation of active intermediates and subsequently accelerates the oxidation process.26,27 Based on the effects and influences of the aforementioned components, the molecular structure of the phenolic substance being tested is more readily disrupted, leading to misjudgment during the evaluation. To date, few studies have investigated the determination of Magnolia officinalis extracts in cosmetics using LC-MS/MS. With our ongoing research interest in the analysis of these extracts as well as the prevailing trends in the cosmetic industry, herein, we report our latest progress on the simultaneous quantification of magnolol, honokiol, magnocurarine and magnoflorine in cosmetic formulations for optimizing extraction methodologies and enhancing analytical techniques using liquid chromatography-tandem mass spectrometry (LC-MS/MS).
2 Materials and methods
2.1 Chemicals and reagents
Standard substances, magnolol, honokiol, and magnoflorine, were purchased from Anpel-Trace Standard Technical Services Co., Ltd (Shanghai, China). Magnocurarine was purchased from Yuanye Bio-Technology Co., Ltd (Shanghai, China). Ammonium formate (HCOONH4, >99%), HPLC-grade formic acid (HCOOH, >99%) and phosphoric acid (H3PO4, ≥85 wt% in H2O) were purchased from Macklin Biochemical Technology Co., Ltd (Shanghai, China). HPLC-grade acetonitrile, n-hexane and methanol were purchased from Guangzhou Towin Chemical Technology Co., Ltd (Guangdong, China). QuEChERS/SPE sorbents, including PSA, GCB, C18, NH2, ALN, PLS-A, and HLB-pro, as well as silica, were purchased from ANPEL Laboratory Technologies Inc. (Shanghai, China). Light-protected standard stock solutions (c = 10 mg l−1) of each Magnolia officinalis extract in methanol were prepared and stored at −20 °C until use.2.2 Theoretical calculation methods
The geometric structures of magnolol, honokiol, magnocurarine and magnoflorine were optimized in the Solvation Model based on Density (SMD) with the B3LYP/cc-pvdz level28,29 of the Gaussian 16 package by density functional theory (DFT) study. TD-DFT (n states = 100) calculations at identical theoretical levels were also performed to assess the optical energy gaps by computing the lowest singlet–singlet electronic transitions of each molecule. Acetone, dichloromethane, ethanol, acetonitrile, methanol, chlorobenzene, and tetrahydrofuran were used to account for the solvation effect and evaluate the performance of the extract solvent.2.3 HPLC-MS/MS conditions
LC-MS/MS analysis was conducted using an Agilent (CA, USA) HPLC system interfaced with a SCIEX (MA, USA) 4000 QTRAP triple quadrupole mass spectrometer. Mixed standard solutions and sample solutions were separated by (1) an Agilent (CA, USA) Eclipse XDB-C18 column (3.5 µm, 4.6 × 150 mm) using a mobile phase of 10% aqueous solution (0.1% HCOOH) in acetonitrile at a flow rate of 0.5 mL min−1 in negative ion mode for the evaluation of magnolol and honokiol; (2) an Agilent (CA, USA) Extend-C18 column (3.5 µm, 3.0 × 150 mm) using a mobile phase of 20% aqueous solution (0.1% HCOOH + 5 mmol l−1 HCOONH4) in acetonitrile at a flow rate of 0.7 mL min−1 in positive ion mode for monitoring magnoflorine and magnocurarine. The MRM parameters of the four natural products and their conditions are summarized in Table 1. Representative chromatograms, MRM mass spectra, and corresponding mass spectra are presented in Fig. S1–S6.| Moleculesa | Ion mode | Precursor ion [m/z] | Product ion [m/z] | C.V.c [V] | C.E.d [eV] | Retention time [min] |
|---|---|---|---|---|---|---|
| a The moiety in bold in the molecular structure represents a phenolic hydroxyl group with higher reactivity. b Quantitative ions. c Cone voltage. d Collision energy. e Other mass spectrometric conditions were as follows: ion source temperature, 550 °C; ion spray voltage, 4000 V; curtain gas flow, 170 kPa; collision gas flow, 68 kPa; and auxiliary gas flow, 340 kPa. | ||||||
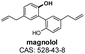
|
Negative | 264.900 | 247.100 | −80.00 | −31.00 | 3.85 |
| 244.800b | −35.00 | |||||
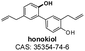
|
Negative | 265.000 | 248.800b | −88.00 | −41.00 | 3.47 |
| 223.000 | ||||||
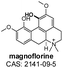
|
Positive | 342.000 | 265.000b | 80.00 | 34.00 | 0.99 |
| 297.100 | ||||||
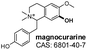
|
Positive | 314.000 | 175.100 | 110.00 | 37.00 | 0.93 |
| 107.100b | ||||||
2.4 UPLC confirmation
Ultra performance liquid chromatography (UPLC) analysis was conducted using a Shimadzu (Kyoto, Japan) UPLC system, and mixed standard solutions and sample solutions were separated by an Athena (Shanghai, China) C18 column (5.0 µm, 4.6 × 250 mm) using a linear gradient of 10–50% aqueous solution (0.1% H3PO4) in acetonitrile at a flow rate of 1.0 mL min−1 (λ = 280 nm). The gradient programs for the mobile phase and chromatographic parameters of 4 Magnolia officinalis extracts are detailed in Tables S1, S2 and Fig. S7.3 Results and discussion
DFT calculations were performed to investigate the extraction performance of several common solvents (Fig. 1). Initially, structural optimizations and assessments of the highest occupied molecular orbital (HOMO) energy levels for magnolol and honokiol were conducted to assess the oxidative properties of these extracts in the extraction solvents (Fig. 1a). In accordance with molecular orbital theory, it is conventionally recognized that the values of the HOMO energy level bear a direct and significant correlation to a molecule's propensity for being oxidized.30,31 Specifically, the oxidation propensity among the magnolol and honokiol in different extraction solvents ranks in the approximate order chlorobenzene (PhCl) > tetrahydrofuran (THF) > dichloromethane (DCM) > acetone > acetonitrile (ACN) > ethanol > methanol, which suggests that the extracts exhibit less oxidizability in methanol, underscoring the role of extract media in the inhibition of oxidative processes. Remarkably, the calculated results of HOMO–LUMO gaps (Fig. 1b) and optical gaps (Fig. 1c) indicate that solvent methanol exhibits higher gap values during extraction, which indicates that methanol may enhance the thermostability and photostability of extracts by solvent effects.32–34 Furthermore, the stability of highly reactive aryl hydroxyl groups can be assessed through their free O–H stretching vibration frequencies.23 Calculation data reveal that in alcoholic solvents, the optimized molecular structures exhibit a higher frequency of free O–H stretching vibrations, which indicates that alcoholic solvents (Fig. 1d), particularly methanol, can suppress the reactivity of hydroxyl groups from bond-cleavage through solvation effects, thereby restricting the tendency of deprotonation.In order to optimize the performance of the mass spectrometer and HPLC, parameters were systematically adjusted with a focus on enhancing sensitivity, improving Signal-to-Noise Ratio (SNR) and peak shape, and reducing sample analysis time while maintaining good resolution (Fig. S1). Particularly, for the mass spectrometer, key configurations such as ion polarity, cone voltage (CV), collision energy (CE) and declustering potential (DP) were meticulously evaluated (Table 1). In the preliminary experiments, significant matrix effects were observed across diverse cosmetic formulations during the analysis of Magnolia officinalis extracts via LC-MS/MS. The single-point recovery rates relative to a methanol standard solution (100 µg l−1) ranged from 33.9% to 136.0% for cream-based matrices, 23.5% to 102.0% for mask-based matrices, 21.0% to 93.2% for aqueous-based matrices, and 17.7% to 114.0% for oil-based matrices across 32 blank samples (4 matrices × 8 samples), with all labels failing to indicate the addition of Magnolia officinalis extract (Table S3). Consequently, proper pretreatment procedures before instrumental analysis, especially effective purification methods, are essential. Initially, SPE/QuEChERS sorbents were employed to purify the samples and reduce matrix effects. For magnolol and honokiol, PSA demonstrated optimal purification performance for cream-based matrices, while silica performed best for aqueous solutions and mask-based matrices because (1) GCB possesses a unique π–π interaction, enabling it to effectively adsorb planar molecules such as phenols; (2) acid–base interactions and hydrogen bonding effects promote the adsorption of phenolic compounds by NH2 and Al–N; and (3) the limited purification efficiency of biological-sample-specific sorbents, like PLS-A and HLB-pro, for cosmetic matrices. However, SPE/QuEChERS adsorbents failed to achieve satisfactory single-point recovery rates and linearity for oil-based samples, particularly at high spike concentration (>5.00 mg kg−1) levels, where deviations of up to 20-fold in the recovery values were observed during the linear regression. Accordingly, further investigation of liquid–liquid extraction methods is imperative to address these limitations. Based on the principle of “like dissolves like” and considering the significant matrix interference caused by lipid substances and lipophilic disruptors in mass spectrometric detection, a purification strategy utilizing immiscible solvent systems was employed. Following a systematic evaluation of multiple co-solvents, an immiscible solvent system using n-hexane/methanol proved effective for oil-based sample purification, achieving robust recovery rates and calibration curves. Similarly, for magnocurarine and magnoflorine, PSA remained optimal for cream and mask-based matrices, while C18 excelled in aqueous systems. Notably, despite the persistent matrix effects caused by hydrophobic compounds and lipid-soluble interferents during conventional QuEChERS procedures in the positive-ion detection mode, the low-temperature LLE purification method consistently maintained effective oil-based sample cleanup, enabling the establishment of reliable quantification regression curves. These findings highlight the effectiveness of the selected sample preparation strategies in mitigating matrix interference, which lays a solid foundation for subsequent quantitative analysis.
All analyses were carried out by employing a matrix-matched approach, wherein the limits of detection (LODs) and quantification (LOQs) for the four Magnolia officinalis extracts were derived by spiking them into cosmetic samples. Table 2 illustrates the validation outcomes, confirming good linearities across five concentration levels ranging from 0.50 to 10.00 mg kg−1, with LOQs of 0.50 mg kg−1 (ppm) and calibration curves displaying a correlation coefficient (R2 ≥ 0.9994). Method performance, encompassing recovery, accuracy, and precision, was verified at low (0.50 mg kg−1), mid (1.00 mg kg−1), and high (5.00 mg kg−1) fortified levels. Recovery rates for all target compounds spanned 90.69–106.25% (n = 8), accompanied by relative standard deviations (RSDs ≤ 4.15%). Intraday accuracy (90.90–106.33%) and precision (RSDs ≤ 3.27%) were evaluated within 24 hours (n = 6), while interday accuracy (90.58–106.00%) and precision (RSDs ≤ 4.07%) were verified for 72 hours (n = 6). Collectively, these findings substantiate the reliability of the LC-MS/MS method for concurrent identification and quantification of Magnolia officinalis extracts in cream, aqueous solutions, masks, and oil-based cosmetic products.
| Matrices | Compound | Linearityc | LOD [mg kg−1] | LOQ [mg kg−1] | Conc.d [mg kg−1] | Recovery [%] | Intra-day (%) | Inter-day (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Slope | Intercept | R 2 | Mean | RSD | Accuracy | Precision | Accuracy | Precision | |||||
| a Unless otherwise noted, 0.20 g of cosmetic sample was combined with standard and methanol, diluted to a final volume of 10.00 mL, and ultrasonicated at room temperature (25.0 ± 2.0 °C) for 15 minutes. After centrifugation, 1.0 mL of supernatant was transferred to a clean tube, mixed with 50.0 mg of QuEChERS adsorbent, and subsequently vortexed for 15 minutes. The mixture was then filtered through a 0.22 µm membrane prior to LC-MS/MS analysis. b After ultrasonic extraction in methanol, 10 mL of n-hexane was added to the extract. The mixture was vortexed and then frozen at −25 °C for 3 hours to achieve complete phase-separation. The upper n-hexane layer was carefully discarded, while the lower layer was filtered through a 0.22 µm membrane prior to LC-MS/MS analysis. c Mass concentration range for linearity: LOQ, × 2, × 5, × 10, × 20. d Concentration. | |||||||||||||
| Creama | Magnolol | 5370 | −4600 | 0.9994 | 0.15 | 0.50 | 0.50 | 101.25 | 2.93 | 100.17 | 2.40 | 100.67 | 2.65 |
| 1.00 | 100.44 | 2.53 | 100.75 | 1.90 | 99.83 | 2.66 | |||||||
| 5.00 | 100.21 | 3.41 | 101.72 | 2.27 | 99.28 | 3.46 | |||||||
| Honokiol | 3520 | −6240 | 0.9996 | 0.15 | 0.50 | 0.50 | 105.00 | 3.30 | 106.00 | 3.27 | 104.33 | 3.46 | |
| 1.00 | 92.19 | 3.49 | 91.67 | 3.00 | 90.58 | 1.58 | |||||||
| 5.00 | 99.65 | 4.15 | 101.65 | 2.16 | 98.37 | 4.07 | |||||||
| Magnoflorine | 2890 | −3500 | 0.9999 | 0.15 | 0.50 | 0.50 | 98.25 | 1.56 | 98.48 | 0.81 | 98.10 | 1.70 | |
| 1.00 | 98.50 | 0.86 | 98.67 | 0.89 | 98.58 | 0.87 | |||||||
| 5.00 | 98.28 | 0.68 | 98.27 | 0.62 | 98.32 | 0.80 | |||||||
| Magnocurarine | 2860 | −1310 | 0.9999 | 0.15 | 0.50 | 0.50 | 96.68 | 1.87 | 97.08 | 1.98 | 96.43 | 1.91 | |
| 1.00 | 94.38 | 1.83 | 94.50 | 2.12 | 94.00 | 1.99 | |||||||
| 5.00 | 94.10 | 0.92 | 94.18 | 0.80 | 93.75 | 0.70 | |||||||
| Aqueous solutionsa | Magnolol | 4190 | 2830 | 0.9997 | 0.15 | 0.50 | 0.50 | 104.25 | 0.85 | 104.17 | 0.39 | 104.33 | 0.99 |
| 1.00 | 101.13 | 1.73 | 101.25 | 1.46 | 100.75 | 1.82 | |||||||
| 5.00 | 106.25 | 1.40 | 106.33 | 1.14 | 106.00 | 1.58 | |||||||
| Honokiol | 2620 | 1340 | 0.9999 | 0.15 | 0.50 | 0.50 | 104.75 | 1.75 | 104.83 | 1.85 | 104.33 | 1.88 | |
| 1.00 | 100.06 | 0.78 | 100.00 | 0.77 | 99.75 | 0.61 | |||||||
| 5.00 | 105.63 | 1.82 | 105.83 | 1.73 | 105.50 | 2.05 | |||||||
| Magnoflorine | 2680 | −7620 | 0.9999 | 0.15 | 0.50 | 0.50 | 100.38 | 1.95 | 99.83 | 1.89 | 100.10 | 1.92 | |
| 1.00 | 97.13 | 1.60 | 97.17 | 1.37 | 97.83 | 1.00 | |||||||
| 5.00 | 98.49 | 1.05 | 98.02 | 0.68 | 98.63 | 1.04 | |||||||
| Magnocurarine | 1160 | 627 | 0.9998 | 0.15 | 0.50 | 0.50 | 102.13 | 1.10 | 102.00 | 1.24 | 101.83 | 1.15 | |
| 1.00 | 100.69 | 0.37 | 100.58 | 0.37 | 100.67 | 0.41 | |||||||
| 5.00 | 97.69 | 1.70 | 98.15 | 1.61 | 98.12 | 1.63 | |||||||
| Maska | Magnolol | 4420 | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
0.9996 | 0.15 | 0.50 | 0.50 | 99.60 | 2.89 | 98.80 | 2.89 | 98.47 | 2.37 |
| 1.00 | 100.94 | 2.02 | 101.92 | 0.90 | 100.42 | 2.12 | |||||||
| 5.00 | 91.06 | 1.14 | 91.33 | 1.18 | 90.60 | 0.71 | |||||||
| Honokiol | 2680 | 6070 | 0.9997 | 0.15 | 0.50 | 0.50 | 102.34 | 1.92 | 103.33 | 0.79 | 101.78 | 1.95 | |
| 1.00 | 104.88 | 2.41 | 105.92 | 1.83 | 104.50 | 2.69 | |||||||
| 5.00 | 92.03 | 0.91 | 92.25 | 0.85 | 92.12 | 1.02 | |||||||
| Magnoflorine | 3520 | −2720 | 0.9999 | 0.15 | 0.50 | 0.50 | 101.88 | 0.82 | 101.83 | 0.74 | 102.00 | 0.88 | |
| 1.00 | 99.75 | 1.37 | 100.42 | 0.66 | 99.42 | 1.37 | |||||||
| 5.00 | 95.26 | 0.71 | 95.42 | 0.75 | 95.13 | 0.66 | |||||||
| Magnocurarine | 3160 | −3110 | 0.9997 | 0.15 | 0.50 | 0.50 | 102.00 | 1.28 | 101.67 | 1.19 | 102.17 | 1.44 | |
| 1.00 | 100.94 | 2.02 | 101.33 | 1.42 | 100.42 | 2.12 | |||||||
| 5.00 | 91.06 | 1.14 | 90.90 | 0.90 | 90.60 | 0.71 | |||||||
| Oilb | Magnolol | 6520 | −42700 | 0.9998 | 0.15 | 0.50 | 0.50 | 103.88 | 1.31 | 104.17 | 1.41 | 103.33 | 1.00 |
| 1.00 | 91.50 | 1.31 | 91.33 | 1.08 | 91.17 | 1.13 | |||||||
| 5.00 | 102.25 | 0.87 | 102.00 | 0.88 | 102.17 | 0.96 | |||||||
| Honokiol | 2560 | 5040 | 0.9999 | 0.15 | 0.50 | 0.50 | 101.74 | 3.61 | 102.23 | 2.07 | 102.57 | 2.49 | |
| 1.00 | 102.94 | 2.43 | 103.75 | 1.69 | 103.50 | 1.67 | |||||||
| 5.00 | 105.50 | 1.90 | 105.50 | 2.22 | 105.17 | 2.12 | |||||||
| Magnoflorine | 3410 | −3060 | 0.9999 | 0.15 | 0.50 | 0.50 | 101.63 | 1.04 | 101.83 | 1.15 | 101.33 | 1.02 | |
| 1.00 | 100.50 | 0.46 | 100.58 | 0.49 | 100.50 | 0.44 | |||||||
| 5.00 | 94.78 | 1.25 | 95.15 | 1.19 | 94.22 | 0.73 | |||||||
| Magnocurarine | 2110 | 2040 | 0.9999 | 0.15 | 0.50 | 0.50 | 90.69 | 0.86 | 90.97 | 0.75 | 90.60 | 0.48 | |
| 1.00 | 100.56 | 0.49 | 100.42 | 0.49 | 100.75 | 0.42 | |||||||
| 5.00 | 100.63 | 0.51 | 100.83 | 0.40 | 100.67 | 0.51 | |||||||
The original intention of the study is twofold: first, to evaluate the content of functional components, magnolol and honokiol, derived from Magnolia officinalis extracts, and second, to monitor magnoflorine and magnocurarine, compounds subject to regulatory restrictions or bans in different countries and territories, due to potential illegal incorporation or residue into cosmetics commercialized in China (Tables 3 and S4). For cream-based cosmetics, especially European brand samples, the purified extracting solutions required up to 100-fold dilution prior to LC-MS/MS analysis, with both magnolol and honokiol contents measuring around 0.5‰ (∼500 mg kg−1). Given the potential attenuation in matrix effects from high dilution, control experiments using matrix-free standard curves for calibration (LC-MS/MS) and UPLC analysis conclusively demonstrated substantial persistent matrix effects. Notably, following the concentration of the purified extract from an anti-acne cream product, magnoflorine (0.0168 mg kg−1) and magnocurarine (0.0206 mg kg−1) were detected, demonstrating the capability of the method to identify the potential prohibited/restricted substances. For aqueous-based cosmetics containing the extract, the typically low dosage necessitates concentration of the extraction solutions before LC-MS/MS analysis. Particularly, in aqueous solution B, only magnolol was detected, with honokiol, magnoflorine, and magnocurarine absent, indicating the situation of mislabelling. Commercial facial masks show considerable variability in extract content, requiring either dilution or concentration for magnolol and honokiol quantification. As evidenced by facial mask E-testing, inter-data comparisons reveal that matrix effects persisted even in diluted mask liquids. The evaluation of oil-based cosmetics and oily raw materials via LC-MS/MS revealed distinct situations. For commercially available oil-based cosmetics, dilution was required prior to detection, yet matrix effects remained significant even after dilution, with magnolol and honokiol content ranging from 0.01‰ to 0.25‰. In stark contrast, the oily raw materials contained substantially higher concentrations of honokiol and magnolol, necessitating multiple dilutions (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000-fold) for instrument testing. At this dilution level, matrix effects became abrogated, and the corresponding detected values aligned closely with the UPLC results. Furthermore, analysis via direct injection of unpurified oil-based raw materials using both LC-MS/MS and UPLC platforms revealed undetectable levels of magnoflorine and magnocurarine, indicating the reliability and safety of domestically sourced raw materials.
000-fold) for instrument testing. At this dilution level, matrix effects became abrogated, and the corresponding detected values aligned closely with the UPLC results. Furthermore, analysis via direct injection of unpurified oil-based raw materials using both LC-MS/MS and UPLC platforms revealed undetectable levels of magnoflorine and magnocurarine, indicating the reliability and safety of domestically sourced raw materials.
| Samples | Magnolol [mg kg−1] | Honokiol [mg kg−1] | Magnocurarine [mg kg−1] | Magnoflorine [mg kg−1] | ||
|---|---|---|---|---|---|---|
| LC-MS/MSa | HPLCm | LC-MS/MS | HPLCm | |||
a Unless otherwise noted, 0.20 g of cosmetic sample was diluted to a final volume of 10.00 mL with solvent methanol and ultrasonicated at room temperature (25.0 ± 2.0 °C) for 15 minutes. After centrifugation, 1.0 mL of supernatant was transferred to a clean tube, mixed with 50.0 mg of QuEChERS adsorbent, and subsequently vortexed for 15 minutes. The mixture was then filtered through a 0.22 µm membrane prior to the LC-MS/MS analysis.
b The purified extraction solution underwent a 100-fold dilution.
c The purified extraction solution underwent a 20-fold concentration.
d The purified extraction solution underwent a 100-fold concentration.
e The purified extraction solution underwent a 10-fold concentration.
f The purified extraction solution underwent a 50-fold dilution.
g The purified extraction solution underwent a 10-fold dilution.
h The purified extraction solution underwent a 50-fold concentration.
i The purified extraction solution underwent a 5-fold dilution.
j The purified extraction solution underwent a 20-fold dilution.
k The unpurified extraction solution underwent a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000-fold dilution.
l Quantification using a non-matrix matched calibration curve (methanol standard solution).
m The supernatant was filtered through a 0.22 µm membrane without purification and directly subjected to UPLC analysis.
n The cosmetic packaging states that Magnolia officinalis extract is present as a kind of trace ingredient.
o The unpurified filtrate underwent a 100-fold dilution. 000-fold dilution.
l Quantification using a non-matrix matched calibration curve (methanol standard solution).
m The supernatant was filtered through a 0.22 µm membrane without purification and directly subjected to UPLC analysis.
n The cosmetic packaging states that Magnolia officinalis extract is present as a kind of trace ingredient.
o The unpurified filtrate underwent a 100-fold dilution.
|
||||||
| Cream A | 4.55 × 102b | 4.61 × 102 | 5.35 × 102b | 5.02 × 102 | n.d.a,m | n.d.a,m |
| 3.35 × 102l | 4.35 × 102l | |||||
| Cream B | 6.45 × 102b | 6.46 × 102 | 4.72 × 102b | 4.24 × 102 | n.d.a,m | n.d.a,m |
| 3.03 × 102l | 3.19 × 102l | |||||
| Cream C | 5.58 | 1.05 | 2.06 × 10−2h | 1.68 × 10−2h | ||
| Cream D | 7.95 × 10−2c | 4.93 × 10−2c | n.d. | n.d. | ||
| Cream E | 1.80 × 102b | 1.74 × 102 | 5.20 × 102b | 4.88 × 102 | n.d.a,m | n.d.a,m |
| 1.06 × 102l | 3.91 × 102l | |||||
| Aqueous A | n.d.a,d | n.d.a,d | n.d. | n.d. | ||
| Aqueous Bn | 1.27 × 10−2d | n.d.d | n.d. | n.d. | ||
| Aqueous C | 7.65 × 10−2e | 2.20 × 10−1e | n.d. | n.d. | ||
| Aqueous D | 8.25 × 10−2e | 2.13 × 10−1e | n.d. | n.d. | ||
| Aqueous En | 8.60 × 10−2d | n.d.a,d | n.d. | n.d. | ||
| Mask An | 7.85 × 10−2e | 3.31 × 10−1e | n.d. | n.d. | ||
| Mask Bn | n.d.a,d | n.d.a,d | n.d. | n.d. | ||
| Mask C | 1.32 × 10−2d | 1.70 × 10−2d | n.d. | n.d. | ||
| Mask Dn | 2.19 | 2.64 | n.d. | n.d. | ||
| Mask E | 1.15 × 102f | 1.26 × 102 | 3.25 × 102f | 3.28 × 102 | n.d.a,m | n.d.a,m |
| 7.85 × 101l | 2.70 × 102l | |||||
| Oil An | 1.49 × 101g | 1.31 × 101g | n.d. | n.d. | ||
| Oil B | 2.81 × 102b | 2.77 × 102 | 2.51 × 102b | 2.67 × 102 | n.d. | n.d. |
| 4.19 × 102l | 4.03 × 102l | |||||
| Oil C | 4.35 × 10−2h | 5.45 × 10−2h | n.d. | n.d. | ||
| Oil Dn | 1.01 × 101i | 4.85 × 101i | n.d. | n.d. | ||
| Oil E | 1.70 × 101j | 1.11 × 102j | n.d. | n.d. | ||
| Raw material A | 2.32 × 104k,l | 2.25 × 104o | 1.92 × 104k,l | 2.05 × 104o | n.d.a,m | n.d.a,m |
| Raw material B | 7.15 × 104k,l | 7.34 × 104o | 9.15 × 104k,l | 9.50 × 104o | n.d.a,m | n.d.a,m |
| Raw material C | 1.63 × 104k,l | 1.87 × 104o | 1.49 × 104k,l | 1.68 × 104o | n.d.a,m | n.d.a,m |
4 Conclusions
A robust LC-MS/MS method was developed to determine 4 Magnolia officinalis bioactive compounds in cosmetic formulations. The systematic optimization of chromatographic conditions and sample pretreatment protocols enabled baseline separation of the structurally analogous phenolic isomers magnolol and honokiol on an Eclipse XDB-C18 column, without requiring unconventional mobile phase compositions or gradient elution programs. Additionally, separation of the alkaloids magnoflorine and magnocurarine was achieved using an Extend-C18 column. The QuEChERS method and low-temperature LLE method were optimized to mitigate matrix interferences. Validation of the developed LC-MS/MS method assessed the LOD, LOQ, linearity, recovery rate, accuracy, and precision of all four analytes. The application of this method to a commercial anti-acne cream confirmed magnoflorine and magnocurarine as potential prohibited/restricted substances; when applied to a commercial skin softener, it revealed mislabeling practices with the sole addition of magnolol instead of Magnolia officinalis (bark) extracts. Furthermore, a UPLC method evaluated matrix effects for LC-MS/MS under varying dilution conditions, revealing effects dependent on both the sample matrix and dilution extent. Although UPLC achieved adequate separation of the four Magnolia officinalis extracts, quantitative analysis was limited to high-concentration samples due to significant sensitivity limitations. Therefore, the developed LC-MS/MS method is considered reliable from the perspective of practicality and sensitivity. Further investigations into cosmetic sample pretreatment strategies for UPLC analysis, as well as the evaluation of rinse-off cosmetics samples, are ongoing in our laboratory.Author contributions
Yongqi Yao: conceptualization, methodology, writing – review & editing, writing – original draft, funding acquisition, formal analysis, investigation. Guangfeng Zeng: formal analysis, investigation. Zhiyuan Wang: conceptualization. Jianjun Xie: methodology, funding acquisition. Cheng Chen: visualization, theoretical calculation. Yingye Hou: validation. Keyin Xiao: formal analysis. Cuiling Cai: data curation. Jie Dong: resources. Qing Liu: funding acquisition. Longyan Xu: resources.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ay01630d.Acknowledgements
This work was financially supported by the Research Project of General Administration of Customs of China (2025HK137, 2024HK085), the Guangdong Basic and Applied Basic Research Foundation (2021A1515110160) and the Quality Improvement Strategy Special Project of Dongguan City (KYHZ2023A18).References
- H. Zhang, J. Tang, H. Cao, C. Wang, C. Shen and J. Liu, J. Ethnopharmacol., 2025, 338, 119007 CrossRef CAS PubMed.
- R. Yan, W. Wang, J. Guo, H. Liu, J. Zhang and B. Yang, Molecules, 2013, 18, 7739–7750 CrossRef CAS PubMed.
- M. Rădulescu, C. Jianu, A. T. Lukinich-Gruia, M. Mioc, A. Mioc, C. Soica and L. G. Stana, Antioxidants, 2021, 10, 1081 CrossRef.
- K. Xu, C. Li, C. Li, J. Ma, Y. Zang, F. Ye and D. Zhang, Org. Chem. Front., 2021, 8, 4833–4838 RSC.
- Z. Xue, X. Zhang, H. Peng, S. Zhu, J. You, T. Zhou, L. Yu, C. Song and B. Yang, Phytomedicine, 2022, 98, 153957 CrossRef CAS.
- H. Li, Y.-H. He, Y.-M. Hu, Q.-R. Chu, Y.-J. Chen, Z.-R. Wu, Z.-J. Zhang, Y.-Q. Liu, C.-J. Yang, H.-J. Liang and Y.-F. Yan, J. Agric. Food Chem., 2021, 69, 11781–11793 CrossRef CAS PubMed.
- X. Song, L. Liu, X. Wu, Y. Liu and J. Yuan, Int. J. Mol. Sci., 2021, 22, 7769 CrossRef CAS.
- L. Luo, T. Wu, M. Ji, J. Xiang, Y. Zou and Y. Liao, Int. Immunopharmacol., 2024, 133, 112098 CrossRef CAS PubMed.
- J. M. Sampieri-Morán, D. A. Bravo-Alfaro, E. Uribe-Lam, G. Luna-Bárcenas, M. Montiel-Sánchez, L. del C. Velasco-Rodríguez, A. A. Acosta-Osorio, M. Ferrer and H. S. García, Eur. J. Pharm. Biopharm., 2025, 208, 114627 CrossRef PubMed.
- T.-H. Tsai and C.-F. Chen, J. Chromatogr. A, 1992, 598, 143–146 CrossRef CAS.
- C. Y. C. Chou, T. H. Tsai, M. F. Lin and C. F. Chen, J. Liq. Chromatogr. Relat. Technol., 1996, 19, 1909–1915 CrossRef CAS.
- Y.-T. Wu, L.-C. Lin and T.-H. Tsai, Biomed. Chromatogr., 2006, 20, 1076–1081 CrossRef CAS PubMed.
- Y.-L. Sheng, J.-H. Xu, C.-H. Shi, W. Li, H.-Y. Xu, N. Li, Y.-Q. Zhao and X.-R. Zhang, J. Ethnopharmacol., 2014, 155, 1568–1574 CrossRef CAS.
- D.-D. Hu, Q.-B. Han, L. L.-D. Zhong, Y.-H. Li, C.-Y. Lin, H.-M. Ho, M. Zhang, S.-H. Lin, L. Zhao, T. Huang, H. Mi, H.-S. Tan, H.-X. Xu and Z.-X. Bian, J. Chromatogr. B, 2015, 1000, 136–146 CrossRef CAS.
- K. Luo, Q. Shi and F. Feng, J. Chromatogr. B, 2017, 1040, 260–272 CrossRef CAS.
- K. Guo, C. Tong, Q. Fu, J. Xu, S. Shi and Y. Xiao, J. Pharm. Biomed. Anal., 2019, 170, 153–160 CrossRef CAS.
- D. Bui, L. Li, T. Yin, X. Wang, S. Gao, M. You, R. Singh and M. Hu, J. Agric. Food Chem., 2020, 68, 6576–6587 CrossRef CAS PubMed.
- J. L. Benedé, E. Rodríguez, A. Chisvert and A. Salvador, Anal. Lett., 2021, 54, 1510–1521 CrossRef.
- H. Li, Y. Lou, Y. Shu, W. Jin, X. Yao, J. Song, Y. Chen and B. Nie, Chin. J. Anal. Chem., 2025, 53, 100488 Search PubMed.
- H. Yao, M. Fan, Y. Wang, G. Luo and W. Fei, J. Mater. Chem. A, 2015, 3, 17511–17524 RSC.
- M. M. Mention, A. L. Flourat, C. Peyrot and F. Allais, Green Chem., 2020, 22, 2077–2085 RSC.
- C. Kuroda, T. Tsuchida, C. Tsunoda, M. Minamide, R. Hiroshige and S. Goto, Langmuir, 2025, 41, 1429–1445 CrossRef CAS.
- A. Baschieri, L. Pulvirenti, V. Muccilli, R. Amorati and C. Tringali, Org. Biomol. Chem., 2017, 15, 6177–6184 RSC.
- N. Y. Shin, E. Tsui, A. Reinhold, G. D. Scholes, M. J. Bird and R. R. Knowles, J. Am. Chem. Soc., 2022, 144, 21783–21790 CrossRef CAS.
- H. Ohta, K. Tobayashi, A. Kuroo, M. Nakatsuka, H. Kobayashi, A. Fukuoka, G. Hamasaka, Y. Uozumi, H. Murayama, M. Tokunaga and M. Hayashi, Chem.–Eur. J., 2019, 25, 14762–14766 CrossRef CAS.
- L. Zhang, R. Liang, C. Hang, H. Wang, L. Sun, L. Xu, D. Liu, Z. Zhang, X. Zhang, F. Chang, S. Zhao and W. Huang, Green Chem., 2020, 22, 2498–2504 RSC.
- Y. Yao, S. Su, N. Wu, W. Wu and H. Jiang, Org. Chem. Front., 2022, 9, 5125–5132 RSC.
- Y. Yao, W. Yang, C. Wang, F. Gu, W. Yang and D. Yang, Asian J. Org. Chem., 2019, 8, 506–513 CrossRef CAS.
- Y. Yao, W. Yang, Q. Lin, W. Yang, H. Li, L. Wang, F. Gu and D. Yang, Org. Chem. Front., 2019, 6, 3360–3364 RSC.
- C. Chen, X. Yan and L. Zhang, ChemElectroChem, 2022, 9, e202101628 CrossRef CAS.
- C. Chen, J. Guo, C. Wu, X. Duan and L. Zhang, ACS Appl. Mater. Interfaces, 2024, 16, 8733–8741 CrossRef CAS.
- A. Stachowiak, K. Kędzierski, B. Barszcz, K. Kotwica and D. Wróbel, J. Mol. Liq., 2021, 341, 116800 CrossRef CAS.
- K. Kawabata and K. Takimiya, Chem.–Eur. J., 2021, 27, 15660–15670 CrossRef CAS PubMed.
- Y. Wang, Y. Ji, Y. Yang, Z. Chen, H. Sun, X. Wang, Z. Zou and H. Huang, ACS Energy Lett., 2024, 9, 336–345 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |

