A FRET-based NCA fluorescent probe for highly sensitive and selective detection of hydrogen sulfide
Lingxiao
Xiong
a,
Xin
Liu
a,
Wanmeng
Li
a,
Zhiyu
Xie
a,
Shaowu
Lv
*b and
Guodong
Feng
 *a
*a
aCollege of Chemistry, Jilin University, Changchun, Jilin 130021, China. E-mail: fenggd@jlu.edu.cn
bLaboratory for Molecular Enzymology and Engineering of the Ministry of Education, School of Life Science, Jilin University, Changchun, Jilin 130021, China. E-mail: lvsw@jlu.edu.cn
First published on 11th December 2025
Abstract
A Förster resonance energy transfer (FRET)-based fluorescent probe (NCA) was developed for highly sensitive and selective detection of hydrogen sulfide (H2S). In this design, coumarin (CA) serves as the fluorescent donor and nitrobenzoxadiazole (NB) acts as an energy acceptor and reactive site. Upon exposure to H2S, cleavage of the NB unit interrupts the FRET process and restores CA fluorescence, producing a 16-fold signal enhancement at 463 nm with a detection limit of 0.11 µM. Under optimized conditions (Britton–Robinson (BR) buffer, pH 8, 15 min), NCA enables rapid and selective detection of H2S over competing biological species. Practical applications were demonstrated by integrating NCA into a smartphone-assisted test strip platform, which achieved recoveries of 98.7–109.9% in tap and lake water. Furthermore, NCA successfully imaged both endogenous and exogenous H2S in human lung carcinoma (A549) cells with low cytotoxicity. These results highlight the potential of NCA as a versatile tool for environmental monitoring and biological studies of H2S.
1 Introduction
Hydrogen sulfide (H2S) is an essential gaseous signaling molecule that regulates diverse physiological and pathological processes, including oxidative stress, inflammation, and mitochondrial bioenergetics.1–5 Dysregulated H2S levels are associated with neurodegenerative and cardiovascular diseases as well as environmental toxicity, highlighting the importance of accurate detection in both biological and environmental systems.6–10 Conventional techniques such as electrochemical assays and gas chromatography provide quantitative analysis but lack the spatial and temporal resolution required for real-time monitoring in living organisms.11–14Fluorescent probes have therefore emerged as attractive alternatives due to their high sensitivity, noninvasiveness, and compatibility with bioimaging.15–19 However, many reported H2S probes still suffer from drawbacks such as strong background interference, slow response kinetics, or poor selectivity against endogenous thiols like cysteine (Cys) and glutathione (GSH).20–23 The rational design of donor–acceptor pairs is thus critical to improving sensitivity, selectivity, and signal-to-noise ratio.
Förster resonance energy transfer (FRET) is a powerful strategy for developing ratiometric or “turn-on” probes, since fluorescence output can be finely modulated by donor–acceptor energy transfer.24–26 Yet existing FRET probes for H2S often exhibit insufficient spectral overlap, sluggish reaction rates, or cross-reactivity with biothiols.27–29 Careful fluorophore selection is therefore necessary, as spectral overlap, quantum yield, and photostability dictate overall performance.30,31
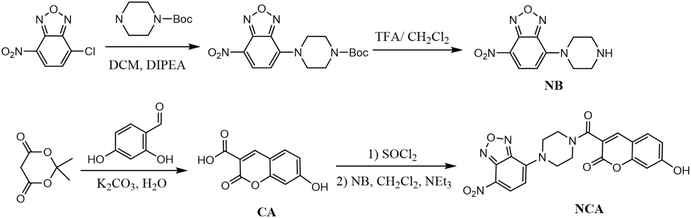
In this work, a FRET-based probe (NCA) was designed. In the probe, coumarin (CA) acts as a high-quantum-yield donor32,33 and nitrobenzoxadiazole (NB) serves as a non-emissive acceptor and H2S-reactive site.34,35 As illustrated in Scheme 1, the NB unit is selectively cleaved by H2S, leading to disruption of the FRET process and restoration of CA fluorescence. This design minimizes background emission, enables rapid fluorescence recovery, and ensures high selectivity toward H2S over other biological species. A 16-fold fluorescence enhancement with a detection limit of 0.11 µM was achieved, and broad applicability was demonstrated in environmental water analysis, smartphone-assisted test strips, and cellular imaging.
2 Materials and methods
2.1 Reagents, instruments, and synthetic procedure
Specific details regarding chemical reagents, instrumentation, and all synthesis procedures are provided in the SI. The fluorescent probe NCA was synthesized based on the route outlined in Scheme 1.2.2 Smartphone imaging
Fluorescence images were acquired using a high-resolution smartphone, and corresponding RGB values were analyzed with the Color Coll app. The non-fluorescent test strips (1 × 3 cm) were used as substrates. These strips were immersed in a 10 µM NCA solution and air-dried to serve as fluorescence sensors before sample application. Images were captured under 365 nm UV illumination.2.3 Environmental water sample analysis
Environmental water samples were collected from tap water, Yan Lake, and Qing Lake, followed by filtration. A water solution system was prepared, in which NCA was dissolved to a final concentration of 10 µM. Na2S was then added to obtain solutions with different concentrations (10, 20, and 50 µM). The fluorescence spectra were recorded at an emission wavelength of 463 nm.2.4 Bioimaging in cells
A549 cells were cultured in DMEM supplemented with 9% fetal bovine serum and 1% penicillin-streptomycin at 37 °C in a humidified incubator with 5% CO2. For cytotoxicity assessment, cells were seeded in 96-well plates at 1 × 105 cells per mL (100 µL per well) and incubated for 20 hours, followed by exposure to NCA at concentrations of 0, 2.5, 5, 10, 20, 40, and 80 µM for 24 hours. Cell viability was determined using the CCK-8 assay, with 10 µL of CCK-8 solution added per well, a 2-hour incubation, and absorbance measurement at 450 nm.For fluorescence imaging of H2S, A549 cells were seeded in six-well plates and divided into four groups: (1) control: incubation with 10 µM NCA for 24 hours, followed by two PBS washes and fixation with 4% paraformaldehyde for 20 minutes before imaging. (2) Endogenous H2S detection: pre-treatment with 100 µM Cys for 1 hour to induce H2S generation, followed by incubation with 10 µM NCA for 24 hours. (3) Exogenous H2S detection (low concentration): pre-treatment with 50 µM Na2S for 1 hour, washing, and incubation with 10 µM NCA for 24 hours. (4) Exogenous H2S detection (high concentration): pre-treatment with 75 µM Na2S for 1 hour, followed by identical washing and incubation procedures with 10 µM NCA for 24 hours. Following treatment, all samples were processed identically, and fluorescence images were acquired using an inverted fluorescence microscope equipped with blue, green, and red channels.
3 Results and discussion
3.1 Fluorescence spectral properties of NCA
The fluorescence and absorption spectra of the coumarin (CA) and nitrobenzoxadiazole (NB) moieties of the probe NCA were examined, and the results are presented in Fig. 1A. The fluorescence emission spectrum of CA shows substantial overlap with the absorption spectrum of NB, satisfying the spectral prerequisites for Förster resonance energy transfer (FRET). Consequently, NCA exhibits nearly complete quenching of the CA emission at 463 nm (Fig. 1B, purple curve). The negligible fluorescence signal confirms that NB is intrinsically non-emissive in aqueous solution and that the transferred energy from CA is predominantly dissipated through non-radiative pathways.Upon H2S-triggered cleavage of the NB group, NBD is released, interrupting the FRET process and resulting in a 16-fold fluorescence enhancement of the CA fluorophore (Fig. 1B, blue curve). Absolute fluorescence quantum yields (Φ) of NCA and its H2S-reacted product were measured in Britton–Robinson buffer (pH 8.0) using an integrating sphere coupled to an Edinburgh Instruments FLS-1000 spectrometer, as described in the SI. The intact probe displayed a very low Φ of 1.34%, whereas the reaction product exhibited a markedly increased Φ of 48.99%, demonstrating substantial restoration of fluorescence following H2S activation.
The sensing mechanism of NCA toward H2S was proposed based on experimental observations and analytical data. As illustrated in Fig. 1C, H2S acts as a nucleophile to cleave the electron-deficient C–N bond in NCA via thiolysis, thereby disrupting the donor–acceptor energy transfer system. This bond cleavage abolishes the Förster resonance energy transfer (FRET) effect, leading to fluorescence recovery at 463 nm.
To confirm the proposed sensing mechanism, NCA was treated with Na2S in DMSO-d6 and analyzed by 1H NMR analysis (400 MHz). Upon reaction completion, the diagnostic aromatic proton signals of the NBD fluorophore (δ 8.55–6.67) completely disappeared, while new resonances corresponding to the released coumarin analogue (CAA) emerged, including its characteristic doublet peaks at δ 5.41 and 5.36 (Fig. S4). These findings align with previously reported H2S-responsive NBD probes, reinforcing that the sensing mechanism is driven by the nucleophilic attack of HS− at the C–N bond, triggering NBD release and restoring the fluorescence of CA at 463 nm.
3.2 Optimization of test condition for H2S detection
The effect of water content on the fluorescence response of NCA (10 µM) toward H2S (50 µM) was systematically evaluated (Fig. 2A). In the absence of Na2S, the fluorescence intensity of NCA remained nearly constant across various water content levels (10–99%), demonstrating excellent probe stability. Upon the addition of Na2S, fluorescence intensity increased progressively with rising water content, reaching a maximum enhancement at 99% water content. This phenomenon was attributed to dual synergistic effects mediated by hydration: elevated water content not only enhances the conformational flexibility of NCA molecules to reduce steric hindrance around the electron-deficient C–N bond, but also facilitates H2S deprotonation to generate nucleophilic HS− species. Consequently, a 99% water content was selected as the optimal condition for subsequent H2S detection.The pH dependence of the fluorescence response was systematically investigated (Fig. 2B). At pH < 6, the fluorescence change (ΔI) remained negligible, as H2S exists mainly in its protonated and less nucleophilic form (pKa1 ≈ 6.98). As the pH increased from 6 to 8, gradual deprotonation shifted the equilibrium toward HS− formation, enhancing nucleophilic attack on the C–N bond and leading to a pronounced fluorescence increase via release of the NBD group. Above pH 8, the response approached saturation, as the system predominantly existed in the HS− form and the nucleophile concentration reached its effective maximum (pKa2 ≈ 13.9). Therefore, pH 8 was selected as the optimal condition for sensing. Under these conditions, the probe responds to the total dissolved sulfide species (H2S/HS− equilibrium), in accordance with the widely adopted usage of the term “H2S probe” in analytical sensing.
Finally, the stability and response time of NCA were assessed under the optimized conditions by monitoring its fluorescence intensity over time in a BR buffer (pH 8) system (Fig. 2C). In the absence of Na2S, the fluorescence intensity remained unchanged, reaffirming the probe's stability. Upon addition of Na2S (50 µM), the fluorescence intensity increased progressively, reaching a plateau within 15 minutes. Based on these results, a reaction time of 15 minutes was adopted for subsequent detection experiments.
Unless otherwise stated, all subsequent fluorescence sensing and selectivity experiments were conducted under the optimized conditions of Britton–Robinson (BR) buffer (pH 8.0).
3.3 Selectivity and anti-interference performance of NCA
To assess the selectivity and anti-interference capability of NCA for H2S, its fluorescence response was measured in the presence of various potential interfering species. These interferents included metal cations (Na+, Ca2+, K+, Mg2+, Cu2+, Al3+, Zn2+, NH4+, Fe3+, Fe2+, Ni2+, Li+, Ba2+), anions (SO32−, SO42−, Br−, HCO3−, Cl−, PO43−, CO32−), amino acids (Cys, Hcy, GSH), and reactive oxygen species (S2O32−). In addition, Se and Te standard solutions were examined under identical sensing conditions.Each analyte was individually introduced into the probe solution, and the fluorescence intensity of NCA at 463 nm was recorded. As shown in Fig. S5, negligible fluorescence changes were observed with the addition of any of the tested species—even at high concentrations (250 µM for Cys, Hcy, and GSH)—whereas a significant fluorescence enhancement was obtained upon the introduction of Na2S. This selective response is attributed to the strong nucleophilicity of H2S, which triggers a thiolysis reaction of NCA. Furthermore, the anti-interference ability of NCA was evaluated by spiking Na2S into solutions containing these potential interfering species. As illustrated in Fig. 2D, the fluorescence response toward H2S remained robust and unaffected by the coexisting analytes. These results confirm that NCA exhibits high selectivity and a strong anti-interference capability, underscoring its potential utility for H2S detection in biologically and environmentally relevant samples.3.4 Detection performance of NCA for H2S detection
Fluorescence titration experiments were performed to evaluate the sensitivity of NCA toward H2S. A series of solutions containing 10 µM NCA and varying concentrations of Na2S (0–500 µM) were prepared, and the corresponding fluorescence spectra were recorded. As depicted in Fig. 2E, the fluorescence intensity at 463 nm increased in a concentration-dependent manner with rising Na2S levels. This progressive enhancement of blue fluorescence is indicative of a rapid “turn-on” response resulting from the immediate disruption of the FRET process upon H2S-mediated cleavage of the NB acceptor.Moreover, a good linear relationship (R2 = 0.98) was established between the fluorescence intensity and H2S concentrations in the range of 0.5–100 µM (Fig. 2F). At concentrations above 100 µM, the fluorescence response gradually approached a saturation plateau. The limit of detection (LOD) was calculated to be 0.11 µM based on a signal-to-noise ratio (S/N = 3), demonstrating the high sensitivity of NCA toward H2S. Compared with recently reported H2S probes (Table S1), NCA exhibits a lower detection limit, rapid response, and broad applicability, ranging from aqueous and environmental water analysis to gaseous H2S sensing and cellular imaging. These results confirm that NCA is a robust and versatile probe for quantitative H2S detection in complex environments.
3.5 Integrated smartphone platform for H2S detection
To establish a field-deployable platform for H2S across aqueous and atmospheric matrices, we developed an integrated strategy combining smartphone-assisted test strips and vapor-phase quantification.For aqueous sample analysis, NCA-functionalized test strips were fabricated by immobilizing the probe on test strips. Upon exposure to H2S-containing solutions (1–50 µM), these strips exhibited concentration-dependent fluorescence enhancement from pale to intense blue under 365 nm UV illumination. Quantitative analysis via smartphone RGB extraction revealed a linear response between the B/(R + G + B) ratio and H2S concentration (y = 0.0036x + 0.4051, R2 = 0.99), enabling rapid field quantification (Fig. 3A).
Given the critical need to detect hazardous levels of H2S where rapid olfactory fatigue occurs, we extended the platform to vapor-phase quantification. Initial qualitative analysis confirmed NCA's reactivity: exposure to H2S from 0.5 mM Na2S/HCl induced a distinct yellow-to-purple color transition (Fig. 3a and b) and fluorescence turn-on (Fig. 3c and d), verifying FRET disruption. Subsequent quantitative assessment using controlled Na2S concentrations (0.05–0.5 mM) with NCA solution revealed concentration-dependent fluorescence enhancement (Fig. 3e), with RGB analysis demonstrating robust linear correlation within 0.05–0.2 mM (y = 0.5820x + 0.5461, R2 = 0.99).
This validated platform delivers rapid, cost-effective H2S monitoring, showing significant potential for environmental surveillance and biological studies requiring real-time sulfide assessment.
3.6 Detection of H2S in environmental water samples
H2S is a highly toxic, water-soluble gas that poses significant risks to aquatic ecosystems and living organisms. To assess the practical applicability of NCA for environmental water analysis, its ability to detect H2S in real water samples was investigated. Specifically, Na2S at concentrations of 10, 20, and 50 µM was spiked into three different water sources (tap water, Yan Lake, and Qing Lake), with parallel blank samples analyzed. The fluorescence intensity at 463 nm was recorded (Fig. S6). As summarized in Table 1, the recoveries of Na2S in these samples ranged from 98.7% to 109.9%, with relative standard deviations (RSDs, n = 3) below 6.34%, thereby confirming the high accuracy and reproducibility of the method. These results demonstrate that NCA effectively detects H2S in diverse aqueous environments, highlighting its potential as a reliable tool for environmental water quality monitoring.| Samples | Na2S added (µM) | Na2S found (µM) | RSD (%) | Recovery (%) |
|---|---|---|---|---|
| Tap water | 0 | Not detected | ||
| 10 | 10.1 | 1.15 | 100.9 | |
| 20 | 21.0 | 3.51 | 105.0 | |
| 50 | 50.3 | 6.08 | 100.5 | |
| Yan lake | 0 | Not detected | ||
| 10 | 10.9 | 6.34 | 109.9 | |
| 20 | 20.8 | 1.22 | 103.8 | |
| 50 | 50.3 | 4.04 | 100.6 | |
| Qing lake | 0 | Not detected | ||
| 10 | 10.6 | 1.90 | 106.2 | |
| 20 | 21.1 | 2.91 | 105.3 | |
| 50 | 49.4 | 5.03 | 98.7 | |
3.7 Bioimaging of NCA in living cells
H2S plays a crucial role in various physiological processes. To explore the potential of NCA for biological imaging applications, we systematically evaluated its ability to detect H2S in live cells. The cytotoxicity of NCA was first assessed in A549 cells using the CCK-8 assay. As shown in Fig. S7, cell viability remained above 80% when NCA concentrations ranged from 2.5 to 20 µM, indicating low toxicity at these levels. In contrast, concentrations above 40 µM led to a decline in cell viability, reaching 60% at 80 µM. Based on these findings, a concentration of 10 µM NCA was selected for subsequent imaging experiments, as it is well below the cytotoxicity threshold.To further assess the probe's performance, A549 cells were incubated with 10 µM NCA and imaged with an inverted fluorescence microscope. The cells exhibited a weak blue fluorescence signal (Fig. 4a–c), likely corresponding to the basal level of endogenous H2S. In biological systems, H2S is endogenously produced from cysteine (Cys) via the enzymatic actions of cystathionine β-synthase (CBS) and cystathionine γ-lyase (CSE). To evaluate NCA's response to endogenous H2S, cells were pretreated with 100 µM Cys, which resulted in a marked fluorescence enhancement (Fig. 4d–f), confirming that Cys stimulates H2S generation that is effectively detected by the probe. Additionally, when cells were exposed to increasing concentrations of exogenous H2S (via Na2S), the fluorescence intensity increased in a dose-dependent manner (Fig. 4g–l), demonstrating that NCA can reliably image both endogenous and exogenous H2S in live cells.
4 Conclusion
In summary, we developed a FRET-based fluorescent probe (NCA) that combines coumarin (CA) as a donor with nitrobenzoxadiazole (NB) as an acceptor and H2S-reactive site. NCA exhibits a 16-fold fluorescence enhancement with a low detection limit of 0.11 µM, along with rapid response and strong anti-interference capability. Its versatility is demonstrated across multiple application scenarios, including aqueous solutions, environmental water, gaseous H2S, and living cells, and further extended through integration with a smartphone-assisted test strip platform for cost-effective on-site analysis. Looking forward, in vivo imaging and benchmarking against commercial probes will be pursued to expand its applicability. Collectively, these features establish NCA as a robust and versatile tool for exploring H2S dynamics in both environmental monitoring and biological research.Author contributions
X. L. X.: conceptualization, investigation, formal analysis, writing – original draft. L. X.: investigation, methodology. L. W. M.: investigation, validation. X. Z. Y.: formal analysis. L. S. W.: conceptualization, supervision, writing – review & editing. F. G. D.: conceptualization, supervision, writing – review & editing.Conflicts of interest
The authors declare no competing interests.Data availability
Data will be made available on request.Supplementary information (SI): experimental procedures, synthesis of compounds, and additional spectroscopic and biological data. See DOI: https://doi.org/10.1039/d5ay01647a.
Acknowledgements
The authors would like to thank all those who contributed to this research.References
- Y. H. Chan, S. S. M. Lock, M. K. Wong, C. L. Yiin, A. C. M. Loy, K. W. Cheah, S. Y. W. Chai, C. Li, B. S. How, B. L. F. Chin, Z. P. Chan and S. S. Lam, Environ. Pollut., 2022, 314, 120219 CrossRef CAS PubMed.
- J. L. Obeso, D. R. Amaro, C. V. Flores, A. Gutiérrez-Alejandre, R. A. Peralta, C. Leyva and I. A. Ibarra, Coord. Chem. Rev., 2023, 485, 215135 CrossRef CAS.
- R. J. Lewis and G. B. Copley, Crit. Rev. Toxicol., 2015, 45, 93–123 CrossRef CAS PubMed.
- E. Panza, I. Bello, M. Smimmo, V. Brancaleone, E. Mitidieri, M. Bucci, G. Cirino, R. Sorrentino and R. d'Emmanuele di Villa Bianca, Biomed. Pharmacother., 2022, 151, 113137 CrossRef CAS PubMed.
- N. H. Khan, D. Wang, W. Wang, M. Shahid, S. Khattak, E. E. Ngowi, M. Sarfraz, X.-Y. Ji, C.-Y. Zhang and D.-D. Wu, Molecules, 2022, 27, 4049 CrossRef CAS PubMed.
- J. W. Elrod, J. W. Calvert, J. Morrison, J. E. Doeller, D. W. Kraus, L. Tao, X. Jiao, R. Scalia, L. Kiss, C. Szabo, H. Kimura, C. Chow and D. J. Lefer, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 15560–15565 CrossRef CAS PubMed.
- L. Cao, X. Cao, Y. Zhou, B. V. Nagpure, Z.-Y. Wu, L. F. Hu, Y. Yang, G. Sethi, P. K. Moore and J.-S. Bian, Brain Behav. Immun., 2018, 73, 603–614 CrossRef CAS PubMed.
- L. Chang, B. Geng, F. Yu, J. Zhao, H. Jiang, J. Du and C. Tang, Amino Acids, 2008, 34, 573–585 CrossRef CAS PubMed.
- S. Wang, Y. Chen and N. Chen, et al. , Cell Death Dis., 2017, 8, e2688 CrossRef CAS PubMed.
- G. Cirino, C. Szabo and A. Papapetropoulos, Physiol. Rev., 2023, 103, 31–276 CrossRef CAS PubMed.
- S. Arndt, C. D. Baeza-Garza, A. Logan, T. Rosa, R. Wedmann, T. A. Prime, J. L. Martin, K. Saeb-Parsy, T. Krieg, M. R. Filipovic, R. C. Hartley and M. P. Murphy, J. Biol. Chem., 2017, 292, 7761–7773 CrossRef CAS PubMed.
- L. E. Santos-Figueroa, C. de la Torre, S. El Sayed, F. Sancenón, R. Martínez-Máñez, A. M. Costero, S. Gil and M. Parra, Eur. J. Inorg. Chem., 2014, 2014, 41–45 CrossRef CAS.
- J. E. Doeller, T. S. Isbell, G. Benavides, J. Koenitzer, H. Patel, R. P. Patel, J. R. Lancaster, V. M. Darley-Usmar and D. W. Kraus, Anal. Biochem., 2005, 341, 40–51 CrossRef CAS PubMed.
- J. K. Fogo and M. Popowsky, Anal. Chem., 1949, 21, 732–734 CrossRef CAS.
- H. Chen, X. Li, P. Gao, Y. Pan and J. Liu, J. Mol. Struct., 2022, 1262, 133015 CrossRef CAS.
- Z. Xu, Y. Zheng, R. Qiao, S. Cao, Y. Fang, X. Bo, H. Zhu, C. Ying, Y. Sun and J. Zheng, Chem. Eng. J., 2023, 456, 141000 CrossRef CAS.
- H. Yan, F. Huo, Y. Yue, J. Chao and C. Yin, J. Am. Chem. Soc., 2021, 143, 318–325 CrossRef CAS PubMed.
- J. Yang, R. Zhang, Y. Zhao, J. Tian, S. Wang, C. P. Gros and H. Xu, Spectrochim. Acta, Part A, 2021, 248, 119199 CrossRef CAS PubMed.
- Y. Zhang, Y. Zhao, A. Zhou, Q. Qu, X. Zhang, B. Song, K. Liu, R. Xiong and C. Huang, Spectrochim. Acta, Part A, 2021, 261, 120014 CrossRef CAS PubMed.
- Y. Liu, Y. Ding, J. Huang, X. Zhang, T. Fang, Y. Zhang, X. Zheng and X. Yang, Dyes Pigm., 2017, 138, 112–118 CrossRef CAS.
- X. Yu, Y. Huang, N. Zhang, Q. Zan, X. Wang, Z. Jin, L. Fan, C. Dong and Y. Zhang, Anal. Chim. Acta, 2023, 1277, 341679 CrossRef CAS PubMed.
- Y. Ji, L.-J. Xia, L. Chen, X.-F. Guo, H. Wang and H.-J. Zhang, Talanta, 2018, 181, 104–111 CrossRef CAS PubMed.
- L. Ji, A. Fu, Y. Zhang, Y. Xu, Y. Xi, S. Cui, N. Gao, L. Yang, W. Shang, Z. Yang and G. He, Anal. Chim. Acta, 2025, 1334, 343425 CrossRef CAS.
- X. Tian, L. C. Murfin, L. Wu, S. E. Lewis and T. D. James, Chem. Sci., 2021, 12, 3406–3426 RSC.
- L. Wu, C. Huang, B. P. Emery, A. C. Sedgwick, S. D. Bull, X.-P. He, H. Tian, J. Yoon, J. L. Sessler and T. D. James, Chem. Soc. Rev., 2020, 49, 5110–5139 RSC.
- X. Jia, Q. Chen, Y. Yang, Y. Tang, R. Wang, Y. Xu, W. Zhu and X. Qian, J. Am. Chem. Soc., 2016, 138, 10778–10781 CrossRef CAS PubMed.
- P. Sontisiri, D. Promrug, D. Arthan, N. Choengchan and P. Thongyoo, Spectrochim. Acta, Part A, 2025, 326, 125170 CrossRef CAS.
- D. Liu, W. Hessler and M. Henary, Molecules, 2023, 28, 1295 CrossRef CAS PubMed.
- P. Liu, Y. Liu, H. Huang, G. Bai and Y. Peng, Spectrochim. Acta, Part A, 2023, 289, 122223 CrossRef CAS PubMed.
- L. He, B. Dong, Y. Liu and W. Lin, Chem. Soc. Rev., 2016, 45, 6449–6461 RSC.
- H. Fang, Y. Chen, Z. Jiang, W. He and Z. Guo, Acc. Chem. Res., 2023, 56, 258–269 CrossRef CAS PubMed.
- D. Cao, Z. Liu, P. Verwilst, S. Koo, P. Jangjili, J. S. Kim and W. Lin, Chem. Rev., 2019, 119, 10403–10519 CrossRef CAS PubMed.
- X. Sun, T. Liu, J. Sun and X. Wang, RSC Adv., 2020, 10, 10826–10847 RSC.
- L. Yang, F. Wang, X. Luo, X. Kong, Z. Sun and J. You, Talanta, 2020, 209, 120517 CrossRef CAS.
- Y. Zhang, Y. Chen, Y. Bai, X. Xue, W. He and Z. Guo, Analyst, 2020, 145, 4233–4238 RSC.
| This journal is © The Royal Society of Chemistry 2026 |