Open Access Article
Open Access ArticleSupramolecular assemblies of tetravalent terbium complex units: syntheses, structure, and materials properties†
Tianjiao
Xue
ab,
Qing-Song
Yang
ab,
Lei
Li
ab,
Xiao-Yong
Chang
a,
You-Song
Ding
 *ab and
Zhiping
Zheng
*ab and
Zhiping
Zheng
 *ab
*ab
aDepartment of Chemistry, Southern University of Science and Technology, Shenzhen, Guangdong 518055, China. E-mail: dingys@sustech.edu.cn; zhengzp@sustech.edu.cn
bKey Laboratory of Rare Earth Chemistry of Guangdong Higher Education Institutes, Southern University of Science and Technology, Shenzhen, Guangdong 518055, China
First published on 11th March 2025
Abstract
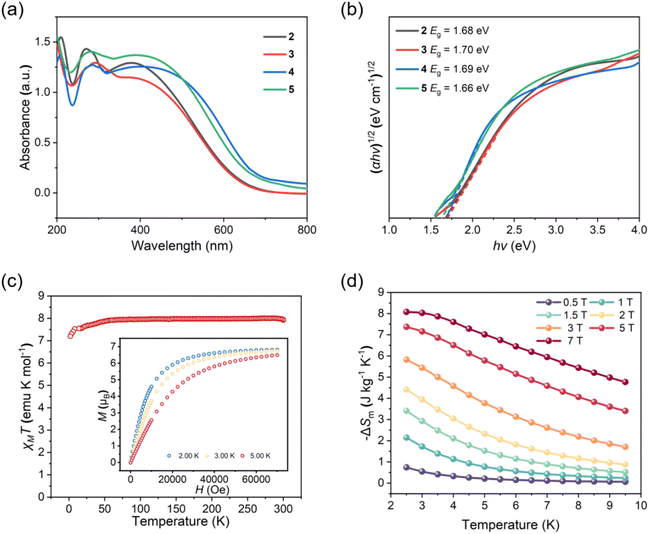
There is a growing interest in lanthanide complexes exhibiting unconventional oxidation states, primarily due to their unique electronic structures and accompanying physicochemical properties. Herein, likely the first examples of supramolecular assemblies of non-Ce(IV) tetravalent lanthanide complexes, with the general formula [Tb(OSiPh3)4Lx]n [1 (n = 2, L1 = 1,2-bis(4-pyridyl)ethane); 2 (L2 = 4,4′-bipyridine), 3 (L3 = 1,2-bis(4-pyridyl)acetylene), 4 (L4 = 1,2-bis(4-pyridyl)ethylene), and 5 (L5 = 1,4-bis(4-pyridyl)benzene)], are reported. Cyclic voltammetry studies show two successive redox events, indicating electronic interactions between the two Tb(IV) centers in the dimeric metallomacrocycle 1. Compounds 2–5 are zig-zag structured coordination polymers featuring complex units of Tb(OSiPh3)4 bridged by their respective pyridyl-based ditopic ligands. These tetravalent lanthanide species display impressive stability in air, which is believed to result from the stabilization effect of ligand Lx and the extensive multifarious interactions involving the aromatic rings of the anionic (Ph3SiO−) and bridging ligands. UV-vis absorption spectroscopic studies show that 2–5 are semiconducting, each with a narrow bandgap of ca. 1.7 eV. Magnetic property studies yielded magnetic entropy changes of ca. 8.0 J (kg K)−1 at 2.5 K and 7T, which is reasonable for a complex with high-molecular-weight ligands, suggesting the potential development of Tb(IV) complexes as molecular refrigerants due to their f7 electronic configuration.
Introduction
Complexes of metal ions in unconventional oxidation states are of fundamental scientific interest and practical significance due to their unique properties and potential materials applications.1,2 For the lanthanide (Ln) elements, chemistry has been dominated by trivalent ions. With a few exceptions, complexes of lanthanide ions in other oxidation states, specifically divalent and tetravalent, are notoriously sensitive to air and moisture, making their synthesis challenging. For example, most Ln(II) complexes are unstable at room temperature and should be kept at low temperatures and in glove boxes.3–39 Some Ln(II) complexes can even activate the C–H bonds (sp3) of the ligands or solvent molecules.7,14,18,19,25–27,34,39 However, thanks to the collective efforts of Evans, Schumann, Ziller, Protchenko, Junk, and others, complexes of all Ln(II) ions have been obtained and structurally characterized,4–10 some of which exhibit interesting luminescence,12,31,40–43 magnetic,11,22,28,32,36 or catalytic properties.21 In comparison, the analogous research on Ln(IV) has lagged far behind, except for the well-known Ce(IV), for which some exciting new chemistry44,45 and applications46–52 have been reported recently.Collectively responsible for the hiatus noted above are the scarcity of appropriate oxidants, the highly demanding synthetic procedures, and the lack of suitable ligands for the stabilization of high-valence lanthanide ions. The extreme sensitivity of such complexes to air and moisture renders their characterization and property studies difficult; there is an inherently high propensity for their decomposition in solution, including reduction by solvent molecules.53–64 Thus so far, only a handful of Tb(IV) complexes exist and an even smaller number of complexes of Pr(IV) (Table S1†),53–64 with only one complex of Pr(V) having being recently reported.65
Recently, we have made efforts to enhance the stability of complexes of Tb(IV) and Pr(IV) by replacing the coordinated solvent molecules in the benchmarking complex Tb(OSiPh3)4S2 (S = MeCN or THF) with 1,2-dimethoxyethane (DME) and several pyridyl-based chelating ligands.62,63 Surprisingly, the DME chelate is only marginally more stable than its THF congener, while the use of the latter group of ligands leads to markedly enhanced stability of the corresponding chelates. Experimental and computational studies indicate that the pyridyl (π)-to-Tb(IV) (5d) donation and the intramolecular π–π interactions between the aromatic rings of OSiPh3 and these ancillary ligands are primarily responsible for the enhanced stability observed. These results indicate that the contribution of a ligand being merely chelating is insignificant. A corollary is that one may use almost any pyridyl-based ligand, chelating or not, to realize a great variety of Ln(IV) complexes for property studies and application exploration.
With this recognition, we set out to prepare supramolecular assemblies featuring complex units of Tb(IV) bridged by ditopic pyridyl-based ligands (L1–L5) and to explore the possible interactions between the individual complex units and the resulting properties. These ligands include (1,2-bis(4-pyridyl)ethane) (L1); 4,4′-bipyridine (L2); 1,2-bis(4-pyridyl)ethylene (L3); 1,2-bis(4-pyridyl)acetylene (L4), and 1,4-bis(4-pyridyl)benzene (L5). A straightforward replacement of MeCN in Tb(OSiPh3)4(MeCN)2 with Lx affords a cyclic dimeric complex and four coordination polymers of the general formula [Tb(OSiPh3)4Lx]n [1(L1, n = 2); 2(L2), 3(L3), 4(L4), and 5(L5)]. Compounds 1–5 are likely the first polynuclear complexes of a non-Ce(IV) tetravalent lanthanide ion,66 for which we carried out magnetic property studies originating from the f7 electronic configuration of the Tb(IV) center.
Results and discussion
Syntheses and crystallographic studies
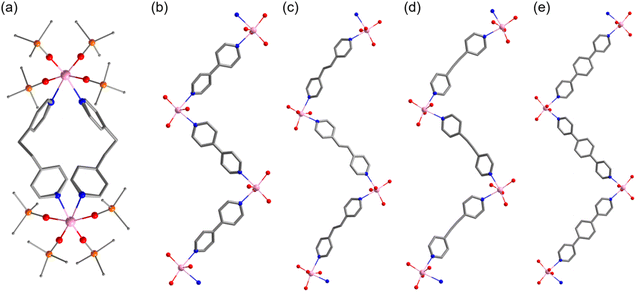
The title complexes were obtained by the ligand exchange reaction of Tb(OSiPh3)4(MeCN)2 with the bridging ligands (L1–L5). Different from the literature synthesis, the tetravalent MeCN solvate was obtained by oxidizing the trivalent precursor complex Tb(OSiPh3)3(THF)3 with thianthrene tetrafluoroborate in an acetonitrile solution of KOSiPh3 (Scheme 1); a profound color change of the reaction mixture from pale-yellow green to orange-red was observed as the reaction progressed.67,68 Compared with the previously used oxidant [N(C6H4Br)3][SbCl6], thianthrene tetrafluoroborate offers several advantages, including its ease of synthesis and use, low cost, and straightforward isolation of the solvate product by filtration from the reduction byproduct (thianthrene).69 All the crystals are intensely colored, with dark red for 1, orange for 2, and red for 3–5. This red-orange hue is consistent with previously reported Tb(IV) complexes.53–55,61–63Crystallographic studies (Table S2†) revealed the dimeric cyclic structure of 1 featuring two complex units of Tb(OSiPh3)4 bridged by two L1 ligands (Fig. 1a). The hexacoordinate Tb atom displays a roughly octahedral coordination sphere formed by four O (Ph3SiO−) atoms and two pyridyl N atoms of the cis-disposed L1 ligands, each adopting a gauche–gauche conformation to facilitate the formation of the metallomacrocycle.70–73 With the more rigid L2–L5, zigzag-chain structured coordination polymers 2–5 were obtained (Fig. 1b–e). The distortion of the coordination geometry from a perfect octahedron in these polynuclear Tb(IV) complexes is estimated by continuous shape measures analysis74 to be 0.566, 1.113–1.148, 0.849–0.940, 0.798–0.920, and 0.675 for 1–5, respectively (Table S3†). The Tb(IV)–O bonds, ranging from 2.024(5) to 2.090(3) Å (Table S4†), are comparable to those of previously reported Tb(IV) complexes but significantly shorter than their Tb(III)-OSiPh3 counterparts. The N–Tb–N angles range from 79.58(14)° to 89.4(5)° in 1–5, which is in good agreement with previously reported Tb(IV) complexes.53–55,61–63 The two Tb atoms in the uniquely structured 1 are separated by a distance of 10.446 Å, while the neighboring Tb atoms within the polymeric chains are separated by 12.054, 14.083, 14.207, and 16.320 Å, respectively in 2–5, in approximate accordance with the increase in the size of the different bridging ligands.
In our recently reported Tb(IV) chelates with pyridyl-based ligands, the intramolecular π–π and C–H⋯π interactions between the aromatic rings of Ph3SiO− and the chelating ligands are believed to be critically important in improving the stability of the chelates. Similar interactions are identified in the discrete dimeric complex 1 (Fig. S1 and Table S5†).62,63 For the coordination polymers (2–5), such interactions, in addition to the ones within each of the complex units, exist copiously between individual polymer chains (Fig. S2–S4†). The extensive non-covalent interactions within the complex building unit and between the polymeric chains can be appreciated from the crystal packing of 5 (Fig. 2) – a representative of the coordination polymers. Within the complex unit, one or two phenyl groups of the Ph3SiO− ligands are disposed in such a way that strong face-to-face π–π interactions between the pyridyl ligands are present (green-colored fragments, Fig. 2). Corroborating such interactions is the pronounced deviation of the axial coordination motif from linearity (151.44(16)° to 159.03(15)°, Table S4†). Moreover, inter-chain interactions, mostly of the C–H⋯π type, exist amply between the Ph3SiO− phenyl groups (plum-colored fragments, Fig. 2). The same type of interactions are also found between the Ph3SiO− phenyl groups in one chain and the central aromatic ring of L5 in neighboring chains (black-colored fragments, Fig. 2). The extent of such inter-chain interactions, up to 27% of the total non-covalent interactions, is verified by Hirshfeld surface analysis (Fig. S5–S26†).75 Although individually weak, such interactions collectively contribute to the stability observed for many supramolecular systems. For example, extensive π-interactions have been found to stabilize the organization of porphyrin and m-xylylene panels, affording the robust supramolecular constructs of π-Diamond.76,77 In the present case, the extensive C–H⋯π interactions bring together individual polymeric chains into an intimate arrangement, resulting in significant shielding of the Tb(IV) centers and the enhanced stability observed (vide infra).
The four coordination polymers (2–5) display impressive stability when exposed to air (Fig. 3 and S27–S29†). Against the simulated pattern, the powder X-ray diffraction (PXRD) pattern of a pristine sample of 2 is shown in Fig. 3, together with the ones obtained after the sample was exposed to air for 24 and 48 hours. The solid retained its crystallinity over this extended period of air exposure, and more importantly, the recovered solids showed essentially the same PXRD patterns as the pristine sample. This air-stability facilitates sample handling and any property studies of these Tb(IV) complexes.
 | ||
| Fig. 3 Power X-ray diffraction patterns for a sample of 2 (pristine and with air exposure for 24 and 48 hours) against the one simulated based on its single-crystal structure. | ||
The Tb(IV) ion, with a half-filled 4f7 electronic configuration, is isoelectronic Eu(II) and Gd(III) ions. Temperature- and field-dependent magnetic susceptibility measurements were conducted for these polymeric species. The results for the representative 3 are shown in Fig. 5c, and those for the other polymeric species are provided in Fig. S30–S32.† The χT values at 300 K are 7.90 (2), 7.83 (3), 7.84 (4), and 7.94 cm3 K mol−1 (5), in good agreement with the value calculated for mononuclear 4f7-complexes.53–55,57,58,60–62,81 The χT value decreases slowly as the temperature is lowered to ca. 20 K, where a sudden drop occurs, reaching a minimum of 6.50 cm3 K mol−1. The drop in the low-temperature region is indicative of varied zero-field splitting.60–62,81 Field-dependent magnetization measurements were subsequently performed at low temperatures with the field up to a maximum of 7T (inset in Fig. 5c for 3). The maximum magnetization values at 2 K and 7T are 6.52 (2), 6.82 (3), 6.54 (4), and 6.47 μB (5), which are close to the saturation magnetization value of 7 μB calculated for an ion with the 4f7 electronic configuration.53–55,57,58,60–62,81 With the same large ground-state spin of 7/2 and magnetic isotropy as Gd(III) and Eu(II) complexes, it is entirely reasonable to explore the potential of Tb(IV) complexes as molecule-based magnetic refrigerants.82,83 The magnetic entropy changes (−ΔSm) for the representative 3 were calculated by applying Maxwell's equation using the low-temperature field-dependent magnetization values (Fig. 5d), producing a maximum value of 8.07 J (kg K)−1 at 2.5 K and 7T. This experimentally obtained −ΔSm is smaller than the theoretical value of 10.15 J (kg K)−1 (−ΔSm = R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2S + 1); R is the gas constant and S is the state spin number). Comparable magnetic entropies were obtained for the other coordination polymers, as they closely resemble 3, both in structure and composition. However, due to the significantly higher molecular weights of the complex building unit of [Tb(OSiPh3)4Lx] (Lx = L2–L5),84 the magnetocaloric effects displayed by the present Tb(IV) complexes are noticeably smaller than those of Gd(III) or Eu(II) complexes.82,83 Possible enhancement in this regard may be achieved by increasing the mass percentage of Tb(IV) in its complexes with lower-molecular-weight ligands.
ln(2S + 1); R is the gas constant and S is the state spin number). Comparable magnetic entropies were obtained for the other coordination polymers, as they closely resemble 3, both in structure and composition. However, due to the significantly higher molecular weights of the complex building unit of [Tb(OSiPh3)4Lx] (Lx = L2–L5),84 the magnetocaloric effects displayed by the present Tb(IV) complexes are noticeably smaller than those of Gd(III) or Eu(II) complexes.82,83 Possible enhancement in this regard may be achieved by increasing the mass percentage of Tb(IV) in its complexes with lower-molecular-weight ligands.
Conclusions
In this work, we report what is likely the first supramolecular assemblies composed of complex units of non-Ce(IV) tetravalent lanthanide ions. A total of four coordination polymers have been obtained with the largely rigid dipyridyl bridging ligands, while the use of a more flexible ditopic ligand afforded a dimeric metallamacrocycle. Cyclic voltammetry studies revealed significant electronic interactions between the Tb(IV) centers within the macrocycle, highlighting the rich redox chemistry enabled by uniquely structured tetravalent lanthanide complexes. The four coordination polymers survived extended air-exposure, due to a combination of the ligand stabilizing effect and the extensive intra- and inter-chain non-covalent interactions that involve the aromatic rings of both the anionic ligands in the complex unit and the bridging ligands. Physical property studies show that the coordination polymers possess optical band gaps comparable to those of some narrow bandgap semiconductors. Thanks to the f7 electronic configuration of Tb(IV), we propose that it is possible to design unprecedented molecule-based magnetic semiconductors or magnetic refrigerants.Data availability
The data supporting this article have been included as part of the ESI.†Author contributions
TJX synthesized and characterized the compounds with the assistance of QSY and LL. TJX and XYC assisted in the X-ray crystallographic analyses. ZPZ and YSD designed the research project and directed the experiments. The manuscript was written with contributions from all authors, who have also approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors gratefully acknowledge the financial support for this work by the National Natural Science Foundation of China (92261203, 22101116, and 21971106), the Key Laboratory of Rare Earth Chemistry of Guangdong Higher Education Institutes (2022KSYS006), the Stable Support Plan Program of Shenzhen Natural Science Fund (20200925161141006), and the Shenzhen Fundamental Research Program (JCYJ20220530115001002 and JCYJ20220818100417037).Notes and references
- J. C. Wedal and W. J. Evans, J. Am. Chem. Soc., 2021, 143, 18354–18367 CrossRef CAS PubMed.
- Y. Wang and W. Huang, Sci. China Chem., 2020, 50, 1504–1559 Search PubMed.
- W. J. Evans, N. T. Allen and J. W. Ziller, J. Am. Chem. Soc., 2001, 123, 7927–7928 CrossRef CAS PubMed.
- M. N. Bochkarev, I. L. Fedushkin, S. Dechert, A. A. Fagin and H. Schumann, Angew. Chem., Int. Ed., 2001, 40, 3176–3178 CrossRef CAS PubMed.
- W. J. Evans, N. T. Allen and J. W. Ziller, Angew. Chem., Int. Ed., 2002, 41, 359–361 CrossRef CAS PubMed.
- P. B. Hitchcock, M. F. Lappert, L. Maron and A. V. Protchenko, Angew. Chem., Int. Ed., 2008, 47, 1488–1491 CrossRef CAS PubMed.
- F. Jaroschik, A. Momin, F. Nief, X.-F. Le Goff, G. B. Deacon and P. C. Junk, Angew. Chem., Int. Ed., 2009, 48, 1117–1121 CrossRef CAS PubMed.
- M. R. MacDonald, J. W. Ziller and W. J. Evans, J. Am. Chem. Soc., 2011, 133, 15914–15917 CrossRef CAS PubMed.
- M. R. MacDonald, J. E. Bates, M. E. Fieser, J. W. Ziller, F. Furche and W. J. Evans, J. Am. Chem. Soc., 2012, 134, 8420–8423 CrossRef CAS PubMed.
- M. R. MacDonald, J. E. Bates, J. W. Ziller, F. Furche and W. J. Evans, J. Am. Chem. Soc., 2013, 135, 9857–9868 CrossRef CAS PubMed.
- K. R. Meihaus, M. E. Fieser, J. F. Corbey, W. J. Evans and J. R. Long, J. Am. Chem. Soc., 2015, 137, 9855–9860 CrossRef CAS PubMed.
- A. N. W. Kuda-Wedagedara, C. Wang, P. D. Martin and M. J. Allen, J. Am. Chem. Soc., 2015, 137, 4960–4963 CrossRef CAS PubMed.
- M. E. Fieser, M. R. MacDonald, B. T. Krull, J. E. Bates, J. W. Ziller, F. Furche and W. J. Evans, J. Am. Chem. Soc., 2015, 137, 369–382 CrossRef CAS PubMed.
- J. F. Corbey, D. H. Woen, C. T. Palumbo, M. E. Fieser, J. W. Ziller, F. Furche and W. J. Evans, Organometallics, 2015, 34, 3909–3921 CrossRef CAS.
- L. A. Ekanger, D. R. Mills, M. M. Ali, L. A. Polin, Y. Shen, E. M. Haacke and M. J. Allen, Inorg. Chem., 2016, 55, 9981–9988 CrossRef CAS PubMed.
- M. Xémard, A. Jaoul, M. Cordier, F. Molton, O. Cador, B. Le Guennic, C. Duboc, O. Maury, C. Clavaguéra and G. Nocton, Angew. Chem., Int. Ed., 2017, 56, 4266–4271 CrossRef PubMed.
- R. P. Kelly, L. Maron, R. Scopelliti and M. Mazzanti, Angew. Chem., Int. Ed., 2017, 56, 15663–15666 CrossRef CAS PubMed.
- M. E. Fieser, C. T. Palumbo, H. S. La Pierre, D. P. Halter, V. K. Voora, J. W. Ziller, F. Furche, K. Meyer and W. J. Evans, Chem. Sci., 2017, 8, 7424–7433 RSC.
- C. T. Palumbo, D. P. Halter, V. K. Voora, G. P. Chen, J. W. Ziller, M. Gembicky, A. L. Rheingold, F. Furche, K. Meyer and W. J. Evans, Inorg. Chem., 2018, 57, 12876–12884 CrossRef CAS PubMed.
- D. P. Halter, C. T. Palumbo, J. W. Ziller, M. Gembicky, A. L. Rheingold, W. J. Evans and K. Meyer, J. Am. Chem. Soc., 2018, 140, 2587–2594 CrossRef CAS PubMed.
- F. D. White, C. Celis-Barros, J. Rankin, E. Solís-Céspedes, D. Dan, A. N. Gaiser, Y. Zhou, J. Colangelo, D. Páez-Hernández, R. Arratia-Pérez and T. E. Albrecht-Schmitt, Inorg. Chem., 2019, 58, 3457–3465 CrossRef CAS PubMed.
- C. A. Gould, K. R. McClain, J. M. Yu, T. J. Groshens, F. Furche, B. G. Harvey and J. R. Long, J. Am. Chem. Soc., 2019, 141, 12967–12973 CrossRef CAS PubMed.
- D. N. Huh, S. R. Ciccone, S. Bekoe, S. Roy, J. W. Ziller, F. Furche and W. J. Evans, Angew. Chem., Int. Ed., 2020, 59, 16141–16146 CrossRef CAS PubMed.
- S. A. Moehring, M. Miehlich, C. J. Hoerger, K. Meyer, J. W. Ziller and W. J. Evans, Inorg. Chem., 2020, 59, 3207–3214 CrossRef CAS PubMed.
- A. J. Ryan, J. W. Ziller and W. J. Evans, Chem. Sci., 2020, 11, 2006–2014 RSC.
- T. F. Jenkins, S. Bekoe, J. W. Ziller, F. Furche and W. J. Evans, Organometallics, 2021, 40, 3917–3925 CrossRef CAS.
- A. B. Chung, A. J. Ryan, M. Fang, J. W. Ziller and W. J. Evans, Inorg. Chem., 2021, 60, 15635–15645 CrossRef CAS PubMed.
- J. Moutet, J. Schleinitz, L. La Droitte, M. Tricoire, F. Pointillart, F. Gendron, T. Simler, C. Clavaguéra, B. Le Guennic, O. Cador and G. Nocton, Angew. Chem., Int. Ed., 2021, 60, 6042–6046 CrossRef CAS PubMed.
- D. N. Huh, J. P. Bruce, S. Ganesh Balasubramani, S. R. Ciccone, F. Furche, J. C. Hemminger and W. J. Evans, J. Am. Chem. Soc., 2021, 143, 16610–16620 CrossRef CAS PubMed.
- A. B. Chung, D. Rappoport, J. W. Ziller, R. E. Cramer, F. Furche and W. J. Evans, J. Am. Chem. Soc., 2022, 144, 17064–17074 CrossRef CAS PubMed.
- R. M. Diaz-Rodriguez, D. A. Gálico, D. Chartrand, E. A. Suturina and M. Murugesu, J. Am. Chem. Soc., 2022, 144, 912–921 CrossRef CAS PubMed.
- K. Kundu, J. R. K. White, S. A. Moehring, J. M. Yu, J. W. Ziller, F. Furche, W. J. Evans and S. Hill, Nat. Chem., 2022, 14, 392–397 CrossRef CAS PubMed.
- L. M. Anderson-Sanchez, J. M. Yu, J. W. Ziller, F. Furche and W. J. Evans, Inorg. Chem., 2023, 62, 706–714 CrossRef CAS PubMed.
- C. R. Stennett, J. Q. Nguyen, J. W. Ziller and W. J. Evans, Organometallics, 2023, 42, 696–707 CrossRef CAS.
- W. N. G. Moore, T. J. McSorley, A. Vincent, J. W. Ziller, L. A. Jauregui and W. J. Evans, ACS Appl. Nano Mater., 2023, 6, 6461–6466 CrossRef CAS.
- P.-B. Jin, Q.-C. Luo, G. K. Gransbury, I. J. Vitorica-Yrezabal, T. Hajdu, I. Strashnov, E. J. L. McInnes, R. E. P. Winpenny, N. F. Chilton, D. P. Mills and Y.-Z. Zheng, J. Am. Chem. Soc., 2023, 145, 27993–28009 CrossRef CAS PubMed.
- P. W. Smith, J. Hrubý, W. J. Evans, S. Hill and S. G. Minasian, J. Am. Chem. Soc., 2024, 146, 5781–5785 CrossRef CAS PubMed.
- D. Errulat, K. L. M. Harriman, D. A. Gálico, E. V. Salerno, J. van Tol, A. Mansikkamäki, M. Rouzières, S. Hill, R. Clérac and M. Murugesu, Nat. Commun., 2024, 15, 3010 CrossRef CAS PubMed.
- J. D. Queen, L. M. Anderson-Sanchez, C. R. Stennett, A. Rajabi, J. W. Ziller, F. Furche and W. J. Evans, J. Am. Chem. Soc., 2024, 146, 3279–3292 CrossRef CAS PubMed.
- G. Zhan, L. Wang, Z. Zhao, P. Fang, Z. Bian and Z. Liu, Angew. Chem., Int. Ed., 2020, 59, 19011–19015 CrossRef CAS PubMed.
- W. Yan, T. Li, Z. Cai, H. Qi, R. Guo, P. Huo, Z. Liu and Z. Bian, Inorg. Chem. Front., 2022, 9, 4794–4800 RSC.
- W. Yan, Z. Zhao, G. Yu, H. Qi, R. Guo, N. Zheng, Z. Bian and Z. Liu, Sci. China Chem., 2023, 66, 1750–1757 CrossRef CAS.
- R. M. Diaz-Rodriguez, D. A. Gálico, D. Chartrand and M. Murugesu, J. Am. Chem. Soc., 2024, 146, 34118–34129 CrossRef CAS PubMed.
- Y. Wang, J. Liang, C. Deng, R. Sun, P.-X. Fu, B.-W. Wang, S. Gao and W. Huang, J. Am. Chem. Soc., 2023, 145, 22466–22474 CrossRef CAS PubMed.
- Y. Guo, X.-L. Jiang, Q.-Y. Wu, K. Liu, W. Wang, K.-Q. Hu, L. Mei, Z.-F. Chai, J. K. Gibson, J.-P. Yu, J. Li and W.-Q. Shi, J. Am. Chem. Soc., 2024, 146, 7088–7096 CrossRef CAS PubMed.
- Y. Qiao and E. J. Schelter, Acc. Chem. Res., 2018, 51, 2926–2936 CrossRef CAS PubMed.
- A. Hu, J.-J. Guo, H. Pan and Z. Zuo, Science, 2018, 361, 668–672 CrossRef CAS PubMed.
- Q. Yang, Y.-H. Wang, Y. Qiao, M. Gau, P. J. Carroll, P. J. Walsh and E. J. Schelter, Science, 2021, 372, 847–852 CrossRef CAS PubMed.
- G. B. Panetti, D.-C. Sergentu, M. R. Gau, P. J. Carroll, J. Autschbach, P. J. Walsh and E. J. Schelter, Nat. Commun., 2021, 12, 1713 CrossRef CAS PubMed.
- Q. An, Y.-Y. Xing, R. Pu, M. Jia, Y. Chen, A. Hu, S.-Q. Zhang, N. Yu, J. Du, Y. Zhang, J. Chen, W. Liu, X. Hong and Z. Zuo, J. Am. Chem. Soc., 2023, 145, 359–376 CrossRef CAS PubMed.
- H. Tateyama, A. C. Boggiano, C. Liao, K. S. Otte, X. Li and H. S. La Pierre, J. Am. Chem. Soc., 2024, 146, 10268–10273 CrossRef CAS PubMed.
- Q. An, L. Chang, H. Pan and Z. Zuo, Acc. Chem. Res., 2024, 57, 2915–2927 CrossRef CAS PubMed.
- C. T. Palumbo, I. Zivkovic, R. Scopelliti and M. Mazzanti, J. Am. Chem. Soc., 2019, 141, 9827–9831 CrossRef CAS PubMed.
- A. R. Willauer, C. T. Palumbo, R. Scopelliti, I. Zivkovic, I. Douair, L. Maron and M. Mazzanti, Angew. Chem., Int. Ed., 2020, 59, 3549–3553 CrossRef CAS PubMed.
- A. R. Willauer, I. Douair, A.-S. Chauvin, F. Fadaei-Tirani, J.-C. G. Bünzli, L. Maron and M. Mazzanti, Chem. Sci., 2022, 13, 681–691 RSC.
- A. R. Willauer, C. T. Palumbo, F. Fadaei-Tirani, I. Zivkovic, I. Douair, L. Maron and M. Mazzanti, J. Am. Chem. Soc., 2020, 142, 5538–5542 CrossRef CAS PubMed.
- N. T. Rice, I. A. Popov, D. R. Russo, J. Bacsa, E. R. Batista, P. Yang, J. Telser and H. S. La Pierre, J. Am. Chem. Soc., 2019, 141, 13222–13233 CrossRef CAS PubMed.
- N. T. Rice, I. A. Popov, D. R. Russo, T. P. Gompa, A. Ramanathan, J. Bacsa, E. R. Batista, P. Yang and H. S. La Pierre, Chem. Sci., 2020, 11, 6149–6159 RSC.
- N. T. Rice, I. A. Popov, R. K. Carlson, S. M. Greer, A. C. Boggiano, B. W. Stein, J. Bacsa, E. R. Batista, P. Yang and H. S. La Pierre, Dalton Trans., 2022, 51, 6696–6706 RSC.
- T. P. Gompa, S. M. Greer, N. T. Rice, N. Jiang, J. Telser, A. Ozarowski, B. W. Stein and H. S. La Pierre, Inorg. Chem., 2021, 60, 9064–9073 CrossRef CAS PubMed.
- M. Tricoire, F.-C. Hsueh, M. Keener, T. Rajeshkumar, R. Scopelliti, I. Zivkovic, L. Maron and M. Mazzanti, Chem. Sci., 2024, 15, 6874–6883 RSC.
- T. Xue, Y.-S. Ding, X.-L. Jiang, L. Tao, J. Li and Z. Zheng, Precis. Chem., 2023, 1, 583–591 CrossRef CAS PubMed.
- T. Xue, Y.-S. Ding and Z. Zheng, Dalton Trans., 2024, 53, 5779–5783 RSC.
- A. C. Boggiano, S. R. Chowdhury, M. D. Roy, M. G. Bernbeck, S. M. Greer, B. Vlaisavljevich and H. S. La Pierre, Angew. Chem., Int. Ed., 2024, 63, e202409789 CAS.
- A. Boggiano, C. Studvick, S. Roy Chowdhury, J. Niklas, H. Tateyama, H. Wu, et al., Praseodymium in the Formal +5 Oxidation State, ChemRxiv, 2024, preprint, DOI:10.26434/chemrxiv-2024-cb3zj, This content is a preprint and has not been peer-reviewed.
- Z. Guo, G. B. Deacon and P. C. Junk, J. Coord. Chem., 2024, 1–6 Search PubMed.
- M. J. McGeary, P. S. Coan, K. Folting, W. E. Streib and K. G. Caulton, Inorg. Chem., 1991, 30, 1723–1735 CrossRef CAS.
- B. Boduszek and H. J. Shine, J. Org. Chem., 1988, 53, 5142–5143 CrossRef CAS.
- N. G. Connelly and W. E. Geiger, Chem. Rev., 1996, 96, 877–910 CrossRef CAS PubMed.
- W.-H. Zhang, Y.-L. Song, Z.-H. Wei, L.-L. Li, Y.-J. Huang, Y. Zhang and J.-P. Lang, Inorg. Chem., 2008, 47, 5332–5346 CrossRef CAS PubMed.
- V. Chandrasekhar, T. Hajra, J. K. Bera, S. M. W. Rahaman, N. Satumtira, O. Elbjeirami and M. A. Omary, Inorg. Chem., 2012, 51, 1319–1329 CrossRef CAS PubMed.
- W. Zheng, S.-T. Jiang, B. Jiang and H.-B. Yang, in Metallomacrocycles: From Structures to Applications, ed. H.-B. Yang, The Royal Society of Chemistry, 2018, pp. 1–19, 10.1039/9781788013123-00001.
- G. J. Douglas, E. Richards and S. Sproules, Chem. Commun., 2025, 61, 685–688 RSC.
- S. Alvarez, D. Avnir, M. Llunell and M. Pinsky, New J. Chem., 2002, 26, 996–1009 RSC.
- P. R. Spackman, M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, D. Jayatilaka and M. A. Spackman, J. Appl. Crystallogr., 2021, 54, 1006–1011 CrossRef CAS PubMed.
- K. Liang, Y. Liang, M. Tang, J. Liu, Z.-B. Tang and Z. Liu, Angew. Chem., Int. Ed., 2024, 63, e202409507 CrossRef CAS PubMed.
- P. Harding and D. J. Harding, Trends Chem., 2024, 6, 575–576 CrossRef CAS.
- P. Makuła, M. Pacia and W. Macyk, J. Phys. Chem. Lett., 2018, 9, 6814–6817 CrossRef PubMed.
- Y. H. Lee, Science, 2023, 382, eadl0823 CrossRef PubMed.
- L. Li, Y.-S. Ding and Z. Zheng, Angew. Chem., Int. Ed., 2024, 63, e202410019 CrossRef CAS PubMed.
- J. B. Petersen, Y.-S. Ding, S. Gupta, A. Borah, E. J. L. McInnes, Y.-Z. Zheng, R. Murugavel and R. E. P. Winpenny, Inorg. Chem., 2023, 62, 8435–8441 CrossRef CAS PubMed.
- B. Wang, X. Liu, F. Hu, J.-t. Wang, J. Xiang, P. Sun, J. Wang, J. Sun, T. Zhao, Z. Mo, J. Shen, Y. Chen, Q. Huang and B. Shen, J. Am. Chem. Soc., 2024, 146, 35016–35022 CrossRef CAS PubMed.
- Q. Xu, B. Liu, M. Ye, G. Zhuang, L. Long and L. Zheng, J. Am. Chem. Soc., 2022, 144, 13787–13793 CrossRef CAS PubMed.
- Y.-S. Ding and Y.-Z. Zheng, J. Rare Earths, 2021, 39, 1554–1559 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Materials and methods, synthesis, detailed crystallographic data and characterization, figures S1–S32, and tables S1–S10. CCDC 2410273 (1), 2337622 (2), 2351767 (3), 2351768 (4), and 2337623 (5). For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4sc08731c |
| This journal is © The Royal Society of Chemistry 2025 |