Unique catalytic role of intermolecular electric fields that emanate from Lewis acids in a ring closing carbonyl olefin metathesis reaction†
Lopita
Swain
 ,
Karthik
Gopakumar
,
Karthik
Gopakumar
 and
Rajeev
Ramanan
and
Rajeev
Ramanan
 *
*
Department of Chemistry, National Institute of Technology Rourkela, Odisha 769008, India. E-mail: rrajeev@nitrkl.ac.in
First published on 15th February 2025
Abstract
Electric field (EF) catalysis has evolved as an effective tool for controlling reactivity and selectivity of reactions. While EF catalysis brings precise control over reactivity, it also challenges the concept's practical realization due to the difficulties in juxtaposing reactants with directional EF. The present density functional theory (DFT) studies demonstrate the catalytic role of the inherent intermolecular EFs that originate from Lewis acids (LA) during a ring-closing carbonyl–olefin metathesis (RCCOM) reaction. The specificity of LA coordination to reactants generates specifically oriented intermolecular EF components along the reaction axis which is defined parallel to the direction of flow of electrons wherein the influence of the EF would be at maximum. By examining the thermal [2+2] cycloaddition and carbonyl–ene reaction steps in a RCCOM reaction as model systems, the results revealed the pivotal role of intermolecular EF in mixing some of the dormant ionic structures into normal covalent structures and facilitating a partial rotation of the nonbonding orbitals at the carbonyl oxygen to enhance an ionic pseudo-pericyclic pathway. The unique role of intermolecular EF is further verified by modelling the pristine reaction, in the absence of LAs, under oriented external EFs. The conspicuous intermolecular EF component adds to other modes of catalysis, such as conventional Lewis acidity, to result in the gross catalytic effect. The findings offer insights into the practical realization of EF catalysis by harnessing the intermolecular EFs and point out the need to include intermolecular EF as an inevitable factor for a holistic explanation of any catalytic mechanism.
1. Introduction
Oriented external electric fields (EFs) are future smart reagents and invisible catalysts for chemical reactions.1 The mechanism of oriented external EFs acting as an invisible catalyst in chemical reactions has been explained by theory and demonstrated by experiments.2–12 Oriented external EF as a stimulus imparts changes in structure, bonding, electronic states, and the reaction outcome.2 The details on the impact of an oriented external EF on chemical reactions such as the Diels–Alder reaction,7,8 C–H bond activation,9 methyl transfer,10 disproportionation of methanol into methane and formaldehyde,11 nucleophilic substitution,12 electrophilic aromatic substitution,13 modulating entropy,14 and pH effects15 in water, oxidative addition,16 and catalyst free hydrogen synthesis17 were reported. EF can selectively polarize molecules, modify solvation dynamics, and stabilize reactive intermediates, improving catalytic efficiency. EF-induced electro-freezing can also restrict the dynamics of the hydrogen bond network in water and result in a ferroelectric amorphous phase.18 Recent advancements in utilizing the inherent internal EFs in water microdroplet chemistry have generated a novel approach to harness EF catalysis in chemical reactions.19–24 The naturally occurring high EFs at the peripheries of water microdroplets serve as a highly efficient catalyst for a range of reactions such as proton transfer,25 halogen-bond breaking,20 nucleophilic addition,21 alkylation,22 photolysis of H2O2,23 methane oxidation24 and cross-coupling reactions between C–H and N–H bonds.26 However, some reports suggest that Diels–Alder reactions in water microdroplets which are driven by rapid evaporation and reagent enrichment have minor EF contribution towards the gross catalysis.27 Nature utilizes a preorganised intrinsic EF within enzymes to bring efficient catalysis.6 Tailoring charged functional groups on the porphyrin ring was effective in catalysing hydrogen atom transfer reactivity of the ferryl [Fe(IV)-oxo] in metalloenzyme analogues due to the generation of an oriented intrinsic EF.28,29 As such, catalysis that utilizes the intrinsic EF is a promising area and it opens up a new avenue for the practical use of EF catalysis.The present study provides a comprehensive analysis of the role of intermolecular EF emanating from a Lewis acid (LA) in a [2+2] cycloaddition as well as in the competitive carbonyl–ene reaction that occurs concurrently in a ring-closing carbonyl–olefin metathesis (RCCOM) reaction. In the following discussion, we quantified the intermolecular EF generated by two LA catalysts, [AlCl2]+[SbF6]− and its neutral counterpart [AlCl3], in the reaction and subsequently demonstrated the catalytic role of the intermolecular EF. The intermolecular EFs showed unique catalytic activity by enhancing the conventional LA catalysis as well as stabilising some of the dormant ionic structures involved in a pseudo-pericyclic pathway.2,30
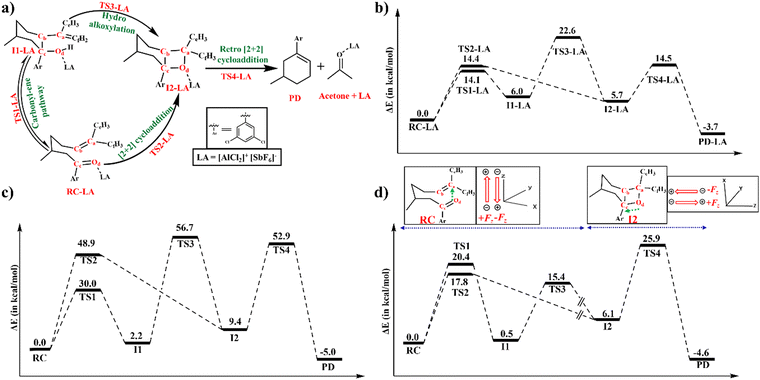
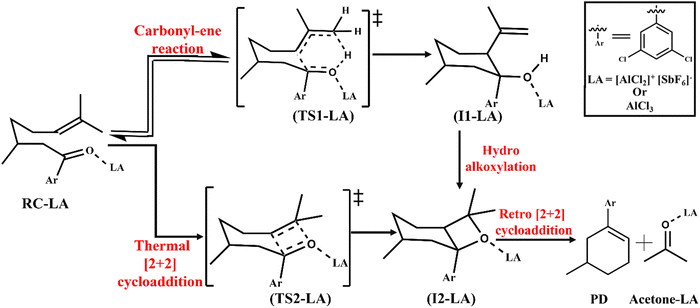
The LA catalysed RCCOM proceeds via two pathways under thermal conditions as shown in Scheme 1. The other most used LAs include GaCl3,31 FeCl3,32 tropylium cations,33 Brønsted acids,34 and various other organic reagents.35Scheme 1 shows a [2+2] cycloaddition reaction in the carbonyl–olefin metathesis with a competing carbonyl–ene reaction pathway. The carbonyl–ene reaction pathway proceeds through a six-membered cyclic transition state (TS1-LA) where the β-hydrogen shifts from an olefin to carbonyl oxygen to generate the intermediate I1-LA. Subsequently, a hydroalkoxylation step yields an oxetane (I2-LA) as shown in Scheme 1. Alternatively, the thermal [2+2] cycloaddition, through TS2-LA, can also lead to the formation of the oxetane (I2-LA). The retro [2+2] cycloaddition of the oxetane results in the formation of the carbonyl–olefin metathesis product (PD).36 The present study is focused on the catalysis imparted by the intermolecular EFs on the thermal [2+2] cycloaddition and carbonyl–ene reaction.
2. Computational details
All calculations were performed with the Gaussian 16 suite program.37 All geometries were optimized at the B97-D38,39 density functional theory (DFT) with the Def2-SVP basis set (B1).40,41 Geometries were optimized at the SMD implicit solvation model considering 1,2-dichloroethane as the solvent.42 Frequency calculations were performed at the B1 level of theory where the transition states (TSs) were characterised by one imaginary frequency corresponding to the reaction coordinate, whereas the reactants (RCs) and intermediates (Is) were devoid of any imaginary frequency. The intrinsic reaction coordinate (IRC) calculations were used for further verification of the optimized TSs.43–45 Energies were further refined at the B97-D/Def2-TZVP40 level of theory (B2) in a similar solvation model as in B1 optimization. The EF calculations were performed by single point calculations on the B1 optimized geometries at the B2 level of theory, as well as on geometries optimized at the B2 level of theory in the presence of the EF. The intermolecular EFs were quantified using the TITAN code.46 Implicit solvation models in the present study under higher EFs are aimed to mimic the intermolecular EF generated by the LAs. However, such solvation models lack solvent specific charge transfers under high EFs15,18 which are more relevant in a real reaction in solvents in an external EF experimental setup.3. Results and discussion
The introduction of an LA into the RCCOM reaction results in efficient catalysis under mild experimental conditions.47 The ion-pair ([AlCl2]+[SbF6]−) LA is generated from the precursors AgSbF6 and AlCl3 as an active catalyst for the reaction. The in situ generated LA acts as a strong Lewis acidic super-electrophile to stimulate relatively unreactive RC in RCCOM reactions as in Scheme 1.363.1. Quantification of the intermolecular EF and catalysis
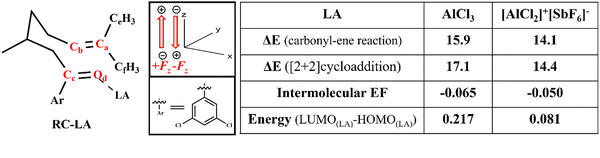
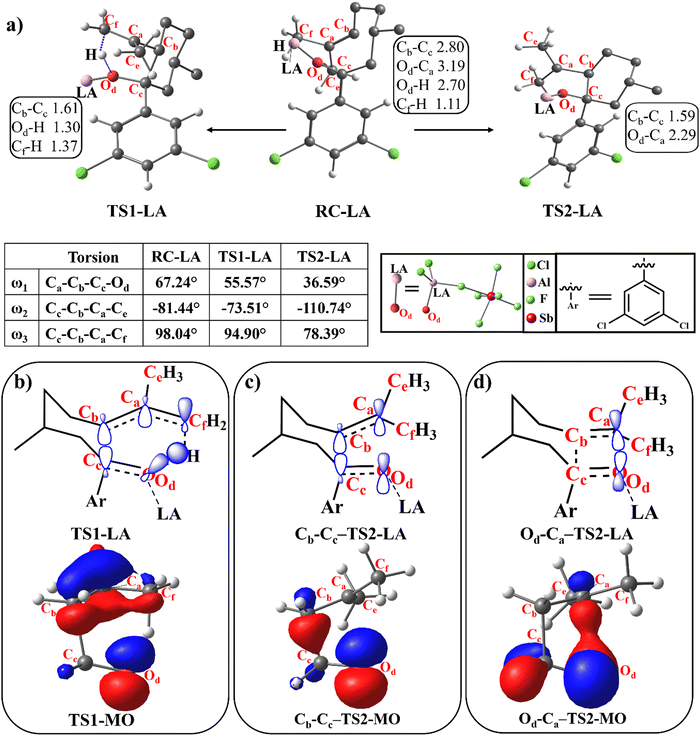
The proximity of the LA can impart an extreme (>0.01 a.u.5) EF on the reactants and the resultant mechanistic changes that lead to catalysis are of prime importance.48 The quantification of intermolecular EF as a function of LA proximity and its catalytic activity is summarised in Fig. 1. The equilibrium distance, defined as ‘d’, between an aluminium atom in the LA ([AlCl2]+[SbF6]−) and the RC carbonyl oxygen (Od) is 1.79 Å in both RC-LA and TS1-LA, while it is 1.74 Å in TS2-LA. In the case of LA (AlCl3), the equilibrium distance ‘d’ is found to be 1.82 Å in both RC-LA and TS1-LA, and 1.77 Å in TS2-LA. The proximity effect of the LA to the reactant was analysed by imposing manual displacement to the equilibrium distance ‘d’ along the Od–LA direction. Fig. 1b shows the amount of intermolecular EF along the reaction axis as a function of the proximity of the LA to the reactant. The intermolecular electric field (EF) is computed using the TITAN code, as illustrated in Fig. S1 in the ESI.† The intermolecular EF due to the LA is mapped onto the axis that starts from oxygen (Od) and passes through the carbon (Ca) atom as shown in Fig. 1a. In the optimized geometry of the LA ([AlCl2]+[SbF6]−)-coordinated RC, the intermolecular EF along the reaction axis is −0.0507 a.u. and for LA (AlCl3), the value is −0.0658 a.u. As the distance increases to (d + 1) Å, the intermolecular EF value decreases to −0.0141 a.u. for LA([AlCl2]+[SbF6]−) and −0.0213 a.u. for LA(AlCl3), respectively. The results also underscore that the use of an ion-pair ([AlCl2]+[SbF6]−) may not always bring enhanced intermolecular EF components along the reaction axis. The net intermolecular EF component along the reaction axis, which is a vector, depends on the relative orientation of charges in space. The intermolecular EF displays non-homogeneity within a reaction, suggesting a complex spatial dispersion of electrostatic forces.49,50 The intermolecular EF value approaches near zero at a longer distance of LA as shown in Fig. 1b and Table S1 (ESI†).Both carbonyl–ene and [2+2] cycloaddition reactions are catalysed by AlCl3 and the [AlCl2]+[SbF6]− ion-pair. However, the ion pair catalysed reaction has a lower energy barrier than its neutral counterpart. The energy barriers for the carbonyl–ene and [2+2] cycloaddition reactions are 14.1 and 14.4 kcal mol−1 respectively with the ion-pair LA catalyst and the barriers are further increased to 15.9 and 17.1 kcal mol−1 respectively with the AlCl3 catalyst as shown in Fig. 1c and Fig. S2 in the ESI.† The lower energy barriers for the ion-pair catalyst, irrespective of having a lower magnitude of intermolecular EF along the reaction axis, are due to the enhanced Lewis acidity which is successively discussed in the article after detailing the EF catalysis. Fig. 1c displays the energy barrier of the [AlCl2]+[SbF6]− catalysed reaction as a function of LA proximity. LA-[AlCl3] also showed similar trends between the energy barrier and LA proximity (Fig. S3 in the ESI†). The separation between the RC and the LA directly impacts the energy barrier as shown in Fig. 1c. As the distance extends from the equilibrium distance ‘d’ to (d + 1) Å, the ΔE increases from 13.27 kcal mol−1 to 18.02 kcal mol−1 for the carbonyl–ene reaction and from 13.73 kcal mol−1 to 29.61 kcal mol−1 for the [2+2] cycloaddition reaction (Table S2 in the ESI†). It should be noted that the barrier of the reaction shows a continuous increase as a function of distance between the RC and LA and it does not show any sudden increase while detaching the Lewis coordination. All in all, the distance between the RC and LA affects the catalysis, apart from the conventional LA coordination, by imparting an intermolecular EF along the reaction axis. The so-generated intermolecular EF also enhances, complementarily, the conventional LA catalysis (vide infra).
3.2. Origin of catalysis along the reaction axis due to the intermolecular EF
In RCCOM, the innate covalent structure of RC-LA needs to be changed to a more ionic structure to facilitate the reaction. The covalent electronic structure along with its ionic resonance structure is shown in Scheme 2. Even though the ionic resonance structure is not normally attainable, a properly oriented component of an intermolecular EF or an oriented external EF analogue can stabilise such dormant polar configurations and mix them into the covalent structure. Enhanced stabilisation of the ionic structure brings catalysis in carbonyl–ene reactions and facilitates a pseudo-pericyclic pathway for the thermal [2+2] cycloaddition reaction as detailed in the following discussion.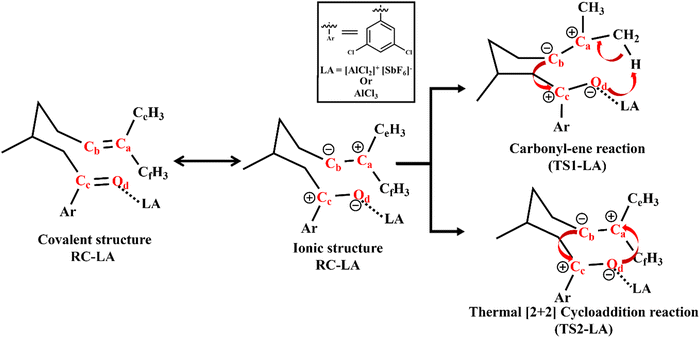 | ||
| Scheme 2 Thermal [2+2] cycloaddition and carbonyl–ene reaction that proceeds through a dormant ionic structure. | ||
The LA coordination stabilises the negatively charged oxygen in the carbonyl–ene reaction, which abstracts the β-hydrogen atom present in the olefin group. The feasibility of both reactions in Scheme 2 is attributed to the stabilisation of the negative charge present in the Od atom and the positive charge in Ca. The carbonyl–ene reaction proceeds through a concerted six-membered TS as shown in Scheme 2.51 The π-orbital of the alkene, Ca![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Cb, overlaps with the π*-orbital of the carbonyl Cc
Cb, overlaps with the π*-orbital of the carbonyl Cc![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Od to form the Cb–Cc bond as shown in Fig. 2b. The molecular orbitals (MO) from a minimalistic model between formaldehyde and isobutene are displayed in Fig. 2b–d as the MOs of the LA coordinated RCCOM reaction are cumbersome. The presence of intermolecular EFs stabilises ionic structures and consequently changes Ca to become more cationic and Od more anionic. During Cb–Cc bond formation, the reactant also undergoes a geometrical change to smoothen the abstraction of the allylic proton by the anionic Od. The π* orbital of the carbonyl group (Cc
Od to form the Cb–Cc bond as shown in Fig. 2b. The molecular orbitals (MO) from a minimalistic model between formaldehyde and isobutene are displayed in Fig. 2b–d as the MOs of the LA coordinated RCCOM reaction are cumbersome. The presence of intermolecular EFs stabilises ionic structures and consequently changes Ca to become more cationic and Od more anionic. During Cb–Cc bond formation, the reactant also undergoes a geometrical change to smoothen the abstraction of the allylic proton by the anionic Od. The π* orbital of the carbonyl group (Cc![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Od) aligns with the σ* orbital of the Cf–H bond for a better overlap during the Od–H bond formation in the concerted carbonyl–ene reaction. Orbital interactions in the carbonyl–ene reaction are shown in Fig. 2b. The geometrical changes are depicted in Fig. 2 where the dihedral angle, ω1, undergoes a significant transition from 67.24° in RC-LA to 55.57° in TS1-LA, to form the Cb–Cc bond. Similarly, the changes in dihedral angles ω2 and ω3 by 7.93° and 3.14°, respectively, facilitate the overlap between the donor π*-orbital at the Cc–Od and the acceptor Cf–H σ* orbital.
Od) aligns with the σ* orbital of the Cf–H bond for a better overlap during the Od–H bond formation in the concerted carbonyl–ene reaction. Orbital interactions in the carbonyl–ene reaction are shown in Fig. 2b. The geometrical changes are depicted in Fig. 2 where the dihedral angle, ω1, undergoes a significant transition from 67.24° in RC-LA to 55.57° in TS1-LA, to form the Cb–Cc bond. Similarly, the changes in dihedral angles ω2 and ω3 by 7.93° and 3.14°, respectively, facilitate the overlap between the donor π*-orbital at the Cc–Od and the acceptor Cf–H σ* orbital.
A pericyclic pathway for the concerted [2+2] cycloaddition is thermally forbidden.52 Instead, the reaction proceeds via an ionic pseudo-pericyclic pathway as given in Scheme 2. The [2+2] cycloaddition proceeds with the formation of two new bonds between Cb and Cc and between Ca and Od as shown in Fig. 2c and d. Along with the Cb–Cc bond formation, the nonbonding orbitals at the Od atom, which is perpendicular to the Cc![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Od π-orbital, rotate and form a bond with the cationic Ca. The overlap of the nonbonding orbital at the Od and the vacant p-orbital at the Ca atom leads to the bond formation and it does not require either Ca
Od π-orbital, rotate and form a bond with the cationic Ca. The overlap of the nonbonding orbital at the Od and the vacant p-orbital at the Ca atom leads to the bond formation and it does not require either Ca![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Cb or Cc
Cb or Cc![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Od bonds to rotate 180°. The partial rotation of the nonbonding orbital at the Od atom and the vacant p-orbital at the Ca atom plays a crucial role in facilitating the Ca–Od bond formation and the corresponding transition state (TS2-LA) is depicted in Fig. 2d. The angle ω1 undergoes a significant transition from 67.24° in the initial RC-LA to 36.59° in the subsequent TS2-LA indicating the rotation of the nonbonding orbital towards the vacant p-orbitals at the Ca atom. Similarly, angles ω2 and ω3 experience shifts of 29.30° and 19.65°, respectively, where the rotations facilitate overlap of the donor nonbonding orbital at the Od and the acceptor vacant p-orbital at the Ca atom.
Od bonds to rotate 180°. The partial rotation of the nonbonding orbital at the Od atom and the vacant p-orbital at the Ca atom plays a crucial role in facilitating the Ca–Od bond formation and the corresponding transition state (TS2-LA) is depicted in Fig. 2d. The angle ω1 undergoes a significant transition from 67.24° in the initial RC-LA to 36.59° in the subsequent TS2-LA indicating the rotation of the nonbonding orbital towards the vacant p-orbitals at the Ca atom. Similarly, angles ω2 and ω3 experience shifts of 29.30° and 19.65°, respectively, where the rotations facilitate overlap of the donor nonbonding orbital at the Od and the acceptor vacant p-orbital at the Ca atom.
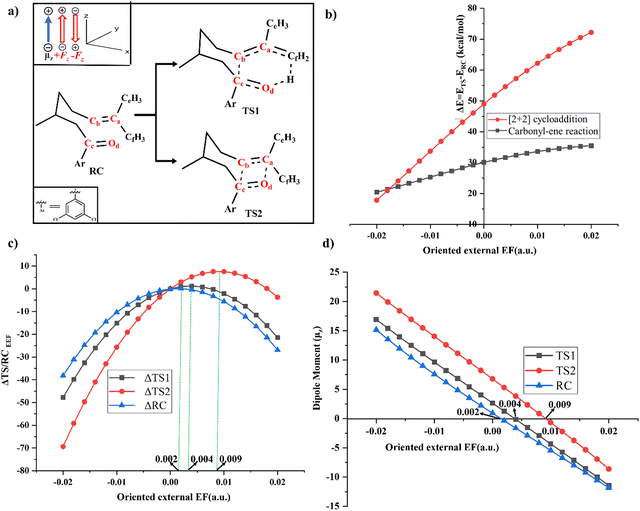
A comparison of the changes in relative energies of RC and TSs explains the observed catalysis along the −Fz direction and inhibition along the +Fz direction. (Fig. 3b and c) Under −0.02 a.u. oriented external EF, ΔE decreases to 20.3 kcal mol−1 for the carbonyl–ene reaction and up to 17.7 kcal mol−1 for the [2+2] cycloaddition reactions. Conversely, with a +0.02 a.u. oriented external EF along the reaction axis, ΔE increases to 35.4 kcal mol−1 for the carbonyl–ene reaction and up to 72.1 kcal mol−1 for the [2+2] cycloaddition reaction (Table S5 in the ESI†). The relative stabilisation of TSs along −Fz is higher as compared to the RC and vice versa along the +Fz direction effectively resulting in catalysis along the −Fz direction and inhibition along the +Fz direction.
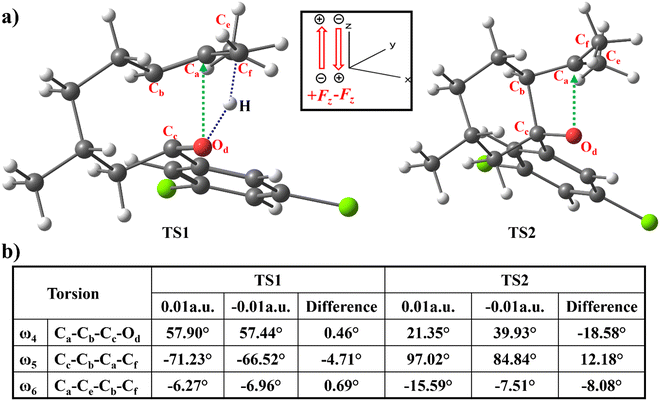
Oriented external EF, analogous to LA, possesses the capability to bring asynchronous TSs and partially rotate the orbitals to proceed with reactions more effectively as shown in Fig. 4. In TS1, the angular displacement of ω4 experiences a notable variation from 57.90° to 57.44° in response to changes in oriented external EF strength, ranging from 0.01 (+Fz) to −0.01 (−Fz). Additionally, angles ω5 and ω6 undergo shifts of −4.71° and 0.69°, respectively. The changes in ω5 facilitate the rotation of the CfH3 group that enables the interaction between the π*-orbital of the Cc–Od atom and the σ* orbital of Cf–H and reduces the energy barrier for TS1.
The angle ω4 as defined in Fig. 4 undergoes a significant transition from 21.35° to 39.93° in TS2 as the amount of oriented external EF varies from 0.01(+Fz) to −0.01(−Fz) for the [2+2] cycloaddition reaction. Additionally, the angles ω5 and ω6 in TS2 also change by 12.18° and −8.08° respectively. The changes in ω6, an improper dihedral angle, indicate that the −Fz tends to promote a more planar cationic structure for the Ca atom. As in Fig. 2 the changes due to the −Fz EF in ω4 and ω5 facilitate a better overlap of the Od nonbonding orbital to the vacant p-orbital at the Ca. As such, the EF along −Fz sets the foundation for a better rotation of the donor Od nonbonding orbital and the acceptor vacant p-orbital, leading to the Od–Ca bond formation and effectively bringing down the barrier in TS2. Geometric comparison between TS1 and TS2 shows that TS1 requires less substantial structural changes and directly correlates with its lower energy. Thus, in the progression of the reaction, the change in the dihedral angle plays a pivotal role in facilitating molecular transformations. It orchestrates the rotation of both the nonbonding donor orbital and the vacant acceptor p-orbital for a smooth pseudo-pericyclic pathway.
An oriented external EF applied along the reaction axis has a direct impact on the energy difference between the lowest unoccupied molecular orbital (LUMO(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)) and the highest occupied molecular orbital (HOMO(C
O)) and the highest occupied molecular orbital (HOMO(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)).54 The HOMO(C
C)).54 The HOMO(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) overlaps with the LUMO(C
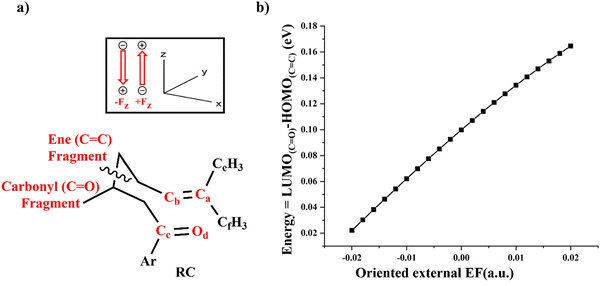
C) overlaps with the LUMO(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) during the Cb–Cc bond formation. To detail the orbital energy changes, the RC was divided into two segments: the ene part and the carbonyl part. To preserve valency, hydrogen atoms were added to both the carbonyl and ene groups as given in Fig. 5a. The impact of an oriented external EF on the MOs of carbonyl and ene groups was measured by considering one group at a time and keeping similar relative orientation of external EF to that in Fig. 5. The ene group is replaced with a dummy atom (see Fig. S6 in the ESI†) to study the EF's effect on the MOs of the carbonyl group and vice versa for the ene group.55 EF analysis between the ene and carbonyl fragments showed that application of external EF along the +Fz direction increased the LUMO–HOMO energy gap, whereas applying it along the −Fz direction decreased the energy gap as visible in Fig. 5b and Table S7 in the ESI.†
O) during the Cb–Cc bond formation. To detail the orbital energy changes, the RC was divided into two segments: the ene part and the carbonyl part. To preserve valency, hydrogen atoms were added to both the carbonyl and ene groups as given in Fig. 5a. The impact of an oriented external EF on the MOs of carbonyl and ene groups was measured by considering one group at a time and keeping similar relative orientation of external EF to that in Fig. 5. The ene group is replaced with a dummy atom (see Fig. S6 in the ESI†) to study the EF's effect on the MOs of the carbonyl group and vice versa for the ene group.55 EF analysis between the ene and carbonyl fragments showed that application of external EF along the +Fz direction increased the LUMO–HOMO energy gap, whereas applying it along the −Fz direction decreased the energy gap as visible in Fig. 5b and Table S7 in the ESI.†
3.3. How does the catalytic component of intermolecular EF add to Lewis acidity?
A comparative analysis of the LAs, AlCl3 and [AlCl2]+[SbF6]−, revealed that [AlCl2]+[SbF6]− consistently shows a lower ΔE value in both carbonyl–ene and [2+2] cycloaddition reactions, as depicted in Fig. 6. Surprisingly, the component of intermolecular EF along the axis that connects Od to Ca is higher for AlCl3 (−0.06 a.u.) than that for [AlCl2]+[SbF6]− (−0.05 a.u.). This result underscores that merely creating ionic fragments may not lead to enhanced EF catalysis, as the strength of intermolecular EF components along a specific bond or reaction axis depends not only on the magnitude of the charges but also on the relative orientations of the charged groups. Comparison of the LUMO–HOMO energy gaps of the LAs revealed a notable distinction between AlCl3 and [AlCl2]+[SbF6]− where the former has a higher 0.217 eV energy gap against 0.081 eV of the latter. The LUMO–HOMO energy gap is an indicator of Lewis acidity, suggesting that a lower value, as seen in ion pairs, represents a more acidic nature than AlCl3.56,57 The reduced intermolecular EF catalysis along the reaction axis is compensated by the enhanced Lewis acid catalysis.A complete picture of the catalytic cycle and the effect of the oriented external EF on the reaction is shown in Fig. 7. The oriented external EF is applied along the reaction axis, parallel to the electron flow in the reaction mechanism. The oriented external EF is applied along Od to Ca in TS1, TS2, and TS3, whereas in TS4, the direction of Fz is changed from Od to Cc to make it parallel to the reaction axis of the retro [2+2] cycloaddition step. Fig. 7a illustrates all the individual steps in the catalytic cycle while Fig. 7b represents the corresponding potential energy profile. The preferred pathway in Fig. 7b, where the ion pair is coordinated to RC, proceeds through TS2-LA, I2-LA, and TS4-LA to the metathesis product. Due to the lower barrier of the carbonyl–ene pathway (TS1-LA), I1-LA remains in equilibrium with RC-LA. However, the following hydroalkoxylation step towards I2-LA is not operative due to the higher barrier. Fig. 7c and Table S8 in the ESI† depict the energy profile of the pristine reaction without any oriented external EF or LA where barriers are found to be prohibitively high. The profile in Fig. 7d is recreated by aligning an oriented external EF along the reaction axis at each individual step to mimic the intermolecular EF component along the reaction axes. Applying an oriented external EF along the reaction axis, specifically along the Od–Ca direction, catalyses the carbonyl–ene reaction, hydroalkoxylation, and [2+2] cycloaddition. Similarly, in the retro [2+2] cycloaddition reaction, where the electron flow is along the Od–Cc direction, the reaction is catalysed by an oriented external EF aligned in that direction. The component of intermolecular EF along the Od–Cc bond in I2 is found to be −0.08 a.u. As such, the application of an EF along the reaction axis profoundly influences the catalytic efficiency in the carbonyl–ene reaction, hydroalkoxylation, [2+2] cycloaddition, and retro [2+2] cycloaddition. It also conveys that the intermolecular EF component along the reaction axis, which originates from the LAs, has a unique catalytic effect.
The intermolecular EF generated at the reaction site is non-uniform and its effect and magnitude diminish significantly for groups and atoms located farther from the LA catalyst. In contrast, the externally applied EF is uniform which exerts the same magnitude of influence on all groups and atoms along its applied direction. Due to this difference between uniform external and nonuniform intermolecular EFs, a lower magnitude of external EF is enough to reproduce similar catalytic characteristics by an intermolecular EF. For example, an external EF of 0.022 a.u. is enough to lower the pristine reaction barrier to match that of the LA-catalysed [2+2] cycloaddition reaction where the latter has an intermolecular EF of 0.050 a.u. All in all, along with the conventional LA catalysis characterised by the LUMO–HOMO energies, the intermolecular EF originating from LAs has a distinct and unique contribution towards gross catalysis.
4. Conclusion
Exploring the thermal [2+2] cycloaddition and carbonyl–ene reactions in a ring-closing carbonyl–olefin metathesis (RCCOM) sheds light on the pivotal roles of intermolecular EFs in catalysis. The specific orientations of coordinated LAs with reactants lead to the generation of specifically oriented intermolecular EF components along the reaction axis. Intermolecular EF components along the reaction axis, which is parallel to the electron flow in each reaction step, are found to catalyse the reaction with a unique mechanism. The preferred pathway for the reaction is a [2+2] cycloaddition followed by a retro [2+2] cycloaddition step. A carbonyl–ene reaction step is also operative which makes the starting reactant form an equilibrium with the following intermediate. In the thermal [2+2] cycloaddition, the intermolecular EF promotes the charge transfer from the alkene (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C π-orbital) donor to the carbonyl (C
C π-orbital) donor to the carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O π*-orbital) acceptor to enhance the C–C bond formation. Stabilisation of the resultant charge transferred ionic intermediates further enhances the subsequent C–O bond formation via a partial rotation of nonbonding electrons on oxygen which is necessary to circumvent the thermal forbiddances of [2+2] cycloaddition.
O π*-orbital) acceptor to enhance the C–C bond formation. Stabilisation of the resultant charge transferred ionic intermediates further enhances the subsequent C–O bond formation via a partial rotation of nonbonding electrons on oxygen which is necessary to circumvent the thermal forbiddances of [2+2] cycloaddition.
The carbonyl–ene reaction also follows the initial step of C–C bond formation as in [2+2] cycloaddition and accumulates anionic charge on the carbonyl oxygen. Subsequent Od–H bond formation proceeds via electron transfer to the σ* orbital of the Cf–H bond. The mechanistic importance of intermolecular EF is delineated by modelling reactions under similarly oriented external EFs. Quantification of the intermolecular EF along the reaction axis has revealed that, contrary to common assumption, the neutral LA([AlCl3]) imparts a higher intermolecular EF than its ion pair ([AlCl2]+[SbF6]−) counterpart. The findings exemplify that judicious utilisation of intermolecular EFs can effectively maximize catalytic benefits and a catalytic mechanism is incomplete without delineating the role of intermolecular EFs.
Conflicts of interest
The authors declare no conflicts.Acknowledgements
RR acknowledges the Science and Engineering Research Board (SERB), India, for the grant SRG/2021/000439. LS and KG acknowledge teaching assistantships from NITR.References
- S. Shaik, D. Mandal and R. Ramanan, Nat. Chem., 2016, 8, 1091–1098, DOI:10.1038/nchem.2651.
- S. Shaik, R. Ramanan, D. Danovich and D. Mandal, Chem. Soc. Rev., 2018, 47(14), 5125–5145, 10.1039/c8cs00354h.
- S. Ciampi, N. Darwish, H. M. Aitken, I. Diez-Perez and M. L. Coote, Chem. Soc. Rev., 2018, 47, 5146–5164, 10.1039/C8CS00352A.
- A. C. Aragonès, N. L. Haworth, N. Darwish, S. Ciampi, N. J. Bloomfield, G. G. Wallace, I. Diez-Perez and M. L. Coote, Nature, 2016, 531(7592), 88–91, DOI:10.1038/nature16989.
- S. D. Fried, S. Bagchi and S. G. Boxer, Science, 2014, 346(6216), 1510–1514, DOI:10.1126/science.1259802.
- A. Warshel, Acc. Chem. Res., 1981, 14(9), 284–290, DOI:10.1021/ar00069a004.
- R. Meir, H. Chen, W. Lai and S. Shaik, ChemPhysChem, 2010, 11(1), 301–310, DOI:10.1002/cphc.200900848.
- Z. Wang, D. Danovich, R. Ramanan and S. Shaik, J. Am. Chem. Soc., 2018, 140(41), 13350, DOI:10.1021/jacs.8b08233.
- H. Hirao, H. Chen, M. A. Carvajal, Y. Wang and S. Shaik, J. Am. Chem. Soc., 2008, 130, 3319–3327, DOI:10.1021/ja070903t.
- R. Ramanan, D. Danovich, D. Mandal and S. Shaik, J. Am. Chem. Soc., 2018, 140, 4354–4362, DOI:10.1021/jacs.8b00192.
- G. Cassone, F. Pietrucci, F. Saija, F. Guyot and A. M. Saitta, Chem. Sci., 2017, 8, 2329–2336 RSC.
- A. Yosipof, H. Basch and S. Hoz, J. Phys. Chem. A, 2013, 117, 5023–5027, DOI:10.1021/jp402758u.
- T. Stuyver, D. Danovich, F. De Proft and S. Shaik, J. Am. Chem. Soc., 2019, 141(24), 9719–9730, DOI:10.1021/jacs.9b04982.
- V. Conti Nibali, S. Maiti, F. Saija, M. Heyden and G. Cassone, J. Chem. Phys., 2023, 158, 184501 CrossRef PubMed.
- S. Pullanchery, S. Kulik, T. Schönfeldová, C. K. Egan, G. Cassone, A. Hassanali and S. Roke, Nat. Commun., 2024, 15, 5951 CrossRef PubMed.
- K. Gopakumar, V. Samantaray, M. K. Prusty, L. Swain and R. Ramanan, Chem. Commun., 2023, 2, 13054–13057, 10.1039/D3CC04283A.
- G. Cassone, A. Sofia, G. Rinaldi and J. Sponer, J. Phys. Chem. C, 2019, 123, 9202–9208 CrossRef CAS.
- G. Cassone and F. Martelli, Nat. Commun., 2024, 15, 1856 Search PubMed.
- H. Hao, I. Leven and T. Head-Gordon, Nat. Commun., 2022, 13, 280, DOI:10.1038/s41467-021-27941-x.
- C. Zhu, L. N. Pham, X. Yuan, H. Ouyang, M. L. Coote and X. Zhang, J. Am. Chem. Soc., 2023, 145(39), 21207–21212, DOI:10.1021/jacs.3c08818.
- P. Basuri, L. E. Gonzalez, N. M. Morato, T. Pradeep and R. G. Cooks, Chem. Sci., 2020, 11, 12686–12694, 10.1039/D0SC02467H.
- E. Gnanamani, X. Yan and R. N. Zare, Angew. Chem., Int. Ed., 2020, 59, 3069–3072, DOI:10.1002/anie.201913069.
- Z. P. Rao, Y. G. Fang, Y. S. Pan, W. C. Yu, B. L. Chen, J. S. Francisco, C. Q. Zhu and C. H. Chu, J. Am. Chem. Soc., 2023, 145(45), 24717–24723, DOI:10.1021/jacs.3c08101.
- X. Song, C. Basheer and R. N. Zare, J. Am. Chem. Soc., 2023, 145(50), 27198–27204, DOI:10.1021/jacs.3c08643.
- G. Cassone, J. Phys. Chem. Lett., 2020, 11, 8983–8988 CrossRef PubMed.
- D. Zhang, X. Yuan, C. Gong and X. Zhang, J. Am. Chem. Soc., 2022, 144(35), 16184–16190, DOI:10.1021/jacs.2c07385.
- K. Gong, A. Nandy, Z. Song, Q.-S. Li, A. Hassanali, G. Cassone, S. Banerjee and J. Xie, J. Am. Chem. Soc., 2024, 146(46), 31585–31596 CrossRef PubMed.
- I. K. Mati and S. L. Cockroft, Chem. Soc. Rev., 2010, 39(11), 4195–4205, 10.1039/B822665M.
- T. Stuyver, R. Ramanan, D. Mallick and S. Shaik, Angew. Chem., Int. Ed., 2020, 59(20), 7915, DOI:10.1002/anie.201916592.
- H. Schmalzried and S. Smolin, Ber. Bunsen-Ges. Phys. Chem., 1998, 102, 1740–1746, DOI:10.1002/bbpc.19981021203.
- H. Albright, H. L. Vonesh, M. R. Becker, B. W. Alexander, J. R. Ludwig, R. A. Wiscons and C. S. Schindler, Org. Lett., 2018, 20, 4954–4958, DOI:10.1021/acs.orglett.8b02086.
- J. R. Ludwig, S. Phan, C. C. McAtee, P. M. Zimmerman, J. J. Devery and C. S. Schindler, J. Am. Chem. Soc., 2017, 139, 10832–10842, DOI:10.1021/jacs.7b05641.
- U. P. N. Tran, G. Oss, D. P. Pace, J. Ho and T. V. Nguyen, Chem. Sci., 2018, 9, 5145, 10.1039/C8SC00907D.
- T. Malakar and P. M. Zimmerman, J. Org. Chem., 2021, 86(3), 3008–3016, DOI:10.1021/acs.joc.0c03021.
- L. Ravindar, R. Lekkala, K. P. Rakesh, A. M. Asiri, H. M. Marwani and H.-L. Qin, Org. Chem. Front., 2018, 5, 1381–1391, 10.1039/C7QO01037K.
- A. J. Davis, R. B. Watson, D. J. Nasrallah, J. L. Gomez-Lopez and C. S. Schindler, Nat. Catal., 2020, 3, 787–796, DOI:10.1038/s41929-020-00499-5.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci and G. A. Petersson, Gaussian16, Revision A.03, Gaussian Inc, Wallingford CT, 2016 Search PubMed[See ESI† for full reference].
- S. Grimme, J. Comput. Chem., 2006, 27, 1787–1799, DOI:10.1002/jcc.20495.
- S. Grimme, J. Chem. Phys., 2006, 124, 034108, DOI:10.1063/1.2148954.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297, 10.1039/b508541a.
- A. Schäfer, H. Horn and R. Ahlrichs, J. Chem. Phys., 1992, 97, 2571, DOI:10.1063/1.463096.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378–6396, DOI:10.1021/jp810292n.
- K. Fukui, Acc. Chem. Res., 1981, 14, 363–368, DOI:10.1021/ar00072a001.
- C. Gonzalez and H. B. Schlegel, J. Chem. Phys., 1989, 90, 2154–2161, DOI:10.1063/1.456010.
- C. Gonzalez and H. B. Schlegel, J. Phys. Chem., 1990, 94, 5523–5527, DOI:10.1021/j100377a021.
- T. Stuyver, J. Huang, D. Mallick, D. Danovich and S. Shaik, J. Comput. Chem., 2020, 41(1), 74–82, DOI:10.1002/jcc.26072.
- H. Albright, A. J. Davis, J. L. Gomez-Lopez, H. L. Vonesh, P. K. Quach, T. H. Lambert and C. S. Schindler, Chem. Rev., 2021, 121, 9359–9406, DOI:10.1021/acs.chemrev.0c01096.
- J. L. Alvarez-Hernandez, X. Zhang, K. Cui, A. P. Deziel, S. Hammes-Schiffer, N. Hazari, N. Piekut and M. Zhong, Chem. Sci., 2024, 15(18), 6800–6815, 10.1039/D3SC06177A.
- T. A. Rokob, I. Bakó, A. Stirling, A. Hamza and I. Pápai, J. Am. Chem. Soc., 2013, 135, 4425–4437, DOI:10.1021/ja312387q.
- S. Grimme, H. Kruse, L. Goerigk and G. Erker, Angew. Chem., Int. Ed., 2010, 49, 1402–1405, DOI:10.1002/anie.200905484.
- H. A. Rodríguez, D. A. Cruz, J. I. Padrón and I. Fernández, J. Org. Chem., 2023, 88, 11102–11110, DOI:10.1021/acs.joc.3c01059.
- R. Hoffmann and R. B. Woodward, Acc. Chem. Res., 1968, 1, 17–22, DOI:10.1021/ar50001a003.
- K. Gopakumar, S. Shaik and R. Ramanan, Angew. Chem., Int. Ed., 2023, 135, e202307579, DOI:10.1002/ange.202307579.
- S. Shaik, D. Danovich, J. Joy, Z. Wang and T. Stuyver, J. Am. Chem. Soc., 2020, 142(29), 12551–12562, DOI:10.1021/jacs.0c05128.
- R. J. Burns, I. K. Mati, K. B. Muchowska, C. Adam and S. L. Cockroft, Angew. Chem., Int. Ed., 2020, 59(38), 16717–16724, DOI:10.1002/anie.202006943.
- R. G. Parr and R. G. Pearson, J. Am. Chem. Soc., 1983, 105(26), 7512–7516, DOI:10.1021/ja00364a005.
- W. B. J. C. R. Jensen, Chem. Rev., 1978, 78(1), 1–22, DOI:10.1021/cr60311a002.
Footnote |
| † Electronic supplementary information (ESI) available: The intermolecular EF values, group charges, molecular orbital energies, energies and cartesian coordinates of the optimized geometries. See DOI: https://doi.org/10.1039/d4cp04879b |
| This journal is © the Owner Societies 2025 |


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EEF) −
EEF) −