Open Access Article
Open Access ArticleMechanism and behavior of caffeine sorption: affecting factors
Merve Fakioğlu
 and
Yasemen Kalpaklı
and
Yasemen Kalpaklı
 *
*
Department of Chemical Engineering, Yildiz Technical University, 34220 Davutpaşa, Istanbul, Turkey. E-mail: merve.fakioglu@std.yildiz.edu.tr; kalpakli@yildiz.edu.tr
First published on 16th September 2022
Abstract
Caffeine is one of the emerging pollutants with a diverse chemical composition. It is mixed with the hydrobiota as a result of its high consumption, and when certain dose intervals are exceeded, it re-enters the human body through indirect routes such as plants, animals, soil, water, and the food chain, causing health problems that are difficult or impossible to treat, and irreversible environmental problems. This situation raises concerns about the presence of pollutants emerging in water resources, igniting interest in water treatment processes and the development of alternative methods. Although there are several methods for removing caffeine from aqueous media, adsorption is the most popular because it is less expensive than other methods and has the highest removal efficiency. Furthermore, it has the benefit of selectively attaching the molecules in solution. In this article, studies on the caffeine adsorption process have been examined, and the caffeine adsorption efficiency of various adsorbents has been summarized by compiling information such as pH, contact time, temperature, and concentration of adsorbent and adsorbate, which are considered as optimum processing conditions. The binding mechanism was investigated, and it was clearly stated how caffeine adheres to the adsorbent surface. Among the equilibrium adsorption isotherms, the isotherm model with the best agreement with the experimental data was attempted to be determined. Many studies clearly show that the process of developing environmentally friendly and high-capacity adsorbents in sustainable processes and in harmony with the circular economy is increasing day by day.
1. Introduction
In recent years, the increase in the concentration of pollution parameters in the hydrobiota, which forms the basis of life in the ecosystem, has emerged as a serious problem.1 As a result of the increase in the human population and the increasing technological and industrial developments in parallel, many pollutants are mixed into the receiving waters. The concern about the possibility of experiencing water scarcity due to the risk of depletion of water resources in the future or the risk of it being insufficient is one of the biggest reasons for the rapid increase in research on dangerous and toxic compounds called “new emerging pollutants”.2,3Emerging pollutants are defined by the United States Environmental Protection Agency (USEPA) as chemical compounds that do not have a status yet.4 They are pollution parameters such as caffeine, which have been present in our environment before now, but cannot be determined because the analytical methods used are not sensitive enough. Although caffeine is an emerging pollutant, it is accepted as the most important representative pollutant of pharmaceutical active substances.5
Caffeine is an organic chemical compound that can be found in many plant species, its chemical formula is C8H10N4O2, its systematic name is 1,3,7-trimethylxanthine, and its molecular weight is 194 g mol−1.6–9 Caffeine, a natural alkaloid, is a stimulant found both in daily foods and beverages (coffee, tea, soda, energy drinks, chocolates, etc.) and in many pharmaceutical products, since it is obtained from many plant species.10 Some cold and flu remedies may also contain caffeine.11 Furthermore, over-the-counter drugs that do not require a prescription and are illegal to sell and use, such as drugs, contain caffeine.12 As a result, caffeine has been identified as one of nature's most common pharmaceutically active compounds.13,14 Pharmacovigilance has become an important application for assessing the environmental risks posed by caffeinated drugs and implementing the necessary actions. Specifically, ecopharmacovigilance (EPV), a subfield of pharmacovigilance, has taken its place today as an important environmental management program that operates to minimize the hidden dangers posed by pharmaceutical pollutants in the environment.12
Caffeine is consumed by approximately 90% of the adult population. The main sources of caffeine as a result of this consumption are metabolic excretion wastes formed as a result of caffeine consumption, rinsing of dishes used for caffeinated beverages, caffeinated beverages that are not consumed by throwing them in the sink, improper accumulation of expired, or unused pharmaceutical products containing caffeine, and the production facility wastes of these products originate from hospital wastes and the like.15–17
After consumption of foods, beverages or drugs containing caffeine, the part that cannot be metabolized is excreted from the body. Discarded caffeine first reaches the sewer and then to the wastewater treatment plants.18 Waste caffeine, which is not biodegradable or is not destroyed during treatment in the treatment plant, enters the water cycle and may even mix with drinking water.19,20 The presence of caffeine and its metabolites in surface waters or groundwater, which can be removed up to 99% during wastewater treatment, indicates that they can escape treatment systems or that re-pollution from human activities contributes.21
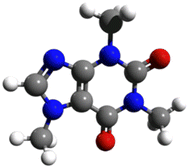
Furthermore, caffeine detected in the environment in the absence of natural caffeine sources (tea and coffee) is a sign of contamination caused by humans.13 Caffeine is thus an anthropogenic biomarker (Fig. 1).12,22
Chronic toxicity, endocrine disruption, the development of bacteria resistant species, and congenital anomalies are known to occur in organisms as a result of exposure of aquatic flora to pollutants such as caffeine.23,24 The water flora, as well as the quality of life and health of living things, are adversely affected by wastewater discharges that do not meet the wastewater discharge criteria. The wastewater treatment plant's effluent can also be used to irrigate agricultural land. Mixing these waters with the soil reduces soil fertility, and by mixing with groundwater, they can accumulate in plant tissues, eventually reaching humans and animals via the food chain.25
Although caffeine has potential side effects, the Federal Drug Administration has classified it as “Generally Safe” and it is now legal in most countries.26 Caffeine is a hydrophilic and lipophilic substance that can disperse into all body fluids and pass through all biological membranes in humans and animals. Caffeine consumption in moderation does not pose a health risk, as long as it is not excessive.27
It is true that daily caffeine consumption varies by country and by age group. In addition, the amount of caffeine consumed on a daily basis should be considered. For healthy adults, doses up to 400 mg are safe, but doses exceeding 400 mg may cause difficult or incurable side effects.7 The right amount of caffeine helps to keep the metabolism active. Capillaries expand, blood flow accelerates, and you feel more vigorous and lively as a result of caffeine stimulating the central nervous system.28–30 Caffeine, while necessary to keep people awake in these situations, has been shown to increase anxiety, raise blood pressure, and reduce hand stability.31 The maximum daily dose for pregnant women is 300 mg. Women who consume more than 300 mg of caffeine per day are known to have a lower fertility rate and a higher risk of miscarriage.28,30 It is recommended not to exceed 25–50 mg d−1 for children aged 2–5 and 50–75 mg d−1 for children aged 10–15.29 In other words, caffeine consumption in children and adolescents should not exceed <100 mg d−1.32
Due to the potential negative effects of caffeine on human and environmental health, special methods are needed to efficiently remove it from wastewater before discharge.25 When compared to methods such as membrane filtration or oxidative processes, which are among the different alternative technologies for caffeine purification, adsorption stands out due to its easy applicability, high efficiency and lower cost.33,34 The high (80–95%) removal efficiency of pollutants,35 as well as the ability to selectively adsorb targeted substances, are the important advantages of the method.36
Because adsorption is a surface treatment, the ability to functionalize an adsorbent using physical and/or chemical methods is the most important factor in increasing its efficiency. The pore structure of the adsorbent is another important factor that influences adsorption efficiency. The adsorbent surface's large and porous structure allows for a more efficient adsorption process. Natural adsorbents and adsorbents obtained through engineering studies are the two types of adsorbents available. Although natural adsorbents are used without being functionalized, their surface area and pore size are usually limited. Engineering adsorbents, on the other hand, are adsorbents whose surfaces have been modified to make them more efficient for the adsorption process. Acid treatment, organic modification, surfactant modification, polymer modification, heat treatment, and other activation processes are examples.13,17,37–39 The most commonly used and preferred methods are heat treatment and acid treatment to functionalize the adsorbent.
Among the studies that can be used effectively in caffeine adsorption, there are many different adsorbents and activation studies, some of them: activated carbon obtained from grape stalk,17 thermally modified Verdo-lodo bentonite,25 activated carbon from date stone,37 biochar obtained from pine needles,40 oxidized activated carbon from Luffa cylindrica41 and organically modified saponites.42
In this study, the 3D molecular structure in Fig. 2 and the adsorption of caffeine molecules, whose chemical properties are given in Table 1, from the aqueous solution by adsorption were investigated.
| Molecular weight | 194.19 g mol−1 |
| Molecular formula | C8H10N4O2 |
| pKa | 14 (25 °C) |
Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kow Kow |
−0.07 |
| Water solubility | 21.600 mg L−1 |
| pH | 6.55 |
2. Caffeine adsorption and affecting parameters
The adsorption process is divided into two stages: “film diffusion”, in which dissolved substances in the solution pass through the solvent liquid film surrounding the adsorbent, and “pore diffusion”, in which substances approaching the adsorbent surface enter the interior of the pores. The pH of the solution, the ambient temperature, the properties of the adsorbent, the adsorbed substance, and the solvent are all factors that affect adsorption during this process.Table 2 lists the adsorbents used in current caffeine adsorption studies, as well as experimental conditions, inlet adsorbate and adsorbent concentrations, and the maximum adsorption capacity obtained under these conditions.
| Adsorbent | Max. adsorption capacity (mg g−1) | Experimental conditions | Equilibrium adsorption isotherms | Adsorbent concentration (g L−1) | Adsorbate concentration (mg L−1) | ||
|---|---|---|---|---|---|---|---|
| pH | Contact time (min) | Temperature (°C) | |||||
| Grape stalk17 | 89.2 | 2 | 40 | 25 | S | 25.0 | — |
| Modified grape stalk with phosphoric acid17 | 129.6 | 2 | 30 | 25 | S | 15.0 | — |
| Grape stalk activated carbon17 | 395.0 | 4 | 30 | 25 | S | 1.0 | — |
| Pineapple plant leaves activated carbon18 | 155.5 | 5.8 | 240 | 25 | L | 1.0 | 500 |
| Thermally modified Verdo-lodo bentonite25 | 31.1 | — | 2400 | 25 | L | 160.0 | 143 |
| Date stone activated carbon37 | 28.0 | — | 80 | 25–50 | — | 8.0 | 12 |
| Sargassum activated carbon38 | 221.6 | 6 | 90 | 25 | L, F | 0.6 | 500 |
| Active biochar from açai seed39 | 176.8 | 7 | 180 | 30 | BET | 1.0 | 300 |
| Oxidized biochar obtained from pine needles40 | 5.4 | 4 | 150 | 25 | L, F | 1.67 | 50 |
| Oxidized carbon derived from Luffa cylindrica41 | 59.9 | 4 | 80 | 25 | L, F | 1.67 | 45 |
| Organically modified saponites42 | 88.0 | 6 | 240 | 25 | L, F | 10.0 | 20 |
| Klinoptilolit43 | 1.7 | — | 1440 | 25 | L, F | 0.12 | 0.1 |
| Rice husk charcoal44 | 8.0 | 4 | 1440 | — | L, F | 1.0 | 70 |
| Babassu Coco activated carbon45 | 205.0 | 3 | 120 | 23 | L, F | 0.50 | 100 |
| Dende Coco activated carbon45 | 164.0 | 3 | 120 | 23 | L, F | 0.50 | 100 |
| Modified Babassu coconut activated carbon in inert atmosphere45 | 235.0 | 3 | 120 | 23 | L, F | 0.50 | 100 |
| Modified Dende coconut activated carbon in inert atmosphere45 | 165.0 | 3 | 120 | 23 | L, F | 0.50 | 100 |
| Modified coconut biomass activated carbon in inert atmosphere45 | 306.0 | 3 | 120 | 23 | L, F | 0.50 | 100 |
| Peach stone activated carbon46 | 260.0 | 6.3 | 120–180 | 30 | S | 2.4 | 100 |
| Oxidized peach stone activated carbon46 | 126.0 | 6.3 | 120–180 | 30 | S | 2.4 | 100 |
| Granular activated carbon47 | 88.0 | 6 | 40 | 25 | L, F, T | 0.05 | 15 |
| Carbon xerogels48 | 79.0 | 6.9 | 2880 | 30 | S | 3.0 | 100 |
| Graphene49 | 22.7 | 6.4 | 120 | 25 | L | 0.1 | 0.5 |
| Nickel modified inorganic–organic pillared clays50 | 5.5 | 6.5 | 480 | 25 | F | 6.67 | 14 |
| Pine activated carbon51 | 362.0 | 5 | 300 | 30 | L, F | 0.3 | 120 |
| Calcined bentonite52 | 41.7 | 2 | 1440 | 25 | L | 0.5 | 20 |
| Water hyacinth biochar53 | 2.48 | 2 | — | 25 | L, F | 2.0 | 5 |
| Fix-bed bentonite54 | 43.53 | — | — | 25 | — | — | 194.19 |
2.1. Effect of pH
The adsorbent surface charge and degree of ionization of the adsorbate molecules are affected by the pH of the solution, which is very important in the adsorption process.41,55,56 The adsorbent surface can be positively or negatively charged depending on the pH of the solution. In general, because the adsorbent surface is positively charged at acidic pH values, it is more suitable for negatively charged ions, and because the surface is negatively charged at high pH, it is more suitable for positively charged ions. Because caffeine is a weak electrolyte, its ionization is highly influenced by pH.56,57 It has a pH of 6.55 in nature.50 Adsorption experiments were carried out at various pH values in many studies, and the optimum pH value for the chosen adsorbent was discovered. The optimum pH is determined by the adsorbent's surface charge and the degree of ionization of the adsorbate molecules. Adsorption increases as long as the ionization of adsorbed molecules is prevented, and adsorption reaches its maximum when the molecules are not completely ionized.It is possible to see different pH values as optimum when evaluating various adsorbents used for caffeine removal. The results of the studies reviewed, however, show that caffeine is absorbed more efficiently in an acidic environment.17,40,41,44,45,50
When the pH of the solution is lower than the pH of the adsorbent with zero surface charge, the adsorbent becomes positively charged, which aids in the attraction of molecules in the electron donor.51 The hydrogen bonding forces between the adsorbent and the adsorbate explain the increase in caffeine removal efficiency at low pH values.58
Couto et al. investigated the effects of Babassu and Dende coconut activated carbons functionalized in an inert atmosphere on caffeine removal in relation to pH in a paper published in 2015. The results showed that 3 was the best working pH for this study.45 Grape stalk were used to remove caffeine in another study based on this one.17 The researchers compared three types of grape stalk: unfunctionalized grape stalk (GS), modified grape stalk with phosphoric acid (MGS), and activated carbon (GSAC) obtained from grape stalk. GS and MGS had the best pH at 2, while GSAC had the best pH at 4. Lenzi et al. used bentonite clay calcined at 773 K as an adsorbent in a study published in 2020. This study shows that caffeine is adsorbed more efficiently in an acidic environment, revealing that pH 2 is the best pH for the adsorption process.52
In the study published by W. A. Cabrera-Lafaurie et al. in 2012; investigated the effect of clays functionalized with transition metals and calcination on the caffeine removal, and as seen in Table 3, when the caffeine adsorption of different adsorbents at different pH conditions was evaluated, it was concluded that better efficiency was achieved in an acidic or neutral environment.50
| Ci (mg L−1) | |||
|---|---|---|---|
| Low pH | Normal pH | High pH | |
| Al–NaBt | 0.95 | 0.94 | 0.80 |
| AlOr–NaBt | 0.45 | 0.32 | 0.29 |
| AlOr–NaBt (partial calc.) | 0.82 | 0.85 | 0.06 |
| CoAlOr–NaBt (partial calc.) | 1.81 | 1.88 | 0.00 |
| CuAlOr–NaBt (partial calc.) | 1.79 | 1.88 | 0.00 |
| NiAlOr–NaBt (partial calc.) | 1.77 | 1.75 | 0.00 |
| CoAlOr–NaBt | 1.31 | 0.47 | 0.29 |
| CuAlOr–NaBt | 1.30 | 0.53 | 0.36 |
| NiAlOr–NaBt | 1.37 | 1.37 | 0.72 |
Anastopoulos et al., in their optimization study of caffeine adsorption with oxidized biochar obtained from pine needles in 2020, confirmed that the adsorption capacity increases with increases up to 4 in solution pH, and then begins to decrease after 4. In addition, another study confirming that caffeine adsorption occurs with higher efficiency in acidic medium by catching the highest adsorption efficiency at pH 4.40
Ngeno et al. in their study published in 2016, investigated the adsorption of caffeine on pyrolytically derived water hyacinth biochar and determined the highest pH value of qe (mg g−1). They concluded that it is possible in 2. They revealed that the qe value decreased at pH values after 2.53
The majority of studies have found that caffeine adsorption is more efficient in an acidic or neutral environment. Caffeine adsorption is relatively consistent in the pH range of 5–8, according to studies. Because hydroxyl ions, which may exist in higher concentrations, compete with caffeine for sorption to the acidic sites on the adsorbent at a more basic pH, a decrease in yield is expected.59
2.2. Effect of reaction time (adsorption equilibrium time)
It's a crucial parameter because it determines the adsorbate removal rate and adsorption equilibrium time during the reaction. At the beginning of the adsorption process, the adsorption rate is higher due to the empty pore structure of the adsorbent, and it decreases as the time progresses.60 The pore ratio in the adsorbent structure decreases as the number of molecules attached to the surface increases over time. The adsorption rate may vary depending on the material's adsorption capacity.61,62 With the decrease of the solvent accessible surface area, the adsorbent reaches the saturation value, and the adsorption takes place by inclusion, and the increased reaction time decreases the adsorption efficiency due to the smaller inner surface area. It can be said that the small increase in adsorption capacity over time is due to the increasing difficulty of occupying the vacant active sites due to the repulsive effects of the caffeine molecules on the adsorbed molecules.58Different optimum reaction times were determined in the studies listed in Table 2. Mohammed Danish studied the removal of caffeine from date stone using activated carbon, explaining that the adsorption efficiency increased rapidly for the first 15 minutes, then began to decline, and the adsorption reached equilibrium in 80 minutes. Initially, the abundance of empty spaces suitable for adsorption on the adsorbent surface and the amount of adsorbate caused the adsorption to be faster.37
Grape stalk (GS), grape stalk modified with phosphoric acid (MGS), and activated carbon (GSAC) obtained from grape stalk were used to adsorb caffeine in a study conducted by Portinho et al. in 2017. The equilibrium times of the reactions were found to be 40 minutes, 30 minutes, and 30 minutes, respectively, in experiments using three different types of adsorbent obtained from grape stalk. Later in the reaction, after reaching equilibrium, a small increase in adsorption capacity was observed. The caffeine molecules are attached to the active sites on the adsorbent at the start of the reaction. The small increase in adsorption capacity is due to the difficulties that arise because the caffeine molecules clinging to the pores create a repulsive force against the caffeine molecules in the solution that want to cling to the empty active sites.58 Furthermore, it is stated that the main reason MGS and GSAC reach equilibrium in a shorter time is because they have improved pore volume as a result of being treated, and GSAC has a high carbon content.63
In the studies carried out by Anastopoulos and Pashalidis in 2019, caffeine adsorption experiments were carried out using oxidized carbon derived from Luffa cylindrica. In the studies carried out, it was stated that the adsorption took place very quickly in the first 10 minutes and the adsorption rate decreased gradually with increasing contact time until equilibrium was reached after 80 minutes. It was determined that there was no significant change in the adsorption capacity in the trials made up to 240 min for contact times higher than 80 min. This result is due to the reduction of active pores on the adsorbent over time.41
Caffeine adsorption with 773 K bentonite reached equilibrium in 1440 minutes, according to Lenzi et al. They also looked into the effect of calcined bentonite in calcium alginate on the adsorption process, and discovered that compared to bentonite in suspension, caffeine adsorption was reduced by 19%.52
The adsorption equilibrium time was 40 hours, according to Oliveira et al., who used thermally modified Verdo-lodo bentonite as an adsorbent. The adsorption kinetics showed the best agreement with the pseudo second-order model.25 In just 24 hours, approximately 90% of the total caffeine adsorbed has been removed.
S. Álvarez et al. and colleagues synthesized carbon xerogel and investigated the effect of caffeine on adsorption in a study published in 2015. Caffeine adhered to the xerogel to a large extent in the first hour and then gradually decreased until the total removal capacity was reached after 48 hours, according to the findings. The pore structure is known to be an important parameter in the adsorption process. The mesoporous carbon xerogel used in the study was compared to microporous materials tested in a previous study, such as F-400 activated carbon.23
2.3. Effect of ambient temperature
The ambient temperature, which is another factor affecting the adsorption, affects the adsorption rate. The dependence of adsorption on temperature is either endothermic or exothermic.Table 2 shows the optimum reaction temperature values determined in previous caffeine adsorption studies. When the studies were examined, it was seen that the reaction showed endothermic reaction characteristics in some studies and exothermic reaction characteristics in some studies.
Among the studies carried out using different adsorbents in Table 2, the ability of water hyacinth biochar produced by the pyrolytic method to retain the caffeine molecules in the solution was evaluated at different temperatures. It was found that increasing the reaction temperature from 298 K to 338 K decreased the adsorption efficiency from 86.05% to 59.75%.53
In the article published by Anastopoulos and Pashalidis in 2019, in the adsorption studies performed during the adsorption of caffeine on oxidized carbon derived from Luffa cylindrica, the highest adsorption capacity was 298 K, qmax = 59.9 mg g−1, and the lowest adsorption capacity was 323 K they also obtained qmax = 49.3 mg g−1. These results confirm that the adsorption is exothermic.41
The maximum adsorption capacities were found to be 5.4 mg g−1, 6.5 mg g−1, and 6.26 mg g−1, respectively, in the adsorption study carried out by Anastopoulos et al. in 2020, using biochar obtained by the oxidation of pine needles selected for caffeine adsorption. When these results are analyzed, it is clear that as the temperature rises from 298 K to 313 K, the adsorption capacity increases slightly, but as the temperature rises to 323 K, the adsorption capacity decreases. In contrast to the study they carried out in 2019, the data obtained from this study reveal that the adsorption that took place was endothermic and entropy-induced.40
The adsorption efficiency of caffeine adsorption with activated carbon obtained from the date stone increased as the ambient temperature increased from 298 K to 323 K, according to a study published by Mohammed Danish in 2020. With the increase in temperature, the adsorption efficiency, which was around 70% at 298 K, increased to 78 percent. The adsorption capacity increased from around 9.11 mg g−1 to 10.18 mg g−1. The fact that caffeine adsorption increases with increasing temperature indicates that this is an endothermic reaction.37
When the examined caffeine adsorption studies are evaluated, the results obtained confirm that the adsorption capacity can change whether the adsorption process is exothermic or endothermic. In some of the studies carried out, the decrease in the adsorption capacity with the increase in temperature confirms that the caffeine adsorption process is an exothermic reaction.40,41,53
There are studies in the literature that show that when caffeine is physically absorbed, its capacity for adsorption diminishes with temperature.41,53,64,65 When the surface is highly functionalized with oxygen, however, the adsorption capacity may increase with temperature due to chemical absorption of caffeine.37,40 Temperature has a significant impact on both selectivity and adsorption capacity.66
The solubility is influenced by the solute's concentration and the system's temperature. The solute's solubility can be increased by raising the ambient temperature.67 Caffeine is partially polar. The polarity of the molecule is largely influenced by the two carbonyl groups and the single electron pair on the nitrogen. Caffeine is thus soluble in both water and polar organic solvents, but significantly less so in non-polar solvents.68
The degree of adsorption of the solute and its solubility in the solvent where adsorption occurs, on the other hand, generally have an inverse relationship. Because the higher the solute solubility, the stronger the solute–solvent bond and the less the adsorption.69 However, as the temperature increases, caffeine becomes more soluble in water. In this case, it is concluded that adsorption can be positively affected by the increase in ambient temperature.68
At lower temperatures, physisorption is usually the dominant phenomenon, and the amount of solute removed by adsorption decreases as temperature rises. Chemisorption becomes much more dominant at higher temperatures, and the amount of adsorbed material begins to increase with temperature.25
2.4. Determination of equilibrium adsorption isotherms
The differences between the isotherm models are critical for selecting a theoretical model that is compatible with experimental data and allows for the calculation of the materials' adsorption capacity.The Langmuir isotherm is an isotherm model in which the adsorbent's surface is homogeneous and a monolayer surface coating forms without adsorbed molecules interacting. On the other hand, the Freundlich isotherm model is an adsorption isotherm in which a heterogeneous surface is formed and molecular interactions between adsorbate molecules are observed.
The Toth isotherm is an isotherm used in the definition of adsorption systems using heterogeneous adsorbents. It was developed from the Langmuir isotherm.
Another name for the Sips isotherm is the Langmuir–Freundlich isotherm. It exhibits the Freundlich isotherm characteristics at low adsorbate concentrations and the Langmuir isotherm characteristics with monolayer capacity at high concentrations.70
When Table 2 is examined, it is concluded that most of the adsorption studies are in agreement with the Langmuir and Freundlich isotherm models.
The Sips isotherm was found to be the best match in caffeine adsorption with grape stalk in a study published by Portinho et al. in 2017. The best match is obtained with the Sips isotherm in all three studies: grape stalk (GS), phosphoric acid modified grape stalk (MGS), and grape stalk activated carbon (GSAC). The Freundlich isotherm has been found to be inconsistent because of its inconsistency at high pollutant concentrations, whereas the Langmuir isotherm has incompatibility due to the homogeneity of the surface.17
The experimental data of caffeine adsorption is best explained by the Sips adsorption isotherm, according to a study published by Silvia Álvarez Torrellas et al. in 2015. Because the target compound's adsorption isotherm has a sigmoidal profile, standard models like the Langmuir or Freundlich equations have been inadequate.46
Another study by Silvia Álvarez et al., with carbon xerogel, determined that the most suitable isotherm for caffeine adsorption was the Sips isotherm. The explanation of the Sips equation, which is more complex due to the heterogeneity of the solid surface at this point, is the reason why the Sips model shows the best fit.48
According to the studies in the literature, the adsorption capacity values obtained from the Langmuir and Sips models are close to the experimental adsorption capacity values. Therefore, it can be inferred from the data that a homogeneous surface is used for caffeine adsorption.36,71,72
In addition, when the kinetic models calculated as a result of the experimental data of caffeine adsorption studies using different adsorbents were examined, it was determined that most of the studies were compatible with the pseudo second order kinetic model.40,41,45
2.5. Effect of water hardness
Couto et al. investigated the effects of water hardness on caffeine removal with coconut activated carbons from various families in a study published in 2015.45 Caffeine retention tendencies of functionalized activated carbons in an inert atmosphere were investigated at various water hardnesses, and the results revealed that the caffeine molecules' ability to adhere to the adsorbent gradually decreases as the hardness increases. The increase in the solubility of caffeine in parallel with the increase in water hardness is the most important reason for this result. Ca2+ and Mg2+ are ions that contribute significantly to the hardness of water. In the experiments, adsorption efficiency was investigated in water containing 0 mg L−1 (W0), 80 mg L−1 (W80), 200 mg L−1 (W200) CaCO3, respectively, deionized water, medium hard water and hard water, using water with different levels of hardness.In Table 4, the adsorption amounts of functionalized coconut activated carbons depending on the water hardness were examined.
| Activated carbon sample | Water hardness | Qeq. cal. (mg g−1) |
|---|---|---|
| BI | 0 | 100.20 |
| BI | 80 | 83.28 |
| BI | 200 | 80.78 |
| DI | 0 | 92.96 |
| DI | 80 | 82.07 |
| DI | 200 | 79.76 |
| NI | 0 | 107.30 |
| NI | 80 | 94.39 |
| NI | 200 | 92.28 |
When the values in Table 4 are examined, it confirms the decrease in caffeine yield as the water hardness increases. This is due to the tendency of free ions such as Ca2+ and Mg2+ in hard water to compete with the adsorbent.
2.6. Attachment mechanism
It's crucial to understand how caffeine binds to the adsorbent surface in order to explain the adsorption mechanism. The presence of some functional groups and properties such as ionization, in particular, affect the adsorbate's binding mechanism to the adsorbent. However, some constraints must be overcome, such as particle size and crystal morphology, where the lack of functional groups on the surface reduces adsorbate molecule removal efficiency.73The majority of studies have found that caffeine adsorption is more efficient in an acidic or neutral environment. Caffeine adsorption is relatively consistent in the pH range of 5–8, according to studies. At a more alkaline pH, higher concentrations of hydroxyl ions compete with caffeine for sorption to acidic sites (phosphate groups) on the adsorbent.59
According to the researchers, while caffeine adsorption takes place between pH 2–7, π–π interactions and hydrogen bonds are responsible for the adsorption of adsorbate molecules to the adsorbent surface, while a decrease in surface adhesion is observed in the 8–9 range after pH 7. This is due to the repulsive forces between the caffeine molecules and the adsorbent with a negative surface charge. Caffeine is generally positively charged when the pH is lower than 8 and therefore attaches to the negatively charged adsorbent surface via electrostatic interactions.18
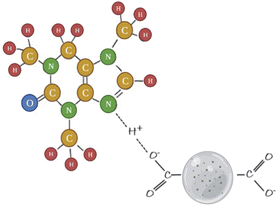
Caffeine is known to have a high dipole moment.74 The positively charged nitrogen atom in the caffeine molecule forms an electrostatic bond with the adsorbent's negatively charged functional groups.75,76 This electrostatic attraction is due to the dipole moment.77
On the carbon surface of carbonaceous materials, there are many polar groups with hydrophilic behavior, such as –NH, –OH, –O, and –COO. As a result, caffeine adsorption can occur through dipole–dipole interactions, in which the π-electrons and 2-nitrophenol aromatic rings of caffeine interact with π-aromatic electrons on the adsorbent surface.36,49,78
Furthermore, hydroxyl groups (OH) are the most dominant bonds between adsorbents and caffeine in coffee husk and caffeine biosorption, followed by carboxyl groups (COOH). Coffee husk biochar meets this requirement thanks to a large number of polar groups with hydrophilic behavior, such as –NH, –OH, –O, and –COO.43 As a result, caffeine adsorption can occur through dipole–dipole interactions, in which the π-electrons and 2-nitrophenol aromatic rings of caffeine interact with aromatic electrons on the biochar adsorbent surface.79
A dipole–dipole interaction occurs between the deprotonated caffeine molecule and the protonated groups on the surface of carbonaceous materials when the pH is below the pKa of caffeine.33
There is a continuous decrease in the adsorption efficiency with the increase of the ionic strength of the solutions, indicating that the adsorption of caffeine by the adsorbent is based on pure electrostatic interactions. The results of caffeine adsorption studies carried out with biochar obtained from pine needles revealed that the negative carbosilicate parts on the adsorbent surface at pH 4 and the positively charged amino groups of caffeine molecules are active sites, due to the effect of ionic forces, and that caffeine adsorption occurs through electrostatic attraction.40
The electrostatic attraction between the negatively charged carbosilicates on the adsorbent surface and the positively charged amino groups of caffeine is depicted in Fig. 3.
2.7. Adsorbent and adsorbate concentration
The efficiency of the adsorbents selected for adsorbing different adsorbate materials is very important to determine the optimum amount of adsorbent and to prevent unnecessary adsorbent waste. Each adsorption event has its own optimum adsorbent concentration. In addition, determining the optimum adsorbent dose is one of the important ways to reduce the cost in the adsorption process, and makes the adsorption process more attractive.Table 2 lists a number of previous studies on caffeine adsorption, which is the focus of this research. For each study, different optimum conditions were determined.
Adsorbents have different ability to adsorb various active substances. If the material used in the adsorption process and the adsorbed material are in harmony with each other, the efficiency will increase. In general, high molecular weight substances adsorb more easily than low molecular weight substances (the larger the molecule, the greater the van der Waals force). Each adsorbent has a maximum adsorbate holding capacity.
When Table 2 is examined, it is clear that the adsorption capacities of activated carbons prepared from different types of coconut shells modified in an inert atmosphere have radically different values. The material structure influences the capacities of different starting materials sintered in the same environment.45
While preparing the adsorbent, the selected material, sintering temperature, time and atmosphere are effective parameters on the properties and capacity of the final product18,25,39,45
The initial adsorbent concentrations chosen by the researchers and, as a result, the change in adsorption capacity (qm) per unit adsorbant were investigated in the studies listed in Table 2. Thus, the ideal amount of adsorbent for caffeine adsorbent can be compared.
3. Conclusion and discussion
In this review, adsorbents that have been effectively used in caffeine adsorption until now have been grouped together based on the parameters that affect the adsorption process, such as pH, contact time, temperature, adsorbate concentration, and water hardness.As a result of the researches, the following data was obtained:
• Caffeine is adsorbed more efficiently in an acidic environment.
• Many studies usually give effective results at room temperature (298 K).
• The maximum adsorption capacity values vary in a wide range such as 5 to 395 mg g−1.
• In addition, among the equilibrium adsorption isotherms studied, the Langmuir isotherm model showed the best agreement with the experimental data obtained in caffeine adsorption in most studies.
• The binding mechanism is that the negative carbosilicate parts on the adsorbent surface and the positively charged amino groups of caffeine molecules are active sites, and caffeine adsorption also occurs through electrostatic attraction.
Conflicts of interest
The authors declare no intellectual as well as financial conflict of interests.Acknowledgements
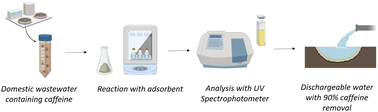
The authors would like to thank Yıldız Technical University BAPKA project no. FLY-2021-4619 for financial support of this work. Caffeine removal by adsorption was demonstrated using https://BioRender.com.References
- T. Malchi, Y. Maor, G. Tadmor, M. Shenker and B. Chefetz, Environ. Sci. Technol., 2014, 48, 9325–9333 CrossRef CAS PubMed.
- M. Taheran, M. Naghdi, S. Brar, M. Verma and R. Surampalli, Environ. Nanotechnol. Monit. Manag., 2018, 10, 122–126 Search PubMed.
- V. Lakshmi and N. Das, Removal of caffeine from industrial wastewater using Trichosporon asahii, J. Environ. Biol., 2013, 34(4), 701 CAS.
- T. Deblonde, C. Cossu-Leguille and P. Hartemann, Int. J. Hyg. Environ. Health, 2011, 214, 442–448 CrossRef CAS PubMed.
- S. Bachmann, T. Calvete and L. Féris, Sci. Total Environ., 2021, 767, 144229 CrossRef CAS.
- M. Asghar, Q. Zhu, S. Sun, Y. Peng and Q. Shuai, Sci. Total Environ., 2018, 635, 828–837 CrossRef CAS PubMed.
- C. Willson, Toxicol. Rep., 2018, 5, 1140–1152 CrossRef CAS PubMed.
- K. Fent, A. Weston and D. Caminada, Aquat. Toxicol., 2006, 76, 122–159 CrossRef CAS.
- A. Palo and R. Choudhury, Environ. Toxicol. Pharmacol., 2006, 21, 254–259 CrossRef CAS PubMed.
- I. Buerge, T. Poiger, M. Müller and H. Buser, Environ. Sci. Technol., 2003, 37, 691–700 CrossRef CAS PubMed.
- W. Li, Environ. Pollut., 2014, 187, 193–201 CrossRef CAS.
- S. Li, B. He, J. Wang, J. Liu and X. Hu, Chemosphere, 2020, 243, 125343 CrossRef CAS.
- R. Dafouz, N. Cáceres, J. Rodríguez-Gil, N. Mastroianni, M. López de Alda, D. Barceló, Á. de Miguel and Y. Valcárcel, Sci. Total Environ., 2018, 615, 632–642 CrossRef CAS PubMed.
- B. Özpalas and E. Özer, Nevsehir Bilim ve Teknol. Derg., 2017, 6, 297–305 CrossRef.
- O. Hillebrand, K. Nödler, T. Licha, M. Sauter and T. Geyer, Water Res., 2012, 46, 395–402 CrossRef CAS.
- K. Knee, R. Gossett, A. Boehm and A. Paytan, Mar. Pollut. Bull., 2010, 60, 1376–1382 CrossRef CAS.
- R. Portinho, O. Zanella and L. Féris, J. Environ. Manage., 2017, 202, 178–187 CrossRef CAS PubMed.
- K. Beltrame, A. Cazetta, P. de Souza, L. Spessato, T. Silva and V. Almeida, Ecotoxicol. Environ. Saf., 2018, 147, 64–71 CrossRef CAS.
- E. S. Gonçalves, S. V. Rodrigues and E. V. D. Silva-Filho, Rev. Ambiente Agua, 2017, 12, 192–202 CrossRef.
- S. Kurissery, N. Kanavillil, S. Verenitch and A. Mazumder, Ecol. Indic., 2012, 23, 501–508 CrossRef CAS.
- W. Warner, T. Licha and K. Nödler, Sci. Total Environ., 2019, 686, 75–89 CrossRef CAS PubMed.
- K. Katam, T. Shimizu, S. Soda and D. Bhattacharyya, Sci. Total Environ., 2020, 707, 135987 CrossRef CAS.
- J. Sotelo, A. Rodríguez, S. Álvarez and J. García, Chem. Eng. Res. Des., 2012, 90, 967–974 CrossRef CAS.
- F. Al-Qaim, Z. Mussa, M. Othman and M. Abdullah, J. Hazard. Mater., 2015, 300, 387–397 CrossRef CAS PubMed.
- M. Oliveira, M. da Silva and M. Vieira, Appl. Clay Sci., 2019, 168, 366–373 CrossRef CAS.
- J. Temple, J. Am. Acad. Child Adolesc. Psychiatry, 2019, 58, 36–45 CrossRef PubMed.
- A. Sobstyl, M. Dobosz, A. Chałupnik, Z. Chilimoniuk, A. Borkowska and H. Szczęsna, National Scientific e-Conference, Lodz, 2020, p. 80 Search PubMed.
- M. Diego, T. Field, M. Hernandez-Reif, Y. Vera, K. Gil and A. Gonzalez-Garcia, J. Child Adolesc. Subst. Abuse, 2008, 17, 41–49 CrossRef PubMed.
- L. Shilo, H. Sabbah, R. Hadari, S. Kovatz, U. Weinberg, S. Dolev, Y. Dagan and L. Shenkman, Sleep Med., 2002, 3, 271–273 CrossRef.
- A. Wilcox, C. Weinberg and D. Baird, Lancet, 1988, 332, 1453–1456 CrossRef.
- Taking sides: Clashing views on controversial issues in drugs and society, R. Goldberg, Dushkin Publishing Group/Brown & Benchmark Publishers, 1996 Search PubMed.
- S. Heatherley, K. Hancock and P. Rogers, J. Child Psychol. Psychiatry, 2006, 47, 135–142 CrossRef PubMed.
- M. N. Rigueto, C. De Souza, J. Cadore, V. Brião and J. Piccin, J. Water Process Eng., 2020, 35, 101231 CrossRef.
- M. Ahmaruzzaman, Adv. Colloid Interface Sci., 2008, 143, 48–67 CrossRef CAS PubMed.
- S. Dhaka, R. Kumar, A. Deep, M. Kurade, S. Ji and B. Jeon, Coord. Chem. Rev., 2019, 380, 330–352 CrossRef CAS.
- I. Anastopoulos, I. Pashalidis, A. Orfanos, I. Manariotis, T. Tatarchuk, L. Sellaoui, A. Bonilla-Petriciolet, A. Mittal and A. Núñez-Delgado, J. Environ. Manage., 2020, 261, 110236 CrossRef CAS PubMed.
- M. Danish, Mater. Today: Proc., 2020, 31, 18–22 CAS.
- M. Francoeur, A. Ferino-Pérez, C. Yacou, C. Jean-Marius, E. Emmanuel, Y. Chérémond, U. Jauregui-Haza and S. Gaspard, J. Environ. Chem. Eng., 2021, 9, 104795 CrossRef CAS.
- A. d. S. V. de Almeida, W. Vieira, M. Bispo, S. de Melo, T. da Silva, T. Balliano, M. Vieira and J. Soletti, J. Environ. Chem. Eng., 2021, 9, 104891 CrossRef.
- I. Anastopoulos, A. Katsouromalli and I. Pashalidis, J. Mol. Liq., 2020, 304, 112661 CrossRef CAS.
- I. Anastopoulos and I. Pashalidis, J. Mol. Liq., 2019, 296, 112078 CrossRef CAS.
- L. Marçal, E. de Faria, E. Nassar, R. Trujillano, N. Martín, M. Vicente, V. Rives, A. Gil, S. Korili and K. Ciuffi, ACS Appl. Mater. Interfaces, 2015, 7, 10853–10862 CrossRef PubMed.
- M. Leal, V. Martínez-Hernández, R. Meffe, J. Lillo and I. de Bustamante, Chemosphere, 2017, 175, 534–542 CrossRef CAS PubMed.
- M. Bernardo, C. Madeira, N. dos Santos Nunes, D. Dias, D. Godinho, M. de Jesus Pinto, I. do Nascimento Matos, A. Carvalho and I. de Figueiredo Ligeiro Fonseca, Environ. Sci. Pollut. Res., 2017, 24, 22698–22708 CrossRef CAS PubMed.
- O. Couto, I. Matos, I. da Fonseca, P. Arroyo, E. da Silva and M. de Barros, Can. J. Chem. Eng., 2014, 93, 68–77 CrossRef.
- S. Á. Torrellas, R. G. Lovera, N. Escalona, C. Sepúlveda, J. L. Sotelo and J. García, Chem. Eng. J., 2015, 279, 788–798 CrossRef CAS.
- A. Gil, N. Taoufik, A. García and S. Korili, Environ. Technol., 2018, 40, 3017–3030 CrossRef PubMed.
- S. Álvarez, R. Ribeiro, H. T. Gomes, J. L. Sotelo and J. García, Chem. Eng. Res. Des., 2015, 95, 229–238 CrossRef.
- G. Yang and P. Tang, Water Sci. Technol., 2016, 73, 2268–2274 CrossRef CAS PubMed.
- W. A. Cabrera-Lafaurie, F. R. Román and A. J. Hernández-Maldonado, J. Colloid Interface Sci., 2012, 386, 381–391 CrossRef CAS PubMed.
- M. Galhetas, A. Mestre, M. Pinto, I. Gulyurtlu, H. Lopes and A. Carvalho, J. Colloid Interface Sci., 2014, 433, 94–103 CrossRef CAS PubMed.
- G. G. Lenzi, M. E. K. Fuziki, M. Z. Fidelis, Y. B. Fávaro, M. A. Ribeiro, E. S. Chaves and E. K. Lenzi, Braz. Arch. Biol. Technol., 2020, 63, 1–10 Search PubMed.
- E. Ngeno, F. Orata, L. Baraza, V. Shikuku and S. Kimosop, J. Chem. Chem. Eng., 2016, 10, 185–194 CAS.
- M. Oliveira, V. de Souza, M. da Silva and M. Vieira, Ind. Eng. Chem. Res., 2018, 57, 17480–17487 CrossRef CAS.
- M. Yagub, T. Sen, S. Afroze and H. Ang, Adv. Colloid Interface Sci., 2014, 209, 172–184 CrossRef CAS PubMed.
- C. Barbas, A. García, L. Saavedra and M. Castro, J. Chromatogr. A, 2000, 870, 97–103 CrossRef CAS.
- A. Rao, A. Kumar, R. Dhodapkar and S. Pal, Environ. Sci. Pollut. Res., 2021, 28, 21347–21358 CrossRef CAS PubMed.
- J. Sotelo, A. Rodríguez, M. Mateos, S. Hernández, S. Torrellas and J. Rodríguez, J. Environ. Sci. Health, Part B, 2012, 47, 640–652 CrossRef CAS PubMed.
- S. Ravi, Y. Choi and J. Choe, Chem. Eng. J., 2020, 379, 122290 CrossRef CAS.
- L. Ahmad, L. Linh, M. Akimoto, Y. Kaneki, M. Honda, M. Suda and K. Kunimoto, Food Sci. Technol. Res., 2013, 19, 697–703 CrossRef CAS.
- S. Gupta and A. Kumar, Appl. Water Sci., 2019, 9(4), 1–11 Search PubMed.
- D. Xu, C. Gu and X. Chen, Procedia Environ. Sci., 2013, 18, 127–134 CrossRef CAS.
- A. Deiana, M. Sardella, H. Silva, A. Amaya and N. Tancredi, J. Hazard. Mater., 2009, 172, 13–19 CrossRef CAS PubMed.
- M. Bahrami, M. Amiri, M. Mahmoudi and S. Koochaki, J. Water Health, 2017, 15, 526–535 CrossRef PubMed.
- L. Al-Khateeb, S. Almotiry and M. Salam, Chem. Eng. J., 2014, 248, 191–199 CrossRef CAS.
- A. Gotzias, E. Kouvelos and A. Sapalidis, Surf. Coat. Technol., 2018, 350, 95–100 CrossRef CAS.
- Q. Vuong and P. Roach, Sep. Purif. Rev., 2013, 43, 155–174 CrossRef.
- Q. Edwards, S. Kulikov, L. Garner-O'Neale and I. Lunat, Int. J. Chem. Sci., 2015, 13(3), 1218–1226 CAS.
- C. van Oss, Interface Sci. Technol., 2008, 85–111 Search PubMed.
- L. Melo, A. Ide, J. Duarte, C. Zanta, L. Oliveira, W. Pimentel and L. Meili, Environ. Sci. Pollut. Res., 2020, 27, 27048–27060 CrossRef CAS PubMed.
- S. Sharma, P. Nandal and A. Arora, Waste Biomass Valorization, 2018, 10, 3427–3434 CrossRef.
- T. Medina-Espinosa, C. Asimbaya, S. Galeas, N. Rosas-Laverde, A. Debut and V. Guerrero, IOP Conf. Ser.: Earth Environ. Sci., 2021, 897, 012020 CrossRef.
- A. Benjamine, B. Lucie, Y. Denis and B. Sawaliho, Comput. Chem., 2019, 07, 95–105 CrossRef CAS.
- S. Keerthanan, A. Bhatnagar, K. Mahatantila, C. Jayasinghe, Y. Ok and M. Vithanage, Environ. Technol. Innov., 2020, 19, 100847 CrossRef.
- M. Ptaszkowska-Koniarz, J. Goscianska and R. Pietrzak, Chem. Eng. J., 2018, 345, 13–21 CrossRef CAS.
- K. Yamamoto, T. Shiono, Y. Matsui and M. Yoneda, Part. Sci. Technol., 2018, 37, 325–332 CrossRef.
- J. Bedia, M. Peñas-Garzón, A. Gómez-Avilés, J. Rodriguez and C. Belver, C, 2018, 4, 63 CAS.
- T. Fraga, M. Carvalho, M. Ghislandi and M. Motta Sobrinho, Brazilian J. Chem. Eng., 2019, 36, 1–31 CrossRef CAS.
- M. H. Rodiguez, J. Yperman, R. Carleer, J. Maggen, D. Dadi, G. Gryglewicz, B. Van der Bruggen, J. F. Hernández and A. O. Calvis, J. Environ. Chem. Eng., 2018, 6, 1161–1170 CrossRef.
| This journal is © The Royal Society of Chemistry 2022 |