Open Access Article
Open Access ArticleEfficient synthesis of cyclic amidine-based fluorophores via 6π-electrocyclic ring closure†
Guofeng
Li‡
 a,
Man
Zhao‡
a,
Junqiu
Xie‡
b,
Ying
Yao
a,
Lingyun
Mou
b,
Xiaowei
Zhang
b,
Xiaomin
Guo
b,
Wangsheng
Sun
a,
Man
Zhao‡
a,
Junqiu
Xie‡
b,
Ying
Yao
a,
Lingyun
Mou
b,
Xiaowei
Zhang
b,
Xiaomin
Guo
b,
Wangsheng
Sun
 b,
Zheng
Wang
b,
Zheng
Wang
 c,
Jiecheng
Xu
a,
Jianzhong
Xue
a,
Tao
Hu
a,
Ming
Zhang
b,
Min
Li
*a and
Liang
Hong
c,
Jiecheng
Xu
a,
Jianzhong
Xue
a,
Tao
Hu
a,
Ming
Zhang
b,
Min
Li
*a and
Liang
Hong
 *a
*a
aGuangdong Key Laboratory of Chiral Molecular and Drug Discovery, School of Pharmaceutical Sciences, Sun Yat-Sen University, Guangzhou 510006, China. E-mail: limin65@mail.sysu.edu.cn; hongliang@sysu.edu.cn
bKey Laboratory of Preclinical Study for New Drugs of Gansu Province, School of Basic Medical Sciences, Lanzhou University, Lanzhou 730000, China
cGuangdong Key Lab of Nano-Micro Material Research, School of Chemical Biology and Biotechnology, Peking University Shenzhen Graduate School, Shenzhen 518055, China
First published on 13th March 2020
Abstract
Novel 10π-electron cyclic amidines with excellent fluorescence properties were synthesized by a general and efficient 6π-electrocyclic ring closure of ketenimine and imine starting from N-sulfonyl triazoles and arylamines. The photophysical properties of cyclic amidine fluorophores have been studied in detail and have shown good properties of a large Stokes shift, pH insensitivity, low cytotoxicity and higher photostability, which have great potential for biological imaging. Furthermore, this novel fluorophore was successfully applied to the localization of the NK-1 receptor in living systems.
Introduction
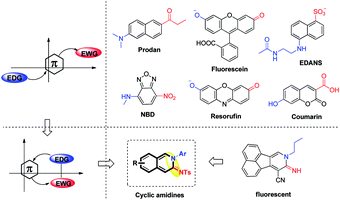
Small organic fluorophores have been used widely in biological science and drug discovery due to their easy handling, high sensitivity and minimal disruption to living systems.1 Although a large variety of fluorescent small molecules have been developed, their core scaffolds are still limited to several types such as prodan, fluorescein, coumarin etc.,2 most of which usually possess a π-conjugated system with an electron-donating and an electron-withdrawing group located at the opposite position of the fluorescent moiety (Fig. 1).3 On the other hand, the ideal fluorophores for living systems with good photophysical properties including a large Stokes shift, an applicable pH environment, good stability and low biological toxicity are in high demand. Consequently, the development of small organic fluorophores with a new framework and good photophysical properties is an appealing and challenging task.4To develop a new fluorescent framework, we wondered whether it is possible to incorporate an electron-donating and an electron-withdrawing group at the adjacent position of a π-system. Considering the particularity of this structure, we tried to choose π-system cyclic amidines as the core structure of novel fluorophores, not only due to their potent fluorescence properties,5 but also due to the electronic specificity of amidines (Fig. 1). However, how to incorporate an amidine into a π-system is a challenge, since the synthesis of such a scaffold is limited and difficult. Moreover, it usually needs a variety of cyclic amidines with structural diversity to study the fluorescence–structure relationship. Thus, it is necessary yet challenging to develop a simple and short synthetic route to access various cyclic amidines.
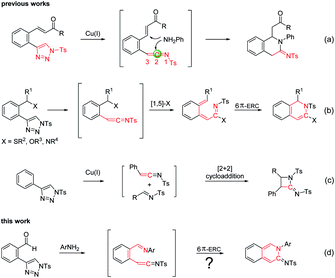
N-Sulfonyl-1,2,3-triazoles have become an important class of intermediates for accessing a wide variety of complex molecular scaffolds.6 Metal-bound imino carbenes, readily generated from N-sulfonyl 1,2,3-triazoles, have found wide application in many useful transformations, including cycloaddition, X–H insertion, alkyl migration, sigmatropic rearrangement and some other carbine induced reactions.6,7 In addition to imino carbenes, N-sulfonyl-1,2,3-triazoles could also form active ketenimine intermediates, which would readily undergo nucleophilic additions at C2.8 In particular, the ketenimines containing another electrophilic site would undergo double nucleophilic addition with amines to give cyclic amidines (Scheme 1a).9 Moreover, ketenimines could also in whole or in part participate in some cyclization reactions. For example, in the [1,5]-X sigmatropic shift/6π-electrocyclic ring closure (6π-ERC), the whole ketenimines (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NTs) take part in the reaction to give dihydroisoquinolines (Scheme 1b),8f,10 while part of the ketenimines (C
NTs) take part in the reaction to give dihydroisoquinolines (Scheme 1b),8f,10 while part of the ketenimines (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) in the [2 + 2] cycloadditions reacts to give amidines (Scheme 1c).11 From these studies, we found that cyclic amidines could be constructed by the intramolecular conjunction of a nitrogen atom to the C2 of the ketenimine. Accordingly, we designed the intermediate A functionalized with an adjacent imine and ketenimine. The imine bond (C
C) in the [2 + 2] cycloadditions reacts to give amidines (Scheme 1c).11 From these studies, we found that cyclic amidines could be constructed by the intramolecular conjunction of a nitrogen atom to the C2 of the ketenimine. Accordingly, we designed the intermediate A functionalized with an adjacent imine and ketenimine. The imine bond (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NAr) was introduced for the formation of 6π-substrates suitable for ERC and might react with part of the ketenimines to give cyclic amidine products (Scheme 1d).
NAr) was introduced for the formation of 6π-substrates suitable for ERC and might react with part of the ketenimines to give cyclic amidine products (Scheme 1d).
Results and discussion
With this reaction design in mind, we first investigated the reaction of N-sulfonyl triazoles 1a with phenylamine 2a as the model reaction in the presence of a catalyst at 120 °C in a sealed tube for 2 h (Table 1). A low yield of 17% was observed in the presence of a CuI catalyst (entry 1). We also examined some other catalysts including Cu(OAc)2, CuTc, Rh2(OAc)4, Rh2(Oct)4 and Pd(OAc)2, and they gave yields ranging from 12% to 54% (entries 2–6). Interestingly, when the reaction was conducted in the absence of any catalyst, the reaction could give a significant improvement of the yield to 83% (entry 7). Increasing the reaction time to 4 h could further improve the yield to 91% (entry 8), but further increasing the reaction time to 10 h reduced the yield to 86% (entry 9). After screening several solvents, CHCl3 was found to be the optimum solvent for this reaction (entries 8–13).| Entry | Cat. | Solvent | t (h) | Yieldb (%) |
|---|---|---|---|---|
| a Conditions: 1a (0.10 mmol, 1.0 equiv.), 2a (0.12 mmol, 1.2 equiv.), the catalyst (0.005 mmol, 5 mol%), and solvent (2.0 mL) in a sealed tube. b Isolated yield. | ||||
| 1 | CuI | CHCl3 | 2 | 17 |
| 2 | Cu(OAc)2 | CHCl3 | 2 | 26 |
| 3 | CuTC | CHCl3 | 2 | 46 |
| 4 | Rh2(OAc)4 | CHCl3 | 2 | 23 |
| 5 | Rh2(Oct)4 | CHCl3 | 2 | 54 |
| 6 | Pd(OAc)2 | CHCl3 | 2 | 54 |
| 7 | — | CHCl3 | 2 | 83 |
| 8 | — | CHCl 3 | 4 | 91 |
| 9 | — | CHCl3 | 10 | 86 |
| 10 | — | Toluene | 4 | 40 |
| 11 | — | DCE | 4 | 77 |
| 12 | — | THF | 4 | 30 |
| 13 | — | 1,4-Dioxane | 4 | 44 |
Under optimal conditions, the scopes of the reaction were examined as shown in Scheme 2. We first examined the scope of benzenamines with various substituents at different positions. The reaction proceeded efficiently to afford the corresponding cyclic amidines 3aa–3ar in good to excellent yields, though higher reactivity was observed with electron-donating groups. Significantly, the benzenamines with functional groups such as azide, hydroxy and carboxyl groups were well tolerated to give 3as–3au, which could be modified easily for the purpose of bioconjugate chemistry. Some other amines, including naphthyl amine, 2-aminopyridine and phenylhydrazine were also suitable substrates for the reaction, leading to the desired products 3av–3ax in moderate yields. The structure of 3aw was determined by X-ray crystallography.12 The scope of N-sulfonyl-1,2,3-triazoles was subsequently investigated. The reaction took place with moderate to excellent yields with substrates bearing different substituents 3ba–3be.
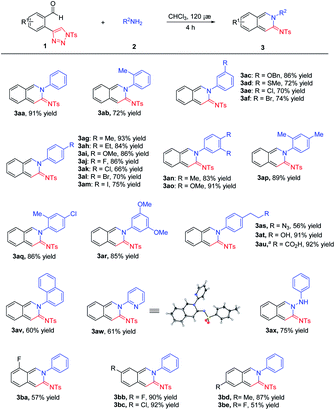 | ||
| Scheme 2 Scope of the reaction. Conditions: 1 (0.10 mmol, 1.0 equiv.), 2 (0.12 mmol, 1.2 equiv.) and the solvent (2.0 mL) in a sealed tube for 4 h at 120 °C. a1 mmol scale. | ||
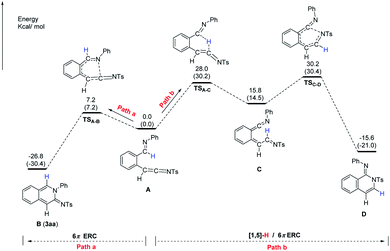
To further understand the formation of cyclic amidine 3aa, analysis by DFT calculations was performed. As shown in Fig. 2, starting from ketenimine intermediate A, there are two pathways to proceed. By path a, the intermediate A could undergo 6π-electrocyclic ring closure (6π-ERC) via TSA–B to give B (3aa); while by path b, the intermediate A could undergo the first [1,5]-H shift to form the intermediate C, followed by the 6π-ERC via TSC–D to afford the isoquinolone D. The energy for the formation of TSA–B (ΔG = 7.2 kcal mol−1) is lower than that for the TSC–D (ΔG = 30.2 kcal mol−1). Therefore, the intermediate A favors path a to furnish the observed product 3aa.
With a series of 10π-electron cyclic amidines in hand, we measured their photophysical properties including absorptions, emissions, extinction coefficients, Stokes shifts and quantum yields. As shown in Table 2, the maximum absorption wavelengths (λmax) varied from 424 to 443 nm with moderate extinction coefficients (2110–4364 M−1 cm−1), while the maximum emission wavelengths (λem) varied from 525 to 552 nm with quantum yields from 1% to 23%. In addition, cyclic amidines 3as–3au with functional groups exhibited similar photophysical properties, which could be conveniently modified for further fluorophore tagging.
| Compound | λ max (nm) | λ em (nm) | ε (M−1 cm−1) | Stokes shift (nm) | Φ (%) |
|---|---|---|---|---|---|
| a Measured in CH3CN at 200 μM. b Molar extinction coefficient. c Absolute fluorescence quantum yield determined with an integrating sphere system. d The structures of coumarin and fluorescein are shown in Fig. 1. | |||||
| Coumarin13d | 386 | 448 | 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
62 | 70 |
| Fluorescein3bd | 490 | 512 | 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
22 | 95 |
| 3aa | 433 | 535 | 2110 | 102 | 17 |
| 3ab | 432 | 527 | 3066 | 95 | 23 |
| 3ac | 434 | 542 | 3613 | 108 | 14 |
| 3ad | 434 | 538 | 3790 | 104 | 13 |
| 3ae | 436 | 543 | 3588 | 107 | 11 |
| 3af | 437 | 545 | 3440 | 108 | 12 |
| 3ag | 434 | 538 | 3558 | 104 | 16 |
| 3ah | 434 | 541 | 3430 | 107 | 17 |
| 3ai | 434 | 535 | 3312 | 101 | 19 |
| 3aj | 435 | 536 | 2770 | 101 | 15 |
| 3ak | 435 | 540 | 3209 | 105 | 14 |
| 3al | 436 | 542 | 3786 | 106 | 14 |
| 3am | 435 | 543 | 3418 | 108 | 12 |
| 3an | 433 | 536 | 4364 | 103 | 19 |
| 3ao | 434 | 537 | 3874 | 103 | 18 |
| 3ap | 432 | 525 | 2804 | 93 | 22 |
| 3aq | 434 | 530 | 3917 | 96 | 18 |
| 3ar | 433 | 539 | 3578 | 106 | 14 |
| 3as | 435 | 535 | 2915 | 100 | 18 |
| 3at | 434 | 538 | 3250 | 104 | 18 |
| 3au | 434 | 536 | 2935 | 102 | 17 |
| 3av | 434 | 532 | 3170 | 98 | 18 |
| 3aw | 437 | 552 | 3099 | 115 | 1 |
| 3ax | 425 | 528 | 3865 | 103 | 16 |
| 3ba | 440 | 542 | 2285 | 102 | 09 |
| 3bb | 443 | 550 | 3771 | 107 | 09 |
| 3bc | 443 | 547 | 2796 | 104 | 10 |
| 3bd | 427 | 536 | 3853 | 109 | 17 |
| 3be | 424 | 535 | 2458 | 111 | 12 |
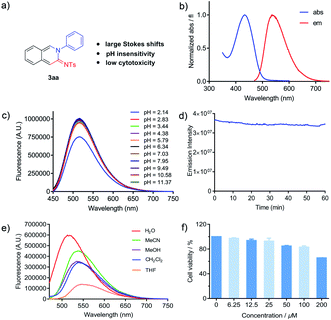
A good fluorophore for living systems has good properties including a remarkable Stokes shift, an applicable pH environment, high photostability and low biological toxicity. Keeping this in mind, we further examined these photophysical properties (Fig. 3). Based on the absorption and emission wavelengths in Table 2, all cyclic amidines exhibited remarkable Stokes shifts of ∼100 nm, which could minimize self-absorption and provide better fluorescence imaging. We next studied the effect of pH on cyclic amidine 3aa. The results showed that the pH value of the environment ranging from 2.8–11.4 had no effect on the emissive properties (Fig. 3c). In addition, amidine 3aa showed excellent resistance to photobleaching with only 3.4% fluorescence lost after one hour at the wavelength of maximum excitation (Fig. 3d).14 Subsequently, the impact of different solvent environments was investigated. As shown in Fig. 3e, 3aa worked well in aqueous environments, indicating that it could be used as a polarity probe. In addition, the emission intensity could be maintained in aqueous solution without loss after 24 h (see the ESI†). Moreover, cytotoxicity was another important factor for its application in living systems. We evaluated the cytotoxicity of 3aa by using CCK-8 assays for HeLa cells, and it exhibited no significant cytotoxicity at concentrations of up to 100 μM (Fig. 3f). These results demonstrated that the novel amidine fluorophore has good properties of a large Stokes shift, pH insensitivity, low cytotoxicity and good photostability, which has great potential for biological imaging.
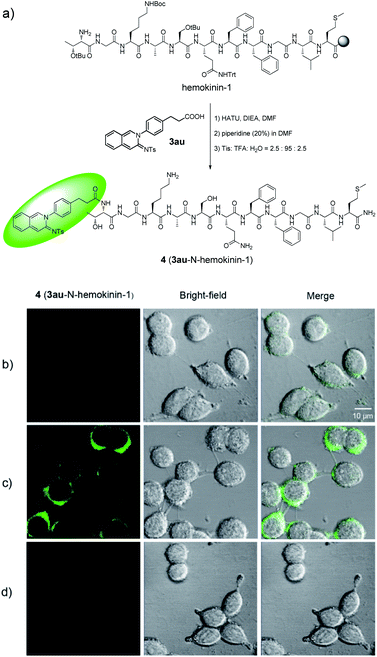
Ligand-based probes15 have received extensive attention and have extensive applications due to their high selectivity and affinity in the visualization of receptor–ligand interactions and drug evaluation. In order to evaluate the potential of cyclic amidines for cell imaging, we synthesized a fluorescent probe 4 by conjugating cyclic amidine 3au with the hemokinin-1 (HK-1) peptide (TGKASQFFGLM-NH2), which was highly selective to the NK-1 receptor (Fig. 4a).16 The neurokinin-1 (NK-1) receptor, as a member of the G-protein-coupled receptor (GPCR) family, is located at the cell membrane. If a fluorescent ligand binds to the NK-1 receptor, it would give fluorescence signals at the cell membrane. To test whether fluorescent probe 4 could bind to the NK-1 receptor, WT 22RV1 and NK-1-overexpressing 22RV1 cells were treated with 4 respectively. As Fig. 4 shows, green fluorescence could only be observed in the cell membrane of NK-1-overexpressing 22RV1, not in the cell membrane of WT 22RV1. Furthermore, the fluorescence could be blocked with aprepitant, a selective NK-1 receptor antagonist, indicating that the fluorescent ligand 4 was specifically bound to the NK-1 receptor. These preliminary results demonstrated that amidine fluorophores could be used as potential bioprobes.
Conclusions
In summary, we have developed a general and efficient 6π-electrocyclic ring closure of ketenimine and imine starting from N-sulfonyl triazoles and arylamines. The method provides expeditious access to a variety of 10π-electron cyclic amidines in moderate to excellent yields. Through a fluorescence–structure relationship study, we found that cyclic amidine fluorophores have the advantages of large Stokes shifts, pH insensitivity, low cytotoxicity and higher photostability. Furthermore, they can be used efficiently in developing new fluorescent probes for imaging in living systems.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We would like to dedicate this to Professor Albert S. C. Chan on the occasion of his 70th birthday. We are grateful for financial support from the NSFC (21871296 and 21907111), the Guangdong Natural Science Funds for Distinguished Young Scholars (2017A030306017) and the Fundamental Research Funds for the Central Universities (19ykpy128).Notes and references
- (a) J. Chan, S. C. Dodani and C. J. Chang, Nat. Chem., 2012, 4, 973–984 CrossRef CAS PubMed; (b) Q. Shao and B. Xing, Chem. Soc. Rev., 2010, 39, 2835–2846 RSC; (c) W. Sun, S. Guo, C. Hu, J. Fan and X. Peng, Chem. Rev., 2016, 116, 7768–7817 CrossRef CAS PubMed; (d) M. Vendrell, D. Zhai, J. C. Er and Y. T. Chang, Chem. Rev., 2012, 112, 4391–4420 CrossRef CAS PubMed; (e) J. Zhang, R. E. Campbell, A. Y. Ting and R. Y. Tsien, Nat. Rev. Mol. Cell Biol., 2002, 3, 906–918 CrossRef CAS PubMed; (f) H. Zhu, J. Fan, J. Du and X. Peng, Acc. Chem. Res., 2016, 49, 2115–2126 CrossRef CAS PubMed; (g) I. Johnson and M. T. Z. Spence, The Molecular Probes Handbook, A Guide to Fluorescent Probes and Labeling Technologies, Molecular Probes, Eugene, OR, 11th edn, 2010 Search PubMed.
- (a) S. M. Barbon, V. N. Staroverov and J. B. Gilroy, J. Org. Chem., 2015, 80, 5226–5235 CrossRef CAS PubMed; (b) E. Benedetti, L. S. Kocsis and K. M. Brummond, J. Am. Chem. Soc., 2012, 134, 12418–12421 CrossRef CAS PubMed; (c) A. N. Butkevich, M. V. Sednev, H. Shojaei, V. N. Belov and S. W. Hell, Org. Lett., 2018, 20, 1261–1264 CrossRef CAS PubMed; (d) R. Greiner, D. S. Ziegler, D. Cibu, A. C. Jakowetz, F. Auras, T. Bein and P. Knochel, Org. Lett., 2017, 19, 6384–6387 CrossRef CAS PubMed; (e) V. Jamsheena, R. K. Mishra, K. S. Veena, S. Sini, P. Jayamurthy and R. S. Lankalapalli, ACS Omega, 2018, 3, 856–862 CrossRef CAS PubMed; (f) E. Kim, M. Koh, B. J. Lim and S. B. Park, J. Am. Chem. Soc., 2011, 133, 6642–6649 CrossRef CAS PubMed.
- (a) L. D. Lavis and R. T. Raines, ACS Chem. Biol., 2008, 3, 142–155 CrossRef CAS PubMed; (b) A. P. Demchenko, Advanced Fluorescence Reporters in Chemistry and Biology I, Springer-Verlag: Berlin, Heidelberg, 2010 Search PubMed.
- (a) M. Arribat, E. Remond, S. Clement, A. V. Lee and F. Cavelier, J. Am. Chem. Soc., 2018, 140, 1028–1034 CrossRef CAS PubMed; (b) O. N. Burchak, L. Mugherli, M. Ostuni, J. J. Lacapere and M. Y. Balakirev, J. Am. Chem. Soc., 2011, 133, 10058–10061 CrossRef CAS PubMed; (c) Y. Cheng, G. Li, Y. Liu, Y. Shi, G. Gao, D. Wu, J. Lan and J. You, J. Am. Chem. Soc., 2016, 138, 4730–4738 CrossRef CAS PubMed; (d) Y. Lian, R. G. Bergman, L. D. Lavis and J. A. Ellman, J. Am. Chem. Soc., 2013, 135, 7122–7125 CrossRef CAS PubMed; (e) D. B. Zhao, J. Y. Hu, N. J. Wu, X. L. Huang, X. R. Qin, J. B. Lan and J. S. You, Org. Lett., 2011, 13, 6516–6519 CrossRef CAS PubMed.
- (a) A. R. Longstreet, R. R. Chandler, T. Banerjee, L. Zane Miller, K. Hanson and D. Tyler McQuade, Photochem. Photobiol. Sci., 2017, 16, 455–458 RSC; (b) A. R. Longstreet, M. Jo, R. R. Chandler, K. Hanson, N. Zhan, J. J. Hrudka, H. Mattoussi, M. Shatruk and D. T. McQuade, J. Am. Chem. Soc., 2014, 136, 15493–15496 CrossRef CAS PubMed.
- (a) H. M. Davies and J. S. Alford, Chem. Soc. Rev., 2014, 43, 5151–5162 RSC; (b) Y. Jiang, R. Sun, X. Y. Tang and M. Shi, Chem.–Eur. J., 2016, 22, 17910–17924 CrossRef CAS PubMed; (c) Y. Li, H. Yang and H. Zhai, Chem.–Eur. J., 2018, 24, 12757–12766 CrossRef CAS PubMed.
- Selected examples: (a) J. S. Alford and H. M. Davies, J. Am. Chem. Soc., 2014, 136, 10266–10269 CrossRef CAS PubMed; (b) K. Chen, Z. Z. Zhu, Y. S. Zhang, X. Y. Tang and M. Shi, Angew. Chem., Int. Ed., 2014, 53, 6645–6649 CrossRef CAS PubMed; (c) S. Chuprakov, B. T. Worrell, N. Selander, R. K. Sit and V. V. Fokin, J. Am. Chem. Soc., 2014, 136, 195–202 CrossRef CAS PubMed; (d) J. He, Y. Shi, W. Cheng, Z. Man, D. Yang and C. Y. Li, Angew. Chem., Int. Ed., 2016, 55, 4557–4561 CrossRef CAS PubMed; (e) S. W. Kwok, L. Zhang, N. P. Grimster and V. V. Fokin, Angew. Chem., Int. Ed., 2014, 53, 3452–3456 CrossRef CAS PubMed; (f) V. N. Lindsay, H. M. Viart and R. Sarpong, J. Am. Chem. Soc., 2015, 137, 8368–8371 CrossRef CAS PubMed; (g) S. Y. Liu, W. F. Yao, Y. Liu, Q. H. Wei, J. H. Chen, X. Wu, F. Xia and W. H. Hu, Sci. Adv., 2017, 3, e1602467 CrossRef PubMed; (h) P. Mi, R. Kiran Kumar, P. Liao and X. Bi, Org. Lett., 2016, 18, 4998–5001 CrossRef CAS PubMed; (i) T. Miura, T. Tanaka, T. Biyajima, A. Yada and M. Murakami, Angew. Chem., Int. Ed., 2013, 52, 3883–3886 CrossRef CAS PubMed; (j) E. E. Schultz, V. N. Lindsay and R. Sarpong, Angew. Chem., Int. Ed., 2014, 53, 9904–9908 CrossRef CAS PubMed; (k) Z.-F. Xu, H. Dai, L. Shan and C.-Y. Li, Org. Lett., 2018, 20, 1054–1057 CrossRef CAS PubMed; (l) J. M. Yang, C. Z. Zhu, X. Y. Tang and M. Shi, Angew. Chem., Int. Ed., 2014, 53, 5142–5146 CAS; (m) Y. Yang, M. B. Zhou, X. H. Ouyang, R. Pi, R. J. Song and J. H. Li, Angew. Chem., Int. Ed., 2015, 54, 6595–6599 CrossRef CAS PubMed; (n) Y. Yu, L. Zhu, Y. Liao, Z. Mao and X. Huang, Adv. Synth. Catal., 2016, 358, 1059–1064 CrossRef CAS; (o) H. Yuan, J. Gong and Z. Yang, Chem. Commun., 2017, 53, 9089–9092 RSC; (p) M. Zibinsky and V. V. Fokin, Angew. Chem., Int. Ed., 2013, 52, 1507–1510 CrossRef CAS PubMed.
- For reviews: (a) I. Bae, H. Han and S. Chang, J. Am. Chem. Soc., 2005, 127, 2038–2039 CrossRef CAS PubMed; (b) S. H. Cho and S. Chang, Angew. Chem., Int. Ed., 2007, 46, 1897–1900 CrossRef CAS PubMed; (c) S. H. Cho and S. Chang, Angew. Chem., Int. Ed., 2008, 47, 2836–2839 CrossRef CAS PubMed; (d) S. H. Cho, E. J. Yoo, L. Bae and S. Chang, J. Am. Chem. Soc., 2005, 127, 16046–16047 CrossRef CAS PubMed; (e) R. H. Dodd and K. Cariou, Chem.–Eur. J., 2018, 24, 2297–2304 CrossRef CAS PubMed; (f) Y. Jiang, R. Sun, Q. Wang, X. Y. Tang and M. Shi, Chem. Commun., 2015, 51, 16968–16971 RSC; (g) S. H. Kim, S. H. Park, J. H. Choi and S. Chang, Chem.–Asian J., 2011, 6, 2618–2634 CrossRef CAS PubMed; (h) H. D. Xu, Z. H. Jia, K. Xu, M. Han, S. N. Jiang, J. Cao, J. C. Wang and M. H. Shen, Angew. Chem., Int. Ed., 2014, 53, 9284–9288 CrossRef CAS PubMed; (i) Y. Zhang, K. A. DeKorver, A. G. Lohse, Y. S. Zhang, J. Huang and R. P. Hsung, Org. Lett., 2009, 11, 899–902 CrossRef CAS PubMed.
- (a) S. Chang, M. Lee, D. Y. Jung, E. J. Yoo, S. H. Cho and S. K. Han, J. Am. Chem. Soc., 2006, 128, 12366–12367 CrossRef CAS PubMed; (b) Z. Chen, C. Ye, L. Gao and J. Wu, Chem. Commun., 2011, 47, 5623–5625 RSC; (c) Y. Huang, W. Yi, Q. Sun, L. Zhang and F. Yi, RSC Adv., 2018, 8, 74–79 RSC.
- L. Sun, Y. Zhu, P. Lu and Y. Wang, Org. Lett., 2013, 15, 5894–5897 CrossRef CAS PubMed.
- (a) W. Lu, W. Song, D. Hong, P. Lu and Y. Wang, Adv. Synth. Catal., 2009, 351, 1768–1772 CrossRef CAS; (b) E. Romero, C. Minard, M. Benchekroun, S. Ventre, P. Retailleau, R. H. Dodd and K. Cariou, Chem.–Eur. J., 2017, 23, 12991–12994 CrossRef CAS PubMed; (c) M. Whiting and V. V. Fokin, Angew. Chem., Int. Ed., 2006, 45, 3157–3161 CrossRef CAS PubMed; (d) Y. Xing, H. Zhao, Q. Shang, J. Wang, P. Lu and Y. Wang, Org. Lett., 2013, 15, 2668–2671 CrossRef CAS PubMed.
- CCDC 1942349.†.
- W.-C. Sun, K. R. Gee and R. P. Haugland, Bioorg. Med. Chem. Lett., 1998, 8, 3107–3110 CrossRef CAS PubMed.
- The photobleaching of fluorescein is about 17% after 33 min, see: W.-C. Sun, K. R. Gee, D. H. Klaubert and R. P. Haugland, J. Org. Chem., 1997, 62, 6469–6475 CrossRef CAS.
- (a) Z. Ma, L. Du and M. Li, J. Med. Chem., 2014, 57, 8187–8203 CrossRef CAS PubMed; (b) H. Shi, R. T. Kwok, J. Liu, B. Xing, B. Z. Tang and B. Liu, J. Am. Chem. Soc., 2012, 134, 17972–17981 CrossRef CAS PubMed; (c) C. Zhang, S. Jin, K. Yang, X. Xue, Z. Li, Y. Jiang, W. Q. Chen, L. Dai, G. Zou and X. J. Liang, ACS Appl. Mater. Interfaces, 2014, 6, 8971–8975 CrossRef CAS PubMed.
- (a) F. Bellucci, F. Carini, C. Catalani, P. Cucchi, A. Lecci, S. Meini, R. Patacchini, L. Quartara, R. Ricci, M. Tramontana, S. Giuliani and C. A. Maggi, Br. J. Pharmacol., 2002, 135, 266–274 CrossRef CAS PubMed; (b) J. Li, Q. Zeng, Y. Zhang, X. Li, H. Hu, X. Miao, W. Yang, W. Zhang, X. Song, L. Mou and R. Wang, Eur. J. Cell Biol., 2016, 95, 368–377 CrossRef CAS PubMed; (c) L. Mou, Y. Xing, Z. Kong, Y. Zhou, Z. Chen and R. Wang, Biochem. Pharmacol., 2011, 81, 661–668 CrossRef CAS PubMed; (d) Y. Zhang, L. W. Lu, C. Furlonger, G. E. Wu and C. J. Paige, Nat. Immunol., 2000, 1, 392–397 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Details of the experimental procedure, structural characterization and spectral data of all new compounds and crystallographic data (CIF). CCDC 1942349. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/d0sc00798f |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2020 |