Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Highly crystalline CsPbI2Br films for efficient perovskite solar cells via compositional engineering
Fang
He†
ab,
Wenzhan
Xu†
ab,
Meng
Zhang
ab,
Xuan
Zhang
ab,
Baofu
Ding
ab,
Guodan
Wei
 *ab and
Feiyu
Kang
*abc
*ab and
Feiyu
Kang
*abc
aTsinghua-Berkeley Shenzhen Institute (TBSI), Tsinghua University, Shenzhen, 518000, China. E-mail: weiguodan@sz.tsinghua.edu.cn; fykang@sz.tsinghua.edu.cn
bTsinghua Shenzhen International Graduate School, Tsinghua University, Shenzhen, 518000, China
cSchool of Materials Science and Engineering, Tsinghua University, Beijing 100084, China
First published on 26th September 2019
Abstract
All-inorganic CsPbI2Br shows high thermal stability for promising application in perovskite solar cells (PSCs). The performance of PSCs is significantly affected by their morphology and crystallinity induced by compositional ratio, solvent/anti-solvent engineering and post thermal annealing. In this study, the compositional ratio effect of two precursors, PbI2 and CsBr, on the power conversion efficiency (PCE) of a device with ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au structure was investigated. With the assistance of anti-solvent chlorobenzene, perovskite with a PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr ratio of 1.05
CsBr ratio of 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 showed a high quality thin film with higher crystallinity and larger grain size. In addition, the molar ratio of precursors PbI2 and CsBr improved the PCE of the PSCs, and the PSCs fabricated using the perovskite with an optimal ratio of PbI2 and CsBr exhibited a PCE of 13.34%.
1 showed a high quality thin film with higher crystallinity and larger grain size. In addition, the molar ratio of precursors PbI2 and CsBr improved the PCE of the PSCs, and the PSCs fabricated using the perovskite with an optimal ratio of PbI2 and CsBr exhibited a PCE of 13.34%.
1. Introduction
Due to its appropriate optical band gap, high optical absorption coefficient, good ion migration ability and long carrier life, organic–inorganic hybrid perovskite is considered as an ideal material for use in the light absorption layer of solar cells.1–5 In the past decade, new breakthroughs have been made in the research of perovskite solar cells (PSCs).6–8 So far, the highest power conversion efficiency (PCE) of organic–inorganic hybrid perovskite solar cells has reached 25.2%,9 which is higher than the efficiency of commercial silicon solar cells. However, the poor long-term stability seriously limits the large-scale commercial use of organic–inorganic hybrid perovskite solar cells.10–12 Although some studies have effectively improved the stability of hybrid perovskite solar cells,13,14 they still cannot meet the requirements for practical applications. But the all-inorganic halide perovskite with good thermal stability can be obtained by completely replacing the organic components (MA+, FA+) of mixed perovskite with Cs+.15Typical all-inorganic halide perovskite materials mainly include CsPbI3,16 CsPbI2Br,17 CsPbBr3 (ref. 18) and CsSnI3.19 Among them, CsPbI3 and CsPbI2Br of cubic phase have appropriate optical band gaps, 1.73 eV and 1.92 eV, respectively.16,20 Therefore, these two halide perovskites have been extensively studied and the PCE of devices has been rapidly improved.20 Nevertheless, there is still a big gap between the current maximum efficiency and the theoretical schottky limit,20 which suggests that the all-inorganic perovskite solar cells still have great room for improvement and research value in photoelectric conversion efficiency. On the other hand, the all-inorganic halide perovskite usually suffers a severe phase transition problem. In ambient air environment, CsPbI3 and CsPbI2Br of cubic phase are easily converted into non-perovskite phase, which will seriously affect their photoelectric performance and practical utilization of devices.21
Studies have shown that, compared with CsPbI3, CsPbI2Br is easier to maintain the cubic phase structure, which has intrigued great attention.22 Chen et al. confirmed that the a-phase and b-phase CsPbI2Br have better thermal stability and phase stability than CsPbI3 due to the mixing of halide elements through theoretical calculations.23 Through interface engineering,24 solvent engineering,25 precursor engineering26 and other strategies, the photoelectric performance of CsPbI2Br has been effectively improved. For example, Guo et al.27 successfully synthesized Rb-doped CsPbI2Br film with improved crystallinity and light absorption. Based on this, the prepared all-inorganic perovskite solar cells without hole transport layer achieved stabilized PCE of over 11%. Bai et al.28 improved the crystallinity of the CsPbBrI2 film by preheating the precursor solution at 100 °C, and reached a high efficiency of up to 14.81% with the device structure of FTO/TiO2/CsPbBrI2/CsPbBrI2 QDs/PTAA/Au. Chen et al.29 precisely controlled the growth of α-CsPbI2Br crystal by the synergistic effect of gradient thermal annealing and anti-solvent method, and obtained a high-quality film with few defects and good stability, achieving a record PCE of 16.07%. Liu et al.30 successfully designed a novel structure of solar cell by using ZnO@C60 bilayer as the electron transport layer, achieving a high PCE of 13.3% with good thermal stability. Dong et al.31 used EtOH anti-solvent and optimized CsBr deposition cycle to obtain a high-purity phase and high-quality CsPbI2Br film, successfully achieving a record PCE of 10.21% for the α-CsPbI2Br PSCs without hole transporting material.
Herein, we studied the effect of raw material stoichiometric ratio on the properties of CsPbI2Br. Four perovskite solar cells with ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au were fabricated by changing the molar ratios of PbI2 and CsBr (0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.00
1, 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.05
1, 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.10
1, 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). We found that excessive PbI2 would lead to obvious PbI2 peak in the XRD pattern, and a small amount of PbI2 would significantly affect the morphology and crystallinity of as-prepared CsPbI2Br perovskite films. In addition, different proportions of PbI2 and CsBr will also lead to changes in optical band gap and light absorption properties of perovskite thin films, as well as differences in CsPbI2Br phase stability. Our results show that solar cell devices have the best power conversion efficiency when the molar ratio of PbI2 and CsBr is 1.05
1). We found that excessive PbI2 would lead to obvious PbI2 peak in the XRD pattern, and a small amount of PbI2 would significantly affect the morphology and crystallinity of as-prepared CsPbI2Br perovskite films. In addition, different proportions of PbI2 and CsBr will also lead to changes in optical band gap and light absorption properties of perovskite thin films, as well as differences in CsPbI2Br phase stability. Our results show that solar cell devices have the best power conversion efficiency when the molar ratio of PbI2 and CsBr is 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The enhanced PCE originates from the higher crystallinity and defects-free of perovskite thin film with appropriate grain size characterized by X-ray diffraction (XRD) and can electronic microscopy (SEM).
1. The enhanced PCE originates from the higher crystallinity and defects-free of perovskite thin film with appropriate grain size characterized by X-ray diffraction (XRD) and can electronic microscopy (SEM).
2. Experimental
2.1 Materials
Lead iodide (PbI2, 99.999%) and tin(IV) oxide (SnO2) colloid precursor were purchased from Alfa Aesar; cesium bromide (CsBr, 99.999%), chlorobenzene (CB, 99.8%), anhydrous dimethyl sulfoxide (DMSO, ≥99.9%), N,N-dimethylformamide (DMF, 99.8%), 4-tert-butylpyridine (tBP), tris(2-(1H-pyrazol-1-yl)-4-tert-butylpyridine)cobalt(III)-tris(bis(trifluoromethylsulfonyl) imide) (FK209) and bis(trifluoromethylsulfonyl)amine lithium salt (Li-TFSI) were purchased from Sigma Aldrich. Anhydrous methanol, ethanol (>99.5%) and isopropyl alcohol (IPA, 99.5%) were purchased from Acros; Spiro-MeOTAD (99.8%) was purchased from Xi'an Polymer Light Technology Corp. All materials and reagents were used as received without further purification.2.2 Precursor preparation
1 M CsBr and X M PbI2 (X = 0.95, 1.00, 1.05, 1.10) were dissolved in a mixed solvent (DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMF = 9
DMF = 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) according to certain stoichiometric ratios, and stirred at 70 °C for 12 h. The CsPbI2Br precursor solution was then obtained by filtration through a 0.22 μm PTFE filter.
1) according to certain stoichiometric ratios, and stirred at 70 °C for 12 h. The CsPbI2Br precursor solution was then obtained by filtration through a 0.22 μm PTFE filter.
2.3 Solar cells fabrication
The ITO glasses were successively cleaned with acetone, ethanol and deionized water respectively by ultrasonic cleaning for 15 minutes, respectively. Before the spin coating, each ITO glass was blown dry with nitrogen gun and cleaned with plasma for 60 s. SnO2 was selected as the electron transport layer. We diluted the SnO2 colloid precursor to 5% with deionized water, then spin-coated at 3000 rpm for 30 s, and thermal annealed at 150 °C for 30 min to obtain an electron transport layer. Then the 1 M CsPbI2Br precursor solution was deposited on the SnO2 layer at the speed of 1000 rpm for 10 s and 4500 rpm for 35 s in a glove box, annealing at 260 °C for 10 minutes. For deposition of hole-transporting material, Spiro-MeOTAD was dissolved in chlorobenzene with a concentration of 80 mg mL−1, then 35 μL of lithium bis (trifluoromethanesulfonyl)imide in acetonitrile (260 mg mL−1) and 30 μL of 4-tert-butylpyridine was added into the Spiro-MeOTAD solution. The mixture was coated onto the perovskite film at 3500 RPM for 30 s to form a Spiro-MeOTAD hole-transporting layer. Finally, the counter electrode was deposited by thermal evaporation of 80 nm-thick gold under a pressure of 2 × 10−6 mbar. The active area was measured to be 0.045 cm2.2.4 Characterization of perovskite thin films
UV-vis absorption and photoluminescence (PL) spectra of thin films were recorded on a HP 8453 spectrophotometer and FLS920 spectrofluorimeter (Edinburgh Instruments), respectively. A 150 W, ozone-free xenon arc lamp was used in PL measurements. Scanning electron microscope (SEM) images were obtained by using a field emission scanning electron microscope (JEOL-7401). Thicknesses of thin films were measured by Dektak 150 surface profilometer. X-Ray Diffraction (XRD) patterns were measured by an X-ray diffractometer Bruker D8 Advance using Cu Kα radiation source with a scan rate of 10° min−1.2.5 Characterization of perovskite solar cells
The J–V characteristics of PSCs were measured by a Keithley model 2400 source measure unit (Newport, Oriel AM 1.5 G, 100 mW cm−2). The light intensity of 100 mW cm−2 was calibrated by a silicon reference cell. The external quantum efficiency (EQE) spectra of PSCs were performed on a DSR100UV-B spectrometer with a bromine tungsten light source, a SR830 lock-in amplifier and a calibrated Si detector.3. Results and discussion
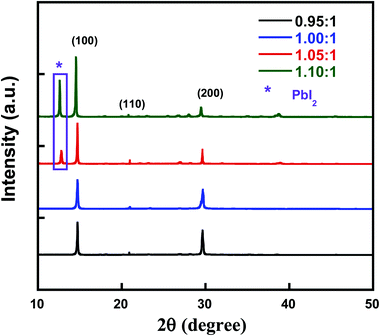
To investigate the influence of compositional ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr on the structural evolution of all-inorganic perovskite, X-ray diffraction (XRD) of these thin films are conducted in Fig. 1. Similar to perovskite with PbI2
CsBr on the structural evolution of all-inorganic perovskite, X-ray diffraction (XRD) of these thin films are conducted in Fig. 1. Similar to perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.00
CsBr of 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, all other perovskite with PbI2
1, all other perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr (0.95
CsBr (0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.05
1, 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.10
1 and 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) thin films exhibited peaks at 14.7°, 20.9° and 29.6°, which correspond to the (100), (110) and (200) crystal planes of CsPbI2Br perovskite thin film, respectively, demonstrating prepared perovskites retain cubic phase structure, which is favorable for reducing charge recombination and promoting device performance.26,32 The corresponding average grain size were 61.34, 61.25, 53.75 and 55.83 nm calculated based on Scherrer formula, indicating the small adjusted component ratio of the PbI2 added into the precursor solution doesn't change crystal size significantly which is beneficial to improve the charge transport in the solar cells. The new peak located at 12.5° detected from both perovskite with PbI2
1) thin films exhibited peaks at 14.7°, 20.9° and 29.6°, which correspond to the (100), (110) and (200) crystal planes of CsPbI2Br perovskite thin film, respectively, demonstrating prepared perovskites retain cubic phase structure, which is favorable for reducing charge recombination and promoting device performance.26,32 The corresponding average grain size were 61.34, 61.25, 53.75 and 55.83 nm calculated based on Scherrer formula, indicating the small adjusted component ratio of the PbI2 added into the precursor solution doesn't change crystal size significantly which is beneficial to improve the charge transport in the solar cells. The new peak located at 12.5° detected from both perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr (1.05
CsBr (1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.10
1 and 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) is likely to due to the peak of excess unreacted PbI2. It was reported that moderate excess PbI2 can functioned as role of passivating the surface defects for reducing the charge carrier recombination.
1) is likely to due to the peak of excess unreacted PbI2. It was reported that moderate excess PbI2 can functioned as role of passivating the surface defects for reducing the charge carrier recombination.
Fig. 2a showed the absorption spectrum of perovskite thin films, all of which exhibited the typical absorption profile for cubic phase CsPbI2Br thin film with the typical absorption peak of 625 nm, which is consistent with other report.31,33 However, the perovskite film deposited with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr ratio of 1.10
CsBr ratio of 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 has the absorption peak at 635 nm. The slightly red shifted absorption peak indicates the excess PbI2 actually influences the CsPbI2Br perovskite thin films. In addition, the peak is observed at 405 nm for the δ-perovskite with PbI2
1 has the absorption peak at 635 nm. The slightly red shifted absorption peak indicates the excess PbI2 actually influences the CsPbI2Br perovskite thin films. In addition, the peak is observed at 405 nm for the δ-perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.10
CsBr of 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, implying that the black α-CsPbI2Br phase could have been partially transitioned into the yellow δ-CsPbI2Br phase. Fig. 2b depicted the steady-state photoluminescence (PL) spectra of these four perovskite thin films. The emission peak of the CsPbI2Br thin film is at 653 nm is well consistent with absorption edges for corresponding perovskite thin films (Fig. 2a). It is also observed that a red shifted PL peak for perovskites with the molar ratio of 1.10
1, implying that the black α-CsPbI2Br phase could have been partially transitioned into the yellow δ-CsPbI2Br phase. Fig. 2b depicted the steady-state photoluminescence (PL) spectra of these four perovskite thin films. The emission peak of the CsPbI2Br thin film is at 653 nm is well consistent with absorption edges for corresponding perovskite thin films (Fig. 2a). It is also observed that a red shifted PL peak for perovskites with the molar ratio of 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
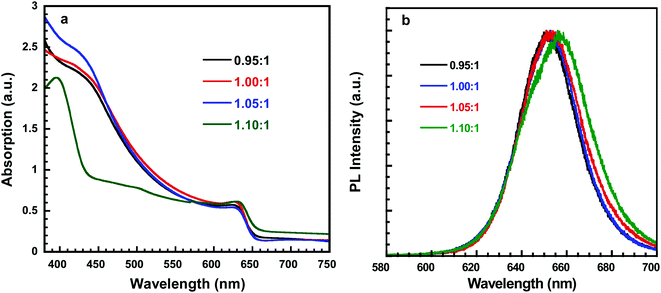 | ||
Fig. 2 (a) Absorption of perovskite thin films with different molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr. (b) Photoluminescence (PL) spectra of perovskite thin films with different molar ratios of PbI2 CsBr. (b) Photoluminescence (PL) spectra of perovskite thin films with different molar ratios of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr. CsBr. | ||
The surface morphology and crystalline size of CsPbI2Br thin films deposited with these four different molar ratios of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr is further analyzed by SEM images, as shown in Fig. 3. It is obviously that all samples display densely stacked grains on their surfaces which is consistently with the calculated average crystal size in nanoscale range. However, brighter dots on the surface of the perovskite grains are clearly observed when the molar ratio of PbI2
CsBr is further analyzed by SEM images, as shown in Fig. 3. It is obviously that all samples display densely stacked grains on their surfaces which is consistently with the calculated average crystal size in nanoscale range. However, brighter dots on the surface of the perovskite grains are clearly observed when the molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr is 1.10
CsBr is 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Fig. 3d), proofing the existence of the unreacted PbI2 particles could affect the thin film quality.
1 (Fig. 3d), proofing the existence of the unreacted PbI2 particles could affect the thin film quality.
 | ||
Fig. 3 (a) The top-view SEM image of CsPbI2Br thin film with molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of (a) 0.95 CsBr of (a) 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (b) 1.00 1, (b) 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (c) 1.05 1, (c) 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and (d) 1.10 1 and (d) 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1. | ||
To investigate the effect of composition ratios of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr on the photovoltaic performance of PSCs, devices with a planar heterojunction structure of ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au (Fig. 4a) with the CsPbI2Br shown in Fig. 4b were fabricated where ITO acts as the cathode electrode, solution-processed SnO2 thin film is used as the electron extraction layer, Spiro-MeOTAD acts as the hole extraction layer and Au acts as the anode electrode, respectively (Fig. 4c). The current densities versus voltages (J–V) characteristics of PSCs under one-sun illumination with the light intensity of 100 mW cm−2, at the scan rate of 0.20 V s−1 and the reverse scan direction, are shown in Fig. 5a. The device performance parameters are summarized in Table 1. The PSCs based on the perovskite with PbI2
CsBr on the photovoltaic performance of PSCs, devices with a planar heterojunction structure of ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au (Fig. 4a) with the CsPbI2Br shown in Fig. 4b were fabricated where ITO acts as the cathode electrode, solution-processed SnO2 thin film is used as the electron extraction layer, Spiro-MeOTAD acts as the hole extraction layer and Au acts as the anode electrode, respectively (Fig. 4c). The current densities versus voltages (J–V) characteristics of PSCs under one-sun illumination with the light intensity of 100 mW cm−2, at the scan rate of 0.20 V s−1 and the reverse scan direction, are shown in Fig. 5a. The device performance parameters are summarized in Table 1. The PSCs based on the perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 0.95
CsBr of 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.00
1 and 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 show PCE of 11.49% (VOC of 1.09 V, JSC of 14.8 mA cm−2 and FF of 71.2%) and 12.52% (VOC of 1.12 V, JSC of 15.01 mA cm−2 and FF of 74.5%). The PSCs based on the perovskite with PbI2
1 show PCE of 11.49% (VOC of 1.09 V, JSC of 14.8 mA cm−2 and FF of 71.2%) and 12.52% (VOC of 1.12 V, JSC of 15.01 mA cm−2 and FF of 74.5%). The PSCs based on the perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.05
CsBr of 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 shows an enhanced performance upon slightly increasing with PbI2. Specifically, the PCE is increased to 13.34% with a VOC, JSC, and FF of 1.12 V, 15.78 mA cm−2, and 75.5%, respectively. It is worth noted that the simultaneously enhanced VOC, JSC and FF is achieved. These enhancements are attributed to the denser, higher crystallinity CsPbI2Br grains which could effectively suppress charge recombination (Fig. 3c). However, further addition of much more PbI2 causes a negative performance change. For example, when the molar ratio of PbI2
1 shows an enhanced performance upon slightly increasing with PbI2. Specifically, the PCE is increased to 13.34% with a VOC, JSC, and FF of 1.12 V, 15.78 mA cm−2, and 75.5%, respectively. It is worth noted that the simultaneously enhanced VOC, JSC and FF is achieved. These enhancements are attributed to the denser, higher crystallinity CsPbI2Br grains which could effectively suppress charge recombination (Fig. 3c). However, further addition of much more PbI2 causes a negative performance change. For example, when the molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr is increased to 1.10
CsBr is increased to 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the PCE is decreased to 10.91% (VOC of 1.10 V, JSC of 14.01 mA cm−2, FF of 70.8%). The excess PbI2 has reduced the sunlight absorption efficiency (Fig. 2a) and could act as recombination or trap centers with unnecessary spots on the active CsPbI2Br thin film (Fig. 3d), severally reducing the photocurrent and FF (Table 1). The strong correlation between JSC and series resistance (Rs) could be validated that with more and more PbI2 added, the measured Rs has consistently reduced when the molar ratio changes from 0.95 to 1.05 (Table 1), indicating the improved crystallinity does increase the charge separation and carrier transport efficiency. Thus the molar ratio of PbI2
1, the PCE is decreased to 10.91% (VOC of 1.10 V, JSC of 14.01 mA cm−2, FF of 70.8%). The excess PbI2 has reduced the sunlight absorption efficiency (Fig. 2a) and could act as recombination or trap centers with unnecessary spots on the active CsPbI2Br thin film (Fig. 3d), severally reducing the photocurrent and FF (Table 1). The strong correlation between JSC and series resistance (Rs) could be validated that with more and more PbI2 added, the measured Rs has consistently reduced when the molar ratio changes from 0.95 to 1.05 (Table 1), indicating the improved crystallinity does increase the charge separation and carrier transport efficiency. Thus the molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr in the precursor solution is a crucial parameter for determining device performance.
CsBr in the precursor solution is a crucial parameter for determining device performance.
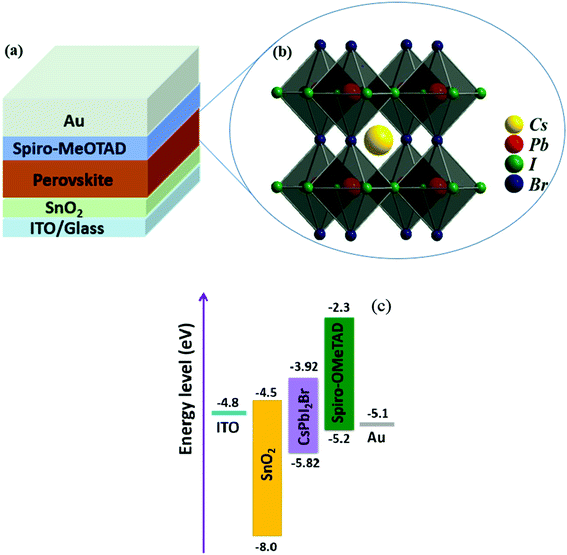 | ||
| Fig. 4 (a) Device structure of the PSCs. (b) Crystal structure of the perovskite CsPbI2Br. (c) the schematic energy diagram of glass/ITO/SnO2/perovskite/Spiro-OMeTAD/Au device. | ||
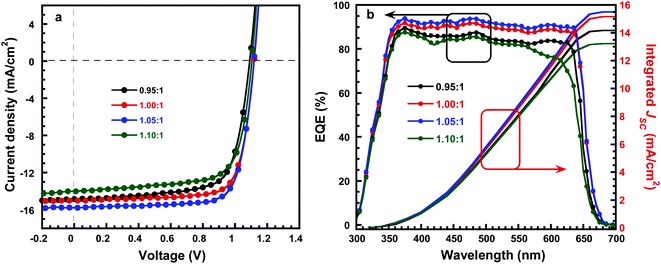 | ||
Fig. 5 (a) J–V characteristics of CsPbI2Br thin film with molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr. (b) EQE spectrum and integrated JSC of CsPbI2Br thin film with molar ratio of PbI2 CsBr. (b) EQE spectrum and integrated JSC of CsPbI2Br thin film with molar ratio of PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr. CsBr. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr
CsBr
PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr (molar ratio) CsBr (molar ratio) |
V OC (V) | J SC (mA cm−2) | FF (%) | PCE (%) (Best) | PCE (%) (Average) | R s (Ω cm2) |
|---|---|---|---|---|---|---|
0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.09 | 14.80 | 71.2 | 11.49 | 10.43 | 53.26 |
1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.12 | 15.01 | 74.5 | 12.52 | 11.85 | 43.87 |
1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.12 | 15.78 | 75.5 | 13.34 | 12.78 | 35.55 |
1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1.10 | 14.01 | 70.8 | 10.91 | 9.85 | 48.12 |
EQE of all the perovskite solar cells is shown in Fig. 5b. The perovskite solar cells exhibit a photoresponse in the wavelength ranging from 300 to 650 nm, suggesting absorbed photos being converted into charge carriers, and further generates into photocurrent in the solar cells. In addition, the champion device shows a maximum value of about 93.4% at λ = 480 nm, the integrated JSC values from the EQE spectra are 14.17 mA cm−2, 15.16 mA cm−2, 15.51 and 13.22 mA cm−2 for the PSCs fabricated by perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 0.95
CsBr of 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.00
1, 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.05
1, 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.10
1 and 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively, which is about 5% error between the JSC from EQE spectra and J–V curve, implying the accuracy of device performance.
1, respectively, which is about 5% error between the JSC from EQE spectra and J–V curve, implying the accuracy of device performance.
Furthermore, as the stability is the priority concern for the PSCs, the shelf stability of the PSCs is investigated. The devices are stored in N2 atmosphere without any encapsulation. As shown in Fig. 6, the PSCs fabricated by perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.05
CsBr of 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 showed a better shelf stability than that of PSCs fabricated by perovskite with PbI2
1 showed a better shelf stability than that of PSCs fabricated by perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.00
CsBr of 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. After being stored for 240 h, PSCs fabricated by perovskite with PbI2
1. After being stored for 240 h, PSCs fabricated by perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.05
CsBr of 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 retained 84% of the initial PCE, while the PCE of PSCs fabricated by perovskite with 1
1 retained 84% of the initial PCE, while the PCE of PSCs fabricated by perovskite with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of the two precursors dropped to 75% of the initial value in the same storage condition and time. The enhanced shelf stability which we attribute to the surface passivation of unreacted PbI2.34–36
1 ratio of the two precursors dropped to 75% of the initial value in the same storage condition and time. The enhanced shelf stability which we attribute to the surface passivation of unreacted PbI2.34–36
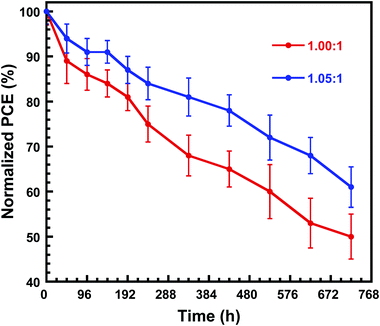 | ||
Fig. 6 Normalized PCEs of the unencapsulated PSCs fabricated by perovskite with PbI2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CsBr of 1.00 CsBr of 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.05 1 and 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 functioned as the time under in N2-filled glove box. 1 functioned as the time under in N2-filled glove box. | ||
4. Conclusions
In summary, we investigated the effect of compositional engineering on the film properties and device performance of inorganic perovskite CsPbI2Br by changing the stoichiometric ratios of PbI2 and CsBr (0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.00
1, 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1.05
1, 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1.10
1 and 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The results show that the molar ratio of the raw materials is 1.05
1). The results show that the molar ratio of the raw materials is 1.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the CsPbI2Br film shows the best crystallinity and the corresponding solar cell (ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au) achieves the highest efficiency of 13.34%. XRD and SEM results show that once the molar ratio of raw materials is increased to 1.10
1, the CsPbI2Br film shows the best crystallinity and the corresponding solar cell (ITO/SnO2/CsPbI2Br/Spiro-MeOTAD/Au) achieves the highest efficiency of 13.34%. XRD and SEM results show that once the molar ratio of raw materials is increased to 1.10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the resultant film contains more PbI2, which may provide more recombination centers for free electrons and holes, thus significantly reducing the efficiency of the device. Our study highlights the importance of compositional engineering and offers a meaningful reference for the preparation of stable and efficient solar cell devices.
1, the resultant film contains more PbI2, which may provide more recombination centers for free electrons and holes, thus significantly reducing the efficiency of the device. Our study highlights the importance of compositional engineering and offers a meaningful reference for the preparation of stable and efficient solar cell devices.
Conflicts of interest
There are no conflicts of interest to declare.Acknowledgements
We thank Shenzhen Municipal Development and Reform Commission, and New Energy Technology Engineering Laboratory (Grant number: SDRC [2016]172).References
- S. D. Stranks, G. E. Eperon, G. Grancini, C. Menelaou, M. J. Alcocer, T. Leijtens, L. M. Herz, A. Petrozza and H. J. Snaith, Science, 2013, 342, 341–344 CrossRef CAS.
- S. D. Stranks, V. M. Burlakov, T. Leijtens, J. M. Ball, A. Goriely and H. J. Snaith, Phys. Rev. Appl., 2014, 2, 034007 CrossRef.
- C. S. Ponseca Jr, T. J. Savenije, M. Abdellah, K. Zheng, A. Yartsev, T. Pascher, T. Harlang, P. Chabera, T. Pullerits and A. Stepanov, J. Am. Chem. Soc., 2014, 136, 5189–5192 CrossRef.
- Q. Dong, Y. Fang, Y. Shao, P. Mulligan, J. Qiu, L. Cao and J. Huang, Science, 2015, 347, 967–970 CrossRef CAS.
- D. Shi, V. Adinolfi, R. Comin, M. Yuan, E. Alarousu, A. Buin, Y. Chen, S. Hoogland, A. Rothenberger and K. Katsiev, Science, 2015, 347, 519–522 CrossRef CAS.
- M. A. Green, A. Ho-Baillie and H. J. Snaith, Nat. Photonics, 2014, 8, 506–514 CrossRef CAS.
- J.-P. Correa-Baena, A. Abate, M. Saliba, W. Tress, T. Jesper Jacobsson, M. Grätzel and A. Hagfeldt, Energy Environ. Sci., 2017, 10, 710–727 RSC.
- S. Gholipour and M. Saliba, Small, 2018, 14, e1802385 CrossRef.
- Laboratory, T. N. R. E., http://https://www.nrel.gov/pv/cell-efficiency.html, 2019.
- T. Leijtens, G. E. Eperon, N. K. Noel, S. N. Habisreutinger, A. Petrozza and H. J. Snaith, Adv. Energy Mater., 2015, 5, 1500963 CrossRef.
- B. W. Park and S. I. Seok, Adv. Mater., 2019, 31, e1805337 CrossRef.
- T. A. Berhe, W.-N. Su, C.-H. Chen, C.-J. Pan, J.-H. Cheng, H.-M. Chen, M.-C. Tsai, L.-Y. Chen, A. A. Dubale and B.-J. Hwang, Energy Environ. Sci., 2016, 9, 323–356 RSC.
- Q. Fu, X. Tang, B. Huang, T. Hu, L. Tan, L. Chen and Y. Chen, Adv. Sci., 2018, 5, 1700387 CrossRef.
- Z. Zhao, F. Gu, H. Rao, S. Ye, Z. Liu, Z. Bian and C. Huang, Adv. Energy Mater., 2019, 9, 1802671 CrossRef.
- W. Ahmad, J. Khan, G. Niu and J. Tang, Sol. RRL, 2017, 1, 1700048 CrossRef.
- G. E. Eperon, G. M. Paternò, R. J. Sutton, A. Zampetti, A. A. Haghighirad, F. Cacialli and H. J. Snaith, J. Mater. Chem. A, 2015, 3, 19688–19695 RSC.
- Q. Zeng, X. Zhang, C. Liu, T. Feng, Z. Chen, W. Zhang, W. Zheng, H. Zhang and B. Yang, Sol. RRL, 2019, 3, 1800239 CrossRef.
- H. Yuan, Y. Zhao, J. Duan, Y. Wang, X. Yang and Q. Tang, J. Mater. Chem. A, 2018, 6, 24324–24329 RSC.
- P. Zhu, C. Chen, S. Gu, R. Lin and J. Zhu, Sol. RRL, 2018, 2, 1700224 CrossRef.
- Q. Tai, K.-C. Tang and F. Yan, Energy Environ. Sci., 2019, 2375–2405 RSC.
- Y. Zhou and Y. Zhao, Energy Environ. Sci., 2019, 12, 1495–1511 RSC.
- C. Dong, X. Han, W. Li, Q. Qiu and J. Wang, Nano Energy, 2019, 59, 553–559 CrossRef CAS.
- Y. Chen, T. Shi, P. Liu, W. Xie, K. Chen, X. Xu, L. Shui, C. Shang, Z. Chen, H.-L. Yip, G. Zhou and X. Wang, J. Mater. Chem. A, 2019, 7, 20201–20207 RSC.
- L. Zhou, X. Guo, Z. Lin, J. Ma, J. Su, Z. Hu, C. Zhang, S. Liu, J. Chang and Y. Hao, Nano Energy, 2019, 60, 583–590 CrossRef CAS.
- S. Zhang, S. Wu, W. Chen, H. Zhu, Z. Xiong, Z. Yang, C. Chen, R. Chen, L. Han and W. Chen, Materials today energy, 2018, 8, 125–133 CrossRef.
- G. Yin, H. Zhao, H. Jiang, S. Yuan, T. Niu, K. Zhao, Z. Liu and S. F. Liu, Adv. Funct. Mater., 2018, 28, 1803269 CrossRef.
- Y. Guo, F. Zhao, J. Tao, J. Jiang, J. Zhang, J. Yang, Z. Hu and J. Chu, ChemSusChem, 2019, 12, 983–989 CrossRef CAS.
- D. Bai, H. Bian, Z. Jin, H. Wang, L. Meng, Q. Wang and S. F. Liu, Nano energy, 2018, 52, 408–415 CrossRef CAS.
- W. Chen, H. Chen, G. Xu, R. Xue, S. Wang, Y. Li and Y. Li, Joule, 2019, 3, 191–204 CrossRef CAS.
- C. Liu, W. Li, C. Zhang, Y. Ma, J. Fan and Y. Mai, J. Am. Chem. Soc., 2018, 140, 3825–3828 CrossRef CAS.
- C. Dong, X. Han, W. Li, Q. Qiu and J. Wang, Nano Energy, 2019, 59, 553–559 CrossRef CAS.
- J. K. Nam, M. S. Jung, S. U. Chai, Y. J. Choi, D. Kim and J. H. Park, J. Phys. Chem. Lett., 2017, 8, 2936–2940 CrossRef CAS.
- Z. Zhang, J. Zhang, X. Gan, H. Sun, M. Shang, D. Hou, C. Lu, R. Chen, Y. Zhu and L. Han, Adv. Energy Mater., 2018, 8, 1801050 CrossRef.
- Y. Zhao, Q. Li, W. Zhou, Y. Hou, Y. Zhao, R. Fu, D. Yu, X. Liu and Q. Zhao, Sol. RRL, 2019, 3, 1800296 CrossRef.
- D. H. Cao, C. C. Stoumpos, C. D. Malliakas, M. J. Katz, O. K. Farha, J. T. Hupp and M. G. Kanatzidis, APL Mater., 2014, 2, 091101 CrossRef.
- S. Wang, W. Dong, X. Fang, Q. Zhang, S. Zhou, Z. Deng, R. Tao, J. Shao, R. Xia and C. Song, Nanoscale, 2016, 8, 6600–6608 RSC.
Footnote |
| † Fang He and Wenzhan Xu are contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2019 |