Efficient removal of uranium(VI) from simulated seawater with hyperbranched polyethylenimine (HPEI)-functionalized polyacrylonitrile fibers†
Guoqing
Huang‡
ab,
Wenting
Li‡
a,
Qi
Liu
 a,
Jingyuan
Liu
a,
Jingyuan
Liu
 a,
Hongsen
Zhang
c,
Rumin
Li
a,
Hongsen
Zhang
c,
Rumin
Li
 *ad,
Zhanshuang
Li
a,
Xiaoyan
Jing
a and
Jun
Wang
*ad,
Zhanshuang
Li
a,
Xiaoyan
Jing
a and
Jun
Wang
 *ad
*ad
aKey Laboratory of Superlight Material and Surface Technology, Ministry of Education, Harbin Engineering University, Harbin, 150001, China. E-mail: zhqw1888@sohu.com; Fax: +86 451 8253 3026; Tel: +86 451 8253 3026
bHandan Purification Equipment Research Institute, Handan, 056027, P. R. China
cModern Analysis, Test and Research Center, Heilongjiang University of Science and Technology, Harbin 150027, P. R. China
dInstitute of Advanced Marine Materials, Harbin Engineering University, 150001, China
First published on 20th November 2017
Abstract
Capturing uranium(VI) from seawater is of paramount interest for the rapid expansion of energy needs. We report on materials covalently functionalized with amine groups to improve gas adsorption and remove metal ions. In this paper, polyacrylonitrile fibers (PANFs) covalently modified with hyperbranched polyethylenimine (HPEI) of different grafting degrees were facilely synthesized, and their performance to capture U(VI) in the presence of competitive ions and simulated seawater evaluated. In addition, the adsorption properties of the as-prepared adsorbents, including thermodynamics, kinetics and the influence of critical factors affecting U(VI) adsorption from aqueous solution were examined. The fibers with the best performance were characterized by X-ray photoelectron spectroscopy, scanning electron microscopy and Fourier transformed infrared spectroscopy. The high density of amine groups on the modified fiber surface improves the adsorption ability by electrostatic attraction and surface interaction during the reaction process. From our findings, PAN–HPEI offers wide-ranging potential in the wide field of the removal of metal ions, and, in particular, the removal of U(VI) from seawater with high throughput.
Introduction
Sustainable development of energy remains a predominant scientific and technological challenge facing humanity.1 Today, with the rapidly growing demand for energy and uncertainty in fossil fuel-based energy sources, the use of nuclear power, which is highly efficient and clean, has captured increasing attention.2 As one of the most crucial and effective raw materials of a nuclear reactor, U(VI) plays an indispensable role in the ongoing pursuit of alternative energies to the commonly used energies.3 U(VI) resources from terrestrial ores are limited, so an efficient extraction of U(VI) from aqueous systems, especially seawater in which the quantity of U(VI) is around 4.5 billion tons,4 is of great significance for sustainable energy development.5 Adsorption is one of various techniques to extract U(VI) from aqueous solution; it is considered to be an effective and economic way forward.6In recent years, extensive effort has been dedicated to this research field and many adsorbents have been reported for U(VI) recovery from simulated seawater or seawater. Wang et al. prepared acylamide- and carboxyl-functionalized metal–organic frameworks to extract U(VI) from seawater.7 Yamazaki et al. synthesized novel phenol-type resins embedded in highly-porous silica beads to explore the adsorption behavior in simulated seawater and seawater.8 Górka et al. investigated sonochemical functionalized mesoporous carbon to extract uranium from seawater.9 Among the potential materials, fabric and fiber adsorbents are being comprehensively researched due to their low cost and feasibility for industrialization. Dietz et al. prepared polyamide 6 fabrics modified with diallyl oxalate to remove U(VI) from seawater.10 Xie et al. designed an electrospun nanofibrous adsorbent for U(VI) extraction from seawater.11 Ladshaw et al. studied amidoxime functionalized fibers to extract U(VI) in the presence of other ions in simulated seawater.12 Herein, we use a commercial product of polyacrylonitrile fibers (PANFs) because they possesses good mechanical strength and stability, and are resistant to corrosion under concentrated salt conditions;13 the fibers have been used for the removal of heavy metal ions. To activate a tight binding with metal ions, the conversion of nitriles to carboxylic groups was necessary for the grafting of various functional moieties14 containing N and O and other atoms (amide, carboxyl, aminoxime, etc.) that have been reported to interact with U(VI) easily.
Hyperbranched polyethyleneimine (HPEI), which is believed to bind metal ions tightly owing to the substantial primary and secondary amine groups per molecule and its non-reaction with transitional metal ions, is commonly used to functionalize the surface of adsorbents to improve the adsorption ability.15,16 Recently, PEI-based adsorbents have been extensively investigated for metal binding. Tian et al. prepared PEI modified halloysite nanotubes for the adsorption of Cr(VI), in which the uptake capacity was sixty-four times higher than those of the raw materials.17 Lindén fabricated cross-linked PEI coatings for copper adsorption from seawater.18
In the present work, PANF modified with HPEI (PAN–HPEI) with different grafting degrees was facilely synthesized using a hydrothermal method. First, we explore the adsorption behavior of the obtained fibrous materials in the presence of several competing cations, and investigate the adsorption performance of the adsorbents to remove U(VI) from aqueous solution and simulated seawater; we ascertain the adsorbent with the best grafting degree. In addition, we study the parameters affecting uranium adsorption, such as pH, time and concentration, and the kinetic models and thermodynamics, to further understand the process. Finally, we propose hypothetical adsorption mechanisms.
Experimental
Chemicals
PANFs of lengths 2–3 cm, HPEI with Mn 1.0 × 104 g mol−1 and 1-hydroxybenzotriazole (HOBt) were obtained from Aladdin. Sodium hydroxide was acquired from Tianjin Chemical Reagent Co., Ltd. 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDCI, 98%) was obtained from Xiya reagent.The sea salt was purchased from Haiyang Guangzhou aquarium Technology Co., Ltd. The HNO3 (UPS, 68%) was obtained from Suzhou Crystal clear chemical Co., Ltd. Na2EDTA (AR), NaOH (AR), HCl (AR, 36%), NaHCO3 (AR), and Na2SO4 (AR) were acquired from Tianjin Dongli chemical Co., Ltd.
The water used was deionized.
Preparation of PAN–HPEI adsorbent
The synthesis of PAN–HPEI was carried out in two steps as follows, as depicted in Scheme 1.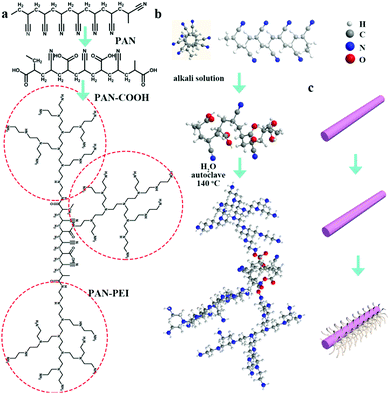 | ||
| Scheme 1 Synthesis routine of PAN–HPEI based on PAN: (a) in the structural formula of the molecule of which the red circle is HPEI; (b) in the stick type; (c) in 3D representation. | ||
First, dried PAN (1.0 g), NaOH (10 g), and deionized water (100 mL) were added to a conical flask and stirred in a bath oscillator at 80 °C for 15 min. Then the hydrolyzed adsorbent was immersed in HCl until the supernatant was neutral, followed by drying at 60 °C under vacuum overnight to give PAN–COOH.
Quintessentially, the as-prepared fibers and HPEI (mass ratio of the fiber to HPEI was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) were put into a 100 mL Teflon-lined autoclave. Ample de-ionized water was poured into the mixture and stirred for 2 h to make sure that all HPEI was dissolved. Then the autoclave was put into an oven at 140 °C for a specified time to obtain PAN–HPEI with different grafting degrees. After cooling, the fibers were separated and washed with warm water until the wash water was neutral, followed by drying at 60 °C under vacuum overnight.
3) were put into a 100 mL Teflon-lined autoclave. Ample de-ionized water was poured into the mixture and stirred for 2 h to make sure that all HPEI was dissolved. Then the autoclave was put into an oven at 140 °C for a specified time to obtain PAN–HPEI with different grafting degrees. After cooling, the fibers were separated and washed with warm water until the wash water was neutral, followed by drying at 60 °C under vacuum overnight.
The grafting degrees (GDs) of HPEI onto the PANFs were assessed by the mass increase of the raw fibres from the following eqn (1):19
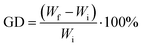 | (1) |
Batch equilibrium U(VI) adsorption experiments
We first verified adsorption selectivity in the presence of a number of competing ions to determine if the adsorbents can be used to remove U(VI) from seawater.Then adsorption tests in simulated seawater were carried out for 48 h to determine the adsorption performance. Simulated seawater was prepared according to a previous specification in the literature20 with a minor change. By dissolving 2.11 g uranyl nitrate and 33 g sea salt in 1 L DI water, artificial seawater with a concentration of 1000 mg L−1 was synthesized. Herein, the as-prepared seawater was diluted to the μg L−1 level ranging from 3 to 30 μg L−1 to mimic the seawater environment.
To further study the adsorption process, batch experiments were implemented. Classically, U(VI) removal was executed in a Erlenmeyer flask (100 mL) in which a specified dosage of the fibres was vibrated with U(VI)-containing solution (50 mL) of an assigned pH and concentration in a thermostatic oscillator at a speed of 150 rpm. The solution pH was monitored with trace volumes of 0.5 M HNO3 or NaOH solution at first. After the fibrous material separated, the concentration of U(VI) in the supernatant was measured for further calculation.
The loading capacity Qe (mg g−1), solid phase distribution coefficient Kd (mL g−1), and removal rate R (%) were determined on the basis of eqn (2)–(4):21,22
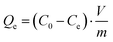 | (2) |
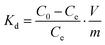 | (3) |
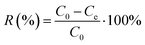 | (4) |
Characterization
The characterization of the adsorbents was carried out by scanning electron microscopy (SEM), and Fourier-transform infrared (FT-IR) and X-ray photoelectron spectroscopy (XPS). And the detailed device types are displayed in the ESI.†The concentration of U(VI) at mg L−1 or μg L−1 level was determined using Inductively Coupled Plasma-Atomic Emission Spectroscopy (ICP-AES) with an IRIS Intrepid II XSP instrument, or using Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) with a X SeriesII instrument. All the experiments mentioned above were carried out in triplicate, and the reported results were the mean values of three data sets.
Results and discussion
The grafting efficiencies
The degree of grafting of HPEI onto the PANFs was assessed by the mass addition of the PANFs (Fig. 1), which shows that the grafting degrees of 7.19, 17.53, 33.29, and 66.08, denoted as PAN–HPEI-GD7.19, PAN–HPEI-GD17.53, PAN–HPEI-GD33.29, and PAN–HPEI-GD66.08, respectively, increase with the prolongation of reaction time; the grafting reaction is performed in the heterogeneous mode.23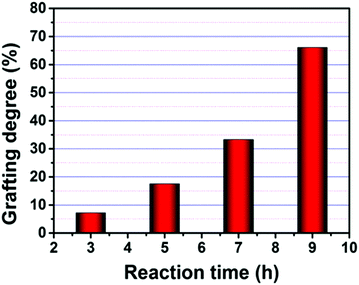 | ||
Fig. 1 Grafting degree of HPEI to PAN influenced by reaction time at 140 °C with a mass ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1. | ||
As an adsorbent material, hydrophilicity and porosity are paramount factors that influence the adsorption performance of the absorbents and directly relate to the quantity of ions in the water that contacts the functional groups. Taking the practical application into account, the mechanical properties are also very important. Herein, the hydrophilicity, porosity, and mechanical properties have been explored11 and displayed in the ESI.†
Competing ion experiments
Because of the existence of various metal ions in natural seawater, we first carried out ion competition tests in the presence of Sr2+, Ba2+, Ca2+, VO3−, Na+, Mg2+, and Zn2+ to evaluate if the adsorbents can be used in seawater.24,25Fig. 2 displays the results, and Fig. 2A shows the adsorption capacity of the adsorbents with different grafting degrees for the adsorption of uranium, from which we see a maximum uptake of uranium by the adsorbent with the same grafting degree; the one with a grafting degree of 33.29 shows the best uranium adsorption performance. Fig. 2B displays the removal rate and the distribution coefficient of the adsorption. It is clear that the removal rates of U and V are much higher than the other cations, and the distribution coefficient of V is one order higher than that of U. In addition, only when the grafting degree is 33.29%, the removal rate of U is as much as V, reaching up to 99%. From the above, we realize that PAN–HPEI-GD33.29 shows promising potential in the application of uranium removal under the reaction conditions.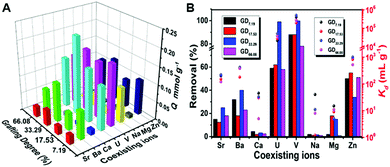 | ||
| Fig. 2 (A) Adsorption capacity and (B) removal rate and Kd for uranium removal with different grafting degrees of PAN–HPEI in the presence of several competing ions. | ||
Uranium adsorption tests in simulated seawater
Based on the above ion competition results, the performance to remove U(VI) under simulated seawater was assessed, as exhibited in Fig. 3. The removal rate of uranium clearly increases first and then decreases as the grafting degree increases. In addition, the best removal rate is achieved with PAN–HPEI-GD33.29 at a specified concentration. The removal rates are 94.8, 96.2, 94.3, 96.2, and 97.6%, respectively, indicating the use of PAN–HPEI as a potential adsorbent under simulated seawater conditions. This might be ascribed to the increasing number of active sites which can effectively coordinate with uranium on the adsorbents with the increase in grafting degrees, and the steric hindrance caused by the grafted HPEI reducing the opportunity to make contact with active sites and U(VI) subsequently. We chose PAN–HPEI-GD33.29 for further study to clarify the adsorption behavior and mechanism.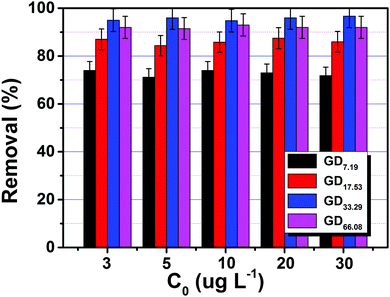 | ||
| Fig. 3 Removal of U(VI) by PAN–HPEI with different grafting degrees under simulated seawater. (V = 50 mL, T = 298 K, m = 0.02 g). | ||
Effect of pH on uranium(VI) adsorption
Solution acidity affects greatly the process of U(VI) extraction and the species of U(VI) in aqueous solution as well as the chemical properties and binding sites on the surface of the fiber.26 The influence of pH on uranyl removal by PAN–HPEI was conducted in the range of 2.0–8.0, and the zeta potential was measured to determine the optimum pH and operating mechanism for the process (Fig. 4). The U(VI) removal efficiency by PAN–HPEI largely depends on the solution pH, and the point of zero charge (pHpzc) is estimated to be 5.8. The quantity of adsorbed U(VI) increases swiftly from pH 2.0 to 6.0, during which the zeta potential is positive, and reduces slowly with further increase in pH, during which the zeta potential is negative; a maximum loading capacity of 416.47 mg g−1 is achieved at pH 6.0.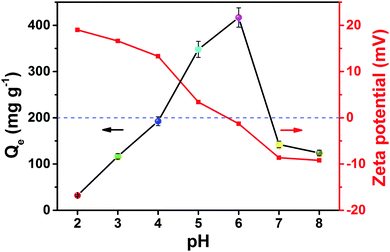 | ||
| Fig. 4 Effect of pH on the adsorption of U(VI). (C0 = 200 mg L−1, V = 50 mL, T = 298 K, m = 0.02 g). | ||
When the pH is under 5.0, UO22+ is the main form of U(VI) existing in the solution. The adsorption process between U(VI) ions and PAN–HPEI is restricted by electrostatic repulsion and the effective competition of H+ on the binding sites.27 With a pH higher than 5.0 and electrostatic repulsion decreasing the coordinating ability of the functional ligands (such as amine, imine, and carbonyl) is promoted,28 chemical complexation takes a major part in the adsorption process. In the pH range of 6.0–8.0, uranium combined with hydroxide and formed different complexes such as UO2(OH)3− and (UO2)3(OH)7−. Meanwhile, it is observed that the ζ-potential value was negative from 6 to 8, leading to electrostatic repulsion between these ions and the adsorbent surface. Accordingly, the optimum pH during the adsorption process is 6.0, which we select for further experiments.
Effect of contact time on uranium sorption
To gauge the equilibrium time, a crucial factor in the adsorption process, the removal of U(VI) by PAN–HPEI was analyzed as a function of contact time, as displayed in Fig. 5. The experiments were carried out with contact times ranging between 1 and 90 min at 298 K. The adsorption rate increases rapidly in the first 10 min and then gradually tends toward a steady state, in which equilibrium is reached in 30 min and the equilibrium uptake is 465 mg g−1. The rapid capture of U(VI) implies that PAN–HPEI possesses numerous active sites on the surface in the initial stage; accordingly, a contact time of 30 min is reasonable for saturated adsorption of uranyl onto PAN–HPEI.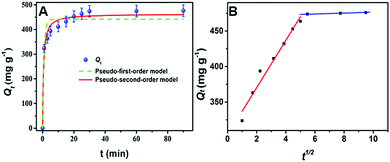 | ||
| Fig. 5 Effect of contact time on the adsorption of U(VI). (C0 = 200 mg L−1, pH = 6, V = 50 mL, T = 298 K, m = 0.02 g). | ||
To deeply understand the physical chemistry process and to establish the kinetics, we investigated the kinetic mechanism of the process. The kinetics data were tested and modeled with pseudo-first order and pseudo-second order rate models, and the intra-particle diffusion model.29,30 The details of these equations are displayed in the ESI.†
The kinetic parameters computed from the above models are listed in Table 1, from which we see that the calculated Qe value in pseudo-second order kinetics is close to the experimental data with a regression coefficient of 0.9772, which verifies that the extraction of uranyl ions onto PAN–HPEI is more accurately depicted by a pseudo-second order equation. These results are suggestive that a pseudo-second order model is the uppermost mechanism and the calculated rate constant seems to be governed by a chemisorption process, which implies that chemical adsorption occurs involving electronic sharing and transfer between PAN–HPEI and U(VI) ions.
| Kinetic model | T (°C) | C 0 (mg L−1) | Q expe (mg g−1) | Q cale (mg g−1) | k 1 (min−1)/k2 (g (mg min)−1) | R 2 |
|---|---|---|---|---|---|---|
| Pseudo-first-order | 25 | 200 | 475 | 441.6 | 1.1518 | 0.9314 |
| Pseudo-second-order | 25 | 200 | 475 | 463.3 | 0.0038 | 0.9772 |
| Intra-particle diffusion | 25 | 200 | 475 | 471.0 | 33.30/0.52 | 0.9622/0.9953 |
As depicted in Fig. 5B, the plot of Qtversus t1/2 presents two straight lines, showing that the adsorption process of uranyl on PAN–HPEI includes two stages. The removal rate is fastest in the initial twenty minutes, in which the uranium ions move toward the surface of PAN–HPEI with a k3 value of 33.30 mg g−1 min−0.5. For the second stage, the fitted curve exhibits a comparatively low slope by virtue of a decrease in available active sites, implying an approaching adsorption equilibrium state, which further indicates that the adsorption on PAN–HPEI is via chemical bonding between the active sites on the surface of PAN–HPEI and U(VI).
Effect of temperature and adsorption thermodynamics
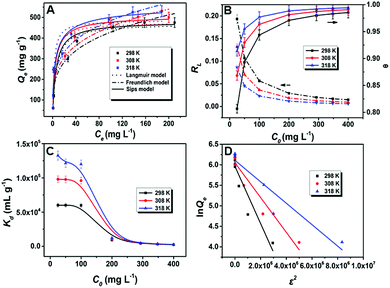
Broadly speaking, the equilibrium studies are constructive and meaningful for both the design of the adsorption process and comprehension of the adsorption mechanism. An adsorbate may be adsorbed from aqueous media onto the surface of an adsorbent by some kind of interaction mode. An effective mechanism is related to the nature of the adsorption sites, the surface, the affinities of the adsorbent, the type of the adsorbate, and the adsorption conditions. In general, the speed of adsorption and adsorption capacity increase as the temperature increases. The impact of temperature on U(VI) sorption onto PAN–HPEI was explored using a water bath at 298, 308, and 318 K, and the adsorption isotherms of U(VI) on PAN–HPEI are shown in Fig. 6A, illustrating that an elevated temperature is profitable for U(VI) sorption. Herein, the Kd values are in the range of 2.0 × 103 to 1.3 × 105 mL g−1 (Fig. 6C). Kd values above 3.0 × 103 are generally recognized as desirable, whereas above 1.0 × 104 as excellent.31 The majority of Kd values of PAN–HPEI being >5.0 × 103 (even >1.0 × 104) implies a high affinity of the modified fibers toward U(VI). Accordingly, PAN–HPEI is regarded as an ideal and potential material for the harvesting of U(VI).The interaction between uranyl and active sites on the surface of PAN–HPEI may be interpreted as a chelation mechanism. For the design and manipulation of extraction systems, coefficient of equilibrium data is essential. Langmuir, Freundlich, Sips and D–R isotherm models were used for the acquired adsorption values to verify the adsorption parameters that drive the adsorption process. The details of these equations are explored in the ESI,† while the results are displayed in Fig. 6A and D. Fig. 6B depicts the relationship of θ, RL and initial concentration of U(VI) ions. Evidently, the adsorption process proceeds fast in the initial stage (low coverage of the fiber surface and plenty of free active sites are available for binding with U(VI) ions) and then tends toward a plateau at a higher surface coverage where most of the active sites are occupied. This reveals the applicability of the Langmuir model to describe the adsorption of uranyl onto PAN–HPEI.
The thermodynamics parameters as well as the correlation coefficients are listed in Table 2, which indicates that the Sips model better fits the experimental results over the experimental range, with high correlation coefficients (>0.97). The Sips model shows that the extraction process is a union of Langmuir and Freundlich models. The maximum loading capacity of PAN–HPEI is determined as 481.26 mg g−1 at 298 K.
| Isotherm parameters | 298 K | 308 K | 318 K |
|---|---|---|---|
| Langmuir isotherm | |||
| q m (mg U g−1) | 470.67 | 476.49 | 486.87 |
| K L (L mg−1) | 0.1669 | 0.2639 | 0.4237 |
| R 2 | 0.9407 | 0.9160 | 0.8724 |
| Freundlich isotherm | |||
| K F (L g−1) | 131.85 | 147.36 | 164.74 |
| N | 0.2561 | 0.2451 | 0.2344 |
| R 2 | 0.9085 | 0.9218 | 0.9171 |
| Dubinin–Radushkevich (D–R) | |||
| B | 6.97 × 10−7 | 4.24 × 10−7 | 2.61 × 10−7 |
| Q DR | 388.23 | 424.02 | 449.78 |
| E | 846.94 | 1086.16 | 1383.27 |
| R 2 | 0.8677 | 0.9199 | 0.8976 |
| Langmuir–Freundlich (Sips) | |||
| Q s | 481.26 | 510.18 | 574.63 |
| K s | 0.2009 | 0.3009 | 0.3730 |
| M | 0.0617 | 0.1599 | 0.3718 |
| R 2 | 0.9733 | 0.9663 | 0.9366 |
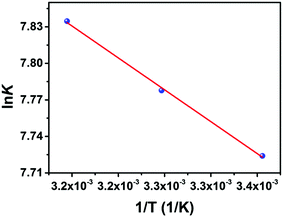
The thermodynamic parameters, enthalpy (ΔH°) and entropy (ΔS°) changes and Gibbs free energy (ΔG°) were also computed and the formulae were listed in the ESI.† The results of ΔS°and ΔH° are appraised from the slope and intercept of the fitting line of ln KDvs. 1/T (Fig. 7). Positive standard entropy (ΔS°) implies that randomness increases at the fiber/solution interface in the course of extraction, which is in virtue of the liberation of free water molecules in the chelation sphere as a consequence of chelation of uranyl at the carboxyl and amine-active sites. A positive value of ΔH° suggests that the essence of the extraction process of UO22+ is endothermic and the binding of U(VI) ions increases as the temperature rises.
ΔG° values at diverse temperatures are also attained. The values of ΔH°, ΔS°, and ΔG° are listed in Table 3. Minus values of ΔG° manifest that the process accords with a spontaneous and feasible tendency. ΔG°, in the range of −25.063 and −23.486, reduces with increase in temperature, which implies that higher temperatures facilitate adsorption of U(VI) ions onto PAN–HPEI due to a greater driving force.
| ΔH° (kJ mol−1) | ΔS° (J mol−1 K−1) | ΔG° (kJ mol−1) | ||
|---|---|---|---|---|
| 4.357 | 78.83 | 298 K | 308 K | 318 K |
| −23.486 | −24.275 | −25.063 | ||
Desorption and reusability study
To prove the feasibility of regeneration of the PAN–HPEI adsorbents after adsorption of uranyl, a desorption experiment was implemented with PAN–HPEI0.33. All of the concentrations of the desorption agents are the same at 0.1 mol L−1 and Fig. 8A shows that the best desorption agent is HCl solution. Hence, multiple concentrations of HCl varying from 0.01 to 1.0 mol L−1 were examined for the elution of adsorbed uranyl using PAN–HPEI (the results are shown in Fig. S3 in the ESI†). The percentages for desorption with HCl concentrations of 0.01, 0.05, 0.1, 0.5, and 1.0 mol L−1 are 65.2%, 74.8%, 79.2%, 88.1%, 88.3%, respectively; therefore, on considering optimization and economic efficiency, we select as the best choice a concentration of 0.5 M HCl.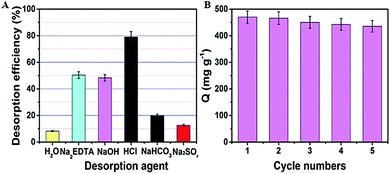 | ||
| Fig. 8 (A) Desorption efficiency for the removal of U(VI) by different desorption agents. (B) Adsorption–desorption cycles of U(VI) from aqueous solutions. | ||
To estimate the cyclic utilization of the fiber, an adsorption–desorption cycle test with 0.5 M HCl was carried out five times, which was shown in Fig. 8B. After five circulations, the elution efficiency of the PAN–HPEI is still more than 82%, which suggests that the fiber can be utilized availably in a real process, for instance, extraction of uranyl from large volumes of aqueous solutions.
Characterization of samples
To research and analyze if the surface functional groups change in the process of modification, adsorption, and desorption, FT-IR spectra of (A) raw PAN, (B) PAN–HPEI0.33, (C) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U (the PAN–HPEI0.33 after U adsorption) and (D) PAN–HPEI0.33
Ad-U (the PAN–HPEI0.33 after U adsorption) and (D) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) De-U (the PAN–HPEI0.33 after U desorption) were recorded and are presented in Fig. 9. Fig. S1 (ESI†) presents the FT-IR spectra of the PAN–HPEI with different grafting degrees. A distinct and gradual weakening peak at 2243 cm−1 in the raw PAN and PAN–HPEI0.33 is attributed to the C
De-U (the PAN–HPEI0.33 after U desorption) were recorded and are presented in Fig. 9. Fig. S1 (ESI†) presents the FT-IR spectra of the PAN–HPEI with different grafting degrees. A distinct and gradual weakening peak at 2243 cm−1 in the raw PAN and PAN–HPEI0.33 is attributed to the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N group in the PANFs,32 which means that not all C
N group in the PANFs,32 which means that not all C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N participates in the hydrolysis and graft reactions. The band at 2923 cm−1 is the stretching vibration of C–H (Curve A), which red-shifts slightly after the graft reaction (Curve B). We speculate that the electron cloud density weakens with a decrease in electronegativity during the process of the conversion of nitrile groups into oxygen-containing functional groups. The band at 1635 cm−1 is assigned to an O
N participates in the hydrolysis and graft reactions. The band at 2923 cm−1 is the stretching vibration of C–H (Curve A), which red-shifts slightly after the graft reaction (Curve B). We speculate that the electron cloud density weakens with a decrease in electronegativity during the process of the conversion of nitrile groups into oxygen-containing functional groups. The band at 1635 cm−1 is assigned to an O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching bond in the amide I band. The new peak located at 1383 cm−1 is the C–N bending vibration bond. Furthermore, the intense C
C stretching bond in the amide I band. The new peak located at 1383 cm−1 is the C–N bending vibration bond. Furthermore, the intense C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O asymmetric stretching bond frequency of COO− is also found at 1538 cm−1, which overlaps with N–H deformation vibrations. The new peak position at 667 cm−1 is ascribed to the bending vibration of N–H. The broad peak at 3700–3050 cm−1 corresponds to the combination of N–H and –OH groups,33 which signifies a successful modification of HPEI. Presumably, the active sites, such as carboxyl groups, and amine and imide groups, are present in abundance in PAN–HPEI, which is indicative of binding with U(VI) ions through a complexation mechanism. Furthermore, minor red-shifts in the spectra are observed, such as O
O asymmetric stretching bond frequency of COO− is also found at 1538 cm−1, which overlaps with N–H deformation vibrations. The new peak position at 667 cm−1 is ascribed to the bending vibration of N–H. The broad peak at 3700–3050 cm−1 corresponds to the combination of N–H and –OH groups,33 which signifies a successful modification of HPEI. Presumably, the active sites, such as carboxyl groups, and amine and imide groups, are present in abundance in PAN–HPEI, which is indicative of binding with U(VI) ions through a complexation mechanism. Furthermore, minor red-shifts in the spectra are observed, such as O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, –OH, and N–H, which are attributed to a change of the electron cloud distribution triggered by the interaction between N/O containing groups and U(VI). In particular, the emergence of a new peak at 904 cm−1 (Curve C) illustrates the antisymmetric stretching vibration of the O
C, –OH, and N–H, which are attributed to a change of the electron cloud distribution triggered by the interaction between N/O containing groups and U(VI). In particular, the emergence of a new peak at 904 cm−1 (Curve C) illustrates the antisymmetric stretching vibration of the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) U
U![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group,7,9 which does not exist in PAN–HPEI. After the desorption of U(VI), the band at UO22+ weakens but does not disappear, implying that U(VI) is not completely removed.
O group,7,9 which does not exist in PAN–HPEI. After the desorption of U(VI), the band at UO22+ weakens but does not disappear, implying that U(VI) is not completely removed.
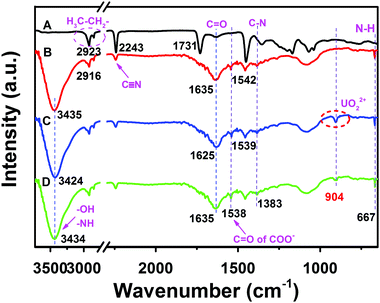 | ||
Fig. 9 FT-IR spectra of (A) raw PAN, (B) PAN–HPEI0.33, (C) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U, and (D) PAN–HPEI0.33 Ad-U, and (D) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) De-U. De-U. | ||
The surface morphologies of (A) PAN–HPEI0.33, (B) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U, and (C) PAN–HPEI0.33
Ad-U, and (C) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) De-U were observed by SEM and are recorded in Fig. 10. Fig. S2 (ESI†) presents the SEM images of raw PAN and PAN–HPEI with different grafting degrees. We observe that the surface of raw PAN is somewhat smooth. After U(VI) adsorption, however, the surface roughens with a covering of a dense layer of small particles, whereas, after desorption, the fiber body restores its smoothness. In the interest of evaluating the change of surface elemental composition, the EDS spectra of (D) PAN–HPEI0.33, (E) PAN–HPEI0.33
De-U were observed by SEM and are recorded in Fig. 10. Fig. S2 (ESI†) presents the SEM images of raw PAN and PAN–HPEI with different grafting degrees. We observe that the surface of raw PAN is somewhat smooth. After U(VI) adsorption, however, the surface roughens with a covering of a dense layer of small particles, whereas, after desorption, the fiber body restores its smoothness. In the interest of evaluating the change of surface elemental composition, the EDS spectra of (D) PAN–HPEI0.33, (E) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U, and (F) PAN–HPEI0.33
Ad-U, and (F) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) De-U were also recorded (Fig. 10). The presence of U(VI) with a wt(%) of 9.45 on the surface of the fiber dramatically changes the morphology of PAN–HPEI0.33, confirming the adsorption of U(VI) onto the fiber surface. After the desorption, the wt(%) of U(VI) decreases to 0.48, signifying an incomplete removal of U(VI), which is agreement with the results of desorption experiments and FT-IR.
De-U were also recorded (Fig. 10). The presence of U(VI) with a wt(%) of 9.45 on the surface of the fiber dramatically changes the morphology of PAN–HPEI0.33, confirming the adsorption of U(VI) onto the fiber surface. After the desorption, the wt(%) of U(VI) decreases to 0.48, signifying an incomplete removal of U(VI), which is agreement with the results of desorption experiments and FT-IR.
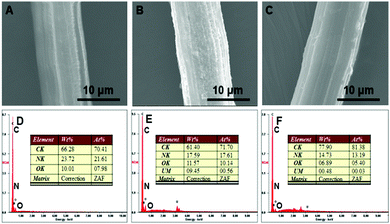 | ||
Fig. 10 SEM images and EDS spectra of (A) and (D) PAN–HPEI0.33, (B) and (E) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U, and (C) and (F) PAN–HPEI0.33 De-U. Ad-U, and (C) and (F) PAN–HPEI0.33 De-U. | ||
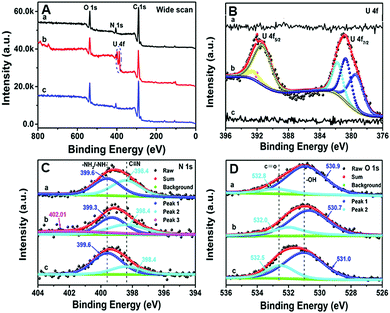
To ascertain the surface chemical composition and bonding environment after U(VI) adsorption and desorption, the XPS spectra of (a) PAN–HPEI0.33, (b) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U, and (c) PAN–HPEI0.33
Ad-U, and (c) PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) De-U were recorded (Fig. 11). The results assist in verifying the binding mechanisms of PAN–HPEI and U(VI) ions. Compared with the survey spectra of PAN–HPEI0.33 (Fig. 11A), the spectra of PAN–HPEI0.33-U obviously show a new and strong double U 4f peak, and a corresponding high resolution of U 4f (Fig. 11B), characterized with U 4f5/2 (∼392 eV) and U 4f7/2 (∼382 eV) core-level spectra, disclosing the existence of U(VI) in PAN–HPEI0.33
De-U were recorded (Fig. 11). The results assist in verifying the binding mechanisms of PAN–HPEI and U(VI) ions. Compared with the survey spectra of PAN–HPEI0.33 (Fig. 11A), the spectra of PAN–HPEI0.33-U obviously show a new and strong double U 4f peak, and a corresponding high resolution of U 4f (Fig. 11B), characterized with U 4f5/2 (∼392 eV) and U 4f7/2 (∼382 eV) core-level spectra, disclosing the existence of U(VI) in PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U,34 respectively. Meanwhile, we observe pronounced shifts of N 1s and O 1s (Fig. 11C and D), which indicate chemical complexing reactions between nitrogen and oxygen atoms and U(VI) ions, respectively. Before the adsorption of U(VI) the N 1s spectra can be fitted to two peaks as seen on Curve a in Fig. 11C, of which peaks 1 and 2 at binding energies of 399.6 and 398.4 eV are ascribed to NH2 or NH groups and C
Ad-U,34 respectively. Meanwhile, we observe pronounced shifts of N 1s and O 1s (Fig. 11C and D), which indicate chemical complexing reactions between nitrogen and oxygen atoms and U(VI) ions, respectively. Before the adsorption of U(VI) the N 1s spectra can be fitted to two peaks as seen on Curve a in Fig. 11C, of which peaks 1 and 2 at binding energies of 399.6 and 398.4 eV are ascribed to NH2 or NH groups and C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N,35 respectively. After the extraction of uranyl, a new peak appears at 402.1 eV (Curve b in Fig. 11C), while peak 1 experiences a positive shift of 0.3 eV and does not change, which further demonstrates that uranyl ions and amine groups chemically interact.36 After desorption, peak 1 and peak 2 return to their original positions. Before the adsorption of U(VI), the O 1s spectra can be fitted to two peaks as seen on Curve a in Fig. 11D, of which peaks 1 and 2 at 399.60 and 398.40 eV belong to –OH and C
N,35 respectively. After the extraction of uranyl, a new peak appears at 402.1 eV (Curve b in Fig. 11C), while peak 1 experiences a positive shift of 0.3 eV and does not change, which further demonstrates that uranyl ions and amine groups chemically interact.36 After desorption, peak 1 and peak 2 return to their original positions. Before the adsorption of U(VI), the O 1s spectra can be fitted to two peaks as seen on Curve a in Fig. 11D, of which peaks 1 and 2 at 399.60 and 398.40 eV belong to –OH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O,20,37 respectively. After the adsorption of U(VI) (Curve b in Fig. 11D), peak 1 encounters a negative shift of 0.2 eV and peak 2 experiences a negative shift as much as 0.8 eV, both of which indicate chemical combinations between uranyl ions and oxygen-containing functional groups on PAN–HPEI.38 After the desorption of U(VI), peak 1 and peak 2 undergo a positive shift to almost their original positions.
O,20,37 respectively. After the adsorption of U(VI) (Curve b in Fig. 11D), peak 1 encounters a negative shift of 0.2 eV and peak 2 experiences a negative shift as much as 0.8 eV, both of which indicate chemical combinations between uranyl ions and oxygen-containing functional groups on PAN–HPEI.38 After the desorption of U(VI), peak 1 and peak 2 undergo a positive shift to almost their original positions.
Proposed mechanism
In general, physisorption and/or chemisorption are the basic principles for solid materials involving the extraction of metal ions. Chemisorption is facilely affected by outside factors, particularly acidity of the medium. One factor is the removal performance of the material which generally weakens due to protonation of the surface functional ligands. However, physisorption is little or nothing, as influenced by medium acidity. Furthermore, taking the composition and structure of PAN–HPEI into account (as shown in Scheme 1), numerous active sites on the as-prepared fibers cause effective complexation with U(VI) ions.In addition, the FT-IR and XPS spectra of PAN–HPEI and PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U were recorded to better understand the mechanism for the adsorption of U(VI) onto PAN–HPEI. Comparing the FT-IR spectra of PAN–HPEI and PAN–HPEI0.33
Ad-U were recorded to better understand the mechanism for the adsorption of U(VI) onto PAN–HPEI. Comparing the FT-IR spectra of PAN–HPEI and PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U it is evident that PAN–HPEI extracts uranyl ions primarily through chemical bonding. Of more importance, the XPS spectra of PAN–HPEI and PAN–HPEI0.33
Ad-U it is evident that PAN–HPEI extracts uranyl ions primarily through chemical bonding. Of more importance, the XPS spectra of PAN–HPEI and PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U strongly confirm the existence of complexation between U(VI) ions and oxygen and nitrogen ligands onto PAN–HPEI.4 Combined with the result of ζ-potential, an electrostatic attraction exists with pH < 5.8, and chemical bonding most likely plays a leading role with partial electrostatic attraction, as displayed in Scheme 2.
Ad-U strongly confirm the existence of complexation between U(VI) ions and oxygen and nitrogen ligands onto PAN–HPEI.4 Combined with the result of ζ-potential, an electrostatic attraction exists with pH < 5.8, and chemical bonding most likely plays a leading role with partial electrostatic attraction, as displayed in Scheme 2.
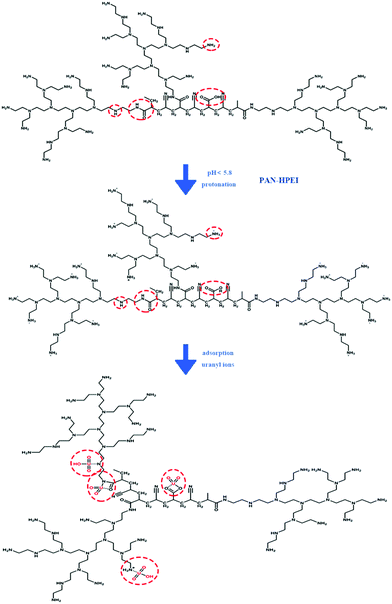 | ||
| Scheme 2 Schematic diagram of the proposed mechanism for the adsorption of uranyl ions onto PAN–HPEI fibers. | ||
Conclusions
In conclusion, we synthesized covalently modified PAN–HPEI fibers with different grafting degrees, of which the best adsorption behavior is 33.29%. Importantly, the removal rate at low concentrations under artificial seawater conditions reaches more than 90%. The extraction process is pH-dependent and is accomplished in no more than 60 min with a high loading capacity of 470.67 mg g−1. Moreover, PAN–HPEI0.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ad-U is facilely separated from solutions and efficaciously regenerated by HCl. Furthermore, the structures before and after U(VI) adsorption are well characterized by SEM, EA, FT-IR, and XPS to infer the nature of the adsorption mechanisms. The ease of operation and impactful adsorption capability are indicative that PAN–HPEI can be used as a highly interactive and prospective adsorption material for the harvesting of uranyl from seawater.
Ad-U is facilely separated from solutions and efficaciously regenerated by HCl. Furthermore, the structures before and after U(VI) adsorption are well characterized by SEM, EA, FT-IR, and XPS to infer the nature of the adsorption mechanisms. The ease of operation and impactful adsorption capability are indicative that PAN–HPEI can be used as a highly interactive and prospective adsorption material for the harvesting of uranyl from seawater.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC 51402065), the International Science & Technology Cooperation Program of China (2015DFR50050), the Fundamental Research Funds of the Central University (HEUCFM), Heilongjiang Province Natural Science Funds for Distinguished Young Scholar (JC201404), and the Major Project of Science and Technology of Heilongjiang Province (GA14A101).Notes and references
- S. Tan, J. Yang, J. Yan, C. Lee, H. Hashim and B. Chen, Appl. Energy, 2017, 185, 1919–1930 CrossRef.
- W. Li, Q. Liu, J. Liu, H. Zhang, R. Li, Z. Li, X. Jing and J. Wang, Appl. Surf. Sci., 2017, 403, 378–388 CrossRef CAS.
- Y. Lu, Nat. Chem., 2014, 6, 175–177 CrossRef CAS PubMed.
- S. Das, S. Brown, R. T. Mayes, C. J. Janke, C. Tsouris, L. J. Kuo, G. Gill and S. Dai, Chem. Eng. J., 2016, 298, 125–135 CrossRef CAS.
- B. Li, C. Y. Bai, S. Zhang, X. S. Zhao, Y. Li, L. Wang, K. Ding, X. Shu, S. J. Li and L. J. Ma, J. Mater. Chem. A, 2015, 3, 23788–23798 CAS.
- S. E. Bailey, T. J. Olin, R. M. Bricka and D. D. Adrian, Water Res., 1999, 33, 2469–2479 CrossRef CAS.
- L. L. Wang, F. Luo, L. L. Dang, J. Q. Li, X. L. Wu, S. J. Liu and M. B. Luo, J. Mater. Chem. A, 2015, 3, 13724–13730 CAS.
- Y. Yamazaki, Y. Tachibana, T. Kaneshiki, M. Nomura and T. Suzuki, Prog. Nucl. Energy, 2015, 82, 74–79 CrossRef CAS.
- F. Li, D. Li, X. Li, J. Liao, S. Li, J. Yang, Y. Yang, J. Tang and N. Liu, Chem. Eng. J., 2016, 284, 630–639 CrossRef CAS.
- T. C. Dietz, C. E. Tomaszewski, Z. Tsinas, D. Poster, A. Barkatt, M. Adel-Hadadi, F. B. Bateman, L. T. Cumberland, E. Schneider, K. Gaskell, J. LaVerne and M. Al-Sheikhly, Ind. Eng. Chem. Res., 2016, 55, 4179–4186 CrossRef CAS.
- S. Xie, X. Liu, B. Zhang, H. Ma, C. Ling, M. Yu, L. Li and J. Li, J. Mater. Chem. A, 2015, 3, 2552–2558 CAS.
- A. P. Ladshaw, S. Das, W. P. Liao, S. Yiacoumi, C. J. Janke, R. T. Mayes, S. Dai and C. Tsouris, Ind. Eng. Chem. Res., 2016, 55, 4241–4248 CrossRef CAS.
- S. Deng, P. Wang, G. Zhang and Y. Dou, J. Hazard. Mater., 2016, 307, 64–72 CrossRef CAS PubMed.
- A. Centrone, Y. Yang, S. Speakman, L. Bromberg, G. C. Rutledge and T. A. Hatton, J. Am. Chem. Soc., 2010, 132, 15687–15691 CrossRef CAS PubMed.
- M. Ratanajanchai, S. Soodvilai, N. Pimpha and P. Sunintaboon, Mater. Sci. Eng., C, 2014, 34, 377–383 CrossRef CAS PubMed.
- M. Ghoul, M. Bacquet and M. Morcellet, Water Res., 2003, 37, 729–734 CrossRef CAS PubMed.
- X. Tian, W. Wang, Y. Wang, S. Komarneni and C. Yang, Microporous Mesoporous Mater., 2015, 207, 46–52 CrossRef CAS.
- J. B. Lindén, M. Larsson, S. Kaur, W. M. Skinner, S. J. Miklavcic, T. Nann, I. M. Kempson and M. Nydén, RSC Adv., 2015, 5, 51883–51890 RSC.
- S. Brown, Y. Yue, L.-J. Kuo, N. Mehio, M. Li, G. Gill, C. Tsouris, R. T. Mayes, T. Saito and S. Dai, Ind. Eng. Chem. Res., 2016, 55, 4139–4148 CrossRef CAS.
- F. Wang, H. Li, Q. Liu, Z. Li, R. Li, H. Zhang, L. Liu, G. A. Emelchenko and J. Wang, Sci. Rep., 2016, 6, 19367 CrossRef CAS PubMed.
- M. J. Manos and M. G. Kanatzidis, J. Am. Chem. Soc., 2012, 134, 16441–16446 CrossRef CAS PubMed.
- M. Al-Shannag, Z. Al-Qodah, K. Bani-Melhem, M. R. Qtaishat and M. Alkasrawi, Chem. Eng. J., 2015, 260, 749–756 CrossRef CAS.
- M. Wang, T. Jiang, Y. Lu, H. Liu and Y. Chen, J. Mater. Chem. A, 2013, 1, 5923–5933 CAS.
- P. Yang, Q. Liu, J. Liu, H. Zhang, Z. Li, R. Li, L. Liu and J. Wang, Ind. Eng. Chem. Res., 2017, 56, 3588–3598 CrossRef CAS.
- Y. Peng, H. Huang, D. Liu and C. Zhong, ACS Appl. Mater. Interfaces, 2016, 8, 8527–8535 CAS.
- J. Yang and B. Volesky, Water Res., 1999, 33, 3357–3363 CrossRef CAS.
- A. Schierz and H. Zänker, Environ. Pollut., 2009, 157, 1088–1094 CrossRef CAS PubMed.
- Y. Zhao, J. Li, S. Zhang, H. Chen and D. Shao, RSC Adv., 2013, 3, 18952–18959 RSC.
- N. Singh and K. Balasubramanian, RSC Adv., 2014, 4, 27691–27701 RSC.
- S. W. Zhang, M. Y. Zeng, J. X. Li, J. Li, J. Z. Xu and X. K. Wang, J. Mater. Chem. A, 2014, 2, 4391–4397 CAS.
- T. Wen, Q. Fan, X. Tan, Y. Chen, C. Chen, A. Xu and X. Wang, Polym. Chem., 2016, 7, 785–794 RSC.
- H. Zhao, X. Liu, M. Yu, Z. Wang, B. Zhang, H. Ma, M. Wang and J. Li, Ind. Eng. Chem. Res., 2015, 54, 3101–3106 CrossRef CAS.
- S. Deng, G. Zhang, X. Wang, T. Zheng and P. Wang, Chem. Eng. J., 2015, 276, 349–357 CrossRef CAS.
- G. Sheng, P. Yang, Y. Tang, Q. Hu, H. Li, X. Ren, B. Hu, X. Wang and Y. Huang, Appl. Catal., B, 2016, 193, 189–197 CrossRef CAS.
- Q. Shi, Y. Su, W. Chen, J. Peng, L. Nie, L. Zhang and Z. Jiang, J. Membr. Sci., 2011, 366, 398–404 CrossRef CAS.
- S. Deng, R. Bai and J. P. Chen, Langmuir, 2003, 19, 5058–5064 CrossRef CAS.
- M. Pakula, S. Biniak and A. Swiatkowski, Langmuir, 1998, 14, 3082–3089 CrossRef CAS.
- T. Classen, M. Lingenfelder, Y. Wang, R. Chopra, C. Virojanadara, U. Starke, G. Costantini, G. Fratesi, S. Fabris and S. de Gironcoli, J. Phys. Chem. A, 2007, 111, 12589–12603 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c7nj03243a |
| ‡ These authors contributed to the work equally and should be regarded as co-first authors. |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 |