Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Six new coordination compounds based on rigid 5-(3-carboxy-phenyl)-pyridine-2-carboxylic acid: synthesis, structural variations and properties†
Jiang-Feng Song *a,
Ying-Ying Jiaa,
Rui-Sha Zhoua,
Si-Zhe Lia,
Xiao-Min Qiua and
Jie Liu*b
*a,
Ying-Ying Jiaa,
Rui-Sha Zhoua,
Si-Zhe Lia,
Xiao-Min Qiua and
Jie Liu*b
aDepartment of Chemistry, North University of China, Taiyuan, Shanxi 030051, P. R. China. E-mail: jfsong0129@nuc.edu.cn
bState Key Laboratory of Polymer Physics and Chemistry, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun, 130022, P. R. China. E-mail: liujie@ciac.ac.cn
First published on 23rd January 2017
Abstract
Six new coordination compounds Ni(cppca)(H2O)4·2H2O (1), Co(cppca)(H2O)4·2H2O (2), {Cd2(cppca)2(H2O)5}n (3), {[Cd3(cppca)2(H2P2O7)(H2O)6]·2H2O}n (4), {Cu(Hcppca)2}n (5) and {[Mn7(cppca)6(Hcppca)2(H2O)10]·2H2O}n (6) have been obtained by reactions of the corresponding metal salts and the rigid ligand 5-(3-carboxy-phenyl)-pyridine-2-carboxylic acid (H2cppca) under solvothermal conditions. All the compounds were fully characterized by single-crystal X-ray diffraction, elemental analysis, IR spectroscopy, thermal analysis and powder X-ray diffraction. The single-crystal X-ray analyses showed that compounds 1–6 have rich structural chemistry ranging from mononuclear (1 and 2), one-dimensional (3 and 4), two-dimensional (5) to three-dimensional (6) structures. Moreover, the framework of compound 6 may be simplified into a 3-nodal net with Schlafli symbol {42·6}{42·82·102}{43·62·8·104}2, which shows an unprecedented (3,4,5)-connected topology net. The fluorescent properties of compounds 3 and 4 were investigated in the solid state and in various solvent emulsions, which indicated that compounds 3 and 4 are both highly sensitive fluorescent probes for the acetone molecules. Variable-temperature magnetic susceptibility measurements indicate weak antiferromagnetic interactions between metal centers in compounds 1, 2, 5 and 6.
Introduction
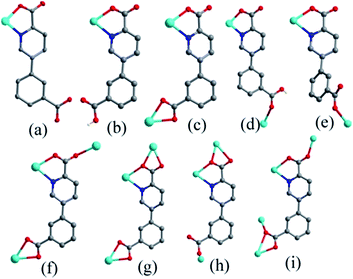
Coordination compounds have attracted considerable attention due to their intriguing structures1 and wide potential applications in gas storage and separation,2 luminescence,3 molecular magnetism,4 catalysis,5 etc. Although lots of effort has been put into developing the ability to control the reactions, and significant progress has also been made, the controllable synthesis of coordination compounds with unique structure and function still remains a long-term challenge because their self-assembly processes are often influenced by compositional (chemical structure of ligands chosen, kind of metal salt, pH value, solvent system, metal-to-ligand ratio, etc.) and process parameters (reaction time, temperature, and pressure).6As is well known, the selection and design of proper organic ligands have a large impact on the construction of coordination compounds with novel structures and unique properties. Among the chosen ligands, N-heterocyclic carboxylates have been widely used to construct coordination compounds due to their multiplicity of coordinated atoms and wide range of binding modes resulting in good ligating ability to metal ions in various arrangements.7 To date, diverse complexes based on N-heterocyclic carboxylates such as imidazole-carboxylic acid,8 pyridine-carboxylic acid,9 pyrimidine-carboxylic acid,10 and so on, have been reported, which possess intriguing topology structures and specific functional characteristics. Here, the conjugated N-heterocyclic dicarboxylic ligand, 5-(3-carboxy-phenyl)-pyridine-2-carboxylic acid (H2cppca), is introduced as a rigid linker for the reasons as follows: (i) it possesses four carboxylic oxygen atoms and one pyridyl nitrogen atom, which can assume many kinds of bridging or chelating modes to meet the geometric coordination requirement of metal centers. (ii) The H2cppca ligand might be partially or completely deprotonated at different pH values to adopt various acidity-dependent binding modes, on the other hand, it can act as hydrogen bond acceptors and donors to benefit the formation of various supramolecular structures.11 (iii) The flexible C–C single bonds between phenyl and pyridyl rings can rotate freely, as results in the formation of cis- and trans-conformations and enrich structures of coordination compounds. (iv) The conjugated phenyl and pyridyl system of the H2cppca ligand is helpful to provide remarkable luminescent performances and stable architecture.12 Based on the above mentioned analysis, the H2cppca ligand should be an effective component in the design of functional coordination complexes.
Herein, we describe the design and synthesis of six new coordination compounds based on H2cppca, Ni(cppca)(H2O)4·2H2O (1), Co(cppca)(H2O)4·2H2O (2), {Cd2(cppca)2(H2O)5}n (3), {[Cd3(cppca)2(H2P2O7)(H2O)6]·2H2O}n (4), {Cu(Hcppca)2}n (5), {[Mn7(cppca)6(Hcppca)2(H2O)10]·2H2O}n (6). All the compounds were fully characterized by single-crystal X-ray diffraction, elemental analysis, IR spectroscopy, thermal analysis and powder X-ray diffraction. The fluorescence properties of compounds 3 and 4 in the solid state and in various solvent emulsions have also been investigated, displaying highly sensitive fluorescent probes for the acetone molecules. The magnetic properties of compounds 1, 2, 5 and 6 have been investigated.
Experimental
Materials and physical measurements
All reagents and solvents were commercially available and used as received without further purification. Elemental analysis (C, H, and N) was performed on a Perkin-Elmer 240C elemental analyzer. Infrared (IR) spectra were obtained with KBr Pellets on a Perkin Elmer Spectrum One FTIR spectrometer in the range of 4000–400 cm−1 with the resolution (4.0 cm−1) and the scan's numbers.10 Powder X-ray diffraction (PXRD) patterns of the samples were recorded by a RIGAKU-DMAX2500 X-ray diffractometer using Cu-Kα radiation (λ = 1.542 Å) with a scanning rate of 10° min−1 and a step size of 0.02°. Thermalgravimetric analysis (TGA) was performed on a Perkin-Elmer TGA-7000 thermogravimetric analyzer with a heating rate of 10 °C min−1. The solvent emulsion and solid fluorescent spectra of compounds 3 and 4 were obtained on a HITACHI F-2700 fluorescence Spectrophotometer at room temperature. Variable-temperature magnetic susceptibility measurements were collected using SQUID magnetometer MPMS XL-7 (Quantum Design) at 1.0 kOe in the temperature range of 2–300 K.Synthesis of compounds 1–6
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 6 mL), which was sealed in a 20 mL Teflon-lined stainless-steel reactor, heated at 80 °C for 72 h under autogenous pressure. After slowly cooling to room temperature, green stick crystals of compound 1 were collected by filtration and washed with distilled water several times. Yield: 60% (based on the H2cppca). Elemental anal. calcd C13H19NNiO10 (408): C, 38.24; H, 4.66; N, 3.43. Found: C, 38.25; H, 4.65; N, 3.45. IR data (KBr, cm−1): 3358(s), 3174(s), 1618(s), 1543(s), 1383(s), 1255(m), 881(m), 759(m).
3, 6 mL), which was sealed in a 20 mL Teflon-lined stainless-steel reactor, heated at 80 °C for 72 h under autogenous pressure. After slowly cooling to room temperature, green stick crystals of compound 1 were collected by filtration and washed with distilled water several times. Yield: 60% (based on the H2cppca). Elemental anal. calcd C13H19NNiO10 (408): C, 38.24; H, 4.66; N, 3.43. Found: C, 38.25; H, 4.65; N, 3.45. IR data (KBr, cm−1): 3358(s), 3174(s), 1618(s), 1543(s), 1383(s), 1255(m), 881(m), 759(m).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 7 mL) was stirred in a 25 mL beaker for 5 minutes at room temperature. The above-mentioned mixtures, in which pyrophosphate aqueous solution (0.1 mL, 0.1 mol L−1) was added, were transferred and sealed in a 20 mL Teflon-lined stainless steel reactor and kept under autogenous pressure at 80 °C for 72 h. After slowly cooling to room temperature, light-yellow block crystals were obtained and washed with distilled water. Yield: 43% (based on the H2cppca). Elemental anal. calcd C26H32Cd3N2O23P2 (1139.68): C, 27.38; H, 2.81; N, 2.46. Found: C, 27.42; H, 2.79; N, 2.50. IR data (KBr, cm−1): 3408(s), 1608(m), 1548(s), 1399(s), 1092(s), 871(m), 771(m).
3, 7 mL) was stirred in a 25 mL beaker for 5 minutes at room temperature. The above-mentioned mixtures, in which pyrophosphate aqueous solution (0.1 mL, 0.1 mol L−1) was added, were transferred and sealed in a 20 mL Teflon-lined stainless steel reactor and kept under autogenous pressure at 80 °C for 72 h. After slowly cooling to room temperature, light-yellow block crystals were obtained and washed with distilled water. Yield: 43% (based on the H2cppca). Elemental anal. calcd C26H32Cd3N2O23P2 (1139.68): C, 27.38; H, 2.81; N, 2.46. Found: C, 27.42; H, 2.79; N, 2.50. IR data (KBr, cm−1): 3408(s), 1608(m), 1548(s), 1399(s), 1092(s), 871(m), 771(m).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 8 mL) solutions, and stirred for 0.5 h, then the mixture was transferred into a 20 mL Teflon-lined stainless steel container and heated at 150 °C for 72 h. After slowly cooling to room temperature, blue block crystals of compound 5 were isolated and washed with distilled water. Yield: 33% (based on the H2cppca). Elemental anal. calcd C26H16CuN2O8 (547.95): C, 56.94; H, 2.92; N, 5.11. Found: C, 56.95; H, 2.90; N, 5.09. IR data (KBr, cm−1): 3438(m), 1684(s), 1612(s), 1377(m), 1249(m), 759(m).
4, 8 mL) solutions, and stirred for 0.5 h, then the mixture was transferred into a 20 mL Teflon-lined stainless steel container and heated at 150 °C for 72 h. After slowly cooling to room temperature, blue block crystals of compound 5 were isolated and washed with distilled water. Yield: 33% (based on the H2cppca). Elemental anal. calcd C26H16CuN2O8 (547.95): C, 56.94; H, 2.92; N, 5.11. Found: C, 56.95; H, 2.90; N, 5.09. IR data (KBr, cm−1): 3438(m), 1684(s), 1612(s), 1377(m), 1249(m), 759(m).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 8 mL) was stirred for 0.5 h to form a suspension where NaOH (0.1 M) was added to adjust pH = 6.0. Then the suspension was sealed in a 20 mL Teflon-lined stainless steel container and heated at 120 °C for 72 h. After slowly cooling to room temperature, light-yellow rod crystals of compound 6 were isolated after washed with water several times. Yield: 35% (based on the H2cppca). Elemental anal. calcd C104H80N8O44Mn7 (2530.34): C, 49.32; H, 3.16; N, 4.43. Found: C, 46.35; H, 3.12; N, 4.45. IR data (KBr, cm−1): 3438(m), 1626(m), 1556(s), 1385(s), 1191(m), 1028(m), 764(m).
4, 8 mL) was stirred for 0.5 h to form a suspension where NaOH (0.1 M) was added to adjust pH = 6.0. Then the suspension was sealed in a 20 mL Teflon-lined stainless steel container and heated at 120 °C for 72 h. After slowly cooling to room temperature, light-yellow rod crystals of compound 6 were isolated after washed with water several times. Yield: 35% (based on the H2cppca). Elemental anal. calcd C104H80N8O44Mn7 (2530.34): C, 49.32; H, 3.16; N, 4.43. Found: C, 46.35; H, 3.12; N, 4.45. IR data (KBr, cm−1): 3438(m), 1626(m), 1556(s), 1385(s), 1191(m), 1028(m), 764(m).Single crystal structure determination
The crystal structures were determined by single-crystal X-ray diffraction. Reflection data were collected on a Bruker SMARTCCD area-detector diffractometer (Mo-Kα radiation, graphite monochromator) at room temperature with ω-scan mode. Empirical adsorption correction was applied to all data using SADABS. The structure was solved by direct methods and refined by full-matrix least squares on F2 using SHELXTL 97 software.13 Non-hydrogen atoms were refined anisotropically. All C-bound H atoms were refined using a riding model. All calculations were carried out using SHELXTL 97. The crystallographic data and pertinent information for compounds 1–6 are contained in Table 1; selected bond lengths and angles are listed in Table S1.†| a R1 = Σ||Fo| − |Fc||/Σ|Fo|; wR2 = [Σw(Fo2 − Fc2)2/Σw(Fo2)2]1/2. | ||||||
|---|---|---|---|---|---|---|
| Compound | 1 | 2 | 3 | 4 | 5 | 6 |
| Empirical formula | C13H19NNiO10 | C13H19CoNO10 | C26H24Cd2N2O13 | C26H32Cd3N2O23P2 | C26H16CuN2O8 | C104H82Mn7N8O44 |
| Formula weight | 408.00 | 408.22 | 797.27 | 1131.70 | 547.95 | 2532.36 |
| Crystal system | Monoclinic | Monoclinic | Triclinic | Monoclinic | Monoclinic | Triclinic |
| Space group | P21/c | P21/c | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
C2/c | P21/c | P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| a/Å | 15.569(3) | 15.5726(4) | 7.7894(16) | 35.360(7) | 8.3091(6) | 13.136(3) |
| b/Å | 7.7545(16) | 7.7978(2) | 12.085(2) | 11.899(2) | 7.1846(6) | 14.882(3) |
| c/Å | 13.712(3) | 13.7900(5) | 14.169(3) | 7.9016(16) | 19.0036(14) | 15.283(3) |
| α/° | 90.00 | 90.00 | 82.94(3) | 90.00 | 90.00 | 116.26(3) |
| β/° | 99.11(3) | 98.943(3) | 81.63(3) | 90.88(3) | 98.168(7)° | 93.42(3) |
| γ/° | 90.00 | 90.00 | 89.49(3) | 90.00 | 90.00 | 108.77(3) |
| V/(Å3) | 1634.5(6) | 1654.19(8) | 1309.5(5) | 3324.2(12) | 1122.96(15) | 2464.2(9) |
| Z | 4 | 4 | 2 | 4 | 2 | 1 |
| ρcalc/g cm−3 | 1.658 | 1.639 | 2.022 | 2.261 | 1.621 | 1.706 |
| Absorption coef. | 1.242 | 1.092 | 1.701 | 2.097 | 1.031 | 0.973 |
| Reflns collected | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 359 359 |
7682 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 699 699 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 602 602 |
3876 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 497 497 |
| Unique reflns (Rint) | 3728(0.0288) | 2955(0.0335) | 5922(0.0208) | 3788(0.0423) | 1995(0.0203) | 8673(0.0317) |
| Completeness | 99.7% | 99.9% | 98.6% | 99.0% | 99.8% | 98.6% |
| GooF | 1.227 | 1.099 | 1.179 | 1.075 | 1.095 | 1.066 |
| R1, wR2 [I > 2σ(I)]a | 0.0330, 0.0854 | 0.0339, 0.0727 | 0.0250, 0.0675 | 0.0629, 0.1801 | 0.0370, 0.0821 | 0.0402, 0.0980 |
| R1, wR2 (all data) | 0.0400, 0.0954 | 0.0428, 0.0776 | 0.0322, 0.0819 | 0.0736, 0.1867 | 0.0484, 0.0902 | 0.0550, 0.1030 |
Results and discussion
Synthesis of compounds 1–6
Due to the rigid structure of the H2cppca ligand, the mixture of metal salt and organic ligand in different solvents usually produced lots of precipitations at room temperature, as makes the crystal complexes difficultly to be obtained, so compounds 1–6 were synthesized under the solvothermal conditions. DMF or acetonitrile is helpful to solve the H2cppca ligand, as provides a facility for the solvothermal reactions. The experimental results indicate that the reaction solvents and temperature play an important role in the preparation of compounds 1–6. Compounds 1–3 were obtained with high yield by the solvothermal reaction of metal ions and H2cppca in a DMF/H2O solution, however, DMF was substituted by the other organic solvents such as methanol, ethanol, acetonitrile, etc., crystalline compounds 1–3 were difficult to be synthesized. Compound 4 has been obtained through adding pyrophosphoric acid methanol solution to DMF/H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) solutions, methanol was crucial for the synthesis of compound 4, small crystals of compounds 4 were obtained if methanol was absent in the reaction. Notably, very small crystals of compounds 5 and 6 were obtained when the reaction solvent is DMF/H2O solutions, while DMF was substituted by CH3CN and further increased the reaction temperature, compounds 5 and 6 with larger crystals were obtained successfully, because high temperature and the pressure during the reaction process are helpful to improve the ligand solubility and enhance the reactivity of reactant.
3) solutions, methanol was crucial for the synthesis of compound 4, small crystals of compounds 4 were obtained if methanol was absent in the reaction. Notably, very small crystals of compounds 5 and 6 were obtained when the reaction solvent is DMF/H2O solutions, while DMF was substituted by CH3CN and further increased the reaction temperature, compounds 5 and 6 with larger crystals were obtained successfully, because high temperature and the pressure during the reaction process are helpful to improve the ligand solubility and enhance the reactivity of reactant.
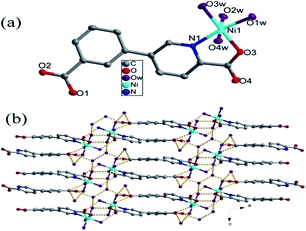 | ||
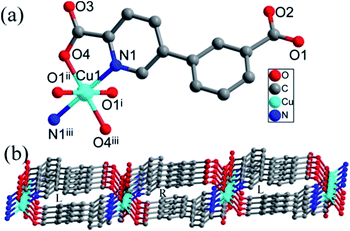
| Fig. 1 (a) Coordination environment of the Ni(II) ion, the hydrogen atoms are omitted for clarity. (b) The 3D supramolecular network in compound 1. | ||
Two Ni(H2O)4 units constructed by the Ni center and four coordinated water are interconnected into a Ni2(H2O)8 dimer with six-membered ring through O1w–H⋯O4w hydrogen bonds (Fig. S1†). In Ni2(H2O)8 dimer, coordinated O1w and O4w interact with lattice water molecules (O5w and O6w) via hydrogen bonds, respectively; there are two hydrogen bonds between coordinated O2w and lattice water molecules (O5w and O6w). The interactions between Ni2(H2O)8 dimers and lattice water molecules (O5w and O6w) result in the formation of a rather complex 2D supramolecular layer via hydrogen bonds, in which O5w and O6w bridge two dimmers (Fig. S2†). The 2D supramolecular layers were joined into a 3D supramolecular network through O–H⋯O hydrogen bonds between uncoordinated carboxyl group of cppca2− anions and water molecules (Fig. 1b).
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . The asymmetric unit of 3 contains two Cd2+ ions, two cppca2− anions, and five coordination water molecules (Fig. 3a). The Cd1 center with a slightly distorted pentagonal bipyramid geometry where the equatorial plane comprises four carboxylate O atoms (O2, O5, O3ii and O4ii) and one N atom (N1) from three different cppca2− anions, and two water molecules (O1w and O2w) occupy the apical sites. Similar with Cd1, Cd2 is bound to six oxygens and one nitrogen from two independent cppca2− anions and three water molecules. The Cd–N/O bond lengths are 2.275(3) and 2.434(3) Å, and the O/N–Cd–O/N bond angles range from 53.77(9) to 172.25(13)°.
. The asymmetric unit of 3 contains two Cd2+ ions, two cppca2− anions, and five coordination water molecules (Fig. 3a). The Cd1 center with a slightly distorted pentagonal bipyramid geometry where the equatorial plane comprises four carboxylate O atoms (O2, O5, O3ii and O4ii) and one N atom (N1) from three different cppca2− anions, and two water molecules (O1w and O2w) occupy the apical sites. Similar with Cd1, Cd2 is bound to six oxygens and one nitrogen from two independent cppca2− anions and three water molecules. The Cd–N/O bond lengths are 2.275(3) and 2.434(3) Å, and the O/N–Cd–O/N bond angles range from 53.77(9) to 172.25(13)°.
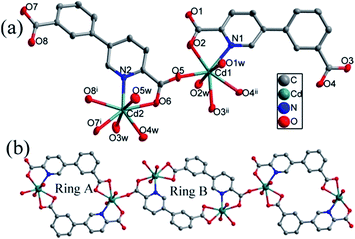 | ||
| Fig. 3 (a) Coordination environment of the Cd(II) ions. The hydrogen atoms are omitted for clarity. (b) The 1D chain constructed by Ring A and Ring B in compound 3. | ||
In compound 3, two different coordination modes of cppca2− anions are observed in the completely deprotonated cppca2− ligands: μ2-κN,O:κO′,O′′ (Fig. 2c) and μ3-κO:κN,O:κO′,O′′ (Fig. 2f). Two symmetry-related Cd1 metal centers are connected into a 18-membered ring by two cppca2− ligands adopting μ2 coordination modes (abbreviated to Ring A). Interestingly, another 18-membered ring is composed of two Cd2 centers and two cppca2− ligands adopting μ3 coordination modes (abbreviated to Ring B). Each Ring B joins two Ring A into an extended 1D chain via Cd–O bonds (Fig. 3b).
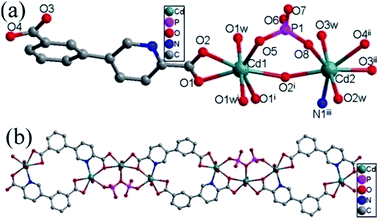 | ||
| Fig. 4 (a) Coordination environment of the Cd(II) ions. The hydrogen atoms are omitted for clarity. (b) The 1D chain in compound 4. | ||
The cppca2− adopting μ3-κN,O:κO′,O′′:κO,O′′′ coordination mode (Fig. 2g) and [H2P2O7]2− anions each connect two Cd1 and one Cd2 centers into a bent Cd3 secondary building units (SBUs). The Cd3 SBUs are further joined into a 1D chain by four cppca2− anions, in which there exist in an 18-membered ring constructed by two cppca2− ligands and two Cd2 centers (Fig. 4b).
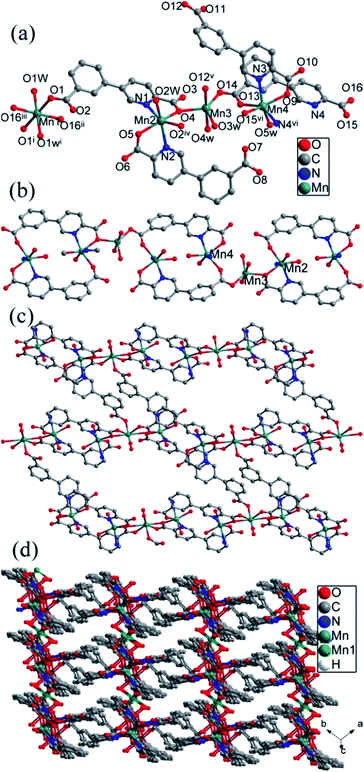
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . The asymmetric unit of compound 6 contains four crystallographically independent manganese(II) ion (Mn1, Mn2, Mn3 and Mn4), three cppca2− anions, one (Hcppca)− anion, five coordinated and one lattice water molecules (Fig. 6a). Mn1 is six-coordinated with six oxygens from four cppca2− anions and two water molecules, forming a distorted octahedral geometry. Mn2 with octahedral geometry is bound to three carboxylate oxygens and two nitrogens from two independent cppca2− and one (Hcppca)− anions and one oxygen from a water molecule, similar with Mn2, Mn4 also coordinates with four oxygens and two nitrogens from three independent cppca2− anions and one water molecule. While Mn3 exhibits a slightly distorted trigonal bipyramidal geometry, which is bound to five oxygens from three different cppca2− anions and two coordination water molecule. The Mn–O/N bond distances around Mn centers are in the range of 2.119(2)–2.284(3) Å, and the O/N–Mn–O/N bond angles range from 72.64(8) to 180.00(1)°.
. The asymmetric unit of compound 6 contains four crystallographically independent manganese(II) ion (Mn1, Mn2, Mn3 and Mn4), three cppca2− anions, one (Hcppca)− anion, five coordinated and one lattice water molecules (Fig. 6a). Mn1 is six-coordinated with six oxygens from four cppca2− anions and two water molecules, forming a distorted octahedral geometry. Mn2 with octahedral geometry is bound to three carboxylate oxygens and two nitrogens from two independent cppca2− and one (Hcppca)− anions and one oxygen from a water molecule, similar with Mn2, Mn4 also coordinates with four oxygens and two nitrogens from three independent cppca2− anions and one water molecule. While Mn3 exhibits a slightly distorted trigonal bipyramidal geometry, which is bound to five oxygens from three different cppca2− anions and two coordination water molecule. The Mn–O/N bond distances around Mn centers are in the range of 2.119(2)–2.284(3) Å, and the O/N–Mn–O/N bond angles range from 72.64(8) to 180.00(1)°.
Interestingly, there exist in four types of coordination modes for organic linker: μ1-κN,O; μ2-κN,O:κO′; μ4-κN,O:κO,κO′:κO′′ and μ4-κN,O:κO′:κO′′:κO′′′ fashions (Fig. 2b, e, h, i). Mn2, Mn3 and Mn4 are assembled into a linear Mn3 secondary building unit (SBU) by carboxyl groups from cppca2− anions. The Mn3 SBUs are joined into a 1D chain by cppca2− anions adopting μ4-κN,O:κO,κO′:κO′′ coordination mode, in which an 18-membered ring constructed by two cppca2− ligands and two Mn centers (Fig. 6b). Then cppca2− ligands adopting μ2-κN,O:κO′ modes bridge Mn3 and Mn4 centers of the adjacent chains into a 2D corrugated layer with an irregular ring in which the two Hcppca− ligands with μ1-κN,O coordination mode are filled and coordinated with Mn2 centers as the terminal chelators (Fig. 6c). The cppca2− anions with μ4-κN,O:κO′:κO′′:κO′′′ modes in 2D corrugated layers joined Mn1 ions to generate a 3D network (Fig. 6d). From the perspective of network topology, cppca2− anions interacting with two Mn ions can be regarded as a line; cppca2− anion with μ4 coordination modes interacting with two Mn3 SBUs and Mn1 center is regarded as a 3-connected node, hexa-coordinated Mn1 center interacting four cppca2− ligands is also a 4-connected node; linear Mn3 SBU is considered as a 5-connected node. So the framework of compound 6 may be simplified into a 3-nodal net with Schlafli symbol {42·6}{42·82·102}{43·62·8·104}2, which shows an unprecedented3–5-connected topology net determined by TOPOS (Fig. 7).
Characterization
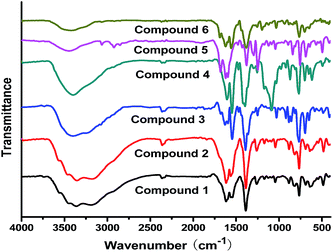
The experimental and simulated PXRD patterns of compounds 1–6 are shown in Fig. S3.† The simulated PXRD patterns from the single-crystal X-ray diffraction data are in agreement with the observed ones, indicating the phase purity of these synthesized crystalline products. The different intensities between the simulated and experimental patterns may be caused by the preferred orientation of the powder samples.The FT-IR spectra of compounds 1–6 are displayed in Fig. 8. The wide peaks at 3445 cm−1 in compounds 1–6 belong to the O–H stretching. The absorption peaks located at 1605 and 1392 cm−1 can be ascribed to asymmetric and symmetric stretching vibrations of carboxylate groups, respectively. The peaks in the range of 1257–1032 cm−1 can be ascribed to the in-plane bending vibration of C–H in the benzene ring, while the out-plane bending stretching vibrations appear in the range of 762 to 694 cm−1. Notably, the peak at 1058 cm−1 for compound 4 corresponds to the stretching vibration of P–O bonds due to the present of [H2P2O7]2− anions.
TG curves of compounds 1–6
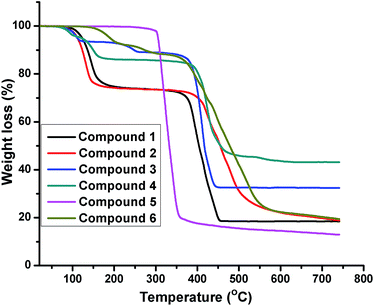
The thermal behaviors of compounds 1–6 were studied from 14 °C to 750 °C under air (Fig. 9). The thermogravimetric analysis (TGA) curves of compounds 1–4 and 6 have similar thermal behavior with two-step weight loss. The first weight loss corresponded to the weight loss of water molecules (obsd 26.32% for 1, 26.38% for 2, 10.98% for 3, 13.31% for 4; 8.25% for 6; calcd 26.49% for 1, 26.45% for 2, 11.29% for 3, 12.64% for 4, 8.43% for 6). The second weight loss of 1–4 and 6 were characteristic of the combustion of organic ligands (obsd 55.11% for 1, 54.94% for 2, 56.46% for 3, 43.43% for 4, 72.05% for 6; calcd 55.20% for 1, 55.16% for 2, 56.50% for 3, 41.06% for 4, 71.94% for 6). The total weight loss of compounds 1–4 and 6 amounted to 81.61%, 81.56%, 67.44%, 56.74%, 80.30%, respectively, and the remaining weight correspond to the formation of NiO, CoO, CdO, Cd3(PO4)2, MnO (obsd 18.57% for 1, 18.68% for 2, 32.56% for 3; 43.26% for 4, 19.70% for 6; calcd 18.31% for 1, 18.36% for 2, 32.21% for 3, 46.30% for 4, 19.63% for 6).The framework of compounds 5 is thermally stable up to 300 °C, on further heating, the (Hcppca)− ligand begins to rapidly decompose, the total weight loss of compounds 5 amounted to 84.95%, and the remaining weight is ascribed to the formation of CuO (obsd 15.05%, calcd 14.59%).
Luminescence property
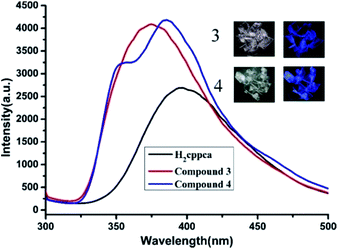
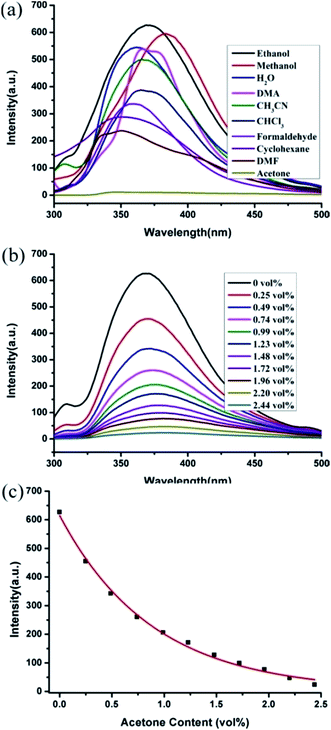
Under UV illumination, H2cppca ligand can't give off light, however, compounds 3 and 4 exhibit strong blue light emission. The solid-state photoluminescence behavior of H2cppca, compounds 3 and 4 were measured at room temperature (Fig. 10). They all exhibit strong blue light emission when excited at 280 nm. The free H2cppca and compound 3 exhibit emission bands at 396 nm and 375 nm, respectively, while a blue emission band at 385 nm with a shoulder peak at 355 nm is observed in compound 4. Compared with H2cppca, the emission maximum of compounds 3 and 4 are slightly blue-shifted by 21 and 11 nm, respectively. So the emission bands of compounds 3 and 4 may be attributed to synergistic effects between intraligand charge transfer (LLCT) and ligand-to-metal transition (LMCT).14 Notably, the fluorescence intensities of compounds 3 and 4 are apparently stronger than that of H2cppca ligand under the same experimental conditions, which may be attributed to the unique coordination of ligand cppca2− to the Cd(II) centers increasing the conformational rigidity, thereby reducing the non-radiative energy loss.15The intense blue light emission of compounds 3 and 4 encouraged us to use them as hosts for the sensing of small molecules. The ground powders of compound 3 or 4 (0.5 mg) were immersed in water and common organic solvents 4.00 mL, methanol (MeOH), ethanol (EtOH), acetonitrile (CH3CN), N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMA), formaldehyde, cyclohexane, trichloromethane (CHCl3) and acetone, then treated by ultrasonication for 0.5 hours, and the corresponding PL spectra were measured as shown in Fig. 11a. The emission spectra of compounds 3 and 4 (λex = 280 nm) dispersed in different solvents revealed that the corresponding maximum emission peaks are largely dependent on the solvent molecules. For 3, the emission peak with slightly shifted compared with its solid state emission spectrum (λem = 375 nm), while the maximum emission peaks for 4 have a subtle blue-shifted by 9 nm compared with its solid state emission spectrum (λem = 385 nm) (Fig. S4†). This phenomenon may be ascribed to the interactions between homogeneously dispersible framework and solvent molecules with different polarities.16
Notably, the fluorescent intensities of compounds 3 and 4 vary with the solvent molecules. The maximum luminescence intensity of 3-solvent emulsions gradually decreased in the order EtOH > MeOH > H2O > DMA > CH3CN > CHCl3 > formaldehyde > cyclohexane > DMF > acetone for 3; while for 4, the order is EtOH > DMA > CH3CN > DMF > cyclohexane > CHCl3 > MeOH > H2O > formaldehyde > acetone. Obviously, ethanol exhibits the most significant enhancing effect of fluorescence, while acetone exerted the significant quenching effect.
In addition, the sensing ability toward acetone for compounds 3 and 4 were further investigated. 0.5 mg samples of compounds 3 or 4 dispersed in ethanol were considered as the standard emulsion, and a batch of emulsions with gradually increased analytes concentration were prepared and monitored through fluorescent spectra. As seen in Fig. 11b, the fluorescence intensity of 3-ethanol emulsion has a significant decrease with the increasing concentration of acetone. The fluorescence quenching percentages (QP) can be calculated using equation:17
| QP = (I0 − I)/I0 × 100% |
In order to investigate the mechanism of quenching behavior by acetone, the UV-Vis spectra of acetone and the other solvents were measured (Fig. S5†). The results revealed that only acetone has a strong absorption ranging from 270 to 330 nm, while other solvents have no significant absorption in this range. Notably, the excitation wavelength of the compound 3 and 4 is 280 nm, which is completely overlaid by the absorbing band of acetone. Upon excitation at 280 nm, there is competition for the absorption of the light source energy between as-synthesized 3 and 4 and acetone molecules. So the efficient quenching of acetone in this system might be ascribed to the physical interaction of the solute and the solvent, which induces the changes in energy transfer.18b,19
Magnetic properties of compounds 1, 2, 5 and 6
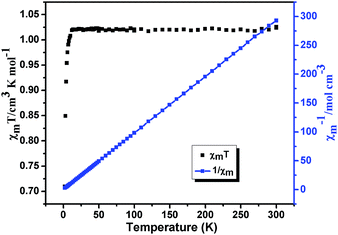
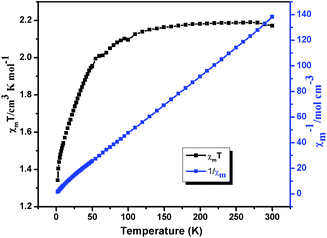
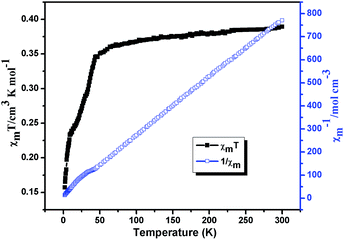
Variable-temperature magnetic susceptibilities for compounds 1, 2, 5 and 6 were measured in the temperature range of 2–300 K under a 1000 Oe applied field. For compound 1, the χmT value is 1.03 cm3 mol−1 K at T = 300 K (Fig. 12), which is close to the spin-only value of 1.00 cm3 K mol−1 for Ni(II) (S = 1 and g = 2.0). The χmT of 1 slightly decreases with decreasing temperature and sharply reach a minimum (0.71 cm3 mol−1 K) at 2 K. In χm−1 versus T curve, the magnetic susceptibilities data follow the Curie–Weiss law (χm = C/(T − θ)) above 20 K with Curie constant C = 1.05 cm3 K mol−1 and the Weiss constant θ = −0.15 K. According to the formulas C = 0.125g2∑S(S + 1), the g value is 2.1 for 1. For 2, the χmT value is 2.16 cm3 mol−1 K at T = 300 K (Fig. 13), which is higher than the spin-only 1.875 cm3 K mol−1 for CoII ion (CoII, S = 3/2 and g = 2.0). The χmT continuously decreases with decreasing temperature and reach a minimum (1.34 cm3 mol−1 K) at 2 K. In χm−1 versus T curve, the magnetic susceptibilities data follow the Curie–Weiss law (χm = C/(T − θ)) above 30 K with Curie constant C = 2.27 cm3 K mol−1 and the Weiss constant θ = −5.80 K. According to the formulas C = 0.125g2∑S(S + 1), the g value is 2.18 for 2. The negative value of θ and the decrease of χmT indicate weak antiferromagnetic interactions in compounds 1 and 2.20For compound 5 (Fig. 14), the χmT value is 0.389 cm3 mol−1 K at T = 300 K, which is higher than the spin-only value of 0.375 cm3 K mol−1 for Cu(II) (S = 1/2 and g = 2.0). Upon cooling, the χmT continuously decreases with decreasing temperature and reach a minimum (0.79 cm3 mol−1 K) at 2 K. In χm−1 versus T curve, the magnetic susceptibilities data follow the Curie–Weiss law (χm = C/(T − θ)) above 50 K with Curie constant C = 1.98 cm3 K mol−1 and the Weiss constant θ = −8.11 K. According to the formulas C = 0.125g2∑S(S + 1), the g value is 2.14 for 5. The negative value of θ and the decrease of χmT indicate weak antiferromagnetic interactions between Cu(II) centers in compounds 5.21 The field dependence of the magnetization for compound 5 at 2 K in the range of −70–70 kOe shows no hysteresis loop (Fig. S6†), which indicates no 3D magnetic ordering behavior exists even at 2 K. The experimental magnetization shows an abrupt increase to 0.89 Nβ before 40 kOe and further steadily increases to 1.03 Nβ at 70 kOe, which is close to the value for one uncoupled CuII ion. This behavior further consist well with the result obtained from χmT vs. T plot, indicating that there are very weak antiferromagnetic interactions between the Cu2+ ions in compound 5.
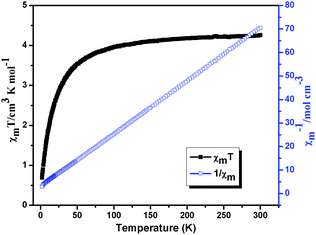
The χmT and χm−1 vs. T plots for 6 are shown in Fig. 15, the χmT value of Mn(II) ion at 300 K is 4.26 cm3 K mol−1, which is close to the expected value (4.38 cm3 K mol−1) for magnetically non-interacting Mn(II) ions with S = 5/2 and g = 2.0. Upon cooling, χmT decreases smoothly to a minimum of 0.69 cm3 K mol−1 at 2 K, and the decrease of χmT indicates a dominant antiferromagnetic interactions among the Mn(II) centers. In χm−1 versus T curve, the magnetic behavior of compound 6 obeys the Curie–Weiss law (χm = C/(T − θ)) in the temperature range of 2–300 K, and the corresponding Curie and Weiss constants are 4.45 cm3 K mol−1 and −13.43 K (θ), respectively. According to the formulas C = 0.125g2∑S(S + 1), the g value is 2.02. The large negative θ value further indicates significant antiferromagnetic interactions exist between neighboring Mn(II) ions.20,22
Conclusion
In summary, six new coordination compounds based on H2cppca ligand have been synthesized and characterized in detail. Compounds 1–6 have rich structural chemistry ranging from mononuclear (1 and 2), one-dimensional (3 and 4), two-dimensional (5) to three-dimensional (6) structures. Moreover, the framework of compound 6 may be simplified into a 3-nodal net with Schlafli symbol {42·6}{42·82·102}{43·62·8·104}2, which shows an unprecedented3–5-connected topology net. Compounds 3 and 4 show efficiently and easily recycled fluorescent probes for the acetone molecules. Weak antiferromagnetic interactions were observed in compounds 1, 2, 5 and 6. The further research for the construction of new coordination compounds based on H2cppca ligand is underway in our laboratory.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21201155), the Natural Science Young Scholars Foundation of Shanxi Province (No. 2012021007-5 and 2013021008-6), Program for the Top Young Academic Leaders of Higher Learning Institutions of Shanxi and 131 Talent Plan of Higher Learning Institutions of Shanxi, respectively.Notes and references
- (a) S. R. Batten and R. Robson, Angew. Chem., Int. Ed., 1998, 37, 1460 CrossRef; (b) L. Carlucci, G. Ciani and D. M. Proserpio, Coord. Chem. Rev., 2003, 246, 247 CrossRef CAS; (c) A. Harada, A. Hashidzume, H. Yamaguchi and Y. Takashima, Chem. Rev., 2009, 109, 5974 CrossRef CAS PubMed; (d) G. E. Kostakis, A. M. Ako and A. K. Powell, Chem. Soc. Rev., 2010, 39, 2238 RSC; (e) R. Chakrabarty, P. S. Mukherjee and P. J. Stang, Chem. Rev., 2011, 111, 6810 CrossRef CAS PubMed; (f) D. Zhao, D. J. Timmons, D. Q. Yuan and H. C. Zhou, Acc. Chem. Res., 2011, 44, 123 CrossRef CAS PubMed; (g) J. Yang, J. F. Ma and S. R. Batten, Chem. Commun., 2012, 48, 7899 RSC; (h) L. Carlucci, G. Ciani, D. M. Proserpio, T. G. Mitina and V. A. Blatov, Chem. Rev., 2014, 114, 7557 CrossRef CAS PubMed.
- (a) A. M. Seayad and D. M. Antonelli, Adv. Mater., 2004, 16, 765 CrossRef CAS; (b) J. L. C. Rowsell and O. M. Yaghi, Angew. Chem., Int. Ed., 2005, 44, 4670 CrossRef CAS PubMed; (c) V. V. Struzhkin, B. Militzer, W. L. Mao, H.-k. Mao and R. J. Hemley, Chem. Rev., 2007, 107, 4133 CrossRef CAS PubMed; (d) J. R. Li, R. J. Kuppler and H. C. Zhou, Chem. Soc. Rev., 2009, 38, 1477 RSC; (e) A. Phan, C. J. Doonan, F. J. Uribe-Romo, C. B. Knobler, M. O'Keeffe and O. M. Yaghi, Acc. Chem. Res., 2010, 43, 58 CrossRef CAS PubMed; (f) G. Ferey, C. Serre, T. Devic, G. Maurin, H. Jobic, P. L. Llewellyn, G. De Weireld, A. Vimont, M. Daturi and J. S. Chang, Chem. Soc. Rev., 2011, 40, 550 RSC; (g) J. Canivet, A. Fateeva, Y. Guo, B. Coasne and D. Farrusseng, Chem. Soc. Rev., 2014, 43, 5594 RSC; (h) B. Van de Voorde, B. Bueken, J. Denayer and D. De Vos, Chem. Soc. Rev., 2014, 43, 5766 RSC; (i) Q. Wang, J. Bai, Z. Lu, Y. Pan and X. You, Chem. Commun., 2016, 52, 443 RSC.
- (a) L. E. Kreno, K. Leong, O. K. Farha, M. Allendorf, R. P. V. Duyne and J. T. Hupp, Chem. Rev., 2012, 112, 1105 CrossRef CAS PubMed; (b) X. Sun, Y. Wang and Y. Lei, Chem. Soc. Rev., 2015, 44, 8019 RSC; (c) J. Wu, B. Kwon, W. Liu, E. V. Anslyn, P. Wang and J. S. Kim, Chem. Rev., 2015, 115, 7893 CrossRef CAS PubMed; (d) L. You, D. Zha and E. V. Anslyn, Chem. Rev., 2015, 115, 7840 CrossRef CAS PubMed; (e) X. Zhou, S. Lee, Z. Xu and J. Yoon, Chem. Rev., 2015, 115, 7944 CrossRef CAS PubMed.
- (a) M. Kurmoo, Chem. Soc. Rev., 2009, 38, 1353 RSC; (b) M. Murrie, Chem. Soc. Rev., 2010, 39, 1986 RSC; (c) G. Rogez, N. Viart and M. Drillon, Angew. Chem., Int. Ed., 2010, 49, 1921 CrossRef CAS PubMed; (d) M. Clemente-Leon, E. Coronado, C. Marti-Gastaldo and F. M. Romero, Chem. Soc. Rev., 2011, 40, 473 RSC; (e) X. Y. Wang, C. Avendano and K. R. Dunbar, Chem. Soc. Rev., 2011, 40, 3213 RSC; (f) D. F. Weng, Z. M. Wang and S. Gao, Chem. Soc. Rev., 2011, 40, 3157 RSC; (g) J. Yuan, Y. Xu and A. H. Muller, Chem. Soc. Rev., 2011, 40, 640 RSC; (h) J. Tao, R. J. Wei, R. B. Huang and L. S. Zheng, Chem. Soc. Rev., 2012, 41, 703 RSC.
- (a) J. Lee, O. K. Farha, J. Roberts, K. A. Scheidt, S. T. Nguyen and J. T. Hupp, Chem. Soc. Rev., 2009, 38, 1450 RSC; (b) A. Corma, H. Garcıa and F. X. LlabresiXamena, Chem. Rev., 2010, 110, 4606 CrossRef CAS PubMed; (c) A. Dhakshinamoorthy and H. Garcia, Chem. Soc. Rev., 2014, 43, 5750 RSC; (d) J. W. Liu, L. F. Chen, H. Cui, J. Y. Zhang, L. Zhang and C. Y. Su, Chem. Soc. Rev., 2014, 43, 6011 RSC; (e) A. Dhakshinamoorthy, A. M. Asiri and H. Garcia, Chem. Soc. Rev., 2015, 44, 1922 RSC.
- (a) N. W. Ockwig, O. Delgado-Friedrichs, M. O'Keeffe and O. M. Yaghi, Acc. Chem. Res., 2005, 38, 176 CrossRef CAS PubMed; (b) A. K. Cheetham, C. N. R. Rao and R. K. Feller, Chem. Commun., 2006, 46, 4780 RSC; (c) I. G. Georgiev and L. R. MacGillivray, Chem. Soc. Rev., 2007, 36, 1239 RSC; (d) O. K. Farha and J. T. Hupp, Acc. Chem. Res., 2010, 43, 1166 CrossRef CAS PubMed; (e) N. Stock and S. Biswas, Chem. Rev., 2012, 112, 933 CrossRef CAS PubMed; (f) M. O'Keeffe and O. M. Yaghi, Chem. Rev., 2012, 112, 675 CrossRef PubMed; (g) N. Stock and S. Biswas, Chem. Rev., 2012, 112, 933 CrossRef CAS PubMed; (h) C. Wang, T. Zhang and W. Lin, Chem. Rev., 2012, 112, 1084 CrossRef CAS PubMed; (i) Y. Han, J. R. Li, Y. Xie and G. Guo, Chem. Soc. Rev., 2014, 43, 5952 RSC; (j) Z. Zhang and M. J. Zaworotko, Chem. Soc. Rev., 2014, 43, 5444 RSCY. X. Sun and W. Y. Sun, Chin. Chem. Lett., 2014, 25, 823 CrossRef CAS.
- (a) F. A. Paz, J. Klinowski, S. M. Vilela, J. P. Tome, J. A. Cavaleiro and J. Rocha, Chem. Soc. Rev., 2012, 41, 1088 RSC; (b) C. Zhang, M. Zhang, L. Qin and H. Zheng, Cryst. Growth Des., 2014, 14, 491 CrossRef CAS; (c) D. Zhao, D. J. Timmons, D. Yuan and H. C. Zhou, Acc. Chem. Res., 2010, 44, 123 CrossRef PubMed; (d) X. L. Zhao and W. Y. Sun, CrystEngComm, 2014, 16, 3247 RSC.
- (a) Y. X. Sunand and W. Y. Sun, CrystEngComm, 2015, 17, 4045 RSC; (b) L. Sun, J. F. Song, R. S. Zhou, J. Zhang, L. Wang, K. L. Cui and X. Y. Xu, J. Coord. Chem., 2014, 67, 822 CAS; (c) J. F. Song, Y. Li, R. S. Zhou, J. Shao, L. Wang and X. B. Cui, J. Coord. Chem., 2015, 68, 3651 CrossRef CAS; (d) B. B. Shi, Y. H. Zhong, L. L. Guo and G. Li, Dalton Trans., 2015, 44, 4362 RSC.
- (a) W. X. Yin, Y. T. Liu, Y. J. Ding, Q. Lin, X. M. Lin, C. L. Wu, X. D. Yao and Y. P. Cai, CrystEngComm, 2015, 17, 3619 RSC; (b) Q. F. Li, D. Yue, G. W. Ge, X. D. Du, Y. C. Gong, Z. L. Wang and J. H. Hao, Dalton Trans., 2015, 44, 16810 RSC; (c) J. J. Hou, X. Q. Li, B. Q. Tian and X. M. Zhang, CrystEngComm, 2016, 18, 2065 RSC; (d) D. S. Deng, H. Guo, G. H. Kang, L. F. Ma, X. He and B. M. Ji, CrystEngComm, 2015, 17, 1871 RSC.
- (a) B. M. Ji, D. S. Deng, L. F. Ma, H. L. Li, X. S. Fan, G. R. Qu, L. Cao and L. Zhou, CrystEngComm, 2013, 15, 4107 RSC; (b) J. Cepeda, S. Pérez-Yáñez, G. Beobide, O. Castillo, A. Luque, P. A. Wright, S. Sneddon and S. E. Ashbrook, Cryst. Growth Des., 2015, 15, 2352 CrossRef CAS; (c) R. G. AbdulHalim, A. Shkurenko, M. H. Alkordi and M. Eddaoudi, Cryst. Growth Des., 2016, 16, 722 CrossRef CAS.
- (a) B. M. Ji, D. S. Deng, X. He, B. Liu, S. B. Miao, N. Ma, W. Z. Wang, L. G. Ji, P. Liu and X. F. Li, Inorg. Chem., 2012, 51, 2170 CrossRef CAS PubMed; (b) L. B. Hamdy, P. R. Raithby, L. H. Thomas and C. C. Wilson, New J. Chem., 2014, 38, 2135 RSC.
- (a) J. Xiao, B. Y. Liu, G. Wei and X. C. Huang, Inorg. Chem., 2011, 50, 11032 CrossRef CAS PubMed; (b) S. W. Zhang, J. G. Ma, X. P. Zhang, E. Y. Duan and P. Cheng, Inorg. Chem., 2015, 54, 586 CrossRef CAS PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112 CrossRef CAS PubMed.
- (a) F. Y. Yi, Y. Wang, J. P. Li, D. Wu, Y. Q. Lan and Z. M. Sun, Mater. Horiz., 2015, 2, 245 RSC; (b) F. Y. Yi, J. P. Li, D. Wu and Z. M. Sun, Chem.–Eur. J., 2015, 21, 11475 CrossRef CAS PubMed; (c) Y. L. Wu, G. P. Yang, Y. Q. Zhao, W. P. Wu, B. Liu and Y. Y. Wang, Dalton Trans., 2015, 44, 3271 RSC; (d) W. H. Huang, J. Z. Li, T. Liu, L. S. Gao, M. Jiang, Y. N. Zhang and Y. Y. Wang, RSC Adv., 2015, 5, 97127 RSC.
- (a) Y. Y. Wang, Q. Jin, S. X. Liu, C. Guo, Y. Y. Liu, B. Ding, X. X. Wu, Y. Li and Z. Z. Zhu, RSC Adv., 2015, 5, 35238 RSC; (b) S. S. Chen, Z. S. Bai, J. Fan, G. C. Lv, Z. Su, M. S. Chen and W. Y. Sun, CrystEngComm, 2010, 12, 3091 RSC.
- (a) S. R. Zhang, D. Y. Du, J. S. Qin, S. J. Bao, S. L. Li, W. W. He, Y. Q. Lan, P. Shen and Z. M. Su, Chem.–Eur. J., 2014, 20, 3589 CrossRef CAS PubMed; (b) L. L. Wen, X. F. Wang, H. Shi, K. L. Lv and C. G. Wang, RSC Adv., 2016, 6, 1388 RSC; (c) S. Pramanik, Z. C. Hu, X. Zhang, C. Zheng, S. Kelly and J. Li, Chem.–Eur. J., 2013, 19, 15964 CrossRef CAS PubMed; (d) Y. X. Zhu, Z. W. Wei, M. Pan, H. P. Wang, J. Y. Zhang and C. Y. Su, Dalton Trans., 2016, 45, 943 RSC; (e) M. J. Zhang, H. X. Li, H. Y. Lia and J. P. Lang, Dalton Trans., 2016, 45, 17759 RSC.
- Z. M. Ju, W. Yan, X. J. Gao, Z. Z. Shi, T. Wang and H. G. Zheng, Cryst. Growth Des., 2016, 16, 2496 CAS.
- (a) F. H. Liu, C. Qin, Y. Ding, H. Wu, K. Z. Shao and Z. M. Su, Dalton Trans., 2015, 44, 1754 RSC; (b) J. A. Hua, Y. Zhao, Y. S. Kang, Y. Lu and W. Y. Sun, Dalton Trans., 2015, 44, 11524 RSC; (c) Z. Y. Guo, H. Xu, S. Q. Su, J. F. Cai, S. Dang, S. C. Xiang, G. D. Qian, H. J. Zhang, M. O'Keeffe and B. L. Chen, Chem. Commun., 2011, 47, 5551 RSC.
- (a) S. T. Zhang, J. Yang, H. Wu, Y. Y. Liu and J. F. Ma, Chem.–Eur. J., 2015, 21, 15806 CrossRef CAS PubMed; (b) Z. M. Hao, X. Z. Song, M. Zhu, X. Meng, S. N. Zhao, S. Q. Su, W. T. Yang, S. Y. Song and H. J. Zhang, J. Mater. Chem. A, 2013, 1, 11043 RSC; (c) Q. Liu, J. M. Yang, F. Guo, L. N. Jin and W. Y. Sun, Dalton Trans., 2016, 45, 5841 RSC.
- (a) R. Patra, H. M. Titi and I. Goldberg, CrystEngComm, 2013, 15, 2863 RSC; (b) R. Patra, H. M. Titi and I. Goldberg, CrystEngComm, 2013, 15, 7257 RSC.
- S. X. Cui, Y. L. Zhao, J. P. Zhang, Q. Liu and Y. Zhang, Cryst. Growth Des., 2008, 8, 3803 CAS.
- F. W. Zhang, Z. F. Li, T. Z. Ge, H. C. Yao, G. Li, H. J. Lu and Y. Y. Zhu, Inorg. Chem., 2010, 49, 3776 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Table S1, Fig. S1–S5 and X-ray crystallographic files in CIF format of compounds 1–6. CCDC 1509277–1509282. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra26966d |
| This journal is © The Royal Society of Chemistry 2017 |