Synthesis and characterization of magnetic copper–iron-titanate and uptake studies of americium from nuclear waste solutions†
Krishnan Muthukumara,
D. Shanthana Lakshmib,
Rajesh B. Gujarc,
Arvind B. Borichaa,
Prasanta K. Mohapatra*cd and
Hari C. Bajaja
aInorganic Materials & Catalysis Division, CSIR-CSMCRI, Bhavnagar, Gujarat, India. E-mail: hcbajaj@csmcri.org
bReverse Osmosis Division, CSIR-CSMCRI, Bhavnagar, Gujarat, India. E-mail: slakshmi@csmcri.org
cRadiochemistry Division, Bhabha Atomic Research Centre, Mumbai-400085, India. E-mail: mpatra@barc.gov.in
dHomi Bhabha National Institute, Anushaktinagar, Mumbai-400094, India
First published on 18th November 2016
Abstract
Magnetic materials, which find enormous applications due to their benign nature, can have their efficiency and stability enhanced by incorporation of foreign materials and controlling the synthesis conditions. Herein carbon containing iron-titanate (TF) and copper–iron-titanate (CTF) magnetic materials were prepared by a solvo-thermal method in methanol at 200 °C/20 h. Physico-chemical analysis techniques, such as BET, PXRD, FE-SEM, HR-TEM, zeta potential, XPS and NH3-TPD, were used to investigate the materials' structure and textural properties. Further, the prepared materials were used as a sorbent for the uptake of 241Am radiotracer from low pH solutions. The adsorption studies showed excellent uptake efficiency for radioactive 241Am by copper–iron-titanate (CTF), and it followed the Langmuir isotherm and pseudo-second order sorption kinetics. In addition, 1/n calculated from the Freundlich isotherm (∼0.33) indicated the favourable sorption of 152,154Eu (surrogate of 241Am) sorption by both CTF and TF. The NH3-TPD studies revealed a change in the electronic properties of the metal centre and supports the chemisorption based sorption behaviour of the CTF magnetic material. Desorption of the loaded metal ion was facile with 0.1 M HNO3 and the CTF nanomaterial can be effectively reused.
Introduction
The contribution of magnetic materials is immensely important in modern science and its widespread applications are growing day by day. Iron based ferrite and titanate materials are more promising for various applications, particularly in areas like catalysis,1 electronics,2 health care3 and waste management.4,5 Also, magnetic iron oxide and iron based ferrite and iron titanate materials are preferred because of features such as different oxidation states, environment-friendliness, abundance and tunable properties. On the other hand, the lack of stability of iron oxides and ferrites at acidic pH and the lack of magnetic properties of iron titanates6 limit their applications in metal ion sorption (actinide partitioning).Actinide separation is one of the key features of the ‘actinide partitioning’ step which is an accepted strategy for the remediation of high level radioactive wastes due to the long live half-lives of the minor actinides such as Np, Am and Cm.7 Most of the reported methods for actinide separation are based on solvent extraction processes which employ selective organic ligands such as TODGA (N,N,N′,N′-tetra-n-octyl diglycolamide), substituted bis-triazinyl pyridine,8 and crown ethers9 having their own limitations like secondary sludge generation, cost and recyclability of the material.
To alleviate the existing limitations, benign carbon-based sorbents are reported in the literature.10 To the best of our knowledge, no report has been made with carbon containing magnetic copper–iron-titanate nanomaterials as an inorganic magnetic sorbent for actinide separations from the radioactive feeds.11 The idea behind the incorporation of Ti and carbon in the ferrite material is to alter the surface charge and enhance the stability.12 These are the essential requirements for radioactive waste remediation under harsh conditions.13 Nevertheless, by retaining the magnetic property of the material with the desired stability and surface charge14 will address some of the serious problems faced in the actinide separation processes.
In this paper, an attempt had been made to synthesize a high defect containing copper–iron-titanium (CTF) metal oxide embedded with carbon material by a solvo-thermal decomposition method.15 The highlights of the method are in situ generation of carbon as a reducing agent to induce the defects and stability in the magnetic material, and non-requirement of post annealing thermal treatment which affects the magnetic property.16 Also it is easy to control the material property by altering the solution volume, initial metal ion concentration, and incorporation of foreign metal ions.17 The above mentioned magnetic CTF materials were tested for Am uptake with a view to recover highly radiotoxic actinides from the nuclear waste solutions which is one of the major challenges to the separation scientists working at the back end of nuclear fuel cycle.
Experimental
Materials
Iron(III) acetate, copper(II) acetate, titanium(IV) isopropoxide (all from Sigma-Aldrich) and specially dried HPLC grade methanol and ethanol (Merck chemical, 99.5% purity) were used as procured. 241Am radiotracer was from a freshly purified laboratory stock after ascertaining its radiochemical purity (ESI†).18 152,154Eu radiotracer was obtained from Board of Radiation and Isotope Technology (BRIT, Mumbai) and was used after checking its radiochemical purity by gamma-ray spectroscopy using an HPGe detector (Canberra).Synthesis of the inorganic ion-exchangers
To 1 g of iron(III) acetate (99.5%) taken in a 250 ml round bottom flask, fitted with a condenser, 65 ml of methanol was added and refluxed at 60 °C for 1 h at 600 rpm. After the solution colour changed from reddish to pale yellow-green, 0.609 g of titanium(IV) isopropoxide was added and the melange was further refluxed for 30 minutes under stirring and transferred to a Teflon-lined autoclave reactor. The reaction mixture was heated at 200 °C for 20 h and was subsequently allowed to cool down to room temperature and was filtered and washed with dry ethanol for several times to remove the organic impurities and dried at 100 °C for 1 h and it was coded as TF. Similarly, copper containing iron titanate was prepared in an identical manner using 0.21 g of copper(II) acetate, 0.304 g of titanium(IV) isopropoxide added with 1 g of iron(III) acetate and coded as CTF.Characterization of the inorganic exchangers
Rigaku MiniflexII Desktop X-ray Diffractometer using Cu Kα1 radiation (λ = 0.15406 nm) at an operating voltage of 30 kV and current of 15 mA was used for the powder X-ray diffraction (PXRD) measurements from 2° to 80° at 2° per minute as the scan speed. Morphology of the material was characterised by field emission-scanning electron microscope (JEOL JSM 7100M) and high resolution-transmission electron microscope (JEOL, JEM 2100). Zeta potential (ζ) of the materials was analysed by Malvern Nano-ZS7090 by using finely dispersed nanomaterials in water taken in the cell. After the 2 minutes, equilibrating time average, results were taken from 25 cycle measurements. Elemental analysis of the samples was carried out using Vario Micro Cube (Elementar) equipment where acetanilide was used as a standard.X-ray photoelectron spectroscopy studies were carried out using SPECS instrument with a PHOBOS 100/150 Delay Line Detector (DLD). Non monochromatic Al Kα radiation (1486 eV) with operating power 385 W was used as the X-ray source. All XPS spectra were first analysed using the Casa XPS software. Peak shift due to any charging were corrected using internal reference (C 1s line from adventitious aliphatic carbon). After the calibration, the background from each spectrum was subtracted using Shirley-type background. Tougaard function was used to deconvolute the high-resolution XPS spectra with least chi-square and R2 values of 0.995.
Nitrogen adsorption–desorption was performed at 77 K after degassing the sample at 200 °C for 4 h using ASAP 2010 (Micromeritics, USA) instrument. Brunauer–Emmett–Teller (BET) equation was used to calculate the surface area and BJH cylindrical pore approximation was used to determine the pore size distribution. NH3-temperature programmed reduction analysis was carried out using Micromeritics AutoChem II (USA). In brief, in situ material activation was done at 120 °C for 2 h, and a mixture of 10% NH3 and pure He gas was passed for 10 more minutes for adsorption. Subsequently, He gas was purged for 10 minute to remove the physisorbed NH3 molecule, followed by heating at a rate of 10 °C per minute (10 to 800 °C) to desorb the remaining NH3 molecules from the sample.
Americium adsorption studies
The uptake of the metal ions was determined by equilibrating a known volume of aqueous phase (1 ml) containing the required radiotracer (vide supra) with a known amount of nanomaterial (adsorbent) (ca. 10 mg) in a thermostated water bath. After equilibration, the tubes were centrifuged and the aqueous layer was separated. Suitable aliquots of the aqueous phase were taken before and after equilibration for the assay of metal ions. Assay of 241Am and 152,154Eu, was performed by gamma ray counting (ESI†) using a NaI(Tl) scintillation counter (Para Electronics) coupled with a multi-channel analyzer (ECIL, India). The concentrations of Am and Eu used in the adsorption studies were 10−7 and 10−5 M, respectively. For the isotherm studies the aqueous solution contained 100 mg ml−1 Eu and was also spiked with 152,154Eu radiotracer. The weight distribution coefficient (Kd) of the metal ions on the nanomaterial was calculated by employing the following formula,
 | (1) |
Results
The TF and CTF nanomaterials were subjected to various characterization methods. BET surface area analysis results indicated surface area of the TF and CTF nanomaterials as 35 m2 g−1 and 118 m2 g−1, respectively suggesting wide variation in their particle sizes. Powder X-ray diffraction (PXRD) pattern of Fe2TiOx (Fig. 1), preparation under solvo-thermal conditions from methanol at 200 °C/20 h, clearly indicates the formation of pseudobrookite iron titanate and it matches very well with the Fe2TiO5 (pseudobrookite JCPDS no.-98-002-4134) and the reported literature for pseudo brookite.19 It was noted that the absence of the peak at 27.3° clearly indicated the absence of Ti in TiO2 form in the prepared nanomaterial.20 To understand the formation and crystalline growth of iron titanate, materials prepared at different time intervals were analysed by PXRD. The time dependent PXRD studies (ESI, Fig. S1†) showed more amorphous nature in the sample prepared in less than 5 h. The observed PXRD for Fe2TiOx (5 h & <5 h) matched well with Fe2TiO3 (ilmenite JCPDS-04-011-8810) whereas with increasing the treatment time (>5 h) showed more crystalline nature of Fe2TiO3. The sample prepared at 10 h and above resembled with Fe2TiO5 (pseudobrookite JCPDS no.-98-002-4134) as reported in the literature.21 In the present study, the possibility of the transformation of ilmenite to pseudobrookite is more feasible due to the thermal treatment and oxygen availability. Nevertheless, PXRD peaks around 62 and 63° are commonly observed in alpha iron oxide and iron titanate and the planes mentioned in the Fig. S1 (ESI†) help in distinguishing the two different iron titanate phases. Further to understand the structural changes, Fe2TiOx sample prepared at different time intervals are characterized by Field Emission-Scanning Electron Microscope (FE-SEM). | ||
| Fig. 1 PXRD of different iron titanate TF-Fe2xTixO5x & CTF-Fe2Ti0.5xCu0.5xO5x prepared at 200 °C for 20 h. | ||
FE-SEM images of copper iron titanate (CTF) and iron titanate (TF) (Fig. 2) showed layered hexagonal plate-like structure. The formation of stable layered hexagonal shape crystal in the absence of any template increases the curiosity to understand the nanomaterial formation in the supercritical solvo-thermal condition. Here, iron alkoxide was present in the heterogeneous phase and titanium precursor was taken in the homogeneous phase. The presence of iron precursor in the heterogeneous phase analysed by FE-SEM (ESI Fig. S2†) depict rods like structure.
 | ||
| Fig. 2 FE-SEM image of different iron titanates prepared at 200 °C for 20 h (CTF-copper–iron titanate, TF-iron titanate). | ||
Also, the time dependent particle growth (characterization by FE-SEM) showed that the structure retains for 5 h treated sample and further particle growth taking place with increasing reaction time (ESI Fig. S3a–d†). From the time depended morphology study, it was apparent that the formation of the nanoparticle took place in two steps. While the first step involved the formation of iron alkoxide which acted as nucleus for the particle growth, Ostwald ripening takes place in the second step.22 Beside, with increasing time the generated small nucleus combined together and formed the hexagonal structure which is the plausible mechanism for the nanomaterial formation.23 So, the possible way of particle growth in the reaction may be the decomposition of the titanium precursor in the solvo-thermal conditions followed by deposition/exchange of titanium over the iron precursor.24
In order to understand and interpret the structural and textural changes of the prepared iron titanate, High Resolution-Transmission Electron Microscope (HR-TEM) analysis was carried out for Fe2TiOx. The observed HR-TEM images (Fig. S4†) are similar to the FE-SEM images. The Fe2TiOx 5 h image (ESI S4a†) showed the small particle domain attached linearly and each linear grown molecules attached together and makes the complex structure, the high magnification image of the sample's fringes are not very clear, indicating the poor crystallinity which supports the PXRD results for the weak peak intensity and the absence of some crystalline peaks. Also, the low-resolution particle size image showed that the crystalline formations initially occur on the surface. Therefore, Ti ion deposition (reaction) on iron precursor may be possible on the bulk surface. The HR-TEM of the 10 h treated surface particles showed more crystalline nature than the sample with 5 h treatment. Similarly, the rest of the (15 h and 20 h treated samples with hexagonal structure) images indicated high crystallinity. The d-spacing value (ca. 4.3 Å) calculated from the crystal fringes for 15 and 20 h thermally treated samples confirmed the crystal growth in the (021) plane of Fe2TiO5. From the PXRD and HR-TEM data, it was confirmed that the formation of pseudobrookite increased with increasing reaction time. The HR-TEM images supported the possibility of Ti deposition/decomposition over the iron cluster which may further result in the formation of Fe2TiOx molecules. Similar type of formation was reported by Agafonov et al.23 Moreover, the Selected Area Electron Diffraction (SAED) pattern (ESI S5†) showed nanocrystalline nature in 5 and 10 h treated Fe2TiOx-sample and polycrystalline nature in more than 10 h treated sample. With increasing reaction time small particle agglomerate were found to form in the bulk material. Besides the particle growth, the oxidation of material/sintering of metal in the bulk would result in the different crystalline formations. With the particle growth, the surface charge of the nanomaterial is expected to differ with increasing reaction time. This was confirmed by zeta potential measurements. Zeta potential (ζ), an indicator of the surface charge of the particle, depicts the positive surface charge on the iron titanate (Table 1) which increased with increasing time up to 15 h indicating more acidic site availability in the prepared material. There was a drop in the positive charge with further increase in the reaction time and a shift towards the negative charge region. The observed positive charge is the expected one, and it evinced the proposal for particle/structure/textural growth from the FE-SEM and HR-TEM results. Because iron titanate is a n-type semiconductor material, here the observed positive charge is a solid evidence for the more availability of Ti over the surface.25 The surface charge of metal oxide was found to rise due to the terminal oxygen/defect and this could be induced inadvertently by the metal ions with different oxidation states.14 During the metal oxide formation, the deliberately added foreign metal ion occupies the interstitial position/added as add-atoms in the host surface, and this exerted a profound effect on the metal oxide charge property.26 However, in iron titanate, the availability of Ti4+ is responsible for the generation of acidic sites. This was reported by Liu et al., who suggested that the higher availability of Ti4+ in the surface might be the reason for the positive nature of the surface.27 Hence, this result supports the inadvertent deposition of homogeneous phase metal precursor into the heterogeneous phase metal. Apart from the afore mentioned reason, the partial oxygen pressure during the synthesis also affects the surface charge, as a lower oxygen partial pressure may impart a major contribution towards the surface charge. In general, metal oxide preparation under high oxygen environment results in more negative charge and partial/reduced oxygen conditions produce more positive charge on the surface.28 Here, the synthesis of metal oxide was carried out in the organic phase with less oxygen availability. Therefore, the possibility for oxygen source is in situ generation by the decomposition of organic counter ions and solvent molecules. In a nutshell, the limited oxygen availability may be the reason for the positive charge formation and further change occurs due to the sustainable availability of oxygen with increasing timing.
| Nano material | Fe2TiO5 (5 h) | Fe2TiO5 (10 h) | Fe2TiO5 (15 h) | Fe2TiO5 (20 h) | Fe2Ti(0.5)Cu(0.5)O5 (20 h) | CuFe2O4 (10 h) |
| Zeta potential (ζ) mV | 19.4 | 19.98 | 33.6 | 17.43 | −6.25 | −14 |
By tailoring the textural property and the surface charge of iron titanate under solvo-thermal treatment time enabled us to utilize the material for the specific application. Because sorption of the metal ion in the solution phase happens due to the complex formation on the adsorbent surface, it governs the type and selectivity of the specific species (adsorbate) in the solution phase.26 Here, the observed positive charge and its shift towards negative with increasing the reaction time evinced the presence of more positive charged metal ion in the surface than the surface oxygen molecules. And decreasing the positive charge after a longer thermal treatment (above 10 h) may be due to the sintering of metal ions/reduction and the formation of more oxygen molecule over the surface which results in the decrease in the positive charge. The zeta potential of the nanomaterial without titanium ions showed higher negative charge which decreases with an increase in the titanium concentration.
This may be due to the presence of copper ion in the solution which reduces the Ti(IV) to lower oxidation states which in turn affect the deposition rate and growth of nanoparticle in a particular plane by competing/enhancing the deposition of the titanium ions on the surface. Thus, it will impact the surface charges in the nanomaterial. In addition, the decomposition of the acetate precursor in solvo-thermal supercritical condition produces in situ carbon in the reaction also has some role in the surface charge. The specific reason for the incorporation of titanium is to alter the surface charge and copper for retaining the magnetic property of the material. The magnetic property of the sorbent material simplifies the separation process in the post adsorption process. Thus, it is expected to impact the surface charges in the nanomaterial. Therefore, to understand the changes in iron titanate by copper incorporation, the prepared iron titanate was analysed by X-ray photoelectron spectroscopy (XPS).
X-Ray Photoelectron Spectroscopy (XPS) full scan results showed the presence of Ti, Cu, O and C in the prepared material and the results were compared with the reported literature. The high-resolution XPS spectra (Fig. 3) gave a peak at 931.1, 952.1 and satellite peak at 943 eV evincing the presence of copper in the +2 oxidation state. Similarly, the high-resolution spectra of Fe (710.2 & 724.3 eV) indicate the existence of Fe3+. In contrast, XPS peak position and the band separation values of Cu and Fe resembled with ferrite type material.29 The broad XPS peak of Ti (Fig. 4) indicates the different oxidation state of Ti in both the samples. However, the deconvoluted spectra and the peak positions revealed the co-existence of Ti2+, Ti3+ and Ti4+ in CTF and Ti3+ and Ti4+ in TF (sample without copper), where Ti3+ is more21–29 predominant in the latter (457.1 eV).30,31 The deconvoluted O 1s peaks of CTF and TF material showed the presence of lattice and free oxygen in the material surface (529.4 and 531.5 eV), and the higher intensity of O 1s for CTF material needs to be mentioned here. Overall, the XPS spectra of Ti revealed the reduced oxidation in the synthesised material; it supports the possibilities for the reduction of metal in the solvo-thermal method. When the Ti atom is present in the +4 oxidation state, it may lead to some inductive effect in the metal–oxygen bonding as reported in the literature.32 Here, the reduced Ti oxidation state may alter the surface charge of the material which may lead to changes in the adsorption-based separation processes.27 So, the prepared material tested for metal ion separation from synthetic acidic feeds containing Am3+ which mimic the radioactive wastes.33 The presence of carbon and titanium in different oxidation states with the magnetic property will be a suitable material for working in acidic feed conditions.34
 | ||
| Fig. 3 X-ray photoelectron spectroscopy spectrum of TF and CTF prepared at 200 °C/20 h (Fe2xTi1xOnx, Fe2xCu0.5x Ti0.5xOnx). | ||
Americium separation
 | ||
| Fig. 5 (a) Kinetics of sorption of 241Am from pH 4 solution at 25 °C; (b) pseudo first order and (c) pseudo second-order data fittings. | ||
To understand the controlling mechanism of the sorption process, the kinetics data were fitted with the commonly used kinetics models such as the pseudo-first order Lagergren model35 and Ho's pseudo-second order kinetics model.36 The pseudo-first order rate kinetics can be described by the following expression,34
ln(qeq − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qmax − k1 × t qmax − k1 × t
| (2) |
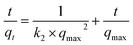 | (3) |
The Langmuir isotherm37 dwells on several fundamental assumptions viz., only one monolayer of a sorbing species is present on the surface of the sorbent; all sites of the sorbent are equivalent; the ability of a molecule to sorb at a given site is independent of the nature of the neighbouring sites, and the enthalpy of the sorption is constant. As shown in Fig. 6a, the linear plot of Ceq/q versus Ceq suggests that the sorption of europium on TF and CTF obeys the Langmuir sorption model. The Langmuir constant qmax and b, obtained by fitting the experimental data are listed in Table 2 (ESI, Table S2†). The calculated values of qmax (5.9 mg g−1 and 20.4 mg g−1) were in close agreement with the experimentally obtained values of 6.1 mg g−1 and 21.3 mg g−1 of TF and CTF, respectively. The Langmuir constant b in the present study was <1 (Table 2), indicating that the sorption process was favourable for both the nanomaterials. The good representation of the sorption equilibrium by the Langmuir model supports the hypothesis that the sorption of Eu(III) onto the nanoparticles proceeds via a chemical reaction and the sorption takes place onto the monolayer.
 | ||
| Fig. 6 Sorption isotherm data obtained with 152,154Eu (used as a surrogate of 241Am) from pH 4 solution at 25 °C; (a) Langmuir isotherm (b) Freundlich isotherm. | ||
| Nanomaterial type | Langmuir model | Freundlich model | |
|---|---|---|---|
| qmax (mg g−1) | exp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) q (mg g−1) q (mg g−1) |
1/n | |
| Iron titanate (TF) | 5.9 ± 0.1 | 6.1 ± 0.2 | 0.31 ± 0.03 |
| Copper–iron titanate (CTF) | 20.4 ± 0.7 | 21.3 ± 0.4 | 0.33 ± 0.01 |
The Freundlich sorption isotherm model was also tested for the present system, which assumes multilayer sorption of metal ions on the surface.38,39 The data fitted in the model did not fit on the calculated data points for TF (Fig. 6b). This observation reflected the absence of multilayer sorption of europium. On the other hand, very good fitting was observed for CTF indicating the presence of multilayer sorption of europium, (Fig. 6b) which supports the higher efficiency of CTF nanomaterial as compared to that of TF. In addition, it is also known that 1/n in the Freundlich model is the adsorption intensity and the values of 1/n indicate the type of isotherm to be irreversible (1/n = 0), favorable (0 < 1/n < 1) or unfavorable (1/n > 1).40 As results are shown in Table 2 (ESI, Table S3†), the 1/n of the Freundlich isotherm obtained was 0.31 for TF and 0.33 for CTF, which revealed that europium was favorably adsorbed onto the nanomaterial. To know the specific reason for the adsorption behaviour of CTF, the nanomaterials were prepared with the higher concentration of Ti and without Ti (simple copper ferrite) and tested at pH 4. It showed the poor adsorption behaviour for the nanomaterials without any Ti (copper ferrite) as well as those containing excessive Ti. Generally, magnetic materials, in particular, iron oxide and copper ferrite based adsorbents have been employed for the metal ion separation.41 However, for most of the magnetic materials, the adsorption properties arise from the defects present in the nano material or from the swinging adsorption behaviour of magnetic material as reported in the literature.41 Besides, the surface electronic property also plays a vital role in the sorption process. As in our case, the experiments have been carried out under mildly acidic condition suggesting fewer possibilities for swinging adsorption of the nanomaterial due to the acidic conditions. Also, the simple copper ferrite sorption behaviour proved the afore mentioned statement (copper ferrite is completely magnetic material). So, to get an idea of the nature of the material, CFT, and TF nanomaterials were characterised by NH3-TPD,42 which indicated drastic changes in the moderated acidic sites of CFT than TF nanomaterial, i.e., less electron deficient nature of the metal centres.43 Hence, the major possibilities for the CFT adsorption may be due to the electron rich metal–oxygen centres present on the surface of the nanomaterials. The less inductive effect from the Ti discussed in the XPS analysis (vide supra) and the surface charge measurement from zeta potential results also support the surface charge based sorption behaviour of the CTF nanomaterial. So, more possibilities for the metal ion reduction in the nanomaterial surface may be the plausible mechanism of adsorption of Am(III) in the nanomaterial surface along with synergic stability from the Ti, and carbon present in the material. Thus, these observations support the possibilities the chemisorbed sorption of Am(III) onto the CTF nanomaterial.
 | ||
| Fig. 7 Desorption and reusability (using the conditions used for sorption studies as mentioned above) results using (a) 1 M HNO3 and (b) 0.1 M HNO3. | ||
The sorption data of the TF and CTF nanomaterials are compared with the results of analogous materials reported in the literature. The comparison, as shown in Table 3, clearly suggests that CTF can be considered as one of the promising sorbents for Am uptake from aqueous radioactive feeds.
| Material | Kd value | Sorption capacity | Operating pH | Ref. |
|---|---|---|---|---|
| Zirconium phosphate | ca. 103 | — | 2.5–3 | 44 |
| Cerium vanadate | ca. 103 | 0.096 mg g−1 | 1–6 | 45 |
| Zirconium vanadate | 2.3 × 102 | 0.1 meg g−1 | 2–4 | 46 |
| Titanosilicate | ca. 104 | 7.1 meq g−1 | Neutral | 47 |
| Iron titanate | 2.3 × 104 | 6.1 mg g−1 | ca. 4 | This work |
| Copper iron titanate | 3.4 × 104 | 21.3 mg g−1 | ca. 4 | This work |
Conclusions
In summary, hexagonal sheet like pseudo brookite iron titanate (TF) and the copper iron titanate (CTF) were synthesised by solvo-thermal conditions using methanol as a low boiling solvent at 200 °C/20 h. This solvo-thermal method was chosen to prepare the material with carbon in order to tune the surface charge as well as defect in the nanomaterial. And it was confirmed by physico-chemical methods like PXDR, FE-SEM, HR-TEM and XPS analysis. The prepared CTF and TF nanomaterials were used as sorbents for radioactive tracer 241Am in nitric acid medium at pH 4. The rate constant (k2) and linear regression analysis suggested that the pseudo-second order kinetic model may be best fitted with the sorption data. The isotherm model fit obtained by the radioactive tracer 152,154Eu indicated the higher efficiency of copper containing iron titanate. Also, the adsorption isotherm fits suggested that the both the material (CTF and TF) followed the Langmuir fit, and the observed and theoretical value were very close. On the other hand, 1/n value calculated Freundlich isotherm was ca. 0.33 for CTF and TF suggested the favourable absorption of 152,154Eu in both (CTF and TF) the nanomaterials. To know the higher efficiency of copper contain iron titanate NH3-temperature programmed reduction experiments were also carried out. The substantial shift in the moderate acid site for CTF indicates the considerable change in the electronic nature in the metal–oxygen site, which corroborates the possibilities of the higher efficiency of CTF is due to the chemisorption based adsorption by the nanomaterial.Acknowledgements
The authors thank CSIR for funding under the network project “Waste to Wealth” CSC-0120, CSIR-CSMCRI institute communication number-128/2016. They (RBG and PKM) are also thankful to Dr P. K. Pujari, Head, Radiochemistry Division for his keen interest. They are also thankful to Dr A. K. Tripathi, Chemistry Division, BARC for XPS analysis of the samples.Notes and references
- A. T. Nguyen, L. T. M. Nguyen, C. K. Nguyen, T. Truong and N. T. S. Phan, ChemCatChem, 2014, 6, 815–823 CrossRef CAS.
- Y. Li, L. Kang, G. Bai, P. Li, J. Deng, X. Liu, Y. Yang, F. Gao and W. Liang, Electrochim. Acta, 2014, 134, 67–75 CrossRef CAS.
- P. Xu, G. M. Zeng, D. L. Huang, C. L. Feng, S. Hu, M. H. Zhao, C. Lai, Z. Wei, C. Huang, G. X. Xie and Z. F. Liu, Sci. Total Environ., 2012, 424, 1–10 CrossRef CAS PubMed.
- M. Kaur, H. Zhang, L. Martin, T. Todd and Y. Qiang, Environ. Sci. Technol., 2013, 47, 11942–11959 CrossRef CAS PubMed.
- P. Homchuen, R. D. Alorro, N. Hiroyoshi, R. Sato, H. Kijitani and M. Ito, Miner. Process. Extr. Metall. Rev., 2016, 1–9, DOI:10.1080/08827508.2016.1181629.
- M. N. Akieh, M. Lahtinen, A. Vaisanen and M. Sillanpaa, J. Hazard. Mater., 2008, 152, 640–647 CrossRef CAS PubMed.
- A. Wilden, C. Schreinemachers, M. Sypula and G. Modolo, Solvent Extr. Ion Exch., 2011, 29, 190–212 CrossRef CAS.
- K. N. Tevepaugh, J. D. Carrick, S. Tai, J. G. Coonce, L. H. Delmau and D. D. Ensor, Solvent Extr. Ion Exch., 2015, 1–13, DOI:10.1080/07366299.2015.1110408.
- N. N. Popovaa, V. I. Zhilova, S. V. Deminb and A. Y. Tsivadze, Russ. J. Inorg. Chem., 2015, 60, 754–758 CrossRef.
- R. B. Gujar and P. K. Mohapatra, RSC Adv., 2015, 5, 24705 RSC.
- I. V. Vasylenko, K. S. Gavrilenko, I. E. Kotenko, O. Cador, L. Ouahab and V. V. Pavlishchuk, Theor. Exp. Chem., 2014, 50, 226–231 CrossRef CAS.
- B. Jang, M. Park, O. B. Chae, S. Park, Y. Kim, S. M. Oh, Y. Piao and T. Hyeon, J. Am. Chem. Soc., 2012, 134, 15010–15015 CrossRef CAS PubMed.
- E. M. Wylie, D. T. Olive and B. A. Powell, Environ. Sci. Technol., 2016, 50, 1853–1858 CrossRef CAS PubMed.
- M. L. Machesky, D. J. Wesolowski, D. A. Palmer and M. K. Ridley, J. Colloid Interface Sci., 2001, 239, 314–327 CrossRef CAS PubMed.
- D. R. Modeshia and R. I. Walton, Chem. Soc. Rev., 2010, 39, 4303–4325 RSC.
- C. Yuan, H. B. Wu, Y. Xie and X. W. Lou, Angew. Chem., Int. Ed., 2014, 53, 1488–1504 CrossRef CAS PubMed.
- R. I. Walton, Chem. Soc. Rev., 2002, 31, 230–238 RSC.
- A. Khaleel, Colloids Surf., A, 2009, 346, 130–137 CrossRef CAS.
- T. W. Kim, H.-W. Ha, M.-J. Paek, S.-H. Hyun, I.-H. Baek, J.-H. Choy and S.-J. Hwang, J. Phys. Chem. C, 2008, 112, 14853–14862 CAS.
- Q. Liu, J. He, T. Yao, Z. Sun, W. Cheng, S. He, Y. Xie, Y. Peng, H. Cheng, Y. Sun, Y. Jiang, F. Hu, Z. Xie, W. Yan, Z. Pan, Z. Wu and S. Wei, Nat. Commun., 2014, 5, 5122 CrossRef CAS PubMed.
- S. V. Pol, V. G. Pol and A. Gedanken, Chemistry, 2004, 10, 4467–4473 CrossRef CAS PubMed.
- L. S. Zhong, J. S. Hu, H. P. Liang, A. M. Cao, W. G. Song and L. J. Wan, Adv. Mater., 2006, 18, 2426–2431 CrossRef CAS.
- A. V. Agafonov, D. A. Afanasyev, T. V. Gerasimova, A. S. Krayev, M. A. Kashirin, V. V. Vinogradov, A. V. Vinogradov and V. G. Kessler, ACS Sustainable Chem. Eng., 2016, 4, 2814–2821 CrossRef CAS.
- C. Li, T. Wang, Z. Luo, S. Liu and J. Gong, Small, 2016, 12, 3415–3422 CrossRef CAS PubMed.
- V. H. Gordon, E. Brown Jr, W. Casey, D. Clark, C. Eggleston, A. F. Andrew, D. W. G. Felmy, M. Gratzel, G. Maciel, M. I. McCarthy, K. H. Nealson, M. T. Dimitri Sverjensky and J. M. Zachara, Chem. Rev., 1999, 99, 77–174 CrossRef.
- F. Liu and H. He, J. Phys. Chem. C, 2010, 114, 16929–16936 CAS.
- J. L. G. Fierro, Metal Oxides-Chemistry and Applications, 2006 Search PubMed.
- R. Jamatia, A. Gupta and A. K. Pal, ChemistrySelect, 2016, 4, 852–860 CrossRef.
- N. S. Leyland, J. Podporska-Carroll, J. Browne, S. J. Hinder, B. Quilty and S. C. Pillai, Sci. Rep., 2016, 6, 1–10 CrossRef.
- M. C. Biesinger, L. W. M. Lau, A. R. Gerson and R. S. C. Smart, Appl. Surf. Sci., 2010, 257, 887–898 CrossRef CAS.
- L. Wang, N. T. Nguyen and P. Schmuki, ChemSusChem, 2016, 9, 2048–2053 CrossRef CAS PubMed.
- H. J. Lee, W. Cho, E. Lim and M. Oh, Chem. Commun., 2014, 50, 5476–5479 RSC.
- M. Rossier, F. M. K. E. K. Athanassiou, R. N. Grass, M. Waelle, K. Birbaum and W. J. S. Detlef Günther, Ind. Eng. Chem. Res., 2010, 49, 9355–9362 CrossRef CAS.
- H. Bessbousse, T. Rhlalou, J.-F. o. Verchère and L. Lebrun, J. Phys. Chem. B, 2009, 113, 8588–8598 CrossRef CAS PubMed.
- S. Lagergren, Kungliga, S. Vetenskapsakademiens and Handlinger, 1898, p. 24 Search PubMed.
- E. Bayram, N. Hoda and E. Ayranci, J. Hazard. Mater., 2009, 168, 1459–1466 CrossRef CAS PubMed.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- G. McKay, H. S. Blair and J. R. Gardner, J. Appl. Polym. Sci., 1982, 27, 3043–3057 CrossRef CAS.
- M. M. Dubinin and L. V. Radushkevich, Proceedings of the Academy of Sciences of the USSR, Physical Chemistry Section, 1947, 55, 331 Search PubMed.
- M. Arami, N. Yousefi Limaee and N. M. Mahmoodi, Chemosphere, 2006, 65, 1999–2008 CrossRef CAS PubMed.
- G. B. Cotten, H. B. Eldredge and J. D. Navratil, Nukleonika, 2003, 48, 17–23 CAS.
- E. W. McFarland and H. Metiu, Chem. Rev, 2013, 113, 4391–4427 CrossRef CAS PubMed.
- K. Saravanan, B. Tyagi, R. S. Shukla and H. C. Bajaj, Appl. Catal., B, 2015, 172–173, 108–115 CrossRef CAS.
- H. Mimura, K. Akiba and N. Yamamoto, J. Nucl. Sci. Technol., 1995, 38, 819–821 CrossRef.
- C. Banerjee, N. Dudwadkar, S. C. Tripathi, P. M. Gandhi, V. Grover, C. P. Kaushik and A. K. Tyagi, J. Hazard. Mater., 2014, 280, 63–70 CrossRef CAS PubMed.
- K. Roy, P. K. Mohapatra, N. Rawat, D. K. Pal, S. Basu and V. K. Manchanda, Appl. Radiat. Isot., 2004, 60, 621–624 CrossRef CAS PubMed.
- L. Al-Attar, A. Dyer and R. Harjula, J. Mater. Chem., 2003, 13, 2963–2968 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra24266a |
| This journal is © The Royal Society of Chemistry 2016 |

