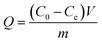Amino acid-based ionic liquid surface modification of magnetic nanoparticles for the magnetic solid-phase extraction of heme proteins†
Binghai Wanga,
Xiong Wanga,
Juanqiang Wanga,
Xue Xuea,
Xingjun Xib,
Qiao Chub,
Genlai Dongb and
Yun Wei *a
*a
aState Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, 15 3rd North East Road, Chaoyang District, Beijing 100029, China. E-mail: weiyun@mail.buct.edu.cn
bChina National Institute of Standardization, No. 4 Zhichun Road, Haidian District, Beijing 100191, P. R. China
First published on 20th October 2016
Abstract
In this study, magnetic nanoparticles with an amino acid-based ionic liquid directly modified onto their surface were fabricated as a new magnetic adsorbent (Fe3O4@DIH–EMIMLpro) for the magnetic solid phase extraction of protein. The synthesized Fe3O4@DIH–EMIMLpro nanoparticles were characterized using Fourier transform infrared spectrometry, transmission electron microscopy, vibrating sample magnetometry, X-ray diffraction and thermogravimetric analysis. Hemoglobin was chosen as a model protein to investigate the extraction performance of the prepared materials. After extraction, the concentration of protein in the supernatant was determined by UV-vis spectrophotometry. The adsorbed hemoglobin could be eluted from the Fe3O4@DIH–EMIMLpro nanoparticles by sodium dodecyl sulfate solution. The Fe3O4@DIH–EMIMLpro nanoparticles could be used for rapid, efficient, and selective adsorption of hemoglobin with a binding capacity as high as 1.58 mg mg−1. Moreover, the extraction process of protein from human whole blood by Fe3O4@DIH–EMIMLpro has been performed to evaluate the practical application of the proposed method.
1. Introduction
The selective separation and detection of protein targets from complex biological samples is one of the most intensively researched subjects owing to its significant applications in diagnostics, proteomics and protein purification.1,2 Several analytical methods such as two aqueous phase extraction,3 solid phase extraction,4 immobilized metal ion affinity chromatography,5,6 electrophoresis7 and molecular imprinting8,9 have been reported for the separation and purification of proteins from cell debris or other complex mixtures. To the best of our knowledge, although these approaches have shown a broad range of bioapplications in the field of protein separation, some limitations including time consuming processes and high consumption of solvents, as well as complicated pre-treatment of samples, still exist. Therefore, it is highly necessary to develop a new alternative for the separation and purification of proteins.In recent years, magnetic solid phase extraction (MSPE), as an improved solid phase extraction (SPE) that is based on the application of magnetic or magnetically modified adsorbents, has emerged as an alternative method.10,11 In MSPE procedures, the magnetic adsorbents are dispersed into the sample solution to capture the analytes and then directly separated from the sample matrix by an external magnetic field, which greatly simplifies the SPE procedure and enhances the extraction efficiency.12 Thus, some efforts have been made in developing various magnetic adsorbents, and further exploiting their potential applications in MSPE.13–16 For example, Ji’s group reported the use of amino acid-based polymer functionalized magnetic nanocomposites (MNPs) as adsorbents for the analysis of plant growth regulators in bean sprouts.17 Wu et al. evaluated graphene-based MNPs for the MSPE of carbamate pesticides from environmental water samples.18 Gao et al. prepared magnetic amino-functionalized polyacrylic acid–nanomagnetite (Fe3O4@PAA@NH2) for hexavalent chromium removal from polluted water.19 Although substantial progress has been made, new magnetic adsorbents with simple preparation processes, low costs and high adsorption efficiencies are still highly desirable.
Ionic liquids (ILs) are a kind of organic salt that possess unique chemical and physical properties, such as high thermal and chemical stability, negligible vapor pressure, electroconductivity and non-inflammability.20 These excellent properties make them promising solvents and functional materials for sample pre-treatment, including liquid–liquid extraction,21 liquid phase microextraction,22 and solid phase microextraction.23 For example, Pino’s group applied the ionic liquid 1-hexadecyl-3-methylimidazolium bromide in a microwave-assisted liquid–liquid extraction system for polycyclic aromatic hydrocarbons (PAHs) in sediments.24 Aliyari et al. investigated modified surface-active ionic liquid-coated magnetic graphene oxide as a new magnetic solid phase extraction sorbent for preconcentration of trace nickel.25 However, ILs directly used for protein separation can lead not only to changes of protein conformation, but also to loss of activity due to long term effects. Recycling and reuse of ILs can also be difficult. The immobilization of ILs on the surface of magnetic nanoparticles (ILs–MNPs) as a new magnetic adsorbent can solve this problem very well. Up to now, ILs–MNPs have been used in many fields, for example in the separation of materials such as natural products26 and environmental contamination.27 It is an inviting prospect that a target protein, specifically adsorbed on magnetic nanoparticles modified by ionic liquids, could be isolated with the help of a magnet, which favours recycling and reusing multiple times.
As is well known, amino acid-based ionic liquids are a kind of functional materials, which have good biocompatibility and are easy available.28 Moreover, this kind of ionic liquid contains multiple functional groups, such as imidazolium cations, carboxylic acid roots, etc. Therefore, we assume that amino acid-based ionic liquid functionalized magnetic nanoparticles should have great potential in the extraction of proteins via multiple interactions.
In this work, an L-proline-based ionic liquid (1-ethyl-3-methyl-imidazolium L-proline, EMIMLpro) was introduced as a fabricated MSPE adsorbent. EMIMLpro-functionalized magnetic nanoparticles Fe3O4@DIH–EMIMLpro were synthesized via a crosslinking reaction in which 1,6-diisocyanatohexane is a crosslinking agent, and were further used as MSPE adsorbents for the separation of heme proteins which have an iron-containing heme group (such as hemoglobin). The resultant EMIMLpro-functionalized nanoparticles show a good adsorption capacity and unique selectivity due to covalent coordination between the ferrous atoms and the imidazolium cations in the ionic liquid,29 and electrostatic forces. Moreover, the proposed nanoparticles were further applied in separating hemoglobin from a human blood sample.
2. Material and methods
2.1 Reagents and chemicals
All reagents used in this work were of analytical grade and commercially available: iron(III) chloride hexahydrate (FeCl3·6H2O) and sodium dodecyl sulfatepolyacrylamide (SDS) were purchased from Tianjin Fuchen Chemical Reagent Factory. Sodium chloride (NaCl), potassium dihydrogen phosphate (KH2PO4), potassium hydrogen phosphate anhydrous (K2HPO4·3H2O), propylene glycol (PG), succinic acid, urea, anhydrous ethanol, aqueous ammonia solution (25%), concentrated hydrochloric acid (37.5%) and dimethyl sulfoxide (DMSO) were bought from Beijing Chemical Factory. Tris(hydroxymethyl) aminomethane (Tris) was purchased from Amresco Company. 1,6-Diisocyanatohexane (DIH) was purchased from J&K Company. 1-Ethyl-3-methyl-imidazolium L-proline (EMIMLpro) was obtained from Shanghai Cheng Jie Chemical Co., Ltd. Hemoglobin (Hb, H8020, pI = 6.9) and lysozyme from chicken egg white (Lys, L8120, pI = 11) were purchased from Solarbio. The human whole blood sample was provided by the Beijing Maternity Hospital. Solvents used in the preparation process were of analytical grade.2.2 Characterization and analytical conditions
The size and morphology of the prepared magnetic nanoparticles were observed by transmission electron microscopy (TEM Philips Tecnai 20, Netherlands). X-ray diffractometry (XRD) was performed on a RIGAKU diffractometer. Fourier transform infrared (FT-IR) spectra were obtained using a Thermo Electron Nexus 8700 FTIR spectrometer. Thermogravimetric analysis (TGA) was performed under a nitrogen atmosphere using STARTe System (TGA/DSC1 SF1100 of METTLER TOLEDO). An elemental analyzer (Elementar, Vario EL cube, Germany) was utilized to estimate the relative percentages of the elements C, H and N in Fe3O4@DIH–EMIMLpro. Room temperature magnetization isotherms of the magnetic nanoparticles were carried out using a vibrating sample magnetometer JDM-13 (vibrating sample magnetometer, VSM, Lake Shore 7410, USA). A water ultrasonicator KH-100E (Kunshan Ultrasonic Instrument, China) could be used to disperse the prepared nanoparticles in solutions. Moreover, a magnet, a mechanical stirrer RZR-2041 (Heidolph, Germany) and a vacuum drying oven DZG-6020 (Shanghai Senxin equipment, China) were used. A UV-vis spectrophotometer U-3010 (Hitachi, Japan) was used to measure the absorbance of the proteins. The maximum absorption wavelength of hemoglobin and Lys were 408 and 280 nm, respectively. All measurements were performed at ambient temperature.2.3 Synthesis of Fe3O4 nanoparticles
A solvent-thermal method was used to synthesize Fe3O4 nanoparticles in our previous work.30 In a typical procedure, 0.81 g (3 mmol) of FeCl3·6H2O, 0.12 g (1 mmol) of succinic acid and 1.8 g (30 mmol) of urea were completely dissolved in 30 mL of propylene glycol by ultrasonication to obtain a homogeneous yellow solution. Then, the resulting mixture was transferred to a Teflon-lined stainless steel autoclave, sealed and maintained at 200 °C for 8 h. After being cooled down to room temperature, the black precipitate that formed was washed five times with ethanol and deionized water with the help of a magnet. Finally, it was dried at 60 °C for 24 h to obtain the freshly prepared magnetic nanoparticles.2.4 Synthesis of Fe3O4@DIH–EMIMLpro nanoparticles
The above obtained Fe3O4 nanoparticles (0.2 g) were subsequently dispersed in 30 mL of DMSO and ultrasonicated for 10 minutes. 3 mL of EMIMLpro ionic liquid was then added to the solution and the mixture was stirred at 80 °C for 4 h. After stirring, 1 mL of DIH was added in batches as a coupling agent, and the resulting mixture was mechanically stirred and reacted for 48 h. As the reaction was completed, the synthesized magnetic nanoparticles were cooled to room temperature, then separated from the solvent using a magnet and washed five times with methanol and acetone to remove excess EMIMLpro ionic liquid and by-products. Finally, the product Fe3O4@DIH–EMIMLpro was dried under vacuum at 70 °C for 24 h.2.5 Magnetic solid phase extraction of hemoglobin
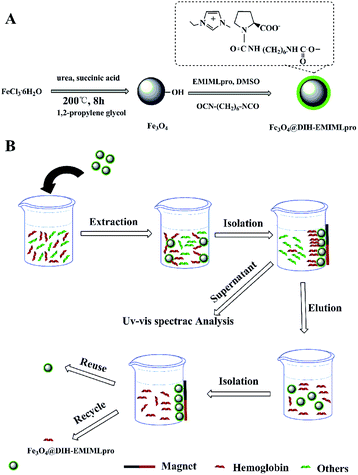
All experiments were performed in 10 mL glass vials. To conduct extraction, 2 mL of the phosphate buffered protein solution or blood samples were added to the bottle, and then a certain amount of Fe3O4@DIH–EMIMLpro nanoparticles was homogeneously dispersed under ultrasonication for 5 min. The extraction was subsequently performed under vortex for 10 min to attain equilibrium. After extraction, an external magnet was placed beside the glass bottle to collect the adsorbents. After removing the phosphate buffered solution, 2 mL of Tris buffer solution containing SDS and NaCl was added to the glass vials as an eluent under vortex for 10 min, and then the magnetic nanoparticles were collected with the help of a magnet. The concentrations of proteins in the supernatant were determined by a UV-vis spectroscopy (the absorbance for hemoglobin being at 408 nm and for Lys at 280 nm).where C0 and V are the added concentration and volume in prepared buffer protein solution, respectively. Ce stands for the equilibrium concentration of protein in the supernatant after magnetic separation. m is the amount of Fe3O4@DIH–EMIMLpro added.
3. Results and discussion
3.1 Synthesis and characterization of Fe3O4@DIH–EMIMLpro nanoparticles
The synthetic route for Fe3O4@DIH–EMIMLpro nanoparticles is shown in Scheme 1. This preparation procedure contains two parts: the first step was the synthesis of Fe3O4 nanoparticles via a solvothermal method; the second step gives EMIMLpro ionic liquid modified magnetic nanoparticles (named as Fe3O4@DIH–EMIMLpro).![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and the bending vibration of N–H, respectively. The band located at 1126 cm−1 belongs to the stretching vibration of C–N. The characteristic band of Fe–O at 600 cm−1 appears in the spectrum of Fe3O4@DIH–EMIMLpro, indicating the successful combination of EMIMLpro with Fe3O4 nanoparticles. Besides, as shown in Fig. S1,† the maximum absorption wavelengths of Fe3O4@DIH–EMIMLpro and EMIMLpro in the ultraviolet spectrum are 268 nm and 230 nm, respectively. This result further confirms the connection between EMIMLpro and the Fe3O4 nanoparticles due to the apparent red shift of the wavelength of Fe3O4@DIH–EMIMLpro nanoparticles.
O and the bending vibration of N–H, respectively. The band located at 1126 cm−1 belongs to the stretching vibration of C–N. The characteristic band of Fe–O at 600 cm−1 appears in the spectrum of Fe3O4@DIH–EMIMLpro, indicating the successful combination of EMIMLpro with Fe3O4 nanoparticles. Besides, as shown in Fig. S1,† the maximum absorption wavelengths of Fe3O4@DIH–EMIMLpro and EMIMLpro in the ultraviolet spectrum are 268 nm and 230 nm, respectively. This result further confirms the connection between EMIMLpro and the Fe3O4 nanoparticles due to the apparent red shift of the wavelength of Fe3O4@DIH–EMIMLpro nanoparticles.
3.2 MSPE procedure
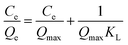 | (1) |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Qe = 1/n Qe = 1/n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce + log Ce + log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KF KF
| (2) |
Compared with the linear correlation coefficients of the two isothermal models in Table 1, we found that the Langmuir isotherm model (r = 0.9981) is more suitable for describing the adsorption process of hemoglobin onto Fe3O4@DIH–EMIMLpro than the Freundlich model (r = 0.5721). This result indicates that the adsorption process of proteins onto Fe3O4@DIH–EMIMLpro follows monolayer adsorption. Moreover, the maximum adsorption capacity of hemoglobin obtained from experiments (1.528 mg mg−1) is also close to the apparent maximum adsorption capacity of hemoglobin (1.581 mg g−1). Therefore, the binding of hemoglobin onto the prepared magnetic nanoparticles follows monolayer adsorption. In addition, the adsorption capacity of our work is much higher than some other reported work, shown in Table S1.†
| Langmuir model | Freundlich model | ||||
|---|---|---|---|---|---|
| Qe (mg mg−1) | KL (mL mg−1) | R2 | KF (mg mg−1) | 1/n | R2 |
| 1.528 | 5.606 | 0.981 | 0.948 | 0.233 | 0.572 |
3.3 Practical isolation of hemoglobin from human whole blood
The practical applicability of the prepared magnetic nanoparticles was investigated through the selective separation of hemoglobin from diluted human whole blood. In this work, SDS-PAGE was used to analyze the removal of proteins by the magnetic absorbent. As seen in Fig. 10, the intensity of the hemoglobin band (B14 kDa) is significantly weaker (Fig. 10, lane 3) after adsorption by the Fe3O4@DIH–EMIMLpro nanoparticles and the band of hemoglobin appeared again as the adsorbent was eluted with a desorption solution (Fig. 10, lane 4). In conclusion, these results prove the practical applicability of the EMIMLpro-modified magnetic nanomaterial for the effective isolation of hemoglobin from complex biological samples.4. Conclusions
In this work, the prepared Fe3O4@DIH–EMIMLpro combines the superiorities of amino acid-based ILs and Fe3O4 nanoparticles, revealing many fascinating properties which could not be achieved by either component alone. An environmentally friendly amino acid-based IL was utilized to directly decorate the Fe3O4 nanoparticles with 1,6-diisocyanatohexane to synthesize Fe3O4@DIH–EMIMLpro. In an MSPE process, several key elements such as solution pH, extraction capacity, selectivity recognition, recyclability and practical application were investigated. High extraction capacity, short extraction time, reusability and selective extraction were the advantages of Fe3O4@DIH–EMIMLpro as an adsorbent compared with traditional extraction materials. This result suggests that it has attractive potential to offer new possibilities in the extraction of proteins and proves that Fe3O4@DIH–EMIMLpro would be an important tool in bioseparation technology, as well as in other biotechnological applications.Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC, Grant No. 21075007), and the Program for New Century Excellent Talents in University (NCET-11-0563) and Beijing Nova program Interdisciplinary Cooperation Project are gratefully acknowledged.Notes and references
- B. Alvarez-Sanchez, F. Priego-Capote and M. D. Castro, TrAC, Trends Anal. Chem., 2010, 29, 120–127 CrossRef CAS.
- H. M. Chen, D. W. Qi, C. H. Deng, P. Y. Yang and X. M. Zhang, Proteomics, 2009, 9, 380–387 CrossRef CAS PubMed.
- N. Li, Y. Z. Wang, K. J. Xu, Y. H. Huang, Q. Wen and X. Ding, Talanta, 2016, 152, 23–32 CrossRef CAS PubMed.
- M. V. Barberan, M. J. L. García, E. F. S. Alfonso and J. M. H. Martínez, Anal. Chim. Acta, 2016, 917, 37–43 CrossRef PubMed.
- J. L. Cao, X. H. Zhang, X. W. He, L. X. Chen and Y. K. Zhang, J. Mater. Chem. B, 2013, 1, 3625–3632 RSC.
- C. Ding, X. D. Ma, X. Yao and L. Jia, J. Chromatogr. A, 2015, 1424, 18–26 CrossRef CAS PubMed.
- B. Yu, M. Chi, Y. X. Han, H. L. Cong, J. B. Tang and Q. H. Peng, Talanta, 2016, 152, 76–81 CrossRef CAS PubMed.
- Y. T. Liu, Y. X. Gu, M. L. Li and Y. Wei, New J. Chem., 2014, 38, 6064–6072 RSC.
- H. M. Duan, X. J. Wang, Y. H. Wang, J. B. Li and C. N. Luo, RSC Adv., 2015, 5, 88492–88499 RSC.
- Q. Liu, J. B. Shi, M. T. Cheng, G. L. Li, D. Cao and G. B. Jiang, Chem. Commun., 2012, 48, 1874–1876 RSC.
- J. Wang, Z. Y. Chen, Z. M. Li and Y. L. Yang, Food Chem., 2016, 204, 135–140 CrossRef CAS PubMed.
- H. Parham and F. Khoshnam, Talanta, 2013, 114, 90–94 CrossRef CAS PubMed.
- R. M. Frizzarin, C. P. Cabello, M. M. Bauza, L. A. Portugal, F. Maya, V. Cerda, J. M. Estela and G. T. Palomino, Anal. Chem., 2016, 88, 6990–6995 CrossRef CAS PubMed.
- M. A. Habila, Z. A. ALOthman, A. M. El-Toni, J. P. Labis and M. Soylak, Talanta, 2016, 154, 539–547 CrossRef CAS PubMed.
- S. L. Zhang, W. X. Yao, J. B. Ying and H. T. Zhao, J. Chromatogr. A, 2016, 1452, 18–26 CrossRef CAS PubMed.
- J. H. Zhu, S. Y. Wei, H. B. Gu, S. B. Rapole, Q. Wang, Z. P. Luo, N. Haldolaarachige, D. P. Young and Z. H. Guo, Environ. Sci. Technol., 2012, 46, 977–985 CrossRef CAS PubMed.
- S. L. Ji, L. Qi, N. Li and M. L. Wang, Talanta, 2016, 158, 229–234 CrossRef CAS PubMed.
- Q. H. Wu, G. Y. Zhao, C. Feng, C. Wang and Z. Wang, J. Chromatogr. A, 2011, 1218, 7936–7942 CrossRef CAS PubMed.
- F. Gao, H. B. Gu, H. W. Wang, X. F. Wang, B. Xiang and Z. H. Guo, RSC Adv., 2015, 5, 60208–60219 RSC.
- N. V. Plechkova and K. R. Seddon, Chem. Soc. Rev., 2008, 37, 123–150 RSC.
- C. C. Chang and S. D. Huang, Anal. Chim. Acta, 2010, 662, 39–43 CrossRef CAS PubMed.
- H. H. Bai, Q. X. Zhou, G. H. Xie and J. P. Xiao, Talanta, 2010, 80, 1638–1642 CrossRef CAS PubMed.
- X. Zhou, P. F. Xie, J. Wang, B. B. Zhang, M. M. Liu, H. L. Liu and X. H. Feng, J. Chromatogr. A, 2011, 1218, 3571–3580 CrossRef CAS PubMed.
- V. Pino, J. L. Anderson, J. H. Ayala, V. González and A. M. Afonso, J. Chromatogr. A, 2008, 1182, 145–152 CrossRef CAS PubMed.
- E. Aliyari, M. Alvand and F. Shemirani, RSC Adv., 2016, 6, 64193–64202 RSC.
- H. He, D. H. Yuan, Z. Q. Gao, D. L. Xiao, H. He, H. Dai, J. Peng and N. Li, J. Chromatogr. A, 2014, 1324, 78–85 CrossRef CAS PubMed.
- E. Yilmaz and M. Soylak, Talanta, 2013, 116, 882–886 CrossRef CAS PubMed.
- D. M. Kroupa, C. J. Brown, L. M. Heckman and T. A. Hopkins, J. Phys. Chem. B, 2012, 116, 4952–4958 CrossRef CAS PubMed.
- Y. Wei, Y. Li, A. L. Tian, Y. T. Fan and X. Wang, J. Mater. Chem. B, 2013, 1, 2066–2071 RSC.
- Y. T. Liu, Y. Li and Y. Wei, J. Sep. Sci., 2014, 37, 3745–3752 CrossRef CAS PubMed.
- Y. H. Huang, Y. Z. Wang, Y. Wang, Q. Pan, X. Q. Ding, K. J. Xu, N. Li and Q. Wen, RSC Adv., 2016, 6, 5718–5728 RSC.
- Y. T. Liu, A. L. Tian, X. Wang, J. Qi, F. K. Wang, Y. Ito and Y. Wei, J. Chromatogr. A, 2015, 1400, 40–46 CrossRef CAS PubMed.
- X. Xue, B. H. Wang, X. J. Xi, Q. Chu and Y. Wei, New J. Chem., 2015, 39, 5735–5742 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra23616b |
| This journal is © The Royal Society of Chemistry 2016 |