Ethanol thermal reduction synthesis of hierarchical MoO2–C hollow spheres with high rate performance for lithium ion batteries†
Lingxing Zeng ac,
Xiaoxia Huanga,
Xi Chena,
Cheng Zhengb,
Renpin Liua,
Gan Chena,
Qingrong Qian*ac,
Qinghua Chenac and
Mingdeng Wei*b
ac,
Xiaoxia Huanga,
Xi Chena,
Cheng Zhengb,
Renpin Liua,
Gan Chena,
Qingrong Qian*ac,
Qinghua Chenac and
Mingdeng Wei*b
aEngineering Research Center of Polymer Green Recycling of Ministry of Education, Fujian Normal University, Fuzhou, Fujian 350007, China. E-mail: qrqian@fjnu.edu.cn; Tel: +86-591-83753180
bInstitute of Advanced Energy Materials, Fuzhou University, Fuzhou, Fujian 350002, China. E-mail: wei-mingdeng@fzu.edu.cn
cFujian Key Laboratory of Pollution Control & Resource Reuse, Fuzhou, Fujian 350007, China
First published on 31st October 2016
Abstract
In the present work, we focus on the development of hierarchical MoO2–C hollow spheres with large reversible capacity and high rate capability in lithium ion batteries. The hierarchical MoO2–C hollow spheres assembled by MoO2 nanoparticles were initially synthesized successfully through ethanol thermal reduction method, followed by annealing process. Due to the synergistic effect between the assembled hierarchical hollow structure and the carbon coating, the sample exhibited excellent electrochemical properties as anode material for lithium-ion batteries. For example, a large reversible capacity of 763 mA h g−1 was obtained after 50 cycles at a current density of 0.2 A g−1. In addition, a large reversible capacity of 573 mA h g−1 can be remained when the current density reached at the high current density of 2 A g−1, indicating that the hierarchical MoO2–C hollow spheres electrode possess an outstanding rate performance.
1 Introduction
Lithium-ion batteries (LIBs) with large energy density and high rate capability have been widely regarded as the evolution of battery technology.1,2 Nowadays, one of the most significant challenges for the prospective application of LIBs is how to design and manufacture new anode materials with better electrochemical properties.3–10 Due to their high theoretical capacity for LIBs, transition metal oxides have recently found great potential to act as high performance electrode materials for LIBs.11–16 Among all these materials, monoclinic MoO2 has recently considered to be an attractive alternatives anode material, since it has high theoretical capacity (838 mA h g−1), low conversion potential and nontoxicity.17–20 However, its structure crack during the repetitive Li+ insertion/extraction process has limited its practical application.21–24 The rate capability and cycling stability are far from satisfactory.Up to now, many approaches to improve the activity of MoO2 have been attempted, such as fabricating carbon hybrid nanocomposite, developing hierarchical micro-nanostructure and designing hollow/porous structure.25–34 Benefiting from higher special surface area to provide more active sites, the shortened electronic-ionic diffusion pathways and enhanced composite conductivity, hierarchical micro-nanostructure or carbon hybrid nanocomposite could exhibit better electrochemical performance than bulk counterparts.35–41 A hierarchical MoO2/graphene nanoarchitectures can maintain a high capacity of 598 mA h g−1 at 1 A g−1 after 50 cycles.42 The hierarchical MoO2 nanotubes maintained a large reversible capacity of 720 mA h g−1 at 0.1 A g−1 after 70 cycles.43 The MoO2–Mo2C–carbon composite remained a reversible capacity of 724 mA h g−1 at 0.2 A g−1 after 50 cycles.44 A hierarchical MoO2 thin film showed a stable discharge capacity of 760 mA h g−1 at 0.4 A g−1 after 100 cycles.45 The MoO2 nanoparticles anchored on graphene delivered a high capacity of 640 mA h g−1 at 0.2 A g−1 after 50 cycles.46 Our previous work also revealed that a MoO2-ordered mesoporous carbon nanocomposite exhibited a stable capacity of 689 mA h g−1 at 0.05 A g−1 after 50 cycles.47 Although the above method has been proved to be effective, but MoO2-based anode materials with high rate performance are still limited and need to be improved.
Recently, the solvent-thermal reduction method as a process for preparing low-valence-state transition metal oxide with hierarchical structures has been drawn much attention and received widely concerns.48–51 On the other hand, this approach is a facile strategy for in situ fabricating carbon composite. We initially fabricated hierarchical MoO2–C hollow spheres by ethanol thermal reduction method followed by annealing process. The hierarchical MoO2–C hollow spheres can be more effective to accommodate the volume expansion, shorten the diffusion distance of Li+ and electron, and also provide more active sites to improve the storage of lithium. The hierarchical MoO2–C hollow spheres electrode delivered a large reversible capacity and high rate performance as anode material of LIBs.
2 Experimental
2.1 Preparation and characterizations
The MoO2–C precursors were synthesized via a facile ethanol thermal reduction method. In a typical procedure, 0.194 g of sodium molybdate and 0.15 g of cetyltrimethylammonium bromide (CTAB) were dissolved in 30 mL of ethanol. After vigorous stirring for 20 min, the resulting mixture was sealed in a 50 mL PTFE-lined stainless steel autoclave and was heated at 200 °C for 1 day or 2 days. By means of centrifugation, the precipitate was collected, repeatedly washed with distilled water and ethanol three times, and then dried overnight in an air oven at 80 °C. Finally, the powders were place in a tube furnace and sintered at 550 °C for 3 h under Ar flow to obtain the MoO2–C composites. The resulting final products were denoted as MoO2–C-1d and MoO2–C-2d for different heating time of ethanol thermal reduction process.X-ray powder diffraction (XRD) measurements were obtained on Bruker D8 diffractometer operated at a step of 5° per min in the range 10–80° with Cu-Kα radiation (λ = 0.15406 nm). The morphology of the products was analyzed by scanning electron microscopy (SEM, Hitachi 4800). The transmission electron microscopy (TEM) images, high-resolution transmission electron microscopy (HRTEM) images and element mapping results were performed on FEI F20 S-TWIN at an acceleration voltage of 200 kV. Thermal gravimetric analysis (TGA) was conducted in air at a heating rate of 10 °C min−1.
2.2 Electrochemical measurements
The coin-type cells were assembled in a glove box full of Ar atmosphere. The as-prepared active materials (70 wt%) was mixed with 20 wt% acetylene black and 10 wt% polyvinylidene difluoride (PVDF) in N-methyl-2-pyrrolidone, painted onto a copper foil, and subsequently dried at 110 °C in vacuum for 12 hours. The mass loading of the active material is around 1.2–1.6 mg per electrode slice. The specific capacity values of composite electrode materials are calculated on the basis of the total mass of MoO2 and carbon. The amount of active material in test cells was ca.1–2 mg. The material in the electrolyte was the LiPF6 (1 M) in a mixture of EC, EMC and DMC with a weight ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and the counter electrodes material were the metallic lithium foil. The material of the separator was the UP 3093 (Japan) microporous polypropylene membrane. Coin cells (2025-type) were assembled in an argon-filled glove box, and the quantity of oxygen and moisture less than 1 ppm. Cyclic voltammograms (CV) were carried out on CHI660C at a scanning rate of 0.5 mV s−1 between 0.01 and 3.0 V. Tests of constant galvanostatic charge–discharge curves and cycling performance were measured with Land CT 2001A electrochemical workstation. The current densities were between 100-2000 mA g−1, and the voltage range was between 0.01 and 3.0 V (vs. Li+/Li) at room temperature.
1, and the counter electrodes material were the metallic lithium foil. The material of the separator was the UP 3093 (Japan) microporous polypropylene membrane. Coin cells (2025-type) were assembled in an argon-filled glove box, and the quantity of oxygen and moisture less than 1 ppm. Cyclic voltammograms (CV) were carried out on CHI660C at a scanning rate of 0.5 mV s−1 between 0.01 and 3.0 V. Tests of constant galvanostatic charge–discharge curves and cycling performance were measured with Land CT 2001A electrochemical workstation. The current densities were between 100-2000 mA g−1, and the voltage range was between 0.01 and 3.0 V (vs. Li+/Li) at room temperature.
3 Results and discussion
The powder X-ray diffraction (XRD) patterns of the MoO2–C-1d and MoO2–C-2d sample synthesized for different times exhibits broad diffraction peaks. As shown in Fig. 1, there are the same diffraction peaks for the MoO2–C-1d and MoO2–C-2d sample. Moreover, there is no impurity peaks in the XRD patterns, and the figures also manifest that the materials are monoclinic MoO2 phases (JCPDS no. 073-1249).The morphologies and microstructures of the MoO2–C-1d and MoO2–C-2d samples were examined by SEM and TEM measurements. Fig. 2a presents the SEM image of MoO2–C-1d sample with this powder having a spherical shape and smooth surface. As shown in Fig. 2b, the MoO2–C-2d sample was composed of uniform spheres with rough surface, and the diameter of a sphere was about 0.8 μm. Furthermore, the broken sphere displayed the hierarchical structure with hollow interior.52,53 The TEM image disclosed that the MoO2–C-1d sample was consisted of solid spheres (Fig. 2c). Fig. 2d shows the TEM image of MoO2–C-2d sample, in which the sample exhibited an obvious hierarchical hollow spherical structure. It can be seen from Fig. 2c and d that the size of the MoO2–C-2d sample was bigger than that of MoO2–C-1d. A magnified TEM image of MoO2–C-2d sample was shown in Fig. 2e, in which the amorphous carbon shell surrounding many crystalline nanoparticles (size of ca. 30 nm) can be observed. The representative high-resolution TEM (HRTEM) image in Fig. 2f shows a lattice fringe in MoO2–C-2d sample. The interplanar spacing was about 0.343 nm, corresponding to the (011) plane of crystalline MoO2 phase.54 Therefore, we can deduce that the surface morphologies and textures of the MoO2–C composite samples can be tailored by varying the solvothermal reaction time. According to the result of thermogravimetric analysis (TGA), the carbon contents in the MoO2–C-1d and MoO2–C-2d samples were evaluated to be about 18.6 and 21.9 wt%, respectively. The porous features of samples are measured by nitrogen adsorption–desorption isotherm (Fig. S1†). It is found that the two samples have BET specific surface areas of 39.2 (MoO2–C-1d) and 102.1 (MoO2–C-2d) m2 g−1, respectively. The MoO2–C-1d sample is consisted of solid spheres; while the MoO2–C-2d sample shows hierarchical hollow structure, exhibiting higher specific surface area. The MoO2–C-2d shows a narrow pore size distribution, while MoO2–C-1d doesn't show the narrow pore size distribution due to its denser structure.
 | ||
| Fig. 2 SEM images of (a) MoO2–C-1d and (b) MoO2–C-2d. TEM images of (c) MoO2–C-1d and (d–f) MoO2–C-2d. | ||
The STEM images of the MoO2–C-2d composite and the corresponding elemental mapping images (Fig. 3a) indicate the homogeneous distribution of carbon over the entire selected MoO2–C-2d particles to form a hierarchical structure.55 The chemical composition of the MoO2–C-2d was measured by EDS, as depicted in Fig. 3b. It was also confirmed that the sample contained carbon, oxygen and molybdenum elements.
 | ||
| Fig. 3 (a) STEM image of the MoO2–C-2d sample and the corresponding elemental mapping results for C (red), O (medium yellow) and Mo (luminous yellow). (b) EDS spectrum obtained from MoO2–C-2d sample. | ||
The cyclic voltammetry (CV) tests were performed to clearly investigate the MoO2–C-1d and MoO2–C-2d electrodes reaction processes between 0.01 and 3.0 V at the scanning rate of 0.5 mV s−1 (Fig. 4). In the first discharge curve, there was an irreversible reduction peak at ca. 0.65 V, which may arise from the formation of solid electrolyte interphase (SEI) film, and then the peak disappeared in subsequent cycles.56 In the following cycles, two pairs of redox peaks at about 1.21/1.43 V and 1.48/1.73 V can be associated with the transformation between monoclinic MoO2 phase and orthorhombic phase of the fractionally lithiated LixMoO2 (0 < x < 1).57,58 In addition, in the voltage below 1 V, it was maybe connected with the transformation reaction from MoO2 to Mo. These electrochemical reaction processes of MoO2–C-1d and MoO2–C-2d electrode can be described as the following equations:59,60
| MoO2 + xLi+ + xe− ↔ LixMoO2 (0 < x < 1) | (1) |
| MoO2 + 4Li+ + 4e− ↔ Mo + 2Li2O | (2) |
 | ||
| Fig. 4 Selected CV curves of (a) MoO2–C-1d and (b) MoO2–C-2d electrodes at a scan rate of 0.5 mV s−1 in the voltage range of 0.01–3.0 V. | ||
As can be seen from Fig. 4, the integrated area and the peak current of the MoO2–C-1d electrode decreased more quickly than those of the MoO2–C-2d electrode, which suggested that the latter exhibited better electrochemical performance than that of former. This deduction can be further confirmed by the galvanostatic discharge and charge voltage profiles of these two samples (Fig. 5).
 | ||
| Fig. 5 The charge and discharge profiles of (a) MoO2–C-1d and (b) MoO2–C-2d electrodes at a current density of 1 A g−1 between 0.01 and 3.0 V. | ||
Fig. 5 depicts the charge and discharge profiles of MoO2–C-1d and MoO2–C-2d electrodes for the 1st, 2nd, 10th, 60th and 100th cycles at a current density of 1 A g−1 in the voltage range of 0.01–3 V. As depicted in Fig. 5a, the MoO2–C-1d electrode showed a gradual degradation of cycling capacity, which was common for conversion-type transition metal oxide electrodes.61 On the other hand, the MoO2–C-2d electrode revealed a original discharge capacity of 1118 mA hg−1 and the charge capacity of 820 mA h g−1, giving a coulombic efficiency of 73%, due to the formation of lithium solid electrolyte interface layer (SEI) as well as the irreversible reaction upon cycling process. In the initial discharge–charge profile of MoO2–C-2d electrode, there were two discharge voltage curves at 1.45 and 1.18 V and two charge ones at 1.42 and 1.71 V, which consistent with the CV results. These observed voltage plateaus were attributed to the phase transition process for the partial and gradual lithiation of the electrode from MoO2 to LixMoO2. The charge and discharge curves of 2nd and 100th cycle were high degree of overlap, suggesting that the electrode material of MoO2–C-2d has good electrochemical stability.62
Fig. 6 displays the cycling performance of the MoO2–C-1d and MoO2–C-2d electrodes at the current density of 0.2 A g−1 with the voltage range of 0.01–3.0 V (vs. Li+/Li). It can be found that the former showed better cycling performance than that of the latter. The high reversible capacity of 763 mA h g−1 was still delivered for MoO2–C-2d electrode after 50 cycles at 0.2 A g−1, which higher than that of MoO2–C-1d electrode (379 mA h g−1). Importantly, the coulombic efficiency of MoO2–C-2d electrode increased to more than 98% after 5 cycles. The specific discharge capacities of MoO2–C-2d electrode were 849, 832 and 763 mA h g−1 for the 5th, 10th and 50th cycles, which indicated that the hierarchical MoO2 hollow microspheres show a high reversible capacity and cycling stability.
 | ||
| Fig. 6 The cycling performance of MoO2–C-1d and MoO2–C-2d electrodes at a current density of 0.2 A g−1 between 0.01 and 3.0 V. | ||
Fig. 7 describes the rate capacity of the MoO2–C-1d and MoO2–C-2d electrodes at different current densities between 0.1 and 2 A g−1 for every ten successive cycles. The reversible specific capacities of MoO2–C-2d electrode were 839, 798, 740, 670 and 573 mA h g−1 at current densities of 0.1, 0.2, 0.5, 1 and 2 A g−1, respectively. Moreover, a large reversible capacity of 573 mA h g−1 can be maintained when the current density reached at the high current density of 2 A g−1, indicating that the MoO2–C-2d electrode possess an outstanding rate performance. Furthermore, even after being subjected to the high current charge–discharge, the MoO2–C-2d electrode was still able to provide a high capacity of 745 mA h g−1 at 0.2 A g−1. By comparison, the capacities of MoO2–C-1d electrode were merely 785, 600, 474, 394 and 305 mA h g−1 at the current densities of 0.1, 0.2, 0.5, 1 and 2 A g−1, respectively. It demonstrates the MoO2–C-2d (hierarchical MoO2–C hollow spheres) electrode showed the excellent cycling stability and high rate performance.
 | ||
| Fig. 7 The rate capability of MoO2–C-1d and MoO2–C-2d electrodes at different current densities between 0.1 and 2 A g−1. | ||
Fig. 8 shows the coulombic efficiency and cycling performance of MoO2–C-1d and MoO2–C-2d at the specific current density of 1 A g−1. It can be found that the MoO2–C-2d electrode presented better coulombic efficiency and cycling stability than those of MoO2–C-1d. The MoO2–C-1d showed fast capacity fading upon long-time cycling process. The coulombic efficiency of MoO2–C-2d electrode reached 98% after 5th cycles. The discharge specific capacities of MoO2–C-2d were 647, 631 and 560 mA h g−1 after 10th, 50th and 100th cycles, which illustrated that the MoO2–C-2d electrode exhibited great coulombic efficiency and good cycling stability.63
 | ||
| Fig. 8 The coulombic efficiency and cycling performance of MoO2–C-1d and MoO2–C-2d electrodes at the specific current density of 1 A g−1. | ||
To verify the stability of the electrode materials, the morphology and structure of MoO2–C-1d and MoO2–C-2d were characterized by SEM and TEM after 100 cycles at the current density of 1 A g−1 (Fig. 9). It can be seen from Fig. 9a and c that the MoO2–C-1d sample was obvious pulverization and agglomeration. However, the MoO2–C-2d sample maintained its hierarchically hollow structures without visible large range of aggregations (Fig. 9b and d). These behaviors confirmed the MoO2–C-2d sample possess the highly reversible structure stability during the lithium storage processes.
 | ||
| Fig. 9 (a, b) SEM and (c, d) TEM images of (a, c) MoO2–C-1d and (b, d) MoO2–C-2d after 100 cycles at the current density of 1 A g−1. | ||
Fig. S2a† displays the Nyquist profiles of MoO2–C-1d and MoO2–C-2d electrodes after 50 cycles from 1 MHz to 0.05 Hz in the fully charged state. In the low frequency regions, the short inclined line of the relationship between Zre and ω−1/2 is corresponding to the ion diffusion within the anode material. Obviously, the semicircle for MoO2–C-2d was much smaller than that of MoO2–C-1d, suggesting that the MoO2–C-2d has lower contact and charge transfer resistances. As depicted in Fig. S2b,† the MoO2–C-2d exhibited lower slope than MoO2–C-1d, indicating that the facile ion transportation of MoO2–C-2d electrode.
The excellent electrochemical performance of MoO2–C-2d electrode originates from the unique hierarchical architecture, as illustrated in Scheme 1. The hierarchical hollow structure could accommodate volume expansion and release the strain during cycling process. The carbon layers scattered around the MoO2 nanoparticles, which provide fast electron transport channel. Moreover, the small MoO2 nanoparticles will provide short path for Li+ diffusion. During the charge–discharge process, the obtained structure of active nanoparticles in an amorphous carbon matrix could efficiently prevent the nanoparticles aggregation, accommodate the volume change, and provide a stable conductive network for ion kinetics, which contributes to the superior rate performance and cycling stability. Therefore, all of these aspects are contributed to the large reversible capacity, good cycling stability and high rate capability of hierarchical MoO2–C hollow spheres.
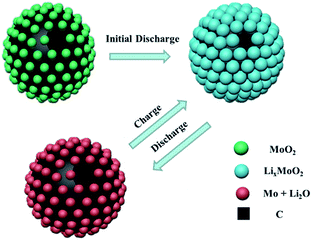 | ||
| Scheme 1 Schematic illustration of the excellent structure stability in hierarchical MoO2 hollow spheres during the electrochemical charge–discharge processes. | ||
4 Conclusions
In summary, we have successfully synthesized the hierarchical MoO2–C hollow spheres via an ethanol thermal reduction method followed by annealing process. The combined effect of hollow micro-spherical structure, carbon hybrid and MoO2 nanoparticles endows the materials with excellent electrochemical performances. With the design, the hierarchical MoO2–C hollow spheres exhibited a large reversible capacity, good cycling stability and high rate performance. For example, a large reversible capacity of 763 mA h g−1 can be reached at current density of 0.2 A g−1 after 50 cycles. In addition, a large reversible capacity of 573 mA h g−1 can be remained at the current density as high as 2 A g−1. The results provide an efficient and facile route to the design of a hierarchical electrode material for other energy storage applications.Acknowledgements
This work was financially supported by National Natural Science Foundation of China (NSFC 51502036, U1505241, 21407025 and 21307012), Natural Science Foundation of Fujian Province (2016J05116 and 2014J01035) and Educational Commission of Fujian Province (JA15115).Notes and references
- S. Srivastava, A. Santos, K. Critchley, K. S. Kim, P. Podsiadlo, K. Sun, J. Lee, C. Xu, G. D. Lilly, S. C. Glotzer and N. A. Kotov, Science, 2010, 327, 1355–1359 CrossRef CAS PubMed.
- A. Magasinki, P. Dixon, B. Hertzberg, A. Kvit, J. Ayala and G. Yushin, Nat. Mater., 2010, 9, 353–358 CrossRef PubMed.
- M. V. Reddy, G. V. Subba Rao and B. V. R. Chowdari, Chem. Rev., 2013, 113, 5364–5457 CrossRef CAS PubMed.
- H. Li, Z. X. Wang, L. Q. Chen and X. J. Huang, Adv. Mater., 2009, 21, 4593–4607 CrossRef CAS.
- C. Z. Wu, F. Feng and Y. Xie, Chem. Soc. Rev., 2013, 42, 5157–5183 RSC.
- Y. L. Liang, Z. L. Tao and J. Chen, Adv. Energy Mater., 2012, 2, 742–769 CrossRef CAS.
- Y. G. Wang, H. Q. Li, P. He, E. Hosono and H. S. Zhou, Nanoscale, 2010, 2, 1294–1305 RSC.
- G. L. Cui, L. Gu, L. J. Zhi, N. Kaskhedikar, P. A. van Aken, K. Mullen and J. Maier, Adv. Mater., 2008, 20, 3079–3083 CrossRef CAS.
- D. J. Xue, S. Xin, Y. Yan, K. C. Jiang, Y. X. Yin, Y. G. Guo and L. J. Wan, J. Am. Chem. Soc., 2012, 134, 2512–2515 CrossRef CAS PubMed.
- L. Q. Mai, X. C. Tian, X. Xu, L. Chang and L. Xu, Chem. Rev., 2014, 114, 11828–11862 CrossRef CAS PubMed.
- Y. M. Chen, Z. Li and X. W. Lou, Angew. Chem., Int. Ed., 2015, 54, 10521–10524 CrossRef CAS PubMed.
- H. Hu, L. Yu, X. H. Gao, Z. Lin and X. W. Lou, Energy Environ. Sci., 2015, 8, 1480–1483 CAS.
- J. G. Cai and L. M. Qi, Mater. Horiz., 2015, 2, 37–53 RSC.
- S. Chen, Y. L. Xin, Y. Y. Zhou, F. Zhang, Y. R. Ma, H. H. Zhou and L. M. Qi, J. Mater. Chem. A, 2014, 2, 15582–15589 CAS.
- L. Q. Mai, B. Hu, W. Chen, Y. Y. Qi, C. S. Lao, R. S. Yang, Y. Dai and Z. L. Wang, Adv. Mater., 2007, 19, 3712–3716 CrossRef CAS.
- L. X. Zeng, C. Zheng, L. C. Xia, Y. X. Wang and M. D. Wei, J. Mater. Chem. A, 2013, 1, 4293–4299 CAS.
- X. L. Hu, W. Zhang, X. X. Liu, Y. N. Mei and Y. H. Huang, Chem. Soc. Rev., 2015, 44, 2376–2404 RSC.
- X. L. Ji, P. S. Herle, Y. Rho and L. F. Nazar, Chem. Mater., 2007, 19, 374–383 CrossRef CAS.
- L. C. Yang, Q. S. Gao, Y. H. Zhang, Y. Tang and Y. P. Wu, Electrochem. Commun., 2008, 10, 118–122 CrossRef CAS.
- J. F. Ni, Y. Zhao, L. Li and L. Q. Mai, Nano Energy, 2015, 11, 129–135 CrossRef CAS.
- H. J. Zhang, K. X. Wang, X. Y. Wu, Y. M. Jiang, Y. B. Zhai, C. Wang, X. Wei and J. S. Chen, Adv. Funct. Mater., 2014, 24, 3399–3404 CrossRef CAS.
- Y. N. Ko, S. B. Park, K. Y. Jung and Y. C. Kang, Nano Lett., 2013, 13, 5462–5466 CrossRef CAS PubMed.
- Y. M. Sun, X. L. Hu, J. C. Yu, Q. Li, W. Luo, L. X. Yuan, W. X. Zhang and Y. H. Huang, Energy Environ. Sci., 2011, 4, 2870–2877 CAS.
- D. Zhao, J. W. Qin, L. R. Zheng and M. H. Cao, Chem. Mater., 2016, 28, 4180–4190 CrossRef CAS.
- Y. F. Shi, B. K. Guo, S. A. Corr, Q. H. Shi, Y. S. Heier, K. R. Hu, L. Q. Chen, R. Seshadri and G. D. Stucky, Nano Lett., 2009, 9, 4215–4220 CrossRef CAS PubMed.
- L. Zhou, H. B. Wu, Z. Y. Wang and X. W. Lou, ACS Appl. Mater. Interfaces, 2011, 3, 4853–4857 CAS.
- Q. S. Gao, L. C. Yang, X. C. Lu, J. J. Mao, Y. H. Zhang, Y. P. Wu and Y. Tang, J. Mater. Chem., 2010, 20, 2807–2812 RSC.
- Q. W. Tang, Z. Q. Shan, L. Wang and X. Qin, Electrochim. Acta, 2012, 79, 148–153 CrossRef CAS.
- L. C. Yang, L. L. Liu, Y. S. Zhu, X. J. Wang and Y. P. Wu, J. Mater. Chem., 2012, 22, 13148–13152 RSC.
- Z. Y. Wang, S. Madhavi and X. W. Lou, J. Phys. Chem. C, 2012, 116, 12508–12513 CAS.
- X. P. Fang, B. K. Guo, Y. F. Shi, B. Li, C. X. Hua, C. H. Yao, Y. C. Zhang, Y. S. Hu, Z. X. Wang, G. D. Stucky and L. Q. Chen, Nanoscale, 2012, 4, 1541–1544 RSC.
- M. Ihsan, H. Q. Wang, S. R. Majid, J. P. Yang, S. J. Kennedy, Z. P. Guo and H. K. Liu, Carbon, 2016, 96, 1200–1207 CrossRef CAS.
- Y. P. Zhu, X. M. Xu, G. Chen, Y. J. Zhong, R. Cai, L. Li and Z. P. Shao, Electrochim. Acta, 2016, 211, 972–981 CrossRef CAS.
- X. Deng, B. T. Zhao, Y. J. Zhong, Y. P. Zhu and Z. P. Shao, J. Mater. Chem. A, 2016, 4, 10403–10408 CAS.
- J. B. Zhou, N. Lin, L. B. Wang, K. L. Zhang, Y. C. Zhu and Y. T. Qian, J. Mater. Chem. A, 2015, 3, 7463–7468 CAS.
- G. Z. Yang, S. Y. Li, M. M. Wu and C. X. Wang, J. Mater. Chem. A, 2016, 4, 10974–10985 CAS.
- G. X. Gao, S. Y. Lu, B. T. Dong, Y. Xiang, K. Xi and S. J. Ding, J. Mater. Chem. A, 2016, 4, 6264–6270 CAS.
- Q. H. Tian, Z. X. Zhang, L. Yang and S.-I. Hirano, Carbon, 2015, 93, 887–895 CrossRef CAS.
- F. K. Butt, C. B. Cao, R. Ahmed, W. S. Khan, T. Cao, N. Bidin, P. Li, Q. Wan, X. H. Qu, M. Tahir and F. Idrees, CrystEngComm, 2014, 16, 894–899 RSC.
- L. X. Zeng, X. X. Huang, C. Zheng, Q. R. Qian, Q. H. Chen and M. D. Wei, Dalton Trans., 2015, 44, 7967–7972 RSC.
- C. Zheng, L. X. Zeng, M. L. Wang, H. Y. Zheng and M. D. Wei, CrystEngComm, 2014, 16, 10309–10313 RSC.
- Y. M. Sun, X. L. Hu, W. Luo and Y. H. Huang, ACS Nano, 2011, 5, 7100–7107 CrossRef CAS PubMed.
- H. J. Zhang, J. Shu, K. X. Wang, X. T. Chen, Y. M. Jiang, X. Wei and J. S. Chen, J. Mater. Chem. A, 2014, 2, 80–86 CAS.
- Y. P. Zhu, S. F. Wang, Y. J. Zhong, R. Cai, L. Li and Z. P. Shao, J. Power Sources, 2016, 307, 552–560 CrossRef CAS.
- Y. L. Liu, H. Zhang, P. Ouyang, W. H. Chen, Y. Wang and Z. C. Li, J. Mater. Chem. A, 2014, 2, 4714–4721 CAS.
- K. H. Seng, G. D. Du, L. Li, Z. X. Chen, H. K. Liu and Z. P. Guo, J. Mater. Chem., 2012, 22, 16072–16077 RSC.
- L. X. Zeng, C. Zheng, C. L. Deng, X. K. Ding and M. D. Wei, ACS Appl. Mater. Interfaces, 2013, 5, 2182–2187 CAS.
- G. Z. Yang, H. Cui, G. W. Yang and C. X. Wang, ACS Nano, 2014, 8, 4474–4487 CrossRef CAS PubMed.
- S. Hu, F. Yin, E. Uchaker, W. Chen, M. Zhang, J. Zhou, Y. Y. Qi and G. Z. Cao, J. Phys. Chem. C, 2014, 118, 24890–24897 CAS.
- B. Liu, X. Y. Zhao, Y. Xiao and M. H. Cao, J. Mater. Chem. A, 2014, 2, 3338–3343 CAS.
- Y. R. Liu, J. Bai, X. J. Ma, J. F. Li and S. L. Xiong, J. Mater. Chem. A, 2014, 2, 14236–14244 CAS.
- X. L. Liu, D. Wu, W. X. Ji and W. H. Hou, J. Mater. Chem. A, 2015, 3, 968–972 CAS.
- X. L. Liu, W. X. Ji, J. Y. Liang, L. M. Peng and W. H. Hou, Phys. Chem. Chem. Phys., 2014, 16, 20570–20577 RSC.
- S. S. Tang, B. Ouyang, L. Y. Yang and W. H. Ji, RSC Adv., 2015, 5, 50705–50710 RSC.
- X. F. Zhang, X. X. Song, S. Gao, Y. M. Xu, X. L. Cheng, H. Zhao and L. H. Huo, J. Mater. Chem. A, 2013, 1, 6858–6864 CAS.
- L. X. Zeng, C. L. Deng, C. Zheng, H. Y. Qiu, Q. R. Qian, Q. H. Chen and M. D. Wei, Mater. Res. Bull., 2015, 71, 42–47 CrossRef CAS.
- Z. Y. Wang, J. S. Chen, T. Zhu, S. Madhavi and X. W. Lou, Chem. Commun., 2010, 46, 6906–6908 RSC.
- Y. Wang, Z. G. Huang and Y. J. Wang, J. Mater. Chem. A, 2015, 3, 21314–21320 CAS.
- L. C. Yang, W. Sun, Z. W. Zhong, J. W. Liu, Q. S. Gao, R. Z. Hu and M. Zhu, J. Power Sources, 2016, 306, 78–84 CrossRef CAS.
- K. Palanisamy, Y. Kim, H. Kim, J. M. Kim and W. S. Yoon, J. Power Sources, 2015, 275, 351–361 CrossRef CAS.
- L. X. Zeng, X. X. Huang, X. Chen, C. Zheng, Q. R. Qian, Q. H. Chen and M. D. Wei, ACS Appl. Mater. Interfaces, 2016, 8, 232–239 CAS.
- D. Y. Park, Y. K. Sun and S. T. Myung, J. Power Sources, 2015, 280, 1–4 CrossRef CAS.
- L. Guo and Y. Wang, J. Mater. Chem. A, 2015, 3, 4706–4715 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra22792a |
| This journal is © The Royal Society of Chemistry 2016 |

