Magnetic solid-phase extraction coupled with HPLC-Q-TOF-MS for rapid analysis of tyrosinase binders from San-Bai decoction by Box–Behnken statistical design†
Abstract
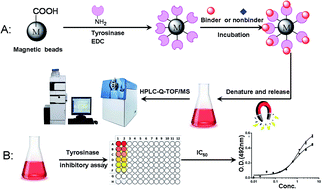
Tyrosinase is the rate-limiting enzyme for controlling the production of melanin. A very precise, sensitive and convenient solid-phase extraction based approach was developed for extract and analysis of tyrosinase binders from San-Bai decoction. Tyrosinase binders were captured by using tyrosinase conjugated magnetic beads. The retained tyrosinase binders were dissociated and analyzed by high performance liquid chromatography and quadrupole-time-of-flight mass spectrometry. The powder X-ray diffraction (XRD), transmission electron microscopy (TEM), vibration sample magnetometer (VSM) and Fourier transform infrared (FT-IR) spectroscopy techniques were used for the characterization of tyrosinase conjugated magnetic beads. A variety of experimental conditions such as incubation temperature, buffer pH and ion strength, which may affect extraction yield, were optimized through Box–Behnken statistical design and response surface methodology by using paeoniflorin as a model compound. The optimal incubation conditions were as follows: wash times: 4, wash solvent: 10% acetonitrile–water, incubation time: 30 min, temperature: 34.84 °C, ion strength: 193.67 mM, pH: 6.98. The established approach was successfully applied for the extraction of tyrosinase binders from San-Bai decoction. The tyrosinase inhibitory assay was employed for validation of the activities of these identified binders. Fifteen tyrosinase inhibitors were identified, and nine of which, including benzoylpaeoniflorin, benzoyloxypaeoniflorin, galloylpaeoniflorin, paeonolide, oxypaeoniflora, albiflorin, ononin, mudanpioside C and apiopaeonoside, were reported for the first time.

 Please wait while we load your content...
Please wait while we load your content...