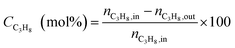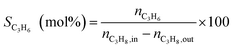N-, P- and B-doped mesoporous carbons for direct dehydrogenation of propane†
Yuejun Songab,
Guishu Liuab and
Zhong-Yong Yuan*ab
aNational Institute for Advanced Materials, School of Materials Science and Engineering, Nankai University, Tongyan Road 38, Haihe Educational Park, Tianjin 300353, China. E-mail: zyyuan@nankai.edu.cn
bKey Laboratory of Advanced Energy Materials Chemistry (Ministry of Education), Collaborative Innovation Center of Chemical Science and Engineering (Tianjin), Nankai University, Weijin Road 94, Tianjin 300071, China
First published on 19th September 2016
Abstract
Mesoporous carbons doped with nitrogen, phosphorous and boron are prepared by a post-treating method or in situ approach. All the samples present a well-defined mesoporous structure and the dopants distribute uniformly with special chemical structures. In a direct propane dehydrogenation reaction, phosphorous- and boron-doped mesoporous carbons (PMC, BMC) exhibit improved propane conversion and propene selectivity, in comparison with undoped mesoporous carbon (MC). Nitrogen-doped mesoporous carbons (NMC) show decreased activity, although having higher surface area and larger pore volume than MC, PMC and BMC. Through comparative analysis of the chemical structures and oxygen-containing groups in these carbon materials, it is found that the amounts of carbonyl and/or quinone groups of the carbons play a pivotal role in direct dehydrogenation of propane. Doping with heteroatoms affects the compositions and contents of the oxygen-containing groups, and then ultimately the catalytic ability of the carbon catalysts.
1. Introduction
Propene, as an important chemical feedstock, is widely used to produce polymer and rubber. Traditionally, steam cracking and fluid catalytic cracking (FCC) of naphtha, liquid petroleum gas and heavier oil fractions are the main methods to obtain propene.1,2 Another potential way is the direct or oxidative dehydrogenation of propane to propene due to high conversion and selectivity.3–5 However, oxidative dehydrogenation of propane has not been commercialized so far, due to high equipmental and technical requirements, as well as oxidation by oxygen, although dehydrogenation is achieved at low reaction temperature.6,7 At present, the main commercial catalysts for direct dehydrogenation of propane are CrOx and Pt–Sn, adopting the Catofin (Lummus) and the Oleflex (UOP) technologies, respectively.1,8 However, these two catalysts are very hazardous for human health or expensive to utilize extensively.Recently, carbon materials have attracted increasing attention due to their low cost, high stability and high electronic conductivity and have been applied as adsorbents, catalysts, catalyst supports, and electrode materials.9–12 Doping carbons with non-metal atoms, such as nitrogen (N), phosphorus (P) and boron (B), can introduce defect sites on the surface, break the electroneutrality of host carbon framework, and further improve the chemical performance.13–15 For example, N-doped mesoporous carbons not only exhibit a significant CO2 adsorption capacity and selectivity,10 but also are potential a counter electrode candidate in dye-sensitized solar cells (DSSC).16 P- and B-doped mesoporous carbons display potential in oxygen reduction reaction (ORR) and supercapacitors with superior electrochemical performance.17,18
Indeed, metal-free carbonaceous materials have been regarded as promising heterogeneous catalysts.19 Carbon materials including active carbons, carbon nanotubes (CNTs), ordered mesoporous carbons (OMCs), carbon onion and nanodiamond, have displayed great efficiencies in oxidative dehydrogenation of ethylbenzene and direct dehydrogenation of alkane.20–26 Several non-metal heteroatom-doped carbon materials have also been studied on alkane dehydrogenation to further improve the catalytic activity. For instance, carbon catalysts doped with N, which has similar atomic radius as C and provides an n-type electronic modification to the carbon framework,27 improved the catalytic activity effectively by speeding up the dissociative adsorption of O2 molecules and decreasing the activation energy of oxidative dehydrogenation.28,29 P- or B-doped CNTs exhibited higher selectivity due to suppression of the formation of electrophilic oxygen species by P or B reacting with CNT sidewall defects.30 For direct dehydrogenation, non-metal-doped carbon materials also exhibited interesting activity. Zhao and co-workers improved the conversion of direct dehydrogenation of ethylbenzene to styrene by N-doped CNTs.31–33 P-doped mesoporous carbons synthesized by the hard-templating approach were also used to directly dehydrogenize propane.34 However, the influence of the doped non-metal heteroatoms on the surface active groups over carbon materials in direct dehydrogenation of light hydrocarbons is still not totally clear.
In this study, N-, P- and B-doped mesoporous carbon catalysts were synthesized by post-treatment or in situ soft-templating approach and their catalytic performances were evaluated by propane dehydrogenation. The effects of the nature of the heteroatoms on the catalytic activity of the doped mesoporous carbons in the direct dehydrogenation of propane were discussed and analyzed in detail by a series of characterizations.
2. Experimental
2.1 Materials
Resorcinol was purchased from Tianjin Yuanli Chemical Co., Ltd. F127 (MW = 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) was obtained from Nanjing Will Chemical Co., Ltd. 1-Hydroxyethylidene-1,1-diphosphonic acid (HEDP, 50%) was received from Shandong Taihe Chemical Co., Ltd. Boric acid, formaldehyde (37%) and HCl (37%) were provided by Tianjin Guangfu Fine Chemical Research Institute Co., Ltd. All chemicals were used without any further purification.
000) was obtained from Nanjing Will Chemical Co., Ltd. 1-Hydroxyethylidene-1,1-diphosphonic acid (HEDP, 50%) was received from Shandong Taihe Chemical Co., Ltd. Boric acid, formaldehyde (37%) and HCl (37%) were provided by Tianjin Guangfu Fine Chemical Research Institute Co., Ltd. All chemicals were used without any further purification.
2.2 Catalyst preparation
Mesoporous carbons (MC) were prepared as per previous reports.35 N-doped mesoporous carbons (NMC) were prepared by a post-treating method with NH3.36 In a typical procedure, 1 g of MC was heated up to 800 °C at 5 °C min−1 under a flow rate of 50 cm3 min−1 NH3 atmosphere and held for 2 h.P-doped mesoporous carbons (PMC) were prepared using HEDP as a phosphorus source.17 15 mmol of resorcinol and 0.2 mmol of F127 were dissolved in 40 ml mixture of ethanol and water (vethanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) vwater = 1
vwater = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), followed by the addition of 7.5 mmol of HEDP (50%). After 1 h of stirring, 30 mmol of formaldehyde (37%) was added dropwise to the abovementioned solution. After another 1 h of tempestuous stirring, the transparent mixture was transferred to a Teflon-lined autoclave and heated at 60 °C for 3 days. The obtained orange polymeric monoliths were washed with water, dried at 60 °C, carbonized to 600 °C at 1 °C min−1 for 3 h under the protection of nitrogen. B-doped mesoporous carbons (BMC) were prepared in the same way except that boric acid (7.5 mmol) was used as the boron source together with 2 mmol of HCl (37%).
1), followed by the addition of 7.5 mmol of HEDP (50%). After 1 h of stirring, 30 mmol of formaldehyde (37%) was added dropwise to the abovementioned solution. After another 1 h of tempestuous stirring, the transparent mixture was transferred to a Teflon-lined autoclave and heated at 60 °C for 3 days. The obtained orange polymeric monoliths were washed with water, dried at 60 °C, carbonized to 600 °C at 1 °C min−1 for 3 h under the protection of nitrogen. B-doped mesoporous carbons (BMC) were prepared in the same way except that boric acid (7.5 mmol) was used as the boron source together with 2 mmol of HCl (37%).
2.3 Catalyst characterization
Nitrogen adsorption–desorption isotherms were recorded on a Quantachrome NOVA 2000e sorption analyzer at −196 °C. Prior to the measurement, the samples were degassed in vacuum at 200 °C for 8 h. The specific surface areas (SBET) were estimated using the Brunauer–Emmett–Teller (BET) method, the pore size distribution curves were calculated by the Barrett–Joyner–Halenda (BJH) method from the adsorption branches of isotherms, and the total pore volume values (Vtotal) were derived from the adsorbed amount at a relative pressure (P/P0) of 0.98. Scanning electron microscopy (SEM) was conducted on a Jeol JSF-7500L microscope at 5 kV. X-ray photoelectron spectroscopy (XPS) was performed on a Kratos Axis Ultra DLD (delay line detector) spectrometer, equipped with a monochromatic Al Kα X-ray source (1486.6 eV). The binding energies (BE) were referenced to the C 1s peak of contaminant carbon at 284.6 eV. Raman spectra were obtained on a Thermo-Fisher Scientific DXR spectrometer equipped with 514 nm wavelength incident laser light. The temperature-programmed desorption (TPD) profiles were operated on a Quantachrome ChemBET 3000 analyzer equipped with a U-shaped tubular micro-reactor placed inside an electrical furnace. The volumetric flow rate of the helium carrier gas and the heating rate of the furnace were controlled with 20 mL min−1 and 10 °C min−1, respectively.2.4 Catalytic activity measurements
Catalytic performances were tested in a fixed-bed quartz tubular microreactor at atmospheric pressure. The catalyst load was 200 mg and the total flow rate of gas reactant was 20 cm3 min−1 containing 5 vol% propane and a balance of nitrogen. The reaction temperature was maintained constant at 600 °C and the products were analyzed on an on-line gas chromatograph equipped with a 6 m column of Porapak Q and a flame ionization detector (FID). Propane conversion and propene selectivity were calculated as follows:3. Results and discussion
N, P and B are the most prevailing non-metal elements as doping atoms to improve the properties of carbon materials. In this study, all the mesoporous carbons were prepared under acidic conditions. Under the acidic system, initially, HEDP and boric acid could participate in self-assembly by combined action of coulombic interactions and hydrogen-bonding interaction, and then PMC and BMC are formed after surfactant template removal and subsequent carbonization; NMC was synthesized by post-treating way, i.e., heat treatment of MC under NH3 flowing at 800 °C. The pore structures of all the heteroatom-doped carbons were characterized by N2 adsorption–desorption analysis. The isotherms of all the samples are of type IV with a clear H1 hysteresis loop at P/P0 = 0.4–0.7 (Fig. 1a), indicating that the well-defined mesoporous characters were maintained after doping.37 The pore sizes, which were calculated using the BJH method from the adsorption branches, are similar (∼6 nm) after doping a heteroatom (Fig. 1b). Compared with MC, both the surface area and pore volume of NMC are enlarged (Table 1) due to etching of carbon fragments by radicals produced from the decomposition of NH3 at high temperature38 and the degradation of some unstable groups under elevated preparation temperature (800 °C). However, the specific surface areas and the total pore volumes of both PMC and BMC decreased in comparison to MC. NMC exhibited the highest surface area and the largest pore volume. | ||
| Fig. 1 N2 sorption isotherms (a) and the corresponding pore size distribution curves (b) of the carbon catalysts. | ||
| Samples | SBET (m2 g−1) | Vtotal (cm3 g−1) | Dpore (nm) | TOFb (mmol g−1 h−1) | Initial conversion (%) | Initial selectivity (%) |
|---|---|---|---|---|---|---|
| a Data in the parentheses correspond to the used catalysts after 24 h of propane dehydrogenation reaction.b Data were obtained after reaction for 2 h depicted as propene formation. | ||||||
| NMC | 908 (750)a | 0.91 | 5.8 | 41.8 | 18.6 | 84.6 |
| PMC | 679 (538) | 0.74 | 6.2 | 68.8 | 37.1 | 89.2 |
| BMC | 690 (571) | 0.68 | 6.2 | 67.1 | 33.9 | 87.2 |
| MC | 736 (541) | 0.80 | 5.4 | 59.8 | 30.9 | 87.6 |
Energy dispersive X-ray spectroscopy (EDS) elemental mapping images (Fig. 2) clearly reveal the uniform distribution of N, P and B throughout NMC, PMC and BMC, respectively, suggesting that the heteroatoms are perfectly doped into the carbon catalysts either by post-treating or an in situ process. Raman spectroscopy was also used to characterize the structural properties of carbon materials (Fig. 3). All the samples display a typical D band (∼1349 cm−1) and G band (∼1591 cm−1), which represent defective structure lattice with A1g symmetry and ideal graphitic lattice with E2g symmetry, respectively.39,40 The peak area ratios (ID/IG) of NMC and PMC are higher than BMC, implying higher degree of disorder. Shift in D band (1340 cm−1) and G band (1603 cm−1) of NMC suggest imperfect structure with further N doping. The defective degree would increase active sites to facilitate catalytic activity. However, BMC presents lower value of ID/IG than MC, suggesting that boron is helpful to increase the degree of graphitization, which may accelerate the electron transfer ability.
Moreover, the surface chemical structures were detected by XPS and the doped-atom peaks were also fitted to investigate their chemical environment. The characteristic peaks of heteroatoms in XPS spectra illustrate that the three different heteroatoms are successfully doped into the carbon skeleton. As shown in Fig. 4, N 1s spectra (Fig. 4b) are deconvoluted into pyridinic N, pyrrolic N, graphene N and oxide N, ascribing to binding energies of 398.5 eV, 400.4 eV, 401.8 eV and 405.5 eV, respectively.36,41 P 2p spectra (Fig. 4c) can be fitted to two peaks: one peak at 132.9 eV is ascribed to P–C bonds in aromatic C ring system, and the other peak at 134.3 eV is due to P–O structure of pentavalent tetra-coordinated phosphorous (PO4 tetrahedra).30,42,43 In the B 1s spectra (Fig. 4d), the peak at 200.3 eV refers to the B atoms replaced in C framework, whereas the one at 192.5 eV is assigned to (BO3)n chain bonded to the zig-zag face of the graphite lattice.30,44 Moreover, the O 1s spectra of all the catalysts were also detected and deconvoluted into three different chemical structures (Fig. 4a), i.e., unsaturated double bonded oxygen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) (∼531.1 eV), O
O) (∼531.1 eV), O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–O in esters and anhydrides (∼532.7 eV), C–OH or C–O in anhydrides (∼534.1 eV), and adsorbed water (∼535.7 eV).45 It is observed that oxygen-containing precursors (HEDP and boric acid) maintain relatively high oxygen contents in PMC and BMC, and the contents of C
C–O in esters and anhydrides (∼532.7 eV), C–OH or C–O in anhydrides (∼534.1 eV), and adsorbed water (∼535.7 eV).45 It is observed that oxygen-containing precursors (HEDP and boric acid) maintain relatively high oxygen contents in PMC and BMC, and the contents of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups are also promoted after P- and B-doping (Table 2). In contrast, low total oxygen contents in NMC may be mainly caused by the reaction of NH3 with some oxygen-containing groups on the NMC surface,38 which also resulted in higher N contents (5.98 at%) than P and B (0.31 at% and 0.20 at% in PMC and BMC, respectively, Table S2†).
O groups are also promoted after P- and B-doping (Table 2). In contrast, low total oxygen contents in NMC may be mainly caused by the reaction of NH3 with some oxygen-containing groups on the NMC surface,38 which also resulted in higher N contents (5.98 at%) than P and B (0.31 at% and 0.20 at% in PMC and BMC, respectively, Table S2†).
 | ||
| Fig. 4 High-resolution XPS spectra of O 1s (a) in all the samples, N 1s (b) in NMC, P 2p (c) in PMC and B 1s (d) in BMC. | ||
| Cat. | Total (at%) | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–O C–O |
C–OH/C–O | Water | ||||
|---|---|---|---|---|---|---|---|---|---|
| (eV) | (at%) | (eV) | (at%) | (eV) | (at%) | (eV) | (at%) | ||
| NMC | 4.92 | 531.1 | 0.48 | 532.7 | 2.99 | 534.1 | 1.11 | 535.7 | 0.34 |
| PMC | 7.19 | 531.5 | 1.68 | 532.5 | 1.50 | 533.7 | 3.34 | 535.3 | 0.66 |
| BMC | 9.89 | 531.4 | 1.29 | 532.6 | 3.46 | 533.8 | 4.70 | 535.6 | 0.44 |
| MC | 10.33 | 531.2 | 0.95 | 532.4 | 2.86 | 533.7 | 6.21 | 535.8 | 0.31 |
Mesoporous carbons after doping with heteroatoms perform different catalytic activity for propane dehydrogenation. Therefore, the catalytic performances of all the samples were measured on a fix-bed microreactor for direct propane dehydrogenation. As shown in Fig. 5, PMC and BMC exhibit higher initial propane conversion (37.1% and 33.9%, respectively) than MC (30.9%), and PMC also shows the highest initial selectivity to propene (89.2%). However, NMC with the highest surface area presents the lowest initial propane conversion and propene selectivity (18.6% and 84.6%, respectively), even inferior to the undoped MC. Furthermore, turnover frequency (TOF) for the amount of propene produced per hour is also considered and presented in Table 1. Likewise, the TOF values of PMC and BMC are 68.81 mmol g−1 h−1 and 67.12 mmol g−1 h−1, respectively, which are higher than that of MC (59.81 mmol g−1 h−1), while NMC displays TOF of 41.83 mmol g−1 h−1. However, all the catalysts showed reduced catalytic behavior with the time on stream. After 20 h time on stream, the activities are more or less similar for all the catalysts, which may be attributed to the coke formation on the surface during the reaction,23,37 accompanying with the decrease of specific surface areas (Table 1). Previous reports23,46,47 have suggested that carbonyl or quinone groups in carbon catalysts are the active site for propane dehydrogenation. From the XPS results, it is found that C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contents in PMC and BMC (Table 2) are indeed higher than those of others, explaining their higher initial conversion reasonably. One may believe that N, as an electron-rich atom, should perform similar behavior to P atom. However, the present result is inconsistent with the previous reports that N would improve the catalytic activity of alkane dehydrogenation.28,48 It is speculated that the C
O contents in PMC and BMC (Table 2) are indeed higher than those of others, explaining their higher initial conversion reasonably. One may believe that N, as an electron-rich atom, should perform similar behavior to P atom. However, the present result is inconsistent with the previous reports that N would improve the catalytic activity of alkane dehydrogenation.28,48 It is speculated that the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contents in NMC decreased so much that the doped N atom could not compensate enough electron for the rest C
O contents in NMC decreased so much that the doped N atom could not compensate enough electron for the rest C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contents.
O contents.
 | ||
| Fig. 5 Propane conversion and propene selectivity in propane dehydrogenation over all the catalysts. | ||
In order to further verify the assumption proposed above, three other N-doped mesoporous carbons, NMC-o, NMC-m and NMC-u, were synthesized by different methods to increase the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contents (see ESI†). NMC-o were prepared by oxidation of NMC with nitric acid aiming at increasing the oxygen-containing groups in carbon materials.26 NMC-m and NMC-u were synthesized by doping with melamine32 and urea,49 respectively. It is reported that during doping N with melamine, melamine can form C3N4 initially to protect some oxygen-containing groups from decomposition. NMC-u were prepared by evaporation-induced self-assembly (EISA) and carbonized under the same condition as MC. By N2 adsorption–desorption measurements, NMC-o, NMC-m and NMC-u have favorable mesoporous character (Fig. S1†), which would rule out the influence from the structure. The increase in oxygen-containing groups is indeed observed as expected (Table S2†) and the initial propane conversion of these three N-doped mesoporous carbons also enhanced with it (Table S1 and Fig. S3†), although it is still lower than that of PMC, BMC and MC. In particular, NMC-m has total oxygen atom percent equal to NMC but higher C
O contents (see ESI†). NMC-o were prepared by oxidation of NMC with nitric acid aiming at increasing the oxygen-containing groups in carbon materials.26 NMC-m and NMC-u were synthesized by doping with melamine32 and urea,49 respectively. It is reported that during doping N with melamine, melamine can form C3N4 initially to protect some oxygen-containing groups from decomposition. NMC-u were prepared by evaporation-induced self-assembly (EISA) and carbonized under the same condition as MC. By N2 adsorption–desorption measurements, NMC-o, NMC-m and NMC-u have favorable mesoporous character (Fig. S1†), which would rule out the influence from the structure. The increase in oxygen-containing groups is indeed observed as expected (Table S2†) and the initial propane conversion of these three N-doped mesoporous carbons also enhanced with it (Table S1 and Fig. S3†), although it is still lower than that of PMC, BMC and MC. In particular, NMC-m has total oxygen atom percent equal to NMC but higher C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O proportion, resulting in higher propane conversion than the latter, which suggests that C
O proportion, resulting in higher propane conversion than the latter, which suggests that C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O contents play a key role in this reaction. The drastic inactivity for NMC-u should be contributed to the collapse of structure (Fig. S2†). Therefore, it is critical to prepare a stable mesoporous structure with high oxygen-containing groups of N-doped carbon materials for direct propane dehydrogenation.
O contents play a key role in this reaction. The drastic inactivity for NMC-u should be contributed to the collapse of structure (Fig. S2†). Therefore, it is critical to prepare a stable mesoporous structure with high oxygen-containing groups of N-doped carbon materials for direct propane dehydrogenation.
The oxygen-containing groups of all the samples can also be defined further through temperature-programmed desorption (TPD) methods. Usually, carboxylic acid and lactones decompose at low temperature (<627 °C), whereas carbonyls (and quinones), hydroxyl groups and ether-type oxygen decompose above 627 °C.37,50 From Fig. 6, it is observed that an obvious peak appeared at around 766 °C for PMC, BMC and MC, illustrating the presence of carbonyls, hydroxyl groups and ether-type oxygen. In contrast, NMC exhibits low signal intensity due to low oxygen-containing group contents. Similar results can also be found in NMC-o, NMC-m and NMC-u (Fig. S5†). These results are also consistent with the data from XPS.
Therefore, carbonyl or quinone groups in carbon materials are regarded as the active sites for a dehydrogenation reaction in many works. The catalytic activity is usually improved by increasing the electronic density of carbonyl or quinone groups with doping electron-rich heteroatoms. However, the mesoporous carbons doped with N did not improve the activity of direct propane dehydrogenation. The reasons may be contributed to (1) the preparation condition of N-doping is harsh. N–H groups are too susceptible to maintain a good mesoporous structure at high temperature by in situ synthesis method, whereas the oxygen-containing groups usually decompose or react with a nitrogen source under a post-treatment approach; (2) reaction mechanisms are different from the direct dehydrogenation to the oxidative dehydrogenation of propane. In direct dehydrogenation, hydrogen atoms are abstracted from C–H bonds of alkanes to the corresponding alkenes by Lewis basic groups (such as, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups). However, in oxidative dehydrogenation, N groups transfer electrons to oxidative molecules such as O2 to form electrophilic oxygen species (O22−, O2−, and O−), which not only favors consumption of coke, but also forms oxygenated groups with C atoms;30 (3) the structure of reactants plays an important role. For example, ethylbenzene is a molecule with an electron-attracting group (phenyl), whereas propane with an electron-donor group. Different electron densities may affect hydrogen leaving from C–H bonds. In comparison, P has same lone electron pair as N but displays different chemical properties. While N can dope both in edge plane and basal plane sites, P doping usually takes place at the edge-plane sites of the graphitic framework due to the larger atomic radius.14 In PMC, P exists as P–C bonds and P–O bonds in PO4 tetrahedra. P not only donate electrons to unsaturated C
O groups). However, in oxidative dehydrogenation, N groups transfer electrons to oxidative molecules such as O2 to form electrophilic oxygen species (O22−, O2−, and O−), which not only favors consumption of coke, but also forms oxygenated groups with C atoms;30 (3) the structure of reactants plays an important role. For example, ethylbenzene is a molecule with an electron-attracting group (phenyl), whereas propane with an electron-donor group. Different electron densities may affect hydrogen leaving from C–H bonds. In comparison, P has same lone electron pair as N but displays different chemical properties. While N can dope both in edge plane and basal plane sites, P doping usually takes place at the edge-plane sites of the graphitic framework due to the larger atomic radius.14 In PMC, P exists as P–C bonds and P–O bonds in PO4 tetrahedra. P not only donate electrons to unsaturated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, but also shares the catalytic activity of dehydrogenation reaction in the form of P
O groups, but also shares the catalytic activity of dehydrogenation reaction in the form of P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups as C
O groups as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups.34 The electron rich properties of P also enhance the propene selectivity. Instead, B is a electron-deficient atom, but incorporating B into carbon skeleton produced additional surface oxygen groups,13,18 particularly C
O groups.34 The electron rich properties of P also enhance the propene selectivity. Instead, B is a electron-deficient atom, but incorporating B into carbon skeleton produced additional surface oxygen groups,13,18 particularly C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, as shown by the results of XPS and TPD (Table 2 and Fig. 6), which facilitate the propane conversion. Thus, B doping could decrease the barrier for the first C–H breaking.29 Consequently, doping mesoporous carbon with P and B improved the conversion of propane dehydrogenation in spite of different electronic structures.
O groups, as shown by the results of XPS and TPD (Table 2 and Fig. 6), which facilitate the propane conversion. Thus, B doping could decrease the barrier for the first C–H breaking.29 Consequently, doping mesoporous carbon with P and B improved the conversion of propane dehydrogenation in spite of different electronic structures.
Furthermore, P or B-doped mesoporous carbons can be prepared by an in situ synthesis approach. Perfect mesoporous structure and uniform distribution of doped heteroatoms are the preconditions for a good catalyst. Moreover, electron-rich phosphorus shared the catalytic activity and lone pair electron to carbonyl or quinone groups, resulting in the improved catalytic performance. Similarly, boron enhanced the oxygen-containing groups of carbon framework, which can also improve the conversion of direct propane dehydrogenation, although boron is an electron-deficient atom. In addition, after doping with P or B, the oxygen-containing groups remained nearly constant and the proportion of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups increased greatly, which effectively contribute to the propane conversion and propene selectivity.
O groups increased greatly, which effectively contribute to the propane conversion and propene selectivity.
4. Conclusions
In summary, mesoporous carbons doped with N, P, and B were prepared by different methods and used on direct dehydrogenation of propane. P- and B-doped mesoporous carbons exhibited improved initial propane conversion (37.1% and 33.9%, respectively) and higher initial propene selectivity for PMC (89.2%), which are due to the increased contents of carbonyl or quinone groups that are usually deemed as the main catalytic active sites. Apart from the low oxygen-containing group contents in NMC, it is also speculated that nitrogen itself or nitrogen-containing structures may not be helpful for direct oxidative dehydrogenation of propane, owing to different reaction mechanism, though it is electron rich as phosphorus. Moreover, whether heteroatom-doped carbon improves the activity or not also depends on the specific reaction process. In a word, this study points a way towards the metal-free carbon catalysts for direct dehydrogenation of propane or some other alkane.Acknowledgements
This study was supported by the National Natural Science Foundation of China (21421001, 21573115) and the Natural Science Foundation of Tianjin (15JCZDJC37100).References
- J. J. H. B. Sattler, J. Ruiz-Martinez, E. Santillan-Jimenez and B. M. Weckhuysen, Chem. Rev., 2014, 114, 10613–10653 CrossRef CAS PubMed.
- R. Wu, P. Xie, Y. Cheng, Y. Yue, S. Gu, W. Yang, C. Miao, W. Hua and Z. Gao, Catal. Commun., 2013, 39, 20–23 CrossRef CAS.
- M. M. Bhasin, J. H. McCain, B. V. Vora, T. Imai and P. R. Pujadó, Appl. Catal., A, 2001, 221, 397–419 CrossRef CAS.
- A. Qiao, V. N. Kalevaru, J. Radnik, A. Düvel, P. Heitjans, A. S. H. Kumar, P. S. S. Prasad, N. Lingaiah and A. Martin, Ind. Eng. Chem. Res., 2014, 53, 18711–18721 CrossRef CAS.
- X. Fan, J. Li, Z. Zhao, Y. Wei, J. Liu, A. Duan and G. Jiang, Catal. Sci. Technol., 2015, 5, 339–350 CAS.
- Z. Zhao, Y. Dai, G. Ge, X. Guo and G. Wang, RSC Adv., 2015, 5, 53095–53099 RSC.
- C. A. Carrero, R. Schloegl, I. E. Wachs and R. Schomaecker, ACS Catal., 2014, 4, 3357–3380 CrossRef CAS.
- S. Sokolov, M. Stoyanova, U. Rodemerck, D. Linke and E. V. Kondratenko, J. Catal., 2012, 293, 67–75 CrossRef CAS.
- T. Y. Ma, L. Liu and Z. Y. Yuan, Chem. Soc. Rev., 2013, 42, 3977–4003 RSC.
- K. Huang, S.-H. Chai, R. T. Mayes, G. M. Veith, K. L. Browning, M. A. Sakwa-Novak, M. E. Potter, C. W. Jones, Y.-T. Wu and S. Dai, Chem. Commun., 2015, 51, 17261–17264 RSC.
- Y. Yang, P. Li, S. Wu, X. Li, E. Shi, Q. Shen, D. Wu, W. Xu, A. Cao and Q. Yuan, Chem.–Eur. J., 2015, 21, 6157–6164 CrossRef CAS PubMed.
- A. Węgrzyniak, S. Jarczewski, A. Wach, E. Hędrzak, P. Kuśtrowski and P. Michorczyk, Appl. Catal., A, 2015, 508, 1–9 CrossRef.
- X. Sun, Y. Ding, B. Zhang, R. Huang and D. S. Su, Chem. Commun., 2015, 51, 9145–9148 RSC.
- T. Panja, D. Bhattacharjya and J.-S. Yu, J. Mater. Chem. A, 2015, 3, 18001–18009 CAS.
- G.-P. Hao, N. R. Sahraie, Q. Zhang, S. Krause, M. Oschatz, A. Bachmatiuk, P. Strasser and S. Kaskel, Chem. Commun., 2015, 51, 17285–17288 RSC.
- M. Chen, L. L. Shao, Y. P. Liu, T. Z. Ren and Z. Y. Yuan, J. Power Sources, 2015, 283, 305–313 CrossRef CAS.
- Y.-P. Zhu, Y. Liu, Y.-P. Liu, T.-Z. Ren, T. Chen and Z.-Y. Yuan, ChemCatChem, 2015, 7, 2903–2909 CrossRef CAS.
- X. Zhao, A. Wang, J. Yan, G. Sun, L. Sun and T. Zhang, Chem. Mater., 2010, 22, 5463–5473 CrossRef CAS.
- L. Liu, Y. P. Zhu, M. Su and Z. Y. Yuan, ChemCatChem, 2015, 7, 2765–2787 CrossRef CAS.
- M. F. R. Pereira, J. J. M. Órfão and J. L. Figueiredo, Appl. Catal., A, 1999, 184, 153–160 CrossRef CAS.
- J. Zhang, D. Su, A. Zhang, D. Wang, R. Schlögl and C. Hébert, Angew. Chem., Int. Ed., 2007, 46, 7319–7323 CrossRef CAS PubMed.
- D. S. Su, J. J. Delgado, X. Liu, D. Wang, R. Schlögl, L. Wang, Z. Zhang, Z. Shan and F.-S. Xiao, Chem.–Asian J., 2009, 4, 1108–1113 CrossRef CAS PubMed.
- L. Liu, Q.-F. Deng, B. Agula, X. Zhao, T.-Z. Ren and Z.-Y. Yuan, Chem. Commun., 2011, 47, 8334–8336 RSC.
- J. Zhang, D. S. Su, R. Blume, R. Schlögl, R. Wang, X. Yang and A. Gajović, Angew. Chem., Int. Ed., 2010, 49, 8640–8644 CrossRef CAS PubMed.
- Z. Zhang, Y. Li, J. Wang, H. Yang, N. Li, C. Ma and Z. Hao, Catal. Sci. Technol., 2016, 6, 4863–4871 CAS.
- L. Liu, Q. F. Deng, Y. P. Liu, T. Z. Ren and Z. Y. Yuan, Catal. Commun., 2011, 16, 81–85 CrossRef CAS.
- K. N. Wood, R. O'Hayre and S. Pylypenko, Energy Environ. Sci., 2014, 7, 1212–1249 CAS.
- C. Chen, J. Zhang, B. Zhang, C. Yu, F. Peng and D. Su, Chem. Commun., 2013, 49, 8151–8153 RSC.
- S. Mao, B. Li and D. Su, J. Mater. Chem. A, 2014, 2, 5287–5294 CAS.
- Y. Marco, L. Roldán, E. Muñoz and E. García-Bordejé, ChemSusChem, 2014, 7, 2496–2504 CrossRef CAS PubMed.
- Z. Zhao, Y. Dai and G. Ge, Catal. Sci. Technol., 2015, 5, 1548–1557 CAS.
- Z. Zhao, Y. Dai, G. Ge, X. Guo and G. Wang, Phys. Chem. Chem. Phys., 2015, 17, 18895–18899 RSC.
- Z. Zhao, Y. Dai, G. Ge and G. Wang, ChemCatChem, 2015, 7, 1135–1144 CrossRef CAS.
- L. Li, W. Zhu, Y. Liu, L. Shi, H. Liu, Y. Ni, S. Liu, H. Zhou and Z. Liu, RSC Adv., 2015, 5, 56304–56310 RSC.
- L. Liu, F.-Y. Wang, G.-S. Shao and Z.-Y. Yuan, Carbon, 2010, 48, 2089–2099 CrossRef CAS.
- L. Liu, Q. F. Deng, T. Y. Ma, X. Z. Lin, X. X. Hou, Y. P. Liu and Z. Y. Yuan, J. Mater. Chem., 2011, 21, 16001–16009 RSC.
- L. Liu, Q. F. Deng, B. Agula, T. Z. Ren, Y. P. Liu, B. Zhaorigetu and Z. Y. Yuan, Catal. Today, 2012, 186, 35–41 CrossRef CAS.
- X. Wang, J. S. Lee, Q. Zhu, J. Liu, Y. Wang and S. Dai, Chem. Mater., 2010, 22, 2178–2180 CrossRef CAS.
- J. Zhu, K. Li, M. Xiao, C. Liu, Z. Wu, J. Ge and W. Xing, J. Mater. Chem. A, 2016, 4, 7422–7429 CAS.
- L. Roldan, A. M. Benito and E. Garcia-Bordeje, J. Mater. Chem. A, 2015, 3, 24379–24388 CAS.
- Z. Wu, P. A. Webley and D. Zhao, J. Mater. Chem., 2012, 22, 11379–11389 RSC.
- Y. P. Zhu, Y. L. Liu, Y. P. Liu, T. Z. Ren, G. H. Du, T. H. Chen and Z. Y. Yuan, J. Mater. Chem. A, 2015, 3, 11725–11729 CAS.
- Y. P. Zhu, T. Z. Ren and Z. Y. Yuan, ACS Appl. Mater. Interfaces, 2015, 7, 16850–16856 CAS.
- J. J. Duan, S. Chen, M. Jaroniec and S. Z. Qiao, ACS Catal., 2015, 5, 5207–5234 CrossRef CAS.
- G. Wen, J. Diao, S. Wu, W. Yang, R. Schlögl and D. S. Su, ACS Catal., 2015, 5, 3600–3608 CrossRef CAS.
- B. Frank, J. Zhang, R. Blume, R. Schlögl and D. S. Su, Angew. Chem., Int. Ed., 2009, 48, 6913–6917 CrossRef CAS PubMed.
- J. Zhang, X. Liu, R. Blume, A. Zhang, R. Schlögl and D. S. Su, Science, 2008, 322, 73–77 CrossRef CAS PubMed.
- J. Wang, H. Liu, X. Gu, H. Wang and D. S. Su, Chem. Commun., 2014, 50, 9182–9184 RSC.
- M. Xie, Y. Xia, J. Liang, L. Chen and X. Guo, Microporous Mesoporous Mater., 2014, 197, 237–243 CrossRef CAS.
- H. F. Gorgulho, J. P. Mesquita, F. Gonçalves, M. F. R. Pereira and J. L. Figueiredo, Carbon, 2008, 46, 1544–1555 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra20726j |
| This journal is © The Royal Society of Chemistry 2016 |