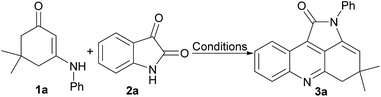
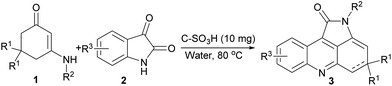
Single step incorporation of isatin to enaminone: a recyclable catalyst towards assembly of diverse four ring fused pyrrolo[2,3,4-kl]acridin-1-ones†
Chunmei Li and
Furen Zhang*
School of Chemistry and Chemical Engineering, Zhejiang Key Laboratory of Alternative Technologies for Fine Chemicals Process, Shaoxing University, Shaoxing, Zhejiang Province 312000, China. E-mail: frzhang@usx.edu.cn; Fax: +86-57588345682; Tel: +86-57588345682
First published on 5th August 2016
Abstract
A series of pyrrolo[2,3,4-kl]acridin-1-one derivatives were successfully synthesized from facile materials (enaminone and isatin) using a reusable solid acid as catalyst in water. A variety of structurally diverse enaminones and various isatins bearing different functional groups tolerated the reaction and gave the corresponding goal products with good to excellent yields. Advantages of this strategy include the mild conditions, operational simplicity, being convenient and environmentally benign, and the reusability of the heterogeneous catalyst. A mechanism was proposed to explain the formation of different substituted pyrrolo[2,3,4-kl]acridin-1-one derivatives.
Introduction
The simple and efficient construction of azaheterocyclic scaffolds, particularly, those of acridine derivatives, is of biological and chemical importance and has been actively pursued in organic and medicinal research for several decades recently.1 The acridine derivatives are an important class of heterocyclic pharmaceuticals and bioactive natural products and the structurally diverse and intriguing acridine family has been found to exhibit diverse biological activities such as antibacterial,2 antiparasitic,3 antifungal,4 and antitumor5 activities. Since 1990 three novel pentacyclic alkaloids,6 which all contain the fused pyrrolo[2,3,4-kl]acridine ring were isolated from a plakortis sponge, many of their biological and pharmacological activities had been found. Thus, the development of these fused acridine skeleton bearing different substituents or functional groups could potentially lead to a series of structurally and biologically interesting compounds.7 The synthesis of functional pyrrolo[2,3,4-kl]acridines and their derivatives has played an important role in organic and medical chemistry due to their therapeutic and pharmacological properties. However, only a few literatures exist concerning fused pyrrolo[2,3,4-kl]acridine syntheses,8 and these synthetic strategies still have many limitations involving multiple steps, harsh conditions, and expensive and non-recycle catalyst. Therefore, it is necessary to develop new economic, green and efficient methodology for their syntheses.As solid acid catalyst, carbonaceous materials have received more and more attention in view of their unique catalytic advantages recently.9 However, the general synthesis of carbonaceous materials usually relies on very harsh conditions, such as electric-arc discharge techniques,10 catalytical chemical vapor deposition,11 catalytic pyrolysis of organic compounds,12 high-temperature (800 °C) hydrothermal conversion from amorphous carbon13 or tedious separation work. Since a novel carbon functionalized with sulfonic acid groups,14 which owned the comparable activities to sulfonic acid, has been synthesized and investigated through esterification, oxathioketalization, and other organic reactions, such carbonaceous materials holding great potential for the environment-friendly processes had been widely used to catalyze organic synthetic reaction. As the continuous efforts on the construction of heterocyclic skeleton,15 here we would like to report a green method for the synthesis of fused pyrrolo[2,3,4-kl]acridin-1-one derivatives using recoverable sulfonated carbonaceous material as heterogeneous catalyst.
Results and discussion
To begin our study, we chose 5,5-dimethyl-3-(phenylamino)cyclohex-2-en-1-one (1a) and isatin (2a) as the model substrates to optimize suitable reaction conditions. The results were summarized in Table 1. As shown in Table 1, no goal product was obtained in the absence of catalyst even in 24 h at reflux. Then, different acidic catalysts, including inorganic acids such as HY zeolite, Amberlyst-15, and carbonaceous materials and organic acids such as TsOH, HOAc, TFA, H2SO4, Sc(OTf)3, and Y(OTf)3 were screened to promote the model reaction (Table 1, entries 2–11). Inorganic solid acid HY zeolite and Amberlyst-15 exhibit low activities and only give goal products with 20–26% yields. The carbonaceous material obtained from the single polyvinyl alcohol (carbon) showed almost no activity. However, the sulfonated carbonaceous material (C-SO3H) displayed a remarkably high activity and gave 4,4-dimethyl-2-phenyl-4,5-dihydropyrrolo[2,3,4-kl]acridin-1(2H)-one (3a) with 85% yield. In addition, the Brønst acids (including TsOH, HOAc, TFA, and H2SO4) and Lewis acids (including Sc(OTf)3 and Y(OTf)3) showed low activities and gave the goal products with 27% to 65% yields (Table 1, entries 6–11). Then, the screening of the amount of solid acid catalyst shown that carbonaceous material (10 mg) was enough to promote this reaction (Table 1, entries 5, 12–13).| Entry | Catalyst (mg) | Solvent | T (°C) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 1a (1.0 mmol), 2a (1.0 mmol), catalysts (x mg), solvent (3 mL), 8 h.b Isolated yields.c This reaction was carried out for 24 h.d Catalyst 10 mol%. | ||||
| 1 | — | H2O | Reflux | —c |
| 2 | Zeolite (HY) (10) | H2O | 80 | 26 |
| 3 | Amberlyst-15 (10) | H2O | 80 | 20 |
| 4 | Carbon (10) | H2O | 80 | 10 |
| 5 | C-SO3H (10) | H2O | 80 | 85 |
| 6 | TsOH (10)d | H2O | 80 | 32 |
| 7 | HOAc (10)d | H2O | 80 | 27 |
| 8 | TFA (10)d | H2O | 80 | 35 |
| 9 | H2SO4 (10)d | H2O | 80 | 42 |
| 10 | Sc(OTf)3 (10)d | H2O | 80 | 65 |
| 11 | Y(OTf)3 (10)d | H2O | 80 | 61 |
| 12 | C-SO3H (20) | H2O | 80 | 87 |
| 13 | C-SO3H (5) | H2O | 80 | 41 |
| 14 | C-SO3H (10) | MeOH | Reflux | 57 |
| 15 | C-SO3H (10) | EtOH | Reflux | 62 |
| 16 | C-SO3H (10) | DMF | 80 | 18 |
| 17 | C-SO3H (10) | THF | Reflux | 26 |
| 18 | C-SO3H (10) | Toluene | 80 | 45 |
| 19 | C-SO3H (10) | DCM | Reflux | 22 |
| 20 | C-SO3H (10) | H2O | 60 | 55 |
| 21 | C-SO3H (10) | H2O | 40 | 12 |
| 22 | C-SO3H (10) | H2O | 20 | —c |
| 23 | C-SO3H (10) | H2O | 100 | 86 |
To optimize the solvent for this method, the model substrates were mixed under different solvent such as MeOH, EtOH, DMF, THF, and toluene at 80 °C to investigate the solvent effect. As can be seen in Table 1, the reaction in water provided the best yield (Table 1, entry 5). Thus, we carried out the reaction in water to synthesize the following desired products. To further optimize the reaction conditions, the same reaction was carried out in water at temperature ranging from 20 to 100 °C (Table 1, entries 5, 20–23) with an increment of 20 °C each time. The yield of desired product was increased when the reaction temperature was increased from 20 °C to 80 °C. However, further increase of the temperature to 100 °C failed to improve the yield of product obviously (Table 1, entry 23). Thus, 80 °C was chosen as the most suitable temperature for all the further reactions.
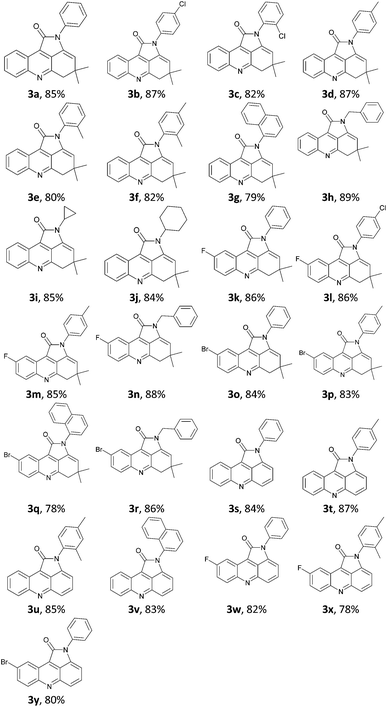
On the basis of optimized conditions described above, the scope of these reactions was examined using various easily available starting materials (Table 2). A range of valuable structure fused pyrrolo[2,3,4-kl]acridines were synthesized in good to excellent yields using sulfonated carbonaceous material as catalyst. Firstly, we chose isatin (2a) to react with enaminone (1) bearing different substituents. The effect of electronic nature of the substituents on aromatic ring of imines did not show strong influence on yields. As illustrated in Table 2, the imines substrates bearing a variety of either electron-rich or electron-deficient functional groups were efficient for the domino reaction, giving the corresponding fused pyrrolo[2,3,4-kl]acridine derivatives in good to excellent yields. Particularly noteworthy was the fact that hydrogen atom of enamine can be substituted with phenyl bearing the bulky groups, such as 1-naphthylamine, o-toluidine, 2,4-dimethylaniline and 2-chloroaniline, also displayed good reactivities but led to corresponding goal products with subtle low yields, respectively. In addition, aliphatic groups on the enaminone ring, such as benzyl, cyclopropyl, and cyclohexyl were well tolerated under the reaction conditions, leading to the desired products in satisfactory yields (84–89%). Then, different substrates (2) bearing fluoro, and bromo substituents on the isatin ring were submitted to react with enaminone bearing various substituents. As the results illustrated, all the reactions proceeded to generate the corresponding fused pyrrolo[2,3,4-kl]acridine derivatives in good to excellent yields, which highlighted the wide scope of this condensation. Thus, we concluded that the nature of the substituents of the isatin and enaminones had no obvious effect on the reaction.
To further explore the scope of this procedure, N-substituted 3-aminocyclohex-2-enone was employed to react with isatin. To our delight, the expected products (3s–3y) were obtained with good yields under the optimized reaction conditions. Similar to N-substituted 5,5-dimethyl-3-aminocyclohex-2-enone, N-substituted 3-aminocyclohex-2-enone bearing same substituents exhibited similar reaction features but with subtle reactivity differences. We attributed the different product structure of 4,5-dihydropyrrolo[2,3,4-kl]acridin-1-ones and pyrrolo[2,3,4-kl]acridin-1-ones to different substituted pattern of enaminone, which indicated that the reaction pathways could been controlled by introducing different enaminone. Additionally, we also noted that the existence of electron withdrawing functional groups such as fluoro and bromo groups of isatin also affect the formation of desired products in a way.
The use of heterogeneous and reusable catalysts represents one of the greenest procedures in chemical technology. Meanwhile, heterogeneous catalysts should not only be stable and active, but also require easy separating and reusing for useful applications. For this propose, after completion of reaction, catalyst which was recovered from reaction between 5,5-dimethyl-3-(phenylamino)cyclohex-2-en-1-one (1a) and isatin (2a) by filtration; after washing with water and methanol, and drying in vacuum oven at 100 °C for 4 h, it was used. To our delight, the catalyst could be recycled efficiently while no decrease in the yield of the goal product was observed during the first recycle. Further in the following run, it was found to be subtle decrease in the yield of goal product. We attribute the slight active loss of solid acid catalyst after the subsequent runs to the minor losses in the recovery process and marginal poisoning of carbonaceous material surface by organic impurities (Fig. 1).
On the basis of above results and the literature reports, possible mechanisms have been proposed for the divergent formations of fused pyrrolo[2,3,4-kl]acridines as shown in Fig. 2. The domino reaction of isatins and enaminones follows the regular mechanism of acid-catalyzed condensations. We assumed that the reaction of C-SO3H with isatins affords radical intermediate A, which is attacked by enaminone to give intermediate B. Under the influence of C-SO3H, intermediate B undergoes imine–enamine tautomerization to give intermediate C, immediately. The intermediate C undergoes rapidly C–C bond rotation (intermediate D) and reacts in an intramolecular cyclization and ring-opening sequence (intermediate E) to give intermediate F. Subsequently, intramolecular imine formation then leads to the formation of intermediate G and the goal product by losing H2O molecule. When the group R1 is hydrogen atom, the compounds 3 would undergo aromatic cyclization and give desired product 2-phenylpyrrolo[2,3,4-kl]acridin-1(2H)-one derivatives (3s–3y).
Conclusions
In summary, we have developed a domino reaction (isatins and enaminones) as an alternative method for divergent synthesis of fused pyrrolo[2,3,4-kl]acridines with different substituted patterns by varying N-substituted enaminone and isatin substrates. The ready accessibility of the starting materials, the broad compatibility of N-substituted enaminone substrates, and the generality of this process make the reaction highly valuable in view of the synthetic and biological importance of fused pyrrolo[2,3,4-kl]acridine derivatives. The typical advantages of this approach include the stable and active sulfonated carbonaceous materials as reusable catalyst and the ready available water as green solvent. Mechanism studies indicated that the domino reaction catalyzed by this solid acid catalyst include ring closure cascade reaction, intramolecular cyclization, ring-opening of isatin, recyclization and dehydration.Experimental
General
The reagents were purchased from Aladdin and Aldrich and were not purified before use. Analytical thin layer chromatography (TLC) was performed using Merck silica gel GF254 plates. Melting points were measured on an X-4 melting point apparatus. 1H NMR spectra were recorded on a 400 MHz instrument (Bruker Avance 400 Spectrometer). Chemical shifts (δ) are given in ppm relative to TMS as the internal reference, with coupling constants (J) in Hz. 13C NMR spectra were recorded at 100 MHz. Chemical shift were reported in ppm with the internal chloroform signal at 77.0 ppm as a standard. Elemental analysis was carried out on EuroEA elemental analyzer.General procedure for the synthesis of compounds 3
Enaminone (1.0 mmol) was introduced in a 10 mL reaction vial, isatin (1.0 mmol) and carbonaceous material (10 mg), and water (3 mL), were then successively added. Then, the reaction vial was closed and stirred on an oil bath at 80 °C for the appropriate time. The mixture was stirred until TLC revealed that the conversion of the starting material was complete about 8 h. Then, the mixture was cooled to room temperature and the catalyst and the product were filtered. The filtered solid catalyst was washed with water and methanol, and dried in vacuum oven at 100 °C for 4 h. The resulting solution was concentrated and recrystallized from EtOH (95%) to give the pure products.Acknowledgements
This work was made possible by grants from the Natural Science Foundation of Zhejiang Province (No. LY16B020007) and the Foundation of Education Department of Zhejiang Province (No. Y201533906).Notes and references
- For recent reviews, see: (a) R. Tohme, N. Darwiche and H. Gali-Muhtasib, Molecules, 2011, 16, 9665 CrossRef CAS PubMed; (b) G. L. Thomas and C. W. Johannes, Curr. Opin. Chem. Biol., 2011, 15, 516 CrossRef CAS PubMed; (c) S. Dandapani and L. A. Marcaurelle, Curr. Opin. Chem. Biol., 2010, 14, 362 CrossRef CAS PubMed; (d) M. E. Welsch, S. A. Snyder and B. R. Stockwell, Curr. Opin. Chem. Biol., 2010, 14, 347 CrossRef CAS PubMed.
- (a) M. J. Wainwright, J. Antimicrob. Chemother., 2001, 47, 1 CrossRef CAS PubMed; (b) B. L. Mylari, J. G. Lombardino and J. M. McManus, US Pat. 5, 624, 929, 1997.
- L. Guetzoyan, F. Ramiandrasoa, H. Dorizon, C. Desprez, A. Bridoux, C. Rogier, B. Pradines and M. Perree-Fauvet, Bioorg. Med. Chem., 2007, 15, 3278 CrossRef CAS PubMed.
- P. J. McCarthy, T. P. Pitts, G. P. Gunawardana, M. Kelly-Borges and S. A. Pomponi, J. Nat. Prod., 1992, 55, 1664 CrossRef CAS.
- (a) A. Koller, A. Rudi, M. Garcia and Y. Kashman, Molecules, 2001, 6, 300 CrossRef CAS; (b) W. Denny, Curr. Med. Chem., 2002, 9, 1655 CrossRef CAS PubMed; (c) E. Gimenez-Arnau, S. Missailidis and M. F. G. Stevens, Anti-Cancer Drug Des., 1998, 13, 431 CAS.
- (a) W. D. Inman, M. O'Neill-Johnson and P. Crews, J. Am. Chem. Soc., 1990, 112, 1 CrossRef CAS; (b) R. R. West, C. L. Mayne, C. M. Ireland, L. S. Brinen and J. Clardy, Tetrahedron Lett., 1990, 31, 3271 CrossRef CAS.
- (a) A. Kuzuya, R. Mizoguchi, T. Sasayama, J.-M. Zhou and M. Komiyama, J. Am. Chem. Soc., 2004, 126, 1430 CrossRef CAS PubMed; (b) I. Antonini, P. Polucci, A. Magnano, S. Sparapani and S. Martelli, J. Med. Chem., 2004, 47, 5244 CrossRef CAS PubMed; (c) J. C. Cookson, R. A. Heald and M. F. G. Stevens, J. Med. Chem., 2005, 48, 7198 CrossRef CAS PubMed; (d) M.-K. Cheng, C. Modi, J. C. Cookson, I. Hutchinson, R. A. Heald, A. J. McCarroll, S. Missailidis, F. Tanious, W. D. Wilson, J.-L. Mergny, C. A. Laughton and M. F. G. Stevens, J. Med. Chem., 2008, 51, 963 CrossRef CAS PubMed; (e) Y. Kitahara, T. Mizuno and A. Kubo, Tetrahedron, 2004, 60, 4283 CrossRef CAS; (f) B. V. Subba Reddy, A. Antony and J. S. Yadav, Tetrahedron Lett., 2010, 51, 3071 CrossRef; (g) Y.-N. Wu, R. Fu, N.-N. Wang, W.-J. Hao, G.-G. Li, S.-J. Tu and B. Jiang, J. Org. Chem., 2016, 81, 4762 CrossRef CAS PubMed; (h) Y.-L. Zhu, B. Jiang, W.-J. Hao, J.-K. Qiu, J. Sun, D.-C. Wang, W. Ping, A.-F. Wang, G.-G. Li and S.-J. Tu, Org. Lett., 2015, 17, 6078 CrossRef CAS PubMed.
- (a) B. Jiang, X.-S. Wang, M.-Y. Li, Q. Wu, Q. Ye, H.-W. Xu and S.-J. Tu, Org. Biomol. Chem., 2012, 10, 8533 RSC; (b) H. Wang, L. Li, W. Lin, P. Xu, Z. Huang and D. Shi, Org. Lett., 2012, 14, 4598 CrossRef CAS PubMed; (c) L. Li, H. Wang, W. Lin, Z. Huang and D. Shi, J. Heterocycl. Chem., 2014, 51, 1778 CrossRef CAS; (d) M.-H. Hu, W. Lin, C.-P. Cao, Z.-B. Huang and D.-Q. Shi, J. Heterocycl. Chem., 2014, 51, E227 CrossRef CAS; (e) C. Cao, C. Xu, W. Lin, X. Lin, M. Hu, J. Wang, Z. Huang, D. Shi and Y. Wang, Molecules, 2013, 18, 1613 CrossRef CAS PubMed; (f) P. Sarkar and C. Mukhopadhyay, Green Chem., 2015, 17, 3452 RSC; (g) S. Ray, A. Bhaumik, M. Pramanik and C. Mukhopadhyay, RSC Adv., 2014, 4, 15441 RSC; (h) V. O. Iaroshenko, S. Dydkin, V. Y. Sosnovskikh, A. Villinger and P. Langer, Synthesis, 2013, 45, 971 CrossRef CAS; (i) W.-J. Hao, J.-Q. Wang, X.-P. Xu, S.-L. Zhang, S.-Y. Wang and S.-J. Ji, J. Org. Chem., 2013, 78, 12362 CrossRef CAS PubMed; (j) B. Jiang, X. Wang, H.-W. Xu, M.-S. Tu, S.-J. Tu and G.-G. Li, Org. Lett., 2013, 15, 1540 CrossRef CAS PubMed.
- (a) V. Budarin, R. Luque, D. J. Macquarrie and J. H. Clark, Chem.–Eur. J., 2007, 13, 6914 CrossRef CAS PubMed; (b) K. Nakajima, M. Okamura, J. N. Kondo, K. Domen, T. Tatsumi and S. Hayashi, Chem. Mater., 2009, 21, 186 CrossRef CAS; (c) J. W. Comerford, J. H. Clark, D. J. Macquarrie and S. W. Breeden, Chem. Commun., 2009, 2562 RSC; (d) M. Toda, A. Takagaki, M. Okamura, J. N. Kondo, K. Domen and S. Hayashi, Nature, 2005, 438, 178 CrossRef CAS PubMed; (e) M. Hara, T. Yoshida, A. Takagaki, T. Takata, J. N. Kondo and K. Domen, Angew. Chem., Int. Ed., 2004, 43, 2955 CrossRef CAS PubMed.
- A. Thess, R. Lee, P. Nikolaev, H. J. Dai, P. Petit and J. Robert, Science, 1996, 273, 483 CAS.
- A. Eftekhari, P. Jafarkhani and F. Moztarzadeh, Carbon, 2006, 44, 1343 CrossRef CAS.
- L. Gherghel, C. Kubel, G. Lieser, H. J. Rader and K. Müllen, J. Am. Chem. Soc., 2002, 124, 1330 CrossRef.
- H. Ye, N. Naguib, Y. Gogotsi, A. G. Yazicioglu and C. M. Megaridis, Nanotechnology, 2004, 15, 232 CrossRef CAS.
- X. Z. Liang, H. Q. Xiao, Y. S. Shen and C. Z. Qi, Mater. Lett., 2010, 64, 953 CrossRef CAS.
- (a) C. M. Li, X. Z. Liang, F. R. Zhang and C. Z. Qi, Catal. Commun., 2015, 62, 6 CrossRef CAS; (b) F. R. Zhang, C. M. Li, C. Wang and C. Z. Qi, Org. Biomol. Chem., 2015, 13, 5022 RSC; (c) Z. Chen, Y. X. Shi, Q. Q. Shen, H. X. Xu and F. R. Zhang, Tetrahedron Lett., 2015, 56, 4749 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra18048e |
| This journal is © The Royal Society of Chemistry 2016 |