pH and thermosensitive 5-fluorouracil loaded poly(NIPAM-co-AAc) nanogels for cancer therapy†
Naga Sravan Kumar Varma V.*a,
Shivakumar H. G.a,
Syeda Juveriya Fathimab,
Vegesna Radhac and
Farhath Khanumb
aDepartment of Pharmaceutics, JSS College of Pharmacy, JSS University, Mysuru, India. E-mail: vnskvarma@gmail.com
bBiochemistry and Nanosciences Discipline, Defence Food Research Laboratory, Defence Research and Development Organisation (DRDO), Mysuru, India
cCentre for Cellular and Molecular Biology, Council of Scientific and Industrial Research (CSIR), Uppal Road, Hyderabad, India
First published on 25th October 2016
Abstract
The aim of the study was to develop 5-FU loaded pH and thermo-sensitive nanogels, capable of sensing change in tumour environment pH and triggering drug release at physiological temperature. Poly(N-isopropylacrylamide-co-acrylic acid) (PNA) nanogels (Ng's) were prepared via free radical polymerization. The prepared nanogels were screened for FT-IR, 1H NMR, pH dependent lower critical solution temperature (LCST), and DLS. Selected Ng's were loaded with 5-FU and screened for FTIR, particle size, TEM, % EE, in vitro drug release, % hemolysis, pH dependent cytotoxicity, in vivo kinetics on Wistar rats, in vivo anti-cancer efficacy and mean life span on Swiss albino mice. FTIR and 1H NMR confirmed the formation of PNA. The prepared nanogels exhibited pH dependent LCST. The particle size of selected PNA and 5-FU–PNA Ng's were found to be 70.25 and 131.5 nm respectively at 25 °C. Drug release from 5-FU–PNA Ng's was found to be extended and highly sustained at pH 5.5, 6.8 and 7.4. However, when compared with respect to individual pH, drug release in pH 7.4 PBS was quite minimal when compared to pH 5.5 and 6.8. The cytotoxicity of 5-FU–PNA Ng's increased with decrease in pH value. Additionally they could extend in vivo circulation time and displayed effective anti-cancer efficacy when compared to 5-FU injection for the same. Our results documented enhanced anti-cancer efficacy and mean life span in EAC bearing mice.
1. Introduction
Even though the advances in treatment and management of cancer have progressed over the years, its complete eradication still remains a conundrum for researchers as well as physicians. According to the WHO cancer report 2014, cancer still remains a leading cause of morbidity and is responsible for at least 8.2 million deaths each year and is accountable for 14 million new cases each year. Research suggests that the number of new cases is expected to rise by approximately 70% in the upcoming two decades.15-Fluorouracil (5-FU), a fluorinated analog of the pyrimidine uracil, is well known for its anti-metabolite and anti-neoplastic actions and applications in the treatment of different types of cancer. Principally, it acts as a thymidylate synthase (TS) inhibitor via blocking the enzyme synthesis of pyrimidine thymidine, a nucleoside required for DNA replication. Consequently, inhibition of TS leads to a scarcity of thymidine monophosphate (dTMP) that ultimately causes cell death.2 A major drawback of 5-FU and current chemotherapeutic regimens has been associated with severe side effects such as cardiac, lung, liver, and nephrotoxicity which also affect emotional status of the individual and higher risk of relapse.3–6 In order to address the aforementioned problems, extensive research on developing targeted drug delivery systems for existing molecules as well as synthesis of new potential anti-neoplastic/anti-metabolites candidates is being carried out.7–11 However, a complete breakthrough for successful treatment still remains a dilemma.
More recently altered tumour microenvironment and pH has generated substantial interest in developing pH responsive drug delivery systems.12–15 It is well known that extracellular nature of tumour cells/tissues is acidic in contrast to physiological pH of healthy cells that are basic.16–19 Therefore, difference in pH could be used as a tool to trigger the release of drug at the tissue/cell of interest.16,20 Thermo-sensitive polymers that are hydrophilic in nature but become hydrophobic beyond its lower critical solution temperature (LCST) can be used as a secondary stimuli for triggering the drug release.21,22 Therefore, a system responding to more than one stimuli would be a boon to the existing mono stimulus systems.23 pH and thermo-sensitive nanocarriers that could alter its LCST, based on the difference in micro-environmental pH, could be utilized as an effective carrier for targeting the drug.24 Amongst the available pH and thermo-sensitive polymer, poly(N-isopropylacrylamide-co-acrylic acid) (PNA) has been extensively reported for its pH dependent thermo-sensitivity property and its application for targeting drugs to the site of choice.25,26
Previously Xiong et al.,27 made an attempt to prepare pH and thermo-sensitive PNA nanoparticles covalently conjugated with doxorubicin and studied its potential application in tumour hyperthermia treatment. Sanoj Rejinold et al., attempted to develop 5-FU loaded pH and thermo-sensitive chitosan-graft-poly(N-isopropylacrylamide)28 and fibrinogen-graft-poly(N-isopropylacrylamide)29 and studied their application for breast cancer therapy using MCF-7 breast cancer cells. Even through the respective studies highlighted the applications of pH and thermo sensitive over cancer therapy, there is no sufficient in vivo anti-cancer efficacy studies highlighting their application. In the present study, an attempt has been made to prepare self regulated pH and thermosensitive PNA Ng's; capable of sensing the change in tumour environmental pH and trigger the drug release at physiological temperature. 5-FU was selected as a model drug loaded into selected PNA Ng's and studied for its in vitro drug release, cytotoxicity, in vivo kinetics, in vivo anticancer efficacy and mean life span.
2. Materials and methods
2.1. Materials
5-FU was a gift sample obtained from Sigma-Aldrich, India. N-Isopropylacrylamide (NIPAM) was procured from Tokyo Chemical Industry Co., Ltd. Tokyo, Japan. Acrylic acid (AAc) and sodium dodecyl sulphate (SDS) were procured from Loba Chemicals, Mumbai. Potassium peroxodisulphate (KPS) was procured from Merck, Mumbai. N,N′-Methylenebis(acrylamide) was procured from Sigma Aldrich, USA. NIPAM was purified by re-crystallization in methanol/n-hexane (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50). All other excipients and chemicals used were of analytical grades.
50). All other excipients and chemicals used were of analytical grades.
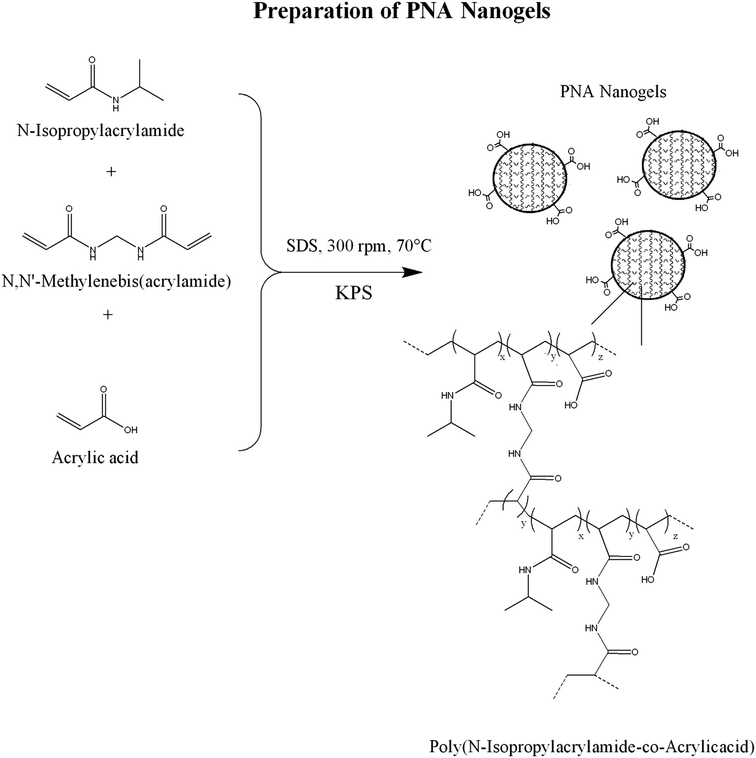
2.2. Development of poly(N-isopropylacrylamide-co-acrylic acid) nanogels
Poly(N-isopropylacrylamide-co-acrylic acid) Ng's were prepared using free radical polymerization. As listed in Table 1, a series of nanogels were prepared by varying the monomer feed composition (NIPAM and AAc).27,30 The total monomer in feed was kept constant at 0.015 mol/100 mL and feed composition ratios were varied accordingly (98 − x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x
x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2; NIPAM
2; NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AAc
AAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MBA). The monomers, crosslinker MBA (46 mg) and surfactant SDS (58 mg) were initially dissolved in 95 mL of distilled water. The prepared solution was heated at 70 ± 5 °C for 1 h and constantly stirred at 300 rpm using magnetic stirrer. Polymerization was initiated by addition of 5 mL distilled water containing 17 mg KPS. Nitrogen was purged for 15 min prior to addition of initiator. The reaction process was continued for next 4 h with above set parameters. Polymerization was terminated by reducing the temperature of the solution to room temperature. The resulting solution was dialysed for 5 days at room temperature and further freeze dried using Christ Alpha 2-4 LD Freeze Dryer. % practical yield was determined gravimetrically (eqn (1)).
MBA). The monomers, crosslinker MBA (46 mg) and surfactant SDS (58 mg) were initially dissolved in 95 mL of distilled water. The prepared solution was heated at 70 ± 5 °C for 1 h and constantly stirred at 300 rpm using magnetic stirrer. Polymerization was initiated by addition of 5 mL distilled water containing 17 mg KPS. Nitrogen was purged for 15 min prior to addition of initiator. The reaction process was continued for next 4 h with above set parameters. Polymerization was terminated by reducing the temperature of the solution to room temperature. The resulting solution was dialysed for 5 days at room temperature and further freeze dried using Christ Alpha 2-4 LD Freeze Dryer. % practical yield was determined gravimetrically (eqn (1)).
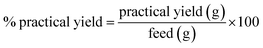 | (1) |
| Code | Feed composition ratioa (NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) AAc AAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MBA) MBA) |
% practical yield | Obtained polymer compositionb mol% | |
|---|---|---|---|---|
| NIPAM | AAc | |||
a Total monomer in feed was 0.015 mol. Feed composition ratio's used (98 − x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).b Obtained polymer composition (mol%) was calculated using 1H-NMR results. 2).b Obtained polymer composition (mol%) was calculated using 1H-NMR results. |
||||
| PNA0 | 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
83.45 | 100 | 0.00 |
| PNA1 | 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
80.25 | 99.05 | 0.95 |
| PNA1.5 | 96.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
87.23 | 99.36 | 0.64 |
| PNA2 | 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
79.07 | 97.16 | 2.84 |
| PNA2.5 | 95.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
78.31 | 91.18 | 8.82 |
2.3. Characterization and evaluation of poly(N-isopropylacrylamide-co-acrylic acid) nanogels
The nanogels were screened for Fourier transform infrared spectroscopy (FT-IR), proton nuclear magnetic resonance (1H NMR), pH dependent lower critical solution temperature (LCST) and change in hydrodynamic diameters in response to pH and temperature.Polymer composition was calculated by comparing peak area of NIPAM C–H at 3.9 with total peak area between 0.7 and 2.2 ppm. Further, % copolymer composition was calculated by comparing ratio of copolymer with that of homopolymer.
2.4. 5-FU loaded PNA Ng's
Entrapment efficacy (% EE) was determined using following eqn (2).
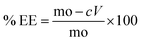 | (2) |
The resulting 5-FU loaded PNA (5-FU–PNA) Ng's dispersion was lyophilized using TFD5503, Ilshin Lab, Co. Ltd., and the % practical yield was quantified gravimetrically (eqn (1)). Freeze dried 5-FU–PNA Ng's were screened for FT-IR, 1H-NMR, and particle size. FT-IR and 1H-NMR were carried out as per the methodology mentioned in Section 2.3. Particle size, PDI and zeta potential were analysis in distilled water was facilitated by DLS (Nano ZS90, Malvern instruments). The screening tests were carried out at 25 °C and 37 °C.
| Plasma Hb = {(2 × A450) − (A380 + A415) × 76.26} | (3) |
2.4.5.1. Sterilization process. Pre calculated amount of 5-FU–PNA Ng's were sealed in ampoules under nitrogen atmosphere and sterilized using gamma rays at a dose of 15.76 kGy cobalt-60 at (Microtrol, Bangalore, India).
2.4.5.2. Sterility test. 5-FU–PNA Ng's dispersion was prepared by dispersing sterilized sample in water for injection. Prepared Ng's dispersion was inoculated to soybean digestive media and fluid thioglycolate medium and examined for sterility. Sterilized medium was used as negative control and un-sterilized medium was used as positive control. Clouding of medium indicates growth of microorganisms and contamination, while clear/transparent medium indicates un-contaminated.
Twelve albino Wistar rats of either sex weighing 230–260 g were divided into two groups and monitored for one week under quarantine conditions prior to studies. The animals were fed a standard chow diet ad libitum and had free access to water. The two pre-divided groups comprising of six in each, were labelled as standard (5-FU) and test (5-FU–PNA). Intra-peritoneal injection of pure 5-FU and 5-FU–PNA (fixed dose 20 mg kg−1 in water for injection) to the standard (5-FU) and test (5-FU–PNA) group's was facilitated using 1 mL syringe equipped with 26 G needle respectively.35 0.5 mL blood samples were withdrawn post administration from the jugular vein at pre-determined time intervals. Samples were collected in K3 EDTA tube (CB Plus®, Quantum Biomedicals, India), followed by centrifugation for 10 min at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm at 4 °C. Plasma was separated and stored at −50 °C for further analysis.
000 rpm at 4 °C. Plasma was separated and stored at −50 °C for further analysis.
Plasma drug concentration was evaluated using ultra-fast liquid chromatography (UFLC), (Shimadzu Prominence LC-20AD), equipped with a 1260 binary pump VL (35 MPa), Prominence SIL-20ACHT auto-sampler and Prominence diode array detector. 5-FU from test plasma was extracted by dispersing 790 μL of acetonitrile, as extracting solvent, to the 200 μL plasma test sample. The resulting samples were vortexed for 10 min and centrifuged for 10 min at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm. Supernatant was filtered using 0.2 μm syringe filter. The obtained solution (10 μL) was analysed using Kinetex® C18 column and pH 6 (20 mM) potassium dihydrogen orthophosphate buffer at a flow rate of 0.8 mL min−1. The retention time (Rt) of 5-FU was found to be 4.9 min.
000 rpm. Supernatant was filtered using 0.2 μm syringe filter. The obtained solution (10 μL) was analysed using Kinetex® C18 column and pH 6 (20 mM) potassium dihydrogen orthophosphate buffer at a flow rate of 0.8 mL min−1. The retention time (Rt) of 5-FU was found to be 4.9 min.
Pharmacokinetic parameters such as maximum plasma concentration (Cmax), half life (t1/2), area under curve (AUC0–t), area under curve extrapolated to infinity (AUC0–∞), area under the first moment curve extrapolated to infinity (AUMC0–∞), and residence time (MRT) were calculated from plasma-concentration time profile.
3. Results and discussion
3.1. Preparation and evaluation of PNA Ng's
3.1.2.1. FT-IR spectral analysis. FT-IR spectral data of NIPAM, AAc, and prepared nanogels were compared and shown in ESI Fig. S1 and S2.† Spectral analytical data of monomer NIPAM showed functional peaks at 2978 cm−1, 2939 cm−1, 2879 cm−1, 1465 cm−1, 1388 cm−1, 1363 cm−1, 1666 cm−1, and 1543 cm−1. Functional peaks at 2978 cm−1, 2939 cm−1 and 2879 cm−1 representing C–H stretching vibrations of methyl and ethyl groups. Functional peaks at 1465 cm−1, 1388 cm−1 and 1363 cm−1 represent asymmetry and symmetry bending of –CH3, respectively. Amide I and amide II bands at 1666 cm−1 and 1543 cm−1 were considered to be from C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching and N–H plane bending. Peak at 1249 cm−1 represent C–N stretching vibration of amide. Monomer AAc showed functional peak at 1720 cm−1 representing C
O stretching and N–H plane bending. Peak at 1249 cm−1 represent C–N stretching vibration of amide. Monomer AAc showed functional peak at 1720 cm−1 representing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration in carboxylic acid. Further, peaks observed at 3000–4000 cm−1 attributed to stretching vibration of acrylic carboxylic –OH.37
O stretching vibration in carboxylic acid. Further, peaks observed at 3000–4000 cm−1 attributed to stretching vibration of acrylic carboxylic –OH.37FT-IR spectral data of homo-polymer PNA0 showed all the functional peaks of monomer NIPAM. Similarly, FT-IR spectral data of all co-polymeric nanogels (PNA1–PNA2.5) showed all the functional peaks of NIPAM and AAc without any major shift. The obtained results confirm that polymerization was successful.
3.1.2.2. 1H NMR spectroscopic studies. 1H NMR spectral analysis of D2O swelled homo-polymer PNA0 and copolymers PNA2 nanogels were sketched and reproduced in Fig. S3–S5.† The 1H NMR spectra of PNA0 showed 4 major peaks at 1.028, 1.464, 1.895 and 3.780. The first 3 represents the following groups present in polymer backbone i.e., –CH3, –CH2, –CH, whereas the fourth peak reflects the isopropyl –CH group in NIPAM respectively. As per previous reports, the peak area of the above observed peaks should fall in the ratio of 6
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. However, as per the result, the measured intensity is in the ratio of 6.07
1. However, as per the result, the measured intensity is in the ratio of 6.07![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.88
1.88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.17
1.17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 respectively. These observations are in agreement with previously reported findings.38
1.00 respectively. These observations are in agreement with previously reported findings.38Similarly, the prepared copolymers PNA1.0–PNA2.5 exhibited similar/infinitesimal difference in peak positions. The difference in peak area is attributed in response to AAc content.
As mentioned earlier % copolymer composition was calculated by comparing peak area at 3.9 ppm, with the total peak area between 0.7 and 2.2 ppm. From the results (Table 1), it is clear that copolymer composition was significantly influenced by feed ratio. However, % composition copolymer PNA1.5 was found to be insignificant. This occurred because of practical failure or because of selected feed ratios of NIPAM/AAc is very narrow.
FT-IR and 1H NMR spectral analysis confirmed copolymerization between NIPAM and AAc was successful.
3.1.2.3. pH dependent LCST. LCST of individual polymers in response to change in pH was evaluated by measuring the absorbance at regular temperature intervals. The results obtained were sketched and reproduced in Fig. 1. In order to mimic tumour endosomal and extracellular pH, pH 5.5 and 6.8 PBS were used, whereas pH 7.4 PBS was used to mimic physiological pH.39–41 We have also studied LCST in 0.9% NaCl saline as physiological blood stream contains about 95–105 mM of Cl− which could affect the release of the drug/LCST.42 From the results (Fig. 2) it is evident that pH of the sample solution and polymer composition had significant influence over LCST.
Effect of sample pH over LCST. The homo-polymer PNA0 Ng (Fig. 1A) showed sharp LCST phase transition between 30–31.5 °C, without being influenced by pH of sample solution. However for all co-polymeric nanogels (PNA1–PNA2.5) (Fig. 1B–E), LCST phase transition was found to be significantly influenced by pH of the sample solution. Increase in PBS pH from 5.5 to 7.4 consequently shifted the LCST to higher temperature. For e.g., LCST of PNA2 in pH 5.5 and 6.8 was found to be between 38 °C and 55 °C respectively, whereas at pH 7.4 it was found to be higher than 55 °C. This characteristic pH dependent LCST of Ng's is because of presence of carboxylic acid derivative (AAc) in the backbone of Ng's which deprotonate easily at higher pH, consequently resulting in increased hydrophilicity of Ng's.27,43,44
Effect of polymer composition. With the increase in AAc to NIPAM ratio, LCST phase transition of prepared nanogels shifted to higher temperature. For e.g., LCST of homo-polymer in saline solution was found to be 31 °C, whereas LCST of PNA1–PNA2.5 shifted to higher temperatures such as 32 °C, 32 °C, 38 °C and 44 °C respectively. Similar behaviour was observed at different pH 5.5, 6.8 and 7.4 PBS respectively. From the results it was also observed that LCST phase transition of homo-polymer PNA0 is very sharp whereas, phase transition of copolymers PNA1–PNA2.5 were found to be broad and extended. This is because with the increase of AAc monomers within the copolymer network, increased hindrance in polymer chain subsequently leading to dehydration/hydrophobic aggregation and further expands the structural breakdown.38
From the results, we can conclude that at 37 °C PNA2 Ng's comprising of 97.16% molar of NIPAM and 2.84% molar of AAc, responded sharply to pH 5.5 and were dormant at pH 6.8 and 7.4 respectively, making it a suitable candidate for further evaluations.
3.1.2.4. pH and temperature dependent hydrodynamic diameter. PNA2 Ng's was studied for pH and temperature dependent hydrodynamic volume phase transition (Fig. 2). Initially, average particle size of prepared Ng's were found to be 68.80, 69.19 and 70.25 nm in pH 5.5, 6.8 and 7.4 respectively at 25 °C. The observed increase in particle size with increasing pH is because of the increase in pH, more and more –COOH deprotonate to anionic –COO–, resulting in internal electrostatic repulsion, and hence increase in particle size. Similarly, with decrease in surrounding pH, intra-chain hydrogen bonding association between NIPAM and AAc leads to shrinking of PNA Ng's at pH 5.5.45,46 The pH and temperature dependent hydrodynamic volume phase transition were found to be in agreement with pH and temperature dependent LCST studies carried out using UV spectrometer. As discussed earlier, decrease in particle size with increase in temperature is because of Ng's transition from hydrophilic to hydrophobic nature, which in-turn resulted in weakening of hydrogen bonding between water and polymer segments resulting in de-swelling/shrinking.46
3.2. 5-FU loaded PNA Ng's
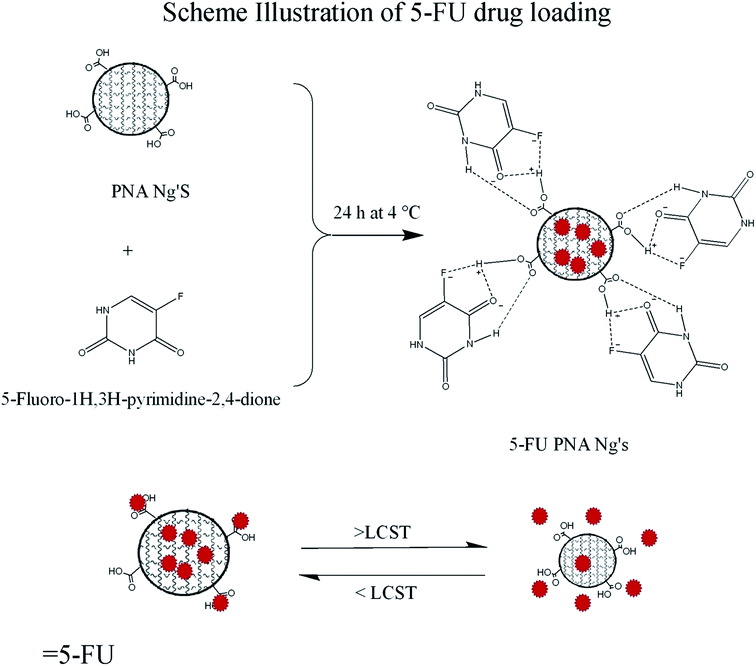
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and fluoro present in 5-FU are δ+, interaction between these groups forms a stable hydrogen bond. Similarly, –NH present in 5-FU and
O and fluoro present in 5-FU are δ+, interaction between these groups forms a stable hydrogen bond. Similarly, –NH present in 5-FU and ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O present in COOH– interacts through hydrogen bonding) (Scheme 2).29 % practical yield of 5-FU PNA Ng's was 79.5%, and % EE of 5-FU loading in PNA2 Ng's was found to be 34.46%. Average particle size, PDI and zeta potential of 5-FU–PNA Ng's in distilled water at 25 °C and 37 °C was found to be 138.7 and 109.1 nm, 0.262 and 0.243, −21 mV and −14 mV respectively. The observed increase of particle size when compared to placebo Ng's might be because of entrapment of 5-FU within the PNA matrix. FTIR spectral analysis confirmed 5-FU and PNA are compatible and formation of 5-FU–PNA (Fig. S6†).
O present in COOH– interacts through hydrogen bonding) (Scheme 2).29 % practical yield of 5-FU PNA Ng's was 79.5%, and % EE of 5-FU loading in PNA2 Ng's was found to be 34.46%. Average particle size, PDI and zeta potential of 5-FU–PNA Ng's in distilled water at 25 °C and 37 °C was found to be 138.7 and 109.1 nm, 0.262 and 0.243, −21 mV and −14 mV respectively. The observed increase of particle size when compared to placebo Ng's might be because of entrapment of 5-FU within the PNA matrix. FTIR spectral analysis confirmed 5-FU and PNA are compatible and formation of 5-FU–PNA (Fig. S6†).
| Pharmacokinetic parameters | 5-FU IP injection | 5-FU PNA Ng's |
|---|---|---|
| a P < 0.05****; when compared with 5-FU IP injection. | ||
| t1/2 (h) | 0.35 ± 0.046 | 5.49 ± 3.83 |
| Cmax (μg mL−1) | 98.19 ± 12.518 | 76.476 ± 7.50 |
| AUC0–t (μg mL−1 h−1) | 56.43 ± 13.18 | 214.19 ± 69.95 |
| AUC0–∞ (μg mL−1 h−1) | 59.61 ± 13.59 | 232.40 ± 82.50 |
| AUMC0–∞ (μg mL−1 h−2) | 40.44 ± 14.14 | 1488.20 ± 1091.30**** |
| MRT0–∞ (h) | 0.66 ± 0.076 | 5.64 ± 3.20 |
EAC bearing control group has shown an uncontrollable and significant increase in body weight. Standard (5-FU) and test (5-FU–PNA) group's injected with free 5-FU and 5-FU–PNA exhibited significant reduction in body weight when compared to control group. However when standard (5-FU) and test (5-FU–PNA) groups were compared with each other, test (5-FU–PNA) group treated with 5-FU–PNA Ng's has significantly delayed and controlled the increase in body weight of EAC bearing mice. Therefore, it can be said that the 5-FU–PNA Ng's prepared were highly effective in anti-cancer efficacy in contrast to its counterpart.
4. Conclusion
pH and thermo-sensitive Ng's with the earlier mentioned aim have been successfully developed by cross-linking monomers of NIPAM and AAc. Co-polymer ratio was altered and studied for their effect on pH dependent LCST. Ng's comprising of 97.16% molar of NIPAM and 2.84% molar of AAc were found to be a suitable candidate fitting under pre-mentioned objective and were loaded with 5-FU. In vitro drug release from 5-FU–PNA Ng's was found to be pH and thermo dependent. Pharmacokinetic data depicted that 5-FU PNA Ng's could sustain the drug release, and extend the half-life. In vivo anti-cancer studies has proven that 5-FU–PNA Ng's were highly effective in anti-cancer efficacy when compared with its counterpart free 5-FU, and MST has significantly improved when compared with control group.Acknowledgements
The author Naga Sravan Kumar Varma V., acknowledges JSS University, Mysuru for financial support through JSS University Junior Research fellowship (JRF) order no. REG/DIR(R)/JSSURF/29(1)/2013-14. Authors are also thankful to (A) Dr H. P. Ramesh, (Retired) Experimental Histopathologist, CSIR—Central Food Technological Research Institute, Mysuru, (B) Dr Hsieh-Chih Tsai, Associate Professor, National Taiwan University of Science and Technology, Taiwan, (C) Mr A. Harikrishna, TEM facility, Centre for Cellular and Molecular Biology, Hyderabad, (D) Mr Rudra Vaghela and Sandeep Kanna, Dept. of Pharmaceutics, JSS College of Pharmacy, Mysuru, (E) Mr Praven, and Mr Preren, Dept. Pharmacology, JSSCP, Mysuru, (F) Dr MVSST. Subbarao and Mr Venu, Dept. of Biochemistry, JSS Medical College, Mysuru, and (G) Dr Dattatri K. Nagesha, Head, Faculty of Life Sciences, JSS University, Mysuru, for their assistance in related to this work. Authors have no conflicts of interest.References
- World Cancer Report 2014, ed. B. W. Stewart and C. P. Wild, IARC, 2014 Search PubMed.
- D. B. Longley, D. P. Harkin and P. G. Johnston, Nat. Rev. Cancer, 2003, 3, 330–338 CrossRef CAS PubMed.
- H. I. El-Sayyad, M. F. Ismail, F. M. Shalaby, R. F. Abou-El-Magd, R. L. Gaur, A. Fernando, M. H. G. Raj and A. Ouhtit, Int. J. Biol. Sci., 2009, 5, 466–473 CrossRef CAS PubMed.
- J. S. Macdonald, Oncology, 1999, 13, 33–34 CAS.
- S. Rashid, N. Ali, S. Nafees, S. K. Hasan and S. Sultana, Food Chem. Toxicol., 2014, 66, 185–193 CrossRef CAS PubMed.
- S. A. Thomas, Z. Grami, S. Mehta and K. Patel, Cancer Cell Microenviron., 2016, 3 Search PubMed.
- R. Dhankhar, S. P. Vyas, A. K. Jain, S. Arora, G. Rath and A. K. Goyal, Artif. Cells, Blood Substitutes, Immobilization Biotechnol., 2010, 38, 230–249 CrossRef CAS PubMed.
- S. P. Egusquiaguirre, M. Igartua, R. M. Hernández and J. L. Pedraz, Clin. Transl. Oncol., 2012, 14, 83–93 CrossRef CAS PubMed.
- M. A. Firer and G. Gellerman, J. Hematol. Oncol., 2012, 5, 70 CrossRef CAS PubMed.
- V. Jain, S. Jain and S. C. Mahajan, Curr. Drug Delivery, 2015, 12, 177–191 CrossRef CAS.
- C. Xu and J. Wang, Asian J. Pharm. Sci., 2015, 10, 1–12 CrossRef.
- Y. Bae, N. Nishiyama, S. Fukushima, H. Koyama, M. Yasuhiro and K. Kataoka, Bioconjugate Chem., 2005, 16, 122–130 CrossRef CAS PubMed.
- G. Chang, C. Li, W. Lu and J. Ding, Macromol. Biosci., 2010, 10, 1248–1256 CrossRef CAS PubMed.
- F.-Q. Hu, Y.-Y. Zhang, J. You, H. Yuan and Y.-Z. Du, Mol. Pharm., 2012, 9, 2469–2478 CrossRef CAS PubMed.
- Y. Liu, W. Wang, J. Yang, C. Zhou and J. Sun, Asian J. Pharm. Sci., 2013, 8, 159–167 CrossRef.
- M. Oishi and Y. Nagasaki, Nanomedicine, 2010, 5, 451–468 CrossRef CAS PubMed.
- J. L. Wike-Hooley, J. Haveman and H. S. Reinhold, Radiother. Oncol., 1984, 2, 343–366 CrossRef CAS PubMed.
- L. E. Gerweck and K. Seetharaman, Cancer Res., 1996, 56, 1194–1198 CAS.
- Y. Kato, S. Ozawa, C. Miyamoto, Y. Maehata, A. Suzuki, T. Maeda and Y. Baba, Cancer Cell Int., 2013, 13, 89 CrossRef CAS PubMed.
- I. F. Tannock and D. Rotin, Cancer Res., 1989, 49, 4373–4384 CAS.
- M. A. Ward and T. K. Georgiou, Polymers, 2011, 3, 1215–1242 CrossRef CAS.
- S. Purushotham, P. E. J. Chang, H. Rumpel, I. H. C. Kee, R. T. H. Ng, P. K. H. Chow, C. K. Tan and R. V. Ramanujan, Nanotechnology, 2009, 20, 305101 CrossRef CAS PubMed.
- C. J. F. Rijcken, O. Soga, W. E. Hennink and C. F. van Nostrum, J. Controlled Release, 2007, 120, 131–148 CrossRef CAS PubMed.
- P. Bawa, V. Pillay, Y. E. Choonara and L. C. du Toit, Biomed. Mater., 2009, 4, 022001 CrossRef PubMed.
- J.-H. Kim and T. Randall Lee, Drug Dev. Res., 2006, 67, 61–69 CrossRef CAS.
- L. A. Perelman, T. Moore, J. Singelyn, M. J. Sailor and E. Segal, Adv. Funct. Mater., 2010, 20, 826–833 CrossRef CAS PubMed.
- W. Xiong, W. Wang, Y. Wang, Y. Zhao, H. Chen, H. Xu and X. Yang, Colloids Surf., B, 2011, 84, 447–453 CrossRef CAS PubMed.
- N. S. Rejinold, K. P. Chennazhi, S. V. Nair and R. Jayakumar, J. Nanopharmaceutics Drug Deliv., 2013, 1, 18–29 CrossRef.
- N. S. Rejinold, T. Baby, K. P. Chennazhi and R. Jayakumar, Colloids Surf., B, 2014, 114, 209–217 CrossRef CAS PubMed.
- T. Gan, Y. Guan and Y. Zhang, J. Mater. Chem., 2010, 20, 5937 RSC.
- A. Prasannan, H.-C. Tsai, Y.-S. Chen and G.-H. Hsiue, J. Mater. Chem. B, 2014, 2, 1988 RSC.
- R. V Joshi, C. E. Nelson, K. M. Poole, M. C. Skala and C. L. Duvall, Acta Biomater., 2013, 9, 6526–6534 CrossRef PubMed.
- Q. Wang, H. Xu, X. Yang and Y. Yang, Int. J. Pharm., 2008, 361, 189–193 CrossRef CAS PubMed.
- W. Xiong, X. Gao, Y. Zhao, H. Xu and X. Yang, Colloids Surf., B, 2011, 84, 103–110 CrossRef CAS PubMed.
- R. L. Sastre, M. D. Blanco, C. Teijón, R. Olmo and J. M. Teijón, Drug Dev. Res., 2004, 63, 41–53 CrossRef CAS.
- N. Kumar, I. Dhamija, P. Vasanth Raj, B. S. Jayashree, V. Parihar, S. N. Manjula, S. Thomas, N. Gopalan Kutty and C. Mallikarjuna Rao, Arabian J. Chem., 2014, 7, 409–417 CrossRef CAS.
- S. Yu, Z. Lü, Z. Chen, X. Liu, M. Liu and C. Gao, J. Membr. Sci., 2011, 371, 293–306 CrossRef CAS.
- R. A. Stile, W. R. Burghardt and K. E. Healy, Macromolecules, 1999, 32, 7370–7379 CrossRef CAS.
- S. D. Kong, A. Luong, G. Manorek, S. B. Howell and J. Yang, Bioconjugate Chem., 2007, 18, 293–296 CrossRef CAS PubMed.
- K.-N. Yang, C.-Q. Zhang, W. Wang, P. C. Wang, J.-P. Zhou and X.-J. Liang, Cancer Biol. Med., 2014, 11, 34–43 CAS.
- L. Zhu and V. P. Torchilin, Integr. Biol., 2013, 5, 96–107 RSC.
- J. Peng, T. Qi, J. Liao, B. Chu, Q. Yang, W. Li, Y. Qu, F. Luo and Z. Qian, Biomaterials, 2013, 34, 8726–8740 CrossRef CAS PubMed.
- J. C. Garbern, A. S. Hoffman and P. S. Stayton, Biomacromolecules, 2010, 11, 1833–1839 CrossRef CAS PubMed.
- J. Zhang, L.-Y. Chu, Y.-K. Li and Y. M. Lee, Polymer, 2007, 48, 1718–1728 CrossRef CAS.
- G. Bokias, G. Staikos and I. Iliopoulos, Polymer, 2000, 41, 7399–7405 CrossRef CAS.
- L.-Y. Chu, R. Xie, X.-J. Ju and W. Wang, Smart Hydrogel Functional Materials, Springer Berlin Heidelberg, Berlin, Heidelberg, 2013 Search PubMed.
- S. Sun, J. Hu, H. Tang and P. Wu, Phys. Chem. Chem. Phys., 2011, 13, 5061–5067 RSC.
- I. A. Hossain, J. A. Khanam, M. Jesmin and M. M. Ali, Exp. Toxicol. Pathol., 2016, 68, 15–25 CrossRef CAS PubMed.
- S. Qureshi, O. A. Al-Shabanah, A. M. Al-Bekairi, M. M. Al-Harbi, N. M. Al-Gharably and M. Raza, Invest. New Drugs, 2000, 18, 221–230 CrossRef CAS PubMed.
- S. M. Choudhury, M. Gupta and U. K. Majumder, Oxid. Med. Cell. Longevity, 2010, 3, 61–70 CrossRef PubMed.
- P. K. Samudrala, B. B. Augustine, E. R. Kasala, L. N. Bodduluru, C. Barua and M. Lahkar, Pharmacogn. Res., 2015, 7, 66–73 CrossRef PubMed.
- P. K. Haldar, B. Kar, A. Bala, S. Bhattacharya and U. K. Mazumder, Pharm. Biol., 2010, 48, 1337–1343 CrossRef PubMed.
- B. R. von Beust, Lab. Anim., 2010, 44, 379–380 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra18034e |
| This journal is © The Royal Society of Chemistry 2016 |