Diastereoselective synthesis of novel tetra-and pentacyclic annulated coumarino-δ-sultam pyrrolidine, pyrrolizidine, pyrrolothiazole and isoxazolidine derivatives via intramolecular 1,3-dipolar cycloadditions†
Mehdi Ghandi*,
Nahid Zarezadeh and
Alireza Abbasi
School of Chemistry, College of Science, University of Tehran, P.O. Box 14155 6455, Tehran, Iran. E-mail: ghandi@khayam.ut.ac.ir
First published on 11th July 2016
Abstract
A facile and efficient strategy towards the novel tetra- and pentacyclic annulated coumarino-δ-sultam pyrrolidine, pyrrolizidine, pyrrolothiazole and isoxazolidine via intramolecular 1,3-dipolar cycloadditions of nitrones or azomethine ylides is described. These highly functionalized polycyclic scaffolds were obtained with high diastereoselectivity in high yields via condensation of the initially prepared coumarin-based bifunctional starting materials with sarcosine or N-phenylglycine, L-proline or L-4-thiazolidinecarboxylic acid, and methyl or benzylhydroxylamine. The proof of the structures relies on analytical investigation and X-ray crystallography.
Introduction
In the domain of cycloaddition,1 intramolecular 1,3-dipolar cycloaddition of azomethine ylides2 and nitrones3 with alkenes on carbo- and heterocyclic systems is one of the most efficient and reliable strategies for the construction of functionalized and stereoisomerically pure five-membered pyrrolidines, pyrrolizidines, pyrrolothiazoles and isoxazolidines. As valuable building blocks, the functionalized derivatives of pyrrolidines,4 pyrrolizidines5 and pyrrolothiazoles6 have outstanding application in the synthesis of natural compounds and pharmaceuticals. On the other hand, the isoxazolidines are valuable precursors, owing to the labile nature of the N–O bond which can be easily modified to biologically active β-amino acids, β-lactams, amino sugars, and alkaloids.7 Thus, it is not surprising that development of synthetic routes to pyrrolidine-, pyrrolizidine-, pyrrolothiazole-, and isoxazolidine-containing heterocycles is an interesting challenge in organic synthesis.Whereas intermolecular 1,3-dipolar cycloadditions employing azomethine ylides8 or nitrones9 lead to substituted pyrrolidines/pyrrolizines/pyrrolothiazoles and isoxazolidines, respectively, in contrast, intramolecular 1,3-dipolar cycloadditions give annulated polycyclic ring analogs.2,3 To access this goal, attention has been dedicated toward synthesis of systems in which both reacting components are tethered to the starting material so that polycyclic molecular architecture can be constructed via intramolecular addition of the in situ generated azomethine ylide or nitrone to the dipolarophile partner. So, design and synthesis of a wide variety of polycyclic scaffolds has been carried out by changing the length or position of the tether between the two reaction centers.
Sulfonamides have a rich chemical and biological history and are an important class of compounds in drug discovery due to their extensive chemical and biological activities.10 Significant interest has been directed toward cyclic sulfonamides, also known as sultams. Although not found in nature,11 sultams are known as privileged structures in drug discovery due to their diverse biological properties.12 Examples include the ampiroxicam (COX-2) anti-inflammatory agent,13 calpain inhibitor 1,14 benzodithiazine dioxides displaying anti-HIV-1 activity,15 antiepileptic agent sulthiame,16 brinzolamide for the treatment of glaucoma,17 and MMP-2 inhibitor.18 Sultams with promising bioactivities such as antiviral,19 anticancer,20 antimicrobial,21 antimalarial,22 and antileukemic,23 are also known. As chemically important materials, sultams have been utilized as efficient chiral auxiliaries and reagents.24
On the other hand, coumarin derivatives have been the subject of recent excellent reviews25a,b due to the presence of this core structure in many pharmaceuticals and biologically active natural compounds with a wide spectrum of considerable biological activities such as anti-inflammatory,25c,d anticancer,25e–g antioxidant,25h and antituberculosis.25i
Given that the synthesis of fused triheterocycles encompassing bioactive coumarin, δ-sultam and five-membered pyrrolidine, pyrrolizidine, pyrrolothiazole or isoxazolidine motifs seemed to be interesting due to their individual widespread known biological activities and uses, we undertook a study of the synthesis of heterocyclic scaffolds containing three motifs into one molecule. In continuation of our previous research based on the cyclization reactions,26 as well as sultam scaffold,27 herein, we report an efficient protocol for the synthesis of novel tetra and pentacyclic coumarin-based δ-sultam annulated pyrrolidine, pyrrolizine, pyrrolothiazole and isoxazolidine scaffolds through in situ formation of nitrones or azomethine ylides followed by an intramolecular 1,3-dipolar cycloaddition reaction sequence using coumarin-based biofunctional starting materials. It is noticeable that two/three new rings and three/four contiguous stereocenters with high regio- and stereoselectivity are efficiently created in a single step in this procedure.
Results and discussion
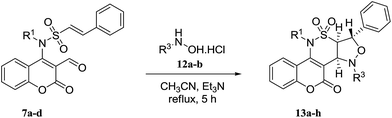
Initially, 4-chloro-3-formyl coumarin (2)28 and (E)-N-alkyl-2-phenylethenesulfonamide 6a–d29 were prepared using the previously reported procedure (Scheme 1a). In the next step, four derivatives of (E)-N-(3-formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-alkylethenesulfonamides 7a–d were synthesized using the method depicted in Scheme 1b. | ||
| Scheme 1 Preparation of (a) aldehyde 2 and (E)-N-alkyl-2-phenylethenesulfonamide 6a–d, and (b) (E)-N-(3-formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-alkylethenesulfonamides 7a–d. | ||
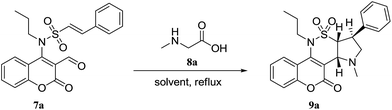
(E)-N-(3-Formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-propylethenesulfonamide (7a) served for our early exploration of 1,3-dipolar cycloaddition reaction. Indeed, 9a was isolated as the sole reaction product in 72% upon treatment of 7a with an equimolar amount of sarcosine 8a in EtOH within 10 h (entry 1, Table 1). Whereas reaction in MeOH and toluene occurred more efficiently (entries 2 and 3, Table 1), it was sluggish in DCE and DCM (entries 4 and 5, Table 1). It was discovered that reaction proceeded to completion within 5 h in MeCN, affording 9a in 96% yield (entry 6, Table 1).
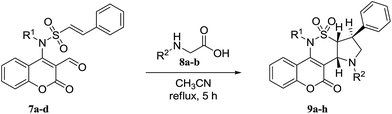
This new method was then applied to a range of 7a–d, sarcosine or N-phenylglycine 8a,b in MeCN at reflux for 5 h. Tetracyclic coumarino-δ-sultam annulated pyrrolidines 9a–h were obtained in excellent yields (Table 2).
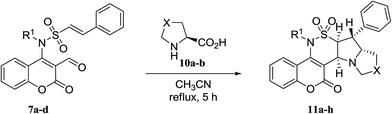
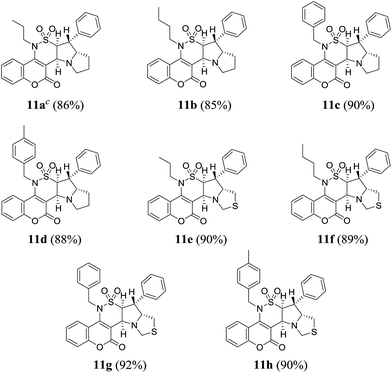
To explore the generality and the substrate scope of the developed condensation, sarcosine and N-phenylglycine 8a,b were replaced with L-proline or L-4-thiazolidinecarboxylic acid 10a,b. The corresponding pyrrolizine and thiopyrrolizine products 11a–h were obtained in satisfactory yields (Table 3).
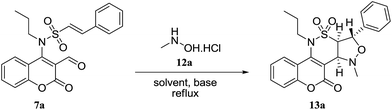
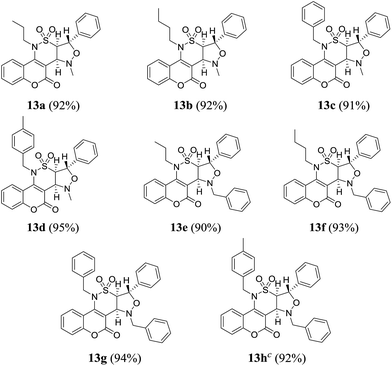
Inspired by these results, we decided to study the 1,3-dipolar cycloaddition reaction of (E)-N-(3-formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-alkylethenesulfonamides 7a–d with N-methyl or N-benzylhydroxylamine hydrochlorides 12a,b. To prepare 13a, (E)-N-(3-formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-propylethene sulfonamide (7a) was treated with N-methylhydroxylamine hydrochloride 12a and potassium carbonate in methanol at reflux. The isoxazolidine 13a was obtained in 54% yield within 15 h (entry 1, Table 4). When the reaction was repeated in ethanol and acetonitrile, isoxazolidine 13a was obtained in 56% and 73% during 15 h and 10 h, respectively (entries 2 and 3, Table 4). Changing the base to cesium carbonate showed comparable result in acetonitrile and lesser yield in toluene (entries 4 and 5, Table 4). It was shown that reaction with sodium methoxide is sluggish in acetonitrile (entry 6, Table 4). Triethylamine proved to be an excellent base especially in acetonitrile on the basis of the obtained results (entries 7 to 10, Table 4). Encouraged by this result, aldehydes 7a–d were then treated with N-methyl or N-benzylhydroxylamine hydrochlorides 12a,b, which successfully yielded desired derivatives 13a–h (Table 5).
| Entry | Solvent | Base | Time (h) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: all reactions were carried out using aldehyde (1 mmol), N-methyl hydroxylamine hydrochloride (1.1 mmol), and base (1.1 mmol) with 4 Å MS and 15 mL of solvent at reflux.b Isolated yields. | ||||
| 1 | MeOH | K2CO3 | 15 | 54 |
| 2 | EtOH | K2CO3 | 15 | 56 |
| 3 | MeCN | K2CO3 | 10 | 73 |
| 4 | MeCN | Cs2CO3 | 10 | 71 |
| 5 | Toluene | Cs2CO3 | 10 | 68 |
| 6 | MeCN | NaOMe | 15 | 52 |
| 7 | EtOH | Et3N | 10 | 68 |
| 8 | MeOH | Et3N | 10 | 70 |
| 9 | Toluene | Et3N | 7 | 81 |
| 10 | MeCN | Et3N | 5 | 92 |
Compounds 9a–h, 11a–h and 13a–h were characterized by elemental analysis, MS, IR, and 1H and 13C NMR spectroscopy. Unambiguous evidence for the proposed structures of 9h,30 11a31 and 13h32 was finally obtained by single crystal X-ray-diffraction analysis and the ORTEP diagrams are shown in Fig. 1–3.
Although the precise mechanism is not known, a mechanistic postulate as shown in Scheme 2 may be invoked to rationalize the diastereoselective formation of cis–trans 9a–h, 11a–h and 13a–h, respectively. The dipolar cycloaddition of an azomethine ylide or nitrone generated from sarcosine, methyl or benzylhydroxylamine and an aldehyde with a dipolarophile could lead to a mixture of stereoisomers. Based on the crystal structures of compounds 9h, 11a and 13h, the relative stereochemistry of the Ph group and the adjacent H atom is in the anti orientation, presumably due to the initial E geometry of the double bond present in compounds 7a–d. Since the trans stereochemistry of the alkene dipolarophiles 7a–d has been maintained in pyrrolidines 9a–h, pyrrolizidines 11a–d, thiopyrrolizines 11e–h and isoxazolidines 13a–h (see Table 2, 3 and 5), the cycloadditions have proceeded concerted with both carbon–carbon σ-bonds being formed at the same time, although not necessarily to the same extent. It seems justified that the cis–trans and trans–trans annealated products are expected to be formed via endo-E-ylide I or endo-Z-nitrone I′ transition states and exo-Z-ylide II or exo-E-nitrone II′ transition states, respectively (Scheme 2a and b). The highly diastereoselective formation of cis–trans pyrrolidines 9a–h and isoxazolidines 13a–h reveals the implication of the endo-E-ylide I or endo-Z-nitrone I′ transition states, perhaps due to the more favorable π-interactions in the 1,3-dipolar cycloaddition reaction. Since both endo-E-ylide I or endo-Z-nitrone I′ transition states correspond to an endo-orientation of the substituent Ph and exo-orientation of the rather bulkier coumarin substituent on the dipolarophile, reactions in these cases proceed faster in comparison to those passing respectively through exo-Z-ylide II or exo-E-nitrone II′ transition states.
 | ||
| Scheme 2 Suggested mechanism for the formation of (a) pyrrolidines 9a–h, (b) isoxazolidines 13a–h and (c) pyrrolizidines and thiopyrrolizines 11a–h. | ||
Notably, the dipolar cycloaddition reaction of an azomethine ylide generated from L-proline or L-4-thiazolidinecarboxylic acid and an aldehyde with a dienophile could lead to a mixture of endo-S-shaped III or exo-W-shaped III′ ylides, respectively (Scheme 2c). Inspection of other reported examples together with highly diastereoselective formation of cis–trans pyrrolizidines 11a–d and thiopyrrolizines 11e–h in this presentation perhaps shows some general preference for cycloaddition through an endo-S-shaped ylide III (Scheme 2c).
Overall, novel tetra-and pentacyclic annulated coumarino-δ-sultam pyrrolidine, pyrrolizidine, pyrrolothiazole and isoxazolidine derivatives were obtained via highly diastereoselective processes. These reactions have presumably been proceeded intramolecularly via the in situ generated azomethine or nitrone ylides, affording the desired products in excellent yields (Table 2, 3 and 5). As such, it can be concluded that azomethine and nitrone ylide intermediates react with dipolarophile with the least decomposition of the ylides under the reaction conditions. It implies in turn that both azomethine and nitrone ylides are rather stable although the nature of the tethered RN–SO2–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHPh group as a good dipolarophile may not be overlooked.
CHPh group as a good dipolarophile may not be overlooked.
Conclusions
In conclusion, a number of bifunctional (E)-N-(3-formyl-2-oxo-2H-chromen-4-yl)-2-phenyl-N-alkylethenesulfonamides 7a–d were initially prepared from 4-chloro-3-formyl coumarin (2) and (E)-N-alkyl-2-phenylethenesulfonamide 6a–d. These starting materials underwent reactions with secondary amino acids or N-substituted methyl or benzylhydroxylamines in acetonitrile at reflux within 5 h. A variety of pyrrolidines 9a–h, pyrrolizidines 11a–h and isoxazolidines 13a–h were obtained in excellent yields. These reactions were designed to initially generate in situ an azomethine or nitrone ylides to accomplish an intramolecular 1,3-dipolar cycloadditions. These new structures broaden the scaffolds that are accessible through 1,3-dipolar cycloadditions reactions and many of them may represent interesting pharmacophores.Experimental
General information
All commercially available chemicals and reagents were purchased from Merck Chemical Company and used without further purification. Melting points were determined with an Electrothermal model 9100 apparatus and are uncorrected. IR spectra were recorded on a Shimadzu 4300 spectrophotometer, in cm−1. 1H and 13C NMR spectra were recorded on a Bruker DRX-500-AVANCE spectrometer at 300 (1H) and 75 MHz (13C) using CDCl3 as solvent. Mass spectra of the products were obtained with an HP (Agilent technologies) 5937 Mass Selective Detector. Elemental analyses were carried out by a CHN-Rapid Heraeus elemental analyzer (Wellesley, MA).General procedure for the synthesis of compounds 6a–d
To a stirring solution of (E)-2-phenylethenesulfonyl chloride 4 (10 mmol) in CH3CN (30 mL) were added primary amines 5a–d (10 mmol) and K2CO3 (1.38 g, 10 mmol) and the reaction mixture was stirred at room temperature (25 °C) for 24 h. After completion of the reaction as indicated by TLC, the solid was filtered, and the filtrate was concentrated under reduced pressure to afford products 6a–d as yellowish oil or white solid.General procedure for the synthesis of compounds 7a–d
To an ice-cooled solution of (E)-N-alkyl-2-phenylethenesulfonamide derivatives 6a–d (10 mmol) in DMF (20 mL) was added 60% sodium hydride in mineral oil (0.88 g, 22 mmol), and the mixture was stirred for 30 min at room temperature. Subsequently, 4-chloro-3-formyl coumarin 2 (2.08 g, 10 mmol) was added, and the mixture was stirred for 2 h at room temperature. After completion of the reaction as indicated by TLC, the reaction mixture was poured into ice-water with constant stirring. The mixture was then extracted with methylene chloride for three times, and the organic phase was washed with water and brine solution, and finally dried over Na2SO4. The solvent was then evaporated under reduced pressure, and the crude product was purified by column chromatography (SiO2, eluent: 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, n-hexane/EtOAc) to afford products 7a–d as yellow solids.
1, n-hexane/EtOAc) to afford products 7a–d as yellow solids.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1343 (–SO2), 1142 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.47; 1H NMR (300 MHz, CDCl3) δ 0.85 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.46–1.55 (m, 1H, MeCHHCH2N), 1.69–1.79 (m, 1H, MeCHHCH2N), 3.38 (ddd, J = 14.6, 11.0, 5.3 Hz, 1H, MeCH2CHHN), 3.73 (ddd, J = 14.5, 11.3, 5.4 Hz, 1H, MeCH2CHHN), 6.93 (d, J = 15.3 Hz, 1H, SO2CH), 7.39–7.62 (8H, m, Ar and PhCH), 7.73 (td, J = 8.5, 1.4 Hz, 1H, Ar), 8.21 (dd, J = 8.1, 1.3 Hz, 1H, Ar), 10.35 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 11.1, 22.8, 54.1, 116.9, 118.6, 120.7, 124.8, 125.2, 128.5 (2C), 129.1 (2C), 129.3, 131.1, 132.2, 135.3, 143.3, 154.0, 154.3, 161.1, 188.0 ppm; m/z (EI, 70 eV) 398 (19, M+ + 1), 333 (25), 304 (74), 230 (100), 212 (32), 200 (15), 186 (25), 175 (8), 167 (14), 161 (30), 146 (8), 119 (25), 103 (89), 91 (26), 77 (46%); anal. calcd for C21H19NO5S: C, 63.46; H, 4.82; N, 3.52. Found: C, 63.23; H, 4.91; N, 3.34%.
O), 1343 (–SO2), 1142 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.47; 1H NMR (300 MHz, CDCl3) δ 0.85 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.46–1.55 (m, 1H, MeCHHCH2N), 1.69–1.79 (m, 1H, MeCHHCH2N), 3.38 (ddd, J = 14.6, 11.0, 5.3 Hz, 1H, MeCH2CHHN), 3.73 (ddd, J = 14.5, 11.3, 5.4 Hz, 1H, MeCH2CHHN), 6.93 (d, J = 15.3 Hz, 1H, SO2CH), 7.39–7.62 (8H, m, Ar and PhCH), 7.73 (td, J = 8.5, 1.4 Hz, 1H, Ar), 8.21 (dd, J = 8.1, 1.3 Hz, 1H, Ar), 10.35 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 11.1, 22.8, 54.1, 116.9, 118.6, 120.7, 124.8, 125.2, 128.5 (2C), 129.1 (2C), 129.3, 131.1, 132.2, 135.3, 143.3, 154.0, 154.3, 161.1, 188.0 ppm; m/z (EI, 70 eV) 398 (19, M+ + 1), 333 (25), 304 (74), 230 (100), 212 (32), 200 (15), 186 (25), 175 (8), 167 (14), 161 (30), 146 (8), 119 (25), 103 (89), 91 (26), 77 (46%); anal. calcd for C21H19NO5S: C, 63.46; H, 4.82; N, 3.52. Found: C, 63.23; H, 4.91; N, 3.34%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1347 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 0.83 (t, J = 7.3 Hz, 3H, MeCH2CH2CH2N), 1.19–1.28 (m, 2H, MeCHHCH2CH2N), 1.40–1.45 (m, 1H, MeCH2CHHCH2N), 1.65–1.69 (m, 1H, MeCH2CHHCH2N), 3.40 (ddd, J = 14.6, 11.0, 5.2 Hz, 1H, MeCH2CH2CHHN), 3.71–3.79 (ddd, J = 14.5, 11.0, 5.5 Hz, 1H, MeCH2CH2CHHN), 6.91 (d, J = 15.3 Hz, 1H, SO2CH), 7.38–7.60 (8H, m, Ar and PhCH), 7.72 (td, J = 8.5, 1.3 Hz, 1H, Ar), 8.19 (dd, J = 8.1, 1.2 Hz, 1H, Ar), 10.33 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 13.5, 19.9, 31.5, 52.3, 117.0, 118.6, 120.9, 124.9, 125.3, 128.5 (2C), 129.1 (2C), 129.4, 131.2, 132.3, 135.3, 143.4, 154.1, 154.4, 161.2, 188.1 ppm; m/z (EI, 70 eV) 412 (11, M+ + 1), 347 (21), 318 (54), 244 (100), 226 (45), 214 (15), 200 (12), 186 (36), 175 (16), 167 (14), 161 (4), 146 (11), 119 (19), 103 (90), 77 (51%); anal. calcd for C22H21NO5S: C, 64.22; H, 5.14; N, 3.40. Found: C, 64.25; H, 5.14; N, 3.51%.
O), 1347 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 0.83 (t, J = 7.3 Hz, 3H, MeCH2CH2CH2N), 1.19–1.28 (m, 2H, MeCHHCH2CH2N), 1.40–1.45 (m, 1H, MeCH2CHHCH2N), 1.65–1.69 (m, 1H, MeCH2CHHCH2N), 3.40 (ddd, J = 14.6, 11.0, 5.2 Hz, 1H, MeCH2CH2CHHN), 3.71–3.79 (ddd, J = 14.5, 11.0, 5.5 Hz, 1H, MeCH2CH2CHHN), 6.91 (d, J = 15.3 Hz, 1H, SO2CH), 7.38–7.60 (8H, m, Ar and PhCH), 7.72 (td, J = 8.5, 1.3 Hz, 1H, Ar), 8.19 (dd, J = 8.1, 1.2 Hz, 1H, Ar), 10.33 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 13.5, 19.9, 31.5, 52.3, 117.0, 118.6, 120.9, 124.9, 125.3, 128.5 (2C), 129.1 (2C), 129.4, 131.2, 132.3, 135.3, 143.4, 154.1, 154.4, 161.2, 188.1 ppm; m/z (EI, 70 eV) 412 (11, M+ + 1), 347 (21), 318 (54), 244 (100), 226 (45), 214 (15), 200 (12), 186 (36), 175 (16), 167 (14), 161 (4), 146 (11), 119 (19), 103 (90), 77 (51%); anal. calcd for C22H21NO5S: C, 64.22; H, 5.14; N, 3.40. Found: C, 64.25; H, 5.14; N, 3.51%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1336 (–SO2), 1142 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 4.60 (d, J = 14.3 Hz, PhCHHN), 4.97 (d, J = 14.3 Hz, PhCHHN), 7.01 (d, J = 15.4 Hz, 1H, SO2CH), 7.17–7.65 (14H, m, Ar and PhCH), 7.87 (d, J = 8.0 Hz, 1H, Ar), 10.22 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 55.4, 116.7, 119.1, 120.3, 124.9, 125.1, 128.6 (4C), 128.7, 129.2 (2C), 129.6 (2C), 131.3, 132.2, 133.8, 135.1, 143.7, 153.4, 154.2, 161.0, 188.2 ppm; m/z (EI, 70 eV) 446 (3, M+ + 1), 381 (8), 352 (9), 278 (92), 262 (5), 248 (6), 220 (4), 167 (4), 103 (31), 91 (100%); anal. calcd for C25H19NO5S: C, 67.40; H, 4.30; N, 3.14. Found: C, 67.65; H, 4.37; N, 3.03%.
O), 1336 (–SO2), 1142 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 4.60 (d, J = 14.3 Hz, PhCHHN), 4.97 (d, J = 14.3 Hz, PhCHHN), 7.01 (d, J = 15.4 Hz, 1H, SO2CH), 7.17–7.65 (14H, m, Ar and PhCH), 7.87 (d, J = 8.0 Hz, 1H, Ar), 10.22 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 55.4, 116.7, 119.1, 120.3, 124.9, 125.1, 128.6 (4C), 128.7, 129.2 (2C), 129.6 (2C), 131.3, 132.2, 133.8, 135.1, 143.7, 153.4, 154.2, 161.0, 188.2 ppm; m/z (EI, 70 eV) 446 (3, M+ + 1), 381 (8), 352 (9), 278 (92), 262 (5), 248 (6), 220 (4), 167 (4), 103 (31), 91 (100%); anal. calcd for C25H19NO5S: C, 67.40; H, 4.30; N, 3.14. Found: C, 67.65; H, 4.37; N, 3.03%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1347 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.22 (s, 3H, Me), 4.60 (d, J = 14.2 Hz, PhCHHN), 4.90 (d, J = 14.2 Hz, PhCHHN), 6.98–7.04 (5H, m, Ar and SO2CH), 7.26–7.65 (9H, m, Ar and PhCH), 7.92 (dd, J = 8.3, 1.4 Hz, 1H, Ar), 10.18 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 21.1, 55.1, 116.8, 119.3, 120.3, 124.96, 125.03, 128.6 (2C), 129.2 (2C), 129.3 (2C), 129.5 (3C), 130.7, 131.3, 132.3, 135.1, 138.6, 143.7, 153.5, 154.2, 160.9, 188.2 ppm; m/z (EI, 70 eV) 460 (3, M+ + 1), 395 (2), 354 (1), 292 (90), 276 (3), 263 (7), 248 (4), 235 (3), 207 (4), 167 (6), 146 (3), 120 (5), 105 (100), 77 (31%); anal. calcd for C26H21NO5S: C, 67.96; H, 4.61; N, 3.05. Found: C, 67.78; H, 5.68; N, 2.90%.
O), 1347 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.22 (s, 3H, Me), 4.60 (d, J = 14.2 Hz, PhCHHN), 4.90 (d, J = 14.2 Hz, PhCHHN), 6.98–7.04 (5H, m, Ar and SO2CH), 7.26–7.65 (9H, m, Ar and PhCH), 7.92 (dd, J = 8.3, 1.4 Hz, 1H, Ar), 10.18 (s, 1H, CHO) ppm; 13C NMR (75 MHz, CDCl3) δ 21.1, 55.1, 116.8, 119.3, 120.3, 124.96, 125.03, 128.6 (2C), 129.2 (2C), 129.3 (2C), 129.5 (3C), 130.7, 131.3, 132.3, 135.1, 138.6, 143.7, 153.5, 154.2, 160.9, 188.2 ppm; m/z (EI, 70 eV) 460 (3, M+ + 1), 395 (2), 354 (1), 292 (90), 276 (3), 263 (7), 248 (4), 235 (3), 207 (4), 167 (6), 146 (3), 120 (5), 105 (100), 77 (31%); anal. calcd for C26H21NO5S: C, 67.96; H, 4.61; N, 3.05. Found: C, 67.78; H, 5.68; N, 2.90%.General procedure for the synthesis of compounds 9a–d and 11a–h
A mixture of aldehyde derivatives 7a–d (1 mmol) and acyclic secondary amino acids 8a,b or cyclic secondary amino acids 10a,b (1.1 mmol) in MeCN (15 mL) containing molecular sieves (500 mg, 4 Å) was heated at reflux with stirring for 5 h. After completion of the reaction as indicated by TLC, the solvent was removed under reduced pressure and the residue was recrystallized from 30% EtOAc in hexane to afford the products 9a–h and 11a–h.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1350 (–SO2), 1145 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.47; 1H NMR (300 MHz, CDCl3) δ 0.97 (t, 3H, J = 7.4 Hz, MeCH2CH2N), 1.85–2.01 (m, 2H, MeCHHCH2N), 2.38 (s, 3H, MeN), 2.61 (dd, J = 10.1, 9.0 Hz, 1H, MeNCHH), 3.55–3.70 {[2H, consisting dd, J = 8.3, 7.9 Hz, 1H (3.58), SO2CH]; and ddd, J = 14.2, 10.9, 5.5 Hz, 1H (3.65), MeCH2CHHN}, 3.97–4.10 {[ddd, J = 14.2, 10.9, 6.0 Hz, 1H (4.02), MeCH2CHHN]; and dd, J = 9.0, 6.5 Hz, 1H (4.08), MeNCHH}, 4.16–4.29 {[2H, consisting ddd, J = 10.1, 7.9, 7.9 Hz, 1H (4.20), PhCH]; and d, J = 8.3 Hz, 1H (4.27), CHN}, 7.28–7.41 (m, 7H, Ar), 7.63 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.81 (dd, J = 8.0, 1.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.4, 23.8, 39.4, 47.6, 56.5, 63.0, 63.5, 72.4, 117.2, 118.0, 119.0, 124.5, 124.8, 127.5, 127.7 (2C), 129.0 (2C), 133.2, 140.0, 153.8, 155.6, 161.5 ppm; m/z (EI, 70 eV) 425 (12, M+), 360 (9), 345 (17), 331 (14), 317 (41), 302 (8), 288 (28), 274 (16), 256 (100), 240 (20), 226 (53), 214 (41), 198 (29), 133 (61), 115 (19), 104 (14), 91 (25%); anal. calcd for C23H24N2O4S: C, 65.07; H, 5.70; N, 6.60. Found: C, 64.98; H, 5.72; N, 6.61%.
O), 1350 (–SO2), 1145 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.47; 1H NMR (300 MHz, CDCl3) δ 0.97 (t, 3H, J = 7.4 Hz, MeCH2CH2N), 1.85–2.01 (m, 2H, MeCHHCH2N), 2.38 (s, 3H, MeN), 2.61 (dd, J = 10.1, 9.0 Hz, 1H, MeNCHH), 3.55–3.70 {[2H, consisting dd, J = 8.3, 7.9 Hz, 1H (3.58), SO2CH]; and ddd, J = 14.2, 10.9, 5.5 Hz, 1H (3.65), MeCH2CHHN}, 3.97–4.10 {[ddd, J = 14.2, 10.9, 6.0 Hz, 1H (4.02), MeCH2CHHN]; and dd, J = 9.0, 6.5 Hz, 1H (4.08), MeNCHH}, 4.16–4.29 {[2H, consisting ddd, J = 10.1, 7.9, 7.9 Hz, 1H (4.20), PhCH]; and d, J = 8.3 Hz, 1H (4.27), CHN}, 7.28–7.41 (m, 7H, Ar), 7.63 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.81 (dd, J = 8.0, 1.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.4, 23.8, 39.4, 47.6, 56.5, 63.0, 63.5, 72.4, 117.2, 118.0, 119.0, 124.5, 124.8, 127.5, 127.7 (2C), 129.0 (2C), 133.2, 140.0, 153.8, 155.6, 161.5 ppm; m/z (EI, 70 eV) 425 (12, M+), 360 (9), 345 (17), 331 (14), 317 (41), 302 (8), 288 (28), 274 (16), 256 (100), 240 (20), 226 (53), 214 (41), 198 (29), 133 (61), 115 (19), 104 (14), 91 (25%); anal. calcd for C23H24N2O4S: C, 65.07; H, 5.70; N, 6.60. Found: C, 64.98; H, 5.72; N, 6.61%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1351 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 0.95 (t, 3H, J = 7.3 Hz, MeCH2CH2CH2N), 1.27–1.48 (m, 2H, MeCHHCH2CH2N), 1.78–1.99 (m, 2H, MeCH2CHHCH2N), 2.38 (s, 3H, MeN), 2.61 (dd, J = 10.1, 9.0 Hz, 1H, MeNCHH), 3.57 (dd, J = 8.6, 7.5 Hz, 1H, SO2CH), 3.70 (ddd, J = 14.2, 11.1, 5.1 Hz, 1H, MeCH2CH2CHHN), 4.02–4.12 {[2H, consisting m, MeCH2CH2CHHN]; and dd, J = 9.0, 6.4 Hz, 1H (4.08), MeNCHH}, 4.17–4.29 {[2H, consisting ddd, J = 10.1, 7.5, 7.5 Hz, 1H (4.20), PhCH]; and d, J = 8.6 (4.28) Hz, 1H, CHN}, 7.28–7.41 (m, 7H, Ar), 7.63 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.81 (dd, J = 8.0, 1.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.2, 32.4, 39.4, 47.7, 54.7, 62.9, 63.5, 72.4, 117.2, 118.1, 119.0, 124.5, 124.8, 127.5, 127.7 (2C), 129.0 (2C), 133.2, 140.0, 153.8, 155.7, 161.4 ppm; m/z (EI, 70 eV) 439 (12, M+), 374 (6), 359 (18), 331 (29), 317 (15), 302 (14), 288 (25), 270 (100), 254 (16), 240 (65), 228 (70), 214 (29), 198 (47), 170 (10), 158 (14), 143 (12), 133 (69), 115 (37), 104 (30), 91 (49%); anal. calcd for C24H26N2O4S: C, 65.73; H, 5.98; N, 6.39. Found: C, 65.62; H, 5.95; N, 6.40%.
O), 1351 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 0.95 (t, 3H, J = 7.3 Hz, MeCH2CH2CH2N), 1.27–1.48 (m, 2H, MeCHHCH2CH2N), 1.78–1.99 (m, 2H, MeCH2CHHCH2N), 2.38 (s, 3H, MeN), 2.61 (dd, J = 10.1, 9.0 Hz, 1H, MeNCHH), 3.57 (dd, J = 8.6, 7.5 Hz, 1H, SO2CH), 3.70 (ddd, J = 14.2, 11.1, 5.1 Hz, 1H, MeCH2CH2CHHN), 4.02–4.12 {[2H, consisting m, MeCH2CH2CHHN]; and dd, J = 9.0, 6.4 Hz, 1H (4.08), MeNCHH}, 4.17–4.29 {[2H, consisting ddd, J = 10.1, 7.5, 7.5 Hz, 1H (4.20), PhCH]; and d, J = 8.6 (4.28) Hz, 1H, CHN}, 7.28–7.41 (m, 7H, Ar), 7.63 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.81 (dd, J = 8.0, 1.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.2, 32.4, 39.4, 47.7, 54.7, 62.9, 63.5, 72.4, 117.2, 118.1, 119.0, 124.5, 124.8, 127.5, 127.7 (2C), 129.0 (2C), 133.2, 140.0, 153.8, 155.7, 161.4 ppm; m/z (EI, 70 eV) 439 (12, M+), 374 (6), 359 (18), 331 (29), 317 (15), 302 (14), 288 (25), 270 (100), 254 (16), 240 (65), 228 (70), 214 (29), 198 (47), 170 (10), 158 (14), 143 (12), 133 (69), 115 (37), 104 (30), 91 (49%); anal. calcd for C24H26N2O4S: C, 65.73; H, 5.98; N, 6.39. Found: C, 65.62; H, 5.95; N, 6.40%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1361 (–SO2), 1143 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 2.40 (s, 3H, MeN), 2.59 (dd, J = 9.6, 8.5 Hz, 1H, MeNCHH), 3.51 (dd, J = 8.4, 7.6 Hz, 1H, SO2CH), 4.06 (dd, J = 8.5, 6.2 Hz, 1H, MeNCHH), 4.17 (ddd, J = 9.6, 7.6, 7.6 Hz, 1H, PhCH), 4.31 (d, J = 8.4 Hz, 1H, CHN), 4.73 (d, J = 14.9 Hz, 1H, PhCHHN), 5.45 (d, J = 14.9 Hz, 1H, PhCHHN), 6.97 (t, J = 7.6 Hz, 1H, Ar), 7.16 (t, J = 7.8 Hz, 1H, Ar), 7.27–7.37 (m, 11H, Ar), 7.48 (t, J = 7.7 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 39.4, 47.5, 58.9, 63.2, 63.5, 72.0, 116.7, 117.8, 119.0, 124.3, 125.4, 127.5, 127.7 (2C), 128.4, 128.6 (2C), 129.0 (2C), 129.4 (2C), 133.0, 135.9, 140.0, 153.6, 156.1, 161.6 ppm; m/z (EI, 70 eV) 473 (0.8, M+), 408 (3), 393 (16), 381 (23), 365 (22), 317 (19), 304 (92), 288 (8), 274 (53), 261 (19), 230 (13), 213 (58), 158 (8), 133 (38), 115 (21), 104 (12), 91 (100%); anal. calcd for C27H24N2O4S: C, 68.62; H, 5.12; N, 5.93. Found: C, 68.61; H, 5.08; N, 5.95%.
O), 1361 (–SO2), 1143 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 2.40 (s, 3H, MeN), 2.59 (dd, J = 9.6, 8.5 Hz, 1H, MeNCHH), 3.51 (dd, J = 8.4, 7.6 Hz, 1H, SO2CH), 4.06 (dd, J = 8.5, 6.2 Hz, 1H, MeNCHH), 4.17 (ddd, J = 9.6, 7.6, 7.6 Hz, 1H, PhCH), 4.31 (d, J = 8.4 Hz, 1H, CHN), 4.73 (d, J = 14.9 Hz, 1H, PhCHHN), 5.45 (d, J = 14.9 Hz, 1H, PhCHHN), 6.97 (t, J = 7.6 Hz, 1H, Ar), 7.16 (t, J = 7.8 Hz, 1H, Ar), 7.27–7.37 (m, 11H, Ar), 7.48 (t, J = 7.7 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 39.4, 47.5, 58.9, 63.2, 63.5, 72.0, 116.7, 117.8, 119.0, 124.3, 125.4, 127.5, 127.7 (2C), 128.4, 128.6 (2C), 129.0 (2C), 129.4 (2C), 133.0, 135.9, 140.0, 153.6, 156.1, 161.6 ppm; m/z (EI, 70 eV) 473 (0.8, M+), 408 (3), 393 (16), 381 (23), 365 (22), 317 (19), 304 (92), 288 (8), 274 (53), 261 (19), 230 (13), 213 (58), 158 (8), 133 (38), 115 (21), 104 (12), 91 (100%); anal. calcd for C27H24N2O4S: C, 68.62; H, 5.12; N, 5.93. Found: C, 68.61; H, 5.08; N, 5.95%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1352 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.35 (s, 3H, Me), 2.40 (s, 3H, MeN), 2.56–2.62 (m, 1H, MeNCHH), 3.48–3.53 (m, 1H, SO2CH), 4.03 (br s, 1H, MeNCHH), 4.13–4.19 (m, 1H, PhCH), 4.30 (d, J = 8.1 Hz, 1H, CHN), 4.70 (d, J = 14.8 Hz, 1H, PhCHHN), 5.40 (d, J = 14.8 Hz, 1H, PhCHHN), 7.00 (t, J = 7.1 Hz, 1H, Ar), 7.11–7.36 (m, 9H, Ar), 7.49 (t, J = 7.3 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 39.4, 47.4, 58.7, 63.2, 63.5, 71.9, 116.6, 117.8, 118.9, 124.3, 125.5, 1257.5, 127.7 (2C), 129.0 (2C), 129.2 (2C), 129.3 (2C), 132.9, 132.9, 138.2, 140.0, 153.6, 156.1, 161.6 ppm; m/z (EI, 70 eV) 487 (7, M+), 422 (4), 407 (17), 381 (31), 318 (76), 302 (4), 288 (32), 275 (22), 213 (31), 133 (26), 105 (100%); anal. calcd for C28H26N2O4S: C, 69.11; H, 5.39; N, 5.76. Found: C, 69.10; H, 5.39; N, 5.81%.
O), 1352 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.35 (s, 3H, Me), 2.40 (s, 3H, MeN), 2.56–2.62 (m, 1H, MeNCHH), 3.48–3.53 (m, 1H, SO2CH), 4.03 (br s, 1H, MeNCHH), 4.13–4.19 (m, 1H, PhCH), 4.30 (d, J = 8.1 Hz, 1H, CHN), 4.70 (d, J = 14.8 Hz, 1H, PhCHHN), 5.40 (d, J = 14.8 Hz, 1H, PhCHHN), 7.00 (t, J = 7.1 Hz, 1H, Ar), 7.11–7.36 (m, 9H, Ar), 7.49 (t, J = 7.3 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 39.4, 47.4, 58.7, 63.2, 63.5, 71.9, 116.6, 117.8, 118.9, 124.3, 125.5, 1257.5, 127.7 (2C), 129.0 (2C), 129.2 (2C), 129.3 (2C), 132.9, 132.9, 138.2, 140.0, 153.6, 156.1, 161.6 ppm; m/z (EI, 70 eV) 487 (7, M+), 422 (4), 407 (17), 381 (31), 318 (76), 302 (4), 288 (32), 275 (22), 213 (31), 133 (26), 105 (100%); anal. calcd for C28H26N2O4S: C, 69.11; H, 5.39; N, 5.76. Found: C, 69.10; H, 5.39; N, 5.81%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1328 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 0.96 (t, J = 7.3 Hz, 3H, MeCH2CH2N), 1.74–1.97 (m, 2H, MeCHHCH2N), 3.47 (ddd, J = 14.0, 11.8, 4.9 Hz, 1H, MeCH2CHHN), 3.40 (d, J = 8.5 Hz, 1H, PhNCHH), 3.95–4.05 (ddd, J = 14.0, 11.4, 5.7 Hz, 1H, MeCH2CHHN), 4.18 (d, J = 5.8 Hz, 1H, SO2CH), 4.36–4.44 (m, 2H, PhCH and PhNCHH), 5.48 (d, J = 5.8 Hz, 1H, CHN), 6.92 (t, J = 7.2 Hz, 2H, Ar), 7.02 (d, J = 7.9 Hz, 2H, Ar), 7.21 (d, J = 6.8 Hz, 1H, Ar), 7.28–7.38 (m, 7H, Ar), 7.60 (t, J = 7.7 Hz, 1H, Ar), 7.78 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.7, 45.1, 54.8, 54.9, 57.5, 74.7, 116.6 (2C), 117.2, 118.1, 120.1, 120.5, 124.4, 124.9, 127.2 (2C), 127.8, 129.3 (2C), 129.6 (2C), 133.3, 139.8, 145.9, 153.7, 154.7, 161.3 ppm; m/z (EI, 70 eV) 486 (9, M+), 443 (4), 318 (100), 289 (32), 276 (48), 260 (12), 240 (13), 226 (42), 213 (13), 195 (30), 115 (28), 106 (58), 91 (42), 77 (74%); anal. calcd for C28H26N2O4S: C, 69.11; H, 5.39; N, 5.76. Found: C, 68.96; H, 5.45; N, 5.62%.
O), 1328 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 0.96 (t, J = 7.3 Hz, 3H, MeCH2CH2N), 1.74–1.97 (m, 2H, MeCHHCH2N), 3.47 (ddd, J = 14.0, 11.8, 4.9 Hz, 1H, MeCH2CHHN), 3.40 (d, J = 8.5 Hz, 1H, PhNCHH), 3.95–4.05 (ddd, J = 14.0, 11.4, 5.7 Hz, 1H, MeCH2CHHN), 4.18 (d, J = 5.8 Hz, 1H, SO2CH), 4.36–4.44 (m, 2H, PhCH and PhNCHH), 5.48 (d, J = 5.8 Hz, 1H, CHN), 6.92 (t, J = 7.2 Hz, 2H, Ar), 7.02 (d, J = 7.9 Hz, 2H, Ar), 7.21 (d, J = 6.8 Hz, 1H, Ar), 7.28–7.38 (m, 7H, Ar), 7.60 (t, J = 7.7 Hz, 1H, Ar), 7.78 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.7, 45.1, 54.8, 54.9, 57.5, 74.7, 116.6 (2C), 117.2, 118.1, 120.1, 120.5, 124.4, 124.9, 127.2 (2C), 127.8, 129.3 (2C), 129.6 (2C), 133.3, 139.8, 145.9, 153.7, 154.7, 161.3 ppm; m/z (EI, 70 eV) 486 (9, M+), 443 (4), 318 (100), 289 (32), 276 (48), 260 (12), 240 (13), 226 (42), 213 (13), 195 (30), 115 (28), 106 (58), 91 (42), 77 (74%); anal. calcd for C28H26N2O4S: C, 69.11; H, 5.39; N, 5.76. Found: C, 68.96; H, 5.45; N, 5.62%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1324 (–SO2), 1138 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.53; 1H NMR (300 MHz, CDCl3) δ 0.91 (t, J = 7.3 Hz, 3H, MeCH2CH2CH2N), 1.25–1.46 (m, 2H, MeCHHCH2CH2N), 1.64–1.91 (m, 2H, MeCH2CHHCH2N), 3.54 (ddd, 1H, J = 14.0, 11.9, 4.8 Hz, 1H, MeCH2CH2CHHN), 3.70 (d, J = 9.2 Hz, 1H, PhNCHH), 4.02 (ddd, 1H, J = 14.0, 11.4, 5.6 Hz, 1H, MeCH2CH2CHHN), 4.18 (d, J = 5.8 Hz, 1H, SO2CH), 4.33–4.43 {[2H, consisting m, 1H, PhCH]; and dd, J = 9.5, 6.8 Hz, 1H (4.41), PhNCHH}, 5.47 (d, J = 5.8 Hz, 1H, CHN), 6.90 (t, J = 7.3 Hz, 1H, Ar), 6.99 (d, J = 8.0 Hz, 2H, Ar), 7.20 (d, J = 6.8 Hz, 2H, Ar), 7.27–7.38 (m, 7H, Ar), 7.60 (t, J = 7.3 Hz, 1H, Ar), 7.76 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.6, 20.2, 32.1, 45.1, 53.0, 54.7, 57.4, 74.9, 116.4 (2C), 117.3, 118.1, 120.1, 120.3, 124.3, 124.9, 127.2 (2C), 127.8, 129.3 (2C), 129.6 (2C), 133.3, 139.8, 145.9, 153.7, 154.6, 161.3 ppm; m/z (EI, 70 eV) 500 (20, M+), 443 (3), 332 (100), 318 (27), 289 (69), 276 (28), 260 (10), 240 (36), 227 (13), 195 (27), 116 (16), 106 (42), 91 (24), 77 (41%); anal. calcd for C29H28N2O4S: C, 69.58; H, 5.64; N, 5.60. Found: C, 69.61; H, 5.58; N, 5.61%.
O), 1324 (–SO2), 1138 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.53; 1H NMR (300 MHz, CDCl3) δ 0.91 (t, J = 7.3 Hz, 3H, MeCH2CH2CH2N), 1.25–1.46 (m, 2H, MeCHHCH2CH2N), 1.64–1.91 (m, 2H, MeCH2CHHCH2N), 3.54 (ddd, 1H, J = 14.0, 11.9, 4.8 Hz, 1H, MeCH2CH2CHHN), 3.70 (d, J = 9.2 Hz, 1H, PhNCHH), 4.02 (ddd, 1H, J = 14.0, 11.4, 5.6 Hz, 1H, MeCH2CH2CHHN), 4.18 (d, J = 5.8 Hz, 1H, SO2CH), 4.33–4.43 {[2H, consisting m, 1H, PhCH]; and dd, J = 9.5, 6.8 Hz, 1H (4.41), PhNCHH}, 5.47 (d, J = 5.8 Hz, 1H, CHN), 6.90 (t, J = 7.3 Hz, 1H, Ar), 6.99 (d, J = 8.0 Hz, 2H, Ar), 7.20 (d, J = 6.8 Hz, 2H, Ar), 7.27–7.38 (m, 7H, Ar), 7.60 (t, J = 7.3 Hz, 1H, Ar), 7.76 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.6, 20.2, 32.1, 45.1, 53.0, 54.7, 57.4, 74.9, 116.4 (2C), 117.3, 118.1, 120.1, 120.3, 124.3, 124.9, 127.2 (2C), 127.8, 129.3 (2C), 129.6 (2C), 133.3, 139.8, 145.9, 153.7, 154.6, 161.3 ppm; m/z (EI, 70 eV) 500 (20, M+), 443 (3), 332 (100), 318 (27), 289 (69), 276 (28), 260 (10), 240 (36), 227 (13), 195 (27), 116 (16), 106 (42), 91 (24), 77 (41%); anal. calcd for C29H28N2O4S: C, 69.58; H, 5.64; N, 5.60. Found: C, 69.61; H, 5.58; N, 5.61%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1353 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.43; 1H NMR (300 MHz, CDCl3) δ 3.73 (dd, J = 9.7, 1.7 Hz, 1H, PhNCHH), 4.13–4.30 {[2H, consisting d, J = 6.0 Hz, 1H (4.23), SO2CH]; and d, J = 15.3 Hz, 1H (4.28), PhCHHN}, 4.39–4.41 (m, 1H, PhCH), 5.47 (dd, J = 9.7, 7.4 Hz, 1H, PhNCHH), 5.52–5.57 {[2H, consisting d, J = 15.3 Hz, 1H (5.544), PhCHHN]; and d, J = 6.0 Hz, 1H (5.540), CHN}, 6.89 (t, J = 7.6 Hz, 1H, Ar), 6.95–7.48 (m, 18H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 45.0, 55.1, 57.0, 57.6, 74.3, 116.7 (3C), 117.8, 120.1, 120.6, 124.4, 125.5, 127.3 (2C), 127.9, 128.4, 128.8 (2C), 128.9 (2C), 129.4 (2C), 129.7 (2C), 133.2, 136.5, 139.7, 146.0, 153.6, 155.5, 161.3 ppm; m/z (EI, 70 eV) 534 (11, M+), 443 (15), 366 (100), 275 (68), 261 (14), 230 (8), 195 (17), 116 (11), 106 (25), 91 (100%); anal. calcd for C32H26N2O4S: C, 71.89; H, 4.90; N, 5.24. Found: C, 71.78; H, 4.94; N, 5.31%.
O), 1353 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.43; 1H NMR (300 MHz, CDCl3) δ 3.73 (dd, J = 9.7, 1.7 Hz, 1H, PhNCHH), 4.13–4.30 {[2H, consisting d, J = 6.0 Hz, 1H (4.23), SO2CH]; and d, J = 15.3 Hz, 1H (4.28), PhCHHN}, 4.39–4.41 (m, 1H, PhCH), 5.47 (dd, J = 9.7, 7.4 Hz, 1H, PhNCHH), 5.52–5.57 {[2H, consisting d, J = 15.3 Hz, 1H (5.544), PhCHHN]; and d, J = 6.0 Hz, 1H (5.540), CHN}, 6.89 (t, J = 7.6 Hz, 1H, Ar), 6.95–7.48 (m, 18H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 45.0, 55.1, 57.0, 57.6, 74.3, 116.7 (3C), 117.8, 120.1, 120.6, 124.4, 125.5, 127.3 (2C), 127.9, 128.4, 128.8 (2C), 128.9 (2C), 129.4 (2C), 129.7 (2C), 133.2, 136.5, 139.7, 146.0, 153.6, 155.5, 161.3 ppm; m/z (EI, 70 eV) 534 (11, M+), 443 (15), 366 (100), 275 (68), 261 (14), 230 (8), 195 (17), 116 (11), 106 (25), 91 (100%); anal. calcd for C32H26N2O4S: C, 71.89; H, 4.90; N, 5.24. Found: C, 71.78; H, 4.94; N, 5.31%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1340 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 2.38 (s, 3H, Me), 3.70 (dd, J = 9.7, 2.3 Hz, 1H, PhNCHH), 4.21 (dd, J = 6.0, 1.5 Hz, 1H, SO2CH), 4.26 (d, J = 15.5 Hz, 1H, PhCHHN), 4.36–4.38 (m, 1H, PhCH), 4.45 (dd, J = 9.7, 7.2 Hz, 1H, PhNCHH), 5.47–5.52 (m, 2H, PhCHHN and CHN), 6.90–7.40 (m, 17H, Ar), 7.46 (t, J = 7.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 45.0, 55.2, 56.7, 57.5, 74.2, 116.6 (2C), 116.7, 117.8, 120.0, 120.5, 124.3, 125.6, 127.3 (2C), 127.83, 128.78 (2C), 129.3 (2C), 129.6 (2C), 129.6 (2C), 133.2, 133.4, 138.3, 139.7, 146.0, 153.6, 155.4, 161.2 ppm; m/z (EI, 70 eV) 548 (7, M+), 443 (20), 380 (62), 288 (41), 275 (80), 260 (8), 195 (10), 116 (9), 105 (100), 77 (42%); anal. calcd for C33H28N2O4S: C, 72.24; H, 5.14; N, 5.11. Found: C, 72.21; H, 5.18; N, 4.97%.
O), 1340 (–SO2), 1139 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.52; 1H NMR (300 MHz, CDCl3) δ 2.38 (s, 3H, Me), 3.70 (dd, J = 9.7, 2.3 Hz, 1H, PhNCHH), 4.21 (dd, J = 6.0, 1.5 Hz, 1H, SO2CH), 4.26 (d, J = 15.5 Hz, 1H, PhCHHN), 4.36–4.38 (m, 1H, PhCH), 4.45 (dd, J = 9.7, 7.2 Hz, 1H, PhNCHH), 5.47–5.52 (m, 2H, PhCHHN and CHN), 6.90–7.40 (m, 17H, Ar), 7.46 (t, J = 7.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 45.0, 55.2, 56.7, 57.5, 74.2, 116.6 (2C), 116.7, 117.8, 120.0, 120.5, 124.3, 125.6, 127.3 (2C), 127.83, 128.78 (2C), 129.3 (2C), 129.6 (2C), 129.6 (2C), 133.2, 133.4, 138.3, 139.7, 146.0, 153.6, 155.4, 161.2 ppm; m/z (EI, 70 eV) 548 (7, M+), 443 (20), 380 (62), 288 (41), 275 (80), 260 (8), 195 (10), 116 (9), 105 (100), 77 (42%); anal. calcd for C33H28N2O4S: C, 72.24; H, 5.14; N, 5.11. Found: C, 72.21; H, 5.18; N, 4.97%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1354 (–SO2), 1159 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.17; 1H NMR (300 MHz, CDCl3) δ 0.86 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.18–1.31 (m, 1H, CHHCH2CH2N), 1.37–1.47 (m, 1H, CHHCH2CH2N), 1.65–1.95 (m, 4H, CH2CHHCH2N and MeCHHCH2N), 3.08–3.17 (m, 1H, CH2CH2CHHN), 3.31–3.39 (m, 1H, CH2CH2CHHN), 3.64 (ddd, J = 14.6, 11.0, 5.7 Hz, 1H, MeCH2CHHN), 3.88 (ddd, J = 14.6, 11.2, 5.8 Hz, 1H, MeCH2CHHN), 4.08 (td, J = 8.6, 7.4 Hz, 1H, NCHCPh), 4.27 (dd, J = 7.4, 6.1 Hz, 1H, PhCH), 4.39 (dd, J = 8.3, 6.1 Hz, 1H, SO2CH), 4.78 (d, J = 8.3 Hz, 1H, CHN), 7.22–7.39 (m, 7H, Ar), 7.56 (t, J = 8.3 Hz, 1H, Ar), 7.73 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.2, 26.4, 27.5, 48.1, 55.0, 56.1, 63.6, 66.4, 70.0, 117.0, 117.2, 119.1, 124.4, 124.6, 127.3, 127.9 (2C), 128.8 (2C), 132.6, 138.1, 149.8, 153.1, 161.1 ppm; m/z (EI, 70 eV) 451 (8, M+), 357 (4), 343 (6), 317 (100), 288 (46), 274 (40), 260 (19), 240 (90), 226 (19), 212 (24), 198 (49), 159 (19), 115 (33), 91 (39%); anal. calcd for C25H26N2O4S: C, 66.64; H, 5.82; N, 6.22. Found: C, 66.61; H, 5.78; N, 6.22%.
O), 1354 (–SO2), 1159 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.17; 1H NMR (300 MHz, CDCl3) δ 0.86 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.18–1.31 (m, 1H, CHHCH2CH2N), 1.37–1.47 (m, 1H, CHHCH2CH2N), 1.65–1.95 (m, 4H, CH2CHHCH2N and MeCHHCH2N), 3.08–3.17 (m, 1H, CH2CH2CHHN), 3.31–3.39 (m, 1H, CH2CH2CHHN), 3.64 (ddd, J = 14.6, 11.0, 5.7 Hz, 1H, MeCH2CHHN), 3.88 (ddd, J = 14.6, 11.2, 5.8 Hz, 1H, MeCH2CHHN), 4.08 (td, J = 8.6, 7.4 Hz, 1H, NCHCPh), 4.27 (dd, J = 7.4, 6.1 Hz, 1H, PhCH), 4.39 (dd, J = 8.3, 6.1 Hz, 1H, SO2CH), 4.78 (d, J = 8.3 Hz, 1H, CHN), 7.22–7.39 (m, 7H, Ar), 7.56 (t, J = 8.3 Hz, 1H, Ar), 7.73 (d, J = 8.0 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.2, 26.4, 27.5, 48.1, 55.0, 56.1, 63.6, 66.4, 70.0, 117.0, 117.2, 119.1, 124.4, 124.6, 127.3, 127.9 (2C), 128.8 (2C), 132.6, 138.1, 149.8, 153.1, 161.1 ppm; m/z (EI, 70 eV) 451 (8, M+), 357 (4), 343 (6), 317 (100), 288 (46), 274 (40), 260 (19), 240 (90), 226 (19), 212 (24), 198 (49), 159 (19), 115 (33), 91 (39%); anal. calcd for C25H26N2O4S: C, 66.64; H, 5.82; N, 6.22. Found: C, 66.61; H, 5.78; N, 6.22%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1353 (–SO2), 1157 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.19; 1H NMR (300 MHz, CDCl3) δ 0.86 (t, J = 7.4 Hz, 3H, MeCH2CH2CH2N), 1.18–1.35 (m, 3H, C
O), 1353 (–SO2), 1157 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.19; 1H NMR (300 MHz, CDCl3) δ 0.86 (t, J = 7.4 Hz, 3H, MeCH2CH2CH2N), 1.18–1.35 (m, 3H, C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) HCH2CH2N and MeC
HCH2CH2N and MeC![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2CH2CH2N), 1.38–1.49 (m, 1H, CHHCH2CH2N), 1.70–1.93 (m, 4H, CH2CHHCH2N and MeCH2CHHCH2N), 3.09–3.18 (m, 1H, CH2CH2CHHN), 3.32–3.40 (m, 1H, CH2CH2CHHN), 3.70 (ddd, J = 14.5, 11.2, 5.3 Hz, 1H, MeCH2CH2CHHN), 3.93 (ddd, J = 14.5, 11.2, 5.3 Hz, 1H, MeCH2CH2CHHN), 4.08 (td, J = 9.0, 7.4 Hz, 1H, NCHCPh), 4.27 (dd, J = 7.4, 6.0 Hz, 1H, PhCH), 4.39 (dd, J = 8.3, 6.0 Hz, 1H, SO2CH), 4.79 (d, J = 8.3 Hz, 1H, CHN), 7.22–7.42 (m, 7H, Ar), 7.59 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.75 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.5, 20.0, 26.5, 27.6, 31.7, 48.1, 53.3, 56.2, 63.6, 66.4, 70.0, 117.0, 117.2, 119.2, 124.5, 124.5, 127.3, 127.9 (2C), 128.8 (2C), 132.5, 138.1, 149.8, 153.1, 161.2 ppm; m/z (EI, 70 eV) 465 (47, M+), 400 (3), 357 (5), 343 (6), 331 (100), 303 (28), 288 (31), 274 (34), 254 (81), 240 (25), 227 (23), 198 (38), 159 (19), 115 (17), 91 (25%); anal. calcd for C26H28N2O4S: C, 67.22; H, 6.07; N, 6.03. Found: C, 67.20; H, 6.12; N, 6.06%.
2CH2CH2N), 1.38–1.49 (m, 1H, CHHCH2CH2N), 1.70–1.93 (m, 4H, CH2CHHCH2N and MeCH2CHHCH2N), 3.09–3.18 (m, 1H, CH2CH2CHHN), 3.32–3.40 (m, 1H, CH2CH2CHHN), 3.70 (ddd, J = 14.5, 11.2, 5.3 Hz, 1H, MeCH2CH2CHHN), 3.93 (ddd, J = 14.5, 11.2, 5.3 Hz, 1H, MeCH2CH2CHHN), 4.08 (td, J = 9.0, 7.4 Hz, 1H, NCHCPh), 4.27 (dd, J = 7.4, 6.0 Hz, 1H, PhCH), 4.39 (dd, J = 8.3, 6.0 Hz, 1H, SO2CH), 4.79 (d, J = 8.3 Hz, 1H, CHN), 7.22–7.42 (m, 7H, Ar), 7.59 (td, J = 8.5, 1.4 Hz, 1H, Ar), 7.75 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.5, 20.0, 26.5, 27.6, 31.7, 48.1, 53.3, 56.2, 63.6, 66.4, 70.0, 117.0, 117.2, 119.2, 124.5, 124.5, 127.3, 127.9 (2C), 128.8 (2C), 132.5, 138.1, 149.8, 153.1, 161.2 ppm; m/z (EI, 70 eV) 465 (47, M+), 400 (3), 357 (5), 343 (6), 331 (100), 303 (28), 288 (31), 274 (34), 254 (81), 240 (25), 227 (23), 198 (38), 159 (19), 115 (17), 91 (25%); anal. calcd for C26H28N2O4S: C, 67.22; H, 6.07; N, 6.03. Found: C, 67.20; H, 6.12; N, 6.06%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1355 (–SO2), 1159 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.13; 1H NMR (300 MHz, CDCl3) δ 1.13–1.18 (m, 1H, CHHCH2CH2N), 1.43–1.46 (m, 1H, CHHCH2CH2N), 1.65–1.67 (m, 2H, CH2CHHCH2N), 2.91–3.00 (m, 1H, CH2CH2CHHN), 3.33–3.40 (m, 1H, CH2CH2CHHN), 3.71 (br s, 1H, NCHCPh), 4.09 (s, 2H, PhCH and SO2CH), 4.49 (d, J = 7.9 Hz, 1H, CHN), 4.78 (d, J = 15.3 Hz, 1H, PhCHHN), 5.24 (d, J = 15.3 Hz, 1H, PhCHHN), 6.93–6.95 (m, 2H, Ar), 7.19–7.39 (m, 10H, Ar), 7.53–7.62 (m, 2H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 26.8, 27.5, 47.1, 56.4, 56.7, 64.3, 67.0, 69.8, 116.3, 117.1, 120.0, 124.6, 125.1, 127.1, 127.8 (2C), 128.7 (2C), 128.8, 128.9 (2C), 129.1 (2C), 132.6, 134.3, 138.3, 148.8, 153.1, 160.8 ppm; m/z (EI, 70 eV) 499 (0.8, M+), 365 (89), 337 (6), 288 (12), 274 (40), 261 (11), 246 (4), 230 (13), 217 (4), 202 (5), 159 (6), 128 (8), 115 (10), 91 (100%); anal. calcd for C29H26N2O4S: C, 69.86; H, 5.26; N, 5.62. Found: C, 69.81; H, 5.31; N, 5.61%.
O), 1355 (–SO2), 1159 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.13; 1H NMR (300 MHz, CDCl3) δ 1.13–1.18 (m, 1H, CHHCH2CH2N), 1.43–1.46 (m, 1H, CHHCH2CH2N), 1.65–1.67 (m, 2H, CH2CHHCH2N), 2.91–3.00 (m, 1H, CH2CH2CHHN), 3.33–3.40 (m, 1H, CH2CH2CHHN), 3.71 (br s, 1H, NCHCPh), 4.09 (s, 2H, PhCH and SO2CH), 4.49 (d, J = 7.9 Hz, 1H, CHN), 4.78 (d, J = 15.3 Hz, 1H, PhCHHN), 5.24 (d, J = 15.3 Hz, 1H, PhCHHN), 6.93–6.95 (m, 2H, Ar), 7.19–7.39 (m, 10H, Ar), 7.53–7.62 (m, 2H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 26.8, 27.5, 47.1, 56.4, 56.7, 64.3, 67.0, 69.8, 116.3, 117.1, 120.0, 124.6, 125.1, 127.1, 127.8 (2C), 128.7 (2C), 128.8, 128.9 (2C), 129.1 (2C), 132.6, 134.3, 138.3, 148.8, 153.1, 160.8 ppm; m/z (EI, 70 eV) 499 (0.8, M+), 365 (89), 337 (6), 288 (12), 274 (40), 261 (11), 246 (4), 230 (13), 217 (4), 202 (5), 159 (6), 128 (8), 115 (10), 91 (100%); anal. calcd for C29H26N2O4S: C, 69.86; H, 5.26; N, 5.62. Found: C, 69.81; H, 5.31; N, 5.61%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1356 (–SO2), 1161 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.17; 1H NMR (300 MHz, CDCl3) δ 1.07–1.20 (m, 1H, CHHCH2CH2N), 1.44–1.52 (m, 1H, CHHCH2CH2N), 1.60–1.76 (m, 2H, CH2CHHCH2N), 2.32 (s, 3H, Me), 2.88–2.96 (m, 1H, CH2CH2CHHN), 3.36–3.43 (m, 1H, CH2CH2CHHN), 3.49 (br s, 1H, NCHCPh), 4.01–4.03 (m, 1H, CHPh), 4.10–4.17 (m, 1H, SO2CH), 4.44 (d, J = 7.8 Hz, 1H, CHN), 4.72 (d, J = 15.1 Hz, 1H, PhCHHN), 5.17 (d, J = 15.1 Hz, 1H, PhCHHN), 6.87–6.89 (m, 2H, Ar), 7.06 (s, 4H, Ar), 7.37 (d, J = 8.2 Hz, 1H, Ar), 7.56 (t, J = 7.6 Hz, 1H, Ar), 7.67 (d, J = 7.7 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.1, 27.1, 27.5, 46.7, 56.2, 57.1, 64.6, 67.1, 69.7, 116.2, 117.1, 120.1, 124.6, 125.1, 127.1, 127.8 (2C), 128.6 (2C), 129.3 (2C), 129.4 (2C), 130.9, 132.6, 138.6, 138.9, 148.4, 153.1, 160.7 ppm; m/z (EI, 70 eV) 512 (0.8, M+), 407 (2), 379 (67), 343 (8), 302 (4), 288 (8), 274 (29), 230 (10), 202 (5), 173 (8), 128 (8), 115 (10), 105 (100), 91 (19%); anal. calcd for C30H28N2O4S: C, 70.29; H, 5.51; N, 5.46. Found: C, 70.33; H, 5.47; N, 5.33%.
O), 1356 (–SO2), 1161 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.17; 1H NMR (300 MHz, CDCl3) δ 1.07–1.20 (m, 1H, CHHCH2CH2N), 1.44–1.52 (m, 1H, CHHCH2CH2N), 1.60–1.76 (m, 2H, CH2CHHCH2N), 2.32 (s, 3H, Me), 2.88–2.96 (m, 1H, CH2CH2CHHN), 3.36–3.43 (m, 1H, CH2CH2CHHN), 3.49 (br s, 1H, NCHCPh), 4.01–4.03 (m, 1H, CHPh), 4.10–4.17 (m, 1H, SO2CH), 4.44 (d, J = 7.8 Hz, 1H, CHN), 4.72 (d, J = 15.1 Hz, 1H, PhCHHN), 5.17 (d, J = 15.1 Hz, 1H, PhCHHN), 6.87–6.89 (m, 2H, Ar), 7.06 (s, 4H, Ar), 7.37 (d, J = 8.2 Hz, 1H, Ar), 7.56 (t, J = 7.6 Hz, 1H, Ar), 7.67 (d, J = 7.7 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.1, 27.1, 27.5, 46.7, 56.2, 57.1, 64.6, 67.1, 69.7, 116.2, 117.1, 120.1, 124.6, 125.1, 127.1, 127.8 (2C), 128.6 (2C), 129.3 (2C), 129.4 (2C), 130.9, 132.6, 138.6, 138.9, 148.4, 153.1, 160.7 ppm; m/z (EI, 70 eV) 512 (0.8, M+), 407 (2), 379 (67), 343 (8), 302 (4), 288 (8), 274 (29), 230 (10), 202 (5), 173 (8), 128 (8), 115 (10), 105 (100), 91 (19%); anal. calcd for C30H28N2O4S: C, 70.29; H, 5.51; N, 5.46. Found: C, 70.33; H, 5.47; N, 5.33%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1345 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 0.95 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.85–1.98 (m, 2H, MeCHHCH2N), 2.51–2.64 (m, 2H, SCH2C), 3.79–3.90 (m, 1H, MeCH2CHHN), 3.94–4.10 (m, 2H, MeCH2CHHN and NCHCPh), 4.27 (d, J = 10.3 Hz, 1H, NCHHS), 4.53 (d, J = 10.3 Hz, 1H, NCHHS), 4.58–4.73 (m, 2H, PhCH and SO2CH), 4.89 (d, J = 9.5 Hz, 1H, CHN), 7.32–7.43 (m, 7H, Ar), 7.63 (td, J = 8.4, 1.4 Hz, 1H, Ar), 7.78 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.7, 32.6, 49.2, 55.1, 56.7, 57.6, 65.7, 74.5, 117.3, 117.6, 118.9, 124.4, 124.8, 127.6 (2C), 127.8, 129.0 (2C), 133.3, 136.3, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 468 (0.8, M+), 357 (11), 318 (100), 288 (36), 274 (31), 260 (14), 240 (68), 228 (25), 214 (19), 198 (50), 177 (58), 156 (16), 130 (63), 115 (69), 103 (25), 91 (69%); anal. calcd for C24H24N2O4S2: C, 61.52; H, 5.16; N, 5.98. Found: C, 61.50; H, 5.16; N, 6.01%.
O), 1345 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 0.95 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.85–1.98 (m, 2H, MeCHHCH2N), 2.51–2.64 (m, 2H, SCH2C), 3.79–3.90 (m, 1H, MeCH2CHHN), 3.94–4.10 (m, 2H, MeCH2CHHN and NCHCPh), 4.27 (d, J = 10.3 Hz, 1H, NCHHS), 4.53 (d, J = 10.3 Hz, 1H, NCHHS), 4.58–4.73 (m, 2H, PhCH and SO2CH), 4.89 (d, J = 9.5 Hz, 1H, CHN), 7.32–7.43 (m, 7H, Ar), 7.63 (td, J = 8.4, 1.4 Hz, 1H, Ar), 7.78 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.7, 32.6, 49.2, 55.1, 56.7, 57.6, 65.7, 74.5, 117.3, 117.6, 118.9, 124.4, 124.8, 127.6 (2C), 127.8, 129.0 (2C), 133.3, 136.3, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 468 (0.8, M+), 357 (11), 318 (100), 288 (36), 274 (31), 260 (14), 240 (68), 228 (25), 214 (19), 198 (50), 177 (58), 156 (16), 130 (63), 115 (69), 103 (25), 91 (69%); anal. calcd for C24H24N2O4S2: C, 61.52; H, 5.16; N, 5.98. Found: C, 61.50; H, 5.16; N, 6.01%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1345 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 0.92 (t, J = 7.3 Hz, 1H, MeCH2CH2CH2N), 1.26–1.45 (m, 2H, MeCHHCH2CH2N), 1.82–1.92 (m, 2H, MeCH2CHHCH2N), 2.51–2.63 (m, 2H, SCH2C), 3.75–3.93 (m, 1H, MeCH2CH2CHHN), 3.98–4.10 (m, 2H, MeCH2CH2CHHN and NCHCPh), 4.26 (d, J = 10.3 Hz, 1H, NCHHS), 4.54 (d, J = 10.3 Hz, 1H, NCHHS), 4.58–4.73 (m, 2H, PhCH and SO2CH), 4.89 (d, J = 9.5 Hz, 1H, CHN), 7.27–7.42 (m, 7H, Ar), 7.62 (td, J = 8.6, 1.5 Hz, 1H, Ar), 7.77 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.1, 32.2, 32.6, 49.2, 53.4, 56.8, 57.7, 65.6, 74.5, 117.3, 117.6, 118.9, 124.4, 124.8, 127.6 (2C), 127.8, 129.0 (2C), 133.2, 136.3, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 482 (5, M+), 371 (16), 361 (14), 332 (100), 315 (8), 302 (16), 288 (25), 274 (30), 254 (58), 240 (36), 228 (23), 198 (51), 177 (68), 156 (13), 130 (46), 115 (48), 103 (19), 91 (53%); anal. calcd for C25H26N2O4S2: C, 62.22; H, 5.43; N, 5.80. Found: C, 62.23; H, 5.40; N, 5.73%.
O), 1345 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 0.92 (t, J = 7.3 Hz, 1H, MeCH2CH2CH2N), 1.26–1.45 (m, 2H, MeCHHCH2CH2N), 1.82–1.92 (m, 2H, MeCH2CHHCH2N), 2.51–2.63 (m, 2H, SCH2C), 3.75–3.93 (m, 1H, MeCH2CH2CHHN), 3.98–4.10 (m, 2H, MeCH2CH2CHHN and NCHCPh), 4.26 (d, J = 10.3 Hz, 1H, NCHHS), 4.54 (d, J = 10.3 Hz, 1H, NCHHS), 4.58–4.73 (m, 2H, PhCH and SO2CH), 4.89 (d, J = 9.5 Hz, 1H, CHN), 7.27–7.42 (m, 7H, Ar), 7.62 (td, J = 8.6, 1.5 Hz, 1H, Ar), 7.77 (dd, J = 8.0, 1.2 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.1, 32.2, 32.6, 49.2, 53.4, 56.8, 57.7, 65.6, 74.5, 117.3, 117.6, 118.9, 124.4, 124.8, 127.6 (2C), 127.8, 129.0 (2C), 133.2, 136.3, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 482 (5, M+), 371 (16), 361 (14), 332 (100), 315 (8), 302 (16), 288 (25), 274 (30), 254 (58), 240 (36), 228 (23), 198 (51), 177 (68), 156 (13), 130 (46), 115 (48), 103 (19), 91 (53%); anal. calcd for C25H26N2O4S2: C, 62.22; H, 5.43; N, 5.80. Found: C, 62.23; H, 5.40; N, 5.73%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1346 (–SO2), 1143 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.34; 1H NMR (300 MHz, CDCl3) δ 2.48 (d, J = 8.3 Hz, 1H, SCH2C), 3.73 (td, J = 8.3, 7.1 Hz, NCHCPh), 4.17 (d, J = 10.2 Hz, 1H, NCHHS), 2.35 (dd, J = 8.5, 7.1 Hz, 1H, PhCH), 4.47–4.53 {[2H, consisting dd, J = 9.3, 8.5 Hz, 1H (4.50), SO2CH]; and d, 1H (4.30), J = 10.2 Hz, NCHHS}, 4.84 (d, J = 9.5 Hz, 1H, CHN), 4.95 (d, J = 14.9 Hz, 1H, PhCHHN), 5.35 (d, J = 14.9 Hz, 1H, PhCHHN), 7.10 (td, J = 8.1, 0.9 Hz, 1H, Ar), 7.20 (d, J = 8.1 Hz, 2H, Ar), 7.30–7.40 (m, 10H, Ar), 7.54 (td, J = 7.4, 1.4 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 32.6, 48.5, 56.7, 56.8, 57.5, 65.5, 74.4, 117.0, 117.1, 118.7, 124.5, 125.2, 127.5 (2C), 127.7, 128.6 (3C), 128.9 (2C), 129.6 (2C), 133.1, 135.6, 136.2, 153.6, 154.2, 161.8 ppm; m/z (EI, 70 eV) 516 (1, M+), 455 (2), 425 (39), 405 (8), 366 (64), 331 (10), 313 (9), 288 (18), 274 (83), 262 (19), 246 (8), 230 (23), 217 (9), 202 (14), 177 (39), 147 (9), 130 (42), 115 (36), 103 (13), 91 (100%); anal. calcd for C28H24N2O4S2: C, 65.09; H, 4.68; N, 5.42. Found: C, 65.17; H, 4.65; N, 5.38%.
O), 1346 (–SO2), 1143 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.34; 1H NMR (300 MHz, CDCl3) δ 2.48 (d, J = 8.3 Hz, 1H, SCH2C), 3.73 (td, J = 8.3, 7.1 Hz, NCHCPh), 4.17 (d, J = 10.2 Hz, 1H, NCHHS), 2.35 (dd, J = 8.5, 7.1 Hz, 1H, PhCH), 4.47–4.53 {[2H, consisting dd, J = 9.3, 8.5 Hz, 1H (4.50), SO2CH]; and d, 1H (4.30), J = 10.2 Hz, NCHHS}, 4.84 (d, J = 9.5 Hz, 1H, CHN), 4.95 (d, J = 14.9 Hz, 1H, PhCHHN), 5.35 (d, J = 14.9 Hz, 1H, PhCHHN), 7.10 (td, J = 8.1, 0.9 Hz, 1H, Ar), 7.20 (d, J = 8.1 Hz, 2H, Ar), 7.30–7.40 (m, 10H, Ar), 7.54 (td, J = 7.4, 1.4 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 32.6, 48.5, 56.7, 56.8, 57.5, 65.5, 74.4, 117.0, 117.1, 118.7, 124.5, 125.2, 127.5 (2C), 127.7, 128.6 (3C), 128.9 (2C), 129.6 (2C), 133.1, 135.6, 136.2, 153.6, 154.2, 161.8 ppm; m/z (EI, 70 eV) 516 (1, M+), 455 (2), 425 (39), 405 (8), 366 (64), 331 (10), 313 (9), 288 (18), 274 (83), 262 (19), 246 (8), 230 (23), 217 (9), 202 (14), 177 (39), 147 (9), 130 (42), 115 (36), 103 (13), 91 (100%); anal. calcd for C28H24N2O4S2: C, 65.09; H, 4.68; N, 5.42. Found: C, 65.17; H, 4.65; N, 5.38%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1334 (–SO2), 1135 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 2.37 (Me), 2.49 (d, J = 8.2 Hz, 1H, SCH2C), 3.75 (td, J = 8.2, 7.0 Hz, NCHCPh), 4.17 (d, J = 10.2 Hz, 1H, NCHHS), 4.36 (dd, J = 8.5, 7.0 Hz, 1H, PhCH), 4.45 (dd, J = 9.1, 8.5 Hz, 1H, SO2CH), 4.53 (d, J = 10.2 Hz, 1H, NCHHS), 4.82 (d, J = 9.1 Hz, 1H, CHN), 4.90 (d, J = 14.9 Hz, 1H, PhCHHN), 5.30 (d, J = 14.9 Hz, 1H, PhCHHN), 7.10–7.43 (m, 12H, Ar), 7.55 (td, J = 8.4, 1.4 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 32.6, 48.3, 56.7, 56.8, 57.5, 65.3, 74.3, 116.99, 117.04, 118.6, 124.4, 125.3, 127.5 (2C), 127.7, 128.9 (2C), 129.2 (2C), 129.5 (2C), 132.4, 133.1, 136.3, 138.5, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 530 (2, M+), 425 (52), 400 (4), 380 (27), 333 (12), 313 (7), 295 (69), 274 (19), 253 (6), 239 (22), 177 (12), 130 (16), 115 (19), 105 (100%); anal. calcd for C29H26N2O4S2: C, 65.64; H, 4.94; N, 5.28. Found: C, 65.43; H, 5.03; N, 5.18%.
O), 1334 (–SO2), 1135 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.38; 1H NMR (300 MHz, CDCl3) δ 2.37 (Me), 2.49 (d, J = 8.2 Hz, 1H, SCH2C), 3.75 (td, J = 8.2, 7.0 Hz, NCHCPh), 4.17 (d, J = 10.2 Hz, 1H, NCHHS), 4.36 (dd, J = 8.5, 7.0 Hz, 1H, PhCH), 4.45 (dd, J = 9.1, 8.5 Hz, 1H, SO2CH), 4.53 (d, J = 10.2 Hz, 1H, NCHHS), 4.82 (d, J = 9.1 Hz, 1H, CHN), 4.90 (d, J = 14.9 Hz, 1H, PhCHHN), 5.30 (d, J = 14.9 Hz, 1H, PhCHHN), 7.10–7.43 (m, 12H, Ar), 7.55 (td, J = 8.4, 1.4 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 32.6, 48.3, 56.7, 56.8, 57.5, 65.3, 74.3, 116.99, 117.04, 118.6, 124.4, 125.3, 127.5 (2C), 127.7, 128.9 (2C), 129.2 (2C), 129.5 (2C), 132.4, 133.1, 136.3, 138.5, 153.6, 154.1, 161.8 ppm; m/z (EI, 70 eV) 530 (2, M+), 425 (52), 400 (4), 380 (27), 333 (12), 313 (7), 295 (69), 274 (19), 253 (6), 239 (22), 177 (12), 130 (16), 115 (19), 105 (100%); anal. calcd for C29H26N2O4S2: C, 65.64; H, 4.94; N, 5.28. Found: C, 65.43; H, 5.03; N, 5.18%.General procedure for the synthesis of compounds 13a–h
A mixture of aldehyde derivatives 7a–d (1 mmol), N-methyl or benzylhydroxylamines hydrochloride 12a,b (1.1 mmol) and Et3N (1.1 mmol) in MeCN (15 mL) containing molecular sieves 4 Å was heated at reflux with stirring for 5 h. After completion of the reaction as indicated by TLC, the solvent was removed under reduced pressure, and then H2O (10 mL) was added and the mixture was extracted with EtOAc (3 × 10 mL). The combined organic phase was dried over Na2SO4, filtered, and evaporated in vacuo and the residue was recrystallized from 30% EtOAc in hexane to afford the products 13a–h.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1328 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 0.99 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.95–2.05 (m, 2H, MeCHHCH2N), 2.85 (s, 3H, MeN), 3.69 (ddd, J = 14.3, 10.2, 6.4 Hz, 1H, MeCH2CHHN), 4.11 (ddd, J = 14.3, 10.2, 6.6 Hz, 1H, MeCH2CHHN), 4.25 (dd, J = 9.1, 6.7 Hz, 1H, SO2CH), 4.55 (d, J = 9.1 Hz, 1H, CHN), 5.71 (d, J = 6.7 Hz, 1H, PhCHO), 7.37–7.46 (m, 5H, Ar), 7.52–7.54 (m, 2H, Ar), 7.65 (td, J = 8.4, 1.2 Hz, 1H, Ar), 7.81 (d, J = 8.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.8, 43.2, 56.8, 66.0, 74.4, 82.0, 115.1, 117.4, 117.6, 124.5, 125.0, 126.7 (2C), 128.9 (2C), 129.1, 133.7, 136.7, 153.9, 156.2, 160.9 ppm; m/z (EI, 70 eV) 426 (49, M+), 380 (19), 338 (31), 316 (100), 304 (13), 288 (21), 274 (80), 255 (36), 240 (22), 227 (80), 214 (94), 198 (51), 185 (18), 134 (70), 115 (25), 105 (35), 91 (35%); anal. calcd for C22H22N2O5S: C, 61.96; H, 5.20; N, 6.57. Found: C, 61.97; H, 5.17; N, 6.61%.
O), 1328 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 0.99 (t, J = 7.4 Hz, 3H, MeCH2CH2N), 1.95–2.05 (m, 2H, MeCHHCH2N), 2.85 (s, 3H, MeN), 3.69 (ddd, J = 14.3, 10.2, 6.4 Hz, 1H, MeCH2CHHN), 4.11 (ddd, J = 14.3, 10.2, 6.6 Hz, 1H, MeCH2CHHN), 4.25 (dd, J = 9.1, 6.7 Hz, 1H, SO2CH), 4.55 (d, J = 9.1 Hz, 1H, CHN), 5.71 (d, J = 6.7 Hz, 1H, PhCHO), 7.37–7.46 (m, 5H, Ar), 7.52–7.54 (m, 2H, Ar), 7.65 (td, J = 8.4, 1.2 Hz, 1H, Ar), 7.81 (d, J = 8.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.8, 43.2, 56.8, 66.0, 74.4, 82.0, 115.1, 117.4, 117.6, 124.5, 125.0, 126.7 (2C), 128.9 (2C), 129.1, 133.7, 136.7, 153.9, 156.2, 160.9 ppm; m/z (EI, 70 eV) 426 (49, M+), 380 (19), 338 (31), 316 (100), 304 (13), 288 (21), 274 (80), 255 (36), 240 (22), 227 (80), 214 (94), 198 (51), 185 (18), 134 (70), 115 (25), 105 (35), 91 (35%); anal. calcd for C22H22N2O5S: C, 61.96; H, 5.20; N, 6.57. Found: C, 61.97; H, 5.17; N, 6.61%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1352 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 0.97 (t, J = 7.4 Hz, 3H, MeCH2CH2CH2N), 1.30–1.50 (m, 2H, MeCHHCH2CH2N), 1.85–2.04 (m, 2H, MeCH2CHHCH2N), 2.85 (s, 3H, MeN), 3.72 (ddd, J = 14.3, 11.0, 5.5 Hz, 1H, MeCH2CH2CHHN), 4.17 (ddd, J = 14.3, 11.0, 5.7 Hz, 1H, MeCH2CH2CHHN), 4.25 (dd, J = 9.0, 6.7 Hz, 1H, SO2CH), 4.55 (d, J = 9.0 Hz, 1H, CHN), 5.71 (d, J = 6.7 Hz, 1H, PhCHO), 7.37–7.45 (m, 5H, Ar), 7.52–7.54 (m, 2H, Ar), 7.66 (td, J = 8.5, 1.3 Hz, 1H, Ar), 7.81 (d, J = 8.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.1, 32.4, 43.2, 55.2, 66.0, 74.4, 82.1, 115.2, 117.4, 117.6, 124.5, 125.0, 126.7 (2C), 128.9 (2C), 129.1, 133.7, 136.8, 153.9, 156.2, 160.9 ppm; m/z (EI, 70 eV) 440 (20, M+), 394 (12), 359 (9), 338 (31), 330 (100), 288 (19), 274 (69), 240 (18), 227 (88), 214 (49), 198 (46), 185 (14), 134 (63), 115 (28), 105 (34), 115 (30), 105 (36), 91 (38), 77 (74%); anal. calcd for C23H24N2O5S: C, 62.71; H, 5.49; N, 6.36. Found: C, 62.74; H, 5.50; N, 5.54%.
O), 1352 (–SO2), 1144 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 0.97 (t, J = 7.4 Hz, 3H, MeCH2CH2CH2N), 1.30–1.50 (m, 2H, MeCHHCH2CH2N), 1.85–2.04 (m, 2H, MeCH2CHHCH2N), 2.85 (s, 3H, MeN), 3.72 (ddd, J = 14.3, 11.0, 5.5 Hz, 1H, MeCH2CH2CHHN), 4.17 (ddd, J = 14.3, 11.0, 5.7 Hz, 1H, MeCH2CH2CHHN), 4.25 (dd, J = 9.0, 6.7 Hz, 1H, SO2CH), 4.55 (d, J = 9.0 Hz, 1H, CHN), 5.71 (d, J = 6.7 Hz, 1H, PhCHO), 7.37–7.45 (m, 5H, Ar), 7.52–7.54 (m, 2H, Ar), 7.66 (td, J = 8.5, 1.3 Hz, 1H, Ar), 7.81 (d, J = 8.1 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.1, 32.4, 43.2, 55.2, 66.0, 74.4, 82.1, 115.2, 117.4, 117.6, 124.5, 125.0, 126.7 (2C), 128.9 (2C), 129.1, 133.7, 136.8, 153.9, 156.2, 160.9 ppm; m/z (EI, 70 eV) 440 (20, M+), 394 (12), 359 (9), 338 (31), 330 (100), 288 (19), 274 (69), 240 (18), 227 (88), 214 (49), 198 (46), 185 (14), 134 (63), 115 (28), 105 (34), 115 (30), 105 (36), 91 (38), 77 (74%); anal. calcd for C23H24N2O5S: C, 62.71; H, 5.49; N, 6.36. Found: C, 62.74; H, 5.50; N, 5.54%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1346 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.89 (s, 3H, MeN), 4.23 (br s, 1H, SO2CH), 4.59 (d, J = 8.9 Hz, 1H, CHN), 4.68 (d, J = 15.2 Hz, 1H, PhCHHN), 5.60 (d, J = 15.2 Hz, 1H, PhCHHN), 5.72 (d, J = 6.5 Hz, 1H, PhCHO), 6.97 (t, J = 7.4 Hz, 1H, Ar), 7.13 (d, J = 7.6 Hz, 1H, Ar), 7.30–7.52 (m, 12H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 43.2, 59.6, 66.3, 74.1, 82.2, 115.2, 116.8, 117.4, 124.5, 125.6, 126.7 (2C), 128.6, 128.8 (2C), 129.0 (2C), 129.1 (3C), 133.5, 135.8, 136.7, 153.7, 157.0, 161.0 ppm; m/z (EI, 70 eV) 474 (39, M+), 364 (72), 319 (8), 303 (43), 286 (8), 274 (49), 246 (10), 230 (24), 213 (52), 202 (14), 136 (88), 105 (25), 91 (100%); anal. calcd for C26H22N2O5S: C, 65.81; H, 4.67; N, 5.90. Found: C, 62.80; H, 5.71; N, 5.79%.
O), 1346 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 2.89 (s, 3H, MeN), 4.23 (br s, 1H, SO2CH), 4.59 (d, J = 8.9 Hz, 1H, CHN), 4.68 (d, J = 15.2 Hz, 1H, PhCHHN), 5.60 (d, J = 15.2 Hz, 1H, PhCHHN), 5.72 (d, J = 6.5 Hz, 1H, PhCHO), 6.97 (t, J = 7.4 Hz, 1H, Ar), 7.13 (d, J = 7.6 Hz, 1H, Ar), 7.30–7.52 (m, 12H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 43.2, 59.6, 66.3, 74.1, 82.2, 115.2, 116.8, 117.4, 124.5, 125.6, 126.7 (2C), 128.6, 128.8 (2C), 129.0 (2C), 129.1 (3C), 133.5, 135.8, 136.7, 153.7, 157.0, 161.0 ppm; m/z (EI, 70 eV) 474 (39, M+), 364 (72), 319 (8), 303 (43), 286 (8), 274 (49), 246 (10), 230 (24), 213 (52), 202 (14), 136 (88), 105 (25), 91 (100%); anal. calcd for C26H22N2O5S: C, 65.81; H, 4.67; N, 5.90. Found: C, 62.80; H, 5.71; N, 5.79%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1346 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 2.36 (s, 3H, Me), 2.88 (s, 3H, MeN), 4.20 (br s, 1H, SO2CH), 4.57 (d, J = 8.9 Hz, 1H, CHN), 4.65 (d, J = 15.2 Hz, 1H, PhCHHN), 5.55 (d, J = 15.2 Hz, 1H, PhCHHN), 5.72 (d, J = 6.5 Hz, 1H, PhCHO), 7.02 (t, J = 7.5 Hz, 1H, Ar), 7.14–7.54 (m, 12H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 43.2, 59.4, 66.3, 74.0, 82.1, 115.1, 116.8, 117.5, 124.4, 125.7, 126.7 (2C), 129.0 (2C), 129.1 (3C), 129.4 (2C), 132.7, 133.5, 136.8, 138.4, 153.7, 157.1, 161.0 ppm; m/z (EI, 70 eV) 488 (9, M+), 378 (39), 317 (14), 289 (14), 274 (30), 246 (8), 230 (19), 213 (18), 202 (13), 136 (51), 105 (100), 91 (21), 77 (78%); anal. calcd for C27H24N2O5S: C, 66.38; H, 4.95; N, 5.73. Found: C, 66.40; H, 4.96; N, 5.71%.
O), 1346 (–SO2), 1141 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.50; 1H NMR (300 MHz, CDCl3) δ 2.36 (s, 3H, Me), 2.88 (s, 3H, MeN), 4.20 (br s, 1H, SO2CH), 4.57 (d, J = 8.9 Hz, 1H, CHN), 4.65 (d, J = 15.2 Hz, 1H, PhCHHN), 5.55 (d, J = 15.2 Hz, 1H, PhCHHN), 5.72 (d, J = 6.5 Hz, 1H, PhCHO), 7.02 (t, J = 7.5 Hz, 1H, Ar), 7.14–7.54 (m, 12H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 43.2, 59.4, 66.3, 74.0, 82.1, 115.1, 116.8, 117.5, 124.4, 125.7, 126.7 (2C), 129.0 (2C), 129.1 (3C), 129.4 (2C), 132.7, 133.5, 136.8, 138.4, 153.7, 157.1, 161.0 ppm; m/z (EI, 70 eV) 488 (9, M+), 378 (39), 317 (14), 289 (14), 274 (30), 246 (8), 230 (19), 213 (18), 202 (13), 136 (51), 105 (100), 91 (21), 77 (78%); anal. calcd for C27H24N2O5S: C, 66.38; H, 4.95; N, 5.73. Found: C, 66.40; H, 4.96; N, 5.71%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1357 (–SO2), 1147 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.43; 1H NMR (300 MHz, CDCl3) δ 1.01 (t, J = 7.3 Hz, 1H, MeCH2CH2N), 1.98–2.10 (m, 2H, MeCHHCH2N), 3.80 (ddd, J = 14.2, 10.8, 6.5 Hz, 1H, MeCH2CHHN), 4.12–4.31 {[4H, consisting s, 2H (4.17), PhCH2N]; [m, 1H, MeCH2CHHN]; and dd, 1H, J = 8.8, 6.5 Hz, 1H (4.28), SO2CH}, 4.85 (d, J = 8.8 Hz, 1H, CHN), 5.74 (d, J = 6.4 Hz, 1H, PhCHO), 7.22–7.54 (m, 12H, Ar), 7.66 (t, J = 7.6 Hz, 1H, Ar), 7.83 (d, J = 7.8 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.9, 56.9, 60.1, 64.6, 74.3, 82.1, 115.3, 117.3, 117.7, 124.6, 125.0, 126.7 (2C), 127.4, 128.2 (2C), 128.4 (2C), 128.9 (2C), 129.0, 133.8, 136.8, 136.9, 153.9, 156.3, 161.2 ppm; m/z (EI, 70 eV) 502 (17, M+), 411 (42), 380 (8), 369 (7), 331 (15), 316 (70), 303 (8), 288 (16), 274 (31), 249 (21), 227 (26), 212 (12), 198 (42), 105 (36), 91 (100%); anal. calcd for C28H26N2O5S: C, 66.91; H, 5.21; N, 5.57. Found: C, 66.90; H, 5.25; N, 5.68%.
O), 1357 (–SO2), 1147 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.43; 1H NMR (300 MHz, CDCl3) δ 1.01 (t, J = 7.3 Hz, 1H, MeCH2CH2N), 1.98–2.10 (m, 2H, MeCHHCH2N), 3.80 (ddd, J = 14.2, 10.8, 6.5 Hz, 1H, MeCH2CHHN), 4.12–4.31 {[4H, consisting s, 2H (4.17), PhCH2N]; [m, 1H, MeCH2CHHN]; and dd, 1H, J = 8.8, 6.5 Hz, 1H (4.28), SO2CH}, 4.85 (d, J = 8.8 Hz, 1H, CHN), 5.74 (d, J = 6.4 Hz, 1H, PhCHO), 7.22–7.54 (m, 12H, Ar), 7.66 (t, J = 7.6 Hz, 1H, Ar), 7.83 (d, J = 7.8 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 11.3, 23.9, 56.9, 60.1, 64.6, 74.3, 82.1, 115.3, 117.3, 117.7, 124.6, 125.0, 126.7 (2C), 127.4, 128.2 (2C), 128.4 (2C), 128.9 (2C), 129.0, 133.8, 136.8, 136.9, 153.9, 156.3, 161.2 ppm; m/z (EI, 70 eV) 502 (17, M+), 411 (42), 380 (8), 369 (7), 331 (15), 316 (70), 303 (8), 288 (16), 274 (31), 249 (21), 227 (26), 212 (12), 198 (42), 105 (36), 91 (100%); anal. calcd for C28H26N2O5S: C, 66.91; H, 5.21; N, 5.57. Found: C, 66.90; H, 5.25; N, 5.68%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1346 (–SO2), 1140 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 0.98 (t, J = 7.3 Hz, 1H, MeCH2CH2CH2N), 1.35–1.49 (m, 2H, MeCHHCH2CH2N), 1.94–2.05 (m, 2H, MeCH2CHHCH2N), 3.87 (ddd, J = 14.0, 11.4, 5.6 Hz, 1H, MeCH2CH2CHHN), 4.12–4.32 {[4H, consisting AB-q, J = 14.9 Hz, 2H (4.17), PhCH2N]; [m, 1H, MeCH2CH2CHHN]; and dd, 1H, J = 8.9, 6.8 Hz, 1H (4.29), SO2CH}, 4.84 (d, J = 9.0 Hz, 1H, CHN), 5.73 (d, J = 6.8 Hz, 1H, PhCHO), 7.22–7.54 (m, 12H, Ar), 7.66 (t, 1H, J = 7.6 Hz, Ar), 7.82 (d, J = 7.8 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.2, 32.5, 55.0, 60.1, 64.6, 74.4, 82.1, 115.3, 117.4117.7, 124.5, 125.0, 126.7 (2C), 127.3, 128.2 (2C), 128.4 (2C), 128.9 (2C), 129.0, 133.8, 136.8, 136.9, 153.9, 156.2, 161.2 ppm; m/z (EI, 70 eV) 516 (5, M+), 425 (36), 394 (7), 369 (8), 345 (14), 330 (95), 303 (22), 288 (17), 274 (44), 249 (28), 220 (29), 198 (65), 105 (58), 91 (100%); anal. calcd for C29H28N2O5S: C, 67.42; H, 5.46; N, 5.42. Found: C, 67.50; H, 5.46; N, 5.48%.
O), 1346 (–SO2), 1140 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.45; 1H NMR (300 MHz, CDCl3) δ 0.98 (t, J = 7.3 Hz, 1H, MeCH2CH2CH2N), 1.35–1.49 (m, 2H, MeCHHCH2CH2N), 1.94–2.05 (m, 2H, MeCH2CHHCH2N), 3.87 (ddd, J = 14.0, 11.4, 5.6 Hz, 1H, MeCH2CH2CHHN), 4.12–4.32 {[4H, consisting AB-q, J = 14.9 Hz, 2H (4.17), PhCH2N]; [m, 1H, MeCH2CH2CHHN]; and dd, 1H, J = 8.9, 6.8 Hz, 1H (4.29), SO2CH}, 4.84 (d, J = 9.0 Hz, 1H, CHN), 5.73 (d, J = 6.8 Hz, 1H, PhCHO), 7.22–7.54 (m, 12H, Ar), 7.66 (t, 1H, J = 7.6 Hz, Ar), 7.82 (d, J = 7.8 Hz, 1H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 13.7, 20.2, 32.5, 55.0, 60.1, 64.6, 74.4, 82.1, 115.3, 117.4117.7, 124.5, 125.0, 126.7 (2C), 127.3, 128.2 (2C), 128.4 (2C), 128.9 (2C), 129.0, 133.8, 136.8, 136.9, 153.9, 156.2, 161.2 ppm; m/z (EI, 70 eV) 516 (5, M+), 425 (36), 394 (7), 369 (8), 345 (14), 330 (95), 303 (22), 288 (17), 274 (44), 249 (28), 220 (29), 198 (65), 105 (58), 91 (100%); anal. calcd for C29H28N2O5S: C, 67.42; H, 5.46; N, 5.42. Found: C, 67.50; H, 5.46; N, 5.48%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1385 (–SO2), 1148 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 4.19–4.31 {[3H, consisting AB-q, J = 14.7 Hz, 2H (4.25) PhCH2N]; and br s, 1H (4.31), SO2CH}, 4.74 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 4.92 (d, J = 8.8 Hz, 1H, CHN), 5.70 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 5.77 (d, J = 6.4 Hz, 1H, PhCHO), 6.92 (t, J = 7.5 Hz, 1H, Ar), 7.05 (d, J = 7.7 Hz, 1H, Ar), 7.27–7.55 (m, 17H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 60.0, 60.1, 65.1, 73.9, 82.4, 115.2, 116.7, 117.6, 124.4, 125.6, 126.7 (2C), 127.5, 128.3 (2C), 128.5 (2C), 128.6, 128.88 (2C), 128.94 (2C), 129.1, 129.2 (2C), 133.6, 135.9, 136.7, 136.8, 153.7, 157.4, 161.3 ppm; m/z (EI, 70 eV) 550 (7, M+), 459 (11), 364 (44), 289 (20), 275 (75), 230 (22), 220 (26), 202 (12), 106 (38), 91 (100%); anal. calcd for C32H26N2O5S: C, 69.80; H, 4.76; N, 5.09. Found: C, 69.82; H, 4.71; N, 4.95%.
O), 1385 (–SO2), 1148 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.41; 1H NMR (300 MHz, CDCl3) δ 4.19–4.31 {[3H, consisting AB-q, J = 14.7 Hz, 2H (4.25) PhCH2N]; and br s, 1H (4.31), SO2CH}, 4.74 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 4.92 (d, J = 8.8 Hz, 1H, CHN), 5.70 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 5.77 (d, J = 6.4 Hz, 1H, PhCHO), 6.92 (t, J = 7.5 Hz, 1H, Ar), 7.05 (d, J = 7.7 Hz, 1H, Ar), 7.27–7.55 (m, 17H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 60.0, 60.1, 65.1, 73.9, 82.4, 115.2, 116.7, 117.6, 124.4, 125.6, 126.7 (2C), 127.5, 128.3 (2C), 128.5 (2C), 128.6, 128.88 (2C), 128.94 (2C), 129.1, 129.2 (2C), 133.6, 135.9, 136.7, 136.8, 153.7, 157.4, 161.3 ppm; m/z (EI, 70 eV) 550 (7, M+), 459 (11), 364 (44), 289 (20), 275 (75), 230 (22), 220 (26), 202 (12), 106 (38), 91 (100%); anal. calcd for C32H26N2O5S: C, 69.80; H, 4.76; N, 5.09. Found: C, 69.82; H, 4.71; N, 4.95%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1345 (–SO2), 1148 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.46; 1H NMR (300 MHz, CDCl3) δ 2.38 (s, 3H, Me), 4.23–4.29 {[3H, consisting s, 2H (4.24) PhCH2N]; and br s, 1H (4.30), SO2CH}, 4.70 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 4.90 (d, J = 8.8 Hz, 1H, CHN), 5.65 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 5.76 (d, J = 6.5 Hz, 1H, PhCHO), 6.96 (t, J = 7.6 Hz, 1H, Ar), 7.14–7.53 (m, 17H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 60.0, 60.1, 65.0, 73.9, 82.3, 115.1, 116.7, 117.6, 124.4, 125.7, 126.7 (2C), 127.4, 128.3 (2C), 128.4 (2C), 128.9 (2C), 129.0, 129.1 (2C), 129.5 (2C), 132.8, 133.5, 136.7, 136.9, 138.4, 153.7, 157.4, 161.3 ppm; m/z (EI, 70 eV) 564 (5, M+), 378 (25), 289 (34), 274 (12), 220 (8), 105 (100), 91 (83%); anal. calcd for C33H28N2O5S: C, 70.19; H, 5.00; N, 4.96. Found: C, 70.23; H, 5.06; N, 5.01%.
O), 1345 (–SO2), 1148 (–SO2) cm−1; Rf (33% EtOAc/hexane) 0.46; 1H NMR (300 MHz, CDCl3) δ 2.38 (s, 3H, Me), 4.23–4.29 {[3H, consisting s, 2H (4.24) PhCH2N]; and br s, 1H (4.30), SO2CH}, 4.70 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 4.90 (d, J = 8.8 Hz, 1H, CHN), 5.65 (d, J = 15.1 Hz, 1H, PhCHHNSO2), 5.76 (d, J = 6.5 Hz, 1H, PhCHO), 6.96 (t, J = 7.6 Hz, 1H, Ar), 7.14–7.53 (m, 17H, Ar) ppm; 13C NMR (75 MHz, CDCl3) δ 21.2, 60.0, 60.1, 65.0, 73.9, 82.3, 115.1, 116.7, 117.6, 124.4, 125.7, 126.7 (2C), 127.4, 128.3 (2C), 128.4 (2C), 128.9 (2C), 129.0, 129.1 (2C), 129.5 (2C), 132.8, 133.5, 136.7, 136.9, 138.4, 153.7, 157.4, 161.3 ppm; m/z (EI, 70 eV) 564 (5, M+), 378 (25), 289 (34), 274 (12), 220 (8), 105 (100), 91 (83%); anal. calcd for C33H28N2O5S: C, 70.19; H, 5.00; N, 4.96. Found: C, 70.23; H, 5.06; N, 5.01%.Acknowledgements
We acknowledge the University of Tehran for financial support of this research.Notes and references
- For reviews on 1,3-dipolar cycloaddition reactions, see: (a) A. Padwa, Angew. Chem., Int. Ed. Engl., 1976, 15, 123 CrossRef; (b) J. W. Lown, in 1,3-Dipolar Cycloaddition Chemistry, ed. A. Padwa, Wiley, New York, NY, 1984, vol. 1, p. 2 Search PubMed; (c) K. V. Gothelf and K. A. Jørgensen, Chem. Rev., 1998, 98, 863 CrossRef CAS PubMed; (d) K. V. Gothelf, in Cycloaddition Reactions in Organic Synthesis, ed. S. Kobayashi and S. K. A. Jorgensen, Wiley-VCH, Weinheim, 2002, p. 211 Search PubMed; (e) I. Coldham and R. Hufton, Chem. Rev., 2005, 105, 2765 CrossRef CAS PubMed; (f) F. Chen and J. Huang, Chem. Rev., 2005, 105, 4671 CrossRef CAS PubMed; (g) G. Pandey, P. Banerjee and S. R. Gadre, Chem. Rev., 2006, 106, 4484 CrossRef CAS PubMed; (h) V. Naira and T. D. Sujaa, Tetrahedron, 2007, 63, 12247 CrossRef; (i) M. S. Singh, S. Chowdhury and S. Koley, Tetrahedron, 2016, 72, 1603 CrossRef CAS.
- For some recent examples on intramolecular 1,3-dipolar cycloadditions of azomethine ylides, see: (a) I. Coldham, B. C. Dobson, A. I. Franklin and S. R. Fletcher, Tetrahedron Lett., 2007, 48, 873 CrossRef CAS; (b) L. Meng, J. C. Fettinger and M. J. Kurth, Org. Lett., 2007, 9, 5055 CrossRef CAS PubMed; (c) M. Bakthadoss, N. Sivakumar, G. Sivakumar and G. Murugan, Tetrahedron Lett., 2008, 49, 820 CrossRef CAS; (d) E. Ramesh and R. Raghunathan, Tetrahedron Lett., 2008, 49, 1125 CrossRef CAS; (e) I. Kim, H.-K. Na, K. R. Kim, S. G. Kim and G. H. Lee, Synlett, 2008, 13, 2069 Search PubMed; (f) S. Kathiravan, E. Ramesh and R. Raghunathan, Tetrahedron Lett., 2009, 50, 2389 CrossRef CAS; (g) S. Kathiravan, D. Vijayarajan and R. Raghunathan, Tetrahedron Lett., 2010, 51, 3065 CrossRef CAS; (h) G. Pandey, N. R. Gupta and S. R. Gadre, Eur. J. Org. Chem., 2011, 740 CrossRef CAS; (i) A. Hazra, Y. P. Bharitkar, A. Maity, S. Mondal and N. B. Mondal, Tetrahedron Lett., 2013, 54, 4339 CrossRef CAS; (j) N. J. Parmar, B. R. Pansuriya, B. M. Labana, R. Kant and V. K. Gupta, RSC Adv., 2013, 3, 17527 RSC; (k) R. Rajesh and R. Raghunathan, Synlett, 2013, 24, 2107 CrossRef CAS; (l) M. Nayak, N. Rastogi and S. Batra, Tetrahedron, 2013, 69, 5029 CrossRef CAS; (m) S. Picard, F. Lecornué and G. Bashiardes, Synlett, 2014, 25, 1106 CrossRef; (n) M. Bakthadoss, D. Kannan, J. Srinivasanb and V. Vinayagam, Org. Biomol. Chem., 2015, 13, 2870 RSC; (o) T. R. Sutariya, B. M. Labana, N. J. Parmar, R. Kant, V. K. Gupta, G. B. Platac and J. M. Padrónc, New J. Chem., 2015, 39, 2657 RSC; (p) E. L. Martínez-Mora, M. A. Caracas, C. H. Escalante, C. Espinoza-Hicks, H. Quiroz-Florentino, F. Delgado and J. Tamariz, Synthesis, 2016, 48, 1055 CrossRef.
- For some recent examples on intramolecular 1,3-dipolar cycloadditions of nitrone ylides, see: (a) U. Chiacchio, A. Corsaro, G. Gumina, V. Pistará, A. Rescifina, M. Alessi, A. Piperno, G. Pomeo and R. Roberto, Tetrahedron, 1997, 53, 13855 CrossRef CAS; (b) E. M. Beccalli, G. Broggini, G. La Rosa, D. Passarella and T. Pilati, J. Org. Chem., 2000, 65, 8924 CrossRef CAS PubMed; (c) Q. Zaho, F. Han and D. L. Romero, J. Org. Chem., 2000, 67, 3317 CrossRef; (d) G. K. Tranmer and W. Tam, J. Org. Chem., 2001, 66, 5113 CrossRef CAS PubMed; (e) K. Singha, A. Roy, P. K. Dutta, S. Tripathi, S. Sahabuddin, B. Achari and S. B. Mandal, J. Org. Chem., 2004, 69, 6507 CrossRef CAS PubMed; (f) P. K. Kalita, B. Baruah and P. J. Bhuyan, Tetrahedron Lett., 2006, 47, 7779 CrossRef CAS; (g) A. Chatterjee and P. K. Bhattacharya, J. Org. Chem., 2006, 71, 345 CrossRef CAS PubMed; (h) G. Singh, M. P. S. Ishar, V. Gupta, G. Singh, M. Kalyan and S. S. Bhella, Tetrahedron, 2007, 63, 4773 CrossRef CAS; (i) M. Bakthadoss and G. Murugan, Eur. J. Org. Chem., 2010, 5825 CrossRef CAS; (j) S. Saubern, J. M. Macdonald, J. H. Ryan, R. C. J. Woodgate, T. S. Louie, M. J. Fuchter, J. M. White and A. B. Holmes, Tetrahedron, 2010, 66, 2761 CrossRef CAS; (k) S. Majumder and P. J. Bhuyan, Tetrahedron Lett., 2012, 53, 762 CrossRef CAS; (l) E. H. Krenske, S. Agopcan, V. Aviyente, K. N. Houk, B. A. Johnson and A. B. Holmes, J. Am. Chem. Soc., 2012, 134, 12010 CrossRef CAS PubMed; (m) M. Bakthadoss, G. Sivakumar and D. S. Sharada, Synthesis, 2013, 45, 237 CrossRef CAS; (n) R. Rajesh, M. Suresh and R. Raghunathan, Tetrahedron Lett., 2014, 55, 69 Search PubMed; (o) M. Bakthadoss and J. Srinivasan, RSC Adv., 2015, 5, 67206 RSC; (p) M. Bakthadoss and A. Devaraj, Tetrahedron Lett., 2015, 56, 3954 CrossRef CAS; (q) T. Zhu, J. Xiang, Z. Liu, Q. Dang and X. Bai, Synlett, 2015, 26, 238 CrossRef CAS; (r) F. Chafaa, D. Hellel, A. K. Nacereddine and A. Djerourou, Tetrahedron Lett., 2016, 57, 67 CrossRef CAS.
- (a) R. R. Kumar, S. M. Rajesh, S. Perumal, D. Banerjee, P. Yogeeswari and D. Sriram, Eur. J. Med. Chem., 2010, 45, 411 CrossRef CAS PubMed; (b) A. S. Girgis, Eur. J. Med. Chem., 2009, 44, 91 CrossRef CAS PubMed.
- (a) G. Periyasami, R. Raghunathan, G. Surendiran and N. Mathivanan, Bioorg. Med. Chem. Lett., 2008, 18, 2342 CrossRef CAS PubMed; (b) A. Thangamani, Eur. J. Med. Chem., 2010, 45, 6120 CrossRef CAS PubMed.
- (a) P. Prasanna, K. Balamurugan, S. Perumal, P. Yogeeswari and D. Sriram, Eur. J. Med. Chem., 2010, 45, 5653 CrossRef CAS PubMed; (b) S. V. Karthikeyan, B. D. Bala, V. P. A. Raja, S. Perumal, P. Yogeeswari and D. Sriram, Bioorg. Med. Chem. Lett., 2010, 20, 350 CrossRef CAS PubMed; (c) S. U. Maheswari, K. Balamurugan, S. Perumal, P. Yogeeswari and D. Sriram, Bioorg. Med. Chem. Lett., 2010, 20, 7278 CrossRef CAS PubMed.
- (a) K. Kasahara, H. Iida and C. Kibayashi, J. Org. Chem., 1989, 54, 2225 CrossRef CAS; (b) R. Alibes and F. J. Busque, Tetrahedron, 1998, 54, 11581 CrossRef CAS; (c) U. Chiacchio, A. Corsaro, D. Iannazzo, A. Piperno, A. Procopio, A. Rescifina, G. Romeo and R. Romeo, J. Org. Chem., 2002, 67, 4380 CrossRef CAS PubMed; (d) K. Takao, R. Munakata and K. Tadano, Chem. Rev., 2005, 105, 4779 CrossRef CAS PubMed; (e) N. Carrillo, E. A. Davalos, J. A. Russak and J. W. Bode, J. Am. Chem. Soc., 2006, 128, 1452 CrossRef CAS PubMed.
- (a) J. A. Xiao, H. G. Zhang, S. Liang, J. W. Ren, H. Yang and X. Q. Chen, J. Org. Chem., 2013, 78, 11577 CrossRef CAS PubMed; (b) K. Revathy and A. Lalitha, RSC Adv., 2014, 4, 279 RSC; (c) P. Dhanalakshmi, S. S. Babu, S. Thimmarayaperumal and S. Shanmugam, RSC Adv., 2015, 5, 33705 RSC; (d) M. Bakthadoss, N. Sivakumar and A. Devaraj, Tetrahedron Lett., 2015, 56, 4980 CrossRef CAS.
- (a) N. Morita, K. Fukui, J. Irikuchi, H. Sato, Y. Takano, I. Okamoto, H. Ishibashi and O. Tamura, J. Org. Chem., 2008, 73, 7164 CrossRef CAS PubMed; (b) K. V. Kumar, K. Dhananjaya and K. S. Rangappa, Synth. Commun., 2002, 32, 1887 CrossRef.
- (a) J. Drews, Science, 2000, 287, 1960 CrossRef CAS PubMed; (b) A. Scozzafava, T. Owa, A. Mastrolorenzo and C. T. Supuran, Curr. Med. Chem., 2003, 10, 925 CrossRef CAS PubMed.
- S. Hanessian, H. Sailes and E. Therrien, Tetrahedron, 2003, 59, 7047 CrossRef CAS.
- For recent reviews on sultam chemistry, see: (a) A. Scozzafava, T. Owa, A. Mastrolorenzo and C. T. Supuran, Curr. Med. Chem., 2003, 10, 925 CrossRef CAS PubMed; (b) M. D. McReynolds, J. M. Dougherty and R. R. Hanson, Chem. Rev., 2004, 104, 2239 CrossRef CAS PubMed; (c) Z. Liu and Y. Takeuchi, Heterocycles, 2009, 78, 1387 CrossRef CAS; (d) K. C. Majumdar and S. Mondal, Chem. Rev., 2011, 111, 7749 CrossRef CAS PubMed; (e) V. A. Rassadin, D. S. Grosheva, A. A. Tomashevskii and V. V. Sokolov, Chem. Heterocycl. Compd., 2013, 49, 47 CrossRef.
- X. Rabasseda and S. J. Hopkins, Drugs Today, 1994, 30, 557 CAS.
- G. J. Wells, M. Tao, K. A. Josef and R. Bihovsky, J. Med. Chem., 2001, 44, 3488 CrossRef CAS PubMed.
- F. Brzozowski, F. Saczewski and N. Neamati, Bioorg. Med. Chem. Lett., 2006, 16, 5298 CrossRef PubMed.
- H. Tanimukai, M. Inui, S. Harigushi and J. Kaneko, Biochem. Pharmacol., 1965, 14, 961 CrossRef CAS PubMed.
- T. Wroblewski, A. Graul and J. Castaner, Drugs Future, 1998, 23, 365 CrossRef CAS.
- R. J. Cherney, R. Mo, D. T. Meyer, K. D. Hardman, R.-Q. Liu, M. B. Covington, M. Qian, Z. R. Wasserman, D. D. Christ, J. M. Trzaskos, R. C. Newton and C. P. Decicco, J. Med. Chem., 2004, 47, 2981 CrossRef CAS PubMed.
- R. Di Santol, R. Costil, M. Articol, S. Massa, M. E. Marongiu, A. G. Loi, A. De Montis and P. La Colla, Antiviral Chem. Chemother., 1998, 9, 127 CrossRef.
- A. Scozzafava, T. Owa, A. Mastrolorenzo and C. T. Supuran, Curr. Med. Chem., 2003, 10, 925 CrossRef CAS PubMed.
- M. Zia-ur-Rehman, J. A. Choudary, S. Ahmad and H. L. Siddiqui, Chem. Pharm. Bull., 2006, 54, 1175 CrossRef CAS PubMed.
- C. Valente, R. C. Guedes, R. Moreira, J. Iley, J. Gut and P. Rosenthal, Bioorg. Med. Chem. Lett., 2006, 16, 4115 CrossRef CAS PubMed.
- (a) R. Silvestri, G. Marfe, M. Artico, G. La Regina, A. Lavecchia, E. Novellino, M. Morgante, C. Di Stefano, G. Catalano, G. Filomeni, E. Abruzzese, M. R. Ciriolo, M. A. Russo, S. Amadori, R. Cirilli, F. La Torre and P. Sinibaldi Salimei, J. Med. Chem., 2006, 49, 5840 CrossRef CAS PubMed; (b) N. Lebegue, S. Gallet, N. Flouquet, P. Carato, B. Pfeiffer, P. Renard, S. Leonce, A. Pierre, P. Chavatte and P. Berthelot, J. Med. Chem., 2005, 48, 7363 CrossRef CAS PubMed.
- (a) K. H. Ahn, H.-H. Baek, S. J. Lee and C.-W. Cho, J. Org. Chem., 2000, 65, 7690 CrossRef CAS PubMed; (b) G. Kumaraswamy, M. Padmaja, B. Markondaiah, N. Jena, B. Sridhar and M. U. Kiran, J. Org. Chem., 2006, 71, 337 CrossRef CAS PubMed.
- For recent reviews on coumarin chemistry, see: (a) F. G. Medina, J. G. Marrero, M. Macías-Alonso, M. C. González, I. Córdova-Guerrero, A. G. Teissier García and S. Osegueda-Robles, Nat. Prod. Rep., 2015, 32, 1472 RSC; (b) E. Calcio Gaudino, S. Tagliapietra, K. Martina, G. Palmisano and G. Cravotto, RSC Adv., 2016, 6, 46394 RSC. For anti-inflammatory activity, see: (c) J. Grover and S. M. Jachak, RSC Adv., 2015, 5, 38892 RSC; (d) G. Kayal, K. Jain, S. Malviya and A. Kharia, Int. J. Pharma Sci. Res., 2014, 5, 3577 CAS. For anticancer activity, see: (e) S. Emami and S. Dadashpour, Eur. J. Med. Chem., 2015, 102, 611 CrossRef CAS PubMed; (f) A. Thakur, R. Singla and V. Jaitak, Eur. J. Med. Chem., 2015, 101, 476 CrossRef CAS PubMed; (g) M. Kaur, S. Kohli, S. Sandhu, Y. Bansal and G. Bansal, Adv. Anticancer Agents Med. Chem., 2015, 15, 1032 CrossRef CAS. For antioxidant activity, see: (h) I. Najmanovà, M. Dosedel, R. Hrdina, P. Anzenbacher, T. Filipsky, M. Riha and P. Mladenka, Curr. Top. Med. Chem., 2015, 15, 830 CrossRef. For antituberculosis activity, see: (i) R. S. Keri, B. S. Sasidhar, B. M. Nagaraja and M. A. Santos, Eur. J. Med. Chem., 2015, 100, 257 CrossRef CAS PubMed.
- (a) M. Ghandi, S. J. Tabatabaei Rezaei, A. Yari and A. Taheri, Tetrahedron Lett., 2008, 49, 5899 CrossRef CAS; (b) M. Ghandi, A. Yari, S. J. Tabatabaei Rezaei and A. Taheri, Tetrahedron Lett., 2009, 50, 4724 CrossRef CAS; (c) M. Ghandi, A. Taheri and A. Abbasi, Tetrahedron, 2010, 66, 6744 CrossRef CAS; (d) M. Ghandi, A. Taheri and A. Abbasi, J. Heterocycl. Chem., 2010, 47, 611 CAS; (e) M. Ghandi, E. Mohammadimehr, M. Sadeghzadeh and A. Hasani Bozcheloei, Tetrahedron, 2011, 67, 8484 CrossRef CAS; (f) M. Ghandi, A. Hasani Bozcheloei, S. H. Nazari and M. Sadeghzadeh, J. Org. Chem., 2011, 76, 9975 CrossRef CAS PubMed; (g) M. Ghandi, A. Taheri, A. Hasani Bozcheloei, A. Abbasi and R. Kia, Tetrahedron, 2012, 68, 3641 CrossRef CAS; (h) M. Ghandi, M. T. Nazeri and M. Kubicki, Tetrahedron, 2013, 69, 4979 CrossRef CAS.
- (a) M. Ghandi, S. H. Nazari, A. Hasani Bozcheloei, M. Sadeghzadeh and R. Kia, Tetrahedron Lett., 2011, 52, 6613 CrossRef CAS; (b) M. Ghandi, S. Feizi, F. Ziaie and B. Notash, Tetrahedron, 2014, 70, 2563 CrossRef CAS; (c) M. Ghandi, M. Asle Kia, M. Sadeghzadegh, A. Hasani Bozcheloei and M. Kubicki, J. Heterocycl. Chem., 2015, 52, 1646 CrossRef CAS.
- K.-T. Li, Y.-B. Lin and D.-Y. Yang, Org. Lett., 2012, 14, 1190 CrossRef CAS PubMed.
- (a) M. R. Mallireddigari, V. R. Pallela, E. P. Reddy and M. V. R. Reddy, Synthesis, 2005, 3639 CAS; (b) B. M. Culbertson and S. Dietz, J. Chem. Soc. C, 1968, 992 RSC.
- The CCDC deposition number for compound 9h is 1433802. Formula: C33H28N2O4S. Unit cell parameters: a = 13.148(3) Å, b = 16.285(3) Å, c = 17.991(6) Å, α = 90.00°, β = 133.170(18)°, γ = 90.00°, space group P21/c.†.
- The CCDC deposition number for compound 11a is 998125. Formula: C25H26N2O4S. Unit cell parameters: a = 8.790(2) Å, b = 15.553(2) Å, c = 16.313(2) Å, α = 90.00°, β = 90.00°, γ = 90.00°, space group P212121.†.
- The CCDC deposition number for compound 13h is 1429726. Formula: C33H28N2O5S. Unit cell parameters: a = 14.898(3) Å, b = 11.159(2) Å, c = 17.179(3) Å, α = 90.00°, β = 108.33(3)°, γ = 90.00°, space group P21/n.†.
Footnote |
| † Electronic supplementary information (ESI) available: Copies of 1H NMR, 13C NMR, MS and IR of all the compounds, and crystallographic data for 9h, 11a and 13h. CCDC 1433802, 998125 and 1429726. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra14169b |
| This journal is © The Royal Society of Chemistry 2016 |