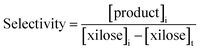Perovskite as nickel catalyst precursor – impact on catalyst stability on xylose aqueous-phase hydrogenation†
Ruddy Moralesa,
Cristian H. Campos a,
J. L. G. Fierroc,
Marco A. Fraga*b and
Gina Pecchi*a
a,
J. L. G. Fierroc,
Marco A. Fraga*b and
Gina Pecchi*a
aDepartamento de Físico-Química, Facultad Ciencias Químicas, Concepción, Chile. E-mail: gpecchi@udec.cl
bInstituto Nacional de Tecnologia/MCTIC, Laboratório de Catálise, Rio de Janeiro, Brazil. E-mail: marco.fraga@int.gov.br
cInstituto Catálisis y Petroleoquímica, CSIC, 28049 Madrid, Spain
First published on 4th July 2016
Abstract
Precursors materials with formula, La1−xCexAl0.18Ni0.82O3 (x = 0.0, 0.1, 0.5, 0.7), were successfully used as precursors to prepare Ni nanoclusters to be used as catalysts in the hydrogenation of xylose to xylitol. For the Ce free and lower Ce content (x = 0.0; 0.1), the perovskite structure was obtained, whereas for a higher Ce content (x = 0.5; 0.7), an ordinary CeO2–La2O3 solid dissolution with no perovskite structure were obtained. Under the reduction conditions, the perovskite structure leads to ∼30% of metallic Ni without any loss of the perovskite structure (x = 0.0; 0.1) and the CeO2–La2O3 solid dissolution allows a Ni reduction of ∼40% of Ni (x = 0.5; 0.7). As expected, the similar reduced Ni content does not show large differences in the aqueous-phase xylose hydrogenation product distribution, or large difference in the precursors. The perovskite structure (x = 0.0; 0.1) and solid dissolution (x = 0.5; 0.7) highlight the importance of the perovskite structure in the remarkable leaching resistance showed by the Ni perovskite-precursor (x = 0.0; 0.1) catalysts, showing no Ni leaching over 6 h of reaction in an aqueous medium.
Introduction
The liquid phase hydrogenation of aldoses on porous metal catalyst is an important process in the synthesis of polyhydric alcohols, which have long been used as natural sweetening agents in the food industry. Xylitol, obtained by the hydrogenation of xylose, is a sweetener molecule, which has been increasingly commercialized1 due to its attractive properties as high solubility in water, stability upon storage and does not caramelize at elevated temperatures. Moreover, the sweetening capacity of xylitol exceeds that of saccharose by 20–25% with no insulin requirement.2,3Xylitol is typically produced by an isothermal hydrogenation process of xylose aqueous solutions in a triphasic batch reactor on a metallic dispersed catalyst. Wisniak et al.3,4 studied different metal catalysts in the hydrogenation of glucose, fructose and xylose, reporting that the activity of the metals decreases in the order, Ru > Ni > Rh > Pd.2 The performance of Ni-based catalysts stands out due to their lower price compared to noble metals and that they can be used efficiently in batch reactors, with high activity and selectivity to polyalcohols. Nevertheless, the major drawback of RANEY® Ni catalysts is their large and rapid deactivation due to accumulation of organic impurities (from the starting material) on the catalyst surface, leading to poisoning of the active sites, and metal leaching.5 These technical hurdles motivate the pursuit of alternative catalysts by changing their chemical formulation and/or their surface architecture through different active surface site assemblies. This can be accomplished by exploring different synthesis procedures and catalyst precursors with distinctive topology or flexible crystalline structure.
Perovskite-type oxides are crystalline structures, represented by the general formula ABO3, where A is a large cation and B is a small cation of the d-transition series. A multitude of chemical compositions is possible because a large number of chemical elements may be accommodated at both A and B positions of such flexible structure. Furthermore, these crystalline structures have a large stability in oxidant atmosphere; and they can be total or partially reduced in a reductive atmosphere. Owing to such interesting properties, they have been exploited extensively as catalyst precursors for many reactions.6,7 The main advantage of perovskite-like oxide as a catalyst precursor is the possible formation of a small and uniform particle size catalyst, which could provide a distinguished catalytic performance.
Ni-containing perovskites have been investigated widely as catalyst precursor for a broad variety of reactions. Natural gas8 and alcohols9 reforming, water–gas shift reaction,10 methane combustion11 and soot oxidation12 can be listed just to mention a few processes. In the case of LaNiO3 perovskite, the reducible element (Ni3+) in the perovskite structure can be reduced easily to metallic Ni0 dispersed on La2O3 under the appropriate H2 reduction conditions.13 In hydrogenation reactions, one strategy to tailor catalyst selectivity is the employment of promoters that can also be accommodated into perovskite catalyst precursor structure leading to a well-dispersed system. Ceria, a well-known promoter with outstanding redox and acid–base properties, has been exploited in this regard.14,15
In this study, perovskite structures were investigated as a strategy to stabilize Ni nanoclusters in metal catalysts for the aqueous-phase hydrogenation of xylose to xylitol. All catalyst presented a nominal Ni loading of 20 wt%, which was ensured by adding Al alongside in the B position. Different Ce loadings were considered to partially substitute La in the A position. A full structural and textural characterization of synthesized La1−xCexAl0.18Ni0.82O3 (x = 0.0, 0.1, 0.5, and 0.7) structures was carried out to explain the activity and selectivity of the oxidized and reduced catalysts.
Experimental
Preparation of the catalysts
La1−xCexAl0.18Ni0.82O3 (x = 0.0, 0.1, 0.5, 0.7) materials, all of them with a 20 wt% nominal Ni loading and different Ce and La contents, were prepared by a self-combustion method.16 Glycine (H2NCH2CO2H), used as the ignition promoter, was added to an aqueous solution of metal nitrates with the appropriate stoichiometry to obtain a NO3−/NH2 (molar ratio) = 1. The resulting solution was slowly evaporated until a vitreous gel was obtained. The gel was heated to more than 265 °C, temperature at which the ignition reaction occurs producing a powdered precursor. After the combustion process, the powders were ground and subjected to air thermal treatment calcination to eliminate the remaining carbon and obtain the perovskite structure. The solids were crushed and sieved to obtain the required particle size (<200 μm) and then finally calcined at a heating rate of 1 °C min−1 up to 700 °C and maintained for 5 h.Because hydrogen was used to activate the solids prior to catalytic activity measurements, the calcined perovskites were first reduced under a pure hydrogen flow (50 mL min−1) at 500 °C for 2 h to carry out ex situ characterization. After reduction, the reactor was purged with nitrogen, cooled down until room temperature and then placed in a cryostatic bath (a mixture of liquid nitrogen and isopropyl alcohol at −70 °C). Once the temperature was reached, a flow of 5% O2/N2 was admitted into the reactor for 1 h. Finally, the bath was removed and the catalysts were stabilized at room temperature for 1 h. These materials are referred to as Ni-reduced solids hereinafter.
Samples were labeled according to their chemical composition: x = 0.0 to La1.0Al0.18Ni0.82O3; x = 0.1 to La0.9Ce0.1Al0.18Ni0.82O3; x = 0.5 to La0.5Ce0.5Al0.18Ni0.82O3; x = 0.7 to La0.3Ce0.7Al0.18Ni0.82O3.
Characterization
Chemical analysis was conducted by atomic absorption spectrometry (AAS) using a Perkin Elmer instrument 3100. The samples were heated in an open Teflon beaker with 2.5 mL of HNO3, 2.5 mL of HCl and 5.0 mL of HF (all in high concentration). Once the samples were dissolved, HF was evaporated to a minimum volume. Finally, a spatula tip of H3BO3(s) and up to 50 mL total volume of water was added in a plastic volumetric flask.The specific areas were calculated using the BET method from the nitrogen adsorption isotherms obtained on a Micromeritics ASAP 2010 apparatus at −196 °C. The samples were previously pretreated at 150 °C under a nitrogen atmosphere for 2 h to dehydrate and clean the catalysts surface from adsorbed gases, followed by vacuum. The isotherms were recorded taking 25 relative pressure points ranging from 0.0 to 1.0 for adsorption and 23 for desorption process.
X-ray powder diffraction (XRD) patterns were obtained with nickel-filtered CuKα1 radiation (λ = 1.5418 Å) using a Rigaku diffractometer and collected in the 2θ range of 20–70° in steps of 2 °C min−1.
Temperature programmed reduction (TPR) experiments were performed in a TPR/TPD 2900 Micromeritics system with a thermal conductivity detector (TCD). Prior to the reduction experiments, the samples (50 mg) were thermally treated under an air stream at 700 °C to remove any contaminants on the catalyst surface. The reduction profiles were recorded under 10% H2/N2 flow at 25 mL min−1 at a heating rate of 5 °C min−1 from room temperature to 1000 °C.
The NH3-TPD experiments were carried out on the reduced samples (2 h at 500 °C) on a TPR/TPD 2010 apparatus, saturating the catalyst surface at 100 °C with ammonia pulses. The samples were cooled to room temperature and once the baseline was restored, the temperature was increased linearly up to 500 °C.
XPS measurements were performed using a VG Escalab 200R electron spectrometer equipped with a hemispherical electron analyzer and a Mg Kα (1253.6 eV) X-ray source. Prior to analysis, the samples were reduced in situ under hydrogen at 500 °C for 1 h and transported to the analysis chamber without contact with air. The charging effects on the samples were corrected by taking the C 1s peak of adventitious carbon at 284.8 eV. The peaks were decomposed into several components assuming a Gaussian/Lorentzian shape.
Catalytic activity
The catalytic activity evaluation in the aqueous-phase hydrogenation of xylose was performed in a semi-batch Parr reactor at conditions (catalyst weight and agitation speed) to ensure the absence of any transport limitations, evaluated by a Weisz–Prater parameter. The catalysts were reduced previously to the reaction in a conventional fixed bed reactor at 36 mL min−1 of H2 and heating of 5 °C min−1 up to 500 °C and maintained at this temperature for 2 h. Subsequently, reducing gas was switched to a nitrogen stream and the catalyst was cooled to room temperature.Preliminary reaction runs were performed to settle the suitable reaction conditions. A selected sample (x = 0.1 catalyst) was used and reaction temperature and H2 pressure in the reactor were modified systematically. High xylose conversion to xylitol and low secondary product formation were taken into consideration to evaluate the catalyst performance of the reaction carried out under three different experimental conditions: (i) 130 °C and 25 bar of H2; (ii) 100 °C and 25 bar of H2; (iii) 100 °C and 15 bar of H2. Hydrogenated products were the main compounds when the reaction was performed at 100 °C and 25 bar of H2; therefore, those were the operation conditions used to assess the behaviour of the synthesized samples.
In a typical run, the reactor was fed with 40 mL of water and 100 mg of catalyst, sealed and purged five times with N2 and then with H2 to avoid contamination with other gases. The reactor was heated to 100 °C and stirred at 600 rpm. Once the desired temperature was reached, 40 mL of xylose aqueous solution was admitted into the reactor, providing a starting reaction medium at 0.083 mol xylose per L. The hydrogen pressure was adjusted and maintained constant at 25 bar. Samples were taken periodically during the reaction and analysed using a Waters HPLC chromatograph model Alliance e2695, coupled to a PDA detector model 2998 at a wavelength of 280 nm and a RID detector model 2414 at 50 °C using a Biorad Aminex HPX-87H column maintained at 65 °C. A H2SO4 aqueous solution at 5 mmol L−1 was used as the mobile phase at 0.7 mL min−1.
The total xylose conversion and product selectivity were calculated as:
Results and discussion
Atomic absorption spectrometry (AAS)
The elemental composition of Ni in the prepared solids determined by AAS is compiled in Table 1. The obtained values are similar to the nominal one (20 wt% Ni), indicating no metal loss during synthesis. For all samples, the aluminium content was 1.8 wt%, added as a non-reducible element to ensure the required 20 wt% of Ni in the perovskite formulation.X-ray diffraction (XRD)
The XRD patterns of the calcined solids shown in Fig. 1a indicate significant differences in the crystalline phases as lanthanum is gradually partially substituted by cerium. Ce-Free sample (x = 0.0) exhibits sharp diffraction peaks indicative of a crystalline iso-structural rhombohedral LaNiO3 perovskite structure (33-0711).17 By replacing La with 6 wt% Ce (x = 0.1), LaNiO3 perovskite structure is maintained but segregated crystalline phases identified as fluorite CeO2 (34-0964) and NiO (44-1159) are also formed. For larger Ce loadings (x = 0.5; 0.7), the formation of a perovskite structure is no longer detected and only CeO2 and NiO isolated phases were detected. None of the diffraction peaks associated with lantana appeared in the XRD patterns, indicating that after calcination at 700 °C, this oxide phase may be highly dispersed and present as an amorphous form or incorporated into CeO2 as solid solution.7 This behavior is similar to that previously reported by Lima et al.18 and Liu et al.10 The limit of the Ce loading in the A site to form a perovskite type structure is indeed ≤0.5. For a larger Ce content, a CeO2–La2O3 solid solution with a fluorite structure was formed. The diffraction peaks associated with CeO2 fluorite structure indeed shift towards higher Bragg angles, which is indicative of lattice contraction (inset Fig. 1a), in line with the previous result for ceria doping with other lanthanides oxides.19,20 Considering that the ionic radii of La3+ (0.117 nm) is larger than Ce4+ (0.097 nm), the contraction of the lattice should be explained by a change to a higher oxidation state of the cations releasing electrons to maintain the compound neutral or the additional insertion of smaller cations such as Ni3+ (0.056 nm). The mean crystallite size calculated by the Debye–Scherrer equation, using the diffraction line at 2θ 32.8° for LaNiO3 and 27.9° for CeO2, indicates no large differences in the obtained dhkl values. The calculated mean particle size was 15 nm for LaNiO3 perovskites and 5.0 nm for the CeO2–La2O3 solid solution. | ||
| Fig. 1 X-ray diffraction patterns of: (a) calcined samples; (b) Ni-reduced catalysts. Inset figures: enlargement of Bragg angle region corresponding to the (111) plane of CeO2. | ||
Surface area
The surface areas shown in Table 1 display lower SBET values for the x = 0.0 and 0.1 perovskite type structures and larger values for those at which CeO2–La2O3 solid solutions are formed (x = 0.5; 0.7). Even though the SBET values are similar to other perovskite type oxides,21,22 differences in the amount of adsorbed, desorbed and the hysteresis cycle can be detected (Fig. S1†). According to IUPAC definition,23 the isotherms corresponds to H3 type, characteristic of solids with slit-shaped pores, also reported for Co perovskite synthesized by a self-combustion method.22 A decrease in the specific area after reduction treatment was observed, as shown in Table 1.Those lower values may be explained by a sintering process upon reduction because the surface areas are almost equal to the geometrical ones as they do not possess internal microporosity.
Temperature programmed reduction (TPR)
As the main objective of this study is the preparation of stable nickel nanoclusters as chemoselective catalysts for the hydrogenation of xylose to xylitol, it is of great importance to define the minimum reduction temperature to obtain reduced nickel nanoparticles without sintering. The complete reduction of non-substituted LaNiO3 perovskite structure has been reported in two well-defined reduction steps; Ni3+ to Ni2+ at around 350 °C followed by Ni2+ to Ni0 at about 470 °C.10,18,24 TPR profiles of calcined materials (Fig. 2) showed different reduction peaks, as it can be expected for complex systems. Taking into account that reduction of Ni3+ to Ni2+ in LaNiO3 occurs at 350 °C, in LaAl0.18Ni0.82O3 (x = 0.0) perovskite, the first reduction peak recorded at lower temperatures (250 °C) can be assigned to the partial reduction of the perovskite structure with formation of La2Ni2O5 (ref. 25) and NiAl2O4 (ref. 26) spinels, and the small reduction peak at 320 °C can be associated with the reduction of surface OH– groups and adsorbed oxygen.27 As for x = 0.1 solid, even though same reduction peaks are observed; they were shifted to lower temperatures. Furthermore, peaks related to the CeO2 phase reduction are also observed.17 Regarding the higher Ce content (x = 0.5; 0.7), the TPR profiles (Fig. 2) show only one asymmetrical and broad reduction peak, corresponding mainly to the reduction of NiO segregated on the CeO2–La2O3 solid solution.28 However, CeO2 bulk reduction could not be ruled out.7,29 From these results, a reduction temperature of 500 °C was selected for all solids, which corresponds to the temperature at which Ni reduction is starting for Ce-free perovskite (x = 0.0).This mild reduction condition leads to Ni reduction but without complete destruction of the perovskite structure, aiming at maximizing metal dispersion while avoiding Ni sintering.
Indeed, after the reduction treatment, XRD patterns of the Ni-reduced solids (Fig. 1b) do not show significant differences compared to the corresponding calcined samples.
A different behavior was reported for LaNiO3 and La0.9CeNiO3 perovskites elsewhere6,8,23,24 and a complete loss of the perovskite structure was evidenced under hydrogen at 500 °C forming Ni metal nanoparticles supported on a La2O3 matrix. Such an apparent discrepancy is indeed an important finding, which might indicate that the presence of only ∼2 wt% of aluminum may avoid the destruction of the perovskite structure. At larger Ce contents (x = 0.5; 0.7), the reductive atmosphere does not produce any structural difference in CeO2–La2O3 solid solution30 with no sensitive effect of the Al content.6
Ammonium temperature programmed desorption (NH3-TPD)
The measurement of acid strength by NH3-TPD assumes that the temperature of maximum desorption rate is a rough measure of the acid strength of the sorption sites. An acid strength distribution is thus usually proposed considering both NH3 physically adsorbed and hydrogen-bound at ≤150 °C (ref. 31) and acid site-bound NH3 at T > 150 °C. In particular, it is generally accepted to classify the acid sites as: (i) weak (T < 250 °C) and moderate to strong (T > 250 °C) acid sites.32NH3 desorption profiles shown in Fig. 3 indicate a wide distribution of acidic site strengths on the surface of Ni-reduced solids. The first desorbed NH3 peak at temperatures ≤150 °C observed for all solids is attributed to physically adsorbed as previously discussed.31 The second broad unresolved NH3 desorption peak recorded for both x = 0.0 and 0.1 Ni-reduced catalysts is similar to the supported alumina catalysts.33 Upon Ce substitution, it is observed that for a 6 wt% of Ce loading (x = 0.1) almost the same TPD pattern is observed compared to the Ce free catalyst. Accordingly, only one of the strong acidic site peaks at around 350 °C is flattened in the x = 0.1 catalyst. The total amount of desorbed NH3 was estimated with a confidence level of 94% by deconvoluting the peaks and the data is also summarized in Table 1.
There is no large difference in the total number of acid sites upon Ce substitution for x ≤ 0.5 catalysts. For the catalyst with a larger substitution degree a small decrease in total acidity is detected according to previous discussions, wherein a large presence of Ce covers the acid sites.
X-ray photoelectron spectroscopy (XPS)
The Ni 2p core-levels spectra for the Ni-reduced solids are shown in Fig. 4, (La 3d, Ce 3d and O 1s in Fig. S2†) and their respective binding energies (BE) are compiled in Table 2. The most intense peak of La 3d5/2 shows two components at 834.3 eV and 838.1 eV, both attributed to La3+.34,35| Catalyst | Binding energy (eV) | |||
|---|---|---|---|---|
| La 3d5/2 | Ce 3d5/2 | Ni 2p3/2 | O 1s | |
| x = 0.0 | 834.3 | — | 852.5 (42) | 529.3 (63) |
| 855.7 (58) | 531.6 (37) | |||
| x = 0.1 | 834.4 | 882.4 (79) | 852.5 (61) | 529.3 (64) |
| 885.4 (21) | 855.7 (39) | 531.6 (36) | ||
| x = 0.5 | 834.3 | 882.6 (82) | 852.4 (58) | 529.7 (62) |
| 885.2 (18) | 855.4 (42) | 531.4 (38) | ||
| x = 0.7 | 834.4 | 882.7 (17) | 852.6 (60) | 529.8 (66) |
| 885.6 (83) | 855.5 (40) | 531.5 (34) | ||
A doublet followed by a satellite peak was observed for Ni 2p3/2. The first peak of the doublet was attributed to Ni0, the largest peak at 852.5 ± 0.1 eV,36 and the second one and the satellite peak were assigned to oxidized (Ni2+ and Ni3+) nickel species. It was not possible to distinguish between those two oxidic species due to their close BE: 857.2 eV and 856.0 eV for Ni2+ and Ni3+,37 respectively. The Ce spectra (Fig. S2b†) displayed a single peak attributed to Ce4+, as well as multiplets and satellite peaks, previously reported for Ce3+.38 The core-level spectra of Ce4+ and Ce3+ at 882.3 (ref. 39) and 885.3 eV,40 respectively, allow differentiating the surface contributions. The lower Ce content sample (x = 0.1) shows the smallest Ce4+ peak, which is in line with previous discussion regarding the formation of a perovskite-type structure (Ce3+) with presence of La2O3–CeO2 (Ce4+) as segregated phase in a lower extent. At larger Ce content (x = 0.5; 0.7), the large increase in the core-level spectra and in the peak corresponding to the CeO2 phase (Ce4+) is also in agreement with previous characterization results.
With regard to the O 1s spectra, no surface core-level spectra higher than 532 eV indicate the total removal of molecular water from the surface of the samples.41 No differences in the BE of the surface hydroxyls/carbonate groups at 531.5 eV was observed (Table 2), and a change from 529.7 eV (x = 0.5; 0.7) to 529.3 eV (x = 0.0; 0.1) was detected for the lattice oxygen.
Accordingly, this BE of O 1s for both x = 0.0 and 0.1 reduced samples fits quite well with the corresponding values expected for a perovskite structure,42,43 which is in line with previously assigned crystalline phases as identified by XRD. Similarly, the upshift to 529.7 eV for x = 0.5 and 0.7 solids has been reported for oxygen species with cations in the lower oxidation state,44,45 which is in agreement with the shift of BE of the Ni 2p3/2 values.44,46
Table 3 displays the surface atomic composition and surface Ni content, calculated from the peaks at 852 eV for the Ni-reduced samples. Unfortunately, the loss of oxygen produced during the reduction treatment prevents a determination of the nominal ratios. The expected decrease in La/La + Ce and increase in the Ce/La + Ce upon Ce content were observed. With regard to Al/La + Ce and Ni/La + Ce ratio, the nominal values for the calcined precursors were 0.18 and 0.82, respectively; therefore, much lower values in the Ni/La + Ce ratio are observed. The decrease in surface Ni content upon Ce addition is in line with the XRD results regarding the contraction of the ceria crystal lattice by the insertion of Ni3+ (0.056 nm) or Ni2+ (0.069 nm) in CeO2 upon reduction treatment.47 An opposite trend observed with other reduced perovskite-type structures, where B-site cations are the ones that are surface enriched.48,49
The lowest value of the reduced Ni surface content (Table 3) for the Ce free Ni-reduced catalyst can be explained considering the higher thermal stability of this perovskite upon ∼2 wt% aluminium insertion (Fig. 3). It is proposed that the higher stability of the as-synthesized La1.0Al0.18Ni0.82O3 perovskite, as well as that of La2Ni2O5 (ref. 25) and NiAl2O4 (ref. 26) reported spinels, which would collapse when the reduction temperature reaches 600 °C, does not allow an increase in the concentration of surface reduced nickel at the reduction temperature of 500 °C used for both x = 0.0 and x = 0.1, obtaining thus a Ni surface extent of ∼30%. Regarding the CeO2–La2O3 solid solution precursors (x = 0.5; 0.7), the reported strong metal support interaction (SMSI) effect for Ni/CeO2 catalysts50 is likely to explain the reducibility extent of Ni of ∼36% to 40%.
Owing to the lower specific surface of the Ni-reduced solids, it can be extrapolated that a similar surface and bulk composition would be present.51 Therefore, considering the surface atomic composition (Table 2) and atomic ratios (Table 3), it can be possible to estimate the metallic Ni0 content. The atomic Ni0 g−1 of reduced sample displayed in Table 3 will be used to further calculate the TOF as an intrinsic measurement of the catalytic activity on the aqueous-phase hydrogenation of xylose (Table 4).
Catalytic activity
The experimental results of the total conversion and product distribution of xylose hydrogenation are depicted in Fig. 5a and 6, respectively. In addition, the successful fit of the experimental data with a pseudo first order reaction3,4 is displayed in Fig. 5b, allowing the global pseudo first order constant to be calculated. The obtained values as well as the initial reaction rate, TOF values and selectivity are shown in Table 4. | ||
| Fig. 5 Xylose conversion during hydrogenation reaction at 100 °C and pH2 = 25 bar. (a) Total conversion, (b) pseudo first order adjustment. | ||
The global constant and reaction rate increased with increasing Ce content up to x = 0.5 and a further decrease for the Ni-reduced catalyst was observed with a larger Ce loading (x = 0.7).
The selectivity to xylitol, on the other hand, did not show significant changes up to x = 0.5, while an increase was observed over the sample with the highest content (x = 0.7).
Regarding product distribution during the reaction time, the hydrogenation reaction is the preferred pathway with the large formation of xylitol along with ethylene glycol and glycerol. To assess the genesis of those lower carbon chain polyalcohols and determine if they are formed straight from xylose in a parallel reaction or from xylitol hydrogenolysis as a cascade reaction, blank experiments were carried out under the same experimental conditions, but feeding the reactor with xylitol or glycerol instead of xylose.
No conversion could be observed in both cases, indicating that under the experimental reaction conditions (reductive atmosphere and presence of metallic sites) xylose hydrogenation is the preferential reaction route and no consecutive reactions appear to occur. It is thus proposed that xylose undergoes retro-aldol condensation to produce both glycolaldehyde and glyceraldehyde, which can be hydrogenated rapidly to ethylene glycol and glycerol, respectively.
Such a side reaction pathway has indeed been previously claimed by some authors elsewhere.52–55 The production of these side reaction products is attributed to metallic sites promoted by basic sites.56–60
Note that Lewis acid sites can isomerise xylose to xylulose52 and in the presence of Brønsted acid sites, xylose and xylulose may produce furfural by dehydration followed by the aldehyde hydrogenation to furfuryl alcohol on metallic sites.53 Moreover, it has been also reported that highly dispersed metallic sites closer to acid sites are able to produce furfuryl alcohol in a single step.53 Based on the abovementioned considerations and the observed trends in the formation of xylulose and furfuryl alcohol in the experiments reported in this study (Fig. 6), xylulose reaches a maximum concentration and then decreases consistently when furfuryl alcohol is formed – a simplified reaction pathway is proposed, as shown in Scheme 1.
According to our reaction pathway scheme, k1, k2, k3 and k4 correspond to parallel reactions steps from xylose, while k5 represents a consecutive reaction from xylulose to furfuryl alcohol. The specific pseudo first order reaction constants for each step were calculated and are collected in Table 5. The higher values of k1, k3 and k4, all corresponding to hydrogenation reactions, confirm the hydrogenation character of these catalysts. This finding is also supported by k5 because only k2 corresponds to a reaction (isomerisation) rather than hydrogenation. To further corroborate k2 dependence with acidity, Fig. 7 shows both total acidity and k2 values. It can be observed that the high correlation between these curves confirm k2 dependence upon acidity.
| Catalyst | k1 (min−1 g−1) | k2 (min−1 g−1) | k3 (min−1 g−1) | k4 (min−1 g−1) | k5 (min−1 g−1) |
|---|---|---|---|---|---|
| x = 0.0 | 0.026 | 0.016 | 0.013 | 0.013 | 0.257 |
| x = 0.1 | 0.047 | 0.033 | 0.029 | 0.029 | 0.394 |
| x = 0.5 | 0.054 | 0.029 | 0.032 | 0.032 | 0.413 |
| x = 0.7 | 0.044 | 0.014 | 0.034 | 0.034 | 0.441 |
The turnover frequency (TOF) calculated from the surface Ni atoms as determined by XPS (Table 4) is in good agreement with some reports in the literature for Pt supported catalysts.61 Little differences in the TOF values for all the studied catalysts shows that intrinsic activity in the catalyst series is the same, without the influence of neither composition nor structural differences of the catalysts. The product selectivity did not change with reaction time as revealed by the experimental data collected at 30% (Table 4) and 98% (Fig. 8) isoconversion levels. Nevertheless, the selectivity to xylitol increases on the x = 0.7 sample. This behaviour may be associated with the lower acidity of this catalyst (Table 1), which was shown to determine the reaction constant of the isomerisation reaction (Fig. 7).
 | ||
| Fig. 8 Selectivity to the detected products at 30% of isoconversion during xylose hydrogenation. T = 100 °C, pH2 = 25 bar. | ||
Indeed the k1/k2 ratio is higher for x = 0.7 catalyst (k1/k2 = 3.14) than for all the other samples with lower amounts of Ce (k1/k2 ranges from 1.63 to 1.86), indicating that straight hydrogenation to xylitol is preferred on this catalyst. To address one of the major drawbacks concerning the use of a Ni-based catalyst in aqueous-phase hydrogenation, the studied catalysts were recovered by filtration after each batch reaction and analyzed in regard to its chemical composition, particularly the Ni loading, and crystalline structure. The AAS results indicate a different behaviour according to the catalyst original structure (Table 1). It can be observed that the Ni content remains unchanged when the catalyst comes from a precursor holding a perovskite-type structure (x = 0.0 and 0.1). On the other hand, metal leaching is evidenced for the catalysts obtained from a mixture of oxide phases (x = 0.5 and 0.7). A metal loss of 15% was consistently observed for both samples, irrespective of their Ce content. This finding is quite promising because it points to a simple bottom-up approach that leaching-resistant catalysts can be obtained through a perovskite structure.
The XRD patterns of the spent catalysts are displayed in Fig. 9. The catalyst obtained from a bare perovskite structure (x = 0.0) undergoes some structural modification under the xylose aqueous-phase hydrogenation conditions because new diffraction lines are now registered. Such peaks are associated with a crystalline La2O3 (22-369) isolated phase. This may have arisen from a previously highly dispersed and/or amorphous lanthanum phase not detected on the fresh sample. The presence of the perovskite structure, as evidenced by their corresponding diffraction peaks, suggests that such a structure is stable, which is consistent with the higher metal leaching-resistance observed for this sample, as already discussed. As for the other systems (x = 0.1; 0.5; 0.7), no significant change was detected, revealing the stability of the single oxides used in the formulation, as expected.
 | ||
| Fig. 9 Post-reaction X-ray diffraction patterns. Inset figure: enlargement of region within 26–30° Bragg angle. | ||
Conclusions
La1−xCexAl0.18Ni0.82O3 (x = 0.0, 0.1, 0.5, 0.7) systems prepared by a self-combustion method were used successfully as precursors to prepare Ni nanoclusters. A perovskite structure was obtained for Ce-free and lower Ce content samples (x = 0.0; 0.1), whereas at higher Ce contents (x = 0.5; 0.7), a mixture of NiO and CeO2–La2O3 solid solution were the main structures. All catalysts were active in the aqueous-phase hydrogenation of xylose, leading mostly to xylitol and other lower carbon polyalcohols (glycerol and ethylene glycol). The highest selectivity toward xylitol was obtained on the CeO2–La2O3 solid solution-supported catalyst (x = 0.7) as a consequence of its lower acidity and higher activity to hydrogenation. The lower stability of this catalyst, measured as a loss of Ni by leaching, spoils its use. The formation of the perovskite structure rendered remarkable leaching-resistant catalysts because no Ni loss was observed at all. Moreover, these catalysts presented only a slightly lower selectivity towards xylitol production, highlighting its potential applications. As the well-known Ni leaching was clear for those catalysts prepared from ordinary metal oxide phases, the promising use of perovskites as catalyst precursors for the aqueous-phase processing of biomass-derived compounds is corroborated.Acknowledgements
The authors acknowledge the support from CONICYT-Fondecyt Grant 1130005 and fellowship 21130572 (Chile) and CNPq and FAPERJ (Brazil).Notes and references
- T. Granström, K. Izumori and M. Leisola, Appl. Microbiol. Biotechnol., 2007, 74, 273 CrossRef PubMed.
- J.-P. Mikkola, R. Sjöholm, T. Salmi and P. Mäki-Arvela, Catal. Today, 1999, 48, 73 CrossRef CAS.
- J. Wisniak, M. Hershkowitz, R. Leibowitz and S. Stein, Prod. R&D, 1974, 13, 75 CAS.
- J. Wisniak, M. Hershkowitz and S. Stein, Prod. R&D, 1974, 13, 232 CAS.
- M. Yadav, D. K. Mishra and J.-S. Hwang, Appl. Catal., A, 2012, 425–426, 110 CrossRef CAS.
- N. Escalona, W. Aranzaez, K. Leiva, N. Martinez and G. Pecchi, Appl. Catal., A, 2014, 481, 1 CrossRef CAS.
- C. A. Franchini, W. Aranzaez, A. M. Duarte de Farias, G. Pecchi and M. A. Fraga, Appl. Catal., B, 2014, 147, 193 CrossRef CAS.
- G. Valderrama, A. Kiennemann and M. R. Goldwasser, J. Power Sources, 2010, 195, 1765 CrossRef CAS.
- S. M. de Lima, A. M. da Silva, L. O. O. da Costa, J. M. Assaf, L. V. Mattos, R. Sarkari, A. Venugopal and F. B. Noronha, Appl. Catal., B, 2012, 121–122, 1 CrossRef CAS.
- F. Liu, L. Zhao, H. Wang, X. Bai and Y. Liu, Int. J. Hydrogen Energy, 2014, 39, 10454 CrossRef CAS.
- R. Hu, Y. Bai, H. Du, H. Zhang, Y. Du, J. Zhang and Q. Zhou, J. Rare Earths, 2015, 33, 1284 CrossRef CAS.
- W. Shan, J. Yang, L. Yang and N. Ma, J. Nat. Gas Chem., 2011, 20, 384 CrossRef CAS.
- T. Maneerung, K. Hidajat and S. Kawi, Catal. Today, 2011, 171, 24 CrossRef CAS.
- N. Ichikawa, S. Sato, R. Takahashi and T. Sodesawa, J. Mol. Catal. A: Chem., 2005, 231, 181 CrossRef CAS.
- B. C. Campo, S. Ivanova, C. Gigola, C. Petit and M. A. Volpe, Catal. Today, 2008, 133, 661 CrossRef.
- L. A. Chick, L. R. Pederson, G. D. Maupin, J. L. Bates, L. E. Thomas and G. J. Exarhos, Mater. Lett., 1990, 10, 6 CrossRef CAS.
- Y.-J. Su, K.-L. Pan and M.-B. Chang, Int. J. Hydrogen Energy, 2014, 39, 4917 CrossRef CAS.
- S. M. Lima, J. M. Assaf, M. A. Peña and J. L. G. Fierro, Appl. Catal., A, 2006, 311, 94 CrossRef CAS.
- G. Sierra Gallego, J. Barrault, C. Batiot-Dupeyrat and F. Mondragón, Catal. Today, 2010, 149, 365 CrossRef CAS.
- A. M. Duarte de Farias, D. Nguyen-Thanh and M. A. Fraga, Appl. Catal., B, 2010, 93, 250 CrossRef CAS.
- N. Rezlescu, E. Rezlescu, P. D. Popa, C. Doroftei and M. Ignat, Composites, Part B, 2014, 60, 515 CrossRef CAS.
- J. A. Villoria, M. C. Alvarez-Galvan, S. M. Al-Zahrani, P. Palmisano, S. Specchia, V. Specchia, J. L. G. Fierro and R. M. Navarro, Appl. Catal., B, 2011, 105, 276 CrossRef CAS.
- K. S. W. Sing, Pure Appl. Chem., 1982, 54, 2201 CrossRef.
- J. Choisnet, N. Abadzhieva, P. Stefanov, D. Klissurski, J. M. Bassat, V. Rives and L. Minchev, J. Chem. Soc., Faraday Trans., 1994, 90, 1987 RSC.
- M. E. Rivas, C. E. Hori, J. L. G. Fierro, M. R. Goldwasser and A. Griboval-Constant, J. Power Sources, 2008, 184, 265 CrossRef CAS.
- C. H. Campos, P. Osorio-Vargas, N. Flores-Gonzalez, J. L. G. Fierro and P. Reyes, Catal. Lett., 2016, 146, 433 CrossRef CAS.
- L. G. Tejuca, J. L. G. Fierro and J. M. D. Tascón, in Advances in Catalysis, ed. H. P. D. D. Eley and B. W. Paul, Academic Press, 1989, vol. 36, p. 237 Search PubMed.
- P. Osorio-Vargas, C. H. Campos, R. M. Navarro, J. L. G. Fierro and P. Reyes, J. Mol. Catal. A: Chem., 2015, 407, 169 CrossRef CAS.
- H. Zhu, W. Wang, R. Ran and Z. Shao, Int. J. Hydrogen Energy, 2013, 38, 3741 CrossRef CAS.
- X. Han, Y. Yu, H. He and W. Shan, Int. J. Hydrogen Energy, 2013, 38, 10293 CrossRef CAS.
- D. Liu, P. Yuan, H. Liu, J. Cai, D. Tan, H. He, J. Zhu and T. Chen, Appl. Clay Sci., 2013, 80–81, 407 CrossRef CAS.
- J. A. Cecilia, A. Infantes-Molina, E. Rodriguez-Castellon, A. Jimenez-Lopez and S. T. Oyama, Appl. Catal., B, 2013, 136, 140 CrossRef.
- K. Jiratova, A. Spojakina, L. Kaluza, R. Palcheva, J. Balabanova and G. Tyuliev, Chin. J. Catal., 2016, 37, 258 CrossRef CAS.
- B. Kucharczyk and W. Tylus, Catal. Today, 2004, 90, 121 CrossRef CAS.
- B. Kucharczyk and W. Tylus, Appl. Catal., A, 2008, 335, 28 CrossRef CAS.
- J. C. Klein and D. M. Hercules, J. Catal., 1983, 82, 424 CrossRef CAS.
- S. M. d. Lima, M. A. Peña, J. L. G. Fierro and J. M. Assaf, in Studies in Surface Science and Catalysis, ed. M. S. Fábio Bellot Noronha and S.-A. Eduardo Falabella, Elsevier, 2007, vol. 167, p. 481 Search PubMed.
- P. S. Bagus, C. J. Nelin, Y. Al-Salik, E. S. Ilton and H. Idriss, Surf. Sci., 2016, 643, 142 CrossRef CAS.
- E. Paparazzo, G. M. Ingo and N. Zacchetti, J. Vac. Sci. Technol., A, 1991, 9, 1416 CAS.
- Y. Ohno, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 48, 5515 CrossRef CAS.
- L. G. Tejuca, J. L. G. Fierro and J. M. D. Tascon, Adv. Catal., 1989, 36, 237 CAS.
- L. G. Tejuca and J. L. G. Fierro, Thermochim. Acta, 1989, 147, 361 CrossRef CAS.
- R. P. Vasquez, Surf. Sci. Spectra, 1992, 1, 361 CrossRef CAS.
- N. S. McIntyre and M. G. Cook, Anal. Chem., 1975, 47, 2208 CrossRef CAS.
- G. C. Allen and K. R. Hallam, Appl. Surf. Sci., 1996, 93, 25 CrossRef CAS.
- V. A. M. Brabers, F. M. van Setten and P. S. A. Knapen, J. Solid State Chem., 1983, 49, 93 CrossRef CAS.
- R. Pereñíguez, V. M. González-DelaCruz, J. P. Holgado and A. Caballero, Appl. Catal., B, 2010, 93, 346 CrossRef.
- G. Pecchi, C. M. Campos, M. G. Jiliberto, E. J. Delgado and J. L. G. Fierro, Appl. Catal., A, 2009, 371, 78 CrossRef CAS.
- G. Pecchi, P. Reyes, R. Zamora, L. E. Cadus and J. L. G. Fierro, J. Solid State Chem., 2008, 181, 905 CrossRef CAS.
- A. Caballero, J. P. Holgado, V. M. Gonzalez-delaCruz, S. E. Habas, T. Herranz and M. Salmeron, Chem. Commun., 2010, 46, 1097 RSC.
- R. Dinamarca, C. Sepúlveda, E. J. Delgado, O. Peña, J. L. G. Fierro and G. Pecchi, J. Catal., 2016, 338, 47 CrossRef CAS.
- V. Choudhary, S. Caratzoulas and D. G. Vlachos, Carbohydr. Res., 2013, 368, 89 CrossRef CAS PubMed.
- R. F. Perez and M. A. Fraga, Green Chem., 2014, 16, 3942 RSC.
- J.-P. Mikkola, T. Salmi and R. Sjöholm, J. Chem. Technol. Biotechnol., 1999, 74, 655 CrossRef CAS.
- D. K. Mishra, A. A. Dabbawala and J.-S. Hwang, J. Mol. Catal. A: Chem., 2013, 376, 63 CrossRef CAS.
- J. Lee, Y. Xu and G. W. Huber, Appl. Catal., B, 2013, 140–141, 98 CrossRef CAS.
- N. Li and G. W. Huber, J. Catal., 2010, 270, 48 CrossRef CAS.
- J. Sun and H. Liu, Green Chem., 2011, 13, 135 RSC.
- J. Sun and H. Liu, Catal. Today, 2014, 234, 75 CrossRef CAS.
- K. Wang, M. C. Hawley and T. D. Furney, Ind. Eng. Chem. Res., 1995, 34, 3766 CrossRef CAS.
- A. Tathod, T. Kane, E. S. Sanil and P. L. Dhepe, J. Mol. Catal. A: Chem., 2014, 388–389, 90 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra13395a |
| This journal is © The Royal Society of Chemistry 2016 |