Synthesis, phase behaviour and photo-optical properties of bent-core methacrylate with azobenzene group and corresponding side-chain polymethacrylate
V. Novotná*a,
A. Bobrovskyb,
V. Shibaevb,
D. Pociechac,
M. Kašpara and
V. Hamplováa
aInstitute of Physics, The Czech Academy of Sciences, Na Slovance 2, 182 21 Prague 8, Czech Republic. E-mail: novotna@fzu.cz
bFaculty of Chemistry, Moscow State University, Leninskie gory, Moscow, 119992 Russia. E-mail: bbrvsky@yahoo.com
cLaboratory of Dielectrics and Magnetics, Chemistry Department, University of Warsaw, ul. Żwirki i Wigury 101, 02-089 Warsaw, Poland
First published on 30th June 2016
Abstract
In the present work, synthesis of a new photosensitive bent-core monomer and corresponding side-chain polymethacrylate is described. The liquid crystalline (LC) properties were studied by texture observation, calorimetric and electro-optical measurements. Structural parameters of the LC phases were established from X-ray diffraction. Due to presence of the azobenzene group both compounds revealed photosensitivity and UV-irradiation effectively induces the E–Z isomerization process. The photo-optical properties of the spin-coated films of polymethacrylate are analysed and it is found that irradiation with polarized light aligns chromophores in the direction perpendicular to the polarization plane of incident light. Photoorientation proceeds more effectively in the amorphousized films in comparison with annealed LC films.
1. Introduction
Liquid crystalline (LC) materials are an example of self-assembling media on a nanoscale level. Composed of anisotropic molecules, they can exhibit a variety of crystalline and mesomorphic phases including an appearance of polar order. The latter can stimulate ferroelectric or antiferroelectric behaviour. In 1996 new polar phases have been discovered in materials composed of bent-core molecules.1 The packing of bent-core molecules in tilted smectic layers creates structural layer chirality even though the molecules are non-chiral ones. Numerous LC phases formed by such mesogens have been described in last 20 years and their structural properties investigated.2,3 The most frequently observed are polar tilted smectics, SmCP. Four structures of the SmCP phases have been distinguished with a synclinic (S) or anticlinic (A) molecular tilt and the ferroelectric (F) or antiferroelectric (A) polar correlation between neighbouring layers.Recently, we have designed and studied bent-core compounds with methoxy group as a lateral substitution on the phenyl attached to central core of the bent-shaped molecules.4,5 The selected compounds exhibit very low coercive field for switching the antiferroelectric to the ferroelectric state and only one peak in switching current in the SmCP phase.5 Additionally, we have detected substantial decrease of the phase transition temperatures in comparison with non-substituted compounds, which leads to the utilization of the methoxy derivatives for preparation of bent-core monomers5 and polymers.6 Besides, the photosensitive bent-core mesogens were prepared and studied7–9 having azo-group as a linkage in the mesogenic core. Controlling of liquid crystalline self-assembling process by means of isomerization of photochromic molecules is relatively new area of research. Photo-isomerization can change the molecule symmetry, which causes modification of the liquid crystalline structure and mesomorphic properties.10,11
Liquid crystalline (LC) side-chain polymers are the unique type of smart responsive materials, which combine the typical features of polymers (e.g. elasticity and mechanical stability) with properties of low-molecular-mass liquid crystals, e.g. switching under applied external field accompanied by optical effects.12,13 Introduction of azobenzene fragments into LC polymer allows one to obtain photoresponsive materials capable to change their supramolecular structure, mesophase type, optical and mechanical properties under the action of polarized or nonpolarized light. Azobenzene fragments undergo E–Z isomerization process accompanied with significant decrease of their anisometry and changing of the dipole moment. These processes are successfully used for the creation of materials with photooptically switchable structure and challenging optical properties.13–27
LC side-chain polymers with bent-core mesogens attract a special attention because they possess a number of unique properties including formation of stable films and macroscopic polar order with variety of useful properties, such as piezo and pyroelectricity.28–31 Macromolecular nature of these systems enables to realize stabilization of the polar order in rigid glassy state. Despite the strong interest in this research field up to now there are only few papers devoted to photosensitive bent-core LC polymers.32,33 Moreover, photoorientation processes in films of these polymers under polarized light action have not been studied yet.
Herein, we report synthesis and mesomorphic behaviour of a new bent-core monomethacrylate and corresponding side-chain polymethacrylate. Photosensitive azobenzene groups were introduced into the internal arms of the bent-core molecules, which are attached to the polymer main chain. Photochromic polymer was prepared from monomethacrylate by a radical polymerization. The main goal of the present paper is the comparative study of mesomorphic properties of both compounds, i.e. bent-core monomer and its polymer derivative. A special attention is paid to the investigation of the photooptical properties of the polymer films under the action of polarized and non-polarized light.
2. Synthesis
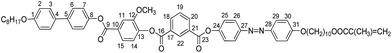
General chemical formulas of presented compounds are shown in Scheme 1. The first step for preparation of new functional polymer is designing and studying of LC monomer. Synthesis of monomer 4-(((4′-(decyloxy)-[1,1′-biphenyl]-4-yl)oxy)carbonyl)-2-methoxyphenyl (4-((4-((5-(methacryloyloxy)pentyl)oxy)phenyl)diazenyl)phenyl)isophthalate, denoted here as M and its corresponding polymethacrylate PM are schematically depicted in Scheme 1 of monomer M and polymethacrylate PM.2.1. Synthesis of intermediates 1 and 2
4-ω-Bromodecyloxy-4′-hydroxyazobenzene 1 was obtained from 4-acetamidophenol by alkylation with 1,10-dibromodecane in dioxan–water solution in the presence of NaOH. The crude product was hydrolyse with dilute H2SO4 (20%) by boiling for 3 hours. After standing at the room temperature for 12 h, the solid sulphuric salt of amine 1 was isolated by filtration, dried and washed twice with n-hexane followed by diazotation and coupling with phenol using standard procedure.1H-NMR (CDCl3, 300 MHz): δ = 7.95 (dd, 4H, ortho to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 6.96 and 7.02 (2d, 2 × 2H, meta to –N
N–), 6.96 and 7.02 (2d, 2 × 2H, meta to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 4.02 (t, 2H, CH2OAr), 3.40 (t, 2H, CH2Br), 1.9 m (4H, CH2), 1.2–1.5 (m, 12H, CH2) ppm (Scheme 2).
N–), 4.02 (t, 2H, CH2OAr), 3.40 (t, 2H, CH2Br), 1.9 m (4H, CH2), 1.2–1.5 (m, 12H, CH2) ppm (Scheme 2).
The bromo derivative 1 was converted into iodide 2 (4-ω-iododecyloxy-4′-hydroxyazobenzene) by boiling with excess sodium iodide in acetone for several hours.
1H-NMR (CDCl3, 300 MHz): δ = 7.95 (dd, 4H, ortho to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 6.96 and 7.02 (2d, 2 × 2H, meta to –N
N–), 6.96 and 7.02 (2d, 2 × 2H, meta to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 4.02 (t, 2H, CH2OAr), 3.20 (t, 2H, CH2I), 1.9 m (4H, CH2), 1.2–1.5 (m, 12H, CH2) ppm.
N–), 4.02 (t, 2H, CH2OAr), 3.20 (t, 2H, CH2I), 1.9 m (4H, CH2), 1.2–1.5 (m, 12H, CH2) ppm.
2.2. Synthesis of methacrylate 3
Iodide 2 (0.1 mol) was dissolved in 150 mL of dry dimethyl sulfoxide and 0.2 mol dry potassium methacrylate (dried 24 h under vacuum at 30 °C) and 0.1 g hydroquinone was added. After reacting at room temperature for five days the solution was poured into 1 dm3 water and crystallized from ethanol. The red product was dried in vacuum at room temperature for two days. 1H-NMR (DMSO-d6, 300 MHz): δ = 7.80 (m, 4H, ortho to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–), 7.02 (d, 2H, ortho to OR), 6.90 (d, 2H, ortho to OH), 6.10 and 5.53 (s, 2H, CH2
N–), 7.02 (d, 2H, ortho to OR), 6.90 (d, 2H, ortho to OH), 6.10 and 5.53 (s, 2H, CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) ), 4.08 (m, 4H, CH2O), 1.96 (s, 3H, CH3C
), 4.08 (m, 4H, CH2O), 1.96 (s, 3H, CH3C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) ), 1.2–1.8 (m, 16H, CH2) ppm.
), 1.2–1.8 (m, 16H, CH2) ppm.
Synthesis of mesogenic phenol 4 and compound 5 has been published recently.4,5
2.3. Synthesis of final monomer M
The final product 6 was obtained by condensation of acid 5 and phenol 3 with dicyclohexylcarbodiimide (DCC) as a condensation agent and dimethylaminopyridine (DMAP) as a catalyst in dichloromethane solution at room temperature. The crude product was crystallized from ethanol–tetrahydrofurane mixture and purified by column chromatography on silica gel (in dark) using a mixture of dichloromethane and acetone (99.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.2) as an eluent and crystallized twice from ethanol. The purity was checked by HPLC, which was carried out using a silica gel column (Biospher Si 5 μm, 4 × 250, Watrex) with a mixture of 99.8% of toluene and 0.2% of methanol as the eluent and detection of the eluting products by a UV-VIS detector (λ = 290 nm). Structure of the final product was confirmed by NMR and elemental analysis.
0.2) as an eluent and crystallized twice from ethanol. The purity was checked by HPLC, which was carried out using a silica gel column (Biospher Si 5 μm, 4 × 250, Watrex) with a mixture of 99.8% of toluene and 0.2% of methanol as the eluent and detection of the eluting products by a UV-VIS detector (λ = 290 nm). Structure of the final product was confirmed by NMR and elemental analysis.
1H-NMR (CDCl3, 300 MHz): 9.08 s (1H, isophtal ortho to –COO); 8.50 d (2H, isophtal. ortho to –COO); 7.90–8.00 m (6H, ortho to –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and ortho and para to –OCH3); 7.73 t (1H, isophtal meta to –COO); 7.50–7.60 dd (4H, ortho to –Ar); 7.40 m (5H, ortho to –OCO); 7.00 m (4H, ortho to –OCH2); 6.10 (s, 1H, trans-CH2
N– and ortho and para to –OCH3); 7.73 t (1H, isophtal meta to –COO); 7.50–7.60 dd (4H, ortho to –Ar); 7.40 m (5H, ortho to –OCO); 7.00 m (4H, ortho to –OCH2); 6.10 (s, 1H, trans-CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) ); 5.56 (s, 1H, cis-CH2
); 5.56 (s, 1H, cis-CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) ); 4.13 (t, J = 7.3, 2H, COOCH2); 4.05 m (4H, CH2OAr); 3.96 s (3H, OCH3); 1.96 (3H, m,
); 4.13 (t, J = 7.3, 2H, COOCH2); 4.05 m (4H, CH2OAr); 3.96 s (3H, OCH3); 1.96 (3H, m, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CCH3); 1.13–1.92 m (28H, CH2); 0.92 t (3H, CH3).
CCH3); 1.13–1.92 m (28H, CH2); 0.92 t (3H, CH3).
13C-NMR (CDCl3, 75 MHz, see figure below for numbering): 167.54 (CCOO); 164.56 (C-16); 163.97 (C-23); 163.24 (C-9); 161.68 (C-31); 158.79 (C-1); 152.07 (C-24); 151.33 (C-12); 150.58 (C-27); 149.71 (C-8); 146.76 (C-28); 144.12 (C-21); 138.85 (C-13); 136.52 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2); 135.30 (C-18); 135.16 (C-20); 132.60 (C-4); 132.02 (C-22); 130.10 (C-21); 129.71 (C-17); 129.20 (C-19); 128.49 (C-10); 128.09 (C-3); 127.76 (C-6); 125.18 (CH2
CH2); 135.30 (C-18); 135.16 (C-20); 132.60 (C-4); 132.02 (C-22); 130.10 (C-21); 129.71 (C-17); 129.20 (C-19); 128.49 (C-10); 128.09 (C-3); 127.76 (C-6); 125.18 (CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C); 124.82 (C-26); 123.82 (C-29); 123.39 (C-14); 123.05 (C-15); 122.20 (C-25); 121.85 (C-7); 114.78 (C-2); 114.68 (C-30); 113.96 (C-11); 68.08 (CH2O); 68.05 (CH2O); 64.81 (CCOOCH2); 56.21 (CH3O); 31.81 (CH2CH2CH3); 29.03–29.73 (6 × CH2); 28.55 (CH2CH2OOC
C); 124.82 (C-26); 123.82 (C-29); 123.39 (C-14); 123.05 (C-15); 122.20 (C-25); 121.85 (C-7); 114.78 (C-2); 114.68 (C-30); 113.96 (C-11); 68.08 (CH2O); 68.05 (CH2O); 64.81 (CCOOCH2); 56.21 (CH3O); 31.81 (CH2CH2CH3); 29.03–29.73 (6 × CH2); 28.55 (CH2CH2OOC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) ); 25.41–25.99 (m, 3 × CH2(CH2)2O); 22.66 (CH2CH3); 18.36 (CCH3); 14.11 (CH2CH3).
); 25.41–25.99 (m, 3 × CH2(CH2)2O); 22.66 (CH2CH3); 18.36 (CCH3); 14.11 (CH2CH3).
Elemental analysis for: C62H68N2O11 calc. C 73.21, H 6.74, found C 71.66, H 5.88%.
2.4. Synthesis of polymethacrylate PM
Photochromic polymer was prepared by a radical polymerization of corresponding acrylic monomer in benzene solution in the presence of 2 wt% (with respect to monomer) of AIBN. 119.5 mg of the monomer and 2 mg of AIBN were placed into ampoule and 1 mL of benzene was added. Then solution was poured by argon for 20 min and ampoule was sealed. After 3 days storage at 65 °C the solvent was evaporated and solid product was washed several times by boiling ethanol. Yield of polymerization was 56.6 mg (47.4%). Such relatively low yield is explained by competing radical transfer reaction promoted by azobenzene fragment. During polymer synthesis a lot of low-molar-mass products (dimers, short oligomers) are formed which were removed by ethanol. Molecular masses (Mw) and polydispersity of polymers (Mw/Mn) as determined by GPC chromatography using instrument Knauer: Mw = 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600, Mw/Mn = 1.96.
600, Mw/Mn = 1.96.
3. Experimental
Phase transition temperatures and enthalpies were determined by DSC studies (Pyris Diamond Perkin-Elmer 7) under cooling and heating runs at a rate of 10 K min−1. The samples of 2–5 mg were hermetically sealed in aluminium pans and placed in the calorimeter chamber inflated by nitrogen. The calorimeter data were calibrated on extrapolated onset temperatures and enthalpy changes of water, indium and zinc. The samples for texture observation and electro-optical studies were filled by capillarity action in the isotropic phase into cells composed of glasses with ITO transparent electrodes. The cell gap was defined by Mylar sheets with thickness 5 or 12 μm. All texture observations were performed using Nikon Eclipse polarizing microscope equipped with a Linkam hot stage.X-ray diffraction experiments were performed with Bruker D8 GADDS (parallel beam of CuKα radiation formed by Goebel mirror monochromator and point collimator, Vantec-2000 area detector, modified Linkam heating stage) and Bruker NanoStar (parallel beam of CuKα radiation formed by cross-coupled Goebel mirrors and three-pinhole collimator, Vantec-2000 area detector, MRI heating stage) systems. Samples were prepared as droplets or thin films on heated surface.
Thin films (100–200 nm) of the polymer for photo-optical experiments were obtained by spin-coating technique using solutions of different concentration in chlorophorm. In order to completely remove any traces of chlorophorm the spin-coated films were kept at room temperature during one day.
Photochemical investigations were performed using an optical set up equipped with a DRSh-350 ultra-high pressure mercury lamp and MBL-N-457 diode laser (457 nm, CNI Laser). To prevent the heating of the samples due to the IR irradiation of the mercury lamp, a water filter was introduced in the optical scheme. To assure the plane-parallel light beam, a quartz lens was applied. Using the filters a light with the wavelengths 365 nm and 436 nm were selected. The intensity of light was measured by LaserMate-Q (Coherent) intensity meter and were equal to ∼4.0 mW cm−2 (365 nm), ∼2.0 mW cm−2 (436 nm) for lamp and ∼0.3 W cm−2 for laser (457 nm).
Spectral measurements were performed using Unicam UV-500 UV-Vis spectrophotometer. The linearly polarized spectra of the film samples were studied with a TIDAS spectrometer (J&M) equipped with rotating polarizer (Glan-Taylor prism controlled by computer program). The dichroism values, D, of the polymer films were calculated from the spectra using the following eqn (1):
| D = (A║ − A⊥)/(A║ + A⊥) | (1) |
4. Results and discussion
4.1. Mesomorphic properties
Both studied compounds, monomethacrylate M and its corresponding side-chain polymethacrylate PM, display one LC mesophase which is observed below the isotropic phase. The phase transition temperatures and associated enthalpy changes obtained by DSC method are shown in Table 1 for both compounds. DSC curves taken at the second heating and the subsequent cooling runs are shown in Fig. 1a and b for M and PM, respectively. For monomer M there are two crystalline phases below liquid crystalline phase on cooling, designated as Cr1 and Cr2, respectively on cooling. On the basis of the texture observation in a polarising microscope, the LC mesophase was identified as tilted smectic phase (SmCP). The phase identification will be confirmed by X-ray diffraction studies later. Clearing temperature of the PM is ca. 60 K higher than monomer, i.e. linking of mesogenic groups to polymer backbone strongly increases mesophase thermostability.| T1/°C [ΔH/J g−1] | T2/°C [ΔH/J g−1] | T3/°C [ΔH/J g−1] | T4/°C [ΔH/J g−1] | ||||
|---|---|---|---|---|---|---|---|
| M | 88 [+1.9]→ | Cr2 | 95 [+1.2]→ | Cr1 | 141 [+18.1]→ | SmCP | 153 [+15.5]→ |
| 91 [−2.2]← | 96 [−1.1]← | 143 [−21.6]← | 153 [−15.8]← | ||||
| PM | 172 [+5.7]→ | SmCP | 212 [+8.5]→ | ||||
| 170 [−6.1]← | 214 [−8.4]← | ||||||
For monomethacrylate M fan-shaped texture is observed (Fig. 2a) on cooling from the isotropic phase, typical for smectic mesophases. The extinction positions of fans are parallel to the smectic layer normal. From the observation in the microscope we can infer that the texture corresponds to anticlinic antiferroelectric structure. Under an electric field 15 V μm−1 the irregular stripe appears (Fig. 2b), being identified as domains with opposite chirality and the texture under field corresponds to synclinic ferroelectric structure. Such textural behaviour under the electric field can be described as the transformation from the SmCAPA to the SmCSPF structure.34 Polymethacrylate PM showed tiny non-characteristic textures (even after several hours of annealing at 200 °C, see Fig. 2c) and no response to the electric field was detected up to the maximum of applied voltage 40 V μm−1. The polymer backbone disrupts probably polar ordering of mesogenic groups and prevents their alignment in the electric field. Nevertheless, for one-free-surface samples (spread droplet on the glass) a schlieren texture was observed, so one can conclude that PM forms a tilted smectic phase (SmCP phase).
4.2. Structural studies
Formation of the tilted smectic phase for both studied materials, M and PM, has been confirmed by X-ray diffraction measurements. For both compounds, X-ray patterns of mesophases exhibited series of commensurate sharp peaks in small diffraction angle range and a diffuse scattering maximum in the wide angle range (Fig. 3a and b for M and PM, respectively). Such patterns evidence lamellar structure with liquid-like molecular packing within the layers, which is inherent to SmA or SmC phase. The smectic layer thickness, d, determined from the positions of the low angle signals, was found almost temperature independent within the mesophase range for both, M (Fig. 4a) and PM (Fig. 4b). The d value obtained for monomer M, about 47.5 Å, is considerably lower than the approximate length of the fully stretched molecule, l ≈ 53 Å (calculated for isolated molecule of monomer M using commercial program Chem3D) indicating tilting of molecules with respect to layer normal by an angle about 26 degrees. The d value of polymer PM being about 96 Å is roughly two times larger than interlayer distance of monomer M. Apparently, PM material exhibit a bilayer-type smectic phase, in which a repeating unit (smectic layer) is formed from two mesogenic molecules – proposed structure is schematically drawn in Fig. 5. X-ray diffraction studies showed that both, monomer M and polymer PM, undergo crystallization on cooling, however the process for polymer is not so sharp comparing to monomer. For crystalline PM, the narrow peaks at high diffraction angle range coexist with a broad diffuse background indicating coexistence of LC and crystalline phases. It means that for polymer PM at low temperatures a crystalline ordering exists only locally. | ||
| Fig. 4 The temperature dependences of the layer spacing value, d, calculated from the position of the scattering peaks for (a) monomer M and (b) polymer PM. | ||
4.3. Photo-optical investigations
Photo-optical investigations were performed using thin (100–200 nm) films obtained by spin-coating technique using solution of polymer PM in chlorophorm. This method allowed us to obtain isotropic (amorphousized) film of the polymer, showing neither birefringence nor light scattering. Absorbance spectra of polymer film just after preparation and after annealing at 150 °C during 30 min are presented in Fig. 6. Two main absorbance maxima are observed in UV spectral range. Peak with maximum at 342 nm corresponds to π–π* electronic transition of the azobenzene chromophores, whereas peak with maximum at 247 nm relates to Φ–Φ* electronic transition of the aromatic rings. Slight absorbance in visible range at ∼450 nm relates to n–π* electronic transition of the azobenzene chromophores. These spectral features are quite typical for the azobenzene-containing systems of such type.35 Annealing of the films results in noticeable blue-shift of peak corresponding to π–π* electronic transition that is explained by process of the chromophores aggregation leading to the formation of the H-aggregates.36 | ||
| Fig. 6 Absorbance spectra of spin-coated film of polymer PM before (black curve) and after annealing (red curve). | ||
UV-irradiation induces E–Z isomerization of azobenzene chromophores that is accompanied by spectral changes presented in Fig. 7. Intensity of peak corresponding to π–π* electronic transition gradually decreases, whereas there is some increase in absorbance in the range of n–π* electronic transition of the azobenzene chromophores (Fig. 7a and c). Subsequent visible light action induces partial recovery of initial concentration of Z-form (Fig. 7b and c). It is noteworthy, that annealing of the film and formation of the LC phase noticeably decreases the rate of E–Z isomerization (Fig. 7d). This effect is explained by the reducing of the available free volume in the polymer film after LC and crystalline phase formation, close packing of molecules in smectic layer prevents formation of strongly kinked Z isomer.
Irradiation of the polymer film with polarized visible light (457 nm) resulted in appearance of significant dichroism in absorbance of azobenzene chromophores (Fig. 8). This effect is associated with photoinduced orientation of the chromophores in direction perpendicular to the polarization plane. This phenomenon is initiated by repetitive E–Z–E isomerization cycles accompanied by rotational diffusion of the chromophores. Using eqn (1) the values of dichroism during light action were calculated. Fig. 9 demonstrates kinetic curves of dichroism growth for the amorphousized and annealed films. For amorphousized film values of dichroism are quite high and comparable with values for previously studied azobenzene-containing polymers with conventional rod-like chromophores.37,38 It is noteworthy that values of the photoinduced dichroism for the amorphousized film are about six times higher than for the annealed LC and crystalline film. This effect is related to the diminished chromophores mobility in ordered LC and crystal state in comparison with amorphous one.
 | ||
| Fig. 8 (a) Polarized absorbance spectra of amorphousized PM film before and after 900 s of polarized light action (457 nm, 380 mW cm−2). (b) Polar plot of absorbance at 342 nm. | ||
 | ||
| Fig. 9 Dichroism growth in amorphousized and annealed PM films under polarized light action (457 nm, 380 mW cm−2). | ||
Subsequent annealing of the irradiated films results in completely different behaviour: for the amorphousized film the dichroism only slightly decreases under heating up to 150 °C and keeping overnight (D = 0.33), whereas for the irradiated LC film the heating results in drop of dichroism values almost to zero (D = 0.02).
The significant difference in D values and its thermostability for amorphousized and annealed films of the polymer can be explained by a coexistence of less ordered SmCP and more ordered crystalline phases in the annealed films. Moreover, we cannot exclude the presence even third disordered isotropic phase in such films. The coexistence of two or three phases is common feature of LC polymers having polymer backbone which is the source of defects in mesogenic groups packing.13 Taking into account the above-mentioned fact one can conclude that photoorientation in the annealed PM films occurs predominantly in amorphous and less ordered SmCP phase domains of the film while it is hardly observed in crystalline phase. Thus, overall values of dichroism are much smaller for the annealed films. In addition, heating of the irradiated films results in complete disorientation of photochromes because crystalline domains were unaffected by light and still “remember” their initial mesogens orientation obtained by annealing of fresh spin-coated film.
5. Conclusions
We have studied mesomorphic and photooptical properties of new bent-core monomer and corresponding side-chain polymer with photosensitive azobenzene group in the molecular core. We established the tilted smectic mesophases SmCP in both studied compounds. It is necessary to point out that by polymerization of studied photosensitive bent-core monomer we reached increase of mesophase thermostability and broadening of the liquid crystallinity phase interval.Synthesized polymethacrylate with bent-core chromophores demonstrates photooptical properties which are in general characteristic for side-chain azobenzene-containing polymers.27,37,38 UV-irradiation effectively induces E–Z isomerization process. Irradiation with polarized light results in appearance of significant dichroism by aligning the chromophores in direction perpendicular to the polarization plane of the incident light. The studied phenomena, E–Z isomerization and photoorientation, occurs more effectively in the amorphousized polymer films in comparison with annealed LC films. Formation of the LC and crystal states results in decrease in free volume in polymer films and diminishes mobility of the chromophores preventing in some extent isomerization and photoorientation process.
Studied liquid crystalline polymethacrylate with bent-core mesogens in the side-chain represent new smart material. Herein, for the first time the photooptical properties of photosensitive polymethacrylate films are presented.
Acknowledgements
This research was supported by projects No. 16-12150S (Czech Science Foundation), LH15305 (Ministry of Education, Youth and Sports of the Czech Republic), Russian Science Foundation (14-13-00379, study of photoorientation processes in polymer films), the Russian Foundation of Fundamental Research (16-03-00455, 16-29-05140, study of E–Z photoisomerization in polymer films). D. P. acknowledges the support from NCN (Poland) project DEC-2013/08/M/ST5/00781.Notes and references
- T. Niori, T. Sekine, J. Watanabe, T. Furukawa and H. Takezoe, Distinct ferroelectric smectic liquid crystals consisting of banana shaped achiral molecules, J. Mater. Chem., 1996, 6, 1231–1233, 10.1039/jm9960601231.
- W. Weissflog, H. Nadasi, U. Dunemann, G. Pelzl, S. Diele, A. Eremin and H. Kresse, Influence of lateral substituents on the mesophase behaviour of banana-shaped mesogens, J. Mater. Chem., 2001, 11, 2748–2758, 10.1039/b104098g.
- W. Weissflog, H. N. Shreenivasa Murthy, S. Diele and G. Pelzl, Relationships between molecular structure and physical properties in bent-core mesogens, Philos. Trans. R. Soc., A, 2006, 364, 2657–2679 CrossRef CAS PubMed.
- M. Kašpar, V. Hamplová, V. Novotná, M. Glogarová and P. Vaněk, New banana-type liquid crystal with a methoxy group substituted near the central ring, J. Mater. Chem., 2002, 12, 2221–2224 RSC.
- V. Novotná, M. Kašpar, V. Hamplová, M. Glogarová, L. Lejček, J. Kroupa and D. Pociecha, Ferroelectric-like behaviour of the SmCP phase in liquid crystalline compounds with asymmetrical bent-core molecules, J. Mater. Chem., 2006, 16, 2031–2038 RSC.
- A. Bubnov, V. Novotná, D. Pociecha, M. Kašpar, V. Hamplová, G. Galli and M. Glogarová, Liquid crystalline co-polysiloxane with asymmetric bent-shaped side chains, Macromol. Chem. Phys., 2011, 212, 181–197 CrossRef.
- N. Gimeno, I. Pintre, M. Martinez-Abadia, J. L. Serano and M. Blanca Ros, Bent-core liquid crystal phase promoted by azo-containing molecules: from monomers to side-chain polymers, RSC Adv., 2014, 4, 19694–19702 RSC.
- Y. Tian, K. Watanabe, X. Kong, J. Abe and T. Iyoda, Synthesis, nanostructures and functionality of amphiphilic liquid crystalline block copolymers with azobenzene moieties, Macromolecules, 2002, 35, 3739–3747 CrossRef CAS.
- A. Bobrovsky, V. Shibaev, A. Bubnov, V. Hamplová, M. Kaspar and M. Glogarová, Effect of molecular structure on chiro-optical and photo-optical properties of smart liquid crystalline polyacrylates, Macromolecules, 2013, 46, 4276–4284 CrossRef CAS.
- Y. Zhao and T. Ikeda, Smart Light-responsive Materials: Azobenzene-containing polymers and liquid crystals, Wiley &Sons: Hoboken, NJ, 2009 Search PubMed.
- H. Duer and H. Bouas-Laurent, Photochromism. Molecules and System, Elsevier, Amsterdam, 1990 Search PubMed.
- A. M. Donald, A. H. Windle and S. Hanna, Liquid Crystalline Polymers, Cambridge University Press, 2006 Search PubMed.
- Liquid Crystalline Polymers, ed. V. Thakur and M. Kessler, Springer, 2016 Search PubMed.
- A. Sobolewska, S. Bartkiewicz, J. Mysliwiec and K. D. Singer, Holographic memory devices based on a single component phototropic liquid crystal, J. Mater. Chem. C, 2014, 2, 1409–1412 RSC.
- J. Jaworska, S. Bartkiewicz and Z. Galewski, Phase Diagram of trans–cis Isomers for Photoactive and Mesogenic 4-Hexyl-4′-propoxyazobenzene, J. Phys. Chem. C, 2013, 117, 27067–27072 CAS.
- R. Hori, D. Furukawa, K. Yamamoto and S. Kutsumizu, Light-Driven Phase Transition in a Cubic-Phase-Forming Binary System Composed of 4′-n-Docosyloxy-3′-nitrobiphenyl-4-carboxylic Acid and an Azobenzene Derivative, Chem.–Eur. J., 2012, 18, 7346–7350 CrossRef CAS PubMed.
- D. Tanaka, H. Ishiguro, Y. Shimizu and K. Uchida, Thermal and photoinduced liquid crystalline phase transitions with a rod-disc alternative change in the molecular shape, J. Mater. Chem., 2012, 22, 25065–25071 RSC.
- E. Uchida, K. Sakaki, Y. Nakamura, R. Azumi, Y. Hirai, H. Akiyama, M. Yoshida and Y. Norikane, Control of the Orientation and Photoinduced phase transitions of macrocyclic azobenzene, Chem.–Eur. J., 2013, 19, 17391–17397 CrossRef CAS PubMed.
- M. Czajkowski, K. Dradrach, S. Bartkiewicz and Z. Galewski, Pattern of liquid crystalline droplets induced by two beam interference in azobenzene derivative, Opt. Mater., 2013, 35, 2449–2455 CrossRef CAS.
- J. Guan, M. Zhang, B. Li, H. Yang and G. Wang, Photocontrolled phase transitions and reflection behaviors of smectic liquid crystals by a chiral azobenzene, ChemPhysChem, 2012, 13, 3812–3818 CrossRef CAS PubMed.
- D.-Y. Kim, S.-A. Lee, H. Kim, S. M. Kim, N. Kim and K.-U. Jeong, An azobenzene-based photochromic liquid crystalline amphiphile for a remote-controllable light shutter, Chem. Commun., 2015, 51, 11080–11083 RSC.
- G. Cipparrone, P. Pagliusi, C. Provenzano and V. P. Shibaev, Reversible photoinduced chiral structure in amorphous polymer for light polarization control, Macromolecules, 2008, 41, 5992–5996, DOI:10.1021/ma801030b.
- V. Shibaev, A. Bobrovsky and N. Boiko, Photoactive liquid crystalline polymer systems with light-controllable structure and optical properties, Prog. Polym. Sci., 2003, 28(5), 729–836, DOI:10.1016/S0079-6700(02)00086-2.
- A. Bobrovsky and V. Shibaev, A study of photooptical processes in photosensitive cholesteric azobenzene-containing polymer mixture under an action of the polarized and nonpolarized light, Polymer, 2006, 47, 4310–4317, DOI:10.1016/j.polymer.2006.04.010.
- A. Bobrovsky and V. Shibaev, Thermo-, chiro- and photo-optical properties of cholesteric azobenzene-containing copolymer in thin films, J. Photochem. Photobiol., A, 2005, 172, 140–145, DOI:10.1016/j.jphotochem.2004.12.002.
- A. Bobrovsky, N. Boiko, V. Shibaev and J. Stumpe, Comparative study of photoorientation phenomena in photosensitive azobenzene-containing homopolymers and copolymers, J. Photochem. Photobiol., A, 2004, 163, 347–358, DOI:10.1016/j.jphotochem.2004.01.021.
- A. Bobrovsky, V. Shibaev, A. Bubnov, V. Hamplova, M. Kaspar, D. Pociecha and M. Glogarova, Effect of molecular structure and thermal treatment on photo-optical properties of photochromic azobenzene-containing polymer films, Macromol. Chem. Phys., 2011, 212, 342–352, DOI:10.1002/macp.201000534.
- G. Galli, S. Demel, C. Slugovc, F. Stelzer, W. Weissflog, S. Diele and K. Fodor-Csorba, Novel liquid crystal admet polymers with banana main-chain mesogens, Mol. Cryst. Liq. Cryst., 2005, 439, 1909–1919 CAS.
- J. Barbera, N. Gimeno, I. Pintre, M. Blanca Ros and J. L. Serrano, Self-assembled bent-core side-chain liquid crystalline polymers, Chem. Commun., 2006, 1212–1214 RSC.
- X. Chen, K. K. Tenneti, C. Y. Li, Y. Bai, X. Wan, X. Fan, Qi.-F. Zhou, L. Rong and B. S. Hsiao, Side-chain liquid crystalline poly(meth)acrylates with bent-core mesogens, Macromolecules, 2007, 40, 840–848 CrossRef CAS.
- L.-Y. Wang, H.-Y. Tsai and H.-C. Lin, Novel supramolecular side-chain banana-shaped liquid crystalline polymers containing covalent- and hydrogen-bonded bent cores, Macromolecules, 2010, 43, 1277–1288 CrossRef CAS.
- M. Vijay Srinivasan, P. Kannan and A. Roy, Photo and Electrically Switchable Behavior of azobenzene containing pendant bent-core liquid crystalline polymers, J. Polym. Sci., Part A: Polym. Chem., 2013, 51, 936–946 CrossRef.
- N. Gimeno, I. Pintre, M. Martinez-Abadia, J. L. Serrano and M. Blanca Ros, Bent-core liquid crystal phases promoted by azo-containing molecules: from monomers to side-chain polymers, RSC Adv., 2014, 4, 19694–19702 RSC.
- D. R. Link, G. Natale, R. Shao, J. E. Maclennan, N. A. Clark, E. Körblova and D. M. Walba, Spontaneous formation of macroscopic chiral domains in a fluid smectic phase of chiral molecules, Science, 1997, 278, 1924–1927 CrossRef CAS PubMed.
- H. M. D. Bandara and S. C. Burdette, Photoisomerization in different classes of azobenzene, Chem. Soc. Rev., 2012, 41, 1809–1825, 10.1039/c1cs15179g.
- M. Kasha, H. R. Rawls and A. M. El-Bayoumi, Pure Appl. Chem., 1965, 11, 371 CrossRef CAS.
- A. Bobrovsky and V. Shibaev, Polarised light-induced orientation and reorientation processes and unexpected ‘memory effect’ in side-chain azobenzene-containing LC polymers, Liq. Cryst., 2012, 39, 339–345, DOI:10.1080/02678292.2011.648665.
- A. Bobrovsky, V. Shibaev, V. Hamplova, M. Kaspar and M. Glogarova, Chirooptical and photooptical properties of a novel side-chain azobenzene-containing LC polymer, Monatsh. Chem., 2009, 140(7), 789–799, DOI:10.1007/s00706-009-0108-8.
| This journal is © The Royal Society of Chemistry 2016 |