POSS–PU electrospinning nanofibers membrane with enhanced blood compatibility
Abstract
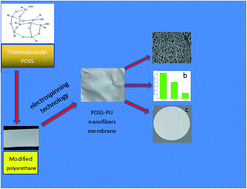
Open cage polyhedral oligomeric silsesquioxane (POSS) was firstly used to modify polyurethane (PU) to prepare a POSS–PU composite. Then a POSS–PU nanofiber membrane was prepared by electrospinning technology. The hydrophilic/hydrophobic properties, fiber morphology and biocompatibility of the POSS–PU nanofibers membrane were investigated. Contact angle increased by 24.3° for 2 wt% POSS–PU nanofibers membrane compared to PU. The ability of the nanofibers membrane surface to repel proteins and platelets was assessed by using platelet adhesion test and BSA static protein-adsorption experiment. Platelet adsorption amount obviously decreased compared with PU and very few platelet was adhered on the surfaces of nanofibers membranes when 1 wt% POSS was added. Protein adsorption was also decreased with addition of POSS. Hemolysis tests showed that hemolysis rate localized in the desired range of values (<5%) for all nanofibers membrane. Moreover, the antibacterial activity of nanofibers membranes was obviously improved after addition of POSS.

 Please wait while we load your content...
Please wait while we load your content...