Luminescence and energy transfer in a color tunable CaY4(SiO4)3O:Ce3+, Mn2+, Tb3+ phosphor for application in white LEDs
Lili Wanga,
Byung Kee Moona,
Sung Heum Parka,
Jung Hwan Kimb,
Jinsheng Shic,
Kwang Ho Kimd and
Jung Hyun Jeong*a
aDepartment of Physics, Pukyong National University, Busan 608-737, Republic of Korea. E-mail: jhjeong@pknu.ac.kr
bDepartment of Physics, Dongeui University, Busan 614-714, Republic of Korea
cDepartment of Chemistry and Pharmaceutical Science, Qingdao Agricultural University, Qingdao 266109, People's Republic of China
dSchool of Materials Science and Engineering, Pusan National University, Busan 609-735, Republic of Korea
First published on 16th August 2016
Abstract
A Ce3+/Mn2+/Tb3+ co-activated CaY4(SiO4)3O phosphor was prepared through high temperature solid state reaction process. By means of dielectric theory of chemical bonding for complex crystals, covalence of chemical bonds in CaY4(SiO4)3O was calculated. Blue emission from Ce3+ in different crystallographic cation sites was discussed quantitatively based on the calculation results. Dual energy transfer of Ce3+ → Mn2+ and Ce3+ → Tb3+ occurs and color tunable emission can be realized by modulation of their relative PL intensity. Energy transfer efficiency has been investigated and the process has been demonstrated to be a resonant type via a dipole–quadrupole mechanism. Critical distance Rc calculated through quenching concentration method and spectral overlap route is 7.23 and 7.55 Å, respectively. As-obtained samples show color tunable emission and the corresponding CIE chromaticity coordinates were given. White light with color coordinates (0.3339, 0.3055) and (0.3553, 0.3338), close to those of the ideal, was realized under lower energy UV light. All results show CaY4(SiO4)3O:Ce3+, Mn2+, Tb3+ phosphor could be a promising candidate for UV chip pumped light emitting diodes.
1. Introduction
Over the past few years, there has been increasing interest for use of light-emitting diodes (LEDs) in display and illumination applications because of their long lifetime, low energy consumption, high luminous efficiency and environment-friendly characteristics.1,2 Present commercial method to generate white light is combining blue LEDs with yellow phosphor Y3Al5O12:Ce3+.3 However, deficiency of a red component leads to low color-rendering index Ra and high correlated color temperature. To enrich red emission, additional red-emitting phosphors are usually blended.4,5 Regrettably, the complement of red emission is usually not enough for utilization. Another alternative approach to white light is using near ultraviolet (UV) or UV LEDs to pump red, green and blue phosphors.6 However, in the system, blue emission efficiency is poor because of strong re-absorption of blue light by green or red phosphors. Therefore, many efforts have been devoted to develop single component white-emitting phosphors based on energy transfer between activators.Recently, single-phased white phosphors realized by energy transfer from Ce3+, Eu2+ to Tb3+, Mn2+, Eu3+ in some hosts has attracted many attention, such as Ca3Sc2Si3O12:Ce3+, Mn2+, Ca2PO4Cl:Eu2+, Mn2+, MgY4Si3O13:Ce3+, Mn2+, NaCaBO3:Ce3+, Tb3+, Mn2+, Ba2(Gd,Tb)2Si4O13:Eu2+, Eu3+, BaY2Si3O10:Ce3+, Tb3+, Eu3+ and so on.7–12 Tb3+ is known to be a common activator in green emitting phosphors due to its predominant 5D4 → 7F5 transition at about 545 nm. Mn2+ activated luminescent materials have wide range emission from 500 to 700 nm depending on crystal field of host.13,14 However, for Tb3+ single doped phosphor, emission is usually very weak due to strictly forbidden 4f–4f transitions of Tb3+.15 A similar situation happened in luminescent materials doped with Mn2+, because its parity and spin forbidden d–d transitions are difficult to pump. Therefore, a sensitizer like Ce3+ has been applied for improving emission intensity of Tb3+ or Mn2+. Ce3+ sensitized Tb3+ or Mn2+ activated phosphors have been widely reported and energy transfer mechanism from Ce3+ to Tb3+ or Mn2+ has been investigated.16–18
As is known, general chemical formula for apatite structure is A10(XO4)6Z2 (A = Ca2+, Ba2+, Ce3+, La3+, Y3+; X = P5+, Si4+ and Z = O2−, F−, Cl−, OH−).19,20 CaY4(SiO4)3O (CYSO), which is isostructural to apatite compound, such as La4.67(SiO4)3O, La5Si2BO13, crystallizes in a hexagonal unit cell with space group P63/m (no. 176) and cell parameters of a = b = 9.36 Å, c = 6.78 Å, V = 514.4 Å3 and Z = 2.21,22 The lattice contains different types of cationic sites, nine-fold coordinated 4f sites with C3 point symmetry and seven-fold coordinated 6h sites with Cs point symmetry. Y atoms occupy both of two sites and Ca atoms are located at 4f site. In view of identical valence, Ce3+/Tb3+ and Mn2+ are expected to substitute for Y3+ and Ca2+ in CYSO lattice, respectively.
In this paper, Ce3+/Mn2+/Tb3+ co-activated CYSO phosphors synthesized by conventional high temperature solid state reaction were reported. Occupation of Ce3+ ions in crystallographic cation sites was analyzed. Energy transfer process from Ce3+ to Mn2+ was investigated and critical distance Rc between them was calculated using concentration quenching or spectral overlap route. Color tunable emission including a blue band from Ce3+, several green lines of Tb3+ and orange band originating from Mn2+ was achieved. White light was generated in CYSO:0.04Ce3+, 0.10Mn2+, 0.05Tb3+ and CYSO:0.04Ce3+, 0.10Mn2+, 0.08Tb3+ under 320 nm excitation.
2. Experimental section
2.1 Preparation
A series of Ca1−yY4−x−z(SiO4)3O:xCe3+, yMn2+, zTb3+ were prepared by conventional high temperature solid state reaction. Doping concentration of Ce3+, Mn2+ and Tb3+ is x = 0.004–0.08, y = 0.005–0.50 and z = 0.02–0.10, respectively. Chemicals CaCO3 (A.R.), Y2O3 (99.9%), SiO2 (99.99%), CeO2 (99.9%), MnCO3 (99.99%) and Tb4O7 (99.9%) were used as raw materials without further purification. Starting materials were first weighed in appropriate molar ratio and mixing was carried out by wet milling in ethanol for 20 min. After being mixed well, precursors were transferred to corundum crucible, heated in a tube furnace under a reducing atmosphere of 5% H2 and 95% N2. Temperature of furnace was increased to 1400 °C followed by holding the temperature for 6 h.2.2 Characterization
Powder X-ray diffraction (XRD) patterns of synthesized phosphor were recorded on a Philips XPert/MPD diffraction system with Cu Kα (λ = 1.5405 Å) radiation. Photoluminescence excitation (PLE) and emission (PL) spectra were collected using a Photon Technology International (PTI) spectrofluorimeter with a 60 W Xe arc lamp, and lifetimes were measured using a phosphorimeter attached to the main system with a Xe flash lamp (25 W).3. Results and discussion
3.1 Phase identification and crystal structure
Fig. 1 is comparison of representative XRD patterns of Ce3+, Mn2+ or Tb3+ doped CYSO samples with standard pattern of Y4.67(SiO4)3O (YSO, JCPDS #30-1457). Both of structure prototype of CYSO and YSO is La4.67(SiO4)3O and their detailed crystal structures have been obtained from MatNavi NIMS Materials and Springer Materials Database. Their crystal structures are almost same except for cell parameters. Peak positions in XRD patterns of CYSO would have some shifts compared with that of YSO because they are determined by distance between parallel planes of atoms in crystals. It can be found from Fig. 1 that all diffraction peaks are in good agreement with those of YSO except for a slight shift resulting from different cell parameters between them. It also can be concluded that as-prepared samples are single phase and incorporations of Ce3+, Mn2+ and Tb3+ do not bring any significant changes in host lattice structure. | ||
| Fig. 1 XRD patterns of Ce3+, Mn2+, Tb3+ doped or co-doped CaY4(SiO4)3O and Y4.67(SiO4)3O standard card. | ||
After phase identification was done, in order to know site occupation of Ce3+, Mn2+, Tb3+ in CYSO host lattice, detailed crystal structure is presented in Fig. 2, from which it can be observed that there are four kinds of cationic sites. Y3+ ions occupy two kinds of cationic sites, in which 6h sites (labeled by Y1) with seven oxygen coordinated and 4f sites (labeled by Y2) with nine. It is interesting that 4f sites are occupied by Ca2+ and Y3+ ions with equal probability. Si4+ (r = 0.26 Å, CN = 4) are too small to be replaced, and Ce3+ and Tb3+ ions are expected to substitute Ca2+ or Y3+ sites based on their ionic radii [r(Ce3+) = 1.196 Å, r(Tb3+) = 1.095 Å, r(Ca2+) = 1.18 Å, and r(Y3+) = 1.075 Å for coordination number CN = 9; r(Ce3+) = 1.07 Å, r(Tb3+) = 0.98 Å, and r(Y3+) = 0.96 Å for CN = 7]. Mn2+ ions intend to enter Ca2+ sites because of their same valence and similar ionic radii. In the following, substitution by Ce3+ will be discussed from quantitative point of view.
3.2 Ce3+ site preference analysis
PL excitation and emission spectra of CYSO:0.04Ce3+ are presented in Fig. 3. As-prepared samples exhibit a broad emission band in wavelength range of 300–600 nm with peak at about 400 nm under 320 nm UV light excitation. Excitation spectrum consists of a strong broad band at 320 nm and a weak shoulder band peaking at about 360 nm. The asymmetric excitation band can be fitted by a sum of three Gaussian peaks at 300, 326 and 361 nm. As illustrated in Fig. 3, PL spectrum is also asymmetric and can be deconvoluted into four Gaussian components, with peaks at 373 nm (26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 810 cm−1), 396 nm (25
810 cm−1), 396 nm (25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 253 cm−1), 426 nm (23
253 cm−1), 426 nm (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 474 cm−1) and 472 nm (21
474 cm−1) and 472 nm (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 186 cm−1), respectively. Energy difference for the first two is 1557 cm−1 and that for the other two is 2288 cm−1. It is known that energy difference of splitting of 2F7/2 and 2F5/2 ground states of Ce3+ is about 2000 cm−1. Energy difference between 426 nm (23
186 cm−1), respectively. Energy difference for the first two is 1557 cm−1 and that for the other two is 2288 cm−1. It is known that energy difference of splitting of 2F7/2 and 2F5/2 ground states of Ce3+ is about 2000 cm−1. Energy difference between 426 nm (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 474 cm−1) and 472 nm (21
474 cm−1) and 472 nm (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 186 cm−1) is in agreement with that between 2F7/2 and 2F5/2 energy levels of Ce3+. Combined with three excitation bands in Fig. 3, it can be concluded that broad emission band is originated from three different sites occupied by Ce3+ ions. Hexagonal CYSO provides three different cationic sites, 4f site filled up with equivalent amount of Ca and Y, 6h site occupied only by Y atoms. Among them, 6h site has a free oxygen ion (not linked to any SiO4 group) to coordinate and therefore is expected to be of higher covalency than 4f site. In order to compare intuitively, we calculated covalency fC of every chemical bond and give them in Table 1. As shown in Table 1, covalency is 0.1855 for Y1–O4, and the value for other Y1–O bonds are also higher than that of Y2–O and Ca–O chemical bonds. Therefore, three excitation bands at 300, 326 and 361 nm can be assigned to 4f → 5d transitions of Ce3+ ions in Y2, Ca and Y1 site, respectively.
186 cm−1) is in agreement with that between 2F7/2 and 2F5/2 energy levels of Ce3+. Combined with three excitation bands in Fig. 3, it can be concluded that broad emission band is originated from three different sites occupied by Ce3+ ions. Hexagonal CYSO provides three different cationic sites, 4f site filled up with equivalent amount of Ca and Y, 6h site occupied only by Y atoms. Among them, 6h site has a free oxygen ion (not linked to any SiO4 group) to coordinate and therefore is expected to be of higher covalency than 4f site. In order to compare intuitively, we calculated covalency fC of every chemical bond and give them in Table 1. As shown in Table 1, covalency is 0.1855 for Y1–O4, and the value for other Y1–O bonds are also higher than that of Y2–O and Ca–O chemical bonds. Therefore, three excitation bands at 300, 326 and 361 nm can be assigned to 4f → 5d transitions of Ce3+ ions in Y2, Ca and Y1 site, respectively.
| Bond | d (Å) | fC | Bond | d (Å) | fC | Bond | d (Å) | fC |
|---|---|---|---|---|---|---|---|---|
| Y1–O1 | 2.3607 | 0.0941 | Y2–O1 | 2.7328 | 0.0537 | Ca–O1 | 2.7328 | 0.082 |
| Y1–O1 | 2.4232 | 0.0935 | Y2–O2 | 2.4148 | 0.0556 | Ca–O2 | 2.4148 | 0.0864 |
| Y1–O2 | 2.4265 | 0.0934 | Y2–O3 | 2.3998 | 0.0557 | Ca–O3 | 2.3998 | 0.0867 |
| Y1–O3 | 2.6973 | 0.0917 | ||||||
| Y1–O4 | 2.2442 | 0.1855 |
3.3 Photoluminescence properties and energy transfer
To optimize doping concentration of Ce3+ and evaluate its influence on emission intensity, Fig. 4(a) and (c) give PL spectra of CYSO:xCe3+ (x = 0.004, 0.012, 0.02, 0.03, 0.04, 0.06) under 290 and 320 nm excitations, respectively. Emission intensities firstly increase with increasing Ce3+ concentration, and reach the maximum value at x = 0.04, then decrease as a result of concentration quenching. Therefore, doping concentration of Ce3+ of samples was fixed at x = 0.04. | ||
| Fig. 4 (a) and (c) PL spectra of CaY4−x(SiO4)3O:xCe3+; (b) and (d) PL intensity of CaY4−x(SiO4)3O:xCe3+ as a function of Ce3+ concentration. | ||
After Ce3+ was incorporated, Mn2+ was introduced into CYSO host lattice. Due to forbidden transitions of Mn2+, there must exists energy transfer from Ce3+ to Mn2+ to get bright red luminescence. Therefore PL spectra of CYSO:0.04Ce3+ and CYSO:0.2Mn2+, and PL excitation spectrum of CYSO:0.2Mn2+ are presented in Fig. 5 in a comparable way. Mn2+ single doped sample exhibits an orange emission band centered at about 588 nm and its excitation bands are in near UV and visible regions. The excitation spectrum consists of several bands centering at 322 (342), 364, 406 and 470 nm, corresponding to transitions of Mn2+ ion from ground state 6A1(6S) to 4E(4D), 4T2(4D), [4A1(4G), 4E(4G)], and 4T1(4G), respectively.23 Broad emission band from 500 to 750 nm peaking at 588 nm is ascribed to spin-forbidden 4T1(4G) → 6A1(6S) transition of Mn2+. As shown in Fig. 5, there is a significant overlap between emission spectra of Ce3+ and excitation spectrum of Mn2+, indicating possibility of effective resonant energy transfer from Ce3+ to Mn2+. Therefore, a series of samples with fixed Ce3+ concentration and variable Mn2+ content was prepared to study luminescent properties and energy transfer process.
 | ||
| Fig. 5 PL excitation spectra of Ca0.8Y4(SiO4)3O:0.2Mn2+ and PL spectra of CaY3.96(SiO4)3O:0.04Ce3+ and Ca0.8Y4(SiO4)3O:0.2Mn2+. | ||
Fig. 6 depicts PL spectra of CYSO:0.04Ce3+, yMn2+ (y = 0, 0.05, 0.1, 0.2, 0.25, 0.3, 0.4, 0.5) under excitation of 320 nm. As content of Mn2+ increases from 0 to 0.5, PL intensity of Mn2+ increases sharply firstly, then gets to the maximum, and at last quenches. In addition, one can also find that emission band of Mn2+ shifts to longer wavelength with its increasing concentration. The peak is located at about 580 nm at y = 0.05 and 588 nm at y = 0.5. This phenomenon can be explained by enhancement of crystal field strength surrounding Mn2+ ions. Crystal field strength around Mn2+ has been proposed to be:
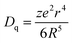 | (1) |
Different from Mn2+, PL intensity of Ce3+ decreases monotonically as increasing Mn2+ content, which means there exists resonant energy transfer from Ce3+ to Mn2+ in CYSO:0.04Ce3+, yMn2+ phosphors. It is well known that critical distance for energy transfer from sensitizer to activator by exchange interaction should be shorter than 4 Å.24 Critical distance Rc among activators can be expressed by:25
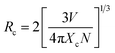 | (2) |
On basis of Dexter's energy transfer formula of exchange and multipolar interactions and Reisfeld's approximation, following relation can be satisfied:26,27
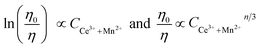 | (3) |
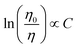 corresponds to exchange interaction, and
corresponds to exchange interaction, and 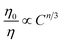 with n = 6, 8 and 10, are related to dipole–dipole, dipole–quadrupole, and quadrupole–quadrupole interactions, respectively. Value of η0/η can be approximately calculated by the ratio of relative luminescence intensities:
with n = 6, 8 and 10, are related to dipole–dipole, dipole–quadrupole, and quadrupole–quadrupole interactions, respectively. Value of η0/η can be approximately calculated by the ratio of relative luminescence intensities:
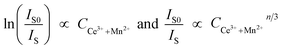 | (4) |
We plot ln(IS0/IS) versus CCe3++Mn2+, as well as IS0/IS against CCe3++Mn2+n/3, and illustrate them in Fig. 7. It is obvious that a better linear behavior can be found when n = 6 and 8, which implying that energy transfer mechanism from Ce3+ to Mn2+ is not a quadrupole–quadrupole interaction type.
 | ||
| Fig. 7 Dependence of ln(IS0/IS) of Ce3+ on CCe3++Mn2+ (a), IS0/IS on CCe3++Mn2+n/3, n = 6, 8, 10 for (b)–(d). | ||
In order to further confirm the type of energy transfer, decay curves of CYSO:0.04Ce3+, yMn2+ (y = 0, 0.05, 0.20, 0.25, 0.30) phosphors excited at 320 nm and monitored at 400 nm are shown in Fig. 8. The curves were well fitted by a single exponential equation:
I = A1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) exp(−t/τ1)
| (5) |
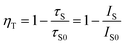 | (6) |
 | ||
| Fig. 8 Decay curves of Ce3+ emission in Ca1−yY3.96(SiO4)3O:0.04Ce3+, yMn2+ excited at 320 nm and monitored at 400 nm. The inset shows energy transfer efficiency ηT on Mn2+ concentration. | ||
From eqn (3), (4) and (6), following equation can be presented:
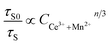 | (7) |
Plots of τS0/τS and CCe3++Mn2+n/3 (n = 6, 8) based on above equation are shown in Fig. 9. Better linear behavior was observed when n = 8, implying energy transfer from Ce3+ to Mn2+ occurred via dipole–quadrupole mechanism.
In view of this kind of interaction, critical distance from sensitizer to activator can be calculated through spectral overlap route and its formula is displayed as follows:26,29
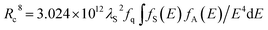 | (8) |
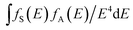 represents spectral overlap between normalized shapes of sensitizer Ce3+ emission fS(E) and activator Mn2+ excitation fA(E). In this case, the value is determined to be 0.21766 eV−5. As a result, critical distance Rc is calculated to be 7.55 Å, which is in good agreement with that obtained using concentration quenching method. This result can further testify electric dipole–quadrupole interaction is responsible for energy transfer process from Ce3+ to Mn2+ in CYSO samples.
represents spectral overlap between normalized shapes of sensitizer Ce3+ emission fS(E) and activator Mn2+ excitation fA(E). In this case, the value is determined to be 0.21766 eV−5. As a result, critical distance Rc is calculated to be 7.55 Å, which is in good agreement with that obtained using concentration quenching method. This result can further testify electric dipole–quadrupole interaction is responsible for energy transfer process from Ce3+ to Mn2+ in CYSO samples.
3.4 Luminescence and chromaticity
Until here, energy transfer mechanism from Ce3+ to Mn2+ has been confirmed, in order to obtain white light, Tb3+ was introduced as a co-dopant. Fig. 10 gives PL spectra of CYSO:0.04Ce3+, 0.10Mn2+, zTb3+ phosphors with different Tb3+ concentration of z = 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, and 0.10. The spectra under 320 nm excitation consist of several emission bands in visible region. Among them one peaking at 400 nm is from transition of Ce3+, and sharp bands at 485, 543 (549), 584 (590) and 620 nm are due to transitions of Tb3+ from 5D4 to 7F6, 7F5, 7F4, 7F3 energy levels, respectively. Broad orange emission band of Mn2+ from 500 to 700 nm overlaps with that of Tb3+. From curves in Fig. 10, it can be seen that with increasing Tb3+ concentration, PL intensity of Tb3+ increased. In order to compare chromaticity of samples with and without Tb3+ dopants, x and y values of Commission Internationale de l'Eclairage (CIE) 1931 chromaticity coordinates for CYSO:0.04Ce3+, yMn2+ (y = 0, 0.05, 0.1, 0.2, 0.25, 0.3, 0.4) and CYSO:0.04Ce3+, 0.10Mn2+, zTb3+ (z = 0.05, 0.08) samples under 320 nm excitation were calculated and presented in Table 2. Corresponding chromaticity diagram was shown in Fig. 11.| No. of points | Sample composition | CIE (x, y) |
|---|---|---|
| 1 | CYSO:0.04Ce3+ | (0.1649, 0.1040) |
| 2 | CYSO:0.04Ce3+, 0.05Mn2+ | (0.2808, 0.2195) |
| 3 | CYSO:0.04Ce3+, 0.10Mn2+ | (0.3379, 0.2745) |
| 4 | CYSO:0.04Ce3+, 0.20Mn2+ | (0.4278, 0.3564) |
| 5 | CYSO:0.04Ce3+, 0.25Mn2+ | (0.4569, 0.3798) |
| 6 | CYSO:0.04Ce3+, 0.30Mn2+ | (0.4792, 0.3974) |
| 7 | CYSO:0.04Ce3+, 0.40Mn2+ | (0.4968, 0.4035) |
| 8 | CYSO:0.04Ce3+, 0.10Mn2+, 0.05Tb3+ | (0.3339, 0.3055) |
| 9 | CYSO:0.04Ce3+, 0.10Mn2+, 0.08Tb3+ | (0.3553, 0.3338) |
 | ||
| Fig. 11 CIE chromaticity diagram for Ca1−yY3.96(SiO4)3O:0.04Ce3+, yMn2+ (y = 0, 0.05, 0.10, 0.20, 0.25, 0.30, 0.40) and Ca0.90Y3.96−z(SiO4)3O:0.04Ce3+, 0.10Mn2+, zTb3+ (z = 0.05, 0.08) samples. | ||
From chromaticity diagram in Fig. 11, it can be observed that light emitted by Ce3+ single doped CYSO phosphor is blue. After incorporation of Mn2+, chromaticity coordinates for CYSO:0.04Ce3+, yMn2+ can be tuned from blue (0.1649, 0.1040) to light purple red and finally to orange (0.4968, 0.4035), and that for CYSO:0.04Ce3+, 0.10Mn2+ (point 3, (0.3379, 0.2745)) is very close to ideal white light. Therefore, concentration of Mn2+ was fixed to be y = 0.10 and green component from Tb3+ is increased with its increasing content from z = 0.02 to 0.10. A near white light was achieved in CYSO:0.04Ce3+, 0.10Mn2+, 0.05Tb3+ and CYSO:0.04Ce3+, 0.10Mn2+, 0.08Tb3+, as given in Table 2.
4. Conclusion
In conclusion, Ce3+/Mn2+/Tb3+ co-activated CaY4(SiO4)3O phosphors were synthesized and their photoluminescence properties were investigated. Ce3+ ions were confirmed to substitute three kinds of cationic sites and generated three excitation bands. Spectral overlap between emission of Ce3+ and excitation of Mn2+ indicates energy transfer occurred among them. Mechanism of the energy transfer is a resonant type through a dipole–quadrupole interaction. Critical distance calculated using concentration quenching method and spectral overlap route was to be 7.23 and 7.55 Å, respectively. Color tunable emission from blue to orange was realized after introduction of Mn2+. White light with CIE chromaticity coordinates (0.3339, 0.3055) and (0.3553, 0.3338) was achieved in sample CaY4(SiO4)3O:0.04Ce3+, 0.10Mn2+, 0.05Tb3+ and CaY4(SiO4)3O:0.04Ce3+, 0.10Mn2+, 0.08Tb3+, respectively. Preliminary studies have implied that the Ce3+/Mn2+/Tb3+ co-activated CaY4(SiO4)3O phosphors may serve as a potential phosphor for UV chip pumped white light emitting diodes.Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (No. 2015060315). This work also supported by the Global Frontier R&D Program (2013-073298) on Center for Hybrid Interface Materials (HIM) funded by the Ministry of Science, ICT & Future Planning. The Ce3+/Mn2+/Tb3+ activated CaY4(SiO4)3O phosphor was supplied by the Display and Lighting Phosphor Bank at Pukyong National University.Notes and references
- M. S. Shur and R. Zukauskas, Proc. IEEE, 2005, 93, 1691–1703 CrossRef CAS.
- Y. Narukawa, J. Narita, T. Sakamoto, T. Yamada, H. Narimatsu, M. Sano and T. Mukai, Phys. Status Solidi A, 2007, 204, 2087–2093 CrossRef CAS.
- Y. Jia, Y. Huang, Y. Zheng, N. Guo, H. Qiao, Q. Zhao, W. Lv and H. You, J. Mater. Chem., 2012, 22, 15146–15152 RSC.
- Q. Shao, H. Li, K. Wu, Y. Dong and J. Jiang, J. Lumin., 2009, 129, 879–883 CrossRef CAS.
- P. Pust, V. Weiler, C. Hecht, A. Tücks, A. S. Wochnik, A. K. Henß, D. Wiechert, C. Scheu, P. J. Schmidt and W. Schnick, Nat. Mater., 2014, 13, 891–896 CrossRef CAS PubMed.
- U. Kaufmann, M. Kunzer, K. Köhler, H. Obloh, W. Pletschen, P. Schlotter, R. Schmidt, J. Wagner, A. Ellens, W. Rossner and M. Kobusch, Phys. Status Solidi A, 2001, 188, 143–146 CrossRef CAS.
- C. H. Hsu, S. Das and C. H. Lu, J. Electrochem. Soc., 2012, 159, J193 CrossRef CAS.
- Y. Liu, X. Zhang, Z. Hao, Y. Luo, X. Wang, L. Ma and J. Zhang, J. Lumin., 2013, 133, 21–24 CrossRef CAS.
- X. Zhang and M. Gong, Dalton Trans., 2014, 43, 2465–2472 RSC.
- P. Li, Z. Wang, Z. Yang and Q. Guo, J. Mater. Chem. C, 2014, 2, 7823 RSC.
- J. Zhou, Z. Xia, M. Bettinelli and Q. Liu, RSC Adv., 2016, 6, 2046–2054 RSC.
- J. Zhou and Z. Xia, J. Mater. Chem. C, 2015, 3, 7552–7560 RSC.
- D. T. Palumbo and J. J. Brown, J. Electrochem. Soc., 1970, 117, 1184–1188 CrossRef CAS.
- D. T. Palumbo and J. J. Brown, J. Electrochem. Soc., 1971, 118, 1159–1164 CrossRef CAS.
- Y. Chen, J. Wang, X. Zhang, G. Zhang, M. Gong and Q. Su, Sens. Actuators, B, 2010, 148, 259–263 CrossRef CAS.
- X. Xu, Y. Wang, X. Yu, Y. Li and Y. Gong, J. Am. Ceram. Soc., 2011, 94, 160–163 CrossRef CAS.
- D. Geng, H. Lian, M. Shang, Y. Zhang and J. Lin, Inorg. Chem., 2014, 53, 2230–2239 CrossRef CAS PubMed.
- H. Y. Chung, C. H. Lu and C. H. Hsu, J. Am. Ceram. Soc., 2010, 93, 1838–1841 CAS.
- Q. Guo, L. Liao and Z. Xia, J. Lumin., 2014, 145, 65–70 CrossRef CAS.
- H. Liu, L. Liao and Z. Xia, RSC Adv., 2014, 4, 7288–7295 RSC.
- N. F. Fedorov, I. F. Andreev and N. S. Meliksetyan, Inorg. Mater., 1975, 11, 1137–1138 Search PubMed.
- Z. Xia, M. S. Molokeev, W. B. Im, S. Unithrattil and Q. Liu, J. Phys. Chem. C, 2015, 119, 9488–9495 CAS.
- W. J. Yang, L. Luo, T. M. Chen and N. S. Wang, Chem. Mater., 2005, 17, 3883–3888 CrossRef CAS.
- B. M. Antipenk, I. Bataev, V. Ermolaev, E. Lyubimov and T. A. Privalov, Opt. Spectrosc., 1970, 29, 177 Search PubMed.
- G. Blasse, Phys. Lett. A, 1968, 28, 444–445 CrossRef CAS.
- G. Blasse, Philips Res. Rep., 1969, 24, 131–144 CAS.
- D. L. Dexter, J. Chem. Phys., 1953, 21, 836–850 CrossRef CAS.
- P. Paulose, G. Jose, V. Thomas, N. Unnikrishnan and M. Warrier, J. Phys. Chem. Solids, 2003, 64, 841–846 CrossRef CAS.
- D. L. Dexter, J. Chem. Phys., 1953, 21, 836–850 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |





