Effect of few-layer graphene films as electrodes on the electrical properties of ferroelectric capacitors
Hong Jing Han,
Yan Na Chen and
Zhan Jie Wang*
Shenyang National Laboratory for Materials Science, Institute of Metal Research (IMR), Chinese Academy of Sciences (CAS), 72 Wenhua Road, Shenyang 110016, China. E-mail: wangzj@imr.ac.cn
First published on 5th July 2016
Abstract
Electrode materials have a great influence on the performance of ferroelectric film capacitors, and it is still a challenge to find new electrode materials. In this work, highly conductive few-layer graphene (FLG) films were prepared on the surface of the PZT films as top electrodes by dip coating and mechanical exfoliation and their effect on the electric properties of the PZT film capacitors were investigated. The polarization property of the FLG/PZT/Pt capacitor is almost the same as that of the Pt/PZT/Pt capacitor. However, the magnitude of leakage current density for the FLG/PZT/Pt capacitor is largely decreased due to the van der Waals (vdW) gap at the FLG/PZT interface. The FLG films have relatively little oxygen functional groups due to the microwave irradiation treatment, and are more suitable for utilization as the electrode of the PZT capacitors compared with the few-layer-graphene oxide (FLGO) films. Our results demonstrate that the FLG films might be a promising electrode material for application in integrated ferroelectric devices.
Introduction
Graphene, as a single-atom-thick layer of graphite, has became one of the most promising electronic materials because of its excellent optical transparency, flexibility and electrical conductivity. Due to its unique properties, graphene has been applied in various electric devices, including field-effect transistors (FETs),1–3 touch screens,4,5 sensors6,7 and so on. Usually, graphene with high quality can be obtained by micromechanical exfoliation of highly oriented pyrolytic graphite1 or epitaxial growth on SiC,8 but the high cost of such production methods prevented their applications in electric devices. In contrast, reduced graphene oxide (rGO) is a widely used material for transparent conductive electrodes due to the low-cost and solution-processed fabrication,9–11 although the electrical conductivity of reduced graphene oxide is decreased in some extent owing to the disruption of π-conjugated structures by oxygen function groups. So far, the rGO films with a sheet resistance about 102 to 104 Ω □−1 have been suggested to be appropriate materials for transparent conductive electrodes applying in flexible displays,4,12 solar cells13,14 and optoelectronic devices.15,16 The rGO films with a low sheet resistance show the trend to be the next-generation conductive electrodes which may possibly replace traditional electrodes commonly used in modern electric devices. Therefore, to further expand the application of rGO films, it is expected that the rGO films with a low resistance can be used as electrodes in other devices.On the other hand, ferroelectric films have been extensively studied for many applications in the integrated ferroelectric devices in recent years, such as nonvolatile ferroelectric random-access memory (FeRAM),17 atomic force microscopy cantilevers and MEMS shunt switches.18 Ferroelectric film capacitor with a sandwich structure of electrode/ferroelectric-film/electrode is an elementary unit for the integrated ferroelectric devices. Among the mostly used typical ferroelectric films, lead zirconate titanate (Pb(ZrxTi1−x)O3: PZT) has attracted great interest due to its large remnant polarization, small leakage current density and high reliability.19 It is widely reported that the performance of PZT capacitors is highly dependent on top and bottom electrodes. Noble metals of Pt and Au, and conductive perovskite oxide, such as La0.5Sr0.5CoO3 (LSCO),20 La0.7Sr0.3MnO3 (LSMO)21 and LaNiO3 (LNO)22 are the most commonly used materials for electrodes of PZT capacitors. However, the noble metals used as electrodes for PZT capacitors are prone to produce ferroelectric polarization fatigue. On the other hand, the application of conductive perovskite electrodes (LSCO, LSMO, LNO and so on) can improve the performance for PZT capacitors,23 but there still exist some barriers that may impede their practical application in the integrated ferroelectric devices. For example, the property of PZT films may be deteriorated by the compositional diffusion at the electrode/film interface.24 The high resistivity for such conductive oxides is another drawback for them to be used in PZT capacitors.25 Therefore, alternative materials that can be used as electrodes for PZT capacitors are still extremely expected. In our previous study,26 we have successfully prepared the high-conductive few-layer graphene (FLG) films by reducing few-layer-graphene oxide (FLGO) films through microwave irradiation. In this study, we try to prepare the high-conductive FLG films on the PZT films to further confirm their possibility as electrode materials for ferroelectric capacitors.
The FLG films as top electrodes are prepared on the surface of the PZT films by dip coating and mechanical exfoliation. And then the electric properties of the ferroelectric capacitor with a sandwich structure of FLG/PZT/Pt characterized by measuring the polarization–electric field (P–E) hysteresis loops, capacitance–voltage (C–V) curves and leakage current density–voltage (J–V) curves. The results show that the FLG/PZT/Pt ferroelectric capacitors have good polarization properties and a low leakage current density, indicating that FLG films might be an alternative electrode material for the integrated ferroelectric devices.
Experimental procedure
The PZT films were spin-coated on the Pt/Ti/SiO2/Si (20 × 20 mm2) substrate by a sol–gel method. The sol–gel precursor solution was prepared from lead acetate, zirconium-n-propoxide and titanium teraisopropoxide. 2-Propanol was used as the solvent. The composition of the solution was adjusted to 0.4 M with the atomic ratio of Pb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zr
Zr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ti = 1.1
Ti = 1.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.52
0.52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.48. The amorphous PZT films were prepared by spin coating, drying and pyrolizing processes. The process details have been reported in our previous research.27 Then the amorphous PZT film was annealed in an electric furnace at 600 °C for 30 min with the heating rate of 35 °C s−1. At last, the crystallized PZT film was divided into four pieces with the same size of 10 × 10 mm2 for fabrication of different top electrodes on them.
0.48. The amorphous PZT films were prepared by spin coating, drying and pyrolizing processes. The process details have been reported in our previous research.27 Then the amorphous PZT film was annealed in an electric furnace at 600 °C for 30 min with the heating rate of 35 °C s−1. At last, the crystallized PZT film was divided into four pieces with the same size of 10 × 10 mm2 for fabrication of different top electrodes on them.
The few-layer graphene (FLG) films used for top electrodes were prepared by reducing few-layer-graphene oxide (FLGO) films through microwave irradiation. The FLGO dispersion was prepared by electrochemical exfoliation of the highly oriented pyrolytic graphite (HOPG). The process details for the preparation of FLGO and FLG have been described in our previous publication.26 It has been known that over 50% of the graphene sheets have 2–5 layers through the AFM statistical analysis. Therefore, compared with other graphene sheets with more than 5 layers, the graphene sheets obtained in our experiment are named few-layer graphene oxide (FLGO). After microwave irradiation, large quantities of oxygen functional groups are removed and such graphene sheets are named few-layer graphene (FLG). Fig. 1 shows the process for the fabrication of the FLG film on the PZT film. Firstly, the surface of the PZT film was covered by the FLG film with a thickness of about 30 nm (Fig. 1(a)). For preparation of the FLG film on the surface of the PZT/Pt/Ti/SiO2/Si substrate, the FLGO dispersion in DMF solution was mixed with DI water by volume ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The FLGO sheets aggregated on the surface of the solution and formed a thin film. And then the FLGO thin film was transferred onto the surface of PZT/Pt/Ti/SiO2/Si substrate. The as-deposited FLGO film was heated at 120 °C for 5 min on a hot plate to remove water and organic matter. After that, the FLGO film was reduced to the FLG film by microwave irradiation using a single mode microwave generator with the frequency of 2.45 GHz at the output power of 42 W for 5 min. The effect of microwave radiation with the above experimental conditions has been confirmed in our previous study.26 Compared with the conventional treatment in an electric furnace at 250 °C for 30 min, microwave irradiation showed a high efficiency in the reduction of graphene oxide. Moreover, such operating conditions ensure that the elevated temperature of the sample is below 250 °C and will not affect the well-crystallized PZT films under the graphene electrodes. The above processes were repeated for several times to obtain the 30 nm-thick FLG film. Secondly, as shown in Fig. 1(b), the integrated FLG film was divided into square electrodes with the same lateral size of about 0.5 mm by mechanical exfoliation using a narrow scotch tape. For comparison, the FLGO film as top electrodes on the PZT film was made using the same procedure but without the process of microwave reduction. In addition, Pt top electrodes (diameter: 0.5 mm) were also sputtered on the surface of the PZT film for comparison.
1. The FLGO sheets aggregated on the surface of the solution and formed a thin film. And then the FLGO thin film was transferred onto the surface of PZT/Pt/Ti/SiO2/Si substrate. The as-deposited FLGO film was heated at 120 °C for 5 min on a hot plate to remove water and organic matter. After that, the FLGO film was reduced to the FLG film by microwave irradiation using a single mode microwave generator with the frequency of 2.45 GHz at the output power of 42 W for 5 min. The effect of microwave radiation with the above experimental conditions has been confirmed in our previous study.26 Compared with the conventional treatment in an electric furnace at 250 °C for 30 min, microwave irradiation showed a high efficiency in the reduction of graphene oxide. Moreover, such operating conditions ensure that the elevated temperature of the sample is below 250 °C and will not affect the well-crystallized PZT films under the graphene electrodes. The above processes were repeated for several times to obtain the 30 nm-thick FLG film. Secondly, as shown in Fig. 1(b), the integrated FLG film was divided into square electrodes with the same lateral size of about 0.5 mm by mechanical exfoliation using a narrow scotch tape. For comparison, the FLGO film as top electrodes on the PZT film was made using the same procedure but without the process of microwave reduction. In addition, Pt top electrodes (diameter: 0.5 mm) were also sputtered on the surface of the PZT film for comparison.
The crystalline structure and orientation of the PZT films were analyzed by X-ray diffraction (XRD; D/max-2000, Cu Kα radiation, Rigaku, Japan). The cross-sectional microstructures of the ferroelectric capacitors with the top electrodes of FLG, FLGO and Pt were studied by transmission electron microscopy (TEM; Tecnai G2 F20, FEI, Eindhoven, The Netherlands). The TEM specimens were ground and polished to a thickness of 20 mm and were further thinned to perforation by using a Gatan precision ion-polishing system (PIPS 691, Gatan, Pleasanton, USA). The polarization versus electric field (P–E) hysteresis loop, capacitance versus voltage (C–V) hysteresis loop and leakage current density versus voltage (J–V) characteristics of the ferroelectric capacitors were measured by using a standard ferroelectric testing system (TF2000E, Axiacct, Aachem, Germany).
Results and discussion
Fig. 2 shows the XRD patterns of the PZT films with the top electrodes of Pt, FLGO and FLG. All the PZT films show only one diffraction peak for (111) planes of the perovskite phase and no peak of the pyrochlore phase can be detected. These results indicate that the PZT films crystallized well into the perovskite phase, and have a nearly completed (111)-preferred orientation, which is consist with our previous results.27 In addition, the intensity of diffraction peak for the three films is almost same, meaning that the three films have almost the same crystal quality. These results indicate that the three PZT films should have the same intrinsic properties, thus we can study the influence of these three top electrodes on the electronic properties of the PZT capacitors.Fig. 3 shows cross-sectional TEM images of the PZT films with the top electrodes of FLG and Pt. The PZT films crystallized well into the perovskite phase, and are composed of columnar grains. As shown in Fig. 3(a), the FLG layer with a thickness of 30 nm is in close contact with the PZT film. The inset in Fig. 3(a) is a HRTEM image showing the FLG/PZT interface. No parasitic layer at the FLG/PZT interface can be observed. By TEM study, we also confirmed that the FLGO/PZT interface has a similar nature. Fig. 3(b) shows a TEM image of the Pt/PZT/Pt capacitor. The top electrode of Pt layer with a thickness of 30 nm is in close contact with PZT film, which can also be confirmed by the HRTEM observation on the Pt/PZT interface (the inset in Fig. 3(b)). Pintilie et al.28 reported that sometimes there may be a parasitic layer at the interface between the metal electrode and the ferroelectric film, which may introduce an additional potential barrier at the interface and affect the property of the ferroelectric film. The above experimental results reveal that there is no parasitic layer at the interfaces between the top electrode layer and the PZT film.
The ferroelectric properties of the PZT capacitors with the top electrodes of FLG, FLGO and Pt were investigated by measuring the P–E hysteresis loops at a frequency of 1 kHz, and the results are shown in Fig. 4. The P–E hysteresis loops of the PZT capacitors with the top electrodes of FLG and Pt show a similar shape. The average remanent polarization (Pr) is 47 μC cm−2 and 48 μC cm−2 for the top electrodes of FLG and Pt, respectively. Baeumer et al.3 reported that the remanent polarization of the Pd/graphene/Pb(Zr0.2Ti0.8)O3/SrRuO3 capacitor was reduced to 23 μC cm−2 from 80 μC cm−2 by using the single-layer graphene as a part of the top electrode compared with the Pd electrode. They thought that the reduced polarization was attributed to the low carrier density of the single-layer graphene or the existence of a dielectric layer caused by absorbed molecules at the graphene/PZT interface. In this study, the thickness of the FLG layer used is about 30 nm as shown as the TEM results and is much larger than that of the single-layer graphene, so enough of the carriers can provide a complete compensation of the polarization. Moreover, the thicker FLG layer also avoided direct contact between the probe and the PZT film. In addition, microwave irradiation was used for preparation of the FLG layer, which may eliminate the absorbed molecules at the FLG/PZT interface as well. In contrast, the P–E hysteresis loop of the PZT capacitor with the FLGO top electrode is quite different from that with the FLG top electrode, and the polarization property is reduced greatly. A major difference between the FLG layer and the FLGO layer is the content of oxygen functional groups contained in them. In our previous work, the effect of microwave irradiation on the content of oxygen functional groups has been investigated by the X-ray photoelectron spectroscopy (XPS) study.26 Two peaks corresponding to C 1s at 284.6 eV and O 1s at 532.6 eV appeared in all the spectra for the as-prepared graphene films (FLG), the graphene films that after conventional annealing (FLG-CA) and microwave irradiation (FLG-MWI). The presence of the O peak in the FLG film indicates that the FLG sheets were still somewhat oxidized during the electrochemical exfoliation process. After conventional annealing or microwave irradiation, the C/O ratio is increased from 7.8 to 9.3 and 17.5, respectively, indicating that both treatment methods can remove oxygen functional groups. Moreover, microwave irradiation shows a better reduction effect than conventional annealing. Such a high value of 17.5 for C/O ratio is almost comparable to that of reduced graphene oxide (rGO) obtained by chemical or thermal reduction method.29–32 The further characterization of C 1s spectrum indicated that these functional groups are assigned to C–O bonds (such as hydroxyl or epoxide groups) at around 286 eV and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds relating to carboxylic acid groups at around 288.8 eV. After treated by microwave irradiation, the intensity of the peaks for C–O and C
O bonds relating to carboxylic acid groups at around 288.8 eV. After treated by microwave irradiation, the intensity of the peaks for C–O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O greatly reduced, indicating that most of oxygen functional groups has been eliminated. In terms of the distribution of oxygen functional groups, Erickson33 and Bachmatiuk34 have made detailed study by using aberration-corrected transmission electron microscopy and have drawn the conclusion that these oxygen functional groups dispersively distribute on the surface of the graphene oxide sheets. In addition, the conductivity of graphene films is mainly related to the number of oxygen functional groups. It has been reported that graphene oxide shows a poor electrical conductivity due to the disruption of π-conjugated structures by oxygen functional groups. The sheet resistance for the as-prepared FLG film is about 107 Ω □−1, and largely reduced to ∼6 × 103 Ω □−1 by microwave irradiation, which can be attributed to the removal of oxygen functional groups caused by microwave irradiation.26 A higher number of oxygen functional groups in the FLGO layer lead to a poor electrical conductivity. Additionally, there may be some absorbed molecules existing at the FLGO/PZT interface, but not at the FLG/PZT interface due to the microwave irradiation treatment.
O greatly reduced, indicating that most of oxygen functional groups has been eliminated. In terms of the distribution of oxygen functional groups, Erickson33 and Bachmatiuk34 have made detailed study by using aberration-corrected transmission electron microscopy and have drawn the conclusion that these oxygen functional groups dispersively distribute on the surface of the graphene oxide sheets. In addition, the conductivity of graphene films is mainly related to the number of oxygen functional groups. It has been reported that graphene oxide shows a poor electrical conductivity due to the disruption of π-conjugated structures by oxygen functional groups. The sheet resistance for the as-prepared FLG film is about 107 Ω □−1, and largely reduced to ∼6 × 103 Ω □−1 by microwave irradiation, which can be attributed to the removal of oxygen functional groups caused by microwave irradiation.26 A higher number of oxygen functional groups in the FLGO layer lead to a poor electrical conductivity. Additionally, there may be some absorbed molecules existing at the FLGO/PZT interface, but not at the FLG/PZT interface due to the microwave irradiation treatment.
To further investigate the effects of the FLG and FLGO films as the top electrode on the PZT capacitor, the capacitance–voltage (C–V) characteristics were measured. As shown in Fig. 5, the C–V hysteresis loops of the PZT capacitors with three different top electrodes show a typical butterfly shape. However, a shift of the symmetry axis of the C–V loops can be clearly observed. The cross points of the C–V loops are marked as XPt, XFLG and XFLGO, and their positions are −0.32 V, 0.29 V and 0.89 V, respectively. Chung et al.35 reported that a shift of the symmetry axis for the C–V loops may be attributed to different work functions for the top and bottom electrodes or charges at the electrode/film interface. The work function for the reduced graphene oxide (rGO) film ranges from 4.7 eV to 5.2 eV according the number of oxygen functional groups containing in it.36 A higher number of oxygen functional groups in the FLGO layer lead to a larger work function. Hence, we can assume that the work function of FLG and FLGO is 4.7 eV and 5.2 eV, respectively. The work function of Pt is 5.3 eV as reported by Nagaraj et al.37 FLGO and Pt have a similar work function, but for the PZT capacitor with the FLGO top electrode, the position of the symmetry axis is largely shifted towards the positive direction of the X axis. As mentioned above, there are large numbers of oxygen functional groups in the FLGO layer. When an extra electric field is applied on the FLGO layer, these oxygen functional groups will turn to negatively charged ions (OH−, COOH− and so on) through acquiring electrons under the extra electric field. This phenomenon has been reported in the study on resistive switching characteristics of the graphene oxide film.38,39 Gopalan et al.40 reported that absorption of OH− in LiTaO3 crystals led to an additional built-in electric field and caused a positive shift for the C–V curve. Chung et al.35 also mentioned that charges at the electrode/film interface would shift the symmetry axis of the C–V curves positively. Therefore, these negatively charged ions at the FLGO/PZT interface lead to the positive shift of the position for X. Namely, these negatively charged ions resulted in an additional built-in electric field, and a part of the extra electric field was offset by this built-in electric field, and may cause the poor polarization performance of the PZT capacitors with the FLGO top electrodes (Fig. 4). In contrast, for the PZT capacitor with the FLG top electrode, the symmetry axis of the C–V curves only shifted to 0.29 eV along the X axis. Few number of oxygen functional groups in the FLG avoided the appearance of the negatively charged ions at the FLG/PZT interface, so the positive shift of the symmetry axis was largely reduced. It is clear that the FLG films as top electrodes can ensure a better polarization performance for the PZT capacitor.
Fig. 6 shows the leakage current density–voltage (J–V) characteristics for the PZT capacitors with the top electrodes of FLG, FLGO and Pt. The PZT capacitors with the top electrodes of FLG and FLGO possess a comparable magnitude of the leakage current density, which are two orders lower than that with the Pt top electrodes. Due to the similar work function between PZT and conductive perovskite oxides, the leakage current density in the PZT films with oxide electrodes is much higher than that with the metal electrodes, which has been reported by Tang et al.41 and Zhou et al.42 This is also another barrier that may impede the practical application of conductive perovskite oxide electrodes in the integrated ferroelectric devices. So compared with oxide and metal electrodes, the leakage current density is reduced in large extent when use the FLG films as electrodes. It has been reported that a Schottky emission mechanism is dominant for the PZT film with Pt electrode.43 As shown in Fig. 6, the similar shape of the J–V curves may hint that a Schottky emission mechanism is also dominant for the PZT capacitors with the top electrodes of FLG and FLGO. For the interface-controlled Schottky emission mechanism, the charge injection is determined by the height of the potential barrier at the electrode/ferroelectric interface, and the magnitude of leakage current density can be estimated in the following equation:43
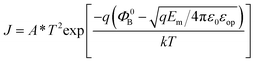 | (1) |
 | (2) |
 | ||
Fig. 6 (a) Leakage current density–voltage (J–V) characteristics for Pt/PZT/Pt, FLGO/PZT/Pt and FLG/PZT/Pt capacitors, (b) relationship between ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) J/T2 and V1/2 for the three capacitors. J/T2 and V1/2 for the three capacitors. | ||
According to eqn (2), it can be assumed that the graphical representation for ln(J/T2) − V1/2 at a constant temperature should be a straight line. Fig. 6(b) shows the fitting results of eqn (2) for the PZT capacitors with the top electrodes of FLG, FLGO and Pt at room temperature. The fitted straight lines for the three electrodes show a similar slope, confirming that an interface-controlled Schottky emission mechanism is dominated for these PZT capacitors. According to the Schottky theory for a metal–semiconductor contact, the Schottky potential barrier height at zero applied field can be estimated using the following equation:43
| Φ0B = S(Φm − χ) + (1 − S)(Eg − Φ0) | (3) |
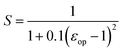 | (4) |
The value of S obtained for the PZT film is about 0.25 considering a εop value of 6.5. By using the parameters of χ = 3.5 ± 0.2 eV,45 Eg = 3.7 eV,46 Φ0 = 1.9 eV,47 ΦFLG = 4.7 eV, ΦFLGO = 5.2 eV, and ΦPt = 5.3 eV, the value of Φ0B for the FLG/PZT, FLGO/PZT and Pt/PZT interfaces can be calculated to be Φ0B(FLG/PZT) = 1.8 eV, Φ0B(FLGO/PZT) = 1.925 eV and Φ0B(Pt/PZT) = 1.95 eV, respectively. According to the Φ0B values of the FLG/PZT, FLGO/PZT and Pt/PZT interfaces, the leakage current density (J) for the PZT capacitors with the top electrodes of FLG, FLGO and Pt is assumed to obey the relationship: JFLG/PZT > JFLGO/PZT > JPt/PZT, which is inconsistent with the above experimental results as shown in Fig. 6(a). Therefore, except the difference in the work function between the FLG/PZT, FLGO/PZT and Pt/PZT interfaces, there may be additional factors that affect the height of the potential barrier at the electrode/PZT interfaces.
The contact for the PZT film with the Pt layer is through forming electrovalent or metallic bonds, but for the PZT film with the FLG or FLGO is through van der Waals interaction. For the Pt/PZT interface, the height of the Schottky barrier is determined by the work functions of Pt and the electron affinity of the PZT film, but for the FLG/PZT and FLGO/PZT interfaces, it is not the case. Allain et al.48 reported that if single or few-layer 2D MoS2 contacted with a bulk (3D) metal through van der Waals interaction, a van der Waals (vdW) gap can be formed at the interface in most situations. This vdW gap acts as an additional barrier for charge carriers before the inherent Schottky barrier and largely decreases the charge injection from the metal. In this study, the FLG or FLGO film and the PZT film is also connected with each other through van der Waals interaction, thus a vdW gap is assumed to exist at the FLG/PZT and FLGO/PZT interfaces. Combining the above analysis, the energy band diagrams of the FLG/PZT, FLGO/PZT and Pt/PZT heterostructures before and after contact are drawn schematically in Fig. 7. It can be seen that after the contact of FLG or FLGO with the PZT film, the potential barrier Φ0B at the interface is composed of two parts: the van der Waals (vdW) gap (ΦvdW) and the inherent Schottky barrier (ΦSB). According to the calculation results mentioned above, the inherent Schottky barrier ΦSB is 1.8 eV, 1.925 eV and 1.95 eV for the FLG/PZT, FLGO/PZT and Pt/PZT interfaces, respectively. With the additional barrier ΦvdW caused by vdW gap for the FLG/PZT and FLGO/PZT interfaces, it can be concluded that the height of the potential barriers at zero applied filed for three interfaces have the relationship as follows: Φ0B(FLGO/PZT) > Φ0B(FLG/PZT) >Φ0B(Pt/PZT). Thus the magnitude of leakage current density for the PZT capacitors with the top electrodes of FLG, FLGO and Pt should be JPt/PZT > JFLG/PZT > JFLGO/PZT, which in complete agreement with the experimental results. Therefore, the magnitude of the leakage current density in the PZT capacitors with the electrodes of FLG and FLGO is reduced.
 | ||
| Fig. 7 Schematic energy band diagrams of (a) FLG/PZT, (b) FLGO/PZT and (c) Pt/PZT heterostructures before and after contact. | ||
Conclusions
In this study, the FLG and FLGO films as top electrodes were prepared by dip coating and mechanical exfoliation on the surface of the PZT films, and their effects on the electric properties of the PZT capacitors were investigated. The polarization property of the FLG/PZT/Pt capacitor is almost the same as that of the Pt/PZT/Pt capacitor. However, the magnitude of leakage current density for the FLG/PZT/Pt capacitors is largely decreased due to the vdW gap at the FLG/PZT interface. The FLG films are more suitable for utilization as the electrode of the PZT capacitors, because they have relatively little oxygen functional groups compared with the FLGO films. These results demonstrate that the FLG films might be a promising electrode material using in integrated ferroelectric devices.Acknowledgements
This research was supported by the Hundred Talents Program of the Chinese Academy of Sciences and the National Natural Science Foundation of China (No. 51172238).References
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, Y. Zhang, S. V. Dubonos, I. V. Grigorieva and A. A. Firsov, Science, 2004, 306, 666–669 CrossRef CAS PubMed.
- W. Jie, Y. Y. Hui, N. Y. Chan, Y. Zhang, S. P. Lau and J. Hao, J. Phys. Chem. C, 2013, 117, 13747–13752 CAS.
- C. Baeumer, S. P. Rogers, R. Xu, L. W. Martin and M. Shim, Nano Lett., 2013, 13, 1693–1698 CAS.
- S. Bae, H. Kim, Y. Lee, X. Xu, J.-S. Park, Y. Zheng, J. Balakrishnan, T. Lei, H. Ri Kim, Y. I. Song, Y.-J. Kim, K. S. Kim, B. Ozyilmaz, J.-H. Ahn, B. H. Hong and S. Iijima, Nat. Nanotechnol., 2010, 5, 574–578 CrossRef CAS PubMed.
- J. Wang, M. Liang, Y. Fang, T. Qiu, J. Zhang and L. Zhi, Adv. Mater., 2012, 24, 2874–2878 CrossRef CAS PubMed.
- Y. Dan, Y. Lu, N. J. Kybert, Z. Luo and A. T. C. Johnson, Nano Lett., 2009, 9, 1472–1475 CrossRef CAS PubMed.
- J. D. Fowler, M. J. Allen, V. C. Tung, Y. Yang, R. B. Kaner and B. H. Weiller, ACS Nano, 2009, 3, 301–306 CrossRef CAS PubMed.
- P. W. Sutter, J.-I. Flege and E. A. Sutter, Nat. Mater., 2008, 7, 406–411 CrossRef CAS PubMed.
- G. Eda, Y.-Y. Lin, S. Miller, C.-W. Chen, W.-F. Su and M. Chhowalla, Appl. Phys. Lett., 2008, 92, 233305 CrossRef.
- C.-Y. Su, A.-Y. Lu, Y. Xu, F.-R. Chen, A. N. Khlobystov and L.-J. Li, ACS Nano, 2011, 5, 2332–2339 CrossRef CAS PubMed.
- D. Sun, L. Jin, Y. Chen, J. R. Zhang and J. J. Zhu, ChemPlusChem, 2013, 78, 227–234 CrossRef CAS.
- G. Eda, G. Fanchini and M. Chhowalla, Nat. Nanotechnol., 2008, 3, 270–274 CrossRef CAS PubMed.
- K. Saranya, N. Sivasankar and A. Subramania, RSC Adv., 2014, 4, 36226–36233 RSC.
- L. J. Larsen, C. J. Shearer, A. V. Ellis and J. G. Shapter, RSC Adv., 2015, 5, 38851–38858 RSC.
- L. Gomez De Arco, Y. Zhang, C. W. Schlenker, K. Ryu, M. E. Thompson and C. Zhou, ACS Nano, 2010, 4, 2865–2873 CrossRef CAS PubMed.
- T.-H. Han, Y. Lee, M.-R. Choi, S.-H. Woo, S.-H. Bae, B. H. Hong, J.-H. Ahn and T.-W. Lee, Nat. Photonics, 2012, 6, 105–110 CrossRef CAS.
- J. F. Scott, Science, 2007, 315, 954–959 CrossRef CAS PubMed.
- G. L. Smith, J. S. Pulskamp, L. M. Sanchez, D. M. Potrepka, R. M. Proie, T. G. Ivanov, R. Q. Rudy, W. D. Nothwang, S. S. Bedair, C. D. Meyer and R. G. Polcawich, J. Am. Ceram. Soc., 2012, 95, 1777–1792 CrossRef CAS.
- N. Izyumskaya, Y. I. Alivov, S. J. Cho, H. Morkoç, H. Lee and Y. S. Kang, Crit. Rev. Solid State, 2007, 32, 111–202 CrossRef CAS.
- T. Ohno, T. Matsuda, T. Nukina, N. Sakamoto, N. Wakiya, S. Tokuda and H. Suzuki, Mater. Lett., 2010, 64, 1742–1744 CrossRef CAS.
- D. Mukherjee, R. Hyde, M. Hordagoda, N. Bingham, H. Srikanth, S. Witanachchi and P. Mukherjee, J. Appl. Phys., 2012, 112, 064101 CrossRef.
- N. Sama, C. Soyer, D. Remiens, C. Verrue and R. Bouregba, Sens. Actuators, A, 2010, 158, 99–105 CrossRef CAS.
- X. J. Lou, J. Appl. Phys., 2009, 105, 024101 CrossRef.
- D. Bao, N. Wakiya, K. Shinozaki, N. Mizutani and X. Yao, Appl. Phys. Lett., 2001, 78, 3286–3288 CrossRef CAS.
- H. L. Wang, X. K. Ning and Z. J. Wang, RSC Adv., 2015, 5, 76783–76787 RSC.
- H. J. Han, Y. N. Chen and Z. J. Wang, RSC Adv., 2015, 5, 92940–92946 RSC.
- H. J. Han, Y. N. Chen and Z. J. Wang, Ceram. Int., 2015, 41, 15208–15216 CrossRef CAS.
- I. Pintilie, C. M. Teodorescu, C. Ghica, C. Chirila, A. G. Boni, L. Hrib, I. Pasuk, R. Negrea, N. Apostol and L. Pintilie, ACS Appl. Mater. Interfaces, 2014, 6, 2929–2939 CAS.
- S. Stankovich, D. A. Dikin, R. D. Piner, K. A. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S. T. Nguyen and R. S. Ruoff, Carbon, 2007, 45, 1558–1565 CrossRef CAS.
- W. Gao, L. B. Alemany, L. Ci and P. M. Ajayan, Nat. Chem., 2009, 1, 403–408 CrossRef CAS PubMed.
- I. K. Moon, J. Lee, R. S. Ruoff and H. Lee, Nat. Commun., 2010, 1, 73 Search PubMed.
- Y. Liang, J. Frisch, L. Zhi, H. Norouzi-Arasi, X. Feng, J. P. Rabe, N. Koch and K. Mullen, Nanotechnology, 2009, 20, 434007 CrossRef PubMed.
- K. Erickson, R. Erni, Z. Lee, N. Alem, W. Gannett and A. Zettl, Adv. Mater., 2010, 22, 4467–4472 CrossRef CAS PubMed.
- A. Bachmatiuk, J. Zhao, S. M. Gorantla, I. G. G. Martinez, J. Wiedermann, C. Lee, J. Eckert and M. H. Rummeli, Small, 2015, 11, 515–542 CrossRef CAS PubMed.
- S. O. Chung, J. W. Kim, S. T. Kim, G. H. Kim and W. J. Lee, Mater. Chem. Phys., 1998, 53, 60–66 CrossRef CAS.
- N. T. Ho, V. Senthilkumar, H.-S. Cho, S. H. Nho, S. Cho, M. C. Jung, Y. B. Qi and Y. S. Kim, Phys. Status Solidi A, 2014, 211, 1873–1876 CrossRef CAS.
- B. Nagaraj, S. Aggarwal and R. Ramesh, J. Appl. Phys., 2001, 90, 375–382 CrossRef CAS.
- C. L. He, F. Zhuge, X. F. Zhou, M. Li, G. C. Zhou, Y. W. Liu, J. Z. Wang, B. Chen, W. J. Su, Z. P. Liu, Y. H. Wu, P. Cui and R.-W. Li, Appl. Phys. Lett., 2009, 95, 232101 CrossRef.
- S. M. Jilani, T. D. Gamot, P. Banerji and S. Chakraborty, Carbon, 2013, 64, 187–196 CrossRef CAS.
- V. Gopalan and M. C. Gupta, J. Appl. Phys., 1996, 80, 6099 CrossRef CAS.
- Z. H. Tang, M. H. Tang, X. S. Lv, H. Q. Cai, Y. G. Xiao, C. P. Cheng, Y. C. Zhou and J. He, J. Appl. Phys., 2013, 113, 164106 CrossRef.
- Y. Zhou, X. Zou, L. You, R. Guo, Z. Shiuh Lim, L. Chen, G. Yuan and J. Wang, Appl. Phys. Lett., 2014, 104, 012903 CrossRef.
- L. Pintilie, I. Boerasu, M. J. M. Gomes, T. Zhao, R. Ramesh and M. Alexe, J. Appl. Phys., 2005, 98, 124104 CrossRef.
- W. Monch, Phys. Rev. Lett., 1987, 58, 1260–1263 CrossRef PubMed.
- J. F. Scott, K. Watanabe, A. J. Hartmann and R. N. Lamb, Ferroelectrics, 1999, 225, 83–90 CrossRef.
- L. Pintilie, I. Vrejoiu, G. Le Rhun and M. Alexe, J. Appl. Phys., 2007, 101, 064109 CrossRef.
- J. Robertson and C. W. Chen, Appl. Phys. Lett., 1999, 74, 1168–1170 CrossRef CAS.
- A. Allain, J. Kang, K. Banerjee and A. Kis, Nat. Mater., 2015, 14, 1195–1205 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2016 |





